Abstract
Mediastinal masses represent a heterogeneous group of entities characterized by a variety of histopathological and radiological features. Imaging plays a pivotal role in the detection and interpretation of mediastinal abnormalities. CT remains the modality of choice due to its high spatial and temporal resolution and its ability to assess tissue composition, including the detection of fluid, fat, and calcifications. MRI represents a complementary tool in specific scenarios, such as differentiating complicated cysts from solid lesions or identifying intracellular fat content, as seen in thymic hyperplasia. The differential diagnosis of mediastinal masses relies primarily on the location of the mass and tissue composition, integrated with clinical characteristics of the patient. This review discusses the most common mediastinal masses in adults, providing a practical approach to their differentiation mainly based on the predominant density pattern and location.
Keywords: mediastinal tumours, CT, MRI
Introduction
Mediastinal masses include a variety of entities with a broad spectrum of histopathological and radiological characteristics. Consequently, a multidisciplinary assessment is recommended to integrate clinical data with imaging findings.1 The assessment of anatomical location and tissue characterization of mediastinal lesions plays a pivotal role in enabling the radiologist to provide a differential diagnosis. In this review, we present the most common mediastinal lesions and offer a practical approach for differentiating them based on their predominant density pattern and anatomical location.
Imaging modalities
The conventional imaging approach to mediastinal masses is based on a stepwise assessment from chest radiograph (CXR) to Computed Tomography (CT), then to positron emission tomography (PET)/CT and/or Magnetic Resonance Imaging (MRI), and diagnostic intervention (ie, biopsy) if needed.2,3 The suspicion for mediastinal lesions is often raised on CXR performed for other purposes but it is of limited value for characterizing these lesions.4,5 The American College of Radiology (ACR) Appropriateness Criteria recommend either contrast-enhanced CT or MRI as the imaging modality of choice for patients with an mediastinal mass, as both allow the evaluation of size, location, margins, density or intensity, and enhancement.2 Additionally, multiplanar reconstructions (MPRs) are extremely useful in identifying the origin of mediastinal masses and their spatial relationships with adjacent structures. The main advantages of CT consist of higher spatial and temporal resolution and fast acquisition time, while MRI provides superior tissue characterization and ability to detect invasion across tissue planes.2,6 Thus, except for specific cases (eg, distinguishing complicated cysts from solid lesions or thymic hyperplasia from other thymic masses), the diagnostic performance of CT is comparable to MRI and remains the imaging modality of choice for the initial assessment of a mediastinal lesion.7–9 18-Fluoro-2-deoxy-D-glucose (FDG)-PET/CT offers limited additional value beyond CT, except for primary mediastinal lymphoma staging and treatment response evaluation.2,10 Additionally, 18-F-FDG-PET/CT shows higher standardized uptake values (SUVs) in thymic epithelial tumours (TETs) than in thymic hyperplasia or low-risk thymoma.2,11,12
Diagnostic approach
To accurately characterize mediastinal masses and propose a meaningful differential diagnosis, it is crucial to consider the anatomic location, predominant density pattern, and relevant clinical factors.7 Age is particularly important, as the prevalence and types of mediastinal abnormalities vary significantly across different age groups.7
Localization of mediastinal abnormalities
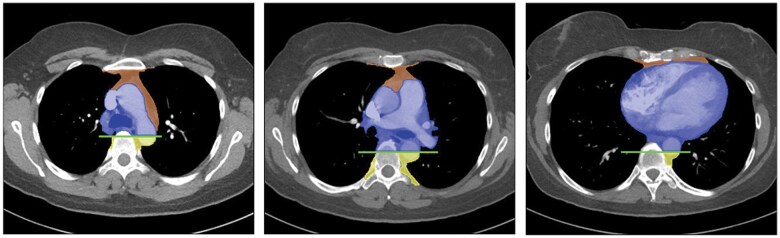
The mediastinum is the central thoracic anatomic compartment situated between the lungs, extending longitudinally from the thoracic inlet to the diaphragm, and containing several vital structures.4 Despite the absence of physical boundaries, it is conventionally divided into compartments to facilitate differential diagnoses and surgical treatment plans.4 Over the years, several classifications have been proposed and Felson’s CXR-based classification has been widely used over time13 but cannot easily applied to cross-sectional imaging. In 2014, the International Thymic Malignancy Interest Group (ITMIG) proposed a classification scheme based on anatomical landmarks easily identified on cross-sectional imaging, particularly on CT.4 By this classification system, the mediastinum is divided into three compartments: the prevascular, visceral, and paravertebral compartment.4,7 (Table 1, Figure 1).
Table 1.
ITMIG CT-based classification of mediastinal compartments (adapted from Carter et al).
| Compartment | Boundaries | Major Content |
|---|---|---|
| Prevascular |
|
|
| Visceral |
|
|
| Paravertebral |
|
|
Figure 1.
International Thymic Malignancy Interest Group (ITMIG) classification of mediastinal compartments (orange: prevascular; blue: visceral; yellow: paravertebral).
Density pattern
The main density patterns in mediastinal lesions include soft-tissue, fat, water, calcium, and high contrast enhancement. On CT fat-containing lesions typically show an average attenuation of −40 to −120 Hounsfield Units (HU), while on MRI they appear as areas of high-intensity on T1- and T2-weighted images, with loss of signal on fat-saturated sequences.6 CT features of typical cystic lesions include: (a) an oval or tubular mass with a well-defined thin wall; (b) homogeneous water attenuation (0-20 HU); (c) no content enhancement; (d) no evidence of adjacent structures infiltration.6,14 When CT findings are indeterminate, further evaluation with MRI may be recommended to demonstrate high-signal intensity of fluid-containing cysts on T2-weighted images.14 Hypervascular mediastinal masses, characterized by intense contrast enhancement, require dynamic imaging for the characterization of specific enhancement patterns.15 Finally, soft-tissue components typically measure 40-60 HU on non-contrast CT with variable contrast enhancement, whereas calcifications are characterized by high attenuation (around 1000 HU) and different forms (eg, punctate, coarse, curvilinear).4
The following sections describe the most common mediastinal masses in adults, grouped according to their compartment of origin and predominant density pattern, as summarized in Table 2.
Table 2.
Classification of the most common mediastinal lesions according to their predominant density pattern.
| Compartment | Fat | Water | Soft-tissue | Intense contrast-enhancement | Calcification |
|---|---|---|---|---|---|
| Prevascular | Mature teratoma | Thymic cyst | Thymic hyperplasia | Hypervascular nodal metastasis | Primary tumour (eg, teratoma, lymphoma) |
| Thymolipoma | Pericardial cyst | Thymoma | Paraganglioma | Metastasis from mucinous tumours | |
| Immature teratoma | Cystic thymoma | Thymic carcinoma | Hemangioma | Thyroid goitre | |
| Teratocarcinoma | Cystic teratoma | Thymic carcinoid | Ectopic parathyroid adenoma | Granulomatous processes or pneumoconiosis | |
| Lipoma | Lymphangioma | Lymphoma | Arteriovenous malformation | ||
| Liposarcoma | Cystic tumour degeneration | Thyroid goitre | |||
| Fat necrosis | Abscess | SMARCA-4 deficient tumours | |||
| Lipomatosis | Castleman disease | ||||
| Congenital hernia (morgagni hernia) | |||||
| Visceral | Bronchogenic cysts | Oesophagus neoplasms | Hypervascular nodal metastasis | Metastasis from mucinous tumours | |
| Oesophageal duplication cysts | Hiatal hernia | Arteriovenous malformation | Granulomatous processes or pneumoconiosis | ||
| Paraganglioma | |||||
| Paravertebral | Lipoma | Intrathoracic meningocele | Neurogenic tumours (eg, schwannoma, neurofibroma) | Hypervascular nodal metastasis | Metastasis from mucinous tumours |
| Liposarcoma | Neuroenteric cyst | Extra-medullary haematopoiesis | Paraganglioma | ||
| Lipomatosis | Mullerian cyst | Infectious spondylodiscitis | Hemangioma | ||
| Congenital hernia (bochdalek hernia) | Cystic schwannoma | Arteriovenous malformation |
Prevascular compartment
Fat-containing lesions
Teratoma
Teratomas are the most common mediastinal germ-cell neoplasms (GCNs). They mostly occur during the second to fourth decades and contain more than one of the primitive germ-cell layers (ectoderm, mesoderm, endoderm).16,17 The vast majority of teratomas are mature teratomas, hence histologically well-differentiated and benign.16 Rarely, they may contain undifferentiated neuroectodermal tissue (immature teratoma), which is considered potentially malignant, or components of carcinoma/sarcoma (teratocarcinoma), regarded as malignant.16
On cross-sectional imaging, teratomas exhibit well-defined, rounded, or lobular margins and a heterogeneous appearance.5,16,17 Fat is present in 75% of lesions, often with characteristic fat-fluid levels, and fat-suppressed MR sequences can be helpful to confirm this content.5,16,17 Rim-like or tooth-like calcifications are commonly observed16 (Figure 2). Some mature teratomas can exhibit a predominantly cystic morphology with internal septations, referred to as cystic teratomas.16 Differentiating between mature and immature teratomas is challenging on imaging as the immature elements can be represented only by small solid-tissue components. A combination of fluid, soft-tissue, calcium, and fat attenuations within a well-defined mass is highly specific for a mature teratoma. In contrast, a predominantly solid, enhancing lesion with necrotic or haemorrhagic areas raises suspicion for teratocarcinoma.16
Figure 2.
Mature teratoma. (A) Contrast-enhanced axial CT image shows a heterogeneous mass in the left prevascular mediastinum, characterized by encapsulated fatty tissue (arrow) intermixed with soft-tissue components (asterisk). (B) Contrast-enhanced axial CT image shows a right prevascular mediastinal mass with well-defined margins, heterogeneous density, containing both fluid and fat (asterisk) with the characteristic fat-fluid level (solid arrow) and rim-like calcifications (yellow arrowhead).
Thymolipoma
Thymolipomas are benign, slow-growing lesions, composed of a mixture of adipose and thymic tissue in variable proportions, with the fat content constituting up to 50%-85% of the lesion.18,19 They typically occur in young adults and are often associated with myasthenia gravis or thyroid disorders.18 On CT, thymolipomas appear as encapsulated fatty lesions with linear whorls of soft-tissue thymic component.5,17 Similarly, on MRI, these lesions demonstrate high-signal intensity on T1- and T2-weighted images due to the fat content, with signal suppression on fat-suppressed sequences, alongside intermediate intensity from the thymic tissue5,17 (Figure 3).
Figure 3.
Thymolipoma. (A) Non-contrast axial CT image shows a fatty mass in the right prevascular compartment intermixed with linear whorls of soft-tissue (arrow), corresponding to adipose and thymic tissue, respectively. (B) Axial T2-weighted MR image of the chest shows the prevascular mass (arrow), characterized by predominant high-signal intensity due to the fat component and intermediate signal intensity from the thymic tissue.
Lipoma and liposarcoma
Lipomatous neoplasms are mesenchymal tumours that can occur in both prevascular and paravertebral compartments.
Lipomas are benign lesions made up of mature adipocytes. They account for approximately 2% of mediastinal neoplasms, with a low risk of malignant degeneration.5 On CT, they appear as encapsulated, homogeneous, fat-attenuated lesions without contrast enhancement.5 Conversely, the presence of soft-tissue components, fibrous bands, or internal septa with contrast enhancement are features consistent with a diagnosis of liposarcoma.5,20
Liposarcomas are rare primary malignant mediastinal tumours (accounting for 0.1%-0.75% of all mediastinal lesions), typically found in individuals in the fifth to sixth decades of life.5,21 Depending on their grade of differentiation, liposarcomas can demonstrate a variable appearance ranging from fatty lesions nearly indistinguishable from lipomas, to heterogeneous enhancing masses.20 On 18F-FDG-PET, liposarcomas usually exhibit higher FDG uptake compared to lipomas.20
Differential diagnoses for lipomatous tumours include mediastinal lipomatosis, mediastinal fat necrosis, and omental hernia. Mediastinal lipomatosis is a benign condition of excessive fat deposition often linked to steroid use, obesity, or Cushing syndrome.5 Mediastinal fat necrosis is an inflammatory self-limiting condition, causing acute chest pain, usually after trauma or surgery.5 On CT, it appears as a juxta-pericardial fat-attenuation area with surrounding soft-tissue stranding and thickening of the adjacent pericardium.22 On 18-F-FDG-PET/CT, fat necrosis could show uptake of FDG, which is expected to resolve on follow-up.6 Finally, omental hernia refers to herniation of omental fat into the mediastinum, either due to congenital (ie, Morgagni and Bochdalek hernias for prevascular and paravertebral compartment, respectively) or acquired defects (trauma or postoperative complications).5,17,20 MPRs help identify diaphragm discontinuity, which allows to differentiate hernias from lipomatous tumours.5,20
Cystic lesions
Thymic cyst
A purely cystic mass with no soft-tissue components or septa in the thymic bed can be reliably classified as a thymic cyst.4 Thymic cysts account for 1%-3% of all mediastinal lesions and can be either congenital or acquired, arising after radiation therapy (RT), thoracotomy, or in relation to autoimmune conditions (eg, Sjogren syndrome).5
Congenital cysts are typically unilocular, thin-walled lesions filled with clear fluid, whereas acquired cysts are more often multilocular and may have variable wall thickness.5 Distinguishing between these types is important as acquired cysts may be associated with malignancies and tend to recur after resection. When haemorrhage or infection occurs, the cysts exhibit higher CT attenuation, potentially leading to a misdiagnosis as a solid mass. In such cases, further characterization with MR imaging should be performed5,16 (Figure 4).
Figure 4.
Thymic cyst. (A) Contrast-enhanced axial CT image of the chest shows a prevascular uniloculated thin-walled lesion (white asterisk) with fluid attenuation, in the typical location of the thymic structure. (B) Axial T2-weighted MR image shows hyperintense signal intensity of the lesion (black asterisk), maintained in fat-suppressed sequence (C) and confirming its water content.
Lymphangioma
Lymphangioma is a rare congenital vascular malformation resulting from an embryologic failure of lymphatic drainage into the venous system.23 It is typically found in the cervical and axillary regions, occasionally extending into the mediastinum.23 Less than 1% of cases are confined to the chest, more often in the prevascular mediastinum.23 Although lymphangiomas are generally diagnosed in childhood, a few adult cases have been reported, possibly due to slow-growing congenital forms or secondary to chronic lymphatic obstruction (eg, following RT, chronic infection, or surgery).23,24 Lymphangiomas are classified based on the size of their dilated lymphatic channels into simple, cavernous, or cystic forms (ie, cystic hygromas) and can be either unilocular or multilocular, with cysts of variable size.5
On CT, lymphangiomas appear as lobulated lesions with fluid attenuation, which envelop rather than displace adjacent structures.5,14 Calcification and contrast enhancement are uncommon.5,14 If CT findings are ambiguous, MRI can be used to confirm the fluid content; however, on T1-weighted images, the lesions can exhibit heterogeneous signal intensity, depending on their content.5,14 A multilocular cystic lesion with septations or soft-tissue components in the prevascular mediastinum should also be differentiated from cystic teratoma and cystic thymoma.4
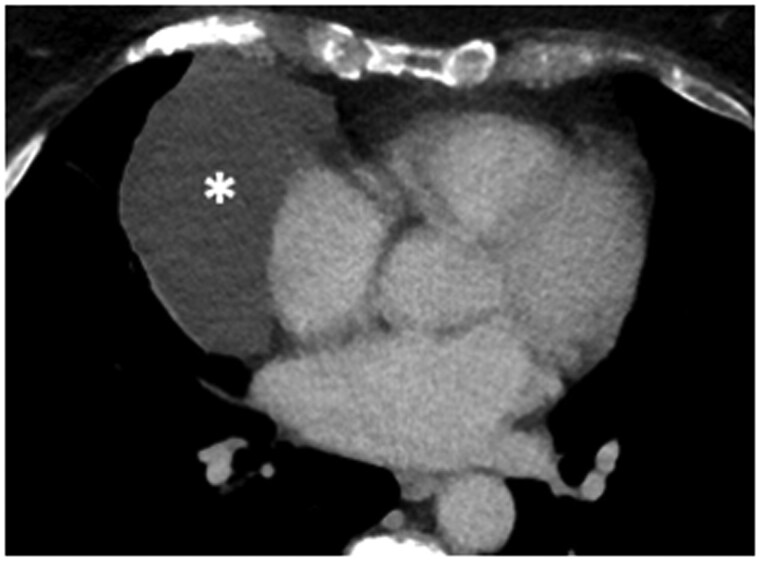
Pericardial cyst
A well-defined unilocular lesion of fluid attenuation with imperceptible walls in the cardiophrenic angle is highly suggestive of a pericardial cyst.6 Pericardial cysts are benign congenital anomalies resulting from abnormal formation of coelomic (somatic) cavities.6,25 They can develop anywhere along the pericardial lining, but most commonly occur in the anterior cardiophrenic angles, particularly on the right side.6,25 Pericardial cysts exhibit the typical imaging features of cystic lesions on both CT and MRI16,26 (Figure 5).
Figure 5.
Pericardial cyst. Contrast-enhanced axial CT image of the chest reveals a lesion (asterisk) in the right cardiophrenic angle, characterized by fluid density and direct contiguity with the pericardial surface.
Soft-tissue lesions
Thymic hyperplasia
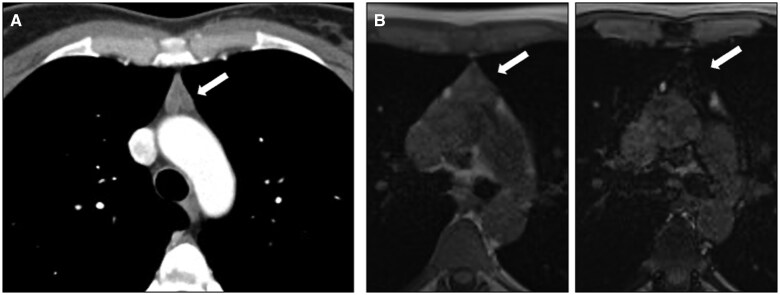
Thymic hyperplasia typically manifests in younger adults as uniform thymic enlargement, while in patients over 40 years, it may appear as a soft-tissue in the thymic bed resembling normal thymus.4 Histologically, it is classified into 2 subtypes: true (rebound) and lymphoid (follicular) hyperplasia. True hyperplasia is characterized by more than a 50% increase in thymic volume with normal histology, often triggered by stressors such as chemotherapy, RT, or corticosteroids.4,5,16 Lymphoid hyperplasia, on the other hand, is distinguished by the presence of lymphoid follicles, with or without glandular enlargement, and is commonly associated with immunologic disorders like myasthenia gravis or collagen vascular disease.4,5,16 On CT, true hyperplasia presents as diffuse symmetric enlargement, whereas lymphoid hyperplasia may appear as a normal thymus, diffuse enlargement, or occasionally as a focal mass.4
In this setting, in-phase and opposed-phase gradient-echo MRI sequences can be used to detect microscopic fat, which is typical for thymic hyperplasia and normal thymus, and is absent in TETs4,7,27 (Figure 6).
Figure 6.
Thymic hyperplasia. (A) Contrast-enhanced axial CT shows a diffuse enlargement of the thymus (arrow). (B) Chemical-shift MR imaging demonstrates in-phase homogeneous intermediate signal intensity of the thymus (left) and signal drop on out-of-phase imaging (right), consistent with thymic hyperplasia.
Thymic epithelial tumours
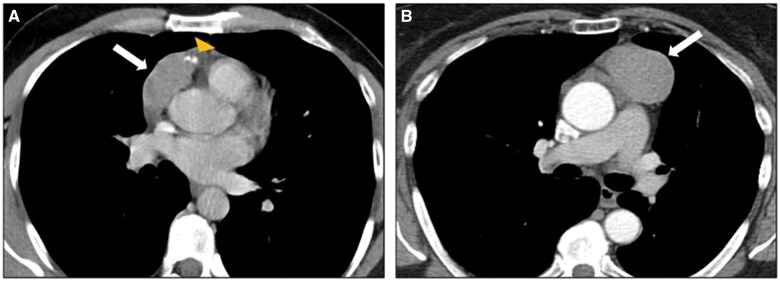
TETs are the most frequent prevascular solid tumours and include thymomas, thymic carcinomas, and thymic carcinoids.28 Thymomas account for approximately 20% of these lesions in adults,29 with a peak of incidence between 55 and 65 years, and are frequently associated with autoimmune disorders, especially myasthenia gravis.30 On CT, thymomas typically present as spherical or ovoid, encapsulated, off-midline soft-tissue lesions with homogeneous attenuation and contrast enhancement (Figure 7). Heterogeneity within the lesion may be due to necrosis, haemorrhage, or cystic change (cystic thymoma).29,31 Calcifications in thymoma exhibit various patterns, including curvilinear (along the capsule or fibrous septa), punctate, or coarse.29 In advanced stages, pleural or pericardial dissemination (drop metastases) may occur.4,5
Figure 7.
Thymoma. (A, B) Contrast-enhanced axial CT images show soft-tissue enhancing lesions (arrow) of the prevascular mediastinum, with punctate peripheral calcifications (A, yellow arrowhead). Biopsy revealed in both cases a thymoma (B2-B3, stage I Masaoka-Koga).
Although MRI may be more effective in assessing local invasion, it is not routinely used due to the lack of distinctive features and superior spatial resolution of CT.29,31 On imaging, a reliable differentiation among TET is not feasible.32 However, a diagnosis of thymic carcinoma or thymic carcinoid should be considered when a lesion shows aggressive features like increased heterogeneity, irregular margins, local invasion, lymphadenopathy, and pleural or pericardial effusion.4,16,29 On 18F-FDG-PET/CT, thymic carcinomas and carcinoids typically demonstrate greater FDG uptake than thymomas.33,34 In the diagnostic evaluation of carcinoids, the nuclear medicine technique targeting somatostatin receptor (SSTR) expression has been recently recognized as a valuable tool.28 Thymic malignancies are staged using CT according to the ninth edition TNM staging system.35 However, the Masaoka-Koga staging system, which strongly correlates prognosis with clinical stage, remains widely used for thymomas.36,37
Lymphoma
Lymphomas account for up to 20% of mediastinal masses.16 Primary mediastinal lymphomas are rare, comprising 1% of all lymphomas16 with Hodgkin lymphoma and primary mediastinal large B-cell lymphoma as the main histologic subtypes.5,16 On contrast-enhanced CT, they typically appear as lobulated soft-tissue masses with mild-to-moderate enhancement, although cystic changes and necrosis may be present.5 Calcifications are uncommon prior to treatment.30 Differentiating lymphoma from other prevascular masses can be challenging; however, unlike other malignancies, lymphomas tend to encase adjacent structures without direct infiltration.4,32 Young age of patients—second to third decades—also supports the diagnosis.4,5 18-F-FDG-PET/CT remains the preferred imaging modality for lymphoma staging and monitoring disease activity.38–40 However, whole-body (WB)-MRI has emerged as viable, radiation-free alternative particularly for younger patients.41,42 Differential diagnosis, particularly in young patients (third to fourth decades), includes Castleman disease, a non-clonal lymphoproliferative disorder. Its most common form is the unicentric hyaline vascular type, which typically presents as a non-invasive mass (in 50% of cases), but may also appear as an infiltrative mass with lymphadenopathy or as matted lymph nodes without a discrete mass.43
Another rare differential diagnosis is SMARCA4-deficient thoracic sarcomas (SMARCA4-DTS), a recently described undifferentiated sarcoma that most commonly occur in the mediastinum of young-middle aged male smokers.44,45 These tumours present as ill-defined masses with heterogeneous enhancement, frequently crossing anatomical compartments and demonstrating strong avidity on 18-F-FDG-PET/CT.45,46 At diagnosis, confluent, necrotic, ill-defined lymphadenopathies with surrounding infiltrate are observed, highlighting the lymphatic spread of these tumours, which is uncommon in other types of sarcomas, and further complicating differentiation from lymphomas.46,47 Thus, diagnosis requires histopathological and immunohistochemical confirmation.
Thyroid goiter
An encapsulated, lobulated, heterogeneous mass that is intrinsically hyperdense (70-85 HU) and continuous with the cervical thyroid gland can be reliably diagnosed as mediastinal goitre.4 Intrathoracic goitre accounts for 3%-6% of mediastinal masses, occurring more commonly in adults. 1%-2% can be ectopic without any connection to the cervical thyroid.4,5,16 On contrast-enhanced CT, these lesions typically exhibit homogeneous, sustained enhancement, although cystic changes and calcifications are common.5,16 Thyroid goitres can extend into the visceral compartment and cause tracheal displacement or compression.4 Loss of fascial planes or the presence of adjacent lymphadenopathies should raise suspicion for thyroid malignancy.7
Visceral compartment
Cystic lesions
Foregut duplication cysts
Foregut duplication cysts are congenital anomalies arising from abnormal budding of the embryonic foregut and tracheobronchial tree. These lesions include bronchogenic and oesophageal duplication cysts.48,49 Bronchogenic cysts account for 50%-60% of mediastinal cystic lesions and are most commonly located in the visceral compartment (less frequently in the paravertebral mediastinum) near the tracheal carina or, less commonly, in the paratracheal or hilar region, often right-sided.49,50 Although most cases are detected during the neonatal period, incidental findings in adults are also possible.51 These cysts exhibit variable attenuation on CT—due to their mixed content of fluid and proteinaceous mucoid material—and heterogeneous signal intensity on T1-weighted MR images49,50 (Figure 8). Air within the cyst is uncommon and suggests infection.14,49,50
Figure 8.
Bronchogenic cyst. (A) Non-contrast CT axial image shows a homogeneous lesion (asterisk) in the right visceral mediastinum under the level of the carina, with attenuation values slightly higher than fluid. (B, C) Axial T1-weighted and coronal T2-weighted MR images show high-signal intensity, consistent with a cystic lesion with a heterogeneous content.
Oesophageal duplication cysts are more commonly detected in children, within or adjacent to the oesophageal wall. A 99mTechnetium-pertechnetate scan can help identify oesophageal cysts containing ectopic gastric mucosa (present in up to one-third of cases), which are at higher risk for complications such as perforation, infection, or haemorrhage. CT and MRI findings closely resemble those of bronchogenic cysts, though oesophageal cysts typically have thicker walls and are generally closer to the oesophagus.6,14,52
Soft-tissue lesions
Oesophagus neoplasms
Up to 80% of oesophagus neoplasms are malignant, primarily squamous cell carcinoma and adenocarcinoma, both of which are more common in the seventh decade of life.53,54 These tumour types are indistinguishable on imaging, but most adenocarcinomas involve the distal oesophagus and the stomach. Although CT is not the primary imaging modality for the oesophagus, it can demonstrate focal asymmetric thickening (>5 mm) or a soft-tissue mass in the oesophageal wall, which may progress to circumferential involvement and invasion of adjacent structures.53,54 In contrast, mesenchymal neoplasms, such as leiomyomas and gastrointestinal stromal tumours (GISTs), typically exhibit well-defined smooth margins without peritumoral invasion or lymphadenopathies; however, differentiating between them is crucial due to the malignant potential of GISTs.53,54 Differential diagnoses for oesophageal wall thickening also include inflammatory conditions, such as gastroesophageal reflux disease, associated with hiatal hernia.55
Paravertebral compartment
Cystic lesions
Intrathoracic meningocele
Intrathoracic meningocele is an anomalous saccular protrusion of the leptomeninges through an intervertebral foramen or bone defects containing cerebrospinal fluid.14,20 More common in adults, it can be associated with neurofibromatosis type 1.20 Meningoceles typically occur in the thoracic spine, especially between T3 and T7, and are usually right-sided, probably due to the aorta’s position on the left. CT scans reveal sharply defined, homogeneous, low-attenuation lesions, with adjacent vertebral abnormalities or enlarged foramina.4,14,20 However, MRI is the gold standard for differentiating meningocele from other paravertebral lesions (eg, neurogenic tumours), as it confirms their cystic nature and connection to the thecal sac.4,14,20
Extramedullary cystic schwannomas should be considered in the differential diagnosis, as they may present cystic components or cystic degeneration, appearing as areas of high-signal intensity on T2-weighted sequences with peripheral enhancement on T1-weighted images.14,56 Differential diagnoses should also include neuroenteric and Müllerian cysts, both of which display similar imaging features to intrathoracic meningocele. Neurenteric cysts are a rare type of foregut cysts resulting from abnormal portioning of the embryonic notochordal plate.57 They are typically intradural and extramedullary, located ventral to the spinal cord, most commonly in the cervical spine.57 These cysts are frequently diagnosed during childhood or in the second to third decades, due to respiratory symptoms or associated radiculopathy.20,57 Lastly, Müllerian cysts—a recently described benign entity33 occurring only in females—can mimic a cystic neural lesion and require pathologic specimens and immunohistochemical staining for diagnosis.1,58
Soft-tissue lesions
Neurogenic tumours
Neurogenic tumours are the most common paraspinal masses. The malignancy rate is higher in infants and young children, while in young adults (aged <40 years), nerve sheath tumours are the most common subtype, including benign schwannomas, neurofibromas, and malignant peripheral nerve sheath tumours (MPNSTs).20,59–61 These tumours typically originate from spinal nerve roots or intercostal nerve59 and often assume a characteristic “dumbbell” shape when extending through intervertebral foramina.20 Benign lesions usually appear as well-defined, fusiform, or spherical soft-tissue lesions spanning several rib interspaces, while larger lesions may become eccentric to the nerve, lobulated, and more heterogeneous.20,59
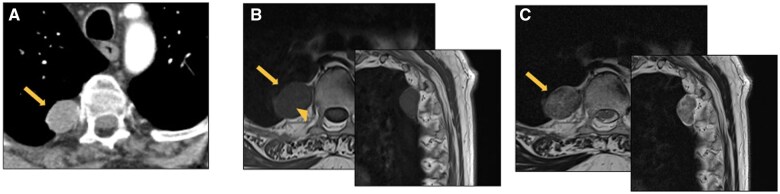
CT may reveal the “split fat sign” indicating a non-infiltrating mass by showing displaced but intact fat surrounding the neuromuscular bundle. Peripheral calcifications can be seen in about 10% of schwannomas, while adjacent osseous erosion is more common in neurofibromas.59 Both schwannomas and neurofibromas show low- to intermediate signal intensity on T1-weighted images. On T2-weighted sequences, neurofibromas may display the “target sign”, characterized by a central low-signal zone surrounded by higher intensity.20,59 Schwannomas may show hypointense small foci with a ring-like shape in a more hyperintense mass (“fascicular sign”).20,26,59 MRI is also valuable for assessing intraspinal extension and neural invasion20,59 (Figure 9).
Figure 9.
Schwannoma. (A) Contrast-enhanced CT axial image (arterial phase) shows a well-defined rounded soft-tissue lesion (arrow) in the right paravertebral mediastinum. This mass abuts the adjacent spinal nerve (arrowhead), appears hypointense on T1-weighted MR images (B) and moderately hyperintense with central hypointense foci on T2-weighted images (C), representing thickened fascicles (fascicular sign).
Rapid growth, necrosis, and local invasion are suggestive of malignancy20,59 and 18F-FDG/PET helps in distinguishing malignant tumours from benign neurofibromas.62 Additionally, Wasa et al reported high suspicion of MPNSTs in nerve sheath tumours displaying 2 or more of the following MRI features: size >5 cm, peripheral enhancement, perilesional oedema, or intratumoral cystic change.63
Infectious diseases
Infectious spondylodiscitis usually originates from pyogenic or tuberculous (Pott’s disease) infection via haematogenous spread. It can extend into pre- and paravertebral soft-tissues, affecting single or multiple spinal segments.20,64 Osteolytic destruction may cause anterior vertebral collapse, resulting in the characteristic gibbus deformity in Pott’s disease, while paravertebral abscess forms a “horseshoe” mass around the affected vertebra.20,64,65 CT and MRI are highly sensitive for detecting early osteolytic changes, with contrast media enhancing paraspinal abscess and disc involvement. On MRI, spondylodiscitis typically shows hypo- to isointense T1-signals and hyperintense T2-signals in the subchondral end plates and intervertebral discs.20,64,65
Extramedullary haematopoiesis
Extramedullary haematopoiesis occurs in response to insufficient bone marrow function (eg, myelofibrosis, leukaemia, haemolytic anemias) with the expansion of haematopoietic tissue outside the bone marrow and formation of paravertebral soft-tissue masses, usually in the lower thoracic spine.66 These masses are typically bilateral, well-defined, with mild contrast enhancement.20,67 The appearance on CT and MRI varies with the lesion’s haematologic activity. Active lesions show soft-tissue density on CT and intermediate T1- and T2-signals on MRI, while inactive lesions, containing fat and/or iron deposits, exhibit variable CT attenuation and MR signal intensity based on the proportion of these components68 (Figure 10).
Figure 10.
Extramedullary haematopoiesis. Non-contrast axial CT image shows bilateral spinal soft-tissue (arrows) in a patient with beta-thalassemia.
Any compartments
Cystic-like lesions
Cystic degeneration can occur in a variety of tumours, including thymomas, Hodgkin lymphoma, GCNs, metastatic lymph nodes, and neurogenic tumours.14 It is more likely after RT or chemotherapy but may also present before treatment. Extensive degeneration results in mixed solid and cystic components on CT and MRI and can make it difficult to differentiate between tumour types and other mediastinal cysts.14
Mediastinal abscesses should also be considered in patients with low-attenuation masses on CT following recent surgery, oesophageal perforation, or infections (eg, retropharyngeal abscess, osteomyelitis, empyema).5,14 Air bubbles and a thick rim-enhancing wall may be present.14
(Intense) Enhancing lesions
Hypervascular nodal metastases
Hypervascular nodal metastases can originate from intra- or extrathoracic tumours, such as renal cell carcinoma, melanoma, neuroendocrine tumours, and thyroid carcinoma (with the first two being the most prevalent).15 PET/CT is often recommended for identifying the primary tumour and staging the disease.15
Mediastinal hemangioma
Mediastinal haemangioma is a rare benign vascular tumour, accounting for less than 0.5% of mediastinal masses.15 It can affect patients of all ages, with a higher incidence in younger individuals (mean age of 35 years).69 Histologically, haemangiomas consist of vascular spaces interspersed with various stromal tissues (eg, fat, myxoid or fibrous tissue), leading to a heterogeneous appearance on both CT and MRI. Dynamic imaging reveals gradually increasing enhancement with a centripetal fill-in pattern and persistent delayed enhancement.6,15,69 Thrombus formation within the vessels is frequent and may calcify as phlebolith, which is a helpful diagnostic feature on CT,6,70 along with the presence of aberrant draining veins.69
Paragangliomas
Paragangliomas are rare neuroendocrine tumours arising from extra-adrenal chromaffin cells in the parasympathetic or sympathetic ganglia. Mediastinal paraganglia are often located in the prevascular compartment along the great vessels (especially in the aortic-pulmonary window) or in the paravertebral compartment along the sympathetic chain.20 Aortopulmonary paragangliomas often occur in patients older than 40 years, while aortosympathetic ones typically affect younger individuals (second to third decades).15
On dynamic imaging, paragangliomas show early intense enhancement after contrast administration (Figure 11) but they may also present calcifications or areas of cystic degeneration. MRI reveals a “salt and pepper” pattern, with hyperintense regions (salt) representing haemorrhage (on T1- and T2-weighted images) or slow flow within vessels (on T2-weighted images), while the focal signal voids (pepper) correlate with the high-velocity flow in other vessels.6,15 PET/CT with 68-Ga DOTATATE is increasingly used for the diagnosis, staging, and follow-up.71
Figure 11.
Paraganglioma. (A) Plain (left) and contrast-enhanced axial CT (right) shows a well-defined hypodense lesion (arrow) in the visceral mediastinum (aortopulmonary window) with intense contrast enhancement. (B) Multiple paragangliomas have been identified in other sites of the same patient: at the division of the right common carotid artery (left image) and retrocaval (right image).
Ectopic parathyroid adenoma
Parathyroid adenomas are benign, functioning tumours typically found in the neck, but they can be ectopically located in approximately 10% of cases, most often near or within the thymus.16 They can occur at any age, but more commonly between fifth and seventh decades.72 On both CT and MRI, parathyroid adenomas appear as rounded lesions with smooth margins, early intense enhancement in the arterial phase, and a rapid washout in the delayed phase, allowing for differentiation from lymph nodes and thyroid tissue.6 A suspicion can be confirmed by single photon emission CT (SPECT) using 99mTc-sestamibi.15
Arteriovenous malformation
Arteriovenous malformations (AVMs) are high-flow vascular anomalies. On dynamic CT and MRI, they present dilated feeding arteries and draining veins, with or without a central nidus, and no significant soft-tissue components.15,73 After contrast administration, early vein opacification is observed, consistent with shunting.15,73
Calcified lesions
Calcifications in mediastinal masses can be observed across all compartments and may be associated with both malignant and benign conditions.7
Primary tumours, such as teratomas, often exhibit rim-like or tooth-like calcifications.5,74 Metastatic mediastinal lymph nodes from mucinous tumours (eg, ovarian, gastrointestinal cancers) and bone-forming sarcoma may present with calcific deposits8 as well as treated malignancies, in particular lymphomas.5 Benign conditions can also lead to calcifications, as seen in up to 75% of thyroid goiters5 (Figure 12).
Figure 12.
Thyroid goitre. (A) Non-contrast axial CT shows a large, lobulated mass in the visceral mediastinum with cystic changes and peripheral calcifications (arrowhead). (B) Coronal contrast-enhanced CT reconstruction shows the continuity with the right lobe of the cervical thyroid and the compression of the tracheal lumen (arrow).
Finally, calcified lymph nodes may indicate granulomatous processes (eg, sarcoidosis, tuberculosis) or pneumoconiosis (eg, silicosis). Calcification patterns can vary, including complete nodal calcifications in tuberculosis and “egg-shell” or “icing sugar” patterns in sarcoidosis and silicosis.5,75,76
Conclusion
A wide spectrum of conditions can occur in the mediastinum, making a standardized approach challenging. In this review, we highlight the importance of imaging findings—particularly tissue characteristics and localization within mediastinal compartments—as valuable tools for radiologists in formulating meaningful differential diagnoses.
Contributor Information
Rebecca Mura, Department of Medicine and Surgery (diMeC), University of Parma, Parma 43125, Italy.
Svitlana Pochepnia, Christian Doppler Laboratory for Machine Learning Driven Precision Imaging, Department of Biomedical Imaging and Image-guided Therapy, Medical University of Vienna, Vienna 1090, Austria.
Daria Kifjak, Christian Doppler Laboratory for Machine Learning Driven Precision Imaging, Department of Biomedical Imaging and Image-guided Therapy, Medical University of Vienna, Vienna 1090, Austria.
Natallia Khenkina, Postgraduation School in Radiodiagnostics, University of Milan, Milan 20122, Italy.
Helmut Prosch, Christian Doppler Laboratory for Machine Learning Driven Precision Imaging, Department of Biomedical Imaging and Image-guided Therapy, Medical University of Vienna, Vienna 1090, Austria.
Funding
None declared.
Conflicts of interest
None declared.
References
- 1. Ghigna MR, Thomas De Montpreville V. Mediastinal tumours and pseudo-tumours: a comprehensive review with emphasis on multidisciplinary approach. Eur Respir Rev. 2021;30:200309. 10.1183/16000617.0309-2020 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Ackman JB, Chung JH, Walker CM, et al. Expert Panel on Thoracic Imaging. ACR appropriateness criteria® imaging of mediastinal masses. J Am Coll Radiol. 2021;18:S37-S51. 10.1016/j.jacr.2021.01.007 [DOI] [PubMed] [Google Scholar]
- 3. Whitten CR, Khan S, Munneke GJ, Grubnic S. A diagnostic approach to mediastinal abnormalities. RadioGraphics. 2007;27:657-671. 10.1148/rg.273065136 [DOI] [PubMed] [Google Scholar]
- 4. Carter BW, Benveniste MF, Madan R, et al. ITMIG classification of mediastinal compartments and multidisciplinary approach to mediastinal masses. RadioGraphics. 2017;37:413-436. 10.1148/rg.2017160095 [DOI] [PubMed] [Google Scholar]
- 5. Shahrzad M, Le TSM, Silva M, Bankier AA, Eisenberg RL. Anterior mediastinal masses. Am J Roentgenol. 2014;203:W128-W138. 10.2214/AJR.13.11998 [DOI] [PubMed] [Google Scholar]
- 6. Archer JM, Ahuja J, Strange CD, et al. Multimodality imaging of mediastinal masses and mimics. Mediastinum. 2023;7:27-27. 10.21037/med-22-53 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Carter BW, Okumura M, Detterbeck FC, Marom EM. Approaching the patient with an anterior mediastinal mass: a guide for radiologists. J Thorac Oncol. 2014;9:S110-S118. 10.1097/JTO.0000000000000295 [DOI] [PubMed] [Google Scholar]
- 8. Erasmus JJ, McAdams HP, Donnelly LF, Spritzer CE. MR imaging of mediastinal masses. Magn Reson Imaging Clin North Am. 2000;8:59-89. 10.1016/S1064-9689(21)00042-8 [DOI] [PubMed] [Google Scholar]
- 9. Tomiyama N, Honda O, Tsubamoto M, et al. Anterior mediastinal tumors: diagnostic accuracy of CT and MRI. Eur J Radiol. 2009;69:280-288. 10.1016/j.ejrad.2007.10.002 [DOI] [PubMed] [Google Scholar]
- 10. Proli C, De Sousa P, Jordan S, et al. UK Thoracic Surgery Research Collaborative. A diagnostic cohort study on the accuracy of 18-fluorodeoxyglucose (18FDG) positron emission tomography (PET)-CT for evaluation of malignancy in anterior mediastinal lesions: the DECiMaL study. BMJ Open. 2018;8:e019471. 10.1136/bmjopen-2017-019471 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Luzzi L, Campione A, Gorla A, et al. Role of fluorine-fluorodeoxyglucose positron emission tomography/computed tomography in preoperative assessment of anterior mediastinal masses. Eur J Cardio-Thorac Surg. 2009;36:475-479. 10.1016/j.ejcts.2009.03.055 [DOI] [PubMed] [Google Scholar]
- 12. Kitami A, Sano F, Ohashi S, et al. The usefulness of positron-emission tomography findings in the management of anterior mediastinal tumors. Ann Thorac Cardiovasc Surg. 2017;23:26-30. 10.5761/atcs.oa.16-00205 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Reeder MM. Chest. In: Reeder and Felson’s Gamuts in Radiology. Springer New York; 1993:357-436. 10.1007/978-1-4757-2269-7_6 [DOI] [Google Scholar]
- 14. Jeung MY, Gasser B, Gangi A, et al. Imaging of cystic masses of the mediastinum. RadioGraphics. 2002;22 Spec No:S79-S93. 10.1148/radiographics.22.suppl_1.g02oc09s79 [DOI] [PubMed] [Google Scholar]
- 15. Cabral FC, Trotman-Dickenson B, Madan R. Hypervascular mediastinal masses: action points for radiologists. Eur J Radiol. 2015;84:489-498. 10.1016/j.ejrad.2014.11.039 [DOI] [PubMed] [Google Scholar]
- 16. Garrana SH, Rosado-de-Christenson ML. Imaging of the anterior/prevascular mediastinum. Radiol Clin North Am. 2021;59:155-168. 10.1016/j.rcl.2020.10.003 [DOI] [PubMed] [Google Scholar]
- 17. Gaerte SC, Meyer CA, Winer-Muram HT, Tarver RD, Conces DJ. Fat-containing lesions of the chest. RadioGraphics. 2002;22 Spec No:S61-S78. 10.1148/radiographics.22.suppl_1.g02oc08s61 [DOI] [PubMed] [Google Scholar]
- 18. Brcic L, Roden AC. Histopathological features of giant mediastinal tumors—a literature review. Mediastinum. 2023;7:37-37. 10.21037/med-23-23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19. Rosado-de-Christenson ML, Pugatch RD, Moran CA, Galobardes J. Thymolipoma: analysis of 27 cases. Radiology. 1994;193:121-126. 10.1148/radiology.193.1.8090879 [DOI] [PubMed] [Google Scholar]
- 20. Occhipinti M, Heidinger BH, Franquet E, Eisenberg RL, Bankier AA. Imaging the posterior mediastinum: a multimodality approach. Diagn Interv Radiol. 2015;21:293-306. 10.5152/dir.2014.14467 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Wong GS, Bass D, Chen IY, Thomas R, Velez MJ, Hobbs SK. Imaging and clinical findings in a series of six cases of rare primary mediastinal liposarcoma. Radiol: Cardiothorac Imaging. 2022;4:e210259. 10.1148/ryct.210259 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22. Bhatt MY, Martínez-Jiménez S, Rosado-de-Christenson ML, Watson KR, Walker CM, Kunin JR. Imaging manifestations of mediastinal fat necrosis. Case Rep Radiol. 2013;2013:323579-323575. 10.1155/2013/323579 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23. Faul JL, Berry GJ, Colby TV, et al. Thoracic lymphangiomas, lymphangiectasis, lymphangiomatosis, and lymphatic dysplasia syndrome. Am J Respir Crit Care Med. 2000;161:1037-1046. 10.1164/ajrccm.161.3.9904056 [DOI] [PubMed] [Google Scholar]
- 24. Miyake H, Shiga M, Takaki H, Hata H, Onishi R, Mori H. Mediastinal lymphangiomas in adults: CT findings. J Thorac Imaging. 1996;11:83-85. 10.1097/00005382-199601110-00009 [DOI] [PubMed] [Google Scholar]
- 25. Feigin DS, Fenoglio JJ, McAllister HA, Madewell JE. Pericardial cysts: a radiologic-pathologic correlation and review. Radiology. 1977;125:15-20. 10.1148/125.1.15 [DOI] [PubMed] [Google Scholar]
- 26. Park JW, Jeong WG, Lee JE, et al. Pictorial review of mediastinal masses with an emphasis on magnetic resonance imaging. Korean J Radiol. 2021;22:139-154. 10.3348/kjr.2019.0897 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Inaoka T, Takahashi K, Mineta M, et al. Thymic hyperplasia and thymus gland tumors: differentiation with chemical shift MR imaging. Radiology. 2007;243:869-876. 10.1148/radiol.2433060797 [DOI] [PubMed] [Google Scholar]
- 28. Koyasu S. Imaging of thymic epithelial tumors—a clinical practice review. Mediastinum. 2024;8:41-41. 10.21037/med-23-66 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Rosado-de-Christenson ML, Strollo DC, Marom EM. Imaging of thymic epithelial neoplasms. Hematol/Oncol Clin North Am. 2008;22:409-431. 10.1016/j.hoc.2008.03.011 [DOI] [PubMed] [Google Scholar]
- 30. Nakazono T, Yamaguchi K, Egashira R, et al. MRI findings and differential diagnosis of anterior mediastinal solid tumors. Magn Reson Med Sci. 2023;22:415-433. 10.2463/mrms.rev.2021-0098 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31. Maher MM, Shepard JAO. Imaging of thymoma. Semin Thorac Cardiovasc Surg. 2005;17:12-19. 10.1053/j.semtcvs.2004.12.003 [DOI] [PubMed] [Google Scholar]
- 32. Prosch H, Röhrich S, Tekin ZN, Ebner L. The role of radiological imaging for masses in the prevascular mediastinum in clinical practice. J Thorac Dis. 2020;12:7591-7597. 10.21037/jtd-20-964 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33. Endo M, Nakagawa K, Ohde Y, et al. Utility of 18FDG-PET for differentiating the grade of malignancy in thymic epithelial tumors. Lung Cancer. 2008;61:350-355. 10.1016/j.lungcan.2008.01.003 [DOI] [PubMed] [Google Scholar]
- 34. Inoue A, Tomiyama N, Tatsumi M, et al. 18F-FDG PET for the evaluation of thymic epithelial tumors: correlation with the World Health Organization classification in addition to dual-time-point imaging. Eur J Nucl Med Mol Imaging. 2009;36:1219-1225. 10.1007/s00259-009-1082-4 [DOI] [PubMed] [Google Scholar]
- 35. Klug M, Kirshenboim Z, Truong MT, et al. RadioGraphics update: the ninth edition TNM staging system for thymic epithelial tumors. RadioGraphics. 2024;44:e240196. 10.1148/rg.240196 [DOI] [PubMed] [Google Scholar]
- 36. Masaoka A, Monden Y, Nakahara K, Tanioka T. Follow-up study of thymomas with special reference to their clinical stages. Cancer. 1981;48:2485-2492. 10.1002/1097-0142(19811201)48:11<2485::AID-CNCR2820481123>3.0.CO; 2-R [DOI] [PubMed] [Google Scholar]
- 37. Markowiak T, Hofmann HS, Ried M. Classification and staging of thymoma. J Thorac Dis. 2020;12:7607-7612. 10.21037/jtd-2019-thym-01 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Cheson BD. Staging and response assessment in lymphomas: the new Lugano classification. Chin Clin Oncol. 2015;4:5. 10.3978/j.issn.2304-3865.2014.11.03 [DOI] [PubMed] [Google Scholar]
- 39. Meignan M. Quantitative FDG-PET: a new biomarker in PMBCL. Blood. 2015;126:924-926. 10.1182/blood-2015-07-653386 [DOI] [PubMed] [Google Scholar]
- 40. Nagle SJ, Chong EA, Chekol S, et al. The role of FDG‐PET imaging as a prognostic marker of outcome in primary mediastinal B‐cell lymphoma. Cancer Med. 2015;4:7-15. 10.1002/cam4.322 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41. Spijkers S, Littooij AS, Beishuizen A, Lam MGEH, Nievelstein RAJ. A meta-analysis on the diagnostic performance of whole-body MRI for the initial staging of Hodgkin lymphoma in children and adults using FDG-PET/CT as a reference standard. EJC Paediatr Oncol. 2023;1:100016. 10.1016/j.ejcped.2023.100016 [DOI] [Google Scholar]
- 42. Maccioni F, Alfieri G, Assanto GM, et al. Whole body MRI with diffusion weighted imaging versus 18F-fluorodeoxyglucose-PET/CT in the staging of lymphomas. Radiol Med. 2023;128:556-564. 10.1007/s11547-023-01622-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Bonekamp D, Horton KM, Hruban RH, Fishman EK. Castleman disease: the great mimic. RadioGraphics. 2011;31:1793-1807. 10.1148/rg.316115502 [DOI] [PubMed] [Google Scholar]
- 44. Perret R, Chalabreysse L, Watson S, et al. SMARCA4-deficient thoracic sarcomas: clinicopathologic study of 30 cases with an emphasis on their nosology and differential diagnoses. Am J Surg Pathol. 2019;43:455-465. 10.1097/PAS.0000000000001188 [DOI] [PubMed] [Google Scholar]
- 45. Roden AC. Thoracic SMARCA4-deficient undifferentiated tumor—a case of an aggressive neoplasm—case report. Mediastinum. 2021;5:39-39. 10.21037/med-20-15 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Crombé A, Alberti N, Villard N, et al. Imaging features of SMARCA4-deficient thoracic sarcomas: a multi-centric study of 21 patients. Eur Radiol. 2019;29:4730-4741. 10.1007/s00330-019-06017-x [DOI] [PubMed] [Google Scholar]
- 47. Le Loarer F, Watson S, Pierron G, et al. SMARCA4 inactivation defines a group of undifferentiated thoracic malignancies transcriptionally related to BAF-deficient sarcomas. Nat Genet. 2015;47:1200-1205. 10.1038/ng.3399 [DOI] [PubMed] [Google Scholar]
- 48. Walker CM, Chung Jonathan H. Muller’s Imaging of the Chest. 2nd ed. Elsevier; 2019. [Google Scholar]
- 49. Berrocal T, Madrid C, Novo S, Gutiérrez J, Arjonilla A, Gómez-León N. Congenital anomalies of the tracheobronchial tree, lung, and mediastinum: embryology, radiology, and pathology. RadioGraphics. 2004;24:e17. 10.1148/rg.e17 [DOI] [PubMed] [Google Scholar]
- 50. McAdams HP, Kirejczyk WM, Rosado-de-Christenson ML, Matsumoto S. Bronchogenic cyst: imaging features with clinical and histopathologic correlation. Radiology. 2000;217:441-446. 10.1148/radiology.217.2.r00nv19441 [DOI] [PubMed] [Google Scholar]
- 51. Gross DJ, Briski LM, Wherley EM, Nguyen DM. Bronchogenic cysts: a narrative review. Mediastinum. 2023;7:26-26. 10.21037/med-22-46 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Wahi JE, Safdie FM. Esophageal duplication cysts: a clinical practice review. Mediastinum. 2023;7:1-1. 10.21037/med-22-33 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Lewis RB, Mehrotra AK, Rodriguez P, Levine MS. From the radiologic pathology archives: esophageal neoplasms: radiologic-pathologic correlation. RadioGraphics. 2013;33:1083-1108. 10.1148/rg.334135027 [DOI] [PubMed] [Google Scholar]
- 54. Lichtenberger JP, Zeman MN, Dulberger AR, Alqutub S, Carter BW, Manning MA. Esophageal neoplasms. Radiol Clin North Am. 2021;59:205-217. 10.1016/j.rcl.2020.11.002 [DOI] [PubMed] [Google Scholar]
- 55. Andrici J, Tio M, Cox MR, Eslick GD. Hiatal hernia and the risk of Barrett’s esophagus. J Gastro Hepatol. 2013;28:415-431. 10.1111/j.1440-1746.2012.07199.x [DOI] [PubMed] [Google Scholar]
- 56. De La Peña NM, Amrami KK, Spinner RJ. Totally cystic schwannoma: a misnomer. World Neurosurg. 2022;157:21-29. 10.1016/j.wneu.2021.09.098 [DOI] [PubMed] [Google Scholar]
- 57. Savage J, Casey J, McNeill I, Sherman J. Neurenteric cysts of the spine. J Craniovertebr Junction Spine. 2010;1:58-63. 10.4103/0974-8237.65484 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58. Sekimura A, Iwai S, Funasaki A, Motono N, Usuda K, Uramoto H. Müllerian cyst in posterior mediastinum: a report of a case. Int J Surg Case Rep. 2018;49:163-165. 10.1016/j.ijscr.2018.06.037 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Pavlus JD, Carter BW, Tolley MD, Keung ES, Khorashadi L, Lichtenberger JP. Imaging of thoracic neurogenic tumors. Am J Roentgenol. 2016;207:552-561. 10.2214/AJR.16.16018 [DOI] [PubMed] [Google Scholar]
- 60. Reeder LB. Neurogenic tumors of the mediastinum. Semin Thorac Cardiovasc Surg. 2000;12:261-267. 10.1053/stcs.2000.16738 [DOI] [PubMed] [Google Scholar]
- 61. Takeda S, Miyoshi S, Minami M, Matsuda H. Intrathoracic neurogenic tumors—50 years’experience in a Japanese institution. Eur J Cardio-Thorac Surg. 2004;26:807-812. 10.1016/S1010-7940(04)00529-9 [DOI] [PubMed] [Google Scholar]
- 62. Bredella MA, Torriani M, Hornicek F, et al. Value of PET in the assessment of patients with neurofibromatosis type 1. Am J Roentgenol. 2007;189:928-935. 10.2214/AJR.07.2060 [DOI] [PubMed] [Google Scholar]
- 63. Wasa J, Nishida Y, Tsukushi S, et al. MRI features in the differentiation of malignant peripheral nerve sheath tumors and neurofibromas. Am J Roentgenol. 2010;194:1568-1574. 10.2214/AJR.09.2724 [DOI] [PubMed] [Google Scholar]
- 64. Kawashima A, Fishman EK, Kuhlman JE, Nixon MS. CT of posterior mediastinal masses. RadioGraphics. 1991;11:1045-1067. 10.1148/radiographics.11.6.1749849 [DOI] [PubMed] [Google Scholar]
- 65. Crombé A, Fadli D, Clinca R, et al. Imaging of spondylodiscitis: a comprehensive updated review—multimodality imaging findings, differential diagnosis, and specific microorganisms detection. Microorganisms. 2024;12:893. 10.3390/microorganisms12050893 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Sohawon D, Lau KK, Lau T, Bowden DK. Extra‐medullary haematopoiesis: a pictorial review of its typical and atypical locations. J Med Imaging Radiat Oncol. 2012;56:538-544. 10.1111/j.1754-9485.2012.02397.x [DOI] [PubMed] [Google Scholar]
- 67. Georgiades CS, Neyman EG, Francis IR, Sneider MB, Fishman EK. Typical and atypical presentations of extramedullary hemopoiesis. Am J Roentgenol. 2002;179:1239-1243. 10.2214/ajr.179.5.1791239 [DOI] [PubMed] [Google Scholar]
- 68. Tsitouridis J, Stamos S, Hassapopoulou E, Tsitouridis K, Nikolopoulos P. Extramedullary paraspinal hematopoiesis in thalassemia: CT and MRI evaluation. Eur J Radiol. 1999;30:33-38. 10.1016/S0720-048X(98)00101-6 [DOI] [PubMed] [Google Scholar]
- 69. Li JL, Liu HJ, Cui YH, et al. Mediastinal hemangiomas: spectrum of CT and MRI findings—retrospective case series study and systematic review of the literature. Eur J Radiol. 2020;126:108905. 10.1016/j.ejrad.2020.108905 [DOI] [PubMed] [Google Scholar]
- 70. Li SM, Hsu HH, Lee SC, Gao HW, Ko KH. Mediastinal hemangioma presenting with a characteristic feature on dynamic computed tomography images. J Thorac Dis. 2017;9:E412-E415. 10.21037/jtd.2017.03.165 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Chang CA, Pattison DA, Tothill RW, et al. 68Ga-DOTATATE and 18F-FDG PET/CT in paraganglioma and pheochromocytoma: utility, patterns and heterogeneity. Cancer Imaging. 2016;16:22. 10.1186/s40644-016-0084-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Wolfe SA, Sharma S. Parathyroid adenoma [updated 2023 Jun 12]. In: StatPearls [Internet]. StatPearls Publishing; 2025. https://www.ncbi.nlm.nih.gov/books/NBK507870/. [PubMed] [Google Scholar]
- 73. Ota Y, Lee E, Sella E, Agarwal P. Vascular malformations and tumors: a review of classification and imaging features for cardiothoracic radiologists. Radiol: Cardiothorac Imaging. 2023;5:e220328. 10.1148/ryct.220328 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74. Apter S, Avigdor A, Gayer G, Portnoy O, Zissin R, Hertz M. Calcification in lymphoma occurring before therapy: CT features and clinical correlation. Am J Roentgenol. 2002;178:935-938. 10.2214/ajr.178.4.1780935 [DOI] [PubMed] [Google Scholar]
- 75. Silva M, Nunes H, Valeyre D, Sverzellati N. Imaging of sarcoidosis. Clinic Rev Allerg Immunol. 2015;49:45-53. 10.1007/s12016-015-8478-7 [DOI] [PubMed] [Google Scholar]
- 76. Gawne-Cain ML, Hansell DM. The pattern and distribution of calcified mediastinal lymph nodes in sarcoidosis and tuberculosis: a CT study. Clin Radiol. 1996;51:263-267. 10.1016/S0009-9260(96)80343-6 [DOI] [PubMed] [Google Scholar]