Abstract
There are substantial sex differences in substance use disorders (SUDs), and a key feature of SUD is pathologically high economic demand for drug. The hypothalamic neuropeptide oxytocin (OXT) is heavily implicated in the modern treatment of SUDs. Using a within-session threshold behavioral economics (BE) procedure, we quantified demand elasticity (a, inverse motivation) and free consumption (Q0) in male and female rats to investigate the effect of OXT on cocaine demand. Results showed that OXT decreased motivation for cocaine; an effect greater during the high-demand phase (diestrus, low progesterone, P4) vs low demand phases (proestrus, high P4). We confirmed our prior findings that P4 attenuates cocaine demand in female rats and that chronic cocaine self-administration disrupts estrus cyclicity. Following each injection, OXT at either 0.1mg/kg or 0.3mg/kg restored estrous cycling in intact females with prior cocaine experience for one week and remained effective with up to 4 weeks of injections. Fos reactivity in OXT+ neurons was greater when rats were in proestrus compared to diestrus and significantly correlated to motivation and circulating levels of P4. Finally, using ovariectomized females with P4 replacement, we show that P4’s demand attenuating effects are reversed by atosiban (1.0 mg/kg, IP), an OXT antagonist. These data show an interaction between oxytocin and progesterone in female rats that may underlie differences in cocaine demand between sexes. Additionally, we show critical periods for using OXT as a treatment to reduce cocaine demand in females. Our results indicate novel therapeutic treatments for SUDs must be tailored to hormonal states.
Keywords: Sex differences, Oxytocin, Behavioral economics, Progesterone, Cocaine
Introduction
There is growing evidence for sex differences in substance use disorders (SUDs) with important treatment implications. In particular, there is a relationship between stressful life events, stress reactivity and SUDs that differs by gender [1–4]. Abundant evidence indicates that women progress more quickly from casual drug use to dependence [5], have greater difficulty quitting [6], and shorter periods of abstinence [7, 8]. These behavioral sex differences in SUDs have been well modelled in rodents. Female rats exhibit enhanced behavioral sensitization to cocaine [9] and acquire cocaine conditioning [10] and self-administration more rapidly and at lower doses than males [11–13]. In animal models of relapse, female rats demonstrate greater context-induced cocaine seeking [11,14,15], greater extinction resistance [16], and greater cocaine-primed [17,18] and stress-induced [19] reinstatement of cocaine seeking than males. Thus, mechanisms driving SUDs in women are of particular interest and are well modelled in female rodents.
Prior studies show that women have increased drug-seeking behaviors during menstrual cycle phases with high estradiol (E2) levels [20]. Conversely, progesterone (P4) may attenuate some of the subjective effects of cocaine in women [21]. Pre-clinical studies have replicated the effects of E2 and P4 on drug-seeking behaviors in female rats [9,22]. A recent publication from our lab using the within-session behavioral economics (BE) paradigm distinguished between the intake-promoting effects of estradiol and the demand-attenuating effects of P4, within-subject, while both steroids were high [23]. Furthermore, we showed that cocaine’s endocrine disrupting effects to suppress proestrus epochs may drive extinction resistance and potentiate demand in female rats [23]. Thus, the BE model when used in female rats permits measuring demand while controlling for intake, to more accurately recapitulate behavioral-endocrine effects of cocaine observed in women.
Oxytocin (OXT) is a neurohormone that is released into the neurohypophysis from the paraventricular (PVN) and supraoptic nuclei (SON). There also are central OXT projections from PVN to brain nuclei implicated in SUDs, including the ventral tegmental area, amygdala, nucleus accumbens and prefrontal cortex [24]. In addition, exogenous OXT has rewarding effects on social behavior in human [25] and produces a conditioned place preference (CPP) in animals [26]. As well, OXT directly infused into nucleus accumbens, amygdala, or hippocampus attenuates morphine tolerance and dependence [27]. OXT administration also reduces cocaine-induced hyperactivity [28] and reinstatement/seeking [16,29,30] in both sexes, and increases demand elasticity (decreases motivation) in males [31]. Together, these data provide strong support for the involvement of OXT signaling in SUD-like behaviors. Here, we extend this body of results by examining interactions between OXT and estrous cycle phase on cocaine use disorder (CUD)-like behaviors in females.
Methods
Animals
Female (225–300 g, n = 23) and male (325–350 g, n = 7) Sprague-Dawley rats (Envigo; ~ 55–60 days of age) were singly housed under a reversed 12 h:12 h light/dark cycle (lights off 0600 h); all experiments were performed during the active cycle. Rats had free access to food and water and were housed in animal facilities at Rutgers University. All experiments were approved by the Institutional Animal Care and Use Committees and conducted in accordance with the National Institutes of Health specifications outlined in their Guide for the Care and Use of Laboratory Animals: Eighth Edition (2011).
Estrous cycle determination
Estrous cycle phases were recorded daily via vaginal cytology. Vaginal cytology and behavioral testing were performed in the afternoon (1–4pm) of the reverse light-dark cycle to capture afternoon peaking P4 levels in proestrus as per prior reports [11,23,32]. Cocaine effects on the estrous cycle were tracked as in our prior report [23].
Jugular catheter implantation
At ~55 days of age, all rats underwent implantation of chronic indwelling catheters. The rats were anesthetized using an intraperitoneal injection of ketamine (56.5 mg/kg) and xylazine (8.7 mg/kg), per previously reported methods [16,23]. Post-operative care included the administration of Rimadyl (1 mg/kg, ip) for analgesia over the course of 3 days. Catheters were maintained by daily flushing with cefazolin (0.1 ml; 100 mg/ml) and heparin (0.1 ml; 100 U/ml). The rats were given a 1-week recovery period before beginning self-administration training.
Ovariectomy
Ovariectomy (OVX) was carried out as previously described [23,33]. Rats were anesthetized with ketamine/xylazine (56.5/8.7 mg/kg, IP) and small incisions were made in the skin and abdominal wall of the rats’ dorsal sides. The fallopian tube was ligated, and the ovaries were isolated and removed. Rats were administered meloxicam (Mobic) daily as an analgesic. OVX was performed in the same surgical session as jugular catherization. Self-administration began one week following surgical recovery.
Hormone-replacement
OVX rats received safflower oil-vehicle or hormone replacement with E2 and/or P4 before BE testing. Administration of E2 (0.09 mg/kg, S.C.) occurred 44–48 h before BE testing, and administration of P4 (4.0 mg/kg S.C.) occurred 4–6 h before BE testing as per prior reports [23,33]. Rats had 7 days of washout between hormone treatments while continuing BE sessions daily.
Drugs
Cocaine hydrochloride (NIDA, Research Triangle Park, NC) was dissolved in 0.9 % sterile saline. Oxytocin (OXT; Tocris, Minneapolis, MN; Cat # 1910) was dissolved in 0.9 % saline and administered at 0.1 or 0.3 mg/kg, intraperitoneally (IP), 30 min prior to testing in all behavioral paradigms. Atosiban (Tocris, Minneapolis, MN; Cat #6332; 1.0 mg/mL) was dissolved in saline and administered 30 min prior to testing. P4 (Tocris, Minneapolis, MN; Cat #2835; 4.0 mg/mL) and E2 (Tocris, Minneapolis, MN; Cat #2824; 0.09 mg/mL) were dissolved with heat in safflower oil and administered as described above.
Cocaine self-administration
Self-administration sessions were conducted in standard operant conditioning chambers housed in sound attenuating cubicles and controlled via MED-PC IV software (Med-Associates, St Albans, VT) as described previously [11,14,16,23]. Rats were trained in daily 2-h sessions to press an active lever for intravenous cocaine (0.2 mg/50 μl infusion for males, 0.16 mg/50 μl infusion for females). Different doses in infusions allowed females and males to achieve the same average number of lever presses, and cocaine-cue pairings, across the last 3 days of self-administration [11,16,23]. Rats were trained for at least 10 days on a fixed ratio 1 (FR-1) schedule of reinforcement to reach a criterion of > 10 cocaine infusions/day wherein light + tone cocaine-associated cues were presented with active lever presses. Each cocaine infusion was followed by a 20 s time-out period in which lever pressing produced neither cocaine nor cues. An inactive lever was also present; presses on it were tabulated but had no consequence.
Within-session behavioral economics (BE) protocol
Within-session BE training used procedures described in our previous papers [23,31,34,35]. BE sessions were conducted 5 day/week and consisted of an FR-1 schedule of reinforcement in a descending series of cocaine doses (421, 237, 133, 75, 41, 24, 13, 7.5, 4.1, 2.4 and 1.3 ug/injection) that were produced by holding the concentration constant and adjusting pump duration. Each infusion was paired with both a stimulus light and tone. The initial 10-min bin was a ‘loading bin’ wherein rats had free access to high-dose cocaine (0.4 mg/injection) to allow animals to establish their preferred brain cocaine concentration at low effort. As bins progressed, rats exerted more effort (more presses) to maintain their cocaine intake until the unit price to defend their baseline cocaine concentration became too expensive (required too much effort/pressing) and the maximum price point was met (PMax). An exponential demand equation [35,36] was used to fit a demand curve to the data and extract important BE parameters, particularly α (demand elasticity) and Q0 (free cocaine consumption). Rats performed daily BE sessions until they exhibited stable behavior, i.e., until α and Q0 values over the last 3 sessions (3 estrous cycles in intact females [23]) were within 20 % of their means. Rats displayed stable (baseline) α and Q0 typically within 6 BE sessions (males, OVX females) or 3 cycles (3 weeks, females). To control for duration of cocaine exposure across groups, all rats were stabilized for 3 weeks minimum prior to the start of testing.
Sucrose self-administration
Female (n = 8) rats were trained in an operant conditioning chamber to lever press on an FR-1 schedule for sucrose pellets. Following a minimum of 10 days of FR-1 for sucrose, rats were tested in a counter-balanced fashion for the effects of OXT on sucrose FR-1 in either proestrus or diestrus. Rats were administered saline vehicle, 0.1 or 0.3 mg/kg OXT, IP, 30 mins prior to testing.
Immunohistochemistry
Immunohistochemistry was carried out as previously described [16, 37]. Sections serially incubated with a rabbit primary antibody to Fos (1:10,000 Synaptic Systems, Goettingen, Germany, Cat # 226–003) then biotinylated donkey anti-rabbit secondary antibody (1:500, Jackson ImmunoResearch Laboratories, West Grove, PA). Sections were visualized via 32,32-diaminobenzidine (DAB) with 0.0002 % H2O2 in 0.05 M Tris buffer plus nickel ammonium sulfate for ~ 2 min to produce a purple reaction product [11,16,37]. Next, sections were incubated with a mouse primary antibody to OXT (1:5000 MAB5296, EMD Millipore, Burlington, MA) then in biotinylated donkey anti-mouse secondary antibody (1:500, Jackson ImmunoResearch Laboratories, West Grove, PA). Sections were visualized for OXT via 32,32-diaminobenzidine (DAB) with 0.0002 % H2O2 in 0.05 M Tris buffer without nickel ammonium sulfate for ~ 4.5 min to produce a brown reaction product, and then mounted, dehydrated, and cover slipped for imaging. The rat paraventricular nucleus (PVN) was separated into 3 the rostral (PVNR; Fig. 1A), mid (PVNM; Fig. 1B), and caudal (PVNC; Fig. 1C) planes [38]. The supraoptic nucleus (SON; Fig. 1D), which projects peripherally, was also analyzed in the same rats. At least three sections per rat per region were analyzed for Fos+ and OXT+ labeling in the same sections.
Fig. 1.

Representative images from frontal brain sections of OXT and Fos expression in the paraventricular nucleus (PVN). In all images, brown staining indicates OXT-positive neurons (brown arrows), and purple staining indicates Fos-positive nuclei (red arrows). In all images, black arrows indicate double-labeled neurons. PVN subregions were identified using landmarks established by Swanson and Sawchenko (1980). A. PVNr. Representative image of OXT/Fos dual labeling in PVNr (AP – 1.60; Paxinos and Watson 2007). B. PVNm. Representative images of OXT/Fos dual labeling in PVNm (AP – 1.88; Paxinos and Watson 2007). C. PVNc. Representative images of OXT/Fos dual labeling in PVNc (AP – 2.12; Paxinos and Watson 2007) D. Representative image of SON OXT/Fos dual labeling (AP – 1.80; Paxinos and Watson 2007).
Statistical analyses
We used repeated measures two-way analysis of variance (ANOVA) to determine interactions between estrous cycle phase and oxytocin dose to affect cocaine demand parameters. We used repeated measures one-way ANOVAs to assess the effects of cocaine self-administration on estrous cycle over time. Pearson R correlation analyses were used to assess the relationship between OXT neuron Fos+ cells and cocaine demand parameters. Post-hoc t-tests were used to determine effects of estrus cycle phase or OXT on cocaine demand where reported.
Results
Fos activation in oxytocin neurons at maximal effort for cocaine is region specific
First, female Sprague-Dawley rats (n = 15) were trained to self-administer cocaine (Fig. 2A) as described in Methods. Rats were then subjected to BE training, as also described in Methods. Following BE stabilization (~ 3 weeks), rats performed a final BE test in either proestrus or diestrus and were sacrificed 90 min following the point of maximal effort, PMax. Rats in proestrus had higher α values (lower motivation) for cocaine compared to diestrus females, as per prior reports (t = 3.116, df =18, p = 0.0060; [23]). No significant differences were found in Q0 (free consumption) between diestrus and proestrus females (t = 0.6806, df=17, p = 0.5053; [23]).
Fig. 2.
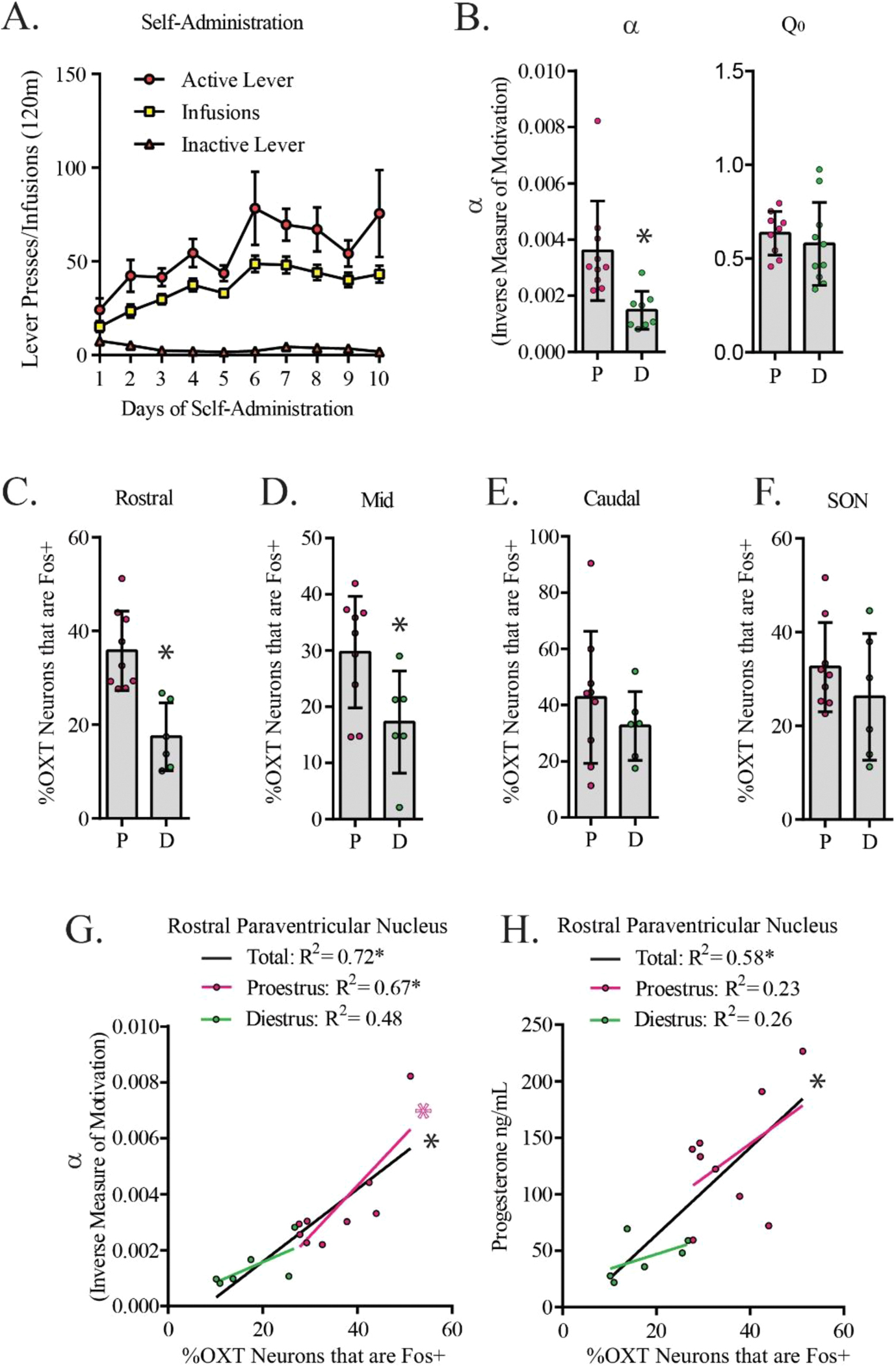
The percentage of Oxytocin/Fos+ neurons in the rostral paraventricular nucleus negatively predicts economic demand for cocaine in intact female rats. A. Self-administration behaviors over the 10 days of FR-1 acquisition prior to starting behavioral economics training. B. Demand elasticity (α) and consumption at null effort (Q0) in proestrus (P) and diestrus (D) rats. Demand elasticity (α; inverse to motivation for cocaine) was lower when females were in diestrus compared to when they were in proestrus. No significant differences were observed in Q0. *p < 0.05. C. Female rats in diestrus have significantly less Fos+ OXT neurons in the rostral region of the paraventricuar nucleus (PVNR). * p < 0.05. D. Female rats in diestrus have significantly less Fos+ OXT neurons in the middle region of the paraventricuar nucleus (PVNM). * p < 0.05. E. There were no significant differences between cycle phases in Fos+ OXT neurons of the caudal region of the paraventricular nucleus (PVNC). F. There were no significant differences between cycle phases in Fos+ neuron reactivity in OXT neurons of the supraoptic nucleus (SON). G. Fos+ OXT neurons in the PVNR positively correlated to α values. Linear regressions are plotted (black lines for collapsed females across cycle, pink lines and symbols for females in proestrus, green lines and symbols for females in diestrus). * p < 0.05. H. Fos+ OXT neurons in the PVNR positively correlated to circulating progesterone levels. Linear regressions are plotted (black lines for collapsed females across cycle, pink lines and symbols for females in proestrus, green lines and symbols for females in diestrus). * p < 0.05.
Immunohistochemistry was performed on PVN sections from female rats sacrificed 90 min after PMax while in either proestrus or diestrus; Fig. 1 shows sample images. We found that the percentage of OXT neurons that were Fos+ (OXT/Fos+) was lower when females were in diestrus compared to proestrus in PVNR (t = 4.340, df =13, p = 0.0008; Fig. 2C) and PVNM (t = 2.461, df=13, p = 0.0286; Fig. 2D). Although trends were apparent, there were no significant effects of cycle phase on OXT/Fos+ neurons in the PVNC (t = 0.9695, df=13, p = 0.3500; Fig. 2E) or SON (t = 1.072, df=13, p = 0.3033; Fig. 2F). Using multiple regression, we found that variances in α values (Fig. 2G) and serum progesterone (Fig. 2H) are explained by a significant amount of the variance in Fos+ OXT neurons in PVNR (F(2, 14) = 23.10, p < 0.0001, R2 = 0.89, R2Adjusted = 0.76), but not in PVNm or PVNc.
Oxytocin reduces economic demand for cocaine at lower doses in females than in males
We next investigated the therapeutic potential of OXT to reduce demand for cocaine across cycle phases and between sexes. We previously found that high doses of OXT (i.e. 1.0mg/kg, IP) reduced motivation for cocaine in male rats [31]. Here we found that, independent of cycle, low dose OXT (e.g. 0.1 and 0.3 mg/kg) increased α (reduced motivation) in females, but not males. A two-way ANOVA comparing effects of OXT dose and cycle phase on demand elasticity indicated a main effect of OXT to increase α (reduce motivation) for cocaine at the 0.3mg/kg dose in particular, independent of cycle phase (F(2,42) = 16.74, p < 0.0001; Fig. 3A). There was an interaction between OXT dose and cycle phase to influence Q0, wherein 0.1 mg/kg OXT increased Q0 in proestrus females (F(2,42) = 3.569, p = 0.0489; Fig. 3B).
Fig. 3.
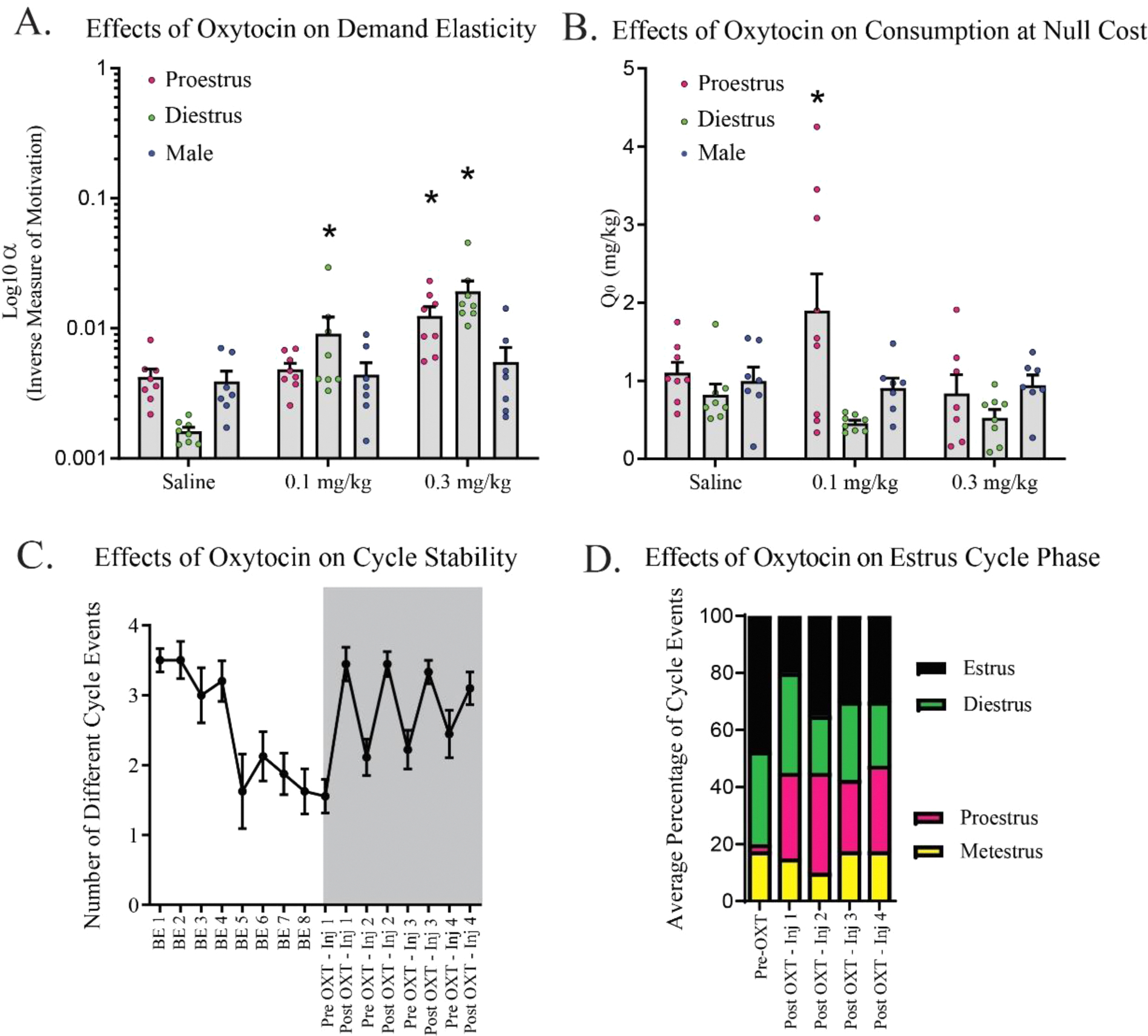
Oxytocin significantly decreases economic demand for cocaine in female rats and restores estrous cyclicity. A. Demand elasticity (α; inverse to motivation for cocaine) was significantly attenuated by oxytocin at 0.1 and 0.3 mg/kg while females were in diestrus, and 0.3 mg/kg only while in proestrus. * p < 0.05. B. Consumption at null effort (Q0) is increased by 0.1 mg/kg oxytocin only during proestrus. * p < 0.05. C. FR-1 or BE cocaine self-administration disrupted estrous cyclicity, as measured by the observed number of different cycle events. Oxytocin treatments occurred in the time period indicated by the grey overlay. On the week following oxytocin treatment, cycle events normalized as compared to the prior week on BE self-administration of cocaine. * p < 0.05. D. Color coded bar graph depiciting the percentage of each cycle phase observed. Following OXT, proestrus epochs return to 25 % of observed cycle phases across subjects in each week. Statistics are described in text.
In a separate group of rats, we tested the motor effects of OXT on low-effort (FR-1) lever pressing behavior for sucrose. Rats were first trained to self-administer sucrose on an FR-1 schedule. Following stabilization, rats were administered OXT at 0.1 or 0.3mg/kg IP 30 mins prior to testing. There was a main effect of cycle phase wherein females self-administered more sucrose in diestrus than when they were in proestrus (F(1,42)=6.594, p = 0.0139), but we observed no effects of OXT at 0.1 or 0.3 mg/kg on sucrose self-administration in either cycle phase. There were no effects of cycle phase or OXT on inactive lever presses (F (2,42)=0.8595, p = 0.4307) or the number of pellet rewards obtained (F (2,42)=0.0415, p = 0.9593) in sucrose FR-1.
Oxytocin ameliorates some of the endocrine disrupting effects of cocaine
Cocaine substantially affects female hormone levels and estrous/menstrual cyclicity [23,25,32,39,40]. Herein, we recapitulate these results and show that the hormone cycle is disrupted after 8 weeks of BE testing with cocaine. With one-week washout on a randomized double-blind schedule of administration, we measured the effects of OXT treatment (0.1 or 0.3 mg ip) on estrous cyclicity during BE testing. We analyzed the effect of OXT on cycle stability during pre-treatment periods to that during OXT treatment periods, and post-treatment periods, over the following 8 weeks using a one-way repeated measures ANOVA with factor matching for rate (eg. comparing week 1 pre-treatment to week 1 post-treatment). There was an interaction between time and OXT dose during once weekly injections of OXT, such that the number of different cycle phases increased to pre-cocaine levels (F(7,63)=13.73, p < 0.0001; Fig. 3C). Interestingly, the rate of increase in cycle number with OXT treatment was more rapid than the rate at which cocaine disrupted estrous cyclicity over time (F(7,63)=13.71, p < 0.0001; Fig. 3D). A repeated measures two-way ANOVA comparing cycle phase epochs (diestrus, proestrus, estrus, metestrus) across OXT pre-treatment vs. post-treatment epochs revealed a significant effect of OXT to impact the estrous cycle (F(12,112)=5.113, p < 0.0001). Post-hoc tests using a Bonferroni correction for multiple comparisons revealed that this effect was driven selectively by OXT’s effect to restore proestrus epochs in the week following each injection (inj 1: df=7, t = 1.929, p = 0.0122; inj 2: df=7, t = 1.256, p = 0.0154; inj 3: df=7, t = 1.369, p = 0.0163; inj 4: df=7, t = 1.825, p = 0.0014; Fig. 3F).
Progesterone’s demand-attenuating effects may be dependent on oxytocin receptors
We previously reported that progesterone attenuates demand for cocaine, both when progesterone is endogenously high, and when exogenously administered to either intact or ovariectomized rats [23]. Therefore, we tested if progesterone’s demand-attenuating effects are dependent on OXT receptors using the OXT receptor antagonist atosiban (1.0mg/kg, IP) in ovariectomized rats. First, our results confirmed our prior findings [23] that progesterone attenuates motivation (increases α) in OVX rats (F(1, 5) =30.19, p = 0.0027; Fig. 4B). Furthermore, there was an interaction between progesterone and OXT administration in these subjects, such that progesterone’s demand-attenuating effects were partially diminished by co-administration of atosiban (F(1, 5) =34.24, p = 0.0021; post-hoc t: P4+veh vs veh+veh (df=6, t = 5.189, p = 0.0035), P4+veh vs P4+atosiban (df=6, t = 4.341, p = 0.0074); Fig. 4B). Similarly, we confirmed our prior finding [23] that progesterone increases Q0 in OVX females (F(1, 5) =19.98, p = 0.0066). We also found that atosiban diminishes this increase of Q0 by progesterone (F(1, 5) =9.887; p = 0.0225; post-hoc t: P4+veh vs veh+veh (df=6, t = 11.92, p < 0.0001), P4+veh vs P4+atosiban (df=6, t = 8.272, p = 0.0004); Fig. 4C). Atosiban in the absence of P4 had no effect on α (post-hoc t: veh+veh vs veh + atosiban (df=6, t = 0.003, p = 0.9975) nor did it affect Q0 (post-hoc t: veh+veh vs veh + atosiban (df=6, t = 0.1500, p = 0.8161).
Fig. 4.
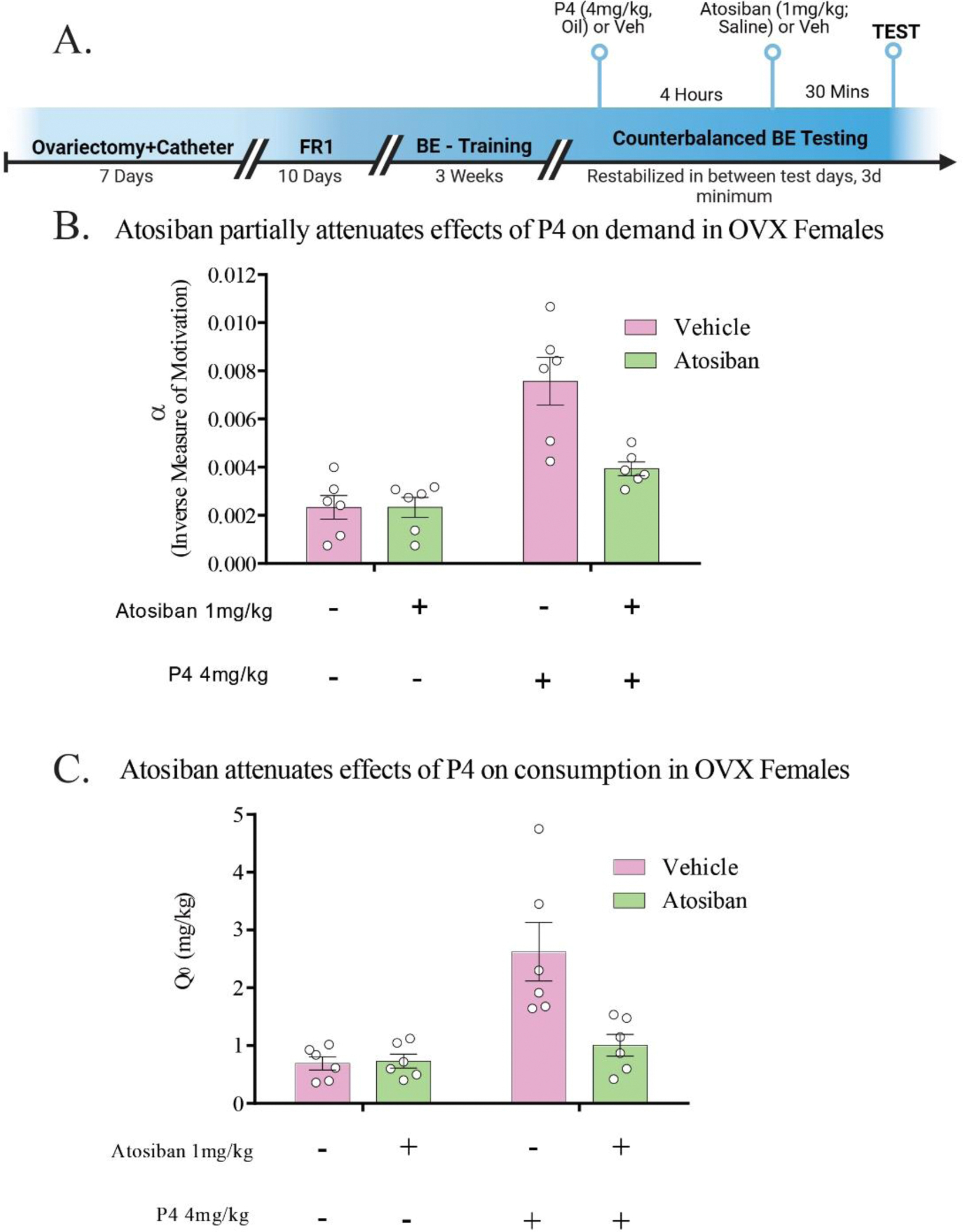
Atosiban, an oxytocin receptor antagonist, partially attenuates progesterone’s treatment efficacy on cocaine demand. A. Timeline depicting testing schedule. B. Progesterone administration increases demand elasticity (α; inverse to motivation for cocaine) in ovariectomized rats, an effect which is partially attenuated by co-administration with atosiban 1.0 mg/kg, an oxytocin antagonist. Atosiban did not impact α in the absence of progesterone. * p < 0.05. C. Cocaine consumption at null effort (Q0) was increased by administration of 4mg/kg progesterone, which was attenuated by co-administration of atosiban 1.0 mg/kg. Atosiban did not impact Q0 in the absence of progesterone. * p < 0.05.
Discussion
Our studies confirm and extend prior results indicating that female rats show higher motivation for cocaine than males, in a hormone-dependent manner. We show that OXT, a potential therapeutic for treating cocaine use disorder, may have greater efficacy for decreasing demand in females than in males. We confirm prior reports that show P4 decreases economic demand for cocaine and extend these findings to indicate that some of P4’s demand attenuating effects may be via interactions with neural oxytocin production and/or signaling.
OXT and P4 vary with endogenous hormonal milieu and are expressed in a region-specific manner within the hypothalamus. P4, P4 metabolites, and P4 receptors are found diffusely in the rat hypothalamus [41–45], and in the PVN throughout its subregions [41,42]. In addition, numerous studies reveal region-, sex-, and cycle-specific differences in expression of OXT and OXT receptors associated with stress and hormonal state. Most relevant to our studies, OXT mRNA expression in female rats is higher during proestrus than diestrus [46]. Importantly, rostral PVN–OXT neurons contain P4 receptors, whereas mid and caudal PVN–OXT neurons do not [44,46], and region-specific differences in hormone receptor expression in OXT neurons are found across species [43]. These data indicate that OXT neuron activity may play a role in motivational properties of cocaine, dependent on P4, consistent with effects of exogenous OXT administration as we observed here and in prior studies [31].
While our results further support and expand on research that indicates tight hypothalamic regulation of OXT neuron activity by P4, additional studies are needed to strengthen the proposed mechanism. Specifically, demonstrating P4’s effects on Fos expression in OXT neurons in OVX females to parallel our estrous phase Fos data would strengthen the interpretation of our findings. Similarly, understanding the interactions between P4, E2, and OXT in the absence of cocaine is crucial for comprehending the underlying mechanisms of this treatment. Future experiments can address this gap by directly assessing P4’s regulatory role in oxytocin neuron activity.
Our understanding of the interactions between P4 and OXT is primarily driven by peripheral responses. However, these relationships during normal daily conditions, or during dysfunctional states, are largely understudied. To add further complexity, within neural circuits prostaglandins are primarily produced by microglia, and P4 originates from neurons and astrocytes. As such, evidence on how these hormones interact in the reproductive tract may not translate to neurosteroidogenic responses. Notably, we observed a lack of effect of the OXT receptor antagonist, atosiban, in OVX females. This lack of effect may result from a floor effect, as ovarian hormone depletion reduces baseline oxytocinergic activity and receptor expression. Atosiban’s efficacy could be greater in intact, hormonally active rats, especially during proestrus. Future research should investigate atosiban’s potential to impact cocaine demand in intact, hormonally active rats, particularly during proestrus, to clarify the roles of OXT and P4 in neural and behavioral regulation of CUDs.
The complex interactions between central and peripheral hormone signaling, including neurosteroidogenic responses, remain understudied. Our results are supported by prior reports showing that acute dosing of cocaine stimulates P4 [40], whereas chronic administration suppresses the hypothalamic-pituitary-gonadal axis and thereby attenuates circulating P4 production long-term [23,39]. Our results underscore the important, bidirectional relationship between peripheral ovarian hormones and neural systems in CUDs. One challenge we face in interpreting our findings is determining the directionality of the effects between peripheral P4 and central OXT. Our data show that P4 impacts OXT, and OXT may also influence P4. Further studies are required to track the progression of changes in both systems to clarify how each system loses functionality in response to cocaine exposure and/or how functionality is restored in both systems following OXT administration. Investigating the interactions between peripheral P4 and neural OXT signaling in cocaine use is required, as it has significant implications for developing CUDs therapeutics in women.
Footnotes
Declaration of competing interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
CRediT authorship contribution statement
Amy S. Kohtz: Writing – review & editing, Writing – original draft, Visualization, Validation, Supervision, Project administration, Methodology, Investigation, Funding acquisition, Formal analysis, Data curation, Conceptualization. Hannah Davies: Writing – original draft, Validation, Methodology, Investigation, Formal analysis, Data curation. Belle Lin: Writing – original draft, Investigation, Data curation, Conceptualization. Gary Aston-Jones: Writing – review & editing, Supervision, Resources, Methodology, Funding acquisition, Data curation, Conceptualization.
Data availability
Data will be made available on request.
References
- [1].Sinha R, Catapano D, O’Malley S, Stress-induced craving and stress response in cocaine dependent individuals, Psychopharmacology 142 (1999) 343–351 (Berl.). [DOI] [PubMed] [Google Scholar]
- [2].Brady KT, Sinha R, Co-occurring mental and substance use disorders: the neurobiological effects of chronic stress, Am. J. Psychiat 162 (2005) 1483–1493. [DOI] [PubMed] [Google Scholar]
- [3].Sinha R, Chronic stress, drug use, and vulnerability to addiction, Ann. N. Y. Acad. Sci. 1141 (2008) 105–130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Sinha R, Fuse T, Aubin LR, O’Malley SS, Psychological stress, drug-related cues and cocaine craving, Psychopharmacology 152 (2000) 140–148 (Berl.). [DOI] [PubMed] [Google Scholar]
- [5].McCance-Katz EF, Carroll KM, Rounsaville BJ, Gender differences in treatment-seeking cocaine abusers - Implications for treatment and prognosis, Am. J. Addict. 8 (1999) 300–311. [DOI] [PubMed] [Google Scholar]
- [6].Becker JB, Hu M, Sex differences in drug abuse, Front. Neuroendocrinol. 29 (2008) 36–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Griffin ML, Weiss RD, Mirin SM, Lange U, A comparison of male and female cocaine abusers, Arch. Gen. Psychiatry 46 (1989) 122–126. [DOI] [PubMed] [Google Scholar]
- [8].Kosten TA, Gawin FH, Kosten TR, Rounsaville BJ, Gender differences in cocaine use and treatment response, J. Subst. Abuse Treat. 10 (1993) 63–66. [DOI] [PubMed] [Google Scholar]
- [9].Hu M, Becker JB, Effects of sex and estrogen on behavioral sensitization to cocaine in rats, J. Neurosci. Off. J. Soc. Neurosci. 23 (2003) 693–699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Russo SJ, Festa ED, Fabian SJ, Gazi FM, Kraish M, Jenab S, et al. , Gonadal hormones differentially modulate cocaine-induced conditioned place preference in male and female rats, Neuroscience 120 (2003) 523–533. [DOI] [PubMed] [Google Scholar]
- [11].Kohtz AS, Aston-Jones G, Cocaine seeking during initial abstinence is driven by noradrenergic and serotonergic signaling in hippocampus in a sex-dependent manner, Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 42 (2017) 408–418. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Lynch WJ, Acquisition and maintenance of cocaine self-administration in adolescent rats: effects of sex and gonadal hormones, Psychopharmacology 197 (2008) 237–246 (Berl.). [DOI] [PubMed] [Google Scholar]
- [13].Hu M, Crombag HS, Robinson TE, Becker JB, Biological basis of sex differences in the propensity to self-administer cocaine, Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 29 (2004) 81–85. [DOI] [PubMed] [Google Scholar]
- [14].Cason AM, Kohtz A, Aston-Jones G, Role of corticotropin releasing factor 1 signaling in cocaine seeking during early extinction in female and male rats, PLoS One 11 (2016) e0158577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [15].Feltenstein MW, Henderson AR, See RE, Enhancement of cue-induced reinstatement of cocaine-seeking in rats by yohimbine: sex differences and the role of the estrous cycle, Psychopharmacology 216 (2011) 53–62 (Berl.). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [16].Kohtz AS, Lin B, Smith ME, Aston-Jones G, Attenuated cocaine-seeking after oxytocin administration in male and female rats, Psychopharmacology 235 (2018) 2051–2063 (Berl.). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [17].Kippin TE, Fuchs RA, Mehta RH, Case JM, Parker MP, Bimonte-Nelson HA, et al. , Potentiation of cocaine-primed reinstatement of drug seeking in female rats during estrus, Psychopharmacology 182 (2005) 245–252 (Berl.). [DOI] [PubMed] [Google Scholar]
- [18].Lynch WJ, Carroll ME, Reinstatement of cocaine self-administration in rats: sex differences, Psychopharmacology 148 (2000) 196–200 (Berl.). [DOI] [PubMed] [Google Scholar]
- [19].Anker JJ, Carroll ME, Sex differences in the effects of allopregnanolone on yohimbine-induced reinstatement of cocaine seeking in rats, Drug Alcohol Depend. 107 (2010) 264–267. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [20].Anker JJ, Carroll ME, Females are more vulnerable to drug abuse than males: evidence from preclinical studies and the role of ovarian hormones, Curr. Top. Behav. Neurosci. 8 (2011) 73–96. [DOI] [PubMed] [Google Scholar]
- [21].Evans SM, Foltin RW, Exogenous progesterone attenuates the subjective effects of smoked cocaine in women, but not in men, Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. 31 (2006) 659–674. [DOI] [PubMed] [Google Scholar]
- [22].Larson EB, Anker JJ, Gliddon LA, Fons KS, Carroll ME, Effects of estrogen and progesterone on the escalation of cocaine self-administration in female rats during extended access, Exp. Clin. Psychopharmacol. 15 (2007) 461–471. [DOI] [PubMed] [Google Scholar]
- [23].Kohtz AS, Lin B, Davies H, Presker M, Aston-Jones G, Hormonal milieu drives economic demand for cocaine in female rats, Neuropsychopharmacol. Off. Publ. Am. Coll. Neuropsychopharmacol. (2022). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [24].Sawchenko PE, Swanson LW, Immunohistochemical identification of neurons in the paraventricular nucleus of the hypothalamus that project to the medulla or to the spinal cord in the rat, J. Comp. Neurol. 205 (1982) 260–272. [DOI] [PubMed] [Google Scholar]
- [25].Mello NK, Mendelson JH, Cocaine’s effects on neuroendocrine systems: clinical and preclinical studies, Pharmacol. Biochem. Behav. 57 (1997) 571–599. [DOI] [PubMed] [Google Scholar]
- [26].Teoh SK, Lex BW, Mendelson JH, Mello NK, Cochin J, Hyperprolactinemia and macrocytosis in women with alcohol and polysubstance dependence, J. Stud. Alcohol 53 (1992) 176–182. [DOI] [PubMed] [Google Scholar]
- [27].Schmittner J, Schroeder JR, Epstein DH, Preston KL, Menstrual cycle length during methadone maintenance, Addiction 100 (2005) 829–836. [DOI] [PubMed] [Google Scholar]
- [28].Leong KC, Zhou L, Ghee SM, See RE, Reichel CM, Oxytocin decreases cocaine taking, cocaine seeking, and locomotor activity in female rats, Exp. Clin. Psychopharmacol. 24 (2016) 55–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [29].Leong KC, Freeman LR, Berini CR, Ghee SM, See RE, Reichel CM, Oxytocin reduces cocaine cued Fos activation in a regionally specific manner, Int. J. Neuropsychopharmacol. /Off. Sci. J. Coll. Int. Neuropsychopharmacol. 20 (2017) 844–854. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [30].Weber RA, Logan CN, Leong KC, Peris J, Knackstedt L, Reichel CM, Regionally Specific effects of oxytocin on reinstatement of cocaine seeking in male and female rats, int. j. neuropsychopharmacol. /off. sci. j. Coll. Int. Neuropsychopharmacol. (2018). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [31].Bentzley BS, Jhou TC, Aston-Jones G, Economic demand predicts addiction-like behavior and therapeutic efficacy of oxytocin in the rat, Proc. Natl. Acad. Sci. U. S. A. 111 (2014) 11822–11827. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [32].Kohtz AS, Paris JJ, Frye CA, Low doses of cocaine decrease, and high doses increase, anxiety-like behavior and brain progestogen levels among intact rats, Horm. Behav. 57 (2010) 474–480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [33].Frye CA, Walf AA, Kohtz AS, Zhu Y, Membrane progestin receptors in the midbrain ventral tegmental area are required for progesterone-facilitated lordosis of rats, Horm. Behav. 64 (2013) 539–545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [34].Bentzley BS, Aston-Jones G, Inhibiting subthalamic nucleus decreases cocaine demand and relapse: therapeutic potential, Addict. Biol. (2016). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [35].Bentzley BS, Fender KM, Aston-Jones G, The behavioral economics of drug self-administration: a review and new analytical approach for within-session procedures, Psychopharmacology 226 (2013) 113–125 (Berl.). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [36].Hursh SR, Silberberg A, Economic demand and essential value, Psychol. Rev. 115 (2008) 186–198. [DOI] [PubMed] [Google Scholar]
- [37].Mahler SV, Aston-Jones GS, Fos activation of selective afferents to ventral tegmental area during cue-induced reinstatement of cocaine seeking in rats, J. Neurosci. Off. J. Soc. Neurosci. 32 (2012) 13309–13326. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [38].Swanson LW, Kuypers HGJM, The paraventricular nucleus of the hypothalamus - cytoarchitectonic subdivisions and organization of projections to the pituitary, dorsal vagal complex, and spinal-cord as demonstrated by retrograde fluorescence double-labeling methods, J. Comp. Neurol. 194 (1980) 555. -&. [DOI] [PubMed] [Google Scholar]
- [39].King TS, Schenken RS, Kang IS, Javors MA, Riehl RM, Cocaine disrupts estrous cyclicity and alters the reproductive neuroendocrine axis in the rat, Neuroendocrinology 51 (1990) 15–22. [DOI] [PubMed] [Google Scholar]
- [40].Mello NK, Mendelson JH, Kelly M, Bowen CA, The effects of cocaine on basal and human chorionic gonadotropin-stimulated ovarian steroid hormones in female rhesus monkeys, J. Pharmacol. Exp. Ther. 294 (2000) 1137–1145. [PubMed] [Google Scholar]
- [41].Karavolas, Subcellular distribution and properties of progesterone (delta4-steroid) 5alpha-reductase in rat medial basal hypothalamus, J. Biol. Chem. (1975). [PubMed] [Google Scholar]
- [42].Li X, Bertics PJ, Karavolas HJ, Regional distribution of cytosolic and particulate 5alpha-dihydroprogesterone 3alpha-hydroxysteroid oxidoreductases in female rat brain, J. Steroid Biochem. Mol. Biol. 60 (1997) 311–318. [DOI] [PubMed] [Google Scholar]
- [43].Goldsmith PC, Boggan JE, Thind KK, Estrogen and progesterone receptor expression in neuroendocrine and related neurons of the pubertal female monkey hypothalamus, Neuroendocrinology 65 (1997) 325–334. [DOI] [PubMed] [Google Scholar]
- [44].Francis K, Meddle SL, Bishop VR, Russell JA, Progesterone receptor expression in the pregnant and parturient rat hypothalamus and brainstem, Brain Res. 927 (2002) 18–26. [DOI] [PubMed] [Google Scholar]
- [45].Romeo RD, Bellani R, McEwen BS, Stress-induced progesterone secretion and progesterone receptor immunoreactivity in the paraventricular nucleus are modulated by pubertal development in male rats, Stress 8 (2005) 265–271. [DOI] [PubMed] [Google Scholar]
- [46].Bale TL, Dorsa DM, Johnston CA, Oxytocin receptor mRNA expression in the ventromedial hypothalamus during the estrous cycle, J. Neurosci. Off. J. Soc. Neurosci. 15 (1995) 5058–5064. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Data will be made available on request.


