Abstract
When endovascular treatment (EVT) is administered via the anterior spinal artery (ASA) for high cervical pial arteriovenous fistulas (PAVFs), severe diaphragmatic paralysis, although rare, may occur. We report a 46-year-old male who experienced subarachnoid hemorrhage. The Hunt–Hess scale score was Grade II. Angiography revealed a high cervical PAVF supplied by the C2 radiculomedullary artery and the ASA at the C4 vertebra level. EVT was performed. A microcatheter introduced through the ASA was placed close to the PAVF. After the Onyx agent was cast, the PAVF was obliterated; however, Onyx reflux into the ASA trunk was observed. Postoperatively, the patient had hemiparesis and insufficient autonomous respiration and mechanical ventilation was provided. Fifteen days after EVT, the patient’s hemiparesis completely resolved. Ultrasound examination revealed diaphragmatic paralysis. Magnetic resonance imaging revealed cervical cord infarction at the C2-C3 vertebral level, which supported the diagnosis of diaphragmatic paralysis due to phrenic nerve injury from cervical cord infarction above the C4 level. Respiratory function training was performed daily. Eighty-seven days after EVT, his autonomous respiration function further improved. However, 93 days after EVT, the patient died while asleep because the portable ventilator was not used. Therefore, for EVT for high cervical PAVFs, excessive Onyx reflux into the ASA can result in ventral cervical cord infarction, causing severe respiratory insufficiency due to diaphragmatic paralysis. We reported this case to alert neurointerventionists to the lessons learned from experience.
Keywords: Pial arteriovenous fistula, Anterior spinal artery, Embolization, Cervical cord, Diaphragmatic paralysis
Introduction
High cervical pial arteriovenous fistulas (PAVFs) at the C1-C2 vertebral levels are uncommon entities characterized by a direct arteriovenous shunt without an intervening nidus on the surface of the spinal cord surface consisting of single or multiple holes and drainage into the perimedullary vein [1]. Patients with high cervical PAVFs can experience subarachnoid hemorrhage (SAH) due to rupture of the fistula point or draining vein or suffer from spinal edema due to high pressure in the spinal vein system [2].
Hemorrhagic or symptomatic high cervical PAVFs cannot be left untreated due to their poor prognosis. Currently, high cervical PAVFs can be managed via endovascular treatment (EVT) [2]. During EVT, the anterior spinal artery (ASA) can serve as a transarterial approach. However, it may be dangerous to embolize high cervical PAVFs via the cervical ASA because excessive ASA occlusion can result in high cervical cord infarction, which rarely results in severe diaphragmatic paralysis. After EVT, the patient must depend on ventilators or die. We reported such a case to alert neurointerventionists to this complication. When high cervical PAVFs are embolized via the ASA, excessive ASA occlusion must be avoided because phrenic nerve injury from the cervical cord infarction above the C4 level can occur.
Case presentation
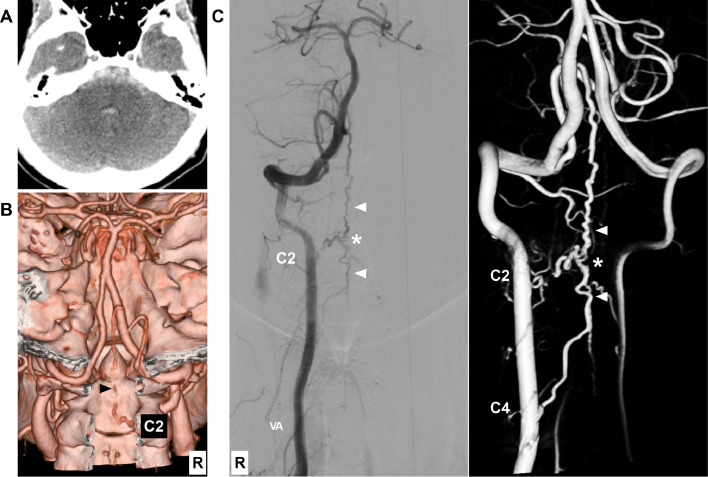
A 46-year-old male experienced severe headache for 4 hours. He was a healthy Chinese Han with no history of systemic hypertension, diabetes mellitus, recent infectious disease or surgery. During the physical examination, the patient was awake and able to answer questions correctly. He had meningeal irritation signs with nuchal rigidity, grade V muscle strength in his limbs, and no abnormal reflexes or hypoesthesia. The Hunt–Hess scale score was Grade II. Computed tomography (CT) revealed an SAH centered on the foramen magnum region and in front of the brainstem (Fig. 1A). CT angiography revealed an abnormal tortuous vessel extending from the cervical canal into the cranium (Fig. 1B).
Fig. 1.
Diagnostic images. (A) CT image showing subarachnoid hemorrhage in front of the brainstem and fourth ventricular hemorrhage. (B) CT angiography image showing an abnormal, tortuous vessel extending into the cranium (arrowhead). (C) DSA (left panel) and reconstructive image (right panel) showing a PAVF (asterisks) with cranial and caudal drainages (arrowheads). The feeding arteries derive from the C2 radiculomedullary artery and the ASA from the C4 vertebra level. Abbreviations: ASA, anterior spinal artery; C2 and 4, second and fourth cervical vertebrae; CT, computed tomography; DSA, digital subtraction angiography; PAVF, pial arteriovenous fistula; R, right; VA, vertebral artery.
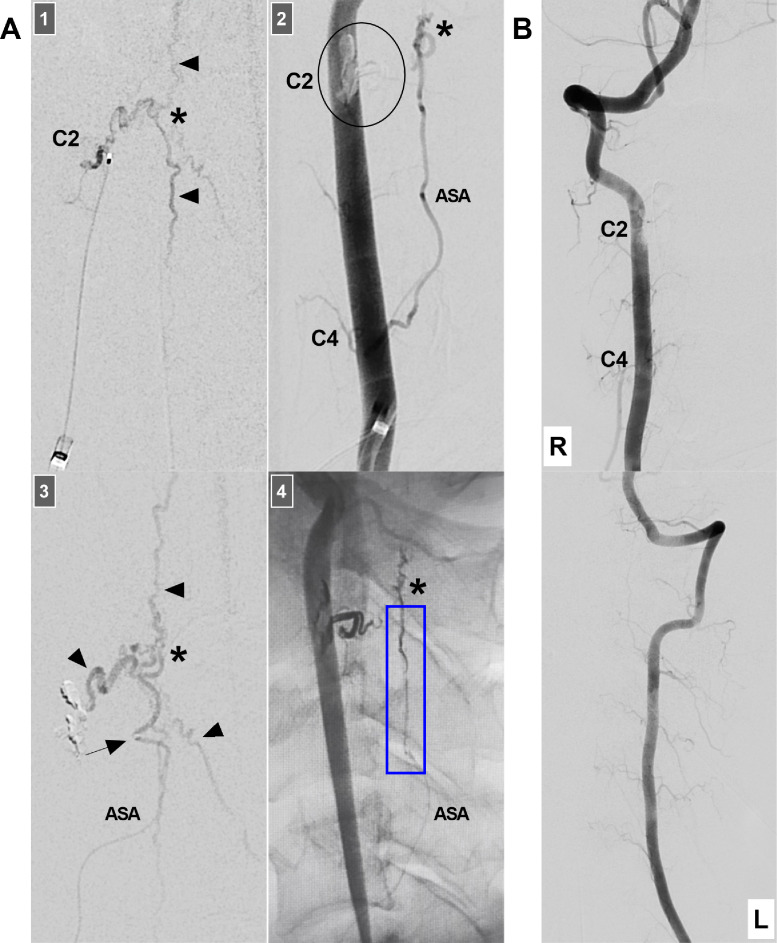
Under general anesthesia, digital subtraction angiography (DSA) revealed a high cervical PAVF at the C2 vertebral level. The PAVF was supplied by the C2 radiculomedullary artery and cervical ASA from the C4 vertebral level and drained into the cervical perimedullary vein system cranially and caudally (Fig. 1C). EVT was subsequently performed. First, the C2 radiculomedullary artery was catheterized with a Marathon microcatheter (Medtronic, Minneapolis, MI, USA). An Onyx-18 liquid embolic system (Medtronic, Irvine, California, USA) was used to occlude the origin of the C2 radiculomedullary artery. Another Marathon microcatheter was then introduced through the cervical ASA to access the PAVF. However, it was unable to reach the shunt point. After the Onyx was cast, the PAVF was penetrated and obliterated. However, Onyx reflux into the distal ASA was observed (Fig. 2).
Fig. 2.
Treatment course. (A) Panel 1: Microcatheter angiography via the C2 radiomedullary artery showing the PAVF (asterisk) and its venous drainages (arrowheads); Panel 2: DSA showing that the ASA from the C4 vertebra still feeds the PAVF (asterisk) after the occlusion of the origin of the C2 radiculomedullary artery by casting Onyx (circle); Panel 3: Microcatheter angiography showing the microcatheter tip (arrow) in the ASA close to but not accessing the PAVF (asterisk), with the arrowheads indicating venous drainages; Panel 4: X-ray image showing the Onyx casting within the PAVF (asterisk) and Onyx reflux in the distal ASA (frame). (B) Upper panel: DSA of the right VA showing obliteration of the PAVF; however, the ASA cannot be observed. Lower panel: DSA of the left VA showing no sign of a PAVF. Abbreviations: ASA, anterior spinal artery; C2 and 4, second and fourth cervical vertebrae; DSA, digital subtraction angiography; L, left; PAVF, pial arteriovenous fistula; R, right; VA, vertebral artery.
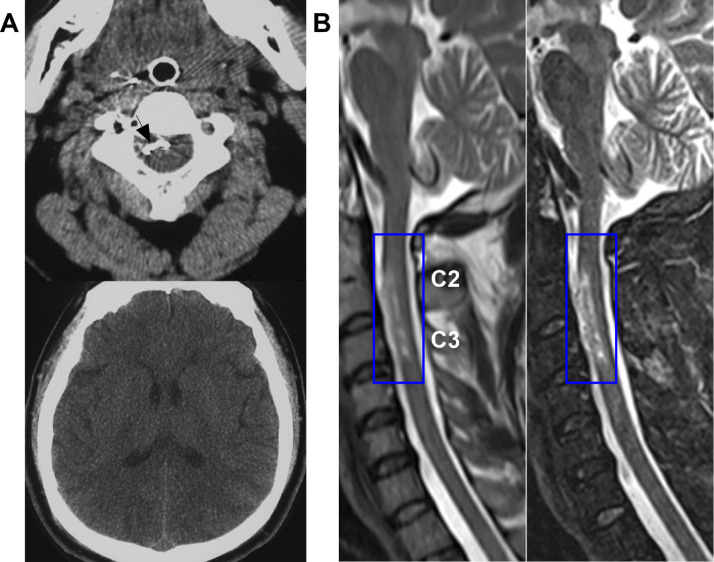
The patient awoke postoperatively and had right hemiparesis with grade III muscle strength. His autonomous respiration was 3–5 times/minute, after which mechanical ventilation was provided. Lumbar puncture drainage was performed to decrease intracranial pressure and drain bloody craniospinal fluid. Three days after EVT, tracheotomy was performed. Four days after EVT, CT revealed a reduction in the SAH (Fig. 3A). The patient’s hemiparesis subsequently began to gradually resolve, and his respiratory function gradually improved. Fifteen days after EVT, the patient’s hemiparesis had completely resolved, and his autonomous respiration frequency increased to 20 breaths/minute.
Fig. 3.
Postoperative images. (A) CT image on the fourth postoperative day showing the Onyx (arrow) in the spinal canal and the absorption of subarachnoid hemorrhage. (B) T1-weighted (left panel) and T2-weighted (right panel) MR images taken on the 30th postoperative day revealed infarction (frames) of the cervical cord at the C2-C3 level. Abbreviations: C2-C3, second-third cervical vertebrae; CT, computed tomography; MR, magnetic resonance.
However, the patient experienced dyspnea at rest, particularly when the patient assumed the supine position. At this time, intermittent assisted mechanical ventilation was provided, and diaphragmatic paralysis was considered. With the patient in the supine position, after mechanical ventilation was stopped, an ultrasound examination was performed, which revealed significantly less respiratory movement of the bilateral diaphragm, leading to a diagnosis of diaphragmatic paralysis. Low-frequency electrical stimulation and respiratory function training were subsequently provided every day. Thirty days after EVT, magnetic resonance imaging revealed an infarction in the high cervical spinal cord at the C2-C3 vertebra level (Fig. 3B), suggesting that diaphragmatic paralysis was caused by injury to the phrenic nerve due to cervical cord infarction. Eighty-seven days after EVT, his autonomous respiration function further improved, and only a portable home ventilator was needed while asleep. After 1 week of observation, the patient had no symptoms of concern and was discharged. However, 93 days after EVT, the patient died while asleep because no portable ventilator was used.
Discussion
High cervical PAVFs are rare type IV spinal cord arteriovenous malformations. Most are located ventral or ventrolateral to the cervical cord. They may have multiple feeding arteries derived from the radiculomedullary artery and/or spinal pial arteries and directly drain into the perimedullary veins [3]. In our case, the feeding arteries included the C2 radiculomedullary artery and the C4 ASA. Owing to hemodynamic stress, PAVFs are often associated with venous varices at the fistula site or the draining vein. In our patient, the draining vein was tortuous, dilated and extended into the cranium. Once a PAVF forms, bleeding or spinal cord dysfunction can occur [4]. In our case, the fistula site ruptured.
For low-flow PAVFs, surgical removal alone or in combination with EVT is a good option. For high-flow PAVFs, EVT alone is often effective [4]. Successful EVT requires penetration of the fistulous points and proximal veinous drainage by a casting liquid embolic agent. In our case, after the minor feeding artery from the C2 radiculomedullary artery was occluded by the casting Onyx, the major feeding artery of the ASA was catheterized by the Marathon microcatheter, after which the PAVF was completely obliterated by casting the Onyx. However, unfortunately, Onyx reflux occluded too much of the distal ASA, and a cervical cord infarction occurred, resulting in severe diaphragmatic paralysis and the patient’s death.
The diaphragm is innervated by the phrenic nerve, which is composed of the C3, C4, and C5 cervical nerves, which are all parts of the cervical plexus. Proper diaphragm function is essential for adequate ventilation and functioning of the lungs. The diaphragm contracts downward to cause negative intrathoracic pressure, allowing lung expansion and passive airflow into the lungs for gas exchange. Therefore, patients with spinal cord injury above the C5 level almost invariably need ventilatory support. Spinal cord injuries below the C5 level result in paralysis of the intercostal and abdominal musculatures; many patients cannot sustain independent ventilation and require ventilatory support [5]. In our patient, respiratory dysfunction occurred due to a spinal cord infarction at the C2-C3 level.
For respiratory dysfunction due to spinal cord injury, high-tidal volume ventilation, high-frequency percussive ventilation, and mechanical insufflation‒exsufflation are helpful for stabilizing the respiratory status of these individuals [6]. However, in our patient, although assisted mechanical ventilation, low-frequency electrical stimulation and respiratory function training were provided daily, the patient’s respiratory function did not recover, and he eventually died. We reported this case to alert neurointerventionists to the lessons learned from experience. Therefore, this case report is valuable.
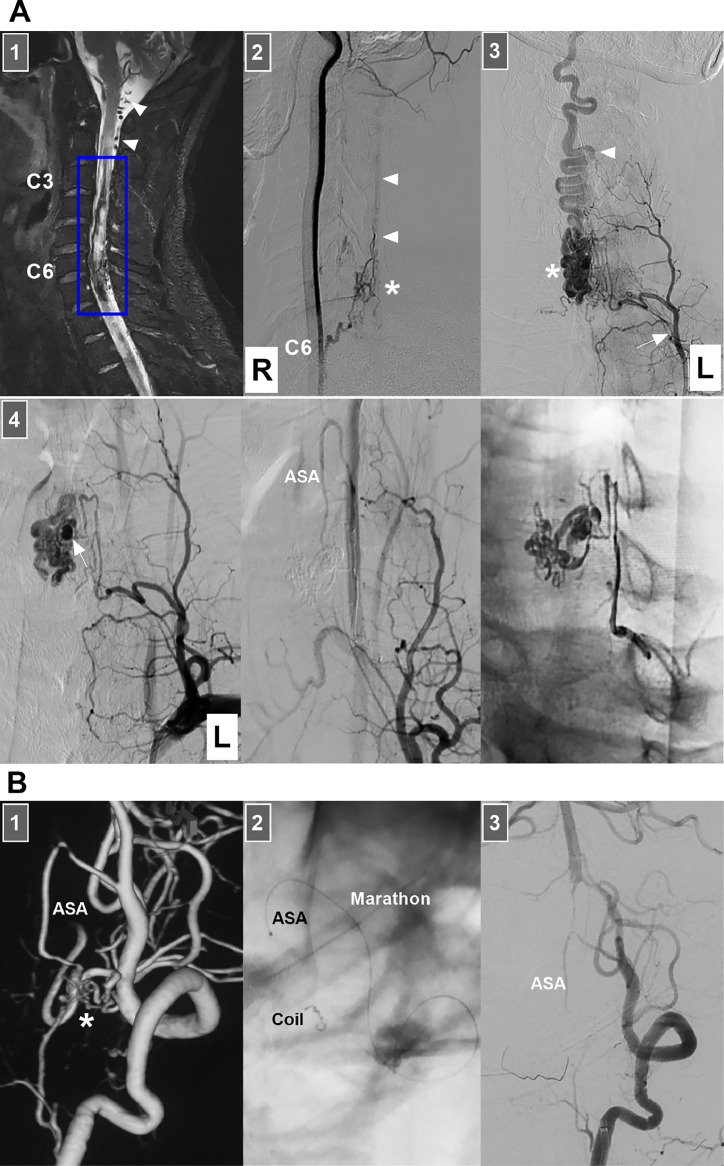
The PAVF described in our case report was fed primarily by the ASA. Consequently, the ASA may not be the most appropriate route of EVT because ASA injury can result in severe ventral cervical spinal cord infarction, disconnecting the descending inspiratory drive of the phrenic nucleus [7]. If embolization must be performed via the ASA, Onyx reflux should be limited; in our patient, unfortunately, the Onyx reflux was too long, and the distal ASA trunk was occluded. If the ventral cervical cord can be partially preserved, severe inspiratory dysfunction can be avoided (Fig. 4A).
Fig. 4.
EVTs for 2 high cervical cord AVFs. (A) EVT in a 35-year-old male. Panel 1: T2-weighted MR image showing hemorrhage (frame) of the cervical cord at the C2–C6 vertebra level; arrowheads indicate dilated veins. Panel 2: DSA of the right VA showing a perimedullary AVF (asterisk) supplied by the C6 radiculomedullary artery with caudal venous drainage (arrowheads). Panel 3: DSA of the left thyrocervical trunk (arrow) showing the ascending cervical artery supplying the AVF (asterisk) with caudal, tortuous venous drainage (arrowhead). Panel 4: Left panel: DSA of the left thyrocervical trunk showing aneurysmal dilation (arrow) in the AVF; Middle panel: DSA showing that the AVF was obliterated by Onyx cast via the ascending cervical artery; Right panel: X-ray image showing the Onyx agent in the AVF and feeding artery. Preoperatively and postoperatively, the patient did not experience respiratory dysfunction. (B) EVT in a 49-year-old male: Panel 1: DSA image showing an AVF ( asterisk) supplied by the ASA; Panel 2: X-ray image showing a detached coil in the distal ASA by the delivery of a Marathon microcatheter; Panel 3: Postoperative DSA showing obliteration of the AVF and preservation of the ASA trunk. Postoperatively, the patient experienced hemiparesis, but 1 month later, he recovered well. Abbreviations: AVF, arteriovenous fistula; ASA, anterior spinal artery; C2-C6, second‒sixth cervical vertebra; DSA, digital subtraction angiography; EVT, endovascular treatment; L, left; MR, magnetic resonance; R, right; VA, vertebral artery.
To reduce Onyx reflux, the “pressure cooker” technique can be used; however, the ASA is too thin to contain a balloon catheter or another microcatheter to coil the distal ASA, making the use of the “pressure cooker” technique in the ASA difficult. To avoid ASA trunk injury from Onyx reflux, only coiling the distal ASA with a Marathon microcatheter may be helpful (Fig. 4B). However, coiling of the feeding artery alone may result in partial retention of the PAVF. When the ASA acts as the only feeding artery, it is helpful to use a flow diverter to cover the ASA orifice to occlude the ASA chronically. In 2024, Albiña-Palmarola et al. reported this novel technique for treating a craniocervical junction perimedullary AVF; they used multiple flow diverters to cover the origin of the ASA over 2 sessions, completely occluding the ASA and reorganizing the blood flow of the cervical spinal cord to obliterate the AVF [8]. However, we did not use this nonroutine technique.
Conclusion
For high cervical PAVFs, when embolization is performed via the ASA, excessively long Onyx reflux in the ASA can result in ventral cervical cord infarction above the C4 vertebra level and phrenic nerve injury, which may cause disastrous diaphragmatic paralysis and even patient death. We reported such a case to alert neurointerventionists to this potential complication.
Ethics approval and consent to participate
Ethics approval was not needed at our institution, as the manuscript was a case report.
Patient consent
Written informed consent was obtained from the family of the patient and the patients involved for publication in this case report and any accompanying images. Written consent is available for review by the Editor-in-Chief of this journal.
Footnotes
Competing Interests: The authors declare no competing interests.
Acknowledgments: No funding was received.
References
- 1.Su H., Yu J. Treatment of high cervical arteriovenous fistulas in the craniocervical junction region. Front Neurol. 2023;14 doi: 10.3389/fneur.2023.1164548. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Ji T., Guo Y., Shi L., Yu J. Study and therapeutic progress on spinal cord perimedullary arteriovenous fistulas. Biomed Rep. 2017;7(3):214–220. doi: 10.3892/br.2017.951. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Meng X., Zhang H., Wang Y., Ye M., He C., Du J., et al. Perimedullary arteriovenous fistulas in pediatric patients: clinical, angiographical, and therapeutic experiences in a series of 19 cases. Childs Nerv Syst. 2010;26(7):889–896. doi: 10.1007/s00381-009-1071-8. [DOI] [PubMed] [Google Scholar]
- 4.Sasamori T., Hida K., Yano S., Asano T., Iwasaki Y. Cervical perimedullary arteriovenous fistula in an infant presenting with subarachnoid hemorrhage–case report. Neurol Med Chir (Tokyo) 2008;48(9):409–413. doi: 10.2176/nmc.48.409. [DOI] [PubMed] [Google Scholar]
- 5.Charco-Roca L.M., Simón-Polo E., PC Cuesta-Montero. Ultrasound evaluation of diaphragm function in patients with cervical spinal cord injury: case report. Braz J Anesthesiol. 2024;74(1) doi: 10.1016/j.bjane.2021.12.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Wong S.L., Shem K., Crew J. Specialized respiratory management for acute cervical spinal cord injury: a retrospective analysis. Top Spinal Cord Inj Rehabil. 2012;18(4):283–290. doi: 10.1310/sci1804-283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Warren P.M., Alilain WJ. The challenges of respiratory motor system recovery following cervical spinal cord injury. Prog Brain Res. 2014;212:173–220. doi: 10.1016/B978-0-444-63488-7.00010-0. [DOI] [PubMed] [Google Scholar]
- 8.Albiña-Palmarola P., Khanafer A., El Mekabaty A., Forsting M., Ganslandt O., Henkes H. A ruptured craniocervical junction perimedullary arteriovenous fistula successfully treated through flow diversion: a case report. Surg Neurol Int. 2024;15:381. doi: 10.25259/SNI_631_2024. [DOI] [PMC free article] [PubMed] [Google Scholar]