Abstract
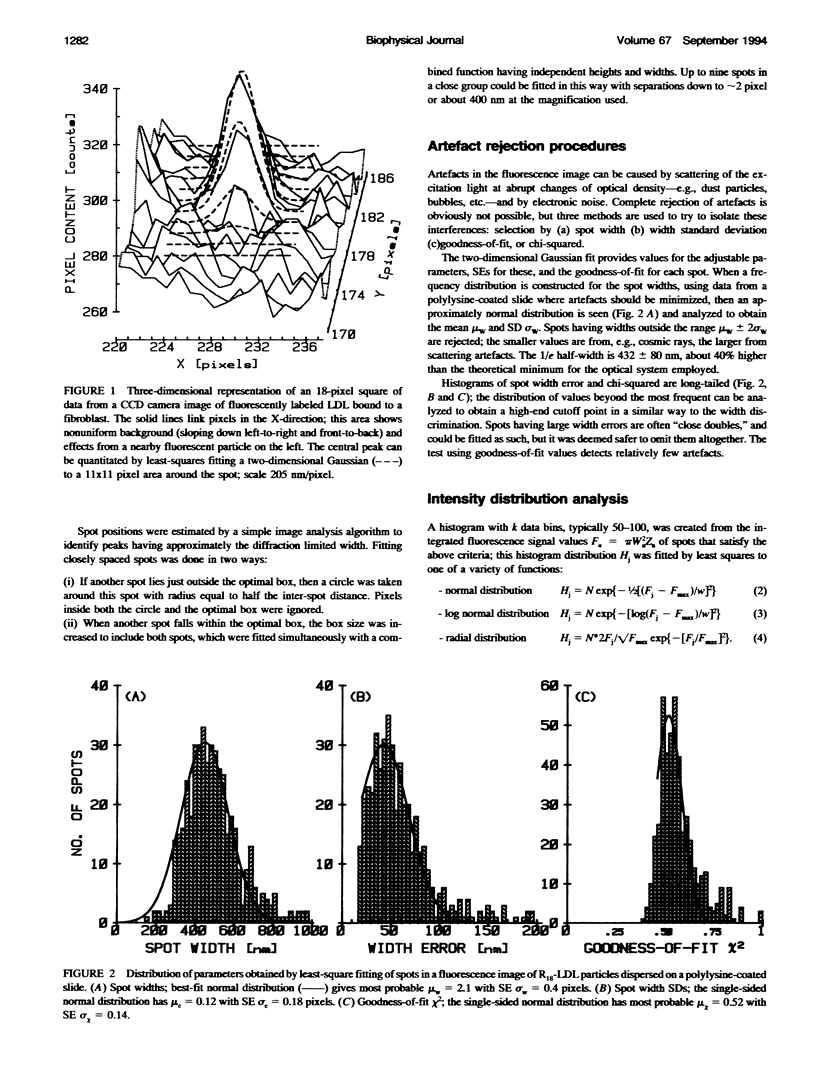
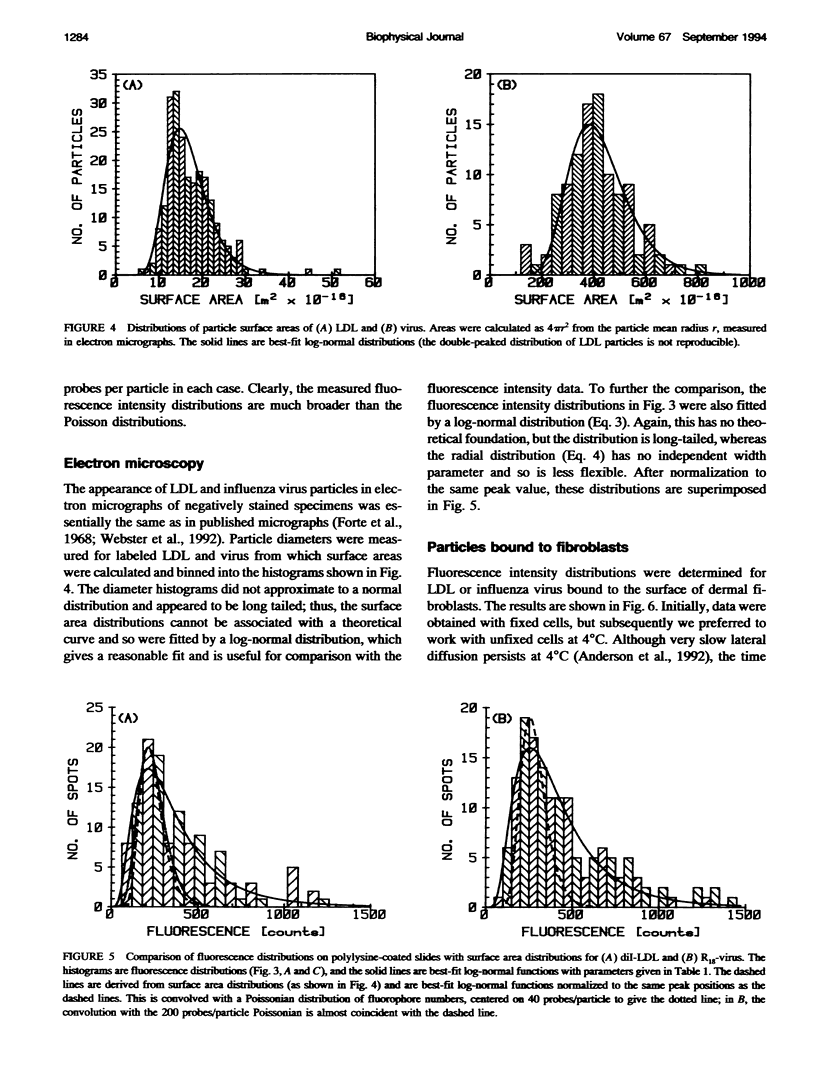
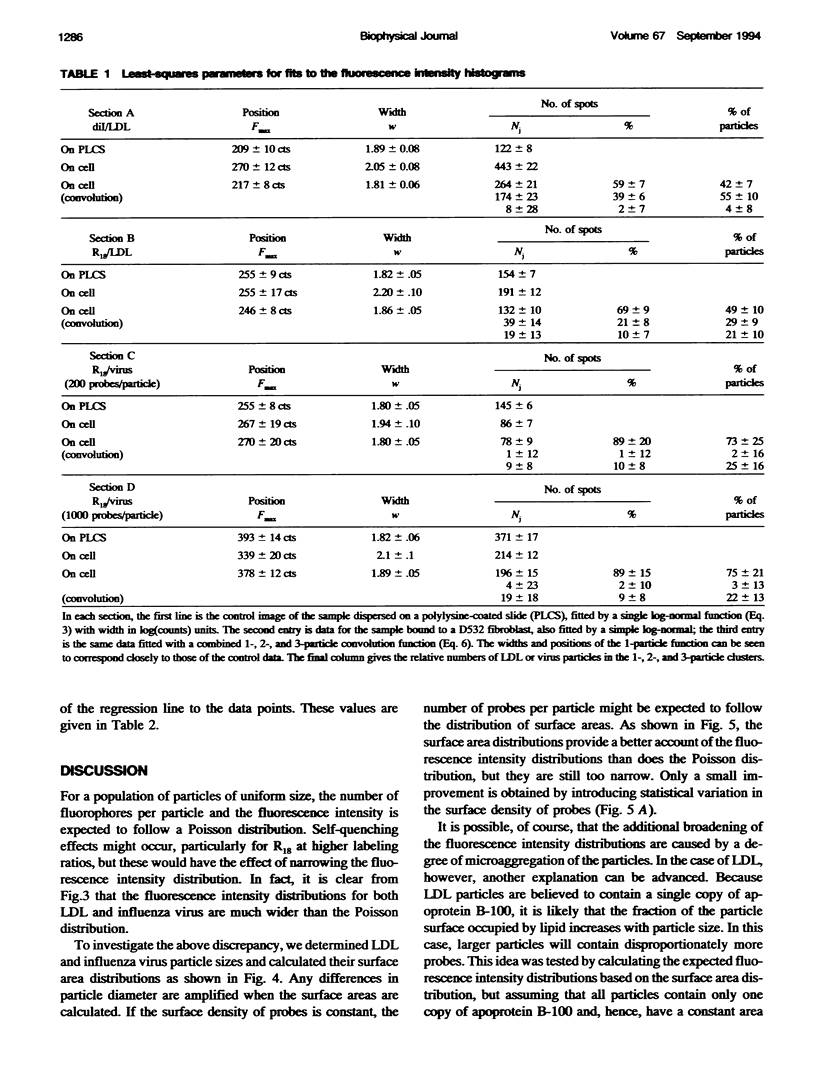
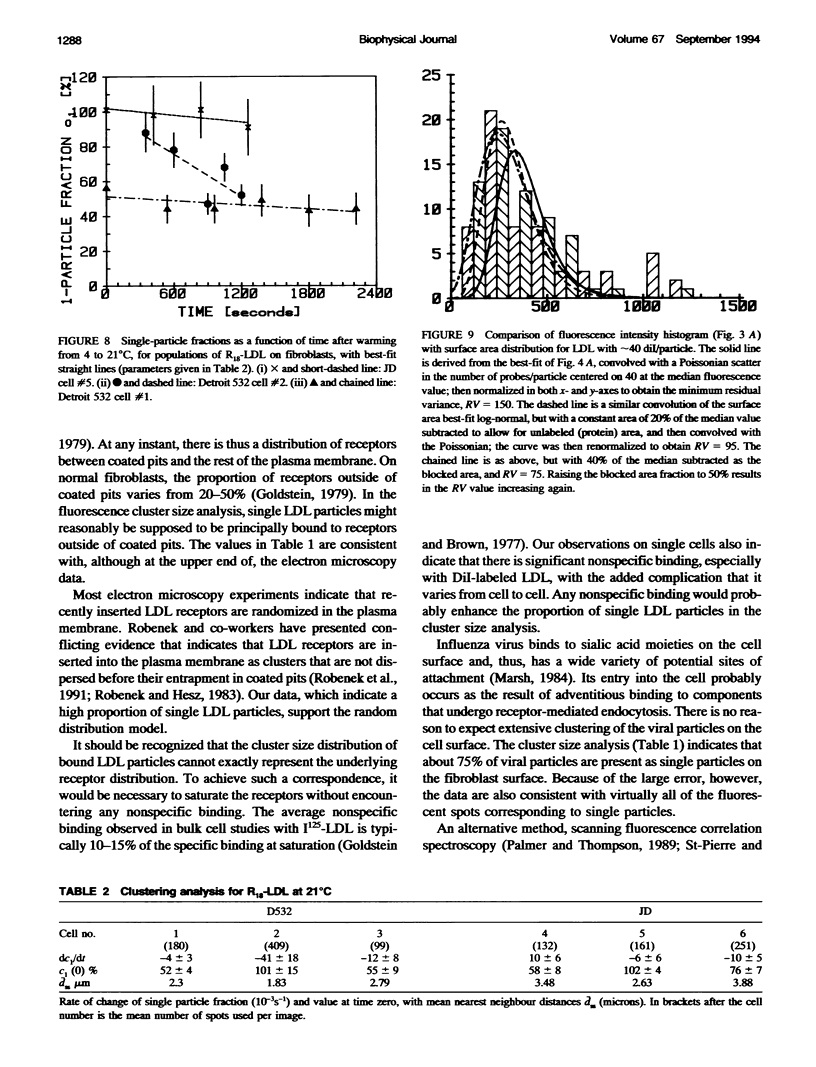
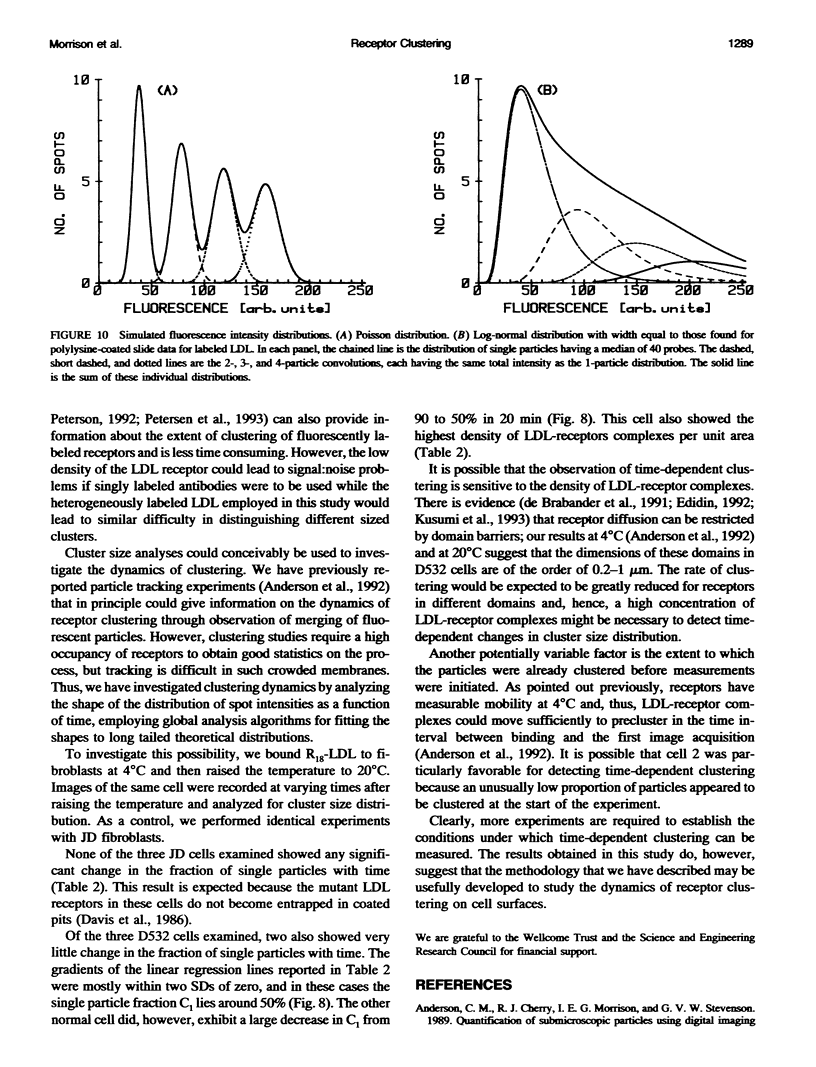
Fluorescently labeled low density lipoproteins (LDL) and influenza virus particles were bound to the surface of human fibroblasts and imaged with a cooled slow-scan CCD camera attached to a fluorescence microscope. Particles were also imaged after attachment to polylysine-coated microscope slides. The digital images were analyzed by fitting data points in the region of fluorescent spots by a two-dimensional Gaussian function, thus obtaining a measure of spot intensity with correction for local background. The intensity distributions for particles bound to polylysine slides were mainly accounted for by particle size distributions as determined by electron microscopy. In the case of LDL, the intensity distributions for particles bound to fibroblasts were considerably broadened, indicative of clustering. The on-cell intensity distributions were deconvolved into 1-particle, 2-particle, 3-particle, etc. components using the data obtained with LDL bound to polylysine-coated slides as an empirical measure of the single particle intensity distribution. This procedure yielded a reasonably accurate measure of the proportion of single particles, but large errors were encountered in the proportions of larger cluster sizes. The possibility of studying the dynamics of clustering was investigated by binding LDL to cells at 4 degrees C and observing changes in the intensity distribution with time after warming to 20 degrees C.
Full text
PDF




Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Anderson C. M., Cherry R. J., Morrison I. E., Stevenson G. V. Quantification of submicroscopic particles using digital imaging fluorescence microscopy: application to low-density lipoprotein receptors. Biochem Soc Trans. 1989 Dec;17(6):1100–1101. doi: 10.1042/bst0171100. [DOI] [PubMed] [Google Scholar]
- Anderson C. M., Georgiou G. N., Morrison I. E., Stevenson G. V., Cherry R. J. Tracking of cell surface receptors by fluorescence digital imaging microscopy using a charge-coupled device camera. Low-density lipoprotein and influenza virus receptor mobility at 4 degrees C. J Cell Sci. 1992 Feb;101(Pt 2):415–425. doi: 10.1242/jcs.101.2.415. [DOI] [PubMed] [Google Scholar]
- Barak L. S., Webb W. W. Fluorescent low density lipoprotein for observation of dynamics of individual receptor complexes on cultured human fibroblasts. J Cell Biol. 1981 Sep;90(3):595–604. doi: 10.1083/jcb.90.3.595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bormann B. J., Engelman D. M. Intramembrane helix-helix association in oligomerization and transmembrane signaling. Annu Rev Biophys Biomol Struct. 1992;21:223–242. doi: 10.1146/annurev.bb.21.060192.001255. [DOI] [PubMed] [Google Scholar]
- Brown M. S., Dana S. E., Goldstein J. L. Regulation of 3-hydroxy-3-methylglutaryl coenzyme A reductase activity in cultured human fibroblasts. Comparison of cells from a normal subject and from a patient with homozygous familial hypercholesterolemia. J Biol Chem. 1974 Feb 10;249(3):789–796. [PubMed] [Google Scholar]
- Carpentier J. L., Gorden P., Goldstein J. L., Anderson R. G., Brown M. S., Orci L. Binding and internalization of 125I-LDL in normal and mutant human fibroblasts. A quantitative autoradiographic study. Exp Cell Res. 1979 Jun;121(1):135–142. doi: 10.1016/0014-4827(79)90453-1. [DOI] [PubMed] [Google Scholar]
- Davis C. G., Lehrman M. A., Russell D. W., Anderson R. G., Brown M. S., Goldstein J. L. The J.D. mutation in familial hypercholesterolemia: amino acid substitution in cytoplasmic domain impedes internalization of LDL receptors. Cell. 1986 Apr 11;45(1):15–24. doi: 10.1016/0092-8674(86)90533-7. [DOI] [PubMed] [Google Scholar]
- Edidin M. Patches, posts and fences: proteins and plasma membrane domains. Trends Cell Biol. 1992 Dec;2(12):376–380. doi: 10.1016/0962-8924(92)90050-w. [DOI] [PubMed] [Google Scholar]
- Goldstein J. L., Anderson R. G., Brown M. S. Coated pits, coated vesicles, and receptor-mediated endocytosis. Nature. 1979 Jun 21;279(5715):679–685. doi: 10.1038/279679a0. [DOI] [PubMed] [Google Scholar]
- Goldstein J. L., Brown M. S. The low-density lipoprotein pathway and its relation to atherosclerosis. Annu Rev Biochem. 1977;46:897–930. doi: 10.1146/annurev.bi.46.070177.004341. [DOI] [PubMed] [Google Scholar]
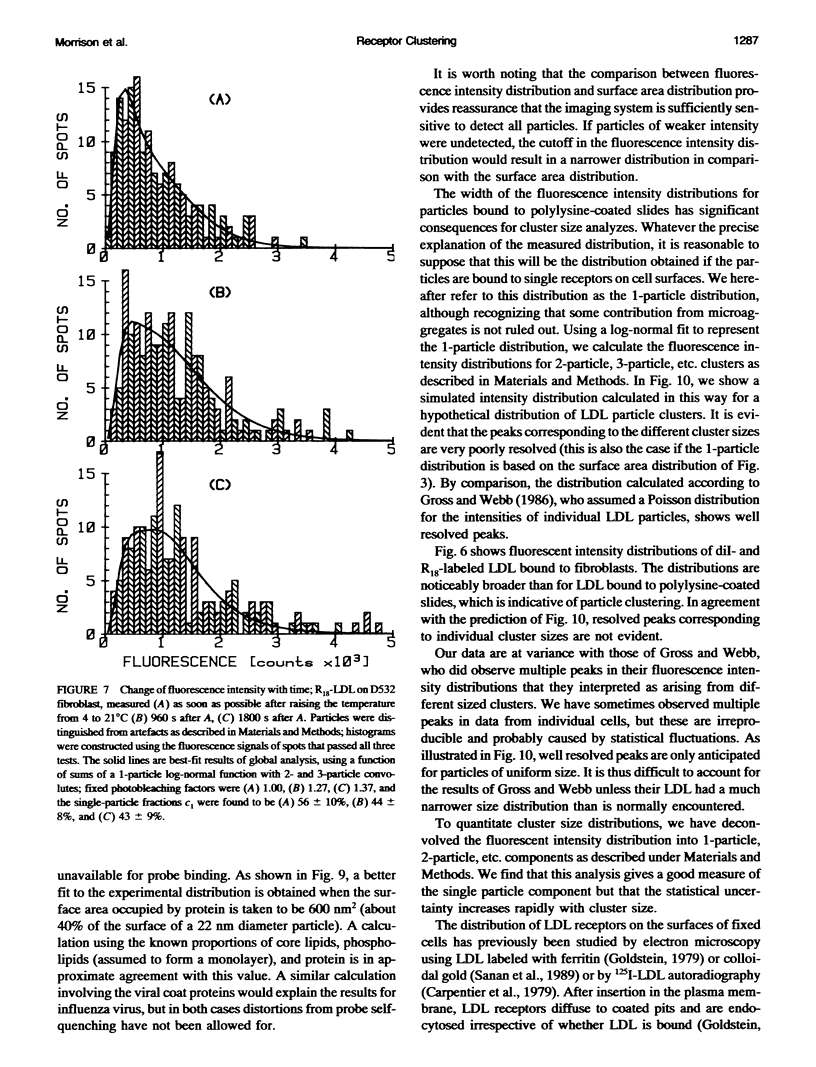
- Gross D., Webb W. W. Molecular counting of low-density lipoprotein particles as individuals and small clusters on cell surfaces. Biophys J. 1986 Apr;49(4):901–911. doi: 10.1016/S0006-3495(86)83718-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- HILLYARD L. A., ENTENMAN C., FEINBERG H., CHAIKOFF I. L. Lipide and protein composition of four fractions accounting for total serum lipoproteins. J Biol Chem. 1955 May;214(1):79–90. [PubMed] [Google Scholar]
- Hatch F. T. Practical methods for plasma lipoprotein analysis. Adv Lipid Res. 1968;6:1–68. [PubMed] [Google Scholar]
- Hiraoka Y., Sedat J. W., Agard D. A. The use of a charge-coupled device for quantitative optical microscopy of biological structures. Science. 1987 Oct 2;238(4823):36–41. doi: 10.1126/science.3116667. [DOI] [PubMed] [Google Scholar]
- Hoekstra D., de Boer T., Klappe K., Wilschut J. Fluorescence method for measuring the kinetics of fusion between biological membranes. Biochemistry. 1984 Nov 20;23(24):5675–5681. doi: 10.1021/bi00319a002. [DOI] [PubMed] [Google Scholar]
- Inoué S. Imaging of unresolved objects, superresolution, and precision of distance measurement with video microscopy. Methods Cell Biol. 1989;30:85–112. doi: 10.1016/s0091-679x(08)60976-0. [DOI] [PubMed] [Google Scholar]
- Kane J. P. Apolipoprotein B: structural and metabolic heterogeneity. Annu Rev Physiol. 1983;45:637–650. doi: 10.1146/annurev.ph.45.030183.003225. [DOI] [PubMed] [Google Scholar]
- Kusumi A., Sako Y., Yamamoto M. Confined lateral diffusion of membrane receptors as studied by single particle tracking (nanovid microscopy). Effects of calcium-induced differentiation in cultured epithelial cells. Biophys J. 1993 Nov;65(5):2021–2040. doi: 10.1016/S0006-3495(93)81253-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Markwell M. A., Haas S. M., Bieber L. L., Tolbert N. E. A modification of the Lowry procedure to simplify protein determination in membrane and lipoprotein samples. Anal Biochem. 1978 Jun 15;87(1):206–210. doi: 10.1016/0003-2697(78)90586-9. [DOI] [PubMed] [Google Scholar]
- Marsh M. The entry of enveloped viruses into cells by endocytosis. Biochem J. 1984 Feb 15;218(1):1–10. doi: 10.1042/bj2180001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Metzger H. Transmembrane signaling: the joy of aggregation. J Immunol. 1992 Sep 1;149(5):1477–1487. [PubMed] [Google Scholar]
- Palmer A. G., 3rd, Thompson N. L. High-order fluorescence fluctuation analysis of model protein clusters. Proc Natl Acad Sci U S A. 1989 Aug;86(16):6148–6152. doi: 10.1073/pnas.86.16.6148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Petersen N. O., Höddelius P. L., Wiseman P. W., Seger O., Magnusson K. E. Quantitation of membrane receptor distributions by image correlation spectroscopy: concept and application. Biophys J. 1993 Sep;65(3):1135–1146. doi: 10.1016/S0006-3495(93)81173-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Robenek H., Harrach B., Severs N. J. Display of low density lipoprotein receptors is clustered, not dispersed, in fibroblast and hepatocyte plasma membranes. Arterioscler Thromb. 1991 Mar-Apr;11(2):261–271. doi: 10.1161/01.atv.11.2.261. [DOI] [PubMed] [Google Scholar]
- Robenek H., Hesz A. Dynamics of low-density lipoprotein receptors in the plasma membrane of cultured human skin fibroblasts as visualized by colloidal gold in conjunction with surface replicas. Eur J Cell Biol. 1983 Sep;31(2):275–282. [PubMed] [Google Scholar]
- Sanan D. A., van der Westhuyzen D. R., Gevers W., Coetzee G. A. Early appearance of dispersed low density lipoprotein receptors on the fibroblast surface during recycling. Eur J Cell Biol. 1989 Apr;48(2):327–336. [PubMed] [Google Scholar]
- St-Pierre P. R., Petersen N. O. Average density and size of microclusters of epidermal growth factor receptors on A431 cells. Biochemistry. 1992 Mar 10;31(9):2459–2463. doi: 10.1021/bi00124a004. [DOI] [PubMed] [Google Scholar]
- Webster R. G., Bean W. J., Gorman O. T., Chambers T. M., Kawaoka Y. Evolution and ecology of influenza A viruses. Microbiol Rev. 1992 Mar;56(1):152–179. doi: 10.1128/mr.56.1.152-179.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Brabander M., Nuydens R., Ishihara A., Holifield B., Jacobson K., Geerts H. Lateral diffusion and retrograde movements of individual cell surface components on single motile cells observed with Nanovid microscopy. J Cell Biol. 1991 Jan;112(1):111–124. doi: 10.1083/jcb.112.1.111. [DOI] [PMC free article] [PubMed] [Google Scholar]


