Abstract
Vertebrates organ development often takes place in two phases: initial formation and subsequent maturation into the adult form. This is exemplified by the intestine. In mouse, the intestine at birth has villus, where most differentiated epithelial cells are located, but lacks any crypts, where adult intestinal stem cells reside. The crypt is formed during the first 3 weeks after birth when plasma thyroid hormone (T3) levels are high. Similarly, in anurans, the intestine undergoes drastic remodeling into the adult form during metamorphosis in a process completely dependent on T3. Studies on Xenopus metamorphosis have revealed important clues on the formation of the adult intestine during metamorphosis. Here we will review our current understanding on how T3 induces the degeneration of larval epithelium and de novo formation of adult intestinal stem cells. We will also discuss the mechanistic conservations in intestinal development between anurans and mammals.
1. Introduction
Thyroid hormone (T3) is critical for not only the physiology and function of many adult organs but also their development, especially the maturation into the adult form during vertebrate postembryonic development when plasma T3 level peaks. This period corresponds to several weeks or months around birth in mouse and human, respectively (Shi, 1999; Tata, 1993). The maternal dependence of newborns/neonates and uterus-enclosed nature of the embryos in mammals has made it difficult to study how T3 functions during postembryonic development or how adult organs are formed during this period. Interestingly, this period resembles amphibian metamorphosis. In anurans Xenopus laevis, T3 is low or not detectable before stage 54, about 4 weeks old and the onset of metamorphosis, reaches the peak level around stage 62 (about 7 weeks old), and drops to a lower, adult level by the end of metamorphosis (about 8 weeks old or stage 66) (Leloup & Buscaglia, 1977; Nieuwkoop & Faber, 1965). The high levels of T3 during this period triggers the transformations of essentially every organ/tissue in the tadpole. These changes include de novo formation of adult organs such as limbs, total degeneration of larval specific organs such as the tail and gills, and drastic remodeling of most organs, including the intestine, from the larval forms into their adult forms (Dodd & Dodd, 1976; Shi, 1999; Tata, 1993). Many of these changes resemble what happen during mammalian postembryonic development (Shi, 1999; Tata, 1993). Importantly, anuran metamorphosis can be easily manipulated by controlling the availability of T3 to tadpoles or even organ or primary cell cultures, making anuran metamorphosis a highly valuable model for studying T3 action and organ maturation during postembryonic development in vertebrates. This is further enhanced by the recent development of transgenic and gene-editing technologies suitable for genetic studies in the highly related anuran species Xenopus laevis and tropicalis (Blitz, Biesinger, Xie, & Cho, 2013; Fu, Buchholz, & Shi, 2002; Kroll & Amaya, 1996; Lei, Guo, Deng, Chen, & Zhao, 2013; Lei et al., 2012; Nakade et al., 2014; Nakayama et al., 2013; Shi et al., 2015; Wang et al., 2015; Young et al., 2011).
2. T3 and intestinal stem cell development
The vertebrate intestine has long been used as a model system for investigating the property and function of adult stem cells in vertebrates. Organ-specific adult stem cells are essential for organ homeostasis and function (Bao, Shi, & Shi, 2020; Clevers, 2013; Shi, Hasebe, Fu, Fujimoto, & Ishizuya-Oka, 2011; Sirakov, Kress, Nadjar, & Plateroti, 2014; Sun, Fu, & Shi, 2014; Sun & Shi, 2012; van der Flier & Clevers, 2009). In adult mammalian intestine, epithelial stem cells are located at the bottom of the crypts (Fig. 1). After proliferation, their daughter cells migrate along the crypt-villus axis and differentiate into different types of epithelial cells. The differentiated epithelial cells eventually die via programmed cell death, mostly at the villus tip. Such a self-renewing cycle occurs once every 1–6 days in adult mammals (MacDonald, Trier, & Everett, 1964; Toner, Carr, & Wyburn, 1971; van der Flier & Clevers, 2009) and every 2 weeks in the anuran Xenopus laevis (McAvoy & Dixon, 1977).
Fig. 1.
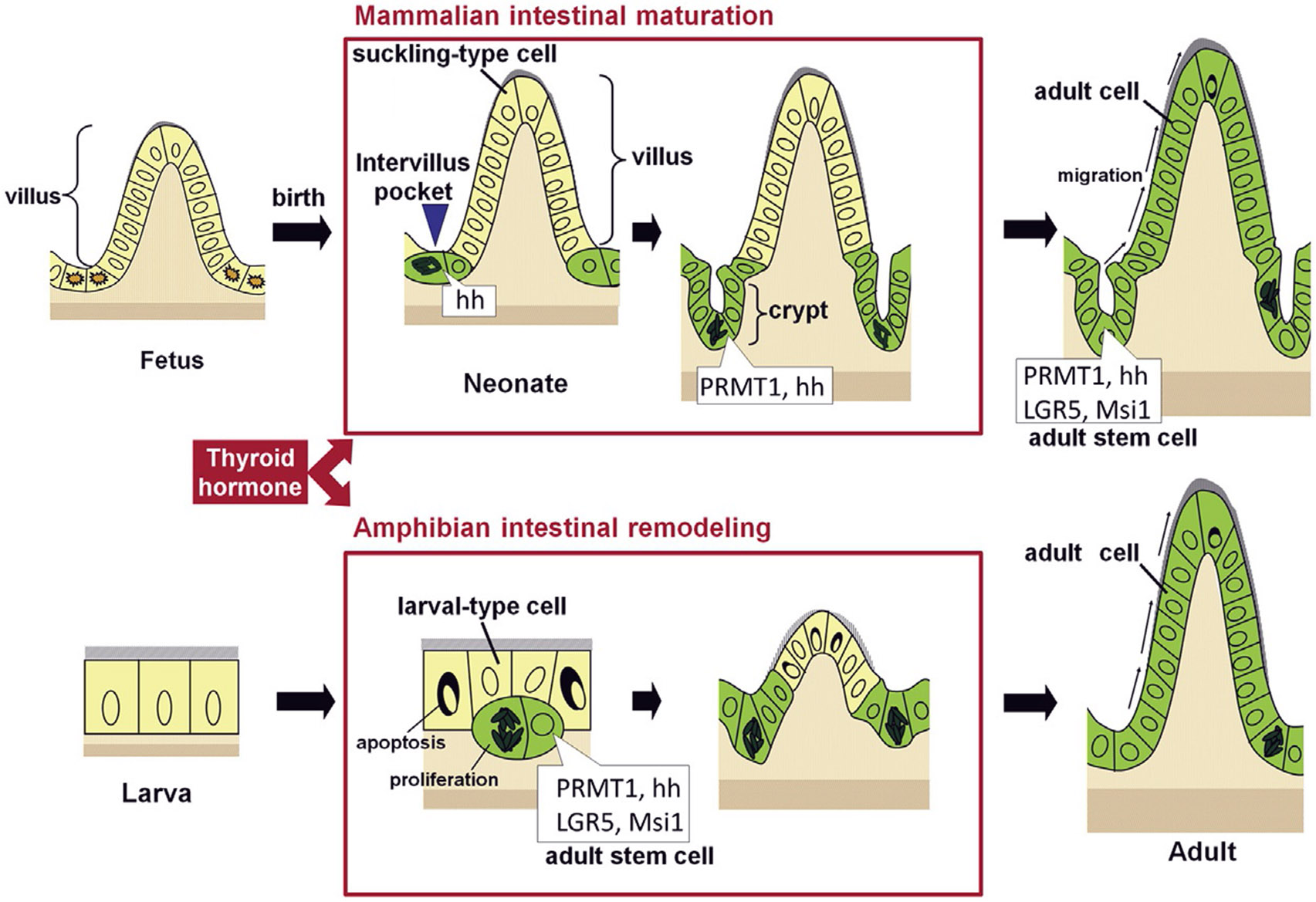
Intestinal maturation takes place during postembryonic development in vertebrates. Both mouse intestinal maturation (upper panel) and frog intestinal remodeling during Xenopus metamorphosis (lower panel) involve the formation of adult epithelial stem cells. Some of the pre-existing epithelial cells develop into adult stem cells during the period when plasma thyroid hormone (T3) levels are high. In mouse, intervillus, sonic hedgehog (hh)-expressing epithelial cells develop into stem cells that express both hh and protein arginine methyltransferase 1 (PRMT1) in the newly formed intestinal crypts within the first 3 weeks after birth. The Xenopus adult intestinal stem cells are formed during metamorphosis through dedifferentiation of some larval epithelial cells to also express high levels of PRMT1 and Shh. The adult stem cell markers LGR5 and Msi1 are expressed in the developing adult intestinal stem cells at the climax of metamorphosis, although they have not been analyzed in the maturing neonatal mouse intestine. Modified after Ishizuya-Oka, A., & Shi, Y.B. (2011). Evolutionary insights into postembryonic development of adult intestinal stem cells. Cell & Bioscience 1, 37.
While there have been extensive studies on the adult intestinal stem cells in mammals, revealing interesting properties of and molecular pathways important for adult intestinal stem cells (Sancho, Eduard Batlle, & Clevers, 2004; van der Flier & Clevers, 2009), much less is known about their development. Recent studies have suggested that the formation of mouse adult intestinal stem cells occurs during the neonatal period when plasma thyroid hormone (T3) level peaks (Fig. 1) (Harper, Mould, Andrews, Bikoff, & Robertson, 2011; Matsuda & Shi, 2010; Muncan et al., 2011; Sun & Shi, 2012). In addition, mutations in T3 receptors (TRs) and deficiency in T3 or TR in mouse cause intestinal defects, including reduced cell proliferation (Bao et al., 2019; Flamant et al., 2002; Kress, Rezza, Nadjar, Samarut, & Plateroti, 2009; Plateroti et al., 1999, 2001; Plateroti, Kress, Mori, & Samarut, 2006). Thus, it is very likely that T3 regulates adult intestinal stem cell development during intestinal maturation.
Similarly, the adult frog intestine is formed during T3-dependent amphibian metamorphosis (Dodd & Dodd, 1976; Shi, 1999; Tata, 1993). In premetamorphic tadpoles, the intestine consists of mainly a monolayer of larval epithelial cells with a single epithelial fold, the typhlosole, surrounded by thin layers of connective tissue and muscles (Figs. 1 and 2) (Shi & Ishizuya-Oka, 1996; Sterling, Fu, Matsuura, & Shi, 2012). As T3 level rises during metamorphosis, most larval epithelial cells undergo apoptosis but some, through yet unknown mechanisms, undergoes dedifferentiation to be highly proliferative cells expressing well-known adult intestinal stem cell markers including leucine-rich repeat-containing G-protein coupled receptor 5 (Lgr5) and Musashi-1 (Msi-1) (Figs. 1 and 2) (Ishizuya-Oka, Shimizu, Sakakibara, Okano, & Ueda, 2003; Okada, Wen, Miller, Su, & Shi, 2015; Wen, Hasebe, Miller, Ishizuya-Oka, & Shi, 2015). By the end of metamorphosis, the proliferating cells differentiate to form a multiply folded adult epithelium surrounded by thick layers of connective tissue and muscles. Accompanying these changes is drastic reduction in the length of the intestine (Ishizuya-Oka et al., 2009; Schreiber, Cai, & Brown, 2005; Shi et al., 2011; Shi & Ishizuya-Oka, 1996; Sterling et al., 2012). In the adult frog, the intestinal stem cells are localized in the trough of the epithelial fold while cell death occurs mainly at the crest of the fold, resembling those occurring in the mammalian crypt-villus unit (Ishizuya-Oka & Shi, 2011; Shi et al., 2011; Shi & Ishizuya-Oka, 1996). Importantly, like all other events during anuran metamorphosis, intestinal remodeling, including adult epithelial stem cell formation, requires T3 signaling. Blocking the synthesis of endogenous T3 in tadpoles inhibits metamorphosis, including intestinal remodeling (Gilbert, Tata, & Atkinson, 1996; Shi, 1999). On the other hand, T3-treatment of premetamorphic tadpoles or even intestinal organ cultures results in premature intestinal metamorphosis, including larval epithelial cell death and de novo formation of adult stem cells (Shi & Ishizuya-Oka, 1996). Thus, T3 controls the de novo formation of the adult epithelial stem cells, which originate organ-autonomously within the larval intestine. Furthermore, T3 treatment of recombinant organ cultures made of wild type intestinal epithelium and transgenic non-epithelial tissues (the rest of the intestine) expressing GFP results in adult epithelium lacking GFP expression while GFP positive adult stem cells are produced if the transgenic epithelium expressing GFP is used in combination with either wild type or transgenic non-epithelial tissues in the organ cultures, demonstrating the epithelial origin of the adult stem cells (Ishizuya-Oka et al., 2009). Given the lack of epithelial stem cells in the tadpole intestine and the differentiated larval epithelial cells are capable of proliferation (Ishizuya-Oka & Shi, 2008; Shi & Ishizuya-Oka, 1996), T3 appears to induce the dedifferentiation of some larval epithelial cells into adult stem cells.
Fig. 2.
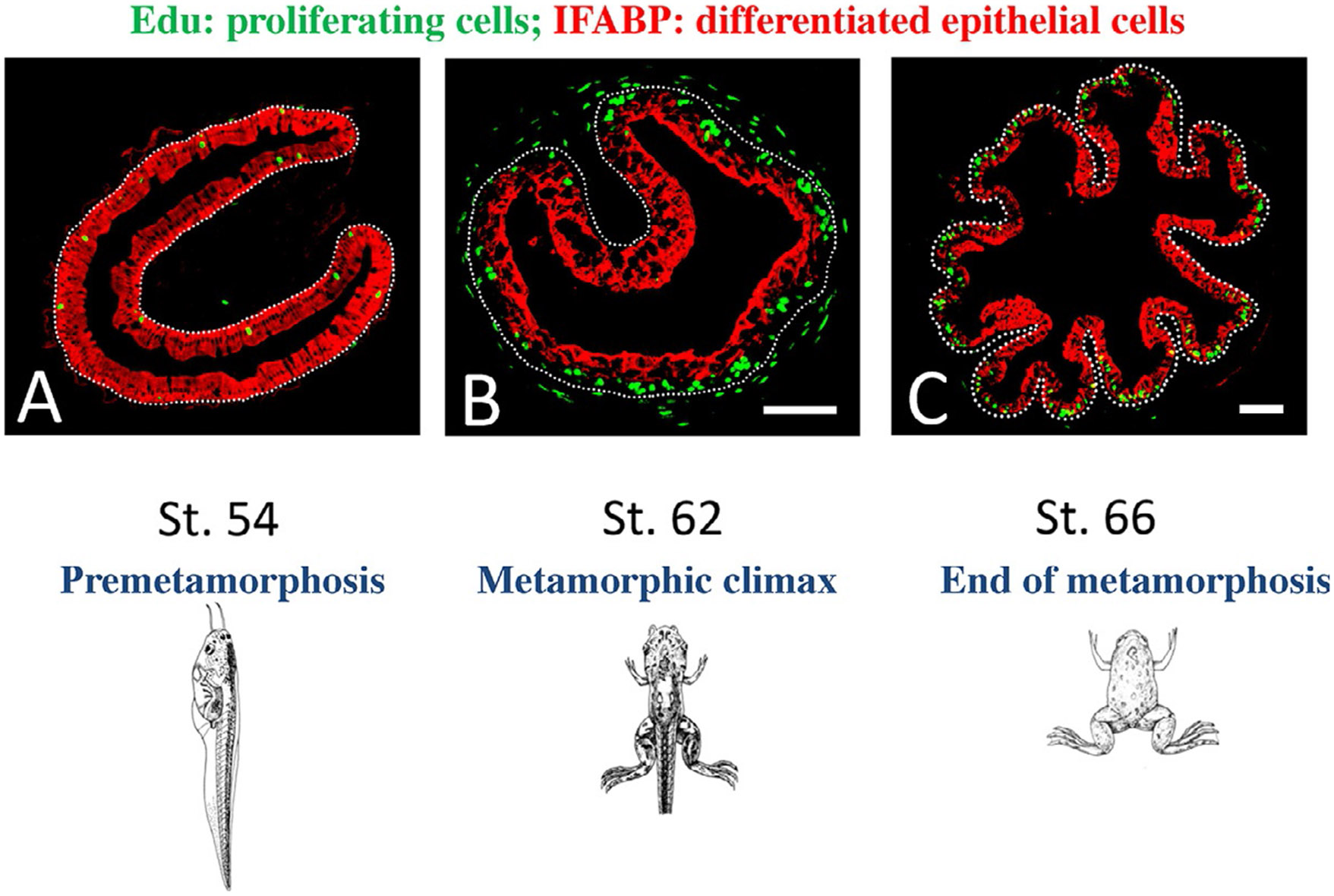
Proliferating adult epithelial stem cells are formed de novo at the climax of Xenopus metamorphosis. Edu (5-ethynyl-2′-deoxyuridine, labeling newly synthesized DNA) were injected into tadpoles at premetamorphic stage 54 (A), metamorphic climax (B, stage 62), or the end of metamorphosis (C, stage 66) to label proliferating cells. One hour later, the animals were sacrificed for preparation of intestinal cross-sections, which were then double-stained for EdU and differentiated intestinal epithelial cell marker IFABP (intestinal fatty acid binding protein) by immunohistochemistry. The epithelium-mesenchyme boundary is marked by dotted lines. Note that at the climax, high levels of EdU-labeled, IFABP-negative proliferating cells were present as clusters between the connective tissue and differentiated cells expressing IFABP (B). After metamorphosis, the proliferating cells were in the troughs where IFABP was low (C), mimicking that in the adult mammalian crypt. Modified after Okada, M., Wen, L., Miller, T.C., Su, D., & Shi, Y.B. (2015). Molecular and cytological analyses reveal distinct transformations of intestinal epithelial cells during Xenopus metamorphosis. Cell & Bioscience 5, 74.
3. Gene regulation by TR and its roles during anuran metamorphosis
T3 is believed to function mainly through its genomic effects via binding to T3 receptors (TRs), although it also has non-genomic effects, at least in cell cultures (Buchholz, Paul, Fu, & Shi, 2006; Davis & Davis, 1996; Evans, 1988; Laudet & Gronemeyer, 2002; Lazar, 1993; Shi, 1999; Tsai & O’Malley, 1994; Yen, 2001). All vertebrates have two TR genes, TRα and TRβ. Extensive biochemical and molecular studies in vitro and in cell cultures have shown that both TRs can activate or repress the transcription of T3-inducible genes in the presence or absence of T3 respectively. TRs do so by mainly functioning as heterodimers with 9-cis retinoic acid receptors (RXRs) to bind to specific DNA sequences called T3-response elements (TREs) within target genes (Lazar, 1993; Mangelsdorf et al., 1995; Tsai & O’Malley, 1994; Wong et al., 1998; Wong, Shi, & Wolffe, 1995, 1997; Yen, 2001). Unliganded TR can recruit corepressor complexes containing histone deacetylases to the target genes to repress their expression (Chen & Evans, 1995; Horlein et al., 1995; Jones, Sachs, Rouse, Wade, & Shi, 2001; Jones & Shi, 2003; Li et al., 2002; Perissi, Jepsen, Glass, & Rosenfeld, 2010; Stewart, Li, & Wong, 2005; Stewart, Tomita, Shi, & Wong, 2006; Tomita, Buchholz, & Shi, 2004; Yoon et al., 2003), while liganded TRs recruit coactivator complexes such as those containing histone acetyltransferases and histone methyltransferases to activate transcription, likely via epigenetic modifications of histones and chromatin remodeling (Bulynko & O’Malley, 2011; Demarest et al., 2002; O’Malley, Malovannaya, & Qin, 2012; Sheppard, Harries, Hussain, Bevan, & Heery, 2001; Shi, Matsuura, Fujimoto, Wen, & Fu, 2012; Yen, 2001). These properties have led to a dual function model for TRs during amphibian metamorphosis (Sachs et al., 2000). That is, during premetamorphosis, TRs are mainly in the unliganded state when T3 is absent or at low levels, and thus recruit corepressors to repress T3-inducible genes. This helps to prevent precocious initiation of metamorphosis to ensure a proper growth period prior to metamorphosis. When T3 levels rise during metamorphosis, T3 binds to TR, which in turn releases the corepressors and recruit coactivators, leading to the de-repression and further activation of the T3-inducible genes, thus triggering metamorphic transformations of different tissues and organ.
With the development of chromatin immunoprecipitation (ChIP) assays and genetic tools for in vivo studies, we and others have tested this dual function model and investigated the roles of TRs during Xenopus metamorphosis. Indeed, TR and RXR have been found to be associated with T3-inducible genes in pre- and metamorphosing Xenopus laevis and tropicalis tadpoles (Sachs & Shi, 2000; Wang, Matsuda, & Shi, 2008) and recruits corepressors or coactivators to modify histones and remodel chromatin in the absence or presence of T3, respectively (Bilesimo et al., 2011; Bulynko & O’Malley, 2011; Demarest et al., 2002; Grimaldi, Buisine, Miller, Shi, & Sachs, 2013; Havis, Sachs, & Demeneix, 2003; Heimeier, Hsia, & Shi, 2008; Huang, Li, Sachs, Cole, & Wong, 2003; Matsuda, Paul, Choi, Hasebe, & Shi, 2009; Matsuda, Paul, Choi, & Shi, 2007; Matsuura, Fujimoto, Fu, & Shi, 2012; O’Malley et al., 2012; Paul, Buchholz, Fu, & Shi, 2005, 2007; Paul, Fu, Buchholz, & Shi, 2005; Sachs, Amano, Rouse, & Shi, 2001; Sachs, Amano, & Shi, 2001; Sachs et al., 2002; Sachs & Shi, 2000; Sheppard et al., 2001; Shi et al., 2012; Tomita et al., 2004; Wang et al., 2008; Wen, Fu, & Shi, 2017; Yen, 2001). More importantly, genetic studies have now demonstrated that TRs are necessary and sufficient for T3-dependent amphibian metamorphosis. First, transgenic expression of dominant negative mutant TRs that cannot bind to T3 inhibits Xenopus laevis metamorphosis while heat shock-inducible transgenic expression of a dominant positive TR that does not bind to T3 but functions like liganded TR mimics T3 induction of metamorphosis in wild type tadpoles, indicating that TR is sufficient to mediate most if not all effects of T3 during amphibian metamorphosis (Bagamasbad, Howdeshell, Sachs, Demeneix, & Denver, 2008; Brown & Cai, 2007; Buchholz et al., 2006; Buchholz, Hsia, Fu, & Shi, 2003; Buchholz, Tomita, Fu, Paul, & Shi, 2004; Denver, Hu, Scanlan, & Furlow, 2009; Nakajima & Yaoita, 2003; Sachs et al., 2000; Schreiber, Das, Huang, Marsh-Armstrong, & Brown, 2001; Schreiber, Mukhi, & Brown, 2009; Shi, 1994, 2009). More recently, with the adaptation of gene-editing technologies to knockout endogenous genes in the diploid Xenopus tropicalis, we and others have shown that both TRα and TRβ are important for metamorphosis (Buchholz & Shi, 2018; Choi, Ishizuya-Oka, & Buchholz, 2017; Choi et al., 2015; Nakajima, Tazawa, & Shi, 2019; Nakajima, Tazawa, & Yaoita, 2018; Sachs, 2015; Sakane et al., 2018; Shibata, Tanizaki, & Shi, 2020; Shibata, Wen, Okada, & Shi, 2020; Wen et al., 2017; Wen & Shi, 2015, 2016; Yen, 2015). While animals lacking either TRα or TRβ can complete metamorphosis and develop into mature frogs, they have various developmental abnormalities during metamorphosis, suggesting that TRα and TRβ have distinct roles in coordinating tissue transformation during metamorphosis but nonetheless can compensate for each other during development. When both TR genes are knocked out, the resulting TR double knockout tadpoles initiate metamorphosis earlier (at younger age) and subsequently progress much slower through metamorphosis (Shibata, Wen, et al., 2020). Thus, consistent with the dual function model, de-repression of the T3-inducible genes leads to premature initiation of metamorphosis and that TRs are important for metamorphic progression. Interestingly and surprisingly, tadpoles lacking both TRα and TRβ can develop up to the climax of metamorphosis (stage 61, Fig. 3) (Shibata, Wen, et al., 2020), suggesting that TRs are not required for the initiation of metamorphosis and de-repression of T3 target genes due to the loss of TRs is sufficient for many of the metamorphic processes. On the other hand, compared to wild type tadpoles at stage 61, the tadpoles lacking any TR have larger gills at stage 61 (Fig. 3), suggesting inhibition of gill resorption due to the TR knockout. Furthermore, these TR double knockout tadpoles are stalled at stage 61 for 2 weeks or so and then die, in contrast to the wild type tadpoles at stage 61, which need only about 1 week to complete the remaining metamorphic changes to become tailless froglets. Further analyses suggest that the development of many of the adult tissues/organs can occur or even takes place earlier in the TR double knockout tadpoles compared to the wild type animals, while the resorption of many larval tissues/organs is inhibited due to the lack of TR (Shibata, Wen, et al., 2020). These finding suggest that de-repression of T3 target genes is sufficient for adult organ development while larval tissue degeneration requires gene activation by liganded TR.
Fig. 3.
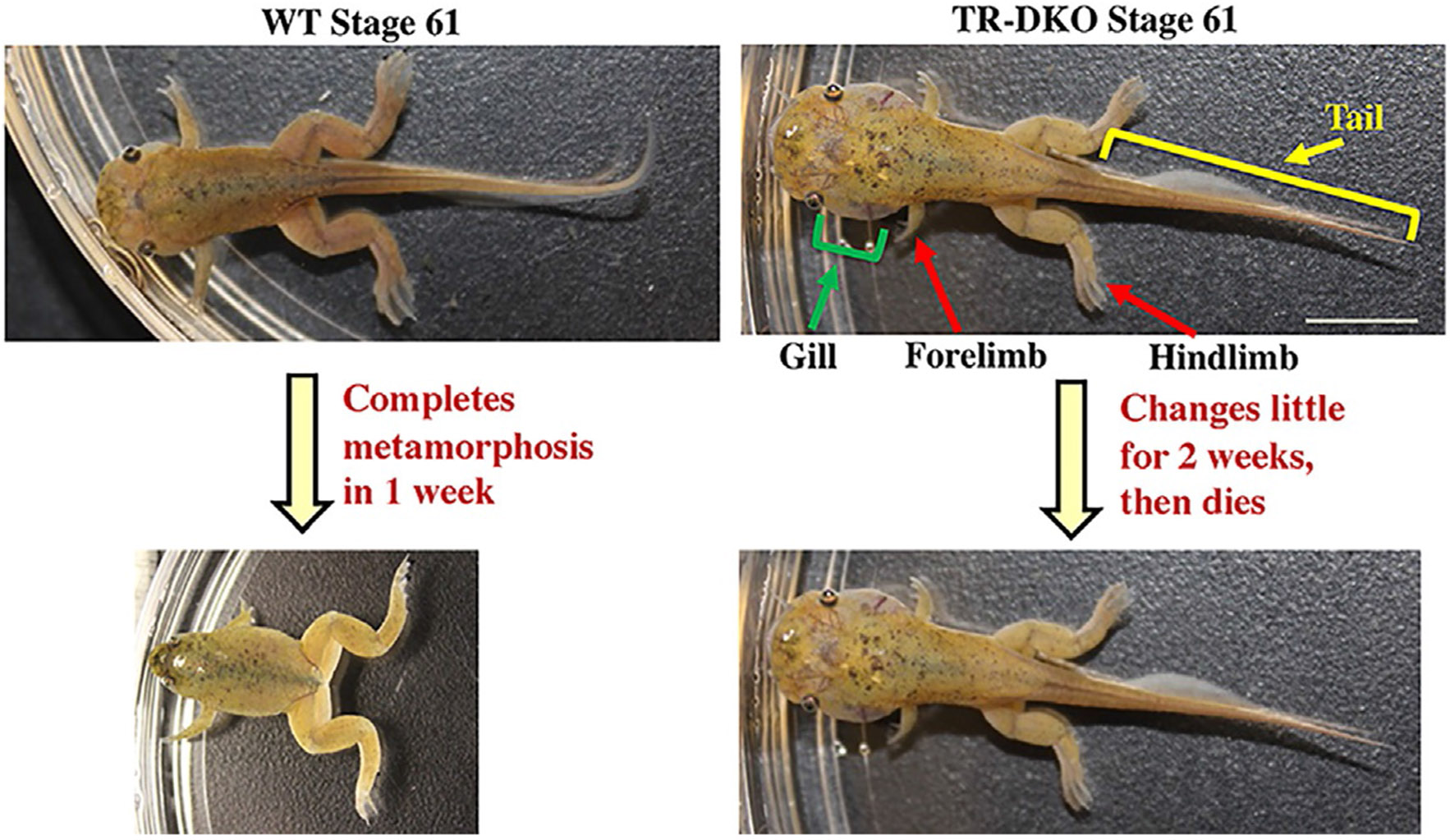
TRs are not required for the initiation of metamorphosis but essential for animal survival and the completion of metamorphosis. TRα and TRβ double knockout (TR-DKO) Xenopus tropicalis tadpoles can develop to the climax stage of 61. Compared to wild type (WT) tadpoles at stage 61, the tadpoles lacking any TR have larger gills, suggesting inhibition of gill resorption due to the TR knockout. Interestingly, these TR-DKO tadpoles are stalled at stage 61 for 2 weeks or so and then die while the WT tadpoles at stage 61 need only about 1 week to complete the remaining metamorphic changes to become tailless froglets. See Shibata, Y., Wen, L., Okada, M., & Shi, Y.B. (2020). Organ-specific requirements for thyroid hormone receptor ensure temporal coordination of tissue-specific transformations and completion of xenopus metamorphosis. Thyroid 30, 300–313 for more details.
4. An essential role of TR in adult intestinal stem cell development
Recent gene knockout studies in Xenopus tropicalis indicate that both TRα and TRβ are important for intestinal metamorphosis (Choi et al., 2015, 2017; Nakajima et al., 2018; Sakane et al., 2018; Shibata, Tanizaki, & Shi, 2020; Shibata, Wen, et al., 2020; Wen & Shi, 2015, 2016; Wen, Shibata, et al., 2017). TRα knockout delays intestinal remodeling (Choi et al., 2017; Wen, Shibata, et al., 2017), while there are only relatively subtle differences in intestinal remodeling between TRβ knockout tadpoles and wild type siblings during natural metamorphosis (Nakajima et al., 2018; Sakane et al., 2018), despite a strong upregulation of TRβ expression during intestinal metamorphosis. These findings suggest that there is a strong compensation between TRα and TRβ during the lengthy natural metamorphosis. This was supported by the recent analysis of intestinal remodeling in TRβ knockout during T3-induced metamorphosis (by treating premetamorphic tadpoles at stage 54 with exogenous but physiological levels of T3), where the larval epithelial cell death and adult intestinal stem cell formation occur within 2–3 days, much shorter than the 2–3 weeks required for the premetamorphic tadpoles at stage 54 to reach stages 60–62 when larval cell death and adult epithelial stem cell formation take place during the natural metamorphosis (Shibata, Tanizaki, & Shi, 2020). Consistent with the reduction in T3-induction of TR target genes in the intestine in TRβ knockout tadpoles, T3-induced reduction in intestinal length, larval epithelial cell death, and adult epithelial stem cell proliferation are all delayed/inhibited in the TRβ knockout tadpoles compared to the wild type siblings. These findings not only reveal a critical role of TRβ for intestinal metamorphosis but also demonstrate that the compensation for the loss of TRβ by TRα is too slow to prevent the defects caused by TRβ knockout during the fast intestinal remodeling induced by T3 treatment.
As described above, TRα and TRβ double knockout animals died at the climax (stage 61) of metamorphosis. Analyses of the intestine have revealed that unlike the wild type intestine at stage 61, little larval epithelial cell death or adult epithelial stem cell proliferation is present in the intestine of TRα and TRβ double knockout animals at stage 61, although adult epithelial folds are formed precociously (Shibata, Y. and Shi, Y.-B., unpublished observation). Furthermore, T3 treatment of premetamorphic TRα and TRβ double knockout animals does not induce any metamorphic changes, including the reduction of intestinal length, accompanied by the lack of the induction of T3 target genes (Shibata, Wen, et al., 2020). Thus, TRs are essential for adult epithelial stem cell development during intestinal remodeling.
A role of TR in adult stem cell development during intestinal metamorphosis has also been supported by recombinant organ culture studies involving animals containing a heat shock-inducible transgenic dominant positive TR (dpTR) that cannot bind to T3 but functions as a constitutively activated TR (Buchholz et al., 2004; Hasebe, Buchholz, Shi, & Ishizuya-Oka, 2011). By recombining wild type intestinal epithelium with transgenic non-epithelium or vice versa, T3 target genes can be selectively activated in either the epithelium or non-epithelium by heat shock treatment of the organ cultures. Analyses of such recombinant organ cultures have shown that dpTR expression in both epithelium and non-epithelium can induce larval epithelial degeneration and development of the adult intestine in the absence of T3, indicating that TR is sufficient for mediating all the effects of T3 for intestinal remodeling (Hasebe et al., 2011). Interestingly, dpTR expression in either the epithelium or non-epithelium can induce larval epithelial cell death, suggesting T3 can induce epithelial apoptosis both cell autonomously and via cell-cell interaction. On the other hand, dpTR in the non-epithelium fails to induce any adult stem cell development while its expression in the epithelium alone leads to the dedifferentiation of some larval epithelial cells, i.e., activating sonic hedgehog gene expression while repressing the expression of the differentiation marker gene encoding intestinal fatty acid binding protein (Hasebe et al., 2011). Interestingly, such dedifferentiated cells lack the expression of Msi-1, a well-known adult intestinal stem cell marker, indicating that adult stem cell development requires T3 signaling in both the epithelium and the non-epithelium (Hasebe et al., 2011). Thus, T3 signaling in the epithelium induces some larval epithelial cells to dedifferentiate and such cells develop further into adult stem cells if T3 signaling is also present in the non-epithelium, which presumably functions to ensure a proper stem cell niche for the developing stem cells (Hasebe et al., 2011; Ishizuya-Oka & Hasebe, 2013; Ishizuya-Oka & Shimozawa, 1992; Schreiber et al., 2009).
5. TR target genes in intestinal stem cell development
TR regulates target gene transcription. Thus, a key to understand how T3 induces the formation of adult stem cells is to identify and functionally characterize TR target genes in the intestine, especially within the epithelium. Over the years, different methods have been used to identify many T3-regulated genes in Xenopus laevis intestine and the involvement of many such genes in intestinal remodeling has been implicated by their expression profiles during metamorphosis (Amano & Yoshizato, 1998; Buchholz, Heimeier, Das, Washington, & Shi, 2007; Fu et al., 2017; Hasebe, Fujimoto, Kajita, & Ishizuya-Oka, 2017; Hasebe, Kajita, Fu, Shi, & Ishizuya-Oka, 2012; Heimeier, Das, Buchholz, Fiorentino, & Shi, 2010; Ishizuya-Oka et al., 2001; Luu et al., 2013; Luu, Fu, Fujimoto, & Shi, 2017; Miller et al., 2013; Na, Fu, Luu, & Shi, 2020; Okada, Miller, Fu, & Shi, 2015; Okada, Miller, Wen, & Shi, 2017; Okada & Shi, 2018a, 2018b; Shi & Brown, 1993; Sun et al., 2013; Sun, Fu, Wen, & Shi, 2014; Wen, Fu, & Shi, 2017). Perhaps not surprisingly, many genes in signaling pathways known to be important for stem cell proliferation and function are induced by T3 during intestinal remodeling. Among them include hedgehog pathway (Hasebe et al., 2012; Hasebe, Kajita, Shi, & Ishizuya-Oka, 2008; Ishizuya-Oka, Ueda, Inokuchi, et al., 2001; Stolow & Shi, 1995; Wen et al., 2015), wnt signaling (Hasebe, Fujimoto, Kajita, & Ishizuya-Oka, 2016; Ishizuya-Oka, Kajita, & Hasebe, 2014; Shi, 2018), Notch pathway (Hasebe et al., 2017), and BMP signaling (Ishizuya-Oka et al., 2001; Ishizuya-Oka, Hasebe, Shimizu, Suzuki, & Ueda, 2006), etc.
If and how such T3-regulated genes participate in adult intestinal stem cell development largely remain to be investigated. Limited genetic studies for some of the genes support their involvement. For example, transgenic overexpression of protein arginine methyltransferase 1 (PRMT1), a known TR coactivator that is upregulated during intestinal remodeling (Fujimoto, Matsuura, Hu-Wang, Lu, & Shi, 2012; Matsuda et al., 2009), leads to increased intestinal stem cells during metamorphosis while antisense morpholino oligonucleotide-mediated knockdown of endogenous PRMT1 reduces proliferating epithelial stem cells (Matsuda & Shi, 2010). PRMT1 likely exerts its effect on intestinal stem cells by functioning as a TR coactivator to increase T3 signaling (Matsuda et al., 2009), although PRMT1 may also act as a coactivator for other transcription factors during stem cell development and/or proliferation or via methylation of non-histone proteins. Similarly, oncogene ectopic viral integration site 1 (EVI) and its variant myelodysplastic syndrome 1 (MDS)/EVI are highly induced by T3 at the climax of intestinal remodeling in Xenopus laevis and Xenopus tropicalis (Miller et al., 2013; Okada & Shi, 2018b). Knocking out the genes by targeting a common region with gene editing causes severe inhibition of tadpole growth and development during metamorphosis and results in partial lethality at metamorphic climax (Okada & Shi, 2018b). Importantly, the knockout tadpoles have reduced adult intestinal epithelial stem cell proliferation at the end of metamorphosis (for the few surviving animals) or during T3-induced metamorphosis (Okada & Shi, 2018b), revealing a novel function of EVI and/or MDS/EVI during vertebrate development.
A surprising finding among the T3-induced genes having an effect on intestinal stem cell development is Mad1, an antagonist of c-Myc in the Myc/Mad/Max network (Okada et al., 2017). Both Mad1 and c-Myc heterodimerize with Max and bind to the same target genes with opposing effect. While c-Myc is a well-known oncogene that activate target gene transcription and promote cell proliferation, Mad represses the expression of c-Myc target genes and is associated with quiescence or cell differentiation. In addition, c-Myc overexpression can induce cell death (Amati & Land, 1994; Dang, 1999; Koskinen & Alitalo, 1993; Kuchino, Asai, & Kitanaka, 1996; McMahon, 2014; Nieminen, Partanen, & Klefstrom, 2007; Packham & Cleveland, 1995; Pelengaris, Rudolph, & Littlewood, 2000; Shi et al., 1992; Thompson, 1998), while Mad is often anti-apoptotic (Amati & Land, 1994; Grandori, Cowley, James, & Eisenman, 2000; Luscher, 2012; McArthur et al., 1998). Consistent with these opposing roles, Mad1 and c-Myc are expressed in distinct epithelial cells during intestinal metamorphosis, with c-Myc expressed in the proliferating adult stem cells while Mad1 in the apoptotic larval epithelial cells (Fig. 4), implicating a surprising role of Mad1 in developmental cell death (Okada et al., 2017). Indeed, knocking out Mad1 inhibits/delays larval epithelial cell death during T3-induced intestinal metamorphosis but unexpectedly, the knockout tadpoles have increased adult epithelial stem cell proliferation during T3-induced metamorphosis (Okada et al., 2017). While the mechanism remains to be determined, one likely scenario is that Mad1 knockout inhibits/delays larval epithelial cell death, allowing more larval epithelial cells or giving larval epithelial cells, which otherwise would undergo T3-induced apoptosis, more time to undergo dedifferentiation into adult epithelial stem cells. Alternatively, Mad1 may be expressed at low levels, not detectable by in situ hybridization as shown in Fig. 4, in developing adult stem cells that express c-Myc. The removal of this low level of Mad1 in the knockout animals thus alters the balance of Mad-Myc, leading to increased stem cell proliferation during T3-induced metamorphosis.
Fig. 4.
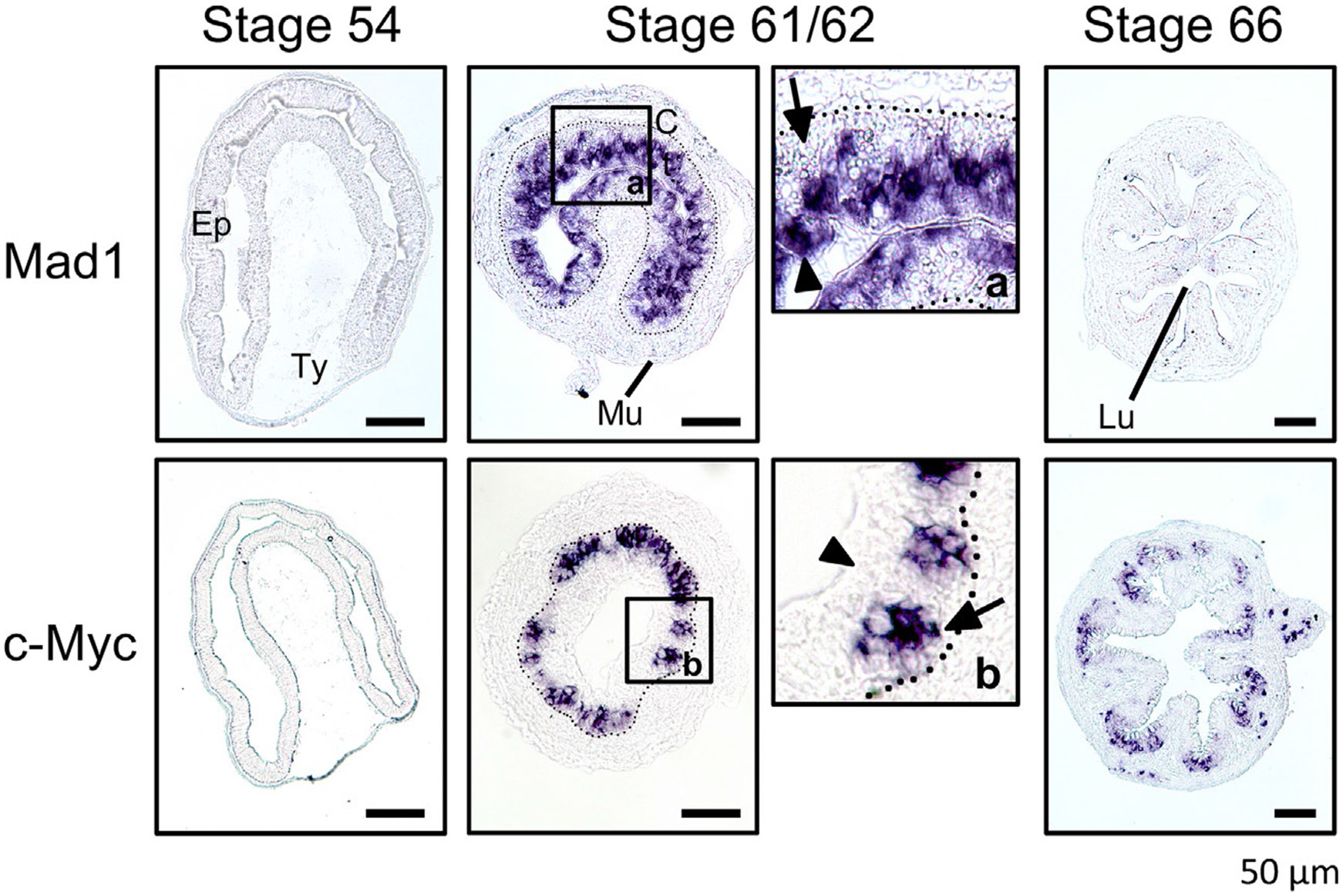
Distinct expression patterns for the oncogene c-Myc and its antagonist Mad1 in the intestinal epithelium during metamorphosis. in situ hybridization analysis of Mad1 and c-Myc mRNAs was carried out on intestinal cross-sections of Xenopus laevis premetamorphic tadpoles (stage 54), metamorphosing tadpoles (stages 61/62 or climax), and post metamorphic froglets (stage 66). Panels a and b are higher magnification photos of the boxed region at stage 61/62 for Mad1 and c-Myc, respectively. Note that both Mad1 and c-Myc have little expression during premetamorphosis but are strongly activated at the climax (stages 61/62) with Mad expressed in the dying larval epithelial cells (arrowheads) while c-Myc expressed in the epithelial region close to the connective tissue (arrows). The dotted lines mark the approximate boundary between the epithelium (Ep) and the connective tissue (C). Scale bar, 50μm. Lu, lumen; Mu, muscle; Ty, typhlosole. Modified after Okada, M., Miller, T.C., Wen, L., & Shi, Y.B. (2017). A balance of Mad and Myc expression dictates larval cell apoptosis and adult stem cell development during Xenopus intestinal metamorphosis. Cell Death & Disease 8, e2787.
The involvement of Myc/Mad/Max network in both larval epithelial cell death and adult stem cell formation and/or proliferation is also consistent with recent RNA-seq analysis of genes regulated by T3 in the intestine of premetamorphic Xenopus tropicalis tadpoles after 1 day T3 treatment (Tanizaki, Shibata, Zhang, & Shi, 2020). T3 treatment of premetamorphic tadpoles at stage 54 is known to induce larval epithelial cell death after 2 days and formation and proliferation of adult intestinal stem cells after 3 days. Interestingly, gene ontology and biological pathway analyses of the T3-regulated genes revealed that the gene ontology terms and signaling pathways related to cell cycle regulation were highly enriched among the regulated genes after 1 day of T3 treatment, which was too short for T3 to induce any larval cell death or stem cell formation/proliferation in the tadpole intestine. By comparing the RNA-seq data of the wild type TRα knockout tadpole intestine, it was found that the endogenous TRα was important for the regulation of the cell cycle program. The intestine in premetamorphic tadpoles are made of predominantly larval epithelial cells, surrounded by thin layers of connective tissue and muscles (Fig. 2). Furthermore, most cell death and proliferation during metamorphosis occur in the epithelium (Fig. 2) (Okada, Wen, et al., 2015). The observed activation of the cell cycle program after 1 day of T3 treatment presumably reflected the changes in gene expression within the larval epithelium. The findings from the RNA-seq analyses thus suggest a novel model for T3 function during intestinal remodeling. That is, T3, mainly via TRα, which is expressed at a much higher level compared to TRβ in premetamorphic tadpole intestine (Wang et al., 2008), induces the cell cycle program in the larval epithelial cells. This, in turn, may facilitate the differentiated larval intestinal epithelial cells to initiate cellular changes toward two alternative fates: death via apoptosis or dedifferentiation to become adult stem cells. Clearly, it would be interesting to test this model.
6. Conservations between Xenopus intestinal metamorphosis and mammalian intestinal maturation
As mentioned above, mouse intestinal maturation occurs within the first 3 weeks or so after birth when plasma T3 concentration peaks, just like during amphibian metamorphosis. In addition, T3 or TR deficiency leads to adult intestinal defects in mouse, including reduced proliferating crypt cells (Flamant et al., 2002; Kress et al., 2009; Plateroti et al., 1999, 2001, 2006). Furthermore, human patients with TRα mutations have constipations, implicating intestinal defects, unlike patients with TRβ mutations (Bochukova et al., 2012; Moran & Chatterjee, 2015; Refetoff, Weiss, & Usala, 1993; van Mullem et al., 2012), supporting a specific role of TRα in the intestine. Consistently, a knockin mutation in the mouse TRα gene mimicking human TRα mutant patients also results in constipations in the adult mice (Bao et al., 2019). More importantly, analyses of the adult intestine in the knockin mutant have revealed drastically reduced cell proliferation in the crypts and altered epithelial cell differentiation (Bao et al., 2019), although only minor effect on the morphology of the villus-crypt axis due to compensatory reduction in epithelial cell death on the villus (Bao et al., 2019, 2020). All these point to a role of T3 and TR in intestinal maturation and adult stem cell function.
Interestingly, PRMT1, a gene that is induced by T3 and important for intestinal stem cell formation/proliferation (see above), is also upregulated during intestinal maturation in mouse and zebrafish when T3 levels are high (Matsuda & Shi, 2010). There is little or no PRMT1 mRNA in the zebrafish larval intestine or mouse neonatal intestine. During intestinal maturation when T3 levels peaks (Brown, 1997; Friedrichsen et al., 2003), PRMT1 is upregulated specifically in the bottom of the developing epithelial fold in zebrafish intestine and the developing crypt in mouse intestine, suggesting that conserved roles of T3 and PRMT1 for the formation and/or proliferation of the adult stem cells in vertebrates. Further support for the conservation in adult intestinal stem cell development came from genetic studies on B lymphocyte-induced maturation protein 1 (Blimp1) (Harper et al., 2011; Muncan et al., 2011). Blimp1, a transcriptional repressor, is strongly and uniformly expressed in the intestinal epithelium up to newborn mice before crypts are formed. With rising T3 concentration after birth, the crypt begins to form within the inter-villus pockets of the intestine where Blimp1 expression is reduced. As the intestine develops further, Blimp1 expression is repressed in all crypt cells and eventually the rest of the epithelium in the adult mice, likely due to the gradual replacement of the neonatal epithelium by the newly differentiated cells originated from the crypts. These findings suggest that Blimp1 repression is an early event in the epithelial transformation to form the adult stem cells. The distinct expression profiles of PRMT1 and Blimp1 in the neonatal intestine compared to the adult intestinal stem cells in the crypts suggest that like anuran intestinal metamorphosis, mouse intestinal maturation involves de novo formation of adult stem cells in a conserved process regulated by T3.
7. Conclusion
The ability to easily manipulate amphibian metamorphosis and the recent advancement in molecular and genetic studies in Xenopus have made this process highly valuable model for studying postembryonic organ development in vertebrates (Bilesimo et al., 2011; Brown & Cai, 2007; Buchholz et al., 2006; Buchholz & Shi, 2018; Grimaldi et al., 2013; Matsuura et al., 2012; Shi, 2013; Shi et al., 2012). Important insights have been gained from studying intestinal metamorphosis in Xenopus laevis and Xenopus tropicalis. Among them are the discovery that adult intestinal epithelial stem cells are formed de novo via T3-induced dedifferentiation of some larval epithelial cells and that this process resembles postembryonic intestinal maturation in mouse. Furthermore, a number of novel genes important for adult stem cell development/proliferation have been discovered from the identification and functional analyses of some of T3 target genes during intestinal remodeling. Given the important role of T3 in intestinal maturation and adult intestinal physiology, these novel adult stem cell genes will likely have conserved roles in mammalian intestinal maturation and function. The availability of many T3-induced genes in the developing intestinal stem cells, i.e., candidate stem cell genes, and the recent advancements in genetic analysis in Xenopus (Blitz et al., 2013; Lei et al., 2012, 2013; Nakade et al., 2014; Nakayama et al., 2013; Shi et al., 2015; Wang et al., 2015; Young et al., 2011) will ensure intestinal metamorphosis to be as a highly valuable model for further studying adult stem cell development in vertebrates.
Acknowledgment
This work was supported by the intramural Research Program of NICHD, NIH.
References
- Amano T, & Yoshizato K (1998). Isolation of genes involved in intestinal remodeling during anuran metamorphosis. Wound Repair and Regeneration, 6, 302–313. [DOI] [PubMed] [Google Scholar]
- Amati B, & Land H (1994). Myc-Max-Mad: A transcription factor network controlling cell cycle progression, differentiation and death. Current Opinion in Genetics & Development, 4, 102–108. [DOI] [PubMed] [Google Scholar]
- Bagamasbad P, Howdeshell KL, Sachs LM, Demeneix BA, & Denver RJ (2008). A role for basic transcription element-binding protein 1 (BTEB1) in the autoinduction of thyroid hormone receptor beta. The Journal of Biological Chemistry, 283, 2275–2285. [DOI] [PubMed] [Google Scholar]
- Bao L, Roediger J, Park S, Fu L, Shi B, Cheng SY, et al. (2019). Thyroid hormone receptor alpha mutations lead to epithelial defects in the adult intestine in a mouse model of resistance to thyroid hormone. Thyroid, 29, 439–448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bao L, Shi B, & Shi YB (2020). Intestinal homeostasis: A communication between life and death. Cell & Bioscience, 10, 66. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bilesimo P, Jolivet P, Alfama G, Buisine N, Le Mevel S, Havis E, et al. (2011). Specific histone lysine 4 methylation patterns define TR-binding capacity and differentiate direct T3 responses. Molecular Endocrinology, 25, 225–237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blitz IL, Biesinger J, Xie X, & Cho KW (2013). Biallelic genome modification in F(0) Xenopus tropicalis embryos using the CRISPR/Cas system. Genesis, 51, 827–834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bochukova E, Schoenmakers N, Agostini M, Schoenmakers E, Rajanayagam O, Keogh JM, et al. (2012). A mutation in the thyroid hormone receptor alpha gene. The New England Journal of Medicine, 366, 243–249. [DOI] [PubMed] [Google Scholar]
- Brown DD (1997). The role of thyroid hormone in zebrafish and axoloft development. Proceedings of the National Academy of Sciences of the United States of America, 94, 13011–13016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown DD, & Cai L (2007). Amphibian metamorphosis. Developmental Biology, 306, 20–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchholz DR, Heimeier RA, Das B, Washington T, & Shi Y-B (2007). Pairing morphology with gene expression in thyroid hormone-induced intestinal remodeling and identification of a core set of TH-induced genes across tadpole tissues. Developmental Biology, 303, 576–590. [DOI] [PubMed] [Google Scholar]
- Buchholz DR, Hsia VS-C, Fu L, & Shi Y-B (2003). A dominant negative thyroid hormone receptor blocks amphibian metamorphosis by retaining corepressors at target genes. Molecular and Cellular Biology, 23, 6750–6758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchholz DR, Paul BD, Fu L, & Shi YB (2006). Molecular and developmental analyses of thyroid hormone receptor function in Xenopus laevis, the African clawed frog. General and Comparative Endocrinology, 145, 1–19. [DOI] [PubMed] [Google Scholar]
- Buchholz DR, & Shi YB (2018). Dual function model revised by thyroid hormone receptor alpha knockout frogs. General and Comparative Endocrinology, 265, 214–218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buchholz DR, Tomita A, Fu L, Paul BD, & Shi Y-B (2004). Transgenic analysis reveals that thyroid hormone receptor is sufficient to mediate the thyroid hormone signal in frog metamorphosis. Molecular and Cellular Biology, 24, 9026–9037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bulynko YA, & O’Malley BW (2011). Nuclear receptor coactivators: Structural and functional biochemistry. Biochemistry, 50, 313–328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen JD, & Evans RM (1995). A transcriptional co-repressor that interacts with nuclear hormone receptors. Nature, 377, 454–457. [DOI] [PubMed] [Google Scholar]
- Choi J, Ishizuya-Oka A, & Buchholz DR (2017). Growth, development, and intestinal remodeling occurs in the absence of thyroid hormone receptor alpha in tadpoles of Xenopus tropicalis. Endocrinology, 158, 1623–1633. [DOI] [PubMed] [Google Scholar]
- Choi J, Suzuki KI, Sakuma T, Shewade L, Yamamoto T, & Buchholz DR (2015). Unliganded thyroid hormone receptor alpha regulates developmental timing via gene repression as revealed by gene disruption in Xenopus tropicalis. Endocrinology, 156, 735–744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clevers H. (2013). The intestinal crypt, a prototype stem cell compartment. Cell, 154, 274–284. [DOI] [PubMed] [Google Scholar]
- Dang CV (1999). c-Myc target genes involved in cell growth, apoptosis, and metabolism. Molecular and Cellular Biology, 19, 1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Davis PJ, & Davis FB (1996). Nongenomic actions of thyroid hormone. Thyroid, 6, 497–504. [DOI] [PubMed] [Google Scholar]
- Demarest SJ, Martinez-Yamout M, Chung J, Chen H, Xu W, Dyson HJ, et al. (2002). Mutual synergistic folding in recruitment of CBP/p300 by p160 nuclear receptor coactivators. Nature, 415, 549–553. [DOI] [PubMed] [Google Scholar]
- Denver RJ, Hu F, Scanlan TS, & Furlow JD (2009). Thyroid hormone receptor subtype specificity for hormone-dependent neurogenesis in Xenopus laevis. Developmental Biology, 326, 155–168. [DOI] [PubMed] [Google Scholar]
- Dodd MHI, & Dodd JM (1976). The biology of metamorphosis. In Lofts B (Ed.), Physiology of the amphibia (pp. 467–599). New York: Academic Press. [Google Scholar]
- Evans RM (1988). The steroid and thyroid hormone receptor superfamily. Science, 240, 889–895. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Flamant F, Poguet AL, Plateroti M, Chassande O, Gauthier K, Streichenberger N, et al. (2002). Congenital hypothyroid Pax8(−/−) mutant mice can be rescued by inactivating the TRalpha gene. Molecular Endocrinology, 16, 24–32. [DOI] [PubMed] [Google Scholar]
- Friedrichsen S, Christ S, Heuer H, Schäafer MKH, Mansouri A, Bauer K, et al. (2003). Regulation of iodothyronine deiodinases in the Pax8 −/− mouse model of congenital hypothyroidism. Endocrinology, 144, 777–784. [DOI] [PubMed] [Google Scholar]
- Fu L, Buchholz D, & Shi YB (2002). Novel double promoter approach for identification of transgenic animals: A tool for in vivo analysis of gene function and development of gene-based therapies. Molecular Reproduction and Development, 62, 470–476. [DOI] [PubMed] [Google Scholar]
- Fu L, Das B, Matsuura K, Fujimoto K, Heimeier RA, & Shi YB (2017). Genome-wide identification of thyroid hormone receptor targets in the remodeling intestine during Xenopus tropicalis metamorphosis. Scientific Reports, 7, 6414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujimoto K, Matsuura K, Hu-Wang E, Lu R, & Shi YB (2012). Thyroid hormone activates protein arginine methyltransferase 1 expression by directly inducing c-Myc transcription during xenopus intestinal stem cell development. The Journal of Biological Chemistry, 287, 10039–10050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilbert LI, Tata JR, & Atkinson BG (1996). Metamorphosis: Post-embryonic reprogramming of gene expression in amphibian and insect cells. New York: Academic Press. [Google Scholar]
- Grandori C, Cowley SM, James LP, & Eisenman RN (2000). The Myc/Max/Mad network and the transcriptional control of cell behavior. Annual Review of Cell and Developmental Biology, 16, 653–699. [DOI] [PubMed] [Google Scholar]
- Grimaldi A, Buisine N, Miller T, Shi YB, & Sachs LM (2013). Mechanisms of thyroid hormone receptor action during development: Lessons from amphibian studies. Biochimica et Biophysica Acta, 1830, 3882–3892. [DOI] [PubMed] [Google Scholar]
- Harper J, Mould A, Andrews RM, Bikoff EK, & Robertson EJ (2011). The transcriptional repressor Blimp1/Prdm1 regulates postnatal reprogramming of intestinal enterocytes. Proceedings of the National Academy of Sciences of the United States of America, 108, 10585–10590. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasebe T, Buchholz DR, Shi YB, & Ishizuya-Oka A (2011). Epithelial-connective tissue interactions induced by thyroid hormone receptor are essential for adult stem cell development in the Xenopus laevis intestine. Stem Cells, 29, 154–161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasebe T, Fujimoto K, Kajita M, Fu L, Shi YB, & Ishizuya-Oka A (2017). Thyroid hormone-induced activation of notch signaling is required for adult intestinal stem cell development during Xenopus laevis metamorphosis. Stem Cells, 35, 1028–1039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasebe T, Fujimoto K, Kajita M, & Ishizuya-Oka A (2016). Thyroid hormone activates Wnt/beta-catenin signaling involved in adult epithelial development during intestinal remodeling in Xenopus laevis. Cell and Tissue Research, 365, 309–318. [DOI] [PubMed] [Google Scholar]
- Hasebe T, Fujimoto K, Kajita M, & Ishizuya-Oka A (2017). Essential roles of thyroid hormone-regulated hyaluronan/CD44 signaling in adult stem cell development during Xenopus laevis intestinal remodeling. Stem Cells, 35, 2175–2183. [DOI] [PubMed] [Google Scholar]
- Hasebe T, Kajita M, Fu L, Shi YB, & Ishizuya-Oka A (2012). Thyroid hormone-induced sonic hedgehog signal up-regulates its own pathway in a paracrine manner in the Xenopus laevis intestine during metamorphosis. Developmental Dynamics, 241, 403–414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hasebe T, Kajita M, Shi YB, & Ishizuya-Oka A (2008). Thyroid hormone-up-regulated hedgehog interacting protein is involved in larval-to-adult intestinal remodeling by regulating sonic hedgehog signaling pathway in Xenopus laevis. Developmental Dynamics, 237, 3006–3015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Havis E, Sachs LM, & Demeneix BA (2003). Metamorphic T3-response genes have specific co-regulator requirements. EMBO Reports, 4, 883–888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heimeier RA, Das B, Buchholz DR, Fiorentino M, & Shi YB (2010). Studies on Xenopus laevis intestine reveal biological pathways underlying vertebrate gut adaptation from embryo to adult. Genome Biology, 11, R55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heimeier RA, Hsia VS-C, & Shi Y-B (2008). Participation of BAF57 and BRG1-containing chromatin remodeling complexes in thyroid hormone-dependent gene activation during vertebrate development. Molecular Endocrinology, 22, 1065–1077. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horlein AJ, Naar AM, Heinzel T, Torchia J, Gloss B, Kurokawa R, et al. (1995). Ligand-independent repression by the thyroid hormone receptor mediated by a nuclear receptor co-repressor. Nature, 377, 397–404. [DOI] [PubMed] [Google Scholar]
- Huang Z-Q, Li J, Sachs LM, Cole PA, & Wong J (2003). A role for cofactor–cofactor and cofactor–histone interactions in targeting p300, SWI/SNF and mediator for transcription. The EMBO Journal, 22, 2146–2155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishizuya-Oka A, & Hasebe T (2013). Establishment of intestinal stem cell niche during amphibian metamorphosis. Current Topics in Developmental Biology, 103, 305–327. [DOI] [PubMed] [Google Scholar]
- Ishizuya-Oka A, Hasebe T, Buchholz DR, Kajita M, Fu L, & Shi YB (2009). Origin of the adult intestinal stem cells induced by thyroid hormone in Xenopus laevis. The FASEB Journal, 23, 2568–2575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishizuya-Oka A, Hasebe T, Shimizu K, Suzuki K, & Ueda S (2006). Shh/BMP-4 signaling pathway is essential for intestinal epithelial development during Xenopus larval-to-adult remodeling. Developmental Dynamics, 235, 3240–3249. [DOI] [PubMed] [Google Scholar]
- Ishizuya-Oka A, Kajita M, & Hasebe T (2014). Thyroid hormone-regulated Wnt5a/ Ror2 signaling is essential for dedifferentiation of larval epithelial cells into adult stem cells in the Xenopus laevis intestine. PLoS One, 9, e107611. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishizuya-Oka A, & Shi YB (2008). Thyroid hormone regulation of stem cell development during intestinal remodeling. Molecular and Cellular Endocrinology, 288, 71–78. [DOI] [PubMed] [Google Scholar]
- Ishizuya-Oka A, & Shi YB (2011). Evolutionary insights into postembryonic development of adult intestinal stem cells. Cell & Bioscience, 1, 37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishizuya-Oka A, Shimizu K, Sakakibara S, Okano H, & Ueda S (2003). Thyroid hormone-upregulated expression of Musashi-1 is specific for progenitor cells of the adult epithelium during amphibian gastrointestinal remodeling. Journal of Cell Science, 116, 3157–3164. [DOI] [PubMed] [Google Scholar]
- Ishizuya-Oka A, & Shimozawa A (1992). Connective tissue is involved in adult epithelial development of the small intestine during anuran metamorphosis in vitro. Roux’s Archives of Developmental Biology, 201, 322–329. [DOI] [PubMed] [Google Scholar]
- Ishizuya-Oka A, Ueda S, Amano T, Shimizu K, Suzuki K, Ueno N, et al. (2001). Thyroid-hormone-dependent and fibroblast-specific expression of BMP-4 correlates with adult epithelial development during amphibian intestinal remodeling. Cell and Tissue Research, 303, 187–195. [DOI] [PubMed] [Google Scholar]
- Ishizuya-Oka A, Ueda S, Inokuchi T, Amano T, Damjanovski S, Stolow M, et al. (2001). Thyroid hormone-induced expression of Sonic hedgehog correlates with adult epithelial development during remodeling of the Xenopus stomach and intestine. Differentiation, 69, 27–37. [DOI] [PubMed] [Google Scholar]
- Jones PL, Sachs LM, Rouse N, Wade PA, & Shi YB (2001). Multiple N-CoR complexes contain distinct histone deacetylases. The Journal of Biological Chemistry, 276, 8807–8811. [DOI] [PubMed] [Google Scholar]
- Jones PL, & Shi Y-B (2003). N-CoR-HDAC corepressor complexes: Roles in transcriptional regulation by nuclear hormone receptors. In Workman JL (Ed.), Current topics in microbiology and immunology: Protein complexes that modify chromatin (pp. 237–268). Berlin: Springer-Verlag. [DOI] [PubMed] [Google Scholar]
- Koskinen PJ, & Alitalo K (1993). Role of myc amplification and overexpression in cell growth, differentiation and death. Seminars in Cancer Biology, 4, 3–12. [PubMed] [Google Scholar]
- Kress E, Rezza A, Nadjar J, Samarut J, & Plateroti M (2009). The frizzled-related sFRP2 gene is a target of thyroid hormone receptor alpha1 and activates beta-catenin signaling in mouse intestine. The Journal of Biological Chemistry, 284, 1234–1241. [DOI] [PubMed] [Google Scholar]
- Kroll KL, & Amaya E (1996). Transgenic Xenopus embryos from sperm nuclear transplantations reveal FGF signaling requirements during gastrulation. Development, 122, 3173–3183. [DOI] [PubMed] [Google Scholar]
- Kuchino Y, Asai A, & Kitanaka C (1996). Myc-mediated apoptosis. Progress in Molecular and Subcellular Biology, 16, 104–129. [DOI] [PubMed] [Google Scholar]
- Laudet V, & Gronemeyer H (2002). The nuclear receptor FactsBook. San Diego: Academic Press. [Google Scholar]
- Lazar MA (1993). Thyroid hormone receptors: Multiple forms, multiple possibilities. Endocrine Reviews, 14, 184–193. [DOI] [PubMed] [Google Scholar]
- Lei Y, Guo X, Deng Y, Chen Y, & Zhao H (2013). Generation of gene disruptions by transcription activator-like effector nucleases (TALENs) in Xenopus tropicalis embryos. Cell & Bioscience, 3, 21. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lei Y, Guo X, Liu Y, Cao Y, Deng Y, Chen X, et al. (2012). Efficient targeted gene disruption in Xenopus embryos using engineered transcription activator-like effector nucleases (TALENs). Proceedings of the National Academy of Sciences of the United States of America, 109, 17484–17489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leloup J, & Buscaglia M (1977). La triiodothyronine: Hormone de la métamorphose des amphibiens. Comptes Rendus. Académie des Sciences, 284, 2261–2263. [Google Scholar]
- Li J, Lin Q, Yoon HG, Huang ZQ, Strahl BD, Allis CD, et al. (2002). Involvement of histone methylation and phosphorylation in regulation of transcription by thyroid hormone receptor. Molecular and Cellular Biology, 22, 5688–5697. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luscher B. (2012). MAD1 and its life as a MYC antagonist: An update. European Journal of Cell Biology, 91, 506–514. [DOI] [PubMed] [Google Scholar]
- Luu N, Fu L, Fujimoto K, & Shi YB (2017). Direct regulation of histidine ammonia-lyase 2 gene by thyroid hormone in the developing adult intestinal stem cells. Endocrinology, 158, 1022–1033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Luu N, Wen L, Fu L, Fujimoto K, Shi YB, & Sun G (2013). Differential regulation of two histidine ammonia-lyase genes during Xenopus development implicates distinct functions during thyroid hormone-induced formation of adult stem cells. Cell & Bioscience, 3, 43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacDonald WC, Trier JS, & Everett NB (1964). Cell proliferation and migration in the stomach, duodenum, and rectum of man: Radioautographic studies. Gastroenterology, 46, 405–417. [PubMed] [Google Scholar]
- Mangelsdorf DJ, Thummel C, Beato M, Herrlich P, Schutz G, Umesono K, et al. (1995). The nuclear receptor superfamily: The second decade. Cell, 83, 835–839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuda H, Paul BD, Choi CY, Hasebe T, & Shi Y-B (2009). Novel functions of protein arginine methyltransferase 1 in thyroid hormone receptor-mediated transcription and in the regulation of metamorphic rate in Xenopus laevis. Molecular and Cellular Biology, 29, 745–757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuda H, Paul BD, Choi CY, & Shi Y-B (2007). Contrasting effects of two alternative splicing forms of coactivator-associated arginine methyltransferase 1 on thyroid hormone receptor-mediated transcription in Xenopus laevis. Molecular Endocrinology, 21, 1082–1094. [DOI] [PubMed] [Google Scholar]
- Matsuda H, & Shi YB (2010). An essential and evolutionarily conserved role of protein arginine methyltransferase 1 for adult intestinal stem cells during postembryonic development. Stem Cells, 28, 2073–2083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsuura K, Fujimoto K, Fu L, & Shi Y-B (2012). Liganded thyroid hormone receptor induces nucleosome removal and histone modifications to activate transcription during larval intestinal cell death and adult stem cell development. Endocrinology, 153, 961–972. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McArthur GA, Laherty CD, Queva C, Hurlin PJ, Loo L, James L, et al. (1998). The Mad protein family links transcriptional repression to cell differentiation. Cold Spring Harbor Symposia on Quantitative Biology, 63, 423–433. [DOI] [PubMed] [Google Scholar]
- McAvoy JW, & Dixon KE (1977). Cell proliferation and renewal in the small intestinal epithelium of metamorphosing and adult Xenopus laevis. The Journal of Experimental Zoology, 202, 129–138. [Google Scholar]
- McMahon SB (2014). MYC and the control of apoptosis. Cold Spring Harbor Perspectives in Medicine, 4, a014407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller TC, Sun G, Hasebe T, Fu L, Heimeier RA, Das B, et al. (2013). Tissue-specific upregulation of MDS/EVI gene transcripts in the intestine by thyroid hormone during Xenopus metamorphosis. PLoS One, 8, e55585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moran C, & Chatterjee K (2015). Resistance to thyroid hormone due to defective thyroid receptor alpha. Best Practice & Research. Clinical Endocrinology & Metabolism, 29, 647–657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muncan V, Heijmans J, Krasinski SD, Buller NV, Wildenberg ME, Meisner S, et al. (2011). Blimp1 regulates the transition of neonatal to adult intestinal epithelium. Nature Communications, 2, 452. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Na W, Fu L, Luu N, & Shi YB (2020). Direct activation of tRNA methyltransferase-like 1 (Mettl1) gene by thyroid hormone receptor implicates a role in adult intestinal stem cell development and proliferation during Xenopus tropicalis metamorphosis. Cell & Bioscience, 10, 60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakade S, Tsubota T, Sakane Y, Kume S, Sakamoto N, Obara M, et al. (2014). Microhomology-mediated end-joining-dependent integration of donor DNA in cells and animals using TALENs and CRISPR/Cas9. Nature Communications, 5, 5560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakajima K, Tazawa I, & Shi YB (2019). A unique role of thyroid hormone receptor beta in regulating notochord resorption during Xenopus metamorphosis. General and Comparative Endocrinology, 277, 66–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakajima K, Tazawa I, & Yaoita Y (2018). Thyroid hormone receptor alpha- and beta-knockout Xenopus tropicalis tadpoles reveal subtype-specific roles during development. Endocrinology, 159, 733–743. [DOI] [PubMed] [Google Scholar]
- Nakajima K, & Yaoita Y (2003). Dual mechanisms governing muscle cell death in tadpole tail during amphibian metamorphosis. Developmental Dynamics, 227, 246–255. [DOI] [PubMed] [Google Scholar]
- Nakayama T, Fish MB, Fisher M, Oomen-Hajagos J, Thomsen GH, & Grainger RM (2013). Simple and efficient CRISPR/Cas9-mediated targeted mutagenesis in Xenopus tropicalis. Genesis, 51, 835–843. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nieminen AI, Partanen JI, & Klefstrom J (2007). c-Myc blazing a trail of death: Coupling of the mitochondrial and death receptor apoptosis pathways by c-Myc. Cell Cycle, 6, 2464–2472. [DOI] [PubMed] [Google Scholar]
- Nieuwkoop PD, & Faber J (1965). Normal table of Xenopus laevis. Amsterdam: North Holland Publishing. [Google Scholar]
- Okada M, Miller TC, Fu L, & Shi YB (2015). Direct activation of amidohydrolase domain-containing 1 gene by thyroid hormone implicates a role in the formation of adult intestinal stem cells during Xenopus metamorphosis. Endocrinology, 156, 3381–3393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okada M, Miller TC, Wen L, & Shi YB (2017). A balance of Mad and Myc expression dictates larval cell apoptosis and adult stem cell development during Xenopus intestinal metamorphosis. Cell Death & Disease, 8, e2787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okada M, & Shi YB (2018a). The balance of two opposing factors Mad and Myc regulates cell fate during tissue remodeling. Cell & Bioscience, 8, 51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okada M, & Shi YB (2018b). EVI and MDS/EVI are required for adult intestinal stem cell formation during postembryonic vertebrate development. The FASEB Journal, 32, 431–439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okada M, Wen L, Miller TC, Su D, & Shi YB (2015). Molecular and cytological analyses reveal distinct transformations of intestinal epithelial cells during Xenopus metamorphosis. Cell & Bioscience, 5, 74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O’Malley BW, Malovannaya A, & Qin J (2012). Minireview: Nuclear receptor and coregulator proteomics—2012 and beyond. Molecular Endocrinology, 26, 1646–1650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Packham G, & Cleveland JL (1995). c-Myc and apoptosis. Biochimica et Biophysica Acta, 1242, 11–28. [DOI] [PubMed] [Google Scholar]
- Paul BD, Buchholz DR, Fu L, & Shi Y-B (2005). Tissue- and gene-specific recruitment of steroid receptor coactivator-3 by thyroid hormone receptor during development. The Journal of Biological Chemistry, 280, 27165–27172. [DOI] [PubMed] [Google Scholar]
- Paul BD, Buchholz DR, Fu L, & Shi Y-B (2007). SRC-p300 coactivator complex is required for thyroid hormone induced amphibian metamorphosis. The Journal of Biological Chemistry, 282, 7472–7481. [DOI] [PubMed] [Google Scholar]
- Paul BD, Fu L, Buchholz DR, & Shi Y-B (2005). Coactivator recruitment is essential for liganded thyroid hormone receptor to initiate amphibian metamorphosis. Molecular and Cellular Biology, 25, 5712–5724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pelengaris S, Rudolph B, & Littlewood T (2000). Action of Myc in vivo—Proliferation and apoptosis. Current Opinion in Genetics & Development, 10, 100–105. [DOI] [PubMed] [Google Scholar]
- Perissi V, Jepsen K, Glass CK, & Rosenfeld MG (2010). Deconstructing repression: Evolving models of co-repressor action. Nature Reviews. Genetics, 11, 109–123. [DOI] [PubMed] [Google Scholar]
- Plateroti M, Chassande O, Fraichard A, Gauthier K, Freund JN, Samarut J, et al. (1999). Involvement of T3Ralpha- and beta-receptor subtypes in mediation of T3 functions during postnatal murine intestinal development. Gastroenterology, 116, 1367–1378. [DOI] [PubMed] [Google Scholar]
- Plateroti M, Gauthier K, Domon-Dell C, Freund JN, Samarut J, & Chassande O (2001). Functional interference between thyroid hormone receptor alpha (TRalpha) and natural truncated TRDeltaalpha isoforms in the control of intestine development. Molecular and Cellular Biology, 21, 4761–4772. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Plateroti M, Kress E, Mori JI, & Samarut J (2006). Thyroid hormone receptor alpha1 directly controls transcription of the beta-catenin gene in intestinal epithelial cells. Molecular and Cellular Biology, 26, 3204–3214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Refetoff S, Weiss RE, & Usala SJ (1993). The syndromes of resistance to thyroid hormone. Endocrine Reviews, 14, 348–399. [DOI] [PubMed] [Google Scholar]
- Sachs LM (2015). Unliganded thyroid hormone receptor function: Amphibian metamorphosis got TALENs. Endocrinology, 156, 409–410. [DOI] [PubMed] [Google Scholar]
- Sachs LM, Amano T, Rouse N, & Shi YB (2001). Involvement of histone deacetylase at two distinct steps in gene regulation during intestinal development in Xenopus laevis. Developmental Dynamics, 222, 280–291. [DOI] [PubMed] [Google Scholar]
- Sachs LM, Amano T, & Shi YB (2001). An essential role of histone deacetylases in postembryonic organ transformations in Xenopus laevis. International Journal of Molecular Medicine, 8, 595–601. [DOI] [PubMed] [Google Scholar]
- Sachs LM, Damjanovski S, Jones PL, Li Q, Amano T, Ueda S, et al. (2000). Dual functions of thyroid hormone receptors during Xenopus development. Comparative Biochemistry and Physiology. Part B, Biochemistry & Molecular Biology, 126, 199–211. [DOI] [PubMed] [Google Scholar]
- Sachs LM, Jones PL, Havis E, Rouse N, Demeneix BA, & Shi Y-B (2002). N-CoR recruitment by unliganded thyroid hormone receptor in gene repression during Xenopus laevis development. Molecular and Cellular Biology, 22, 8527–8538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sachs LM, & Shi Y-B (2000). Targeted chromatin binding and histone acetylation in vivo by thyroid hormone receptor during amphibian development. PNAS, 97, 13138–13143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sakane Y, Iida M, Hasebe T, Fujii S, Buchholz DR, Ishizuya-Oka A, et al. (2018). Functional analysis of thyroid hormone receptor beta in Xenopus tropicalis founders using CRISPR-Cas. Biology Open, 7, bio030338. 10.1242/bio.030338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sancho E, Eduard Batlle E, & Clevers H (2004). Signaling pathways in intestinal development and cancer. Annual Review of Cell and Developmental Biology, 20, 695–723. [DOI] [PubMed] [Google Scholar]
- Schreiber AM, Cai L, & Brown DD (2005). Remodeling of the intestine during metamorphosis of Xenopus laevis. Proceedings of the National Academy of Sciences of the United States of America, 102, 3720–3725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreiber AM, Das B, Huang H, Marsh-Armstrong N, & Brown DD (2001). Diverse developmental programs of Xenopus laevis metamorphosis are inhibited by a dominant negative thyroid hormone receptor. PNAS, 98, 10739–10744. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreiber AM, Mukhi S, & Brown DD (2009). Cell-cell interactions during remodeling of the intestine at metamorphosis in Xenopus laevis. Developmental Biology, 331, 89–98. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheppard HM, Harries JC, Hussain S, Bevan C, & Heery DM (2001). Analysis of the steroid receptor coactivator 1 (SRC1)-CREB binding protein interaction interface and its importance for the function of SRC1. Molecular and Cellular Biology, 21, 39–50. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi Y-B (1994). Molecular biology of amphibian metamorphosis: A new approach to an old problem. Trends in Endocrinology and Metabolism, 5, 14–20. [DOI] [PubMed] [Google Scholar]
- Shi Y-B (1999). Amphibian metamorphosis: From morphology to molecular biology. New York: John Wiley & Sons, Inc. [Google Scholar]
- Shi Y-B (2009). Dual functions of thyroid hormone receptors in vertebrate development: The roles of histone-modifying cofactor complexes. Thyroid, 19, 987–999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi YB (2013). Unliganded thyroid hormone receptor regulates metamorphic timing via the recruitment of histone deacetylase complexes. Current Topics in Developmental Biology, 105, 275–297. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi YB (2018). Thyroid hormone and intestinal tumor: A Wnt connection. Oncotarget, 9, 31941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi Y-B, & Brown DD (1993). The earliest changes in gene expression in tadpole intestine induced by thyroid hormone. The Journal of Biological Chemistry, 268, 20312–20317. [PubMed] [Google Scholar]
- Shi Y, Glynn JM, Guilbert LJ, Cotter TG, Bissonnette RP, & Green DR (1992). Role for c-myc in activation-induced apoptotic cell death in T cell hybridomas. Science, 257, 212–214. [DOI] [PubMed] [Google Scholar]
- Shi YB, Hasebe T, Fu L, Fujimoto K, & Ishizuya-Oka A (2011). The development of the adult intestinal stem cells: Insights from studies on thyroid hormone-dependent amphibian metamorphosis. Cell & Bioscience, 1, 30. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi Y-B, & Ishizuya-Oka A (1996). Biphasic intestinal development in amphibians: Embryogenesis and remodeling during metamorphosis. Current Topics in Developmental Biology, 32, 205–235. [DOI] [PubMed] [Google Scholar]
- Shi YB, Matsuura K, Fujimoto K, Wen L, & Fu L (2012). Thyroid hormone receptor actions on transcription in amphibia: The roles of histone modification and chromatin disruption. Cell & Bioscience, 2, 42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi Z, Wang F, Cui Y, Liu Z, Guo X, Zhang Y, et al. (2015). Heritable CRISPR/ Cas9-mediated targeted integration in Xenopus tropicalis. The FASEB Journal, 29, 4914–4923. [DOI] [PubMed] [Google Scholar]
- Shibata Y, Tanizaki Y, & Shi YB (2020). Thyroid hormone receptor beta is critical for intestinal remodeling during Xenopus tropicalis metamorphosis. Cell & Bioscience, 10, 46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shibata Y, Wen L, Okada M, & Shi YB (2020). Organ-specific requirements for thyroid hormone receptor ensure temporal coordination of tissue-specific transformations and completion of xenopus metamorphosis. Thyroid, 30, 300–313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sirakov M, Kress E, Nadjar J, & Plateroti M (2014). Thyroid hormones and their nuclear receptors: New players in intestinal epithelium stem cell biology? Cellular and Molecular Life Sciences, 71, 2897–2907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sterling J, Fu L, Matsuura K, & Shi Y-B (2012). Cytological and morphological analyses reveal distinct features of intestinal development during Xenopus tropicalis metamorphosis. PLoS One, 7, e47407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart MD, Li J, & Wong J (2005). Relationship between histone H3 lysine 9 methylation, transcription repression, and heterochromatin protein 1 recruitment. Molecular and Cellular Biology, 25, 2525–2538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stewart D, Tomita A, Shi YB, & Wong J (2006). Chromatin immunoprecipitation for studying transcriptional regulation in Xenopus oocytes and tadpoles. Methods in Molecular Biology, 322, 165–181. [DOI] [PubMed] [Google Scholar]
- Stolow MA, & Shi YB (1995). Xenopus sonic hedgehog as a potential morphogen during embryogenesis and thyroid hormone-dependent metamorphosis. Nucleic Acids Research, 23, 2555–2562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun G, Fu L, & Shi Y-B (2014). Epigenetic regulation of thyroid hormone-induced adult intestinal stem cell development during anuran metamorphosis. Cell & Bioscience, 4, 73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun G, Fu L, Wen L, & Shi YB (2014). Activation of sox3 gene by thyroid hormone in the developing adult intestinal stem cell during Xenopus metamorphosis. Endocrinology, 155, 5024–5032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun G, Heimeier RA, Fu L, Hasebe T, Das B, Ishizuya-Oka A, et al. (2013). Expression profiling of intestinal tissues implicates tissue-specific genes and pathways essential for thyroid hormone-induced adult stem cell development. Endocrinology, 154, 4396–4407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun G, & Shi Y-B (2012). Thyroid hormone regulation of adult intestinal stem cell development: Mechanisms and evolutionary conservations. International Journal of Biological Sciences, 8, 1217–1224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanizaki Y, Shibata Y, Zhang H, & Shi Y-B (2020). Analysis of thyroid hormone receptor α knockout tadpoles reveals that the activation of cell cycle program is involved in thyroid hormone-induced larval epithelial cell death and adult intestinal stem cell development during Xenopus tropicalis metamorphosis. Thyroid, 31, 128–142. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tata JR (1993). Gene expression during metamorphosis: An ideal model for post-embryonic development. BioEssays, 15, 239–248. [DOI] [PubMed] [Google Scholar]
- Thompson EB (1998). The many roles of c-Myc in apoptosis. Annual Review of Physiology, 60, 575–600. [DOI] [PubMed] [Google Scholar]
- Tomita A, Buchholz DR, & Shi Y-B (2004). Recruitment of N-CoR/ SMRT-TBLR1 corepressor complex by unliganded thyroid hormone receptor for gene repression during frog development. Molecular and Cellular Biology, 24, 3337–3346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Toner PG, Carr KE, & Wyburn GM (1971). The digestive system: An ultrastructural atlas and review. London: Butterworth. [Google Scholar]
- Tsai MJ, & O’Malley BW (1994). Molecular mechanisms of action of steroid/thyroid receptor superfamily members. Annual Review of Biochemistry, 63, 451–486. [DOI] [PubMed] [Google Scholar]
- van der Flier LG, & Clevers H (2009). Stem cells, self-renewal, and differentiation in the intestinal epithelium. Annual Review of Physiology, 71, 241–260. [DOI] [PubMed] [Google Scholar]
- van Mullem A, van Heerebeek R, Chrysis D, Visser E, Medici M, Andrikoula M, et al. (2012). Clinical phenotype and mutant TRalpha1. The New England Journal of Medicine, 366, 1451–1453. [DOI] [PubMed] [Google Scholar]
- Wang X, Matsuda H, & Shi Y-B (2008). Developmental regulation and function of thyroid hormone receptors and 9-cis retinoic acid receptors during Xenopus tropicalis metamorphosis. Endocrinology, 149, 5610–5618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang F, Shi Z, Cui Y, Guo X, Shi YB, & Chen Y (2015). Targeted gene disruption in Xenopus laevis using CRISPR/Cas9. Cell & Bioscience, 5, 15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wen L, Fu L, & Shi YB (2017). Histone methyltransferase Dot1L is a coactivator for thyroid hormone receptor during Xenopus development. The FASEB Journal, 31, 4821–4831. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wen L, Hasebe T, Miller TC, Ishizuya-Oka A, & Shi YB (2015). A requirement for hedgehog signaling in thyroid hormone-induced postembryonic intestinal remodeling. Cell & Bioscience, 5, 13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wen L, & Shi YB (2015). Unliganded thyroid hormone receptor alpha controls developmental timing in Xenopus tropicalis. Endocrinology, 156, 721–734. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wen L, & Shi YB (2016). Regulation of growth rate and developmental timing by Xenopus thyroid hormone receptor alpha. Development, Growth & Differentiation, 58, 106–115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wen L, Shibata Y, Su D, Fu L, Luu N, & Shi Y-B (2017). Thyroid hormone receptor α controls developmental timing and regulates the rate and coordination of tissue specific metamorphosis in Xenopus tropicalis. Endocrinology, 158, 1985–1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong J, Patterton D, Imhof D, Guschin D, Shi Y-B, & Wolffe AP (1998). Distinct requirements for chromatin assembly in transcriptional repression by thyroid hormone receptor and histone deacetylase. The EMBO Journal, 17, 520–534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wong J, Shi YB, & Wolffe AP (1995). A role for nucleosome assembly in both silencing and activation of the Xenopus TR beta A gene by the thyroid hormone receptor. Genes & Development, 9, 2696–2711. [DOI] [PubMed] [Google Scholar]
- Wong J, Shi Y-B, & Wolffe AP (1997). Determinants of chromatin disruption and transcriptional regulation instigated by the thyroid hormone receptor: Hormone-regulated chromatin disruption is not sufficient for transcriptional activation. The EMBO Journal, 16, 3158–3171. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yen PM (2001). Physiological and molecular basis of thyroid hormone action. Physiological Reviews, 81, 1097–1142. [DOI] [PubMed] [Google Scholar]
- Yen PM (2015). Unliganded TRs regulate growth and developmental timing during early embryogenesis: Evidence for a dual function mechanism of TR action. Cell & Bioscience, 5,8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yoon H-G, Chan DW, Huang ZQ, Li J, Fondell JD, Qin J, et al. (2003). Purification and functional characterization of the human N-CoR complex: The roles of HDAC3, TBL1 and TBLR1. The EMBO Journal, 22, 1336–1346. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Young JJ, Cherone JM, Doyon Y, Ankoudinova I, Faraji FM, Lee AH, et al. (2011). Efficient targeted gene disruption in the soma and germ line of the frog Xenopus tropicalis using engineered zinc-finger nucleases. Proceedings of the National Academy of Sciences of the United States of America, 108, 7052–7057. [DOI] [PMC free article] [PubMed] [Google Scholar]


