Abstract
Objectives
G protein-coupled receptors (GPCRs) are the most druggable targets in biology due to their cell-type specificity, ligand binding, and cell surface accessibility. Underscoring this, agonists for GPCRs have recently revolutionized the treatment of diabetes and obesity. The rampant success of these compounds has invigorated interest in identifying additional GPCRs that modulate appetite and body weight homeostasis. One such potential therapeutic target is G-protein couped receptor 45 (Gpr45), an orphan GPCR expressed both centrally and peripherally. We aimed to explore the role of Gpr45 as well as neurons expressing Gpr45 in energy balance.
Methods
Three novel transgenic mouse models were engineered to investigate the functional contribution of Gpr45 to body weight and appetite regulation: 1) a global Gpr45 knockout, 2) a conditional floxed Gpr45 allele, and 3) a Gpr45-CreERT2 knock-in. Metabolic profiling was performed in global Gpr45 knockout animals including body weight, food intake, body mass, energy expenditure, and body temperature measurements. Animals harboring a conditional floxed Gpr45 allele were bred to mice expressing Cre-recombinase in excitatory neurons labeled via Vesicular glutamate transporter 2 (Vglut2), inhibitory cells expressing Vesicular GABA transporter (Vgat), or neurons marked by the transcription factor Single-minded 1 (Sim1) and monitored for body weight and food consumption. Additionally, floxed Gpr45 mice were bilaterally injected with AAV-Cre targeting the paraventricular nucleus of the hypothalamus (PVH) and body weight and food intake were evaluated. The Gpr45-CreERT2 knock-in model was used to express chronic and acute actuators to the PVH to assess the role of PVHGpr45 neurons in energy homeostasis.
Results
Global Gpr45 disruption caused marked weight gain, increased food intake and fat mass, but no detectable alterations in core temperature or energy output. Selective deletion of Gpr45 from Sim1+ or excitatory Vglut2+ but not inhibitory Vgat+, neurons produced obesity and hyperphagia. Targeted deletion of Gpr45 from the PVH phenocopies these metabolic changes suggesting a major site of action of Gpr45 signaling is glutamatergic neurons residing in the PVH. Tetanus toxin light chain (TeNT) was used to permanently silence PVHGpr45 neuronal activity in Gpr45-CreER mice leading to rapid weight accumulation and escalated food intake. These experiments highlight the critical role of both Gpr45 signaling and neural network activity in the regulation of body weight and appetite. A mutated version of the bacterial sodium channel, NaChBac, was used to constitutively activate PVHGpr45 neuronal activity in Gpr45-CreER mice with limited to no effect on body weight and food consumption, implicating redundant circuitry acting in concert to bias weight loss protection. Acute chemogenetic stimulation of PVHGpr45 neurons durably suppressed food intake regardless of caloric need state or food palatability demonstrating the capacity of these cells to curb appetite.
Conclusions
Gpr45 is a putative therapeutic candidate that could be targeted to combat obesity and overeating.
Keywords: G protein-coupled receptor 45, body weight regulation, food intake, behavioral neuroscience
Highlights
-
•
Global Gpr45 disruption causes massive weight gain, increased food intake and fat mass, but no detectable alterations in core temperature or energy output.
-
•
Selective deletion of Gpr45 from Sim1+ or excitatory Vglut2+ but not inhibitory Vgat+, neurons produce obesity and hyperphagia.
-
•
Targeted PVH deletion of Gpr45 phenocopies these metabolic changes.
-
•
Silencing PVHGpr45 neuronal activity leads to rapid weight accumulation and escalated food intake.
-
•
Constitutive PVHGpr45 activation has limited to no effect on body weight and food consumption.
-
•
Stimulation of PVHGpr45 neurons durably suppresses food intake regardless of caloric need state or food palatability.
1. Introduction
Anti-obesity medications, spearheaded by glucagon-like peptide-1 receptor agonists (GLP-1RAs), have revolutionized the treatment of metabolic disorders, particularly obesity and type 2 diabetes, by demonstrating significant efficacy in reducing body weight, improving glycemic control, and enhancing cardiovascular outcomes [1,2]. These drugs work by enhancing insulin secretion, suppressing appetite, and slowing gastric emptying, highlighting the critical role of G protein-coupled receptor (GPCR) signaling in metabolic regulation [1]. The success of GLP-1RAs underscores both the success and increasing potential of targeting GPCRs in drug development, especially orphan GPCRs, whose endogenous ligands and functions remain unknown. Directing ligands and small molecules to orphan GPCRs offers an untapped avenue for therapeutic innovation, as these receptors may regulate diverse physiological processes, including appetite control, energy expenditure, and glucose homeostasis. Developing drugs that modulate orphan GPCRs could complement existing treatments like GLP-1RAs, offering new mechanisms to tackle obesity and metabolic diseases, especially in patients who are resistant or unresponsive to current therapies.
G protein-coupled receptor 45 (Gpr45) is an orphan receptor predominantly expressed in the central nervous system, with notable expression in the hypothalamus (https://www.proteinatlas.org/ENSG00000135973-GPR45/brain/hypothalamus). Although its endogenous ligand remains unidentified, emerging research suggests that Gpr45 plays a significant role in regulating energy homeostasis and body weight. Studies have indicated that Gpr45 may influence neuronal pathways associated with appetite control and metabolic processes. Recent investigations have demonstrated that disruption of the Gpr45 gene in mice causes [3,4] increased adiposity, glucose intolerance, and hepatic steatosis. These metabolic anomalies are accompanied by a reduction in hypothalamic pro-opiomelanocortin (POMC) expression, a critical protein involved in appetite suppression and energy expenditure [3]. POMC neuronal activation drives weight loss through diminished food consumption [5,6]. Thus, Gpr45 may positively regulate POMC signaling pathways, thereby influencing energy balance.
The studies referenced above suggests Gpr45 activation interacts with the melanocortin system, however, the site of Gpr45 action in governing body weight homeostasis is unclear. In addition to its high brain expression, Gpr45 has been detected in the periphery including the stomach, liver, kidneys, thyroid gland, and testis (https://www.proteinatlas.org/ENSG00000135973-GPR45/tissue, https://celltypes.brain-map.org/rnaseq/mouse_ctx-hpf_10x?selectedVisualization=Scatter+Plot&colorByFeature=Gene+Expression&colorByFeatureValue=Gpr45, http://dropviz.org/) [7,8]. Determining the area of influence mandates a genetic approach to downregulate GPCR expression in either molecularly defined cells and/or anatomical structures. Thus, mice harboring conditional floxed alleles of the GPCR of interest are required to establish the necessity of GPCR signaling in the brain. This strategy has been employed to pinpoint the site of action of both GLP-1R and melanocortin-4 receptor (Mc4R) in metabolic regulation [[9], [10], [11], [12], [13], [14]]. Here, we engineered a conditional floxed Gpr45 model and found that perturbing signaling in excitatory neurons marked by Vesicular glutamate transporter 2 (Vglut2), but not inhibitory cells expressing Vesicular GABA transporter (Vgat), results in robust weight gain and hyperphagia. Furthermore, selective Gpr45 deletion from the paraventricular nucleus of the hypothalamus (PVH), a predominantly glutamatergic region [15], mostly recapitulates this positive energy balance.
While manipulating receptor levels isolate the molecular effects of a single protein, neuron-focused experiments investigate how those effects translate into coordinated neural activity and behavior. A host of neuroscience techniques can be utilized to either chronically or acutely manipulate neural activity as subsequent putative alterations in behavior and/or physiology are monitored [16,17]. These approaches generally rely on specific targeting of viral payloads to cells labeled by Cre-recombinase, requiring a transgenic mouse line expressing Cre under the endogenous regulatory elements of a gene of interest. This strategy has been used to determine the role of both GLP-1R- and Mc4R-expressing PVH neurons in the regulation of appetite and body weight [[18], [19], [20], [21]]. To explore how PVHGpr45 neurons fit into larger neural networks and their overall contribution to energy balance, we generated a Gpr45-CreER knock-in model. These mice allowed us to direct tools to either acutely or chronically stimulate or inhibit PVHGpr45 neurons to determine their functional role in modulating metabolic homeostasis. We found that prolonged silencing of PVHGpr45 cells significantly increased body weight and food intake, while chronic activation failed to change these parameters, supporting a role for redundant signaling pathways in maintaining appropriate food consumption and fat stores obligatory for survival [21]. Notably, we found that acute PVHGpr45 stimulation strongly suppressed eating irrespective of caloric need state or food palatability, demonstrating the ability of these neurons to inhibit consummatory behavior. Collectively, we have refined our understanding of the anatomical expression and functional contribution of Gpr45 that could potentially be leveraged to therapeutically curb appetite and obesity.
2. materials and methods
2.1. Experimental animals
All animal protocols and procedures were approved by the National Institute of Health Animal Care and Use Committee. All animal experiments performed at Pfizer were in accordance with federal, state, local and institutional guidelines governing the use of laboratory animals in research. The procedures used in this study were reviewed and approved by Pfizer's Institutional Animal Care and Use Committee. The facilities that supported all animal studies are fully accredited by the Association for Assessment and Accreditation of Laboratory Animal Care International. Mice were housed with a 12-h light/dark cycle and provided ad libitum access to food (standard chow diet (SD), Envigo 7017 NIH-31) and water unless otherwise noted. All experiments were carried out in adult (>8 weeks) mice that were group-housed until experiments began.
Vglut2-ires-Cre [15], Vgat-ires-Cre [15], Sim1-Cre [22], AgRP-ires-Cre [23] and B6.Cg-Gt(ROSA)26Sortm14(CAG-tdTomato)Hze/J (Ai14-tdTom) [24] mice were generated and maintained as previously described. All mice are on a C57BL/6J background and were used as heterozygotes.
2.1.1. Generation of Gpr45 KO mice
Mice with loss of Gpr45 function (Gpr45 KO) were generated at the University of Texas Southwestern Medical Center using CRISPR/Cas9 technology. Knockout mice were confirmed to have a 1 bp insertion within the exon 2 of the Gpr45 gene resulting in a frame shift mutation. Genotyping was performed by amplifying the region containing the mutation using PCR, followed by sequencing of the amplified region to detect the mutation. Littermate C57BL/6J wild-type and Gpr45 KO mice were housed individually in Innovive cages (Innorack IVC Mouse 3.5) in a temperature- and humidity-controlled environment (room temperature (22 ± 1 °C) or 27 ± 1 °C, as indicated), maintained on a 12-h light, 12-h dark cycle with ad libitum access to water and food (Teklad 2920).
Genotyping primers.
| Primer | Sequence (5′ to 3′) |
|---|---|
| Forward primer | TCAGGCTTCCAATATCTGATTCCTCT |
| Reverse primer | GCTGAAAGCCGACAAAAGTGGT |
| Sequencing primer | AGAAACAGGACAAGGATGTGACTGATG |
2.1.2. Generation of Gpr45 flox/flox mice
Gpr45 flox/flox mice were generated using CRISPR/Cas9 technology. LoxP sites were inserted in frame with the mouse Gpr45 locus 254 bp upstream of exon 2 and 35 bp downstream of the stop codon. LoxP sites were introduced by microinjecting sgRNAs, oligo donors containing loxP sequences and Cas9 protein (IDT cat#1081059) into one cell-stage C57BL/6J embryos. Embryos were implanted to pseudo-pregnant female mice to develop to term. Insertion of loxP sites was validated by genotyping and DNA sequencing. Selected founders were bred to C57BL/6J mice.
Genotyping primers.
| Primer | Sequence (5′ to 3′) |
|---|---|
| 5'loxP forward primer | TATCCCTGTCCTCTTGAAGCTG |
| 5'loxP reverse primer | TTCATCAGTCACATCCTTGTCC |
| Sequencing primer | GTCCTGTTTCTGTGGTAGCC |
| Primer | Sequence (5′ to 3′) |
|---|---|
| 3'loxP forward primer | GCAGGTGCTTTGCTCAAGG |
| 3'loxP reverse primer | ATGCTGGCATCAGAGTGAAC |
| Sequencing primer | TGCCACACTCAGTCTACAGC |
2.1.3. Generation of Gpr45-CreERT2 mice
Gpr45-CreERT2 knock-in model was developed using CRISPR/Cas9 technology. A T2A-CreERT2 cassette was inserted at the stop codon of the mouse Gpr45 locus. Single guide RNAs (sgRNAs) containing sequences 5′-CGAAAACCAATCCGCTGTCT-3′ and 5′-CTGACCTGCTCCCTAGACAG-3′, pGEMTeasy donor plasmid containing GPR45-T2A-CreERT2 sequence, and Cas9 protein (IDT cat#1081059) were delivered to one cell-stage C57BL/6J embryos by microinjection. Embryos were implanted to pseudo-pregnant female mice to develop to term. Insertion of CreERT2 cassette was confirmed by genotyping and DNA sequencing. Selected founders were bred to C57BL/6J mice.
Genotyping primers.
| Primer | Sequence (5′ to 3′) |
|---|---|
| Gpr45 forward primer | ACCTCAGACAGCTGACCG |
| Gpr45 reverse primer | TGAAAGCCACAGATCACCTG |
| Cre reverse primer | TTCGAACGCTAGAGCCTG |
2.2. Body weight and food intake measurements
For prolonged body weight and food intake studies, experimental and control mice were singly housed for accurate measurements. Animals and food were weighed weekly. Food was provided ad libitum. The weekly food consumption total was then divided by 7 to calculate the average daily food intake for each animal. In designated experiments, animals were provided 60 kcal % lard-based high-fat diet (60% HFD, Research Diets D12492).
2.3. Energy expenditure measurements
Whole-body energy metabolism was evaluated using a comprehensive laboratory animal monitoring system home cage (CLAMS-HC) (Columbus Instruments, Columbus, OH, USA) following manufacturer's protocol in 16-week-old littermate C57BL/6J wild-type and Gpr45 KO mice.
2.4. Body composition measurements
Body composition was analyzed using EchoMRI in 16-week-old littermate C57BL/6J wild-type and Gpr45 KO mice at 27 °C.
2.5. Body temperature measurements
Core body temperature was measured during light phase at 27 °C using a rectal thermometer in 16-week-old littermate C57BL/6J wild-type and Gpr45 KO mice.
2.6. RNA analysis
Male and female mice, 5–6 months old, were euthanized with isoflurane. Brains were removed and put on an ice-chilled brain matrix, and 1 mm coronal brain slices were cut with a razor blade and transferred to a microscope slide. The mediobasal hypothalamus with a specific focus on the PVH (from Bregma −0.3 to 1.3) were microdissected with a scalpel and immediately frozen on dry ice and stored at −80 °C until processed for RNA. RNA from microdissected tissue was isolated as reported [25] and quantitated using QuantStudio 7 Flex Real-Time PCR System (Applied Biosystems), normalized to TATA-box binding protein (Tbp). The primers are as follows: Gpr45, x966: (5′-GAAGTGACAAGTCCCCTGGG) and (x967: 5′-GTGCCATCCAGCCTTTCTCT); Brs3, x575 (5′-GCACCCTGAACATACCGACT) and x576 (5′-ACAGGAGATGATTCGGCAAC); Mc4r, x581 (5′-ATCTGTAGCTCCTTGCTCGC) and x582 (5′-TGCAAGCTGCCCAGATACAA).
2.7. Brain tissue preparation and imaging
Mice were terminally anesthetized with chloral hydrate (Sigma Aldrich) and transcardially perfused with phosphate-buffered saline (PBS) followed by 10% neutral buffered formalin (Fisher Scientific). Brains were extracted, cryoprotected in 20% sucrose, and sectioned coronally on a freezing sliding microtome (Leica Biosystems) at 50 μm and collected in four equal series. Sections were mounted onto gelatin-coated slides and fluorescent images were captured with an Olympus VS120 slide scanner microscope.
2.8. Multiplex in situ hybridization preparation and imaging
Samples were prepared as previously described [26]. Briefly, mice were anesthetized with isoflurane and decapitated. Brains were then extracted from mice, frozen on dry ice, and stored at −80 °C until they were sliced using a Cryostat (Leica Biosystems) at 14-μm thickness at −20 °C and mounted on microscope slides. Multiplex RNAscope Fluorescent kit v2 (Advanced Cell Diagnostics) was used according to manufacturer's instructions with the following probes: Mm-Gpr45, Mm-Slc17a6-C2, and Mm-Slc32a1-C3. Samples were imaged using a confocal microscope (LSM700 Zeiss) with a 20X objective. Images were processed on ImageJ 1.54f by removing the background noise. Signal quantification for each probe per cell was performed using CellProfiler v4.2.8 [27].
2.9. Viral injections
Stereotaxic injections were performed as previously described [28]. Mice were anaesthetized with isoflurane and placed into a stereotaxic apparatus (Stoelting Just for Mice). For postoperative care, mice were injected intraperitoneally with meloxicam (0.5 mg per kg). After exposing the skull via small incision, a small hole was drilled for injection. A pulled-glass pipette with 20–40 mm tip diameter was inserted into the brain and virus was injected by an air pressure system. A micromanipulator (Grass Technologies, Model S58 Stimulator) was used to control injection speed at 25 nl min−1 and the pipette was withdrawn 5 min after injection. For Gpr45 deletion experiments, AAV9-hSyn-Cre-P2A-dTomato (Addgene, 107738), AAV9.CMV.HI.eGFP-Cre.WPRE.SV40 (Addgene 105545), or AAV9.CMV.PI.EGFP.WPRE.bGH (Addgene 105530) was bilaterally injected into the PVH (20–40 nl, bregma: AP: −0.70 mm, DV: −4.70 mm, L: +/−0.20 mm). For chronic neural perturbation experiments AAV-DJ-CMV-DIO-eGFP-2A-TeNT was bilaterally injected into the PVH (20–40 nl, bregma: AP: −0.70 mm, DV: −4.70 mm, L: +/−0.20 mm). For chronic constitutive neural activity experiments AAV9-EF1a-Flex-EGFP-P2A-mNaChBac [29] was bilaterally injected into the PVH (20–40 nl, bregma: AP: −0.70 mm, DV: −4.70 mm, L: +/−0.20 mm). For chemogenetic experiments, AAV8-hSyn-DIO-hM3dq (Addgene, 44361) was bilaterally injected into the PVH (20–40 nl, bregma: AP: −0.70 mm, DV: −4.70 mm, L: +/−0.20 mm).
2.10. Drugs
Clozapine N-oxide (CNO, Tocris, 4936) was dissolved in saline at stock concentrations and stored at −20 °C until use. For in vivo experiments, on the day of testing, the CNO stock solution was thawed, diluted with saline, and delivered intraperitoneally (i.p.) at a volume of 10 ml/kg at a 1.0 mg/kg dose.
Cre-mediated recombination in Gpr45-CreER animals was achieved via tamoxifen treatment (110 mg/kg i.p. in corn oil, Sigma Aldrich) daily for 5 consecutive days as previously described [30]. Mice were used for studies ∼2 weeks following the last tamoxifen dose.
2.11. Chemogenetic food intake studies
Food intake studies on chow were performed as previously described [20,28]. Animals were singly housed for at least 2.5 weeks following surgery and handled for 10 consecutive days before the assay to reduce stress response. Studies were conducted in a home cage environment with ad libitumfood access. A full trial consisted of assessing food intake from the study subjects after they received injections of saline or CNO (0.3 mg/kg) in a crossover design across the course of 3 days to ensure the effects of CNO-mediated stimulation had dissipated. Mice with ‘missed’ injections or expression outside the area of interest were excluded from analysis after post hoc examination of mCherry expression. In this way, all food intake measurements were randomized and blind to the experimenter.
Light-cycle hM3Dq ad libitum feeding studies were conducted between 9:00am to 1:00pm (zeitgeber time (ZT) 3h–7h) and food intake was monitored, 1 h, 2 h, 3 h and 4 h after i.p. injection. For post-fast refeed hM3Dq studies, animals were fasted overnight at 5:00pm (ZT 11h) and food returned the following morning at 9:00am (ZT 3h). Food intake was monitored 1 h, 2 h, 3 h and 4 h after i.p. injection. CNO or saline was injected 30 min before food was introduced.
2.12. Electrophysiology
To verify the functionality of NaChBac channels, the virus AAV9-EF1a-Flex-EGFP-P2A-mNaChBac was injected and expressed unilaterally in the PVH of adult male Gpr45-CreERT2::Ai14td-Tom reporter mice. Eight to twelve days after intracranial injection, whole-cell patch clamp recordings were made in the PVH. Brain slices were obtained and stored at 30 °C for 1 h in a heated, oxygenated artificial cerebrospinal fluid (aCSF) chamber, containing (in mM) 126 NaCl, 2.5 KCl, 2.4 CaCl2, 1.2 MgSO4, 1.2 NaH2PO4, 10.0 glucose, 21.4 sodium bicarbonate, pH 7.4, 305–312 mOsmol. Following incubation, brain slices were then transferred to a submerged recording chamber maintained at 30 °C (Warner Instruments). Recording electrodes (3.2–5.5 MΩ) were pulled with a Flaming-Brown Micropipette Puller (Sutter Instruments) using thin-walled borosilicate glass capillaries. Electrodes were filled with an intracellular recording solution containing (in mM) 130 KGluconate, 10 KCl, 0.3 CaCl2, 1 MgCl2, 10 HEPES, 1 EGTA, 10 Na-Phosphocreatine, 3 Mg-ATP, 0.3 Na-GTP, pH to 7.4, 290–295 mOsmol. In current clamp mode, action potentials, membrane properties, and rheobase were recorded. In voltage clamp mode, input resistance and action potentials in depolarization steps were recorded. For all electrophysiology experiments, signals were acquired via a Multiclamp 700B amplifier (Molecular Devices), digitized at 20 kHz, filtered at 3 kHz, and analyzed using Clampfit 11.4 software (Molecular Devices). Liquid junction potential was accounted for in all data analysis.
2.13. Single-cell RNA sequencing bioinformatics analysis
Single-cell RNA sequencing data of the arcuate nucleus (ARC) from our previous study [31] was analyzed using the R Package Seurat v4 (https://satijalab.org/seurat/) [32]. Cells from all mice (fed and fasted virgin and lactating females, 14 weeks old) were integrated using RPCA and normalized using SCTransform. Cells were first classified as neurons using well-established neuronal marker genes via DropViz (http://dropviz.org) [33], and only neurons were used for clustering at 0.5 resolution. Clusters were then filtered based on quality control metrics (mitochondrial/erythrocyte gene expression and sequencing saturation), high expression of select neuronal markers, and low expression of select non-neuronal markers. Cluster marker genes were identified using Seurat v4. Initial clustering revealed two agouti-related peptide (Agrp)+ clusters that showed a high degree of similarity, which were then combined into a single cluster.
3. Results
3.1. Generation and metabolic phenotyping of global Gpr45 knockout mouse model
Recent work found that disruption of Gpr45 via a random, single-copy piggyBac insertion resulted in obesity primarily due to reduced energy expenditure [3]. Despite the significant weight gain in these mutants, food intake was surprisingly not affected at the early stages of obesity. To better understand the metabolic function of this orphan GPCR and explore its potential contribution to appetite regulation, we generated a global Gpr45 knockout line employing a site-directed CRISPR-Cas9 platform. Knockout (KO) mice were confirmed to have a 1 bp insertion within the only coding exon of the Gpr45 locus resulting in a frameshift mutation.
Gpr45 KO mice and age/sex-matched wildtype (WT) controls had comparable body weights at and up to 6 weeks post-weaning. Afterwards the body weight of Gpr45 KO mice quickly diverged from WT controls on a standard chow diet (SD), leading to considerable weight gain (more than double) by 15 weeks of age (Figure 1A). This elevated body weight was strongly driven by a substantial increase in SD food intake (Figure 1B). Both fat mass and lean mass were greater in Gpr45 KO versus WT groups (Figure 1C). To avoid the confounding effects that temperature stress can have on metabolism and energy balance, we ran another cohort of Gpr45 KO and age/sex-matched WT controls at 27 °C and again observed rapid weight accumulation in the Gpr45 KO mice compared to controls (Figure 1D). Core body temperature at 27 °C between the groups was comparable (Figure 1E). Energy expenditure in the Gpr45 KO mice was as expected for their body weight (Figure 1F).
Figure 1.
Metabolic phenotyping of global Gpr45 knockout mouse model.
(A) Gpr45 knockout (KO) mice gain significantly more weight at room temperature on standard chow diet than age/sex-matched wildtype (WT) controls (n = 23 Gpr45 KO, n = 29 WT, mixed sex; repeated measures two-way ANOVA followed by Šídák's multiple comparison test). (B) Gpr45 knockout (KO) mice consume significantly more standard chow diet than age/sex-matched wildtype (WT) controls (n = 6 Gpr45 KO, n = 8 WT; repeated measures two-way ANOVA followed by Šídák's multiple comparison test). (C) Gpr45 knockout (KO) mice accrue significantly more fat and lean mass on standard chow diet than age/sex-matched wildtype (WT) controls (n = 10 Gpr45 KO, n = 12 WT; unpaired t test, two-tailed). (D) Gpr45 knockout (KO) mice gain significantly more weight at 27 °C on standard chow diet than age/sex-matched wildtype (WT) controls (n = 10 Gpr45 KO, n = 12 WT, mixed sex; repeated measures two-way ANOVA followed by Šídák's multiple comparison test). (E) Gpr45 knockout (KO) mice and age/sex-matched wildtype (WT) controls exhibit comparable core body temperatures (n = 8 Gpr45 KO, n = 6 WT; unpaired t test, two-tailed). (F) Gpr45 knockout (KO) mice and age/sex-matched wildtype (WT) controls exhibit comparable total energy expenditure values considering body weight differences (n = 6 Gpr45 KO, n = 8 WT; simple linear regression). All error bars represent s.e.m. ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001.
3.2. Generation and validation of a conditional Gpr45 floxed mouse model
Given the severe obesity phenotype observed in the global Gpr45 KO animals, we sought to determine the central role of Gpr45 in modulating energy balance. Single-cell RNA sequencing data sets and in situ hybridizations suggest widespread Gpr45 expression in the brain (https://celltypes.brain-map.org/rnaseq/mouse_ctx-hpf_10x?selectedVisualization=Scatter+Plot&colorByFeature=Gene+Expression&colorByFeatureValue=Gpr45, http://dropviz.org/) [7,8]. Concordantly, we detected high levels of Gpr45 transcript throughout the hypothalamus and hindbrain in both Vglut2 excitatory and Vgat inhibitory cells (Figure S1). To this end, we engineered Gpr45 flox/flox mice using CRISPR/Cas9 technology. LoxP sites were inserted in the mouse Gpr45 locus 254 basepairs upstream of exon 2 and 35 basepairs downstream of the stop codon (Figure S2A). Firstly, to validate the efficacy of this line, we deleted Gpr45 from all excitatory or inhibitory neurons marked by the expression of Vesicular glutamate transporter 2 (Vglut2) or Vesicular GABA transporter (Vgat) using Vglut2-ires-Cre (Vglut2-ires-Cre; Gpr45 flox/flox) or Vgat-ires-Cre mice (Vgat-ires-Cre; Gpr45 flox/flox), respectively. Quantitative real time-polymerase chain reaction was employed to assess Gpr45 transcript levels, and two other hypothalamic GPCRs, Brs3 or Mc4r, as controls in the mediobasal hypothalamus. We detected significantly lower Gpr45 RNA in both Vglut2-ires-Cre; Gpr45 flox/flox and Vgat-ires-Cre; Gpr45 flox/flox animals compared to Gpr45 flox/flox controls (Figure S2B-C). Importantly, we found that this manipulation was specific to Gpr45 and had no effect on Brs3 or Mc4r transcript levels, other hypothalamic GPCRs (Figure S2B-C). The residual Gpr45 transcript levels in each knockout model are likely due to the high hypothalamic expression in both subtypes (Figure S1A-B).
3.3. Selective deletion of Gpr45 from excitatory, hypothalamic (Vglut2 and/or Sim1) neurons results in obesity and hyperphagia
To uncover the neuronal contribution of Gpr45 to energy balance as well as ascertain the neurochemical identity of putative cell-types expressing Gpr45 that are necessary for body weight and appetite regulation, we assessed the bodyweight and food intake effects of selective deletion of Gpr45 in Vglut2-ires-Cre; Gpr45 flox/flox and Vgat-ires-Cre; Gpr45 flox/flox animals (Figure 2A,D) [15]. Experimental animals were heterozygous for the relevant Cre and homozygous for the Gpr45 floxed allele, while controls were either heterozygous for the relevant Cre or homozygous for the Gpr45 floxed allele. Genetically defined deletion of Gpr45 in glutamatergic neurons (Figure 2A) of both male and female mice resulted in a significant increase in body weight on SD (Figure 2B), which was associated with marked hyperphagia (Figure 2C). However, selective deletion of Gpr45 in GABAergic neurons (Figure 2D) of both male and female mice had no effect on body weight (Figure 2E) or SD food intake (Figure 2F). These results demonstrate that Gpr45 on Vglut2+, but not Vgat+, neurons are necessary for the appropriate maintenance of body weight and food consumption.
Figure 2.
Selective deletion of Gpr45 from Vglut2+ excitatory hypothalamic neurons results in obesity and hyperphagia.
(A) Expression pattern of anatomical brain regions marked by Vesicular glutamate transporter 2 (Vglut2) where Gpr45 is deleted including the cerebral cortex (CTX), piriform cortex (PIR), paraventricular nucleus of the hypothalamus (PVN), lateral olfactory tract (LOT), thalamus (TH), basal lateral amygdala (BLA), posterior hypothalamus (PH), periaqueductal grey (PAG), dorsal raphe (DR), lateral parabrachial nucleus (LPB), and nucleus of the solitary tract (NTS). (B–C) Mice with specific deletion of Gpr45 from excitatory Vglut2+ neurons (B) gain significantly more weight and (C) consume significantly more standard chow diet than age/sex-matched heterozygous controls (n = 8 Vglut2-ires-Cre; Gpr45 flox/flox, n = 7 Vglut2-ires-Cre, n = 6 Gpr45 flox/flox, mixed sex; repeated measures two-way ANOVA followed by Tukey's multiple comparison test). (D) Expression pattern of anatomical brain regions marked by Vesicular GABA transporter (Vgat) where Gpr45 is deleted, including the caudate putamen (CP), nucleus accumbens (ACB), olfactory tubercle (TU), lateral septum (LS), bed nucleus of the stria terminalis (BST), suprachiasmatic nucleus (SCh), arcuate nucleus of the hypothalamus (ARC), central amygdala (CeA), substantia nigra pars reticulata (SNr), superior colliculus (SC), and dorsal cochlear nucleus (DC). (E–F) Mice with specific deletion of Gpr45 from inhibitory Vgat + neurons exhibit comparable (E) body weight and (F) standard chow diet intake as age/sex-matched heterozygous controls (n = 8 Vgat-ires-Cre; Gpr45 flox/flox, n = 6 Vgat-ires-Cre, n = 6 Gpr45 flox/flox, mixed sex; repeated measures two-way ANOVA followed by Tukey's multiple comparison test). (G) Expression pattern of anatomical brain regions marked by Single-minded 1 (Sim1) where Gpr45 is deleted including the paraventricular nucleus of the hypothalamus (PVH), nucleus of lateral olfactory tract (NLOT), medial amygdala (MeA), and dorsal motor vagus (DMV). (H–I) Mice with specific deletion of Gpr45 from excitatory Sim1+ neurons (H) gain significantly more weight and (I) consume significantly more standard chow diet than age/sex-matched heterozygous controls (n = 10 Vglut2-ires-Cre; Gpr45 flox/flox, n = 7 Sim1-ires-Cre, n = 5 Gpr45 flox/flox, mixed sex; repeated measures two-way ANOVA followed by Tukey's multiple comparison test). Panels (A) and (D) adapted from [15]. Panel (G) adapted from [22]. All error bars represent s.e.m. ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001.
The arcuate nucleus of the hypothalamus (ARC) is comprised of discrete subsets of neurons that regulate appetite including those marked by Agouti-related peptide (AgRP) [17]. To determine whether Gpr45 expression may play an important role in the ARC, we first examined the expression pattern of Gpr45 in the ARC by analyzing our previous single cell RNA sequencing data set [31]. Similar to prior datasets [34], we found Gpr45 was ubiquitously expressed throughout each ARC cluster (Figure S3D), including neurons co-labeled with excitatory (Figure S3A) and inhibitory (Figure S3B) gene markers and Agrp (Figure S3C). Therefore, we conditionally deleted Gpr45 from AgRP neurons but found no significant effects on bodyweight compared to controls (Figure S3E). Given that AgRP neurons are composed of GABAergic cells, this is consistent with the lack of phenotype observed after deletion from all GABAergic cells (Figure 2D–F).
Due to the widespread expression of Vglut2 throughout the rostral–caudal axis of the rodent brain (Figure 2A, Figure S1) [15], we aimed to narrow the expression window to the hypothalamus. To achieve this, we selectively deleted Gpr45 from neurons marked by Single-minded 1 (Sim1), a gene encoding a transcription factor involved in the development of the hypothalamus (Figure 2G) [35]. This Sim1-mediated manipulation provides specificity to the hypothalamus. As in above, experimental animals were heterozygous for Sim1-Cre mice, which express Cre in the paraventricular hypothalamus (PVH), as well as supraoptic (SON), medial preoptic (MPO), anterior and posterior lateral (LHA) and posterior (PH) hypothalamic nuclei [22], and homozygous for the Gpr45 floxed allele (Sim1-Cre; Gpr45 flox/flox), while controls were either heterozygous for Sim1-Cre or homozygous for the Gpr45 floxed allele. Deletion of Gpr45 in Sim1 neurons of both male and female mice led to rapid weight gain (Figure 2H), accompanied by escalated food intake (Figure 2I) compared to controls. These findings implicate the importance of Gpr45 located in the hypothalamus for energy homeostasis. Notably, the final body weights at week 18 of Vglut2-ires-Cre; Gpr45 flox/flox were significantly higher than Sim1-Cre; Gpr45 flox/flox subjects, although both were heavier than Vgat-ires-Cre; Gpr45 flox/flox mice (Figure S2D). This suggests that other Vglut2+, Sim1-neurons may contribute to the site of Gpr45 action in regulating body weight, such as those located in the hindbrain. Alternatively, a subset of Vglut2-, Sim1+ neurons expressing Gpr45 may coordinate body weight in the opposing direction, masking the full effect observed in the Vglut2-ires-Cre; Gpr45 flox/flox model.
3.4. Selective deletion of Gpr45 from PVH neurons results in obesity and hyperphagia
To establish the role of Gpr45 in the PVH, Gpr45 deletion was targeted specifically to the PVH by stereotaxic injections of AAV (adeno-associated virus) vectors driving Cre recombinase expression (AAV-Cre) in Gpr45 flox/flox mice (Figure 3A). As the Cre was tagged to a fluorophore, native fluorescence signal was used to identify each injection site (Figure 3B). A recent study found that a specific preparation of AAV-Cre injected into the PVH led to neuronal cell death and gliosis at the site of viral vector injections resulting in obesity and hyperphagia [36]. To control for this, an age/sex-matched wildtype cohort was injected with the same AAV-Cre virus used in the floxed animals (Figure 3A). Both groups exhibited comparable body weights and daily food intake measurements prior to cranial surgeries (Figure 3C–D). However, weight gain was prominent shortly after Cre-mediated excision of Gpr45 in the PVH (Figure 3C), aligned with augmented food consumption (Figured 3D) compared to controls. These data argue that while stereotaxic AAV-injections into the PVH have no effect on energy balance on their own, anatomically defined deletion of Gpr45 from the PVH modulates body weight and appetite control.
Figure 3.
Selective deletion of Gpr45 from PVH neurons results in obesity and hyperphagia.
(A) Brain schematic representing bilateral injection of AAV-hSyn-mCherry-P2A-Cre-WPRE into the PVH of Gpr45 flox/flox or age/sex-matched WT controls. (B) Representative epifluorescent image of a brain slice after bilateral injection of AAV-hSyn-mCherry-P2A-Cre-WPRE into the PVH of a Gpr45 flox/flox mouse. (C–D) Mice with specific deletion of Gpr45 from the PVH (C) gain significantly more weight and (D) consume significantly more standard chow diet than age/sex-matched WT controls (n = 15 Gpr45 flox/flox, n = 9 WT, mixed sex; repeated measures two-way ANOVA followed by Šídák's multiple comparison test). All error bars represent s.e.m. ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001.
3.5. Generation of knock-in Gpr45-CreERT2 mice
The experiments above highlight the vital role Gpr45 play in governing food intake and weight regulation, particularly in Vglut2/Sim1+ excitatory neurons residing in the PVH. However, the relative contribution of the neurons themselves expressing Gpr45 has not been resolved. To address this line of questioning, we generated a Gpr45-CreER knock-in model using the CRISPR/Cas9 platform. A CreERT2 cassette was inserted in frame with the mouse Gpr45 locus just after the stop codon (Figure S4A). Here, the Cre recombinase enzyme is fused to a mutant estrogen receptor (ERT2) domain to allow for spatial and temporal control of gene expression while avoiding potential developmental complications and/or compensation.
3.6. Synaptic silencing of PVHGpr45 neurons via synaptic transmission blockade results in obesity and hyperphagia
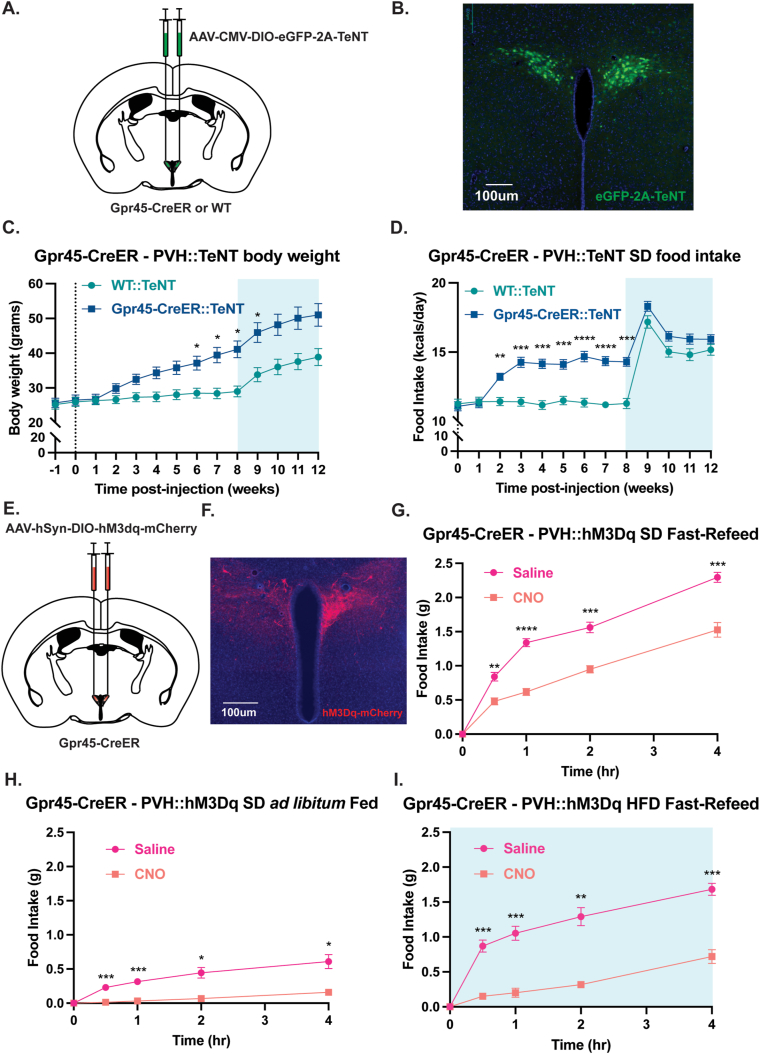
Given the relatively high expression of Gpr45 in the PVH (Figure S1A) and our findings that Gpr45 function in this anatomical region is critical for body weight management (Figure 3), we explored the capacity of Gpr45-expressing PVH (PVHGpr45) neurons in modulating energy balance. To begin, we permanently perturbed signaling from PVHGpr45 by bilaterally targeting the synaptic silencer tetanus toxin light chain (TeNT) to these cells of Gpr45-CreER mice (Figure 4A). This manipulation interferes with neurotransmitter release indefinitely disrupting downstream communication. As the AAV-TeNT is linked to a GFP reporter, we were able to confirm the accuracy of our injections (Figure 4B). The same concentration and volume of virus was injected in age/weight/sex-matched WT littermates to serve as controls. We found that on SD, body weights started to diverge 2 weeks after TeNT injections and reached statistical significance by week 6 between the two cohorts (Figure 4C). Predictively, food consumption during this period was heightened in the Gpr45-CreER::TeNT versus WT::TeNT mice (Figure 4D). Challenging animals with 60% high-fat diet (HFD) amplified weight gain and food intake in both groups at comparable rates, although Gpr45-CreER::TeNT remained significantly heavier during this month-long HFD exposure (Figure 4C–D).
Figure 4.
PVHGpr45 neurons bidirectionally modulate appetite.
(A) Brain schematic representing bilateral injection of AAV-CMV-DIO-eGFP-2A-TeNT into the PVH of Gpr45-CreER or age/sex-matched WT controls. (B) Representative epifluorescent image of a brain slice after bilateral injection of AAV-CMV-DIO-eGFP-2A-TeNT into the PVH of a Gpr45-CreER mouse. (C–D) Synaptic silencing of PVHGpr45 neurons in Gpr45-CreER mice injected with AAV-CMV-DIO-eGFP-2A-TeNT into the PVH (C) gain significantly more weight and (D) consume significantly more standard chow diet than age/sex-matched WT controls (n = 9 Gpr45-CreER, n = 9 WT, mixed sex; repeated measures two-way ANOVA followed by Šídák's multiple comparison test). (E) Brain schematic representing bilateral injection of AAV-hSyn-DIO-hM3dq-mCherry into the PVH of Gpr45-CreER. (F) Representative epifluorescent image of a brain slice after bilateral injection of AAV-hSyn-DIO-hM3dq-mCherry into the PVH of a Gpr45-CreER mouse. (G–I) Acute, CNO-induced chemogenetic activation of PVHGpr45 neurons in Gpr45-CreER mice injected with AAV-hSyn-DIO-hM3dq-mCherry into the PVH significantly attenuates (G) standard chow diet food intake in a fast-refeed paradigm (n = 7 Gpr45-CreER, mixed sex; repeated measures two-way ANOVA followed by Šídák's multiple comparison test), (H) standard chow diet food intake in an ad libitum fed paradigm (n = 7 Gpr45-CreER; repeated measures two-way ANOVA followed by Šídák's multiple comparison test), and (I) high fat diet (HFD) food intake in a fast-refeed paradigm (n = 6 Gpr45-CreER; repeated measures two-way ANOVA followed by Šídák's multiple comparison test), compared to the same animals following a vehicle saline injection. Shaded blue area in (C–D) and (I) represent HFD homecage availability. All error bars represent s.e.m. ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001. (For interpretation of the references to color in this figure legend, the reader is referred to the Web version of this article.)
When designing knock-in Cre animals, the Cre cassette is often targeted just after the stop codon of the gene of interest to minimize alterations in endogenous expression levels. However, it has been reported that this strategy may also perturb gene expression levels, which alone may contribute to any observed changes in energy balance [37,38]. To ensure that Gpr45-CreER mice displayed normal body weight over time separated from any neural activity manipulation, we bilaterally injected a Cre-dependent GFP virus to the PVH of Gpr45-CreER and WT animals and monitored their body weights for 3 months (Figure S4A-B). We found that Gpr45-CreER::GFP mice exhibited normal weight gain on both SD and HFDs compared to controls; a far different body weight trajectory than animals in which these PVHGpr45 neurons were permanently silenced (Figure S4C).
3.7. Constitutive PVHGpr45 neuronal activity via chronic hyperexcitability fails to alter body weight
As chronic inactivation of PVHGpr45 neurons robustly enhanced body weight and food intake (Figure 4C–D), we predicted that chronic activation would have the opposite effect by reducing weight gain and food consumption. To test this, we expressed a mutated version of the bacterial sodium channel NaChBac [29] via stereotaxic bilateral delivery of a conditional AAV-Flex-NaChBac-GFP virus to the PVH of Gpr45-CreER mice (Figure S5A). The accuracy of injections was confirmed through visualization of native GFP expression linked to the NaChBac construct (Figure S5B). NaChBac-expressing neurons have been shown to display altered and prolonged action potentials, reduced input resistance, and a lower rheobase [21,29]. To confirm these NaChBac-induced alterations, we compared the electrophysiological properties of PVHGpr45 neurons with and without NaChBac expression. This analysis revealed prolonged action potential overshoot depolarization (Figure S5D), hyperpolarized resting membrane potential (Figure S5E), lower input resistance (Figure S5F), and increased excitability evident in shifts in action potential threshold (Figure S5G-H) and rheobase (Figure S5I) in NaChBac-expressing versus non-expressing PVHGpr45 neurons. Similar to observations made in anorectic cell populations of the melanocortin system [21], constitutive activation of PVHGpr45 neurons failed to impact body weight either when mice remained on a SD chow diet or were challenged with a 60% HFD compared to age/weight/sex-matched wildtype littermate controls (Figure S5C). Together, these results suggest that while reduced activity levels of PVHGpr45neurons drive obesity, prolonged increased activity levels do not alter body weight.
3.8. Acute activation of PVHGpr45 neurons potently reduces appetite
It is plausible that the lack of a lean phenotype when PVHGpr45 neurons were chronically activated stems from redundant pathways that promote positive energy balance. This notion is in line with prior reports demonstrating that persistent activation of either POMC or Mc4R neurons is ineffective for weight loss [21], but acute activation of either cell type robustly attenuates feeding over the course of minutes to hours [5,6,20]. To assess whether short-term stimulation of PVHGpr45 neurons is sufficient to reduce food intake, we bilaterally delivered the Cre-dependent chemogenetic actuator, hM3Dq, to the PVH of Gpr45-CreER mice (Figure 4E). Targeting precision was verified via visualization of native mCherry expression fused to the hM3Dq construct (Figure 4F). The otherwise pharmacologically inert ligand clozapine-N-oxide (CNO) binds to and stimulates PVHGpr45 activity through the Gq-signaling pathway [28]. We found that CNO-mediated, acute activation of PVHGpr45 neurons significantly curbed SD consumption in calorically deprived mice following an overnight fast compared to the same animals following a saline vehicle injection (Figure 4G). As fasting conditions are not normally experienced in laboratory research animals fed ad libitum, we also assessed SD intake in calorically replete mice during the light cycle, a period when food consumption is typically low. Despite the modest amount of SD ingested during this phase after a vehicle injection, feeding was further diminished when PVHGpr45 neurons were stimulated (Figure 4H) further supporting their appetite suppressing role of these cells. Finally, we asked whether acute PVHGpr45 activation could subdue the intake of palatable HFD in motivated, hungry mice. We found that acute, chemogenetic activation of PVHGpr45 neurons strongly decreased HFD intake in an overnight fast-refeed assay using the same within-subject design (Figure 4I). Collectively, these results illustrate the proficiency of PVHGpr45 cells to transiently blunt appetite independent of caloric need and food type.
4. Discussion
The global obesity epidemic is a complex and multifaceted issue, impacting millions worldwide. Its causes are deeply intertwined, stemming from factors such as dietary choices, sedentary lifestyles, genetic predispositions, and socioeconomic conditions. Over the past few years, anti-obesity medications have become an essential tool in the fight against obesity. The most successful drugs are mimetics of naturally acting agonists of GPCRs, highlighting the importance of identifying novel druggable targets that could contribute to the revolutionary treatment of obesity. One such GPCR, Gpr45, is expressed in various tissues, including the brain, adipose tissue, and muscle [39,40], which are key players in energy metabolism, suggesting a possible influence on both central and peripheral pathways involved in metabolic control. Here, we provide direct evidence that Gpr45 functions to regulate body weight homeostasis in mice, particularly within the PVH, making it a potential therapeutic target of interest. This discovery is timely given the rapid rise, efficacy, and popularity of anti-obesity medications acting as GLP-1 mimetics [2,41]. Notably, both depletion of GLP-1R in the PVH or permanent synaptic silencing of PVH neurons delineated by GLP-1R result in obesity [18,42]. How these drugs reach the hypothalamus is less understood, although the mechanisms underlying GLP-1R agonist function are starting to come to light [43,44].
The functional contribution of Mc4r to energy balance has been well characterized [12,20,22,[45], [46], [47], [48], [49], [50], [51]]. Interestingly, the class A orphan GPCR Gpr45 shares several similarities with Mc4r. Global knockout mice exhibit obesity and hyperphagia, effects that can be recapitulated by restricting gene deletion from excitatory cells marked by Vglut2 or via targeted deletion from the PVH. Additionally, acute chemogenetic activation of PVH neurons labeled by Mc4r- or Gpr45-knock-in Cre animals robustly attenuates food intake regardless of appetite state [20], while irreversible synaptic silencing of these cells rapidly drives eating and subsequent weight gain [18]. This may suggest Mc4r and Gpr45 are expressed in the same PVH cells, however it should be noted that Mc4r signals through canonical Gs and Gq pathways [52], whereas Gpr45 has been suggested to signal via Gi pathways [53]. Since these pathways have opposing effects on cellular processes, primarily through their influence on adenylate cyclase (AC) activity and cyclic AMP (cAMP) levels, it may be more common that these two GPCRs act independently to regulate energy homeostasis. Further work exploring the putative overlap of these two receptors throughout the brain utilizing single-cell RNA sequencing and/or in situ hybridization strategies will help clarify their functional expression patterns.
Gpr45 is expressed broadly throughout the brain in both anatomical regions comprised of predominantly inhibitory GABAergic cells such as the striatum and bed nucleus of the stria terminalis (BNST) and primarily excitatory glutamatergic cells including the PVH and medial amygdala (MeA) as well as structures with mixed cell types like the PLH and DMH. Despite its widespread distribution, only selective deletion from Vglut2+, but not Vgat+, neurons mimic the obesity and hyperphagic phenotypes observed in the global Gpr45 knockout model, suggesting the locus of activity resides in Vglut2 excitatory cells. Reinforcing this notion, targeted deletion of Gpr45 from Sim1-expressing neurons, encompassing glutamatergic cells in the PVH, MPO, PH, LHA, and SON [22], reproduce the increases in body weight and food consumption. Finally, directed Gpr45 deletion from the PVH mirrored the obesity and overeating phenotypes, further pinpointing the precise site of action of this system. While these results demonstrate the necessity of Gpr45 in Vglut2+/Sim1+/PVH cells in modulating energy balance, it does not rule out additional glutamatergic effector sites, such as the hindbrain. Importantly, at the conclusion of experiments, 4.5-month-old mice with Gpr45 deleted from Vglut2+ neurons were significantly heavier than mice with Gpr45 deleted from Sim1+ neurons, implicating an alternative Vglut2+/Sim1-brain region(s) in facilitating energy balance. A targeted deletion of Gpr45 in these putative sites (ie. hindbrain), using the same viral Cre-mediated strategy employed for the PVH in this study, could help determine the potential role of intact Gpr45 signaling in these regions for body weight regulation.
While long-term inhibition of PVHGpr45 neurons led to marked obesity and elevated food intake, chronic activation failed to cause a significant reduction in body weight or obesity development. Like observations made in the melanocortin pathway [21], Gpr45 action seems biased toward obesity development while having no or limited roles in obesity prevention or reversal. Interestingly, we report that acute activation of PVHGpr45 neurons strongly inhibits feeding irrespective of internal state and/or food palatability. The difference between acute and chronic manipulations may arise from overlapping pathways that support a positive energy balance. Comparable redundant mechanisms have been described in GABAergic ARC neurons, including agouti-related peptide (AgRP) cells, as well as PVHMc4r neurons [21,29]. Acute inhibition of feeding caused by altering one pathway can be offset by adaptive changes in other feeding–promoting pathways over time. From an evolutionary standpoint, it is logical that the brain is wired this way to conserve energy reserves and prolong survival.
While animal models and cell culture studies have provided some insight into Gpr45's prospective role in metabolic processes, human studies are limited. Unlike mutations in melanocortin pathway genes that are associated with human obesity [54], there are no significant GWAS associations with metabolic traits identified for Gpr45. However, considering the recessive nature of rodent KO phenotypes, it may be more difficult to find associations for rare mutations mainly found in heterozygote carriers. To gain a more complete understanding of Gpr45's physiological role, the development of specific agonists and/or antagonists is essential. These tools would help clarify how Gpr45 signaling influences body weight regulation and assess its potential as a therapeutic target for obesity. Despite the dearth of translational evidence, here we substantiate the functional contribution of intact rodent Gpr45 signaling in coordinating appetite and body weight maintenance.
CRediT authorship contribution statement
Michael J. Krashes: Writing – original draft, Supervision, Investigation, Funding acquisition, Conceptualization. Eva O. Karolczk: Writing - review and editing, Formal analysis, Data curation, Conceptualization. Chia Li: Writing - review and editing, Data curation, Conceptualization. Ivan C. Alcantara: Writing - review and editing, Data curation. Isabel M. Cohen: Writing - review and editing, Data curation. Cuiying Xiao: Writing- review and editing, Data curation. Abigail I. Goldschmidt: Writing- review and editing, Data curation. Cynthia A Pinkus, Junjie Li, Monica M. Li, Ryan M. Esquejo, Jean-Philippe Fortin, Kendra K. Bence, Marc L. Reitman: Conceptualization.
Declaration of competing interest
The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: C.A.P., J.L., M.M. L., R.M.E., J-P.F., and K.K.B. are employees of Pfizer at the time the study was conducted. M.J.K. receives research support from Pfizer. All other authors have no competing interests to declare
Acknowledgements
This research was supported by the Intramural Research Program of the National Institutes of Health, the National Institutes of Diabetes and Digestive and Kidney Diseases (DK075088, CRADA 21-0947 to M.J.K. and DK075087-06 to M.J.K.). We thank Dr. Qingchun Tong and Dr. Benjamin Arenkiel for the EF1a-Flex-EGFP-P2A-mNaChBac plasmid and the NIEHS Viral Vector Core Facility for packaging the virus.
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.molmet.2025.102174.
Appendix A. Supplementary data
The following are the Supplementary data to this article.
Fig. S1.
Expression pattern of Gpr45, Vglut2, and Vgat transcripts in the hypothalamus and hindbrain.
(A) DAPI (blue) expression highlighting hypothalamic structures including the paraventricular nucleus of the hypothalamus (PVH), anterior hypothalamic area (AHA), suprachiasmatic nucleus (SCN), and zona incerta (ZI). Vglut2 (red), Vgat (magenta), and Gpr45 (green) expression in these regions. (B) DAPI (blue) expression highlighting hypothalamic structures including the dorsal medial hypothalamus (DMH), ventral medial hypothalamus (VMH), and arcuate nucleus (ARC). Vglut2 (red), Vgat (magenta), and Gpr45 (green) expression in these regions. (C) DAPI (blue) expression highlighting hindbrain structures including the are postrema. (AP) and nucleus of the solitary tract (NTS). Vglut2 (red), Vgat (magenta), and Gpr45 (green) expression in these regions.
Fig. S2.
Generation and validation of conditional Gpr45 floxed mouse model.
(A) Schematic of Gpr45 flox/flox allele generated using CRISPR/Cas9 technology. LoxP sites were inserted in frame with the mouse Gpr45 locus 254 bp upstream of exon 2 and 35 bp downstream of the stop codon. (B) Relative expression levels in the mediobasal hypothalamus detected via RT-PCR of Gpr45, but not Brs3, transcript levels are significantly lower in Vglut2-ires-Cre; Gpr45 flox/flox animals compared to Gpr45 flox/flox controls (n = 3 Vglut2-ires-Cre; Gpr45 flox/flox, n = 3 Gpr45 flox/flox; unpaired t test, two-tailed). (C) Relative expression levels in the mediobasal hypothalamus detected via RT-PCR of Gpr45, but not Mc4r, transcript levels are significantly lower in Vgat-ires-Cre; Gpr45 flox/flox animals compared to Gpr45 flox/flox controls (n = 3 Vgat-ires-Cre; Gpr45 flox/flox, n = 3 Gpr45 flox/flox; unpaired t test, two-tailed). (D) Body weights at week 18 of Vglut2-ires-Cre; Gpr45 flox/flox mice are significantly higher than Sim1-Cre; Gpr45 flox/flox subjects, but both are significantly higher than Vgat-ires-Cre; Gpr45 flox/flox mice (n = 8 Vglut2-ires-Cre; Gpr45 flox/flox, n = 8 Vgat-ires-Cre; Gpr45 flox/flox, n = 10 Sim1-Cre; Gpr45 flox/flox, mixed sex; repeated measures one-way ANOVA followed by Tukey's multiple comparison test). All error bars represent s.e.m. ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001.
Fig. S3.
Gpr45 is ubiquitously expressed in the ARC but selective deletion of Gpr45 from AgRP neurons has no effect on body weight regulation.
(A–D) FeaturePlots showing ARC neurons and clusters in UMAP space with (A) Slc17a6 (Vglut2), (B) Slc32a1 (Vgat), (C)Agrp, and (D)Gpr45 expression. (E). Mice with specific deletion of Gpr45 from AgRP + neurons exhibit comparable body weight as age/sex-matched heterozygous controls (n = 5 AgRP-ires-Cre; Gpr45 flox/flox, n = 5 AgRP-ires-Cre, n = 5 Gpr45 flox/flox, mixed sex; repeated measures two-way ANOVA followed by Tukey's multiple comparison test). All error bars represent s.e.m. ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001.
Fig. S4.
Generation and body weight characterization of knock-in Gpr45-CreER mice.
(A) Schematic of Gpr45-CreER knock-in model generated using CRISPR/Cas9 technology. The CreERT2 cassette was inserted in frame with the mouse Gpr45 locus and after the coding sequence. (B) Brain schematic representing bilateral injection of AAV-CMV-DIO-GFP into the PVH of Gpr45-CreER or WT littermate mice. (C) Gpr45-CreER mice injected with AAV-CMV-DIO-GFP into the PVH exhibit comparable bodyweight as littermate WT controls injected with AAV-CMV-DIO-GFP into the PVH. (n = 7 Gpr45-CreER::GFP, n = 7 WT::GFP, mixed sex; repeated measures two-way ANOVA followed by Šídák's multiple comparison test). Shaded blue area in (C) represent HFD homecage availability. All error bars represent s.e.m. ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001.
Fig. S5.
Chronic PVHGpr45 neuronal hyperexcitability fails to alter body weight despite pronounced alterations in electrical activity.
(A) Brain schematic representing bilateral injection of AAV-EF1a-FLEX-eGFP-P2A-mNaChBac into the PVH of Gpr45-CreER or age/sex-matched WT controls. (B) Representative epifluorescent image of a brain slice after bilateral injection of AAV-EF1a-FLEX-eGFP-P2A-mNaChBac into the PVH of a Gpr45-CreER mouse. (C) Mice with constitutive PVHGpr45 neural activity in Gpr45-CreER mice injected with AAV-EF1a-FLEX-eGFP-P2A-mNaChBac into the PVH show comparable body weights to age/sex-matched WT controls injected with AAV-EF1a-FLEX-eGFP-P2A-mNaChBac into the PVH on both a standard chow diet and 60% HFD (n = 7 Gpr45-CreER, n = 7 WT, mixed sex; repeated measures two-way ANOVA followed by Šídák's multiple comparison test). (D) Representative traces showing resting membrane potential (RMP) and action potentials in control (top) and NaChBach-expressing cells (bottom). (E) Quantification of RMP (n = 16 in 3 mice for control, n = 15 in 5 mice for NaChBac, unpaired two-tailed t-test, p = 0.023) and (F) Input resistance (n = 12 in 3 mice for control, n = 16 in 5 mice for NaChBac, unpaired two-tailed t-test, p = 0.026). (G) Representative traces of elicited action potentials by injecting step-wise depolarizing currents in control cells (top) and NaChBac-expressing cells (bottom). First steps that elicited AP highlighted in red. (H) Quantification of action potential threshold (n = 11 in 3 mice for control, n = 11 in 3 mice for NaChBac, unpaired two-tailed t-test, p < 0.0001) and (I) Rheobase (n = 11 in 3 mice for control, n = 11 in 3 mice for NaChBac, unpaired two-tailed t-test, p = 0.007). Shaded blue area in (C) represent HFD homecage availability. All error bars represent s.e.m. ∗P < 0.05, ∗∗P < 0.01, ∗∗∗P < 0.001, ∗∗∗∗P < 0.0001.
Data availability
Data will be made available on request.
References
- 1.Drucker D.J. Efficacy and safety of GLP-1 medicines for type 2 diabetes and obesity. Diabetes Care. 2024;47(11):1873–1888. doi: 10.2337/dci24-0003. [DOI] [PubMed] [Google Scholar]
- 2.Krashes M. Glucagon-like peptide-1 receptor. Curr Biol. 2024;34(23):R1163–R1164. doi: 10.1016/j.cub.2024.10.039. [DOI] [PubMed] [Google Scholar]
- 3.Cui J., Ding Y., Chen S., Zhu X., Wu Y., Zhang M., et al. Disruption of Gpr45 causes reduced hypothalamic POMC expression and obesity. J Clin Investig. 2016;126(9):3192–3206. doi: 10.1172/JCI85676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Powell D.R., Revelli J.P., Doree D.D., DaCosta C.M., Desai U., Shadoan M.K., et al. High-throughput screening of mouse gene knockouts identifies established and novel high body fat phenotypes. Diabetes Metab Syndr Obes. 2021;14:3753–3785. doi: 10.2147/DMSO.S322083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Aponte Y., Atasoy D., Sternson S.M. AGRP neurons are sufficient to orchestrate feeding behavior rapidly and without training. Nat Neurosci. 2011;14(3):351–355. doi: 10.1038/nn.2739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Zhan C., Zhou J., Feng Q., Zhang J.E., Lin S., Bao J., et al. Acute and long-term suppression of feeding behavior by POMC neurons in the brainstem and hypothalamus, respectively. J Neurosci. 2013;33(8):3624–3632. doi: 10.1523/JNEUROSCI.2742-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Liu Y., Savier E.L., DePiero V.J., Chen C., Schwalbe D.C., Abraham-Fan R.J., et al. Mapping visual functions onto molecular cell types in the mouse superior colliculus. Neuron (Camb, Mass) 2023;111(12):1876–1886 e5. doi: 10.1016/j.neuron.2023.03.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schwalbe D.C., Stornetta D.S., Abraham-Fan R.J., Souza G.M.P.R., Jalil M., Crook M.E., et al. Molecular organization of autonomic, respiratory, and spinally-projecting neurons in the mouse ventrolateral medulla. J Neurosci. 2024;44(31) doi: 10.1523/JNEUROSCI.2211-23.2024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sisley S., Gutierrez-Aguilar R., Scott M., D’Alessio D.A., Sandoval D.A., Seeley R.J. Neuronal GLP1R mediates liraglutide’s anorectic but not glucose-lowering effect. J Clin Investig. 2014;124(6):2456–2463. doi: 10.1172/JCI72434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Burmeister M.A., Ayala J.E., Smouse H., Landivar-Rocha A., Brown J.D., Drucker D.J., et al. The hypothalamic Glucagon-like peptide 1 receptor is sufficient but not necessary for the regulation of energy balance and glucose homeostasis in mice. Diabetes. 2017;66(2):372–384. doi: 10.2337/db16-1102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Chen Z., Deng X., Shi C., Jing H., Tian Y., Zhong J., et al. GLP-1R-positive neurons in the lateral septum mediate the anorectic and weight-lowering effects of liraglutide in mice. J Clin Investig. 2024;134(17) doi: 10.1172/JCI178239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Shah B.P., Vong L., Olson D.P., Koda S., Krashes M.J., Ye C., et al. MC4R-expressing glutamatergic neurons in the paraventricular hypothalamus regulate feeding and are synaptically connected to the parabrachial nucleus. Proc Natl Acad Sci U S A. 2014;111(36):13193–13198. doi: 10.1073/pnas.1407843111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Berglund E.D., Lie T., Kong X., Soh J.W., Vong L., Deng Z., et al. Melanocortin 4 receptors in autonomic neurons regulate thermogenesis and glycemia. Nat Neurosci. 2014;17(7):911–913. doi: 10.1038/nn.3737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Guo H., Xin Y., Wang S., Zhang X., Ren Y., Qiao B., et al. Hypothalamic POMC neuron-specific knockout of MC4R affects insulin sensitivity by regulating Kir2.1. Mol Med. 2024;30(1):34. doi: 10.1186/s10020-024-00804-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Vong L., Ye C., Yang Z., Choi B., Chua Jr S., Lowell B.B. Leptin action on GABAergic neurons prevents obesity and reduces inhibitory tone to POMC neurons. Neuron (Camb, Mass) 2011;71(1):142–154. doi: 10.1016/j.neuron.2011.05.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Webber E.S., Bonci A., Krashes M.J. The elegance of energy balance: insight from circuit-level manipulations. Synapse (N Y) 2015;69(9):461–474. doi: 10.1002/syn.21837. [DOI] [PubMed] [Google Scholar]
- 17.Alcantara I.C., Miranda Tapia A.P., Aponte Y., Krashes M.J. Acts of appetite: neural circuits governing the appetitive, consummatory, and terminating phases of feeding. Nat Metab. 2022;4(7):836–847. doi: 10.1038/s42255-022-00611-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Li C., Navarrete J., Liang-Guallpa J., Lu C., Funderburk S.C., Chang R.B., et al. Defined paraventricular hypothalamic populations exhibit differential responses to food contingent on caloric state. Cell Metab. 2019;29(3):681–694 e5. doi: 10.1016/j.cmet.2018.10.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Li M.M., Madara J.C., Steger J.S., Krashes M.J., Balthasar N., Campbell J.N., et al. The paraventricular hypothalamus regulates satiety and prevents obesity via two genetically distinct circuits. Neuron (Camb, Mass) 2019;102(3):653–667 e6. doi: 10.1016/j.neuron.2019.02.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Garfield A.S., Li C., Madara J.C., Shah B.P., Webber E., Steger J.S., et al. A neural basis for melanocortin-4 receptor-regulated appetite. Nat Neurosci. 2015;18(6):863–871. doi: 10.1038/nn.4011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Li H., Xu Y., Jiang Y., Jiang Z., Otiz-Guzman J., Morrill J.C., et al. The melanocortin action is biased toward protection from weight loss in mice. Nat Commun. 2023;14(1):2200. doi: 10.1038/s41467-023-37912-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Balthasar N., Dalgaard L.T., Lee C.E., Yu J., Funahashi H., Williams T., et al. Divergence of melanocortin pathways in the control of food intake and energy expenditure. Cell. 2005;123(3):493–505. doi: 10.1016/j.cell.2005.08.035. [DOI] [PubMed] [Google Scholar]
- 23.Tong Q., Ye C.P., Jones J.E., Elmquist J.K., Lowell B.B. Synaptic release of GABA by AgRP neurons is required for normal regulation of energy balance. Nat Neurosci. 2008;11(9):998–1000. doi: 10.1038/nn.2167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Madisen L., Zwingman T.A., Sunkin S.M., Wook Oh S., Zariwala H.A., Gu H., et al. A robust and high-throughput Cre reporting and characterization system for the whole mouse brain. Nat Neurosci. 2010;13(1):133–140. doi: 10.1038/nn.2467. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Xiao C., Goldgof M., Gavrilova O., Reitman M.L. Anti-obesity and metabolic efficacy of the beta3-adrenergic agonist, CL316243, in mice at thermoneutrality compared to 22 degrees C. Obesity (Silver Spring. Md) 2015;23(7):1450–1459. doi: 10.1002/oby.21124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gao C., Gohel C.A., Leng Y., Ma J., Goldman D., Levine A.J., et al. Molecular and spatial profiling of the paraventricular nucleus of the thalamus. eLife. 2023;12 doi: 10.7554/eLife.81818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Stirling D.R., Swain-Bowden M.J., Lucas A.M., Carpenter A.E., Cimini B.A., Goodman A. CellProfiler 4: improvements in speed, utility and usability. BMC Bioinf. 2021;22(1):433. doi: 10.1186/s12859-021-04344-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Krashes M.J., Koda S., Ye C., Rogan S.C., Adams A.C., Cusher D.S., et al. Rapid, reversible activation of AgRP neurons drives feeding behavior in mice. J Clin Investig. 2011;121(4):1424–1428. doi: 10.1172/JCI46229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhu C., Jiang Z., Xu Y., Cai Z.L., Jiang Q., Xu Y., et al. Profound and redundant functions of arcuate neurons in obesity development. Nat Metab. 2020;2(8):763–774. doi: 10.1038/s42255-020-0229-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pinol R.A., Zahler S.H., Li C., Saha A., Tan B.K., Skop V., et al. Brs3 neurons in the mouse dorsomedial hypothalamus regulate body temperature, energy expenditure, and heart rate, but not food intake. Nat Neurosci. 2018;21(11):1530–1540. doi: 10.1038/s41593-018-0249-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Alcantara I.C., Li C., Mickelsen L.E., Mazzone C.M., de Araujo Salgado I., Gao C., et al. A hypothalamic circuit that modulates feeding and parenting behaviors. bioRxiv. 2024 Jul;23 [Google Scholar]
- 32.Hao Y., Hao S., Andersen-Nissen E., Mauck W.M., Zheng S., Butler A., et al. Integrated analysis of multimodal single-cell data. Cell. 2021;184(13):3573–3587 e29. doi: 10.1016/j.cell.2021.04.048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Saunders A., Macosko E.Z., Wysoker A., Goldman M., Krienen F.M., de Rivera H., et al. Molecular diversity and specializations among the cells of the adult mouse brain. Cell. 2018;174(4):1015–1030 e16. doi: 10.1016/j.cell.2018.07.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Campbell J.N., Macosko E.Z., Fenselau H., Pers T.H., Lyubetskaya A., Tenen D. alA molecular census of arcuate hypothalamus and median eminence cell types. Nat Neurosci. 2017;20(3):484–496. doi: 10.1038/nn.4495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fan C.M., Kuwana E., Bulfone A., Fletcher C.F., Copeland N.G., Jenkins N.A., et al. Expression patterns of two murine homologs of drosophila single-minded suggest possible roles in embryonic patterning and in the pathogenesis of Down syndrome. Mol Cell Neurosci. 1996;7(1):1–16. doi: 10.1006/mcne.1996.0001. [DOI] [PubMed] [Google Scholar]
- 36.Savani R., Park E., Busannagari N., Lu Y., Kwon H., Wang L., et al. Metabolic and behavioral alterations associated with viral vector-mediated toxicity in the paraventricular hypothalamic nucleus. Biosci Rep. 2024;44(1) doi: 10.1042/BSR20231846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Xiao C., Liu N., Province H., Pinol R.A., Gavrilova O., Reitman M.L. BRS3 in both MC4R- and SIM1-expressing neurons regulates energy homeostasis in mice. Mol Metabol. 2020;36:100969. doi: 10.1016/j.molmet.2020.02.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Harno E., Cottrell E.C., White A. Metabolic pitfalls of CNS Cre-based technology. Cell Metab. 2013;18(1):21–28. doi: 10.1016/j.cmet.2013.05.019. [DOI] [PubMed] [Google Scholar]
- 39.Uhlen M., Fagerberg L., Hallstrom B.M., Lindskog C., Oksvold P., Mardinoglu A., et al. Proteomics. Tissue-based map of the human proteome. Sci Technol Humanit. 2015;347(6220) doi: 10.1126/science.1260419. [DOI] [PubMed] [Google Scholar]
- 40.Lein E.S., Hawrylycz M.J., Ao N., Ayres M., Bensinger A., Bernard A., et al. Genome-wide atlas of gene expression in the adult mouse brain. Nature. 2007;445(7124):168–176. doi: 10.1038/nature05453. [DOI] [PubMed] [Google Scholar]
- 41.Drucker D.J. GLP-1 physiology informs the pharmacotherapy of obesity. Mol Metabol. 2022;57 doi: 10.1016/j.molmet.2021.101351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Liu J., Conde K., Zhang P., Lilascharoen V., Xu Z., Lim B.K., et al. Enhanced AMPA receptor trafficking mediates the anorexigenic effect of endogenous glucagon-like Peptide-1 in the paraventricular hypothalamus. Neuron (Camb, Mass) 2017;96(4):897–909 e5. doi: 10.1016/j.neuron.2017.09.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Huang K.P., Acosta A.A., Ghidewon M.Y., McKnight A.D., Almeida M.S., Nyema N.T., et al. Dissociable hindbrain GLP1R circuits for satiety and aversion. Nature. 2024;632(8025):585–593. doi: 10.1038/s41586-024-07685-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Webster A.N., Becker J.J., Li C., Schwalbe D.C., Kerspern D., Karolczak E.O., et al. Molecular connectomics reveals a glucagon-like peptide 1-sensitive neural circuit for satiety. Nat Metab. 2024;6(12):2354–2373. doi: 10.1038/s42255-024-01168-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Cone R.D. Studies on the physiological functions of the melanocortin system. Endocr Rev. 2006;27(7):736–749. doi: 10.1210/er.2006-0034. [DOI] [PubMed] [Google Scholar]
- 46.Krashes M.J., Lowell B.B., Garfield A.S. Melanocortin-4 receptor-regulated energy homeostasis. Nat Neurosci. 2016;19(2):206–219. doi: 10.1038/nn.4202. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Cone R.D. Anatomy and regulation of the central melanocortin system. Nat Neurosci. 2005;8(5):571–578. doi: 10.1038/nn1455. [DOI] [PubMed] [Google Scholar]
- 48.Mountjoy K.G., Mortrud M.T., Low M.J., Simerly R.B., Cone R.D. Localization of the melanocortin-4 receptor (MC4-R) in neuroendocrine and autonomic control circuits in the brain. Mol Endocrinol. 1994;8(10):1298–1308. doi: 10.1210/mend.8.10.7854347. [DOI] [PubMed] [Google Scholar]
- 49.Kishi T., Aschkenasi C.J., Lee C.E., Mountjoy K.G., Saper C.B., Elmquist J.K. Expression of melanocortin 4 receptor mRNA in the central nervous system of the rat. J Comp Neurol. 2003;457(3):213–235. doi: 10.1002/cne.10454. [DOI] [PubMed] [Google Scholar]
- 50.Farooqi I.S., Yeo G.S., Keogh J.M., Aminian S., Jebb S.A., Butler G., et al. Dominant and recessive inheritance of morbid obesity associated with melanocortin 4 receptor deficiency. J Clin Investig. 2000;106(2):271–279. doi: 10.1172/JCI9397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Farooqi I.S., Keogh J.M., Yeo G.S.H., Lank E.J., Cheetham T., O’Rahilly S. Clinical spectrum of obesity and mutations in the melanocortin 4 receptor gene. N Engl J Med. 2003;348(12):1085–1095. doi: 10.1056/NEJMoa022050. [DOI] [PubMed] [Google Scholar]
- 52.Li Y.Q., Shrestha Y., Pandey M., Chen M., Kablan A., Gavrilova O., et al. G(q/11)alpha and G(s)alpha mediate distinct physiological responses to central melanocortins. J Clin Investig. 2016;126(1):40–49. doi: 10.1172/JCI76348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Martin A.L., Steurer M.A., Aronstam R.S. Constitutive activity among orphan Class-A G protein coupled receptors. PLoS One. 2015;10(9) doi: 10.1371/journal.pone.0138463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Loos R.J.F., Yeo G.S.H. The genetics of obesity: from discovery to biology. Nat Rev Genet. 2022;23(2):120–133. doi: 10.1038/s41576-021-00414-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Data will be made available on request.