Abstract
This review paper investigated existing literature on the relationship between physical activity, exercise, and gynecological cancers, focusing on ovarian, endometrial, and cervical cancers. Epidemiological studies consistently demonstrate an inverse association between physical activity levels and the risk of developing gynecological cancers, as well as improved survival outcomes among cancer survivors engaging in regular exercise. Mechanistic insights suggest that physical activity and exercise may modulate key pathways involved in carcinogenesis, tumor progression, and treatment response, offering promising avenues for preventive and adjunctive therapeutic interventions. However, despite a growing body of research in exercise oncology, there is a lack of disease-specific synthesis addressing how exercise can be tailored to the distinct pathophysiological, treatment-related, and psychosocial challenges of gynecological cancers. Current guidelines often generalize exercise recommendations across all cancer types, overlooking the unique symptom burdens, such as pelvic floor dysfunction, sarcopenia, and menopausal symptoms, specific to gynecologic malignancies. This review addresses this gap by examining the evidence base for exercise in ovarian, endometrial, and cervical cancers, with an emphasis on mechanistic, clinical, and rehabilitative dimensions. By integrating epidemiological findings with preclinical and intervention data, this work aims to establish a gynecologic-specific framework for exercise prescription. This review underscores the importance of multidisciplinary research efforts to optimize lifestyle interventions, inform evidence-based guidelines, and improve outcomes for individuals affected by gynecological cancers.
Keywords: Gynecological cancers, Ovarian cancer, Endometrial cancer, Cervical cancer, Exercise
Introduction
Gynecological cancers, which include ovarian cancer, endometrial cancer, also called uterine cancer, and cervical cancer, are a major public health issue worldwide, significantly impacting women's well-being and overall health. They impose a considerable burden on healthcare systems and affect the quality of life of those diagnosed [1]. Based on the 2020 worldwide cancer data, ovarian cancer, cervical cancer, and endometrial cancer collectively accounted for 313,959, 604,127, and 417,367 new cases, respectively. The corresponding numbers of deaths were 207,252 for ovarian cancer, 341,831 for cervical cancer, and 97,370 for endometrial cancer [2]. The high mortality rate of ovarian cancer is largely due to late diagnosis and frequent recurrence. In many countries, five-year survival rates remain below 50% [3].
The typical treatment methods for gynecological cancers involve surgical removal of tumors and administration of systemic chemotherapy. Nevertheless, recurrence remains common even after the completion of chemotherapy [4]. While survival rates for gynecological cancer have risen over time, thanks to advancements in early detection, surgical methods, referral processes, and medical approaches, patients frequently encounter physical and psychological symptoms related to cancer and its treatments [5–7]. These symptoms include reduced physical activity, physical and functional limitations, pelvic floor issues, fatigue, and psychological distress [8, 9]. Such symptoms can significantly impair daily functioning and diminish health-related quality of life [10]. Unlike many other solid tumors, gynecological cancers often involve unique treatment-related side effects such as early menopause, sexual dysfunction, pelvic floor instability, and lymphedema following lymphadenectomy or radiotherapy [11]. These sequelae necessitate rehabilitation strategies tailored to the female reproductive anatomy and hormonal context, making the role of exercise in recovery particularly nuanced. With the increasing population of survivors of gynecological cancer, addressing the burden of the disease becomes a paramount concern [12]. Obesity and physical inactivity are the most significant modifiable risk factors for gynecological cancers. These modifiable factors are not only associated with increased cancer risk but also negatively influence treatment outcomes and survivorship. A considerable number of survivors of gynecological cancer are overweight or obese, which exacerbates insulin resistance, systemic inflammation, and hormonal imbalances, all of which are mechanisms implicated in tumor progression [13–15]. Exercise, as a modifiable lifestyle intervention, has the potential to target these pathways by improving metabolic health, reducing inflammatory cytokines, and restoring hormonal regulation. Thus, exercise may offer both preventive and therapeutic benefits [16, 17]. However, despite the promise of exercise as a therapeutic tool, women with gynecologic cancers often face gender-specific barriers to participation. These include caregiving responsibilities, sociocultural expectations, and norms that may deprioritize female engagement in structured physical activity [18, 19]. Additionally, cancer-related fatigue, compounded by hormonal shifts and emotional distress, frequently undermines motivation and adherence. Addressing these barriers is essential to designing equitable and effective exercise interventions that are both physiologically appropriate and socially sensitive [20, 21].
Despite the distinct physiological and treatment-related challenges posed by gynecological cancers, current exercise oncology research has often generalized findings across cancer types, with limited stratification for gynecologic subtypes. Most existing reviews and clinical guidelines focus on breast, prostate, or colorectal cancers, resulting in exercise recommendations that frequently overlook the disease-specific clinical trajectories, treatment toxicities, and rehabilitation priorities unique to gynecologic malignancies [22–24]. This review aims to address that critical gap by synthesizing current evidence on how exercise interventions may alleviate the symptom burden associated with ovarian, endometrial, and cervical cancers, and by highlighting the mechanistic pathways through which physical activity may contribute to improved outcomes in this population.
It is also important to emphasize that gynecological cancers differ considerably in their clinical course, treatment modalities, and symptom burden, factors that critically influence rehabilitation needs. Ovarian cancer is typically diagnosed at advanced stages and managed with extensive cytoreductive surgery followed by multi-agent platinum-based chemotherapy, often leading to prolonged fatigue, immunosuppression, and loss of lean body mass, which can influence exercise timing and tolerance [25]. In contrast, endometrial cancer is frequently treated with minimally invasive hysterectomy and may not require adjuvant therapy in early stages; however, the high prevalence of obesity, metabolic syndrome, and cardiovascular comorbidities in this population necessitates tailored, risk-adapted exercise prescriptions [26]. Cervical cancer is often treated with radical hysterectomy, pelvic lymphadenectomy, and concurrent chemoradiotherapy, resulting in pelvic floor dysfunction, fibrosis, lymphedema, chronic pain, and sexual or fertility-related issues that can limit physical activity and require specialized rehabilitation [27]. These distinct treatment-related challenges highlight the need for tailored exercise interventions beyond generalized oncology protocols. Instead, exercise prescriptions should be specific to the pathophysiology, psychosocial sequelae, and functional impairments characteristic of each gynecologic malignancy. These variations in treatment intensity and side effect profiles influence how each gynecologic cancer population may respond to and tolerate physical activity. For instance, the immunosuppression and sarcopenia following cytoreductive surgery and chemotherapy in ovarian cancer may delay initiation of high-intensity exercise and require prolonged recovery periods. In contrast, endometrial cancer survivors, often older and obese, may benefit more from moderate-intensity aerobic or resistance training aimed at improving metabolic health and reducing fatigue. Cervical cancer patients, due to pelvic radiation and nerve damage, often require specialized regimens focused on pelvic floor rehabilitation and core stabilization. Recognizing these distinctions is essential for designing safe, feasible, and effective exercise interventions aligned with each patient group's physiological and functional limitations. Epidemiological investigations indicate that engaging in leisure-time physical activities can decrease the risk of developing at least 13 different types of cancer [28, 29]. Moreover, these studies offer evidence suggesting that regular exercise is associated with a decreased risk of cancer recurrence for various cancer types [30–32]. Various guidelines advocate exercise training as a safe and effective approach for enhancing functional capabilities, strength of muscles, health-related quality of life, and cancer-related fatigue among cancer survivors [33–36].
Exercise represents a well-established, non-pharmacological, and safe strategy recommended for all patients undergoing cancer treatment, offering numerous advantages for overall health and quality of life. Furthermore, individuals who participate in higher levels of exercise show a considerably decreased relative risk of cancer mortality and recurrence [37, 38]. Research conducted on tumors in animals has revealed numerous biological mechanisms by which exercise can positively influence tumor biology. For instance, it can increase the expression of pro-apoptotic proteins and tumor suppressor genes, as well as enhance the infiltration of immune cells into tumors, which can restrict tumor cell proliferation and viability. Additionally, initial evidence from human studies indicates that exercise may reduce the levels of Ki-67, a pivotal marker of tumor proliferation [39, 40]. Furthermore, exercise has the potential to alleviate the adverse effects commonly experienced by patients undergoing local and systemic cancer treatments. Specifically, aerobic training (AT), resistance training (RT), and combined training consistently demonstrate beneficial effects on various health-related outcomes, including physical fitness, cancer-related fatigue, and quality of life [41].
In the context of gynecologic malignancies, such interventions may be particularly beneficial in mitigating pelvic pain, restoring pelvic floor and core muscle function, managing fatigue, and supporting psychological recovery, especially in the setting of reproductive loss. Given these challenges and the potential of exercise as a therapeutic strategy, this review aims to provide a gynecologic-specific synthesis of current knowledge. It examines the mechanistic foundations of exercise-induced tumor modulation, evaluates its effects across treatment phases, and summarizes clinical and rehabilitative outcomes. The review also explores the contextual factors that facilitate or hinder implementation of exercise interventions during and after treatment, with the ultimate goal of informing tailored, evidence-based exercise oncology approaches for women with ovarian, endometrial, and cervical cancers.
Mechanism of anticancer effects of exercise
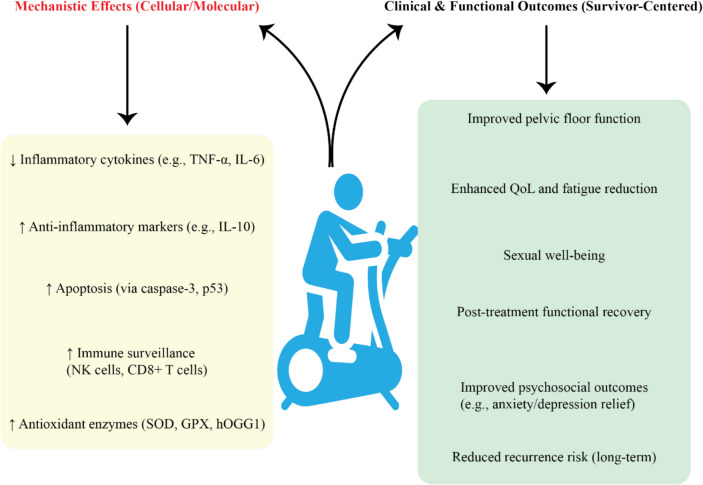
In this section, the multifaceted relationship between exercise and cancer is delved into, with various mechanisms explored through which cancer development and progression are impacted by exercise (Fig. 1). Given the distinct molecular profiles and treatment-related sequelae of ovarian, endometrial, and cervical cancers, understanding how exercise modulates these mechanisms specifically in gynecological malignancies is essential for developing targeted therapeutic strategies [42]. While direct preclinical evidence in gynecologic cancer models remains limited, meaningful insights can still be derived by integrating current knowledge of these cancers’ biology with known exercise-induced mechanisms. For instance, the estrogen-driven pathophysiology of endometrial cancer suggests that exercise-mediated modulation of estrogen signaling, adiposity, and insulin resistance may be particularly relevant in this context [42, 43]. Similarly, the frequent chemoresistance observed in ovarian cancer—often linked to impaired apoptosis and oxidative DNA damage repair—raises the possibility that exercise could enhance treatment efficacy by restoring apoptotic signaling and improving redox homeostasis [44]. In cervical cancer, persistent HPV infection and the resulting immune evasion highlight the potential for exercise to act as an immunological adjuvant, enhancing antiviral immunity and T cell infiltration [44, 45]. Thus, even in the absence of extensive disease-specific models, mechanistic hypotheses grounded in the distinct molecular and immunological features of each gynecological malignancy can inform future experimental research and guide clinical translation.
Fig. 1.
Schematic representation of the mechanistic and clinical benefits of exercise in gynecological cancer patients
The dual role of reactive oxygen species: implications for exercise, oxidative stress, and cancer risk
Reactive oxygen species (ROS) are essential for several physiological functions, such as immune responses, cell signaling, and blood vessel permeability [46]. Nevertheless, the excessive buildup of ROS, either as a result of normal energy metabolism or due to inflammation and exposure to environmental factors like tobacco smoke, has been linked to the development of cancer in both animal studies and human research [47]. Experimental research has shown that ROS can act both as initiators and promoters of tumors by causing damage to important cellular elements such as DNA, proteins, and lipids. Additionally, ROS can function as signaling molecules within cells [48].
Enzymatic and non-enzymatic antioxidant systems, such as superoxide dismutase (SOD), glutathione peroxidase (GPX), catalase (CAT), ascorbic acid (vitamin C), tocopherol (vitamin E), and β-carotene, are vital for removing ROS and shielding cells from oxidative harm [49]. Although external sources of ROS, such as environmental carcinogens, are well-known contributors to cancer, recent evidence suggests that internal sources of ROS, particularly those generated during acute exercise, may also play a role in tumor initiation [50, 51]. However, regular physical activity has consistently been linked to a decreased risk of various cancer types [28, 52]. This seeming contradiction implies that prolonged exercise might trigger physiological changes that offset oxidative harm to DNA [53]. Research has demonstrated that extended periods of exercise training can increase the levels of antioxidant enzymes and non-enzymatic repair mechanisms, which helps alleviate oxidative stress and lowers the likelihood of cancer occurrence [54]. Although most studies focus on skeletal and cardiac tissue, recent evidence shows that exercise also affects antioxidant expression in tissues relevant to cancer [55]. In gynecologic oncology, this is particularly relevant, as oxidative stress has been implicated in ovarian carcinogenesis, while obesity-linked oxidative imbalances are central in endometrial cancer development. Exercise-induced antioxidant adaptation may therefore play differential roles depending on cancer type [56, 57]. Research findings indicate that after exercise training, there is a rise in the levels of antioxidant enzymes such as SOD and GPX in lung tissue [58]. Furthermore, exercise has been observed to boost the effectiveness of DNA repair enzymes like human MutT homologue (hMTH1) and 8-oxoG DNA glycosylase (hOGG1), which are pivotal in averting the mutagenic impacts of oxidative damage [58, 59].
In gynecologic malignancies, oxidative stress plays cancer-type-specific roles. In ovarian cancer, elevated ROS levels not only promote tumor initiation but are also linked to platinum-based chemotherapy resistance, as ROS can impair DNA repair pathways [60]. Preclinical studies suggest that endurance exercise can increase expression of DNA repair enzymes such as hOGG1 in multiple tissues, including skeletal muscle and liver, which may have relevance for reducing oxidative damage in tumors with impaired DNA repair capacity, such as ovarian cancer [61, 62]. In endometrial cancer, particularly in early-stage, hormone-sensitive cases, obesity-driven oxidative stress amplifies estrogen-mediated endometrial proliferation and hyperplasia [63]. Regular exercise may help attenuate this pathway by enhancing antioxidant capacity and lowering systemic ROS levels, thereby reducing proliferative signaling [64]. Although acute exercise may temporarily increase ROS, long-term training elicits adaptive responses that reduce oxidative DNA damage and lower cancer risk [65, 66]. Acute exercise typically involves a single bout of moderate-to-high intensity aerobic or resistance activity, lasting approximately 30 to 60 min at intensities of 60–80% VO₂max or 70–85% one-repetition maximum (1RM) [67, 68]. In contrast, long-term training consists of repeated sessions performed three to five times per week over a minimum duration of 8 to 12 weeks, employing aerobic, resistance, or combined exercise modalities [69, 70]. These chronic exercise regimens stimulate upregulation of endogenous antioxidant enzymes such as SOD and GPX, as well as enhanced DNA repair mechanisms, which collectively mitigate oxidative damage and contribute to reduced cancer risk [71, 72].
Exercise modulation of cellular dynamics: implications for cancer development and progression
The impact of exercise on cancer development is rooted in complex interactions within the cellular microenvironment. Exercise can shift the balance between cell proliferation and apoptosis, both of which are crucial in the development of tumors. Studies indicate that exercise has the potential to affect this equilibrium, potentially hindering the onset and advancement of cancer [73]. Exercise also can elevate apoptosis levels, especially in scenarios such as mammary tumorigenesis in rats [74]. Moreover, exercise might boost apoptosis via pathways not reliant on p53 [75]. Physical activity seems to diminish the proliferation of triple-negative breast cancer cells, lessen the occurrence and proliferation of cancer cells in murine models, and diminish the proliferative traits of specific cancer cells in rodents [76]. Nonetheless, there is no indication supporting the notion that exercise can entirely eradicate malignant tumors [77, 78].
The influence of exercise on tumor occurrence may fluctuate, yet it consistently diminishes both the quantity and dimensions of tumors across diverse models. In endometrial cancer, which is frequently associated with unopposed estrogen stimulation and hyperproliferation of the endometrial lining, exercise-induced modulation of apoptosis may counteract abnormal cell growth in hormone-sensitive contexts [79]. Evidence suggests that moderate aerobic exercise can downregulate estrogen receptor activity and promote apoptosis through Bcl-2 family regulation, thereby offering chemopreventive effects in early-stage endometrial neoplasia [80, 81]. In ovarian cancer, therapeutic resistance is often driven by impaired apoptotic signaling, particularly through overexpression of anti-apoptotic proteins like Bcl-XL and surviving [82]. Preclinical studies suggest that exercise may restore pro-apoptotic signaling via mitochondrial pathways and upregulation of tumor suppressor genes, such as p53 and PTEN, potentially re-sensitizing resistant tumor cells to chemotherapy [83, 84]. This makes the apoptosis-enhancing effects of exercise particularly relevant for improving treatment response and reducing recurrence in ovarian cancer survivors. In cervical cancer, apoptosis dysregulation is frequently linked to HPV oncoproteins (E6 and E7), which suppress p53 and Rb pathways [85]. Exercise may help restore apoptotic sensitivity by modulating these disrupted pathways indirectly through systemic hormonal and immune changes. For example, moderate-intensity training has been linked to lower circulating cortisol and pro-inflammatory cytokines, both of which can influence HPV-related apoptotic resistance [86, 87].
The level of intensity during exercise appears pivotal in its capacity to impede the proliferation of cancer cells. Moderate-intensity workouts have consistently demonstrated the ability to restrain cancer cell expansion and trigger apoptosis, underscoring their protective role in various preclinical models [40]. While some studies suggest that higher intensity training may be less effective or even potentially detrimental, such conclusions should be interpreted with caution [88, 89]. In rodent models, high-intensity exercise is typically conducted using forced modalities such as treadmill running at fixed speeds. These protocols can introduce psychological stress, as evidenced by elevated biological and behavioral markers of chronic distress, which may independently affect tumor biology [90]. Consequently, it remains challenging to disentangle the direct physiological effects of vigorous exercise from those arising due to stress induced by coercion. This methodological limitation highlights the need for more refined experimental designs to accurately evaluate the dose–response relationship between exercise intensity and cancer progression [74]. In addition to promoting apoptosis, exercise activates intracellular signaling cascades that inhibit tumor growth. For instance, moderate swimming has demonstrated an ability to elevate dopamine levels, which in turn regulate kinase phosphorylation and the activity of transforming growth factor-β1 (TGF-β1), thus restraining the proliferation of cancer cells [91]. Moreover, exercise triggers the Hippo signaling pathway, resulting in the suppression of transcription factors implicated in cancer cell proliferation [92]. Furthermore, exercise can modulate the secretion of insulin-like growth factors, target distinct pathways like Akt and mTOR, and prompt apoptosis in cancer cells via diverse mechanisms, relevant in obese endometrial cancer patients where insulin resistance is a key pathogenic driver [93, 94]. Exercise has demonstrated the capacity to reduce the overexpression of the MiT/TFE gene in patients afflicted with renal, pancreatic, and melanoma cancer, resulting in the inhibition of cancer cell proliferation [95]. Moreover, exercise has the potential to trigger apoptosis in tumor cells located within skeletal muscle. This is facilitated through various mechanisms, such as the restoration of autophagy and the enhancement of mitochondrial function [96]. Regular exercise stimulates skeletal muscle to generate SPARC (secreted protein acidic and rich in cysteine) in both mouse models and human patients with colon cancer. This process facilitates the cleavage of caspase-3 and caspase-8, leading to increased cell apoptosis and consequent inhibition of colon cancer growth, the application to gynecologic oncology is emerging [97]. In ovarian cancer, impaired autophagy contributes to chemoresistance, and exercise-induced mitochondrial biogenesis and SPARC expression may represent novel avenues for overcoming therapeutic resistance [98]. In sum, by modulating cell proliferation, promoting apoptosis, and reprogramming survival signaling pathways, exercise offers a mechanistically grounded adjunct to existing treatment strategies in gynecologic malignancies. These effects are particularly pertinent given the molecular heterogeneity and hormone sensitivity of endometrial tumors, chemoresistance in ovarian cancer, and HPV-driven pathogenesis in cervical cancer.
The interplay between chronic inflammation, exercise, and cancer
Chronic inflammation, caused by persistent infections or exposure to non-infectious agents such as smoke or asbestos, has long been associated with the onset and progression of various types of cancer. It contributes to approximately 15% of all cancer cases worldwide [99]. In gynecological cancers, such as endometrial cancer, ovarian cancer, and cervical cancer, chronic inflammation is a key factor in tumor progression, immune suppression, and resistance to treatment [100]. For example, in endometrial cancer, obesity-driven chronic inflammation contributes to insulin resistance and hormonal dysregulation, both of which are associated with tumor growth [101]. In ovarian cancer, pro-inflammatory ascitic fluid promotes immune suppression and metastasis, while persistent HPV infection in cervical cancer creates an inflammatory environment that facilitates immune evasion and carcinogenesis [102–104]. This chronic inflammatory state is characterized by tissue damage, cellular proliferation, and the activation of repair pathways, with the recruitment of lymphocytes and macrophages to the inflamed site. The resulting inflammatory microenvironment not only supports tumor growth but also helps cancer cells evade immune surveillance [105, 106]. While acute inflammation serves protective roles in host defense and tissue repair, chronic inflammation, particularly in the context of cancer, fosters an environment conducive to tumor progression and therapy resistance [107]. This chronic low-grade inflammation is associated with elevated levels of pro-inflammatory cytokines such as TNF-α, IL-1β, and IL-6, which are involved in cellular proliferation, immune evasion, and tumor survival. While acute inflammation plays a protective role in host defense and tissue repair, chronic inflammation—especially in the context of gynecological cancers—fosters an environment that promotes tumor survival and resistance to therapies [108, 109].
In gynecologic cancers, chronic inflammation plays a differential role. In endometrial cancer, obesity-driven chronic inflammation is a key contributor to tumorigenesis through elevated levels of circulating pro-inflammatory cytokines such as IL-6 and TNF-α [110, 111]. Exercise may help disrupt this tumor-promoting environment by attenuating systemic inflammation and improving metabolic parameters associated with insulin resistance [16]. In cervical cancer, persistent HPV infection triggers a chronic inflammatory milieu that fosters immune evasion and carcinogenesis; regular physical activity may modulate this response by reducing local pro-inflammatory cytokine levels and enhancing antiviral immune signaling [112, 113]. In ovarian cancer, the accumulation of pro-inflammatory ascitic fluid and tumor-associated macrophages contributes to immune suppression and metastasis—mechanisms that exercise may partially counteract through macrophage phenotype reprogramming and IL-10 induction [114, 115]. These gynecologic-specific pathways underscore the potential of exercise to target the unique inflammatory landscapes of each cancer subtype.
Adipose tissue is thought to contribute to this inflammation, especially in obesity [116]. Physical inactivity has been associated with systemic inflammation, while exercise has been shown to reduce chronic inflammation, potentially through alterations in cytokine levels [117]. Exercise-induced IL-6, mainly from skeletal muscle, may inhibit pro-inflammatory cytokines like TNFα and IL-1β and stimulate anti-inflammatory mediators. Transcription factors like NF-κB (nuclear factor-kappaB) and AP-1 (activator protein-1), implicated in immune responses and inflammation, are activated by exercise and may play a role in mediating its effects on inflammation and tumor development [118]. A study involving postmenopausal breast cancer survivors investigated the impact of exercise on inflammatory markers. While exercise did not significantly alter inflammatory markers overall, exercisers who achieved 80% of the intervention goal showed a notable reduction in IL-6 levels compared to those who did not [119]. Based on the results of a meta-analysis, regular exercise has shown promise in reducing serum levels of certain proinflammatory markers, including IL-6, IL-2, IL-8, and TNFα, in breast cancer survivors [120, 121]. IL-6, linked to fatigue in cancer patients, notably decreased with exercise, potentially explaining the fatigue-alleviating effects of physical activity. IL-6 is also predictive of survival in metastatic breast cancer, suggesting exercise may positively influence survival rates [122–124]. The meta-analysis also revealed positive effects on IL-2 levels, indicative of enhanced immune function, particularly through natural killer cell activity and its critical role in T-cell homeostasis [121]. Despite exercise-induced increases in IL-6, it also triggers IL-10 release, known for its anti-inflammatory properties, potentially modulating tumor growth. While exercise reduced IL-8 and TNFα levels, no significant changes were observed in CRP levels. These findings suggest that exercise can modulate chronic inflammation and potentially improve cancer outcomes, though more research is needed to confirm effects on cytokine profiles and survival [121].
Exercise as an immunomodulatory tool in cancer: unveiling the intricacies of immune regulation
Exercise has emerged as a potential modulator of immune function in cancer, with a focus on enhancing anti-tumor immunity. Recent studies suggest that physical activity may help reverse immune suppression, reactivating immune cells involved in combating cancer [125]. Tumor progression is shaped by complex immune interactions, and exercise engages several physiological pathways to modify these responses [126]. Acute exercise prompts transient immune reactions, like lymphocyte redistribution, enhancing immune preparedness. Conversely, prolonged exercise regimes induce lasting modifications in immune function, exerting prolonged effects on tumor development [127].
Within the array of immune cell types, macrophages emerge as pivotal controllers of tumor progression. These cells exhibit diverse phenotypes, with the M1 phenotype commonly recognized for its anti-tumor properties, while the M2 phenotype is often implicated in tumor advancement [128, 129]. In the context of gynecologic cancers, tumor-associated macrophages (TAMs) exhibit varying densities and phenotypes depending on the cancer type. In ovarian cancer, high M2 macrophage infiltration is linked to immune suppression, ascites formation, and poor prognosis, making exercise-induced M1 polarization a potentially valuable strategy for reprogramming the tumor microenvironment [130, 131]. In endometrial cancer, macrophage-driven inflammation may be intertwined with obesity-induced immune dysfunction, and exercise could help restore immune surveillance by reducing adiposity-related cytokine spillover [130, 131]. Cervical cancer, often driven by HPV-related immune evasion, is characterized by a dysregulated myeloid cell environment; regular physical activity may partially reverse this by promoting antigen presentation and cytotoxic lymphocyte activation [132, 133]. These tumor-specific immunologic features suggest that exercise may exert differential immune effects across gynecologic cancer subtypes, warranting tailored intervention strategies.
Exercise interventions have demonstrated the capacity to adjust the equilibrium between these phenotypes, promoting an M1 polarization that is hostile to tumors, thereby potentially impeding tumor advancement [134]. The decline in overall macrophage numbers within tumors subsequent to exercise regimens indicates a transition toward a less hospitable environment for cancerous cells [135, 136]. Neutrophils, integral to the immune system, display dual roles in tumor behavior contingent on their phenotype [137]. Exercise has been observed to diminish the buildup of neutrophils within tumors, though the exact consequences of this adjustment on tumor progression necessitate further clarification [135, 136]. The intricate impact of exercise on neutrophil function highlights the intricate nature of immune regulation through physical activity amidst cancer conditions. Natural killer (NK) cells are recognized as formidable agents of anti-tumor defense, and physical activity has demonstrated the ability to mobilize and stimulate these cells [138]. The increased penetration of NK cells into tumors subsequent to exercise interventions implies a reinforcement of the anti-tumor immune reaction within the localized tumor environment [139]. Additionally, the heightened activation of NK cells through exercise, relative to other immune pathways, highlights the promise of exercise as a supplementary therapy to augment immune surveillance against cancerous cells [140]. Exercise also reduces myeloid-derived suppressor cells (MDSCs), which inhibit T and NK cell function and contribute to immune escape [141].
Concerning adaptive immunity, exercise may impact CD8+ T cells, elevating their quantity, cytotoxic activity, and expression of activation markers [142]. This influence might be reliant on CXCL9/11-CXCR3 [143]. Moreover, exercise stimulates the movement and buildup of IL15Rα+ CD8 T cells within tumors, consequently bolstering anti-tumor immunity [144]. T cells, notably CD8+ cytotoxic T cells, are pivotal in adaptive immunity against cancer. Short bursts of exercise temporarily elevate the circulation of CD8+ T cells, possibly preparing the immune system for heightened tumor monitoring [145, 146]. Continuous exercise training has been linked to heightened infiltration of CD3+ and CD8+ T cells into tumors, suggesting a prolonged enhancement of the anti-tumor immune response [142, 147]. Nevertheless, the exact connections between exercise-induced alterations in T lymphocytes and tumor development require additional exploration [78, 148].
Regulatory T cells (Tregs), known for their immunosuppressive actions within the tumor microenvironment, present another aspect of immune modulation influenced by exercise in cancer contexts [149, 150]. Extended exercise regimes have demonstrated a capacity to diminish Treg presence within tumors, thereby possibly mitigating their immunosuppressive impact and nurturing an environment more supportive of anti-tumor immunity. The complex interaction between exercise and Tregs underscores the multifaceted character of immune adjustments induced by physical activity in cancer scenarios [151, 152]. Exercise programs have demonstrated potential in augmenting NK cell counts, enhancing their functionality, and modulating the ratio of Tregs in specific cancer types [153]. Moreover, exercise might trigger the movement of immune cells with anticancer properties within the tumor microenvironment (TME), facilitating the entry and effective functioning of NK and T cells, while diminishing the presence of immune cells that inhibit the immune response [126]. Notably, in ovarian cancer, where immunosuppressive tumor microenvironments are common, exercise-induced activation of cytotoxic T cells and NK cells may help overcome immune evasion [131]. In cervical cancer, where HPV-related immune dysfunction is central, exercise’s effect on T cell recruitment may support viral clearance and tumor control [154]. Hence, the exercise demonstrates diverse impacts on tumor progression by intricately adjusting immune function. By encouraging an anti-tumor macrophage profile and stimulating the mobilization and activation of NK cells, exercise readies the immune system for improved tumor monitoring and inhibition.
Harnessing exercise to mitigate physiological and psychological challenges in cancer patients
Physical activity has gained increasing recognition for its capacity to alleviate the physiological and psychological challenges associated with cancer and mitigate the adverse effects of therapy. Gynecological cancer patients, including those with ovarian, endometrial, and cervical cancers, face unique challenges throughout their cancer trajectory. These include sarcopenia, metabolic dysregulation, fatigue, pelvic floor dysfunction, and psychological distress, especially depression and anxiety, all of which can significantly impair overall prognosis and quality of life. For example, sarcopenia, often seen in cancer patients undergoing chemotherapy, results in muscle weakness and fat accumulation, compromising treatment tolerance and survival [155–158]. In ovarian cancer, muscle wasting and the systemic effects of chemotherapy-induced cachexia are common, and these are closely linked to poorer outcomes [159]. Exercise, especially resistance training and aerobic exercise, has been shown to improve muscle mass and functional capacity, even in advanced cancer stages. Several studies have supported these findings, demonstrating the positive effects of exercise on cancer patients, including those with advanced stages of various cancers. For instance, Rutkowska, Jastrzebski [160] conducted a randomized controlled trial (RCT) in Poland involving 30 lung cancer patients. The intervention group, which participated in a 4-week program of supervised physical exercise, including aerobics, strength training, and Nordic walking, showed significant improvements in the 6-min walking test (6MWT), standing up and walking test, and spirometry values such as FEV1 and FEV1/FVC. This study highlights that structured physical exercise during chemotherapy significantly enhances mobility, physical fitness, and lung capacity in patients with advanced cancer [160]. Porter, Carson [161] focused on breast cancer survivors in the United States and found that an 8-week yoga program (with 8 sessions per week) resulted in improvements in fatigue, pain interference, anxiety, and depression. The study demonstrates that exercise, specifically yoga, can improve functional capacity and alleviate common psychological symptoms like anxiety and depression, offering a holistic approach to cancer rehabilitation [161].
Schink, Herrmann [162] conducted an RCT in Germany with 131 cancer patients. The study showed that electrostimulation resistance training led to significant improvements in physical function and performance status, though quality of life and fatigue did not improve significantly. This suggests that electrostimulation may be an effective tool for improving physical function, especially in advanced cancer patients undergoing palliative chemotherapy [162]. Schmidt, Jonat [163] in Germany evaluated the effects of strength and aerobic training on breast cancer patients. The study indicated significant improvements in muscle strength and immune function following resistance training. This study underscores that strength and aerobic exercise can positively influence immune system functioning and reduce fatigue during chemotherapy for cancer patients [163]. Schuler, Hentschel [164] also conducted an RCT with cancer patients and demonstrated that physical exercise significantly reduced severe fatigue in patients with advanced cancer, further supporting the benefits of exercise for fatigue reduction in this population [164].
Dhillon, Bell [165] found no significant improvements in quality of life or fatigue for patients with advanced lung cancer undergoing palliative exercise therapy. However, this study highlights that physical activity interventions are still feasible and safe for patients with advanced-stage disease [165]. Tsianakas, Harris [166] conducted a 12-week self-initiated walking program for cancer patients and observed no significant differences in fatigue or quality of life, but noted the social benefits and increased adherence to the intervention, which is important for long-term patient engagement in physical activity [166]. Ligibel, Giobbie-Hurder [167] in the United States tested moderate-intensity aerobic activity for breast cancer patients, showing no significant improvements in quality of life. Despite this, the study emphasizes the need to explore alternative interventions for improving symptoms in metastatic breast cancer patients [167]. Henke, Cabri [168] found that a combination of aerobic and strength exercises significantly improved quality of life, physical functioning, and muscle strength in lung cancer patients undergoing chemotherapy, making a strong case for the integration of exercise in palliative care settings [168].
Jensen, Baumann [169] evaluated aerobic and strength training in advanced gastrointestinal cancer patients and found significant improvements in muscle strength and quality of life, demonstrating the positive impact of physical activity even in patients receiving palliative chemotherapy [169]. Cheville, Kollasch [170] tested strength training and aerobic exercises for colorectal and lung cancer patients, resulting in improvements in mobility, fatigue, and sleep quality, showing that supervised, structured exercise can be beneficial in advanced cancer [170]. Cormie, Newton [171] focused on prostate cancer patients with bone metastases and found that aerobic and strength training led to significant improvements in physical condition, activity levels, and lean mass, suggesting that exercise interventions tailored to specific cancer types can improve physical performance and overall well-being [171]. Litterini, Fieler [172] conducted a 10-week RCT on aerobic and strength exercises and reported significant improvements in physical performance, walking speed, and fatigue reduction in advanced cancer patients, further confirming the benefits of exercise for functional outcomes [172].
Hwang, Yu [173] in Taiwan tested high-intensity aerobic interval training for lung cancer patients and found improvements in aerobic capacity, reduced fatigue, and dyspnea, suggesting that intensive aerobic exercise can be effective for improving physical capacity and fatigue management [173]. Oldervoll, Loge [174] in Norway found that aerobic and strength exercises significantly improved gait, strength, and walking amplitude in advanced cancer patients, reinforcing the role of exercise interventions in improving physical function in terminal cancer patients. These studies collectively illustrate the potential of exercise interventions—whether aerobic, resistance training, or a combination of both—in addressing the physiological challenges associated with cancer and its treatment. They provide strong evidence for the feasibility and effectiveness of exercise in improving muscle mass, strength, mobility, and psychological symptoms like fatigue and depression. Furthermore, they highlight the importance of tailored exercise programs that address the specific needs of gynecological cancer survivors, including ovarian, endometrial, and cervical cancers [174].
Metabolic abnormalities, such as obesity and insulin resistance, are particularly prevalent in gynecological cancer survivors. These conditions exacerbate chronic inflammation, an established contributor to poor clinical outcomes. For instance, in endometrial cancer, obesity-driven chronic inflammation leads to insulin resistance and contributes to tumor progression [175]. Exercise can effectively counteract these metabolic disturbances by lowering circulating inflammatory markers (such as TNF-α, IL-6, and CRP) and improving insulin sensitivity. Structured exercise interventions such as resistance training (e.g., weightlifting or resistance bands) for 2–3 sessions per week have been shown to improve metabolic health, reduce systemic inflammation, and improve functional capacity [119, 176, 177].
Beyond physiological benefits, exercise may also play a key role in managing psychological symptoms, including depression, anxiety, and cognitive dysfunction, which are common side effects of cancer and its treatment [178]. One proposed mechanism involves tryptophan metabolism, which has been linked to depressive symptoms in cancer patients. Tryptophan is metabolized into kynurenine, a compound capable of crossing the blood–brain barrier and contributing to neurotoxicity and depression [179]. However, skeletal muscle can convert kynurenine into kynurenic acid, a non-neurotoxic metabolite that does not cross into the brain, thereby potentially protecting against depression. This detoxification mechanism is regulated by PGC-1α, a transcription factor whose expression is increased by endurance exercise. Studies in healthy individuals have shown that physical activity enhances kynurenine metabolism within muscle, but it remains unclear whether these effects are replicated in cancer patients or whether they are sufficient to reduce depressive symptoms in this population [180, 181].
While exercise prescription can vary, the current evidence supports moderate-intensity aerobic exercise (e.g., 150 min per week) alongside resistance training (e.g., 2–3 times per week) to effectively manage the physiological and psychological challenges faced by gynecological cancer survivors [182]. Specifically, pelvic floor exercises, important for cervical cancer survivors, can also reduce pelvic floor dysfunction and improve sexual health post-treatment. These exercise interventions should be personalized to the stage of cancer, treatment regimen, and specific challenges of each gynecological cancer type. In particular, early-stage ovarian cancer survivors may benefit more from higher-intensity aerobic exercise, while those with cervical cancer and pelvic radiation may require more focused pelvic floor rehabilitation and low-impact exercises.
The role of exercise in enhancing cancer treatment: from surgery to targeted therapy
Exercise is increasingly recognized as a valuable complement to various cancer treatment strategies, offering multiple benefits across the continuum of care. From diagnosis through post-treatment recovery, structured physical activity programs have demonstrated the potential to improve patient outcomes and overall well-being [183]. For numerous cancer types, surgical removal remains a key therapeutic approach, frequently offering the most favorable prospects for recovery. Nonetheless, the physiological strain resulting from surgery can incite systemic disturbances and localized harm, heightening the likelihood of postoperative issues and cancer reappearance [184]. Exercise is recognized as an important adjunct to cancer treatment, offering significant benefits in both the short and long term. In the context of preoperative treatment, physical activity has been shown to enhance cardiovascular fitness, reduce the risk of postoperative complications, and expedite recovery. This is particularly relevant for lung cancer patients undergoing surgery, where exercise may help mitigate the physiological strain that often accompanies surgical resection. Both short-term and long-term physical activity can regulate critical processes such as tumor spread, immune function, and metabolic stress. For instance, preoperative exercise programs have demonstrated promising effects on reducing complications like postoperative pulmonary issues, improving functional capacity, and enhancing immune responses, thereby lowering the chances of residual disease post-surgery. These findings underscore the potential of structured exercise interventions to improve postoperative outcomes, including reducing the length of hospital stays and improving physical recovery after cancer surgery [185]. Radiotherapy, another essential modality in cancer treatment, depends heavily on adequate tissue oxygenation for therapeutic efficacy [186]. Exercise has been shown to normalize tumor vasculature and enhance oxygen delivery, thereby alleviating hypoxia and improving the response to radiation therapy. These vascular changes may also support immune infiltration, amplifying the anti-tumor effects of radiotherapy. Although most of the research in this area has focused on cancers like prostate cancer, evidence from studies involving exercise during treatment for various cancers, including gynecological cancers, suggests similar benefits. For instance, a study involving resistance training during radiotherapy for patients with spinal bone metastases demonstrated significant improvements in bone density and physical function. In this study, patients engaged in resistance training, with sessions lasting 60 min, twice a week, and showed a substantial increase in bone density compared to those receiving passive physical therapy, indicating the potential for such interventions in improving treatment responses [187]. Furthermore, an aerobic exercise regimen, such as moderate-intensity walking, has shown positive outcomes for fatigue management and physical function in cancer patients with spinal bone metastasis undergoing radiotherapy. Specifically, patients participating in this regimen demonstrated significant improvements in walking capacity, suggesting that structured physical activity programs can play a crucial role in optimizing cancer treatment, particularly in improving functional recovery and reducing fatigue during and post-treatment [188].
In the context of chemotherapy, targeted therapy, and immunotherapy, exercise is emerging as a promising adjunct. Preclinical studies suggest that exercise can enhance drug delivery to tumors, increase immune cell infiltration, and potentiate the therapeutic efficacy of systemic treatments [84]. Exercise is emerging as a promising adjunct in cancer treatment, particularly in enhancing the efficacy of chemotherapy, targeted therapy, and immunotherapy. While much of the data comes from cancers like breast and pancreatic cancer, the relevance to ovarian, endometrial, and cervical cancers is clear, given similar mechanisms of tumor progression. Preclinical studies show that moderate-intensity aerobic exercise, such as walking or cycling, can improve drug delivery to tumors, enhance immune cell infiltration, and potentiate treatment efficacy [83]. For example, voluntary wheel running in mice bearing breast cancer improved doxorubicin's effects by altering the tumor microenvironment and reducing hypoxia while protecting against cardiotoxicity [189]. Similarly, moderate aerobic exercise in pancreatic cancer models improved tumor vasculature, enhancing chemotherapy outcomes [190]. Clinical data reinforce these findings, with observational studies linking regular physical activity to lower recurrence rates and improved overall survival among cancer patients [191]. In a study of high-risk breast cancer patients, those who engaged in physical activity both before and after diagnosis demonstrated improved outcomes, with physical activity prescribed according to the Physical Activity Guidelines. The exercise prescription typically involved moderate-intensity aerobic exercise, such as brisk walking or cycling, for at least 150 min per week, alongside strength training exercises targeting major muscle groups [192]. Similarly, a study of supervised exercise programs in oncology patients bearing different cancer types showed significant benefits, with participants following a structured regimen of aerobic and resistance training. This regimen included activities like walking, stationary cycling, and strength training sessions two to three times per week, with progressive intensity based on individual capabilities. These findings highlight the potential of structured physical activity in enhancing treatment outcomes and supporting survivorship in cancer patients, including those with ovarian, endometrial, and cervical cancers [193]. However, individual variability in treatment response underscores the need for personalized exercise prescriptions tailored to a patient’s cancer type, treatment phase, and physical condition. While targeted therapy and immunotherapy represent important advances in oncology, our understanding of how exercise interacts with these newer modalities remains limited. The lack of robust clinical trials in this area hinders the systematic integration of exercise into these treatment paradigms. Nonetheless, preliminary evidence suggests that exercise may enhance immune responsiveness, improve drug pharmacokinetics, and increase patients'tolerance to side effects, making it a valuable supportive therapy [84, 194]. As such, exercise holds substantial potential as an adjunct across diverse cancer treatment approaches. By leveraging the synergistic benefits of physical activity and medical therapy, clinicians may be able to improve therapeutic outcomes, reduce treatment-related toxicity, and support long-term survivorship [195, 196]. However, ongoing research is essential to elucidate the underlying mechanisms and optimize exercise protocols for integration into standard cancer care.
Physical activity interventions in gynecological cancer survivors: clinical trials and psychosocial rehabilitation
A growing body of research supports the use of physical activity to address the physical, psychological, and functional challenges faced by gynecological cancer survivors. Given the heterogeneity across ovarian, endometrial, and cervical cancers in terms of pathology, treatment, and survivorship trajectories, exercise interventions must be individualized accordingly. However, many existing programs are not sufficiently individualized by cancer type, despite clear differences in treatment regimens, symptomatology, and recovery patterns. Notably, interventions that align with specific cancer-related impairments, such as sexual health counseling for cervical cancer survivors or fatigue-targeted aerobic programs for ovarian cancer patients, may improve both adherence and clinical outcomes [197, 198]. This section synthesizes the clinical evidence for each cancer type, contextualizing key findings from the literature and highlighting unmet needs. A summary of interventions is also provided in Table 1, with additional insights illustrated in Fig. 1.
Table 1.
An overview of intervention studies in gynecological cancer survivors: physical activity and behavioral outcomes
| Type of study | Population | Physical activity | Research outcomes | Ref |
|---|---|---|---|---|
| A prospective randomized controlled trial | Overweight and obese endometrial cancer survivors | The lifestyle intervention (LI) involved gradually increasing walking or other aerobic activity to 5 days a week for at least 45 min, with the encouragement of long-term changes. Participants were provided with pedometers for self-monitoring and feedback | Over the course of one year, implementing a lifestyle intervention program among obese endometrial cancer survivors has shown feasibility and the potential for sustained behavior modification and weight reduction | [199] |
| A prospective randomized controlled trial | Overweight and obese survivors of endometrial cancer | The intervention program followed a phased approach using strategies outlined by social cognitive theory, with sessions covering nutrition and exercise occurring weekly for six weeks, biweekly for one month, and monthly for three months | The preliminary lifestyle program did not impact quality of life or depressive symptoms but demonstrated enhancements in self-efficacy and certain dietary habits | [200] |
| Single-blind, randomized controlled trial | gynecological cancer patients (stage I–III) | Participants in the PA group engaged in a 12-week, home-based, moderate-intensity physical activity intervention, including walking and strengthening exercises, facilitated by a physiotherapist | Implementing a program aimed at modifying physical activity behavior among survivors of gynecological cancer appears viable based on the adherence of participants to the program and its assessment. Furthermore, the intervention shows promising outcomes in reducing fatigue levels | [201] |
| Non-randomized phase 2 trial | Ovarian, peritoneal, or fallopian tube cancer | A walking intervention was implemented before and after chemotherapy. Participants received personalized guidance from an exercise physiologist and recorded their activity in logbooks. Weekly sessions were conducted to monitor progress and adjust targets | The encouraging initial findings indicated that the walking program for ovarian cancer patients undergoing chemotherapy was both safe and feasible, with high levels of acceptability | [202] |
| A controlled randomized trial | Endometrial cancer survivors (ECS) | Participants were split into two groups: one receiving a 6-month lifestyle intervention focusing on nutrition, exercise, and behavior modification (SUCCEED), aiming for a 5% weight loss; the other receiving usual care | Weight reduction and altering behaviors can be attained among overweight and obese ECS. Nevertheless, the clinical significance of these alterations remains uncertain and necessitates a more extensive trial with extended monitoring | [203] |
| A prospective, randomized, controlled trial | Women with gynecological cancer who underwent radical hysterectomy and pelvic lymph node dissection | 4-week supervised pelvic floor rehabilitation program (PFRP) including biofeedback and core exercises; Control: Provided with informative leaflets for home-based pelvic floor exercises | Findings indicate that the PFRP led to enhancements in pelvic floor dysfunction and quality of life (QoL) among patients with gynecological cancer | [8] |
| A randomized controlled trial | Patients diagnosed with stages III–IV lung or gynecological cancer | The intervention involved supportive talks between patients and trained nurses, focusing on mutual understanding, emotional engagement, and well-being. A 5-day residential rehabilitation course was also offered, emphasizing physical activity, communication strategies, and lifestyle education | The preliminary investigation indicated challenges in executing an RCT for a psychosocial rehabilitation program aimed at pairs. Issues concerning participant recruitment and attrition must be tackled effectively | [204] |
| A randomized controlled trial | Overweight or obese women with Stage I or II endometrial cancer (EC) | Lifestyle intervention group (SUCCEED) received 6 months of counseling on nutrition, exercise, and behavioral modification, including group and individual sessions | This study suggests the potential effectiveness of specific interventions aimed at enhancing self-efficacy, consequently leading to improvements in BMI | [205] |
| Cross-sectional study | Gynecologic cancer survivors (GCS) diagnosed with cervical, endometrial, or ovarian cancer | Participants completed a survey to report their engagement in aerobic and strength exercises. Aerobic exercise was assessed by considering the frequency and duration of moderate and vigorous activities, while strength exercise was determined based on activities such as weightlifting, sit-ups, and push-ups | Physical activity represents a controllable aspect of daily life linked to posttraumatic growth among GCS. It is essential to conduct randomized controlled trials to examine the impact of exercise programs on posttraumatic growth in this specific group | [206] |
| Cross-sectional study | GCS diagnosed with cervical, endometrial, or ovarian cancer | Extreme/adventure activities, involving high physical demands like mountaineering, rafting, and rock climbing, were assessed by asking participants if they had engaged in such activities in the past year | GCS who adjusts the frequency, mode, or intensity of their exercise routines following diagnosis might have a higher likelihood of encountering posttraumatic growth | [207] |
| Randomized controlled trial | GCS with cervical, endometrial, or ovarian cancer | 8-week wall climbing program, twice weekly, 120 min/session, supervised by certified instructors | This phase I/II study suggests that wall climbing may enhance posttraumatic growth and mental health in GCS but could also cause bodily discomfort and reduced physical health. Larger trials are needed to fully understand its benefits and risks in GCS | [208] |
| Retrospective cohort | Endometrial cancer survivors (ECS) | Personalized exercise plans to gradually achieve 30 min of moderate exercise per day | A newly discovered association between participating in an exercise program and enhanced sexual well-being among ECS is an original discovery. This observation implies that physical activity may play a part in enhancing the sexual well-being of ECS, a hypothesis our team is currently investigating in a more extensive upcoming study | [209] |
| Single-blind, randomized controlled trial | Obese women (BMI ≥ 30 kg/m2) diagnosed with endometrial cancer | Participants engaged in two weekly sessions involving 30 min of group counseling and 60 min of moderate to vigorous physical activity. These sessions included a 5-min warm-up, 25 min of dance fitness, 20 min of resistance training, and a 10-min cool-down. Moreover, participants were encouraged to walk for a minimum of 90 min per week outside of these sessions | The findings exhibit encouraging impacts on various parameters, underscoring the necessity for validation through a more extensive randomized controlled trial employing enhanced recruitment methodologies | [210] |
| A randomized pilot study | Gynecologic cancer survivors including cervical, endometrial, or ovarian cancer patients | 4-week CR program: supervised exercises in a rehabilitation room for 2 weeks, followed by home-based exercises including stretching, strengthening, core stability, and aerobic exercises. Intensity: 40–76% of predicted max heart rate | Sophisticated rehabilitation enhances physical function, reduces fatigue, and enhances muscular strength in individuals with unilateral lower-limb lymphedema post-gynecologic cancer surgery, without exacerbating edema | [211] |
| A nonrandomized pre-/post-test pilot study | Women with advanced-stage ovarian cancer | Participants were assigned a 26-week high-dose home-based exercise program, targeting 225 min per week. The primary exercise was walking, with options for cycling and using cardiovascular training equipment | An exercise regimen of 225 min per week was found to be practical and well-received among individuals with ovarian cancer. Over the course of the 26-week program, participants demonstrated notable enhancements in their physical activity levels | [212] |
| A randomized controlled trial | Ovarian cancer survivors | Participants engaged in a six-month home-based, moderate-intensity aerobic exercise program, primarily brisk walking, for 150 min per week | A six-month exercise program conducted at home and delivered via telephone, focusing mainly on brisk walking, was linked to enhanced physical Health-Related Quality of Life (HRQOL) among women diagnosed with ovarian cancer | [213] |
| Mixed between-within-subject design | Gynecologic cancer survivors | Participants were engaged in a 16-week physical training program consisting of warm-up, aerobic and strength training, flexibility exercises, and relaxation. Music was tailored to match the intensity of each exercise | Engaging in both aerobic and resistance training twice weekly over 16 weeks has been shown to enhance V̇O2 peak and maximal strength among women who have undergone treatment for gynecological cancer | [214] |
| Subset analysis from a larger randomized controlled trial (WALC) | Ovarian cancer survivors | Subjects participated in a six-month, home-based aerobic exercise regimen, predominantly brisk walking, averaging 150 min weekly | The investigation revealed that involvement in our home-based exercise program did not produce any negative impact on lower limb lymphedema (LLL), aligning with earlier studies conducted on breast cancer | [215] |
| One-group, pre-post design | Endometrial cancer survivors (ECS) | Each participant received a tailored exercise plan based on initial fitness assessments, emphasizing gradual increases in moderate-intensity walking to achieve 30 min of activity on most days | The study indicates that enhancing physical activity levels could potentially alleviate certain physical quality of life issues faced by survivors of endometrial cancer, who are known to be at an elevated risk of such challenges | [216] |
| Observational cohort study | Endometrial cancer survivors (ECS) | Physical activity levels were assessed before and after diagnosis using a modified questionnaire, covering occupational, household, and recreational activities, including sedentary behavior | Engaging in recreational physical activities, particularly after diagnosis, is linked to enhanced survival among individuals who have experienced endometrial cancer | [217] |
| A randomized controlled trial | Ovarian cancer survivors | Participants were randomly assigned to either a six-month exercise program or an attention-control arm. Those in the exercise program were guided to engage in home-based moderate-intensity aerobic exercise for 150 min weekly with support from a certified cancer exercise trainer via weekly phone calls | Survivors of ovarian cancer can engage in exercise at levels recommended by health guidelines, and participating in regular exercise was linked to a notable decrease in symptoms of depression | [218] |
| A Pilot randomized controlled trial | Endometrial cancer survivors | A 10-week home-based resistance training program compared to a waitlist control. The intervention group received coaching and materials for twice-weekly sessions targeting major muscle groups with resistance bands and dumbbells. The control group received print materials about healthy survivorship and exercise materials at week 10 | Muscle-strengthening exercises performed at home resulted in significant enhancements in three out of seven physical function evaluations. These enhancements in fitness were noticeable within a brief duration of 10 weeks | [219] |
| Qualitative study | Endometrial cancer survivors (ECS) | Twice-weekly 90-min sessions, including 30 min of group counseling and 60 min of multi-modal exercise. Participants were also instructed to increase home-based walking to 90 min per week | This study highlighted the significance of autonomy and the supportive, enjoyable, and communal atmosphere in overcoming fatigue, pain, and familial challenges to adhere to the program. They expressed various health advantages and desired a more comprehensive program | [220] |
Endometrial cancer
In 2022, endometrial cancer ranked fourth among cancers affecting women, with an estimated 66,000 new cases reported [221]. With its comparatively favorable survival rates, endometrial cancer survivors constitute the second-largest group of female cancer survivors, totaling nearly 900,000 individuals in the United States alone [222]. Moreover, the prevalence of endometrial cancer is projected to rise further owing to the increasing rates of obesity, a key contributing factor [223]. More than half of women diagnosed with early-stage endometrial cancer succumb to concurrent conditions like diabetes and obesity, yet patient awareness regarding the association between endometrial cancer and comorbidities remains limited [224]. Endometrial cancer is strongly associated with obesity and chronic inflammation, which play a pivotal role in tumor progression. The high levels of circulating pro-inflammatory cytokines such as TNF-α and IL-6 contribute to the insulin resistance and hormonal dysregulation that drive endometrial carcinogenesis. These mechanisms are exacerbated by obesity, which is a significant risk factor for the disease [101, 110, 193]. Exercise has been proposed as a modulator of these pathways, reducing systemic inflammation, improving metabolic health, and potentially impacting tumor progression and therapeutic outcomes in endometrial cancer survivors [225].
Several clinical studies have investigated the effects of exercise on endometrial cancer survivors, showing positive outcomes in various domains. The SUCCEED program, a randomized controlled trial integrating aerobic exercise, nutrition education, and behavior modification over 6 months, demonstrated significant reductions in weight and BMI, improved physical activity levels, and better dietary habits among participants [203]. Additionally, a related trial indicated that the same program enhanced self-efficacy and motivation for long-term lifestyle change, though effects on quality of life and depressive symptoms were less pronounced [205]. Furthermore, physical activity has been shown to have a beneficial effect on sexual health in endometrial cancer survivors. A prospective study found that increased physical activity was associated with improvements in sexual interest and function, particularly among those receiving personalized exercise support. This suggests that tailored exercise interventions could enhance both psychosocial and physical well-being in this population [209]. Community-based interventions have also yielded positive outcomes. A 12-week group program combining dance fitness, resistance training, and walking improved quality of life and walking endurance while reducing waist circumference [210]. While changes in self-reported activity levels were modest, participants emphasized the social and motivational benefits of group settings. A qualitative study of another 12-week program revealed that social support, goal-setting, and instructor encouragement were key facilitators of adherence, while barriers included fatigue, caregiving responsibilities, and lack of time [220]. In terms of physical capacity, home-based resistance training programs have been effective in improving strength and agility. A 10-week pilot trial showed functional gains even in the absence of significant changes in body composition or inflammatory markers [219].
While exercise interventions show promise in improving physical function and quality of life, long-term follow-up studies are needed to assess the sustainability of these benefits. Additionally, further randomized controlled trials with larger sample sizes and diverse populations are required to confirm the clinical efficacy of exercise in endometrial cancer survivors. One major limitation in the current research is the lack of focus on personalized interventions, especially considering comorbidities like diabetes and obesity, which are common in this patient group. Furthermore, there is a need for targeted exercise protocols that address the unique psychosocial challenges of survivors, particularly those related to sexual health and mental well-being.
Ovarian cancer
Globally, approximately 313,000 women receive a diagnosis of ovarian cancer annually, ranking ovarian cancer as the eighth most prevalent cancer type in women. The overall survival rate for ovarian cancer within 5 years ranges from 30 to 40%, primarily due to frequent late-stage diagnoses as reported by the International Federation of Gynaecology and Obstetrics (FIGO). Managing ovarian cancer, from diagnosis to treatment, frequently leads to diminished physical capabilities, heightened fatigue levels, and a decline in Quality of Life (QoL) [226]. These factors underscore the importance of supportive interventions that can aid recovery and improve long-term outcomes. The tumor microenvironment in ovarian cancer is characterized by immune suppression, inflammatory ascitic fluid, and tumor-associated macrophages, all of which contribute to tumor progression and chemoresistance [225]. The chronic inflammation associated with ovarian cancer, driven by pro-inflammatory cytokines and oxidative stress, plays a critical role in metastasis and treatment resistance [227]. Exercise may influence this microenvironment by modulating immune function, reducing systemic inflammation, and improving oxidative stress responses, thereby enhancing chemotherapy efficacy and overall survival.
Multiple studies have assessed the feasibility and efficacy of exercise programs introduced during and after chemotherapy. A non-randomized walking intervention implemented before and during first-line chemotherapy demonstrated high adherence, with improvements in fatigue, functional capacity, and ovarian cancer–specific quality of life [202]. Importantly, participants maintained high relative dose intensity, suggesting that exercise does not compromise treatment completion. Another study targeting patients undergoing second- or third-line chemotherapy for recurrent ovarian cancer employed a 12-week individualized regimen combining aerobic, resistance, and balance training. This intervention led to clinically meaningful improvements in fatigue, sleep quality, and functional well-being without any adverse events, further supporting the safety and acceptability of exercise during active treatment [228]. Longer-term interventions have also demonstrated positive results. In a six-month randomized controlled trial, a home-based aerobic exercise program consisting of brisk walking for 150 min per week significantly improved physical health-related quality of life (HRQOL) and reduced cancer-related fatigue among adherent participants [213]. Interestingly, individuals with lower baseline HRQOL experienced the greatest gains. The WALC trial also confirmed that moderate-intensity exercise does not exacerbate lower-limb lymphedema (LLL), a common concern among ovarian cancer survivors [215]. Beyond aerobic training, resistance exercise has shown benefits in muscle strength and immune modulation. A 12-week program involving twice-weekly resistance sessions significantly improved strength and was associated with changes in T cell subsets and immune markers, indicating potential systemic benefits [229]. A separate pre/post study of a 26-week high-dose walking program (225 min/week) reported strong adherence and sustained increases in physical activity, even among patients with advanced-stage disease [212].
Despite the positive findings, there remain several limitations in the current exercise research for ovarian cancer survivors. While exercise interventions have proven beneficial, larger, randomized trials with long-term follow-up are required to confirm the durability of these benefits and to establish how exercise can specifically impact survival rates, chemoresistance, and recurrence in ovarian cancer patients. Additionally, personalized exercise prescriptions tailored to individual treatment regimens and symptom profiles are still under-researched. Future studies should aim to focus on immune-related endpoints and investigate how exercise might modulate immune function, particularly in chemotherapy-resistant ovarian cancer. Moreover, addressing psychosocial barriers such as fatigue, psychological distress, and caregiving responsibilities will be essential for improving adherence and long-term outcomes.
Cervical and mixed gynecologic cancers
Despite being one of the most common gynecologic cancers globally, cervical cancer remains underrepresented in clinical exercise research. Most available studies include cervical cancer survivors within mixed cohorts, limiting the ability to derive cancer-specific recommendations [201, 208]. Cervical cancer is primarily driven by persistent HPV infection, which leads to chronic inflammation and immune evasion, significantly contributing to tumor progression. The inflammatory response associated with HPV-related cancers promotes a microenvironment that is conducive to immune suppression and carcinogenesis [102, 104]. Exercise may modulate these immune responses, potentially enhancing viral clearance and improving immune function, which is critical in the management of cervical cancer. Additionally, radiation therapy, surgical interventions, and chemotherapy in cervical cancer survivors often cause pelvic floor dysfunction, sexual health issues, and nerve damage, which exercise can help alleviate by improving muscle strength and psychosocial well-being [102, 230].
Preliminary findings, however, suggest that cervical cancer survivors may benefit from targeted interventions. In a single-blind randomized controlled trial involving a mixed group of gynecological cancer patients, a 12-week home-based program focusing on walking and strength exercises reduced fatigue and modestly improved affective outcomes [201]. Another study examined the effects of a pelvic floor rehabilitation program (PFRP) following radical hysterectomy and lymphadenectomy, reporting significant improvements in pelvic muscle strength, sexual function, and quality of life [8]. Psychosocial rehabilitation has also received attention in this context. A wall climbing program for gynecologic cancer survivors improved posttraumatic growth and mental health, though effects on physical function were variable [208]. Similarly, a randomized controlled trial evaluating a home-based program among cervical, endometrial, and ovarian cancer survivors found moderate improvements in fatigue and positive affect, with adherence rates around 50% [201]. The current evidence on exercise interventions in cervical cancer survivors is limited, particularly in terms of targeted exercise protocols for this population. While interventions like pelvic floor rehabilitation and home-based exercise programs show promise, these studies are hindered by small sample sizes, heterogeneity in design, and a lack of cancer-specific protocols. Future trials must develop and test cervical cancer-specific exercise protocols, particularly addressing the unique side effects of pelvic radiation, nerve damage, and sexual dysfunction. Additionally, longitudinal studies with larger sample sizes and more homogenous cohorts are needed to confirm the long-term benefits of exercise in improving physical and psychosocial outcomes. Emphasis should also be placed on personalized exercise prescriptions that consider the unique treatment regimens and psychosocial challenges faced by cervical cancer survivors.
General psychosocial and rehabilitation interventions
Beyond cancer-type-specific exercise programs, a significant aspect of survivorship care for gynecological cancer patients involves addressing psychosocial and functional needs that affect both physical and emotional well-being. Several interventions have sought to integrate exercise and rehabilitation programs with psychosocial support to help gynecologic cancer survivors better cope with treatment-related challenges and improve their overall quality of life. These programs aim not only to enhance physical fitness but also to address the emotional and psychological distress that often accompanies cancer treatment and recovery.
For instance, one randomized controlled trial in Denmark evaluated a comprehensive program that combined group discussions, peer support, and physical activity education. Participants reported improved well-being, particularly in terms of mental health and coping with the psychological impacts of cancer, though the study's methodological limitations and high attrition rates affected the robustness of its conclusions [204]. Another rehabilitation study incorporating complex decongestive therapy (CDT) alongside home-based exercise showed positive results in improving fatigue, leg strength, and mobility, particularly among patients with secondary lymphedema, without exacerbating edema [211]. Furthermore, exercise programs that combine aerobic and resistance training have consistently demonstrated benefits in muscle strength, cardiorespiratory fitness, and fatigue reduction in diverse cancer survivor populations [214]. A Norwegian study involving a 16-week intervention aligned with ACSM guidelines found that improvements in VO₂ peak and muscular endurance were sustained even one year after program completion. These findings underline the long-term benefits of exercise in enhancing functional outcomes and reducing cancer-related fatigue during survivorship [214]. Equally important are the behavioral support mechanisms incorporated into many exercise programs, such as goal setting, peer engagement, and fostering autonomy in survivors. One trial highlighted that the most critical facilitators of adherence and perceived benefit were a supportive group environment and a shared sense of purpose [220]. This suggests that psychosocial factors are integral to exercise adherence and the successful integration of exercise into survivorship care for gynecologic cancer patients.
These studies collectively emphasize that exercise interventions, when combined with psychosocial support, are crucial for improving physical health while also addressing the psychological and emotional burdens faced by gynecologic cancer survivors. Therefore, integrating flexible, behaviorally supported, and symptom-specific rehabilitation strategies into routine oncology care is essential to optimize both physical and mental health outcomes in these patients.
Limitations and future perspectives
Despite significant progress in understanding the impact of physical activity and exercise on gynecological cancer outcomes, there remain notable limitations in the existing research that warrant further exploration. These limitations primarily stem from gaps in the literature and the scope of the current review itself. A key limitation within the literature is the lack of preclinical and animal studies that investigate the mechanistic underpinnings of how exercise influences gynecological cancer progression and treatment responses. While clinical and epidemiological studies offer valuable insights, they often fail to delve deeply into the biological pathways involved, such as how exercise modulates the immune response, inflammation, or tumor microenvironment in these specific cancers. Future research should focus on preclinical models that explore these mechanisms, especially in gynecologic malignancies like ovarian, endometrial, and cervical cancers. Such studies will be instrumental in informing targeted exercise interventions that address cancer-specific molecular features.
Another important gap in the literature is the disproportionate lack of studies specifically addressing cervical cancer. Compared to ovarian and endometrial cancers, cervical cancer remains underrepresented in mechanistic and interventional exercise oncology research. This imbalance hampers the development of cervical cancer–specific exercise guidelines and limits our understanding of how exercise may influence HPV-related oncogenesis, immune responses, and treatment-related complications unique to this cancer type. Future studies must prioritize cervical cancer in both clinical trials and preclinical investigations to close this gap and provide more robust exercise recommendations tailored to this subtype. Additionally, the current review itself highlights a gap in molecular-level studies in human subjects, particularly regarding how exercise affects the molecular dynamics of gynecological cancer progression and response to treatment. The lack of such studies limits our ability to fully understand how exercise can modulate tumor biology, immune function, and overall patient outcomes. Future research should include molecular studies that explore the effects of exercise on immune-related endpoints and genetic markers of response and resistance, especially in the context of ovarian and endometrial cancers.
To address these gaps, future research directions must be more multidisciplinary, integrating clinical, epidemiological, preclinical, and molecular investigations to provide a comprehensive understanding of exercise’s role in gynecological cancers. These studies should also focus on immune-related endpoints to better understand how exercise may enhance immune function and potentially synergize with immunotherapy in gynecologic oncology. Longitudinal studies with larger sample sizes and diverse patient populations will be critical in validating findings and elucidating the impact of exercise on long-term survival, recurrence, and quality of life in gynecological cancer survivors. Moreover, subgroup analyses will be necessary to explore differential responses to exercise interventions based on tumor histology, stage, molecular subtype, and patient demographics. Such analyses will provide personalized, cancer-specific exercise prescriptions that can be tailored to the individual’s treatment regimen and functional status. The growing body of research should also consider the unique barriers faced by women with gynecological cancers, including caregiving roles, psychological distress, and fatigue, which can impact program adherence. Future interventions must be culturally appropriate, accessible, and flexible enough to address these barriers and enhance patient engagement.
In clinical practice, future guidelines must move beyond generalized recommendations and incorporate cancer-specific and stage-specific parameters. Exercise prescriptions should account for treatment-related toxicities, organ function impairments, and other challenges unique to each gynecologic malignancy. Clinical implementation science plays a pivotal role here, identifying barriers to integrating exercise into routine oncology care and ensuring its feasibility in real-world settings. Embedding exercise professionals within multidisciplinary oncology teams and developing referral pathways for individualized exercise support will be crucial for bridging the gap between research and clinical practice. Finally, digital health platforms and tele-exercise interventions should be explored as scalable tools to reach underserved populations, particularly in rural or resource-limited settings. Virtual and home-based exercise programs offer flexibility for patients to participate in tailored interventions despite geographical or social constraints.
Ultimately, the next generation of research must not only refine mechanistic insights and optimize exercise intervention protocols but also ensure that exercise becomes a core component of holistic cancer care. By addressing the gaps in molecular understanding, clinical application, and real-world implementation, future research can fulfill the promise of exercise as a powerful tool for prevention, symptom management, and survivorship enhancement in women affected by gynecological cancers.
Conclusions
In conclusion, the burgeoning body of evidence underscores the pivotal role of physical activity and exercise in the prevention, treatment, and survivorship of gynecological cancers. Epidemiological data consistently demonstrate the protective effects of regular physical activity against gynecological cancer incidence and mortality. Moreover, intervention studies highlight the potential for exercise regimens to enhance treatment tolerance, mitigate treatment-related side effects, and improve quality of life among gynecological cancer survivors. Mechanistic insights suggest that physical activity and exercise may exert their effects through modulation of inflammation, hormonal balance, immune function, and metabolic pathways implicated in cancer pathogenesis. However, critical knowledge gaps persist, and further research is required to elucidate the underlying mechanisms and optimize intervention strategies. By adopting a multidimensional research framework and addressing these gaps, the full therapeutic potential of lifestyle interventions may be realized to improve clinical outcomes and survivorship. Emphasis should also be placed on advancing molecular and translational research to bridge mechanistic understanding with clinical application, thereby informing more precise and biologically grounded exercise prescriptions in gynecologic oncology.
Acknowledgements
Not applicable.
Authors’ contributions
YW and MM contributed in data collection and manuscript drafting. Both authors approved the final version for submission. All authors reviewed the manuscript."
Funding
Not applicable.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Yubing Wang, Email: wyb_1258@163.com.
Melika Malek, Email: melikamalek862@gmail.com.
References
- 1.Shrestha P, Poudyal B, Yadollahi S, Wright DE, Gregory AV, Warner JD, et al. A systematic review on the use of artificial intelligence in gynecologic imaging–background, state of the art, and future directions. Gynecol Oncol. 2022;166:596–605. [DOI] [PubMed] [Google Scholar]
- 2.Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, et al. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49. [DOI] [PubMed] [Google Scholar]
- 3.Chandra A, Pius C, Nabeel M, Nair M, Vishwanatha JK, Ahmad S, et al. Ovarian cancer: Current status and strategies for improving therapeutic outcomes. Cancer Med. 2019;8:7018–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Salani R, Khanna N, Frimer M, Bristow RE, Chen L-M. An update on post-treatment surveillance and diagnosis of recurrence in women with gynecologic malignancies: Society of Gynecologic Oncology (SGO) recommendations. Gynecol Oncol. 2017;146:3–10. [DOI] [PubMed] [Google Scholar]
- 5.Holcakova J, Bartosik M, Anton M, Minar L, Hausnerova J, Bednarikova M, et al. New trends in the detection of gynecological precancerous lesions and early-stage cancers. Cancers. 2021;13:6339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Penson RT, Wenzel LB, Vergote I, Cella D. Quality of life considerations in gynecologic cancer. Int J Gynecol Obstet. 2006;95:S247–57. [DOI] [PubMed] [Google Scholar]
- 7.Prue G, Allen J, Gracey J, Rankin J, Cramp F. Fatigue in gynecological cancer patients during and after anticancer treatment. J Pain Symptom Manage. 2010;39:197–210. [DOI] [PubMed] [Google Scholar]
- 8.Yang EJ, Lim J-Y, Rah UW, Kim YB. Effect of a pelvic floor muscle training program on gynecologic cancer survivors with pelvic floor dysfunction: a randomized controlled trial. Gynecol Oncol. 2012;125:705–11. [DOI] [PubMed] [Google Scholar]
- 9.Schofield C, Newton RU, Cohen PA, Galvão DA, McVeigh JA, Mohan GR, et al. Health-related quality of life and pelvic floor dysfunction in advanced-stage ovarian cancer survivors: associations with objective activity behaviors and physiological characteristics. Support Care Cancer. 2018;26:2239–46. [DOI] [PubMed] [Google Scholar]
- 10.Asakitogum DA, Nutor JJ, Pozzar R, Hammer M, Miaskowski C. Systematic Review of the Literature on Multiple Co-occurring Symptoms in Patients Receiving Treatment for Gynecologic Cancers. Semin Oncol Nurs. 2024;40: 151572. [DOI] [PubMed] [Google Scholar]
- 11.Barcellini A, Dominoni M, Dal Mas F, Biancuzzi H, Venturini SC, Gardella B, et al. Sexual Health Dysfunction After Radiotherapy for Gynecological Cancer: Role of Physical Rehabilitation Including Pelvic Floor Muscle Training. Front Med. 2022;8:2021. 10.3389/fmed.2021.813352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lin KY, Frawley HC, Denehy L, Feil D, Granger CL. Exercise interventions for patients with gynaecological cancer: a systematic review and meta-analysis. Physiotherapy. 2016;102:309–19. [DOI] [PubMed] [Google Scholar]
- 13.Beesley VL, Eakin EG, Janda M, Battistutta D. Gynecological cancer survivors’ health behaviors and their associations with quality of life. Cancer Causes Control. 2008;19:775–82. [DOI] [PubMed] [Google Scholar]
- 14.Smits A, Smits E, Lopes A, Das N, Hughes G, Talaat A, et al. Body mass index, physical activity and quality of life of ovarian cancer survivors: time to get moving? Gynecol Oncol. 2015;139:148–54. [DOI] [PubMed] [Google Scholar]
- 15.Basen-Engquist K, Scruggs S, Jhingran A, Bodurka DC, Lu K, Ramondetta L, et al. Physical activity and obesity in endometrial cancer survivors: associations with pain, fatigue, and physical functioning. Am J Obstet Gynecol. 2009;200:288. e1-e8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Koelwyn GJ, Zhuang X, Tammela T, Schietinger A, Jones LW. Exercise and immunometabolic regulation in cancer. Nat Metab. 2020;2:849–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Thyfault JP, Bergouignan A. Exercise and metabolic health: beyond skeletal muscle. Diabetologia. 2020;63:1464–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Morrison KS, Paterson C, Coltman CE, Toohey K. What are the Barriers and Enablers to Physical Activity Participation in Women with Ovarian Cancer? A Rapid Review of the Literature. Semin Oncol Nurs. 2020;36:151069. [DOI] [PubMed] [Google Scholar]
- 19.Farrokhzadi L, Dhillon HM, Goumas C, Young JM, Cust AE. Physical Activity Correlates, Barriers, and Preferences for Women With Gynecological Cancer. Int J Gynecol Cancer. 2016;26:1530–7. [DOI] [PubMed] [Google Scholar]
- 20.Kim S, Han J, Lee MY, Jang MK. The experience of cancer-related fatigue, exercise and exercise adherence among women breast cancer survivors: Insights from focus group interviews. J Clin Nurs. 2020;29:758–69. [DOI] [PubMed] [Google Scholar]
- 21.Blaney J, Lowe-Strong A, Rankin J, Campbell A, Allen J, Gracey J. The Cancer Rehabilitation Journey: Barriers to and Facilitators of Exercise Among Patients With Cancer-Related Fatigue. Phys Ther. 2010;90:1135–47. [DOI] [PubMed] [Google Scholar]
- 22.Demark-Wahnefried W, Peterson B, McBride C, Lipkus I, Clipp E. Current health behaviors and readiness to pursue life-style changes among men and women diagnosed with early stage prostate and breast carcinomas. Cancer. 2000;88:674–84. [PubMed] [Google Scholar]
- 23.Anderson AS, Steele R, Coyle J. Lifestyle issues for colorectal cancer survivors—perceived needs, beliefs and opportunities. Support Care Cancer. 2013;21:35–42. [DOI] [PubMed] [Google Scholar]
- 24.Williams K, Beeken RJ, Wardle J. Health behaviour advice to cancer patients: the perspective of social network members. Br J Cancer. 2013;108:831–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Polen-De C, Fadadu P, Weaver A, McGree M, Jatoi A, Cliby W, et al. Sarcopenia in ovarian cancer: Identifying changes in muscle quantity and quality during treatment of advanced disease (214). Gynecol Oncol. 2022;166:S119. [Google Scholar]
- 26.Yang X, Wang J. The Role of Metabolic Syndrome in Endometrial Cancer: A Review. Front Oncol. 2019;9:2019. 10.3389/fonc.2019.00744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Miguel TP, Laurienzo CE, Faria EF, Sarri AJ, Castro IQ, Júnior RJA, et al. Chemoradiation for cervical cancer treatment portends high risk of pelvic floor dysfunction. PLoS ONE. 2020;15:e0234389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Moore SC, Lee I-M, Weiderpass E, Campbell PT, Sampson JN, Kitahara CM, et al. Association of leisure-time physical activity with risk of 26 types of cancer in 1.44 million adults. JAMA internal medicine. 2016;176:816–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Matthews CE, Moore SC, Arem H, Cook MB, Trabert B, Håkansson N, et al. Amount and Intensity of Leisure-Time Physical Activity and Lower Cancer Risk. J Clin Oncol. 2019;38:686–97. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Lahart IM, Metsios GS, Nevill AM, Carmichael AR. Physical activity, risk of death and recurrence in breast cancer survivors: a systematic review and meta-analysis of epidemiological studies. Acta Oncol. 2015;54:635–54. [DOI] [PubMed] [Google Scholar]
- 31.McTiernan A, Friedenreich CM, Katzmarzyk PT, Powell KE, Macko R, Buchner D, et al. Physical activity in cancer prevention and survival: a systematic review. Med Sci Sports Exerc. 2019;51:1252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Friedenreich CM, Stone CR, Cheung WY, Hayes SC. Physical Activity and Mortality in Cancer Survivors: A Systematic Review and Meta-Analysis. JNCI Cancer Spectr. 2020;4:pkz080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Buffart L, Galvão DA, Brug J, Chinapaw M, Newton RU. Evidence-based physical activity guidelines for cancer survivors: current guidelines, knowledge gaps and future research directions. Cancer Treat Rev. 2014;40:327–40. [DOI] [PubMed] [Google Scholar]
- 34.Vardar Yağlı N, Şener G, Arıkan H, Sağlam M, İnal İnce D, Savcı S, et al. Do yoga and aerobic exercise training have impact on functional capacity, fatigue, peripheral muscle strength, and quality of life in breast cancer survivors? Integr Cancer Ther. 2015;14:125–32. [DOI] [PubMed] [Google Scholar]
- 35.McMillan EM, Newhouse IJ. Exercise is an effective treatment modality for reducing cancer-related fatigue and improving physical capacity in cancer patients and survivors: a meta-analysis. Appl Physiol Nutr Metab. 2011;36:892–903. [DOI] [PubMed] [Google Scholar]
- 36.Schmitz KH, Campbell AM, Stuiver MM, Pinto BM, Schwartz AL, Morris GS, et al. Exercise is medicine in oncology: Engaging clinicians to help patients move through cancer. CA Cancer J Clin. 2019;69:468–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Cormie P, Zopf EM, Zhang X, Schmitz KH. The impact of exercise on cancer mortality, recurrence, and treatment-related adverse effects. Epidemiol Rev. 2017;39:71–92. [DOI] [PubMed] [Google Scholar]
- 38.D’Ascenzi F, Anselmi F, Fiorentini C, Mannucci R, Bonifazi M, Mondillo S. The benefits of exercise in cancer patients and the criteria for exercise prescription in cardio-oncology. Eur J Prev Cardiol. 2021;28:725–35. [DOI] [PubMed] [Google Scholar]
- 39.Rao R, Cruz V, Peng Y, Harker-Murray A, Haley BB, Zhao H, et al. Bootcamp during neoadjuvant chemotherapy for breast cancer: a randomized pilot trial. Breast Cancer: Basic and Clinical Research. 2012;6:BCBCR. S9221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Ruiz-Casado A, Martín-Ruiz A, Pérez LM, Provencio M, Fiuza-Luces C, Lucia A. Exercise and the hallmarks of cancer. Trends in cancer. 2017;3:423–41. [DOI] [PubMed] [Google Scholar]
- 41.Malveiro C, Correia IR, Cargaleiro C, Magalhães JP, de Matos LV, Hilário S, et al. Effects of exercise training on cancer patients undergoing neoadjuvant treatment: A systematic review. J Sci Med Sport. 2023;26(11):586–92. [DOI] [PubMed] [Google Scholar]
- 42.Hanahan D, Weinberg RA. Hallmarks of Cancer: The Next Generation. Cell. 2011;144:646–74. [DOI] [PubMed] [Google Scholar]
- 43.Mu N, Zhu Y, Wang Y, Zhang H, Xue F. Insulin resistance: A significant risk factor of endometrial cancer. Gynecol Oncol. 2012;125:751–7. [DOI] [PubMed] [Google Scholar]
- 44.Kobayashi H, Imanaka S, Shigetomi H. Revisiting therapeutic strategies for ovarian cancer by focusing on redox homeostasis (Review). Oncol Lett. 2022;23:80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Conesa-Zamora P. Immune responses against virus and tumor in cervical carcinogenesis: Treatment strategies for avoiding the HPV-induced immune escape. Gynecol Oncol. 2013;131:480–8. [DOI] [PubMed] [Google Scholar]
- 46.Checa J, Aran JM. Reactive oxygen species: drivers of physiological and pathological processes. J Inflamm Res. 2020;13:1057–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Caliri AW, Tommasi S, Besaratinia A. Relationships among smoking, oxidative stress, inflammation, macromolecular damage, and cancer. Mutation Research/Reviews in Mutation Research. 2021;787:108365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Prasad S, Gupta SC, Tyagi AK. Reactive oxygen species (ROS) and cancer: Role of antioxidative nutraceuticals. Cancer Lett. 2017;387:95–105. [DOI] [PubMed] [Google Scholar]
- 49.Rajendran P, Nandakumar N, Rengarajan T, Palaniswami R, Gnanadhas EN, Lakshminarasaiah U, et al. Antioxidants and human diseases. Clin Chim Acta. 2014;436:332–47. [DOI] [PubMed] [Google Scholar]
- 50.Shah MA, Rogoff HA. Implications of reactive oxygen species on cancer formation and its treatment. Semin Oncol. 2021;48:238–45. [DOI] [PubMed] [Google Scholar]
- 51.Tafani M, Sansone L, Limana F, Arcangeli T, De Santis E, Polese M, et al. The Interplay of Reactive Oxygen Species, Hypoxia, Inflammation, and Sirtuins in Cancer Initiation and Progression. Oxid Med Cell Longev. 2016;2016:3907147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Kushi LH, Byers T, Doyle C, Bandera EV, McCullough M, Gansler T, et al. American Cancer Society Guidelines on Nutrition and Physical Activity for cancer prevention: reducing the risk of cancer with healthy food choices and physical activity. CA Cancer J Clin. 2006;56:254–81. [DOI] [PubMed] [Google Scholar]
- 53.Radak Z, Torma F, Berkes I, Goto S, Mimura T, Posa A, et al. Exercise effects on physiological function during aging. Free Radical Biol Med. 2019;132:33–41. [DOI] [PubMed] [Google Scholar]
- 54.Pingitore A, Lima GPP, Mastorci F, Quinones A, Iervasi G, Vassalle C. Exercise and oxidative stress: Potential effects of antioxidant dietary strategies in sports. Nutrition. 2015;31:916–22. [DOI] [PubMed] [Google Scholar]
- 55.Kruk J, Aboul-Enein HY, Kładna A, Bowser JE. Oxidative stress in biological systems and its relation with pathophysiological functions: the effect of physical activity on cellular redox homeostasis. Free Radical Res. 2019;53:497–521. [DOI] [PubMed] [Google Scholar]
- 56.Saed GM, Diamond MP, Fletcher NM. Updates of the role of oxidative stress in the pathogenesis of ovarian cancer. Gynecol Oncol. 2017;145:595–602. [DOI] [PubMed] [Google Scholar]
- 57.Agnew HJ, Kitson SJ, Crosbie EJ. Gynecological malignancies and obesity. Best Pract Res Clin Obstet Gynaecol. 2023;88: 102337. [DOI] [PubMed] [Google Scholar]
- 58.Hatao H, Oh-ishi S, Itoh M, Leeuwenburgh C, Ohno H, Ookawara T, et al. Effects of acute exercise on lung antioxidant enzymes in young and old rats. Mech Ageing Dev. 2006;127:384–90. [DOI] [PubMed] [Google Scholar]
- 59.Radak Z, Bori Z, Koltai E, Fatouros IG, Jamurtas AZ, Douroudos II, et al. Age-dependent changes in 8-oxoguanine-DNA glycosylase activity are modulated by adaptive responses to physical exercise in human skeletal muscle. Free Radical Biol Med. 2011;51:417–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Yang L, Xie H-J, Li Y-Y, Wang X, Liu X-X, Mai J. Molecular mechanisms of platinum-based chemotherapy resistance in ovarian cancer (Review). Oncol Rep. 2022;47:82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Soares JP, Silva AI, Silva AM, Almeida V, Teixeira JP, Matos M, et al. Effects of physical exercise training in DNA damage and repair activity in humans with different genetic polymorphisms of hOGG1 (Ser326Cys). Cell Biochem Funct. 2015;33:519–24. [DOI] [PubMed] [Google Scholar]
- 62.Ovejero-Sánchez M, González-Sarmiento R, Herrero AB. DNA Damage Response Alterations in Ovarian Cancer: From Molecular Mechanisms to Therapeutic Opportunities. Cancers (Basel). 2023;15(2):448. 10.3390/cancers15020448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Wise MR, Jordan V, Lagas A, Showell M, Wong N, Lensen S, et al. Obesity and endometrial hyperplasia and cancer in premenopausal women: A systematic review. Am J Obstetrics & Gynecology. 2016;214:689-.e1-.e17. [DOI] [PubMed] [Google Scholar]
- 64.Na H-K, Oliynyk S. Effects of physical activity on cancer prevention. Ann N Y Acad Sci. 2011;1229:176–83. [DOI] [PubMed] [Google Scholar]
- 65.Radak Z, Chung HY, Goto S. Systemic adaptation to oxidative challenge induced by regular exercise. Free Radical Biol Med. 2008;44:153–9. [DOI] [PubMed] [Google Scholar]
- 66.Rebillard A, Lefeuvre-Orfila L, Gueritat J, Cillard J. Prostate cancer and physical activity: Adaptive response to oxidative stress. Free Radical Biol Med. 2013;60:115–24. [DOI] [PubMed] [Google Scholar]
- 67.Brown WMC, Davison GW, McClean CM, Murphy MH. A Systematic Review of the Acute Effects of Exercise on Immune and Inflammatory Indices in Untrained Adults. Sports Medicine - Open. 2015;1:35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Engeroff T, Banzer W, Niederer D. The impact of regular activity and exercise intensity on the acute effects of resistance exercise on cognitive function. Scand J Med Sci Sports. 2022;32:94–105. [DOI] [PubMed] [Google Scholar]
- 69.Ho SS, Dhaliwal SS, Hills AP, Pal S. The effect of 12 weeks of aerobic, resistance or combination exercise training on cardiovascular risk factors in the overweight and obese in a randomized trial. BMC Public Health. 2012;12:704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Schroeder EC, Franke WD, Sharp RL, Lee D-C. Comparative effectiveness of aerobic, resistance, and combined training on cardiovascular disease risk factors: A randomized controlled trial. PLOS ONE. 2019;14:e0210292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Gomez-Cabrera M-C, Domenech E, Viña J. Moderate exercise is an antioxidant: Upregulation of antioxidant genes by training. Free Radical Biol Med. 2008;44:126–31. [DOI] [PubMed] [Google Scholar]
- 72.Gomez-Cabrera MC, Salvador-Pascual A, Cabo H, Ferrando B, Viña J. Redox modulation of mitochondriogenesis in exercise. Does antioxidant supplementation blunt the benefits of exercise training? Free Radical Biology and Medicine. 2015;86:37–46. [DOI] [PubMed]
- 73.Schwappacher R, Schink K, Sologub S, Dieterich W, Reljic D, Friedrich O, et al. Physical activity and advanced cancer: evidence of exercise-sensitive genes regulating prostate cancer cell proliferation and apoptosis. J Physiol. 2020;598:3871–89. [DOI] [PubMed] [Google Scholar]
- 74.Westerlind K, McCarty H, Gibson K, Strange R. Effect of exercise on the rat mammary gland: implications for carcinogenesis. Acta Physiol Scand. 2002;175:147–56. [DOI] [PubMed] [Google Scholar]
- 75.Radák Z, Naito H, Kaneko T, Tahara S, Nakamoto H, Takahashi R, et al. Exercise training decreases DNA damage and increases DNA repair and resistance against oxidative stress of proteins in aged rat skeletal muscle. Pflugers Arch. 2002;445:273–8. [DOI] [PubMed] [Google Scholar]
- 76.De Santi M, Baldelli G, Lucertini F, Natalucci V, Brandi G, Barbieri E. A dataset on the effect of exercise-conditioned human sera in three-dimensional breast cancer cell culture. Data Brief. 2019;27: 104704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Theriau CF, Shpilberg Y, Riddell MC, Connor MK. Voluntary physical activity abolishes the proliferative tumor growth microenvironment created by adipose tissue in animals fed a high fat diet. J Appl Physiol. 2016;121:139–53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Pedersen L, Idorn M, Olofsson GH, Lauenborg B, Nookaew I, Hansen RH, et al. Voluntary running suppresses tumor growth through epinephrine-and IL-6-dependent NK cell mobilization and redistribution. Cell Metab. 2016;23:554–62. [DOI] [PubMed] [Google Scholar]
- 79.Yeramian A, Moreno-Bueno G, Dolcet X, Catasus L, Abal M, Colas E, et al. Endometrial carcinoma: molecular alterations involved in tumor development and progression. Oncogene. 2013;32:403–13. [DOI] [PubMed] [Google Scholar]
- 80.Zhang R, He Y, Zhang X, Xing B, Sheng Y, Lu H, et al. Estrogen receptor-regulated microRNAs contribute to the BCL2/BAX imbalance in endometrial adenocarcinoma and precancerous lesions. Cancer Lett. 2012;314:155–65. [DOI] [PubMed] [Google Scholar]
- 81.Smith AJ, Phipps WR, Thomas W, Schmitz KH, Kurzer MS. The Effects of Aerobic Exercise on Estrogen Metabolism in Healthy Premenopausal Women. Cancer Epidemiol Biomark Prev. 2013;22:756–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Ali AY, Farrand L, Kim JY, Byun S, Suh J-Y, Lee HJ, et al. Molecular determinants of ovarian cancer chemoresistance: new insights into an old conundrum. Ann N Y Acad Sci. 2012;1271:58–67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Yang L, Morielli AR, Heer E, Kirkham AA, Cheung WY, Usmani N, et al. Effects of Exercise on Cancer Treatment Efficacy: A Systematic Review of Preclinical and Clinical Studies. Can Res. 2021;81:4889–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Ashcraft KA, Warner AB, Jones LW, Dewhirst MW. Exercise as Adjunct Therapy in Cancer. Seminars in Radiation Oncology. 2019;29:16–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Almeida AM, Queiroz JA, Sousa F, Sousa Â. Cervical cancer and HPV infection: ongoing therapeutic research to counteract the action of E6 and E7 oncoproteins. Drug Discovery Today. 2019;24:2044–57. [DOI] [PubMed] [Google Scholar]
- 86.Mooren FC, Krüger K. Exercise, Autophagy, and Apoptosis. Prog Mol Biol Transl Sci. 2015;135:407–22. [DOI] [PubMed] [Google Scholar]
- 87.Krüger K, Agnischock S, Lechtermann A, Tiwari S, Mishra M, Pilat C, et al. Intensive resistance exercise induces lymphocyte apoptosis via cortisol and glucocorticoid receptor-dependent pathways. J Appl Physiol. 2011;110:1226–32. [DOI] [PubMed] [Google Scholar]
- 88.Siewierska K, Malicka I, Kobierzycki C, Paslawska U, Cegielski M, Grzegrzolka J, et al. The Impact of Exercise Training on Breast Cancer. In Vivo. 2018;32:249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Gomes-Santos IL, Kumar AS, Hausmann F, Meyer MN, Shiferaw SZ, Amoozgar Z, et al. Exercise intensity governs tumor control in mice with breast cancer. Front Immunol. 2024;15:2024. 10.3389/fimmu.2024.1339232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Radahmadi M, Alaei H, Sharifi MR, Hosseini N. Stress biomarker responses to different protocols of forced exercise in chronically stressed rats. J Bodyw Mov Ther. 2017;21:63–8. [DOI] [PubMed] [Google Scholar]
- 91.Zhang Q, Zhang B, Zhang K, Meng X, Jia Q, Bu Y, et al. Moderate swimming suppressed the growth and metastasis of the transplanted liver cancer in mice model: with reference to nervous system. Oncogene. 2016;35:4122–31. [DOI] [PubMed] [Google Scholar]
- 92.Dethlefsen C, Hansen LS, Lillelund C, Andersen C, Gehl J, Christensen JF, et al. Exercise-induced catecholamines activate the hippo tumor suppressor pathway to reduce risks of breast cancer development. Can Res. 2017;77:4894–904. [DOI] [PubMed] [Google Scholar]
- 93.Han J-K, Kim G. Role of physical exercise in modulating the insulin-like growth factor system for improving breast cancer outcomes: A meta-analysis. Exp Gerontol. 2021;152: 111435. [DOI] [PubMed] [Google Scholar]
- 94.Kurgan N, Tsakiridis E, Kouvelioti R, Moore J, Klentrou P, Tsiani E. Inhibition of Human Lung Cancer Cell Proliferation and Survival by Post-Exercise Serum Is Associated with the Inhibition of Akt, mTOR, p70 S6K, and Erk1/2. Cancers. 2017;9:46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Di Malta C, Siciliano D, Calcagni A, Monfregola J, Punzi S, Pastore N, et al. Transcriptional activation of RagD GTPase controls mTORC1 and promotes cancer growth. Science. 2017;356:1188–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Ballarò R, Beltrà M, De Lucia S, Pin F, Ranjbar K, Hulmi JJ, et al. Moderate exercise in mice improves cancer plus chemotherapy-induced muscle wasting and mitochondrial alterations. FASEB J. 2019;33:5482–94. [DOI] [PubMed] [Google Scholar]
- 97.Aoi W, Naito Y, Takagi T, Tanimura Y, Takanami Y, Kawai Y, et al. A novel myokine, secreted protein acidic and rich in cysteine (SPARC), suppresses colon tumorigenesis via regular exercise. Gut. 2013;62:882–9. [DOI] [PubMed] [Google Scholar]
- 98.Ren F, Shen J, Shi H, Hornicek FJ, Kan Q, Duan Z. Novel mechanisms and approaches to overcome multidrug resistance in the treatment of ovarian cancer. Biochimica et Biophysica Acta (BBA) - Reviews on Cancer. 2016;1866:266–75. [DOI] [PubMed]
- 99.Coussens LM, Werb Z. Inflammation and cancer. Nature. 2002;420:860–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Szewczyk G, Maciejewski TM, Szukiewicz D. Current progress in the inflammatory background of angiogenesis in gynecological cancers. Inflamm Res. 2019;68:247–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Kiesel L, Eichbaum C, Baumeier A, Eichbaum M. Obesity Epidemic—The Underestimated Risk of Endometrial Cancer. Cancers (Basel). 2020;12(12). 10.3390/cancers12123860. [DOI] [PMC free article] [PubMed]
- 102.Mangino G, Chiantore MV, Iuliano M, Fiorucci G, Romeo G. Inflammatory microenvironment and human papillomavirus-induced carcinogenesis. Cytokine Growth Factor Rev. 2016;30:103–11. [DOI] [PubMed] [Google Scholar]
- 103.Heeren AM, Kenter GG, Jordanova ES, de Gruijl TD. CD14+ macrophage-like cells as the linchpin of cervical cancer perpetrated immune suppression and early metastatic spread: A new therapeutic lead? OncoImmunology. 2015;4:e1009296. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Fernandes JV, Fernandes TAAdM, de Azevedo JCV, Cobucci RNO, de Carvalho MGF, Andrade VS, et al. Link between chronic inflammation and human papillomavirus-induced carcinogenesis (Review). Oncol Lett. 2015;9:1015–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Raziyeva K, Kim Y, Zharkinbekov Z, Kassymbek K, Jimi S, Saparov A. Immunology of acute and chronic wound healing. Biomolecules. 2021;11:700. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Denk D, Greten FR. Inflammation: the incubator of the tumor microenvironment. Trends in Cancer. 2022;8:901–14. [DOI] [PubMed] [Google Scholar]
- 107.Khandia R, Munjal A. Interplay between inflammation and cancer. Adv Protein Chem Struct Biol. 2020;119:199–245. [DOI] [PubMed] [Google Scholar]
- 108.Ziemkiewicz N, Hilliard G, Pullen NA, Garg K. The role of innate and adaptive immune cells in skeletal muscle regeneration. Int J Mol Sci. 2021;22:3265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Chung HY, Kim DH, Lee EK, Chung KW, Chung S, Lee B, et al. Redefining chronic inflammation in aging and age-related diseases: proposal of the senoinflammation concept. Aging Dis. 2019;10:367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Dossus L, Rinaldi S, Becker S, Lukanova A, Tjonneland A, Olsen A, et al. Obesity, inflammatory markers, and endometrial cancer risk: a prospective case–control study. Endocr Relat Cancer. 2010;17:1007–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Wallace AE, Gibson DA, Saunders PTK, Jabbour HN. Inflammatory events in endometrial adenocarcinoma. J Endocrinol. 2010;206:141–57. [DOI] [PubMed] [Google Scholar]
- 112.Sadri Nahand J, Moghoofei M, Salmaninejad A, Bahmanpour Z, Karimzadeh M, Nasiri M, et al. Pathogenic role of exosomes and microRNAs in HPV-mediated inflammation and cervical cancer: A review. Int J Cancer. 2020;146:305–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Karsten K, Frank-Christoph M, Christian P. The Immunomodulatory Effects of Physical Activity. Curr Pharm Des. 2016;22:3730–48. [DOI] [PubMed] [Google Scholar]
- 114.Schweer D, McAtee A, Neupane K, Richards C, Ueland F, Kolesar J. Tumor-Associated Macrophages and Ovarian Cancer: Implications for Therapy. Cancers (Basel). 2022;14(9). 10.3390/cancers14092220. [DOI] [PMC free article] [PubMed]
- 115.Callegari IOM, Rocha GZ, Oliveira AG. Physical exercise, health, and disease treatment: The role of macrophages. Front Physiol. 2023;14:2023. 10.3389/fphys.2023.1061353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Reilly SM, Saltiel AR. Adapting to obesity with adipose tissue inflammation. Nat Rev Endocrinol. 2017;13:633–43. [DOI] [PubMed] [Google Scholar]
- 117.Beavers KM, Brinkley TE, Nicklas BJ. Effect of exercise training on chronic inflammation. Clin Chim Acta. 2010;411:785–93. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Daou HN. Exercise as an anti-inflammatory therapy for cancer cachexia: a focus on interleukin-6 regulation. American Journal of Physiology-Regulatory, Integrative and Comparative Physiology. 2020;318:R296–310. [DOI] [PubMed] [Google Scholar]
- 119.Jones SB, Thomas GA, Hesselsweet SD, Alvarez-Reeves M, Yu H, Irwin ML. Effect of Exercise on Markers of Inflammation in Breast Cancer Survivors: The Yale Exercise and Survivorship Study. Cancer Prev Res. 2013;6:109–18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Meneses-Echavez JF, Ramirez-Velez R, Gonzalez-Jimenez E, Schmidt Rio-Valle J, Sanchez Perez MJ, Molina Montes E. Exercise training, inflammatory cytokines, and other markers of low-grade inflammation in breast cancer survivors: A systematic review and meta-analysis. J Clinical Oncology. 2014;32:121. [Google Scholar]
- 121.Meneses-Echávez JF, Correa-Bautista JE, González-Jiménez E, Schmidt Río-Valle J, Elkins MR, Lobelo F, et al. The Effect of Exercise Training on Mediators of Inflammation in Breast Cancer Survivors: A Systematic Review with Meta-analysis. Cancer Epidemiol Biomark Prev. 2016;25:1009–17. [DOI] [PubMed] [Google Scholar]
- 122.Cohen M, Levkovich I, Katz R, Fried G, Pollack S. Low physical activity, fatigue and depression in breast cancer survivors: Moderation by levels of IL-6 and IL-8. Int J Psychophysiol. 2020;158:96–102. [DOI] [PubMed] [Google Scholar]
- 123.Salgado R, Junius S, Benoy I, Van Dam P, Vermeulen P, Van Marck E, et al. Circulating interleukin-6 predicts survival in patients with metastatic breast cancer. Int J Cancer. 2003;103:642–6. [DOI] [PubMed] [Google Scholar]
- 124.Bachelot T, Ray-Coquard I, Menetrier-Caux C, Rastkha M, Duc A, Blay JY. Prognostic value of serum levels of interleukin 6 and of serum and plasma levels of vascular endothelial growth factor in hormone-refractory metastatic breast cancer patients. Br J Cancer. 2003;88:1721–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.De Araújo AL, Silva LC, Fernandes JR, Benard G. Preventing or reversing immunosenescence: can exercise be an immunotherapy? Immunotherapy. 2013;5:879–93. [DOI] [PubMed] [Google Scholar]
- 126.Spiliopoulou P, Gavriatopoulou M, Kastritis E, Dimopoulos MA, Terzis G. Exercise-Induced Changes in Tumor Growth via Tumor Immunity. Sports. 2021;9:46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Pedersen L, Christensen JF, Hojman P. Effects of exercise on tumor physiology and metabolism. The Cancer Journal. 2015;21:111–6. [DOI] [PubMed] [Google Scholar]
- 128.Komohara Y, Fujiwara Y, Ohnishi K, Takeya M. Tumor-associated macrophages: Potential therapeutic targets for anti-cancer therapy. Adv Drug Deliv Rev. 2016;99:180–5. [DOI] [PubMed] [Google Scholar]
- 129.Rolny C, Mazzone M, Tugues S, Laoui D, Johansson I, Coulon C, et al. HRG inhibits tumor growth and metastasis by inducing macrophage polarization and vessel normalization through downregulation of PlGF. Cancer Cell. 2011;19:31–44. [DOI] [PubMed] [Google Scholar]
- 130.Macciò A, Gramignano G, Cherchi MC, Tanca L, Melis L, Madeddu C. Role of M1-polarized tumor-associated macrophages in the prognosis of advanced ovarian cancer patients. Sci Rep. 2020;10:6096. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Zhang X, Ashcraft KA, Betof Warner A, Nair SK, Dewhirst MW. Can Exercise-Induced Modulation of the Tumor Physiologic Microenvironment Improve Antitumor Immunity? Can Res. 2019;79:2447–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Galliverti G, Wullschleger S, Tichet M, Murugan D, Zangger N, Horton W, et al. Myeloid Cells Orchestrate Systemic Immunosuppression, Impairing the Efficacy of Immunotherapy against HPV+ Cancers. Cancer Immunol Res. 2020;8:131–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Turner JE, Brum PC. Does Regular Exercise Counter T Cell Immunosenescence Reducing the Risk of Developing Cancer and Promoting Successful Treatment of Malignancies? Oxid Med Cell Longev. 2017;2017:4234765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 134.Abdalla DR, Aleixo AAR, Murta EF, Michelin MA. Innate immune response adaptation in mice subjected to administration of DMBA and physical activity. Oncol Lett. 2014;7:886–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Zielinski MR, Muenchow M, Wallig MA, Horn PL, Woods JA. Exercise delays allogeneic tumor growth and reduces intratumoral inflammation and vascularization. J Appl Physiol. 2004;96:2249–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Almeida PWM, Gomes-Filho A, Ferreira AJ, Rodrigues CEM, Dias-Peixoto MF, Russo RC, et al. Swim training suppresses tumor growth in mice. J Appl Physiol. 2009;107:261–5. [DOI] [PubMed] [Google Scholar]
- 137.Galdiero MR, Varricchi G, Loffredo S, Mantovani A, Marone G. Roles of neutrophils in cancer growth and progression. J Leukoc Biol. 2018;103:457–64. [DOI] [PubMed] [Google Scholar]
- 138.Simpson RJ, Kunz H, Agha N, Graff R. Exercise and the regulation of immune functions. Prog Mol Biol Transl Sci. 2015;135:355–80. [DOI] [PubMed] [Google Scholar]
- 139.Wang B, Xu H, Hu X, Ma W, Zhang J, Li Y, et al. Synergetic inhibition of daidzein and regular exercise on breast cancer in bearing-4T1 mice by regulating NK cells and apoptosis pathway. Life Sci. 2020;245: 117387. [DOI] [PubMed] [Google Scholar]
- 140.Walsh NP, Gleeson M, Shephard RJ, Gleeson M, Woods JA, Bishop N, et al. Position statement part one: immune function and exercise. 2011. [PubMed]
- 141.Wennerberg E, Lhuillier C, Rybstein MD, Dannenberg K, Rudqvist N-P, Koelwyn GJ, et al. Exercise reduces immune suppression and breast cancer progression in a preclinical model. Oncotarget. 2020;11:452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Rundqvist H, Veliça P, Barbieri L, Gameiro PA, Bargiela D, Gojkovic M, et al. Cytotoxic T-cells mediate exercise-induced reductions in tumor growth. ELife. 2020;9:e59996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Gomes-Santos IL, Amoozgar Z, Kumar AS, Ho WW, Roh K, Talele NP, et al. Exercise training improves tumor control by increasing CD8+ T-cell infiltration via CXCR3 signaling and sensitizes breast cancer to immune checkpoint blockade. Cancer Immunol Res. 2021;9:765–78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Kurz E, Hirsch CA, Dalton T, Shadaloey SA, Khodadadi-Jamayran A, Miller G, et al. Exercise-induced engagement of the IL-15/IL-15Rα axis promotes anti-tumor immunity in pancreatic cancer. Cancer Cell. 2022;40(720–37):e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Rundqvist H, Veliça P, Barbieri L, Gameiro PA, Bargiela D, Gojkovic M, et al. Cytotoxic T-cells mediate exercise-induced reductions in tumor growth. Elife. 2020;9:e59996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Schauer T, Djurhuus SS, Simonsen C, Brasso K, Christensen JF. The effects of acute exercise and inflammation on immune function in early-stage prostate cancer. Brain, Behavior, & Immunity - Health. 2022;25:100508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Spiliopoulou P, Gavriatopoulou M, Kastritis E, Dimopoulos MA, Terzis G. Exercise-Induced Changes in Tumor Growth via Tumor Immunity. Sports (Basel). 2021;9(4). 10.3390/sports9040046. [DOI] [PMC free article] [PubMed]
- 148.Renman D, Gylling B, Vidman L, Bodén S, Strigård K, Palmqvist R, et al. Density of CD3+ and CD8+ Cells in the Microenvironment of Colorectal Cancer according to Prediagnostic Physical Activity. Cancer Epidemiol Biomark Prev. 2021;30:2317–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Chaudhary B, Elkord E. Regulatory T Cells in the Tumor Microenvironment and Cancer Progression: Role and Therapeutic Targeting. Vaccines (Basel). 2016;4(3). 10.3390/vaccines4030028. [DOI] [PMC free article] [PubMed]
- 150.Zou W. Regulatory T cells, tumour immunity and immunotherapy. Nat Rev Immunol. 2006;6:295–307. [DOI] [PubMed] [Google Scholar]
- 151.Bianco TM, Abdalla DR, Desidério CS, Thys S, Simoens C, Bogers J-P, et al. The influence of physical activity in the anti-tumor immune response in experimental breast tumor. Immunol Lett. 2017;190:148–58. [DOI] [PubMed] [Google Scholar]
- 152.Hagar A, Wang Z, Koyama S, Serrano JA, Melo L, Vargas S, et al. Endurance training slows breast tumor growth in mice by suppressing Treg cells recruitment to tumors. BMC Cancer. 2019;19:1–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Idorn M, Hojman P. Exercise-Dependent Regulation of NK Cells in Cancer Protection. Trends Mol Med. 2016;22:565–77. [DOI] [PubMed] [Google Scholar]
- 154.Lyu D-w. Immunomodulatory effects of exercise in cancer prevention and adjuvant therapy: a narrative review. Front Physiol. 2024;14:2023. 10.3389/fphys.2023.1292580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Stein KD, Syrjala KL, Andrykowski MA. Physical and psychological long-term and late effects of cancer. Cancer. 2008;112:2577–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Emery J, Butow P, Lai-Kwon J, Nekhlyudov L, Rynderman M, Jefford M. Management of common clinical problems experienced by survivors of cancer. The Lancet. 2022;399:1537–50. [DOI] [PubMed] [Google Scholar]
- 157.Nipp RD, Fuchs G, El-Jawahri A, Mario J, Troschel FM, Greer JA, et al. Sarcopenia is associated with quality of life and depression in patients with advanced cancer. Oncologist. 2018;23:97–104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Baracos VE, Martin L, Korc M, Guttridge DC, Fearon KC. Cancer-associated cachexia Nature reviews Disease primers. 2018;4:1–18. [DOI] [PubMed] [Google Scholar]
- 159.Rutten IJG, van Dijk DPJ, Kruitwagen RFPM, Beets-Tan RGH, Olde Damink SWM, van Gorp T. Loss of skeletal muscle during neoadjuvant chemotherapy is related to decreased survival in ovarian cancer patients. J Cachexia Sarcopenia Muscle. 2016;7:458–66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Rutkowska A, Jastrzebski D, Rutkowski S, Żebrowska A, Stanula A, Szczegielniak J, et al. Exercise Training in Patients With Non-Small Cell Lung Cancer During In-Hospital Chemotherapy Treatment: A RANDOMIZED CONTROLLED TRIAL. J Cardiopulm Rehabil Prev. 2019;39:127–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Porter LS, Carson JW, Olsen M, Carson KM, Sanders L, Jones L, et al. Feasibility of a mindful yoga program for women with metastatic breast cancer: results of a randomized pilot study. Support Care Cancer. 2019;27:4307–16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Schink K, Herrmann HJ, Schwappacher R, Meyer J, Orlemann T, Waldmann E, et al. Effects of whole-body electromyostimulation combined with individualized nutritional support on body composition in patients with advanced cancer: a controlled pilot trial. BMC Cancer. 2018;18:886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Schmidt T, Jonat W, Wesch D, Oberg H-H, Adam-Klages S, Keller L, et al. Influence of physical activity on the immune system in breast cancer patients during chemotherapy. J Cancer Res Clin Oncol. 2018;144:579–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Schuler MK, Hentschel L, Kisel W, Kramer M, Lenz F, Hornemann B, et al. Impact of Different Exercise Programs on Severe Fatigue in Patients Undergoing Anticancer Treatment—A Randomized Controlled Trial. J Pain Symptom Manage. 2017;53:57–66. [DOI] [PubMed] [Google Scholar]
- 165.Dhillon HM, Bell ML, van der Ploeg HP, Turner JD, Kabourakis M, Spencer L, et al. Impact of physical activity on fatigue and quality of life in people with advanced lung cancer: a randomized controlled trial. Ann Oncol. 2017;28:1889–97. [DOI] [PubMed] [Google Scholar]
- 166.Tsianakas V, Harris J, Ream E, Van Hemelrijck M, Purushotham A, Mucci L, et al. CanWalk: a feasibility study with embedded randomised controlled trial pilot of a walking intervention for people with recurrent or metastatic cancer. BMJ Open. 2017;7:e013719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Ligibel JA, Giobbie-Hurder A, Shockro L, Campbell N, Partridge AH, Tolaney SM, et al. Randomized trial of a physical activity intervention in women with metastatic breast cancer. Cancer. 2016;122:1169–77. [DOI] [PubMed] [Google Scholar]
- 168.Henke CC, Cabri J, Fricke L, Pankow W, Kandilakis G, Feyer PC, et al. Strength and endurance training in the treatment of lung cancer patients in stages IIIA/IIIB/IV. Support Care Cancer. 2014;22:95–101. [DOI] [PubMed] [Google Scholar]
- 169.Jensen W, Baumann FT, Stein A, Bloch W, Bokemeyer C, de Wit M, et al. Exercise training in patients with advanced gastrointestinal cancer undergoing palliative chemotherapy: a pilot study. Support Care Cancer. 2014;22:1797–806. [DOI] [PubMed] [Google Scholar]
- 170.Cheville AL, Kollasch J, Vandenberg J, Shen T, Grothey A, Gamble G, et al. A Home-Based Exercise Program to Improve Function, Fatigue, and Sleep Quality in Patients With Stage IV Lung and Colorectal Cancer: A Randomized Controlled Trial. J Pain Symptom Manage. 2013;45:811–21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Cormie P, Newton RU, Spry N, Joseph D, Taaffe DR, Galvão DA. Safety and efficacy of resistance exercise in prostate cancer patients with bone metastases. Prostate Cancer Prostatic Dis. 2013;16:328–35. [DOI] [PubMed] [Google Scholar]
- 172.Litterini AJ, Fieler VK, Cavanaugh JT, Lee JQ. Differential Effects of Cardiovascular and Resistance Exercise on Functional Mobility in Individuals With Advanced Cancer: A Randomized Trial. Arch Phys Med Rehabil. 2013;94:2329–35. [DOI] [PubMed] [Google Scholar]
- 173.Hwang C-L, Yu C-J, Shih J-Y, Yang P-C, Wu Y-T. Effects of exercise training on exercise capacity in patients with non-small cell lung cancer receiving targeted therapy. Support Care Cancer. 2012;20:3169–77. [DOI] [PubMed] [Google Scholar]
- 174.Oldervoll LM, Loge JH, Lydersen S, Paltiel H, Asp MB, Nygaard UV, et al. Physical Exercise for Cancer Patients with Advanced Disease: A Randomized Controlled Trial. Oncologist. 2011;16:1649–57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Hursting SD, Hursting MJ. Growth signals, inflammation, and vascular perturbations: mechanistic links between obesity, metabolic syndrome, and cancer. Arterioscler Thromb Vasc Biol. 2012;32:1766–70. [DOI] [PubMed] [Google Scholar]
- 176.Dethlefsen C, Pedersen KS, Hojman P. Every exercise bout matters: linking systemic exercise responses to breast cancer control. Breast Cancer Res Treat. 2017;162:399–408. [DOI] [PubMed] [Google Scholar]
- 177.van Gemert WA, Monninkhof EM, May AM, Elias SG, van der Palen J, Veldhuis W, et al. Association between changes in fat distribution and biomarkers for breast cancer. Endocr Relat Cancer. 2017;24:297–305. [DOI] [PubMed] [Google Scholar]
- 178.Mishra SI, Scherer RW, Geigle PM, Berlanstein DR, Topaloglu O, Gotay CC, et al. Exercise interventions on health‐related quality of life for cancer survivors. Cochrane Database of Systematic Reviews. 2012(8):CD007566. 10.1002/14651858.CD007566.pub2. [DOI] [PMC free article] [PubMed]
- 179.Lanser L, Kink P, Egger EM, Willenbacher W, Fuchs D, Weiss G, et al. Inflammation-Induced Tryptophan Breakdown is Related With Anemia, Fatigue, and Depression in Cancer. Frontiers in Immunology. 2020;11.10.3389/fimmu.2020.00249. [DOI] [PMC free article] [PubMed]
- 180.Agudelo LZ, Femenía T, Orhan F, Porsmyr-Palmertz M, Goiny M, Martinez-Redondo V, et al. Skeletal muscle PGC-1α1 modulates kynurenine metabolism and mediates resilience to stress-induced depression. Cell. 2014;159:33–45. [DOI] [PubMed] [Google Scholar]
- 181.Martin KS, Azzolini M, Lira RJ. The kynurenine connection: How exercise shifts muscle tryptophan metabolism and affects energy homeostasis, the immune system, and the brain. Am J Physiol Cell Physiol. 2020;318:C818–30. [DOI] [PubMed] [Google Scholar]
- 182.Avancini A, Borsati A, Toniolo L, Ciurnelli C, Belluomini L, Budolfsen T, et al. Physical activity guidelines in oncology: A systematic review of the current recommendations. Crit Rev Oncol Hematol. 2025;210: 104718. [DOI] [PubMed] [Google Scholar]
- 183.Duncan M, Moschopoulou E, Herrington E, Deane J, Roylance R, Jones L, et al. Review of systematic reviews of non-pharmacological interventions to improve quality of life in cancer survivors. BMJ Open. 2017;7:e015860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 184.Hiller JG, Perry NJ, Poulogiannis G, Riedel B, Sloan EK. Perioperative events influence cancer recurrence risk after surgery. Nat Rev Clin Oncol. 2018;15:205–18. [DOI] [PubMed] [Google Scholar]
- 185.Cavalheri V, Granger C. Preoperative exercise training for patients with non‐small cell lung cancer. Cochrane Database of Systematic Reviews. 2017.6(6). 10.1002/14651858.CD012020.pub2. [DOI] [PMC free article] [PubMed]
- 186.Graham K, Unger E. Overcoming tumor hypoxia as a barrier to radiotherapy, chemotherapy and immunotherapy in cancer treatment. Int J Nanomed. 2018;13:6049–58. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 187.Windsor PM, Nicol KF, Potter J. A randomized, controlled trial of aerobic exercise for treatment‐related fatigue in men receiving radical external beam radiotherapy for localized prostate carcinoma. Cancer: Interdisciplinary International J Am Cancer Society. 2004;101:550–7. [DOI] [PubMed]
- 188.Rief H, Petersen LC, Omlor G, Akbar M, Bruckner T, Rieken S, et al. The effect of resistance training during radiotherapy on spinal bone metastases in cancer patients–a randomized trial. Radiother Oncol. 2014;112:133–9. [DOI] [PubMed] [Google Scholar]
- 189.Wakefield ZR, Tanaka M, Pampo C, Lepler S, Rice L, Guingab-Cagmat J, et al. Normal tissue and tumor microenvironment adaptations to aerobic exercise enhance doxorubicin anti-tumor efficacy and ameliorate its cardiotoxicity in retired breeder mice. Oncotarget. 2021;12:1737. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 190.Florez Bedoya CA, Cardoso ACF, Parker N, Ngo-Huang A, Petzel MQ, Kim MP, et al. Exercise during preoperative therapy increases tumor vascularity in pancreatic tumor patients. Sci Rep. 2019;9:13966. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Brown JC, Ligibel JA. The Role of Physical Activity in Oncology Care. JNCI Monographs. 2017;2017:lgx017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Cannioto RA, Hutson A, Dighe S, McCann W, McCann SE, Zirpoli GR, et al. Physical activity before, during, and after chemotherapy for high-risk breast cancer: relationships with survival. JNCI: J National Cancer Institute. 2021;113:54–63. [DOI] [PMC free article] [PubMed]
- 193.Salamon G, Dougherty D, Whiting L, Crawford GB, Stein B, Kotasek D. Effects of a prescribed, supervised exercise programme on tumour disease progression in oncology patients undergoing anti-cancer therapy: a retrospective observational cohort study. Intern Med J. 2023;53:104–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Gustafson MP, Wheatley-Guy CM, Rosenthal AC, Gastineau DA, Katsanis E, Johnson BD, et al. Exercise and the immune system: taking steps to improve responses to cancer immunotherapy. J Immunother Cancer. 2021;9:e001872. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Pennington KP, McTiernan A. The role of physical activity in breast and gynecologic cancer survivorship. Gynecol Oncol. 2018;149:198–204. [DOI] [PubMed] [Google Scholar]
- 196.Parker NH, Arlinghaus KR, Johnston CA. Integrating Physical Activity Into Clinical Cancer Care. Am J Lifestyle Med. 2018;12:220–3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Arring N, Barton DL, Reese JB. Clinical Practice Strategies to Address Sexual Health in Female Cancer Survivors. J Clin Oncol. 2023;41:4927–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Beesley VL, Alemayehu C, Webb PM. A systematic literature review of trials of survivorship interventions for women with gynaecological cancer and their caregivers. Eur J Cancer Care. 2019;28: e13057. [DOI] [PubMed] [Google Scholar]
- 199.von Gruenigen VE, Courneya KS, Gibbons HE, Kavanagh MB, Waggoner SE, Lerner E. Feasibility and effectiveness of a lifestyle intervention program in obese endometrial cancer patients: A randomized trial. Gynecol Oncol. 2008;109:19–26. [DOI] [PubMed] [Google Scholar]
- 200.von Gruenigen VE, Gibbons HE, Kavanagh MB, Janata JW, Lerner E, Courneya KS. A randomized trial of a lifestyle intervention in obese endometrial cancer survivors: quality of life outcomes and mediators of behavior change. Health Qual Life Outcomes. 2009;7:1–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Donnelly C, Blaney J, Lowe-Strong A, Rankin J, Campbell A, McCrum-Gardner E, et al. A randomised controlled trial testing the feasibility and efficacy of a physical activity behavioural change intervention in managing fatigue with gynaecological cancer survivors. Gynecol Oncol. 2011;122:618–24. [DOI] [PubMed] [Google Scholar]
- 202.Newton MJ, Hayes SC, Janda M, Webb PM, Obermair A, Eakin EG, et al. Safety, feasibility and effects of an individualised walking intervention for women undergoing chemotherapy for ovarian cancer: a pilot study. BMC Cancer. 2011;11:389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.von Gruenigen V, Frasure H, Kavanagh MB, Janata J, Waggoner S, Rose P, et al. Survivors of uterine cancer empowered by exercise and healthy diet (SUCCEED): A randomized controlled trial. Gynecol Oncol. 2012;125:699–704. [DOI] [PubMed] [Google Scholar]
- 204.Ledderer L, la Cour K, Mogensen O, Jakobsen E, dePont CR, Kragstrup J, et al. Feasibility of a psychosocial rehabilitation intervention to enhance the involvement of relatives in cancer rehabilitation: pilot study for a randomized controlled trial. The Patient-Patient-Centered Outcomes Research. 2013;6:201–12. [DOI] [PubMed] [Google Scholar]
- 205.McCarroll ML, Armbruster S, Frasure HE, Gothard MD, Gil KM, Kavanagh MB, et al. Self-efficacy, quality of life, and weight loss in overweight/obese endometrial cancer survivors (SUCCEED): A randomized controlled trial. Gynecol Oncol. 2014;132:397–402. [DOI] [PubMed] [Google Scholar]
- 206.Crawford JJ, Vallance JK, Holt NL, Courneya KS. Associations between exercise and posttraumatic growth in gynecologic cancer survivors. Support Care Cancer. 2015;23:705–14. [DOI] [PubMed] [Google Scholar]
- 207.Crawford JJ, Holt NL, Vallance JK, Courneya KS. Prevalence and interest in extreme/adventure activities among gynecologic cancer survivors: Associations with posttraumatic growth. Ment Health Phys Act. 2015;9:35–40. [Google Scholar]
- 208.Crawford JJ, Vallance JK, Holt NL, Steed H, Courneya KS. A phase I/II pilot study assessing the preliminary efficacy of wall climbing for improving posttraumatic growth and quality of life in gynecologic cancer survivors. Ment Health Phys Act. 2016;11:60–6. [Google Scholar]
- 209.Armbruster SD, Song J, Bradford A, Carmack CL, Lu KH, Basen-Engquist KM. Sexual health of endometrial cancer survivors before and after a physical activity intervention: A retrospective cohort analysis. Gynecol Oncol. 2016;143:589–95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Rossi A, Garber CE, Ortiz M, Shankar V, Goldberg GL, Nevadunsky NS. Feasibility of a physical activity intervention for obese, socioculturally diverse endometrial cancer survivors. Gynecol Oncol. 2016;142:304–10. [DOI] [PubMed] [Google Scholar]
- 211.Do JH, Choi KH, Ahn JS, Jeon JY. Effects of a complex rehabilitation program on edema status, physical function, and quality of life in lower-limb lymphedema after gynecological cancer surgery. Gynecol Oncol. 2017;147:450–5. [DOI] [PubMed] [Google Scholar]
- 212.Zhang X. Exercise among women with ovarian cancer: A feasibility and pre-/post-test exploratory pilot study. Oncol Nurs Forum. 2017;44(3):366–74. [DOI] [PubMed] [Google Scholar]
- 213.Zhou Y, Cartmel B, Gottlieb L, Ercolano EA, Li F, Harrigan M, et al. Randomized Trial of Exercise on Quality of Life in Women With Ovarian Cancer: Women’s Activity and Lifestyle Study in Connecticut (WALC). JNCI: Journal of the National Cancer Institute. 2017;109. 10.1093/jnci/djx072. [DOI] [PMC free article] [PubMed]
- 214.Hausmann F, Iversen V, Kristoffersen M, Gundersen H, Johannsson E, Vika M. Combined aerobic and resistance training improves physical capacity in women treated for gynecological cancer. Support Care Cancer. 2018;26:3389–96. [DOI] [PubMed] [Google Scholar]
- 215.Iyer NS, Cartmel B, Friedman L, Li F, Zhou Y, Ercolano E, et al. Lymphedema in ovarian cancer survivors: Assessing diagnostic methods and the effects of physical activity. Cancer. 2018;124:1929–37. [DOI] [PubMed] [Google Scholar]
- 216.Robertson MC, Lyons EJ, Song J, Cox-Martin M, Li Y, Green CE, et al. Change in physical activity and quality of life in endometrial cancer survivors receiving a physical activity intervention. Health Qual Life Outcomes. 2019;17:91. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 217.Friedenreich CM, Cook LS, Wang Q, Kokts-Porietis RL, McNeil J, Ryder-Burbidge C, et al. Prospective Cohort Study of Pre- and Postdiagnosis Physical Activity and Endometrial Cancer Survival. J Clin Oncol. 2020;38:4107–17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 218.Cartmel B, Hughes M, Ercolano EA, Gottlieb L, Li F, Zhou Y, et al. Randomized trial of exercise on depressive symptomatology and brain derived neurotrophic factor (BDNF) in ovarian cancer survivors: The Women’s Activity and Lifestyle Study in Connecticut (WALC). Gynecol Oncol. 2021;161:587–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Gorzelitz JS, Stoller S, Costanzo E, Gangnon R, Koltyn K, Dietz AT, et al. Improvements in strength and agility measures of functional fitness following a telehealth-delivered home-based exercise intervention in endometrial cancer survivors. Support Care Cancer. 2022;30:447–55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Rossi A, Ortiz Rossi M, Torres-Panchame R, Brojan Heyman N, Garber CE, Nevadunsky N. Perceptions of a comprehensive physical activity behavior intervention among endometrial cancer survivors with obesity. A qualitative study Gynecologic Oncology Reports. 2023;50: 101313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA: a cancer journal for clinicians. 2022;72. 10.3322/caac.21708. [DOI] [PubMed]
- 222.Miller KD, Nogueira L, Devasia T, Mariotto AB, Yabroff KR, Jemal A, et al. Cancer treatment and survivorship statistics, 2022. CA Cancer J Clinicians. 2022;72:409–36. [DOI] [PubMed] [Google Scholar]
- 223.Felix AS, Brinton LA. Cancer progress and priorities: uterine cancer. Cancer Epidemiol Biomark Prev. 2018;27:985–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Binder PS, Peipert JF, Kallogjeri D, Brooks RA, Massad LS, Mutch DG, et al. Adult Comorbidity Evaluation 27 score as a predictor of survival in endometrial cancer patients. American journal of obstetrics and gynecology. 2016;215:766-e1-. e9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 225.Smits A, Lopes A, Das N, Bekkers R, Massuger L, Galaal K. Exercise Programme in Endometrial Cancer; Protocol of the Feasibility and Acceptability Survivorship Trial (EPEC-FAST). BMJ Open. 2015;5:e009291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226.Hartman YAW, Kenkhuis M-F, Stelten S, Brouwer CG, van Lonkhuijzen LRCW, Kenter GG, et al. Demographic, clinical, and sociocognitive determinants related to physical activity and dietary intake in patients with ovarian cancer: A cross-sectional study. Gynecol Oncol. 2024;183:39–46. [DOI] [PubMed] [Google Scholar]
- 227.Savant SS, Sriramkumar S, O’Hagan HM. The Role of Inflammation and Inflammatory Mediators in the Development, Progression, Metastasis, and Chemoresistance of Epithelial Ovarian Cancer. Cancers (Basel). 2018;10(8). 10.3390/cancers10080251. [DOI] [PMC free article] [PubMed]
- 228.Mizrahi D, Broderick C, Friedlander M, Ryan M, Harrison M, Pumpa K, et al. An exercise intervention during chemotherapy for women with recurrent ovarian cancer: a feasibility study. International J Gynecologic Cancer. 2015;25 (6). 985-992 [DOI] [PubMed]
- 229.Lee JK, Jee YS. Effect of Resistance Exercise on Acquired Immunocytes in Cancer Survivors: A Pilot Study. Int Neurourol J. 2021;25:S96-105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Brennen R, Lin K-Y, Denehy L, Frawley HC. The Effect of Pelvic Floor Muscle Interventions on Pelvic Floor Dysfunction After Gynecological Cancer Treatment: A Systematic Review. Phys Ther. 2020;100:1357–71. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No datasets were generated or analysed during the current study.