Abstract
Background
This study aimed to compare the effects of teriflunomide and ocrelizumab on clinical and MRI endpoints related to smoldering activity in relapsing–remitting multiple sclerosis (RRMS).
Methods
In this observational, longitudinal, multicenter study, we included 128 people with RRMS (pwRRMS) treated with teriflunomide and 495 treated with ocrelizumab. Outcomes included time to progression independent of relapse activity (PIRA). In a subset, we also assessed brain volume loss (BVL), longitudinal changes in diffusion tensor imaging (DTI) metrics, and the burden of paramagnetic rim lesions (PRLs). Propensity score matching was used for between-group comparisons.
Results
Over a median follow-up of 3.1 years in the ocrelizumab group and 1.9 years in the teriflunomide group, there were no significant differences in the risk of PIRA (HR for teriflunomide vs. ocrelizumab: 0.80 [95%-CI:0.40–1.60]; p = 0.53). PwRRMS treated with teriflunomide exhibited lower annualized rates of BVL (−0.80 [95%-CI: −0.91; −0.69] vs. −1.06 [95%-CI: −1.25; −0.86]; p = 0.025) and gray matter volume loss (−0.92 [95%-CI: −1.05; −0.79] vs. −1.20 [95%-CI: −1.43; −0.97]; p = 0.035). No differences were observed in DTI metrics or PRL count.
Conclusions
This real-world study suggests that teriflunomide shows similar efficacy to ocrelizumab on smoldering activity, with a potentially greater effect in reducing BVL. Further research is needed to confirm these findings and understand their long-term implications.
Supplementary Information
The online version contains supplementary material available at 10.1007/s00415-025-13221-x.
Keywords: Disease-modifying therapies, Ocrelizumab, Teriflunomide, PIRA, Smoldering MS, PRLs
Introduction
Multiple sclerosis (MS) is an immune-mediated neurological disease characterized by inflammation, demyelination, and neurodegeneration, ultimately leading to progressive disability. Traditionally, relapsing–remitting MS (RRMS) was thought to be defined solely by acute inflammatory events interspersed with periods of stability. However, it is now increasingly recognized that smoldering disease processes play a critical role in RRMS [1, 2]. A key concept in this evolving understanding is progression independent of relapse activity (PIRA), which refers to the gradual accumulation of disability driven by underlying disease processes that are unrelated to acute inflammatory events [3]. PIRA has been shown to occur frequently in people with RRMS (pwRRMS), where it is the primary determinant of disability accrual in the modern treatment era [3, 4].
The smoldering mechanisms underlying disease progression include non-resolving inflammation, neurodegeneration, oxidative stress, and mitochondrial dysfunction, which may occur independently or interact with each other [1]. In addition, the failure of compensatory mechanisms, such as remyelination and neuroplasticity, together with aging exacerbate these processes [1]. Some of these smoldering processes can be detected in vivo using advanced MRI biomarkers derived from both conventional and non-conventional techniques. These include the estimation of global and regional brain volume loss (BVL), particularly in gray matter structures, which serves as a proxy for the overall neurodegenerative burden of the disease [5]. Paramagnetic rim lesions (PRLs) are another important biomarker, gaining increasing importance for their ability to identify chronic active lesions, which are associated with worse clinical outcomes [6]. Diffusion MRI, which is sensitive to microstructural tissue integrity, provides indirect measures of demyelination and axonal loss [7, 8]. Notably, all these MRI measures have demonstrated sensitivity to the pathological changes occurring in patients with PIRA [9–13].
Together, the smoldering neurodegenerative and chronic inflammatory processes shape the clinical trajectories of MS, emphasizing the need for therapeutic strategies that address the complex nature of the disease. The currently approved disease-modifying therapies (DMTs) vary in their efficacy in providing neuroprotection. Teriflunomide, an oral immunomodulator approved for the treatment of relapsing forms of MS, targets dihydroorotate dehydrogenase, a mitochondrial enzyme involved in pyrimidine synthesis, thereby reducing the activity of proliferating B and T cells. In phase II and phase III clinical trials, teriflunomide showed improvements over placebo in annualized relapse rate (ARR), MRI lesions, and disability accumulation [14]. Additionally, teriflunomide has been shown to reduce BVL in both clinical trials and real-world studies, suggesting a potential effect on neurodegenerative processes [14]. Ocrelizumab, a recombinant humanized anti-CD20 monoclonal antibody, is approved for the treatment of both relapsing forms of MS and primary progressive MS (PPMS). In RRMS, ocrelizumab has demonstrated efficacy in reducing ARR, MRI lesions, and disability accumulation as compared to interferon beta-1a [15]. However, while ocrelizumab demonstrated superiority over placebo in reducing BVL in patients with PPMS, it did not show greater efficacy compared to interferon beta-1a in pwRRMS [15]. Notably, a post-hoc analysis of the pooled OPERA I and II trials showed that ocrelizumab is effective in reducing the incidence of PIRA compared to interferon beta-1a [16].
To date, no head-to-head comparisons have been conducted between teriflunomide and ocrelizumab, and the impact of these treatments on clinical and MRI measures of smoldering activity remains poorly explored.
In this study, we aimed to compare the association of teriflunomide and ocrelizumab with PIRA and MRI measures of smoldering disease activity in an observational multicenter cohort of pwRRMS.
Patients and methods
Participants and study design
In this observational study, participants were selected from the Swiss Multiple Sclerosis Cohort (SMSC), a prospective, multicenter study conducted across eight Swiss academic centers that features standardized collection of demographic, clinical, and MRI data [17]. For our investigation, we retrospectively included all adult SMSC participants diagnosed with RRMS who were treated with either teriflunomide or ocrelizumab during follow-up. We analyzed all longitudinal clinical and MRI examinations for each patient from the initiation of teriflunomide or ocrelizumab treatment, censoring data at the last visit, or at treatment discontinuation. Patients who received both treatments during SMSC follow-up were included only with data corresponding to the first treatment chronologically.
Specific analysis sets, with propensity score matching between treatment groups, were defined for each study endpoint based on data availability. The study endpoints included:
Time to PIRA, analyzed in the entire study cohort.
Global and regional BVL, analyzed in participants with at least one brain MRI scan including three-dimensional (3D) magnetization-prepared rapid gradient echo (MPRAGE).
Longitudinal changes in diffusion tensor imaging (DTI) metrics within white matter lesions (WMLs), normal-appearing white matter (NAWM), and cerebral cortex, analyzed in participants with at least one brain MRI scan including diffusion-weighted imaging.
Longitudinal changes in paramagnetic rim lesion (PRL) burden, analyzed in participants with at least one brain MRI scan including 3D segmented echo planar imaging (EPI).
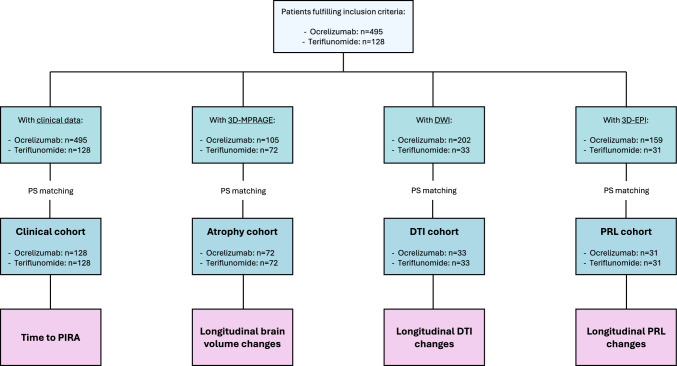
The study design is graphically displayed in Fig. 1. The study was approved by the local ethics committee, and all patients provided written informed consent before study entry.
Fig. 1.
Study design. Abbreviations: 3D-EPI = three-dimensional echo planar imaging; 3D-MPRAGE = three-dimensional magnetization-prepared rapid gradient echo; DTI = diffusion tensor imaging; DWI = diffusion-weighted imaging; PIRA = progression independent of relapse activity; PRL = paramagnetic rim lesion; PS = propensity score
Clinical data
All participants underwent regular clinical evaluations, conducted at least annually. Standardized assessments included the calculation of the Expanded Disability Status Scale (EDSS) score (https://www.neurostatus.net/), performed by certified raters.
The occurrence of PIRA during follow-up was defined as an increase in EDSS score (≥ 1.5, ≥ 1.0, or ≥ 0.5 points if baseline EDSS was 0, 1.0–5.5, or > 5.5, respectively) using a roving baseline [18], confirmed at least after 6 months, in the absence of relapses (1) between the EDSS increase and the preceding reference visit (conducted ≥ 90 days before the EDSS increase) and (2) between the EDSS increase and its confirmation [3].
MRI acquisition and analysis
Brain MRI scans were performed at each center with protocols optimized to ensure a homogeneous signal-to-noise ratio. Protocol details are provided in eTables 1–3.
Segmentation of T2-hyperintense WMLs on 3D fluid attenuated inversion recovery (FLAIR) images was performed using a deep learning-based tool [19], followed by manual correction.
Brain volumetric measurements were obtained on 3D-MPRAGE images using the longitudinal pipeline of SAMSEG, with 3D-FLAIR images included as an additional input to optimize automatic segmentation [20]. The volumes of interest considered in the study included total brain volume, cortical volume, thalamic volume, and total gray matter volume. Total intracranial volume (TIV) was also calculated to account for between-subject differences in head size in the statistical analyses.
DTI metrics were derived from diffusion-weighted images after denoising and correction for ringing artifacts, eddy current distortions, misalignments, and bias field [21]. Fractional anisotropy (FA), radial diffusivity (RD), mean diffusivity (MD), and axial diffusivity (AD) maps were generated using MRtrix [21]. For each map, mean values within WMLs, NAWM, and cerebral cortex were derived.
The presence of PRLs was assessed by an experienced rater (A.Ca.), who was blinded to patient identity. PRLs were defined as discrete FLAIR-hyperintense lesions either completely or partially encircled by a rim of paramagnetic signal, clearly evident in at least one contrast between unwrapped phase and quantitative susceptibility mapping (QSM), as previously described [10]. High inter-rater agreement in PRL detection was previously demonstrated in the same cohort [10].
In patients with PIRA, we assessed the accumulation of new or enlarged T2-hyperintense WMLs to identify events occurring without accompanying MRI activity, thereby fulfilling the criteria for progression independent of relapse and MRI activity (PIRMA) [3, 22]. This analysis was conducted using a semi-automated approach to systematically compare all 3D-FLAIR images acquired during routine follow-up in the SMSC [23].
Statistical analysis
All statistical analyses were conducted using R (version 4.3.1).
To mitigate bias from the non-random assignment to treatments in the SMSC study, we performed 1:1 propensity score matching between treatment groups. The matching criteria included the following variables at baseline: age, sex, disease duration, number of previous DMTs, number and volume of WMLs, raw and Z score levels of serum neurofilament light chain (sNfL) [24], EDSS score, and time under current treatment. Separate matched sets were constructed for each endpoint to maximize sample size.
We compared the incidence of PIRA between groups using Cox proportional hazard models to assess time to PIRA. Longitudinal changes in brain volumes, DTI metrics, and PRL burden were analyzed using mixed-effect models. These models included an interaction term between treatment group and time to examine group differences in longitudinal changes, with participants included as random intercepts. When modeling BVL, scanner magnetic field strength and TIV were included as additional covariates. Time was log-transformed to derive the annualized percentage change (APC) in MRI metrics. Model assumptions were assessed through visual inspection of residuals.
For time to PIRA, the following sensitivity analyses were performed: (1) censoring patients at the same time as their matching pair rather than at their last observation (pairwise censoring); (2) including only PIRA episodes where the initial EDSS increase occurred after treatment initiation; and (3) exploring time to PIRMA.
For BVL, the following sensitivity analyses were conducted: (1) including baseline age, sex, and disease duration, along with their interaction with time, as covariates in the mixed-effect models; (2) including ARR during MRI follow-up, along with its interaction with time, as covariates in the mixed-effect models; (3) including only patients with at least two MRI scans for volumetric analysis; and (4) including only scans obtained at least 6 months after treatment initiation.
All statistical analyses were conducted by a statistician (S.S.) who was not involved in the MRI analyses, while the authors conducting the MRI analyses did not have access to patient information, including treatment details.
Results
A total of 623 pwRRMS met the inclusion criteria and were enrolled in the study. The key demographic and clinical characteristics are summarized in Table 1. Separate match sets were created for each outcome of interest, and the baseline MRI characteristics showed no significant differences between the propensity score-matched groups (Table 2).
Table 1.
Key baseline characteristics of the cohort
| n | 623 |
| Age, mean (SD), years | 43.1 (12.5) |
| Females, No. (%) | 408 (65.5) |
| Disease duration, median [IQR], years | 9.2 [3.6; 16.7] |
| EDSS, median [IQR] | 2.5 [1.5; 3.5] |
| Number of previous DMTs, median [IQR] | 1 [0; 3] |
| Time under current treatment, median [IQR], years | 2.2 [0.0; 5.1] |
| sNfL, median [IQR], pg/ml | 10.2 [7.3; 14.8] |
| sNfL Z-score, median [IQR] | 0.8 [−0.1; 1.7] |
| T2LV, median [IQR], ml | 7.4 [3.4; 14.2] |
| T2L count, median [IQR] | 32.0 [21.7; 42.0] |
| SMSC centers | |
|
– Aarau, No. (%) – Basel, No. (%) – Bern, No. (%) – Geneva, No. (%) – Lausanne, No. (%) – Lugano, No. (%) – St. Gallen, No. (%) – Zurich, No. (%) |
71 (11.4) 315 (50.6) 17 (2.7) 64 (10.3) 46 (7.4) 52 (8.3) 42 (6.7) 16 (2.6) |
DMT disease modifying treatment, EDSS Expanded Disability Status Scale, IQR interquartile range, SD standard deviation, SMSC Swiss Multiple Sclerosis Cohort, sNfL serum neurofilament light chain, T2L T2-lesion, T2LV T2-lesion volume
Table 2.
MRI characteristics at baseline
| Ocrelizumab | Teriflunomide | Comparison | |
|---|---|---|---|
| Atrophy cohort | |||
| n | 72 | 72 | |
| Brain parenchymal fraction, median [IQR] | 0.69 [0.66;0.71] | 0.68 [0.65;0.73] | ap = 0.85; SMD = 0.067 |
| Cortical fraction, median [IQR] | 0.31 [0.29;0.33] | 0.31 [0.29;0.33] | ap = 0.99; SMD = 0.028 |
| Thalamic fraction, median [IQR] | 0.01 [0.01;0.01] | 0.01 [0.01;0.01] | ap = 0.53; SMD = 0.217 |
| Gray matter fraction, median [IQR] | 0.40 [0.38;0.43] | 0.41 [0.38;0.44] |
ap = 0.56; SMD = 0.088 |
| DTI cohort | |||
| n | 33 | 33 | |
| AD WMLs, median [IQR], mm2/s | 0.00149 [0.00141;0.00158] | 0.00145 [0.00141;0.00157] | ap = 0.77; SMD = 0.188 |
| AD NAWM, median [IQR], mm2/s | 0.00106 [0.00105;0.00108] | 0.00107 [0.00105;0.00109] | ap = 0.79; SMD = 0.023 |
| AD Cortex, median [IQR], mm2/s | 0.00106 [0.00104;0.00109] | 0.00106 [0.00103 0.00108] | ap = 0.69; SMD = 0.057 |
| FA WMLs, median [IQR] | 0.300 [0.277;0.320] | 0.303 [0.287;0.340] | ap = 0.24; SMD = 0.364 |
| FA NAWM, median [IQR] | 0.376 [0.363;0.388] | 0.386 [0.375;0.396] | ap = 0.12; SMD = 0.440 |
| FA Cortex, median [IQR] | 0.170 [0.158;0.175] | 0.169 [0.163;0.173] | ap = 0.88; SMD = 0.237 |
| MD WMLs, median [IQR], mm2/s | 0.00113 [0.00107;0.00121] | 0.00109 [0.00103;0.00121] | ap = 0.31; SMD = 0.274 |
| MD NAWM, median [IQR], mm2/s | 0.00074 [0.00073;0.00076] | 0.00074 [0.00073;0.00076] | ap = 0.88; SMD = 0.201 |
| MD Cortex, median [IQR], mm2/s | 0.00091 [0.00090;0.00093] | 0.00091 [0.00088;0.00092] | ap = 0.39; SMD = 0.173 |
| RD WMLs, median [IQR], mm2/s | 0.00095 [0.00091;0.00102] | 0.00090 [0.00084;0.00102] | ap = 0.21; SMD = 0.307 |
| RD NAWM, median [IQR], mm2/s | 0.00058 [0.00057;0.00060] | 0.00057 [0.00056;0.00060] | ap = 0.57; SMD = 0.271 |
| RD Cortex, median [IQR], mm2/s | 0.00083 [0.00082;0.00085] | 0.00083 [0.00081;0.00085] | ap = 0.30; SMD = 0.214 |
| PRL cohort | |||
| n | 31 | 31 | |
| PRL count, median [IQR] | 0 [0; 2] | 1 [0; 3.25] | ap = 0.17; SMD = 0.036 |
Brain parenchymal fraction, cortical fraction, thalamic fraction, and gray matter fraction were obtained by dividing total brain volume, cortical volume, thalamic volume, and gray matter volume by the total intracranial volume
aObtained with Mann–Whitney U test
AD axial diffusivity, DTI diffusion tensor imaging, FA fractional anisotropy, IQR interquartile range, MD mean diffusivity, NAWM normal-appearing white matter, PRL paramagnetic rim lesions, RD radial diffusivity, SMD standardized mean difference, SD standard deviation, WMLs white matter lesions
Incidence of PIRA
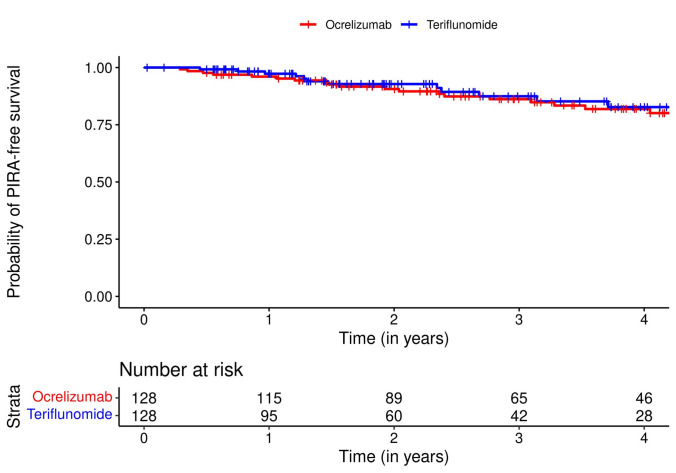
Baseline characteristics of the clinical cohort before and after propensity score matching are reported in eTable 4. A total of 128 pwRRMS per treatment group contributed to the analysis. The median (IQR) follow-up time was 3.1 (1.4; 4.6) years for the ocrelizumab group and 1.9 (1.0; 3.7) years for the teriflunomide group. During the observation period, there were 35 PIRA events, with 21 occurring in the ocrelizumab group and 14 in the teriflunomide group. The time to PIRA was not significantly different between treatment groups (hazard ratio [HR] for teriflunomide vs. ocrelizumab: 0.80 [95% CI: 0.40; 1.60]; p = 0.53). Kaplan–Meier curves for time to PIRA are displayed in Fig. 2.
Fig. 2.
Probability of PIRA-free survival. Abbreviations: PIRA = progression independent of relapse activity
Results remained consistent in sensitivity analyses applying pairwise censoring (HR for teriflunomide vs. ocrelizumab: 0.90 [95% CI: 0.37; 2.21]; p = 0.82) and excluding PIRA events with onset before treatment initiation (HR for teriflunomide vs. ocrelizumab: 0.93 [95% CI: 0.43; 2.02]; p = 0.86). 32 out of 35 PIRA events qualified as PIRMA; the time to PIRMA was not different between treatment groups (HR for teriflunomide vs. ocrelizumab: 0.75 [95% CI: 0.36; 1.56]; p = 0.44).
Brain volume loss
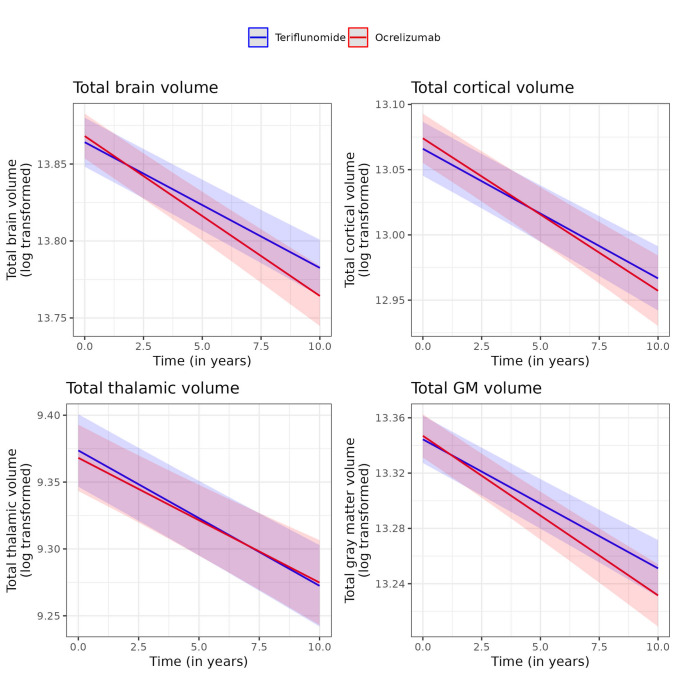
Baseline characteristics of the brain atrophy cohort before and after propensity score matching are reported in eTable 5. A total of 72 pwRRMS per treatment group contributed to the analysis. The median (IQR) follow-up time was 1.7 (0.6; 2.5) years for the ocrelizumab group and 1.0 (0.0; 3.8) years for the teriflunomide group. Compared to pwRRMS treated with ocrelizumab, those treated with teriflunomide exhibited reduced rates of total BVL (APC: −0.80 [95% CI: −0.91; −0.69] vs. −1.06 [95% CI: −1.25; −0.86]; p = 0.025) and gray matter volume loss (APC: −0.92 [95% CI: −1.05; −0.79] vs. −1.20 [95% CI: −1.43; −0.97]; p = 0.035) during the observation period (Fig. 3). There were no significant differences in the rates of cortical volume and thalamic volume loss between groups (Table 3).
Fig. 3.
Between group comparison of brain volume loss rates
Table 3.
Longitudinal changes in MRI metrics
| Ocrelizumab (APC) | Teriflunomide (APC) | Comparison | |
|---|---|---|---|
| Atrophy cohort | |||
| n | 72 | 72 | |
| Total brain volume change, mean (95% CI) | −1.06 (−1.25; −0.86) | −0.80 (−0.91; −0.69) | ap = 0.025 |
| Cortical volume change, mean (95% CI) | −1.24 (−1.51; −0.97) | −0.97 (−1.12, −0.82) | ap = 0.09 |
| Thalamic volume change, mean (95% CI) | −0.97 (−1.23; −0.71) | −1.04 (−1.19; −0.90) | ap = 0.64 |
| Gray matter volume change, mean (95% CI) | −1.20 (−1.43; −0.97) | −0.92 (−1.05; −0.79) | ap = 0.035 |
| No. of MRI follow-ups, median [IQR] | 2.5 [2.0;3.0] | 2.0 [1.0;4.3] | bp = 0.81; SMD = 0.295 |
| Follow-up time, median [IQR], years | 1.7 [0.6; 2.5] | 1.0 (0.0; 3.8) | bp = 0.83; SMD = 0.246 |
| DTI cohort | |||
| n | 33 | 33 | |
| AD WMLs change, mean (95% CI) | −0.05 (−0.72; 0.62) | 0.79 (0.15; 1.44) | ap = 0.08 |
| AD NAWM change, mean (95% CI) | 0.10 (−0.18; 0.38) | −0.00 (−0.27; 0.26) | ap = 0.60 |
| AD Cortex change, mean (95% CI) | −0.30 (−0.68; 0.09) | 0.08 (−0.28; 0.44) | ap = 0.17 |
| FA WMLs change, mean (95% CI) | −0.04 (−1.29; 1.20) | 0.23 (−0.94; 1.41) | ap = 0.76 |
| FA NAWM change, mean (95% CI) | 0.62 (−0.11; 1.34) | −0.27 (−0.95; 0.40) | ap = 0.08 |
| FA Cortex change, mean (95% CI) | −0.43 (−1.36; 0.52) | 0.33 (−0.58; 1.24) | ap = 0.26 |
| MD WMLs change, mean (95% CI) | 0.14 (−0.65; 0.93) | 0.84 (0.08; 1.60) | ap = 0.21 |
| MD NAWM change, mean (95% CI) | −0.11 (−0.55; 0.33) | 0.10 (−0.31; 0.51) | ap = 0.50 |
| MD Cortex change, mean (95% CI) | −0.24 (−0.56; 0.09) | 0.01 (−0.30; 0.32) | ap = 0.29 |
| RD WMLs change, mean (95% CI) | 0.30 (−0.69; 1.30) | 0.89 (−0.07; 1.85) | ap = 0.41 |
| RD NAWM change, mean (95% CI) | −0.29 (−0.92; 0.34) | 0.19 (−0.40; 0.79) | ap = 0.28 |
| RD Cortex change, mean (95% CI) | −0.20 (−0.52; 0.12) | −0.06 (−0.36; 0.25) | ap = 0.52 |
| No. of MRI follow-ups, median [IQR] | 3 [2; 4] | 3 [2; 4] | bp = 0.75; SMD = 0.050 |
| Follow-up time, median [IQR], years | 2.0 [1.7; 2.9] | 2.0 [1.0; 2.9] | bp = 0.91; SMD = 0.104 |
| PRL cohort | |||
| n | 31 | 31 | |
| Longitudinal changes over follow-up: | |||
| – Subjects with new PRLs, No (%) | 1 (3) | 2 (6) | ap = 0.96 |
| – Subjects with resolving PRLs, No (%) | 0 (0) | 0 (0) | |
| No. of MRI follow-ups, median [IQR] | 2 [2; 3] | 3 [2; 4] | bp = 0.25; SMD = 0.171 |
| Follow-up time, median [IQR], years | 1.5 [1.0; 2.2] | 2.0 [1.2; 2.9] | bp = 0.07; SMD = 0.452 |
aObtained from the mixed-effect models
bObtained with Mann–Whitney U test
AD axial diffusivity, APC annualized percentage change, DTI diffusion tensor imaging, FA fractional anisotropy, IQR interquartile range, MD mean diffusivity, MRI magnetic resonance imaging, NAWM normal-appearing white matter, PRL paramagnetic rim lesions, RD radial diffusivity, SMD standardized mean difference, SD standard deviation, WMLs white matter lesions
Significant p-values are displayed in bold
A significantly lower rate of BVL in patients treated with teriflunomide was confirmed in sensitivity analyses adjusting for baseline age, sex, and disease duration, as well as for ARR during the observation period, and in the subset of patients with at least 2 MRI time points (eTables 6–8). When including only scans performed at least 6 months after treatment initiation, the between-group differences in rates of BVL lost statistical significance (eTable 9).
DTI metrics
Baseline characteristics of the DTI cohort before and after propensity score matching are reported in eTable 10. A total of 33 pwRRMS per treatment group contributed to the analysis. The median (IQR) follow-up time was 2.0 (1.7; 2.9) years for the ocrelizumab group and 2.0 (1.0; 2.9) years for the teriflunomide group. No significant differences were observed in the rates of DTI metrics changes across any of the regions of interest during the observation period (Table 3).
PRL count
Baseline characteristics of the PRL cohort before and after propensity score matching are reported in eTable 11. A total of 31 pwRRMS per treatment group contributed to the analysis. The median (IQR) follow-up time was 1.5 (1.0; 2.2) years for the ocrelizumab group and 2.0 (1.2; 2.9) years for the teriflunomide group. During the observation period, three pwRRMS exhibited an increase in PRL count: two treated with teriflunomide and one with ocrelizumab. No patients showed resolution of PRLs. Longitudinal changes in PRL count did not differ between treatment groups (p = 0.96) (Table 3).
Discussion
In this large observational study, we found no differences in the incidence of PIRA between pwRRMS treated with teriflunomide or ocrelizumab. Similarly, longitudinal changes in PRL burden and DTI metrics did not differ between the two groups. However, pwRRMS treated with teriflunomide exhibited reduced rates of whole brain and gray matter volume loss compared to those treated with ocrelizumab.
PIRA, a relatively recent clinical concept, has garnered considerable attention for highlighting that disability can accumulate independently of clinical relapses, even in pwRRMS with short disease duration and low disability [3, 4, 16]. This understanding has driven research into mechanisms underlying disease progression and underscored the urgent need for treatments targeting these processes [1, 2]. This is relevant as PIRA is associated with unfavorable long-term outcomes, particularly when it occurs early in the disease course [25].
Currently, data on the impact of DMTs on PIRA incidence remain limited. A pivotal analysis of the pooled OPERA I and II trials demonstrated that ocrelizumab reduced the risk of PIRA by 22% compared to interferon beta-1a [16]. Post-hoc analyses of the ASCLEPIOS I and II trials found teriflunomide significantly inferior to ofatumumab, which reduced the risk of PIRA by 56% [26]. However, comparative data on teriflunomide versus ocrelizumab have been lacking. Our study addresses this gap in an observational real-world cohort using propensity score-matched treatment groups and reveals no differences in PIRA incidence between the two treatments, suggesting comparable impacts on the mechanisms underlying disability progression. Despite potential biases in implementing the PIRA concept in observational studies, our study benefitted from regular clinical follow-up and used a specific definition for PIRA optimized for providing high specificity in the context of observational studies [3].
During follow-up, patients treated with teriflunomide exhibited significantly reduced BVL compared to those treated with ocrelizumab. In randomized controlled trials, teriflunomide has proved superior to placebo [27], not significantly different from ofatumumab and ublituximab [28, 29], and inferior to ponesimod [30] in reducing BVL. Ocrelizumab, on the other hand, demonstrated superiority over placebo in PPMS [31], though differences compared to interferon beta-1a were non-confirmatory in the OPERA I trial and non-significant in the OPERA II trial [32]. In our study, whole brain and gray matter atrophy rates were significantly higher in the ocrelizumab group. Conversely, regional analyses showed no differences in cortex or thalamus atrophy rates. While the results were robust after adjusting for additional confounders, statistical significance was lost in sensitivity analyses restricted to scans obtained after six months of treatment initiation. This loss of significance may suggest a potential influence of pseudoatrophy on the observed results. However, it is important to acknowledge that the sensitivity analyses were limited by reduced statistical power due to a smaller dataset, making direct comparisons challenging. Collectively, these findings suggest teriflunomide may exert a greater impact on reducing neurodegenerative processes leading to macroscopic BVL, though further data are needed to confirm this observation. Supporting our findings, a recent network meta-analysis of clinical trials found teriflunomide to be superior to placebo in reducing BVL, while this was not the case for ocrelizumab. Moreover, monoclonal antibodies overall did not appear to outperform most other DMTs in this domain [33].
No significant differences in DTI metrics were observed between treatment groups. Diffusion imaging examines the dynamics of water molecules, offering indirect insights into the microstructural organization of tissues [7, 8]. Among diffusion MRI approaches, DTI is the simplest model, providing measures sensitive to both macrostructural and microstructural tissue integrity, which reflect processes such as demyelination and axonal damage [8]. Despite its limitations—such as an inability to accurately model complex tissue architectures, particularly in regions with crossing fibers—DTI has been widely utilized in MS research. Quantitative maps derived from DTI have demonstrated clinical relevance, serving as biomarkers at both the lesional level and in normal-appearing tissue [34]. The lack of significant differences in longitudinal DTI metrics between treatment groups aligns with the observed lack of differences in clinical outcomes.
Similarly, there were no differences in PRL burden changes between groups. Data on DMT effects on PRLs remain limited, with existing studies suggesting that current DMTs have minimal impact on PRL resolution [35]. Observational studies have shown no reduction in PRL count with teriflunomide [36] or anti-CD20 therapies, including ocrelizumab [11]. Our findings are therefore consistent with these observations.
Our study has several limitations. First, its observational design and the non-random allocation of patients to treatments introduce potential bias. While we attempted to mitigate this by performing propensity score matching, incorporating various demographic, clinical, and paraclinical factors, the study design cannot eliminate residual bias, particularly given the differing characteristics of patients treated with teriflunomide and ocrelizumab in clinical practice. Moreover, some relevant clinical variables, such as the burden of spinal cord lesions, could not be included as matching criteria. Additionally, we did not include pre-enrollment ARR as a criterion for matching, opting instead for sNfL levels as a potentially more sensitive marker of inflammatory activity. Second, although the overall cohort size was large, the number of patients included in certain MRI analyses was relatively small, with a relatively short follow-up period; this limitation is due to the recent introduction of systematic 3D-EPI and diffusion MRI data collection in the SMSC study. Third, due to the limited sample size, we were unable to stratify patients further based on prior treatment sequences or systematically account for potential temporal lags in reaching treatment nadir. Fourth, given the exploratory nature of this study, we chose not to adjust for multiple comparisons, which may have increased the risk of type I errors. Fifth, PIRA was defined exclusively by changes in EDSS scores, without incorporating additional measures such as upper and lower limb dexterity, potentially overlooking treatment-specific impacts on these outcomes. Sixth, the follow-up period was unbalanced between groups in some analyses; however, to address this potential bias, we used mixed-effect models specifically designed to account for differences in follow-up duration.
Despite these limitations, our findings are of potential clinical relevance, demonstrating that teriflunomide is non-inferior to ocrelizumab in clinical and MRI measures reflecting smoldering disease activity in patients with RRMS. However, further data are needed to confirm these findings and better understand their long-term implications.
Supplementary Information
Below is the link to the electronic supplementary material.
Funding
Open access funding provided by University of Basel. This study was funded by Sanofi, which had no role in data analysis and interpretation.
Data availability
The data that support the findings of this study are available on reasonable request.
Declarations
Conflict of interest
A. Cagol has received speaker honoraria from Novartis and Roche. S. Schadelin, M. Ocampo-Pineda, P. Benkert, L. Melie-Garcia, L. Luchetti, C. Pot, S. Finkener, G. Disanto, J. M. Lieb, F. Wagner, O. Chan-Hi Kim have nothing to disclose. Ö. Yaldizli received grants from ECTRIMS/MAGNIMS, University of Basel, Pro Patient Stiftung University Hospital Basel, Free Academy Basel, Swiss Multiple Sclerosis Society and advisory board/lecture and consultancy fees from Roche, Sanofi Genzyme, Allmirall, Biogen and Novartis. J. Oechtering received research support by the Swiss MS Society and served on advisory boards for Roche and Merck. M. D’Souza, Michael Diepers, R. Du Pasquier, M. Battaglini have nothing to disclose in relation to this work. B. Fisher-Barnicol reported personal fees from Biogen outside the submitted work. S. Müller received honoraria for travel, honoraria for lectures/consulting, and/or grants for studies from Almirall, Biogen, Celgene, Novartis, Teva, Merck Serono, Genzyme, Roche, and Bayer Schweiz. J. Vehoff received honoraria for travel, honoraria for lectures/consulting and/or grants for studies from Allmiral, Biogen, Novartis, Teva, Merck-Serono, Genzyme, Roche and Bayer Schweiz AG, none related to this work. A. Chan has served on advisory boards for, and received funding for travel or speaker honoraria from, Actelion-Janssen, Almirall, Bayer, Biogen, Celgene, Sanofi-Genzyme, Merck, Novartis, Roche, and Teva, all for hospital research funds; and research support from Biogen, Genzyme and UCB. A. Chan is associate editor of the European Journal of Neurology and serves on the editorial board for Clinical and Translational Neuroscience and as topic editor for the Journal of International Medical Research. C. Zecca’ institution the Department of Neurology, Regional Hospital Lugano (EOC), Lugano, Switzerland receives financial support from Teva, Merck Serono, Biogen, Genzyme, Roche, Celgene, Bayer and Novartis. T. Derfuss received speaker fees, research support, travel support, and/or served on Advisory Boards or Steering Committees of Alexion, Biogen, Celgene, GeNeuro, MedDay, Merck, Novartis, Roche and Sanofi-Genzyme; he received research support from Alexion, Biogen, Novartis, Roche, Swiss National Research Foundation, University of Basel, and Swiss MS Society. P. H. Lalive received honoraria for speaking and or travel expense from Biogen, Merck, Novartis, Roche; consulting fees from Biogen, GeNeuro, Merck, Novartis, Roche; research support from Biogen, Merck, Novartis. None were related to this work. E. Pravatà received honoraria for serving on scientific advisory boards from Bayer AG, unrelated to the present work. R. Hoepner received speaker/advisor honorary from Merck, Novartis, Roche, Biogen, Alexion, Sanofi, Janssen, Bristol-Myers Squibb, Teva/Mepha and Almirall. He received research support within the last 5 years from Roche, Merck, Sanofi, Biogen, Chiesi, and Bristol-Myers Squibb. He also received research grants from the Swiss MS Society, the SITEM Insel Support Fund and is a member of the Advisory Board of the Swiss and International MS Society. He also serves as deputy editor in chief for Journal of Central Nervous System disease and is part of the ECTRIMS Young Investigator Committee. P. Roth has received honoraria for lectures or advisory board participation from Alexion, Bristol-Myers Squibb, Boehringer Ingelheim, CDR-Life, Debiopharm, Galapagos, Laminar, Midatech Pharma, Novocure, OM Pharma, QED, Roche, Sanofi and Servier and research support from Merck Sharp and Dohme and TME Pharma. C. Gobbi’s institution, the Department of Neurology, Regional Hospital Lugano (EOC), Lugano, Switzerland received financial support from Teva, Merck Serono, Biogen, Genzyme, Roche, Celgene, Bayer and Novartis. PHL received honoraria for speaking from Biogen-Idec, CSL Bering, Merck Serono, Novartis, Sanofi-Aventis, Teva; consulting fees from Biogen-Idec, Geneuro, Genzyme, Merck Serono, Novartis, Sanofi-Aventis, Teva; research grants from Biogen-Idec, Merck Serono, Novartis. D. Leppert is Chief Medical Officer of GeNeuro. L. Kappos has received no personal compensation. His institutions (University Hospital Basel/Foundation Clinical Neuroimmunology and Neuroscience Basel) have received and used exclusively for research support: payments for steering committee and advisory board participation, consultancy services, and participation in educational activities from: Actelion, Bayer, BMS, df-mp Molnia & Pohlmann, Celgene, Eli Lilly, EMD Serono, Genentech, Glaxo Smith Kline, Janssen, Japan Tobacco, Merck, MH Consulting, Minoryx, Novartis, F. Hoffmann-La Roche Ltd, Senda Biosciences Inc., Sanofi, Santhera, Shionogi BV, TG Therapeutics, and Wellmera, and license fees for Neurostatus-UHB products; grants from Novartis, Innosuisse, and Roche. M. P. Sormani received consulting fees from Biogen, Merck, Novartis, Roche, Sanofi, Immunic, Alexion. J. Kuhle received speaker fees, research support, travel support, and/or served on advisory boards by Swiss MS Society, Swiss National Research Foundation (320030_212534/1), University of Basel, Progressive MS Alliance, Alnylam, Bayer, Biogen, Bristol Myers Squibb, Celgene, Immunic, Merck, Neurogenesis, Novartis, Octave Bioscience, Quanterix, Roche, Sanofi, Stata DX. C. Granziera: The University Hospital Basel (USB) and the Research Center for Clinical neuroimmunology and Neuroscience (RC2NB), as the employers of Cristina Granziera, have received the following fees which were used exclusively for research support from Siemens, GeNeuro, Genzyme-Sanofi, Biogen, Roche. They also have received advisory board and consultancy fees from Actelion, Genzyme-Sanofi, Novartis, GeNeuro, Merck, Biogen and Roche; as well as speaker fees from Genzyme-Sanofi, Novartis, GeNeuro, Merck, biogen and Roche.
References
- 1.Kuhlmann T, Moccia M, Coetzee T et al (2023) Multiple sclerosis progression: time for a new mechanism-driven framework. Lancet Neurol 22(1):78–88. 10.1016/S1474-4422(22)00289-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Scalfari A, Traboulsee A, Oh J et al (2024) Smouldering-associated worsening in multiple sclerosis: an international consensus statement on definition, biology, clinical implications, and future directions. Ann Neurol 96(5):826–845. 10.1002/ANA.27034 [DOI] [PubMed] [Google Scholar]
- 3.Müller J, Cagol A, Lorscheider J et al (2023) Harmonizing definitions for progression independent of relapse activity in multiple sclerosis: a systematic review. JAMA Neurol 80(11):1232–1245. 10.1001/jamaneurol.2023.3331 [DOI] [PubMed] [Google Scholar]
- 4.Lublin FD, Häring DA, Ganjgahi H et al (2022) How patients with multiple sclerosis acquire disability. Brain 145:3147–3161. 10.1093/brain/awac016 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Sastre-Garriga J, Pareto D, Battaglini M et al (2020) MAGNIMS consensus recommendations on the use of brain and spinal cord atrophy measures in clinical practice. Nat Rev Neurol 16(3):171–182. 10.1038/s41582-020-0314-x [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Bagnato F, Sati P, Hemond CC et al (2012) Imaging chronic active lesions in multiple sclerosis: a consensus statement. Brain 139(4):16–17. 10.1093/BRAIN/AWAE013 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Caranova M, Soares JF, Batista S, Castelo-Branco M, Duarte JV (2023) A systematic review of microstructural abnormalities in multiple sclerosis detected with NODDI and DTI models of diffusion-weighted magnetic resonance imaging. Magn Reson Imaging 104:61–71. 10.1016/j.mri.2023.09.010 [DOI] [PubMed] [Google Scholar]
- 8.Schmierer K, Wheeler-Kingshott CAM, Boulby PA et al (2007) Diffusion tensor imaging of post mortem multiple sclerosis brain. Neuroimage 35(2):467–477. 10.1016/j.neuroimage.2006.12.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cagol A, Schaedelin S, Barakovic M et al (2022) Association of brain atrophy with disease progression independent of relapse activity in patients with relapsing multiple sclerosis. JAMA Neurol 79(7):682–692. 10.1001/jamaneurol.2022.1025 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Cagol A, Benkert P, Melie-Garcia L et al (2024) Association of spinal cord atrophy and brain paramagnetic rim lesions with progression independent of relapse activity in people with MS. Neurology 102(1):e207768. 10.1212/WNL.0000000000207768 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Maggi P, Bulcke CV, Pedrini E et al (2023) B cell depletion therapy does not resolve chronic active multiple sclerosis lesions. EBioMedicine 94:104701. 10.1016/j.ebiom.2023.104701 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Cree BAC, Hollenbach JA, Bove R et al (2019) Silent progression in disease activity–free relapsing multiple sclerosis. Ann Neurol 85(5):653–666. 10.1002/ana.25463 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ocampo-Pineda M, Cagol A, Benkert P et al (2025) White matter tract degeneration in multiple sclerosis patients with progression independent of relapse activity. Neurol Neuroimmunol Neuroinflamm 12(3):e200388. 10.1212/NXI.0000000000200388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Miller AE (2021) An updated review of teriflunomide’s use in multiple sclerosis. Neurodegener Dis Manag 11(5):387–409. 10.2217/NMT-2021-0014 [DOI] [PubMed] [Google Scholar]
- 15.Lamb YN (2022) Ocrelizumab: a review in multiple sclerosis. Drugs 82(3):323–334. 10.1007/S40265-022-01672-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kappos L, Wolinsky JS, Giovannoni G et al (2020) Contribution of relapse-independent progression vs relapse-associated worsening to overall confirmed disability accumulation in typical relapsing multiple sclerosis in a pooled analysis of 2 randomized clinical trials. JAMA Neurol 77(9):1132–1140. 10.1001/jamaneurol.2020.1568 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Disanto G, Benkert P, Lorscheider J et al (2016) The Swiss Multiple Sclerosis Cohort-Study (SMSC): a prospective Swiss wide investigation of key phases in disease evolution and new treatment options. PLoS ONE 11(3):e0152347. 10.1371/journal.pone.0152347 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kappos L, Butzkueven H, Wiendl H et al (2018) Greater sensitivity to multiple sclerosis disability worsening and progression events using a roving versus a fixed reference value in a prospective cohort study. Mult Scler J 24(7):963–973. 10.1177/1352458517709619 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.La Rosa F, Abdulkadir A, Fartaria MJ et al (2020) Multiple sclerosis cortical and WM lesion segmentation at 3T MRI: a deep learning method based on FLAIR and MP2RAGE. Neuroimage Clin 27:102335. 10.1016/j.nicl.2020.102335 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Cerri S, Greve DN, Hoopes A et al (2023) An open-source tool for longitudinal whole-brain and white matter lesion segmentation. Neuroimage Clin 38:103354. 10.1016/j.nicl.2023.103354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Tournier JD, Smith R, Raffelt D et al (2019) MRtrix3: a fast, flexible and open software framework for medical image processing and visualisation. Neuroimage 202:116137. 10.1016/j.neuroimage.2019.116137 [DOI] [PubMed] [Google Scholar]
- 22.Ciccarelli O, Barkhof F, Calabrese M et al (2024) Using the progression independent of relapse activity framework to unveil the pathobiological foundations of multiple sclerosis. Neurology 103(1):e209444. 10.1212/WNL.0000000000209444/SUPPL_FILE/SUPPLEMENTARY_TABLE1.PDF [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Todea AR, Melie-Garcia L, Barakovic M et al (2023) A multicenter longitudinal MRI study assessing LeMan-PV software accuracy in the detection of white matter lesions in multiple sclerosis patients. J Magn Reson Imaging 58(3):864–876. 10.1002/jmri.28618 [DOI] [PubMed] [Google Scholar]
- 24.Benkert P, Meier S, Schaedelin S et al (2022) Serum neurofilament light chain for individual prognostication of disease activity in people with multiple sclerosis: a retrospective modelling and validation study. Lancet Neurol 21(3):246–257. 10.1016/S1474-4422(22)00009-6 [DOI] [PubMed] [Google Scholar]
- 25.Tur C, Carbonell-Mirabent P, Cobo-Calvo Á et al (2022) Association of early progression independent of relapse activity with long-term disability after a first demyelinating event in multiple sclerosis. JAMA Neurol 80(2):151–160. 10.1001/jamaneurol.2022.4655 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Gärtner J, Hauser SL, Bar-Or A et al (2022) Efficacy and safety of ofatumumab in recently diagnosed, treatment-naive patients with multiple sclerosis: results from ASCLEPIOS I and II. Mult Scler 28(10):1562–1575. 10.1177/13524585221078825 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Radue EW, Sprenger T, Gaetano L et al (2017) Teriflunomide slows BVL in relapsing MS: a reanalysis of the TEMSO MRI data set using SIENA. Neurol Neuroimmunol Neuroinflamm 4:e390 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Hauser SL, Bar-Or A, Cohen JA et al (2020) Ofatumumab versus teriflunomide in multiple sclerosis. N Engl J Med 383(6):546–557. 10.1056/nejmoa1917246 [DOI] [PubMed] [Google Scholar]
- 29.Steinman L, Fox E, Hartung HP et al (2022) Ublituximab versus teriflunomide in relapsing multiple sclerosis. N Engl J Med 387(8):704–714. 10.1056/nejmoa2201904 [DOI] [PubMed] [Google Scholar]
- 30.Kappos L, Fox RJ, Burcklen M et al (2021) Ponesimod compared with teriflunomide in patients with relapsing multiple sclerosis in the active-comparator phase 3 OPTIMUM study: a randomized clinical trial. JAMA Neurol 78(5):558–567. 10.1001/jamaneurol.2021.0405 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Montalban X, Hauser SL, Kappos L et al (2017) Ocrelizumab versus placebo in primary progressive multiple sclerosis. N Engl J Med 376(3):209–220. 10.1056/NEJMoa1606468 [DOI] [PubMed] [Google Scholar]
- 32.Hauser SL, Bar-Or A, Comi G et al (2017) Ocrelizumab versus interferon beta-1a in relapsing multiple sclerosis. N Engl J Med 376(3):221–234. 10.1056/nejmoa1601277 [DOI] [PubMed] [Google Scholar]
- 33.Zivadinov R, Keenan AJ, Le HH et al (2024) Brain volume loss in relapsing multiple sclerosis: indirect treatment comparisons of available disease-modifying therapies. BMC Neurol 24(1):1–12. 10.1186/S12883-024-03888-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cagol A, Tsagkas C, Granziera C (2024) Advanced brain imaging in central nervous system demyelinating diseases. Neuroimaging Clin 34:335–357. 10.1016/J.NIC.2024.03.003 [DOI] [PubMed] [Google Scholar]
- 35.Martire MS, Moiola L, Rocca MA, Filippi M, Absinta M (2022) What is the potential of paramagnetic rim lesions as diagnostic indicators in multiple sclerosis? Expert Rev Neurother 22:829–837. 10.1080/14737175.2022.2143265 [DOI] [PubMed] [Google Scholar]
- 36.Tan H, Li X, Li Y et al (2024) Real-world experience of teriflunomide in relapsing multiple sclerosis: paramagnetic rim lesions may play a role. Front Immunol 15:1343531. 10.3389/FIMMU.2024.1343531/BIBTEX [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data that support the findings of this study are available on reasonable request.