Abstract
Background
Glucagon-like peptide-1 (GLP-1) agonists have established their efficacy and safety in the treatment of obesity. In this study, we aimed to assess the efficacy and safety of survodutide, a new GLP-1 agonist.
Methods
We searched PubMed, Web of Science, Scopus, and Cochrane Library databases until May 2024 for randomized controlled trials using survodutide obesity treatment. Continuous data were pooled as standardized mean difference (SMD), while dichotomous data were pooled as risk ratios (RR) with 95% confidence intervals.
Results
There was a significant relative body weight reduction in the survodutide group (SMD: −1.5; 95% CI: −2.05 to −0.95; P < 0.00001). Overall adverse events in the four included studies weren’t significantly different between the survodutide group and placebo group (RR: 1.18; 95% CI: 0.98 to 1.41; I2 = 72%; P = 0.08). Survodutide was associated with a higher risk of developing diarrhea than the placebo group in the four included trials (RR: 1.89; 95% CI: 1.2 to 2.97; I2=0%; P < 0.00001).
Conclusion
Survodutide effectively reduced relative body weight, absolute body weight, and hemoglobin A1c (P < 0.00001). The incidence of adverse events was comparable between the two groups, while gastrointestinal adverse events were higher in the survodutide group.
Keywords: Meta-analysis, obesity, review, survodutide
Obesity is a medical condition that leads to serious complications and bad health outcomes such as poor quality of life and premature death. Also, it is considered the basis for developing many other diseases such as cardiovascular diseases, kidney diseases, type 2 diabetes mellitus, and some cancers such as pancreatic and liver cancer.1 Obesity poses a threat to healthcare systems in populations of high income, and its prevalence is expected to increase from 10% to 20% by the end of 2035.2 Obesity’s economic burden in the United States is immense, leading to annual spending of hundreds of billions of dollars on obesity-related medical needs.1 Although the prevalence is currently stabilized, there is no guarantee that the stability will be maintained.2 Obesity has three classes based on body mass index (kg/m2): (a) class I, for BMI between 30 and 34.9 kg/m2, (b) class II, for BMI between 35 and 39.9 kg/m2, and (c) class III, for BMI above 40 kg/m2.3
Obesity is a multifactorial disease, as it is linked to genetic factors4 and environmental factors such as modern lifestyles5 and quality of diet regardless of other dimensions of lifestyle.6 Based on that, guidelines have agreed on the multidisciplinary management of obesity, with pharmacotherapy, behavioral therapy, lifestyle modifications, and bariatric surgery.7–9 Bariatric surgery is restricted to those with class II or III obesity and, while efficacious, it is impossible to perform for all patients due to the stigma associated with it, access difficulties, and the fear of complications.8 Medical weight management remains one of the options for the treatment of excess weight, and recent advances have revolutionized how we treat it, and more importantly how we will be treating obesity soon.
A study by Rubino and his team in the US compared two glucagon-like peptide-1 (GLP-1) receptor agonists with different durations of action: once-weekly semaglutide at a dose of 2.4 mg versus once-daily liraglutide at 3.0 mg, in conjunction with lifestyle modifications. Over 68 weeks, the trial evaluated the efficacy of these treatments in promoting weight loss. The results indicated that semaglutide was significantly more effective, demonstrating an average weight reduction from baseline of −15.8%, in contrast to −6.4% for liraglutide.
Survodutide, a dual agonist to glucagon receptor (GCGR)/GLP-1R, mimics oxyntomodulin, the natural gut hormone. It can lower body weight through the combination of the GCGR agonist action, which can increase energy expenditure, with GLP-1R agonists, which have anorectic actions.10,11 Lower death rates were observed in patients receiving survodutide, making it a preferred option over both semaglutide and tirzepatide. In this review, we aim to assess the efficacy and safety of survodutide as a medical treatment option for obesity.
METHODS
In this systematic review and meta-analysis, we followed the 2020 updated version of the Preferred Reporting Items for Systematic Reviews and Meta‐analyses (PRISMA).12 We registered the protocol in the Open Science Framework database with a registration number of 10.17605/OSF.IO/GB39W.
Study selection
We searched PubMed, Web of Science, Scopus, and Cochrane Library databases until May 2024 for randomized controlled trials using the search strategy provided in the supplementary material. Additionally, we reinforced our search by reviewing the references of our final included studies to include other relevant studies.
We included randomized controlled trials evaluating the safety and efficacy of survodutide, conducted on human participants of all ages and published in English. Nonrandomized trials, other study designs, animal studies, and studies published in other languages were excluded.
After searching the mentioned databases, we imported the search results into EndNote.13 Afterwards, duplicates were eliminated, and the remaining unique references were exported to an Excel spreadsheet. Two researchers independently screened the titles and abstracts of all identified studies. Full-text articles of potentially eligible studies were assessed for inclusion. Conflicts were resolved by discussion or by a third reviewer.
Data extraction
Two independent researchers extracted the data from the included studies into Microsoft Excel spreadsheets to ensure the accuracy of the data. Any conflicts were resolved by discussion or by a third reviewer. We extracted the following data: (a) summary of the included studies (study phase, drug doses, country, year, sample size, and treatment duration); (b) baseline characteristics (age, weight, BMI, waist circumference, hemoglobin A1c [HbA1c], fasting blood glucose, systolic blood pressure, and diastolic blood pressure); and (c) outcomes. Percentage change from baseline in body weight was our primary outcome. Our secondary outcomes included the mean change in waist circumference, HbA1c, and safety outcomes.
Risk of bias and quality assessment
We used the Cochrane Risk of Bias 2 tool (ROB2) to assess the risk of bias for the included studies.14 Two independent researchers assessed the quality using the tool across its five domains (randomization process, deviation from intended intervention, missing outcome data, selection of the reported results, and overall bias) for each paper. Any disagreements were resolved by consensus.
Statistical analysis
We conducted the analysis using RevMan 5.4 software. The groups of different doses of survodutide were combined to be compared with placebo. The outcome measures for efficacy and adverse effects were the standardized mean difference (SMD) and the risk ratio (RR), respectively; each result was accompanied by a 95% confidence interval (CI). We investigated the heterogeneity using forest plots generated for each outcome of interest. We assessed statistical heterogeneity between trials using the I2 statistic. An I2 value > 50% indicates significant heterogeneity. A random effect model (inverse variance) was adopted rather than a fixed effect model, yielding a more conservative estimate of the pooled effect and generalizable results. When significant heterogeneity appeared, we conducted a sensitivity analysis and evaluated the methodology of each trial to determine the reason for this heterogeneity.
RESULTS
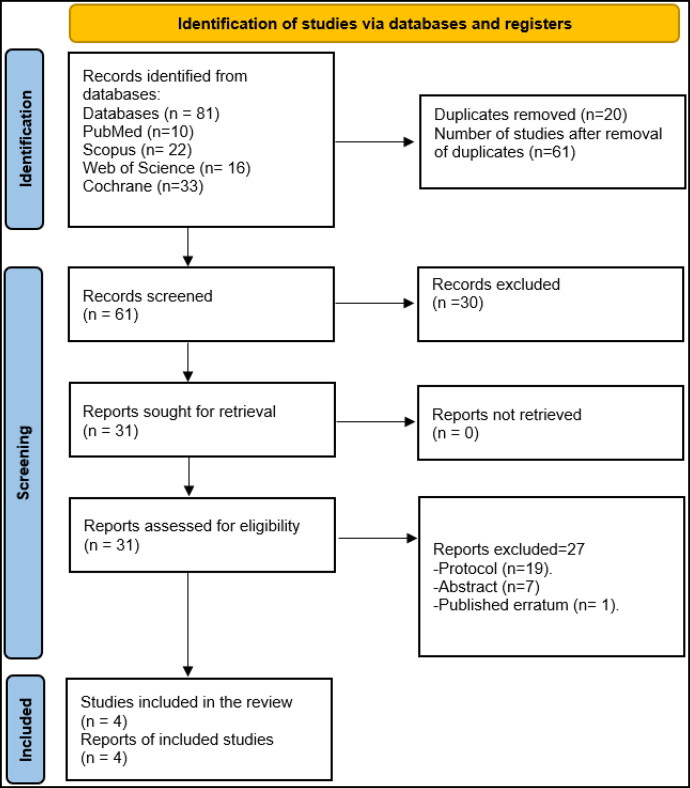
After searching four databases (PubMed, Scopus, Web of Science, and Cochrane), 81 papers were identified; 20 papers were duplicates, so 61 records entered the title and abstract screening phase. Thirty were excluded as they did not meet our eligibility criteria. Thirty-one records entered the full-text screening phase, which ended with four studies15–18 that were included in our analysis (Figure 1).
Figure 1.
PRISMA chart of the reported studies showing the search selection strategy and exclusion criteria.
The included studies were published between 2022 and 2024 with a total sample size of 980 patients with a mean age of 46.3 years; 558 (56.93%) were females. Two studies were conducted in Germany (Jungnik 2022; Blüher 2023),17,18 one was conducted in Japan (Yazawa 2023),16 and one was performed across 11 countries with a sample size of 387 participants (Roux 2024).15
The subgroups were divided based on the survodutide dose, ranging from a minimum of 0.3 mg to a maximum of 4.8 mg, and compared with a placebo group. The administration route was a subcutaneous injection given once weekly, except in the Blüher study,18 where the 1.2 and 1.8 mg doses were administered twice weekly. The treatment duration varied across studies: 6 weeks in Jungnik 2022 Part A17; 16 weeks in Jungnik 2022 Part B,17 Blüher 2023,18 and Yazawa 202316; and 46 weeks in Roux 2024.15 Follow-up duration was only specified in the Roux15 and Blüher18 studies, at 3 weeks and 4 weeks, respectively. The characteristics of the included studies are summarized in Tables 1 and 2.
Table 1.
Baseline characteristics of the included studies
| Study | Dose (mg) | Sample size | Age, mean (SD) | Female, n (%) | Race, n (%) | Metabolic parameters |
|||
|---|---|---|---|---|---|---|---|---|---|
| Weight (kg) | BMI (kg/m2) | WC (cm) | HbA1c (%) | ||||||
| Roux 202415 | 0.6 | 77 | 48.6 (12.6) | 51 (66%) | W: 59 (77%); A: 8 (10%); B/AA: 10 (13%); Multiple: 0; NH/PI: 0; AI/AN: 0 | 107.0 (18.7) | 37.8 (6.3) | 115.3 (13.4) | 5.51 ± 0.43 |
| 2.4 | 78 | 49.0 (13.1) | 54 (69%) | W: 60 (77%); A: 9 (12%); B/AA: 8 (10%); Multiple: 1 (1%); NH/PI: 0; AI/AN: 0 | 106.6 (23.0) | 37.6 (7.3) | 115.3 (17.0) | 5.48 ± 0.46 | |
| 3.6 | 76 | 50.3 (11.8) | 51 (67%) | W: 63 (83%); A: 9 (12%); B/AA: 3 (4%); Multiple: 1 (1%); NH/PI: 0; AI/AN: 0 | 104.7 (19.6) | 37.0 (5.7) | 112.8 (13.9) | 5.56 ± 0.38 | |
| 4.8 | 76 | 47.6 (13.5) | 53 (70%) | W: 59 (78%); A: 7 (9%); B/AA: 8 (11%); Multiple: 0; NH/PI: 1 (1%); AI/AN: 1 (1%) | 105.9 (17.4) | 37.6 (6.0) | 112.8 (13.0) | 5.50 ± 0.42 | |
| Placebo | 77 | 50.0 (13.5) | 53 (69%) | W: 60 (78%); A: 7 (9%); B/AA: 8 (10%); Multiple: 1 (1%); NH/PI: 1 (1%); AI/AN: 0 | 104.3 (23.0) | 35.8 (5.0) | 110.4 (14.6) | 5.45 ± 0.36 | |
| Yazawa 202316 | 1.8 qw | 9 | 37.0 (8.0) | 0 | A: 9 (100%) | 73.8 (6.8) | 25.6 (1.9) | 89.51 (7.15) | 5.20 (0.25) |
| 4.8 qw | 9 | 31.9 (5.8) | 0 | A: 9 (100%) | 72.2 (5.3) | 24.1 (0.8) | 86.44 (2.77) | 5.12 (0.38) | |
| 2.4 biw | 9 | 31.6 (7.5) | 0 | A: 9 (100%) | 77.8 (7.9) | 25.1 (1.9) | 90.11 (7.09) | 5.12 (0.21) | |
| Placebo | 9 | 36.2 (8.5) | 0 | A: 9 (100%) | 76.6 (7.1) | 25.9 (2.0) | 90.90 (6.67) | 5.21 (0.20) | |
| Jungnik 202217 | SRD 0.3 | 6 | 39.5 (3.7) | 6 (100.0) | W: 6 (100.0) | 84.8 (10.2) | 27.4 (1.9) | ||
| SRD 0.5 | 6 | 34.0 (6.1) | 6 (100.0) | W: 6 (100.0) | 85.3 (10.8) | 26.1 (2.2) | |||
| SRD 1.2 | 6 | 31.5 (7.9) | 6 (100.0) | W: 6 (100.0) | 84.2 (10.5) | 26.0 (2.7) | |||
| Placebo | 6 | 29.2 (4.3) | 6 (100.0) | W: 6 (100.0) | 79.7 (2.1) | 24.2 (1.4) | |||
| MRD A: DS1 | 15 | 48.1 (7.7) | 15 (100.0) | W: 15 (100.0) | 98.0 (9.3) | 30.7 (2.9) | |||
| MRD A: DS2 | 17 | 42.6 (11.8) | 17 (100.0) | W: 16 (94.1); B/AA: 1 (5.9) | 97.9 (10.8) | 30.3 (2.5) | |||
| MRD A: DS3 | 18 | 49.6 (9.7) | 18 (100.0) | W: 6 (100.0) | 95.2 (12.4) | 30.3 (2.9) | |||
| MRD A: DS4 | 17 | 39.1 (12.6) | 17 (100.0) | W: 6 (100.0) | 97.4 (9.7) | 30.3 (2.3) | |||
| MRD A: Placebo | 13 | 53.2 (8.5) | 13 (100.0) | W: 6 (100.0); NH/PI: 1 (7.7) | 92.7 (6.2) | 29.7 (1.6) | |||
| MRD B: DS5 | 13 | 45 (16.8) | 4 (30.8) | W: 13 (100) | 87.7 (9.9) | 29.6 (1.9) | |||
| MRD B: DS6 | 11 | 48.9 (14) | 4 (36.4) | W: 11 (100) | 94.2 (19.2) | 30.5 (3.7) | |||
| MRD B: DS7 | 12 | 42.2 (12.7) | 3 (25.0) | W: 12 (100) | 95.8 (12.6) | 31.7 (3.7) | |||
| MRD B: Placebo | 9 | 44.6 (10.3) | 3 (33.3) | W: 9 (100) | 101 (17.2) | 32.3 (4.3) | |||
| Blüher 202318 | 0.3 qw | 50 | 56.1 (10.2) | 24 (48) | W: 42 (84); A: 4 (8); B/AA: 3 (6); AI/AN: 1 (2) | 97.6 (19.7) | 33.8 (6.1) | 110.6 (12.8) | 8.09 (0.76) |
| 0.9 qw | 50 | 58.2 (9.6) | 22 (44) | W: 44 (88); A: 5 (10); B/AA: 1 (2) | 100.1 (19.8) | 34.9 (5.2) | 111.5 (15.6) | 7.89 (0.80) | |
| 1.8 qw | 52 | 55.3 (10.3) | 25 (48.1) | W: 42 (82.8); A: 8 (15.4); B/AA: 2 (3.8) | 95.9 (22.8) | 33.6 (5.8) | 107.2 (20.0) | 8.14 (0.86) | |
| 2.7 qw | 50 | 59.6 (8.5) | 17 (34) | W: 43 (86); A: 4 (8); B/AA: 2 (4); missing: 1 (2) | 96.6 (22.8) | 34.0 (6.8) | 110.7 (16.4) | 8.18 (0.97) | |
| 1.2 biw | 51 | 58.3 ± 8.8 | 24 (47.1) | W: 41 (80.4); A: 5 (9.8); B/AA: 4 (7.8); missing: 1 (2) | 95.0 ± 22.1 | 33.0 ± 5.0 | 109.0 ± 18.2 | 7.97 ± 0.71 | |
| 1.8 biw | 49 | 57.7 ± 9.4 | 22 (44.9) | W: 42 (85.7); A: 3 (6.1); B/AA: 3 (6.1); AI/AN: 1 (2) | 98.3 ± 24.4 | 34.9 ± 7.0 | 115.1 ± 28.7 | 8.03 ± 0.82 | |
| 1.0 qw | 50 | 55.8 ± 10.5 | 16 (32) | W: 43 (86.0); A: 5 (10.0); B/AA: 2 (4.0); AI/AN: 1 (2) | 96.7 ± 20.0 | 33.4 ± 6.1 | 108.1 ± 13.5 | 8.15 ± 0.85 | |
| Placebo | 59 | 57.5 (10.5) | 28 (47.5) | W: 47 (79.7); A: 8 (13.6); B/AA: 3 (5.1); NH/PI: 1 (1.7) | 93.0 (21.0) | 33.4 (5.9) | 110.4 (16.5) | 8.15 (0.85) | |
A indicates Asian; AI/AN, American Indian or Alaskan Native; B/AA, Black or African American; biw, twice weekly; BMI, body mass index; DS, dosage schedule; HbA1c, hemoglobin A1c; MRD, multiple rising dose; NH/PI, Native Hawaiian or Pacific Islander; qw, once weekly; SRD, single rising dose; W, White; WC, waist circumference.
Table 2.
Summary of the included studies
| Study ID | Roux 202415 | Yazawa 202316 | Jungnik 202217 | Blüher 202318 |
|---|---|---|---|---|
| Phase (NCT number) | Phase 2 (NCT04667377) | Phase I (NCT04384081) | Phase 1b (NCT03591718) | NCT04153929 |
| Sample size | 384 | 36 | 125 | 411 |
| Country | USA, Australia, Belgium, Canada, China, Germany, South Korea, Netherlands, New Zealand, Poland, Sweden, and UK | Japan | Germany | Germany |
| Treatment doses (mg) | 0.6, 2.4, 3.6, 4.8 | 1.8, 4.8 | Multiple rising doses | 0.3 qw, 0.9 qw, 1.8 qw, 2.7 qw, 1.2 biw, 1.8 biw |
| Route of administration | Subcutaneous | Subcutaneous | Subcutaneous | Subcutaneous |
| Frequency of administration | Once weekly | Once weekly, twice weekly | Once weekly | Once weekly, twice weekly |
| Treatment duration | 46 weeks | 16 weeks | Part A: 6 weeks; Part B: 16 weeks | 16 weeks |
| Follow-up duration | 3 weeks | 4 weeks | ||
| Main inclusion criteria | Adults (≥18 to <75 years) with a BMI ≥ 27 kg/m², a stable body weight ≥ 70 kg (females) or ≥80 kg (males), and with HbA1c < 6.5% (without diabetes) at screening | Men 20 to 45 years, with a BMI of 23–40 kg/m2, stable (≤5% change within 3 months) body weight of ≥65 kg, and glycated hemoglobin (HbA1c) < 6.5% | Adults (18–70 years) with a BMI of 27–40 kg/m2 and stable body weight (≤5% change within 3 months prior to screening) of ≥70 kg (females) or ≥80 kg (males) | Adults 18–75 years, diagnosed with type 2 diabetes for ≥6 months, had HbA1c value of 53–86 mmol/mol (7.0–10.0%) and a BMI of 25–50 kg/m2 at screening, treated with a stable dose of metformin of ≥1000 mg/day (immediate or extended-release) for ≥3 months before screening; exclusion criteria listed in the Methods |
| Conclusion | All tested survodutide doses significantly reduced body weight in a dose-dependent manner relative to placebo in participants with a BMI ≥27 kg/m² | No unexpected tolerability concerns; reduced placebo-corrected body weight by up to 12.37% in Japanese men with overweight/obesity after 16 weeks of treatment | Produced a placebo-corrected body weight loss of 13.8% (week 16), highlighting its potential to promote clinically meaningful body weight loss in people with overweight/obesity | Reduced HbA1c levels and body weight after 16 weeks of treatment in participants with type 2 diabetes; dose-related gastrointestinal AEs could be mitigated with slower dose escalations |
AE indicates adverse event; biw, twice weekly; BMI, body mass index; HbA1c, hemoglobin A1c; qw, once weekly.
Risk of bias assessment
After evaluating the included studies using ROB2, we found that two studies showed a low risk of bias.15,18 Jungnik’s study showed some concerns, mainly in the randomization process and outcomes measurement.17 Yazawa’s study also showed some concerns regarding outcomes measurement and deviations from the intended intervention16 (Supplemental Figures 1 and 2).
Body weight changes
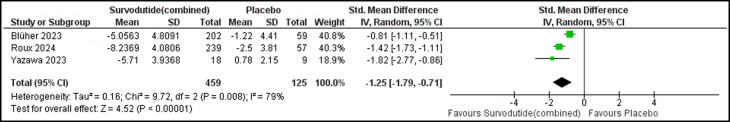
Testing the relative reduction of weight in the four included studies15–18 showed a significant reduction among the survodutide group (SMD: −1.5; 95% CI: −2.05 to −0.95; I2: 78%; P < 0.00001) (Figure 2). This analysis included three studies15,16,18 and revealed a significant absolute weight reduction (kg) among the survodutide group (SMD: −1.25; 95% CI: −1.79 to −0.71; I2: 79%; P < 0.00001) (Figure 3). The subgroup analysis based on the percentage of weight reduction by 5% or 10% between the two studies15,18 showed a significant reduction in weight in the survodutide group: RR: 3.41; 95% CI: 2.25 to 5.17; I2=0%; P < 0.00001 for ≥5% weight loss and RR: 5.89; 95% CI: 2.82 to 12.32; I2=0%; P < 0.00001 for ≥10% weight loss (Supplemental Figure 3). The mean change from baseline in waist circumference (cm) among three studies15,16,18 showed a significant reduction in the survodutide group (SMD: −0.83; 95% CI: −1.56 to −0.1; I2=90%; P = 0.03) (Supplemental Figure 4).
Figure 2.
A forest plot showing the relative change in body weight from baseline (%).
Figure 3.
A forest plot showing the absolute change from baseline in body weight (kg).
Hemoglobin A1c level
The pooled analysis of the mean change from baseline HbA1c (%) among three studies15,16,18 revealed a significant reduction (MD: −1.12; 95% CI: −1.33 to −0.9; I2=0%; P < 0.00001) (Supplemental Figure 5).
Adverse events
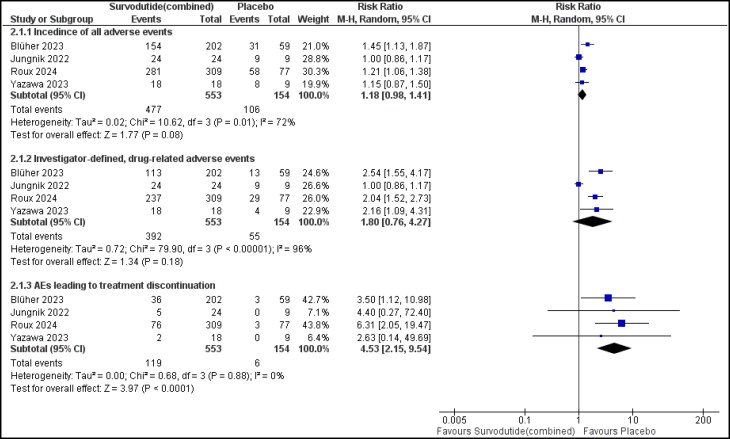
Overall adverse events in the four included studies15–18 weren’t significantly different between the survodutide group and placebo group (RR: 1.18; 95% CI: 0.98 to 1.41; I2=72%; P = 0.08) (Figure 4). The adverse effects defined by the investigator also reported in the four included studies15–18 revealed no significant difference between the two groups (RR: 1.8; 95% CI: 0.76 to 4.27; I2=96%; P = 0.18) (Figure 4). The survodutide group was associated with a higher risk of adverse events that led to discontinuation of treatment in the four included studies15–18 (RR: 4.53; 95% CI: 2.15 to 9.54; I2=0%; P < 0.00001) (Figure 4).
Figure 4.
A forest plot for all adverse events.
Among the four studies,15–18 survodutide was associated with a higher risk of vomiting (RR: 6.64; 95% CI: 3 to 14.7; I2=0%; P < 0.00001) (Supplemental Figure 6), as well as nausea (RR: 3.3; 95% CI: 2.23 to 4.88; I2=0%; P < 0.00001) (Supplemental Figure 7) and diarrhea (RR: 1.89; 95% CI: 1.2 to 2.97; I2=0%; P < 0.00001) (Supplemental Figure 8). Three studies reported dyspepsia, and an increased risk was associated with the survodutide group compared to the placebo group (RR: 6.93; 95% CI: 1.78 to 26.98; I2=0%; P = 0.005) (Supplemental Figure 9). Early satiety, reported in two studies,16,17 showed a significant difference between the two groups, as the survodutide showed a positive association with early satiety (RR, 6.44; 95% CI: 1.72 to 24.16; I2=0%; P = 0.006) (Supplemental Figure 10). Our meta-analysis of the four included studies15–18 revealed no significant difference in decrease in appetite between the two groups (RR: 4.55; 95% CI: 0.68 to 30.32; I2=86%; P = 0.12) (Supplemental Figure 11). Constipation was examined in three studies,15–17 and no significant difference was reported between the two groups (RR: 1.99; 95% CI: 0.29 to 13.48; I2=78%; P = 0.48) (Supplemental Figure 12).
Abdominal pain was reported in two studies,16,17 and there was no significant difference between the survodutide group and the placebo group (RR: 4.74; 95% CI: 0.65 to 34.66; I2= 0%; P = 0.132) (Supplemental Figure 13). Abdominal distention was reported in two studies,16,17 and there was no significant difference between the survodutide group and the placebo group (RR: 2.04; 95% CI: 0.38 to 10.79; I2=0%; P = 0.4) (Supplemental Figure 14). Two studies17,18 reported headache. Our meta-analysis revealed no significant difference between the two groups (RR: 1.46; 95% CI: 0.69 to 3.09; I2 = 0%; P = 0.32) (Supplemental Figure 15).
DISCUSSION
In the domain of weight reduction and diabetes control, the polyagonist therapies have advantages compared to monoagonist therapies due to multiple mechanisms of action according to the targeted receptor. Dual GCGR/GLP-1R agonists are newly developed pharmacological agents used in the management of obesity and type 2 diabetes. They have several mechanisms of action, as they inhibit food intake by secreting glucagon-like peptide and delayed stomach emptying, regulate insulin and glucagon levels, and increase energy expenditure.19
Survodutide is a long-acting GCGR/GLP-1R dual agonist given subcutaneously as an antiobesity and antidiabetic drug;15 it also has shown promising results in treating metabolic dysfunction–associated steatohepatitis (MASH), as it significantly decreases inflammation and improves fibrosis.20
Our meta-analysis shows that survodutide has promising results in managing obesity. In terms of efficacy, survodutide significantly reduces weight (MD: −1.25; 95% CI: −1.79 to −0.71; I2: 79%; P < 0.00001) and decreases HbA1c (MD: −1.12; 95% CI: −1.33 to −0.9; I2=0%; P < 0.00001) compared to placebo. Regarding safety, survodutide had a relatively increased risk of developing side effects compared to the placebo group; the most frequently reported side effects were gastrointestinal symptoms. None of included studies reported deaths, which suggests the relative safety of survodutide as a dual agonist compared to tirzepatide. Survodutide was associated with a high risk of adverse events that led to discontinuation of the drug.
A study led by Rubino and his team in the US sought to compare two GLP-1R agonists with varying durations of action: once-weekly semaglutide at 2.4 mg versus once-daily liraglutide at 3.0 mg, alongside lifestyle modifications. Over the course of a 68-week trial, they assessed the efficacy of these treatments in weight reduction. The findings revealed that semaglutide was more effective, with an average weight reduction from baseline of −15.8%, compared to −6.4% for liraglutide.21 These results indicate the effectiveness of long-acting drugs in weight reduction, offering the advantage of avoiding the risks associated with daily subcutaneous injections, such as injection site pain and issues with patient compliance.22
Tirzepatide is a recently approved weight loss drug, with a dual mechanism of action: glucose-dependent insulinotropic polypeptide (GIP) and GLP-1. It is long-acting and is given subcutaneously once weekly.23 Many studies have tested the efficacy and safety of tirzepatide in weight reduction and glycemic control. Jastreboff and her colleagues conducted a study with 2539 adults with a BMI ≥ 30 kg/m2 in a 72-week randomized clinical trial; the participants were enrolled blindly to 5, 10, or 15 mg tirzepatide groups or to a placebo group. The authors reported a mean body weight reduction at the end of the trial of −15.0% for 5 mg, −19.5% for 10 mg, and −20.9% for 15 mg, compared to the placebo group, which was −3.1%.24 Tirzepatide was also compared with semaglutide to test the efficacy of mono versus dual agonist therapy in 1879 patients with type 2 diabetes. Frías and colleagues found that the mean change in HbA1c level from baseline in the 5, 10, and 15 mg tirzepatide groups was −2.01%, −2.24%, and −2.30, respectively, while in semaglutide the change was only −1.86%. The study also noted the mean reductions in body weight at 40 weeks, which were −7.6, −9.3, and −11.2 kg for tirzepatide at doses of 5, 10, and 15 mg, respectively, while the weight reduction was −5.7 kg with semaglutide. Thus, tirzepatide was more effective than semaglutide at all doses, which suggests that dual agonists more effectively reduce body weight and improve glycemic control compared to monoagonist therapies. Most reported adverse events were gastrointestinal and more likely to occur with tirzepatide at 10 mg and 15 mg doses compared to tirzepatide 5 mg and semaglutide. Thirteen cases of death were reported in the study, with 12 in the tirzepatide groups and 1 in the semaglutide group.25
Our findings highlight incretin-based therapy as an important treatment option for obesity and its associated comorbidities, as survodutide significantly reduced the Hb1Ac. SYNCHRONIZE™-1 and −2 trials, phase 3 clinical trials, will investigate the efficacy, safety, and tolerability of survodutide in the presence or absence of diabetes.26 The SYNCHRONIZE Cardiovascular Outcomes Trial will assess the cardiovascular efficacy and safety of survodutide in patients at increased risk for cardiovascular diseases.27 The LIVERAGE trial also will evaluate the long-term efficacy and safety of survodutide weekly injections in adult participants with noncirrhotic MASH and the F2 and F3 stages of liver fibrosis.28
Our meta-analysis has several strengths, with the inclusion of only randomized controlled trials, double-arm comparisons, and a low overall risk of bias in the included trials. We faced some limitations, as there was heterogeneity in some results, probably due to different doses of survodutide, different durations of drug intake, different sample sizes, and different times when patients were reassessed. Another limitation was that only one study reported blood pressure measurements and heart rate, which hindered the ability to determine the effect of survodutide on the cardiovascular system.
In conclusion, current evidence suggests that survodutide could reduce weight and HbA1c effectively. In terms of safety, survodutide is associated with a high risk of gastrointestinal adverse events. However, no death was reported, making it relatively safe in the treatment of obesity. Future randomized controlled trials with larger populations are needed.
Supplementary Material
Disclosure statement/Funding
The authors report no funding or conflicts of interest.
References
- 1.Patient well-being and the clinical and economic burdens associated with obesity in the United States. Am J Managed Care. 2022;28(Suppl 15):S279–S287. [DOI] [PubMed] [Google Scholar]
- 2.Koliaki C, Dalamaga M, Liatis S.. Update on the obesity epidemic: after the sudden rise, is the upward trajectory beginning to flatten? Curr Obes Rep. 2023;12(4):514–527. doi: 10.1007/s13679-023-00527-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.World Health Organization . Preventing and Managing the Global Epidemic of Obesity. Geneva: World Health Organization; 1997. [PubMed] [Google Scholar]
- 4.Sanghera DK, Bejar C, Sharma S, Gupta R, Blackett PR.. Obesity genetics and cardiometabolic health: potential for risk prediction. Diabetes Obes Metab. 2019;21(5):1088–1100. doi: 10.1111/dom.13641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Verde L, Frias-Toral E, Cardenas D.. Editorial: environmental factors implicated in obesity. Front Nutr. 2023;10:1171507. doi: 10.3389/fnut.2023.1171507. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Barrea L, Frias-Toral E, Aprano S, et al. The clock diet: a practical nutritional guide to manage obesity through chrononutrition. Minerva Med. 2022;113(1):172–188. doi: 10.23736/S0026-4806.21.07207-4. [DOI] [PubMed] [Google Scholar]
- 7.Coutinho W, Halpern B.. Pharmacotherapy for obesity: moving towards efficacy improvement. Diabetol Metab Syndr. 2024;16(1):6. doi: 10.1186/s13098-023-01233-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Roomy MA, Hussain K, Behbehani HM, et al. Therapeutic advances in obesity management: an overview of the therapeutic interventions. Front Endocrinol (Lausanne). 2024;15:1364503. doi: 10.3389/fendo.2024.1364503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Abdalla Ahmed MA, Ssemmondo E, Mark-Wagstaff C, Sathyapalan T.. Advancements in the management of obesity: a review of current evidence and emerging therapies. Expert Rev Endocrinol Metab. 2024;19(3):257–268. doi: 10.1080/17446651.2024.2347258. [DOI] [PubMed] [Google Scholar]
- 10.Melson E, Ashraf U, Papamargaritis D, Davies MJ.. What is the pipeline for future medications for obesity? Int J Obes. 2024. doi: 10.1038/s41366-024-01473-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Klein T, Augustin R, Hennige AM.. Perspectives in weight control in diabetes – survodutide. Diabetes Res Clin Pract. 2024;207:110779. doi: 10.1016/j.diabres.2023.110779. [DOI] [PubMed] [Google Scholar]
- 12.Page MJ, McKenzie JE, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. doi: 10.1136/bmj.n71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.The EndNote Team . EndNote. 20th ed. Philadelphia, PA: Clarivate; 2013. [Google Scholar]
- 14.Sterne JAC, Savović J, Page MJ, et al. RoB 2: a revised tool for assessing risk of bias in randomised trials. BMJ. 2019;366:l4898. doi: 10.1136/bmj.l4898. [DOI] [PubMed] [Google Scholar]
- 15.Le Roux CW, Steen O, Lucas KJ, Startseva E, Unseld A, Hennige AM.. Glucagon and GLP-1 receptor dual agonist survodutide for obesity: a randomised, double-blind, placebo-controlled, dose-finding phase 2 trial. Lancet Diabet Endocrinol. 2024;12(3):162–173. doi: 10.1016/S2213-8587(23)00356-X. [DOI] [PubMed] [Google Scholar]
- 16.Yazawa R, Ishida M, Balavarca Y, Hennige AM.. A randomized Phase I study of the safety, tolerability, pharmacokinetics and pharmacodynamics of BI 456906, a dual glucagon receptor/glucagon‐like peptide‐1 receptor agonist, in healthy Japanese men with overweight/obesity. Diabetes Obes Metab. 2023;25(7):1973–1984. doi: 10.1111/dom.15064. [DOI] [PubMed] [Google Scholar]
- 17.Jungnik A, Arrubla Martinez J, Plum-Mörschel L, et al. Phase I studies of the safety, tolerability, pharmacokinetics and pharmacodynamics of the dual glucagon receptor/glucagon‐like peptide‐1 receptor agonist BI 456906. Diabetes Obes Metab. 2023;25(4):1011–1023. doi: 10.1111/dom.14948. [DOI] [PubMed] [Google Scholar]
- 18.Blüher M, Rosenstock J, Hoefler J, Manuel R, Hennige AM.. Dose–response effects on HbA1c and bodyweight reduction of survodutide, a dual glucagon/GLP-1 receptor agonist, compared with placebo and open-label semaglutide in people with type 2 diabetes: a randomised clinical trial. Diabetologia. 2024;67(3):470–482. doi: 10.1007/s00125-023-06053-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ma T, Huo S, Xu B, et al. A novel long-acting oxyntomodulin analogue eliminates diabetes and obesity in mice. Eur J Med Chem. 2020;203:112496. doi: 10.1016/j.ejmech.2020.112496. [DOI] [PubMed] [Google Scholar]
- 20.Sanyal AJ, Bedossa P, Fraessdorf M, et al. A phase 2 randomized trial of survodutide in MASH and fibrosis. N Engl J Med. 2024;391(4):311–319. doi: 10.1056/NEJMoa2401755. [DOI] [PubMed] [Google Scholar]
- 21.Rubino DM, Greenway FL, Khalid U, et al. Effect of weekly subcutaneous semaglutide vs daily liraglutide on body weight in adults with overweight or obesity without diabetes. JAMA. 2022;327(2):138–150. doi: 10.1001/jama.2021.23619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Usach I, Martinez R, Festini T, Peris J-E.. Subcutaneous injection of drugs: literature review of factors influencing pain sensation at the injection site. Adv Ther. 2019;36(11):2986–2996. doi: 10.1007/s12325-019-01101-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Syed YY. Tirzepatide: first approval. Drugs. 2022;82(11):1213–1220. doi: 10.1007/s40265-022-01746-8. [DOI] [PubMed] [Google Scholar]
- 24.Jastreboff AM, Aronne LJ, Ahmad NN, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387(3):205–216. doi: 10.1056/NEJMoa2206038. [DOI] [PubMed] [Google Scholar]
- 25.Frías JP, Davies MJ, Rosenstock J, et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N Engl J Med. 2021;385(6):503–515. doi: 10.1056/NEJMoa2107519. [DOI] [PubMed] [Google Scholar]
- 26.Wharton S, Le Roux CW, Kosiborod MN, et al. Survodutide for treatment of obesity: rationale and design of two randomized phase 3 clinical trials (SYNCHRONIZETM-1 and -2). Obesity (Silver Spring). 2025;33(1):67–77. doi: 10.1002/oby.24184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kosiborod MN, Platz E, Wharton S, et al. Survodutide for the treatment of obesity: rationale and design of the SYNCHRONIZE cardiovascular outcomes trial. JACC Heart Fail. 2024;12(12):2101–2109. doi: 10.1016/j.jchf.2024.09.004. [DOI] [PubMed] [Google Scholar]
- 28.Boehringer Ingelheim . A randomised, double-blind, placebo-controlled, multicentre, phase III trial evaluating long-term efficacy and safety of survodutide weekly injections in adult participants with noncirrhotic non-alcoholic steatohepatitis/metabolic associated steatohepatitis (NASH/MASH) and (F2) - (F3) stage of liver fibrosis. clinicaltrials.gov. February 2025. Report No.: NCT06632444. https://clinicaltrials.gov/study/NCT06632444.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.