Abstract
Although the left and right human cerebral hemispheres differ both functionally and anatomically, little is known about the environmental or genetic factors that govern central nervous system asymmetry. Nevertheless, cerebral asymmetry is strongly correlated with handedness, and handedness does have a significant genetic component. To explore the relative contribution of environmental and genetic influences on cerebral asymmetry, we examined the volumes of left and right cerebral cortex in a large cohort of aging identical and fraternal twins and explored their relationship to handedness. Cerebral lobar volumes had a major genetic component, indicating that genes play a large role in changes in brain volume that occur with aging. Shared environment, which likely represents in utero events, had about twice the effect on the left hemisphere as on the right, consistent with less genetic control over the left hemisphere. To test the major genetic models of handedness and cerebral asymmetry, twin pairs were divided into those with two right handers and those with at least one left hander (nonright handers). Genetic factors contributed twice the influence to left and right cerebral hemispheric volumes in right-handed twin pairs, suggesting a large decrement in genetic control of cerebral volumes in the nonright-handed twin pairs. This loss of genetic determination of the left and right cerebral hemispheres in the nonright-handed twin pairs is consistent with models postulating a right-hand/left-hemisphere-biasing genetic influence, a “right-shift” genotype that is lost in nonright handers, resulting in decreased cerebral asymmetry.
The central nervous system develops through the complex interplay of genetic and environmental factors. Although much attention has focused on mechanisms of anterior–posterior nervous system patterning during development, little attention has been paid to the left–right axis in the brain. Because language and other cognitive functions, such as spatial–constructional abilities, are organized along the left–right axis in the human cerebral hemispheres and possibly more caudal structures, understanding the genetic contributions to the development of cerebral asymmetry has significant implications for cognitive neuroscience.
Functional asymmetry for language is correlated with the structural asymmetry of two perisylvian regions—anteriorly, the pars triangularis of the inferior frontal gyrus (Broca's area); and, more posteriorly, the planum temporale of the superior temporal lobe [Wernicke's area (1–7)]. These focal asymmetries are stable throughout the life span, beginning at birth (7, 8), and are correlated with more gross cerebral hemispheric asymmetries involving both anterior and posterior portions of the cerebral cortex (3, 4, 9). In some cases, the corresponding language-related asymmetric structures are several times larger on the left side than on the right.
Left-hemispheric language lateralization and the associated brain asymmetries are strongly associated with hand preference, and about 97% of right-handers demonstrate predominant left-hemisphere language localization (4, 10–12). By contrast, only about 60% of left handers (defined as nonright handers) demonstrate left-hemisphere language dominance, 30% show bihemispheric language, and about 10% show right-hemisphere language (4, 13). The decreased tendency for left-hemisphere language dominance in nonright handers was found to be accompanied by more structural symmetry in perisylvian language regions (2, 14–17) and loss of other related gross cerebral asymmetries in most (3, 16, 18) but not all studies (19). Although every variety of genetic study, including twin, family, adoption, and cross-fostering studies, demonstrates that handedness in humans has a significant genetic component (20–25), little is known about the genetics of cerebral asymmetry, although handedness and cerebral asymmetry are clearly related (3, 12, 26).
To assess the roles of genetic and environmental contributions to cerebral structural asymmetries, we have analyzed cerebral lobar volumes measured by MRI in a population of 72 monozygotic (MZ) and 67 dizygotic (DZ) older male twin pairs (32). Twin studies offer the opportunity to determine the relative contribution of genes versus the environment in a trait of interest, such as brain asymmetry. Studies in small groups of twins have demonstrated considerable similarities in brain morphology in MZ twins but do not fully distinguish between environmental or genetic etiologies (e.g., refs. 27 and 28). Comparisons with DZ twins are necessary to separate in utero environmental effects from genetic effects (e.g., refs. 29–31). In this study, we were able to evaluate the relative contribution of genetic and environmental influences to frontal, temporal, parietal, and whole-hemispheric volumes of the left and right cerebral hemispheres and to determine the extent to which variation in hand preference has an effect on twin pair similarity and difference on lobar brain volumes. Strikingly, these results lend support to several genetic models of handedness, including the right-shift model of Annett (12), that propose a loss of bias or tendency toward a more random left–right brain asymmetry in those lacking this right-shift genetic influence, such as most left handers (22, 33).
Methods
Study Population.
Data for this study were collected in the ongoing investigation of the genetic and environmental influences on brain structure and function in the National Heart, Lung, and Blood Institute (NHLBI) Twin Study (32, 34). Twins in this study are World War II veterans, born during 1917–1927, and were 42–56 years old when first examined in 1969–1972 (35). In the most recent follow-up (1995–1997) of the NHLBI Twin Study, brain MRI and a comprehensive battery of cognitive function tests were given to these subjects. Analyses in the present study are limited to intact twin pairs who participated in the latest NHLBI examination cycle and for whom MRI data were available. To define left and right handers, we used a questionnaire-based approach that is a relatively standard way to determine hand preference. Handedness was determined by using items similar to 10 items from the Edinburgh Handedness Inventory and confirmed by the subject's writing-hand preference (22, 36–38). The inventory items focused on fine motor function and included: writing, throwing a ball, cutting with scissors, using a knife, using a screwdriver, using a hammer, combing hair, brushing teeth, using chopsticks, and removing a splinter from the foot. Right handers were those who showed right-hand preference in 70% or more of the tasks. All of those subjects classified as nonright-handed wrote with their left hand, a single measure that is most correlated with handedness skill (22, 36), whereas those classified as right-handed wrote with their right hand.
Cerebral MRI Scans and Image Analysis.
MRI (1.5 T) scanning on General Electric scanners was performed at four study sites by using a conventional spin-echo, T1-weighted image with TR 600, TE 17, 24-cm field of view and 5-mm contiguous slices from the nasum to the occiput imaged in a 256 × 192 matrix and interpolated to 256 × 256 with one excitation. Coronal images were acquired at an angle approximately perpendicular to the anterior commissure-posterior commissure line. After acquisition of the MRI scans, the digital information was transferred to a central location for processing and analysis. Volumetric analysis of the MRI scans was performed with a custom-written program operating on a Sun Microsystems (Mountain View, CA) Ultra 5 work station. Image evaluation was based on a semiautomated segmentation analysis that involves operator-guided removal of nonbrain elements, as described (39). Image intensity nonuniformities were then removed from the image, and the resulting corrected image was modeled as a mixture of two Gaussian probability functions (39, 40). The segmentation threshold was determined at the minimum probability between the modeled cerebrospinal fluid and brain matter intensity distribution (39).
After image segmentation, the operator returned to the image for determination of lobar volumes. Lobar volumes were defined by individual regions of interest identified on each image slice through the entire image in which that particular lobar region appeared. To standardize analysis across individuals, all images were rotated into a standard anatomical space by using internal landmarks of the interhemispheric fissure and the anterior commissure–posterior commissure line. Lobar brain regions were obtained as described (41, 42). In brief, frontal lobar regions were defined as all supratemporal structures anterior to the aqueduct of Sylvius. Temporal lobe volume was traced from the anterior pole of the temporal lobe to the aqueduct of Sylvius. The superior-medial temporal lobe boundary was defined as a straight line drawn from the angle of the medial temporal lobe (where it attaches to the temporal stem) to the midpoint of the operculum (see DeCarli et al., ref. 38, for example). The dura of the middle cranial fossa was then traced around each temporal lobe to complete the temporal lobe region. The parietal lobes were defined as the brain matter posterior to the aqueduct of Sylvius, extending to the medial transverse fissure of the striate cortex. The remaining caudal portions of the cerebral hemispheres were defined as occipital.
Analysis.
To determine the proportion of variance attributable to genetic influences, heritability analyses were performed separately for frontal, temporal, parietal, and occipital brain volumes and for right and left hemisphere. Under the assumption of an underlying polygenic model, a heritability estimate near 0% implies no genetic effects, whereas values close to 100% imply strong genetic influences. Heritability analysis was performed by using three different statistical approaches: (i) classical heritability analysis calculated as twice the difference between the MZ and DZ intraclass correlation (Falconer estimate); (ii) an analysis of variance estimate of heritability; and (iii) maximum likelihood estimation (MLE). In the third approach, heritability estimates were obtained by fitting a series of models to the data, assuming that variation in brain volumes was attributable to a combination of additive gene effects (A), common environment (C), and nonshared environmental influences (E). The selection of the most appropriate model fitting the data was based on likelihood statistics. In cases where we could not distinguish the ACE model from the AE model on the basis of MLE statistics, we chose the ACE model as the preferred model when the correlation rDZ was greater than 0.5 rMZ.
The degree of asymmetry was determined by the coefficient (R − L)/[0.5(R + L)] and was calculated separately for each lobar brain volume. Handedness was determined by using the subjects' responses to an inventory of 10 items described above. By using this classification, 61 MZ pairs were concordant for right handedness (RR), one MZ pair was left-handed concordant (LL), and 10 pairs were right–left discordant (RL). In all analyses, LL and RL pairs were grouped together as nonright-handed. The distribution in the 67 DZ pairs was similar: 59 RR, 8 RL, and no LL concordant. The overall frequency of left handedness in this sample of twins was 8.3%, and no relationship of handedness with zygosity was found. In addition, there was no evidence for significant concordance for left handedness for either zygosity. The ratio of concordant (RR + LL) to discordant pairs (RL) was 6.2 in MZ and 7.4 in DZ pairs.
Results
MZ and DZ twins were well matched in terms of age, education, and lobar brain volumes (Table 1). Mean age of the MZ twin pairs was 72.3 ± 2.9 SD years, and that of DZ twin pairs was 71.8 ± 2.8 SD years. No significant differences in the means and standard deviations of lobar brain volumes were observed between MZ twins and DZ twins. Age was negatively and significantly correlated with frontal and temporal brain volume (−0.21 and −0.19, respectively, both P < 0.001) but was not significantly correlated with parietal and occipital brain volumes. Education (number of years of education) was positively and significantly correlated with frontal and temporal brain volumes but showed no significant correlation with parietal and occipital brain volumes.
Table 1.
Mean and SD of sample characteristics by zygosity
| MZ twins | DZ twins | |
|---|---|---|
| Age, yr | 71.8 ± 2.9 | 71.2 ± 2.7 |
| Education, yr | 13.9 ± 3.0 | 13.6 ± 3.0 |
| MMSE | 27.4 ± 2.2 | 26.9 ± 2.9 |
| Word associations (COWA) | 34.8 ± 12.7 | 33.1 ± 13.1 |
| Right-hemisphere brain, cm3 | 468.7 ± 46.8 | 471.2 ± 47.6 |
| Left-hemisphere brain, cm3 | 469.5 ± 45.4 | 470.9 ± 47.7 |
| Right-frontal brain, cm3 | 205.7 ± 24.3 | 206.3 ± 23.3 |
| Left-frontal brain, cm3 | 203.4 ± 23.7 | 203.4 ± 22.9 |
| Right-temporal brain, cm3 | 63.5 ± 8.4 | 63.3 ± 8.5 |
| Left-temporal brain, cm3 | 61.5 ± 8.0 | 60.7 ± 8.1 |
| Right-parietal brain, cm3 | 152.0 ± 24.9 | 152.7 ± 25.7 |
| Left-parietal brain, cm3 | 153.1 ± 26.0 | 153.5 ± 26.7 |
Heritability of Lobar Volumes.
Fig. 1 shows the gross morphological boundaries used in this study. Intraclass or within-twin pair correlations and heritability estimates for lobar brain regions on the left and right brain side are summarized in Table 2. All of the MZ intraclass correlations were statistically significant (all P < 0.01) and significantly greater than the DZ correlations. For both the right and left lobar brain regions, heritability estimates were significant, indicating that a substantial portion of the observed variability in regional brain volumes is due to genetic influences. The brain region with the lowest heritability estimates was the occipital lobe.
Figure 1.
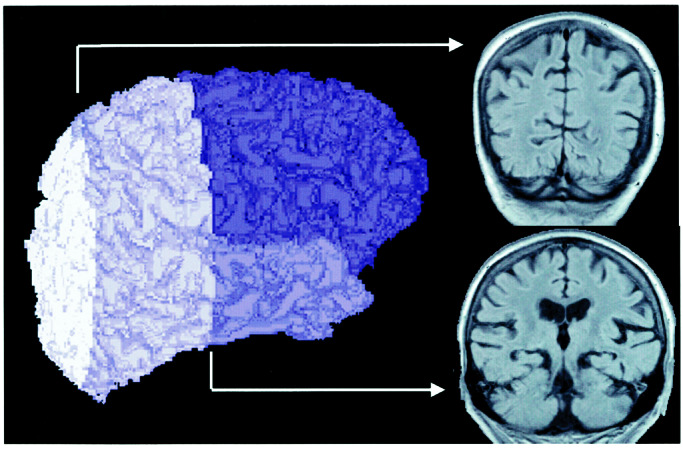
Three-dimensional display of lobar regions. The regions are shaded in order of decreasing darkness from frontal to temporal, parietal, and occipital lobes. Coronal images indicate brain landmarks for distinguishing parietal from occipital and frontal-temporal from parietal lobes. All supratemporal structures anterior to the aqueduct of Sylvius were considered frontal. Temporal lobe volume was traced from the anterior pole of the temporal lobe to the aqueduct of Sylvius. The superior-medial temporal lobe boundary was defined as a straight line drawn from the angle of the medial temporal lobe, where it attaches to the temporal stem, to the midpoint of the operculum. The dura of the middle cranial fossa was then traced around each temporal lobe to complete the temporal lobe region. The parietal lobes were defined as posterior to the aqueduct of Sylvius, extending to the medial transverse fissure of the striate cortex. Occipital cortex was the remaining caudal portion of the hemispheres.
Table 2.
Intraclass correlations and estimates of heritability of lobar brain volumes in the full sample of twins
| Brain volume | Twin correlation
|
Heritability
estimates
|
|||||
|---|---|---|---|---|---|---|---|
| MZ (n = 72) | DZ (n = 67) | Falconer | Within pair | A | MLE
|
||
| C | E | ||||||
| R frontal | 0.76 | 0.43 | 0.66** | 0.60** | 0.56 | 0.16 | 0.28 |
| L frontal | 0.77 | 0.49 | 0.56** | 0.51** | 0.52 | 0.22 | 0.26 |
| R temporal | 0.69 | 0.40 | 0.59** | 0.61** | 0.52 | 0.15 | 0.33 |
| L temporal | 0.68 | 0.47 | (0.41) | 0.42* | 0.40 | 0.26 | 0.34 |
| R parietal | 0.54 | 0.34 | (0.41) | 0.47* | 0.45 | 0.10 | 0.45 |
| L parietal | 0.57 | 0.33 | 0.47* | 0.52* | 0.49 | 0.06 | 0.45 |
| R occipital | 0.38 | 0.24 | (0.28) | 0.49* | 0.27 | 0.07 | 0.66 |
| L occipital | 0.37 | 0.02 | 0.69* | 0.56* | 0.29 | 0.01 | 0.70 |
| R hemisphere | 0.86 | 0.56 | 0.60** | 0.62** | 0.64 | 0.22 | 0.14 |
| L hemisphere | 0.87 | 0.57 | 0.60** | 0.66** | 0.67 | 0.21 | 0.12 |
| Total cerebral hemispheres | 0.87 | 0.56 | 0.61** | 0.65** | 0.64 | 0.23 | 0.13 |
Falconer, 2*(RMZ − RDZ); within pair, 2*(WDZ − WMZ)/[(SMZ + SDZ)/4], where WDZ and WMZ stand for within mean square, and SMZ and SDZ stand for sum of mean square. MLE, maximum likelihood estimate, determined from the best biometric model fitting the observed twin variances and covariances. A, additive gene effects; C, common environment; E, nonshared environmental influences. Heritability estimates in parentheses are statistically not significant.
, P < .05,
, P < .01.
Shared environmental effects contributed significantly to the variation of the left-hemispheric temporal brain volume (26%) and of the left frontal brain volume (22%). But shared environment had less of an effect on right-sided frontal and temporal brain volumes and essentially no effect on dorsal brain volumes. This increased effect of shared environment is consistent with the slightly lower heritability of left frontal and temporal volumes, relative to the homologous regions on the right. These results indicate that these right-hemisphere structures are more genetically determined than those on the left, and the left hemisphere is more susceptible to environmental factors than the right hemisphere.
Relationship Between Asymmetry and Handedness.
We also examined the degree of asymmetry in lobar brain volumes (the differences in volume between the right and left sides of the brain within individual subjects) and the relationship of handedness to twin-pair similarity on regional brain volumes. Table 3 summarizes the calculated mean and SD of the asymmetry coefficients for different brain regions in the sample as a whole and stratified by handedness. For the sample as a whole, and in right-handed twin pairs (RR), we observed significant rightward asymmetry of frontal and temporal brain volumes and significant leftward asymmetry of parietal and occipital volumes. In contrast, twin pairs with one or more left-handed twins [nonright-handed pairs (non-RR)] showed significant leftward asymmetry in occipital brain volumes but no significant asymmetry in frontal, temporal, and parietal brain regions. The loss of asymmetry was observed equally in the right- and left-handed individuals from among the nonright-handed twin pairs. This overall loss of asymmetry is what is observed in singleton left handers and is consistent with previous reports showing that left handers have more symmetrical brains than do right handers (17).
Table 3.
Mean and SD of the asymmetry coefficient (R − L)/[0.5(R + L)] in the full sample of MZ and DZ twins stratified by handedness
| Brain structure | Full sample n = 278 | Twins of RR pairs n = 240 | Twins of non-RR pairs
|
|
|---|---|---|---|---|
| Right-handed twin | Left-handed twin | |||
| Frontal | 1.25 ± 3.58** | 1.37 ± 3.60** | 0.18 ± 3.50 | 0.57 ± 3.18 |
| Temporal | 3.81 ± 9.23** | 3.92 ± 9.37** | 2.64 ± 7.78 | 2.19 ± 7.42 |
| Parietal | −0.45 ± 3.99* | −0.58 ± 3.93* | 0.20 ± 3.77 | 0.34 ± 4.79 |
| Occipital | −9.19 ± 18.01** | −9.20 ± 18.19** | −8.03 ± 19.1** | −8.81 ± 16.35** |
| Total hemisphere | −0.06 ± 1.93 | −0.07 ± 1.90 | −0.12 ± 1.33 | −0.10 ± 2.19 |
In these analyses, subjects were treated as genetically unrelated subjects and standard errors were calculated by using 1,000 bootstrap samples.
Significantly different than zero at P < 0.05;
Significantly different than zero at P < 0.01.
To further explore the relationship of cerebral asymmetry to handedness in this sample of twins, we evaluated the correlation between the right- and left-side brain volume in the sample as a whole and grouped by handedness. As shown in Table 4, all correlations between right and left cerebral volumes were significant and of the same order of magnitude for frontal, parietal, and occipital regions. The correlation, however, appears to be lower (0.76) between the right and left temporal brain volume. Moreover, when we stratified the data by handedness, we observed that the correlation between right and left temporal brain volume was higher in nonright-handed pairs than in the concordant right-handed pairs (0.86 vs. 0.74, ts = 1.89, P = 0.03), which indicates a trend toward more symmetry in the temporal lobe of nonright-handed twin pairs, as has been observed in nontwin cohorts.
Table 4.
Pearson correlations between right and left lobar brain volumes in the full sample stratified by handedness and treating twins as genetically unrelated individuals
| Brain structure | Full sample n = 278 | RR pairs n = 240 | Non-RR pairs n = 38 |
|---|---|---|---|
| Frontal | 0.95 | 0.95 | 0.95 |
| Temporal | 0.75 | 0.74 | 0.86 |
| Parietal | 0.97 | 0.97 | 0.97 |
| Occipital | 0.87 | 0.87 | 0.87 |
| Total hemisphere | 0.98 | 0.98 | 0.98 |
All Pearson correlations significant at the P < 0.01 level.
Testing Genetic Models of Cerebral Asymmetry and Handedness.
Several major genetic models of handedness propose that right handers inherit a directional bias for cerebral language dominance and motor dominance (12, 22, 33). The seminal right-shift gene theory postulated by Annett (12) and a similar corrective model proposed by McManus (22) state that right handers inherit a specific directional bias in brain asymmetries related to motor and cognitive functions (specifically language) that is not inherited by most left handers. This genetic bias strongly predisposes individuals to becoming right-handed and to carrying language in the left hemisphere. The loss of genetic bias postulated in left handers does not lead to an absolute reversal in the typical asymmetry, but rather a relative randomness; this finding is consistent with the observed relatively random or less biased lateralization of language and other cognitive functions in left handers (3, 4, 26). Because it is loss of bias, rather than a change in directionality, that is inherited, some of those lacking the putative right-shift genotype (R−/−) will be right-handed. However, the majority of right handers will carry the right-shift genotype, either as homozygotes or heterozygotes (R+/+ or R+/−), because the right-shift acts dominantly.
Because these major genetic models of handedness postulate that the genetic influences on handedness are not absolute, but impose a bias in right handers that is lost in most left handers, those lacking the right-shift genotype may be either right-handed, ambidextrous, or left-handed. Furthermore, they may have left-hemisphere, bilateral, or right-hemisphere language, but they are less likely than those with the right-handed genotype to be strongly right-handed and have left-hemisphere restricted language (3, 13). Therefore, to test the right-shift and related genetic models, MZ and DZ twins were divided into two groups: those with the postulated right-shift genotype, consisting of RR pairs, and those lacking the right-shift genotype, consisting of nonright-handed pairs. Because the group lacking the right-shift genotype includes those that are concordant for left-handedness (LL pair), in addition to discordant RL pairs, we labeled this latter subgroup of twin pairs “nonright-handers (non-RR),” to distinguish them from the concordant, RR group. Supporting this grouping and the notion that the discordant RL pairs were equivalent to LL pairs, both the right- and left-handed twins among the RL twin pairs had no significant asymmetry in frontal and temporal brain regions, in contrast to the RR pairs, who demonstrated significant asymmetry (Table 3). This assumption also fits with the concordance data for handedness in MZ and DZ twins (43).
Table 5 shows the correlation between the same lobar region or hemisphere within MZ twin pairs and the relationship of this correlation to handedness. Intraclass correlation coefficients for lobar brain volumes of identical twins who were both right-handed (RR pairs) and those who were non-RR [consisting of those that were either both left-handed (LL pair) or discordant for handedness (RL pairs)] are depicted. There is a clear trend for greater within-twin-pair similarity on frontal brain volumes among RR pairs than non-RR pairs. A greater twin-pair similarity on whole-hemispheric volumes was also observed among the RR twin pairs, compared with the non-RR pairs. These differences between RR pairs versus non-RR pairs reached significance for right- and left-side total hemisphere brain volume and left-frontal brain volume. The greater within-twin-pair similarity of hemispheric volumes observed in RR pairs demonstrates that there is more genetic control over these volumes in RR pairs, who carry the putative right-shift genotype, than in the non-RR pairs, who lack the putative right-shift genotype. This finding supports the model that the non-RR pairs inherit a diminished genetic control over left and right cerebral volumes and thus have lost the usual directional bias that is inherited by most right handers. The tendency toward relative symmetry observed in frontal and temporal brain regions in the non-RR twin pairs also bolsters this observation.
Table 5.
Comparisons of the MZ intraclass correlations in the RR versus non-RR pairs (rRR-MZ vs. rnon-RR-MZ)
| Brain volume | rRR-MZ (n = 61) | rnon-RR-MZ (n = 11) | F(WRL/WRR) | P value |
|---|---|---|---|---|
| R frontal | 0.79 | 0.47 | 1.59 | 0.12 |
| L frontal | 0.81 | 0.46 | 1.86* | <0.05 |
| R temporal | 0.67 | 0.76 | 1.19 | 0.40 |
| L temporal | 0.64 | 0.78 | 0.92 | 0.55 |
| R parietal | 0.52 | 0.66 | 1.07 | 0.45 |
| L parietal | 0.54 | 0.68 | 1.09 | 0.44 |
| R hemisphere | 0.89 | 0.67 | 2.91** | <0.01 |
| L hemisphere | 0.89 | 0.72 | 2.27** | <0.02 |
| Total hemisphere | 0.90 | 0.70 | 2.70** | <0.01 |
rRR-MZ, intraclass correlation in RR MZ twin pairs. rnon-RR-MZ, intraclass correlation in non-RR MZ twin pairs. WRR, within-pair mean square in RR MZ twin pairs. WRL, within-pair mean square in non-RR MZ twin pairs. P values given are one-tailed tests. P = one-tailed P value for the test of rRR-MZ > rnon-RR-MZ.
, P < 0.05,
, P < 0.01.
Although these data clearly fit with the genetic models discussed, an alternative interpretation would be that noninherited in utero factors (e.g., maternal hormone levels) underlie the loss of asymmetry and lower intraclass correlations between left and right lobar volumes in non-RR MZ twin pairs. If this were so, a similar pattern of lower intraclass correlation between lobar or whole-hemispheric volumes would be observed in DZ non-RR twin pairs. As shown in Table 6, this is not the case. Although the number of non-RR DZ twins is small (n = 8), there is not even a trend toward lower correlations in the non-RR twin pairs. This difference between MZ and DZ twins provides strong support that there is a genetic basis for the loss of asymmetry and lower correlations between hemispheric and some lobar volumes in those lacking the right-shift genotype. This genetic influence is not strictly deterministic but leads to a relative loss of the typical strong bias toward right-handedness and brain asymmetry.
Table 6.
Comparisons of the DZ intraclass correlations in the RR versus non-RR twin pairs (rRR-DZ vs. rnon-RR-DZ)
| Brain volume | rRR-DZ (n = 59) | rnon-RR-DZ (n = 8) | F(WRL/WRR) | P value |
|---|---|---|---|---|
| R frontal | 0.37 | 0.76 | 0.58 | 0.60 |
| L frontal | 0.45 | 0.73 | 0.73 | 0.65 |
| R temporal | 0.39 | 0.46 | 1.60 | 0.08 |
| L temporal | 0.46 | 0.63 | 0.44 | 0.80 |
| R parietal | 0.32 | 0.60 | 0.46 | 0.79 |
| L parietal | 0.30 | 0.68 | 0.40 | 0.85 |
| R hemisphere | 0.55 | 0.63 | 0.85 | 0.50 |
| L hemisphere | 0.56 | 0.64 | 0.75 | 0.70 |
| Total hemisphere | 0.56 | 0.63 | 0.81 | 0.52 |
rRR-DZ, intraclass correlation in RR DZ twin pairs. rnon-RR-DZ, intraclass correlation in the eight non-RR DZ twin pairs. WRR, within-pair mean square in RR DZ twin pairs. WRL, within-pair mean square in non-RR DZ twin pairs. P values given are one-tailed tests. P, one-tailed P value for the test of rRR-DZ > rnon-RR-DZ.
The Relationship of Total Brain Volume to Handedness.
In addition to the lower intraclass correlation of left and right hemispheric volume among the non-RR MZ pairs (Table 5), overall brain volume showed significantly lower intraclass correlation, as one might expect based on the separate right and left hemisphere results. This result suggested that perhaps asymmetry is related to overall cerebral cortical volume (44, 45). There was a significant difference in both left and right frontal lobar volumes between the right- and left-handed twins from the RL discordant pairs (t test, P < 0.001), consistent with the lower intraclass correlations in these pairs. However, there was no difference in overall cerebral or individual lobar volumes between the non-RR and RR twin groups or between total right or left cerebral hemispheric volume in the right- or left-handed twin from the non-RR twin pairs (see supplemental materials, which include plots of lobar and hemispheric volume by twin-handedness category, at http://geschwindlab.medsch.ucla.edu).
Discussion
Cerebral Cortex Heritability, Environmental Factors, and Aging.
We have analyzed the heritability of lobar and cerebral hemispheric volumes in a large cohort of aging twins and studied the correlation of these measures with handedness. All major brain structures studied showed significant heritabilities, consistent with previous results in less aged populations (46–48). Because the majority of these twins' lives have been spent apart from one another, these high heritabilities suggest that genetic background plays a larger role than environmental influences in the changes in brain structure that occur with aging. Whether these genetic factors act during early development to shape the brain, or later as a response to aging, is unknown, but both genetic influences are likely at work.
Surprisingly, the contribution of shared environmental factors, most of which are related to in utero and early familial factors in an aged twin population (30, 49), accounts for a significant amount of the environmental influence on frontal, temporal, and whole-hemispheric volumes. The influence of a shared environment was almost twice as strong on the left in frontal and temporal regions as on the right, regardless of twin handedness. One plausible explanation for the increased malleability of these left-hemisphere regions is that the left hemisphere develops over a longer time period than the right (50), thus potentially making it more vulnerable to environmental perturbations such as the in utero hormonal environment (3). By having its major effect on the left hemisphere, the in utero environment would still be expected to alter structures relevant to language, as has been proposed (3). The contribution of shared environment further suggests that early developmental events that shape the brain contribute significantly to the brain's response to aging, because these are all elderly subjects (51, 52).
It is interesting to note that frontal and temporal lobar volumes were correlated with educational level, whereas parietal and occipital volumes were not. One can speculate that this may reflect the bias inherent in the educational system, which primarily taps and rewards functions most localized in frontal and temporal regions, such as language, and executive function, rather than more posterior or parietal functions, such as constructional abilities.
Heritability of Cerebral Asymmetry and Genetic Models of Handedness.
To test the major genetic models of handedness, cerebral lobar volumes in twin pairs lacking the putative right-shift genotype (non-RR pairs) were compared with lobar volumes in right-handed twin pairs (RR), most of whom are predicted to carry the right-shift genotype. A significantly stronger tendency toward gross cerebral asymmetry was observed in the concordant right-handed twin pairs. Furthermore, more symmetry was observed in both the left- and right-handed twin from the non-RR pairs. The gross trend toward structural symmetry observed in non-RR pairs was similar to what is observed in left handers, even though one twin from these non-RR pairs was often right-handed. This trend is consistent with current models of cerebral asymmetry and its relationship to handedness, in which the right-handed phenotype should be frequently observed in those with the left-handed genotype (12, 33, 43). It is possible that a “left” genotype twin pair could both be phenotypically right-handed. These rare right-phenotype–left-genotype twin pairs cannot be identified by using current methods. Such bias would weaken, rather than enhance, our ability to detect a genetic influence because of heterogeneity within our categories. The presence of “pathologic” left handedness would also weaken our ability to detect genetic factors rather than increase such a bias. In addition, pathological left handedness is unlikely in this study, given that these are healthy twin pairs, without a history of perinatal or other insult or imaging evidence of brain damage.
Strikingly, a lower intraclass correlation between the frontal lobes and whole-hemispheric volumes in MZ twins was observed in the non-RR MZ pairs, relative to the RR pairs. These findings indicate that the genetic influences on right- and left-hemisphere volumes are greater in RR MZ twins. Conversely, genetic factors have only half the influence on hemispheric volumes in non-RR pairs, compared with RR pairs, indicating a large decrement in genetic determination over this critical process in cerebral pattern formation in non-RR pairs. This loss of genetic determination of the left and right cerebral hemispheres is consistent with models postulating a lost right-shift gene or a gain in randomness in those with the left-handed genotype (12, 22, 33, 57). That this is a genetic effect is strongly supported in the present study by the lack of any decrement in total hemispheric or lobar volume interclass correlation in non-RR DZ twin pairs, which would be the case if shared environmental influences were the major factor. These results do not specify whether such an influence is a major gene, or many genes, but strongly support the concept that bias to the right is what is inherited in most right handers in the general population, and that this bias is lost in most left handers.
This concept of loss of positional determinism runs parallel to a large body of data from studies on the molecular genetic mechanisms of visceral asymmetry (reviewed in refs. 56 and 58). In one well known animal model of visceral asymmetry, the iv mouse, a mutation in left-right dynein leads to random (either left or all-right) organ placement along the left–right axis rather than a totally determined mirror image (53, 54). The example of the iv mouse has been used to support the genetic model of handedness postulated by Klar (33), which is similar to the other major gene models of handedness in proposing a major gene influence (12, 22, 51). Loss of or reduction in directional bias, as well as total loss of positional information, can occur in mouse laterality models as well (55, 56).
Lobar Volumes and More Discrete Language-Related Asymmetries.
Structural brain asymmetry of the planum temporale has been studied previously in a smaller MZ twin cohort stratified by handedness, and no genetic component underlying the degree of asymmetry (asymmetry coefficient) was identified (17). However, there are important differences between the two studies and, although the authors reached different conclusions, when viewed from the current perspective, the data in the earlier study do not contradict the current findings. First, because even in the present large study the numbers are barely sufficient to provide statistical significance, it is not surprising that such significance was not obtained in the smaller study. Second, in the previous work, no MZ versus DZ comparisons were done, and the intraclass correlation of the degree of asymmetry was the metric used to determine heritability, rather than lobar volumes (17). However, it is not the degree of asymmetry but rather the loss of its genetic control that we presumed to be inherited. Thus, one does not expect high-interclass correlations of an asymmetry coefficient in non-RR MZ twins but rather a lower intraclass correlation than is observed in RR pairs. This trend was observed in both studies. Furthermore, in both studies, the left-handed (or non-RR) twins tended toward more symmetric brains than the RR cohort, a finding that is consistent with loss of directional bias or relative randomness. Finally, we observed a clear difference in these measures in MZ and DZ twin pairs consistent with an underlying genetic etiology.
It is also interesting to note that the total hemispheric volume differences in intraclass correlations were more significant than individual lobar differences. We postulate that this is probably because of the contribution of measurement error in the smaller lobar regions and is one reason why such gross measures were used in this study. Measurement error, or bias in regional partitioning, is also likely the explanation for the lower heritabilities observed in occipital cortex in this study, as occipital cortex was defined on the basis of exclusionary criteria. So, although the regions studied are large and extend far beyond the asymmetric language regions, gross lobar and hemispheric asymmetries are strongly correlated with anatomically defined language asymmetries (3, 9). Although in future studies with higher-resolution MRI scans it may be preferable to study the planum temporale and inferior frontal regions in isolation, several issues led us to concentrate on gross regions rather than the smaller regions implicated in language by lesion and functional imaging studies. By using whole volumes, we minimized potential problems inherent in the measurement of specific brain regions with MRI images of the resolution available for this study (5 mm). Typically, these regions are defined by sulcal boundaries, but their measurement is a source of controversy in many cases. For example, the definition of the posterior boundary of the planum temporale is debated and can vary greatly between studies. Additionally, the boundaries of cortical regions defined by cortical surface morphology do not correspond to functionally meaningful cytoarchitectonic boundaries, and the relationship between cytoarchitecture and sulcal markings is quite variable between individuals, posing an unanswered challenge for even the most high-resolution structural imaging methods (59). The approach that we have taken is further supported by the observation that similarity in gross brain morphology is strongly correlated with increased sulcal similarities in MZ twins and that the MZ twins have high correlations in the patterning of deep sulci but less correlation between smaller and less deep sulci (31). However, it clearly is still a reasonable goal to study in the future more discrete brain regions that have clearer functional correlations than the regions used in this study. The results of the current analysis provide an important rationale for such approaches, as well as confirmation of these results in large independent samples.
Acknowledgments
We gratefully acknowledge Roger Woods for generous and invaluable discussions and critical comments on the manuscript and Bonita Porch for editorial assistance. We also thank two anonymous reviewers for helpful suggestions and Harley Kornblum and Michael Geschwind for comments on the manuscript. We acknowledge support from the National Institute of Mental Health (Grant MH60233) (D.H.G.); the National Alliance for Research on Schizophrenia and Depression (D.H.G.); the National Heart, Lung, and Blood Institute (D.C., C.D., B.M.), and the James S. McDonnell Foundation (D.H.G.).
Abbreviations
- MZ
monozygotic
- DZ
dizygotic
- RR
right-handed concordant
- LL
left-handed concordant
- RL
right–left discordant
Footnotes
This paper was submitted directly (Track II) to the PNAS office.
References
- 1.Foundas A L, Leonard C M, Gilmore R L, Fennell E B, Heilman K M. Proc Natl Acad Sci USA. 1996;93:719–722. doi: 10.1073/pnas.93.2.719. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Foundas A L, Eure K F, Luevano L F, Weinberger D R. Brain Lang. 1998;64:282–296. doi: 10.1006/brln.1998.1974. [DOI] [PubMed] [Google Scholar]
- 3.Geschwind N, Galaburda A. Arch Neurol. 1985;42:428–458. doi: 10.1001/archneur.1985.04060050026008. .; 521–552. [DOI] [PubMed] [Google Scholar]
- 4.Geschwind N. Science. 1970;170:940–944. doi: 10.1126/science.170.3961.940. [DOI] [PubMed] [Google Scholar]
- 5.Galaburda A M. In: Biological Asymmetry and Handedness. Marsh J, Bock M J, editors. New York: Wiley; 1991. pp. 219–226. [Google Scholar]
- 6.Galaburda A. In: Brain Asymmetry. Davidson R, Hugdahl K, editors. Cambridge, MA: MIT Press; 1995. pp. 51–73. [Google Scholar]
- 7.Witelson S F, Pallie W. Brain. 1973;96:641–646. doi: 10.1093/brain/96.3.641. [DOI] [PubMed] [Google Scholar]
- 8.Preis S, Jancke L, Schmitz-Hillebrecht J, Steinmetz H. Brain Cogn. 1999;40:441–452. doi: 10.1006/brcg.1998.1072. [DOI] [PubMed] [Google Scholar]
- 9.Zilles K, Dabringhaus A, Geyer S, Amunts K, Qu M, Schleicher A, Gilissen E, Schlaug G, Steinmetz H. Neurosci Biobehav Rev. 1996;20:593–605. doi: 10.1016/0149-7634(95)00072-0. [DOI] [PubMed] [Google Scholar]
- 10.Damasio A R, Geschwind N. Annu Rev Neurosci. 1984;7:127–147. doi: 10.1146/annurev.ne.07.030184.001015. [DOI] [PubMed] [Google Scholar]
- 11.Benson D F. Cortex. 1986;22:71–86. doi: 10.1016/s0010-9452(86)80033-8. [DOI] [PubMed] [Google Scholar]
- 12.Annett M. Left, Right, Hand and Brain: The Right Shift Theory. London: Erlbaum; 1985. [Google Scholar]
- 13.Hécaen H, Consoli S. Neuropsychologia. 1973;11:377–388. doi: 10.1016/0028-3932(73)90024-9. [DOI] [PubMed] [Google Scholar]
- 14.Chiu H C, Damasio A R. J Neurol Neurosurg Psychiatry. 1980;43:873–878. doi: 10.1136/jnnp.43.10.873. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Foundas A L, Hong K, Leonard C M, Heilman K M. Neuropsychiatry Neuropsychol Behav Neurol. 1998;11:65–71. [PubMed] [Google Scholar]
- 16.Steinmetz H, Volkmann J, Jancke L, Freund H J. Ann Neurol. 1991;29:315–319. doi: 10.1002/ana.410290314. [DOI] [PubMed] [Google Scholar]
- 17.Steinmetz H, Herzog A, Schlaug G, Huang Y, Jancke L. Cereb Cortex. 1995;5:296–300. doi: 10.1093/cercor/5.4.296. [DOI] [PubMed] [Google Scholar]
- 18.Geschwind N. Sci Am. 1972;226:76–83. doi: 10.1038/scientificamerican0472-76. [DOI] [PubMed] [Google Scholar]
- 19.Witelson S F, Kigar D L. J Comp Neurol. 1992;323:326–340. doi: 10.1002/cne.903230303. [DOI] [PubMed] [Google Scholar]
- 20.Hicks R E, Hinsborne M. Science. 1976;192:908–910. doi: 10.1126/science.1273577. [DOI] [PubMed] [Google Scholar]
- 21.Longstreth L E. J Gen Psychol. 1980;137:275–283. [Google Scholar]
- 22.McManus I C. Ciba Found Symp. 1991;162:251–267. doi: 10.1002/9780470514160.ch15. [DOI] [PubMed] [Google Scholar]
- 23.Annett M. Br J Psychol. 1974;65:129–131. doi: 10.1111/j.2044-8295.1974.tb02778.x. [DOI] [PubMed] [Google Scholar]
- 24.Carter S L, Scarr S S, Barker W B, Katz S. Behav Genet. 1976;6:189–203. doi: 10.1007/BF01067148. [DOI] [PubMed] [Google Scholar]
- 25.Chamberlain H D. J Hered. 1928;19:557–559. [Google Scholar]
- 26.Alexander M P, Annett M. Brain Lang. 1996;55:213–239. doi: 10.1006/brln.1996.0102. [DOI] [PubMed] [Google Scholar]
- 27.Oppenheim J S, Skerry J E, Tramo M J, Gazzaniga M S. Ann Neurol. 1989;26:100–104. doi: 10.1002/ana.410260117. [DOI] [PubMed] [Google Scholar]
- 28.Tramo M J, Loftus W C, Stukel T A, Green R L, Weaver J B, Gazzaniga M S. Neurology. 1998;50:1246–1252. doi: 10.1212/wnl.50.5.1246. [DOI] [PubMed] [Google Scholar]
- 29.Pfefferbaum A, Sullivan E V, Carmelli D. NeuroReport. 2001;12:1677–1681. doi: 10.1097/00001756-200106130-00032. [DOI] [PubMed] [Google Scholar]
- 30.Pfefferbaum A, Sullivan E V, Swan G E, Carmelli D. Neurobiol Aging. 2000;21:63–74. doi: 10.1016/s0197-4580(00)00086-5. [DOI] [PubMed] [Google Scholar]
- 31.Lohmann G, von Cramon D Y, Steinmetz H. Cereb Cortex. 1999;9:754–763. doi: 10.1093/cercor/9.7.754. [DOI] [PubMed] [Google Scholar]
- 32.Carmelli D, Swan G E, Reed T, Wolf P A, Miller B L, DeCarli C. Neurology. 1999;52:1119–1124. doi: 10.1212/wnl.52.6.1119. [DOI] [PubMed] [Google Scholar]
- 33.Klar A J. Cold Spring Harbor Symp Quant Biol. 1996;61:59–65. [PubMed] [Google Scholar]
- 34.Carmelli D, Swan G E, Reed T, Miller B, Wolf P A, Jarvik G P, Schellenberg G D. Neurology. 1998;50:1580–1585. doi: 10.1212/wnl.50.6.1580. [DOI] [PubMed] [Google Scholar]
- 35.Feinleib M, Garrison R J, Fabsitz R, Christian J C, Hrubec Z, Borhani N O, Kannel W B, Rosenman R, Schwartz J T, Wagner J O. Am J Epidemiol. 1977;106:284–285. doi: 10.1093/oxfordjournals.aje.a112464. [DOI] [PubMed] [Google Scholar]
- 36.Corey D M, Hurley M M, Foundas A L. Neuropsychiatry Neuropsychol Behav Neurol. 2001;14:144–152. [PubMed] [Google Scholar]
- 37.Oldfield R C. Neuropsychologia. 1971;9:97–113. doi: 10.1016/0028-3932(71)90067-4. [DOI] [PubMed] [Google Scholar]
- 38.Raczkowski D, Kalat J W, Nebes R. Neuropsychologia. 1974;12:43–47. doi: 10.1016/0028-3932(74)90025-6. [DOI] [PubMed] [Google Scholar]
- 39.DeCarli C, Maisog J, Murphy D G, Teichberg D, Rapoport S I, Horwitz B. J Comput Assist Tomogr. 1992;16:274–284. doi: 10.1097/00004728-199203000-00018. [DOI] [PubMed] [Google Scholar]
- 40.DeCarli C, Murphy D G, Teichberg D, Campbell G, Sobering G S. J Magn Reson Imaging. 1996;6:519–528. doi: 10.1002/jmri.1880060316. [DOI] [PubMed] [Google Scholar]
- 41.DeCarli C, Murphy D G, Gillette J A, Haxby J V, Teichberg D, Schapiro M B, Horwitz B. Am J Neuroradiol. 1994;15:689–696. [PMC free article] [PubMed] [Google Scholar]
- 42.Murphy D G, DeCarli C, McIntosh A R, Daly E, Mentis M J, Pietrini P, Szczepanik J, Schapiro M B, Grady C L, Horwitz B, Rapoport S I. Arch Gen Psychiatry. 1996;53:585–594. doi: 10.1001/archpsyc.1996.01830070031007. [DOI] [PubMed] [Google Scholar]
- 43.Sicotte N, Woods R, Mazziotta J. Laterality. 1999;4:265–286. doi: 10.1080/713754339. [DOI] [PubMed] [Google Scholar]
- 44.Jancke L, Preis S, Steinmetz H. NeuroReport. 1999;10:2981–2985. doi: 10.1097/00001756-199909290-00020. [DOI] [PubMed] [Google Scholar]
- 45.Jancke L, Staiger J F, Schlaug G, Huang Y, Steinmetz H. Cereb Cortex. 1997;7:48–56. doi: 10.1093/cercor/7.1.48. [DOI] [PubMed] [Google Scholar]
- 46.Alarcón M, Pennington B F, Filipek P A, DeFries J C. Dev Neuropsychol. 2000;17:339–360. doi: 10.1207/S15326942DN1703_4. [DOI] [PubMed] [Google Scholar]
- 47.Bartley A J, Jones D W, Weinberger D R. Brain. 1997;120:257–269. doi: 10.1093/brain/120.2.257. [DOI] [PubMed] [Google Scholar]
- 48.Pennington B F, Filipek P A, Lefly D, Chhabildas N, Kennedy D N, Simon J H, Filley C M, Galaburda A, DeFries J C. J Cognit Neurosci. 2000;12:223–232. doi: 10.1162/089892900561850. [DOI] [PubMed] [Google Scholar]
- 49.Machin G A. Am J Med Genet. 1996;61:216–228. doi: 10.1002/(SICI)1096-8628(19960122)61:3<216::AID-AJMG5>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- 50.Chi J G, Dooling E C, Gilles F H. Ann Neurol. 1976;1:86–93. doi: 10.1002/ana.410010109. [DOI] [PubMed] [Google Scholar]
- 51.Geschwind D H, Miller B L. Am J Med Genet. 2001;101:370–381. [PubMed] [Google Scholar]
- 52.Geschwind D H, Robidoux J, Alarcón M, Miller B L, Wilhelmsen K C, Cummings J L, Nasreddine Z S. Ann Neurol. 2001;50:741–746. doi: 10.1002/ana.10024. [DOI] [PubMed] [Google Scholar]
- 53.Supp D M, Witte D P, Potter S S, Brueckner M. Nature (London) 1997;389:963–966. doi: 10.1038/40140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Layton W M., Jr J Hered. 1976;67:336–338. doi: 10.1093/oxfordjournals.jhered.a108749. [DOI] [PubMed] [Google Scholar]
- 55.Bisgrove B W, Essner J J, Yost H J. Development (Cambridge, UK) 2000;127:3567–3579. doi: 10.1242/dev.127.16.3567. [DOI] [PubMed] [Google Scholar]
- 56.Yost H J. Int Rev Cytol. 2001;203:357–381. doi: 10.1016/s0074-7696(01)03011-x. [DOI] [PubMed] [Google Scholar]
- 57.Klar A J. Schizophr Res. 1999;39:207–218. doi: 10.1016/s0920-9964(99)00075-4. [DOI] [PubMed] [Google Scholar]
- 58.Yost H J. Dev Genet. 1998;23:159–163. doi: 10.1002/(SICI)1520-6408(1998)23:3<159::AID-DVG1>3.0.CO;2-1. [DOI] [PubMed] [Google Scholar]
- 59.Rajkowska G, Goldman-Rakic P S. Cereb Cortex. 1995;5:307–322. doi: 10.1093/cercor/5.4.307. [DOI] [PubMed] [Google Scholar]


