ABSTRACT
We previously reported that elevated levels of diazepam binding inhibitor (DBI), also called ‘endozepine’ because it acts as an endogenous benzodiazepine equivalent on the gamma-aminobutyric acid type A receptor, constitutes a potential risk factor for the diagnosis of non-small cell lung cancer (NSCLC). Antibody-mediated neutralization of DBI improved the immunosurveillance of NSCLC in preclinical models with and without immunotherapy targeting programmed cell death protein 1 (PD-1). A pilot study in a small French-Canadian cohort (n = 205) suggested that benzodiazepine (BZD) use correlates with reduced progression-free survival in NSCLC patients receiving PD-1/PD-L1 blockade. Here, we report a retrospective analysis of the nation-wide French registry of advanced NSCLC patients treated with pembrolizumab. Among the eligible NSCLC patients surviving ≥2 months after treatment initiation (n = 31,479), 37.7% (n = 11,878) received at least two prescriptions of benzodiazepines within 90 days before to 30 days after treatment initiation. Compared to non-users (n = 19,601), BZD users had significantly reduced overall survival (hazard ratio = 1.08, 95% CI: 1.04–1.12, p < 0.001), an effect that persisted after correction using inverse probability of treatment weighting (IPTW) on sociodemographic, clinical, oncologic, and comedication variables. In a subset of 556 patients from the ONCOBIOTICS study, benzodiazepine use was associated with signs of intestinal dysbiosis and alterations in the TOPOSCORE, a prognostic marker linked to poorer outcomes in cancer patients receiving immunotherapy. We conclude that benzodiazepine use may be an independent negative prognostic factor for NSCLC patients under pembrolizumab-based immunotherapy. Future studies must determine whether withdrawal of benzodiazepines or neutralization of DBI improves the clinical response to immunotherapy.
KEYWORDS: ACBP, acyl CoA binding protein, NSCLC, lung adenocarcinoma, lung squamous carcinoma, psychotropic agents, benzodiazepines
Introduction
Mounting evidence indicates that one quintessential mechanism of oncogenesis and tumor progression is the failure of cancer immunosurveillance.1–3 Accordingly, treatments designed to enhance the anticancer immune response are now occupying a central stage in the clinical management of multiple cancer types including non-small cell lung cancer (NSCLC),4–6 which is one of the most frequent malignancies affecting both men and women.7 In France, patients with advanced, inoperable NSCLC expressing high PD-L1 levels (on ≥50% of the malignant cells) receive the anti-PD1 monoclonal antibody (mAb) pembrolizumab, while patients expressing lower PD-L1 levels (<50%) receive pembrolizumab together with chemotherapy. A close-to-comprehensive analysis of the French population, the ATHENA cohort,8 has confirmed the efficacy of pembrolizumab irrespective of treatment duration (< or ≥2 years) and has unraveled covariables that were independently associated with worse overall survival (OS), including male sex with chemo-immunotherapy, advanced age, treatment in hospitals with low recruitment of NSCLC patients, high deprivation index, inpatient hospitalization for the first pembrolizumab treatment, a history of diabetes, or prescription of diuretics, beta blockers, or painkillers.8 These observations support the idea that sociodemographic features, the general health state, and perhaps comedications affect the efficacy of immunotherapy in NSCLC patients, likely by modulating the anticancer immune response.9 Among the potential mechanisms, growing evidence suggests that shifts in microbiota composition – often associated with poor health and aging, a condition known as ‘dysbiosis’10–12– may influence NSCLC immunotherapy outcomes. In an effort to define clinically relevant signatures,13 we demonstrated that an increase in harmful fecal bacteria from the unfavorable species-interacting group (SIG) SIG1, a decrease in beneficial bacteria from the favorable group SIG2, or any deviation from the optimal relative abundance of Akkermansia species, as epitomized in the TOPOSCORE, could predict the success of immunotherapy in NSCLC.14,15 Recently, we also identified the elevation of the circulating levels of diazepam binding inhibitor (DBI, also known as acyl CoA binding protein, ACBP, or ‘endozepine’) as a biomarker of biological aging.16,17 The plasma concentration of DBI does not only increase with chronological aging18 but also correlates with cardiometabolic risk factors (such as an increase in body mass index, hyperglycemia, hyperinsulinemia, dyslipidemia, hypertension, and metabolism-associated steatohepatitis)19–21 and actually precedes, in apparently still healthy individuals, the imminent advent of major cardiovascular events20 and the diagnosis of cancers including NSCLC.22 To establish causality in these patient-relevant statistical correlations, we performed preclinical experiments showing that antibody-mediated neutralization of DBI can reduce high fat diet-induced weight gain and diabetes,23 prevent or reverse steatohepatitis,21,24 combat myocardium infarction and heart failure,20,24 and improve immunosurveillance of various cancers including NSCLC.22 Thus, injections of neutralizing anti-DBI mAb reduce cancer progression as a monotherapy and enhance tumor growth control by anti-PD-1 mAb in murine models.22 Accordingly, anti-DBI mAb increases the infiltration of tumors by activated, non-exhausted cytotoxic T lymphocytes but reduces the infiltration of such tumors by regulatory T cells.22,25
As its name indicates, diazepam binding inhibitor (DBI) displaces the prototypic benzodiazepine diazepam from its cell surface receptor, which is a combination of specific subunits (alpha-1 and gamma-2) of the gamma-aminobutyric acid (GABA) A type receptor (GABARA).26 A specific single amino acid substitution (F77I) in gamma-2 (gene/protein symbol: GABRG2) abolishes the binding of both DBI and diazepam to cells.27–30 Mice bearing this mutation in homozygosity (genotype: Gabrg2F77I/F77I) bear a similar phenotype as mice subjected to the conditional knockout of Dbi or receiving systemic injections of anti-DBI mAb.24 Importantly, administration of diazepam to mice fully reversed the beneficial effects of anti-DBI mAb on tumor progression in the context of chemoimmunotherapy,31 underscoring the functional relationship between the exogenous benzodiazepine and its endogenous equivalent (DBI or ‘endozepine’). In line with these preclinical results, we observed in a small French-Canadian cohort of NSCLC patients under immunotherapy targeting PD-1 or PD-L1 (n = 205) that benzodiazepine use correlated with reduced progression-free survival (PFS). Importantly, in this cohort, other psychotropic drugs than benzodiazepines did not affect PFS, suggesting a ‘specific’ effect of benzodiazepines.31 However, this cohort was too heterogeneous (e.g., because patients from two different countries received a range of antibodies targeting PD-1 or PD-L1) and too small (n = 205) to reach definitive conclusions.
For this reason, we decided to interrogate the French nation-wide ATHENA cohort, the largest real-world dataset on pembrolizumab treatment (n > 50,000),8 to identify possible effects of benzodiazepine medication on the clinical outcome of NSCLC patients treated with one single PD-1 antibody, pembrolizumab. In addition, we show that benzodiazepine use correlates with an unfavorable TOPOSCORE.
Methods
ATHENA study
Ethics and data protection
ATHENA is a retrospective cohort study using a comprehensive administrative database aimed firstly at exploring the optimal duration of pembrolizumab and secondly real-life prognosis factors in patients with advanced NSCLC.8 This study was conducted in accordance with the Declaration of Helsinki, as well as institutional and ethical rules concerning research regarding patient data. In accordance with French regulations applicable to the French National Health Insurance database (Système National des Données de Santé, SNDS), a strictly anonymous database,32 no informed consent was required. This study has been declared and approved prior to data extraction on the Health Data Hub online platform of the French data protection agency (Commission nationale de l’informatique et des libertés – CNIL, No. F20230713113749).
Study population
The database33 included (i) demographic data, (ii) hospital discharge reports, (iii) outpatient care, and (iv) long-term illness records. The cohort initially enrolled 50,083 patients with advanced stage metastatic non-small cell lung cancer treated with pembrolizumab. Among those 41,529 patients received pembrolizumab as first line (alone or under chemo-immunotherapy) between January 1, 2015, and December 31, 2022. Only patients who survived a minimum of 2 months after the initiation of pembrolizumab-based immunotherapy and without missing data were included in the analysis (n = 31,479) to study the possible effects of benzodiazepine exposure.
Concomitant benzodiazepine medication
Concomitant medications with benzodiazepines (indicated according to the World Health Organization Anatomical Therapeutic Chemical [ATC] classification as ATC N05BA) were identified from outpatient drug delivery data as a minimum of two prescriptions between day 90 before and day 30 after the first administration of pembrolizumab. This time interval was chosen because it corresponds to the one during which antibiotic use compromises the efficacy of immunotherapy in meta-analyses.34,35 ATC N05BA lists benzodiazepine derivatives including adinazolam, alprazolam, bentazepam, bromazepam, camazepam, chlordiazepoxide, clobazam, clotiazepam, cloxazolam, diazepam, fludiazepam, ethyl loflazepate, etizolam, halazepam, ketazolam, lorazepam, medazepam, mexazolam nordazepam, oxazepam, pinazepam, potassium clorazepate, prazepam, and tofisopam.
Statistical methods
We adjusted the population by means of a Cox model pondered by the Inverse Probability of Treatment Weighting (IPTW) score constructed with the following variables: sex, immunotherapy or chemoimmunotherapy, age, year of diagnosis, hospital type according to the volume of patient recruitment (high in upper decile, intermediate, or volume in the bottom 69%), full or day hospitalization, history of myocardial infarction, history of heart failure, history of stroke, history of liver disease, peripheral artery disease, chronic-obstructive pulmonary disease, history of renal failure, history of diabetes, use of antihypertensive drugs such as angiotensin converting enzyme and angiotensin receptor 2 antagonists, lipid-lowering agent, antiplatelet, anticoagulant, diuretic drugs, beta blockers, non-steroidal anti-inflammatory drugs, antipsychotics, antidepressants, thyroid hormone replacement, painkillers, opiate replacement therapy, exposure to antibiotics, proton pump inhibitors, corticosteroids, antiepileptic drugs, history of radiotherapy, as well as precariousness index of the municipality of residence (to correct for sociodemographic parameters). Confidence intervals were estimated with the bootstrap procedure. Time-to-event endpoints were estimated using the Kaplan–Meier method. All analysis was performed with R (v4.1.2).
ONCOBIOTICS cohort
Ethics and study population
The Oncobiotics trial (NCTC04567446) is a multicenter study assessing the impact of the microbiome on outcomes in advanced NSCLC patients treated with anti-PD-(L)1 therapies, either alone or in combination with chemotherapy or tyrosine kinase inhibitors. Conducted across 14 centers in France and Canada, the study followed standard care until disease progression, intolerable side effects, or a maximum of 2 years of ICI treatment. Eligibility criteria and baseline data, including recent medications, are outlined in the trial protocol and recorded in electronic case reports.14 All participants provided written informed consent. GDPR and anonymization procedures were adhered to, in accordance with Oncobiome H2020 at ClinicObiome, Gustave Roussy. Data and sample collection complied with regulatory and ethical requirements, as well as ICH E6(R2) Good Clinical Practice (GCP) and the Declaration of Helsinki. Information on ancestry, race, ethnicity, and socioeconomic status was excluded due to France’s Data Protection Act No. 78–17.
Concomitant benzodiazepine and antibiotics medication
Concomitant medications with benzodiazepines (indicated according to the World Health Organization Anatomical Therapeutic Chemical [ATC] classification as ATC N05BA) and antibiotics (ATC J01) were identified from outpatient drug delivery data. We focused on patients with a minimum of two prescriptions within 60 days prior to the first administration of immune checkpoint inhibitors. This time interval was selected because it corresponds to the one during which antibiotic use has been shown to compromise the efficacy of immunotherapy in meta-analyses. Among 556 patients, stool samples were available from all participants before ICI initiation and, in some cases, after the use of antibiotics or benzodiazepines. These samples were used for shotgun metagenomics analysis to define microbiota composition (Table S1).
Microbiota analyses
Stool samples were processed for DNA extraction and sequencing using Ion Proton technology, following the MetaGenoPolis protocols. Prokaryotic taxonomic profiling at species level was performed using MetaPhlAn4 (vJan21_CHOCOPhlAn) (doi:https://doi.org/10.1038/s41587–023–01688-w). TOPOSCORE classification was based on publicly available algorithms (https://github.com/valerioiebba/TOPOSCORE), as previously published.14 Microbial alpha diversity (Shannon and Richness metrics) was computed with the vegan package in R environment. For beta diversity, statistical significance was determined using a PERMANOVA model, accounting for age, gender, body mass index (BMI), Eastern Cooperative Oncology Group (ECOG) performance status, antibiotics, and benzodiazepine (BZD) use. Discriminant species were identified using the Wilcoxon–Mann–Whitney test and by considering false discovery rate for multiple hypothesis testing correction.
Statistical methods
Progression free survival (PFS) was defined as time to progression or death and overall survival (OS) as the time to death, with patients censored at the last follow-up time if no event occurred. Survival analyses were performed using Kaplan–Meier curves, log-rank tests, and Cox regression, with significance defined as p < 0.05. Variables included in the multivariate model are reported in Supplemental Table S2. Least discriminant analysis effect size (LEfSe) analysis was used to determine discriminant species, which were identified using the Wilcoxon–Mann–Whitney test and by considering false discovery rate for multiple hypothesis testing correction.
Results
Pembrolizumab-treated lung cancer patients and benzodiazepine comedication
Our nation-wide database includes 50,083 patients with NSCLC that were treated with pembrolizumab. Among these patients, we only included those receiving pembrolizumab alone or together with chemotherapy in first line, who survived >2 months after treatment initiation and for which records on comedication were available to be able to measure the potential effect of benzodiazepine medication (n = 31,479). Among these 31,479 participants, 11,878 (37.7%) received at least two prescriptions of benzodiazepines within 90 days before and 30 days after treatment initiation. We focused on this interval because it has been pinpointed as particularly critical for the long-term impact of antibiotic medication on immunotherapy outcome.34,35 The characteristics of this population are listed in Table 1. Benzodiazepine users were more often women (40.9% vs 28.9% in non-users), treated with chemo-immunotherapy (69.6% vs 63.5% in non-users), exposed to antibiotics (47.9% vs 39.6% in non-users), to steroids (67.5% vs 54.1% in non-users), and to proton pump inhibitors (63.9% vs 47.7% in non-users).
Table 1.
Patient’s characteristics.
| Receiving benzodiazepine (N = 11878) |
Not receiving benzodiazepine (N = 19601) |
Overall (N = 31479) |
|
|---|---|---|---|
| Sex | |||
| Female | 4858 (40.9%) | 5668 (28.9%) | 10526 (33.4%) |
| Male | 7020 (59.1%) | 13933 (71.1%) | 20953 (66.6%) |
| Age at diagnosis | |||
| Mean (SD) | 63.1 (9.59) | 65.6 (9.77) | 64.7 (9.78) |
| Type of hospital | |||
| High volume | 4853 (40.9%) | 8423 (43.0%) | 13276 (42.2%) |
| Intermediate volume | 5314 (44.7%) | 8234 (42.0%) | 13548 (43.0%) |
| Low volume | 1711 (14.4%) | 2944 (15.0%) | 4655 (14.8%) |
| Deprivation index | |||
| First quintile | 1920 (16.2%) | 3351 (17.1%) | 5271 (16.7%) |
| Second quintile | 2356 (19.8%) | 4063 (20.7%) | 6419 (20.4%) |
| Third quintile | 2480 (20.9%) | 4032 (20.6%) | 6512 (20.7%) |
| Fourth quintile | 2639 (22.2%) | 4153 (21.2%) | 6792 (21.6%) |
| Fifth quintile | 2483 (20.9%) | 4002 (20.4%) | 6485 (20.6%) |
| Strategy | |||
| Pembrolizumab alone | 3605 (30.4%) | 7164 (36.5%) | 10769 (34.2%) |
| Pembrolizumab plus chemotherapy | 8273 (69.6%) | 12437 (63.5%) | 20710 (65.8%) |
| Type of first hospitalization | |||
| Day hospital | 8685 (73.1%) | 15233 (77.7%) | 23918 (76.0%) |
| Inpatient hospitalization | 3193 (26.9%) | 4368 (22.3%) | 7561 (24.0%) |
| Exposition to antibiotics at baseline | |||
| No | 6188 (52.1%) | 11850 (60.4%) | 18038 (57.3%) |
| Yes | 5690 (47.9%) | 7754 (39.6%) | 13444 (42.7%) |
| Exposition to steroid at baseline | |||
| No | 3862 (32.5%) | 8996 (45.9%) | 12858 (40.8%) |
| Yes | 8016 (67.5%) | 10605 (54.1%) | 18621 (59.2%) |
| Exposition to PPI at baseline | |||
| No | 4292 (36.1%) | 10260 (52.3%) | 14552 (46.2%) |
| Yes | 7586 (63.9%) | 9341 (47.7%) | 16927 (53.8%) |
| Diabetes at diagnosis | |||
| No | 10593 (89.2%) | 16878 (86.1%) | 27471 (87.3%) |
| Yes | 1285 (10.8%) | 2723 (13.9%) | 4008 (12.7%) |
| Antipsychotic at diagnosis | |||
| No | 11236 (94.6%) | 19182 (97.9%) | 30418 (96.6%) |
| Yes | 642 (5.4%) | 419 (2.1%) | 1061 (3.4%) |
| Antidepressant at diagnosis | |||
| No | 8728 (73.5%) | 17561 (89.6%) | 26289 (83.5%) |
| Yes | 3150 (26.5%) | 2040 (10.4%) | 5190 (16.5%) |
Impact of benzodiazepines on overall survival
As compared to control patients not receiving benzodiazepines (n = 19,601), benzodiazepine users (n = 11,878) exhibited diminished OS (Figure 1, hazard ratio [HR] = 1.08, 95% confidence interval [CI] 1.04 to 1.12). This statistically significant (p < 0.001) negative impact of benzodiazepine medication survived correction of the Cox model by means of an IPTW score pondering sociodemographic variables (age, sex, year of diagnosis, deprivation index), the history of arteriosclerotic, cardiometabolic, hepatic, renal or pulmonary disease, anticancer treatments including chemotherapy and radiotherapy, as well as other comedications with antibiotics, antihypertensive, antidiabetic, lipid-lowering, antiplatelet, anticoagulant, diuretic, beta receptor blocking, anti-inflammatory, analgesic, antipsychotic, or antidepressant drugs. This is particularly important, as previous studies have documented the negative effects of several of these variables on the OS of NSCLC cancer patients. Such observations apply to old age,8,36 male sex in the group of patients treated with chemoimmunotherapy,8,36 treatment in hospitals with a low volume of NSCLC cancer patients,8 residence in medically neglected areas,8 inpatient hospitalization during the first cycle of pembrolizumab treatment,8 past or present diabetes,8 treatment with antibiotics,37 diuretics,8 beta blockers,8,38 and painkillers including paracetamol.8,39,40
Figure 1.
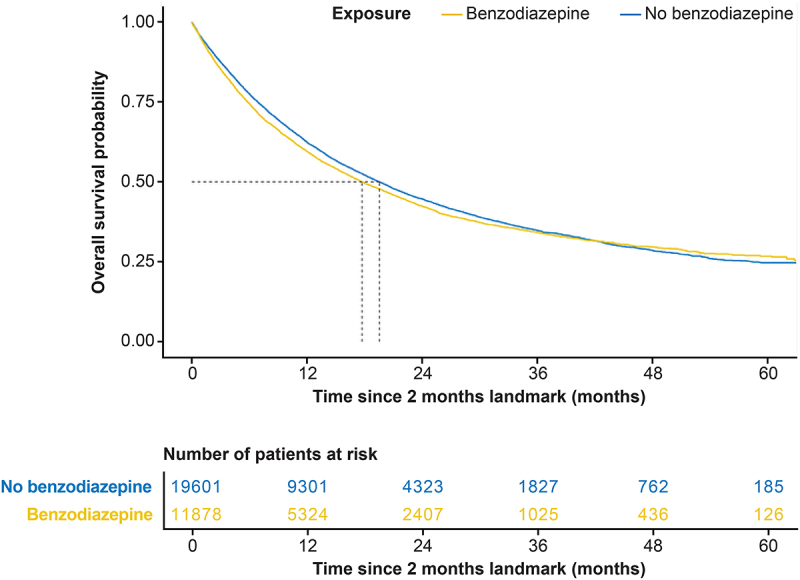
Impact of benzodiazepines on overall survival of lung cancer patient under pembrolizumab-based immunotherapy in the French national database including patients with non-small cell lung cancer that received immunotherapy. The graph compares the survival of patients after the 2 month-landmark that received benzodiazepines (orange line) or not (blue line). The number of surviving patients is indicated below the graph for selected time points.
In conclusion, in this cohort, benzodiazepine use could be considered a negative prognostic factor for NSCLC patients treated with pembrolizumab, even after statistical correction for sociodemographic and health-related variables.
Impact of benzodiazepines on the intestinal microbiota
To investigate the impact of benzodiazepine use on the composition of the fecal microbiota, we analyzed data from our previous study (NCT03084471), which included microbial data from 556 patients with NSCLC. In this cohort, benzodiazepine use was associated with a significant reduction in OS (p = 0.023, Figure 2a), although effect size was smaller than that observed for the use of antibiotics (p = 0.008) in univariate analyses (Figure 2b). Indeed, in this relatively small cohort, benzodiazepine use was significantly associated with several confounding factors (Supplemental Table S1). Statistically, in this smaller cohort, the impact of benzodiazepines on OS did not remain significant in multivariate analyses (Supplemental Table S2). However, the combined use of benzodiazepines and antibiotics had a markedly stronger (and independent) negative effect on OS compared to either drug category alone (Figure 2c). The use of benzodiazepines had a significant effect on beta diversity after adjusting for age, gender, body mass index, and ECOG performance status (Supplemental Fig. S1A). When restricting the analysis to non-antibiotic-treated patients, a difference in fecal microbial composition was observed between patients consuming benzodiazepines compared to non-users, as determined by metagenomic shotgun sequencing and linear discriminant analysis effect size (LEfSe) analyses (Supplemental Fig. S1B). Indeed, in this comparative analysis, benzodiazepine use turned out to be associated with a significant increase in the abundance of four bacterial species belonging to the unfavorable species-interacting group (SIG) SIG1, but a decrease in species belonging to the favorable group SIG2. We recently demonstrated the prognostic value of classifying cancer patients based on their fecal microbiota into three groups: those with a predominantly unfavorable microbiota (SIG1), a mostly favorable microbiota (SIG2), or an intermediate (‘grey’) microbiota.14 Notably, among non-antibiotic users, benzodiazepine consumption was associated with a significant shift from SIG2 toward SIG1, a trend that was further exacerbated by antibiotic use (Figure 3a). Building on this observation, we calculated the TOPOSCORE, which combines information on the presence of bacterial species falling into the SIG1 and SIG2 categories with the abundancy of decisive Akkermansia species, to establish a dichotomic classification of cancer patients into SIG1+ and SIG2+ individuals. SIG1+ cancer patients under immunotherapy have a reduced OS compared to SIG2+ patients, as documented for NSCLC as well as other cancer types.14 Of note the SIG1/SIG2 prognostic ratio was increased in patients using both antibiotics and benzodiazepines (p = 0.00021) (Figure 3b).
Figure 2.
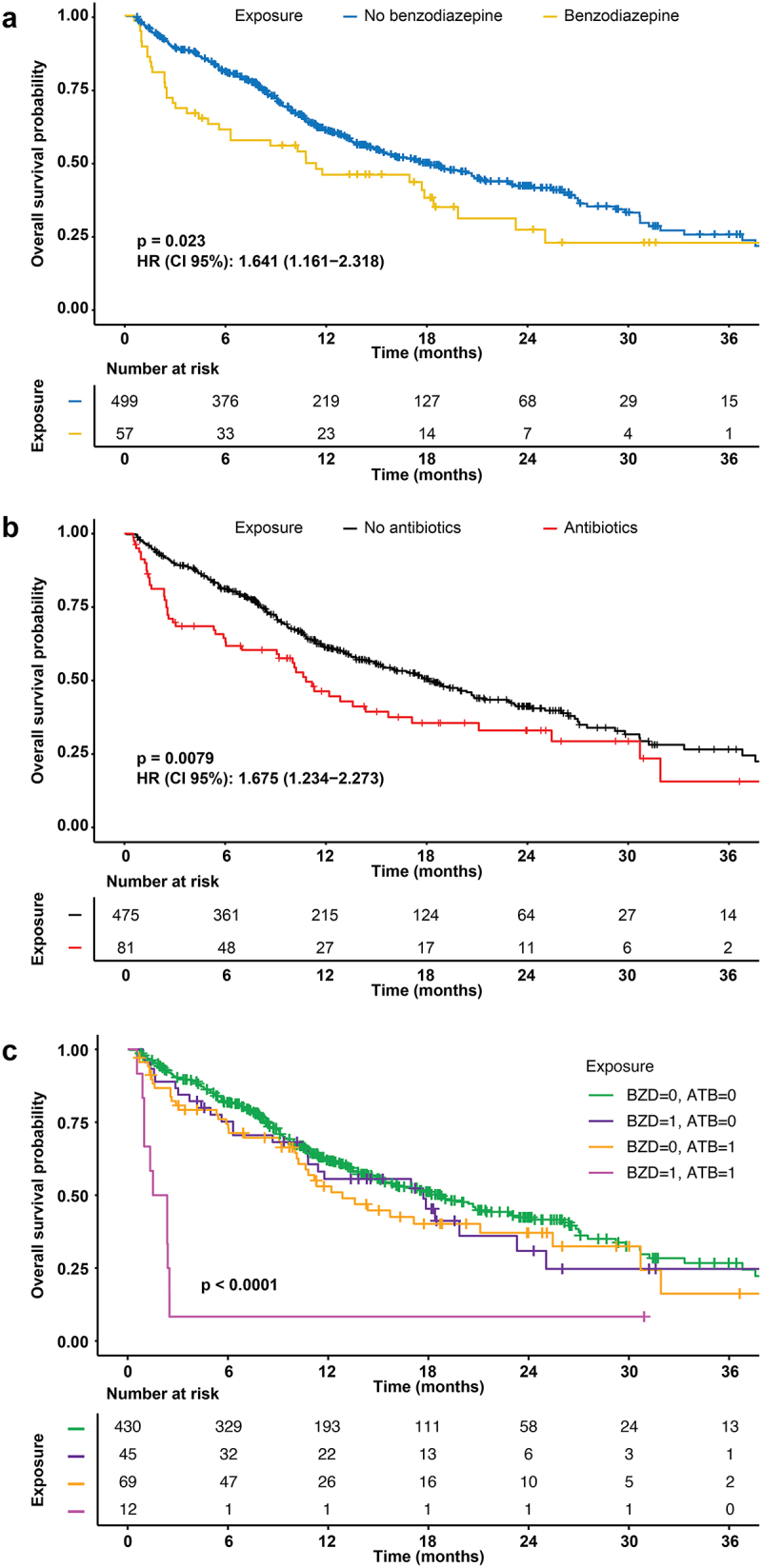
Impact of benzodiazepine and antibiotic use on overall survival of patients enrolled in the ONCOBIOTICS trial.
(a) Impact of benzodiazepines (BZD) on overall survival (OS). (b) Impact of antibiotics (ATB) use on OS. (c) Combined use of antibiotics and benzodiazepines on OS. Use of benzodiazepines or antibiotics within the 60 days prior to treatment initiation is coded ‘1’, while the absence of prescription is coded ‘0’. p-values were calculated by means of the log-ranks (Cox–Mantel test). The number of surviving patients is indicated below the graph for selected time points.
Figure 3.
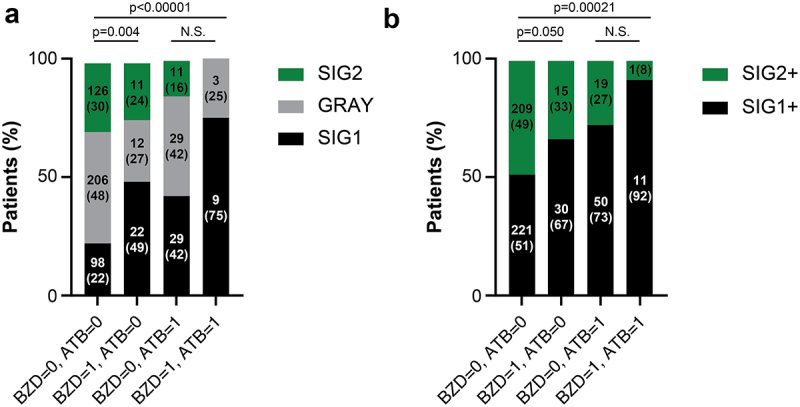
Impact of benzodiazepine and antibiotic use on the fecal microbiota in patients enrolled in the ONCOBIOTICS trial.
(a) Classification of patients into three groups (SIG1, SIG2, Gray) according to their benzodiazepine and antibiotic use. (b) TOPOSCORE classification of patients into two groups (SIG1+, SIG2+). Use of benzodiazepines (BZD) or antibiotics (ATB) within the 60 days prior to treatment initiation is coded ‘1’, while the absence of prescription is coded ‘0’. Classifications were based on algorithms published in.14 Statistics were calculated by means of the chi-square test while replacing 0 values by 1. Absolute number of patients and relative percentages are displayed within bar plots without and within parentheses, respectively.
In conclusion, it appears that benzodiazepine use is associated with shifts in the fecal microbiota that are linked to poor immunotherapy outcome.
Discussion
Our study suggests that benzodiazepines have a negative impact on NSCLC cancer immunotherapy outcome in a French nationwide cohort involving patients treated with pembrolizumab. Hence, this retrospective analysis supports the idea that benzodiazepines subvert immunosurveillance in NSCLC patients. Of note, as shown here, more than half of the patients of the ATHENA cohort were prescribed benzodiazepines during the critical time window (90 days before until 30 days after initiation of pembrolizumab immunotherapy), reflecting the highly prevalent use of this class of drugs in France.41,42 Current epidemiological estimates indicate that, in Western countries, 4–8% of the adult population regularly uses benzodiazepines,43 commensurate with the high level of addictiveness of this class of psychotropic drugs.44,45
Beyond the epidemiological association between benzodiazepine use and reduced efficacy of immunotherapy, the present study reveals a correlation between benzodiazepine consumption and stigmata of intestinal dysbiosis. The direction of causality between these two phenomena is still ambiguous since, on the one hand, oral benzodiazepine administration may affect the intestinal microbiota,46,47 and on the other hand, gut intestinal dysbiosis may affect immunity, general and mental health, hence increasing the need for benzodiazepine medication.48 Thus, future preclinical and clinical studies are needed to disentangle the potential mechanistic links between benzodiazepine use, dysbiosis, and poor immunotherapy outcome.
At this stage, we do not know whether the negative impact of benzodiazepines use would be independent of other established modulatory effects of the pembrolizumab response such as performance status, tumor burden, PD-L1 expression, tumor mutational burden, or gene expression signatures within the tumor49 (which all are not available in the SNDS database). In addition to cancer-related variables, other unmeasured confounders such as anxiety severity, insomnia, alcohol or tobacco consumption, food intake, physical activity, or the need for palliative care may influence the response to PD-1 therapy and hence were not corrected by our IPTW adjustment, If benzodiazepines had an immunosuppressive effect, one would expect that their prolonged use is associated with a surge of serious infectious diseases as well as cancer diagnosis. Indeed, there are preclinical and patient-centered epidemiological studies suggesting that this is the case.
The use of benzodiazepine has been linked to an increased risk of lung infections. Diazepam enhances the susceptibility of hamsters to respiratory infection with Mycobacterium bovis,50 as well as that of mice to infection with cowpox (CPXV), vaccinia virus (VACV),51 and Streptococcus pneumoniae.52 Accordingly, a meta-analysis of clinical studies found that benzodiazepine and benzodiazepine-related drugs increased risk of pneumonia among current users (with the most recent prescription within 30 days of the index data; OR = 1.4; 95% CI, 1.22–1.6), recent users (with the prescription within 31–90 days of the index date; OR = 1.38; 95% CI, 1.06–1.8) users, but not past users (prescription beyond 90 days; OR = 1.11; 95% CI, 0.96–1.27).53 This effect was particularly strong for the population younger than 65 years, with an increase in lung infections by 80%.53 Pneumonia has also been reported for hospitalized schizophrenic patients, in whom several benzodiazepines (such a diazepam, lorazepam, midazolam, and triazolam)54 and the benzodiazepine derivative clozapine55,56 were associated with increased risk.57
Of note, the effects of benzodiazepine are not limited to lung infection. Thus, diazepam sensitizes mice to intraperitoneal infection with Salmonella typhimurium.58 A retrospective population-based cohort study in South Korea revealed that long-term benzodiazepine use observed in 16 686 adult patients with sepsis was associated with increased 90-day mortality compared with propensity score-matched non-users (HR = 1.75, 95% CI 1.70–1.81).59 A recent study performed on the Swedish national registry focusing on the population under 65 years found that the use of benzodiazepines and related drugs (BZRD) was associated with an 83% relative increase in risk of serious viral or bacterial infections in any location of the body.60 This was found for 713,896 BZDR recipients compared to 713,896 age, sex, and residence-matched controls (HR = 1.83, 95% CI 1.79–1.89). The risk of serious infection was also found increased in a co-twin cohort involving 9197 BZRD-treated twins with their BZRD-untreated siblings (HR = 1.55, 95% CI 1.23–1.97) and when comparing 434,900 BZRD recipients to 428,074 patients taking selective serotonin reuptake inhibitors (HR = 1.33, 95% CI 1.30–1.35). The observed risks were similar across different types of BZDRs, with a dose–response association between cumulative BZRD dosage and risk of serious infections.60
Beyond their association with infectious diseases, prolonged benzodiazepine (ab)use has also been linked to an increase in the incidence of malignant disease. A pioneering study was conducted on a rural US population matching users of hypnotics with control subjects by sex, age ±5 years, smoking status, and start of period of observation. High-dose zolpidem ( > 800 mg/year, mean 3600, N = 1427) was found to be associated with higher cancer risk (HR = 1.28, 95% CI 1.03–1.59), and this was also observed for two dose levels of temazepam, an intermediate dose (240–1640 mg/year, mean 683, N = 613; HR = 1.44, 95% CI 1.05–1.98) and a high dose ( > 1640 mg/year, mean 7777, N = 665; HR = 1.99; 95% CI 1.57–2.52).61 A population-based retrospective analysis matching 59,647 benzodiazepine users with controls by age and sex revealed an increase in cancer risk in Taiwan. This study found an increase in overall cancer risk (HR = 1.19, 99.6% CI 1.08–1.32) and in particular liver cancer (HR = 1.45; 99.6% CI 1.10–1.90), prostate cancer (HR = 1.72, 99.6% CI 1.10–2.70), and bladder and kidney cancer (HR = 1.76, 99.6% CI 1.16–2.67).62 In the Danish nationwide registers, 152 510 cases with a first-time cancer who were matched (1:8) by age and gender to 1,220,317 cancer-free controls. In this study, long-term use of BZRD was defined by a cumulative amount of ≥500 defined daily doses of BZRD within a period of 1–5 years prior to the index date. The adjusted odds ratio (OR) of long-term BZRD for all cancers was 1.09 (95% CI 1.04–1.14). ORs were particularly high in descending order for liver (OR = 1.81, 95% CI 1.18–2.80), esophagus (OR = 1.43, 95% CI 1.01–2.02), stomach (OR = 1.40, 95% CI 1.05–1.88), kidney (OR = 1.39, 95% CI 1.01–1.91), lung (OR = 1.38, 95% CI 1.23–1.54) and pancreas (OR = 1.35, 95% CI 1.02–1.79).63 A later study on the same cohort included 94 923 patients with cancer and 759 334 age- and sex-matched (1:8) population controls, then implemented propensity score (PS) calibration to eliminate confounding factors and concluded similarly that benzodiazepine use was associated with an increased cancer risk (OR = 1.09, 95% CI 1.00–1.19).64
A meta-analysis of observational studies including a total of 18 case–control studies and 4 cohort studies (213,823 patients with cancer and 1,683,780 controls) found that benzodiazepine use was significantly associated with a dose-dependent increased risk of all cancers (pooled OR or relative risk [RR] 1.19, 95% CI 1.16–1.21). This effect was particularly strong – in descending order – for brain cancer (OR/RR = 2.08, 95% CI 1.77–2.44), esophagus cancer (OR/RR = 1.55, 95% CI 1.30–1.85), pancreatic cancer (OR/RR = 1.39, 95% CI 1.17–1.64), renal cancer (OR/RR = 1.30, 95% CI 1.14–1.49), prostate cancer (OR/RR = 1.26, 95% CI 1.16–1.37), liver cancer (OR/RR = 1.22, 95% CI 1.13–1.31), and lung cancer (OR/RR = 1.20, 95% CI 1.12–1.28).65 Another meta-analysis came to a similar conclusion (RR = 1.25, 95% CI 1.15–1.36), again revealing the highest risk for brain cancer (RR = 2.06, 95% CI 1.76–2.43),66 perhaps reflecting the fact that benzodiazepines have been designed to cross the blood–brain barrier and to accumulate in the central nervous system to exert their anxiolytic and sedative effects.67
Altogether, multiple retrospective studies suggest immunosuppressive effects of benzodiazepines that increase the incidence of a wide array of infectious and malignant diseases. The present study extends this conclusion to cancer immunotherapy that, in the case of NSCLC and its treatment by the anti-PD-1 antibody pembrolizumab, apparently is compromised by benzodiazepines and associated with intestinal dysbiosis. It will be important to broaden this kind of exploration to other cancer types and other modalities of immunotherapy including PD-L1 or CTLA-4 blockade, CAR-T cells, oncolytic viruses, or therapeutic vaccination.
Limitations of the study
The present study reports a statistical association between death from any cause and the use of benzodiazepine over the period studied in very large cohort ( >50 000 patients). Hence, the study does not explore disease-specific survival (rather than overall survival) and only evaluates benzodiazepine use within a limited time window. Although it appears plausible that, given their high level of addictiveness, benzodiazepine use is continued in most of the individuals, no information on possible cumulative effects has been retrieved. Moreover, patients with more advanced NSCLC or more severe comorbidities could be particularly exposed to benzodiazepines. The ATHENA cohorts do not report major factor that influence immunotherapy benefit such as performance status, PD-L1 expression, tumor burden, presence of liver metastases, or tumor mutational burden. The need for an anxiolytic treatment could be related to a more symptomatic disease, reflecting a higher tumor burden and a worse outcome. The mechanisms through which benzodiazepines cause immunosuppression, which may include intestinal dysbiosis, require further experimental and clinical exploration.
Irrespective of these limitations, the available data from this and other cohort studies indicate that benzodiazepine (ab)use by NSCLC patients under PD-1/PD-L1 blockade is associated with reduced PFS, OS, and even immune-related adverse events, in line with general immunosuppressive effects of this class of psychotropic drugs.
Supplementary Material
Acknowledgments
We thank Stéphanie Foulon (Biostatistics and Epidemiology Department, Gustave Roussy, Paris-Saclay University, Villejuif, France) for granting us access to the SNDS data base. LM participated to the conceptualization, analysis, and writing of the study. AR and BB, respectively, conducted and supervised the analyses on the ATHENA cohort. CU, LD, MF, CACS, and EP collected and analyzed the data from the ONCOBIOTICS cohort and participated to the editing of the manuscript. BR and LZ provided resources, supervision, and revising input. GK conceptualized and wrote the study.
Funding Statement
LD is supported by Gustave Roussy Foundation, European Union’s Horizon 2020 research and innovation program [under grant agreement number: 825410, Project Acronym: ONCOBIOME, project title: Gut OncoMicrobiome Signatures (GOMS) associated with cancer incidence, prognosis and prediction of treatment response], French Ministry of Health PIA5, ANR, program RHU5 ‘ANR-21-RHUS-0017 IMMUNOLIFE’ (project title: ‘effects of antibiotics on the clinical outcome to immunotherapy’), SIGN’IT ARC Foundation [MICROBIONT-PREDICT (2021), Made-IT (2023)]. LZ is supported by European Union’s Horizon 2020 research and innovation program [under grant agreement number: 825410, Project Acronym: ONCOBIOME, project title: Gut OncoMicrobiome Signatures (GOMS) associated with cancer incidence, prognosis and prediction of treatment response], French Ministry of Health PIA5, ANR, program RHU5 ‘[ANR-21-RHUS-0017] IMMUNOLIFE’ (project title: ‘effects of antibiotics on the clinical outcome to immunotherapy’), European Research Council [under grant agreement number [ERC-2023-ADG - MADCAM – 101142062], project acronym: MADCAM, project title: Microbiota-induced trafficking of immunosuppressive intestinal cells into tumors], The IHMCSA [project ID 964590] project has received funding from the European Union’s Horizon 2020 research and innovation program and Institut National du Cancer MAdCAM INCA_ 16698. GK is supported by the Ligue contre le Cancer (équipe labellisée); Agence National de la Recherche [ANR-22-CE14-0066 VIVORUSH, ANR-23-CE44-0030 COPPERMAC, ANR-23-R4HC-0006 Ener-LIGHT]; Association pour la recherche sur le cancer (ARC); Cancéropôle Ile-de-France; Fondation pour la Recherche Médicale (FRM); a donation by Elior; European Joint Program on Rare Diseases (EJPRD) Wilsonmed; European Research Council Advanced Investigator Award [ERC-2021-ADG, Grant No. 101052444; project acronym: ICD-Cancer, project title: Immunogenic cell death (ICD) in the cancer-immune dialogue]; The ERA4 Health Cardinoff Grant Ener-LIGHT; European Union Horizon 2020 research and innovation programmes Oncobiome [grant agreement number: 825410, Project Acronym: ONCOBIOME, Project title: Gut OncoMicrobiome Signatures [GOMS] associated with cancer incidence, prognosis and prediction of treatment response, Prevalung [grant agreement number 101095604, Project Acronym: PREVALUNG EU, project title: Biomarkers affecting the transition from cardiovascular disease to lung cancer: towards stratified interception], Neutrocure [grant agreement number 861878: Project Acronym: Neutrocure; project title: Development of ‘smart’ amplifiers of reactive oxygen species specific to aberrant polymorphonuclear neutrophils for treatment of inflammatory and autoimmune diseases, cancer and myeloablation]; National support managed by the Agence Nationale de la Recherche under the France 2030 programme [reference number 21-ESRE-0028, ESR/Equipex+ Onco-Pheno-Screen]; Hevolution Network on Senescence in Aging [reference HF-E Einstein Network]; Institut National du Cancer (INCa); Institut Universitaire de France; LabEx Immuno-Oncology ANR-18-IDEX-0001; a Cancer Research ASPIRE Award from the Mark Foundation; PAIR-Obésité INCa_1873, the RHUs Immunolife and LUCA-pi [ANR-21-RHUS-0017 and ANR-23-RHUS-0010, both dedicated to France Relance 2030]; Seerave Foundation; SIRIC Cancer Research and Personalized Medicine [CARPEM, SIRIC CARPEM INCa-DGOS-Inserm-ITMO Cancer_18006 supported by Institut National du Cancer, Ministère des Solidarités et de la Santé and INSERM]. This study contributes to the IdEx Université de Paris Cité [ANR-18-IDEX-0001].
Disclosure statement
LZ has held research contracts with Glaxo Smyth Kline, Incyte, Lytix, Kaleido, Innovate Pharma, Daiichi Sankyo, Pileje, Merus, Transgene, 9 m, Tusk and Roche, was on the on the Board of Directors of Transgene, is a cofounder of everImmune, and holds patents covering the treatment of cancer and the therapeutic manipulation of the microbiota. The funders had no role in the design of the study, in the writing of the manuscript, or in the decision to publish the results. BB received Grants from AbbVie, Amgen, AstraZeneca, Chugai Pharmaceutical, Daiichi-Sankyo, Ellipse, EISAI, Genmab, Genzyme Corporation, Hedera Dx, Inivata, IPSEN, Janssen, MSD, Pharmamar, Roche-Genentech, Sanofi, Socar Research, Tahio Oncology, and Turning Point Therapeutics. GK has been holding research contracts with Daiichi Sankyo, Eleor, Kaleido, Lytix Pharma, PharmaMar, Osasuna Therapeutics, Samsara Therapeutics, Sanofi, Sutro, Tollys, and Vascage. GK is on the Board of Directors of the Bristol Myers Squibb Foundation France. GK is a scientific co-founder of everImmune, Osasuna Therapeutics, Samsara Therapeutics, and Therafast Bio. GK is in the scientific advisory boards of Hevolution, Institut Servier, Longevity Vision Funds, and Rejuveron Life Sciences/Centenara Labs AG. GK’s brother, Romano Kroemer, was an employee of Sanofi and now consults for Boehringer-Ingelheim. The other authors do not declare any conflict of interest related to this study.
Authors contributions
LM participated to the conceptualization, analysis, and writing of the study. AR and BB, respectively, conducted and supervised the analyses on the ATHENA cohort. CU, LD, MF, CACS, and EP collected and analyzed the data from the ONCOBIOTICS cohort and participated to the editing of the manuscript. BR and LZ provided resources, supervision, and revising input. GK conceptualized and wrote the study.
Data availability statement
Individual patient data cannot be shared due to protection of privacy, but access to the ATHENA cohort can be requested to the National Health Data System on the French Data Hub platform. Metagenomics data from the ONCOBIOTICS will be made available upon reasonable request.
Supplementary material
Supplemental data for this article can be accessed online at https://doi.org/10.1080/2162402X.2025.2528955
References
- 1.Schreiber RD, Old LJ, Smyth MJ.. Cancer immunoediting: integrating immunity’s roles in cancer suppression and promotion. Science. 2011. 331(6024):1565–15. doi: 10.1126/science.1203486. [DOI] [PubMed] [Google Scholar]
- 2.Mascaux C, Angelova M, Vasaturo A, Beane J, Hijazi K, Anthoine G, Buttard B, Rothe F, Willard-Gallo K, Haller A, et al. Immune evasion before tumour invasion in early lung squamous carcinogenesis. Nature. 2019;571(7766):570–575. doi: 10.1038/s41586-019-1330-0. [DOI] [PubMed] [Google Scholar]
- 3.Swanton C, Bernard E, Abbosh C, André F, Auwerx J, Balmain A, Bar-Sagi D, Bernards R, Bullman S, DeGregori J, et al. Embracing cancer complexity: hallmarks of systemic disease. Cell. 2024;187(7):1589–1616. doi: 10.1016/j.cell.2024.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Sharma P, Goswami S, Raychaudhuri D, Siddiqui BA, Singh P, Nagarajan A, Liu J, Subudhi SK, Poon C, Gant KL, et al. Immune checkpoint therapy-current perspectives and future directions. Cell. 2023. Apr 13;186(8):1652–1669. doi: 10.1016/j.cell.2023.03.006. [DOI] [PubMed] [Google Scholar]
- 5.Kroemer G, Chan TA, Eggermont AMM, Galluzzi L. Immunosurveillance in clinical cancer management. CA Cancer J Clin. 2024. Mar;74(2):187–202. doi: 10.3322/caac.21818. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Meyer ML, Fitzgerald BG, Paz-Ares L, Cappuzzo F, Jänne PA, Peters S, Hirsch FR. New promises and challenges in the treatment of advanced non-small-cell lung cancer. Lancet. 2024. Aug 24;404(10454):803–822. doi: 10.1016/S0140-6736(24)01029-8. [DOI] [PubMed] [Google Scholar]
- 7.Siegel RL, Giaquinto AN, Jemal A. Cancer statistics, 2024. CA Cancer J Clin. 2024. Jan;74(1):12–49. doi: 10.3322/caac.21820. [DOI] [PubMed] [Google Scholar]
- 8.Rousseau A, Michiels S, Simon-Tillaux N, Lolivier A, Bonastre J, Planchard D, Barlesi F, Remon J, Lavaud P, Aldea M, et al. Impact of pembrolizumab treatment duration on overall survival and prognostic factors in advanced non-small cell lung cancer: a nationwide retrospective cohort study. Lancet Reg Health Eur. 2024. Aug;43:100970. doi: 10.1016/j.lanepe.2024.100970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kroemer G, McQuade JL, Merad M, Andre F, Zitvogel L. Bodywide ecological interventions on cancer. Nat Med. 2023. Jan;29(1):59–74. doi: 10.1038/s41591-022-02193-4. [DOI] [PubMed] [Google Scholar]
- 10.Kroemer G, Montegut L, Kepp O, Zitvogel L. The danger theory of immunity revisited. Nat Rev Immunol. 2024. Dec;24(12):912–928. doi: 10.1038/s41577-024-01102-9. [DOI] [PubMed] [Google Scholar]
- 11.El Tekle G, Garrett WS. Bacteria in cancer initiation, promotion and progression. Nat Rev Cancer. 2023. Sep;23(9):600–618. doi: 10.1038/s41568-023-00594-2. [DOI] [PubMed] [Google Scholar]
- 12.Lopez-Otin C, Pietrocola F, Roiz-Valle D, Galluzzi L, Kroemer G. Meta-hallmarks of aging and cancer. Cell Metab. 2023. Jan 3;35(1):12–35. doi: 10.1016/j.cmet.2022.11.001. [DOI] [PubMed] [Google Scholar]
- 13.Lynch SV, Pedersen O, Phimister EG. The human intestinal microbiome in health and disease. N Engl J Med. 2016. Dec 15;375(24):2369–2379. doi: 10.1056/NEJMra1600266. [DOI] [PubMed] [Google Scholar]
- 14.Derosa L, Iebba V, Silva CAC, Piccinno G, Wu G, Lordello L, Routy B, Zhao N, Thelemaque C, Birebent R, et al. Custom scoring based on ecological topology of gut microbiota associated with cancer immunotherapy outcome. Cell. 2024. June 20;187(13):3373–3389.e16. doi: 10.1016/j.cell.2024.05.029. [DOI] [PubMed] [Google Scholar]
- 15.Derosa L, Routy B, Thomas AM, Iebba V, Zalcman G, Friard S, Mazieres J, Audigier-Valette C, Moro-Sibilot D, Goldwasser F, et al. Intestinal Akkermansia muciniphila predicts clinical response to PD-1 blockade in patients with advanced non-small-cell lung cancer. Nat Med. 2022. Feb;28(2):315–324. doi: 10.1038/s41591-021-01655-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Tonon MC, Vaudry H, Chuquet J, Guillebaud F, Fan J, Masmoudi-Kouki O, Vaudry D, Lanfray D, Morin F, Prevot V, et al. Endozepines and their receptors: structure, functions and pathophysiological significance. Pharmacol Ther. 2020. Apr;208:107386. doi: 10.1016/j.pharmthera.2019.06.008. [DOI] [PubMed] [Google Scholar]
- 17.Montegut L, Abdellatif M, Motino O, Madeo F, Martins I, Quesada V, López-Otín C, Kroemer G. Acyl coenzyme a binding protein (ACBP): an aging- and disease-relevant “autophagy checkpoint”. Aging Cell. 2023. Sep;22(9):e13910. doi: 10.1111/acel.13910. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Joseph A, Chen H, Anagnostopoulos G, Montégut L, Lafarge A, Motiño O, Castedo M, Maiuri MC, Clément K, Terrisse S, et al. Effects of acyl-coenzyme a binding protein (Acbp)/diazepam-binding inhibitor (DBI) on body mass index. Cell Death Dis. 2021. June 9;12(6):599. doi: 10.1038/s41419-021-03864-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Joseph A, Moriceau S, Sica V, Anagnostopoulos G, Pol J, Martins I, Lafarge A, Maiuri MC, Leboyer M, Loftus J, et al. Metabolic and psychiatric effects of acyl coenzyme a binding protein (Acbp)/diazepam binding inhibitor (DBI). Cell Death Dis. 2020. Jul 6;11(7):502. doi: 10.1038/s41419-020-2716-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Montegut L, Joseph A, Chen H, Abdellatif M, Ruckenstuhl C, Motiño O, Lambertucci F, Anagnostopoulos G, Lachkar S, Dichtinger S, et al. High plasma concentrations of acyl-coenzyme a binding protein (ACBP) predispose to cardiovascular disease: evidence for a phylogenetically conserved proaging function of ACBP. Aging Cell. 2023. Jan;22(1):e13751. doi: 10.1111/acel.13751. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Motino O, Lambertucci F, Joseph A, Durand S, Anagnostopoulos G, Li S, Carbonnier V, Nogueira-Recalde U, Montégut L, Chen H. et al. ACBP/DBI neutralization for the experimental treatment of fatty liver disease. Cell Death Differ. 2024. Nov 16;32(3):434–446. doi: 10.1038/s41418-024-01410-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Montegut L, Liu P, Zhao L, Pérez-Lanzón M, Chen H, Mao M, Zhang S, Derosa L, Naour JL, Lambertucci F, et al. Acyl-coenzyme a binding protein (ACBP) - a risk factor for cancer diagnosis and an inhibitor of immunosurveillance. Mol Cancer. 2024. Sep 6;23(1):187. doi: 10.1186/s12943-024-02098-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.bravo-San Pedro JM, Sica V, Martins I, Pol J, Loos F, Maiuri MC, Durand S, Bossut N, Aprahamian F, Anagnostopoulos G. et al. Acyl-CoA-binding protein is a lipogenic factor that triggers food intake and obesity. Cell Metabolism. 2019. Oct 1;30(4):754–767.e9. doi: 10.1016/j.cmet.2019.07.010. [DOI] [PubMed] [Google Scholar]
- 24.Motino O, Lambertucci F, Anagnostopoulos G, Li S, Nah J, Castoldi F, Senovilla L, Montégut L, Chen H, Durand S. et al. ACBP/DBI protein neutralization confers autophagy-dependent organ protection through inhibition of cell loss, inflammation, and fibrosis. Proc Natl Acad Sci USA. 2022. Oct 11;119(41):e2207344119. doi: 10.1073/pnas.2207344119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Montegut L, Martins I, Kroemer G. Neutralization of the autophagy-repressive tissue hormone DBI/ACBP (diazepam binding inhibitor, acyl-CoA binding protein) enhances anticancer immunosurveillance. Autophagy. 2024. Dec;20(12):2836–2838. doi: 10.1080/15548627.2024.2411854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Guidotti A, Forchetti CM, Corda MG, Konkel D, Bennett CD, Costa E. Isolation, characterization, and purification to homogeneity of an endogenous polypeptide with agonistic action on benzodiazepine receptors. Proc Natl Acad Sci USA. 1983. June;80(11):3531–3535. doi: 10.1073/pnas.80.11.3531. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cope DW, Wulff P, Oberto A, Aller MI, Capogna M, Ferraguti F, Halbsguth C, Hoeger H, Jolin HE, Jones A, et al. Abolition of zolpidem sensitivity in mice with a point mutation in the GABAA receptor gamma2 subunit. Neuropharmacology. 2004. Jul;47(1):17–34. doi: 10.1016/j.neuropharm.2004.03.007. [DOI] [PubMed] [Google Scholar]
- 28.Ogris W, Poltl A, Hauer B, Ernst M, Oberto A, Wulff P, Höger H, Wisden W, Sieghart W. Affinity of various benzodiazepine site ligands in mice with a point mutation in the GABA(A) receptor gamma2 subunit. Biochem Pharmacol. 2004. Oct 15;68(8):1621–1629. doi: 10.1016/j.bcp.2004.07.020. [DOI] [PubMed] [Google Scholar]
- 29.Dumitru I, Neitz A, Alfonso J, Monyer H. Diazepam binding inhibitor promotes stem cell expansion controlling environment-dependent neurogenesis. Neuron. 2017. Apr 5;94(1):125–137 e5. doi: 10.1016/j.neuron.2017.03.003. [DOI] [PubMed] [Google Scholar]
- 30.Anagnostopoulos G, Saavedra E, Lambertucci F, Motiño O, Dimitrov J, Roiz-Valle D, Quesada V, Alvarez-Valadez K, Chen H, Sauvat A, et al. Inhibition of acyl-CoA binding protein (ACBP) by means of a GABA(A)Rgamma2-derived peptide. Cell Death Dis. 2024. Apr 6;15(4):249. doi: 10.1038/s41419-024-06633-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Montegut L, Derosa L, Messaoudene M, Chen H, Lambertucci F, Routy B, Zitvogel L, Martins I, Kroemer G. Benzodiazepines compromise the outcome of cancer immunotherapy. Oncoimmunology. 2024;13(1):2413719. doi: 10.1080/2162402X.2024.2413719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Tuppin P, Rudant J, Constantinou P, Gastaldi-Ménager C, Rachas A, de Roquefeuil L, Maura G, Caillol H, Tajahmady A, Coste J, et al. Value of a national administrative database to guide public decisions: from the systeme national d’information interregimes de l’assurance maladie (SNIIRAM) to the systeme national des donnees de sante (SNDS) in France. Rev Epidemiol Sante Publique. 2017. Oct;65(Suppl 4):S149–S167. doi: 10.1016/j.respe.2017.05.004. [DOI] [PubMed] [Google Scholar]
- 33.Bousquet PJ, Lefeuvre D, Tuppin P, BenDiane MK, Rocchi M, Bouée-Benhamiche E, Viguier J, Le bihan-Benjamin C. Cancer care and public health policy evaluations in France: usefulness of the national cancer cohort. PLOS ONE. 2018;13(10):e0206448. doi: 10.1371/journal.pone.0206448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Derosa L, Routy B, Desilets A, Daillère R, Terrisse S, Kroemer G, Zitvogel L. Microbiota-centered interventions: the next breakthrough in immuno-oncology? Cancer Discov. 2021. Oct;11(10):2396–2412. doi: 10.1158/2159-8290.CD-21-0236. [DOI] [PubMed] [Google Scholar]
- 35.Elkrief A, Mendez-Salazar EO, Maillou J, Vanderbilt CM, Gogia P, Desilets A, Messaoudene M, Kelly D, Ladanyi M, Hellmann MD, et al. Antibiotics are associated with worse outcomes in lung cancer patients treated with chemotherapy and immunotherapy. NpJ Precis Oncol. 2024. Jul 16;8(1):143. doi: 10.1038/s41698-024-00630-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Liu W, Huo G, Chen P. Clinical benefit of pembrolizumab in treatment of first line non-small cell lung cancer: a systematic review and meta-analysis of clinical characteristics. BMC Cancer. 2023. May 19;23(1):458. doi: 10.1186/s12885-023-10959-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Routy B, Le Chatelier E, Derosa L, Duong CPM, Alou MT, Daillère R, Fluckiger A, Messaoudene M, Rauber C, Roberti MP, et al. Gut microbiome influences efficacy of PD-1-based immunotherapy against epithelial tumors. Science. 2018. Jan 5;359(6371):91–97. doi: 10.1126/science.aan3706. [DOI] [PubMed] [Google Scholar]
- 38.Leshem Y, Etan T, Dolev Y, Nikolaevski-Berlin A, Miodovnik M, Shamai S, Merimsky O, Wolf I, Havakuk O, Tzuberi M, et al. The prognostic value of beta-1 blockers in patients with non-small-cell lung carcinoma treated with pembrolizumab. Int J Cardiol. 2024. Feb 15;397:131642. doi: 10.1016/j.ijcard.2023.131642. [DOI] [PubMed] [Google Scholar]
- 39.Bessede A, Marabelle A, Guegan JP, Danlos FX, Cousin S, Peyraud F, Chaput N, Spalato M, Roubaud G, Cabart M, et al. Impact of acetaminophen on the efficacy of immunotherapy in cancer patients. Ann Oncol. 2022. Sep;33(9):909–915. doi: 10.1016/j.annonc.2022.05.010. [DOI] [PubMed] [Google Scholar]
- 40.Nelli F, Virtuoso A, Giannarelli D, Fabbri A, Giron Berrios JR, Marrucci E, Fiore C, Ruggeri EM. Effects of acetaminophen exposure on outcomes of patients receiving immune checkpoint inhibitors for advanced non-small-cell lung cancer: a propensity score-matched analysis. Curr Oncol. 2023. Sep 1;30(9):8117–8133. doi: 10.3390/curroncol30090589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Alabaku O, Yang A, Tharmarajah S, Suda K, Vigod S, Tadrous M, Fadare JO. Global trends in antidepressant, atypical antipsychotic, and benzodiazepine use: a cross-sectional analysis of 64 countries. PLOS ONE. 2023;18(4):e0284389. doi: 10.1371/journal.pone.0284389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lefebvre-Durel C, Bailly I, Hunault J, Jovic L, Novic M, Vorspan F, Bellivier F, Drunat O, Kerever S. Benzodiazepine and Z drug cessation in elderly patients: a qualitative study on the perception of healthcare providers and the place of advanced practice nurses. Int J Ment Health Nurs. 2021. June;30(3):653–666. doi: 10.1111/inm.12831. [DOI] [PubMed] [Google Scholar]
- 43.Ma TT, Wang Z, Qin X, Ju C, Lau WCY, Man KKC, Castle D, Chung Chang W, Chan AYL, Cheung ECL, et al. Global trends in the consumption of benzodiazepines and Z-drugs in 67 countries and regions from 2008 to 2018: a sales data analysis. Sleep. 2023. Oct 11;46(10). doi: 10.1093/sleep/zsad124. [DOI] [PubMed] [Google Scholar]
- 44.Votaw VR, Geyer R, Rieselbach MM, McHugh RK. The epidemiology of benzodiazepine misuse: a systematic review. Drug Alcohol Depend. 2019. Jul 1;200:95–114. doi: 10.1016/j.drugalcdep.2019.02.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Vinkers CH, Kupka RW, Penninx BW, Ruhé HG, van Gaalen JM, van Haaren PCF, Schellekens AFA, Jauhar S, Ramos-Quiroga JA, Vieta E, et al. Discontinuation of psychotropic medication: a synthesis of evidence across medication classes. Mol Psychiatry. 2024. Aug;29(8):2575–2586. doi: 10.1038/s41380-024-02445-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Qian L, He X, Liu Y, Gao F, Lu W, Fan Y, Gao Y, Wang W, Zhu F, Wang Y, et al. Longitudinal gut microbiota dysbiosis underlies olanzapine-induced weight gain. Microbiol Spectr. 2023. Aug 17;11(4):e0005823. doi: 10.1128/spectrum.00058-23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Vich Vila A, Collij V, Sanna S, Sinha T, Imhann F, Bourgonje AR, Mujagic Z, Jonkers DMAE, Masclee AAM, Fu J, et al. Impact of commonly used drugs on the composition and metabolic function of the gut microbiota. Nat Commun. 2020. Jan 17;11(1):362. doi: 10.1038/s41467-019-14177-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Lopez-Otin C, Kroemer G. The missing hallmark of health: psychosocial adaptation. Cell Stress. 2024;8:21–50. doi: 10.15698/cst2024.03.294. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Xu S, Chen X, Ying H, Chen J, Ye M, Lin Z, Zhang X, Shen T, Li Z, Zheng Y, et al. Multi-omics identification of a signature based on malignant cell-associated ligand-receptor genes for lung adenocarcinoma. BMC Cancer. 2024. Sep 12;24(1):1138. doi: 10.1186/s12885-024-12911-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Domingues-Junior M, Pinheiro SR, Guerra JL, Palermo-Neto J. Effects of treatment with amphetamine and diazepam on Mycobacterium bovis-induced infection in hamsters. Immunopharmacol Immunotoxicol. 2000. Aug;22(3):555–574. doi: 10.3109/08923970009026012. [DOI] [PubMed] [Google Scholar]
- 51.Huemer HP, Lassnig C, Nowotny N, Irschick EU, Kitchen M, Pavlic M. Diazepam leads to enhanced severity of orthopoxvirus infection and immune suppression. Vaccine. 2010. Aug 3128(38):6152–6158. doi: 10.1016/j.vaccine.2010.07.032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Sanders RD, Godlee A, Fujimori T, Goulding J, Xin G, Salek-Ardakani S, Snelgrove RJ, Ma D, Maze M, Hussell T, et al. Benzodiazepine augmented gamma-amino-butyric acid signaling increases mortality from pneumonia in mice. Crit Care Med. 2013. Jul;41(7):1627–1636. doi: 10.1097/CCM.0b013e31827c0c8d. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Sun GQ, Zhang L, Zhang LN, Wu Z, Hu DF. Benzodiazepines or related drugs and risk of pneumonia: a systematic review and meta-analysis. Int J Geriatric Psychiatry. 2019. Apr;34(4):513–521. doi: 10.1002/gps.5048. [DOI] [PubMed] [Google Scholar]
- 54.Cheng SY, Chen WY, Liu HC, Yang T-W, Pan C-H, Yang S-Y, Kuo C-J. Benzodiazepines and risk of pneumonia in schizophrenia: a nationwide case-control study. Psychopharmacol (Berl). 2018. Nov;235(11):3329–3338. doi: 10.1007/s00213-018-5039-9. [DOI] [PubMed] [Google Scholar]
- 55.Milano VR, Kayhart BM, Morgan RJ, DeSimone DC, Mara KC, Leung JG. Second-generation antipsychotics and pneumonia-related hospitalizations. Prim Care Companion CNS Disord. 2020. Aug 6;22(4). doi: 10.4088/PCC.20m02594. [DOI] [PubMed] [Google Scholar]
- 56.Yang Y, Kong D, Li Q, Chen W, Zhao G, Tan X, Huang X, Zhang Z, Feng C, Xu M, et al. Non-antipsychotic medicines and modified electroconvulsive therapy are risk factors for hospital-acquired pneumonia in schizophrenia patients. Front Psychiatry. 2022;13:1071079. doi: 10.3389/fpsyt.2022.1071079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Chen YH, Ren CY, Liao Y. Analysis of risk factors for hospital-acquired pneumonia in schizophrenia. Front Psychiatry. 2024;15:1414332. doi: 10.3389/fpsyt.2024.1414332. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Galdiero F, Bentivoglio C, Nuzzo I, Ianniello R, Capasso C, Mattera S, Nazzaro C, Galdiero M, Carratelli CR. Effects of benzodiazepines on immunodeficiency and resistance in mice. Life Sci. 1995. Nov 17;57(26):2413–2423. doi: 10.1016/0024-3205(95)02199-0. [DOI] [PubMed] [Google Scholar]
- 59.Oh TK, Park HY, Han JY, Song IA. Prior benzodiazepine use and mortality among adult patients with sepsis: a retrospective population-based cohort study in South Korea. Int J Clin Pract. 2021. Oct;75(10):e14517. doi: 10.1111/ijcp.14517. [DOI] [PubMed] [Google Scholar]
- 60.Wang X, Isomura K, Lichtenstein P, Kuja-Halkola R, D’Onofrio BM, Brikell I, Quinn PD, Zhu N, Jayaram-Lindström N, Chang Z, et al. Incident benzodiazepine and Z-drug use and subsequent risk of serious infections. CNS Drugs. 2024. Oct;38(10):827–838. doi: 10.1007/s40263-024-01108-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kripke DF, Langer RD, Kline LE. Hypnotics’ association with mortality or cancer: a matched cohort study. BMJ Open. 2012;2(1):e000850. doi: 10.1136/bmjopen-2012-000850. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Kao CH, Sun LM, Su KP, Chang S-N, Sung F-C, Muo C-H, Liang J-A. Benzodiazepine use possibly increases cancer risk: a population-based retrospective cohort study in Taiwan. J Clin Psychiatry. 2012. Apr;73(4):e555–60. doi: 10.4088/JCP.11m07333. [DOI] [PubMed] [Google Scholar]
- 63.Pottegard A, Friis S, Andersen M, Hallas J. Use of benzodiazepines or benzodiazepine related drugs and the risk of cancer: a population-based case-control study. Br J Clin Pharmacol. 2013. May;75(5):1356–1364. doi: 10.1111/bcp.12001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Thygesen LC, Pottegard A, Ersboll AK, Friis S, Sturmer T, Hallas J. External adjustment of unmeasured confounders in a case-control study of benzodiazepine use and cancer risk. Br J Clin Pharmacol. 2017. Nov;83(11):2517–2527. doi: 10.1111/bcp.13342. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Kim HB, Myung SK, Park YC, Park B. Use of benzodiazepine and risk of cancer: a meta-analysis of observational studies. Int J Cancer. 2017. Feb 1;140(3):513–525. doi: 10.1002/ijc.30443. [DOI] [PubMed] [Google Scholar]
- 66.Zhang T, Yang X, Zhou J, Liu P, Wang H, Li A, Zhou Y. Benzodiazepine drug use and cancer risk: a dose-response meta analysis of prospective cohort studies. Oncotarget. 2017. Nov 24;8(60):102381–102391. doi: 10.18632/oncotarget.22057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.El Balkhi S, Abbara C. Designer benzodiazepines: effects, toxicity, and interactions. Ther Drug Monit. 2023. Aug 1;45(4):494–507. doi: 10.1097/FTD.0000000000001071. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
Individual patient data cannot be shared due to protection of privacy, but access to the ATHENA cohort can be requested to the National Health Data System on the French Data Hub platform. Metagenomics data from the ONCOBIOTICS will be made available upon reasonable request.


