Abstract
Introduction
Metabolic and bariatric surgery (MBS) has been an established treatment option for patients with Type 2 diabetes mellitus (T2DM), but there is a relative paucity of evidence-based guidelines on preoperative, operative, and postoperative considerations concerning metabolic surgery for T2DM patients. To address this gap, we initiated a Delphi consensus process with a diverse group of international multidisciplinary experts.
Method
We embarked on a Delphi consensus-building exercise to propose an evidence-based expert consensus covering various aspects of MBS in patients with T2DM. We defined the scope of the exercise and proposed statements and surveyed the literature through electronic databases. The literature summary and voting process were conducted by 52 experts, who evaluated 44 statements. The quality of evidence was assessed using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) criteria.
Results
Consensus, defined as > 80% agreement, was reached for 43 out of 44 statements. The experts reached an agreement on the nature, terminology, and mechanisms of action of MBS. The currently available scores for predicting remission of T2DM after surgery are not robust enough for routine clinical use, and there is a need for further research to enable more personalized treatment. Additionally, they agreed that metabolic surgery for T2DM is cost-effective, and MBS procedures for treating T2DM vary in their safety and efficacy.
Conclusion
This Delphi expert consensus statement guides clinicians on various aspects of metabolic surgery for T2DM and also grades the quality of the available evidence for each of the proposed statements.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12902-025-01961-w.
Keywords: Metabolic Surgery, Bariatric Surgery, Type 2 diabetes mellitus
Introduction
Metabolic and Bariatric surgery (MBS) is an established treatment for patients with type 2 diabetes mellitus (T2DM) [1, 2]. Obesity is one of the most important risk factors for T2DM [1, 3], and significant weight loss achieved through MBS or other approaches can lead to improvement and remission of T2DM [1, 4]. Improvements in T2DM post-MBS, however, may also be mediated through non-weight-loss dependent mechanisms [4]. Both weight loss and T2DM improvement show considerable variation after MBS [5]. The most commonly performed surgical procedures, Roux-en-Y gastric bypass (RYGB) and sleeve gastrectomy (SG), are increasingly incorporated into treatment algorithms for T2DM [6, 7]. Despite this, there is a relative lack of evidence-based and clear guidelines on perioperative considerations concerning MBS for patients with T2DM.
The modified Delphi methodology is a recognized process for developing consensus amongst experts on topics that require additional high-quality evidence to inform current practice [8]. Since no consensus statement covers various aspects of MBS in patients with T2DM, we embarked on a Delphi consensus-building exercise. This work aimed to achieve a consensus amongst a multidisciplinary, international group of experts on multiple aspects of metabolic surgery for T2DM.
Methods
We established a multidisciplinary group of 52 experts, including metabolic and bariatric surgeons, endocrinologists, GPs with specialist interest, dietitians, psychologists, anaesthesiologists, and a patient representative from 23 countries (Supplementary Material 1), to carry out this consensus-building exercise. The selection of experts was based on their expertise, academic qualifications, proficiency in the English language, and willingness to participate in the exercise. Of 61 experts invited to join the project, 52 (85.0%) accepted the invitation. The Delphi methodology was shared with the group in advance, and two moderators (MK and KM) moderated all communication.
This adopted methodology has been used previously [8]. As the first step, experts were invited to submit statements covering various aspects of MBS for T2DM. These were then compiled by the moderators and presented to the group for further discussion. Group members were advised to only communicate through moderators to prevent any influence on opinions. Communication was through emails and online meetings with the moderators. The levels of agreement/disagreement were classified using a six-point Likert scale. Moderators assigned 2–3 statements to each expert in their areas of expertise to compile the evidence.
This work was further supplemented by a core study team of non-voting members (IO, SP, SS, and SK), who summarised the relevant literature. The core team researched literature and focused on high-quality evidence from levels 1 and 2. The moderators then reviewed the list of publications. This summary of evidence was then graded using the Grading of Recommendations, Assessment, Development, and Evaluation (GRADE) guideline criteria [9] (Table 1). The summary and the evidence grade were then shared with the consensus-building team.
Table 1.
GRADE criteria
| Code | Quality of Evidence | Definition |
|---|---|---|
| A | High | Further research is very unlikely to change our confidence in the estimate of effect; the statement can be supported by several high-quality studies with consistent results or, in special cases, by one large, high-quality multicentre trial |
| B | Moderate | Further research is likely to have an important effect on our confidence in the estimate of effect and might change the estimate; the statement can be supported by one high-quality study or several studies with some limitations |
| C | Low | Further research is very likely to have an important effect on our confidence in the estimate of effect and is likely to change the estimate; the statement can be supported by one or more studies with severe limitations |
| D | Very Low | Any estimate of effect is very uncertain; the statement can be supported by expert opinion or one or more studies with very severe limitations, or there might be no direct research evidence |
The committee discussed the proposed statements and went through several rounds of modifications. Ultimately, the committee agreed on a set of statements to vote on using the SurveyMonkey® platform. The first round of voting took place between July 17, 2022, to August 20, 2022. An agreement of ≥ 80% of experts was regarded as consensus. The results of the voting were then shared with the consensus building team.
The whole process was then repeated for the statements that the committee did not reach a consensus on after the first round of voting. During the second round, committee members were also able to add new statements, modify previous statements, or eliminate statements where no consensus was possible. After discussion over several rounds, a set of statements was proposed for second-round voting between October 21, 2022 and October 30, 2022.
Results
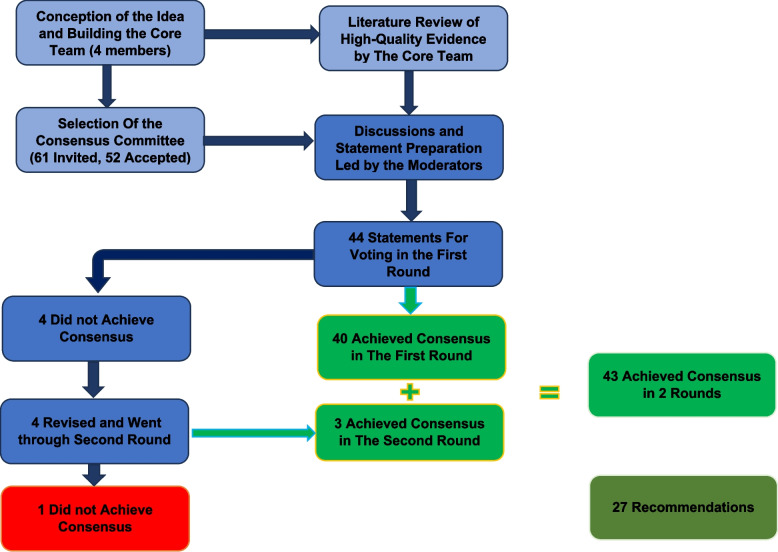
Fifty-two experts agreed on 43 statements, yielding 27 recommendations (Table 2) on MBS for T2DM after two rounds of the Delphi methodology. Figure 1 shows the study flowchart.
Table 2.
Statements and recommendations
| Statement | First Round | Second Round | Grade | Relevant Recommendations | |
|---|---|---|---|---|---|
| I | Nature, Terminology, and Mechanisms of Action | ||||
| 1 | Surgery for Type 2 Diabetes Mellitus (T2DM) is largely similar to bariatric surgery, the only difference being the primary focus, which is diabetes improvement/remission and prevention of complications of diabetes in this case, with weight loss being considered a secondary benefit. This allows patients with diabetes to be considered for surgery at a lower BMI than historically recommended thresholds for bariatric surgery and may influence the choice of surgical procedure. Care must, however, be taken to ensure that this does not result in stigmatization and under-treatment of people with other serious obesity-associated conditions | 96% Agree (Consensus) | - | Grade A | Surgery for Type 2 Diabetes Mellitus (T2DM) is similar to bariatric surgery, but allows patients with diabetes to be considered for surgery at a lower BMI |
| 2 | The term “Type 2 Diabetes Surgery” should be used as a common descriptor for surgery aimed at treating people with T2DM |
57% Agree (No -Consensus) |
61% Agree (Non-Consensus) |
Grade D | The term “Metabolic Surgery for T2DM” should be used as a common descriptor for surgery aimed at treating people with T2DM |
| 3 | The term “Metabolic Surgery for T2DM” should be used as a common descriptor for surgery aimed at treating people with T2DM |
77% Agree (No-Consensus) |
84% Agree (Consensus) |
Grade D | |
| 4 | Surgery has a strong evidence-based role in the treatment of some patients with Type 2 Diabetes Mellitus |
95% Agree (Consensus) |
Grade A | Surgery has a strong evidence-based role in the treatment of patients with T2DM that works through both weight-loss dependent and weight-loss independent mechanisms and changes multiple signals (metabolic, biliary, neural, hormonal) | |
| 5 | Surgery for T2DM works through both weight-loss-dependent and weight-loss-independent mechanisms |
94% Agree (Consensus) |
Grade B | ||
| 6 | The surgery alters how the gut communicates with the rest of the body by changing multiple signals (metabolic, biliary, neural, hormonal) |
92% Agree (Consensus) |
Grade A | ||
| II | Predictors of Remission | ||||
| 7 | The beneficial impact of surgery for T2DM depends on factors such as age, preoperative Body Mass Index (BMI), diabetes duration, preoperative glycaemic control, pancreatic beta-cell reserve, waist circumference, and the presence of other obesity-associated conditions |
96% Agree (Consensus) |
Grade A |
The beneficial impact of surgery for T2DM depends on factors such as younger age, higher baseline BMI, shorter diabetes duration, and better preoperative glycaemic control represented by higher c-peptide level, lower HbA1c, and less insulin usage. Surgery for T2DM ONLY has a role in individuals with a Body Mass Index of ≥ 30 kg/m2, or 27.5 kg/m2 in some Asian populations Despite some existing scores such as ABCD score and DiaRem, It seems that there is an essential need for more accurate predicting scores |
|
| 8 | Surgery for T2DM ONLY has a role in individuals with obesity (i.e., Body Mass Index of ≥ 30 kg/m2, or 27.5 kg/m2 in some Asian populations) |
86% Agree (Consensus) |
Grade A | ||
| 9 | Those with a longer duration of T2DM or on insulin are less likely to benefit from surgery in terms of remission but will benefit from improvement in glycaemic control, obesity, and other associated conditions |
92% Agree (Consensus) |
Grade A | ||
| 10 | Currently, available scores for predicting remission of T2DM after surgery are not robust enough for routine clinical use, and there is a need for greater research for more personalization of treatment |
86% Agree (Consensus) |
Grade B | ||
| III | Preoperative Diabetic Control | ||||
| 11 | HbA1c > 69 mmol/mol (≈ 8.5%) should not be considered an absolute contraindication for patients undergoing surgery for T2DM because of the effect of preoperative diet and surgery on glycemic control |
69% Agree (No- Consensus) |
83% Agree (Consensus) |
Grade B | HbA1c levels matter but all efforts should be made to optimize glycaemic control to an HbA1c level of ≤ 69 mmol/mol (≈ 8.5%) but surgery should not be delayed unduly to achieve this |
| 12 | All efforts should be made to optimize glycaemic control to an HbA1c level of ≤ 69 mmol/mol (≈ 8.5%), but surgery should not be delayed unduly to achieve this |
92% Agree (Consensus) |
Grade A | ||
| 13 | Patients undergoing surgery for T2DM should be reviewed by a diabetologist (or an appropriately trained healthcare professional supported by them) for the formulation of a plan for perioperative and postoperative glycaemic management of diabetes (including the period covering any preoperative liver-reducing diet) |
92% Agree (Consensus) |
Grade B | ||
| IV | Perioperative Care, Monitoring, and Special Considerations | ||||
| 14 | Preoperative liver-reducing diets are safe for these patients with adequate supervision and support from dietitians, pharmacists, and diabetologists as needed. Patients should receive appropriate advice regarding glucose and blood pressure-lowering medications and monitoring |
96% Agree (Consensus) |
Grade B | Patients undergoing surgery for T2DM should be reviewed by a diabetologist for perioperative and postoperative glycaemic management of diabetes (including preoperative liver-reducing diet and adjustment of dosage of glucose and blood pressure-lowering agents after surgery) | |
| 15 | Doses of glucose and blood pressure-lowering agents may need further adjustment after surgery in consultation with diabetologists (or an appropriately trained healthcare professional supported by diabetologists) |
98% Agree (Consensus) |
Grade A | ||
| 16 | Frequent capillary/interstitial glucose and blood pressure monitoring (in those on antihypertensives) are advised in the early postoperative phase to allow for titration of doses of medications used for these conditions |
94% Agree (Consensus) |
Grade A | Frequent capillary/interstitial glucose and blood pressure monitoring (in those on antihypertensives) are advised in the early postoperative phase to allow for titration of doses of medications | |
| 17 | Postprandial hypoglycemia (also known as reactive hypoglycemia) is a mostly mild complication of foregut surgery. It is more common after diversionary procedures, not more common in patients with T2DM, and can be serious |
87% Agree (Consensus) |
Grade A | Postprandial hypoglycemia (also known as reactive hypoglycemia) is more common after diversionary procedures, is no more common in patients with T2DM, and can be serious. These patients should be referred to diabetologists for further investigation and management | |
| 18 | Patients with suspected postprandial hypoglycemia should be referred to diabetologists for further investigation and management |
90% Agree (Consensus) |
Grade A | ||
| 19 | Sodium-Glucose Co-Transporter 2 Inhibitors (SGLT2i) should be stopped before surgery or before starting a preoperative low-energy diet to reduce the risk of ketoacidosis. The risk will persist for several months after surgery, and the decision to restart these drugs in the long term should only be made after a careful discussion of the pros and cons with the patient |
83% Agree (Consensus) |
Grade A | Based on the evidence we suggest stopping SGLT2 inhibitors 48 h prior to surgery to reduce the risk of ketoacidosis | |
| V | Care Pathways and Logistics of Service | ||||
| 20 |
Some of the barriers that prevent patients from accessing surgery for diabetes include (Insufficient awareness of the benefits and risks of surgery amongst many healthcare professionals, Insufficient awareness amongst patients on the safety and efficacy of surgery, Lack of funding or insurance coverage for surgery, Mandatory weight loss targets, Lack of capacity for delivering surgery to eligible patients, Weight bias towards access to healthcare, Stigma toward people with obesity, Shame amongst patients because of obesity) There is a need for targeted measures such as advocacy and evidence-based campaigns to address this issue |
100% Agree (Consensus) |
Grade A | There is a need for targeted measures to address barriers that prevent patients from accessing surgery for diabetes include (Insufficient awareness of the benefits and risks of surgery amongst many healthcare professionals and patients, Lack of funding or insurance coverage for surgery and Stigma toward people with obesity, Shame amongst patients because of obesity) | |
| 21 | All patients with T2DM meeting the criteria for surgery should be offered a choice of energy deficit diets (including meal replacement products) and/or available pharmacological options (if not used previously) with a full discussion of benefits, risks, and duration of treatment in comparison with surgery by a healthcare professional expert in weight management before referral for surgery. Evidence on comparative durability of effect should be specifically provided to ensure fully-informed decision-making |
88% Agree (Consensus) |
Grade A | While surgery is the most effective and durable, patients have a right to be informed about treatment options such as dietary and lifestyle interventions and pharmacological therapy with a full discussion of benefits, risks, and duration of treatment in comparison with surgery by a healthcare professional to make their own decisions about their preferred treatment | |
| 22 | Referral criteria for surgery should not be based on BMI alone and also consider the patient's other obesity-associated medical and mental health conditions and quality of life |
100% Agree (Consensus) |
Grade C | Referral criteria for surgery should not be based on BMI alone and also consider the patient's other obesity-associated medical and mental health conditions, and quality of life | |
| 23 | Mandatory medical weight management should not be a prerequisite for surgery for T2DM, but patients should have seriously attempted to lose weight in the past with the help of appropriate lifestyle/dietary education and available pharmacological interventions |
88% Agree (Consensus) |
Grade B | Medical weight management it’s not necessary for surgery for T2DM but patients should have seriously attempted to lose weight in the past with the help of appropriate lifestyle/dietary education and available pharmacological interventions | |
| 24 | Patients should receive comprehensive dietetic assessment and education, information about the risks and benefits of surgery, and education on living with surgery as part of routine preoperative assessment and planning |
100% Agree (Consensus) |
Grade A | Patients should receive comprehensive dietetic assessment and education, information about the risks and benefits of surgery, and access to a peer support group and MDT including dietitians, psychologists, and diabetologists both before and after surgery. Patients not eligible for surgery on the grounds of mental health contraindications should be referred for appropriate mental health treatment and support | |
| 25 | Patients should have access to a peer support group and multidisciplinary team, including dietitians, psychologists, and diabetologists, both before and after surgery |
98% Agree (Consensus) |
Grade A | ||
| 26 | Mental health contraindications of surgery include conditions such as unstable major affective or psychotic disorders; active substance/alcohol abuse; self-harm or suicidal attempts in the previous 12 months; severe, untreated eating disorder; and severe learning disability (without adequate family/social support making it difficult to cope with demands of surgery). Patients not eligible for surgery on the grounds of mental health contraindications should be referred for appropriate mental health treatment and support |
96% Agree (Consensus) |
Grade B | ||
| VI | Accurate Classification of Diabetes Mellitus | ||||
| 27 | Consider alternative diagnoses for T2DM, such as T1DM or Maturity Onset Diabetes of Young (MODY), before referring patients for surgery |
90% Agree (Consensus) |
Grade B | Consider alternative diagnoses for T2DM such as T1DM or Maturity Onset Diabetes of Young (MODY) before referring patients for surgery | |
| 28 | Surgery can be an option in selected patients with T1DM who also meet BMI criteria for bariatric surgery and may thus benefit from better glycaemic control and weight loss, but the decision for surgery should not be taken without consulting a diabetologist in these patients |
84% Agree (Consensus) |
Grade A | Surgery can be an option in selected patients with T1DM or Latent Autoimmune Diabetes in Adults (LADA) who also meet BMI criteria for bariatric surgery and may thus benefit from better glycaemic control and weight loss but the decision for surgery should not be taken without consulting a diabetologist in these patients | |
| 29 | Surgery can be an option in selected patients with Latent Autoimmune Diabetes in Adults (LADA) who also meet BMI criteria for bariatric surgery and may thus benefit from better glycaemic control and weight loss, but the decision for surgery should not be taken without consulting a diabetologist in these patients |
90% Agree (Consensus) |
Grade A | ||
| 30 | Routine antibody/C peptide level testing to rule out T1DM is unnecessary before surgery for diabetes. This should be left to the discretion of the diabetologists |
83% Agree (Consensus) |
Grade C | Antibody/C-peptide level can help to increase the accuracy of preoperative diabetes classification and rule out T1DM but it is unnecessary before surgery for diabetes | |
| VII | Surgical Considerations | ||||
| 31 | Patients undergoing surgery do not routinely need to be admitted to the hospital before the day of surgery |
98% Agree (Consensus) |
Grade D | Patients undergoing surgery do not routinely need to be admitted to the hospital before the day of surgery, but should be prioritised as the first case of the day if they are on drugs that can cause hypoglycaemia | |
| 32 | Patients undergoing surgery should be prioritized as the first case of the day if they are on drugs that can cause hypoglycemia |
88% Agree (Consensus) |
Grade D | ||
| 33 | Gastric Balloons have no role for patients seeking surgery for T2DM (except when they are used as a bridge to facilitate surgery) |
86% Agree (Consensus) |
Grade B | Gastric Balloons have no role for patients seeking surgery for T2DM (except when they are used as a bridge to facilitate surgery). Additionally, there is insufficient evidence to suggest a role for Endoscopic Sleeve Gastroplasty for patients seeking surgery for T2DM | |
| 34 | Currently, there is insufficient evidence to suggest a role for Endoscopic Sleeve Gastroplasty for patients seeking surgery for T2DM |
83% Agree (Consensus) |
Grade B | ||
| 35 | Surgery for T2DM is a treatment option for adolescent patients who meet the criteria for surgery but should only be carried out in specialized centers with an appropriate multidisciplinary team, including pediatricians, bariatric physicians (or diabetologists), psychologists, and dietitians |
96% Agree (Consensus) |
Grade A | Surgery for T2DM is a treatment option for adolescent patients who meet the criteria for surgery but should only be carried out in specialized centers with an appropriate MDT including pediatricians, bariatric physicians (or diabetologists), psychologists, and dietitians | |
| 36 | Surgery for T2DM is cost-effective |
98% Agree (Consensus) |
Grade A | Surgery for T2DM seems to be cost-effective over 10-year and lifetime horizons | |
| 37 | Surgical procedures aimed at treating T2DM vary in their safety and efficacy |
96% Agree (Consensus) |
Grade A | Surgical procedures aimed at treating T2DM vary in their safety and efficacy. The more weight loss, the better the effect in terms of T2DM remission/improvement: BPD > OAGB > RYGB > LSG. Regarding safety, LSG is safer than RYGB > OAGB > BPD | |
| VIII | Communication with Primary Care and Extended Follow-up | ||||
| 38 | Patients and their General Practitioners should be given clear written as well as verbal information at the time of discharge regarding the nature of the surgical procedure, post-discharge medications/supplements, diabetes management post-surgery, and any changes in the management of diabetes and other associated health conditions |
100% Agree (Consensus) |
Grade D | Patients and their General Practitioners should be given clear written as well as verbal information at the time of discharge regarding the nature of the surgical procedure, post-discharge medications/supplements, and changes in the diabetes management post-surgery to decrease the incidence of preventable medication-induced complications | |
| 39 | Post-surgery for T2DM, patients should have their HbA1c levels checked every 3 months for the first year or until stable and annually after that |
90% Agree (Consensus) |
Grade D | Post-surgery for T2DM, patients should have their HbA1c levels checked every 3 months for the first year or until stable, and annually after that (this strategy may significantly help to monitor the changes in serum glucose after surgery accurately and modify the dosage of the antidiabetic agents) | |
| 40 | Patients should be followed up in a multidisciplinary environment for the first two years after surgery with close input from a diabetologist as needed |
94% Agree (Consensus) |
Grade A | Patients should be followed up in a MDT environment for the first two years after surgery with close input from a diabetologist as needed. Stable patients can be discharged to primary care after 2–3 years of surgery where they should have an annual follow-up including aspects of surgery and diabetes management | |
| 41 | Stable patients can be discharged to primary care after 2–3 years of surgery, where they should have an annual follow-up, including aspects of surgery and diabetes management. There should be clear pathways of care for patients to be referred back to the surgical teams for surgical, dietetic, or mental health issues that may arise later in the course of a patient's life. Such follow-up should be properly funded |
76% Agree (No-Consensus) |
94% Agree (Consensus) |
Grade A | |
| IX | Remission and Recurrence | ||||
| 42 | Remission is defined as an HbA1c of < 48 mmol/mol (< 6.5%) for at least three months in the absence of glucose-lowering pharmacotherapy. When HbA1c is determined to be an unreliable marker of chronic glycaemic control (for example, because of anemia related to surgery), Fasting Plasma Glucose (FPG) < 7.0 mmol/l (< 126 mg/dl) or Estimated Glycosylated Haemoglobin (eA1c) < 6.5% calculated from continuous glucose monitoring values can be used as alternative criteria |
88% Agree (Consensus) |
Grade A |
Remission is defined as an HbA1c of < 48 mmol/mol (< 6.5%) for at least 3 months in the absence of glucose-lowering pharmacotherapy. Furthermore FPG < 7.0 mmol/l (< 126 mg/dl) or eA1c < 6.5% calculated from continuous glucose monitoring values can be used as alternative criteria Patients in remission should continue to receive the usual diabetes care for screening and management of diabetes complications (retinopathy, nephropathy, neuropathy, and macrovascular disease) and early detection of recurrence |
|
| 43 | Patients in remission should continue to receive the usual diabetes care for screening and management of diabetes complications (retinopathy, nephropathy, neuropathy, and macrovascular disease) and early detection of recurrence |
92% Agree (Consensus) |
Grade A | ||
| 44 | The risk of recurrence is multifactorial and depends on variables such as preoperative duration of diabetes, type of surgery, weight regain, and time after surgery |
98% Agree (Consensus) |
Grade A | Restrictive procedures, longer duration of T2DM, higher preoperative HbA1 C level, less postoperative weight loss, female sex, and insulin treatment prior to surgery are risk factors for T2D relapse after initial remission | |
Fig. 1.
Study flowchart
The statements and recommendations covered nine different aspects (Table 2), namely I-Nature, Terminology and Mechanisms of Action, II-Predictors of Remission, III-Preoperative Diabetic Control, IV-Perioperative Care, Monitoring and Special Considerations, V-Care Pathways and Logistics of Service, VI- Accurate Classification of Diabetes Mellitus, VII-Surgical Considerations, VIII-Communication with Primary Care and Extended Follow-up, and IX-Remission and Recurrence.
Ninety-five per cent agreed that surgery has a strong evidence-based role in treating some patients with T2DM, and 94% agreed that metabolic surgery for T2DM works through both weight-loss-dependent and weight-loss-independent mechanisms. The expert panel recommended using the term"Metabolic Surgery for T2DM" as a common descriptor for surgery aimed at treating people with T2DM. Ninety-six per cent of the panel agreed that the beneficial impact of metabolic surgery for T2DM depends on many factors. Additionally, 86% agreed that metabolic surgery for T2DM only has a role in individuals with obesity, and the currently available scores for predicting remission of T2DM after surgery are not robust enough for routine clinical use. However, 92% agreed on the need to optimize glycemic control to an HbA1c level of ≤ 69 mmol/mol (≈ 8.5%), and 83% agreed that HbA1c > 69 mmol/mol should not be considered an absolute contraindication for patients undergoing surgery for T2DM.
The experts agreed on six statements related to perioperative care and monitoring and recommended that patients undergoing metabolic surgery for T2DM should be reviewed by a diabetologist for perioperative management of diabetes. Additionally, they recommended frequent glucose and blood pressure monitoring in the early postoperative phase.
There was complete consensus (100%) on the barriers that prevent patients from accessing surgery for diabetes, including insufficient awareness of the benefits and risks of surgery amongst many healthcare professionals, insufficient awareness amongst patients on the safety and efficacy of surgery, lack of funding or insurance coverage for surgery, mandatory weight loss targets, lack of capacity for delivering surgery to eligible patients, weight bias towards access to healthcare, stigma, and shame of obesity.
A complete consensus was achieved that the referral criteria for surgery should not be based on BMI alone, and the patient's other obesity-associated medical and mental health conditions and quality of life should be considered.
Accurate diagnosis of the type of diabetes was an important domain of the consensus, and the panel recommended considering alternative diagnoses for T2DM, such as T1DM or Maturity Onset Diabetes of Young (MODY), before referring patients for surgery. Additionally, surgery can be an option in selected patients with T1DM or Latent Autoimmune Diabetes in Adults (LADA) who also meet BMI criteria for MBS.
Regarding the surgical considerations, 86% agreed that gastric balloons have no role for patients seeking metabolic surgery for T2DM apart from being used as a bridge to facilitate surgery, and 83% agreed that there is currently insufficient evidence to suggest a role for endoscopic sleeve gastroplasty in these patients.
Ninety-four per cent agreed that the patients should be followed up in a multidisciplinary environment for the first two years after surgery. The panel recommends that remission is defined as an HbA1c of < 48 mmol/mol (< 6.5%) for at least three months in the absence of glucose-lowering pharmacotherapy.
Discussion
Nature, terminology, and mechanisms of action
Surgery performed primarily for T2DM may differ from bariatric surgery; it has different goals from bariatric surgery. There is now a large body of literature, including randomized studies [10, 11] focusing on surgery for patients with T2DM. It may, therefore, be necessary to differentiate between bariatric surgery and metabolic/diabetes surgery [12]. Both terms"metabolic surgery"and"diabetes surgery"have been used in previous consensus-building exercises on this topic [6, 7]. However, the use of the term “Type 2 Diabetes Surgery” did not reach a consensus in this exercise.
Level 1 evidence supports the role of surgery in treating patients with T2DM [2, 13] and international diabetes and obesity organizations have recommended it [7]. Further, high-quality evidence confirms that surgery is an effective treatment of T2DM in patients with obesity [14]. MBS is more likely to achieve resolution of T2DM compared with medical management [15]. The latest version of the American Diabetes Association Standards of Care in Diabetes (2023) incorporates MBS in the management algorithm of T2DM with a lower BMI cut point for patients of Asian ethnic origin [16]. At the same time, evidence suggests that a combination of treatment strategies (i.e., surgery with medication) often yields better outcomes than any single treatment [17, 18].
Weight loss and glycemic effects of MBS for T2DM were traditionally thought to be mediated by “restriction” and/or “malabsorption” of ingested nutrients. Recent studies have challenged this paradigm, and many have suggested that malabsorption by itself is not a sustained mechanism of action, even after RYGB [19]. An increasing body of evidence indicates that neuro-hormonal and other physiological factors affecting hunger, appetite, satiety, and energy expenditure are responsible for the beneficial effects of MBS for T2DM [20, 21].
Predictors of remission
Surgery has variable effectiveness among patients. Several factors have been identified that can help identify individuals who are likely to respond better to surgical treatment, and clinical scores have been developed [22, 23] to predict that response. Younger age, higher BMI, better pancreatic islet beta-cell reserve, shorter duration of diabetes, and better glycemic control before surgery seem to be associated with better clinical outcomes [24].
A nationwide study of the Scandinavian obesity surgery registry [25] included 8,057 patients who had RYGB and showed that increasing age, higher HbA1c levels, and longer diabetes duration decreased the chance of reaching T2DM remission, while there was a positive linear association with postoperative weight loss. However, even those patients who do not achieve T2DM remission after surgery can derive significant benefits through better glycemic control, weight loss, and improvement in other associated health conditions [26].
A nationwide register-based cohort study [26] from Sweden, which included 8,546 patients, showed that the chance of no longer requiring T2DM medication was lower in patients with a longer preoperative diabetes duration at both two and five years after surgery. The chance of achieving sustained T2DM remission correlated negatively with diabetes duration, insulin treatment, age, and HbA1c levels at baseline. T2DM remission was greater among males and those with higher BMI at baseline. Although studies have also shown benefits in those who are living with overweight [27] rather than obesity, there is currently a lack of randomized evidence to inform practice in this area. However, recent evidence with a long-term follow-up period suggests that the accuracy of the available scores in predicting T2D remission in the long term is still suboptimal [28].
Preoperative diabetic control
Hyperglycaemia leads to immune dysfunction and cellular damage, which could potentially lead to perioperative complications [29]. There is no level 1 evidence available on the effect of preoperative HbA1 C levels on the outcomes of MBS. However, attention to glycemic control minimizes the potential risk for adverse outcomes similar to other surgical procedures.
The evidence from large-scale cohort studies showed conflicting results on the association between HBA1 C levels and the complications or mortality rates after MBS or other surgical interventions [30, 31]. This was probably the basis for the American Society for Metabolic and Bariatric Surgery (ASMBS) conclusion that the current evidence does not support delaying or withholding bariatric surgery to achieve specific HbA1 C targets [32].
Hart A et al. [33] studied 31,060 patients who had SG and 13,754 patients who had RYGB. They found that patients with elevated HbA1c levels had a significantly higher rate of composite overall morbidity and mortality rates but did not have a significant difference in 30-day mortality. Unplanned intubation, superficial surgical site infection, postoperative pneumonia, and postoperative sepsis were all higher in patients with high HbA1c.
More recently, Pina L et al. [34] analyzed the Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program (MBSAQIP) database, which included 42,181 patients who had RYGB and SG. They found that there was no significant association between HbA1c levels and 30-day major complications (Clavien-Dindo III/IV). Both the American Diabetes Association (ADA) [16] and the European Association for the Study of Obesity (EASO) [35] recommend that MBS should be performed by multidisciplinary teams with appropriate experience in managing obesity and diabetes to formulate and oversee the diabetes management plans.
Perioperative care, monitoring, and special considerations
Preoperative energy restriction
Preoperative energy restriction regimens have been adopted by Upper Gastrointestinal (UGI) surgeons to reduce liver volume before major UGI surgical procedures [36, 37]. Different regimens have been described, which could be collectively categorized as a very low-energy diet (VLED) with 450–800 kcal per day or a low-energy diet (LED) with 800–1500 kcal per day.
A systematic review [38] including 849 patients from nine studies (three randomized clinical trials and six observational studies) found that while a VLED resulted in liver size reduction (5%—20%), it did not significantly reduce perioperative complications. However, one study (n = 273) reported a protective effect 30 days after surgery. A recent study [39] included 120 candidates for MBS who received a VLED for eight days only and observed a 5% reduction in body weight primarily due to loss of body fat. A recent systematic review by Romeijn MM et al. [40] included eight studies with 251 patients and showed that LED reduced liver volume (12–27%) and achieved a 4–17% weight loss. They concluded that LED had acceptable patient adherence and could be used instead of a VLED for 2 to 4 weeks preoperatively. Besides the technical feasibility of a"liver-shrinking"diet, these regimens have proven to significantly improve liver histological features, including steatosis, inflammation, and hepatocellular ballooning [41]. Dietary modification and nutritional support should be achieved under supervision by experienced teams specialized in managing patients with obesity and T2DM [16].
Blood pressure changes
Recent studies [42, 43] have shown a significant decrease in mean arterial blood pressure (MAP) within ten days to two weeks after MBS in both hypertensive and normotensive patients, with a substantial reduction of blood pressure 6—12 months after MBS [43, 44]. These changes are more pronounced after RYGB [45]. Orthostatic intolerance post-MBS has been reported and can present as light-headedness, dizziness, syncope, and palpitations [46]. These findings highlight the need for early monitoring and management of blood pressure after MBS.
Post-MBS hypoglycaemia
Post-MBS hypoglycaemia has been reported and could be attributed to faster gastric emptying after MBS and rapid circulating glucose surge with consequent excessive postprandial insulin secretion leading to a sharp drop in blood glucose levels. It has been reported in up to 10% of patients after gastric bypass procedures. Patients may present with sweating, tachycardia, hunger, tremor, impaired cognition, loss of consciousness, and seizures. This should be differentiated from dumping syndrome due to intra-intestinal osmotic effects, which usually occur soon after surgery and improve over time. Post-MBS hypoglycaemia usually occurs > 1 year after surgery. The management options include dietary modification, exclusion of surgical complications, and pharmacological management [47, 48].
A recent meta-analysis [49] of 8 studies including 280 patients found that the total weighted mean prevalence (WMP) of post-MBS hypoglycemia (PMH) was 54.3%, and the WMP of nocturnal PMH was 16.4%, with a comparable rate of PMH after RYGB and SG. However, RYGB was associated with a higher glycemic variability than SG. The time elapsed from surgical intervention was positively associated with a higher rate of both total PMH and nocturnal PMH. Another study from the USA [50] included 6,024 patients who had MBS and showed that 118 patients (2.0%) had a postoperative glucose level ≤ 70 mg/dL, and 83 (1.4%) developed symptomatic hypoglycemia.
Early monitoring of glucose levels and adjustment of diabetes medications have been emphasized in the latest guidelines for perioperative care after MBS by the Enhanced Recovery After Surgery (ERAS) Society [51]. More detailed recommendations have been proposed by the Task Force of the European Association for the Study of Obesity for the post-bariatric surgery medical management, including target glucose levels and frequency of blood glucose testing [35]. Also, the latest ADA guidelines recommend a long-term medical review and monitoring of metabolic status. Continuous glucose monitoring has been recommended in cases with suspected post-MBS hypoglycaemia [16].
Sodium-glucose co-transporter-2 inhibitors and MBS
SGLT-2i are increasingly prescribed due to their cardio-renal protective benefits in patients with and without T2DM [52]. However, euglycemic and hyperglycemic diabetic ketoacidosis (DKA) have been reported in patients using these medications. The Food and Drug Administration (FDA) issued a warning on this in 2015, and the British Obesity and Metabolic Surgery Society (BOMSS) patient safety committee released an alert in 2022 highlighting these risks and outlining specific recommendations before and after MBS [53, 54]. The National Institute for Health and Care Excellence (NICE) recommends assessing the risk of DKA before commencing SGLT2i [55].
Care pathways and logistics of service
Utilization of the service and barriers
Despite the safety profile and clear benefits of MBS in treating obesity and related complications and its superiority over the alternatives, MBS has been significantly underutilized in many parts of the world [56, 57]. This has been attributed to a variety of reasons, including patient and public misperceptions [58, 59], ineffective referral pathways [60, 61], logistics, and medical insurance-related issues [62, 63]. Of particular concern is the disparity in bariatric service access related to ethnicity [64, 65].
Dietary modification
Dietary and physical activity modifications are associated with clinically significant weight loss and improvement in weight-related comorbidities, including glycaemia and hypertension [66–68]. LED has demonstrated efficacy over the other alternatives in terms of T2DM remission without significant adverse consequences [69, 70]. The ADA guidelines [16] recommend a short-term LED (800–1,000 kcal/day) for selected individuals by trained staff. Additionally, they advised nutritional modification and physical activity to achieve and maintain at least 5% weight loss for people with T2DM with overweight or obesity. These options should be explored and discussed with patients with T2DM as alternatives to MBS as part of informed consent for the agreed treatment plan.
Referral criteria
Despite being the most commonly used anthropometric measure for obesity, BMI has limitations in assessing body composition and distinguishing between muscle and fat distribution. Other more accurate parameters to diagnose obesity and predict associated risks and comorbidities have been explored [71, 72]. Despite the paucity of evidence around referral criteria apart from BMI, there is a growing body of evidence on the efficacy and safety of MBS in treating T2DM in patients with BMI ≤ 35 kgs/m2 [73, 74]. The ADA [16] recommendations addressed this clearly and identified lower BMI cut points for consideration of MBS in patients with T2DM of Asian origin. The latest guidelines of ASMBS and the International Federation for the Surgery of Obesity and Metabolic Disorders (IFSO) recommended surgery for patients with T2DM and BMI ≥ 30 kg/m2 [74].
Preoperative weight loss
Preoperative weight loss of 5% – 10% of total body weight was recommended [51, 75] and even considered mandatory for obtaining insurance or state coverage in some healthcare settings. However, currently available evidence would challenge such practices. While some studies have shown a positive association between preoperative and postoperative weight loss, reduction of complications, and mortality, others have not found these associations [76]. In addition, the majority of the candidates for MBS have a long history of weight loss attempts. Therefore, forcing them to repeat these treatments, which have previously not resulted in achieving durable outcomes, may be considered an unnecessary barrier to MBS and, therefore, morally indefensible as such practices may exclude those most in need.
Comprehensive assessment and MDT approach
As part of the informed consent process, the different perioperative considerations should be discussed in detail with the patients. Moreover, providing access to support groups can help the patients through their treatment journey [75]. MBS results in fundamental anatomical changes to the patient’s alimentary tract. In addition to reducing intake, they can also affect the absorption of macro/micronutrients. Prevention, early detection, and correction of nutritional and micronutrient deficiencies is imperative for this patient group at high risk of nutritional deficiencies. Different national societies have released dedicated practice guidelines on perioperative biochemical monitoring and micronutrient replacement, such as BOMSS perioperative and postoperative biochemical monitoring and micronutrient replacement guidelines [77], ASMBS Integrated Health Nutritional Guidelines [78] and the more recent French expert consensus guidance on post-MBS nutritional support [79].
Mental health and MBS
The MDT approach and psychological support are paramount and have been emphasized in the NICE [80, 81] and BOMSS guidelines [82] as an integral part of the surgery provided by the UK National Health Service (NHS) and private and public insurers in the United States.
Although MBS in patients with stable psychiatric illnesses is safe [83], mental health disorders may lead to lower adherence to follow-up reviews and postoperative care plans [84], worsening of psychiatric disorders after surgery [85], and the emergence of new mental health and behavioural disorders after MBS in some patients, including maladaptive eating, substance use disorders, self-harm behaviour, and suicidal ideation [86].
A recent nationwide cohort study [87] identified 9,480 patients with psychiatric illnesses within 22,539 who had gastric bypass. The disease spectrum includes bipolar disorder, schizophrenia, depression, neurotic disorders, attention deficit hyperactivity disorder (ADHD), substance use disorder, eating disorder, personality disorder, and self-harm. The authors found that the presence of psychiatric illness was associated with delayed discharge and higher reoperation rates. Another study has linked psychiatric disorders to early readmission and increased length of hospital stay [88]. Moreover, a scoping review of fifty-eight studies [89] reported that substance use history does not influence weight loss after MBS. However, it could be associated with increased substance use after surgery.
All the relevant guidelines emphasize the preoperative psychological assessment and postoperative psychological support as part of the MDT approach for patients with mental health conditions rather than excluding them from surgery on these grounds [16, 80, 81, 90].
Accurate classification of diabetes mellitus
MBS has a demonstrated positive impact on patients with Type 1 Diabetes Mellitus (T1DM) by reducing insulin requirements [91] and reducing the risks of cardiovascular disease, stroke, and mortality. However, it is associated with some risks, including that of postoperative hypoglycaemia and diabetic ketoacidosis (DKA) [92, 93].
Despite the proven benefit of MBS in reducing insulin requirements and resolving comorbidities, its benefit in achieving glycemic control is debatable [91, 93]. The most commonly performed procedures for T1DM are RYGB and SG [93], with no significant differences in the weight loss outcomes or reduction of insulin requirements between both procedures [94]. Studies comparing the outcome of MBS in patients with T2DM and T1DM showed significant differences in the outcomes in favour of T2DM [95]. Accordingly, a clear distinction should be sought for better management of patient expectations and consenting.
The latest guidelines of the American Association of Clinical Endocrinology define diagnostic criteria for different types of diabetes. T1DM is characterized by insulin deficiency, hyperglycaemia, and positive autoantibody tests to glutamic acid decarboxylase (GAD65), pancreatic islet b cells (tyrosine phosphatase IA-2), and IA-2b zinc transporter (ZnT8), and/or insulin [96]. An accurate diagnosis in liaison with the endocrinologist or diabetes specialist team is an essential preliminary step before considering the surgical option in this patient group.
Latent Autoimmune Diabetes in Adults (LADA) and Maturity Onset Diabetes of Young (MODY) are unique variants of glycemic metabolic derangements with different pathophysiological characteristics and genetic and molecular basis [97–99]. Few studies have been conducted on the effect of MBS in patients with these forms of DM. However, it seems that the response of these disease types and T1DM to MBS is less favourable than that of T2DM [100, 101].
Surgical considerations
Day case or regular preoperative admission?
There has been a significant shift in the perioperative care setting across different surgical specialities, with an inclination towards shortening preoperative and postoperative hospitalization. This trend has been driven by the benefits of reducing bed occupancy, avoiding perioperative nosocomial infections, and achieving cost-effective medical care. This idea formed the basis for the concept of Perioperative Anaesthesia and Surgical Home (PASH) [102]. The concept of ambulatory MBS with or without telemonitoring has been investigated and shown to be safe in selected patients [103, 104] with comparable results in terms of patient satisfaction and greater cost-effectiveness [105, 106]. The latest Guidelines for Perioperative Care in Bariatric Surgery of the (ERAS) Society [51] detailed the evidence-based recommendations for preoperative care, which can be implemented on the day of surgery without admitting the patient. However, inadequate diabetes management in the perioperative period after or before MBS has been reported [107]. Great caution should be exercised to prevent fluctuation of blood glucose levels in the perioperative period, including listing patients with diabetes at the top of the list to limit the time of preoperative fasting.
Metabolic endoscopy
Recent technological advances in gastrointestinal endoscopy have led to the evolution of bariatric and metabolic endoscopy (BME) as a less invasive option compared to MBS. Gastric balloons (GB) and endoscopic sleeve gastroplasty (ESG) are the most common applications of BME [108].
Evidence from different studies on various types of gastric balloons confirms the achievement of acceptable short-term weight loss results [109, 110] and the feasibility of utilizing gastric balloons as a bridge to definitive MBS [111]. Despite the promising initial results, the long-term efficacy of gastric balloons has yet to be confirmed [112, 113], and there is a need for more well-designed studies to evaluate long-term outcomes to support it. A recent meta-analysis [114] of 17 studies that included 1,198 patients showed a significant reduction in HbA1c levels six months after inserting an intragastric balloon (IGB). It was concluded that IGB is associated with improvements in insulin resistance and other obesity-related metabolic derangements.
ESG has been introduced as a less invasive variant of sleeve gastrectomy. Current evidence supports its safety, efficacy in achieving weight loss, and some improvement in metabolic parameters [115]. Additionally, it has been proposed as a revisional procedure after sleeve gastrectomy [116]. A recent prospective cohort study of 612 patients who had ESG showed the resolution of T2DM in 51.2% of the cases [117]. A non-inferiority propensity score-matched comparative study [118] showed a resolution of diabetes in 64% of the patients after EGS compared to 82% after LSG. Moreover, a recent systematic review and meta-analysis of 35 studies, including 7,525 patients, showed diabetes resolution in more than 55% of patients who had EGS [119]. Based on the current evidence, NICE recommended EGS as a treatment option in adults with obesity [120].
Surgery for adolescents with T2DM
T2DM in the adolescent population with obesity poses a significant risk for the early development of diabetes complications, poor quality of life, and mortality. Adolescents with T2DM will have a faster decline in pancreatic islet beta-cell function and earlier deterioration of renal function, in addition to worsening retinopathy, hypertension, and neuropsychiatric complications [121, 122]. The landmark study, Teen Longitudinal Assessment of Bariatric Surgery (Teen-LABS) [123], showed that adolescents with T2DM had better glycemic control after MBS compared to adults without the use of diabetes medications. The same observation has been reproduced in subsequent studies [124, 125], which led to increased utilization and an overall uptrend of MBS in the adolescent age group [126]. Both the European Society of Endocrinology and the Pediatric Endocrine Society guidelines [127] and ASMBS pediatric metabolic and bariatric surgery guidelines [128] emphasize the role of MBS in the adolescent population based on clear criteria for referral and the MDT approach.
Costs and burden
Given the high cost of the intervention and management of subsequent potential complications of MBS, cost-effectiveness is a key issue. Different cost-effectiveness analyses of MBS for T2DM have been conducted in different parts of the world with varying healthcare settings; all have reported that MBS is cost-saving in the long run due to reduction of the costs of diabetes medication as well as postoperative medical care and management of T2DM and its related complications [129, 130]. A recent systematic review of economic evaluations [131] included 30 studies and found that MBS for patients with T2DM and obesity is cost-effective in the long term.
Procedure choice
The resolution of T2DM after MBS is multifactorial and achieved through weight loss-dependent and weight loss-independent mechanisms [132]. The most commonly performed procedures include SG, One Anastomosis Gastric Bypass (OAGB), RYGB, and Biliopancreatic Diversion (BPD). A recent systematic review included five RCTs [133] that compared OAGB and SG and found that T2DM remission with OAGB was more efficient at one and five years. Another systematic review and meta-analysis of RCTs compared RYGB to SG [134] and found that the remission rate of T2DM at one year was higher with RYGB than with SG. However, at 2–5 years, there was no significant difference. A network meta-analysis of 20 RCTs [135] included 1,803 patients and showed greater T2DM remission with either RYGB or OAGB compared to SG. However, perioperative complications were higher with RYGB compared to either SG or OAGB.
Another RCT with a 10-year follow-up [136] showed that after SG and RYGB, there was no significant difference in T2DM remission. A recent systematic review [137] compared the safety and efficacy of RYGB, OAGB, and Single Anastomosis Duodeno-Ileal bypass with Sleeve gastrectomy (SADI-S). The review included 18 studies and showed that SADI-S was associated with better resolution of T2DM. However, the early complications and mortality were higher with SADI-S, and late complications were more frequent with RYGB. OAGB was associated with fewer complications.
Communication with primary care and extended follow-up
Postoperative care after MBS requires close collaboration of the specialist MDT in tertiary healthcare facilities with primary care to guarantee continuity of care and safety. Also, patient education and involvement are of paramount importance. This shared-care model of management has been emphasized in the NICE guidance [80, 81]. Documentation of the discharge process and follow-up care arrangements, including detailed written discharge summaries and liaising with primary care, are integral roles of the surgical teams [138].
Postoperative monitoring of the nutritional status and glycemic control is an important phase of the treatment journey [77, 78, 81, 139]. The ADA guidance [139] recommends assessing glycemic status at least quarterly and as needed in patients whose therapy has been recently changed. As MBS induces a dramatic effect on glucose metabolism, there should be regular monitoring, and HBA1c levels should be checked regularly till stabilization of glucose levels and cessation or adjustment of diabetes medications. The NICE guidelines recommend two years of follow-up under bariatric services and MDT, followed by at least annual monitoring of nutritional status in the long term in primary care after discharge from the bariatric service [81].
Remission and recurrence
Remission
Monitoring glycemic control to determine remission can be achieved by measuring HbA1c levels, fasting plasma glucose (FPG), 2-h plasma glucose after an oral glucose challenge, or mean daily glucose from continuous glucose monitoring (CGM). HbA1c is the most practical and commonly used method. However, conditions exist that may interfere with the reliability of HbA1c and include haemoglobin variants, differing rates of glycation, or alterations in erythrocyte survival. Iron deficiency, which is common after MBS, could potentially complicate the picture further by affecting HBA1c levels. Oral glucose tolerance tests may not be feasible or accurate in patients post-MBS [140, 141].
The most used criterion for remission is HbA1c < 6.5% (48 mmol/mol), as used in the DIADEM-I trial [140] and recommended by an expert consensus group [4]; this is consistent with the cut-off value for DM diagnosis [141]. If HbA1c is deemed inaccurate for remission determination, FPG lower than 126 mg/dL (7.0 mmol/L) could be used as an alternative criterion. Testing for remission should be done at least three months after the procedure and three months after cessation of glucose-lowering medications [140].
Diabetes-related complications
MBS reduces, but does not eliminate, the risk of diabetes-related complications, including retinopathy, nephropathy, or neuropathy [142, 143]. A recent meta-analysis [144] included 32,756 participants in 12 studies and showed that MBS reduced the incidence of microvascular complications, diabetic nephropathy, and neuropathy compared with nonsurgical treatments in obesity with T2DM. These findings support the regular follow-up of diabetes-related complications even after T2DM remission.
Recurrence
Recurrence of T2DM after initial remission secondary to MBS has been reported [145]. The PCORNet [14] study showed that T2DM remission rates of patients who had RYGB and SG were 59.2% and 55.9%, respectively, at one year and 86.1% and 83.5% at five years after surgery. Among 6,141 patients who experienced T2DM remission, the estimated relapse rates for those who had RYGB and SG were 8.4% and 11.0% at one year and 33.1% and 41.6% at five years postoperatively. A recent meta-analysis reproduced the same with more favourable results with RYGB compared to SG in terms of relapse of T2DM after initial remission [146]. A registry-based Swedish study [147] included 2,090 patients in complete remission two years after surgery who were followed for a median of 5.9 years. They reported a cumulative T2DM relapse rate of 20.1%. Duration of diabetes, preoperative HbA1c level and preoperative insulin treatment were associated with higher rates of relapse. Other predictors of relapse have been reported and included preoperative insulin use, a lower percentage of total body weight loss at one year, and a greater percentage of total body weight regained after one year [148]. Clear information on T2DM remission and recurrence would formulate the patient's expectations and should be an integral part of the informed consenting process.
Recommendations
According to the results of this expert consensus, the recommendations around metabolic surgery for T2DM are summarised in Table 3.
Table 3.
Recommendations from the Delphi consensus on surgery for type 2 diabetes mellitus
| Recommendations | Grading level |
|---|---|
| Surgery for Type 2 Diabetes Mellitus (T2DM) is similar to bariatric surgery but allows patients with diabetes to be considered for surgery at a lower BMI | Grade A |
| The term “Metabolic Surgery for T2DM” should be used as a common descriptor for surgery aimed at treating people with T2DM | Grade D |
| Surgery has a strong evidence-based role in the treatment of patients with T2DM that works through both weight-loss dependent and weight-loss independent mechanisms and changes multiple signals (metabolic, biliary, neural, hormonal) | Grades A and B |
| The beneficial impact of surgery for T2DM depends on factors such as younger age, higher baseline BMI, shorter diabetes duration, and better preoperative glycaemic control represented by higher c-peptide level, lower HbA1c, and less insulin usage. Surgery for T2DM ONLY has a role in individuals with a Body Mass Index of ≥ 30 kg/m2, or 27.5 kg/m2 in some Asian populations. Despite some existing scores such as ABCD score and DiaRem, It seems that there is an essential need for more accurate predicting scores | Grades A and B |
| HbA1c levels matter, but all efforts should be made to optimize glycaemic control to an HbA1c level of ≤ 69 mmol/mol (≈ 8.5%), but surgery should not be delayed unduly to achieve this | Grades A and B |
| Patients undergoing surgery for T2DM should be reviewed by a diabetologist for perioperative and postoperative glycaemic management of diabetes (including preoperative liver-reducing diet and adjustment of dosage of glucose and blood pressure-lowering agents after surgery) | Grades A and B |
| Frequent capillary/interstitial glucose and blood pressure monitoring (in those on antihypertensives) are advised in the early postoperative phase to allow for titration of doses of medications | Grade A |
| Postprandial hypoglycemia (also known as reactive hypoglycemia) is more common after diversionary procedures, no more common in patients with T2DM, and can be serious. These patients should be referred to diabetologists for further investigation and management | Grade A |
| Based on evidence, we suggest stopping SGLT2 inhibitors 48 h prior to surgery to reduce the risk of ketoacidosis | Grade A |
| There is a need for targeted measures to address barriers that prevent patients from accessing surgery for diabetes, including (Insufficient awareness of the benefits and risks of surgery amongst many healthcare professionals and patients, Lack of funding or insurance coverage for surgery, and Stigma toward people with obesity, Shame amongst patients because of obesity) | Grade A |
| While surgery is the most effective and durable, patients have a right to be informed about treatment options such as dietary and lifestyle interventions and pharmacological therapy with a full discussion of benefits, risks, and duration of treatment in comparison with surgery by a healthcare professional to make their own decisions about their preferred treatment | Grade A |
| Referral criteria for surgery should not be based on BMI alone and should also consider the patient's other obesity-associated medical and mental health conditions and quality of life | Grade C |
| Medical weight management is not necessary for surgery for T2DM, but patients should have seriously attempted to lose weight in the past with the help of appropriate lifestyle/dietary education and available pharmacological interventions | Grade B |
| Patients should receive comprehensive dietetic assessment and education, information about the risks and benefits of surgery, and access to a peer support group and MDT, including dietitians, psychologists, and diabetologists both before and after surgery. Patients not eligible for surgery on the grounds of mental health contraindications | A and B |
| Consider alternative diagnoses for T2DM, such as T1DM or Maturity Onset Diabetes of Young (MODY), before referring patients for surgery | Grade B |
| Surgery can be an option in selected patients with T1DM or Latent Autoimmune Diabetes in Adults (LADA) who also meet BMI criteria for bariatric surgery and may thus benefit from better glycaemic control and weight loss, but the decision for surgery should not be taken without consulting a diabetologist in these patients | Grade A |
| Antibody/C-peptide level can help to increase the accuracy of preoperative diabetes classification and rule out T1DM, but it is unnecessary before surgery for diabetes | Grade C |
| Patients undergoing surgery do not routinely need to be admitted to the hospital before the day of surgery but should be prioritized as the first case of the day if they are on drugs that can cause hypoglycemia | Grade D |
| Gastric Balloons have no role for patients seeking surgery for T2DM (except when they are used as a bridge to facilitate surgery). Additionally, there is insufficient evidence to suggest a role for Endoscopic Sleeve Gastroplasty for patients seeking surgery for T2DM | Grade B |
| Surgery for T2DM is a treatment option for adolescent patients who meet the criteria for surgery but should only be carried out in specialized centers with an appropriate MDT, including pediatricians, bariatric physicians (or diabetologists), psychologists, and dietitians | Grade A |
| Surgery for T2DM seems to be cost-effective over 10-year and lifetime horizons | Grade A |
| Surgical Procedures aimed at treating T2DM vary in their safety and efficacy. The more weight loss, the better the effect in terms of T2DM remission/improvement: BPD > OAGB > RYGB > LSG. Regarding safety, LSG is safer than RYGB > OAGB > BPD | Grade A |
| Patients and their General Practitioners should be given clear written as well as verbal information at the time of discharge regarding the nature of the surgical procedure, post-discharge medications/supplements, and changes in the diabetes management post-surgery to decrease the incidence of preventable medication-induced complications | Grade D |
| Post-surgery for T2DM, patients should have their HbA1c levels checked three monthly for the first year or until stable, and annually after that (this strategy may significantly help to monitor the changes in serum glucose after surgery accurately and modify the dosage of the antidiabetic agents) | Grade D |
| Patients should be followed up in an MDT environment for the first two years after surgery with close input from a diabetologist as needed. Stable patients can be discharged to primary care after 2–3 years of surgery, where they should have an annual follow-up, including aspects of surgery and diabetes management | Grade A |
|
Remission is defined as an HbA1c of < 48 mmol/mol (< 6.5%) for at least three months in the absence of glucose-lowering pharmacotherapy. Furthermore, FPG < 7.0 mmol/l (< 126 mg/dl) or eA1c < 6.5% calculated from continuous glucose monitoring values can be used as alternative criteria Patients in remission should continue to receive the usual diabetes care for screening and management of diabetes complications (retinopathy, nephropathy, neuropathy, and macrovascular disease) and early detection of recurrence |
Grade A |
| Restrictive procedures, longer duration of T2DM, higher preoperative HbA1 C level, less postoperative weight loss, female sex, and insulin treatment prior to surgery are risk factors for T2D relapse after initial remission | Grade A |
Strengths and limitations
This is the first consensus-building exercise on metabolic surgery for T2DM involving experts from 23 countries with significant experience and expertise in the field. The experts followed a robust Delphi methodology.
Several limitations of this work need to be highlighted. The selection of expert clinicians can be considered arbitrary, but due care was taken to represent those with the appropriate international expertise, including contributors to the major national and international obesity and diabetes guidelines. The threshold of 80% for consensus can also be considered arbitrary but has been used in several prior consensus-building exercises [8].
Some of the statements achieved consensus with a low margin of agreement. The lack of high-quality evidence on these recommendations limited the expansion of the relevant discussion section. These areas need more research to enrich the decision-making process and fill the gaps in knowledge.
Moreover, we appreciate the variability in the scope of service provision and the availability of resources. Given the variable resources and expertise in different parts of the world, one of the challenges of these recommendations is their generalisability.
Finally, although very valuable for day-to-day clinical practice, a consensus statement amongst experts is still opinion, and these statements need further confirmation by adequately designed studies.
Conclusion
This Delphi expert consensus statement provides guidance to clinicians on various aspects of metabolic surgery for T2DM and also grades the quality of the contemporary evidence for each of the proposed statements.
Supplementary Information
Acknowledgements
None.
Clinical trial number
Not applicable.
Informed consent
Informed consent does not apply.
Abbreviations
- MBS
Metabolic and Bariatric surgery
- T2DM
Type 2 Diabetes Mellitus
- MDT
Multidisciplinary Team
- BMI
Body Mass Index
- HbA1c
Glycated haemoglobin
- ASMBS
American Society for Metabolic and Bariatric Surgery
- SG
Sleeve Gastrectomy
- RYGB
Roux-en-Y Gastric Bypass
- MBSAQIP
Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program
- ADA
American Diabetes Association
- EASO
European Association for the Study of Obesity
- GI
Gastrointestinal
- UGI
Upper Gastrointestinal
- VLED
Very Low-Energy Diet
- LED
Low-Energy Diet
- MAP
Mean Arterial Blood Pressure
- SGLT-2
Sodium-Glucose Transport Protein 2
- GLP-1
Glucagon-Like Peptide-1
- WMP
Weighted Mean Prevalence
- PBH
Post-Bariatric Hypoglycaemia
- ERAS
Enhanced Recovery After Surgery
- DKA
Diabetic ketoacidosis
- FDA
Food and Drug Administration
- BOMSS
British Obesity and Metabolic Surgery Society
- NICE
The National Institute for Health and Care Excellence
- NHS
National Health Service
- ADHD
Attention Deficit Hyperactivity Disorder
- T1DM
Type 1 Diabetes Mellitus
- LADA
Latent Autoimmune Diabetes in Adults
- MODY
Maturity Onset Diabetes of Young
- GAD65
Glutamic Acid Decarboxylase
- ZnT8
IA-2b Zinc Transporter
- PASH
Perioperative Anaesthesia and Surgical Home
- GB
Gastric Balloons
- ESG
Endoscopic Sleeve Gastroplasty
- IGB
Intragastric Balloon
- Teen-LABS
Teen Longitudinal Assessment of Bariatric Surgery
- OAGB
One Anastomosis Gastric Bypass
- BPD
Biliopancreatic Diversion
- SADI-S
Single Anastomosis Duodeno-Ileal Bypass with Sleeve Gastrectomy
- DIADEM-I
Effect of Intensive Lifestyle Intervention on Body Weight and Glycaemia in Early Type 2 Diabetes
- FPG
Fasting Plasma Glucose
- eA1c
Estimated Glycosylated Hemoglobin
- CGM
Continuous Glucose Monitoring
- PCORnet
Bariatric Study funded through a Patient-Centred Outcomes Research Institute (PCORI)
Authors’ contributions
MK and IO contributed equally to this work and shared the first authorship. MK Conception of the Idea, Moderation of the Consensus building, Investigation, Formal analysis, Writing – original draft. IO Discussion of the Scientific Evidence, Investigation, Formal analysis, Writing – original draft, Writing – review & editing. KM Moderation of the Consensus building, Statement Formulation, Investigation, Writing – review & editing, Supervision. All authors reviewed the manuscript.
Funding
None.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
This article does not contain any studies with human participants or animals performed by any of the authors.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Mohammad Kermansaravi and Islam Omar contributed equally as co-first authors.
Contributor Information
Mohammad Kermansaravi, Email: mkermansaravi@yahoo.com.
Islam Omar, Email: islamfawzyomar@hotmail.com.
References
- 1.Liang Z, Wu Q, Chen B, Yu P, Zhao H, Ouyang X. Effect of laparoscopic Roux-en-Y gastric bypass surgery on type 2 diabetes mellitus with hypertension: a randomized controlled trial. Diabetes Res Clin Pract. 2013;101(1):50–6. [DOI] [PubMed] [Google Scholar]
- 2.Mirghani H, Altedlawi AI. Metabolic surgery versus usual care effects on diabetes remission: a systematic review and meta-analysis. Diabetol Metab Syndr. 2023;15(1):31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Lean MEJ, Leslie WS, Barnes AC, Brosnahan N, Thom G, McCombie L, et al. Durability of a primary care-led weight-management intervention for remission of type 2 diabetes: 2-year results of the DiRECT open-label, cluster-randomised trial. Lancet Diabetes Endocrinol. 2019;7(5):344–55. [DOI] [PubMed] [Google Scholar]
- 4.Riddle MC, Cefalu WT, Evans PH, Gerstein HC, Nauck MA, Oh WK, Rothberg AE, le Roux CW, Rubino F, Schauer P, Taylor R, Twenefour D. Consensus Report: Definition and Interpretation of Remission in Type 2 Diabetes. Diabetes Care. 2021;44(10):2438–44. 10.2337/dci21-0034. Epub ahead of print. PMID: 34462270; PMCID: PMC8929179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Vaz M, Pereira SS, Monteiro MP. Metabolomic signatures after bariatric surgery - a systematic review. Rev Endocr Metab Disord. 2022;23(3):503–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rubino F, Nathan DM, Eckel RH, Schauer PR, Alberti KG, Zimmet PZ, et al. Metabolic Surgery in the Treatment Algorithm for Type 2 Diabetes: A Joint Statement by International Diabetes Organizations. Diabetes Care. 2016;39(6):861–77. [DOI] [PubMed] [Google Scholar]
- 7.Cohen RV, Shikora S, Petry T, Caravatto PP, Le Roux CW. The Diabetes Surgery Summit II Guidelines: a Disease-Based Clinical Recommendation. Obes Surg. 2016;26(8):1989–91. [DOI] [PubMed] [Google Scholar]
- 8.Scarpellini E, Arts J, Karamanolis G, Laurenius A, Siquini W, Suzuki H, et al. International consensus on the diagnosis and management of dumping syndrome. Nat Rev Endocrinol. 2020;16(8):448–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Guyatt G, Oxman AD, Akl EA, Kunz R, Vist G, Brozek J, et al. GRADE guidelines: 1. Introduction-GRADE evidence profiles and summary of findings tables. J Clin Epidemiol. 2011;64(4):383–94. [DOI] [PubMed] [Google Scholar]
- 10.Hofsø D, Fatima F, Borgeraas H, Birkeland KI, Gulseth HL, Hertel JK, et al. Gastric bypass versus sleeve gastrectomy in patients with type 2 diabetes (Oseberg): a single-centre, triple-blind, randomised controlled trial. Lancet Diabetes Endocrinol. 2019;7(12):912–24. [DOI] [PubMed] [Google Scholar]
- 11.Mingrone G, Panunzi S, De Gaetano A, Guidone C, Iaconelli A, Capristo E, et al. Metabolic surgery versus conventional medical therapy in patients with type 2 diabetes: 10-year follow-up of an open-label, single-centre, randomised controlled trial. Lancet. 2021;397(10271):293–304. [DOI] [PubMed] [Google Scholar]
- 12.Rubino F, Shukla A, Pomp A, Moreira M, Ahn SM, Dakin G. Bariatric, metabolic, and diabetes surgery: what’s in a name? Ann Surg. 2014;259(1):117–22. [DOI] [PubMed] [Google Scholar]
- 13.Mingrone G, Panunzi S, De Gaetano A, Guidone C, Iaconelli A, Nanni G, et al. Bariatric-metabolic surgery versus conventional medical treatment in obese patients with type 2 diabetes: 5 year follow-up of an open-label, single-centre, randomised controlled trial. Lancet. 2015;386(9997):964–73. [DOI] [PubMed] [Google Scholar]
- 14.McTigue KM, Wellman R, Nauman E, Anau J, Coley RY, Odor A, Tice J, Coleman KJ, Courcoulas A, Pardee RE, Toh S, Janning CD, Williams N, Cook A, Sturtevant JL, Horgan C, Arterburn D, PCORNet Bariatric Study Collaborative. Comparing the Diabetes outcomes of sleeve gastrectomy and gastric bypass: the national patient-centered clinical research network (PCORNet) bariatric study. JAMA Surg. 2020;155(5):e200087. 10.1001/jamasurg.2020.0087. Epub 2020 May 20. PMID: 32129809; PMCID: PMC7057171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Khorgami Z, Shoar S, Saber AA, Howard CA, Danaei G, Sclabas GM. Outcomes of Bariatric surgery versus medical management for type 2 diabetes mellitus: a meta-analysis of randomized controlled trials. Obes Surg. 2019;29(3):964–74. 10.1007/s11695-018-3552-x. (PMID: 30402804). [DOI] [PubMed] [Google Scholar]
- 16.ElSayed NA, Aleppo G, Aroda VR, Bannuru RR, Brown FM, Bruemmer D, Collins BS, Hilliard ME, Isaacs D, Johnson EL, Kahan S, Khunti K, Leon J, Lyons SK, Perry ML, Prahalad P, Pratley RE, Seley JJ, Stanton RC, Gabbay RA. On behalf of the american diabetes association. 8. obesity and weight management for the prevention and treatment of type 2 diabetes: standards of care in diabetes-2023. Diabetes Care. 2023;46(Suppl 1):S128–39. 10.2337/dc23-S008. PMID: 36507637; PMCID: PMC9810466. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Courcoulas AP, Belle SH, Neiberg RH, Pierson SK, Eagleton JK, Kalarchian MA, DeLany JP, Lang W, Jakicic JM. Three-Year outcomes of bariatric surgery vs lifestyle intervention for type 2 diabetes mellitus treatment: a randomized clinical trial. JAMA Surg. 2015;150(10):931–40. 10.1001/jamasurg.2015.1534. PMID:26132586;PMCID:PMC4905566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ikramuddin S, Korner J, Lee WJ, Connett JE, Inabnet WB, Billington CJ, Thomas AJ, Leslie DB, Chong K, Jeffery RW, Ahmed L, Vella A, Chuang LM, Bessler M, Sarr MG, Swain JM, Laqua P, Jensen MD, Bantle JP. Roux-en-Y gastric bypass vs intensive medical management for the control of type 2 diabetes, hypertension, and hyperlipidemia: the Diabetes Surgery Study randomized clinical trial. JAMA. 2013;309(21):2240–9. 10.1001/jama.2013.5835. PMID:23736733;PMCID:PMC3954742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Miras AD, le Roux CW. Mechanisms underlying weight loss after bariatric surgery. Nat Rev Gastroenterol Hepatol. 2013;10(10):575–84. [DOI] [PubMed] [Google Scholar]
- 20.Buchwald H, Dorman RB, Rasmus NF, Michalek VN, Landvik NM, Ikramuddin S. Effects on GLP-1, PYY, and leptin by direct stimulation of terminal ileum and cecum in humans: implications for ileal transposition. Surgery Obes Relats Dis. 2014;10(5):780–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Buchwald H, Menchaca HJ, Michalek VN, Bertin NT. Ileal effect on blood glucose, HbA1c, and GLP-1 in Goto-Kakizaki rats. Obes Surg. 2014;24(11):1954–60. [DOI] [PubMed] [Google Scholar]
- 22.Lee WJ, Hur KY, Lakadawala M, Kasama K, Wong SK, Chen SC, Lee YC, Ser KH. Predicting success of metabolic surgery: age, body mass index, C-peptide, and duration score. Surg Obes Relat Dis. 2013;9:379–84. [DOI] [PubMed] [Google Scholar]
- 23.Aminian A, Brethauer SA, Andalib A, Nowacki AS, Jimenez A, Corcelles R, et al. Individualised Metabolic Surgery Score: Procedure Selection Based on Diabetes Severity. Ann Surg. 2017;266(4):650–7. [DOI] [PubMed] [Google Scholar]
- 24.Moradi M, Kabir A, Khalili D, et al. Type 2 diabetes remission after Roux-en-Y gastric bypass (RYGB), sleeve gastrectomy (SG), and one anastomosis gastric bypass (OAGB): results of the longitudinal assessment of bariatric surgery study. BMC Endocr Disord. 2022;22:260. 10.1186/s12902-022-01171-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Stenberg E, Olbers T, Cao Y, et al. Factors determining chance of type 2 diabetes remission after Roux-en-Y gastric bypass surgery: a nationwide cohort study in 8057 Swedish patients. BMJ Open Diabetes Res Care. 2021;9: e002033. 10.1136/bmjdrc-2020-002033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jans A, Näslund I, Ottosson J, Szabo E, Näslund E, Stenberg E. Duration of type 2 diabetes and remission rates after bariatric surgery in Sweden 2007–2015: A registry-based cohort study. PLoS Med. 2019;16(11): e1002985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Zhou X, Zeng C. Diabetes remission of bariatric surgery and nonsurgical treatments in type 2 diabetes patients who failure to meet the criteria for surgery: a systematic review and meta-analysis. BMC Endocr Disord. 2023;23:46. 10.1186/s12902-023-01283-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cardoso S, Pereira SS, Almeida RF, et al. Accuracy of prediction models for long-term type 2 diabetes remission after gastric bypass. Acta Diabetol. 2023;60:1019–26. 10.1007/s00592-023-02092-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Duggan EW, Carlson K, Umpierrez GE. Perioperative hyperglycaemia management: an update. Anesthesiology. 2017;126(3):547–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Rollins KE, Varadhan KK, Dhatariya K, Lobo DN. Systematic review of the impact of HbA1c on outcomes following surgery in patients with diabetes mellitus. Clinical Nutr. 2016;35(2):308–16. [DOI] [PubMed] [Google Scholar]
- 31.Basishvili G, Yang J, Nie L, Docimo S, Pryor AD, Spaniolas K. HbA1C is not directly associated with complications of bariatric surgery. Surg Obes Relat Dis. 2021;17(2):271–5. [DOI] [PubMed] [Google Scholar]
- 32.Carter J, Chang J, Birriel TJ, Moustarah F, Sogg S, Goodpaster K, Benson-Davies S, Chapmon K, Eisenberg D. ASMBS position statement on preoperative patient optimization before metabolic and bariatric surgery. Surg Obes Relat Dis. 2021;17(12):1956–76. 10.1016/j.soard.2021.08.024. Epub 2021 Sep 3 PMID: 34629296. [DOI] [PubMed] [Google Scholar]
- 33.Hart A, Goffredo P, Carroll R, et al. Optimizing Bariatric Surgery outcomes: the impact of preoperative elevated hemoglobin A1c levels on composite perioperative outcome measures. Surg Endosc. 2021;35:4618–23. 10.1007/s00464-020-07887-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Pina L, Dove J, Wood GC, et al. Stratified Preoperative A1c is not Significantly Associated With Clavien-Dindo Major Complications Following Bariatric Surgery in the MBSAQIP Database. The American SurgeonTM. 2022;88(11):2760–7. 10.1177/00031348221121551. [DOI] [PubMed] [Google Scholar]
- 35.Busetto L, Dicker D, Azran C, Batterham RL, Farpour-Lambert N, Fried M, Hjelmesæth J, Kinzl J, Leitner DR, Makaronidis JM, Schindler K, Toplak H, Yumuk V. Practical recommendations of the obesity management task force of the European association for the study of obesity for the post-bariatric surgery medical management. Obes Facts. 2017;10(6):597–632. 10.1159/000481825. Epub 2017 Dec 6. PMID: 29207379; PMCID: PMC5836195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Childs J, Mudge LA, Esterman A, Thompson SK. What Is the optimal time on a low-calorie diet prior to laparoscopic anti-reflux surgery? a prospective case-controlled study. J Gastrointest Surg. 2022;26(11):2249–54. 10.1007/s11605-022-05438-2. Epub 2022 Aug 25. PMID: 36008652; PMCID: PMC9643211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Faria SL, Faria OP, de Almeida Cardeal M, Ito MK. Effects of a very low calorie diet in the preoperative stage of bariatric surgery: a randomized trial. Surg Obes Relat Dis. 2015;11(1):230–7. 10.1016/j.soard.2014.06.007. Epub 2014 Jul 23. PMID: 25543310. [DOI] [PubMed] [Google Scholar]
- 38.Holderbaum M, Casagrande DS, Sussenbach S, Buss C. Effects of very low calorie diets on liver size and weight loss in the preoperative period of bariatric surgery: a systematic review. Surg Obes Relat Dis. 2018;14(2):237–44. 10.1016/j.soard.2017.09.531. [DOI] [PubMed] [Google Scholar]
- 39.Serafim MP, Santo MA, Gadducci AV, Scabim VM, Cecconello I, de Cleva R. Very low-calorie diet in candidates for bariatric surgery: change in body composition during rapid weight loss. Clinics (Sao Paulo). 2019;14(74):e560. 10.6061/clinics/2019/e560. PMID:30892414;PMCID:PMC6399661. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Romeijn MM, Kolen AM, Holthuijsen DDB, Janssen L, Schep G, Leclercq WKG, van Dielen FMH. Effectiveness of a low-calorie diet for liver volume reduction prior to bariatric surgery: a systematic review. Obes Surg. 2021;31(1):350–6. 10.1007/s11695-020-05070-6. Epub 2020 Nov 2. PMID: 33140292; PMCID: PMC7808983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Wolf RM, Oshima K, Canner JK, Steele KE. Impact of a preoperative low-calorie diet on liver histology in patients with fatty liver disease undergoing bariatric surgery. Surg Obes Relat Dis. 2019;15(10):1766–72. 10.1016/j.soard.2019.08.013. Epub 2019 Aug 22 PMID: 31558407. [DOI] [PubMed] [Google Scholar]
- 42.Hawkins DN, Faler BJ, Choi YU, Prasad BM. Time Course of Blood Pressure Decrease After Bariatric Surgery in Normotensive and Hypertensive Patients. Obes Surg. 2018;28(7):1845–51. 10.1007/s11695-017-3091-x. PMID: 29725978. [DOI] [PubMed] [Google Scholar]
- 43.Głuszewska A, Gryglewska B, Gąsowski J, Bilo G, Zarzycki B, Dzieża-Grudnik A, Major P, Budzyński A, Faini A, Parati G, Grodzicki T. Reduction of 24-h blood pressure variability in extreme obese patients 10 days and 6 months after bariatric surgery depending on pre-existing hypertension. Eur J Intern Med. 2019;60:39–45. 10.1016/j.ejim.2018.10.022. Epub 2018 Nov 9 PMID: 30420135. [DOI] [PubMed] [Google Scholar]
- 44.Oliveras A, Goday A, Sans L, Arias CE, Vazquez S, Benaiges D, Ramon JM, Pascual J. Changes in Central 24-h ambulatory blood pressure and hemodynamics 12 months after bariatric surgery: the barihta study. Obes Surg. 2020;30(1):195–205. 10.1007/s11695-019-04107-9. PMID: 31346932. [DOI] [PubMed] [Google Scholar]
- 45.Wang L, Lin M, Yu J, Fan Z, Zhang S, Lin Y, Chen X, Peng F. The Impact of bariatric surgery versus non-surgical treatment on blood pressure: systematic review and meta-analysis. Obes Surg. 2021;31(11):4970–84. 10.1007/s11695-021-05671-9. Epub 2021 Sep 14 PMID: 34519991. [DOI] [PubMed] [Google Scholar]
- 46.Al Nou’mani J, Al Alawi AM, Falhammar H, Al Qassabi A. Orthostatic intolerance after bariatric surgery: a systematic review and meta-analysis. Clin Obes. 2021;11(6):e12483. 10.1111/cob.12483. Epub 2021 Aug 19. PMID: 34409762. [DOI] [PubMed] [Google Scholar]
- 47.Sheehan A, Patti ME. Hypoglycaemia after upper gastrointestinal surgery: clinical approach to assessment, diagnosis, and treatment. Diabetes Metab Syndr Obes. 2020;13:4469–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lee D, Dreyfuss JM, Sheehan A, Puleio A, Mulla CM, Patti ME. Glycaemic patterns are distinct in post- bariatric hypoglycaemia after gastric bypass (PBH- RYGB). J Clin Endocrinol Metab. 2021;106:2291–303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Lupoli R, Lembo E, Rainone C, Schiavo L, Iannelli A, Di Minno MND, Capaldo B. Rate of post-bariatric hypoglycaemia using continuous glucose monitoring: A meta-analysis of literature studies. Nutr Metab Cardiovasc Dis. 2022;32(1):32–9. 10.1016/j.numecd.2021.08.047. Epub 2021 Sep 25 PMID: 34802853. [DOI] [PubMed] [Google Scholar]
- 50.Nor Hanipah Z, Punchai S, Birriel TJ, Lansang MC, Kashyap SR, Brethauer SA, Schauer PR, Aminian A. Clinical features of symptomatic hypoglycaemia observed after bariatric surgery. Surg Obes Relat Dis. 2018;14(9):1335–9. 10.1016/j.soard.2018.02.022. Epub 2018 Feb 28 PMID: 30001888. [DOI] [PubMed] [Google Scholar]
- 51.Stenberg E, Dos Reis Falcão LF, O’Kane M, Liem R, Pournaras DJ, Salminen P, Urman RD, Wadhwa A, Gustafsson UO, Thorell A. Guidelines for perioperative care in bariatric surgery: enhanced recovery after surgery (eras) society recommendations: a 2021 update. World J Surg. 2022;46(4):729–51. 10.1007/s00268-021-06394-9. Epub 2022 Jan 4. Erratum in: World J Surg. 2022 Jan 29; PMID: 34984504; PMCID: PMC8885505. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Giugliano D, Longo M, Scappaticcio L, et al. SGLT-2 inhibitors and cardiorenal outcomes in patients with or without type 2 diabetes: a meta-analysis of 11 CVOTs. Cardiovasc Diabetol. 2021;20:236. 10.1186/s12933-021-01430-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.FDA.GOV. 2015. FDA Drug Safety Communication: Available at: < Drug safety and availability > fda drug safety communication: fda warns that sglt2 inhibitors for diabetes may result in a serious condition of too much acid in the blood (archive-it.org). Last accessed 21 May 2023.
- 54.BOMSS Patient safety committee alert on the use of Gliflozins. 2022. https://bomss.org/wp-content/uploads/2022/03/BOMSS-PSA-SGLT-2i.pdf . Last accessed 21 May 2023.
- 55.National Institute for Health and Care Excellence. Type 2 diabetes in adults: management. 2022. https://www.nice.org.uk/guidance/ng28/chapter/Recommendations#drug-treatment. Last accessed 21 May 2023. NICE Guideline No.28.
- 56.Gasoyan H, Tajeu G, Halpern MT, Sarwer DB. Reasons for underutilization of bariatric surgery: The role of insurance benefit design. Surg Obes Relat Dis. 2019;15(1):146–51. 10.1016/j.soard.2018.10.005. Epub 2018 Oct 13. PMID: 30425002; PMCID: PMC6441615. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Sarwer DB, Gasoyan H, Bauerle Bass S, Spitzer JC, Soans R, Rubin DJ. Role of weight bias and patient-physician communication in the underutilisation of bariatric surgery. Surgery for obesity and related diseases : official journal of the American Society for Bariatric Surgery. 2021;17(11):1926–32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Murtha JA, Alagoz E, Breuer CR, Finn A, Raffa SD, Voils CI, et al. Individual-level barriers to bariatric surgery from patient and provider perspectives: a qualitative study. Am J Surg. 2022;224(1 Pt B):429–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zevin B, Sivapalan N, Chan L, Cofie N, Dalgarno N, Barber D. Factors influencing primary care provider referral for bariatric surgery: Systematic review. Can Fam Physician. 2022;68(3):e107–17. 10.46747/cfp.6803e107.PMID:35292475;PMCID:PMC9833187. PMID:35292475;PMCID:PMC9833187 . [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Conaty EA, Denham W, Haggerty SP, Linn JG, Joehl RJ, Ujiki MB. Primary Care Physicians’ Perceptions of Bariatric Surgery and Major Barriers to Referral. Obes Surg. 2020;30(2):521–6. [DOI] [PubMed] [Google Scholar]
- 61.Holmes S, Sarma S, Campbell S, Azab A, Qiang J, Mukerji G. Gaps in Referral to Bariatric Surgery for Patients With Type 2 Diabetes Seen in Endocrinology Clinics. Can J Diabetes. 2022;46(8):835-842.e1. 10.1016/j.jcjd.2022.06.005. Epub 2022 Jun 23 PMID: 36088215. [DOI] [PubMed] [Google Scholar]
- 62.Gasoyan H, Ibrahim JK, Aaronson WE, Sarwer DB. The role of health insurance characteristics in utilization of bariatric surgery. Surg Obes Relat Dis. 2021;17(5):860–8. 10.1016/j.soard.2021.01.024. Epub 2021 Jan 30. PMID: 33664010; PMCID: PMC8096659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Billmeier SE, Atkinson RB, Adrales GL. Surgeon presence and utilization of bariatric surgery in the United States. Surg Endosc. 2020;34(5):2136–42. 10.1007/s00464-019-07001-8. Epub 2019 Jul 30 PMID: 31363893. [DOI] [PubMed] [Google Scholar]
- 64.Iuzzolino E, Kim Y. Barriers impacting an individuals decision to undergo bariatric surgery: a systematic review. Obes Res Clin Pract. 2020;14(4):310–20. [DOI] [PubMed] [Google Scholar]
- 65.Zhang JC, Tomlinson G, Wnuk S, Sockalingam S, Cram P. Disparities in receipt of bariatric surgery in Canada: an analysis of data from an ontario bariatric surgery referral center. Med Care. 2019;57(9):723–7. 10.1097/MLR.0000000000001163. PMID: 31274783. [DOI] [PubMed] [Google Scholar]
- 66.Paula TP, Viana LV, Neto AT, Leitão CB, Gross JL, Azevedo MJ. Effects of the DASH Diet and walking on blood pressure in patients with type 2 diabetes and uncontrolled hypertension: a randomized controlled trial. J Clin Hypertens (Greenwich). 2015;17(11):895–901. 10.1111/jch.12597. Epub 2015 Jun 4. PMID: 26041459; PMCID: PMC8031764. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Papamichou D, Panagiotakos DB, Itsiopoulos C. Dietary patterns and management of type 2 diabetes: a systematic review of randomised clinical trials. Nutr Metab Cardiovasc Dis. 2019;29(6):531–43. 10.1016/j.numecd.2019.02.004. Epub 2019 Feb 25 PMID: 30952576. [DOI] [PubMed] [Google Scholar]
- 68.Wang X, Li Q, Liu Y, Jiang H, Chen W. Intermittent fasting versus continuous energy-restricted diet for patients with type 2 diabetes mellitus and metabolic syndrome for glycaemic control: a systematic review and meta-analysis of randomized controlled trials. Diabetes Res Clin Pract. 2021;179:109003. 10.1016/j.diabres.2021.109003. Epub 2021 Aug 12 PMID: 34391831. [DOI] [PubMed] [Google Scholar]
- 69.Goldenberg JZ, Day A, Brinkworth GD, Sato J, Yamada S, Jönsson T, Beardsley J, Johnson JA, Thabane L, Johnston BC. Efficacy and safety of low and very low carbohydrate diets for type 2 diabetes remission: systematic review and meta-analysis of published and unpublished randomized trial data. BMJ. 2021;13(372):m4743. 10.1136/bmj.m4743. PMID:33441384;PMCID:PMC7804828. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ren M, Zhang H, Qi J, Hu A, Jiang Q, Hou Y, Feng Q, Ojo O, Wang X. An Almond-based low carbohydrate diet improves depression and glycometabolism in patients with type 2 diabetes through modulating gut microbiota and glp-1: a randomized controlled trial. Nutrients. 2020;12(10):3036. 10.3390/nu12103036. PMID:33022991;PMCID:PMC7601479. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Gažarová M, Galšneiderová M, Mečiarová L. Obesity diagnosis and mortality risk based on a body shape index (ABSI) and other indices and anthropometric parameters in university students. Rocz Panstw Zakl Hig. 2019;70(3):267–75. 10.32394/rpzh.2019.0077. (PMID: 31515986). [DOI] [PubMed] [Google Scholar]
- 72.Lima Borges L, de Rodrigues Lima T, Augusto Santos Silva D. Accuracy of anthropometric indicators of obesity to identify high blood pressure in adolescents-systematic review. Peer J. 2022;10:e13590. 10.7717/peerj.13590. PMID: 35966930; PMCID: PMC9373973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Aminian A, Chang J, Brethauer SA, Kim JJ, American Society for metabolic and bariatric surgery clinical issues committee. ASMBS updated position statement on bariatric surgery in class I obesity (BMI 30–35 kg/m2). Surg Obes Relat Dis. 2018;14(8):1071–87. [DOI] [PubMed] [Google Scholar]
- 74.Eisenberg D, Shikora SA, Aarts E, Aminian A, Angrisani L, Cohen RV, De Luca M, Faria SL, Goodpaster KPS, Haddad A, Himpens JM, Kow L, Kurian M, Loi K, Mahawar K, Nimeri A, O’Kane M, Papasavas PK, Ponce J, Pratt JSA, Rogers AM, Steele KE, Suter M, Kothari SN. 2022 American Society for metabolic and bariatric surgery (asmbs) and international federation for the surgery of obesity and metabolic disorders (ifso): indications for metabolic and bariatric surgery. Surg Obes Relat Dis. 2022;18(12):1345–56. 10.1016/j.soard.2022.08.013. Epub 2022 Oct 21 PMID: 36280539. [DOI] [PubMed] [Google Scholar]
- 75.Mechanick JI, Apovian C, Brethauer S, Garvey WT, Joffe AM, Kim J, Kushner RF, Lindquist R, Pessah-Pollack R, Seger J, Urman RD, Adams S, Cleek JB, Correa R, Figaro MK, Flanders K, Grams J, Hurley DL, Kothari S, Seger MV, Still CD. Clinical practice guidelines for the perioperative nutrition, metabolic, and nonsurgical support of patients undergoing bariatric procedures - 2019 update: cosponsored by american association of clinical endocrinologists/american college of endocrinology, the obesity society, american society for metabolic & bariatric surgery, obesity medicine association, and american society of anesthesiologists - executive summary. Endocr Pract. 2019;25(12):1346–59. 10.4158/GL-2019-0406. Epub 2019 Nov 4 PMID: 31682518. [DOI] [PubMed] [Google Scholar]
- 76.Roman M, Monaghan A, Serraino GF, et al. Meta-analysis of the influence of lifestyle changes for preoperative weight loss on surgical outcomes. Br J Surg. 2019;106(3):181–9. [DOI] [PubMed] [Google Scholar]
- 77.O’Kane M, Parretti HM, Pinkney J, Welbourn R, Hughes CA, Mok J, Walker N, Thomas D, Devin J, Coulman KD, Pinnock G, Batterham RL, Mahawar KK, Sharma M, Blakemore AI, McMillan I, Barth JH. British Obesity and Metabolic Surgery Society Guidelines on perioperative and postoperative biochemical monitoring and micronutrient replacement for patients undergoing bariatric surgery-2020 update. Obes Rev. 2020;21(11):e13087. 10.1111/obr.13087. Epub 2020 Aug 2. PMID: 32743907; PMCID: PMC7583474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Parrott J, Frank L, Rabena R, Craggs-Dino L, Isom KA, Greiman L. American Society for Metabolic and Bariatric Surgery Integrated Health Nutritional Guidelines for the Surgical Weight Loss Patient 2016 Update: Micronutrients. Surg Obes Relat Ddis. 2017;13(5):727–41. 10.1016/j.soard.2016.12.018. Epub 2017 Jan 19 PMID: 28392254. [DOI] [PubMed] [Google Scholar]
- 79.Quilliot D, Coupaye M, Ciangura C, Czernichow S, Sallé A, Gaborit B, Alligier M, Nguyen-Thi PL, Dargent J, Msika S, Brunaud L. Recommendations for nutritional care after bariatric surgery: Recommendations for best practice and SOFFCO-MM/AFERO/SFNCM/expert consensus. J Visc Surg. 2021;158(1):51–61. 10.1016/j.jviscsurg.2020.10.013. Epub 2021 Jan 9 PMID: 33436155. [DOI] [PubMed] [Google Scholar]
- 80.The National institute for health and care excellence. Obesity: clinical assessment and management quality standard published: Aug 4 2016. www.nice.org.uk/guidance/qs127. Last Accessed 22 May 2023.
- 81.The national institute for health and care excellence. Obesity: identification, assessment and management Clinical guideline Published: Nov 27 2014 . www.nice.org.uk/guidance/cg189. Last accessed 22 May 2023. [PubMed]
- 82.Ogden J, Ratcliffe D, Snowdon-Carr V. British obesity metabolic surgery society endorsed guidelines for psychological support pre- and post-bariatric surgery. Clin Obes. 2019;9(6):e12339. 10.1111/cob.12339. Epub 2019 Sep 11. PMID: 31512398. [DOI] [PubMed] [Google Scholar]
- 83.Ahmed AT, Warton EM, Schaefer CA, Shen L, McIntyre RS. The effect of bariatric surgery on psychiatric course among patients with bipolar disorder. Bipolar Disord. 2013;15(7):753–63. 10.1111/bdi.12109. Epub 2013 Aug 5. PMID: 23909994; PMCID: PMC3844030. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Friedman KE, Applegate K, Portenier D, McVay MA. Bariatric surgery in patients with bipolar spectrum disorders: selection factors, postoperative visit attendance, and weight outcomes. Surg Obes Relat Dis. 2017;13(4):643–51. 10.1016/j.soard.2016.10.009. Epub 2016 Oct 17. PMID: 28169206; PMCID: PMC5400728. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Kouidrat Y, Amad A, Stubbs B, Moore S, Gaughran F. Surgical management of obesity among people with schizophrenia and bipolar disorder: a systematic review of outcomes and recommendations for future research. Obes Surg. 2017;27(7):1889–95. 10.1007/s11695-017-2715-5. PMID: 28508277. [DOI] [PubMed] [Google Scholar]
- 86.Kalarchian MA, Marcus MD. Psychosocial concerns following bariatric surgery: current status. Curr Obes Rep. 2019;8(1):1–9. 10.1007/s13679-019-0325-3. Erratum In: Curr Obes Rep. 2019 Feb 8;PMID: 30659459. [DOI] [PubMed] [Google Scholar]
- 87.Lagerros YT, Brandt L, Sundbom M, Hedberg J, Bodén R. Risk of delayed discharge and reoperation of gastric bypass patients with psychiatric comorbidity-a nationwide cohort study. Obes Surg. 2020;30(7):2511–8. 10.1007/s11695-020-04483-7. PMID: 32152840; PMCID: PMC7260256. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Jalilvand A, Dewire J, Detty A, Needleman B, Noria S. Baseline psychiatric diagnoses are associated with early readmissions and long hospital length of stay after bariatric surgery. Surg Endosc. 2019;33(5):1661–6. 10.1007/s00464-018-6459-7. Epub 2018 Sep 25. PMID: 30255332; PMCID: PMC6461512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Wong SE, Akioyamen L, Melamed O, Taylor VH. Exploring presurgery and post-surgery substance use disorder and alcohol use disorder in bariatric surgery: a qualitative scoping review. Int J Obes (Lond). 2019;43(9):1659–74. 10.1038/s41366-019-0397-x. Epub 2019 Jun 18. PMID: 31213657. [DOI] [PubMed] [Google Scholar]
- 90.British Obesity and Metabolic Surgery Society (BOMSS). Commissioning guide: weight assessment and management clinics (tier 3) March 2014. https://www.rcseng.ac.uk/-/media/files/rcs/library-and-publications/non-journal-publications/weight-assessment-and-management-tier-services--commissioning-guide.pdf. Last accessed 24 May 2023.
- 91.Kermansaravi M, Valizadeh R, Jazi AD, Shahmiri SS, Martinez JAL, Mousavimaleki A, Eghbali F, Aliakbar A, Atarodi H, Aghajani E, Lainas P. current status of metabolic/bariatric surgery in type 1 diabetes mellitus: an updated systematic review and meta-analysis. Obes Surg. 2022;32(5):1726–33. 10.1007/s11695-022-05980-7. Epub 2022 Feb 24 PMID: 35211844. [DOI] [PubMed] [Google Scholar]
- 92.Höskuldsdóttir G, Ekelund J, Miftaraj M, Wallenius V, Ottosson J, Näslund I, Gudbjörnsdottir S, Sattar N, Svensson AM, Eliasson B. Potential benefits and harms of gastric bypass surgery in obese individuals with type 1 diabetes: a nationwide, matched. Observ Cohort Study Diab Care. 2020;43(12):3079–85. 10.2337/dc20-0388. Epub 2020 Oct 6 PMID: 33023988. [DOI] [PubMed] [Google Scholar]
- 93.Parmar C, Appel S, Lee L, Ribeiro R, Sakran N, Pouwels S. Choice of Bariatric Surgery in Patients with Obesity and Type 1 Diabetes Mellitus? an Up-to-Date Systematic Review. Obes Surg. 2022;32(12):3992–4006. 10.1007/s11695-022-06321-4. Epub 2022 Oct 22 PMID: 36272054. [DOI] [PubMed] [Google Scholar]
- 94.Thaher O, Iaroshevych V, Driouch J, Hukauf M, Croner RS, Stroh C. Current status of metabolic surgery in patients with type I diabetes mellitus and obesity: a nationwide multicenter study. Langenbecks Arch Surg. 2023;408(1):46. 10.1007/s00423-023-02788-6. PMID: 36662321. [DOI] [PubMed] [Google Scholar]
- 95.Maraka S, Kudva YC, Kellogg TA, Collazo-Clavell ML, Mundi MS. Bariatric surgery and diabetes: Implications of type 1 versus insulin-requiring type 2. Obesity. 2015;23:552–7. 10.1002/oby.20992. [DOI] [PubMed] [Google Scholar]
- 96.Blonde L, Umpierrez GE, Reddy SS, McGill JB, Berga SL, Bush M, Chandrasekaran S, DeFronzo RA, Einhorn D, Galindo RJ, Gardner TW, Garg R, Garvey WT, Hirsch IB, Hurley DL, Izuora K, Kosiborod M, Olson D, Patel SB, Pop-Busui R, Sadhu AR, Samson SL, Stec C, Tamborlane WV Jr, Tuttle KR, Twining C, Vella A, Vellanki P, Weber SL. American Association of clinical endocrinology clinical practice guideline: developing a diabetes mellitus comprehensive care plan-2022 update. Endocr Pract. 2022;28(10):923–1049. 10.1016/j.eprac.2022.08.002. Epub 2022 PMID: 35963508; PMCID: PMC10200071. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Maddaloni E, Moretti C, Mignogna C, Buzzetti R. Adult-onset autoimmune diabetes in 2020: an update. Maturitas. 2020;137:37–44. 10.1016/j.maturitas.2020.04.014. Epub 2020 Apr 30 PMID: 32498935. [DOI] [PubMed] [Google Scholar]
- 98.Shepherd MH, Shields BM, Hudson M, et al. A UK nationwide prospective study of treatment change in MODY: genetic subtype and clinical characteristics predict optimal glycaemic control after discontinuing insulin and metformin. Diabetologia. 2018;61(12):2520–7. 10.1007/s00125-018-4728-6[EL2;PCS]. PubMed: 30229274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Warncke K, Kummer S, Raile K, et al. Frequency and characteristics of MODY 1 (HNF4A mutation) and MODY 5 (HNF1B mutation): analysis from the dpv database. J Clin Endocrinol Metab. 2019;104(3):845–55. 10.1210/jc.2018-01696[EL2;ES]. PubMed: 30535056. [DOI] [PubMed] [Google Scholar]
- 100.Guimarães M, Pereira SS, Nora M, Monteiro MP. Bariatric Surgery Outcomes in Patients with Latent Autoimmune Diabetes of the Adult. Obes Facts. 2021;14(4):425–30. 10.1159/000517588. Epub 2021 Jul 28. PMID: 34320499; PMCID: PMC8406337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Aminian A, Sharma G, Wilson RL, Kashyap SR, Lo Menzo E, Szomstein S, Rosenthal RJ, Schauer PR, Vangoitsenhoven R. Bariatric surgery in patients with obesity and latent autoimmune diabetes in adults (LADA). Diabetes Care. 2020;43(5):e56–7. 10.2337/dc19-2388. Epub 2020 Mar 18. PMID: 32188775; PMCID: PMC7171945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Almutairi A, Alkhalaf H, Caswell A, Kolobe LE, Alatassi A, Alzughaibi N, Alnamshan M, Alqanatish J. Impact of a same day admission project in reducing the preoperative bed occupancy demand in a pediatric inpatient hospital. Ann Med Surg (Lond). 2022;6(81):104304. 10.1016/j.amsu.2022.104304. PMID:35991505;PMCID:PMC9386388. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Alqahtani AR, Elahmedi M, Amro N, Abdurabu HY, Abdo N, Alqahtani S, Boutros A, Ebishi A, Al-Darwish A. Laparoscopic sleeve gastrectomy as day-case surgery versus conventional hospitalization: results of the DAYSLEEVE randomized clinical trial. Surg Obes Relat Dis. 2022;18(9):1141–9. 10.1016/j.soard.2022.05.023. Epub 2022 May 25 PMID: 35803849. [DOI] [PubMed] [Google Scholar]
- 104.van Ede ES, Scheerhoorn J, Buise MP, Bouwman RA, Nienhuijs SW. Telemonitoring for perioperative care of outpatient bariatric surgery: Preference-based randomized clinical trial. PLoS ONE. 2023;18(2):e0281992. 10.1371/journal.pone.0281992. PMID:36812167;PMCID:PMC9946229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Ignat M, Ansiaux J, Osailan S, D’Urso A, Morainvillers-Sigwalt L, Vix M, Mutter D. A cost analysis of healthcare episodes including day-case bariatric surgery (roux-en-y gastric bypass and sleeve gastrectomy) versus inpatient surgery. Obes Surg. 2022;32(8):2504–11. 10.1007/s11695-022-06144-3. Epub 2022 Jun 10 PMID: 35689142. [DOI] [PubMed] [Google Scholar]
- 106.Badaoui R, Rebibo L, Kirat K, Alami Y, Hchikat A, Cosse C, Regimbeau JM, Lorne E. Satisfaction rate of patients undergoing sleeve gastrectomy as day-case surgery compared to conventional hospitalization: a prospective non-randomized study. J Anesth. 2018;32(2):227–35. 10.1007/s00540-018-2469-9. Epub 2018 Mar 2 PMID: 29500534. [DOI] [PubMed] [Google Scholar]
- 107.Omar I, Madhok B, Parmar C, Khan O, Wilson M, Mahawar K. Analysis of National Bariatric Surgery Related Clinical Incidents: Lessons Learned and a Proposed Safety Checklist for Bariatric Surgery. Obes Surg. 2021;31(6):2729–42. 10.1007/s11695-021-05330-z. Epub 2021 Mar 5 PMID: 33675021. [DOI] [PubMed] [Google Scholar]
- 108.Mauro A, Lusetti F, Scalvini D, Bardone M, De Grazia F, Mazza S, Pozzi L, Ravetta V, Rovedatti L, Sgarlata C, Strada E, Torello Viera F, Veronese L, Olivo Romero DE, Anderloni A. A Comprehensive Review on Bariatric Endoscopy: Where We Are Now and Where We Are Going. Medicina (Kaunas). 2023;59(3):636. 10.3390/medicina59030636. PMID:36984637;PMCID:PMC10052707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Kotinda APST, de Moura DTH, Ribeiro IB, Singh S, da Ponte Neto AM, Proença IM, Flor MM, de Souza KL, Bernardo WM, de Moura EGH. Efficacy of Intragastric Balloons for Weight Loss in Overweight and Obese Adults: a Systematic Review and Meta-analysis of Randomized Controlled Trials. Obes Surg. 2020;30(7):2743–53. 10.1007/s11695-020-04558-5. PMID: 32300945. [DOI] [PubMed] [Google Scholar]
- 110.Abu Dayyeh BK, Maselli DB, Rapaka B, Lavin T, Noar M, Hussan H, Chapman CG, Popov V, Jirapinyo P, Acosta A, Vargas EJ, Storm AC, Bazerbachi F, Ryou M, French M, Noria S, Molina D, Thompson CC. Adjustable intragastric balloon for treatment of obesity: a multicentre, open-label, randomised clinical trial. Lancet. 2021;398(10315):1965–73. 10.1016/S0140-6736(21)02394-1. Epub 2021 Nov 15. Erratum in: Lancet. 2021 Nov 27;398(10315):1964. PMID: 34793746. [DOI] [PubMed] [Google Scholar]
- 111.Loo JH, Lim YH, Seah HL, Chong AZQ, Tay KV. Intragastric Balloon as Bridging Therapy Prior to Bariatric Surgery for Patients with Severe Obesity (BMI ≥ 50 kg/m2): a Systematic Review and Meta-analysis. Obes Surg. 2022;32(2):489–502. 10.1007/s11695-021-05772-5. Epub 2021 Nov 17 PMID: 34787766. [DOI] [PubMed] [Google Scholar]
- 112.Popov VB, Ou A, Schulman AR, Thompson CC. The Impact of Intragastric Balloons on Obesity-Related Co-Morbidities: A Systematic Review and Meta-Analysis. Am J Gastroenterol. 2017;112(3):429–39. 10.1038/ajg.2016.530. Epub 2017 Jan 24 PMID: 28117361. [DOI] [PubMed] [Google Scholar]
- 113.Tønnesen CJ, Hjelmesæth J, Hofsø D, Tonstad S, Hertel JK, Heggen E, Johnson LK, Mathisen TE, Kalager M, Wieszczy P, Medhus AW, Løberg M, Aabakken L, Bretthauer M. A novel intragastric balloon for treatment of obesity and type 2 diabetes. a two-center pilot trial. Scand J Gastroenterol. 2022;57(2):232–8. 10.1080/00365521.2021.1994641. Epub 2021 Oct 29. PMID: 34714203. [DOI] [PubMed] [Google Scholar]
- 114.Shah RH, Vedantam S, Kumar S, Amin S, Pearlman M, Bhalla S. Intragastric Balloon significantly improves metabolic parameters at 6 months: a meta-analysis. Obes Surg. 2023;33(3):725–32. 10.1007/s11695-022-06437-7. Epub 2023 Jan 12 PMID: 36633759. [DOI] [PubMed] [Google Scholar]
- 115.Abu Dayyeh BK, Bazerbachi F, Vargas EJ, Sharaiha RZ, Thompson CC, Thaemert BC, Teixeira AF, Chapman CG, Kumbhari V, Ujiki MB, Ahrens J, Day C, MERIT Study Group, Galvao Neto M, Zundel N, Wilson EB. Endoscopic sleeve gastroplasty for treatment of class 1 and 2 obesity (MERIT): a prospective, multicentre, randomised trial. Lancet. 2022;400(10350):441–51. 10.1016/S0140-6736(22)01280-6. Epub 2022 Jul 28. PMID: 35908555. [DOI] [PubMed] [Google Scholar]
- 116.Maselli DB, Alqahtani AR, Abu Dayyeh BK, Elahmedi M, Storm AC, Matar R, Nieto J, Teixeira A, Al Khatry M, Neto MG, Kumbhari V, Vargas EJ, Jaruvongvanich V, Mundi MS, Deshmukh A, Itani MI, Farha J, Chapman CG, Sharaiha R. Revisional endoscopic sleeve gastroplasty of laparoscopic sleeve gastrectomy: an international, multicenter study. Gastrointest Endosc. 202;93(1):122–30. 10.1016/j.gie.2020.05.028. Epub 2020 May 27 PMID: 32473252. [DOI] [PubMed] [Google Scholar]
- 117.Bhandari M, Kosta S, Reddy M, Mathur W, Neto MG, Bhandari M. Four-year outcomes for endoscopic sleeve gastroplasty from a single centre in India. J Minim Access Surg. 2023;19(1):101–6. 10.4103/jmas.jmas_3_22. PMID: 36124467; PMCID: PMC10034804. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Alqahtani AR, Elahmedi M, Aldarwish A, Abdurabu HY, Alqahtani S. Endoscopic gastroplasty versus laparoscopic sleeve gastrectomy: a noninferiority propensity score-matched comparative study. Gastrointest Endosc. 2022;96(1):44–50. 10.1016/j.gie.2022.02.050. Epub 2022 Mar 3 PMID: 35248571. [DOI] [PubMed] [Google Scholar]
- 119.Fehervari M, Fadel MG, Alghazawi LOK, Das B, Rodríguez-Luna MR, Perretta S, Wan A, Ashrafian H. Medium-term weight loss and remission of comorbidities following endoscopic sleeve gastroplasty: a systematic review and meta-analysis. Obes Surg. 2023;33(11):3527–38. 10.1007/s11695-023-06778-x. Epub 2023 Sep 13. PMID: 37700147; PMCID: PMC10602997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.National Institute for Health and Care Excellence (NICE) (2024). Endoscopic sleeve gastroplasty for obesity Interventional procedures guidance.www.nice.org.uk/guidance/ipg783. Published: 22 February 2024.
- 121.Viner R, White B, Christie D. Type 2 diabetes in adolescents: a severe phenotype posing major clinical challenges and public health burden. Lancet. 2017;389(10085):2252–60. 10.1016/S0140-6736(17)31371-5. PMID: 28589895. [DOI] [PubMed] [Google Scholar]
- 122.Tryggestad JB, Willi SM. Complications and comorbidities of T2DM in adolescents: findings from the TODAY clinical trial. J Diabetes Complications. 2015;29(2):307–12. 10.1016/j.jdiacomp.2014.10.009. Epub 2014 Oct 29. PMID: 25468310; PMCID: PMC4333013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Michalsky MP, Inge TH, Teich S, et al. Adolescent bariatric surgery program characteristics: the Teen Longitudinal Assessment of Bariatric Surgery (Teen-LABS) study experience. Semin Pediatr Surg. 2014;23(1):5–10. PubMed: 24491361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Khidir N, El-Matbouly MA, Sargsyan D, Al-Kuwari M, Bashah M, Gagner M. Five-year Outcomes of Laparoscopic Sleeve Gastrectomy: a Comparison Between Adults and Adolescents. Obes Surg. 2018;28(7):2040–5. 10.1007/s11695-018-3139-6. PMID: 29430596. [DOI] [PubMed] [Google Scholar]
- 125.Inge TH, Courcoulas AP, Jenkins TM, Michalsky MP, Brandt ML, Xanthakos SA, Dixon JB, Harmon CM, Chen MK, Xie C, Evans ME, Helmrath MA. Five-Year outcomes of gastric bypass in adolescents as compared with adults. N Engl J Med. 2019;380(22):2136–45. 10.1056/NEJMoa1813909. Epub 2019 May 16. PMID: 31116917; PMCID: PMC7345847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Salimi-Jazi F, Chkhikvadze T, Shi J, Pourmehdi-Lahiji A, Moshksar A, Rafeeqi TA, Pratt J, Bruzoni M, Chao S. Trends in Adolescent Bariatric Procedures: a 15-Year Analysis of the National Inpatient Survey. Obes Surg. 2022;32(11):3658–65. 10.1007/s11695-022-06265-9. Epub 2022 Sep 14 PMID: 36103080. [DOI] [PubMed] [Google Scholar]
- 127.Styne DM, Arslanian SA, Connor EL, Farooqi IS, Murad MH, Silverstein JH, Yanovski JA. Pediatric Obesity-Assessment, Treatment, and Prevention: An Endocrine Society Clinical Practice Guideline. J Clin Endocrinol Metab. 2017;102(3):709–57. 10.1210/jc.2016-2573. PMID:28359099;PMCID:PMC6283429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Pratt JSA, Browne A, Browne NT, Bruzoni M, Cohen M, Desai A, Inge T, Linden BC, Mattar SG, Michalsky M, Podkameni D, Reichard KW, Stanford FC, Zeller MH, Zitsman J. ASMBS pediatric metabolic and bariatric surgery guidelines, 2018. Surg Obes Relat Dis. 2018;14(7):882–901. 10.1016/j.soard.2018.03.019. Epub 2018 Mar 23. PMID: 30077361; PMCID: PMC6097871. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Noparatayaporn P, Thavorncharoensap M, Chaikledkaew U, Bagepally BS, Thakkinstian A. incremental net monetary benefit of bariatric surgery: systematic review and meta-analysis of cost-effectiveness evidences. Obes Surg. 2021;31(7):3279–90. 10.1007/s11695-021-05415-9. Epub 2021 Apr 24. PMID: 33893610; PMCID: PMC8175295. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Lauren BN, Lim F, Krikhely A, Taveras EM, Woo Baidal JA, Bellows BK, Hur C. Estimated Cost-effectiveness of Medical Therapy, Sleeve Gastrectomy, and Gastric Bypass in Patients With Severe Obesity and Type 2 Diabetes. JAMA Netw Open. 2022;5(2): e2148317. 10.1001/jamanetworkopen.2021.48317.PMID:35157054;PMCID:PMC8845022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Jordan K, Fawsitt CG, Carty PG, Clyne B, Teljeur C, Harrington P, Ryan M. Cost-effectiveness of metabolic surgery for the treatment of type 2 diabetes and obesity: a systematic review of economic evaluations. Eur J Health Econ. 2023;24(4):575–590. 10.1007/s10198-022-01494-2. Epub 2022 Jul 22. PMID: 35869383; PMCID: PMC10175448. [DOI] [PMC free article] [PubMed]
- 132.Sandoval DA, Patti ME. Glucose metabolism after bariatric surgery: implications for T2DM remission and hypoglycaemia. Nat Rev Endocrinol. 2023;19(3):164–76. 10.1038/s41574-022-00757-5. Epub 2022 Oct 26 PMID: 36289368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Ding Z, Jin L, Song Y, Feng C, Shen P, Li H. Comparison of single-anastomosis gastric bypass and sleeve gastrectomy on type 2 diabetes mellitus remission for obese patients: A meta-analysis of randomized controlled trials. Asian J Surg. 2023:S1015–9584(23)00368–8. 10.1016/j.asjsur.2023.03.062. Epub ahead of print. PMID: 37002039. [DOI] [PubMed]
- 134.Borgeraas H, Hofsø D, Hertel JK, Hjelmesaeth J. Comparison of the effect of Roux-en-Y gastric bypass and sleeve gastrectomy on remission of type 2 diabetes: A systematic review and meta-analysis of randomized controlled trials. Obes Rev. 2020;21(6):e13011. 10.1111/obr.13011. Epub 2020 Mar 11. Erratum in: Obes Rev. 2022 Apr;23(4):e13432. PMID: 32162437; PMCID: PMC7317556. [DOI] [PMC free article] [PubMed]
- 135.Currie AC, Askari A, Fangueiro A, Mahawar K. Network Meta-Analysis of Metabolic Surgery Procedures for the Treatment of Obesity and Diabetes. Obes Surg. 2021;31(10):4528–4541. 10.1007/s11695-021-05643-z. Epub 2021 Aug 7. PMID: 34363144; PMCID: PMC8346344. [DOI] [PMC free article] [PubMed]
- 136.Salminen P, Grönroos S, Helmiö M, Hurme S, Juuti A, Juusela R, Peromaa-Haavisto P, Leivonen M, Nuutila P, Ovaska J. Effect of Laparoscopic Sleeve Gastrectomy vs Roux-en-Y Gastric Bypass on Weight Loss, Comorbidities, and Reflux at 10 Years in Adult Patients With Obesity: The SLEEVEPASS Randomized Clinical Trial. JAMA Surg. 2022;157(8):656–66. 10.1001/jamasurg.2022.2229.PMID:35731535;PMCID:PMC9218929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Balamurugan G, Leo SJ, Sivagnanam ST, Balaji Prasad S, Ravindra C, Rengan V, Arora E, Bindal V. Comparison of Efficacy and Safety Between Roux-en-Y Gastric Bypass (RYGB) vs One Anastomosis Gastric Bypass (OAGB) vs Single Anastomosis Duodeno-ileal Bypass with Sleeve Gastrectomy (SADI-S): a Systematic Review of Bariatric and Metabolic Surgery. Obes Surg. 2023 May 4. 10.1007/s11695-023-06602-6. Epub ahead of print. PMID: 37140720. [DOI] [PubMed]
- 138.Royal College of Surgeons (2018). Surgical Care Team. Guidance Framework. http://www.rcseng.ac.uk/surgicalcareteam Last accessed 28/05/2023
- 139.ElSayed NA, Aleppo G, Aroda VR, Bannuru RR, Brown FM, Bruemmer D, Collins BS, Hilliard ME, Isaacs D, Johnson EL, Kahan S, Khunti K, Leon J, Lyons SK, Perry ML, Prahalad P, Pratley RE, Seley JJ, Stanton RC, Gabbay RA, on behalf of the American Diabetes Association. 6. Glycaemic Targets: Standards of Care in Diabetes-2023. Diabetes Care. 2023;46(Suppl 1):S97-S110. 10.2337/dc23-S006. PMID: 36507646; PMCID: PMC9810469. [DOI] [PMC free article] [PubMed]
- 140.Taheri S, Zaghloul H, Chagoury O, Elhadad S, Ahmed SH, El Khatib N, Amona RA, El Nahas K, Suleiman N, Alnaama A, Al-Hamaq A, Charlson M, Wells MT, Al-Abdulla S, Abou-Samra AB. Effect of intensive lifestyle intervention on bodyweight and glycaemia in early type 2 diabetes (DIADEM-I): an open-label, parallel-group, randomised controlled trial. Lancet Diabetes Endocrinol. 2020;8(6):477–89. 10.1016/S2213-8587(20)30117-0. PMID: 32445735. [DOI] [PubMed] [Google Scholar]
- 141.ElSayed NA, Aleppo G, Aroda VR, Bannuru RR, Brown FM, Bruemmer D, Collins BS, Hilliard ME, Isaacs D, Johnson EL, Kahan S, Khunti K, Leon J, Lyons SK, Perry ML, Prahalad P, Pratley RE, Seley JJ, Stanton RC, Gabbay RA, on behalf of the American Diabetes Association. 2. Classification and Diagnosis of Diabetes: Standards of Care in Diabetes-2023. Diabetes Care. 2023;46(Suppl 1):S19-S40. 10.2337/dc23-S002. Erratum in: Diabetes Care. 2023 Feb 01;: PMID: 36507649; PMCID: PMC9810477. [DOI] [PMC free article] [PubMed]
- 142.Adam S, Azmi S, Ho JH, Liu Y, Ferdousi M, Siahmansur T, Kalteniece A, Marshall A, Dhage SS, Iqbal Z, D'Souza Y, Natha S, Kalra PA, Donn R, Ammori BJ, Syed AA, Durrington PN, Malik RA, Soran H. Improvements in Diabetic Neuropathy and Nephropathy After Bariatric Surgery: a Prospective Cohort Study. Obes Surg. 2021;31(2):554–563. 10.1007/s11695-020-05052-8. Epub 2020 Oct 26. PMID: 33104989; PMCID: PMC7847862. [DOI] [PMC free article] [PubMed]
- 143.Chandru S, Pradeepa R, Poonkodi VP, Pramodkumar TA, Kumar MS, Tiwaskar M, Gokulakrishnan K, Anjana RM, Mohan V, Rajalakshmi R. Effects of Metabolic Surgery on Diabetic Kidney Disease and Diabetic Retinopathy Among Obese Asian Indians with Type 2 Diabetes. J Assoc Physicians India. 2022;70(1):11–2. PMID: 35062810. [PubMed] [Google Scholar]
- 144.Chen X, Zhang J, Zhou Z. The effects of metabolic surgery on microvascular complications in obese patients with type 2 diabetes: a meta-analysis. Surg Obes Relat Dis. 2021;17(2):434–43. 10.1016/j.soard.2020.09.013. Epub 2020 Sep 14 PMID: 33144039. [DOI] [PubMed] [Google Scholar]
- 145.Conte C, Lapeyre-Mestre M, Hanaire H, Ritz P. Diabetes Remission and Relapse After Bariatric Surgery: a Nationwide Population-Based Study. Obes Surg. 2020;30(12):4810–20. 10.1007/s11695-020-04924-3. Epub 2020 Aug 31 PMID: 32869127. [DOI] [PubMed] [Google Scholar]
- 146.Yu Z, Li P, Li P, Zhang H, Zhang Y. Meta-analysis of Long-Term Relapse Rate of Type 2 Diabetes Following Initial Remission After Roux-en-Y Gastric Bypass. Obes Surg. 2021;31(11):5034–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Jans A, Szabo E, Näslund I, Ottosson J, Näslund E, Stenberg E. Factors affecting relapse of type 2 diabetes after bariatric surgery in Sweden 2007–2015: a registry-based cohort study. Surg Obes Relat Dis. 2022;18(3):305–12. 10.1016/j.soard.2021.12.005. Epub 2021 Dec 8 PMID: 34974997. [DOI] [PubMed] [Google Scholar]
- 148.Ilyas S, Al-Refai R, Maharjan R, Diaz Bustamante L, Ghattas KN, Khan S. Bariatric Surgery and Type 2 Diabetes Mellitus: Assessing Factors Leading to Remission. A Systematic Review Cureus. 2020;12(8): e9973. 10.7759/cureus.9973.PMID:32983676;PMCID:PMC7510520. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
No datasets were generated or analysed during the current study.