Abstract
The age-related loss of muscle mass is partly driven by a reduction in serial sarcomere number (SSN), and further SSN loss occurs during immobilization. Serial sarcomere number is associated with optimal force and power production and muscle passive tension, thus immobilization-induced SSN loss is especially a concern for older individuals who are often subjected to forced muscle disuse with illness and injury. We previously showed that submaximal eccentric resistance training increased SSN and improved muscle function in old rats. The present study investigated whether this training could prevent the losses of SSN and function when performed intermittently during immobilization. Ten old (32 months) and 10 young (8 months) rats underwent unilateral casting of the plantar flexors in a shortened position for 2 weeks. Thrice weekly, casts were removed for isokinetic eccentric resistance training. Pre- and post-training we assessed in-vivo maximum isometric torque at ankle angles corresponding to stretched and neutral muscle lengths, the passive torque–angle relationship, and isotonic power. The soleus and medial gastrocnemius were harvested for SSN measurements, with the untrained leg as a control. In old and young rats, muscles of the casted leg had smaller muscle wet weights (− 20%–40%), physiological cross-sectional area (− 16%–20%), and SSN (− 7%–29%) than the control leg. Furthermore, maximum isometric torque (− 37%–46%) and isotonic power (−≈70%) decreased, and passive torque increased (+≈400%) from pre- to post-training for both age groups. Thus, irrespective of age, submaximal eccentric resistance training 3 days/week was ineffective for preventing the losses of muscle contractile tissue and mechanical function during casting.
Keywords: Aging, Casting, Force–length relationship, Passive force, Power, Sarcomerogenesis
Sarcopenia, the age-related loss of muscle mass, is in part driven by the loss of sarcomeres aligned in series along a muscle (1,2). This age-related loss of serial sarcomere number (SSN) has been shown to be 14%–35% in old compared to young rodents (3–6) and helps explain the 7%–35% reductions in muscle fascicle length (FL) as measured by ultrasound observed in older humans (7–17). Serial sarcomere number is also associated with the width of the plateau region of the muscle force–length relationship (eg, the range of motion in which the muscle can generate optimal force), maximum power production, and passive tension (18), thus the age-related loss of SSN likely contributes to impairments in these muscle mechanical properties with aging. Loss of SSN also occurs when muscle is immobilized in a shortened position (19–22). We recently showed in old rats that with 2 weeks of casting in full plantar flexion (ie, shortened soleus muscle), the soleus loses an additional 25% SSN on top of that already lost with aging (6). Furthermore, old rats took longer to recover SSN than young rats following cast removal (6). The compounded losses of SSN with aging and immobilization are thereby especially a concern for older adult humans, who are prone to falls and injuries that could result in extended periods of forced muscle disuse (23,24).
Previously, Williams et al. (25) showed that 0.5–2 h of passive stretching per day during 2 weeks of casting prevented soleus SSN loss in young adult mice. However, passive stretching for 40 min only once (26) or thrice (27) weekly did not prevent soleus SSN loss during casting in young adult rats, suggesting only long and potentially unfeasible passive stretching interventions can prevent SSN loss. Compared to passive stretching, eccentric resistance training has shown better effectiveness for increasing SSN or FL and improving mechanical performance in both animals and humans (28–31), likely due to the greater mechanical tension and associated mechanotransductive response for muscle growth (18,32). We recently showed that 4 weeks of submaximal (60% of maximum) eccentric training for just ~10 min/day 3 days/week increased SSN of the old rat plantar flexors by 8%–11%, notably restoring SSN to a level similar to that of young rats (33). This increase in SSN was accompanied by an improvement in torque production throughout the joint range of motion, a 23% improvement in peak isotonic power, and a reduction in passive torque (33). Similar eccentric resistance training interventions have also yielded performance improvements in older humans (34–36). However, whether submaximal eccentric training 3 days/week can prevent SSN loss during immobilization in young and old rats is unknown.
The present study assessed whether submaximal eccentric training 3 days/week during 2 weeks of casting can prevent the losses of SSN and muscle mechanical performance in old and young rats. Due to the large benefits of submaximal eccentric training for SSN and muscle mechanical performance in old rats that we observed previously (33), we hypothesized that this training would ameliorate the effects of casting on muscle morphology and mechanical function in both old and young rats.
Method
Animals
Ten old (32 months) and 10 young (8 months) male Fisher 344/Brown Norway F1 rats were obtained (Charles River Laboratories, Senneville, QC, Canada). All protocols were approved by the University of Guelph’s Animal Care Committee (AUP #4905) and followed guidelines from the Canadian Council on Animal Care. Rats were housed at 23 °C in groups of 2 or 3 and given ad-libitum access to a Teklad global 18% protein rodent diet (Envigo, Huntington, Cambridgeshire, UK) and room-temperature water. As described in detail below, rats were subjected to 2 weeks of unilateral immobilization of the plantar flexors in a shortened position. Thrice weekly during the casting immobilization period, casts were removed and the plantar flexors were subjected to eccentric training, after which a new cast was immediately applied. Mechanical testing was performed 2–7 days prior to application of casts, then again 48 h following the final training session, immediately following cast removal. Following the final mechanical testing session, rats were euthanized via isoflurane followed by CO2 asphyxiation and cervical dislocation. The hindlimbs were amputated and fixed in formalin for subsequent determination of soleus and medial gastrocnemius (MG) SSN. In accordance with previous studies (20,37–40), the left leg served as the experimental leg while the right leg served as an internal control.
Data Acquisition During Mechanical Testing and Training
A 701C High-Powered, Bi-Phase Stimulator (Aurora Scientific, Aurora, ON, Canada) was used to evoke transcutaneous muscle stimulation via a custom-made electrode holder with steel galvanized pins situated transversely over the popliteal fossa and the calcaneal tendon (5,6,33). During piloting, we determined this stimulation setup produced similar values of maximum isometric tetanic torque across repeated testing sessions, and produced plantar flexion torque values consistent with values in previous literature using direct nerve stimulation (41). Torque, angle, and stimulus trigger data were sampled at 1 000 Hz with a 605A Dynamic Muscle Data Acquisition and Analysis System (Aurora Scientific, Aurora, ON, Canada) with a live torque tracing during all training and mechanical data collection sessions.
Mechanical Testing
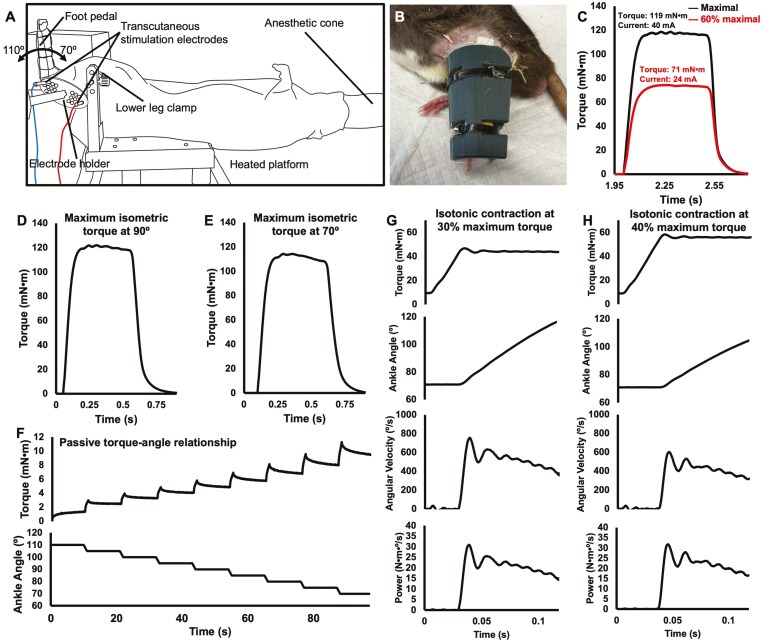
As described previously (5), rats were anesthetized with isoflurane and positioned supine on a heated platform (37 °C; Figure 1A). The left leg was shaved and fixed to a force transducer/length controller foot pedal via tape, with the knee immobilized at 90°. Each mechanical testing session began with determination of the optimal stimulation current for active plantar flexion torque (frequency = 100 Hz, pulse duration = 0.1 ms, train duration = 500 ms) at an ankle angle of 90° (full plantar flexion = 180°; Figure 1D), which was the current used throughout the remainder of the testing session. This stimulation current was confirmed to maximally activate the plantar flexors with minimal spread to the antagonist muscles as described previously (5). A 100 Hz stimulation was then completed at an ankle angle of 70° (Figure 1E). Active torque was measured by subtracting the torque value at baseline (passive torque) immediately prior to stimulation from the total torque during stimulation (42,43). A passive torque–angle relationship was then constructed by recording passive torque following 5s of stress-relaxation at ankle angles of 100°, 95°, 90°, 85°, 80°, 75°, and 70° (Figure 1F). Isotonic (ie, constant torque) contractions were then performed at load clamps equating to 30% and 40% of the maximum isometric torque at 70° (Figure 1G and H), in a randomized order, as we previously determined these loads to represent peak isotonic power for the rat plantar flexors (5,33). Peak angular velocity was recorded as the maximum time derivative of the angular displacement during the isotonic contraction (5), and power was recorded as the torque of the load clamp multiplied by peak angular velocity. Power and angular velocity were averaged between the 30% and 40% isotonic contractions for statistical analyses. Two minutes of rest separated each stimulation to minimize the development of muscle fatigue.
Figure 1.
Experimental setup and representative traces for mechanical testing data from an old rat. A. Experimental setup for mechanical testing and eccentric training. The rat’s left foot was secured to a foot pedal and electrically stimulated to evoke plantar flexion contractions, with the knee fixed at 90°. The ankle had a 70° to 110° range of motion. B. Unilateral cast placing the ankle in full plantar flexion. C. Raw torque traces of a maximum isometric contraction and the corresponding 60% maximum isometric contraction. The stimulation current from the 60% maximum isometric contraction was used for the remainder of that training session during the eccentric contractions. D. Raw torque trace of a maximum isometric contraction at an ankle angle of 90°. E. Raw torque trace of a maximum isometric contraction at an ankle angle of 70°. F. Raw torque and ankle angle traces for construction of the passive torque–angle relationship. Passive torque was recorded immediately prior to each length change. G–H. Raw torque, ankle angle, angular velocity, and power traces for isotonic contractions from 70° to 110° with load clamps set to 30% and 40% of the maximum isometric torque. Peak power was recorded as torque multiplied by the peak angular velocity.
Unilateral Immobilization
Using gauze padding, vet wrap, and a 3D-printed brace and splint, the right hindlimb of each rat was casted in full plantarflexion for 2 weeks (6) (Figure 1B). The toes were left exposed to monitor for swelling (40,44). Casts were inspected daily and repaired/replaced as needed. Since rats were free to walk around while wearing their casts, this intervention merely promoted disuse of the immobilized muscles rather than complete unloading such as that achieved with hindlimb suspension (45).
Isokinetic Eccentric Training
The submaximal eccentric training protocol was replicated directly from our previous study (33), in which we observed 8%–11% increases in SSN and a 23% increase in peak isotonic power in old rats. Training lasted 2 weeks (ie, during the casting intervention) and occurred 3 days/week (Monday, Wednesday, Friday).
At the start of each training session, the stimulus current was first adjusted to produce maximum isometric torque (pulse duration = 0.1 ms, frequency = 100 Hz, train duration = 500 ms) at an ankle angle of 90°. The current was then adjusted to produce an isometric torque that as closely as possible matched 60% of maximum isometric torque (Figure 1C). Maximum isometric torque was recorded for each training session to track changes in muscle strength throughout the training period.
Each eccentric repetition consisted of 3 phases: (a) a 500-ms preactivation at 110°; (b) active lengthening to 70°; and (c) 3 s of deactivation followed by a return to 110° at 20°/s. An additional 3 s of rest were provided before the next repetition. During week 1, rats completed 3 sets of 8 repetitions at 40°/s. In week 2, they completed 3 sets of 9 repetitions at 40°/s. Two minutes of rest were provided between each set.
Serial Sarcomere Number Determination
Following their final mechanical testing session, rats were sacrificed via isoflurane anesthetization followed by CO2 asphyxiation and cervical dislocation. The hindlimbs were amputated, skinned with all muscles overlying the plantar flexors removed, and fixed in 10% phosphate-buffered formalin with the ankle pinned at 90°. After fixation for 1–2 weeks, the soleus and MG were dissected off the lower leg, weighed, then resubmerged in formalin until the commencement of SSN estimations. To commence the process of SSN estimations, the muscles were rinsed with phosphate-buffered saline, then digested in 30% nitric acid for 6–8 h to remove connective tissue and allow for individual muscle fascicles to be teased out (43,46).
For each muscle, 2 fascicles were obtained from each of the proximal, middle, and distal regions (ie, n = 6 fascicles total per muscle). Serial sarcomere number and FL values were averaged across these 6 fascicles for the reporting of data. Dissected fascicles were placed on glass microslides (VWR International, Mississauga, Ontario, Canada), then FLs were measured using ImageJ software (version 1.53f, National Institutes of Health) from pictures captured by a level, tripod-mounted digital camera, with measurements calibrated to a ruler in plane with the fascicles. Sarcomere length (SL) measurements were taken at n = 6 different locations proximal to distal along each fascicle via laser diffraction (Coherent, Santa Clara, CA) with a 5-mW diode laser (~1 mm beam diameter, 635 nm wavelength) and custom LabVIEW program (Version 2011, National Instruments, Austin, TX) (47), for a total of n = 36 SL measurements per muscle. For each fascicle, the 6 SL measurements were averaged to obtain a value of average SL. Given the laser diameter of ~1 mm, 1 SL measurement itself represents an average of hundreds to thousands of SLs. Our total quantity of SL and FL measurements is consistent with previous studies (42,43,46). For each fascicle, the 6 SL measurements were averaged to obtain a value of average SL. Serial sarcomere number of each fascicle was calculated as:
Serial sarcomere number = fascicle length/average sarcomere length
Determination of Physiological Cross-sectional Area
To gain further insight on changes in muscle contractile tissue in parallel, we calculated physiological cross-sectional area (PCSA; in cm2) using the equation (42,48):
Muscle density was assumed to be 1.112 g/cm (3,48). Normalized FL was calculated using FL of dissected fascicles and measured SL in the equation (48):
SLo represents optimal SL of rat muscle at rest, assumed to be ~2.7 μm based on previous literature (42,49). Maximum isometric torque and peak isotonic power normalized to the combined PCSA of the soleus and MG were also recorded, with post-training values normalized to trained muscle PCSA, and pretraining values normalized to untrained muscle PCSA.
Statistical Analyses
All statistical analyses were performed in SPSS Statistics Premium version 28 (IBM SPSS Statistics, Chicago, IL). Normality of data was confirmed using Shapiro–Wilk tests. Differences in SSN, SL, FL, muscle wet weight, and PCSA between the trained and untrained leg were assessed by 2-way repeated measures analysis of variance (ANOVA; Age [young, old] × Cast [uncasted leg, casted leg]). Differences in peak power were also assessed via 2-way repeated measures ANOVA (Age [young, old] × Cast [precast, postcast]). Three-way repeated measures ANOVAs assessed training-induced changes in maximum isometric torque (Age [young, old] × Cast [precast, postcast] × Angle [90°, 70°]) and passive torque (Age [young, old] × Cast [precast, postcast] × Angle [110°–70°]). Three-way ANOVA was used in these cases to allow us to investigate differences in the overall shapes of the active and passive torque–angle relationships. Lastly, to investigate time course changes in maximum isometric torque at 90° across each day of training, we used a 2-way repeated measures ANOVA (Age [young, old] × Training day [precast, training day 1, 2, 3, 4, 5, 6, postcast]). A Greenhouse–Geisser correction for sphericity was applied for all ANOVAs and a Sidak correction was applied to all pairwise comparisons. Significance was set at α = 0.05.
Results
Changes in Muscle Mass and Physiological Cross-sectional Area
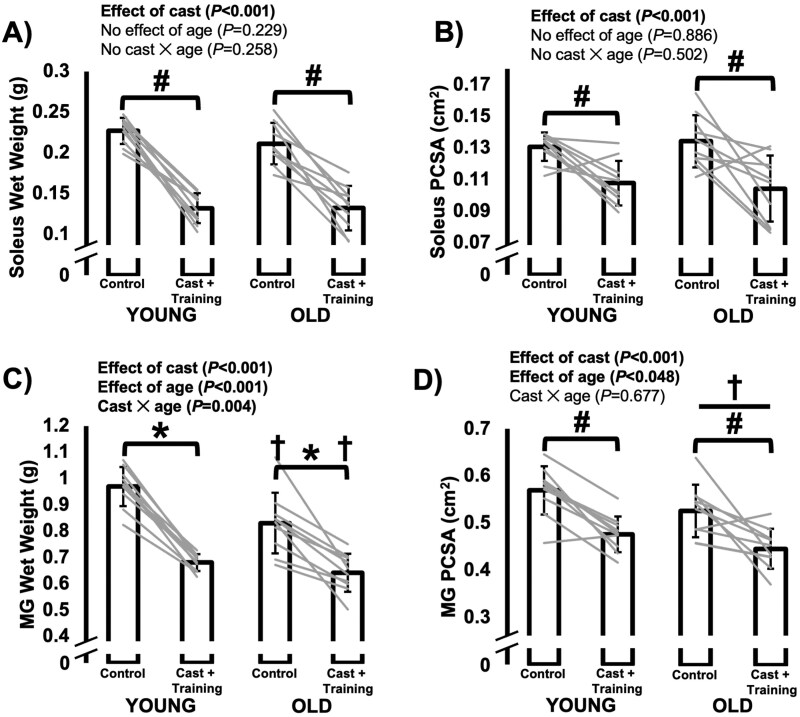
For soleus wet weight, there was an effect of cast (F(1,18) = 154.466, p < .001) but no effect of age (F(1,18) = 1.553, p = .229) such that wet weight was 40% lower in the casted compared to control soleus, with no age-related difference (Figure 2A). Similarly, for soleus PCSA, there was an effect of cast (F(1,18) = 25.204, p < .001) but no effect of age (F(1,18) = 0.021, p = .886) nor an interaction (F(1,18) = 0.470, p = .502). Thus, PCSA of the casted soleus was 20% lower than that of the control soleus, with no age-related difference (Figure 2B).
Figure 2.
Differences in muscle wet weight and physiological cross-sectional area (PCSA) of the soleus (A–B) and medial gastrocnemius (MG; C–D) between the control leg versus the leg that was casted with intermittent submaximal eccentric training, and between old versus young rats. A, B, and D: #Effect of cast with data from young and old rats combined. †Effect of age with data from control and casted combined (p < .05). C. *Difference between control and casted (p < .05). †Difference between young and old rats (p < .05).
For MG wet weight, there was a cast × age interaction (F(1,18) = 10.601, p = .004). Old rats had lower MG wet weight than young in both the control (− 20%, p < .001) and casted leg (− 11%, p = .004). Both age groups exhibited lower MG wet weight in the casted leg than the control leg (young: − 30%, p < .001; old: − 22%, p < .001; Figure 2C). For MG PCSA (Figure 2D) there were effects of cast (F(1,18) = 36.832, p < .001) and age (F(1,18) = 4.511, p = .048) but no interaction (F(1,18) = 0.179, p = .677). Medial gastrocnemius PCSA was 6% smaller in old than young rats and 16% smaller in the casted than the control leg.
Changes in Muscle Fascicle Length Driven by Serial Sarcomere Number Adaptations
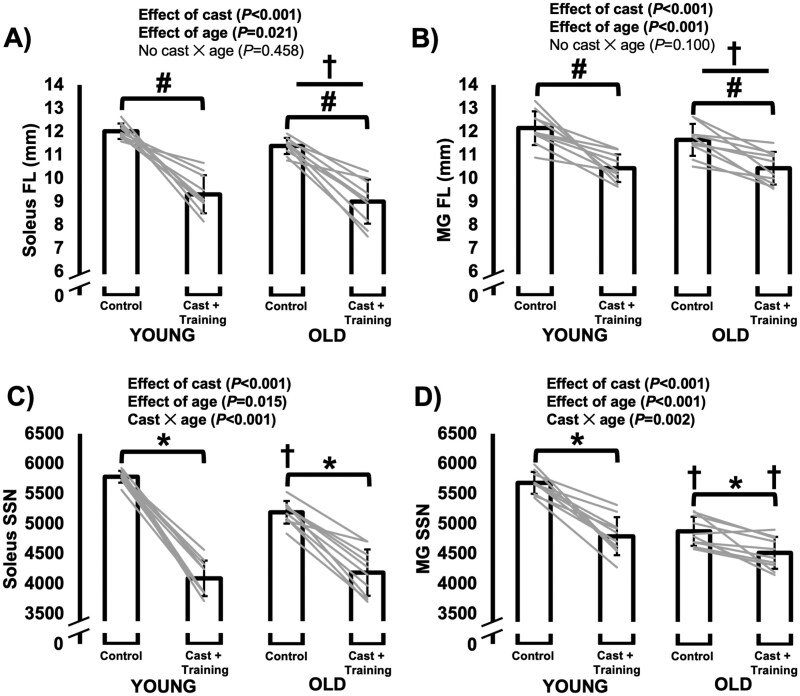
For FL as measured at a 90° ankle angle in both the soleus and MG (Figure 3A and B), there were effects of age (soleus: F(1,18) = 6.351, p = .021; MG: F(1,18) = 19.776, p < .001) and cast (soleus: F(1,18) = 126.945, p < .001; MG: F(1,18) = 54.480, p < .001), but no interaction (soleus: F(1,18) = 0.576, p = .458; MG: F(1,18) = 3.015, p = .100) such that FL was shorter in old than young rats (soleus: −5%; MG: −8%) and shorter in the casted than the control leg (soleus: −22%; MG: −12%).
Figure 3.
Differences in muscle fascicle length (FL) and serial sarcomere number (SSN) of the soleus (A, C) and medial gastrocnemius (MG; B, D) between the control leg versus the leg that was casted with intermittent submaximal eccentric training, and between old versus young rats. A–B: #Effect of cast with data from young and old rats combined. †Effect of age with data from control and casted combined (p < .05). C–D: *Difference between control and casted (p < .05). †Difference between young and old rats (p < .05).
The SSN data showed that the above age-related and cast-related reductions in FL and overstretched SLs (see data in Supplementary Figure 1) were driven by reductions in SSN (Figure 3E and F). For both the soleus and MG, there were cast × age interactions (soleus: F(1,18) = 24.000, p < .001; MG: F(1,18) = 13.524; p = .002). In the soleus, SSN was lower in the casted than the control leg for old (−19%, p < .001) and young rats (−29%, p < .001). The greater magnitude decrease for young rats corresponded to a lower SSN in old than young rats for the control leg (−10%, p < .001) but not the casted leg (p = .544). The MG also exhibited cast-related losses of SSN in both old (−7%, p = .003) and young rats (–16%, p < .001), although at a smaller magnitude than in the soleus. Old rats had a lower MG SSN than young rats in both the control (−14%, p < .001) and casted leg (−6%, p = .048).
Changes in Maximum Isometric Torque as a Function of Joint Angle
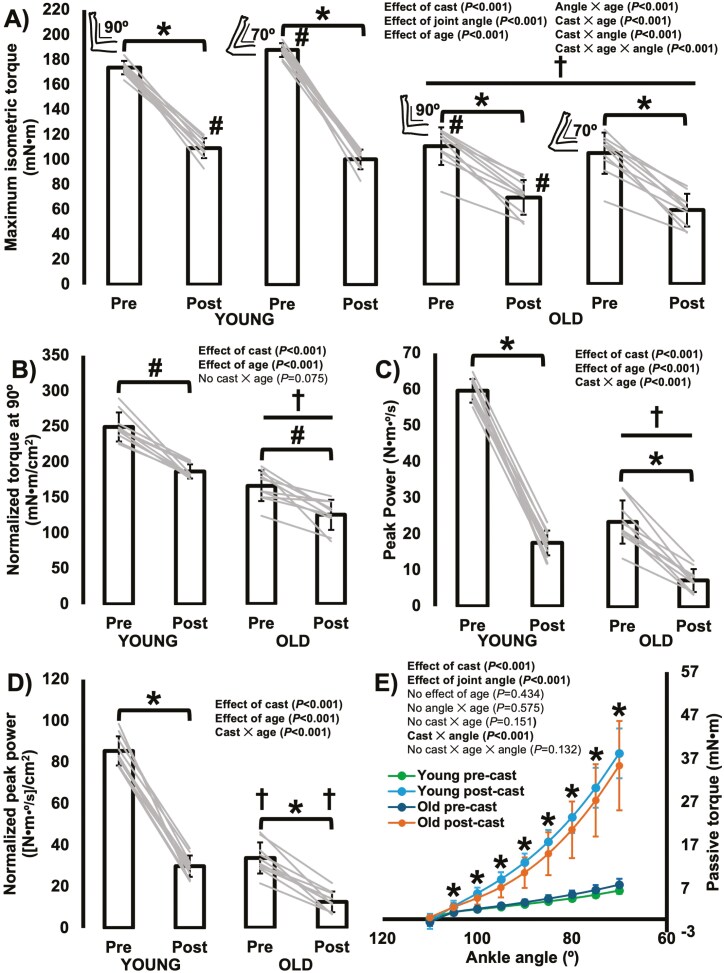
For maximum isometric torque, there was a cast × joint angle × age interaction (F(1,18) = 421.745, p < .001). Old rats produced 36%–44% less isometric torque than young rats at ankle angles corresponding to both neutral (90°) and stretched (70°) muscle lengths pre- and postcast (all p < .001; Figure 4A). Furthermore, both old and young rats experienced decreases in torque at both joint angles from pre- to postcast, with the decreases at 70° noticeably larger in magnitude (old 90°: − 37%; old 70°: − 44%; young 90°: − 37%; young 70°: − 46% all p < .001; Figure 4A). Therefore, intermittent submaximal eccentric training during casting did not prevent the loss of maximum isometric strength, with torque at a more stretched muscle length especially affected. This greater effect on torque at 70° was most noticeable in young rats, because pretraining, torque at 70° was greater than torque at 90° (p < .001), whereas post-training, torque at 90° was instead greater than torque at 70° (p < .001), indicating a leftward shift in the torque–angle relationship (Figure 4A) toward shorter muscle lengths. For old rats, torque was greater at 90° than 70° both pre- and post-training (both p < .001).
Figure 4.
A. Differences in maximum isometric torque as a function of joint angle from pre- to postcast and between young and old rats. *Difference from pre- to postcast (p < .05). †Difference between young and old rats (p < .05). #Difference between ankle angles of 90° and 70° (p < .05). Note that for young rats, 70° (stretched muscle length) was optimal for torque production pretraining but 90° (shorter muscle length) was optimal post-training, and for old rats 90° was optimal both pre- and post-training. B. Differences in maximum isometric torque at 90° normalized to the combined physiological cross-sectional area of the soleus and medial gastrocnemius from pre- to postcast and between young and old rats. #Effect of cast with data from young and old rats combined (p < .05). †Effect of age with data from control and casted combined (p < .05). C. Differences in peak isotonic power from pre- to postcast and between young and old rats. *Difference from pre- to postcast (p < .05). †Difference between young and old rats (p < .05). D. Differences in peak power normalized to the combined physiological cross-sectional area of the soleus and medial gastrocnemius from pre- to postcast and between young and old rats. *Difference from pre- to postcast (p < .05). †Difference between young and old rats (p < .05). E. Differences in passive torque as a function of joint angle from pre- to postcast and between young and old rats. *Difference between pre- and postcast (p < .05) with data from young and old rats pooled, as there was no interaction with age.
After normalizing maximum isometric torque at 90° to the combined PCSA of the soleus and MG, there was still an effect of age (F(1,18) = 133.933, p < .001), with old rats producing 33% lower normalized torque than young (Figure 4B). There was also an effect of cast (F(1,18) = 80.873, p < .001) but no cast × age interaction (F(1,18) = 3.576, p = .075) for normalized torque, with normalized torque decreasing 25% from pre- to postcast. These losses of normalized torque indicate losses of muscle quality both with aging and from pre- to postcast.
Time-course Losses of Maximum Isometric Torque at 90° Throughout the Casting/Intermittent Training Period
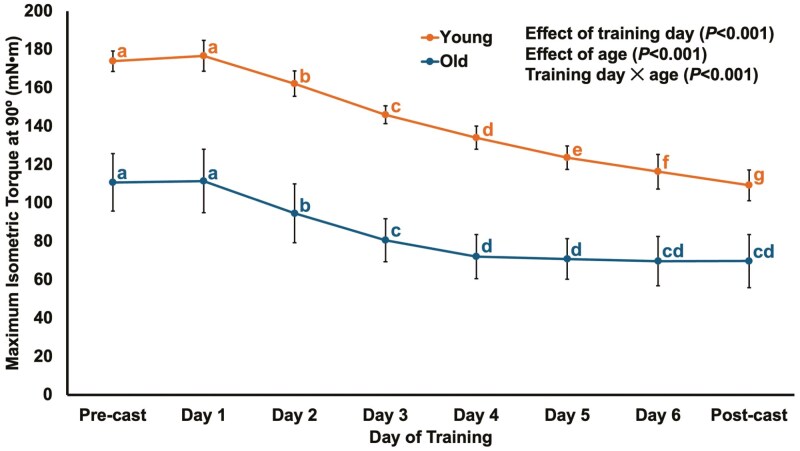
For maximum isometric torque at 90° across the 6 days of intermittent training during casting, there was a training day × age interaction (F(2.24,40.279) = 12.886, p < .001), indicating the time course of torque loss during casting differed between old and young rats. Old rats exhibited torque loss starting on the second day of training (p < .001 compared to precast and day 1 of training), then continued to lose torque up to the fourth day of training (p < .001–.018 compared to all prior days), after which the loss of torque plateaued because torque in the final 3 days of training did not differ compared to each other and compared to postcast (p = .062–1.00; Figure 5). Young rats, conversely, underwent a progressive loss of maximum isometric torque throughout the whole casting period starting after the first day of training, with torque at each subsequent day of training and postcast differing from all other days (p < .001–.002; Figure 5).
Figure 5.
Changes in maximum isometric torque at 90° throughout the 2-week casting with intermittent training period. Training began 2 days after application of casts and occurred 3 days per week (Monday, Wednesday, Friday), amounting to 6 training sessions in total. Casts were removed and postcast testing was completed 2 days following the final training session. Different letters denote a significant difference between time points (p < .05).
Changes in Peak Isotonic Power
For peak isotonic power, there was a cast × age interaction (F(1,18) = 156.35, p < .001). Old rats were 58%–60% less powerful than young rats pre- and post-training (both p < .001; Figure 4C). Old and young rats also both experienced losses of power from pre- to postcast, with a 69% loss in old rats and a 71% loss in young rats (both p < .001; Figure 4C). These losses of power seemed to be due to both the losses of torque and angular velocity, because peak angular velocity during the isotonic contractions also had a cast × age interaction (F(1,18) = 11.885, p = .003) decreasing 47% and 46% from pre- to postcast in old and young rats, respectively (both p < .001). Collectively, intermittent submaximal eccentric training during casting did not prevent the losses of torque, velocity, or power generation, with power undergoing the largest decrease.
After normalizing peak isotonic power to the combined PCSA of the soleus and MG (Figure 4D), there was still a cast × age interaction (F(1,18) = 90.222, p < .001), with lower normalized power in old compared to young rats (pretraining: −59%, p < .001; post-training: −56%, p < .001), and post-training compared to pretraining (young: −65%, p < .001; old: −63%, p < .001). Therefore, the loss of PCSA only accounted for a small portion of the loss of peak power from pre- to postcast.
Changes in Passive Torque as a Function of Joint Angle
For passive torque, there was a cast × joint angle interaction (F(1.03,18.57) = 251.54, p < .001), and no effect of age (F(1,18) = 0.639, p = .434), indicating passive torque changed differently across joint angles from pre- to postcast, but was consistent between old and young rats. As expected, both pre- and postcast, passive torque increased with increasing muscle stretch (all comparisons (p < .001). From pre- to postcast, passive torque underwent increases at all joint angles (all p < .001) other than 110° (p = .645), with the greatest increases of 404% at 70° (ie, the most stretched joint angle tested; Figure 4E). Therefore, intermittent submaximal eccentric training during casting did not prevent the elevation of passive torque that often occurs following casting in a shortened position.
Discussion
Based on our previous observations of serial sarcomerogenesis and improved muscle mechanical performance with submaximal eccentric training in old rats (33), this study aimed to prevent the loss of SSN during a period of immobilization-induced disuse (ie, casting in a shortened muscle position) via intermittent submaximal eccentric training. Following casting, we observed losses of muscle wet weight, SSN, PCSA, and maximal strength and power production in both old and young rats. Thus, submaximal eccentric training 3 days/week did not counteract the effects of casting, and this was not age dependent.
Influence of Aging- and Casting-induced Changes in Muscle Morphology on Mechanical Function
Consistent with previous reports, old rats exhibited smaller muscle wet weights (41,50–52) and shorter FLs driven by a reduced SSN (3–6) compared to young rats. This loss of SSN likely contributed to old rats producing optimal torque at a more shortened muscle length (90° ankle angle) than young rats (70° ankle angle; Figure 4A), and producing lower peak isotonic power (Figure 4C) due to reduced torque production throughout the range of motion (18). These observations align with our previous work (5,6,33) and studies from other groups as well (41,51,53). Notably, there was only an age-related loss of PCSA in the MG and not the soleus. Our previous study also observed no age-related loss of soleus PCSA (6), and given the weaker torque-production capacity in the old compared to young rats, indicates reduced muscle quality (54,55). Reduced muscle quality underscores age-related changes to the intrinsic contractile machinery such as reduced myosin protein content or oxidation of myosin, which reduces crossbridge formation and force production (56,57).
Following 2 weeks of casting with intermittent submaximal eccentric training, we observed losses of muscle wet weight in the soleus and MG, which seemed to be due to losses of contractile tissue both in series (SSN) and in parallel (PCSA; Figures 2 and 3). These muscle morphological changes likely drove the losses of maximum isometric torque and peak isotonic power (Figure 4). The losses of SSN, PCSA, and muscle wet weight were greater for the soleus than the MG, likely because our casts only fully immobilized the ankle while not limiting mobility at the knee. The MG crosses both the knee and ankle whereas the soleus only crosses the ankle, therefore, there was likely some loading of the MG at the knee that allowed better maintenance of MG morphology compared to the soleus.
In young rats, there was also a shift in the optimal angle for torque production from a more stretched muscle length pretraining to a more neutral muscle length post-training (Figure 4A), which is consistent with the loss of SSN due to shifting of the optimal muscle length for actin-myosin overlap (6,20,38,58). As power is the dot product of force and velocity, the loss of torque generating capacity likely contributed to the loss of peak isotonic power. However, since normalizing power to PCSA only slightly reduced the cast-related power loss (from ~70% to ~65%), other factors besides the loss of force-production capacity could have contributed, notably 2 main factors. First, the loss of muscle quality (ie, maximum torque normalized to PCSA; Figure 4B) could have contributed by reducing intrinsic crossbridge function. Second, the loss of SSN could have contributed to power loss by narrowing the torque–angle relationship’s plateau region and reducing torque-production capacity throughout the joint range of motion (18). These findings highlight the functional implications of SSN loss and the importance of SSN maintenance throughout the lifespan for muscle mechanical performance.
It is important to note that, since our mechanical testing system is optimized for testing only the left plantar flexors, we did not perform mechanical function measurements on the uncasted (right) plantar flexors. Therefore, any potential compensatory adaptations in the uncasted leg were not accounted for. With that said, using ultrasound we previously observed no changes in morphology of the uncasted soleus during 2 weeks of casting, as well as no differences between the right and left soleus at precast (40). Furthermore, in the present study, the loss of maximal torque production in the casted leg (−40%) was similar to the loss of muscle wet weight between the uncasted and casted leg (−40% in the soleus and −20% in the MG). Altogether, adaptations in the uncasted leg were likely minimal or absent in the present study.
No Benefit of Casting With Intermittent Submaximal Eccentric Training Compared to Casting Alone
Many of the changes from pre- to postcast we observed here are similar in magnitude to those observed in our previous study following 2 weeks of casting without intermittent training (6), as displayed in Supplementary Figure 2. In fact, it appears that if anything, intermittent submaximal eccentric training during casting was more detrimental than casting alone, as maximum passive torque increased ~3-fold more (+ 343%–478% vs + 68%–180%) and muscle wet weight decreased more (−37% to 42% vs −23% to 25%) with intermittent eccentric training during casting (Supplementary Figure 2). Thus, not only did intermittent submaximal eccentric training not prevent the effects of casting in a shortened position, the 2 together may have had a compounded negative effect on muscle function. While histological assessment of the extracellular matrix was beyond the scope of the present study, the greater increase in passive torque compared to our previous study may be indicative of greater fibrous connective tissue deposition (59–61). It is possible that overstretched sarcomeres due to the loss of SSN following casting could have contributed to the elevated passive torque, however, we only observed a casting-induced increase in SL as measured at 90° in young rats, with no change in old rats (Supplementary Figure 1). Furthermore, a similar increase in SL was also observed in our previous study of casting alone (6) alongside the smaller casting-induced increase in passive torque. Thus, it seems more likely that changes in extracellular matrix morphology contributed to the elevated passive torque here.
While speculative, it is possible some muscle damage was incurred during the eccentric contractions that led to a worsened effect compared to casting alone. Shorter muscles (ie, with fewer sarcomeres in series) can undergo more damage during eccentric contractions for a given magnitude of joint rotation because the stretch imposed on individual sarcomeres would be greater (62,63). Hence, it is possible that as the muscle lost SSN from casting in a shortened position, it became increasingly more susceptible to eccentric contraction-induced damage. In support of this theory, previous studies have shown that the effects of eccentric-induced muscle damage are amplified if preceded by immobilization (64), and that immobilization immediately following eccentric exercise may also prevent recovery from muscle damage (65). Further work is needed to elucidate the role of muscle damage in intermittent submaximal eccentric training during casting.
Comparison to Other Intermittent Training Interventions Employed During Immobilization
We can also look to previous studies that employed intermittent passive stretching during casting to gain insight into why our intermittent eccentric training did not prevent the deleterious effects of casting on muscle. Williams (25) found that exposing the mouse soleus to 30 min to 2 h/day of passive stretching prevented the loss of SSN during casting in a shortened position. They even observed a 10% increase in soleus SSN compared to controls in the legs stretched for 2 h/day during casting. Passive stretch training alone (ie, in nonimmobilized muscle), however, does not always result in increased SSN depending on the duration and intensity (29,30,66,67). It seems in general that shorter duration or less frequent passive stretch training has minimal success for increasing SSN. It follows that Gomes et al. (26) and Coutinho et al. (27) did not prevent SSN loss during casting with intermittent passive stretch training 1 or 3 days/week, respectively in rats. There was reason to believe that intermittent submaximal eccentric training 3 days/week would have been more successful than passive stretch training at preventing SSN loss during casting. While eccentric training interventions have also shown varying levels of success for increasing SSN (5,42,43,46,68,69), the most robust increase in SSN following eccentric training was in our previous study, with 8%–11% increases in soleus and MG SSN following 4 weeks of submaximal isokinetic eccentric training 3 days/week in old rats (33). Furthermore, Van Dyke et al. (70) found that stretch and contraction combined prevented the loss of SSN following tenotomy while passive stretching did not. Collectively, one might expect the conditions of muscle stretch and activation provided by eccentric training to have better success than passive stretch training for preventing SSN loss during casting. However, similar to the findings of Coutinho et al. (27) with passive stretch training 3 days/week, it is possible that eccentric training for ~10 min/day 3 days/week in the present study was not enough to be the dominant stimulus imposed on the muscle compared to casting in a shortened position.
Given the use of transcutaneous electrical stimulation for the present study’s submaximal eccentric training, comparisons can also be drawn to studies that employed “neuromuscular electrical stimulation” (NMES) to counteract the effects of immobilization on skeletal muscle. In NMES trials in humans, transcutaneous electrical stimulation is typically applied to the muscle as isometric contractions in 30- to 60-min sessions once or twice daily, using a stimulation intensity set by the tolerance of the patient (71–76). Neuromuscular electrical stimulation is well-recognized for preventing the losses of myofibrillar protein synthesis and whole-muscle and myofiber cross-sectional area during immobilization, but is less successful at limiting the loss of muscle strength (77). Similar isometric electrical stimulation interventions in rats have also prevented myofiber atrophy and fibrosis during immobilization (78–80). These studies on rats used 15–20 min of tetanic electrical stimulation as few as 3 days/week at a stimulation intensity of up to 60% of the maximum torque development. These training parameters are similar to what was employed in the present study, other than that the present study used eccentric rather than isometric contractions, suggesting tentatively that isometric contractions may be more effective than eccentric contractions during immobilization.
To argue against the above perspective on isometric contractions, however, studies of conventional resistance training (ie, concentric and eccentric contractions together) during bed rest in humans have also shown success for limiting losses of muscle function. Both plantar flexor (81) and leg press (82) resistance training every other day during 2 weeks of bed rest at up to 85% of the 1-repetition maximum prevented losses of 1-repetition maximum weight, concentric and eccentric strength, power production, and myofiber cross-sectional area. Even more impressive, squat and calf press resistance training every 3 days during 90 days of bed rest preserved losses of muscle volume and force and power production (83). Eccentric training of the nonimmobilized limb has also been shown to abolish declines in muscle strength and size during immobilization in humans (84), although these are primarily due to cross-education effects from neural adaptations and thus have limited applicability to the electrically evoked (ie, involuntary) contractions in the present study. Considering the present study’s profound negative effects following casting with intermittent eccentric training, future studies are needed to elucidate optimal training stimuli during forced muscle disuse, especially for older individuals. One potential future direction could be to explore molecular processes associated with mechanosensing proteins in regulating serial sarcomerogenesis (eg, titin, focal adhesion kinase) (2) as potential targets for a better preservation of SSN with intermittent training during casting.
Conclusion
Older adult humans are prone to falls and injuries that could result in extended periods of hospitalization, bed rest and muscle disuse, which can result in the loss of SSN on top of that already lost with aging. Thus, it is prudent to discover interventions to prevent the losses of SSN and associated aspects of mechanical function during immobilization in aged muscle. We found here that intermittent submaximal eccentric training during immobilization provided no positive benefits to muscle morphology or function. Future studies should explore modifications of this intervention to better optimize the ability to maintain SSN and performance during forced muscle disuse.
Supplementary Material
Acknowledgments
This project was supported by the Natural Sciences and Engineering Research Council of Canada (NSERC). The animals were obtained from the National Institute on Aging (NIA) aged rodent colonies.
Contributor Information
Ethan Vlemmix, Department of Human Health and Nutritional Sciences, College of Biological Sciences, University of Guelph, Guelph, Ontario, Canada.
Geoffrey A Power, Department of Human Health and Nutritional Sciences, College of Biological Sciences, University of Guelph, Guelph, Ontario, Canada.
Gustavo Duque, (Biological Sciences Section).
Funding
This project was supported by the Natural Sciences and Engineering Research Council of Canada (NSERC), grant number RGPIN-2024-03782.
Conflict of Interest
None.
Data Availability
All data generated or analyzed during the study are available from the corresponding author upon request.
Author Contributions
A.H. and G.A.P. conceived and designed research; A.H. and E.V. performed experiments; A.H. analyzed data; A.H., E.V., and G.A.P. interpreted results of experiments; A.H. prepared figures; A.H. and G.A.P. drafted manuscript; A.H., E.V., and G.A.P. edited and revised manuscript; A.H., E.V., and G.A.P. approved final version of manuscript.
Ethics Statement
Approval was given by the University of Guelph’s Animal Care Committee and all protocols followed CCAC guidelines (AUP #4905).
References
- 1. Narici MV, Maffulli N.. Sarcopenia: characteristics, mechanisms and functional significance. Br Med Bull. 2010;95(1):139–159. https://doi.org/ 10.1093/bmb/ldq008 [DOI] [PubMed] [Google Scholar]
- 2. Hinks A, Hawke TJ, Franchi MV, Power GA.. The importance of serial sarcomere addition for muscle function and the impact of aging. J Appl Physiol (Bethesda, Md.: 1985). 2023;135(2):375–393. https://doi.org/ 10.1152/japplphysiol.00205.2023 [DOI] [PubMed] [Google Scholar]
- 3. Hooper AC. Length, diameter and number of ageing skeletal muscle fibres. Gerontology. 1981;27(3):121–126. https://doi.org/ 10.1159/000212459 [DOI] [PubMed] [Google Scholar]
- 4. Power GA, Crooks S, Fletcher JR, Macintosh BR, Herzog W.. Age-related reductions in the number of serial sarcomeres contribute to shorter fascicle lengths but not elevated passive tension. J Exp Biol. 2021;224(10):jeb242172. https://doi.org/ 10.1242/jeb.242172 [DOI] [PubMed] [Google Scholar]
- 5. Hinks A, Patterson MA, Njai BS, Power GA.. Age-related blunting of serial sarcomerogenesis and mechanical adaptations following 4 weeks of maximal eccentric resistance training. J Appl Physiol. 2024;136(5):1209–1225. https://doi.org/ 10.1152/japplphysiol.00041.2024 [DOI] [PubMed] [Google Scholar]
- 6. Hinks A, Power GA.. Age-related differences in the loss and recovery of serial sarcomere number following disuse atrophy in rats. Skelet Muscle. 2024;14(1):18. https://doi.org/ 10.1186/s13395-024-00351-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Kubo K, Kanehisa H, Azuma K, et al. Muscle architectural characteristics in young and elderly men and women. Int J Sports Med. 2003;24(2):125–130. https://doi.org/ 10.1055/s-2003-38204 [DOI] [PubMed] [Google Scholar]
- 8. Morse CI, Thom JM, Birch KM, Narici MV.. Changes in triceps surae muscle architecture with sarcopenia. Acta Physiol Scand. 2005;183(3):291–298. https://doi.org/ 10.1111/j.1365-201X.2004.01404.x [DOI] [PubMed] [Google Scholar]
- 9. Thom JM, Morse CI, Birch KM, Narici MV.. Influence of muscle architecture on the torque and power–velocity characteristics of young and elderly men. Eur J Appl Physiol. 2007;100(5):613–619. https://doi.org/ 10.1007/s00421-007-0481-0 [DOI] [PubMed] [Google Scholar]
- 10. Baudry S, Lecoeuvre G, Duchateau J.. Age-related changes in the behavior of the muscle-tendon unit of the gastrocnemius medialis during upright stance. J Appl Physiol (Bethesda, Md.: 1985). 2012;112(2):296–304. https://doi.org/ 10.1152/japplphysiol.00913.2011 [DOI] [PubMed] [Google Scholar]
- 11. Stenroth L, Peltonen J, Cronin NJ, Sipilä S, Finni T.. Age-related differences in Achilles tendon properties and triceps surae muscle architecture in vivo. J Appl Physiol (Bethesda, Md.: 1985). 2012;113(10):1537–1544. https://doi.org/ 10.1152/japplphysiol.00782.2012 [DOI] [PubMed] [Google Scholar]
- 12. Power GA, Makrakos DP, Rice CL, Vandervoort AA.. Enhanced force production in old age is not a far stretch: an investigation of residual force enhancement and muscle architecture. Physiol Rep. 2013;1(1):e00004. https://doi.org/ 10.1002/phy2.4 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Panizzolo FA, Green DJ, Lloyd DG, Maiorana AJ, Rubenson J.. Soleus fascicle length changes are conserved between young and old adults at their preferred walking speed. Gait Posture. 2013;38(4):764–769. https://doi.org/ 10.1016/j.gaitpost.2013.03.021 [DOI] [PubMed] [Google Scholar]
- 14. Baroni BM, Geremia JM, Rodrigues R, et al. Functional and morphological adaptations to aging in knee extensor muscles of physically active men. J Appl Biomech. 2013;29(5):535–542. https://doi.org/ 10.1123/jab.29.5.535 [DOI] [PubMed] [Google Scholar]
- 15. Wu R, Delahunt E, Ditroilo M, Lowery M, De Vito G.. Effects of age and sex on neuromuscular-mechanical determinants of muscle strength. Age (Dordrecht, Netherlands). 2016;38(3):57. https://doi.org/ 10.1007/s11357-016-9921-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16. Conway KA, Franz JR.. Shorter gastrocnemius fascicle lengths in older adults associate with worse capacity to enhance push-off intensity in walking. Gait Posture. 2020;77:89–94. https://doi.org/ 10.1016/j.gaitpost.2020.01.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Kelp NY, Gore A, Clemente CJ, Tucker K, Hug F, Dick TJM.. Muscle architecture and shape changes in the gastrocnemii of active younger and older adults. J Biomech. 2021;129:110823. https://doi.org/ 10.1016/j.jbiomech.2021.110823 [DOI] [PubMed] [Google Scholar]
- 18. Hinks A, Franchi MV, Power GA.. The influence of longitudinal muscle fascicle growth on mechanical function. J Appl Physiol (Bethesda, Md.: 1985). 2022;133(1):87–103. https://doi.org/ 10.1152/japplphysiol.00114.2022 [DOI] [PubMed] [Google Scholar]
- 19. Tabary JC, Tabary C, Tardieu C, Tardieu G, Goldspink G.. Physiological and structural changes in the cat’s soleus muscle due to immobilization at different lengths by plaster casts. J Physiol. 1972;224(1):231–244. https://doi.org/ 10.1113/jphysiol.1972.sp009891 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20. Williams PE, Goldspink G.. Changes in sarcomere length and physiological properties in immobilized muscle. J Anat. 1978;127(3):459–468. [PMC free article] [PubMed] [Google Scholar]
- 21. Spector SA, Simard CP, Fournier M, Sternlicht E, Edgerton’ VR.. Architectural alterations of rat hind-limb skeletal muscles immobilized at different lengths. Exp Neurol. 1982;76:94–110. [DOI] [PubMed] [Google Scholar]
- 22. Witzmann FA, Kim DH, Fitts RH.. Hindlimb immobilization: length-tension and contractile properties of skeletal muscle. J Appl Physiol Respir Environ Exerc Physiol. 1982;53(2):335–345. https://doi.org/ 10.1152/jappl.1982.53.2.335 [DOI] [PubMed] [Google Scholar]
- 23. Surkan MJ, Gibson W.. Interventions to mobilize elderly patients and reduce length of hospital stay. Can J Cardiol. 2018;34(7):881–888. https://doi.org/ 10.1016/j.cjca.2018.04.033 [DOI] [PubMed] [Google Scholar]
- 24. Valenzuela PL, Morales JS, Pareja-Galeano H, et al. Physical strategies to prevent disuse-induced functional decline in the elderly. Ageing Res Rev. 2018;47:80–88. https://doi.org/ 10.1016/j.arr.2018.07.003 [DOI] [PubMed] [Google Scholar]
- 25. Williams PE. Use of intermittent stretch in the prevention of serial sarcomere loss in immobilised muscle. Ann Rheum Dis. 1990;49(5):316–317. https://doi.org/ 10.1136/ard.49.5.316 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26. Gomes ARS, Coutinho EL, França CN, Polonio J, Salvini TF.. Effect of one stretch a week applied to the immobilized soleus muscle on rat muscle fiber morphology. Braz J Med Biol Res. 2004;37(10):1473–1480. https://doi.org/ 10.1590/s0100-879x2004001000005 [DOI] [PubMed] [Google Scholar]
- 27. Coutinho EL, Gomes ARS, França CN, Oishi J, Salvini TF.. Effect of passive stretching on the immobilized soleus muscle fiber morphology. Braz J Med Biol Res. 2004;37(12):1853–1861. https://doi.org/ 10.1590/s0100-879x2004001200011 [DOI] [PubMed] [Google Scholar]
- 28. Konrad A, Tilp M.. Increased range of motion after static stretching is not due to changes in muscle and tendon structures. Clin Biomech (Bristol, Avon). 2014;29(6):636–642. https://doi.org/ 10.1016/j.clinbiomech.2014.04.013 [DOI] [PubMed] [Google Scholar]
- 29. De Jaeger D, Joumaa V, Herzog W.. Intermittent stretch training of rabbit plantarflexor muscles increases soleus mass and serial sarcomere number. J Appl Physiol (Bethesda, Md.: 1985). 2015;118(12):1467–1473. https://doi.org/ 10.1152/japplphysiol.00515.2014 [DOI] [PubMed] [Google Scholar]
- 30. Peviani SM, Guzzoni V, Pinheiro-Dardis CM, et al. Regulation of extracellular matrix elements and sarcomerogenesis in response to different periods of passive stretching in the soleus muscle of rats. Sci Rep. 2018;8:9010. https://doi.org/ 10.1038/s41598-018-27239-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Kruse A, Rivares C, Weide G, Tilp M, Jaspers R.. Stimuli for adaptations in muscle length and the length range of active force exertion—a narrative review. Front Physiol. 2021;12:742034. https://doi.org/ 10.3389/fphys.2021.742034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Franchi MV, Reeves ND, Narici MV.. Skeletal muscle remodeling in response to eccentric vs. concentric loading: morphological, molecular, and metabolic adaptations. Front Physiol. 2017;8:447. https://doi.org/ 10.3389/fphys.2017.00447 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Hinks AA, Vlemmix E, Power GA.. Submaximal eccentric resistance training increases serial sarcomere number and improves dynamic muscle performance in old rats. Physiol Rep. 2024;12(19):e70036. https://doi.org/ 10.14814/phy2.70036 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Reeves ND, Maganaris CN, Longo S, Narici MV.. Differential adaptations to eccentric versus conventional resistance training in older humans: eccentric resistance training in old age. Exp Physiol. 2009;94(7):825–833. https://doi.org/ 10.1113/expphysiol.2009.046599 [DOI] [PubMed] [Google Scholar]
- 35. Quinlan JI, Franchi MV, Gharahdaghi N, et al. Muscle and tendon adaptations to moderate load eccentric vs. concentric resistance exercise in young and older males. GeroScience. 2021;43(4):1567–1584. https://doi.org/ 10.1007/s11357-021-00396-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36. Baxter BA, Baross AW, Ryan DJ, Tkadlec S, Kay AD.. Effects of once- versus twice-weekly eccentric resistance training on muscular function and structure in older adults: a randomised controlled trial. Sci Rep. 2024;14(1):9638. https://doi.org/ 10.1038/s41598-024-59788-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. de la Tour EH, Tabary JC, Tabary C, Tardieu C.. The respective roles of muscle length and muscle tension in sarcomere number adaptation of guinea-pig soleus muscle. J Physiol (Paris). 1979;75(5):589–592. [PubMed] [Google Scholar]
- 38. Heslinga JW, Huijing PA.. Muscle length–force characteristics in relation to muscle architecture: a bilateral study of gastrocnemius medialis muscles of unilaterally immobilized rats. Eur J Appl Physiol 1993;66(4):289–298. https://doi.org/ 10.1007/BF00237771 [DOI] [PubMed] [Google Scholar]
- 39. Kinney MC, Dayanidhi S, Dykstra PB, McCarthy JJ, Peterson CA, Lieber RL.. Reduced skeletal muscle satellite cell number alters muscle morphology after chronic stretch but allows limited serial sarcomere addition: satellite cells and sarcomere addition. Muscle Nerve. 2017;55(3):384–392. https://doi.org/ 10.1002/mus.25227 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. Hinks A, Franchi MV, Power GA.. Ultrasonographic measurements of fascicle length overestimate adaptations in serial sarcomere number. Exp Physiol. 2023;108(10):1308–1324. https://doi.org/ 10.1113/EP091334 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Padilla CJ, Harrigan ME, Harris H, et al. Profiling age-related muscle weakness and wasting: neuromuscular junction transmission as a driver of age-related physical decline. GeroScience. 2021;43(3):1265–1281. https://doi.org/ 10.1007/s11357-021-00369-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42. Chen J, Mashouri P, Fontyn S, et al. The influence of training-induced sarcomerogenesis on the history dependence of force. J Exp Biol. 2020;223(Pt 15):jeb218776. https://doi.org/ 10.1242/jeb.218776 [DOI] [PubMed] [Google Scholar]
- 43. Hinks A, Jacob K, Mashouri P, et al. Influence of weighted downhill running training on serial sarcomere number and work loop performance in the rat soleus. Biol Open. 2022;11(7):bio059491. https://doi.org/ 10.1242/bio.059491 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44. Aoki MS, Soares AG, Miyabara EH, Baptista IL, Moriscot AS.. Expression of genes related to myostatin signaling during rat skeletal muscle longitudinal growth: myostatin and longitudinal growth. Muscle Nerve. 2009;40(6):992–999. https://doi.org/ 10.1002/mus.21426 [DOI] [PubMed] [Google Scholar]
- 45. Warren GL, Stallone JL, Allen MR, Bloomfield SA.. Functional recovery of the plantarflexor muscle group after hindlimb unloading in the rat. Eur J Appl Physiol. 2004;93(1–2):130–138. https://doi.org/ 10.1007/s00421-004-1185-3 [DOI] [PubMed] [Google Scholar]
- 46. Butterfield TA, Leonard TR, Herzog W.. Differential serial sarcomere number adaptations in knee extensor muscles of rats is contraction type dependent. J Appl Physiol. 2005;99:1352–1358. https://doi.org/ 10.1152/japplphysiol.00481.2005 [DOI] [PubMed] [Google Scholar]
- 47. Lieber RL, Yeh Y, Baskin RJ.. Sarcomere length determination using laser diffraction. effect of beam and fiber diameter. Biophys J. 1984;45(5):1007–1016. https://doi.org/ 10.1016/S0006-3495(84)84246-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Ward SR, Lieber RL.. Density and hydration of fresh and fixed human skeletal muscle. J Biomech. 2005;38(11):2317–2320. https://doi.org/ 10.1016/j.jbiomech.2004.10.001 [DOI] [PubMed] [Google Scholar]
- 49. Zuurbier CJ, Heslinga JW, Lee-de Groot MB, Van der Laarse WJ.. Mean sarcomere length–force relationship of rat muscle fibre bundles. J Biomech. 1995;28(1):83–87. https://doi.org/ 10.1016/0021-9290(95)80009-3 [DOI] [PubMed] [Google Scholar]
- 50. Leeuwenburgh C, Gurley CM, Strotman BA, Dupont-Versteegden EE.. Age-related differences in apoptosis with disuse atrophy in soleus muscle. Am J Physiol Regul Integr Comp Physiol. 2005;288(5):R1288–R1296. https://doi.org/ 10.1152/ajpregu.00576.2004 [DOI] [PubMed] [Google Scholar]
- 51. Rader EP, Layner K, Triscuit AM, Chetlin RD, Ensey J, Baker BA.. Age-dependent muscle adaptation after chronic stretch-shortening contractions in rats. Aging Dis. 2016;7(1):1–13. https://doi.org/ 10.14336/AD.2015.0920 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Fuqua JD, Lawrence MM, Hettinger ZR, et al. Impaired proteostatic mechanisms other than decreased protein synthesis limit old skeletal muscle recovery after disuse atrophy. J Cachexia Sarcopenia Muscle. 2023;14(5):2076–2089. https://doi.org/ 10.1002/jcsm.13285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. McBride TA, Gorin FA, Carlsen RC.. Prolonged recovery and reduced adaptation in aged rat muscle following eccentric exercise. Mech Ageing Dev. 1995;83(3):185–200. https://doi.org/ 10.1016/0047-6374(95)01629-e [DOI] [PubMed] [Google Scholar]
- 54. Frontera WR, Suh D, Krivickas LS, Hughes VA, Goldstein R, Roubenoff R.. Skeletal muscle fiber quality in older men and women. Am J Physiol Cell Physiol. 2000;279(3):C611–C618. https://doi.org/ 10.1152/ajpcell.2000.279.3.C611 [DOI] [PubMed] [Google Scholar]
- 55. Lim JY, Frontera WR.. Single skeletal muscle fiber mechanical properties: a muscle quality biomarker of human aging. Eur J Appl Physiol. 2022;122(6):1383–1395. https://doi.org/ 10.1007/s00421-022-04924-4 [DOI] [PubMed] [Google Scholar]
- 56. D’Antona G, Pellegrino MA, Adami R, et al. The effect of ageing and immobilization on structure and function of human skeletal muscle fibres. J Physiol. 2003;552(Pt 2):499–511. https://doi.org/ 10.1113/jphysiol.2003.046276 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57. Moen RJ, Klein JC, Thomas DD.. Electron paramagnetic resonance resolves effects of oxidative stress on muscle proteins. Exerc Sport Sci Rev. 2014;42(1):30–36. https://doi.org/ 10.1249/JES.0000000000000004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Woittiez RD, Huijing PA, Rozendal RH.. Influence of muscle architecture on the length-force diagram of mammalian muscle. Pflügers Arch Eur J Physiol. 1983;399(4):275–279. https://doi.org/ 10.1007/BF00652752 [DOI] [PubMed] [Google Scholar]
- 59. Brashear SE, Wohlgemuth RP, Gonzalez G, Smith LR.. Passive stiffness of fibrotic skeletal muscle in mdx mice relates to collagen architecture. J Physiol. 2020;599:943–962. https://doi.org/ 10.1113/JP280656. Online ahead of print [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Brashear SE, Wohlgemuth RP, Hu LY, Jbeily EH, Christiansen BA, Smith LR.. Collagen cross-links scale with passive stiffness in dystrophic mouse muscles, but are not altered with administration of a lysyl oxidase inhibitor. PLoS One. 2022;17(10):e0271776. https://doi.org/ 10.1371/journal.pone.0271776 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Wohlgemuth RP, Brashear SE, Smith LR.. Alignment, cross-linking, and beyond: a collagen architect’s guide to the skeletal muscle extracellular matrix. Am J Physiol Cell Physiol. 2023;325:C1017–C1030. https://doi.org/ 10.1152/ajpcell.00287.2023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Lieber RL, Fridén J.. Muscle damage is not a function of muscle force but active muscle strain. J Appl Physiol (1985). 1993;74(2):520–526. https://doi.org/ 10.1152/jappl.1993.74.2.520 [DOI] [PubMed] [Google Scholar]
- 63. Hyldahl RD, Chen TC, Nosaka K.. Mechanisms and mediators of the skeletal muscle repeated bout effect. Exerc Sport Sci Rev. 2017;45(1):24–33. https://doi.org/ 10.1249/JES.0000000000000095 [DOI] [PubMed] [Google Scholar]
- 64. Chen TCC, Kang HY, Tseng WC, et al. Muscle damage induced by maximal eccentric exercise of the elbow flexors after 3-week immobilization. Scand J Med Sci Sports. 2023;33(4):382–392. https://doi.org/ 10.1111/sms.14279 [DOI] [PubMed] [Google Scholar]
- 65. Jameson TSO, Kilroe SP, Fulford J, et al. Muscle damaging eccentric exercise attenuates disuse-induced declines in daily myofibrillar protein synthesis and transiently prevents muscle atrophy in healthy men. Am J Physiol Endocrinol Metab. 2021;321(5):E674–E688. https://doi.org/ 10.1152/ajpendo.00294.2021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Peixinho CC, Martins NSF, de Oliveira LF, Machado JC.. Structural adaptations of rat lateral gastrocnemius muscle–tendon complex to a chronic stretching program and their quantification based on ultrasound biomicroscopy and optical microscopic images. Clin Biomech. 2014;29(1):57–62. https://doi.org/ 10.1016/j.clinbiomech.2013.11.002 [DOI] [PubMed] [Google Scholar]
- 67. Zotz TG, Capriglione LGA, Zotz R, et al. Acute effects of stretching exercise on the soleus muscle of female aged rats. Acta Histochem. 2016;118(1):1–9. https://doi.org/ 10.1016/j.acthis.2015.10.004 [DOI] [PubMed] [Google Scholar]
- 68. Koh TJ, Herzog W.. Eccentric training does not increase sarcomere number in rabbit dorsiflexor muscles. J Biomech. 1998;31(5):499–501. https://doi.org/ 10.1016/s0021-9290(98)00032-3 [DOI] [PubMed] [Google Scholar]
- 69. Butterfield TA, Herzog W.. The magnitude of muscle strain does not influence serial sarcomere number adaptations following eccentric exercise. Pflüg Arch—Eur J Physiol. 2006;451(5):688–700. https://doi.org/ 10.1007/s00424-005-1503-6 [DOI] [PubMed] [Google Scholar]
- 70. Van Dyke JM, Bain JLW, Riley DA.. Preserving sarcomere number after tenotomy requires stretch and contraction. Muscle Nerve. 2012;45(3):367–375. https://doi.org/ 10.1002/mus.22286 [DOI] [PubMed] [Google Scholar]
- 71. Gibson JNA, Smith K, Rennie MJ.. Prevention of disuse muscle atrophy by means of electrical stimulation: maintenance of protein synthesis. The Lancet. 1988;332(8614):767–770. https://doi.org/ 10.1016/s0140-6736(88)92417-8 [DOI] [PubMed] [Google Scholar]
- 72. Gibson JNA, Morrison WL, Scrimgeour CM, Smith K, Stoward PJ, Rennie MJ.. Effects of therapeutic percutaneous electrical stimulation of atrophic human quadriceps on muscle composition, protein synthesis and contractile properties. Eur J Clin Invest. 1989;19(2):206–212. https://doi.org/ 10.1111/j.1365-2362.1989.tb00219.x [DOI] [PubMed] [Google Scholar]
- 73. Gerovasili V, Stefanidis K, Vitzilaios K, et al. Electrical muscle stimulation preserves the muscle mass of critically ill patients: a randomized study. Crit Care. 2009;13(5):R161. https://doi.org/ 10.1186/cc8123 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Gruther W, Kainberger F, Fialka-Moser V, et al. Effects of neuromuscular electrical stimulation on muscle layer thickness of knee extensor muscles in intensive care unit patients: a pilot study. J Rehabil Med. 2010;42(6):593–597. https://doi.org/ 10.2340/16501977-0564 [DOI] [PubMed] [Google Scholar]
- 75. Meesen RLJ, Dendale P, Cuypers K, et al. Neuromuscular electrical stimulation as a possible means to prevent muscle tissue wasting in artificially ventilated and sedated patients in the intensive care unit: a pilot study. Neuromodulation: J Int Neuromodul Soc. 2010;13(4):315–320; discussion 321. https://doi.org/ 10.1111/j.1525-1403.2010.00294.x [DOI] [PubMed] [Google Scholar]
- 76. Dirks ML, Wall BT, Snijders T, Ottenbros CLP, Verdijk LB, van Loon LJC.. Neuromuscular electrical stimulation prevents muscle disuse atrophy during leg immobilization in humans. Acta Physiol Oxf Engl. 2014;210(3):628–641. https://doi.org/ 10.1111/apha.12200 [DOI] [PubMed] [Google Scholar]
- 77. Dirks ML, Wall BT, van Loon LJC.. Interventional strategies to combat muscle disuse atrophy in humans: focus on neuromuscular electrical stimulation and dietary protein. J Appl Physiol (Bethesda, Md.: 1985). 2018;125(3):850–861. https://doi.org/ 10.1152/japplphysiol.00985.2016 [DOI] [PubMed] [Google Scholar]
- 78. Honda Y, Tanaka N, Kajiwara Y, et al. Effect of belt electrode-skeletal muscle electrical stimulation on immobilization-induced muscle fibrosis. PLoS One. 2021;16(5):e0244120. https://doi.org/ 10.1371/journal.pone.0244120 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Honda Y, Takahashi A, Tanaka N, et al. Muscle contractile exercise through a belt electrode device prevents myofiber atrophy, muscle contracture, and muscular pain in immobilized rat gastrocnemius muscle. PLoS One. 2022;17(9):e0275175. https://doi.org/ 10.1371/journal.pone.0275175 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80. Brock Symons T, Park J, Kim JH, et al. Attenuation of skeletal muscle atrophy via acupuncture, electro-acupuncture, and electrical stimulation. Integr Med Res. 2023;12(2):100949. https://doi.org/ 10.1016/j.imr.2023.100949 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Bamman MM, Hunter GR, Stevens BR, Guilliams ME, Greenisen MC.. Resistance exercise prevents plantar flexor deconditioning during bed rest. Med Sci Sports Exerc. 1997;29(11):1462– 1468. https://doi.org/ 10.1097/00005768-199711000-00012 [DOI] [PubMed] [Google Scholar]
- 82. Bamman MM, Clarke MS, Feeback DL, et al. Impact of resistance exercise during bed rest on skeletal muscle sarcopenia and myosin isoform distribution. J Appl Physiol (Bethesda, Md.: 1985). 1998;84(1):157–163. https://doi.org/ 10.1152/jappl.1998.84.1.157 [DOI] [PubMed] [Google Scholar]
- 83. Alkner BA, Tesch PA.. Knee extensor and plantar flexor muscle size and function following 90 days of bed rest with or without resistance exercise. Eur J Appl Physiol. 2004;93(3):294–305. https://doi.org/ 10.1007/s00421-004-1172-8 [DOI] [PubMed] [Google Scholar]
- 84. Andrushko JW, Gould LA, Farthing JP.. Contralateral effects of unilateral training: sparing of muscle strength and size after immobilization. Appl Physiol Nutr Metab. 2018;43(11):1131–1139. https://doi.org/ 10.1139/apnm-2018-0073 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
All data generated or analyzed during the study are available from the corresponding author upon request.