Abstract
Objective
Both sarcopenia and arterial wall stiffening are frequent findings among hospitalized older adults, and further insight should be gained to explore their pathophysiological mechanisms and possible correlations.
Methods
90 hospitalized geriatric patients (mean age 83.94 ± 6.6 years, 38.89% female, 36.7% sarcopenic) have been enrolled, and underwent clinical, comorbidity and biochemical assessment. Sarcopenia was investigated following the diagnostic algorithm according to European guidelines assessing muscular strength, using a portable dynamometer, and muscle mass by bio-impedance analysis (BIA); carotid-femoral pulse wave velocity (cfPWV), carotid-radial PWV (crPWV), femoral-pedal PWV (fpPWV) and cardio-ankle vascular index (CAVI) were obtained for each.
Results
Sarcopenic patients (n = 33) were older (p < 0.01) than subjects without sarcopenia (n = 57). Sarcopenic patients presented higher CAVI (12.67 ± 3.12 vs. 10.91 ± 1.4, p < 0.01) and fpPWV, but not cfPWV and crPWV. In backward analysis muscle strength and comorbidity index resulted good indipendent variables of fpPWV (R2 0.11), muscle strength, comorbidity index and sex could predict CAVI (R2 0.22). When examining sarcopenia diagnoses determinants, in a logistic binary regression model and considering several possible covariates, fpPWV resulted a significant indipendent variable of sarcopenia along with age and sex.
Conclusion
increased indexes of arterial stiffness are shown in sarcopenic hospitalized older adults as compared to hospitalized patients without sarcopenia; peripheral arterial segments of the lower limb appeared to be more involved in the stiffening process, as compared to central segments.
Keywords: Sarcopenia, Arterial stiffness, Peripheral arterial stiffness, Femoral-pedal pulse wave velocity, Cardio-ankle vascular index
Introduction
Sarcopenia is a frequent finding among older adults, with even higher prevalence among hospitalized geriatric subjects [1]: it is defined as a progressive skeletal muscle deterioration (in terms of muscle quantity and quality), associated to increased likelihood of adverse outcomes including falls, fractures, physical disability and mortality [2]. Given the raising prevalence of sarcopenia, owing to the progressive aging of the general population, its burden is expected to gain weight [3], and further knowledge is needed to deeply understand its pathophysiology, in order to identify as many modifiable risk factors as possible. Interestingly, sarcopenia and arterial wall stiffening share several common risk factors, namely oxidative stress inflammation and insulin resistance [4, 5]. The underlying mechanisms are not fully understood, but noteworthy, both sarcopenia and arterial stiffness frequently occur with aging.
Along with muscle quantity, muscle quality results to be significantly impaired across aging [6]: in sarcopenic subjects increase in fibrosis, fat deposition inside and outside the muscles and alteration in fibre architecture have been frequently described [7]. Consolidated knowledge demonstrated that oxidative stress and inflammation are contributors to age-related muscle decline [8]. These age-related phenomena concomitantly affect different districts, and similar findings can be observed in arterial walls.
Arterial stiffening results from a multifaceted interplay of inflammatory signaling cascades and vascular remodeling processes [9], which progressively impair arterial wall elasticity and compliance [10], ultimately leading to an increase in Pulse Wave Velocity (PWV).
Increased carotid-femoral PWV (cfPWV), evaluated by applanation tonometer, is a reliable predictor of mortality risk in different subset of patients [11, 12]. The evaluation fo cfPWV is part of the latest European guidelines on arterial hypertension [13]. The same method used for the evaluation of the elastic properties of the aorta (cfPWV) is also used for the functional evaluation of the muscular arteries of the upper limb (carotid-radial PWV, crPWV) and of the lower limb (femoral-pedal PWV, fpPwv). Cardio-ankle vascular index (CAVI) can evaluate arterial stiffness from a larger proportion of the arterial tree and is considered to be less dependent on blood pressure at the time of measurement [14, 15]; this evaluation usually also provides brachial-ankle PWV (baPWV) which is associated to cfPWV [16] although including both central and peripheral segments.
Thus, several different techniques can assess arterial stiffening, focusing on different arterial vessels, and some of them may be more representative of those pathophysiological conditions which connect arterial stiffness to sarcopenia. However, so far, evidence is rather scarce, and albeit previous studies pointed out the relationship between sarcopenia and increased arterial stiffness [17, 18], it should be acknowledged that different sarcopenia diagnostic criteria and different arterial stiffness indexes have been applied. There is need for further research in order to consolidate this knowledge. The aim of the present study is to explore the relationship between sarcopenia and indexes of peripheral and central arterial stiffness, in a cohort of geriatric patients hospitalized due to any reason, in order to widen the perspective on possible pathophysiological mechanisms.
Methods
Study population
90 older patients hospitalized at Geriatric Clinic of Verona University Hospital were enrolled in the present study. Exclusion criteria were: (I) limb amputation or history of surgical treatment to aorta, carotids, or femoral arteries; (II) severe peripheral arterial disease or proximal arterial stenosis; (III) atrial fibrillation or other major arrhythmias; (IV) severe behavioural disorders which could prevent a proper compliance to data collection.
A detailed clinical history, with particular mention to cardiovascular diseases and risk factors, and physical examination were recorded for each patient; the comorbidity burden was estimated by Charlson Comorbidity Index (CCI).
The study was approved by the Ethical Committee of the University of Verona.
Anthropometric variables
Body weight was measured with the patient barefoot and wearing light indoor clothing, to the nearest 0.1 kg (Salus scale, Milan, Italy); height was recorded to the nearest 0.5 cm using a stadiometer (Salus stadiometer, Milan, Italy), however, whenever patients could not stand in erect position, the last anamnestic height was recoded. BMI was calculated as body weight adjusted by stature (kg/m 2).
Arterial stiffness measurements
CAVI, blood pressure and heart rate were measured using VaSera-1500 (Fukuda-Denshi Company, LTD, Tokyo), as per the manufacturer’s recommendations. BP cuffs were placed simultaneously on the four limbs and inflated two by two (right and left side). ECG was obtained by two electrodes placed on both arms; to obtain phonocardiography, a microphone was placed on the sternum (second rib space). Mean arterial Pressure (MAP) was calculated using the following formula: MAP = DBP + 1/3 PP (where DBP stands for Diastolic Blood Pressure and PP for Pulse Pressure).
VaSera device calculates CAVI, on the basis of the Bramwell-Hill Formula [19, 20], by the following equation:
 |
a and b are constants, ρ is considered the blood density, Ps stands for Systolic Blood Pressure (SBP), and Pd stands for DBP. By means of this device, heart-ankle PWV (haPWV) was calculated as the ratio between aortic valve to ankle length (automatically derived by software) and the time T taken by pulse wave to run this distance (T = tb + tba, tb = time from the second heart sound to the dicrotic notch at the brachial pulse wave form, tba = time from brachial to ankle pulse waves) [21].
Pulse Wave Velocity (PWV) was also measured using the portable device PulsePen (Diatecne, Milan, Italy) [22], and its software WPulsePen 2.3.3. A detailed description of PWV calculation was provided in previous studies [23, 24]; two transducers were used simultaneously, recording the arterial pulse curve of an arterial segment at the proximal point and at the distal point respectively.
As recommended by expert consensus [25], the distance between two artery points (central to peripheral) was multiplied by 0.8. Thus, the software calculated carotid-femoral PWV (cfPWV), which is considered representative of central elastic arteries [26], carotid-radial PWV (crPW) and femoral-pedal (fpPWV) which are proxy for peripheral arteries.
Biochemical analysis
Venous blood samples were obtained after the subjects fasted overnight. Plasma glucose was measured with a glucose analyser (Roche Cobas 8000, Monza, Italy). Cholesterol and triacylglycerol concentrations were determined with spectrophotometric method (Roche Cobas 8000, Monza, Italy). High-density-lipoprotein (HDL) cholesterol was measured by using the method of Warnick and Albers. LDL cholesterol was calculated using the Friedwald formula [27]. Creatinine was measured by a modular analyzer (Roche Cobas 8000, Monza, Italy); estimated Glomerular Filtration Rate (eGFR) was calculated by Cockroft-Gault formula. Finally, we evaluated the serum albumin concentration by a spectrophotometric method (Roche Cobas 8000, Monza, Italy).
Sarcopenia diagnosis
Sarcopenia assessment was performed in agreement to latest guidelines [2] (the EWGSOP 2 Consensus for Sarcopenia): subjects at risk for sarcopenia can be identified by the presence of risk factors, clinical symptoms, or validated questionnaires: then, specific tests (such as Handgrip and Chair Stand Test) can assess a reduce muscle strength, which is a pivotal feature of sarcopenia. The diagnosis can be confirmed assessing muscle mass quantity and quality, by means of Dual-energy X-ray absorptiometry (DXA), BIA, computed tomography (CT) scan or magnetic resonance imaging (MRI). All patients received SARC-F questionnaire [1], in order to identify subjects at risk of sarcopenia: SARC-F score > = 4/10 identified patients at risk. Then, Handgrip test was used as proxy for muscular strength, using a portable dynamometer (JAMAR® Hydraulic Hand Dynamometer), measures were repeated three times, and the highest value was finally recorded; the normality threshold was considered 27 kg for male subjects and 16 kg for female [2].
Finally, all patients underwent Bioelectrical Impedance Analysis (BIA) to assess body composition; the impedance measurements were performed with a phase sensitive single frequency analyzer (BIA101 BIVA® PRO, Akern srl, Italy), which applies an alternating current of 250 µA at the frequency of 50 kHz. Measurements were made using tetrapolar configuration as described by Lukaski [28]. Phase angle, resistance and reactance were registered. The appendicular skeletal mass (ASM) was calculated using the raw measurements applied to the Sergi equation [29]. Appendicular skeletral mass has been evaluated and adjusted for patient’s height, obtaining the Appendicular skeletral mass Index (ASMI).
Therefore, sarcopenia diagnosis has been made in presence of a suggestive SARC-F questionnaire, a positive Handgrip Test and ASMI < 7 Kg/m2 for male subjects and < 5.5 Kg/m2 for female [1].
Statistical analyses
Variables are shown as mean value ± standard deviation (SD), or as numbers and percentage. Variables not normally distributed were log-transformed before analysis. Independent samples t-tests were used to compare characteristics of patients with and without sarcopenia. Univariate analysis of covariance (ANCOVA) has been performed to compare arterial stiffness indexes between the sarcopenic and control subgroups, adjusting the effect of covariates, included upon physiological relevance. Backward linear multiple regression analysis was performed to evaluate the effect of selected variables (included upon physiological relevance) on arterial stiffness parameters (CAVI, baPWV, crPWV, fpPWV).
A logistic binary regression model was built to evaluate the role of possible variables (age, female sex, fpPWV. glycemia, cholesterol, hypertension diagnoses, BMI, eGFR and MAP) on sarcopenia diagnoses.
A significance threshold level of 0.05 was used throughout the study. All statistical analyses were performed using SPSS 23.0 version for Windows (IBM, Armonk, New York, USA).
Results
The study population included 90 geriatric subjects (mean age 83.94 ± 6.6 years, of whom 38.89% female). Among them, 33 were diagnosed with sarcopenia, the remaining 57, without sarcopenia, represented the control group. The main characteristics of the study population are listed in Table 1.
Table 1.
Characteristics of the study population
| Mean ± SD | |
|---|---|
| Age (years) | 83.94 ± 6.6 |
| Weight (kg) | 68.63 ± 12.7 |
| BMI (hg/m2) | 24.5 ± 3.8 |
| SBP (mmHg) | 125.17 ± 15.3 |
| DBP (mmHg) | 69.34 ± 8 |
| MAP (mmHg) | 87.95 ± 9.4 |
| CAVI (m/s) | 11.55 ± 2.3 |
| Handgrip (kg) | 20.23 ± 9.6 |
| ASMI (Kg/m2) | 6.08 ± 1 |
| cf. PWV (m/s) | 13.53 ± 5.3 |
| fp PWV (m/s) | 9.15 ± 1.7 |
| cr PWV (m/s) | 7.33 ± 1.4 |
| SARC-F | 4.2 ± 3 |
| CCI | 7.5 ± 2.7 |
| Total Cholesterol (mg/dL) | 127.28 ± 33.4 |
| Glycemia (mg/dL) | 113.31 ± 45.9 |
| LDL Cholesterol (mg/dL) | 65.66 ± 28.5 |
| HDL Cholesterol (mg/dL) | 41 ± 15.4 |
| Triglycerides (mg/dL) | 103.07 ± 45.7 |
| HB (g/dL) | 11.62 ± 2.2 |
| Creatinine (umol/L) | 117.69 ± 74.2 |
| eGFR (ml/min/1.73m2) | 47.49 ± 22 |
| Albumin (g/dL) | 33.72 ± 4.7 |
| Sarcopenia (n,%) | 33; 36.7% |
| Sex Female (n,%) | 35; 38.89% |
| Hypertension (n,%) | 66; 73.3% |
| IHD (n,%) | 15; 16.7% |
| Carotid atheromasia (n,%) | 24; 26.7% |
| Diabetes (n,%) | 21; 23.2% |
| Dyslipidemia (n,%) | 36; 40% |
BMI: body mass index; SBP: systolic blood pressure; DBP: diastolic blood pressure; MAP: mean arterial pressure; CAVI: Cardio Ankle Vascular Index; ASMI: Appendicular Skeletral Mass Index; cf. PWV: carotid-femoral pulse wave velocity; fpPWV: femoral-pedal pulse wave velocity; cr PWV: carotid-radial pulse wave velocity; CCI: Charlson Comorbidity Index; HB: hemoglobin; eGFR: glomerular filtration rate; IHD: Ischemic heart disease
Age was significantly higher in the sarcopenic group (86.42 ± 6.46 years vs. 83.51 ± 6.26 years, p < 0.01; Table 2). CCI resulted to be worse in sarcopenic subgroup (8.58 ± 2.67 vs. 6.88 ± 2.47, < 0.01). The subgroups were well matched with respect to blood tests, hemodynamic parameters and diseases such as hypertension, diabetes mellitus, ischemic heart disease and dyslipidaemia among groups.
Table 2.
Comparison between patients with and without sarcopenia
| WITH SARCOPENIA n = 33 |
WITHOUT SARCOPENIA n = 57 |
||||||
|---|---|---|---|---|---|---|---|
| Mean | SD | Mean | SD | p value | |||
| Age (years) | 86.42 | ± | 6.46 | 82.51 | ± | 6.26 | < 0.01 |
| CCI | 8.58 | ± | 2.67 | 6.88 | ± | 2.47 | < 0.01 |
| Handgrip (kg) | 14.09 | ± | 5.61 | 23.79 | ± | 9.72 | < 0.01 |
| ASMI (Kg/m2) | 5.74 | ± | 0.89 | 6.28 | ± | 1.01 | 0.01 |
| cf. PWV (m/s) | 14.35 | ± | 6.29 | 13.06 | ± | 4.65 | 0.31 |
| fp PWV (m/s) | 9.77 | ± | 1.95 | 8.8 | ± | 1.52 | 0.01 |
| cr PWV (m/S) | 7.32 | ± | 1.73 | 7.33 | ± | 1.26 | 0.97 |
| CAVI (m/s) | 12.67 | ± | 3.12 | 10.91 | ± | 1.4 | < 0.01 |
| MAP (mmHg) | 88.79 | ± | 8.97 | 87.39 | ± | 9.59 | 0.5 |
| SBP (mmHg) | 125.45 | ± | 13.83 | 125 | ± | 16.23 | 0.89 |
| DBP (mmHg) | 70.52 | ± | 8.14 | 68.67 | ± | 7.97 | 0.3 |
CCI: Charlson Comorbidity Index; ASMI: Appendicular Skeletral Mass Index; cf. PWV: carotid-femoral pulse wave velocity; fpPWV: femoral-pedal pulse wave velocity; cr PWV: carotid-radial pulse wave velocity; CAVI: Cardio Ankle Vascular Index; MAP: mean arterial pressure; SBP: systolic blood pressure; DBP: diastolic blood pressure
As regards arterial stiffness indexes, a significant difference was depicted comparing CAVI between subgroups: significantly worse CAVI is described in sarcopenic patients (12.67 ± 3.12 vs. 10.91 ± 1.4, p < 0.01).
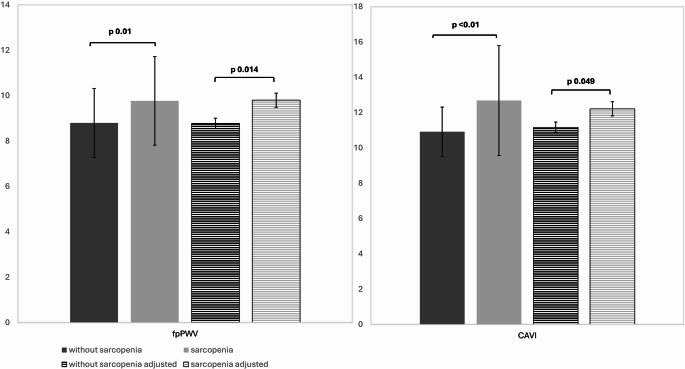
Even after adjustment for age, sex, arterial hypertension and CCI, CAVI remained increased in sarcopenic subjects (p 0.049), as shown in Fig. 1.
Fig. 1.
(left): comparison of fpPWV in patients with and without sarcopenia, before (plain colour) and after (striped) adjustment for age, sex, arterial hypertension and Charlson Comorbidity Index; (right) comparison of CAVI in patients with and without sarcopenia, before (plain colour) and after (striped) adjustment for age, sex, arterial hypertension and Charlson Comorbidity Index. fpPWV: femoral-pedal pulse wave velocity; CAVI: Cardio Ankle Vascular Index
A significant difference was described for tonometric derived pulse wave velocities for arterial segments of the lower limb (fpPWV), which resulted to be higher in the sarcopenia subgroup, as compared to the control group (9.77 ± 1.95 vs. 8.8 ± 1.52 m/s, p 0.01). This difference was confirmed even after adjustment for age, sex, arterial hypertension and Comorbidity Index (p 0.014).
Conversely, pulse wave velocities both for central (cfPWV) and peripheral arterial segments of the upper limb (crPWV), did not significantly differ between groups.
Backward regression analysis (Table 3) demonstrated that when considering fpPWV as a dependent variable and age, sex, arterial hypertension, CCI, strength and muscle mass as independent variables, both Comorbidity Index and muscle strength resulted significant indipendent variables of fpPWV (R2 0.11). Another regression model was built using the same independent variables and considering CAVI as dependent variable: sex, Comorbidity Index and muscle strength resulted significant indipendent variables of CAVI accounting for the 22% of the variance.
Table 3.
Final models of backward regression models considering FpPWV and CAVI as dependent variables and age, sex, arterial hypertension, HR, glycemia, BMI and creatinine clearance as independent variables
| Β | S.E. | standardized β | p | R 2 | ||
|---|---|---|---|---|---|---|
| fpPWV | 0.113 | |||||
| Constant | 9.156 | 0.683 | < 0.001 | |||
| CCI | 0.124 | 0.067 | 0.19 | 0.066 | ||
| Handgrip | -0.046 | 0.018 | -0.255 | 0.014 | ||
| CAVI | 0.216 | |||||
| Constant | 12,418 | 1,043 | < 0.001 | |||
| Sex | -1,54 | 0,515 | -0,323 | 0,004 | ||
| CCI | 0,192 | 0,087 | 0,218 | 0.03 | ||
| Handgrip | -0,084 | 0,026 | -0,347 | 0.002 | ||
fpPWV: femoral-pedal pulse wave velocity; CCI: Charlson Comorbidity Index; CAVI: Cardio Ankle Vascular Index
When examining sarcopenia diagnoses determinants, in a logistic binary regression model (Table 4), and considering several possible covariates, fpPWV resulted a significant indipendent variable of sarcopenia (β 0.415, SE 0.163, 95%CI 1.101–2.083, p 0.01), along with age (β 0.131, SE 0.049, 95%CI 1.036–1.255, p 0.007) and sex (β -1.929, SE 0.675, 95%CI 0.039–0.546, p 0.004).
Table 4.
Logistic Binary Regression, considering Sarcopenia Diagnoses as dependent variable and age, sex (female), fpPWV. Glycemia, cholesterol, Hypertension diagnoses, BMI, eGFR and MAP as independent variable
| β | S.E. | OR | 95% C.I. | p | ||
|---|---|---|---|---|---|---|
| Age | 0.131 | 0.049 | 1.140 | 1.036 | 1.255 | 0.007 |
| Female sex | -1.929 | 0.675 | 0.145 | 0.039 | 0.546 | 0.004 |
| fpPWV | 0.415 | 0.163 | 1.515 | 1.101 | 2.083 | 0.011 |
| glucose | 0.008 | 0.006 | 1.008 | 0.997 | 1.019 | 0.170 |
| cholesterol | 0.000 | 0.010 | 1.000 | 0.982 | 1.019 | 0.967 |
| hypertension | 0.214 | 0.618 | 1.239 | 0.369 | 4.157 | 0.729 |
| BMI | -0.138 | 0.078 | 0.871 | 0.748 | 1.014 | 0.076 |
| eGFR | -0.007 | 0.015 | 0.993 | 0.965 | 1.022 | 0.623 |
| MAP | 0.003 | 0.029 | 1.003 | 0.947 | 1.061 | 0.931 |
fpPWV: femoral-pedal pulse wave velocity; BMI: body mass index, eGFR: estimated glomerular filtration rate; MAP: mean arterial pressure
Discussion
Our study shows that sarcopenic hospitalized older adults display increased indexes of peripheral and systemic arterial stiffness as compared to hospitalized patients without sarcopenia; peripheral arterial segments of the lower limb seem to be more involved in the stiffening process, as compared to central. On the other hand, even sarcopenia, mainly in the legs, could be related to the increased arterial stiffness.
As expected, in our study subjects with sarcopenia were older than controls. This result is in line with consolidated knowledge, which pinpoints increased sarcopenia prevalence across aging [30, 31]. In our study population we found a remarkable prevalence of sarcopenia (36.6%) and this is probably due to the hospital setting of our study population. We showed in a previous study by Rossi et al. [32] a sarcopenia prevalence of about 25% in acute setting. This difference is probably due to the higher age of our study population.
The main finding of the present study is the increased arterial stiffness in sarcopenic subjects. Although there is a certain lack of knowledge regarding the detailed mechanisms connecting arterial stiffening and sarcopenia, our findings complement several previous studies, which underlined the association between impaired muscle quality and different indexes of pulse wave velocity. On the other hand, sarcopenia, mainly in the legs, could be involved in the increased arterial stiffness.
These results may open the perspective on several pathophysiological speculations.
The Health ABC study previously demonstrated that impaired muscle quality, measured by CT scan and DXA, are negatively related to cfPWV [17], suggesting the role of impaired peripheral circulation on muscular decline. Further evidence outlined a negative correlation between muscle mass and PWV, albeit considering different techniques both for muscle mass and for arterial stiffness assessment [33].
According to our data, central arterial stiffness, namely cfPWV, did not significantly differ between patients with and without sarcopenia. On the other hand, we found a significant difference when considering fpPWV and CAVI. The difference is confirmed even after adjustment for multiple variables. According to this evidence, central aorta and peripheral arterial vessels might be differently involved in the stiffening process, and techniques aimed at evaluating muscular (peripheral) arteries might better represent the vascular impairment associated to sarcopenia. Albeit based on different approaches, several previous studies outlined increased arterial stiffness in presence of low muscle mass and quality, actually relying on CAVI (or CAVI-derived baPWV) assessment, which is known to include also peripheral segments [21]; however, to the best of our knowledge, our study is the first showing significant difference in fpPWV. Sampaio and colleagues [18], demonstrated a negative and independent association between CAVI and Skeletal Muscle Index (derived by BIA) in a population of 175 Japanese community-dwelling subjects, older than 65 years. These data have been confirmed even in a European ad Caucasian population [34]: Skeletal muscle Index was found to be related to increased CAVI in a cohort of 366 adults older than 45 years. Another study [35] demonstrated that decreased thigh muscle mass (measured by CT scan) is associated to augmented baPWV in male subjects; the same study outlined that on top of several confounding parameters, baPWV was an independent risk factor for sarcopenia, in male subjects, along with age, height, scarce physical activity and free testosterone levels. Muscle/fat ratio, measured by CT scan and total body DXA, was negatively associated to baPWV in a cohort of 526 apparently healthy adults [36]; similarly, in the J-SHIPP study, led on 1024 older subjects without known cardiovascular diseases, baPWV resulted to be negatively related to muscle mass and positively related to visceral adipose tissue [37]. Our results confirm and complement the above-mentioned evidence: most of the other studies correlated arterial stiffness to muscle mass measurements or to body composition evaluations; in our research, instead, we applied the sarcopenia diagnostic algorithm, as recommended by guidelines [2], focusing on the clinical assessment, on SARC-F questionnaire, Handgrip test and muscle mass evaluation. Furthermore, single indexes of arterial stiffness were provided in previous studies (either CAVI, or baPWV); conversely, we investigated arterial stiffness measuring different parameters, namely CAVI, cfPWV, fpPWV and crPWV. Noteworthy, among the parameters that we presented, more than one (CAVI and fpPWV) appeared to be significantly different in patients with and without sarcopenia.
As above mentioned, CAVI evaluates arterial stiffness considering a wide portion of the arterial tree, taking into account the peripheral segments. Thus, the difference between patients with and without sarcopenia may be potentially explained by the difference in peripheral arterial segments, in particular of the lower limb, which appear to be stiffer in sarcopenic subjects. Noteworthy, age-related increases in peripheral artery stiffness are less marked than in the central arteries [38]. This intriguing result sheds light on a possible pathophysiological mechanism, which may unveil a vicious circle. Impaired vascular perfusion, directly associated to increased arterial stiffening [39] can be interpreted as a cause of muscular deterioration, occurring both as histological and functional abnormalities [39]. From a different perspective, muscular alterations typical of sarcopenia, namely local (and systemic) inflammation, fibrosis and fat infiltration [8], are known to be part of a complex interplay of myokines [7] which potentially affect vascular stiffening as well.
Our data do not allow to draw unique conclusions regarding the causality of these mechanisms. Backward analysis identified muscle strength as a major indipendent variable of CAVI and cfPWV and this may endorse the hypothesis that muscle changings might affect vascular stiffening. However, a reciprocal effect of sarcopenia and arterial stiffness can be hypothesized as we found even that peripheral arterial stiffness (fpPWV) was a significant indipendent variable of sarcopenia.
Moreover, it should be noted that subjects involved in other studies, were substantially considered healthy individuals; conversely, our population was made of hospitalized patients, with a remarkable comorbidity burden, which is known to play a role in vascular stiffness [40]. Anyway, even after adjustment for Comorbidity Index, CAVI and fpPWV remained increased in sarcopenic subjects.
Some limitations should be acknowledged when interpreting our findings. First, the small dimension of the sample size might have affected the significance of some correlations; however, a wide characterization of the stiffness status of these patients has been provided, and different stiffness indexes have been used to corroborate the results. Due to the cross-sectional nature of this study, we were not allowed to draw causality conclusions, nor to evaluate the implications of our findings on clinical outcomes. We acknowledge that the prevalence of sarcopenia might appear rather high in our study, as compared to the general population, but all our subjects were hospitalized, therefore presenting a remarkable risk factor for sarcopenia.
In conclusion, our study described increased indexes of systemic and peripheral arterial stiffness in sarcopenic hospitalized older adults as compared to hospitalized patients without sarcopenia, and lower limb arterial segments appeared to be more involved in the stiffening process, as compared to upper limb and central segments. On the opposite sarcopenia could be, mainly in the legs, a determinant of increased peripheral arterial stiffness.
The findings of this study suggest a potential link between sarcopenia and increased arterial stiffness, particularly at the level of peripheral arteries in the lower limbs. The muscle alterations observed in sarcopenic individuals may contribute biologically to impaired vascular compliance through mechanisms that are not yet fully understood but may involve shared inflammatory, metabolic, and structural pathways.
From a clinical standpoint, the observed association raises the possibility that vascular dysfunction may play a contributory role in the pathophysiology of sarcopenia. This highlights the importance of an integrated approach to the management of older adults, where assessment of vascular health might offer additional insights into musculoskeletal decline. Further longitudinal studies are warranted to clarify the directionality and causality of this relationship, and to explore whether targeting vascular stiffness could represent a novel strategy in the prevention or treatment of sarcopenia.
Author contributions
FF, EZ and MZ wrote the research project. EM and SNP collected data. FF, SNP and AG wrote the main manuscript text. AG prepared the figure. All authors reviewed the manuscript.
Funding
None to declare.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Rossi AP, Caliari C, Urbani S, Fantin F, Brandimarte P, Martini A et al (2021) Sarcopenia risk evaluation in a sample of hospitalized elderly men and women: combined use of the Mini sarcopenia risk assessment (MSRA) and the SARC-F. Nutrients 13. 10.3390/nu13020635 [DOI] [PMC free article] [PubMed]
- 2.Cruz-Jentoft AJ, Bahat G, Bauer J, Boirie Y, Bruyère O, Cederholm T et al (2019) Sarcopenia: revised European consensus on definition and diagnosis. Age Ageing 48:16–31 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Beaudart C, Rizzoli R, Bruyère O, Reginster J-Y, Biver E (2014) Sarcopenia: burden and challenges for public health. Arch Public Health 72:45 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Chhetri JK, de Souto Barreto P, Fougère B, Rolland Y, Vellas B, Cesari M (2018) Chronic inflammation and sarcopenia: A regenerative cell therapy perspective. Exp Gerontol 103:115–123 [DOI] [PubMed] [Google Scholar]
- 5.Hingorani AD, Cross J, Kharbanda RK, Mullen MJ, Bhagat K, Taylor M et al (2000) Acute systemic inflammation impairs endothelium-dependent dilatation in humans. Circulation 102:994–999 [DOI] [PubMed] [Google Scholar]
- 6.Correa-De-Araujo R, Rossi AP, Zamboni M, Addison O, Miljkovic I, Goodpaster B (2022) Editorial: muscle quality in skeletal muscle function deficit: recent advances and potential clinical and therapeutic implications. Front Physiol 13:847883 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Zamboni M, Mazzali G, Brunelli A, Saatchi T, Urbani S, Giani A et al (2022) The role of crosstalk between adipose cells and myocytes in the pathogenesis of sarcopenic obesity in the elderly. Cells 11:3361 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Budui SL, Rossi AP, Zamboni M (2015) The pathogenetic bases of sarcopenia. Clin Cases Min Bone Metab 12:22–26 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Bank AJ (1997) Physiologic aspects of drug therapy and large artery elastic properties. Vascular Med 2:44–50 [DOI] [PubMed] [Google Scholar]
- 10.Vlachopoulos C, Xaplanteris P, Aboyans V, Brodmann M, Cífková R, Cosentino F et al (2015) The role of vascular biomarkers for primary and secondary prevention. A position paper from the European society of cardiology working group on peripheral circulation. Endorsed by the association for research into arterial structure and physiology (ARTERY. Atherosclerosis 241:507–532 [DOI] [PubMed] [Google Scholar]
- 11.Patoulias D, Papadopoulos C, Stavropoulos K, Imprialos K, Doumas M (2020) Updated Meta-Analysis of trials assessing the cardiovascular efficacy of Sodium-Glucose Co-Transporter-2 inhibitors and Glucagon-Like Peptide-1 receptor agonists in black patients. Am J Cardiol 137:133–135 [DOI] [PubMed] [Google Scholar]
- 12.Laurent S, Cockcroft J, Van Bortel L, Boutouyrie P, Giannattasio C, Hayoz D et al (2006) Expert consensus document on arterial stiffness: methodological issues and clinical applications. Eur Heart J 27:2588–2605 [DOI] [PubMed] [Google Scholar]
- 13.McEvoy JW, McCarthy CP, Bruno RM, Brouwers S, Canavan MD, Ceconi C et al (2024) 2024 ESC guidelines for the management of elevated blood pressure and hypertension. Eur Heart J 45:3912–4018 [DOI] [PubMed] [Google Scholar]
- 14.Shirai K, Song M, Suzuki J, Kurosu T, Oyama T, Nagayama D et al (2011) Contradictory effects of β1- and α1- aderenergic receptor blockers on Cardio-Ankle vascular stiffness index (CAVI). J Atheroscler Thromb 18:49–55 [DOI] [PubMed] [Google Scholar]
- 15.Asmar R (2017) Principles and usefulness of the cardio-ankle vascular index (CAVI): a new global arterial stiffness index. Eur Heart J Supplements 19:B4–B10 [Google Scholar]
- 16.Tanaka H, Munakata M, Kawano Y, Ohishi M, Shoji T, Sugawara J et al (2009) Comparison between carotid-femoral and brachial-ankle pulse wave velocity as measures of arterial stiffness. J Hypertens 27:2022–2027 [DOI] [PubMed] [Google Scholar]
- 17.Abbatecola AM, Chiodini P, Gallo C, Lakatta E, Sutton-Tyrrell K, Tylavsky FA et al (2012) Pulse wave velocity is associated with muscle mass decline: health ABC study. Age (Omaha) 34:469–478 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sampaio RAC, Sewo Sampaio PY, Yamada M, Yukutake T, Uchida MC, Tsuboyama T et al (2014) Arterial stiffness is associated with low skeletal muscle mass in Japanese community-dwelling older adults. Geriatr Gerontol Int 14(Suppl 1):109–114 [DOI] [PubMed] [Google Scholar]
- 19.Bramwell JC, Hill AV, Velocity, of transmission of the pulse-wave (1922) Lancet 199:891–892 [Google Scholar]
- 20.Saiki A, Sato Y, Watanabe R, Watanabe Y, Imamura H, Yamaguchi T et al (2016) The role of a novel arterial stiffness parameter, Cardio-Ankle vascular index (CAVI), as a surrogate marker for cardiovascular diseases. J Atheroscler Thromb 23:155–168 [DOI] [PubMed] [Google Scholar]
- 21.Hayashi K, Yamamoto T, Takahara A, Shirai K (2015) Clinical assessment of arterial stiffness with cardio-ankle vascular index. J Hypertens 33:1742–1757 [DOI] [PubMed] [Google Scholar]
- 22.Salvi P, Lio G, Labat C, Ricci E, Pannier B, Benetos A (2004) Validation of a new non-invasive portable tonometer for determining arterial pressure wave and pulse wave velocity: the pulsepen device. J Hypertens 22:2285–2293 [DOI] [PubMed] [Google Scholar]
- 23.Fantin F, Giani A, Macchi F, Amadio G, Rossi AP, Zoico E et al (2021) Relationships between subendocardial perfusion impairment, arterial stiffness and orthostatic hypotension in hospitalized elderly individuals. J Hypertens 39:2379–2387 [DOI] [PubMed] [Google Scholar]
- 24.Fantin F, Giani A, Gasparini L, Rossi AP, Zoico E, Mazzali G et al (2021) Impaired subendocardial perfusion in patients with metabolic syndrome. Diab Vasc Dis Res 18:147916412110471 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Van Bortel LM, Laurent S, Boutouyrie P, Chowienczyk P, Cruickshank JK, De Backer T et al (2012) Expert consensus document on the measurement of aortic stiffness in daily practice using carotid-femoral pulse wave velocity. J Hypertens 30:445–448 [DOI] [PubMed] [Google Scholar]
- 26.McVeigh GE, Bratteli CW, Morgan DJ, Alinder CM, Glasser SP, Finkelstein SM et al (1999) Age-related abnormalities in arterial compliance identified by pressure pulse contour analysis: aging and arterial compliance. Hypertension 33:1392–1398 [DOI] [PubMed] [Google Scholar]
- 27.Friedewald WT, Levy RI, Fredrickson DS (1972) Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 18:499–502 [PubMed] [Google Scholar]
- 28.Lukaski HC, Bolonchuk WW, Hall CB, Siders WA (1986) Validation of tetrapolar bioelectrical impedance method to assess human body composition. J Appl Physiol (1985) 60:1327–1332 [DOI] [PubMed] [Google Scholar]
- 29.Sergi G, De Rui M, Veronese N, Bolzetta F, Berton L, Carraro S et al (2015) Assessing appendicular skeletal muscle mass with bioelectrical impedance analysis in free-living Caucasian older adults. Clin Nutr 34:667–673 [DOI] [PubMed] [Google Scholar]
- 30.Volpato S, Bianchi L, Cherubini A, Landi F, Maggio M, Savino E et al (2014) Prevalence and clinical correlates of sarcopenia in community-dwelling older people: application of the EWGSOP definition and diagnostic algorithm. J Gerontol Biol Sci Med Sci 69:438–446 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Alexandre TdaS, Duarte YA, de Santos O, Wong JLF, Lebrão R (2014) Prevalence and associated factors of sarcopenia among elderly in brazil: findings from the SABE study. J Nutr Health Aging 18:284–290 [DOI] [PubMed] [Google Scholar]
- 32.Rossi AP, Fantin F, Micciolo R, Bertocchi M, Bertassello P, Zanandrea V et al (2014) Identifying sarcopenia in acute care setting patients. J Am Med Dir Assoc 15:303e7–30312 [DOI] [PubMed] [Google Scholar]
- 33.Rodríguez AJ, Karim MN, Srikanth V, Ebeling PR, Scott D (2017) Lower muscle tissue is associated with higher pulse wave velocity: A systematic review and meta-analysis of observational study data. Clin Exp Pharmacol Physiol 44:980–992 [DOI] [PubMed] [Google Scholar]
- 34.Kirkham FA, Bunting E, Fantin F, Zamboni M, Rajkumar C (2019) Independent association between Cardio-Ankle vascular index and sarcopenia in older U.K. Adults. J Am Geriatr Soc 67:317–322 [DOI] [PubMed] [Google Scholar]
- 35.Ochi M, Kohara K, Tabara Y, Kido T, Uetani E, Ochi N et al (2010) Arterial stiffness is associated with low thigh muscle mass in middle-aged to elderly men. Atherosclerosis 212:327–332 [DOI] [PubMed] [Google Scholar]
- 36.Kim TN, Park MS, Lim K, Il, Yang SJ, Yoo HJ, Kang HJ et al (2011) Skeletal muscle mass to visceral fat area ratio is associated with metabolic syndrome and arterial stiffness: the Korean sarcopenic obesity study (KSOS). Diabetes Res Clin Pract 93:285–291 [DOI] [PubMed] [Google Scholar]
- 37.Kohara K, Ochi M, Tabara Y, Nagai T, Igase M, Miki T (2012) Arterial stiffness in sarcopenic visceral obesity in the elderly: J-SHIPP study. Int J Cardiol 158:146–148 [DOI] [PubMed] [Google Scholar]
- 38.Yu S, McEniery CM (2020) Central versus peripheral artery stiffening and cardiovascular risk. Arterioscler Thromb Vasc Biol 40:1028–1033 [DOI] [PubMed] [Google Scholar]
- 39.Jeon YK, Shin MJ, Saini SK, Custodero C, Aggarwal M, Anton SD et al (2021) Vascular dysfunction as a potential culprit of sarcopenia. Exp Gerontol 145:111220 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fantin F, Giani A, Trentin M, Rossi AP, Zoico E, Mazzali G et al (2022) The correlation of arterial stiffness parameters with aging and comorbidity burden. J Clin Med 11. 10.3390/jcm11195761 [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No datasets were generated or analysed during the current study.