Abstract
Background
Finerenone, a novel non-steroidal mineralocorticoid receptor antagonist, has shown promising efficacy and safety profiles in the management of chronic kidney disease (CKD) associated with type 2 diabetes mellitus (T2DM). However, evidence for its role in non-diabetic CKD patients require further investigation.
Methods
This retrospective, real-world study involved non-diabetic CKD patients from April 2023 to June 2024. Participants received finerenone alongside standard CKD treatment. Primary clinical results included changes in the urinary albumin-to-creatinine ratio (UACR), estimated glomerular filtration rate (eGFR), and serum potassium (sK+) levels. The data were collected initially and during follow-ups at 1, 3, 6, and 12 months.
Results
In total, 37 non-diabetic CKD patients were included in the population; 21 individuals (56.8%) were male, and the mean age was 48.84 ± 14.69 years. During the follow-up, there was a notable decrease in UACR, with a median reduction of 664.95 mg/g (IQR, 196.60-1226.70, P = 0.002). The baseline average eGFR was 70.80 ± 27.97 mL/min/1.73m2, with no notable alterations observed during the follow-up (P > 0.05). In terms of safety analysis, the sK + levels were within the 3.5–5.5 mmol/L range, with no significant difference from the baseline (P > 0.05). No patients discontinued treatment or were hospitalized because of hyperkalemia.
Conclusion
Real-world practice indicates that finerenone is effective and safe for non-diabetic CKD patients, but further large-scale, prospective studies are needed to confirm these findings.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12882-025-04241-w.
Keywords: Finerenone, Chronic kidney disease, Non-diabetic
Introduction
Chronic kidney disease (CKD) is a substantial burden threatening global health [1]. Aldosterone is one of the multiple factors contributing to the pathophysiological development of CKD [2]. Considerable evidence supports that aldosterone and overactivation of the mineralocorticoid receptor provoke inflammation, fibrosis, and eventual kidney dysfunction [3].
Currently, mineralocorticoid receptor antagonists (MRAs) have become a novel treatment strategy to alleviate the progression of CKD, the mechanism of which includes a reduction in renal collagen deposition and fibrosis, downregulation of proinflammatory mediators and cytokines, and a decrease in oxidative stress [4–6]. Spironolactone, a first-generation steroidal MRA, and eplerenone, a second-generation one, have shown promise in reducing proteinuria in CKD, though conclusive evidence for long-term renal outcome benefits remains limited [7, 8]. However, owing to apprehensions regarding potential complications such as hyperkalemia, worsening renal function, gynecomastia, impotence in men, and menstrual disorders in women, the use of these agents in CKD management is limited [9]. More recently, third-generation MRA has been successfully developed, and the alterations in receptor affinity and tissue tropism enable them to more selectively enhance efficacy and reduce risks [10].
Finerenone, a non-steroidal MRA, exhibits distinct receptor selectivity and has demonstrated albuminuria reduction in diabetic CKD [11, 12], but comparative efficacy against steroidal MRAs in proteinuria reduction requires further investigation. In the past few years, numerous clinical trials have proven the effectiveness and safety of finerenone in managing diabetic CKD [13–19]. The successful finerenone trials indicate its potential to revolutionize therapeutic approaches for CKD. Recent empirical evidence increasingly substantiates the therapeutic potential of finerenone across a diverse range of renal dysfunctions. A study involving patients with diabetic kidney disease and an estimated glomerular filtration rate (eGFR) below 25 mL/min/1.73 m² revealed that finerenone significantly mitigated eGFR decline without causing hyperkalemia, even among populations with advanced chronic kidney disease [20]. Furthermore, in patients with nephrotic-range proteinuria secondary to diabetic kidney disease, finerenone treatment was linked to a clinically significant reduction in the urinary protein-to-creatinine ratio [21]. These findings imply that the renoprotective effects of finerenone may transcend glycemic status, offering potential benefits for proteinuric CKD with severe functional impairment, but these findings are limited by small sample sizes, retrospective designs, and short follow-up periods. In non-diabetic CKD, MRAs like spironolactone and eplerenone have been shown to protect the heart and kidneys. Research has found that eplerenone can decrease proteinuria and blood pressure in these patients [22]. These findings highlight the possible benefits of MRAs for this patient group and emphasize the necessity for more information on the effectiveness of finerenone in non-diabetic CKD.
Currently, the effectiveness and safety of finerenone in non-diabetic CKD have been explored in a limited number of retrospective studies [23, 24]. However, the evidence for finerenone’s use in non-diabetic CKD patients is still not conclusive. Therefore, we conducted a real-world retrospective study investigating the efficacy and safety of finerenone in individuals with non-diabetic CKD.
Methods
Study design and patient population
This single-center, real-world study was conducted retrospectively at the Department of Nephrology, Zhongshan Hospital of Xiamen University from April 2023 to June 2024. Criteria for inclusion: (1) patients aged 18 to 85 years; (2) patients diagnosed with CKD with a baseline eGFR ≥ 25 mL/min/1.73m2; (3) patients undergoing finerenone treatment for CKD. Criteria for exclusion: (1) patients with diabetic nephropathy, or Addison’s disease; (2) patients lacking complete medical records; (3) patients with severe hepatic impairment (Child-Push grade C).
This study was approved by the Ethics Committee of Zhongshan Hospital of Xiamen University (IRB approval No. 2025-026), with the requirement for informed consent waived in accordance with the retrospective observational study design. Ethics approval was applied in accordance with the Declaration of Helsinki.
Treatment strategy
Patients received standard CKD treatment tailored to their specific disease type and comorbidities, including hypertension, hyperlipidemia, and hyperuricemia. Individualized regimes were implemented.
The initial dose of finerenone was determined based on the patient’s eGFR. Individuals with an eGFR equal to or greater than 60 mL/min/1.73m2 were recommended to start with a daily dose of 20 mg. In contrast, a lower starting dose of 10 mg per day is suggested for those with an eGFR between 25 and 60 mL/min/1.73 m². Patients with an eGFR of less than 25 mL/min/1.73 m² were contraindicated for finerenone. Serum potassium (sK+) levels must be closely monitored and dosage adjustments made within four weeks following the commencement of treatment. If sK+ levels were 4.8 mmol/L or lower, increasing the dose to 20 mg daily might be considered. Should sK+ levels fall within the range of 4.8 to 5.5 mmol/L, it was advisable to maintain the current dosage regimen. Conversely, if sK + levels were higher than 5.5 mmol/L, finerenone should be discontinued temporarily until sK + levels were at or below 5.0 mmol/L; following this, therapy may be restarted with a lower dose of 10 mg daily.
Clinical data collection
The demographic and laboratory characteristics of patients include age, gender, serum creatinine, eGFR, UACR, and sK+ levels. Clinical data collection will occur at baseline, and then at 1, 3, 6, and 12 months following treatment. The CKD-EPI formula was used to assess eGFR [25].
Sample size calculation
Given the retrospective nature of this real-world study, the sample size was inherently constrained by the availability of eligible patients who received finerenone during the study period (April 2023–June 2024). However, to assess the adequacy of the observed sample size for detecting a clinically meaningful effect, a post hoc power analysis was conducted based on the primary endpoint of UACR reduction. Key assumptions and statistical parameters: Effect size: 30% UACR reduction; SD: 25%; α = 0.05, power = 80%. Using a paired t-test, the required sample size was n = 18. Our cohort (n = 37) exceeded this threshold.
Statistical analysis
Categorical data were expressed as frequencies and percentages. Continuous data that followed a normal distribution were presented as the mean ± standard deviation, whereas those that did not were presented as the median and interquartile range. Repeated measure ANOVA and the generalized estimating equation were employed to analyze the data with repeated measurements. Statistical analyses employed SPSS version 26.0 (Chicago, IL, USA), with a significance level established at P < 0.05 (two-tailed).
Results
Patient characteristics
The baseline characteristics of the patients are presented in Table 1. A total of 37 patients were included in the analysis (21 males, mean age 48.84 ± 14.69 years). The most frequent etiologic diagnoses were IgA nephropathy (54.1%) and membranous nephropathy (24.3%). Other etiologies included Henoch–Schönlein purpura nephritis, minimal change nephropathy, hepatitis B virus–associated nephritis, mesangial proliferative glomerulonephritis, and obesity-related glomerulopathy. The baseline median UACR was 1091.90 mg/g (IQR, 494.50-1860.05), and the mean eGFR was 70.80 ± 27.97 mL/min/1.73m2. The baseline mean serum potassium level was 4.30 ± 0.46 mmol/L. Concomitant medications included renin-angiotensin system inhibitors (RASi) in 67.6% of patients and sodium-glucose cotransporter-2 inhibitors (SGLT2i) in 67.6% of the cohort. The baseline characteristics stratified by concomitant medications (RASi/SGLT2i) was presented in Supplemental Table 1.
Table 1.
Demographic and baseline characteristics of the patients
| Characteristics | All (n = 37) |
|---|---|
| Age (years) | 48.84 ± 14.69 |
| Male sex | 21 (56.8%) |
| Kidney diseases (etiological diagnosis)* | |
| IgA nephropathy | 20 (54.1%) |
| Membranous nephropathy | 9 (24.3%) |
| Henoch-Schönlein purpura nephritis | 2 (5.4%) |
| Minimal change nephropathy | 1 (2.7%) |
| Hepatitis B virus-associated nephritis | 1 (2.7%) |
| Mesangial proliferative glomerulonephritis | 1 (2.7%) |
| Obesity-related glomerulopathy | 1 (2.7%) |
| Baseline UACR (mg/g) | 1091.90 (494.50-1860.05) |
| ≤ 300 | 5 (13.5%) |
| > 300 | 32 (86.5%) |
| Baseline eGFR (mL/min/1.73m2) | 70.80 ± 27.97 |
| 25–45 | 8 (21.6%) |
| 45–60 | 8 (21.6%) |
| > 60 | 21 (56.8%) |
| Baseline serum potassium (mmol/L) | 4.30 ± 0.46 |
| Concomitant medications | |
| RASi | 25 (67.6%) |
| SGLT2i | 25 (67.6%) |
| Prednisone | 7 (18.9%) |
| Tacrolimus | 1 (2.7%) |
| Antithrombotic/anticoagulant drugs | 5 (13.5%) |
| Statins | 20 (54.1%) |
| Febuxostat | 13 (35.1%) |
UACR: urinary albumin-to-creatinine ratio; eGFR: estimated glomerular filtration rate; RASi: renin-angiotensin system inhibitors; SGLT2i: sodium-glucose cotransporter-2 inhibitors. *Two patients had unclassified glomerulonephritis (5.4%)
Therapeutic outcomes
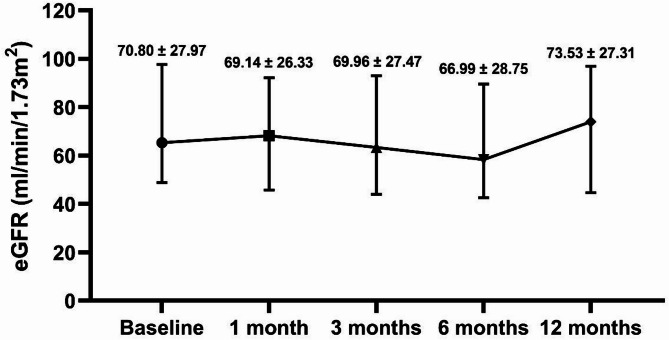
The UACR was decreased compared with baseline at 3 months (P = 0.018), 6 months (P = 0.020), and a significant reduction of 664.95 mg/g (IQR, 196.60-1226.70, P = 0.002) was further observed at 12 months. The reduction percentage of UACR was 60.90% (IQR, 39.76-65.95%) at 12 months, as shown in Fig. 1. The eGFR was lower at 6 months (66.99 ± 28.75 mL/min/1.73m2), but there was no significant difference at baseline, 1, 3, 6, or 12 months (all P > 0.05), as shown in Fig. 2.
Fig. 1.
The effect of finerenone on the relative changes in urinary albumin-to-creatinine ratio (UCAR) from baseline to 12 months follow-up
Fig. 2.
The effect of finerenone on the relative changes in estimated glomerular filtration rate (eGFR) from baseline to 12 months follow-up
The comparative analysis of UACR in the two most prevalent pathological subgroups, IgA nephropathy and membranous nephropathy, was presented in Supplemental Table 2, Supplemental Fig. 1, and Supplemental Fig. 2. In the IgA nephropathy subgroup, UACR demonstrated a decreasing trend within the 12-month follow-up; however, this decrease was not statistically significant compared to baseline (P > 0.05). In contrast, the membranous nephropathy subgroup exhibited a significant progressive reduction in UACR compared with baseline at 3 months (P = 0.012) and 12 months (P = 0.013). After 12 months of treatment, the percentage reduction of UACR from baseline was 40.26% (IQR, 24.27-70.66%) in the IgA nephropathy subgroup and 75.70% (IQR, 51.08-83.80%) in the membranous nephropathy subgroup. In patients with other glomerulopathies (Henoch-Schönlein purpura nephritis, minimal change nephropathy, etc.), the UACR a declining trend over the 12-month follow-up period, but this reduction did not reach statistical significance when compared to baseline measurements (P > 0.05), as shown in Supplemental Fig. 3.
Safety
For safety analysis, the levels of sK + remained within the 3.5–5.5 mmol/L range, with no significant differences compared to baseline (P > 0.05), as shown in Fig. 3. No treatment discontinuation or hospitalization occurred due to hyperkalemia.
Fig. 3.
The effect of finerenone on the relative changes in serum potassium (sK+) from baseline to 12 months follow-up
Discussion
This real-world, observational retrospective study indicated the potential for finerenone in the clinical treatment of non-diabetic CKD patients. Our findings revealed that patients undergoing finerenone therapy experienced a notable reduction in UACR and maintained a stable eGFR throughout the treatment, indicating the drug might be conducive to alleviating the deterioration of renal function. Another crucial aspect of this study was the stable sK+ levels in the non-diabetic CKD population during treatment, supporting the low hyperkalemia risk with finerenone, and manifesting acceptable safety and tolerability profile. Currently, the clinical evidence related to the utilization of finerenone in this patient population is extremely limited, and this study provided initial yet valuable evidence of both the efficacy and safety of finerenone in the treatment of non-diabetic CKD patients.
The reduction of UACR implies an enhancement in renal function and a deceleration in the progression of kidney disease. Studies have reported that a decreased UACR is correlated with superior clinical outcomes [26, 27]. Prior investigations have revealed that finerenone treatment leads to a reduction in UACR, demonstrating the renal protective effect in diabetic CKD patients [28]. The notable decrease in UACR seen in our group indicates that finerenone may effectively enhance kidney function in non-diabetic CKD patients. Pitt et al. [10] conducted an earlier study that examined the effectiveness of finerenone in individuals with heart failure and reduced left ventricular ejection fraction, in conjunction with mild or moderate CKD. In their study, two-thirds of the participants were non-diabetic, and a reduction in UACR was noted one month following finerenone therapy. The findings revealed a UACR reduction trend that matches our research. However, Pitt’s study did not conduct a diabetes-based subgroup analysis, thereby limiting the applicability of the findings to non-diabetic CKD patients. In our cohort, a reduction in UACR was noted within the first month of treatment initiation; nevertheless, this decrease was not statistically significant, which might be attributed to the limited sample size. On later follow-up, significant UACR reductions at 3 months were sustained over 12 months.
The more pronounced UACR reduction in membranous nephropathy compared to IgA nephropathy (75.7% vs. 40.26%) may reflect differences in molecular pathology: MR pathway overactivation likely plays a central role in MN podocyte injury, whereas IgAN fibrosis involves a broader signaling network (e.g., Ang II/Smad1). However, the small MN subgroup (n = 9) and higher baseline UACR may exaggerate observed treatment effects. Future studies with larger cohorts are needed to validate pathology-specific efficacy and explore predictive biomarkers.
The renoprotective mechanisms of MRAs in non-diabetic CKD may involve multi-target regulation of fibrotic and inflammatory pathways. Experimental studies have identified that angiotensin II-dependent activation of Src and Smad1 signaling drives extracellular matrix deposition in renal glomeruli [29]. The delayed but sustained UACR reduction observed from month 3 onward in our cohort aligns with this molecular cascade, as Src/Smad1-mediated collagen synthesis typically requires 8–12 weeks to manifest histologically detectable matrix remodeling. As a selective MRA, finerenone may interrupt this pathological process by blocking mineralocorticoid receptor-mediated transcriptional activation, thereby suppressing collagen synthesis and glomerulosclerosis. Furthermore, mineralocorticoid receptor activation has been shown to upregulate transcription factor 7-like 2 (TCF7L2), which potentiates the activin receptor-like kinase 1 (ALK1)/Smad1 pathway leading to tubular interstitial fibrosis [30]. This second-wave fibrotic mechanism explains why significant UACR improvements in our study emerged only after 3 months of therapy, as TCF7L2-ALK1 axis modulation requires prolonged mineralocorticoid receptor inhibition to reverse established fibrotic lesions. Importantly, non-diabetic CKD shares common pathogenic features with diabetic nephropathy, particularly regarding inflammation and oxidative stress amplification through mineralocorticoid receptor overactivation [30]. The early non-significant UACR changes (month 1) may reflect transient hemodynamic effects, whereas the later sustained improvements correlate with finerenone’s dual action: its non-steroidal structure may provides enhanced suppression of nuclear factor-κB (NF-κB) and NADPH oxidase activity compared to traditional steroidal MRAs [31], addressing both inflammatory cell infiltration and oxidative damage markers.
In CKD, renal function deterioration is usually marked by a gradual decrease in eGFR [32]. Previous meta-analysis on CKD indicated that while finerenone could slow down the deterioration of kidney function, patients still experienced a decrease in eGFR [33]. The FIDELIO-DKD study found that eGFR decline in diabetic CKD patients treated with finerenone followed a biphasic pattern [14]. In the initial stage, the finerenone group had a faster reduction in eGFR than the placebo group, but after 4 months, the decline rate slowed, causing the eGFR levels of the two groups to cross over. In our study, the average eGFR value moderately declined at the 6-month follow-up but subsequently moderately ascended at the 12-month follow-up. Despite the absence of statistically significant differences at various follow-up time points, the change in eGFR demonstrates a distinct pattern in non-diabetic CKD patients.
Finerenone, functioning as an MRA, is capable of influencing the retention of sodium and the excretion of potassium, which might result in hyperkalemia, particularly among CKD patients with compromised excretion function. Hyperkalemia is one of the specific adverse events linked to the utilization of MRA drugs [10]. Previous studies have indicated that finerenone therapy was correlated with an elevated risk of hyperkalemia. Nevertheless, through monitoring and adjusting the dosage of the medication, the risk of hyperkalemia can be effectively mitigated in diabetic CKD patients [14–16]. In our cohort, the sK+ level remained relatively stable throughout the 12-month follow-up period, and no serious adverse events related to the treatment were noted, thereby confirming the favorable safety profile of finerenone in the non-diabetic CKD patient population.
Recent advancements have identified four key therapeutic pillars for diabetic kidney disease (DKD) management: RASi, SGLT2i, glucagon-like peptide-1 receptor agonists (GLP-1RA), and MRAs [34]. These agents target complementary pathways: RASi reduces glomerular hypertension, SGLT2i enhances tubuloglomerular feedback, GLP-1RA decreases inflammation, and finerenone inhibits mineralocorticoid receptor-mediated fibrosis. The FIDELIO-DKD trial notably demonstrated that the addition of RASi therapy resulted in an 18% reduction in renal composite endpoints, without a significant increase in severe hyperkalemia [34]. This finding underscores the compatibility of finerenone with baseline RASi treatment and suggests potential synergistic benefits of combination therapies. Emerging evidence suggests synergistic effects of SGLT2i and MRAs in non-diabetic CKD. A recent open-label randomized trial by Mårup et al. [35] demonstrated that 8 weeks of combined dapagliflozin and finerenone therapy in non-diabetic CKD patients reduced albuminuria by 36%, with additive effects exceeding individual monotherapy responses (–24% for finerenone vs. − 8% for dapagliflozin). Notably, this combination was well-tolerated without severe hyperkalemia, supporting its feasibility in non-diabetic populations. In our study, despite 67.6% of patients received RASi/SGLT2i treatment, our findings underscore finerenone’s additional benefits in a real-world setting among non-diabetic CKD patients, aligning with emerging evidence supporting combination therapies.
This study has certain important limitations. First, the retrospective design and lack of a control group preclude causal attribution of UACR reduction to finerenone. Confounding by concomitant RASi/SGLT2i use and immunosuppressive therapies may partially explain observed outcomes. Second, the homogeneous, single-center cohort limits generalizability to global populations with diverse CKD etiologies. Finally, the short follow-up and small sample size restrict our ability to evaluate long-term renal and cardiovascular outcomes. Therefore, large-scale prospective randomized controlled trials with stratified designs and extended observation periods are needed to confirm these findings among diverse populations and in different regions of the world. The FIND-CKD trial (NCT05047263; registered on September 21, 2021, at ClinicalTrials.gov) currently underway promises to further elucidate the prolonged effects of finerenone on renal function in the non-diabetic CKD patient population.
Based on the observed efficacy and safety profile in this real-world cohort, finerenone may be considered as a viable therapeutic option for non-diabetic CKD patients with persistent proteinuria (UACR ≥ 300 mg/g) and mildly to moderately impaired renal function (eGFR ≥ 25 mL/min/1.73 m²). However, close monitoring of serum potassium levels and renal function is essential, particularly in patients with comorbid conditions. Dose adjustments should therefore be guided by potassium levels and tolerability to optimize therapeutic outcomes. Furthermore, combination therapy with RASi/SGLT2i could be considered to achieve synergistic renoprotection, though this approach requires validation through prospective clinical trials. Importantly, these findings should be interpreted with caution until confirmed by additional studies involving diverse patient populations. Future research should prioritize prospective randomized trials to establish generalizability and refine clinical implementation strategies.
In conclusion, finerenone demonstrates favorable efficacy and safety profiles in non-diabetic CKD patients within real-world clinical settings. This study offers significant insights and a promising perspective on the application of finerenone for non-diabetic CKD patients.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
We are grateful for the subjects in our hospital. We also thank all colleagues who collected all medical information and clinical data.
Author contributions
B.X., Y.L. and T.G. designed and conducted the study, acquired and analyzed data, and wrote the manuscript. J.F., X.G., W.Z, and T.Y. acquired and analyzed data. All authors read and approved the final manuscript.
Funding
This study was funded by grants from the Xiamen Municipal Bureau of Science and Technology (Grant No. 3502Z20244ZD1061).
Data availability
The datasets generated and analyzed in this study can be obtained from the authors upon reasonable request.
Declarations
Ethics approval and consent to participate
This study was approved by the Ethics Committee of Zhongshan Hospital of Xiamen University (IRB approval No. 2025-026), with the requirement for informed consent waived in accordance with the retrospective observational study design. Ethics approval was applied in accordance with the Declaration of Helsinki.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yu Li and Jiaxing Feng contributed equally to this work.
References
- 1.Webster AC, Nagler EV, Morton RL, Masson P. Chronic Kidney Disease Lancet. 2017;389(10075):1238–52. [DOI] [PubMed] [Google Scholar]
- 2.Barrera-Chimal J, Girerd S, Jaisser F. Mineralocorticoid receptor antagonists and kidney diseases: pathophysiological basis. Kidney Int. 2019;96(2):302–19. [DOI] [PubMed] [Google Scholar]
- 3.Epstein M. Aldosterone and mineralocorticoid receptor signaling as determinants of cardiovascular and renal injury: from Hans Selye to the present. Am J Nephrol. 2021;52(3):209–16. [DOI] [PubMed] [Google Scholar]
- 4.Patel V, Joharapurkar A, Jain M. Role of mineralocorticoid receptor antagonists in kidney diseases. Drug Dev Res. 2021;82(3):341–63. [DOI] [PubMed] [Google Scholar]
- 5.Barrera-Chimal J, Lima-Posada I, Bakris GL, Jaisser F. Mineralocorticoid receptor antagonists in diabetic kidney disease - mechanistic and therapeutic effects. Nat Rev Nephrol. 2022;18(1):56–70. [DOI] [PubMed] [Google Scholar]
- 6.Lainscak M, Pelliccia F, Rosano G, et al. Safety profile of mineralocorticoid receptor antagonists: spironolactone and eplerenone. Int J Cardiol. 2015;200:25–9. [DOI] [PubMed] [Google Scholar]
- 7.Quach K, Lvtvyn L, Baigent C, et al. The safety and efficacy of mineralocorticoid receptor antagonists in patients who require dialysis: A systematic review and Meta-analysis. Am J Kidney Dis. 2016;68(4):591–8. [DOI] [PubMed] [Google Scholar]
- 8.Hasegawa T, Nishiwaki H, Ota E, Levack WM, Noma H. Aldosterone antagonists for people with chronic kidney disease requiring dialysis. Cochrane Database Syst Rev. 2021;2(2):CD013109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bakris GL, Filippatos GS, Farmakis D, Epstein M, Pitt B. (2021). Aldosterone Antagonists and CVD. Washington, DC: American College of Cardiology. Available at: https://www.acc.org/latest-in-cardiology/articles/2021/07/19/13/42/aldosterone-antagonists-and-cvd (Accessed July 19, 2021).
- 10.Pitt B, Kober L, Ponikowski P, et al. Safety and tolerability of the novel non-steroidal mineralocorticoid receptor antagonist BAY 94-8862 in patients with chronic heart failure and mild or moderate chronic kidney disease: a randomized, double-blind trial. Eur Heart J. 2013;34(31):2453–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Georgianos PI, Agarwal R. Mineralocorticoid receptor antagonism in chronic kidney disease. Kidney Int Rep. 2021;6(9):2281–91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Grune J, Beyhoff N, Smeir E, et al. Selective mineralocorticoid receptor cofactor modulation as molecular basis for finerenone’s antifibrotic activity. Hypertension. 2018;71(4):599–608. [DOI] [PubMed] [Google Scholar]
- 13.Bakris GL, Agarwal R, Chan JC, et al. Effect of finerenone on albuminuria in patients with diabetic nephropathy: A randomized clinical trial. JAMA. 2015;314(9):884–94. [DOI] [PubMed] [Google Scholar]
- 14.Agarwal GL, Anker R. Effect of finerenone on chronic kidney disease outcomes in type 2 diabetes. N Engl J Med. 2020;383(23):2219–29. [DOI] [PubMed] [Google Scholar]
- 15.Zhang H, Xie J, Hao C, et al. Finerenone in patients with chronic kidney disease and type 2 diabetes: the FIDELIO-DKD subgroup from China. Kidney Dis (Basel). 2023;9(6):498–506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pitt B, Filippatos G, Agarwal R, et al. Cardiovascular events with finerenone in kidney disease and type 2 diabetes. N Engl J Med. 2021;385(24):2252–63. [DOI] [PubMed] [Google Scholar]
- 17.Agarwal R, Filippatos G, Pitt B, et al. Cardiovascular and kidney outcomes with finerenone in patients with type 2 diabetes and chronic kidney disease: the FIDELITY pooled analysis. Eur Heart J. 2022;43(6):474–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ghosal S, Sinha B. Finerenone in type 2 diabetes and renal outcomes: A random-effects model meta-analysis. Front Endocrinol (Lausanne). 2023;14:1114894. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Zhang MZ, Bao W, Zheng QY, Wang YH, Sun LY. Efficacy and safety of finerenone in chronic kidney disease: A systematic review and Meta-Analysis of randomized clinical trials. Front Pharmacol. 2022;13:819327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Mima A, Lee R, Murakami A, Gotoda H, Akai R, Kidooka S, Nakamoto T, Kido S, Lee S. Effect of finerenone on diabetic kidney disease outcomes with estimated glomerular filtration rate below 25 mL/min/1.73 m2. Metabol Open. 2023;19:100251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Mima A, Saito Y, Matsumoto K, Nakamoto T, Lee S. Effect of finerenone on nephrotic syndrome in patients with diabetic kidney disease. Metabol Open. 2024;22:100294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Tsuboi N, Kawamura T, Okonogi H, Ishii T, Hosoya T. The long-term antiproteinuric effect of eplerenone, a selective aldosterone blocker, in patients with non-diabetic chronic kidney disease. J Renin Angiotensin Aldosterone Syst. 2012;13(1):113–7. [DOI] [PubMed] [Google Scholar]
- 23.Zhou L, Li W. Effectiveness and safety of finerenone in Chinese CKD patients without diabetes: a retrospective, real-world study. Int Urol Nephrol. 2024;56(12):3877–85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Wang X. WCN24-933 effectiveness and safety of finerenone in Chinese CKD patients without diabetes mellitus: A real-world retrospective study. Kidney Int Rep. 2024;9(4):S56–7. [Google Scholar]
- 25.Levey AS, Stevens LA, Schmid CH, et al. A new equation to estimate glomerular filtration rate. Ann Intern Med. 2009;150(9):604–12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Neuen BL, Weldegiorgis M, Herrington WG, Ohkuma T, Smith M, Woodward M. Changes in GFR and albuminuria in routine clinical practice and the risk of kidney disease progression. Am J Kidney Dis. 2021;78(3):350–e3601. [DOI] [PubMed] [Google Scholar]
- 27.Levey AS, Gansevoort RT, Coresh J, et al. Change in albuminuria and GFR as end points for clinical trials in early stages of CKD: A scientific workshop sponsored by the National kidney foundation in collaboration with the US food and drug administration and European medicines agency. Am J Kidney Dis. 2020;75(1):84–104. [DOI] [PubMed] [Google Scholar]
- 28.Agarwal R, Tu W, Farjat AE, et al. Impact of Finerenone-Induced albuminuria reduction on chronic kidney disease outcomes in type 2 diabetes: A mediation analysis. Ann Intern Med. 2023;176(12):1606–16. [DOI] [PubMed] [Google Scholar]
- 29.Mima A, Matsubara T, Arai H, Abe H, Nagai K, Kanamori H, Sumi E, Takahashi T, Iehara N, Fukatsu A, Kita T, Doi T. Angiotensin II-dependent Src and Smad1 signaling pathway is crucial for the development of diabetic nephropathy. Lab Invest. 2006;86(9):927–39. [DOI] [PubMed] [Google Scholar]
- 30.Araoka T, Abe H, Tominaga T, Mima A, Matsubara T, Murakami T, Kishi S, Nagai K, Doi T. Transcription factor 7-like 2 (TCF7L2) regulates activin receptor-like kinase 1 (ALK1)/Smad1 pathway for development of diabetic nephropathy. Mol Cells. 2010;30(3):209–18. [DOI] [PubMed] [Google Scholar]
- 31.Mima A. Inflammation and oxidative stress in diabetic nephropathy: new insights on its Inhibition as new therapeutic targets. J Diabetes Res. 2013;2013:248563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Aklilu AM. Diagnosis of chronic kidney disease and assessing glomerular filtration rate. Med Clin North Am. 2023;107(4):641–58. [DOI] [PubMed] [Google Scholar]
- 33.Bao W, Zhang M, Li N, Yao Z, Sun L. Efficacy and safety of finerenone in chronic kidney disease associated with type 2 diabetes: a systematic review and meta-analysis of randomized clinical trials. Eur J Clin Pharmacol. 2022;78(12):1877–87. [DOI] [PubMed] [Google Scholar]
- 34.Mima A. A narrative review of diabetic kidney disease: previous and current Evidence-Based therapeutic approaches. Adv Ther. 2022;39(8):3488–500. [DOI] [PubMed] [Google Scholar]
- 35.Mårup FH, Thomsen MB, Birn H. Additive effects of Dapagliflozin and finerenone on albuminuria in non-diabetic CKD: an open-label randomized clinical trial. Clin Kidney J. 2023;17(1):sfad249. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets generated and analyzed in this study can be obtained from the authors upon reasonable request.