Abstract
Aronia melanocarpa (Michx.) Elliott is rich in bioactive compounds, but its poor sensory properties limit its acceptance and further utilization. This study examined Lactobacillus plantarum 1243 fermentation impact on fermented A. melanocarpa juice (FAJ)’s quality properties, flavor profile, and metabolites. Bacterial growth peaked then declined, increasing acidity while lowering pH. Total phenolics, flavonoids, and sugars decreased, but DPPH activity remained stable. Sensory scores improved for color and acceptability. Flavor profiling revealed increases in aromatic compounds, benzene derivatives, aliphatic aromatics, and methyl compounds, enriching FAJ's characteristic aroma. Metabolomic analysis identified significant alterations between 0 and 48 h, particularly in organic acids, phenolic acids, and anthocyanins, suggesting improved bioavailability. Increased levels of catechol, flavonoids, and dimethyl fumarate were associated with enhanced bioactivity. These findings indicate that fermentation with L. plantarum 1243 can effectively enhance the flavor, nutritional profile, and functional properties of FAJ.
Keywords: Aronia melanocarpa (Michx.) Elliott, Lactobacillus plantarum 1243, Quality properties, Metabolites, Flavor profile
Graphical abstract
Highlights
-
•
Lactobacillus plantarum 1243 fermentation enhances the nutritional profile of FAJ.
-
•
Differential metabolite shifts reveal key flavor and quality changes.
-
•
pH modulation and sugar utilization drive metabolic diversity in FAJ.
1. Introduction
Aronia melanocarpa (Michx.) Elliott, commonly known as black chokeberry and a member of the Rosaceae family, has garnered global attention as a functional food resource due to its rich phytochemical composition. This deciduous shrub is particularly valued for its exceptional polyphenolic composition, containing proanthocyanidins, anthocyanins, flavonoids, and phenolic acids (Borowska & Brzóska, 2016; Gerasimov et al., 2023). Its pomace contains high concentrations of anthocyanins, typically ranging from 5 to 20 mg/g dry weight, with peak values reported up to 115 mg/g dry weight, which are strongly associated with enhanced antioxidant capacity (Garofulić et al., 2023). Due to its health-promoting properties, A. melanocarpa is utilized in the food industry as an ingredient in functional beverages, syrups, jams, fruit teas, and dietary supplements (Kokotkiewicz et al., 2010).
Among bioprocessing techniques, fermentation with Lactobacillus spp. has proven particularly effective in enhancing the sensory qualities, functional attributes, and bioavailability of food products (Voidarou et al., 2021; Yang, Hao, et al., 2024). Fermented fruit-based beverages have attracted growing interest, as Lactobacillus spp. can enhance the flavor and aroma of juices. For instance, fermentation of purple sweet potato juice with Lactobacillus spp. has been shown to improve sensory characteristics (Wang, Tong, et al., 2024). Additionally, lactic acid fermentation extends shelf life and enhances the nutritional profile of juices (Palencia-Argel et al., 2024). Similarly, fermentation of wolfberry juice with L. plantarum NCU137 increased both free and bound polyphenol content, thereby boosting its antioxidant activity (Liu, He, et al., 2024).
Various Lactobacillus spp., including L. acidophilus, L. plantarum, L. casei, and L. rhamnosus, have been utilized to ferment A. melanocarpa juice (AJ) or its extracts (Frumuzachi et al., 2024). The study demonstrated that while A. melanocarpa extract showed the least growth inhibition against L. plantarum (5 mg/mL) compared to L. rhamnosus, L. paracasei, and L. reuteri. Its incorporation (1–5 %) during oat beverage fermentation with L. plantarum DKK 003 significantly increased phenolic content and antioxidant activity (Marchwińska et al., 2023). Similarly, L. plantarum Pro fermentation of oat drinks with freshly squeezed AJ improved sensory acceptance (Yaneva et al., 2022). Fermentation of AJ with L. plantarum ATCC 14917, subsequently incorporated into freeze-dried yogurt, resulted in improved probiotic viability, antioxidant capacity, total phenolic content, and overall sensory acceptance (Plessas et al., 2024). In another study, L. plantarum EJ2014 fermentation of AJ over nine days significantly increased γ-aminobutyric acid, minerals, polyphenols, and flavonoids (Ali et al., 2021). Additionally, L. paracasei SP5 fermentation enhanced AJ's phenolic content, antioxidant activity, volatile compound profile, and storage stability (Bontsidis et al., 2021). L. plantarum JYLP-375 fermentation improved AJ's flavor profile by reducing aldehydes and other undesirable compounds, as determined by GC-IMS (Wang, Wei, et al., 2024). Furthermore, kefir beverages fortified with AJ demonstrated improved bioavailability in an in vitro model (Du & Myracle, 2018). However, most existing studies have focused on strains or multi-strain fermentation systems, often neglecting detailed analyses of metabolic changes or mechanisms of flavor formation. Few studies have systematically evaluated the physicochemical, sensory, and metabolomic changes of fermented AJ using a well-characterized functional strain. Previous study has shown that L. plantarum 1243 can alleviate frailty in elderly women and enhance motor function (Dong et al., 2024), indicating potential health-promoting properties. Its fermentative performance and influence on AJ's quality attributes and metabolite transformation remain unexplored.
This study aimed to evaluate the fermentative capacity of L. plantarum 1243 in AJ and to investigate the biotransformation of its bioactive constituents. Changes in physicochemical properties, including viable bacterial count, pH, acidity, total sugar content, total phenolic and flavonoid content, antioxidant activity, color difference, sensory attributes, and volatile composition (analyzed via an electronic nose), were assessed throughout fermentation. Ultra-performance liquid chromatography-mass spectrometry was employed to characterize the metabolic profile of fermented A. melanocarpa juice (FAJ), and the potential mechanisms underlying flavor and quality modifications post-inoculation were explored. The study further elucidated the role of L. plantarum 1243 in modulating flavor development and metabolic diversity in FAJ, driven by sugar metabolism and pH dynamics, and provides new insights into its potential as a targeted fermentation strategy to enhance the quality and sensory value of functional berry-based beverages.
2. Material and methods
2.1. Strains, chemicals and reagents
The microbial strain Lactobacillus plantarum 1243, originally isolated from kefir grains at South China Agricultural University (Guangzhou, China), was deposited in the Guangdong Microbial Culture Collection Center (GDMCC No. 62947). The inoculum was prepared at a concentration of 107 CFU/mL. Man-Rogosa-Sharpe (MRS) broth was obtained from Huankai Microbial Sci. & Tech. Co., Ltd. (Guangdong, China). The antioxidant 1,1-diphenyl-2-picrylhydrazyl (DPPH) and rutin standards were purchased from Solarbio Science & Technology Co., Ltd. (Beijing, China), while the gallic acid standard was obtained from Yuanye Bio-Technology Co., Ltd. (Shanghai, China). All other chemicals used in this study were of analytical grade, except for the reagents used for ultra-performance liquid chromatography–tandem mass spectrometry (UPLC–MS/MS), which were of chromatographic grade.
2.2. A. melanocarpa materials and fermentation process
Freshly harvested ripe A. melanocarpa berries were obtained from a local farm in Northeast China, selected based on prior studies highlighting the substantial impact of ripening stage on phenolic compound composition (Dobros et al., 2024). After removing the calyx and leaves, the fruit was immersed in distilled water (1:2 fruit-to-water ratio) and pulped. The resulting juice and pomace were divided into 200 g aliquots to produce AJ with a sugar content of 9 %. The AJ was pasteurized at 80 °C for 15 min, cooled to 26 °C, and inoculated with Lactobacillus plantarum 1243 at a final concentration of 7 lg CFU/mL. The inoculum concentration was selected based on previous studies showing efficient fermentation performance in A. melanocarpa juice systems at this level, while maintaining sensory quality (Wang, Wei, et al., 2024; Wu et al., 2025). Fermentation was carried out at 37 °C for 96 h, with a non-inoculated control incubated under the same conditions. Samples were collected at 0, 24, 48, 72, and 96 h for further analysis. Unfermented juice was used as a control for the same batch of experiments and as a shared control in other studies involving strains.
2.3. Viable cell counts of lactic acid bacteria
Viable cell counts (VCC) during fermentation were determined according to CNIS GB 4789.2–2022, with minor modifications. A 100 μL aliquot of FAJ was diluted in 900 μL saline, followed by serial dilutions from 10−1 to 10−7. The sample was plated on MRS agar supplemented with sodium glutamate and incubated at 37 °C for 24–48 h. Results were expressed as lg CFU/mL.
2.4. Analysis of total sugars, pH, and total acidity
Total sugar content (TSC) was determined using the phenol‑sulfuric acid method (Li et al., 2018), with some modifications. Diluted juice (1.0 mL), 5 % (w/v) phenol solution (1.0 mL), and concentrated sulfuric acid (5.0 mL) were thoroughly mixed, incubated at 25 °C for 10 min, and analyzed at 490 nm using a microplate reader, with a linear standard curve (y = 0.0088x + 0.0815; R2 = 0.9964). TSC was expressed as glucose equivalents (GE) in grams per 100 mL of FAJ (g GE/100 mL). The pH was measured with a digital pH meter (Rex Electric Chemical, PHS-3C), and total acidity (TA) was determined via potentiometric titration using 0.1 M NaOH to neutralize 100 g of sample to pH 8.3, following CNIS GB5009.239–2016.
2.5. Determination of antioxidant parameters
2.5.1. Total phenolic content
Total phenolic content (TPC) in AJ was measured using the Folin-Ciocalteu method with modifications (Škerget et al., 2005). The AJ supernatant was centrifuged and subsequently diluted 20-fold. The reaction mixture containing 0.5 mL water, 0.5 mL diluted sample, and 2.5 mL Folin-Phenol reagent (0.1 mol/L) was incubated for 5 min, followed by the addition of 2 mL Na₂CO₃ (7.5 %, m/v). Absorbance at 765 nm was measured using a microplate reader, and TPC was determined from a standard curve (y = 0.0076x + 0.0684, R2 = 0.9953). The results were expressed as micrograms of gallic acid equivalents (GAE) per milliliter of FAJ (μg GAE/mL).
2.5.2. Total flavonoid content
Total flavonoid content (TFC) in AJ was determined based on a modified aluminum chloride colorimetric method (Zhang et al., 2018). Reagents and samples were diluted to one-tenth of their original volumes. Absorbance was measured at 400 nm using a microplate reader, with rutin as the standard (y = 0.0008× + 0.047, R2 = 0.9884). Results were expressed as micrograms of rutin equivalents (RE) per milliliter of FAJ (μg RE/mL).
2.5.3. DPPH radical scavenging activity
DPPH free radical scavenging activity was evaluated using a DPPH assay kit (Solarbio, Beijing, China), following the manufacturer's instructions. Absorbance was measured at 515 nm using a microplate reader. In the blank control, the sample was replaced with an equal volume of extraction solvent. Antioxidant capacity was quantified based on a vitamin C (VC) standard curve (y = 0.0961x + 1.2348; R2 = 0.9972) and expressed as micrograms of VC per milliliter of sample (μg VC/mL).
2.6. Color quantitative analysis
The color characteristics of FAJ were measured using an ST60 colorimeter (3nh Shenzhen Three NH Technology Co., Ltd., Shenzhen, China). The colorimeter was equipped with a D65 light source, a 10° observer angle, and an 8 mm aperture size. The color indices were defined as L* (brightness on a scale from 0 to 100), a* (greenness-redness on a scale from −100 to 100), and b* (blueness-yellowness on a scale from −100 to 100). Measurements were conducted at a controlled ambient temperature of 25 °C.
2.7. Sensory evaluation
Sensory analysis was carried out according to CNIS GB 7101–2022 standards. Samples were prepared under standardized conditions, including identical vessel type, size, and temperature. The sensory evaluation panel consisted of six evaluators, who evaluated the samples based on color, texture, aroma, taste, and overall acceptability. Assessments were conducted in a well-ventilated room, free from natural light and illuminated with uniform artificial lighting. A 9-point hedonic scale was used to evaluate product preferences, and the mean intensity scores for each sensory attribute were calculated and visualized using a radar plot (Table S1).
2.8. E-nose analysis
The electronic nose (E-nose) PEN3 from AIRSENSE Analytics (Germany) was employed to analyze the aroma profiles of samples collected at 0, 24, 48, 72, and 96 h. Prior to analysis, samples were heated at 80 °C for 15 min to facilitate volatile release. The E-nose was calibrated with an injection flow rate of 400 mL/min, followed by a sequence of 120 s of system cleaning, 10 s of baseline zeroing, 5 s of sample preparation, and 140 s of measurement. Sensor signals within the 134–136 s interval were selected for data analysis. The device is equipped with a 10-sensor MOS array, where specific sensors target various compounds: W1 (CS1) detects benzene derivatives, W5 (SS2) detects nitrogen oxides, W3 (CS3) targets ammonia, W6 (SS4) detects hydrides, and W2 (SS8) responds to alcohols, aldehydes, and ketones. Sensor W5 (CS5) identifies aliphatic aromatics, W1 (SS6) detects methyl compounds, W1 (WS7) targets sulfides, W2 (WS9) detects aromatics and organosulfur compounds, and W3 (SS10) identifies long-chain alkanes. Data analysis was performed using Win-Muster software (Version 1.6.2.22).
2.9. Untargeted metabolite analysis by UPLC-MS/MS
A 50 mg freeze-dried AJ sample was mixed with 1200 μL of 70 % methanol containing an internal standard, vortexed for 15 min, and centrifuged at 12,000 rpm for 3 min at 4 °C. The resulting supernatant was filtered through a 0.22 μm PTFE membrane before LC-MS/MS analysis. A quality control (QC) sample was created by pooling aliquots from each sample. HPLC separation was performed using a Shimadzu LC-30 A system with a Waters ACQUITY Premier HSS T3 C18 column. The mobile phases consisted of water with 0.1 % formic acid (A) and acetonitrile with 0.1 % formic acid (B). The gradient program was as follows: 0–2 min, 5–20 % B; 2–5 min, 20–60 % B; 5–6 min, 60–99 % B; 6–7.5 min, 99 % B; 7.5–7.6 min, 99–5 % B; 7.6–10 min, 5 % B. The flow rate was set to 0.40 mL/min, with a column temperature of 40 °C and an injection volume of 4 μL. Mass spectrometry was conducted using a SCIEX TripleTOF 6600+ system. Typical ion source parameters included: ion spray voltage, +5500/−4000 V; curtain gas, 35 psi; ESI+ source temperature, 550 °C; ESI− source temperature, 450 °C; ion source gas 1, 50 psi; ion source gas 2, 50 psi; declustering potential, ±60 V. Data were converted to mzXML format using ProteoWizard, followed by peak extraction, alignment, and retention time correction with XCMS. Peaks with missing data >50 % were excluded, and missing values were imputed using the KNN method. Peak areas were corrected using the SVR method. Peaks were identified using a laboratory database and public libraries. Substances with an identification score > 0.5 and a coefficient of variation (CV) value of QC < 0.5 were selected for further analysis.
2.10. Statistical analysis
All experiments were conducted in triplicate to ensure data reliability. Quantitative data were processed using Microsoft Excel 2019 and reported as mean values ± standard deviation (SD). Statistical evaluations were conducted employing one-way analysis of variance (ANOVA) with IBM SPSS Statistics 22.0 software, supplemented by post-hoc analyses using the Least Significant Difference (LSD) test for homogeneous variances or Tamhane's T2 test for heterogeneous variances; non-parametric alternatives were implemented when data violated normality assumptions. Graphical representations were generated using OriginPro 2024 and GraphPad Prism 10.0. Untargeted metabolomics profiling was performed using Metware Cloud (https://cloud.metware.cn). Multivariate analytical approaches incorporated unsupervised principal component analysis (PCA) performed in R statistical environment (version 4.2.1), incorporating unit variance scaling for data normalization. Bivariate correlations were quantified through Pearson correlation coefficients (PCC), while metabolic feature clustering was achieved via hierarchical cluster analysis (HCA), both implemented using R base functions and visualized as heatmap matrices. Differential metabolite expression was characterized by fold-change (FC) calculations comparing 48-h versus baseline (0-h) timepoints. Predictive modeling was performed through orthogonal partial least squares-discriminant analysis (OPLS-DA) utilizing the MetaboAnalystR package to derive variable importance in projection (VIP) scores. Metabolite structural identification and functional pathway mapping were conducted through systematic interrogation of the Kyoto Encyclopedia of Genes and Genomes (KEGG) (http://www.kegg.jp/kegg/compound/) and pathway (http://www.kegg.jp/kegg/pathway.html) databases.
3. Results
3.1. Viable counts, total acidity, pH and total sugar
The VCC in FAJ during fermentation are presented in Fig. 1A. Within the first 24 h, the strain exhibited effective adaptation to FAJ, reaching a peak viable count of 7.10 lg CFU/mL. Subsequently, a significant decline in the microbial population was observed, with the viable count decreasing to approximately 6 lg CFU/mL by 48 h of fermentation (P < 0.05). The pH of FAJ decreased overall throughout the fermentation process, from an initial value of 3.75 to 3.58 after 96 h. The initial acidity of FAJ was 29.55 mL/100 g, which significantly decreased to 24.04 mL/100 g after 24 h, followed by a gradual increase (P < 0.05). By the end of fermentation, the TA of FAJ increased to 34.59 mL/100 g (P < 0.05) (Fig. 1C). A significant reduction in the TSC of FAJ, ranging from 9 % to 14 %, was observed between 48 and 96 h of fermentation (P < 0.05), with the lowest value reaching 4.25 g GE/100 mL (Fig. 1B).
Fig. 1.
VCC (A), total sugar (TSC)(B), pH and TA (C) in AJ during fermentation. The different letters indicate statistically significant differences in the results (P < 0.05).
3.2. Total phenolic, Total flavonoid, and DPPH radical scavenging activity
During L. plantarum 1243 fermentation (Fig. 2A and B), FAJ exhibited a progressive decline in both TPC and TFC (P < 0.05). The TPC peaked at 24 h (1394.65 μg GAE/mL), though subsequent fermentation phases showed values significantly lower than initial levels (P < 0.05). In contrast, the TFC of FAJ decreased by 9.6 % at 24 h relative to the initial timepoint. While a slight increase in TFC was observed at 48 h, a continuous decline ensued, resulting in a final TFC concentration of 753.33 μg RE/mL at 96 h, which represented a statistically significant decrease (P < 0.05).
Fig. 2.
Content of total phenol (A), total flavonoid (B), DPPH radical scavenging activity (C) in FAJ. Each data is the mean of three replicate samples. The different letters indicate statistically significant differences in the results (p < 0.05).
The DPPH radical scavenging activity of AJ at 0 h was initially 706.23 μg VC/mL (Fig. 2C), indicating strong antioxidant potential due to its ability to neutralize free radicals. A significant decrease in scavenging activity was observed at 24 and 48 h, with reductions of 10.3 % and 5.9 %, respectively (P < 0.05). Although activity levels improved at 72 and 96 h, they did not significantly differ from those observed at 0 h.
3.3. Color properties and sensory evaluation
The color characteristics of AJ during fermentation with L. plantarum 1243 were evaluated by measuring L*, a*, and b* values. The results indicated that the brightness (L*), redness (a*), and yellowness (b*) of FAJ at 24 h were significantly higher than those at 0 h (P < 0.05) (Fig. 3A–C). However, unlike L* and a*, which remained significantly higher than the 0 h value after 48 h of fermentation (P < 0.05), the b* value, despite being numerically increased, did not show a statistically significant difference from 0 h beyond 48 h. Sensory attributes of FAJ samples fermented with L. plantarum 1243, including hue, texture, flavor, mouthfeel, acidity, sweetness, aftertaste, overall acceptability, and consistency, are presented in Fig. 3D. Fermentation significantly enhanced the hue of FAJ, which was consistent with the color difference analysis. The texture also became more uniform and transparent. The most desirable aroma and aftertaste were observed at 24 h of fermentation, while the highest overall acceptability was recorded at 48 h. Acidity and sweetness were perceived as well-balanced in samples fermented for 24 to 72 h, as acid production during this period improved the sensory appeal of both attributes compared to unfermented samples. Overall, FAJ fermented with L. plantarum 1243 exhibited high sensory acceptance.
Fig. 3.
The color properties and sensory quality of FAJ. (A) The colorimetric values of L* (B) The colorimetric values of a* (C) The colorimetric values of b* (D) The sensory evaluation of AJ during fermentation. Different letters indicated the significant level (p < 0.05).
3.4. E-nose
The E-nose effectively distinguished AJ samples fermented for different durations by utilizing a range of sensors. As shown in Fig. 4A, the response profiles of the 10 sensors varied across fermentation times, with significant changes observed in W1C, W5S, W5C, and W1S. These changes suggest an increased presence of aromatic compounds, including benzene derivatives, nitrogen oxides, aliphatic aromatic compounds, and methyl compounds, which likely contribute to the flavor profile of FAJ. Principal component analysis (PCA) was performed on the E-nose data, with the results presented in Fig. 4B. The first principal component (PC1) accounted for 91.63 % of the variance, while the second principal component (PC2) explained 7.34 %, collectively capturing 98.97 % of the total variance, exceeding the 95 % threshold typically used for dimensionality reduction. These results indicate that the two principal components effectively encapsulate the primary aroma variations associated with different fermentation durations. Notably, FAJ samples fermented for varying durations were clearly separated from the 0-h control along both PC1 and PC2, with no overlapping data points.
Fig. 4.
E-nose data of AJ at different fermentation times. (A) Radar plot (B) PCA plot (C) LDA plot (D) Loading analysis plot.
Linear discriminant analysis (LDA), a classification technique for multivariate data, was employed to enhance the differentiation of fermentation stages. This method optimizes the separation between categories while minimizing within-group variance by projecting high-dimensional data onto a lower-dimensional space (Shidiq Nur et al., 2018). The two primary linear discriminants together accounted for 97.03 % of the total variance, with LD1 and LD2 explaining 74.68 % and 22.35 %, respectively (Fig. 4C). The LDA effectively grouped the 0, 24, 48, and 72-h samples, indicating relatively minor differences in their volatile profiles. Notably, the sample of 24 h and the sample of 72 h exhibited the greatest similarity in aroma. In contrast, the sample of 96 h displayed a distinct aroma profile, setting it apart from the earlier fermentation stages. These findings suggest that both PCA and LDA are effective in distinguishing the volatile organic compound composition of FAJ fermented with L. plantarum 1243, offering valuable insights into the flavor evolution of the juice.
As shown in Fig. 4D, sensor W1S exhibited the greatest displacement along the vertical axis, while W5S demonstrated the most substantial shift along the horizontal axis. These findings suggest that methyl compounds and nitrogen oxides are key contributors to the differentiation of juices fermented for varying durations, consistent with the results observed in the radar chart. Additionally, other volatile compounds, including alcohols, aldehydes, ketones, benzene derivatives, aromatic compounds, sulfides, and aliphatic aromatic hydrocarbons, also played a role in distinguishing different fermentation stages.
3.5. Untargeted metabolomics
The compound composition of AJ and FAJ was analyzed using untargeted metabolomics based on LC-MS. QC is essential for ensuring reliable and high-quality data in mass spectrometry-based metabolomics studies. The PCA score plots for QC samples in both positive and negative ion modes are presented in Fig. S1. The tight clustering of QC samples indicates strong analytical stability, high data quality, and excellent repeatability and reliability of the method.
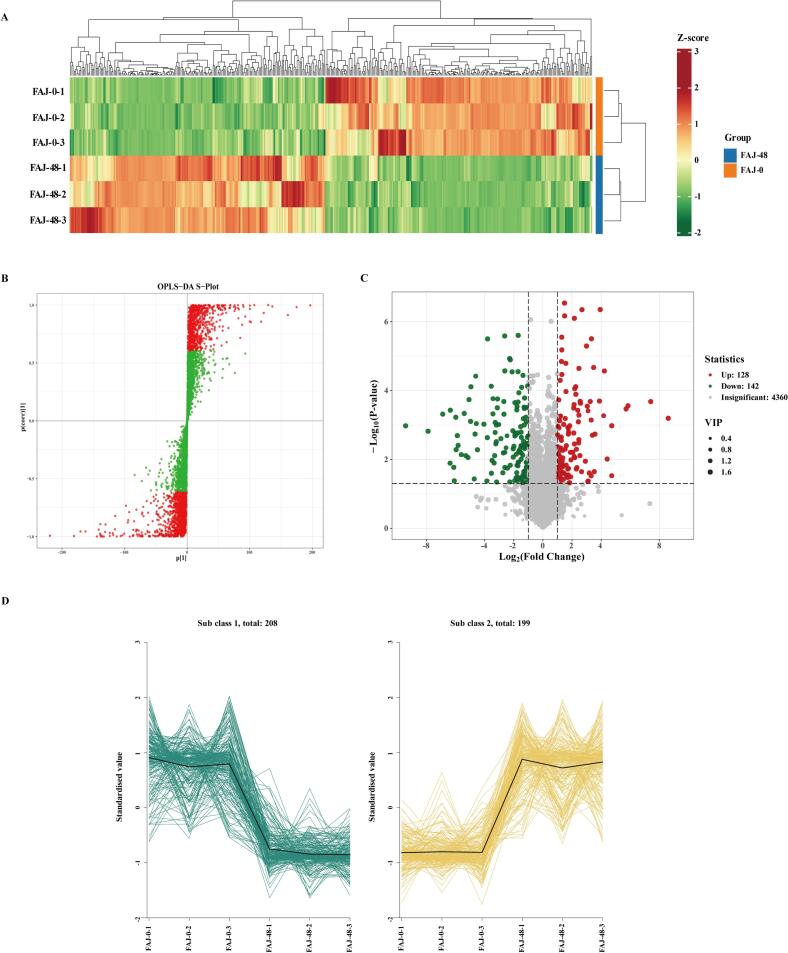
A total of 4630 metabolites were identified across the FAJ-0 (0 h) and FAJ-48 (48 h) groups, with 2849 metabolites detected in positive ion mode and 1781 in negative ion mode. The metabolite composition was predominantly composed of amino acids and their derivatives (24.13 %), organic acids (15.66 %), benzene and substituted derivatives (11.53 %), flavonoids (5.03 %), phenolic acids (4.04 %), and other compounds (Fig. 5A). Heatmap cluster analysis (Fig. 5B and C) revealed distinct differences in metabolite profiles between AJ and FAJ in both ionization modes, as indicated by the clear separation in color patterns.
Fig. 5.
Classification, clustering heatmaps, PCA and OPLS-DA score plots of metabolites. (A) Classification and composition of the 4630 metabolites of the unfermented juice (FAJ-0) and juice fermented for 48 h (FAJ-48). (B) and (C) The heatmap cluster of the FAJ-0 and FAJ-48 in positive and negative ion modes, respectively. The redder the color, the higher the content; and the greener the color, the lower the content. (D) PCA scores of different groups juice of metabolites in two-dimensional graph. (E) Plots of OPLS-DA scores for different groups of metabolites.
3.6. Untargeted metabolomics PCA and OPLS-DA analysis
PCA was performed to evaluate metabolic changes, assess sample repeatability, and visualize group differences. The two principal components, PC1 and PC2, explained 37.52 % and 16.91 % of the total variance, respectively (Fig. 5D). The PCA plot revealed a clear separation between FAJ-0 and FAJ-48, indicating significant differences in their metabolite profiles. To further enhance discrimination, OPLS-DA was applied, optimizing variance separation between the two groups. The high R2Y, R2X, and Q2 values confirmed the robustness of the model, minimizing the risk of overfitting (Fig. S2). As shown in Fig. 5E, FAJ-0 and FAJ-48 samples were distinctly separated, with all FAJ-0 triplicates clustering on the left and FAJ-48 triplicates on the right, further confirming substantial metabolic differences between the two groups.
The OPLS-DA mode was further applied to generate an S-plot (Fig. 6B), highlighting metabolites that contributed significantly to group differentiation. Metabolites positioned in the top-right and bottom-left corners exhibited the strongest influence on separation, with those farther from the origin exerting a more pronounced effect on sample distinction. These metabolites represent potential biomarkers for monitoring fermentation progress.
Fig. 6.
Metabolite OPLS-DA analysis of S plot and differential metabolite clustering heat map, volcano map and K-menas analysis results. (A) The differential metabolites heatmap between FAJ-0 and FAJ-48. (B) S-plot based on OPLS-DA. (C) Differential metabolite volcanic map between FAJ-0 and FAJ-48. (D) K-Means plot of differential metabolites.
3.7. Differential metabolites
To identify differential metabolites between FAJ-0 and FAJ-48, thresholds of fold change (FC ≥ 2 or ≤ 0.5) and variable importance in projection (VIP ≥ 1) were applied. A total of 407 distinct metabolites were identified, including 75 organic acids, 71 amino acids and their derivatives, 55 benzene derivatives, and 22 phenolic acids (Table S2). Heatmap analysis (Fig. 6A) was performed to visualize the relative abundance of these metabolites, with a color gradient from red to green representing high to low concentrations. The heatmap distinctly segregated the samples into two clusters, corresponding to FAJ-0 and FAJ-48, confirming significant differences in metabolite profiles. Additionally, a volcano plot (Fig. 6C) was generated to illustrate metabolite changes, categorizing them as increased, decreased, or not statistically significant. Represented by red, green, and gray dots, respectively, the plot further highlighted significant alterations in metabolite composition after 48 h of fermentation, although with a moderate proportion of changes.
Among the 407 differential metabolites, 199 were significantly increased, while 208 were decreased. Notably, increased metabolites such as N-lactoyl-phenylalanine (FC = 389.71), 2,3,4,9-tetrahydro-1H-carbazole-6‑carbonitrile (FC = 169.28), and 2-hydroxy-3-methylbutanoic acid (FC = 162.26) exhibited substantial increases. In contrast, the concentrations of certain amino acids, including Leu-Asn, phenylalanine, and L-threonine, significantly decreased (FC ≥ 2 or FC ≤ 0.5), whereas derivatives such as N-acetylleucine and N-lactoyl-phenylalanine showed increased levels (Table S2). Among organic acids, 2-hydroxy-3-methylbutyric acid and 3-(3-hydroxyphenyl)propionic acid increased by 162.26-fold and 26.49-fold, respectively, while D-(+)-malic acid, citric acid and pyruvate levels declined. Lactose decreased significantly and L-(+)-fructose increased significantly. Additionally, phenolic acids such as pyrocatechol, 4-hydroxybenzoic acid, 4-hydroxystyrene, hydroxyphenyllactic acid, and dimethyl fumarate, as well as flavonoids including 4′-hydroxyflavanone and 6-hydroxyflavone, were significantly increased. Among anthocyanins, only petunidin-3-(6″-acetylglucoside) exhibited a notable increase. K-means clustering analysis (Fig. 6D) identified two distinct trends in metabolite variation, with 208 metabolites decreasing and 199 increasing, highlighting the significant impact of fermentation on metabolite transformation and biosynthesis.
3.8. The KEGG pathway
Differential metabolites (P < 0.05) between FAJ-0 and FAJ-48 were mapped to the KEGG online database, identifying a total of 68 metabolic pathways that were significantly enriched (Fig. 7A). The top 20 pathways, ranked by P-value from smallest to largest, are presented in Fig. 7B. Additionally, the top 15 increased and decreased metabolic pathways, also ranked by P-value, are shown in Fig. 7C and D. These pathways revealed the metabolic mechanisms underlying the changes in AJ composition, including those related to ascorbate and aldarate metabolism, pentose and glucuronate interconversions, starch and sucrose metabolism, the citrate cycle, phosphonate and phosphinate metabolism, pyruvate metabolism, phenylalanine metabolism, carbon metabolism, and nicotinate and nicotinamide metabolism, among others. Notably, the metabolic pathways of fructose and mannose, propanoate, and the biosynthesis of arginine and histidine were significantly increased (Table S3). Furthermore, fumaric acid, a metabolite involved in the arginine metabolic pathway, exhibited a significant increase following fermentation (Table S2). Significant alterations were observed in the metabolite pathways of amino acids, organic acids, and nucleic acids, particularly the reduction of bitter amino acids such as D-phenylalanine and L-phenylalanine (Table S2). Organic acids, particularly L-lactic acid, played a crucial role in enhancing the flavor profile of sour beverages, with substantial increases observed in fructose and mannose metabolism, propionate metabolism, and pyruvate metabolism (Table S2).
Fig. 7.
The KEGG pathway of FAJ. (A) Classification map of differential metabolite pathways. (B) The top 20 metabolite pathways. The top 15 increased (C) and decreased (D) metabolite pathways. The metabolite pathways were rank by P-value from small to large. The color of the points reflects the size of the P-value, with purple indicating more significant enrichment. The size of the points represents the number of differential metabolites that were enriched. (For interpretation of the references to color in this figure legend, the reader is referred to the web version of this article.)
4. Discussion
The L. plantarum 1243 significantly influenced the physicochemical properties, nutritional value, and flavor of AJ during fermentation, with changes in metabolites strongly correlating with these effects. It demonstrates superior acid resistance compared to L. plantarum JYLP-375, L. acidophilus JYLA-16, and L. rhamnosus JYLR-005, and it can grow at a lower pH than reported in previous FAJ studies (pH = 5.65) (Wang, Wei, et al., 2024). Previous research has indicated that the viability of probiotics in fermented juices is influenced by factors such as the initial pH of the juice and the final pH of the fermented product (Muhialdin et al., 2021). The lower pH likely facilitated probiotic growth, as acidic environments can promote the synthesis of beneficial compounds, such as γ-aminobutyric acid, and enhance antimicrobial activity (Pan et al., 2022). The food industry typically adheres to the recommended probiotic fermentation threshold of 6 lg CFU/mL at the point of consumption (Sheehan et al., 2007). The growth of L. plantarum 1243 within 48 h underscores the market potential of FAJ.
TA serves as a key indicator of metabolic changes occurring in microorganisms during fermentation, providing valuable insights into the fermentation process mediated by L. plantarum. Typically, L. plantarum converts malic acid to lactic acid, which lowers the pH and increases acidity (Wu et al., 2022). However, during the early stages of fermentation, a decrease in TA may occur due to insufficient lactic acid production. These observations are consistent with a previous study, which reported a general decrease in acidity after 12 h of fermentation, followed by an increase, likely due to the decrease of other acids such as malic acid and citric acid at the beginning of fermentation (Wu et al., 2020). As a facultative hetero-fermentative bacterium, L. plantarum utilizes glucose and sucrose to produce lactic acid, acetic acid, and CO₂, depending on the metabolic pathway (Laosee et al., 2022). In this study, L. plantarum consumed 14 % of the sugar, significantly reducing the TSC, which is consistent with previous findings on FAJ (Markkinen et al., 2019).
Previous studies have shown that juice fermented by L. paracasei or L. plantarum exhibits increased antioxidant activity, TPC, and TFC (Ali et al., 2021; Bontsidis et al., 2021). However, this study presents contrasting results, revealing a decrease in both TPC and TFC in AJ. Lactic acid bacteria, such as L. plantarum, are known to degrade phenolic compounds through their enzymatic activities, leading to the breakdown of these compounds (Ankolekar et al., 2012). It has been established that fermentation facilitates the transformation of specific phenolic compounds, including hydroxycinnamic acid and flavonol glycosides, into smaller, more soluble metabolites in AJ (Markkinen et al., 2019). Notably, L. plantarum has been shown to enhance the bioavailability of certain phenolic compounds by promoting their transformation (Landete et al., 2021). In line with this, the findings of this study suggest that L. plantarum 1243 may facilitate the bioavailability of FAJ through similar mechanisms. Similar findings have been reported in other studies (Wu et al., 2020). Importantly, a decrease in TPC and TFC does not necessarily indicate a loss of antioxidant activity. DPPH scavenging assays, commonly used to assess antioxidant activity, showed an initial decrease followed by an increase after 72 h in fruit juices, with the decline likely due to the oxidation or degradation of antioxidant molecules (Chen et al., 2023; Sun et al., 2022). Consequently, while the total levels of TPC and TFC may decrease, their bioavailability could increase, thereby maintaining antioxidant activity. These findings suggest that L. plantarum 1243 may possess distinct properties related to compound structural alteration and enhanced uptake efficiency during fermentation.
In this study, the increase in the a* value of FAJ during fermentation can be attributed to the lowered pH resulting from lactic acid fermentation, which promotes positive ionization and enhances the red hue of the substrate (Sayekti et al., 2017). The observed rise in the b* value is likely due to carotenoid isomerization, which intensifies the yellow coloration. Additionally, the increase in the L* value aligns with findings from other fruit juices, where anthocyanin aggregation leads to noticeable color changes (Ana Lúcia Fernandes et al., 2011). Overall, fermentation with L. plantarum 1243 enhances the visual appeal of FAJ, imparting a vibrant purple-red color.
The flavor profile of lactic acid bacteria-fermented plant-based beverages is influenced by organic acids, amino acids, sugars, and volatile compounds (Yang, Hong, et al., 2024). E-nose analysis revealed an increase in aromatic compounds, consistent with previous findings from FAJ analysis using GC-IMS (Wang, Wei, et al., 2024), indicating that the aromatic compounds produced by L. plantarum 1243 during fermentation contribute to the enhanced flavor profile of the juice. Additionally, prior studies have reported an increase in volatile methyl compounds in FAJ (Bontsidis et al., 2021), further suggesting that L. plantarum 1243 plays a significant role in shaping the juice's aromatic characteristics. The increase in benzene derivatives during fermentation contributes to an enhanced aroma and prolonged flavor in the juice (Liao et al., 2022; Zhao et al., 2014). Interestingly, at 96 h, despite the most pronounced increase in responses from aromatic and methyl compound sensors, the sensory evaluation of aroma did not yield the highest score. This discrepancy may arise from the limitations of the sensors, which do not fully replicate the human olfactory system and may lack the sensitivity and selectivity required to accurately reflect overall sensory perception. This discrepancy may arise from the fact that sensors do not fully replicate the human olfactory system, as they often lack sensitivity and selectivity and may not accurately reflect the overall sensory perception of food (Jung et al., 2017; Smyth & Cozzolino, 2013). However, sensory evaluation provides a holistic perception of fragrance, which may reach an optimal balance at 24 h, despite relatively low individual sensor response values at that time.
During the early stage of fermentation, 9 % sugar was added to enhance the sweetness of the juice, thereby counteracting the inherent bitterness and sourness associated with high procyanidin levels in AJ (Bontsidis et al., 2021). Throughout the fermentation process, glucose degradation and amino acid catabolism contributed to the sweetness of the juice (Wu et al., 2022). As a result, the acidity, sweetness, and overall sensory appeal of FAJ improved, particularly after 48 h. While previous studies have primarily focused on the changes in chemical composition and antioxidant activity of AJ during fermentation, this study extends the analysis by evaluating flavor enhancement following fermentation with L. plantarum 1243 through sensory analysis. The results indicate that L. plantarum 1243 significantly improved the taste and overall flavor of the juice, primarily by reducing acidity and bitterness, likely due to lactic acid production and the formation of other metabolites. Notably, the increased levels of N-lactoyl-phenylalanine and N-acetylleucine, along with a reduction in bitter amino acids, suggest that fermentation converts bitter compounds into kokumi-flavored derivatives (Xie et al., 2023). Moreover, sensory evaluation revealed that the mouthfeel and overall acceptability of the juice after 48 h of fermentation were rated higher than those of the unfermented juice. The breakdown of amino acids also contributed to an increase in volatile compounds, further enhancing both taste and aroma (Peng et al., 2021). Additionally, 13 ketones, including 2-nonone, were increased, further influencing the juice's aroma profile (Duan et al., 2024; Fan et al., 2024). Although no significant statistical correlation was observed between key differential metabolites and sensory attributes in this study (Fig. S3), the overall enhancement in taste and acceptability after fermentation aligns with the accumulation of known flavor-contributing metabolites, such as N-lactoyl-phenylalanine and ketones.
Significant correlations were found between key differential metabolites and physicochemical parameters such as total sugar, pH, TA, and DPPH (Fig. S3). These findings are consistent with the consumption of sugars, acids, and phenolic compounds by L. plantarum 1243during fermentation, contributing to enhanced metabolic diversity. Significantly increased metabolites such as L-lactic acid, L-(+)-fructose, and 4-hydroxystyrene were significantly enriched in KEGG pathways, including pyruvate metabolism, fructose and mannose metabolism, and phenylpropanoid biosynthesis (Table S2). These pathways are associated with carbohydrate degradation, organic acid accumulation, and the transformation of phenolic compounds, which collectively support the observed changes in physicochemical properties.
Further differential metabolite analysis revealed the increase of 34 organic acids, likely resulting from the consumption of sugars by lactic acid bacteria during fermentation (Table S2). KEGG pathway enrichment analysis of significantly differential metabolites pinpointed key functional shifts driven by fermentation. Notably, the significant increase in L-lactic acid, a primary end-product of homolactic fermentation by L. plantarum, was enriched in glycolysis/gluconeogenesis and pyruvate metabolism pathways. In contrast, D-(+)-malic acid and citric acid decreased significantly after 48 h of fermentation, corresponding to reduced activity in the citrate cycle (Table S3). This transition is associated with the shift from stronger sour-tasting malic acid to the milder acidity of lactic acid, thereby reducing perceived sourness (Wei et al., 2018). Conversely, significantly decreased metabolites such as D-(+)-malic acid, citric acid, and phenylalanine were associated with decreased abundance in citrate cycle and phenylpropanoid biosynthesis pathways. This shift may indicate redirection of metabolic flow toward the synthesis of more bioactive or flavor-relevant intermediates, contributing to a more favorable sweet-sour balance. Modulating the balance between sweetness and sourness is crucial for enhancing the palatability of AJ, and sensory evaluation confirmed that the sweet and sour scores at 48 h were significantly higher than those at 0 h. These metabolites were enriched in KEGG pathways including fructose and mannose metabolism, pyruvate metabolism, and phenylpropanoid biosynthesis, highlighting their roles in acid production and phenolic compound conversion during fermentation.
Among the increased metabolites, lactic acid, 4-hydroxybenzoic acid, and hydroxyphenyllactic acid showed a negative correlation with DPPH scavenging activity (Fig. S3), suggesting a decrease in antioxidant capacity but enhanced metabolic transformation activity. Lactic acid is a key product of fructose, mannose, and pyruvate metabolism, and the increase of these metabolic pathways suggests active carbohydrate utilization by L. plantarum during fermentation (Zhang et al., 2024). Organic acids play a crucial role in the release of bound bioactive compounds (Khan et al., 2020). Specifically, lactic acid and other organic acids contribute to the conversion of bound mineral elements into free forms, thereby enhancing their bioaccessibility (Gong et al., 2024). The significant increase in fumarate (enriched in citrate cycle) may also contribute to this acid-mediated solubilization effect.
In contrast to previous reports of anthocyanin degradation during fermentation, no significant decrease in anthocyanin content was observed in this study (Du & Myracle, 2018). The lower pH conditions may have effectively inhibited oxidase activity, thereby reducing anthocyanin degradation (Cano-Lamadrid et al., 2017). Notably, petunia 3-(6″-acetylglucoside) was significantly increased. Its acetylated structure may enhance lipophilicity and facilitate membrane transport, thereby improving anthocyanin bioavailability (Liu et al., 2022; Yang et al., 2019).
Fermentation also led to the synthesis of bioactive phenolic acids, such as 4-hydroxybenzoic acid and hydroxyphenyllactic acid, which increased during the biosynthetic process. The conversion of phenolic compounds into smaller, more metabolically accessible molecules enhances their transformation within the gut microbiome, potentially amplifying their physiological effects (Tang et al., 2023). Additionally, a significant increase in the biosynthesis of arginine was observed. KEGG enrichment analysis revealed significant modulation of amino acid metabolic pathways, including the downregulation of L-phenylalanine in the phenylalanine metabolism pathway. An up-regulation of histidine metabolism was also noted. This reflects a broader reprogramming of amino acid metabolism during fermentation, potentially favoring the synthesis of functional amino acids such as arginine. As L-arginine oxidation by nitric oxide synthase is a primary pathway for nitric oxide production, the increased synthesis of arginine during fermentation may improve its bioavailability, contributing to potential health benefits (Hu et al., 2024). Amino acid metabolism plays a crucial role in these biochemical changes (Xue et al., 2024). The promotion of arginine synthesis helps to reduce liver inflammation (Kim et al., 2023).
Among the differentially metabolized phenolic acids, the majority showed increased levels, with pyrocatechol exhibiting a particularly significant increase. Pyrocatechol is known for its anti-inflammatory and neuroprotective properties (Murata et al., 2024). Additionally, dimethyl fumarate, which has been shown to reduce neuroinflammation and oxidative stress, has proven effective in delaying neurodegeneration (Scuderi et al., 2020). Notably, 4′-hydroxyflavanone and 6-hydroxyflavone, two significantly increased flavonoids, possess strong antioxidant activity, further enhancing the nutritional value of the juice (Guerrero-Rubio et al., 2021). These flavonoids may improve lipid metabolism, boost antioxidant defenses, and modulate gut microbiota (Liu, Ling, et al., 2024). The increase in key bioactive compounds, such as phenolic acids and flavonoids, further elevated the nutritional value of the fermented juice, contributing to its health benefits. In particular, antioxidant-related physicochemical parameters such as DPPH, TPC, and TFC showed negative correlations with several upregulated metabolites including fumarate and 4-hydroxybenzoic acid. These compounds are involved in phenylpropanoid biosynthesis and pyruvate metabolism pathways, suggesting that fermentation not only alters the redox-active compound profile but also promotes the transformation or release of bioavailable phenolic acids, thereby contributing to the antioxidant potential of the fermented juice. Despite the net reduction in total phenolic content, the increased production of specific, bioactive phenolic metabolites highlights a qualitative enhancement of antioxidant functionality, which represents a key outcome of L. plantarum 1243 fermentation.
5. Conclusion
Fermentation with L. plantarum 1243 significantly altered the physicochemical properties, biological activities, and metabolite profile of AJ. During fermentation, the VCC of lactic acid bacteria peaked at 7.10 lg CFU/mL before declining, while the pH decreased and acidity increased. The levels of TSC, TPC, and TFC declined significantly. Despite these changes, the FAJ maintained stable DPPH radical scavenging activity, ranging from 633.07 μg VC/mL to 706.23 μg VC/mL. FAJ exhibited a visually appealing purplish-red hue, with a transparent and homogeneous appearance, and a well-balanced sweet-sour taste, achieving high scores in sensory evaluations. The concentrations of aromatic compounds, benzene derivatives, aliphatic aromatic compounds, and methyl compounds significantly increased during fermentation. Untargeted metabolomic analysis revealed substantial shifts in the metabolite profile of FAJ between 0 and 48 h of fermentation, particularly in organic acids and their derivatives, amino acids and their derivatives, benzene derivatives, and phenolic compounds. Key metabolic pathways, including fructose and mannose metabolism, propionate biosynthesis, and arginine and histidine biosynthesis, were notably increased. Furthermore, metabolomic findings suggest that changes in specific amino acids, organic acids, and ketones may contribute to flavor enhancement. Pathway enrichment analysis indicated enhanced activity in fructose and mannose metabolism, pyruvate metabolism, and amino acid biosynthesis (e.g., arginine and histidine). These pathways were closely associated with flavor modulation, such as sourness reduction and kokumi flavor development, and also with enhanced bioactivity. The metabolic transformations of organic acids, anthocyanins, phenolic acids, and other bioactive compounds indicate an improvement in the bioavailability of FAJ during fermentation. Notably, the increase in dimethyl fumarate, pyrocatechol, and 6-hydroxyflavone, among other metabolites, suggests that FAJ may offer potential health benefits, including antioxidant activity, anti-inflammatory effects, and neuroprotection. These findings provide a theoretical basis for the value-added potential of FAJ.
CRediT authorship contribution statement
Minjun Liu: Writing – original draft, Formal analysis, Data curation. Yingyan Fang: Software, Methodology. Ruihan Chen: Project administration, Methodology. Minyu Cai: Validation, Software. Xiaoping Yang: Supervision, Resources. Ziying Fang: Resources, Project administration, Investigation, Conceptualization. Xiang Fang: Validation, Funding acquisition, Conceptualization. Sashuang Dong: Writing – review & editing, Conceptualization.
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Acknowledgements
This work was supported by the Guangzhou Science and Technology Program (2024B03J1354), Guangdong Provincial Seed Industry Revitalization Special Program (2022-WPY-00-008), South China Agricultural University 2024 College Students' Innovation and Entrepreneurship Training Program (S202310564098).
Footnotes
Supplementary data to this article can be found online at https://doi.org/10.1016/j.fochx.2025.102706.
Contributor Information
Xiang Fang, Email: fxiang@scau.edu.cn.
Sashuang Dong, Email: dsashuang@scau.edu.cn.
Appendix A. Supplementary data
PCA score plots for QC samples in the positive ion mode (A) and negative ion mode (B). Supplementary Fig. 2. Validation diagram of the OPLS-DA model. Supplementary Fig. 3. Correlation matrix analysis between key differential metabolites and physicochemical properties illustrated as heatmap. Red represents a positive correlation and blue represents a negative correlation. Significance levels are indicated by asterisks: ***P < 0.001; **P < 0.01; *P < 0.05.
Scoring criteria for FAJ. Supplementary Table 2. Differential metabolites between FAJ of 0 h and 48 h. Supplementary Table 3. Metabolic pathway enrichment analysis: 68 pathways mapped using the KEGG database for 48 h vs 0 h.
Data availability
No data was used for the research described in the article.
References
- Ali M.S., Lee E.-B., Lee S.-J., Lee S.-P., Boby N., Suk K.…Park S.-C. Aronia melanocarpa extract fermented by Lactobacillus plantarum EJ2014 modulates immune response in mice. Antioxidants. 2021;10(8) doi: 10.3390/antiox10081276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ana Lúcia Fernandes P., Tatiane Cavalcante M., Sueli R. Probiotic beverage from cashew apple juice fermented with Lactobacillus casei. Food Research International. 2011;44:1276–1283. doi: 10.1016/j.foodres.2010.11.035. [DOI] [Google Scholar]
- Ankolekar C., Johnson K., Pinto M., Johnson D., Labbe R.G., Greene D., Shetty K. Fermentation of whole apple juice using lactobacillus acidophilus for potential dietary management of hyperglycemia, hypertension, and modulation of beneficial bacterial responses. Journal of Food Biochemistry. 2012;36(6):718–738. doi: 10.1111/j.1745-4514.2011.00596.x. [DOI] [Google Scholar]
- Bontsidis C., Mallouchos A., Terpou A., Nikolaou A., Batra G., Mantzourani I.…Plessas S. Microbiological and chemical properties of chokeberry juice fermented by novel lactic acid Bacteria with potential probiotic properties during fermentation at 4 °C for 4 weeks. Foods. 2021;10(4) doi: 10.3390/foods10040768. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Borowska S., Brzóska M.M. Chokeberries (Aronia melanocarpa) and their products as a possible means for the prevention and treatment of noncommunicable diseases and unfavorable health effects due to exposure to xenobiotics. Comprehensive Reviews in Food Science and Food Safety. 2016;15(6):982–1017. doi: 10.1111/1541-4337.12221. [DOI] [PubMed] [Google Scholar]
- Cano-Lamadrid M., Trigueros L., Wojdyło A., Carbonell-Barrachina A.Á., Sendra E. Anthocyanins decay in pomegranate enriched fermented milks as a function of bacterial strain and processing conditions. LWT- Food Science and Technology. 2017;80:193–199. doi: 10.1016/j.lwt.2017.02.023. [DOI] [Google Scholar]
- Chen W., Xie C., He Q., Sun J., Bai W. Improvement in color expression and antioxidant activity of strawberry juice fermented with lactic acid bacteria: A phenolic-based research. Food Chemistry: X. 2023;17 doi: 10.1016/j.fochx.2022.100535. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dobros N., Zielińska A., Siudem P., Zawada K.D., Paradowska K. Profile of bioactive components and antioxidant activity of Aronia melanocarpa fruits at various stages of their growth, using Chemometric methods. Antioxidants. 2024;13(4) doi: 10.3390/antiox13040462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dong S., Zeng Q., He W., Cheng W., Zhang L., Zhong R., He W., Fang X., Wei H. Effect of Lactobacillus plantarum BFS1243 on a female frailty model induced by fecal microbiota transplantation in germ-free mice. Food & Function. 2024;15(8) doi: 10.1039/d3fo05282f. [DOI] [PubMed] [Google Scholar]
- Du X., Myracle A.D. Fermentation alters the bioaccessible phenolic compounds and increases the alpha-glucosidase inhibitory effects of aronia juice in a dairy matrix following in vitro digestion. Food & Function. 2018;9(5):2998–3007. doi: 10.1039/c8fo00250a. [DOI] [PubMed] [Google Scholar]
- Duan F., Luo Y., Li D., Zhong D., He G., Wei Z., Jia L. Kaili red sour soup: Correlations in composition/microbial metabolism and flavor profile during post-fermentation. Food Chemistry. 2024;435 doi: 10.1016/j.foodchem.2023.137602. [DOI] [PubMed] [Google Scholar]
- Fan X., Liu M., Shi Z., Zhang T., Du L., Wu Z., Zeng X., Wu X., Pan D. Binary probiotic fermentation promotes signal (cyclic AMP) exchange to increases the number of viable probiotics, anthocyanins and polyphenol content, and the odor scores of wolfberry fermented beverages. Food Chemistry. 2024;448 doi: 10.1016/j.foodchem.2024.139085. [DOI] [PubMed] [Google Scholar]
- Frumuzachi O., Rohn S., Mocan A. Fermented black chokeberry (Aronia melanocarpa (Michx.) Elliott) products - a systematic review on the composition and current scientific evidence of possible health benefits. Food Research International. 2024;196 doi: 10.1016/j.foodres.2024.115094. [DOI] [PubMed] [Google Scholar]
- Garofulić I.E., Repajić M., Zorić Z., Jurendić T., Dragović-Uzelac V. Evaluation of microwave- and ultrasound-assisted extraction techniques for revalorization of black chokeberry (Aronia melanocarpa) fruit pomace anthocyanins. Sustainability. 2023;15(9) doi: 10.3390/su15097047. [DOI] [Google Scholar]
- Gerasimov M.A., Perova I.B., Eller K.I., Akimov M.Y., Sukhanova A.M., Rodionova G.M., Ramenskaya G.V. Investigation of polyphenolic compounds in different varieties of black chokeberry Aronia melanocarpa. Molecules. 2023;28(10) doi: 10.3390/molecules28104101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gong S., Zong A., Wu J., Xu T., Wang Z. Effect of fermentation on color characteristics and metabolite profiles of purple potato flour. Journal of Food Composition and Analysis. 2024;133 doi: 10.1016/j.jfca.2024.106431. [DOI] [Google Scholar]
- Guerrero-Rubio M.A., Hernández-García S., García-Carmona F., Gandia-Herrero F. Flavonoids’ effects on Caenorhabditis elegans’ longevity, fat accumulation, stress resistance and gene modulation involve mTOR, SKN-1 and DAF-16. Antioxidants. 2021;10(3) doi: 10.3390/antiox10030438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hu Y., Xing Y., Fan G., Xie H., Zhao Q., Liu L. L-arginine combination with 5-fluorouracil inhibit hepatocellular carcinoma cells through suppressing iNOS/NO/AKT-mediated glycolysis. Frontiers in Pharmacology. 2024;15 doi: 10.3389/fphar.2024.1391636. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jung H.Y., Kwak H.S., Kim M.J., Kim Y., Kim K.-O., Kim S.S. Comparison of a descriptive analysis and instrumental measurements (electronic nose and electronic tongue) for the sensory profiling of Korean fermented soybean paste (doenjang) Journal of Sensory Studies. 2017;32(5) doi: 10.1111/joss.12282. [DOI] [Google Scholar]
- Khan S.A., Zhang M., Liu L., Dong L., Ma Y., Wei Z.…Zhang R. Co-culture submerged fermentation by lactobacillus and yeast more effectively improved the profiles and bioaccessibility of phenolics in extruded brown rice than single-culture fermentation. Food Chemistry. 2020;326 doi: 10.1016/j.foodchem.2020.126985. [DOI] [PubMed] [Google Scholar]
- Kim D.Y., Park J.Y., Gee H.Y. Lactobacillus plantarum ameliorates NASH-related inflammation by upregulating l-arginine production. Experimental and Molecular Medicine. 2023;55(11):2332–2345. doi: 10.1038/s12276-023-01102-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kokotkiewicz A., Jaremicz Z., Luczkiewicz M. Aronia plants: A review of traditional use, biological activities, and perspectives for modern medicine. Journal of Medicinal Food. 2010;13(2):255–269. doi: 10.1089/jmf.2009.0062. [DOI] [PubMed] [Google Scholar]
- Landete J.M., Plaza-Vinuesa L., Montenegro C., Santamaría L., Reverón I., De Las Rivas B., Muñoz R. The use of Lactobacillus plantarum esterase genes: A biotechnological strategy to increase the bioavailability of dietary phenolic compounds in lactic acid bacteria. International Journal of Food Sciences and Nutrition. 2021;72(8):1035–1045. doi: 10.1080/09637486.2021.1900078. [DOI] [PubMed] [Google Scholar]
- Laosee W., Kantachote D., Chansuwan W., Sirinupong N. Effects of probiotic fermented fruit juice- based biotransformation by lactic acid Bacteria and Saccharomyces boulardii CNCM I-745 on anti-Salmonella and Antioxidative properties. Journal of Microbiology and Biotechnology. 2022;32(10):1315–1324. doi: 10.4014/jmb.2206.06012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y., Mehta R., Messing J. A new high-throughput assay for determining soluble sugar in sorghum internode-extracted juice. Planta. 2018;248(4):785–793. doi: 10.1007/s00425-018-2932-8. [DOI] [PubMed] [Google Scholar]
- Liao J., Zhang S., Zhang X. Effects of mixed adding crude extracts of β-glucosidases from three different non-Saccharomyces yeast strains on the quality of cabernet sauvignon wines. Journal of Fungi. 2022;8(7) doi: 10.3390/jof8070710. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu S., He Y., He W., Song X., Peng Y., Hu X.…Xie M. Exploring the biogenic transformation mechanism of polyphenols by Lactobacillus plantarum NCU137 fermentation and its enhancement of antioxidant properties in wolfberry juice. Journal of Agricultural and Food Chemistry. 2024;72(22):12752–12761. doi: 10.1021/acs.jafc.4c01393. [DOI] [PubMed] [Google Scholar]
- Liu Y., Lin J., Cheng T., Liu Y., Han F. Methylation, hydroxylation, glycosylation and acylation affect the transport of wine anthocyanins in Caco-2 cells. Foods. 2022;11(23) doi: 10.3390/foods11233793. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y.-F., Ling N., Zhang B., Chen C., Mo X.-N., Cai J.-Y.…Yu Q.-M. Flavonoid-rich mulberry leaf extract modulate lipid metabolism, antioxidant capacity, and gut microbiota in high-fat diet-induced obesity: Potential roles of FGF21 and SOCS2. Food & Medicine Homology. 2024;1(2) doi: 10.26599/FMH.2024.9420016. [DOI] [Google Scholar]
- Marchwińska K., Gwiazdowska D., Juś K., Gluzińska P., Gwiazdowska J., Pawlak-Lemańska K. Innovative functional lactic acid Bacteria fermented oat beverages with the addition of fruit extracts and Lyophilisates. Applied Sciences-Basel. 2023;13(23) doi: 10.3390/app132312707. [DOI] [Google Scholar]
- Markkinen N., Laaksonen O., Nahku R., Kuldjärv R., Yang B. Impact of lactic acid fermentation on acids, sugars, and phenolic compounds in black chokeberry and sea buckthorn juices. Food Chemistry. 2019;286:204–215. doi: 10.1016/j.foodchem.2019.01.189. [DOI] [PubMed] [Google Scholar]
- Muhialdin B.J., Hussin A.S.M., Kadum H., Hamid A.A., Jaafar A.H. Metabolomic changes and biological activities during the lacto-fermentation of jackfruit juice using Lactobacillus casei ATCC334. LWT- Food Science and Technology. 2021;141 doi: 10.1016/j.lwt.2021.110940. [DOI] [Google Scholar]
- Murata T., Tago K., Miyata K., Moriwaki Y., Misawa H., Kobata K.…Funakoshi-Tago M. Suppression of Neuroinflammation by coffee component pyrocatechol via inhibition of NF-κB in microglia. International Journal of Molecular Sciences. 2024;25(1) doi: 10.3390/ijms25010316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palencia-Argel M., Rodriguez-Villamil H., Bernal-Castro C., Díaz-Moreno C., Alberto Fuenmayor C. Probiotics in anthocyanin-rich fruit beverages: Research and development for novel synbiotic products. Critical Reviews in Food Science and Nutrition. 2024;64(1):110–126. doi: 10.1080/10408398.2022.2104806. [DOI] [PubMed] [Google Scholar]
- Pan X., Zhang S., Xu X., Lao F., Wu J. Volatile and non-volatile profiles in jujube pulp co-fermented with lactic acid bacteria. LWT- Food Science and Technology. 2022;154 doi: 10.1016/j.lwt.2021.112772. [DOI] [Google Scholar]
- Peng W., Meng D., Yue T., Wang Z., Gao Z. Effect of the apple cultivar on cloudy apple juice fermented by a mixture of Lactobacillus acidophilus, Lactobacillus plantarum, and Lactobacillus fermentum. Food Chemistry. 2021;340 doi: 10.1016/j.foodchem.2020.127922. [DOI] [PubMed] [Google Scholar]
- Plessas S., Mantzourani I., Terpou A., Bekatorou A. Assessment of the physicochemical, antioxidant, microbial, and sensory attributes of yogurt-style products enriched with probiotic-fermented Aronia melanocarpa berry juice. Foods. 2024;13(1) doi: 10.3390/foods13010111. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sayekti W., Lilis W., Muhammad Widyo W., Munawaroh H., Ari Handono R. The effect of pH and color stability of anthocyanin on food colorant. IOP Conference Series: Materials Science and Engineering. 2017;193 doi: 10.1088/1757-899x/193/1/012047. [DOI] [Google Scholar]
- Scuderi S.A., Ardizzone A., Paterniti I., Esposito E., Campolo M. Antioxidant and anti-inflammatory effect of Nrf2 inducer dimethyl fumarate in neurodegenerative diseases. Antioxidants. 2020;9(7) doi: 10.3390/antiox9070630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheehan V.M., Ross P., Fitzgerald G.F. Assessing the acid tolerance and the technological robustness of probiotic cultures for fortification in fruit juices. Innovative Food Science & Emerging Technologies. 2007;8(2):279–284. doi: 10.1016/j.ifset.2007.01.007. [DOI] [Google Scholar]
- Shidiq Nur H., Tri Rini N., Kuwat T. Electronic nose coupled with Chemometrics for monitoring of tempeh fermentation process. International Conference on Science and Technology. 2018 doi: 10.1109/icstc.2018.8528580. [DOI] [Google Scholar]
- Škerget M., Kotnik P., Hadolin M., Hraš H.R., Simonič M., Knez Ž. Phenols, proanthocyanidins, flavones and flavonols in some plant materials and their antioxidant activities. Food Chemistry. 2005;89(2):191–198. doi: 10.1016/j.foodchem.2004.02.025. [DOI] [Google Scholar]
- Smyth H., Cozzolino D. Instrumental methods (spectroscopy, electronic nose, and tongue) as tools to predict taste and aroma in beverages: Advantages and limitations. Chemical Reviews. 2013;113(3):1429–1440. doi: 10.1021/cr300076c. [DOI] [PubMed] [Google Scholar]
- Sun J., Zhao C., Pu X., Li T., Shi X., Wang B., Cheng W. Flavor and functional analysis of Lactobacillus plantarum fermented apricot juice. Fermentation-Basel. 2022;8(10):533. doi: 10.3390/fermentation8100533. [DOI] [Google Scholar]
- Tang S., Luo N., Zeng Q., Dong L., Zhang R., He S., Nag A., Huang F., Su D. Lychee pulp phenolics fermented by mixed lactic acid bacteria strains promote the metabolism of human gut microbiota fermentation in vitro. Food & Function. 2023;14(16):7672–7681. doi: 10.1039/d3fo01668d. [DOI] [PubMed] [Google Scholar]
- Voidarou C., Antoniadou M., Rozos G., Tzora A., Skoufos I., Varzakas T.…Bezirtzoglou E. Fermentative foods: Microbiology, biochemistry, potential human health benefits and public health issues. Foods. 2021;10(1):69. doi: 10.3390/foods10010069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J., Wei B., Xu J., Jiang H., Xu Y., Wang C. Influence of lactic acid fermentation on the phenolic profile, antioxidant activities, and volatile compounds of black chokeberry (Aronia melanocarpa) juice. Journal of Food Science. 2024;89(2):834–850. doi: 10.1111/1750-3841.16899. [DOI] [PubMed] [Google Scholar]
- Wang Z., Tong Y., Tong Q., Liu Y., Xu W. Effects of different lactic acid bacteria on phenolic profiles, antioxidant capacities, and volatile compounds in purple sweet potato juice. Journal of Food Science and Technology-Mysore. 2024 doi: 10.1007/s13197-024-05959-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei M., Wang S., Gu P., Ouyang X., Liu S., Li Y., Zhang B., Zhu B. Comparison of physicochemical indexes, amino acids, phenolic compounds and volatile compounds in bog bilberry juice fermented by Lactobacillus plantarum under different pH conditions. Journal of Food Science and Technology-Mysore. 2018;55(6):2240–2250. doi: 10.1007/s13197-018-3141-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu C., Li T., Qi J., Jiang T., Xu H., Lei H. Effects of lactic acid fermentation-based biotransformation on phenolic profiles, antioxidant capacity and flavor volatiles of apple juice. LWT- Food Science and Technology. 2020;122 doi: 10.1016/j.lwt.2020.109064. [DOI] [Google Scholar]
- Wu D., Xia Q., Cheng H., Zhang Q., Wang Y., Ye X. Changes of volatile flavor compounds in sea buckthorn juice during fermentation based on gas chromatography-ion mobility spectrometry. Foods. 2022;11(21) doi: 10.3390/foods11213471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu Y., Chen R., Liu M., Fang Y., Wu J., Chen J., Yang X., Fang Z., Fang X., Dong S. The effects of Lactobacillus fermentation on the quality changes and flavor characteristics of Aronia melanocarpa juice using physicochemical analysis and electronic nose techniques. Food Innovation and Advances. 2025;4(1):99–107. doi: 10.48130/fia-0025-0010. [DOI] [Google Scholar]
- Xie H., Gao P., Lu Z.-M., Wang F.-Z., Chai L.-J., Shi J.-S., Zhang H.-L., Geng Y., Zhang X.-J., Xu Z.-H. Changes in physicochemical characteristics and metabolites in the fermentation of goji juice by Lactiplantibacillus plantarum. Food Bioscience. 2023;54 doi: 10.1016/j.fbio.2023.102881. [DOI] [Google Scholar]
- Xue Y., Chen J., Wang L., Wang Y., Xu F. Exploring the flavor changes in mung bean flour through Lactobacillus fermentation: Insights from volatile compounds and non-targeted metabolomics analysis. Journal of the Science of Food and Agriculture. 2024;104(12):7238–7248. doi: 10.1002/jsfa.13545. [DOI] [PubMed] [Google Scholar]
- Yaneva T., Dinkova R., Gotcheva V., Angelov A. Modulation of the antioxidant activity of a functional oat beverage by enrichment with chokeberry juice. Journal of Food Processing and Preservation. 2022;46(10) doi: 10.1111/jfpp.16012. [DOI] [Google Scholar]
- Yang H., Hao L., Jin Y., Huang J., Zhou R., Wu C. Functional roles and engineering strategies to improve the industrial functionalities of lactic acid bacteria during food fermentation. Biotechnology Advances. 2024;74 doi: 10.1016/j.biotechadv.2024.108397. [DOI] [PubMed] [Google Scholar]
- Yang W., Kortesniemi M., Ma X., Zheng J., Yang B. Enzymatic acylation of blackcurrant (Ribes nigrum) anthocyanins and evaluation of lipophilic properties and antioxidant capacity of derivatives. Food Chemistry. 2019;281:189–196. doi: 10.1016/j.foodchem.2018.12.111. [DOI] [PubMed] [Google Scholar]
- Yang X., Hong J., Wang L., Cai C., Mo H., Wang J.…Liao Z. Effect of lactic acid Bacteria fermentation on plant-based products. Fermentation-Basel. 2024;10(1) doi: 10.3390/fermentation10010048. [DOI] [Google Scholar]
- Zhang G., Luo S., Huang J., Cai X., Zhang Y. Determination of Total flavonoids in mulberry leaf extract by Aluminium Trichloride colorimetric method. Farm Products Processing. 2018;07(04) doi: 10.16693/j.cnki.1671-9646(X).2018.04.015. 52-54+57. [DOI] [Google Scholar]
- Zhang Y., Yan H., Xu X., Xiao X., Yuan Y., Guo N.…Ye H. Metagenomic/Metaproteomic investigation of the microbiota in Dongbei Suaicai, a traditional fermented Chinese cabbage. Fermentation-Basel. 2024;10(4) doi: 10.3390/fermentation10040185. [DOI] [Google Scholar]
- Zhao Y., Tian T., Li J., Zhang B., Yu Y., Wang Y., Niu H. Variations in Main flavor compounds of freshly distilled brandy during the second distillation. International Journal of Food Engineering. 2014;10(4):809–820. doi: 10.1515/ijfe-2014-0123. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
PCA score plots for QC samples in the positive ion mode (A) and negative ion mode (B). Supplementary Fig. 2. Validation diagram of the OPLS-DA model. Supplementary Fig. 3. Correlation matrix analysis between key differential metabolites and physicochemical properties illustrated as heatmap. Red represents a positive correlation and blue represents a negative correlation. Significance levels are indicated by asterisks: ***P < 0.001; **P < 0.01; *P < 0.05.
Scoring criteria for FAJ. Supplementary Table 2. Differential metabolites between FAJ of 0 h and 48 h. Supplementary Table 3. Metabolic pathway enrichment analysis: 68 pathways mapped using the KEGG database for 48 h vs 0 h.
Data Availability Statement
No data was used for the research described in the article.