Summary
Lisavanbulin is a prodrug of the microtubule-targeting agent avanbulin. Both avanbulin and lisavanbulin have demonstrated significant antitumor activity in several preclinical tumor models including glioblastoma. Previous human studies demonstrated that 48-h infusions of intravenous lisavanbulin were well tolerated with preliminary activity in recurrent glioblastoma. The current phase 1/2a study evaluates the safety and tolerability of once-daily oral lisavanbulin in patients with solid tumors or recurrent glioblastoma or high-grade glioma. Lisavanbulin is associated with profound, durable responses in a subset of patients with recurrent refractory grade 4 astrocytoma or glioblastoma. We present here the clinical and translational results from this trial, including a description of a response-predictive molecular signature that warrants further exploration in these tumor types of significant unmet need. The study is registered at ClinicalTrials.gov (NCT02490800).
Keywords: lisavanbulin, glioma, glioblastoma, astrocytoma
Graphical abstract

Highlights
-
•
Lisavanbulin is an orally dosed brain-penetrant mitotic spindle checkpoint inhibitor
-
•
Our study assesses lisavanbulin in refractory astrocytoma or glioblastoma
-
•
Lisavanbulin is tolerable in patients with these tumors
-
•
A subset of patients have durable efficacy responses to lisavanbulin
Lopez et al. report a phase 1/2a study evaluating once-daily oral lisavanbulin, a brain-penetrant microtubule-targeting agent in patients with solid tumors or recurrent high-grade glioma. Lisavanbulin is tolerable in these patients and is associated with profound, durable responses in a subset of patients with grade 4 astrocytoma or glioblastoma.
Introduction
Lisavanbulin (formerly BAL101553) is a water-soluble, lysine pro-drug of the synthetic small-molecule avanbulin (BAL27862). Avanbulin reversibly binds tubulin heterodimers at the colchicine site, inhibiting microtubule formation and disrupting microtubule organization, with a mechanism of action on microtubule dynamics that differs from that of standard microtubule-targeting agents (MTAs).1 Avanbulin has extensive activity in several in vitro tumor models refractory to existing MTAs2,3 and significant antitumor activity across several tumor xenograft models, including those refractory to standard MTAs and other standard-of-care therapies.4,5,6,7,8,9,10,11,12 Pathologically confirmed cures have also been observed in some cases.7,9 Treatment with avanbulin demonstrated a significant effect on tumor cell proliferation and viability, as well as tumor vasculature, suggesting a dual action on refractory tumor cells and vascular cells.12,13 Importantly, lisavanbulin demonstrates excellent penetration into tissues including the brain, in an autoradiography study in mice.14
End-binding protein 1 (EB1), the prototypic member of microtubule plus-end-tracking proteins controlling microtubule dynamics, had been proposed as a response-predictive biomarker in rodent glioblastoma (GBM) models. EB1 links microtubules to several cellular structures, such as kinetochores and the cell cortex, and participates in microtubule-mediated cell functions, such as cell division, migration, and morphogenesis.15 In vitro data have shown that EB1 proteins sensitize microtubules to the action of microtubule-interacting agents by promoting microtubule catastrophes.16,17
EB1 was shown to be a predictive biomarker of response to lisavanbulin in an orthotopic mouse model with GBM6-derived experimental gliomas.5 When overexpressed in GBM, EB1 is involved in disease progression and has been reported to be correlated with poor overall and progression-free survival (PFS).16 Interestingly, lisavanbulin-treated mice with EB1-high tumors showed a significantly longer survival than those with EB1-downregulated tumors. This survival benefit was attributed to the action of lisavanbulin on GBM stem-like cells, with inhibition of endothelial differentiation and induction of astrocytic differentiation in GBM6 tumor cells.5,6
In a first-in-human phase 1 study, lisavanbulin was administered as a 2-h weekly infusion, but dose-limiting vascular toxicity was observed.18 Dose-limiting toxicity (DLT) appeared to be related to maximum observed plasma concentration (Cmax),18 whereas nonclinical data suggest that the antiproliferative effects are related to avanbulin area under the curve (AUC).19 A regimen of weekly 48-h infusions was therefore assessed.20,21 Vascular toxicity was not observed using 48-h infusions, and one patient with GBM had a confirmed partial response with a >90% decrease in target lesions lasting 16 cycles; another patient with GBM had stable disease (SD) over 10 cycles of treatment.20 The 48-h infusion provided an AUC at maximum tolerated dose (MTD) levels at least 2-fold higher compared to a 2-h infusion. The bioavailability of avanbulin following oral administration of lisavanbulin was assessed in the same study21; the relative oral bioavailability of avanbulin was estimated to be over 80%, suggesting that oral administration of lisavanbulin could efficiently deliver active drug. A recently published study demonstrated that oral lisavanbulin was safe at doses of up to 15 mg daily when used in combination with radiotherapy in the treatment of newly diagnosed GBM with an unmethylated MGMT promoter.22
In the current phase 1/2a study (CDI-CS-002; NCT02490800; EudraCT 2014-003371-34), patients with solid tumors or with recurrent or progressive GBM (per 2016 World Health Organization [WHO] classification of CNS tumors) or high-grade glioma were treated with once-daily oral lisavanbulin, in order to determine the MTD, characterize the safety and tolerability of oral continuous dosing, describe DLTs, and assess efficacy in patients with EB1-positive brain tumors. Here, we present results from both the phase 1 and phase 2a portions of the study, focusing on patients with refractory brain tumors given the significant unmet need. We also report the consolidated biomarker data from these studies proposing a response-predictive biomarker signature that warrants further development.
Results
Patient disposition
The study was conducted between May 20, 2015 and November 24, 2022. The phase 1 portion was conducted at four sites in the UK, and the phase 2a portion at four sites in the UK, four in Switzerland, four in Germany, and one in Belgium. Seventy-two patients were enrolled, 26 with advanced solid tumors and 46 with high-grade glioma or GBM. Patient disposition is summarized in Tables S1 and S2. The overall median age was 57 years, range 25–77 years (median 65 years in patients with advanced solid tumors, 50 years in phase 1 patients with high-grade glioma or GBM, and 57.5 years in phase 2a patients). In total, 31/72 (43.1%) were female, and Eastern Cooperative Oncology Group performance status for all patients was ≤2. Patient demographics and baseline characteristics were as expected for a heavily pretreated patient population and are summarized in Tables S3 and S4. At time of data-lock, the overall median follow-up was 7.4 weeks (range 0.6–236 weeks); median follow-up was 7.6 weeks (range 0.6–40 weeks) in patients with solid tumors in phase 1, 7.1 weeks (range 2.4–236 weeks) for patients with GBM or high-grade glioma in phase 1, and 10.6 weeks (range 3–54 weeks) in phase 2a patients. Eight patients with relapsed GBM/glioma continued treatment in a post-trial access program.
Safety and tolerability
Twenty-six patients with solid tumors were treated in the phase 1 dose-escalation phase in six cohorts receiving oral lisavanbulin at doses from 2 to 30 mg/day (Figure 1). The most common treatment-related adverse events (AEs) were fatigue, diarrhea, and hyponatremia. Two out of the six patients treated at 20 mg/day experienced toxicities that were deemed dose limiting (both reversible grade 4 hyponatremias) as did both patients treated at 30 mg/day (reversible grade 3 hyponatremia and grade 2 hallucinations). The MTD for solid tumor patients was therefore determined to be 16 mg/day.
Figure 1.
Patient flow and determination of MTD in patients with advanced or recurrent solid tumors and in patients with GBM or high-grade glioma in phase 1 (MTD-determining population)
See also Tables S1–S7.
Twenty-eight patients with high-grade glioma/GBM were then enrolled in a parallel dose escalation (Figure 1) and tolerated higher doses than the patients with solid tumors with escalation continuing beyond the solid tumor MTD. The most common treatment-related AEs were similarly nausea and fatigue. DLTs were only experienced by one of six patients treated at 20 mg/day (grade 2 depression and grade 2 fatigue) and none of six patients treated at 30 mg/day. Two of the three patients treated at 35 mg/day experienced DLTs (one patient had grade 3 hallucinations and grade 3 confusion, and another patient had grade 2 confusion and grade 2 gait disturbance). The MTD for the patients with brain tumors was therefore determined to be 30 mg/day.
Treatment-emergent AEs in patients with solid tumors and patients with high-grade glioma/GBM in phase 1 are summarized in Tables S5 and S6, respectively. Table S7 provides the overall safety and tolerability seen in the phase 2a portion of this study, with only one patient (5.6%) experiencing reversible grade 3 hyponatremia confirming the modeling predictions from the dose escalation.
Treatment-related serious AEs (SAEs) and dose discontinuations were seen at doses at or above the respective MTDs for both solid tumors and glioma patients in the phase 1 dose-escalation part of the study. In the phase 2a cohort treated at the recommended phase 2 dose (RP2D), two of 18 patients (11.1%) had treatment-related SAEs; one additional patient discontinued treatment due to related AEs.
Pharmacokinetics
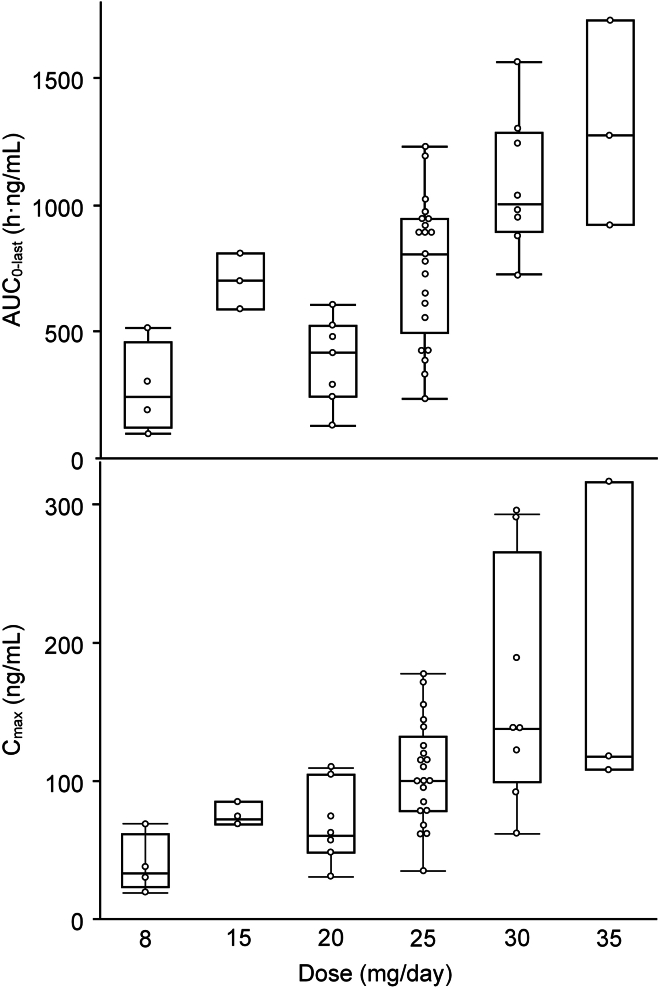
Avanbulin Cmax and AUC0–last increased with increasing dose across the range of 8–35 mg/day lisavanbulin for patients with GBM or high-grade glioma. For a 2-fold increase in dose from 15 to 30 mg/day, there was an approximate 1.9-fold and 1.3-fold increase in the geometric mean Cmax on cycle 1 day 1 and cycle 2 day 1, respectively. The corresponding increases in AUC0–last were 1.5-fold and 1.6-fold, respectively. At the MTD level of 30 mg/day for these patients, cycle 1 day 1 Cmax and AUC0–inf were approximately 147 ng/mL and 1,575 h∗ng/mL, respectively (Figure 2).
Figure 2.
Pharmacokinetic profile of patients with GBM or high-grade glioma (N = 46) on cycle 1 day 1
Circles represent values for each patient. Boxes represent quartiles (25%, 50%, 75%) and whiskers the outliers.
See also Figures S1 and S2; Table S8.
A further analysis was undertaken to better understand the pharmacokinetics profile of phase 1 patients who experienced hallucinations of any grade or significant hyponatremia of grade ≥3 when treated at doses at or above ≥20 mg/day. The total number was 8/31 patients (25.8%), and logistic regression based on a model with avanbulin AUCinf and cohort suggested an increased probability of experiencing any of these events when exposure exceeded 2,375 h∗ng/mL (Table S8; Figure S1). A quantile regression model estimated a 28% and 12% probability of exceeding this threshold at 30 and 25 mg/day, respectively (see Figure S2). Given the totality of available safety, tolerability, and pharmacokinetic modeling, the lower dose of 25 mg/day was therefore recommended to be taken forward in phase 2.
Efficacy
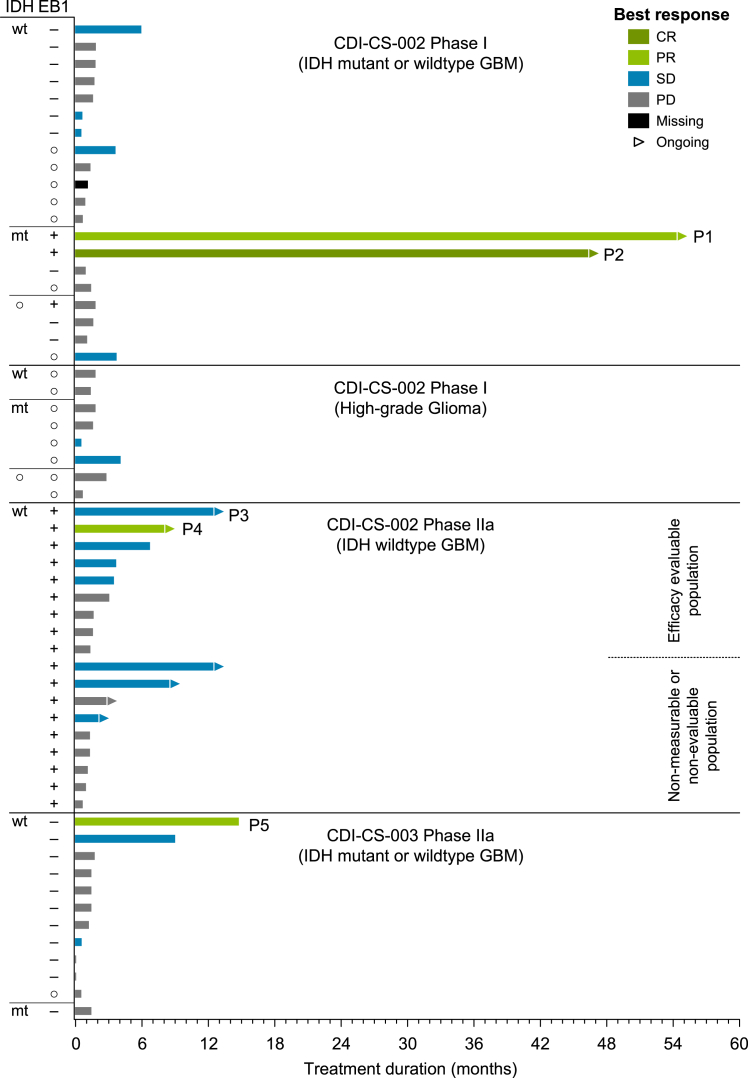
Among 28 evaluable patients with relapsed GBM/recurrent high-grade glioma enrolled in the phase 1, one patient (30 mg/day) had a confirmed radiological complete response, and one patient (25 mg/day) had a confirmed durable partial response (PR) per response assessment in neuro-oncology (RANO) of −94%. Both patients had been on treatment for over 4 years when the study was completed and transitioned to the compassionate access post-trial program (Figure 3). Seven other patients had SD, resulting in a disease control rate of 32.1%. Median PFS was 1.6 months (95% confidence interval [CI] 1.4, 1.7), and the probability of being event-free at 12 months was 11.2% (95% CI 2.3, 28.2). MRI scans for patients showing responses are provided in Figure S3.
Figure 3.
Treatment duration, best objective response, IDH status (wt, wild type; mt, mutation; ○, unknown), and EB1 status (+, positive; −, negative; ○, unknown) of patients with GBM or high-grade glioma in study CDI-CS-002 and study CDI-CS-003
For patients with objective response (P1, P2, P4, and P5) and near objective response (P3), see also Figures 4 and S3. Note: the study design uses the 2016 WHO classification, and therefore, based on the 2021 classification, some patients would not be considered to have GBM.
Both of the exceptional durable responders in phase 1 were retrospectively found to be EB1+ in immunohistochemistry (IHC), leading to the development of a CLIA-certified immunohistochemistry assay to prescreen patients for moderate/high EB1 expression for inclusion in the phase 2a portion of the study.
Thirteen patients with relapsed GBM were enrolled into the first stage of the phase 2a study and were evaluable for efficacy, with nine having measurable disease. There was one confirmed durable PR, again continuing for more than a year when the patient switched to the compassionate access post-trial program. Four other patients had SD radiologically (one patient being close to achieving a PR) with a disease control rate for the patients with measurable disease of 5/9, 55.6% (95% CI 21.2, 86.3). For the 13 patients including those without measurable disease at baseline, the probability of being event free at 12 months in this relapsed population was 32.5% (95% CI 8.9, 59.2) with a 12-month overall survival of 55.6% (95% CI 20.4, 80.5) (Figure 1; Table 1). With only 1 confirmed PR out of 9 efficacy-evaluable patients with GBM, the study did not meet the pre-specified threshold to progress and was closed to further recruitment.
Table 1.
Objective responses and disease control in patients with GBM or high-grade glioma, phase 1 and phase 2a
| Lisavanbulin dose |
||||||||
|---|---|---|---|---|---|---|---|---|
| Phase 1 |
Phase 2a |
|||||||
| 8 mg/day | 15 mg/day | 20 mg/day | 25 mg/day | 30 mg/day | 35 mg/day | Total | 25 mg/day | |
| N | 4 | 3 | 7 | 3 | 8 | 3 | 28 | 9 |
| Best response, n (%) | ||||||||
| Complete response | 0 | 0 | 0 | 0 | 1 (12.5) | 0 | 1 (3.6) | 0 |
| Partial response | 0 | 0 | 0 | 1 (33.3) | 0 | 0 | 1 (3.6) | 1 (11.1) |
| Stable disease | 0 | 1 (33.3) | 2 (28.6) | 1 (33.3) | 1 (12.5) | 2 (66.7) | 7 (25.0) | 4 (44.4) |
| Progressive disease | 4 (100) | 2 (66.7) | 5 (71.4) | 1 (33.3) | 5 (62.5) | 1 (33.3) | 18 (64.3) | 4 (44.4) |
| Missing | 0 | 0 | 0 | 0 | 1 | 0 | 1 | 0 |
| Objective response rate | ||||||||
| n (%) | 0 | 0 | 0 | 1 (33.3) | 1 (12.5) | 0 | 2 (7.1) | 1 (11.1) |
| Exact 95% CI | 0.0, 60.2 | 0.0, 70.8 | 0.0, 41.0 | 0.8, 90.6 | 0.3, 52.7 | 0.0, 70.8 | 0.9, 23.5 | 0.3, 48.2 |
| Disease control rate | ||||||||
| n (%) | 0 | 1 (33.3) | 2 (28.6) | 2 (66.7) | 2 (25.0) | 2 (66.7) | 9 (32.1) | 5 (55.6) |
| Exact 95% CI | 0.0, 60.2 | 0.8, 90.6 | 3.7, 71.0 | 9.4, 99.2 | 3.2, 65.1 | 9.4, 99.2 | 15.9, 52.4 | 21.2, 86.3 |
| Progression-free survival | ||||||||
| Median (months) | 1.6 | 1.8 | 1.6 | 6.0 | 1.5 | 1.4 | 1.6 | 3.5 |
| 95% CI | (0.7, 1.6) | (1.0, 3.4) | (0.8, 3.4) | (1.7, NA) | (0.8, NA) | (NA, NA) | (1.4, 1.7) | (1.6, NA) |
Phase 1: patients with recurrent or progressive glioblastoma or high-grade glioma, full analysis population.
Phase 2a: patients with EB1-positive glioblastoma, efficacy-evaluable population, patients with measurable disease.
There were no objective responses in patients with heavily pre-treated non-glioma enrolled in the phase 1 portion of the study, and this was not explored further.
Biomarker analysis
Thirteen patients with recurrent high-grade glioma/GBM had tissue that could be retrospectively analyzed for EB1 expression. Ten were EB1 negative, with three patients experiencing SD as the best response. Three patients had archival tumor tissue that was EB1 positive, including the two exceptional durable responders.
In the EB1 pre-screening program for the phase 2a component of the study, samples from 64 of 629 patients (10.2%) were EB1 positive (defined as >50% of tumor cells with moderate or strong EB1 staining). Of the 9 evaluable patients with EB1-positive tumors enrolled onto phase 2a, objective antitumor activity was observed in two patients. One patient had a durable target lesion size reduction of 58%, corresponding to PR per RANO, and one patient had a durable reduction of 44% corresponding to SD per RANO. As compared with phase 1 responses, these results suggest that preselection for EB1 expression per IHC did not enrich the patient population for sensitivity to lisavanbulin.
Isocitrate dehydrogenase (IDH) status was not planned to be collected prospectively as part of the trial protocol, but this information was obtained retrospectively. Both the exceptional responders from the phase 1 portion of the study had IDH-mutant tumors (1 patient had the IDH R132H mutation detected by IHC, and another had the IDH R132C mutation detected by RNA sequencing), and according to the updated 2021 WHO classification of CNS tumors, these patients would now be considered as IDH-mutant high-grade astrocytomas. In the phase 2a portion of the study, the exceptional responder was IDH wild type by IHC and sequencing, as were the patient with SD and a lesion size reduction close to a PR and another exceptional responder from study CDI-CS-003.
A responder-non-responder analysis was therefore undertaken using RNA sequencing of archival tissue. RNA sequencing was performed for patients where adequate tissue samples were available for analysis, who had GBM according to WHO 2016 criteria, had received doses where responses had been observed (i.e., 20–30 mg/day), and had not been discontinued for reasons other than radiologically confirmed disease progression. This analysis identified a five-gene response classifier (Figure 4) that is distinct from expression patterns observed with EB1-IHC positivity and predicts current responses irrespective of IDH status. This signature is characterized by homeobox gene downregulation, which may be implicated in the control of microtubule dynamics. A prevalence appraisal using The Cancer Genome Atlas Program (TCGA) revealed that 22% (34/153) of TCGA GBM samples had a call strength of 0.5–1 for response when applying the five-gene classifier. This includes most of the IDH1 mutants but also a sizable proportion of IDH1 wild-type samples (see Figure 5).
Figure 4.
Five-gene response classifier based on GBM samples from study CDI-CS-002 (n = 15) and study CDI-CS-003 (n = 1)
RNA sequencing was performed for patients where adequate tissue samples were available for analysis, who had GBM according to WHO 2016 criteria, had received doses where responses had been observed (i.e., 20–30 mg/day), and had not been discontinued for reasons other than radiologically confirmed disease progression.
Figure 5.
Prevalence of the five-gene response classifier in TCGA GBM database
Discussion
There remains a significant unmet need for novel effective therapies for high-grade gliomas.23,24 MTAs are among the most active cytotoxic anticancer drugs currently in use, inducing either polymerization or depolymerization of microtubules. There are two categories of MTAs: stabilizers (including taxanes and epothilones) and destabilizers (including Vinca alkaloids, halichondrins, and combretastatins). They have a broad spectrum of activity and are used in the treatment of several types of hematologic malignancies and solid tumors. Microtubules play a role in a range of critical cell functions including mitosis. Stabilizing or destabilizing the microtubule polymer results in spindle assembly poisoning and the slowing of mitosis, leading to apoptotic cell death.25 Here, our clinical trial results together with translational studies provide further insights into both the challenges and promise of treating brain tumors with MTAs.
We report our phase 1/2a study of the synthetic MTA lisavanbulin where we describe the safety, tolerability, and preliminary efficacy. The dose selected for patients with brain tumor was 25 mg/day, below the MTD of 30 mg/day given pharmacological modeling of exposures with an increased risk of the more significant toxicities of hallucinations or ≥grade 3 hyponatremia. Overall, this was well tolerated with reversible toxicities at the RP2D.
The population of this study included heavily pretreated patients who had exhausted all prior treatment. In this study, we noted exceptional durable partial or complete responses in three patients with relapsed brain tumors: 2 patients with IDH-mutant GBM (these would now have been reclassified as IDH-mutant astrocytoma per WHO 2021) in phase 1 and 1 patient with IDH-wild-type GBM in phase 2a. The two patients from phase 1 were clinically free of disease symptoms for more than 4 years prior to switching to the compassionate access program. The third patient switched to compassionate access at time of study closure. Overall disease control rate for the patients with relapsed brain tumors treated in this study (in both phase 1 and phase 2a) was 14/37 (37.8%).
Our experience in this study did not confirm EB1 as a reliable predictor of response to lisavanbulin. We could not adequately assess the sensitivity of IDH-mutated tumors vs. IDH-wild-type tumors in this study as the protocol was recruiting prior to the WHO 2021 update and did not prespecify the collection of molecular subtyping data. Additionally, all patients recruited onto the phase 2a portion of the study were IDH wild type. We therefore used an unbiased approach to undertake a responder-non-responder analysis based on RNA sequencing of archival brain tumor (n = 5 responders vs. n = 11 non-responders from this study and study CDI-CS-003). This signature has an expected prevalence of approximately 20% in patients with GBM (IDH wild type) and a higher prevalence in IDH-mutant astrocytoma. This signature would require prospective evaluation in a future clinical study in patients with GBM and/or IDH-mutant astrocytoma.
In conclusion, oral lisavanbulin was well tolerated at doses of up to 30 mg/day in patients with recurrent GBM or high-grade glioma. Lisavanbulin was associated with profound, durable responses and clinical benefit in a small subset of patients with relapsed GBM. Further investigation is warranted to explore the use of our response-predictive molecular signature for the selection of patients with brain tumors for treatment with lisavanbulin.
Limitations of the study
The study was designed in 2014 predating the WHO reclassification of brain tumors, hence allowing for the inclusion of both IDH-wild-type and IDH-mutant brain tumors. Data on IDH status were collected retrospectively; however, the mixed population limits the efficacy readout in each of these subtypes of brain tumors. The 2016 WHO classification used in the study meant that potentially some patients would not be considered to have GBM based on the 2021 classification. However, among the patients with GBM (per 2016 classification) for whom sequencing data were available, all but two patients (identified as P1 and P2 in Figure 4) would be classified as having GBM according to the 2021 classification. The other two patients would be classified as having astrocytoma.
Resource availability
Lead contact
Further information and requests for resources should be directed to and will be fulfilled by the lead contact, Thomas Kaindl (thomas.kaindl@basilea.com).
Materials availability
This study did not generate new unique reagents.
Data and code availability
This paper does not report original code. The individual patient and sequencing data reported cannot be deposited in a public repository secondary to patient privacy concerns. The comparatively low incidence of high-grade gliomas and GBMs overall, the non-standard-of-care usage of study medication, and the limited sample size of the study—all of these factors allow patients to be potentially identified from online publicly available datasets. For access to the data, please submit a request for access to the lead contact (thomas.kaindl@basilea.com). The study sponsor has the primary responsibility of data storage and management. Upon receiving a request, the study sponsor will review the request and if approved, work with the requestor to securely transmit the clinical study data in a GDPR-compliant manner and adhering to the consent agreements established with each study participant. In that context, data recipients are required to enter a formal data-sharing agreement that describes the conditions for release and requirements for data transfer, storage, archiving, publication, and intellectual property. Any additional information required to reanalyze the data reported in this paper is available from the lead contact upon request.
Acknowledgments
This study was funded by Basilea Pharmaceutica International Ltd, Allschwil, Switzerland.
The results shown here are in part based upon data generated by TCGA Research Network: https://www.cancer.gov/tcga.
Author contributions
Conceptualization, M.E. and H. Lane; methodology, M.E. and T.K.; formal analysis, T.K. and M.E.; investigation, J.S.L., S.H., R.P., P.M.C., T.R.J.E., H. Läubli, P.R., R.K., L.B., G.T., A.W., B.W., K.B., J.R.E., P.H., T.H., and J.S.; writing – original draft, J.S.L. and T.K.; writing – review and editing, J.S.L., S.H., R.P., P.M.C., T.R.J.E., H. Läubli, P.R., R.K., L.B., G.T., A.W., B.W., K.B., J.R.E., H. Lane, M.E., T.K., P.H., T.H., and J.S.; visualization, T.K.; project administration, M.E. and T.K.
Declaration of interests
T.K., M.E., and H. Lane are employees of Basilea Pharmaceutica International Ltd.
STAR★Methods
Key resources table
| REAGENT or RESOURCE | SOURCE | IDENTIFIER |
|---|---|---|
| Antibodies | ||
| Purified Mouse Anti-EB1, clone 5/EB1 | BD Transduction Laboratories | Cat#610535; RRID: AB_397892 |
| Biological samples | ||
| Blood | Patients in this study | N/A |
| Formalin-fixed paraffin-embedded (FFPE) archival tumor specimens | Patients in this study | N/A |
| Chemicals, peptides, and recombinant proteins | ||
| Lisavanbulin | Basilea Pharmaceutica International Ltd | BAL101553 |
| Software and algorithms | ||
| SAS software version 9.4 | SAS Institute, Cary, NC, USA | www.sas.com |
Experimental model and study participant details
Study design and participants
This was an open-label, multicenter, Phase I/IIa study in patients with solid tumors or with recurrent or progressive glioblastoma (GBM) or other high-grade glioma. The study was conducted in two portions: The Phase I portion enrolled patients with histologically- or cytologically confirmed advanced or recurrent solid tumors (with measurable disease according to Response Evaluation Criteria in Solid Tumors [RECIST] v1.1 or non-measurable prostate or ovarian cancer) who failed standard therapy, or for whom no effective standard therapy was available, or histologically-confirmed GBM or high-grade glioma (with measurable disease defined by contrast-enhancing MRI), with progressive or recurrent disease after prior radiotherapy, with or without chemotherapy.
The Phase IIa portion that enrolled patients with recurrent, histologically confirmed, GBM with tumor tissue positive for EB1 (this population was selected given the observation of responses in EB1-positive GBM patients in the Phase I portion), either patients with de novo GBM after prior radical chemoradiotherapy or secondary GBM after prior chemotherapy or radiotherapy. Patients had to be neurologically stable, without progression of neurologic symptoms, within 15 days prior to starting study drug. Patients with EB1 positive tumors were identified in a pre-screening program using a CE-marked immunohistochemistry clinical trial assay. EB1 positivity was assessed in a central laboratory by a board certified neuropathologist (Discovery Life Sciences, Targos Molecular Pathology).
For both study portions, adult patients (≥18 years of age), with a life expectancy of ≥12 weeks, acceptable organ and marrow function within 15 days prior to starting study drug.
Key exclusion criteria in patients with recurrent or progressive GBM or high-grade glioma included peripheral neuropathy ≥ CTCAE grade 2; systolic blood pressure ≥160 mmHg or diastolic blood pressure ≥100 mmHg; radiotherapy within 12 weeks, prior anti-tumor chemotherapy within 4 weeks, or within 6 weeks for nitrosoureas, surgical resection within 4 weeks, or a stereotactic biopsy/core biopsy within 1 week. Patients taking more than two antihypertensive medications were also excluded, as were those with significant cardiac disease or abnormality, and clinically significant acute intratumoral hemorrhage. More detailed inclusion and exclusion criteria can be found in the supplemented study protocol in Data S1.
In the Phase Ia portion, it was planned to include up to 42 patients with solid tumors and up to 28 patients with GBM or high-grade glioma, and in the Phase IIa portion it was planned to include up to 34 evaluable EB1-positive GBM patients, up to 19 patients with measurable disease and up to 15 patients with non-measurable disease at baseline.
The Independent Ethics Committees of the fourteen study sites, and relevant authorities in the UK, Switzerland, Germany, and Belgium approved the study protocol. The study was conducted in accordance with the Declaration of Helsinki, International Conference on Harmonisation Guidelines for Good Clinical Practice, and applicable laws and regulations. All patients provided written informed consent prior to study participation.
Method details
Study treatment and assessments
Lisavanbulin was dosed once daily under fasting conditions, as hard capsules containing 1 mg or 5 mg of drug. Each treatment cycle comprised 28 days of continuous, daily oral lisavanbulin administration to patients in a fasted state. In the Phase I portion of the study, each patient was scheduled to receive at least two 28-day treatment cycles. Patients were replaced if the minimum safety evaluation requirements for assessment of the MTD in Cycle 1 were not met. Patients were allowed to receive repeated 28-day treatment cycles until the occurrence of progressive disease, or unacceptable toxicity. The starting dose in the Phase I portion was 2 mg/day in patients with advanced or recurrent solid tumors; starting doses for patients with recurrent or progressive GBM or high-grade glioma, were at least one dose level below the dose level that had already been demonstrated to be safe and well tolerated in patients with advanced or recurrent solid tumors. Dose escalation was conducted using a 3 + 3 titration design. to determine the maximum tolerated dose of oral lisavanbulin and to characterize dose-limiting toxicity. Patients with solid tumors or with recurrent or progressive GBM or other high-grade glioma were enrolled in sequential cohorts of three to six patients with each cohort given an increased lisavanbulin dose, using a flat-fixed dosing approach. Safety, pharmacokinetics and antitumor activity were assessed for each dose level. In the Phase IIa portion of the study, patients received the recommended Phase II dose defined in the Phase I portion.
The Phase IIa dose expansion part of the study aimed to obtain efficacy data in patients with recurrent EB1-positive GBM, and to further characterize the safety and tolerability of lisavanbulin at the recommended Phase II dose.
Safety assessments were conducted throughout the study and included AEs according to the National Cancer Institute CTCAE v4.03/v5.0 (Phase I/IIa), laboratory parameters, ECG, transthoracal echocardiography, chest X-ray (Phase I)/computed tomography (CT)/MRI, vital signs, ECOG performance status, physical examination, and evaluation of concomitant medications.
Pharmacokinetic variables calculated from plasma concentration data using noncompartmental analysis for lisavanbulin (if applicable) and for avanbulin comprised: maximum observed plasma concentration (Cmax), time to maximum concentration (Tmax), area under curve (AUC)0–τ, AUC0–last, AUC0–∞, t½, systemic clearance and volume of distribution. Samples for pharmacokinetic analysis were taken on Day 1 (pre-dose, 0.5, 1, 2, 3, 4, 6, 8 and 24 h after dosing), and Day 8, 15 and 22 (pre-dose) of Cycles 1 and 2. Additional blood samples were taken at the end of the study, and in the Phase I portion at the occurrence of a DLT, and on the first dosing day of the new dose levels from patients undergoing dose escalation or reduction.
Efficacy was assessed in the Phase I portion using RECIST criteria v1.1 in patients with solid tumor and measurable disease. In both the Phase I and Phase IIa portions, for patients with recurrent or progressive GBM or high-grade glioma, evaluation of disease progression and response was assessed by contrast-enhanced MRI, based on the RANO criteria. Clinical progression of the tumor disease was also considered as progressive disease.
Objectives
For the Phase I portion, the primary objectives were to determine the MTD and to characterize DLTs of daily oral lisavanbulin. Secondary objectives were to further assess the safety and tolerability of lisavanbulin, to evaluate the pharmacokinetics of avanbulin and lisavanbulin, and to assess the anti-tumor activity of lisavanbulin.
For the Phase IIa portion, the primary objective was to assess the efficacy of daily oral lisavanbulin, based on the objective response rate (ORR) as per RANO criteria, in patients with recurrent EB1-positive GBM. Secondary objectives were to evaluate the efficacy of lisavanbulin based on overall survival (OS), progression-free survival (PFS) and the proportion of patients with PFS at 6 months after start of study drug treatment (PFS6), to assess the safety and tolerability of lisavanbulin, and to evaluate the pharmacokinetics of avanbulin and lisavanbulin.
Quantification and statistical analysis
The details of the statistical analysis plan were described in Data S1. Statistical analysis was undertaken using SAS version 9.4 (SAS Institute, Cary, NC, USA). The Phase I and Phase IIa portions of the study were analyzed separately. Within the Phase I portion, patients with solid tumors and those with GBM/high grade glioma were treated as separate populations. Analysis populations were defined as follows:
The full analysis population (FAP) included patients who received at least one partial or complete dose of lisavanbulin. The FAP was the primary population for analyzing efficacy in Phase I.
The safety population (FAP with at least one post-baseline safety assessment) was used for the overall analysis of safety.
The PK population (FAP with at least one post-baseline PK assessment) was used to assess PK.
The MTD-determining population consisted of patients who received at least one dose of lisavanbulin and experienced a DLT, or received at least 24 of the scheduled 28 doses of lisavanbulin in Cycle 1 without a DLT; and had been observed for ≥28 days following the first dose and had been evaluated for safety.
The efficacy evaluable population (EEP), used in the Phase IIa portion was a subset of the FAP who had at least one post-baseline RANO assessment after having received at least 6 weeks of study treatment.
Efficacy endpoints were best objective response (complete response (CR), partial response (PR), stable disease (SD) or progressive disease (PD)) per RECIST criteria v1.1 for patients with solid tumors and RANO criteria in patients with GBM/high-grade glioma; the ORR was calculated as the proportion of patients with CR or PR; the disease control rate (DCR) was calculated as the proportion of patients with CR, PR or SD. For ORR and DCR the portion and exact 95% confidence intervals were provided. Patients whose clinical response was unknown or not reported were treated as non-responders.
For the Phase IIa portion a Simon’s two-stage design was used; the null hypothesis that the true response rate was 14% was tested against a one-sided alternative. In the first stage, 9 evaluable patients were accrued. If there were 1 or fewer responses in these 9 patients, the study was to be stopped. Otherwise, 10 additional evaluable patients were accrued for a total of 19. The null hypothesis was rejected if 6 or more responses were observed in 19 patients. This design yields a type I error rate of 3.7% and power of 81% when the true response rate is 40%.
Progression-free survival (PFS) was defined as the interval between the date of first dose and the earliest date of objective disease progression. Overall survival (OS) was the time from first study drug administration to the date of death/or censored at the time the patient was last known to have been alive. Patients or their families were contacted at 3-monthly intervals after the last dose of study drug to ascertain their survival status; more frequent survival updates were collected if patients attended the study centers for other reasons. Kaplan-Meier methods were used for the assessment of OS, PFS and the proportion of patients with PFS at 6 months (PFS6). The 95% confidence intervals were calculated with Brookmeyer-Crowley method.
Additional resources
This study has been registered on https://clinicaltrials.gov/, ID: NCT02490800.
Published: June 17, 2025
Footnotes
Supplemental information can be found online at https://doi.org/10.1016/j.xcrm.2025.102165.
Contributor Information
Juanita Suzanne Lopez, Email: juanita.lopez@icr.ac.uk.
Thomas Kaindl, Email: thomas.kaindl@basilea.com.
Supplemental information
References
- 1.Prota A.E., Danel F., Bachmann F., Bargsten K., Buey R.M., Pohlmann J., Reinelt S., Lane H., Steinmetz M.O. The novel microtubule-destabilizing drug BAL27862 binds to the colchicine site of tubulin with distinct effects on microtubule organization. J. Mol. Biol. 2014;426:1848–1860. doi: 10.1016/J.JMB.2014.02.005. [DOI] [PubMed] [Google Scholar]
- 2.Duran G.E., Lane H., Bachmann F., Sikic B.I. In vitro activity of the novel tubulin active agent BAL27862 in MDR1(+) and MDR1(-) human breast and ovarian cancer variants selected for resistance to taxanes. Cancer Res. 2010;70:4412. doi: 10.1158/1538-7445.AM10-4412. [DOI] [Google Scholar]
- 3.Estève M., Honoré S., McKay N., Bachmann F., Lane H., Braguer D. BAL27862: A unique microtubule-targeted agent that severs microtubules and overcomes multifactorial drug resistance. J. Clin. Oncol. 2010;28:e13589. doi: 10.1200/jco.2010.28.15_suppl.e13589. [DOI] [Google Scholar]
- 4.Bachmann F., Burger K., Duran G.E., Sikic B.I., Lane H.A. Abstract 831: BAL101553 (prodrug of BAL27862): A unique microtubule destabilizer active against drug refractory breast cancers alone and in combination with trastuzumab. Cancer Res. 2014;74:831. doi: 10.1158/1538-7445.AM2014-831. [DOI] [Google Scholar]
- 5.Bergès R., Tchoghandjian A., Honoré S., Estève M.A., Figarella-Branger D., Bachmann F., Lane H.A., Braguer D. The novel tubulin-binding checkpoint activator BAL101553 inhibits EB1-dependent migration and invasion and promotes differentiation of glioblastoma stem-like cells. Mol. Cancer Ther. 2016;15:2740–2749. doi: 10.1158/1535-7163.MCT-16-0252. [DOI] [PubMed] [Google Scholar]
- 6.Bergès R., Tchoghandjian A., Sergé A., Honoré S., Figarella-Branger D., Bachmann F., Lane H.A., Braguer D. EB1-dependent long survival of glioblastoma-grafted mice with the oral tubulin-binder BAL101553 is associated with inhibition of tumor angiogenesis. Oncotarget. 2020;11:759–774. doi: 10.18632/oncotarget.27374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Foster-Gross N., Bachmann F., McSheehy P., Lane H. BAL101553, a novel microtubule-targeting tumor checkpoint controller, synergizes with gemcitabine providing cures in a PDX-pancreatic model. Eur. J. Cancer. 2018;103:e6. doi: 10.1016/S0959-8049(18)31491-6. [DOI] [Google Scholar]
- 8.Kolb E.A., Gorlick R., Keir S.T., Maris J.M., Kang M.H., Reynolds C.P., Lock R.B., Carol H., Wu J., Kurmasheva R.T., et al. Initial testing (stage 1) of BAL101553, a novel tubulin binding agent, by the pediatric preclinical testing program. Pediatr. Blood Cancer. 2015;62:1106–1109. doi: 10.1002/PBC.25329. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lane H., McSheehy P., Bachmann F. BAL101553, a novel microtubule-targeting tumor checkpoint controller, synergizes with gemcitabine providing cures in a PDX-pancreatic model. Eur. J. Cancer. 2018;103:e8. doi: 10.1016/S0959-8049(18)31491-6. [DOI] [Google Scholar]
- 10.Burgenske D.M., Talele S., Pokorny J.L., Mladek A.C., Bakken K.K., Carlson B.L., Schroeder M.A., He L., Hu Z., Gampa G., et al. Preclinical modeling in glioblastoma patient-derived xenograft (GBM PDX) xenografts to guide clinical development of lisavanbulin—a novel tumor checkpoint controller targeting microtubules. Neuro Oncol. 2022;24:384–395. doi: 10.1093/neuonc/noab162. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Sharma A., Broggini-Tenzer A., Nytko-Karouzakis K., Guckenberger M., Bachmann F., Lane H., Pruschy M. The novel microtubule-destabilizing drug BAL101553 acts as radiosensitizing agent in treatment refractory tumor models. Strahlenther. Onkol. 2017;193:870. doi: 10.1007/s00066-017-1195-9. [DOI] [PubMed] [Google Scholar]
- 12.Sharma A., Broggini-Tenzer A., Vuong V., Messikommer A., Nytko K.J., Guckenberger M., Bachmann F., Lane H.A., Pruschy M. The novel microtubule targeting agent BAL101553 in combination with radiotherapy in treatment-refractory tumor models. Radiother. Oncol. 2017;124:433–438. doi: 10.1016/j.radonc.2017.07.024. [DOI] [PubMed] [Google Scholar]
- 13.Bachmann F., Lane H. 421 Dual Mechanism of Action of the Novel Microtubule-targeting Drug BAL27862 (active Moiety of the Prodrug BAL101553): Targeting Tumor and Vascular Cells. Eur. J. Cancer. 2012;48:128. doi: 10.1016/S0959-8049(12)72219-0. [DOI] [Google Scholar]
- 14.Schmitt-Hoffmann A., Klauer D., Gebhardt K., Fullhardt P., Brendle A., Hardgreaves P., Schlaefle C., Bucher C., Spickermann J., Defoin R., et al. BAL27862: A unique microtubule-targeted agent with a potential for the treatment of human brain tumors. Mol. Cancer Ther. 2009;8:C233. doi: 10.1158/1535-7163.TARG-09-C233. [DOI] [Google Scholar]
- 15.Akhmanova A., Steinmetz M.O. Microtubule +TIPs at a glance. J. Cell Sci. 2010;123:3415–3419. doi: 10.1242/jcs.062414. [DOI] [PubMed] [Google Scholar]
- 16.Berges R., Baeza-Kallee N., Tabouret E., Chinot O., Petit M., Kruczynski A., Figarella-Branger D., Honore S., Braguer D. End-binding 1 protein overexpression correlates with glioblastoma progression and sensitizes to Vinca-alkaloids in vitro and in vivo. Oncotarget. 2014;5:12769–12787. doi: 10.18632/ONCOTARGET.2646. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Mohan R., Katrukha E.A., Doodhi H., Smal I., Meijering E., Kapitein L.C., Steinmetz M.O., Akhmanova A. End-binding proteins sensitize microtubules to the action of microtubule-targeting agents. Proc. Natl. Acad. Sci. USA. 2013;110:8900–8905. doi: 10.1073/PNAS.1300395110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kristeleit R., Evans J., Molife L.R., Tunariu N., Shaw H., Slater S., Haris N.R.M., Brown N.F., Forster M.D., Diamantis N., et al. Phase 1/2a trial of intravenous BAL101553, a novel controller of the spindle assembly checkpoint, in advanced solid tumours. Br. J. Cancer. 2020;123:1360–1369. doi: 10.1038/S41416-020-1010-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Pohlmann J., Bachmann F., Schmitt-Hoffmann A., Gebhardt K., Spickermann J., Nuoffer C., Biringer G., O'Reilly T., Pruschy M., Lane H.A. Abstract 1347: BAL101553: An optimized prodrug of the microtubule destabilizer BAL27862 with superior antitumor activity. Cancer Res. 2011;71:1347. doi: 10.1158/1538-7445.AM2011-1347. [DOI] [Google Scholar]
- 20.Joerger M., Hundsberger T., Haefliger S., von Moos R., Hottinger A.F., Kaindl T., Engelhardt M., Marszewska M., Lane H., Roth P., Stathis A. Safety and anti-tumor activity of lisavanbulin administered as 48-hour infusion in patients with ovarian cancer or recurrent glioblastoma: a phase 2a study. Invest. New Drugs. 2023;41:267–275. doi: 10.1007/S10637-023-01336-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Joerger M., Stathis A., Metaxas Y., Hess D., Mantiero M., Mark M., Volden M., Kaindl T., Engelhardt M., Larger P., et al. A Phase 1 study of BAL101553, a novel tumor checkpoint controller targeting microtubules, administered as 48-h infusion in adult patients with advanced solid tumors. Invest. New Drugs. 2020;38:1067–1076. doi: 10.1007/s10637-019-00850-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Holdhoff M., Ye X., Strowd R.E., Nabors B., Walbert T., Lieberman F.S., Bagley S.J., Fiveash J.B., Fisher J.D., Desideri S., et al. Lisavanbulin (BAL101553), a novel microtubule inhibitor, plus radiation in patients with newly diagnosed, MGMT promoter unmethylated glioblastoma. Neurooncol. Adv. 2024;6 doi: 10.1093/noajnl/vdae150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wen P.Y., Weller M., Lee E.Q., Alexander B.M., Barnholtz-Sloan J.S., Barthel F.P., Batchelor T.T., Bindra R.S., Chang S.M., Chiocca E.A., et al. Glioblastoma in adults: a Society for Neuro-Oncology (SNO) and European Society of Neuro-Oncology (EANO) consensus review on current management and future directions. Neuro Oncol. 2020;22:1073–1113. doi: 10.1093/neuonc/noaa106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Weller M., van den Bent M., Preusser M., Le Rhun E., Tonn J.C., Minniti G., Bendszus M., Balana C., Chinot O., Dirven L., et al. EANO guidelines on the diagnosis and treatment of diffuse gliomas of adulthood. Nat. Rev. Clin. Oncol. 2021;18:170–186. doi: 10.1038/s41571-020-00447-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mukhtar E., Adhami V.M., Mukhtar H., Adhami M., Mukhtar H. Targeting Microtubules by Natural Agents for Cancer Therapy. Mol. Cancer Ther. 2014;13:275–284. doi: 10.1158/1535-7163.MCT-13-0791. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
This paper does not report original code. The individual patient and sequencing data reported cannot be deposited in a public repository secondary to patient privacy concerns. The comparatively low incidence of high-grade gliomas and GBMs overall, the non-standard-of-care usage of study medication, and the limited sample size of the study—all of these factors allow patients to be potentially identified from online publicly available datasets. For access to the data, please submit a request for access to the lead contact (thomas.kaindl@basilea.com). The study sponsor has the primary responsibility of data storage and management. Upon receiving a request, the study sponsor will review the request and if approved, work with the requestor to securely transmit the clinical study data in a GDPR-compliant manner and adhering to the consent agreements established with each study participant. In that context, data recipients are required to enter a formal data-sharing agreement that describes the conditions for release and requirements for data transfer, storage, archiving, publication, and intellectual property. Any additional information required to reanalyze the data reported in this paper is available from the lead contact upon request.