Abstract
Extracellular vesicles (EVs) are crucial for facilitating intercellular communication, promoting cell migration, and orchestrating the immune response. Recently, EVs can diagnose and treat tumors. EVs can be measured as biomarkers to provide information about the type of disease and therapeutic efficacy. Furthermore, EVs with lower immunogenicity and better biocompatibility are natural carriers of chemicals and gene drugs. Herein, we review the molecular composition, biogenesis, and separation methods of EVs. We also highlight the important role of EVs from different origins as biomarkers and drug delivery systems in tumor therapy. Finally, we provide deep insights into how EVs play a role in reversing the immunosuppressive microenvironment.
Key words: Extracellular vesicles, Biomarkers, Tumor immunotherapy, Drug delivery systems, Tumor microenvironment, Drug loading methods, Separation technologies, Molecular composition
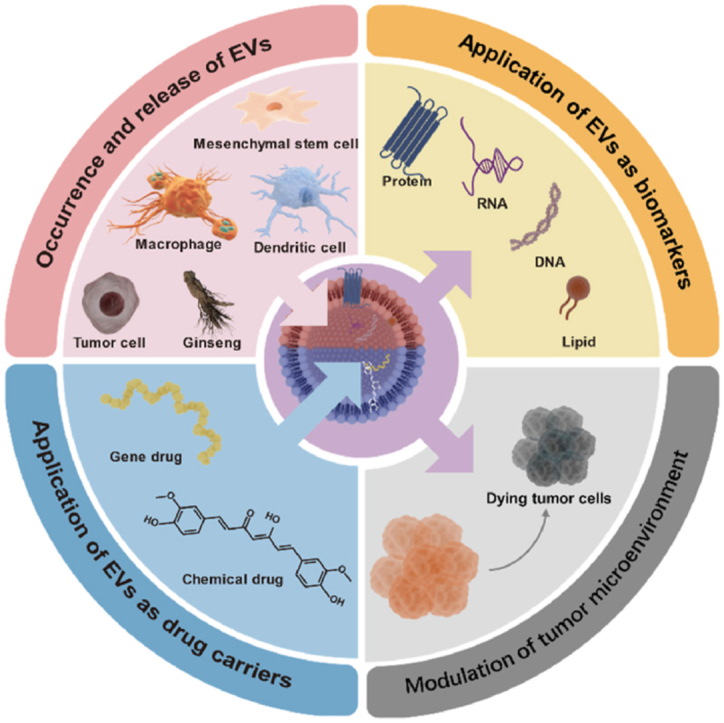
Graphical abstract
This review focuses on the major applications of extracellular vesicles as biomarkers and drug delivery systems in the treatment of tumors.
1. Introduction
The tumor microenvironment (TME), characterized by hypoxia, acidity, and high intertissued fluid pressure, is an immunosuppressive and highly complex system1. Tumor immunotherapy, by activating the patient’s immune response and reversing immunosuppressive TME to eradicate tumor cells, can lower metastatic relapse and prolong the survival time of patients2. Due to the cellular specificity and immunogenicity of extracellular vesicles (EVs), they can modulate TME and are involved in biological processes of tumor development and angiogenesis3. With the natural TME modulation of EVs, it is hoped to prevent the metastasis of oncogenic molecules and inhibit tumor development and angiogenesis.
EVs encompass a diverse range of vesicles that are released by virtually all types of living cells, including both mammalian and plant cells. While mammalian-derived EVs have been extensively studied and reported, it is noteworthy that EVs derived from plants are structurally similar to mammalian-derived EVs, and also can be potentially developed as therapeutic agents4. Recently, EVs have become a major research hotspot as novel biomarkers and drug delivery carriers in cancer therapy5. Since EVs carry various bioactive molecules from donor cells, some specific loading substances may reflect the biological properties and states of the donor cells. EVs circulate stably in re-biotic fluid and can diagnose disease in its early stages6. For example, EVs isolated from ovarian cancer patients carry oncogenic miRNA signatures that can reliably distinguish malignant disease from benign disease.
Additionally, more than 50,000 patients have benefited from exosomal RNA for prostate cancer detection6. Meanwhile, with the advantages of high biocompatibility, cellular specificity, better stability, and limited immunogenicity7, EVs have been applied in drug delivery systems (DDSs) to transport cargo into the cytoplasm of target cells8. CD47, which is widely expressed on the surface of EVs, helps them avoid phagocytosis by the reticuloendothelial system and achieve efficient immune escape. More importantly, EVs can successfully cross physiological barriers to reach target cells and efficiently transport different cargoes to target cells9. Rong et al.10 obtained a targeted DDS by loading CCL2-small interfering RNAs (siRNAs) into EVs via electroporation. To sum up, EVs have promising applications in immunotherapy as new diagnostic biomarkers and DDSs11.
Overall, EVs possess a wide range of biological functions and clinical applications. They have been extensively utilized as biomarkers, therapeutic agents, and drug delivery systems to address specific clinical needs. EVs-based therapeutic regimens have been assessed in numerous preclinical studies with encouraging results. Herein, we not only introduce the molecular composition, biogenesis, separation methods, and drug loading methods of EVs but also summarize the vital role of EVs as biomarkers in tumor diagnosis and treatment. Next, we discuss the advantages of EVs when used as drug delivery systems and therapeutic agents. Of note, we provide deep insights into how EVs reverse the immunosuppressive TME, separately based on endogenous EVs, exogenous EVs, and EVs as drug delivery systems.
2. Biogenesis, molecular composition, and separation of EVs
EVs can cross biological barriers and transmit signals to other cells for intercellular communication. Initially, the term “exosome” was not accurately defined. In the mid-1990s, scientists discovered that exosomes were secreted by dendritic cells (DCs) and B lymphocyte12. On this basis, the potential of exosomes to regulate immune function has been widely explored13. Exosomes are associated with immunomodulation, angiogenesis, cell proliferation, and apoptosis14. Whereas microvesicles are produced by cell membrane outgrowth after stress15. Apoptotic bodies are formed primarily through cell membrane wrinkling and invagination in the process of cell apoptosis16. In recent years, Zhao et al.17 first proved that the chemotherapeutic agent camptothecin induces cell apoptosis and the released apoptotic bodies transport the remaining camptothecin to surrounding tumor cells and exert neighboring effects to enlarge antitumor response.
2.1. Formation, release, and uptake of EVs
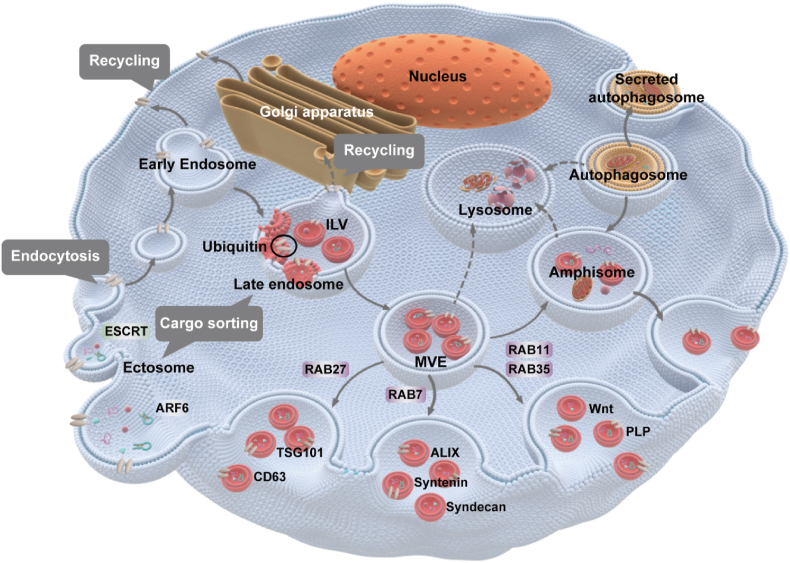
Lymphocytes, nerve cells, tumor cells, and DCs release EVs with different patterns of biogenesis according to the environment in which they live18. The synthesis of exosomes mainly depends on endocytosis. EVs can fuse with early endosomes, and then begin sorting and transport to the extracellular matrix. During this period, early endosomes undergo invagination, budding, and accumulation. Finally, form multivesicular bodies (MVBs) and then integrate with lysosomes. Besides, intracellular vesicles in MVBs can be released to further secrete exosomes (Fig. 1)14. In addition, MVBs form amphisomes through interaction with autophagosomes and then merge with lysosomes5,19. During this process, the ubiquitination of MVB membrane proteins influences exosome secretion by facilitating MVB fusion with lysosomes20. Most organelles in the endomembrane system such as the Golgi apparatus, endoplasmic reticulum, and endosomes carry Rab GTPases. To ensure only mature endosomes are sorted, intracavitary EV formation driven by ESCRT needs to be closely combined with Rab-GTPase transformation21. When apoptosis-related transduction pathways are activated, apoptotic bodies will be released from apoptotic cells through plasma membrane vesiculation and dynamic membrane vesiculation16. Currently, the molecular mechanism of microvesicle formation is not clear. Some studies suggest that binding protein 1 contains an arresting domain, which integrates the endosomal sorting complex protein into the bilayer film to induce budding22. Cytoplasmic calcium influx can disrupt phospholipid asymmetry and cause membrane budding23. The secretion process of exosomes is complex and influenced by various factors, such as long-chain noncoding hot-air (lncRNA) regulating Rab35 and SNAP to increase exosome secretion24, pH value decrease promoting exosome secretion and cell uptake, P53 promoting exosome secretion under stress conditions by inhibiting activation pathways, and heparin overexpression sometimes increasing exosome secretion25. In general, the synthesis and secretion mechanisms of EVs are extremely complex and require further study.
Figure 1.
Vesicles derived from the plasma membrane or Golgi apparatus can fuse with early endosomes, and then begin sorting and transport to the extracellular matrix. During this period, early endosomes invaginate, bud and accumulates intraluminal vesicles to form MVBs, and finally fuse with lysosomes. MVBs can release intracellular vesicles by fusing with the plasma membrane, thus producing exosomes.
EVs can be taken by receptor cells in many ways, including ligand–receptor interactions, direct signal transduction, and direct fusion of membranes26. After interacting with target cells, EVs are taken up by cells through various endocytosis pathways. These pathways such as caveolin-mediated uptake and phagocytosis are characterized by rapid and energy-intensive27. Endocytosis is the main method of EV uptake, but there is almost no consensus on which type of endocytosis mechanism is the most important in the literature. This is because EVs can activate many ways to enter cells. Overall, knowledge about the formation, release, and uptake of EVs is incomplete and worthy of further exploration.
2.2. Molecular composition of EVs
EVs are vesicles derived from endocytic processes and are enclosed by a membrane. Dysregulation of EV expression is implicated in the pathogenesis and progression of numerous diseases, including cancer (Table 1)28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45. Exosomal lipids represent a rich reservoir of unexplored bioactive molecules, which not only contribute to the structural integrity of exosomes but also play pivotal regulatory roles in recipient cells. These lipids hold immense potential as disease biomarkers and are intricately involved in EVs biosynthesis and uptake, immune surveillance, tumor microenvironment modulation, and inflammation regulation. For example, EVs derived from tumor cells are particularly abundant in glycerolipids, sphingolipids, and sterol lipids46,47. Furthermore, proteins derived from the plasma membrane and cytosol serve as the primary sources of EV proteins, which are distributed within the interior of EVs and on their membrane surface48. The surface receptors of EVs facilitate targeting and uptake by recipient cells, allowing the binding of proteins, lipids, and genetic information carried by the vesicles to modulate their physiological state49. However, these markers may not always be suitable as cancer markers. It is suggested that glypican-1 (GPC1) and EphA2 would serve as more effective characteristic proteins for the diagnosis of pancreatic cancer exosomes50. Furthermore, it is now widely recognized that RNAs derived from EVs play a significant role in the transcription, post-transcriptional processing, and translational modification of genes in recipient cells. This regulation leads to the modulation of various cellular functions, including biological processes and disease progression51,52.
Table 1.
Active components and functions of EVs.
| Biochemical characterization | Origin | Component | Function | Ref. |
|---|---|---|---|---|
| Lipid | Grape | PA, PE | Remodeling intestinal tissue, anti-inflammation | 28,29 |
| Grapefruits | PE, PC | Anti-inflammation, anti-oxidant | 30 | |
| Ginger | PA | Anti-inflammation | 31 | |
| Ginseng | PC 28.8%, TG 16.8%, Cer 12.9%, DGMG, PE | Maintaining stability and facilitating preferential uptake by the BBB | 32 | |
| Plasma | Gb3, PEps, PS, SL, BMP, Cer, LacCer | Modulating downstream pathways associated with COVID-19 pathology | 33 | |
| Edible tea flowers | EGCG, EC, ECG | Anti-cancer | 34 | |
| Plasma | PC, SM, CE, TAG, LysoPC | Promoting exosome biogenesis and lymphocyte chemotaxis | 46 | |
| Protein | Serum | GPC1, EphA2 | Noninvasive monitoring and diagnosis of early pancreatic cancer | 50 |
| Urine | EphA2 | Enhancing the migration of tumor cells | 35 | |
| Plasma | CA 15-3, CA 125, CEA, HER2, EGFR, PSMA, EpCAM, VEGF | Diagnosis, monitoring and prediction of metastatic breast cancer | 36 | |
| Nucleic acid | Bitter melon | microRNA | Anti-cancer, anti-inflammation | 37 |
| Apple | miRNA | Intestinal function | 38 | |
| Watermelon | microRNA | Regulation of intestinal secretion groups | 39 | |
| Nut | miRNAs | Anti-inflammation | 40 | |
| Tartary buckwheat | miRNA | Modulation of gut bacteria | 41 | |
| Ginseng | miR396, miR159, miR166, miR156, miR164 | Regulation of apoptotic c-MYC signaling pathway. | 32 | |
| Serum | miR1246, miR141-3p, miR200a-3p, miR200b-3p, miR200c-3p, miR203a-3p, miR-429 | Diagnosis and prognosis of epithelial ovarian carcinoma | 42 | |
| Stem cell | let-7b-5p, miR-24-3p | Reprogramming the dermal microenvironment | 43 | |
| Osteoinductive dental pulp stem cell | miR1246 | Promoting bone tissue regeneration | 44 | |
| Hypoxic lung cancer cells | miR23a | Promoting endothelial cell migration of cancer cells and blood vessel formation | 45 |
BMP, bis(monoacylglycero)phosphate; CA 15-3, carcinoma antigen 15-3; Cer, ceramide; CE, cholesterol esters; CEA, carcinoembryonic antigen; DGMG, digalactosylmonoacylglycerol; EC, epicatechin; ECG, epicatechin gallate; EGCG, epigallocatechin gallate; EGFR, epidermal growth factor receptor; EpCAM, epithelial cell adhesion molecule; EphA2, ephrin type-A receptor 2; Gb3, globotriaosylceramide; GPC1, glypican-1; HER2, human epidermal growth factor receptor 2; LacCer, lactosylceramide; LysoPC, lysophosphatidylcholine; PA, phosphatidic acids; PC, phosphatidylcholine; PE, phosphatidylethanolamines; PEps, phosphatidylethanolamines; PS, phosphatidylserine; PSMA, prostate-specific membrane antigen; SM, sphingomyelin; SL, sulfatide; TAG, triacylglyceride; TG, triglycerides; VEGF, vascular endothelial growth factor.
2.3. Separation of EVs
For the complex secretion process of EVs, common techniques such as ultracentrifugation, ultrafiltration and microfluidization are commonly used to separate EVs. We summarize the features of the mentioned separation technologies (Table 2)53, 54, 55. Ultracentrifugation precipitates and purifies vesicles of the same size from a sample by centrifugation at high speed56. Although the ultracentrifugation method is well-developed, there is some drawbacks, such as the inability to guarantee purity and unstable recovery. More importantly, repeated centrifugation operations may also cause damage to vesicles. Compared with other methods, Ultrafiltration is easy to operate, possess high separation efficiency and keep the biological activity of EVs. However, when using pressure as the driving force for membrane separation in the ultrafiltration process, EVs may well be damaged. And EVs tend to clog the filter, thus affecting the separation57,58.
Table 2.
Comparison of separation technologies of EVs.
| Separation technology | Advantage | Disadvantage | Ref. |
|---|---|---|---|
| Ultracentrifugation | Most commonly used Well developed |
Low purity Possible damage of EVs |
66 |
| Ultrafiltration | Simple and efficient No effect on the biological activity of exosomes |
Low yield Potential for pore blockage |
57,58 |
| Size-based microfluidics | High separation efficiency Size uniformity of yield |
Complicated photolithography fabrication | 53 |
| Microfluidic separation based on immunoaffinity | High purity Specific separation Fast separation |
Isolation of only exosomes containing target proteins | 54 |
| Field-free separation | High reproducibility and purity | Inefficiency | 65 |
| Surface enhancement of Raman scattering | Rapid separation Efficient detection of biomarkers |
Adsorption of non-target EVs | 62 |
| Single vesicle profiling | Specific separation Identification of vesicle heterogeneity |
Complexity of analytical sample handling | 64 |
| Dynamic microfluidics | Size uniformity High purity Fast separation Controllable process |
Operation requiring experienced operators | 55 |
The emerging microfluidic chip technology is a promising separation method. This technology is a high-throughput method, which separates EVs according to their physical and biochemical properties like immunoaffinity and size. In addition to this, it can also combine electromagnetic, acoustic and other external forces with microfluidic technology to realize dynamic microfluidic flow59. Since the first application of microfluidic chips to exosome isolation by Chen et al.60 the studies of efficient isolation of EVs based on microfluidic technology have attracted much attention. For example, Casadei et al.61 successfully isolated and captured liposarcoma-derived EVs using a micro- and nanofluidic device. This device not only reduces processing time and ensured the integrity of the EV cargo, but also greatly improves separation efficiency. In terms of quality control during the large-scale production of EVs, ensuring their efficient and high-purity separation remains a considerable challenge. The intrinsic structure of EVs determines that they differ from the intrinsic charge density or isoelectric point of impurities, which can be exploited to separate EVs by Free-flow electrophoresis (FFE). Lee et al.62 used light-induced electrophoresis for automatic sorting of EVs and successfully separated three kinds of EVs (100–350 nm in diameter) with a purity as high as 86%. Notably, surface enhancement of Raman scattering (SERS) enables simultaneous rapid separation of EVs and efficient detection of biomarkers. Zhang et al.63 construct six SERS tags, enabling the simultaneous detection of six breast cancer biomarkers within 1.5 h. However, the separation method may adsorb non-target EVs carrying the same biomarkers. Single vesicle profiling can accurately identify vesicle heterogeneity between different cell types64. But the complexity of analytical sample handling limits its wide application. Bai et al.65 developed a viscoelastic microfluidic platform DEIC to enable Field-Free Separation and downstream analysis of EVs with minimal sample volume, achieving 100% separation accuracy for selected EpCAM/PD-L1 biomarkers.
Therefore, efforts have been made to design more efficient and convenient exosome separation platforms to provide better preparation for research. Pan et al.66 designed a fast and efficient EVs separation platform, EV-FISHER. EVs from plasma could be induced by cholesterol, followed by centrifugation to obtain. EV-FISHER is optimized for efficient plasma EVs separation compared to ultracentrifugation in terms of processing time, separation efficiency, and separation requirements. In addition, Zong et al.67 showed a dual-switching pH-responsive peptide (referred to as D-S pHLIP) that selectively isolated and enriched cTME-EVs from plasma, which provided an additional option for selective enrichment of EVs for downstream analysis. To improve the isolation of tumor-derived EVs (TDEVs), Niu et al.68 developed a fluidic multivalent magnetic interface (FluidmagFace). Compared with non-fluidic interfaces, FluidmagFace could significantly improve the affinity and separation rate, providing new possibilities for subsequent cancer diagnosis and biomarker discovery. Mature exosome isolation techniques are necessary for the cutting-edge research and clinical application of exosomes. Some traditional isolation methods may not meet the existing needs, so designing isolation methods to adapt to more conditions or to obtain qualified exosomes conveniently and efficiently would be hot research topics in the next few years.
3. Diagnosis of tumors by EVs as biomarkers
Biomarker, a characteristic biochemical index, which can be used for objective measurement and evaluation. Many substances such as serum and exosomal miRNAs are used as biomarkers of cancer, which can be used to diagnose the occurrence of cancer, judge the stage of cancer or assess the safety and efficacy of new therapeutic strategies69. EVs are common intercellular signal transmission media between eukaryotic cells and prokaryotic cells70. Nowadays, they are considered as extremely valuable biomarkers for diagnosing and predicting different diseases because the carried bioactive substance reflect the biological state of the source cells (Table 3)26,71, 72, 73.
Table 3.
EVs as biomarkers in clinical diagnosis, treatment and prognosis for cancer.
| Disease | Biofluid | EV type | Biomarker | Biological function | Ref. |
|---|---|---|---|---|---|
| Melanoma | Plasma | Exosomes and microvesicles | PD-L1 | A predictor of anti-PD-1 therapy and clinical outcomes | 74 |
| Plasma | Exosomes | PD-L1 | 76 | ||
| Plasma | Small extracellular vesicles | PDCD 6 IP | Prognostic biomarkers in melanoma | 80 | |
| Plasma | Exosomes | BRAF RNA | 86 | ||
| Prostate cancer | Urine | Exosomes | ITGA3 and ITGB1 | Diagnostics biomarkers for prostate cancer in a non-invasive manner | 75 |
| Urine | Exosomes | Phosphatidylserine and lactosylceramide | Biomarkers in prostate cancer | 101 | |
| Urine | EVs | Steroid hormone and dehydroepiandrosterone sulphate | A non-invasive source to infer metabolic changes in prostate cancer | 103 | |
| Glioma | Patient-derived glioma stem-like cultures | Microvesicles, exosomes | miR-182-5p, miR-196a-5p and miR-10b-5p | Feasibility on cerebrospinal fluid | 72 |
| Hepatocellular carcinoma | Plasma | EVs | GPX3 and ARHGAP1 | A novel predictive biomarker in hepatocellular carcinoma | 78 |
| Plasma | Exosomes | tRNA-ValTAC-3, tRNA-GlyTCC-5 | Biomarkers | 89 | |
| Serum | Exosomes | circPTGRl, miR-449a-MET | Acting on tumor cell migration and invasion | 92 | |
| Colorectal cancer | Fecal | EVs | CD147 and A33 | Biomarkers in colorectal cancer | 81 |
| Serum | Exosomes | miR-208b | Predictive biomarkers of response to oxaliplatin-based therapy | 90 | |
| Endometrial cancer | Plasma | Exosomes | miR-15a-5p, miR-106b-5p | Diagnostic biomarkers in endometrial cancer | 84 |
| Non-small cell lung cancer | Serum | Exosomes | lncRNA TBILA、AGAP 2-AS 1 and SOX 2 OT | Promising biomarkers in non-small cell lung cancer | 73 |
| Gastric cancer | Plasma | Exosomes | let-7i-5p, miR-181a-5p, miR-1843 and miR-1307-3p, LZIC and SRSF6, lncFTH1–211 and lncPTMA-209 | Identifying responders to fluorouracil-based chemotherapy | 91 |
| Serum | Exosomes | circSHKBP 1 | Diagnostic and prognostic circulating | 93 | |
| Pancreatic cancer | Serum and plasma | Exosomes | GPC1 membrane protein | Biomarkers in gastric cancer | 95 |
| Plasma | Exosomes | Phosphatidylserine | Biomarkers of early malignancy | 102 |
3.1. Protein as a biomarker
It is always a project worthy of intense study to find the unique proteins in EVs. With the implementation of proteomics, many potential biomarkers of proteins have been discovered. Detection of specific protein expression in EVs can diagnose cancer. EVs derived from melanoma impair the maturation of DC and the production of cytokines and promote the TME of immunosuppression. Compared with healthy controls, programmed death-ligand 1 (PD-L1) in plasma circulating EVs derived from patients with metastatic melanoma is significantly upregulated (Fig. 2A and B)74. The analysis of PD-L1 level enables researchers to diagnose cancer against the response and non-response of PD-1 treatment. ITGA3 and ITGB1 in exosomes were highly expressed in patients with metastatic prostate cancer75. Moreover, detecting HSP 70 among patients with cancer can monitor whether there is a malignant solid tumor.
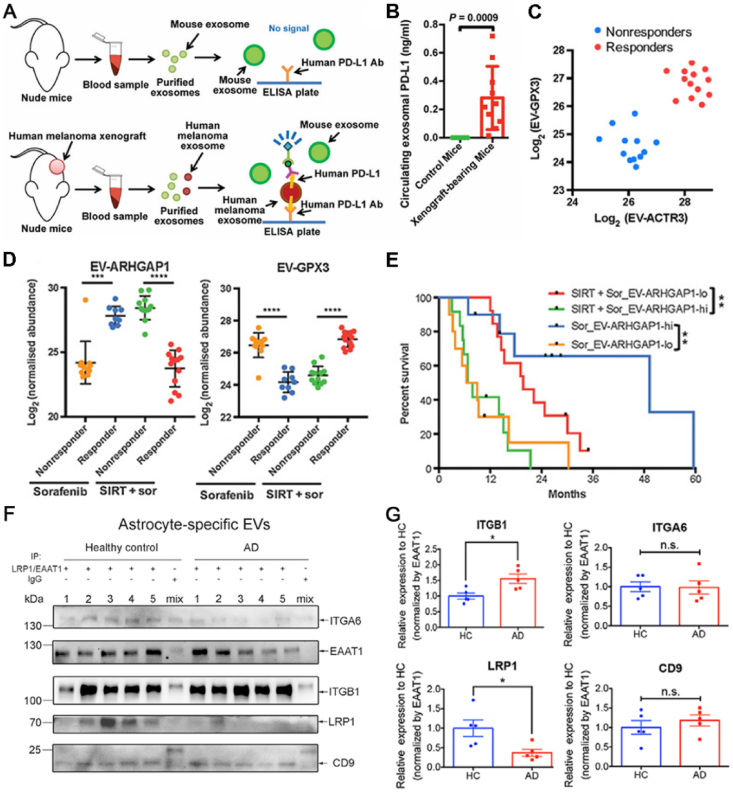
Figure 2.
(A) Schematic representation of ELISA for detecting human exosomal PD-L1. (B) PD-L1 levels on exosomes isolated from plasma samples of nude mice. Reprinted with the permission from Ref. 72. Copyright © 2018 Springer Nature Limited. (C) Levels of EV-GPX3 and EV-ACTR3 and corresponding protein expression in patients before SIRT + sorafenib treatment. (D) Abundance and (E) Percent survival of EV-ARHGAP1 and EV-GPX3. Reprinted with the permission from Ref. 76. Copyright © 2022 American Association for Cancer Research. (F) Western blot analysis of the astrocyte-specific EVs for the ITGB1 in the M7/astrocyte-EV module. (G) Western blot semiquantitative analysis plots. Reproduced with permission from Ref. 77. Copyright © 2021 The Authors.
During the initiation and advancement of cancer, there will be alterations in the composition and types of proteins found in EVs. Marine Cordonnier et al.76 found that the change in the level of exosomal PD-L1 in the peripheral circulatory system could be used as a relevant biomarker in melanoma treatment. Phosphorylation of AKT and ERK 1/2 in EVs of U87 glioma cells and non-small cell tumors decreased significantly after drug treatment in patients with good treatment response77. Timothy et al.78 used sorafenib + selective internal radiation therapy and sorafenib alone to treat patients with hepatocellular carcinoma. The proteomics results showed that high ARHGAP1 and low GPX3 expressed in plasma EVs can indicate the better efficacy of sorafenib monotherapy (Fig. 2C, D and E). Yang et al.79 discovered that astrocyte EV markers were significantly enriched in the M7 module, and exhibited significant positive correlations with Alzheimer’s disease traits (Fig. 2F). This suggests that protein markers integrin-β1 (ITGB1) derived from astrocyte-derived EVs may serve as potential biomarkers for Alzheimer’s disease (Fig. 2G). Additionally, proteins in EVs can judge cancer prognosis. Monica et al.80 compared the proteins in EVs between healthy patients and patients with melanoma after treatment. Among 75 proteins overexpressed in patients with disease progression, PDCD 6 IP had the highest discrimination value which had prognostic significance in melanoma. In colorectal cancer, Zhang et al.81 proved that CD147 and A33, two transmembrane proteins on fecal EVs had the potential to monitor the prognosis of colorectal cancer (Fig. 3A and B). It is necessary to detect several other protein biomarkers in combination.
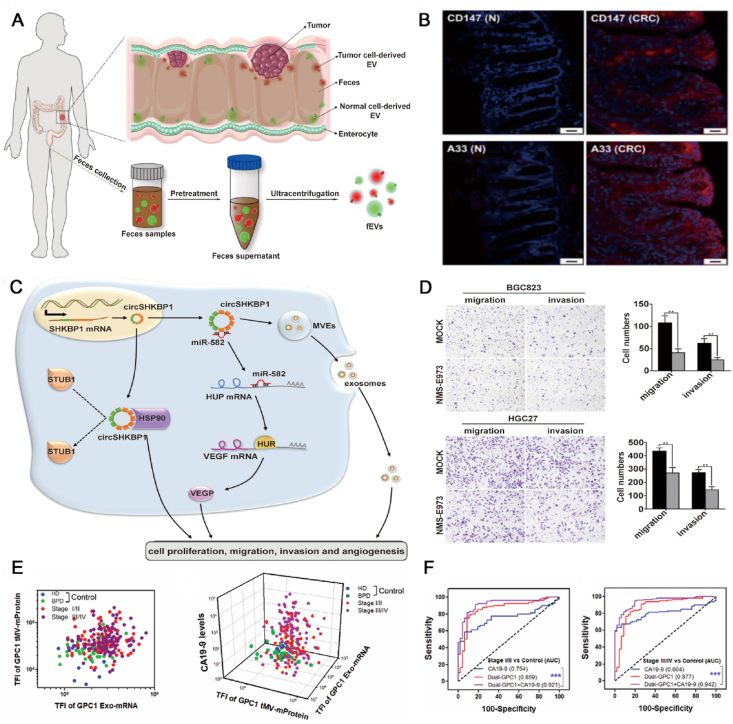
Figure 3.
(A) Scheme of biogenesis and isolation of faeces-derived EVs. (B) The expression levels of CD147 and A33. Reproduced with permission from Ref. 79. Copyright © 2023 Zhang et al. (C) Mechanism of circSHKBP1 regulating the progression of GC. (D) Migration and invasion of BGC823 and HGC27 cells. Reproduced with permission from Ref. 91. Copyright © 2020 Xie et al. (E) Scatter plot of CA19-9, GPC1 Exo-mRNA and tMV-mProtein expression. Reproduced with permission from Ref. 93. Copyright © 2023 Li et al. (F) ROC curves for PDAC stage I/II and III/IV patients. Reproduced with permission from Ref. 93. Copyright 2023 Li et al.
In conclusion, the application of proteins as biomarkers is still limited due to objective conditions. First, a single biomarker cannot accurately predict the immunotherapy response. Secondly, the difference of TIME and heterogeneity of tumor proteins make them have low predictive value in evaluating remission82. At the same time, sample analysis involves many protein extraction and analytical techniques, which makes it more difficult to convert into clinical application.
3.2. RNA as a biomarker
Compared with protein biomarkers, RNA biomarkers are more sensitive and specific which can be used to diagnose cancer more effectively. miRNAs are the most abundant type of foreign RNA cargo. The miRNAs are increasingly regarded as the key factors of tumor occurrence and metastasis and the transport of miRNAs promotes their evaluation as biomarkers83. Zhou et al.84 proved that exosomal miR-15a-5p in patients with endometrial cancer was downregulated and miR-106b-5p were upregulated significantly relative to healthy people. By detecting the content of these exosomes, we can judge the occurrence of cancer. Additionally, RNAs in GSC EVs were associated with angiogenesis in brain endothelial cells, and the presence of gliomas could be judged by monitoring the corresponding RNAs85. Moreover, in patients with melanoma, the content of BRAF RNA in EVs is easier to detect than that in plasma86. In addition, miR-1246, miR-451 and miR-122 in breast cancer MCF 7 exosomes were selectively secreted and enriched relative to other cell contents87.
The miRNAs in EVs isolated from plasma are differentially expressed between immune checkpoint therapy responders and non-responders, which could be used to judge the therapeutic effect88. The potential value of tRNA-derived small RNAs (tsRNAs) in cancer diagnosis has also been tapped. Zhu et al.89 compared exosomal tsRNA levels in hepatocellular carcinoma patients with those in healthy donors and showed a significant increase in plasma exosomes of tsRNAs in hepatocellular carcinoma patients. Additionally, Ning et al.90 found that miR-208b secreted by tumors promoted regulatory T (Treg) cell amplification, which was associated with decreased sensitivity of colorectal cancer to oxaliplatin chemotherapy. And so miR-208b is a possible biomarker for predicting the response to oxaliplatin therapy. In advanced gastric cancer (GC) patients, Guo et al.91 identified and validated the predictive ability of four miRNAs, two mRNAs, and two lncRNAs derived from plasma exosomes in NACT non-responders and responders. CircPTGRl, which could down-regulate miR 449 a-MET in recipient cells was highly abundant in malignant cells and cells of metastatic patients, so it could provide information on the prognosis and treatment of hepatocellular carcinoma92. In GC patients, overexpression of circSHKBP 1 in tumors was strongly associated with advanced pathological stages. CircSHKBP 1 promoted the development of GC through uptake of miR-582-3p and induction of HSP 9093. Zhao et al.94 discovered that the expression of lncRNA HOTTIP was elevated in patients with GC and was correlated with the overall survival rate of these patients (Fig. 3C and D). Li et al.95 discovered that the activation of specific oncogenes resulted in an increased expression of mRNA and its corresponding proteins in extracellular vesicles present in the bloodstream. The scatter plot depicting the expression of double GPC1 Exo-mRNA/tMV-mProtein demonstrated superior diagnostic performance (Fig. 3E and F). Although RNAs need to be further validated and evaluated as biomarkers, they are more suitable for use as circulating markers due to their unique biological properties. Although great progress has been achieved in identifying and characterizing circRNAs, many critical unknowns and limitations need to be addressed in further exploration.
3.3. Other biomarkers
The DNAs in EVs which are protected by double lipid membrane have good stability. They show great potential as cancer biomarkers. For example, Thakur BK et al.96 proved that most DNAs related to tumor exosomes were double-stranded and had high translation value for cancer detection. Maire et al.97 proved that the DNAs extracted from glioma EVs could not only detect tumor-specific genome-wide mutations, but also be used for subtype distribution of glioma. Additionally, liquid biopsies of EVs successfully isolated from NSCLC patients proved higher consistency than conventional tissue biopsies, and the DNAs isolated from plasma EVs of NSCLC patients could also be used to identify KRAS and the EGFR mutations, which proved that DNAs could be wonderful biomarkers98. Cheng et al.99 proved that the level of mtDNA was higher in patients with oral cancer. Thus, the levels of mtDNA can be taken as a biomarker in anti-PD-L1 immunotherapy and diagnosing diseases. As a biomarker for early detection, DNA has great prospects, especially for diseases that have no screening options at present, such as ovarian cancer, pancreatic cancer and GC. When combined with protein, RNA and other biomarkers, DNA can play a deeper role100. Other metabolites such as lipids, phosphatidylserine in EVs can also be used as biomarkers, but there are few studies on them. Tore Skotland et al.101 found the differences in lipid types between healthy people and patients by studying liposomes in urine foreign bodies from prostate cancer, which laid a foundation for liposomes as biomarkers. In addition, Raghava Sharma et al.102 revealed that the content of phosphatidylserine was higher in patients with pancreatic cancer through an ELISA-based determination system. In prostate cancer, the metabolites such as phosphatidylcholine, acyl carnitine, citrate and kynurenine had changed the urinary EVs of patients103.
Many substances in EVs are used as biomarkers, but there are few studies on other related biomarkers except protein and RNA. Due to the limitation of technology and the heterogeneity of cancer, there will be some differences in the expression of single protein and nucleic acid104.
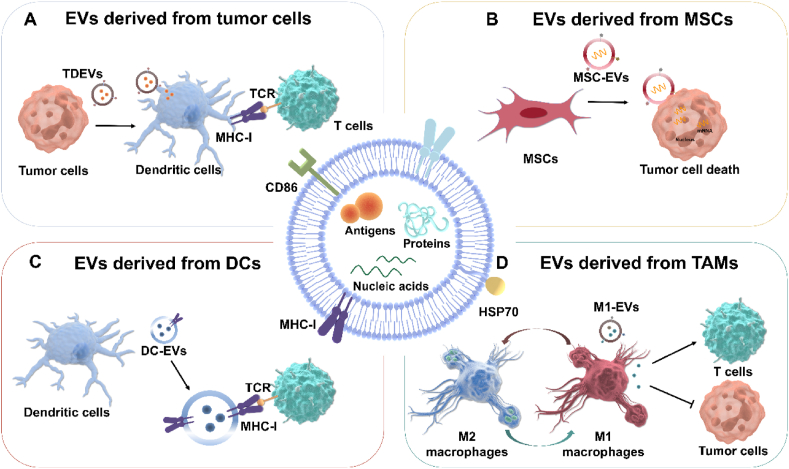
4. EVs from different sources
When used as diagnostic markers, EVs are often extracted from serum, plasma, urine, saliva, and other body fluid samples. EVs derived from tumor cells, MSCs, DCs, macrophages, and plants are often utilized in specific drug delivery systems based on their different biological properties.
4.1. EVs as diagnostic markers
In the clinical field, Tumor-derived EVs (TDEVs) are diagnostic biomarkers of tumor onsets by liquid biopsies (Fig. 4). VCAN and LGALS9, the damage-associated molecular pattern proteins, were highly enriched in pancreatic and lung adenocarcinoma-derived vesicles105. Liquid biopsies are non-invasive, sensitive, and convenient, using information in the blood to suggest tumor-specific information106. Due to the large number of cells present in whole blood, blood derivatives such as plasma and serum are the most studied biofluids in liquid biopsies (Table 3). The blood components are complex, the levels of EVs in whole blood are extremely low, and the sampling procedure can affect the analysis of EVs in the sample in some way107. Recently, the use of serum or plasma samples for biomarker studies has been widely discussed. Serum and plasma contain different subtypes of EVs and the presence of IV transmembrane proteins carried by platelet-derived EV is different. The concentration of EVs derived from platelets is higher in serum since platelets are activated and release substantial amounts of EVs during serum clotting in vitro108. The current findings suggest that plasma samples may be more suitable for tumor biomarker testing, with the choice of specific samples depending on which EVs the study is interested in. In addition, the extraction of plasma from whole blood requires the use of anticoagulants, the common anticoagulants being acid-citrate-dextrose (ACD), ethylenediaminetetraacetic acid (EDTA), and heparin. Zhang et al.109 found that anticoagulants do not influence the concentration of EVs, but the concentration of EVs decreases as time goes on. Therefore, rapid separation of the assay is required after sample extraction. However, the choice of anticoagulant affects the composition of EVs in the sample and alters EV profiles. For example, heparin and EDTA cause an immediate elevation in the number of CD15-positive myeloid EVs. Karimi et al.110 found that EDTA plasma contained more CD9 EVs and residual platelets than ACD-plasma and serum.
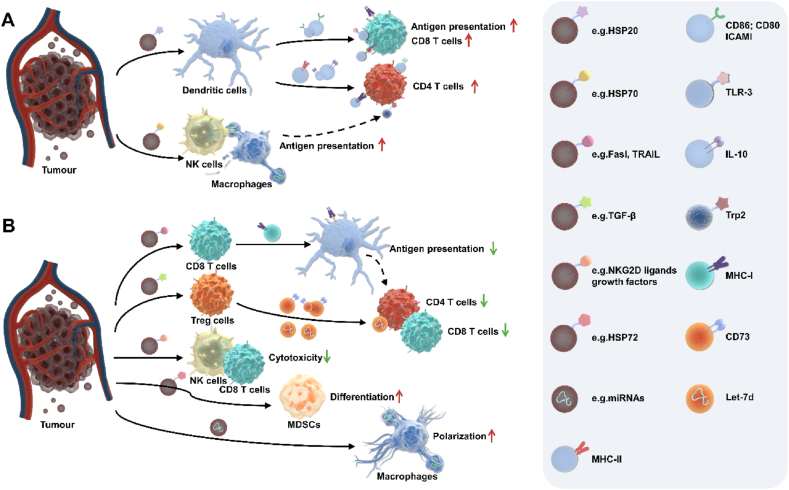
Figure 4.
(A) TDEVs enhance anti-tumor immune response. (B) TDEVs inhibit anti-tumor immune response.
4.2. EV as drug delivery systems
EVs of different cellular origins (e.g., tumor cells, MSCs, DCs, macrophages, and plants) differ significantly from each other and possess properties similar to their derived cells111. This section is to separately summarize their unique biological properties when it comes to drug delivery systems (Table 5 and Fig. 5).
Table 5.
Communication functions of EVs from different sources.
| Source of EVs | Communication object | Transmitted substance | Function |
|---|---|---|---|
| Pancreatic and lung adenocarcinoma-derived EVs | Non-tumor adjacent tissue | DAMP proteins VCAN and LGALS9 | Markers to distinguish cancer versus non-cancer |
| Glioblastoma -derived EVs |
PD-1 | PD-L1 | Stimulating NK-κB |
| Metastatic prostate cancer-derived EVs | Bone cell | CDCP1 protein | Detecting bone metastasis of prostate cancer |
| Breast cancer-derived EVs | Tumor-associated macrophages | miR-138-5p | Promoting the polarization of tumor-associated macrophages |
| Tumor cell-derived EVs | Neutrophils | IL-8, MCP-1 | Regulating their recruitment, activation, and apoptotic processes |
| Lung adenocarcinoma-derived EVs | Tumor-associated macrophages | miR-19b-3p | Facilitating M2 macrophage polarization |
| MSC-EVs | Mitochondrion | Mitochondrial transcription factor A mRNA | Attenuating mtDNA damage and inflammation |
| MSC-EVs | Brain microvascular endothelial cells | miR-612 | Promoting angiogenesis |
| DC-EVs | Hepatoma carcinoma cell | α-Fetoprotein | Triggering antigen-specific antitumor immune responses |
| Tumor-associated macrophages-EVs | Pancreatic ductal adenocarcinoma | miR-365 | Reducing drug sensitivity |
| Tumor-associated macrophages-EVs | Treg cells/Th 17 | miR-21-5p and miR-29-3p | Promoting the progression and metastasis of tumor cells |
| M2 macrophages-EVs | Epithelial cells | Fibromodulin | Protecting the tight junction |
| M2 macrophages-EVs | ECM | ECM enzymes | Degrading collagens |
| Ginseng-derived nanoparticles | Toll-like receptors on M2 macrophages | Ceramide lipids and proteins | Promoting polarization of M2 phenotype towards M1 phenotype and producing ROS |
| Lemon-derived EVs | Gastric cancer cell | Substances in EVs | Inducing cell death |
| Citrus limon-derived nanovesicles | TNF-related apoptosis-inducing ligand-receptor | Substances in EVs | Inhibiting cancer cell proliferation |
Figure 5.
(A) TDEVs activate immune response by promoting T cell expansion. (B) MSC-EVs inhibits tumor cell growth through miRNA-mediated mechanisms. (C) DC-EVs stimulate immune activation by promoting T cell expansion directly. (D) M1-EVs polarize TAMs from the M2 phenotype to the M1 phenotype, thus reversing the TME.
4.2.1. EVs derived from tumor cells
TDEVs have a natural tropism for tumor tissue, possibly because EVs acquire the same surface protein pool as their parental cells. EVs derived from melanoma cells accumulated more significantly in the tumor tissue compared to EVs from other sources112. Similar results were obtained with EVs from sarcoma cells, showing preferential tumor homing113. Accumulating evidence has shown that TDEVs are double-edged swords in the growth and recurrence of tumors. On the one hand, TDEVs carry tumor-associated antigens, which further activate CD8+ T cells to trigger cellular immunity114. Besides, TDEVs can inhibit tumorigenesis by improving the TME. For example, TDEVs could bind to the inhibitory immune receptor CD300a expressed on DCs to prevent Treg cell activation in the TME, thereby preventing melanoma development115. In addition, TDEVs affect the antitumor activity of neutrophils by regulating their recruitment, activation, and apoptotic processes. TDEV produces cytokines and chemokines that lead to neutrophil recruitment at the tumor site and more effective phagocytosis and killing of tumor cells116.
Conversely, TDEVs harbor a range of immunosuppressive factors naturally from the donor cells that suppress the immune response and facilitate the formation of immunosuppressive TMEs. TDEVs can stimulate nuclear factor κB (NF-κB) or intervene in the PD-L1/PD-1 axis to support tumor immune evasion117. In addition, TDEVs not only lead to macrophage polarization toward M2 phenotypes that promote tumor progression in TME but also inhibit the expression of NKG2D of natural killer (NK) cells to suppress the activity and cytotoxicity of them118. TDEVs have the ability to activate myeloid-derived suppressor cells (MDSCs), resulting in significant suppression of T cell function. This suppression leads to tumor proliferation, migration, immune evasion, and resistance to tumor immunotherapy119,120. Taken together, TDEVs can exert a bidirectional regulatory effect on TME by modulating immune cell function.
4.2.2. EVs derived from MSCs
MSCs are adult stem cells with multidirectional differentiation potential121. MSC-derived EVs (MSC-EVs) can function as an immunomodulator by mediating the interaction of bioactive substances such as multiple cytokines, chemokines, and miRNAs with receptors expressed on target cells122. EVs derived from adipose MSCs, acting as immunomodulators, could reduce the level of interferon γ and transcription factors while upregulating the level of immunomodulatory cytokines, thereby inhibiting local inflammatory responses. MSC-EVs containing the inflammation inhibitor IL-1RA could block the release of pro-inflammatory cytokines to induce rapid wound healing123. As for brain diseases, MSC-EVs can cross the blood–brain barrier smoothly and are enriched in cytokines related to nerve regeneration, thus promoting the survival of nerve cells and inhibiting their apoptosis124. MSC-EVs, enriched in enkephalinase, can downregulate the expression of β-amyloid peptide when co-cultured with neuroblastoma cells, implying that MSC-EVs could be an effective treatment for Alzheimer’s disease125. However, in addition to the therapeutic effects, MSC-EVs are at risk of promoting tumor growth. MSC-EVs activate the AKT signaling pathway of GC cells and enhance their invasive ability, thereby inducing epithelial–mesenchymal transition in GC cells126. Cytokines carried by MSC-EVs could activate signal transduction downstream of ERK1/2 and then increase the release of VEGF, thereby stimulating tumor progression127.
4.2.3. EVs derived from DCs
DCs are versatile antigen-presenting cells and build bridges between innate and adaptive immunity128. DC-EVs express MHC peptide complexes and co-stimulatory molecules on their surface, which provides conditions for its action with other immune cells129. DC-EVs can present antigens to initiate T-cells to induce anti-tumor immune responses and interact with antigen-specific CD4+ T lymphocytes to generate an adaptive immune response130. Liu et al.131 found that DCs that migrate to the lymphatic organs can stimulate alloreactive T cells by transferring clusters of EVs containing functional MHC molecules to resident DCs. DC-EVs can also activate CD8+ T cell and B cell responses in vivo antitumor immunity132. Moreover, DC-EVs can induce Treg cell production by producing TGF-β and IL-10, thus inhibiting the activation of antigen-specific T cells and inducing immune tolerance116. EVs derived from donor DCs could induce antigen-specific immune tolerance of the donor and significantly reduce expression of infiltrating leukocytes and interferons (IFN)-γ, thus markedly prolonging the survival of rat heart transplantation133. In addition, DC-EVs express transmembrane on their surface, which can activate NK cells and directly mediate the apoptosis of tumor cells 134. Recently, DC-EVs have been used as an improved strategy for cell-free vaccines with specific antigens that confer a personalized anti-tumor response. DC-EVs-based immunotherapy has shown a strong safety profile to date. Li et al.135 established a nanovaccine delivery platform using DC-EVs-based nanovaccines. Comparing DC-EVs-based personalized antigen carriers with liposomes, they found that DC-EVs improved lymph node delivery efficiency and promoted CD8+ T cell-mediated cytotoxic responses.
4.2.4. EVs derived from macrophages
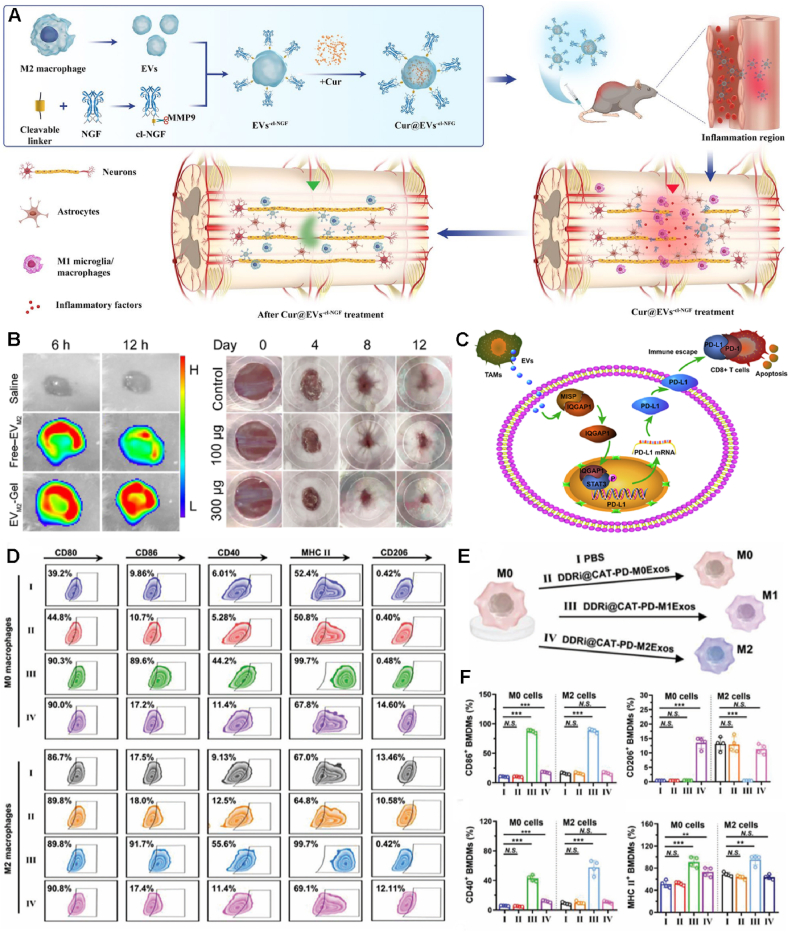
Macrophages can show different phenotypes under different environmental stimuli, including M1 macrophages and M2 macrophages. The balance between these two subtypes is important in fighting pathogens, regulating inflammation, and repairing tissues136. M2-EVs can exert anti-inflammatory effects and alleviate atherosclerosis through surface-bound chemokine receptors137. M2-EVs can promote endothelial cell proliferation, migration, and angiogenesis, with good tissue repair ability138. Moreover, M2-EVs possessed the neuroprotective effects of macrophages and could repair damaged neurons (Fig. 6A)139. Li et al.140 demonstrated bioactive M2-EVs can accelerate skin wound healing by promoting endothelial cell angiogenesis and macrophage polarization to the M2 phenotype in vivo (Fig. 6B).
Figure 6.
(A) Scheme diagram of engineered EVs derived from M2 macrophages for tospinal cord injury therapy. Reproduced with permission from Ref. 139. Copyright © 2021 The Author(s). (B) Proportion of fluorescence intensity and wound size of skin wounds extracted from different treatments. Reproduced with permission from Ref. 140. Copyright © 2023 American Chemical Society. (C) M2-EVs down-regulate MISP levels, up-regulate IQGAP1 levels and then phosphorylated STAT3 and promote PD-L1 transcript expression. Reproduced with permission from Ref. 142. Copyright © 2023 Elsevier B.V. (D–F) Flow cytometry analysis of phenotypes of macrophages. Reproduced with permission from Ref. 143. Copyright © 2022 Wiley-VCH GmbH. (E) Schematic diagram of macrophage polarization. Reproduced with permission from Ref. 143. Copyright © 2022, Wiley-VCH GmbH.
Since the TME is highly relevant to the inflammatory state, macrophage-derived EVs could regulate tumor development. Specifically, M1-EVs have tumor suppressor activity136. M1-EVs can enhance immune responses and suppress tumor growth by promoting the production of Th1 cytokines. In contrast, M2-EVs are not only able to promote tumor growth and migration by secreting chemokines and cytokines that inhibit inflammatory responses, but also promote immunosuppression and immune escape141. Recently, Wang et al.142 verified that M2-EVs upregulate PD-L1 via the MISP/IQGAP1/STAT3 axis to exert immunosuppressive effects and promote immune escape from HCC cells using plasmid transfection technology (Fig. 6C). Ma et al.143 transformed M1-type macrophage-derived exosomes into radiotherapy sensitizers, which loaded three functional molecules, namely catalase, DNA damage repair inhibitor and PD-L1 antibody, to achieve three different functions thus effectively improving the effect of radiotherapy. Since M1-type macrophage-derived exosomes can inherit proteins and nucleic acids from donor cells, it is very likely to promote other types of macrophages to become anti-tumor M1-type. The authors used M1-type exosomes to be uptaken by M0 and M2 macrophages to evaluate whether the polarity changes (Fig. 6E). After incubation with M1-type exosomes, the expressions of M1-type markers CD86, CD40, and MHC-II were increased in both M0 and M2 cells. After incubation with M2-type exosomes, the expression of M2-type marker CD206 was increased in M0 cells, and the expression of M2-type marker CD206 was decreased in M2-type cells after incubation with M1-type exosomes (Fig. 6D‒F).
4.2.5. EVs derived from plants
Research on the biogenesis of plant-derived exosome-like nanovesicles (PDENs) is still in its early stages. Most plant-derived vesicles used in biomedical applications are obtained by disrupting plant tissue, and the exact biogenetic pathway remains unclear144. There are three main biogenetic pathways for plant-derived vesicles: the MVB pathway, similar to exosome production in mammalian cells, and a unique exocyst-positive organelles pathway that differs morphologically from MVB, operates independently of endosomes and autophagosomes, and releases vesicles into extracellular space145. Furthermore, vacuoles are capable of fusing with the plasma membrane to release intraluminal vesicles146. Lipids are essential for the stability of PDENs due to their composition being primarily responsible for the maintenance of the bilayer membrane. Lipidomics analysis has revealed that the main lipid classes present in PDENs are phosphatidylic acid, phosphatidylethanolamine, and phosphatidylcholine147. In contrast, EV lipids of mammalian origin consist mainly of ceramides, cholesterol, glycolipids, sphingomyelin, and phosphatidylserine148. In addition to being rich in lipids, PDENs also contain phytochemicals and exhibit unique biological characteristics (Table 4)149, 150, 151, 152, 153, 154, 155.
Table 4.
Comparison of the molecular composition, and separation methods of EVs from mammalian cells and plant cells.
| Type | Source | Molecular composition |
Separation method | Ref. | |
|---|---|---|---|---|---|
| Lipid | Others | ||||
| Plant-derived exosome-like nanovesicles | Ginseng | DGMG, 59.4%; PE, 16.8%; Cer, 13.8% |
Ginsenoside Rg3 | Differential centrifugation and sucrose density gradient ultracentrifugation | 156 |
| Ginger | PA 37.0%, DGDG 39.9%, MGMG 16.9% | 6-Shogaol, 6-gingerol | Gradient ultracentrifugation | 149 | |
| Catharanthus roseus(L.) Don | EtherPC 16.6%; PG 15.7%; PI 14.0%; EtherPG 9.4% | Vinpocetine | Differential ultracentrifugation and enzyme digestion | 150 | |
| Edible tea flowers | PC, 26.6%; TG, 23.4%; PE, 15.2% | EGCG, ECG, EC, vitexin, myricetin-3-O-rhamnoside | Differential centrifugation and sucrose density gradient ultracentrifugation | 151 | |
| Grapefruit | PE 45.5%; PC 28.5% | Naringenin | Sucrose gradient centrifugation | 152 | |
| Mammalian-derived nanovesicles | Breast cancer cells | Sphingolipids, glycerophospholipids | Differential centrifugation | 153 | |
| Plasma | Cholesterol esters, sphingolipids, PC, PE | Centrifugation and magnetic bead separation | 154 | ||
| Triple-negative breast cancer cell | FA, LPC, LPE, PC, PE | Ultracentrifugation | 155 | ||
Cer, ceramide; DGMG, digalactosyl monoacylglycerol; DG, diglycerols; EC, epicatechin; ECG, epicatechin gallate; EGCG, epigallocatechin-3-gallate; EtherPC, ether-phosphatidylcholines; EtherPG, ether-phosphatidylglycerols; FA, fatty acid; LPC, lysophosphatidylcholine; LPE, lysophosphatidylethanolamine; MG, monoglycerols; PA, phosphatidic acids; PC, phosphatidylcholine; PE, phosphatidyl ethanolamine; PG, phosphatidylglycerols; PI, phosphatidylinositol; TG, triglyceride.
PDENs are membranous vesicles secreted by plants. As natural nano-agents, PDENs show unique functions in anti-tumor, immune function regulation and treatment of intestinal diseases. Studies on the use of PDENs as anticancer therapeutic agents have demonstrated their promising applications in cancer therapy147. PDENs can regulate macrophage polarization and promote to affect tumor growth. Specifically, the EVs-liked ginseng-derived nanoparticles (GDNPs) could induce M1 polarization and increase apoptosis in mouse melanoma cells. This suggested that GDNPs could be involved in the mammalian immune response as immunomodulators, thus providing novel nanomedicines for cancer immunotherapy156. In view of the rich pharmacological effects of citrus fruits, more in-depth studies of the vesicles isolated from them is warranted. Studies have shown that grapefruit-derived nano- and micro-vesicles could induce apoptosis157. In addition, lemon-derived EVs (LDEVs) were able to promote to generate reactive oxygen species (ROS) and promote TRAIL release. The released TRAIL were bind to DR5 and induce apoptosis of tumor cells158,159. Besides, LDEVs could enter tumor cells via caveolin-mediated endocytosis and micropinocytosis to deplete intracellular ATP to overcome multidrug resistance160. Due to the different uptake capacities of different cells for exosome-like nanovesicles derived from Asparagus cochinchinensis (Lour.) Merr. (ACNVs), ACNVs did not significantly damage normal hepatocytes but inhibited proliferation and induced apoptosis in hepatocellular carcinoma cells. Moreover, ACNVs treated with polyethylene glycol had stronger antitumor effects due to prolonged blood accumulation and increased tumor guidance161. The discovery of EVs from Pinus densiflora with antitumor effects suggested that these EVs from plant sap were new candidates for cancer treatment162. Additionally, the specific population of ginger-derived nanoparticles (GDNPs 2) showed favorable therapeutic effects on intestinal epithelial cells. This study proved that GDNPs 2 had good anti-inflammatory properties by down-regulating inflammatory factors, which provided a feasible treatment protocol for the prevention and treatment of colitis-associated cancer163.
5. EVs as drug delivery systems
With the advantages of high biocompatibility, cellular specificity, better stability, and limited immunogenicity compared to traditional synthetic drug delivery vehicles7, EVs have been applied in drug delivery systems to transport cargo into the cytoplasm of target cells8. EVs can protect themselves against the clearance of the immune system due to their small size and specificity of membrane-bound proteins. More importantly, EVs can successfully cross physiological barriers to reach target cells and efficiently transport different cargoes to target cells9.
5.1. Drug loading methods
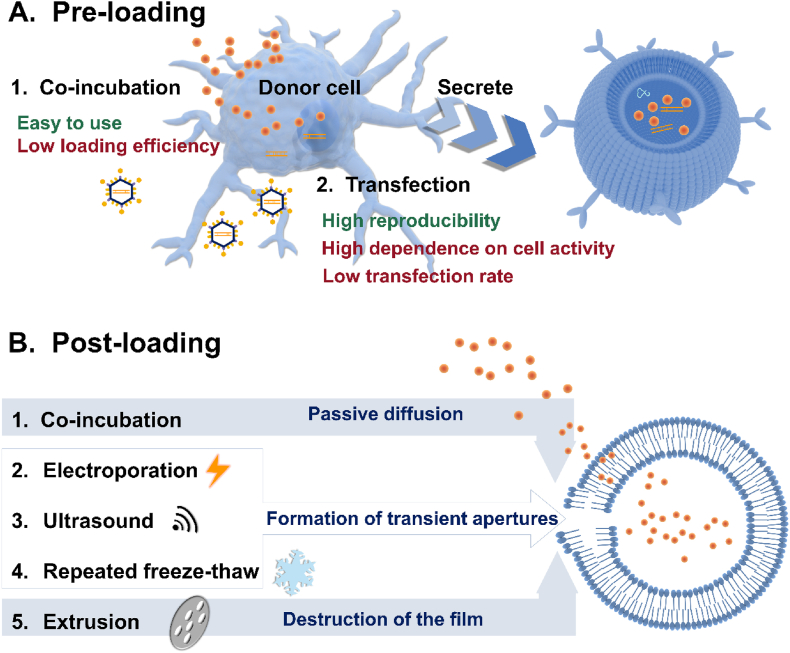
EVs can carry multiple types of drugs, mainly nucleic acid drugs, protein drugs, and small molecule drugs164. Ways to achieve EV loading are divided into post-loading and pre-loading in this section165.
5.1.1. Pre-loading
Pre-loading is the loading of drug molecules into parental cells so that the secreted EVs with membrane integrity carry drug molecules. The most common methods used are co-incubation and transfection (Fig. 7A)166,167. Co-incubation refers to the simultaneous incubation of drug molecules with parent cells that produce extracellular vesicles (EVs) under specific conditions. The drug molecules, particularly hydrophobic drugs, penetrate the cell by interacting with the phospholipid bilayer of the cell membrane and are then encapsulated within the subsequently released EVs168,169. The loading efficiency of curcumin co-incubated with adipose-derived mesenchymal stem cells was 82.26 ± 5.25%9. Transfection refers to parental cells that overexpress therapeutic molecules (e.g., miRNAs, siRNAs, proteins) and load these molecules into subsequent secretion into EVs170. The binding efficiency of carbataxel/TNF-related apoptosis-inducing ligand by transfection was 93.7 ± 1.53%171. Mizrak et al.172 use mRNA/protein transfection of parental cells, releasing EVs carrying suicide mRNA and protein-cytosine deaminase, inducing apoptosis in cancer cells. This loading method has high reproducibility, but the main disadvantage of transfection may be a low transfection rate due to the high dependence on cell activity and RNA species and/or sequence173.
Figure 7.
(A) Common methods of pre-loading. (B) Common methods of post-loading.
5.1.2. Post-loading
Post-loading refers to the passive or active loading of drug molecules directly to the surface bilayer membrane gap or cavity of the EVs after the separation of EVs. Passive loading is mainly achieved through co-incubation, and active loading requires physical or chemical methods to form many transient apertures, allowing drug molecules to enter168,169,174. Co-incubation, electroporation, ultrasound, repeated freeze-thaw, and extrusion are common methods (Fig. 7B).
Electroporation can form transient pores on the membrane by a high-intensity pulsed electric field, allowing drug molecules to enter. In general, the drug loading efficiency is very unstable due to variable parameters such as pulse medium, electric field strength, and pulse interval174. Notably, electroporation may decrease the zeta potential of EVs and thus destabilize the colloid. Similar to electroporation, ultrasound also forms transient pores175, but the efficiency of ultrasound is often higher than electroporation and further improves the incorporation of drug molecules176. However, the powerful mechanical shear force can damage the structure of the vehicles to a certain extent, resulting in a decrease in the drug-loading effect. Therefore, sonication in an ice bath is recommended. Hydrophobic drugs, such as curcumin and viola alcohol, can be directly bound to EVs by co-incubation. Experiments conducted by Kim et al.175 show that co-incubation is not as efficient as electroporation, and ultrasound is the most efficient method among them.
Alternatively, the repeated freeze-thaw and the extrusion method have been widely used in recent years. Repeated freeze-thaw is the mixing of EVs and drug molecules for a freeze-thaw cycle, during which the EVs membrane is temporarily destroyed, allowing drug molecules to enter the EVs and it is greatly affected by temperature, pH, etc.168. So, it has limited loading efficiency and may change the size of EVs and lead to aggregation. The extrusion method is to mix EVs with drugs and then pass the mixture through a nanoporous membrane168,177,178. However, exogenous loading techniques might lead to the accumulation of EVs and may also cause changes in their physicochemical and morphological characteristics. The utilization of different approaches has diverse effects on the drug-loading efficiency of EVs. Generally, the preloading method is simpler but requires more time. compared to ultrasonic-broken electroporation and extrusion. Moreover, while the post-loading method can enhance loading efficiency, it may also lead to decreased stability. Currently, numerous new technologies are being employed to improve drug efficiency, such as the microfluidic device, which effectively enhances DOX loading in U251-GM-derived EVs five times ex vivo. The acoustic fluid device simplifies drug loading. It is important to note that these advanced drug-loading methods offer simple operation, adjustability, quick encapsulation, and high loading efficiency as prominent features, providing a useful tool for efficient delivery179.
5.2. Application of EVs as drug delivery systems
At present, liposomes and polymer nanoparticles are widely used in DDSs. When they carry drugs into the body, they can promote drug transport and extend the half-life of drugs, thus improving their pharmacological properties180.
Firstly, due to the similarity of their biological components to cells, EVs exhibit excellent biocompatibility and are resistant to phagocytosis by macrophages in blood and other body fluids or degradation by lysosomes. Consequently, they have the capability of efficiently delivering drugs to target cells over extended distances181. Furthermore, due to variations in the cellular origin and nucleic acid cargo of different EVs, distinct surface molecules are displayed182. The specialized molecules present on the surface of EVs effectively inhibit immune responses in vivo by obstructing interactions with antibodies, opsonin, and coagulation factors180. Moreover, through engineering modifications or integration with other materials, EVs can exhibit enhanced delivery capabilities. The modified EVs may possess improved cellular absorption properties and significantly enhance drug delivery efficiency183. As gene therapies advance, the utilization of EVs will emerge as a pivotal catalyst for these therapeutic interventions. This section provides a concise overview of various applications of EVs as carriers for drug delivery (Table 6).
Table 6.
Summary of the application of EVs as drug delivery systems.
| Drug | Type of drug | Extracellular vesicle origin | Disease | Therapeutic effect |
|---|---|---|---|---|
| Paclitaxel | Chemical drug | Mesenchymal stem cell line SR4987 | Pancreatic adenocarcinoma | Inhibit the growth of human pancreatic cell line CFPAC-1 |
| Paclitaxel | Chemical drug | Macrophage | Lung cancer | Decreased proliferation of Lewis lung carcinoma cell |
| Paclitaxel | Chemical drug | Prostate cancer cell lines | Autologous prostate cancer | Enhanced drug cytotoxicity to tumor cells |
| Paclitaxel | Chemical drug | Mesenchymal stem cells | Breast cancer | Targeted primary tumors and reduced side effects |
| Paclitaxel | Chemical drug | Macrophages | Breast cancer | Released pro-inflammatory factors and suppressed tumor growth |
| Doxorubicin | Chemical drug | EL4 cells | T-cell lymphoma | Improved cellular uptake and intracellular release of DOX and slowed tumor growth |
| Doxorubicin | Chemical drug | Bone mesenchymal stem cells | Colorectal cancer | Inhibited tumor growth |
| Doxorubicin | Chemical drug | Bone mesenchymal stem cells | Breast cancer | Reduced tumor growth rate |
| Doxorubicin | Chemical drug | Dendritic cells | Breast cancer | Decreased proliferation of breast cancer cell |
| Paclitaxel and doxorubicin | Chemical drug | RAW264.7 cells | Triple negative breast cancer | Inhibit the growth of the orthotopic T11 tumors and human MDA-MB-231 tumors in xenograft model |
| Dopamine | Chemical drug | Kunming mice blood | Parkinson’s disease | Enhanced therapeutic effect resulting from drug delivery targeted to the brain |
| Triptolide | Chemical drug | SKOV3 cells | Ovarian cancer | Inhibited tumor cell proliferation and growth |
| Gemcitabine | Chemical drug | PANC-1 | Pancreatic cancer | Inhibited tumor growth |
| Curcumin | Chemical drug | The EL-4 (mouse lymphoma cell line) and RAW 264.7 (murine macrophage cell line) | Lipopolysaccharide-induced septic shock | Improved curcumin activity and reduced off-target effects |
| miR-146b | Gene drug | Mesenchymal stem cells | Glioblastoma multiforme | Inhibited glioma xenograft growth |
| miRNA-486-5p | Gene drug | Human cord blood endothelial colony-forming cells | Ischemic kidney injury | Reduced ischemic kidney injury |
| miR-122 | Gene drug | Adipose tissue-derived mesenchymal stem cells | Hepatocellular carcinoma | Increased the antitumor efficacy on tumor in vivo |
| miRNA-126 | Gene drug | MDA-MB-231 cells | Non-small cell lung cancer | Inhibited the formulation of lung metastasis |
| miR-195 | Gene drug | Stellate cells | Cholangiocarcinoma | Reduced tumor size of the orthotopic rat model and improved the overall animal survival |
| miRNA-199a | Gene drug | Mesenchymal stem cells | Hepatocellular carcinoma | Improved HCC chemosensitivity |
| miR-335-5p | Gene drug | Stellate cells | Hepatocellular carcinoma | Inhibited tumor cell invasion and reduced tumor size |
| Lamp2b | Gene drug | Dendritic cells | Alzheimer’s disease | Knocked out specific genes |
| si-ciRS-122 | Gene drug | HEK293T | Colorectal cancer | Mediated chemoresistance from drug-resistant cells to drug-sensitive cells |
| Spherical nucleic acids | Gene drug | PC-3 cells | Prostate cancer | Increased three thousand times the intensity of target miR-21 knockdown |
| miR-145-5p | Gene drug | Mesenchymal stem cells | Pancreatic ductal adenocarcinoma | Suppressed the proliferation and invasion of PDAC cells, enhanced apoptosis, and induced cell cycle arrest |
| miR-379 | Gene drug | Mesenchymal stem cells | Breast cancer | Promoted tumor necrosis |
5.2.1. EVs as carriers of chemicals
Currently, the majority of chemical drugs available in the market exhibit poor solubility, high toxicity, and short half-life, thereby resulting in limited absorption, low bioavailability, shortened blood circulation time, and multiple side effects. Although paclitaxel (PTX) demonstrates significant advantages in targeted lung cancer treatment, its systemic toxicity imposes limitations on its utilization184. Temozolomide (TMZ) is the primary pharmacological agent employed in the treatment of glioblastoma (GBM); however, a significant proportion of patients exhibit inherent resistance to this therapeutic intervention185. These challenges can be overcome by leveraging EVs as drug delivery systems to devise sophisticated delivery systems for PTX curcumin, TMZ, doxorubicin, and other therapeutic agents.
Zhu et al.186 developed a hybrid vesicle for the delivery of paclitaxel (PTX), consisting of bi-specific MSLN/PD-L1 CAR-T cell-derived exosomes expressing both mesothelin and PD-L1 single-chain variable fragments and lung-targeted liposomes. This delivery system improved tumor targeting ability and enhanced PTX-induced immunogenic cell death by blocking PD-L1 on the tumor surface, thereby promoting anti-tumor immune response. Wang et al.187 utilized EVs isolated from Raw264.7 cells for encapsulating PTX and subsequently engineered the EVs with RGE peptide to achieve specific targeting towards NRP-1, thereby enabling active glioma targeting of ICG/PTX@RGE-EV. The pH-responsive EVs were prepared by encapsulating sodium bicarbonate and PTX within the EVs. These pH-sensitive EVs exhibit enhanced drug release in response to acidic pH values within the intracellular/lysosomal compartments188. Meanwhile, Passcucci et al.166 made a discovery that MSCs can secrete exosomes when loaded with PTX. These exosomes are then able to deliver PTX to human pancreatic cancer cells, thus providing an effective anti-tumor treatment. TMZ, also the widely used chemotherapy drug, resistance is mainly due to the hyperactivation of O(6)-methylguanine-DNA methyltransferase, which repairs the damaged DNA, thus offsetting the influence of TMZ189. When TMZ is loaded into EVs, its chemotherapeutic resistance and off-target effects are significantly reduced. Due to the overexpression of heme oxygen-1 in TMZ-resistant GBM cells, the modification of EVs derived from bone marrow MSCs with a heme oxygen-1 specific peptide and the encapsulation of TMZ inside these EVs significantly enhanced the drug resistance of tumor cells to TMZ124. Curcumin, a diketone compound extracted from plants of Zingiberaceae and Araceae, has multiple effects of anti-tumor, anti-inflammation, and cholagogue. Sun et al.190 extracted exosomes from the EL-4 cell line and loaded them with curcumin, which significantly increased the solubility and utilization of the drug and reduced the toxicity of the drugs themselves. Exosomal doxorubicin (exoDOX) can avoid the cardiotoxicity of DOX and improve the therapeutic effect191. When DOX is loaded into EVs, its toxicity to the heart is significantly reduced192. Furthermore, Niu et al.193 used natural grapefruit EVs to load DOX, which makes it easier to deliver to tumor tissues.
5.2.2. EVs as carriers of gene-drug
Gene drugs are easily degraded by various nucleases in vivo, while EVs naturally carry nucleic acids. Among them, miRNAs and mRNAs have been extensively studied and applied. At present, complex methods have been developed to load specific RNAs into EVs in order to protect them from extracellular endonucleases, improve their stability and invisibility, and reduce their immunogenicity. These methods also enable the transportation of RNAs to target cells, primarily for the treatment of tumor diseases. Genetic material can be exchanged between cells by RNAs in EVs. When receptor cells ingest these EVs containing RNAs, siRNAs can silence them by interfering with the translation of target genes in the receptor cells. miRNAs can target and regulate the levels of mRNAs in the receptor cells so that they can be translated into the corresponding proteins in the recipient cells.
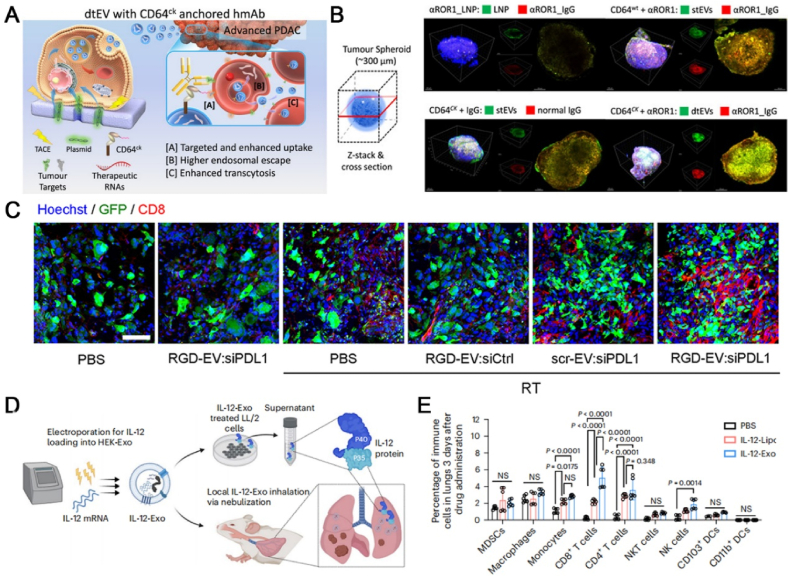
siRNA can induce gene silencing induced to treat various diseases. Because of its hydrophilicity, negative charge, and other characteristics, naked siRNA is easily degraded by serum nuclease which has a short half-life194. Poor targeting and immunogenicity also affect the efficacy of siRNA. However, as natural nucleic acid carriers, EVs can overcome these obstacles. siRNAs were first transported by exosomes to treat diseases in 2011, and their roles in tumor treatment have gradually become significant. Chiang et al.195 engineered double-targeted extracellular vesicles (dtEVs) for the delivery of therapeutic RNAs, resulting in effective inhibition of PDAC tumors (Fig. 8A). The presence of the CD64 protein on the surface of EVs, in combination with tissue-targeting peptides and humanized monoclonal antibodies, facilitates dual targeting to improve receptor-mediated tissue penetration and uptake by tumor cells. dtEVs exhibit a deeper tumor sphere penetration compared to single-targeted vectors carrying αROR1 or CK alone (Fig. 8B). Additionally, Tian et al. developed c(RGDyK)-conjugated extracellular vesicles (RGD-EVs) loaded with siRNA targeting PD-L1, which exhibited enhanced targeted delivery to GBM in a TAM-dependent manner following short-burst radiation therapy. However, the administration of radiotherapy resulted in an upregulation of PD-L1 expression, leading to a compromised state of anti-tumor immunity. Therefore, the delivery of siPD-L1 could offer significant therapeutic benefits for GBM treatment, which significantly augmented the presence of CD8+ T cells within the GBM microenvironment (Fig. 8C). To achieve targeted and efficient delivery of siRNA to specific cells, Pi et al.198 employed RNA nanotechnology to design ligand-expressed EVs as RNA nanoparticles, which exhibited significant tumor regression in models of prostate cancer, breast cancer, and colorectal cancer196. In addition, the bacterial adventitia vesicles contain proteins derived from parental cells and carry a variety of cargos, which can also serve as targeting delivery platforms197. Bioengineered bacterial outer membrane vesicles (OMVs) could deliver siRNAs to target and kill breast cancer cells overexpressing HER2. The laboratory utilized the E. coli K-12 W3110 strain, known for its production of a low-acylated LPS that decreases the immunogenicity of OMVs.
Figure 8.
(A) Scheme of dual-targeted therapeutic EVs containing high copy numbers of TP53 mRNA and siKRASG12D. Reproduced with permission from Ref. 195. Copyright © 2023 Springer Nature Limited. (B) Compared to stEVs or LNPs, dtEVs penetrate deeper into the tumor spheroids of PANC-1 cells. Reproduced with permission from Ref. 206. Copyright © 2023 Springer Nature Limited. (C) CD8 and nuclear immunostaining on brain tumor sections. Reproduced with permission from Ref. 207. Copyright © 2023 The Authors. (D) Analysis of the assessment of IL-12 expression. (E) Flow cytometry analysis of immune cells infiltrating lung tumors after 3 days of treatment. Reproduced with permission from Ref. 203. Copyright © 2024 Springer Nature Limited.
miRNA is most widely used in gene-drug transportation. The interaction between miRNAs and target genes leads to gene silence or translation inhibition, which regulates protein expression levels. Similar to siRNA, EVs can also protect miRNA from degradation, deliver them to target cells, promote cell uptake, and effectively improve drug delivery efficiency. For example, for gliomas, MSC-derived EVs containing miR-146b-5p reduced their expression and further reduced the invasiveness of gliomas by transporting miR-146b-5p to bind to EGFR mRNA in glial cells199. During the growth and invasion of GBM, the expression of miR-1 is decreased. EVs carrying miR-1 were developed to reduce the growth, angiogenesis, and invasion of GBM200. Viñas et al.201 reported that miRNA-486-5p was successfully transferred to exocrine for the treatment of acute renal injury. Let-7, as a miRNA for tumor inhibition, could be specifically delivered by exosomes to EGFR, thereby exerting an anti-breast cancer effect202.
mRNA can be directly translated into the specific target proteins in cells, and proteins can interact with receptors to treat diseases. Direct delivery of mRNAs can produce an obvious protective immune response, which is easily degraded by nuclease in plasma and tissues, while its molecular weight and negative charge make it difficult to cross cell membranes. Endogenous EVs can deliver mRNA/protein specifically and protect mRNA/protein from degradation. At the same time, the bilayer lipid membranes enable them to cross the biological barriers to enhance effective cellular uptake. Mizrak et al.172 successfully loaded therapeutic mRNA and protein-cytosine deaminase fused to uracil phosphoribosyltransferase into EVs for the treatment of related tumor diseases. The prodrug 5-fluorocytosine could be locally converted into an active agent by cytosine deaminase in tumors and cause tumor inhibition at the same time. Cheng et al.203 developed inhalable exosomes to deliver IL-12 mRNA, which could be administered locally in the lungs by inhalation. IL-12 mRNA promotes IFN-γ production resulting in a strongly activated state in the TME and enhances its immunogenicity (Fig. 8D‒E).
The molecules carried by EVs not only reflect the state of parent cells but also have a joint influence on the characteristics and functions of EVs. For instance, when EVs are loaded with mRNAs, they can enter receptor cells and be converted into proteins, thereby altering the biological state and function of the receptor cells204. In conclusion, compared to traditional DDSs, EVs have more prominent advantages in characteristics and structure. Their uptake ability can be enhanced through engineering methods, which makes the prospect of using EVs as DDSs more reliable205.
6. EVs modulate the tumor microenvironment
EVs can regulate tumor progression and microenvironment, and communicate with recipient cells through three different mechanisms: direct protein contact between membranes triggering intracellular signaling cascades206, membrane fusion releasing contents into receptor cells207, and phagocytosis and internalization by target cells (Table 7)208, 209, 210, 211, 212, 213, 214, 215, 216, 217.
Table 7.
EVs from different sources regulate TME.
| Type of EVs | Origin of extracellular vesicles | Cancer type | Mechanism | Ref. |
|---|---|---|---|---|
| Endogenous EVs | Prostate cancer cell | Prostate cancer | Inhibiting T cell activation in draining lymph nodes | 209 |
| Metastatic melanoma | Metastatic melanoma | PD-L1 mediated immune escape on EVs membrane surface | 74 | |
| Non-small cell lung cancer cells | Non-small cell lung cancer | Promoting CD8 T cell dysfunction in NSCLC | 210 | |
| Plasma from hepatocellular carcinoma patients | Hepatocellular carcinoma | Inducing NK cell dysfunction in HCC | 211 | |
| Human melanoma cell lines WM9 and WM164 | Melanoma | Adhesion to activated CD8 T cells via ICAM1-LFA-1 interaction inhibits CD8 T cell proliferation and cytotoxicity | 212 | |
| Exogenous EVs | Anti-MSLN CAR-T cells | Triple-negative breast cancer | The secretion of perforin and granzyme B effectively target MSLN-positive TNBC cells | 221 |
| Bone marrow MSC | Pancreatic ductal carcinoma | Hindering the polarization of tumor-associated macrophages | 213 | |
| Tumor antigen-stimulated dendritic cell | Melanoma | Up-regulating the expression of PD-L1 and activating endogenous T cells | 214 | |
| FAP genetically engineered tumor cells | Colon cancer, lung cancer, melanoma, breast cancer | Promoting the maturation of dendritic cells to target tumor cells | 215 | |
| EVs as drug delivery systems | Serum | Glioblastoma | Facilitating the maturation of DCs and the polarization of M2 TAMs into M1 TAMs. | 186 |
| Dendritic cells | Glioblastoma | Drainage of tumor-associated antigens to lymph nodes to activate classical antigen-presenting cell-induced immune stimulation | 216 | |
| GL261 cells | Glioblastoma | Promoting the polarization of macrophages | 224 | |
| Serum | Glioblastoma | Inhibiting tumor angiogenesis, up-regulatingSHP-2 activity | 217 | |
| Bone marrow-derived dendritic cells | Melanoma | Inducing the upregulation of MHC class I and PD-L1 on tumor cells, and enhancing the invasion of total immune cells | 226 | |
| Dendritic cell | Melanoma | Activation of toll-like receptor 4 signaling stimulates macrophage and DC maturation | 227 |
6.1. Endogenous EVs
EVs from pancreatic cancer have been shown to induce apoptosis of CD8+ T cells by activating the P38 MAPK signaling pathway. Additionally, TAMs lead to inhibition of T cell activity and induction of apoptosis by inhibiting T cell receptor signaling. Moreover, non-coding RNAs in cancer-derived EVs can inhibit immune responses by regulating T cell activation and function218. Exosomal miR-501-3p, derived from M2 macrophages, functions to inhibit the tumor suppressor gene TGFBR3 by activating the TGF-β signaling pathway219.
The DC derivative of EVs within the body has been demonstrated to incite the activity of cytotoxic T cells located within the tumor. This process, in turn, activates the TME. Zhen et al.220 produced exosomes from hepatocellular carcinoma (HCC) antigen-expressing dendritic cells and demonstrated their ability to induce effective antigen-specific immune responses in ectopic or in situ HCC mice. Treatment with these exosomes improved the tumor immune microenvironment of diethylnitrosamine-induced native tumors, along with a decrease in immunosuppressive cytokines and regulatory T cells.
6.2. Exogenous EVs
Recently, EVs derived from CAR-T cells express all surface membrane molecules from the parental cells. This promotes the expression of perforin and granzyme B, which in turn induce cytotoxicity in target cells221. By modifying the stimulation of tumor antigens on dendritic cells (tDC-Exo) with anti-CD3 and resistance to EGFR antibody, the secretion outside of the cell is simulated to activate T cells through CAR-T endogenous therapy. This process enhances T cell infiltration in tumor tissues and ensures specific immune responses targeting cancer antigens222. Additionally, CAR-T-derived EVs delivering RN7SL1 ultimately induced endogenous effector-memory and tumor-specific T cell expansion, generating an immune response in solid tumor cells with CAR antigen loss223. Inspired by CAT-T cell therapy, Fan et al.222 developed tumor antigen-stimulated DC-derived EVs that could provide the same essential signals for T-cell activation as CAR-T cells, as well as anti-CD3 and anti-EGFR to enable precision therapy for solid tumors.
6.3. EVs as drug delivery systems
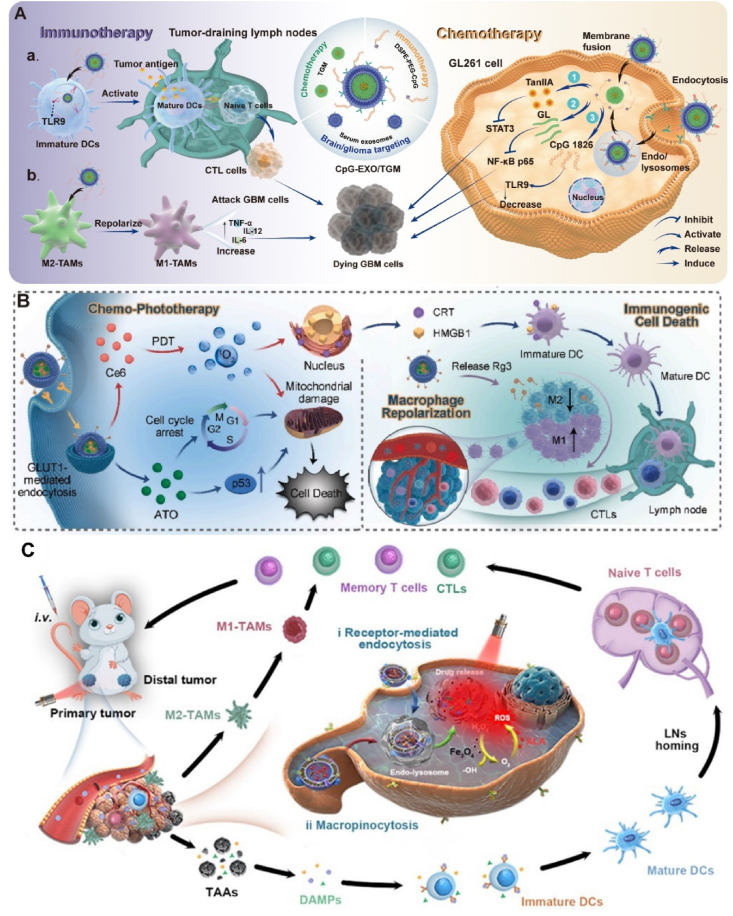
Our research group constructed CpG-EXO/TGM and used them in combination with temozolomide to treat glioma, which proved that it can regulate the immunosuppressive microenvironment (Fig. 9A)186. To kill tumor cells precisely with immunotherapy and chemotherapy, Li et al.224 modified ginsenoside Rg3 on the surface of EVs (Fig. 9B). Similarly, Wang et al.225 constructed a photodynamic therapeutically activated nanovaccine (Dex-HDL/ALA-Fe3O4) that co-delivers 5-aminopentulonic acid and Fe3O4, which achieves significant tumor site aggregation and lymph node homing, and can induce specific and memory anti-tumor immune responses (Fig. 9C).
Figure 9.
(A) The therapeutic mechanism of CpG-EXO/TGM. Reprinted with the permission from Ref. 227. Copyright © 2023 American Chemical Society. (B) Schematic depiction of Rg3-Exo/ATO@Ce6-mediated highly potent and orthotopic GBM-specific synergistic therapy. Reprinted with the permission from Ref. 217. Copyright © 2023 Elsevier B.V. (C) Mechanism diagram of personalized nanovaccine Dex-HDL/ALA-Fe3O4 enhancing lymph node aggregation and reprogramming tumor microenvironment. Reprinted with the permission from Ref. 218. Copyright © 2024 American Chemical Society.
DC-EVs have been demonstrated to elicit an antitumor immune response through the activation of antigen-specific T cells. Veerman et al.226 further demonstrated that the combination of EVs with anti-PD-1 or anti-PD-L1 could improve the response rate to immune checkpoint inhibition therapy. Matsumoto et al.227 activated DCs with LPS and IFN-γ and added ovalbumin to the collected EVs to significantly enhance the immune activity of EVs. Zuo et al.228 also modified the surface of EVs to promote the recruitment, accumulation and activation of DCs. Hansen et al.229 found that EVs derived from CD4+ T cells could be used as immune adjuvants to enhance the immune response to STING agonists. Shin et al.230 found that EVs produced by IL-2-stimulated CD4+ T cells were more effective in inducing CD8+ T cell-mediated antitumor responses without activating Treg cells as IL-2 did. TDEVs contain a large number of neoantigens, which have good immunostimulant effects and can be used to develop personalized immunotherapies. Zuo et al.231 found that multiple tumor-associated antigens of EVs could activate DCs and enhance tumor recognition. In addition, Kim et al.232 loaded misfolded proteins (MPs) into RSVF-expressing EVs and found that bRSVF- EV not only induced tumor cell heterodimerization to trigger the innate immune response, but also delivered MPs directly to the cytoplasm of the cancer cells to induce ICD. Cheng et al.233 genetically engineered natural exosomes and modified them to create genetically engineered exosomes that display antibodies on their surface and contain immunomodulatory proteins. These genetically engineered multifunctional immunomodulatory exosomes demonstrated strong cytotoxicity against triple-negative breast cancer cells, while also exhibiting enhanced stability and biocompatibility.
7. Conclusions and perspectives
Over the past decade, EVs have emerged as valuable biomarkers and DDSs for tumor therapy. This progress is evident in the growing number of high-quality publications and citations. EVs obtained from the body fluids of tumor patients carry specially labeled molecules such as proteins, nucleic acids and specific metabolites, which have shown great value as prognostic and diagnostic biomarkers for tumors234. These methods of detecting EVs are widely accepted by patients due to their real-time and minimally invasive sample collection, facilitating the diagnosis of disease and giving a comprehensive insight to the overall condition of the disease. Additionally, EVs not only reflect the biological properties and states of source cells, but also are common signaling mediators between cells. They can be employed to construct novel targeted DDSs to carry drugs. Compared with conventional DDSs, EVs have better biocompatibility as well as are less likely to elicit an immune response, so they have extended circulation time in the body and are able to deliver drugs to tumor tissues more efficiently8. Moreover, EVs have target specificity, and by altering the surface proteins of EVs can be made to bind to specific receptors on the surface of tumor cells, thus improving drug targeting and therapeutic efficacy. These engineered EVs are more plausible prospects as DDSs235. More surprisingly, EVs have a natural tumor killing ability and can be used as immunostimulation and cancer vaccine for clinical treatment (Table 8).
Table 8.
Summary of EVs in cancer immunotherapy.
| Purpose | Source of EVs | Cancer | Therapeutic effect |
|---|---|---|---|
| Delivery carrier | Red blood cells (RBCs) | Breast cancer | Inhibited tumor growth by activating the RIG-I cascade while increasing immune cell infiltration |
| Immunostimulation | Dendritic cells | – | Enhanced innate immunity and stimulated macrophages and DCs |
| Immunostimulation | Tumor cells | Breast cancer | Release of DNA-containing exosomes, activation of DCs via STING signaling |
| Immunostimulation | Tumor cells | Breast cancer | Induced DC maturation |
| Immunostimulation | Tumor cells | Hepatocellular carcinoma | Generation of long-term memory T cells and activation of potent anti-tumor immunity |
| Cancer vaccine | Dendritic cells | – | Inhibition of tumor growth and extension of survival |
| Cancer vaccine | Dendritic cells | Melanoma | Suppressed tumor growth, eradicated lung metastases, and extended survival |
| Cancer vaccine | Breast cancer | Triple negative breast cancer | Enhanced CD8+ T cell responses in tumor microenvironment |
| Cancer vaccine | Tumor cells and dendritic cells | Neck squamous cell carcinoma | Robust innate immune response |
| Cancer vaccine | Immunogenically dying tumor cells | Pancreatic ductal adenocarcinoma | Delayed tumor growth |
| Cancer vaccine | Melanoma cytomembrane and Salmonella outer membrane | Melanoma | Activated immune system and induced anti-tumour immune response |
| Cancer vaccine | Bacteria | Colon cancer | Inhibited melanoma progression |
| Cancer vaccine | Outer membrane | Pulmonary metastatic melanoma and subcutaneous colon tumor | Inhibited tumor growth and re-invasion |
| Cancer vaccine | Bacterial outer membrane | – | Efficient recognition and uptake by DCs after binding to tumor antigens |
| Cancer vaccine | Bacterial outer membrane | – | Promoted DC maturation and activated antigen presentation function |
‒Not available.
In the clinical settings, the issues of precise drug delivery and standardized production of EVs still exist. Whether surface membrane protein quantification and the number of EVs can be used for indirect quantification remains highly controversial. As research on various EVs continues, new surface markers continue to be discovered. And with that, it has driven the development of new extraction and assay techniques for EVs, which gives the possibility to prepare EVs products with higher purity and less batch variation, as well as to promote precise diagnostics for EVs-based liquid biopsies. It is widely recognized that EVs exhibit a high level of biocompatibility and degradability, and none of the oncology treatments based on EVs have demonstrated significant dose toxicity. However, since some EVs are naturally immunogenic and produce immune rejection or cytokine storm syndrome236, isolating inflammatory substances in EVs by pre-blocking or knocking down their immune proteins may evade immune recognition and allow better targeting. Therefore, researchers still need to pay more attention to the safety of EVs administration, and they are trying to design DDSs with high safety, desirable efficacy, and high clinical translational potential.
EVs-based tumor immunotherapy is also emerging as a cutting-edge strategy to regulate tumor progression and TME. EVs of different origins carry bioactive molecules capable of exerting bi-directional effects of immune activation and immune suppression. For one thing, EVs can enhance the growth, maturation of targeted immune cells, thus exerting a robust anti-tumor efficacy. For another, EVs can support immune escape of tumors and induce drug resistance of tumor cells by promoting suppressive immune cells, including Treg cells, MDSCs, and M2-like TAMs, or by inducing T-cell depletion. Recently, more potential immunotherapeutic strategies are derived based on the mechanism of action of different EVs for TME. For example, TDEVs are more inclined to promote tumor progression, and inhibiting their biogenesis would significantly suppress tumor growth. Rab27a plays a role in the docking of multivesicular endosomes to the plasma membrane and is a commonly used therapeutic target in the clinic. Rab27a silencing inhibits the secretion of TDEVs237.
In the future, considering the powerful role of EVs as biomarkers and DDSs for tumor treatment, we put forward the following views on the development prospect of EVs: (1) Establish EV-TRACK knowledgebase to promote the comparison of different study methods of EVs and make guidelines for experimental design and report in EVs research to reap more vital biomarker findings238. (2) The further development of EVs as new DDSs must be based on a broader and deeper understanding of tumor immune system and the advancement of basic research. (3) The artificial intelligence and machine learning can be applied in solving the heterogeneity of EVs and transforming a large amount of information from omics methods into practical clinical applications239,240. However, despite the wealth of preclinical data suggesting the positive effects of EVs, we are still waiting for comprehensive results from an accumulating number of human clinical trials241.
Author contributions
Xue Wang: Writing – original draft. Wenjing Chen: Writing – original draft. Wei Zeng: Writing – original draft. Kuanhan Feng: Writing – original draft. Yu Zheng: Software. Ping Wang: Software. Fucai Chen: Software. Wen Zhang: Validation. Liuqing Di: Writing – review & editing. Ruoning Wang: Writing – review & editing, Funding acquisition.
Conflicts of interest
The authors declare no conflict of interest.
Acknowledgments
This work was supported by the National Natural Science Foundation of China (No. 82274104, 82074024, 82374042, 82104688, China), National Ten Thousand Talents Program for Young Top-notch Talents, the Natural Science Foundation of Jiangsu Province (No. BK20240144, China), Jiangsu 333 High level Talent Training Project, the Innovation Projects of State Key Laboratory on Technologies for Chinese Medicine Pharmaceutical Process Control and Intelligent Manufacture (No. NZYSKL240103, China), Nanjing University of Chinese Medicine’s Project (No. RC202407, China). This work was supported by the College Students’ Innovative Entrepreneurial Training (No. 202210315056Y, 202310315055Z, China).
Footnotes
Peer review under the responsibility of Chinese Pharmaceutical Association and Institute of Materia Medica, Chinese Academy of Medical Sciences.
Contributor Information
Wen Zhang, Email: wenzhang@njucm.edu.cn.
Liuqing Di, Email: diliuqing@njucm.edu.cn.
Ruoning Wang, Email: ruoningw@njucm.edu.cn.
References
- 1.Pitt J.M., Marabelle A., Eggermont A., Soria J.C., Kroemer G., Zitvogel L. Targeting the tumor microenvironment: removing obstruction to anticancer immune responses and immunotherapy. Ann Oncol. 2016;27:1482–1492. doi: 10.1093/annonc/mdw168. [DOI] [PubMed] [Google Scholar]
- 2.Ando Y., Mariano C., Shen K. Engineered in vitro tumor models for cell-based immunotherapy. Acta Biomater. 2021;132:345–359. doi: 10.1016/j.actbio.2021.03.076. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Maacha S., Bhat A.A., Jimenez L., Raza A., Haris M., Uddin S., et al. Extracellular vesicles-mediated intercellular communication: roles in the tumor microenvironment and anti-cancer drug resistance. Mol Cancer. 2019;18:55. doi: 10.1186/s12943-019-0965-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Feng J., Xiu Q., Huang Y., Troyer Z., Li B., Zheng L. Plant-derived vesicle-like nanoparticles as promising biotherapeutic tools: present and future. Adv Mater. 2023;35 doi: 10.1002/adma.202207826. [DOI] [PubMed] [Google Scholar]
- 5.van Niel G., Carter D.R.F., Clayton A., Lambert D.W., Raposo G., Vader P. Challenges and directions in studying cell-cell communication by extracellular vesicles. Nat Rev Mol Cell Biol. 2022;23:369–382. doi: 10.1038/s41580-022-00460-3. [DOI] [PubMed] [Google Scholar]
- 6.Yu W., Hurley J., Roberts D., Chakrabortty S.K., Enderle D., Noerholm M., et al. Exosome-based liquid biopsies in cancer: opportunities and challenges. Ann Oncol. 2021;32:466–477. doi: 10.1016/j.annonc.2021.01.074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Huang L., Rong Y., Tang X., Yi K., Qi P., Hou J., et al. Engineered exosomes as an in situ DC-primed vaccine to boost antitumor immunity in breast cancer. Mol Cancer. 2022;21:45. doi: 10.1186/s12943-022-01515-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Meng W., He C., Hao Y., Wang L., Li L., Zhu G. Prospects and challenges of extracellular vesicle-based drug delivery system: considering cell source. Drug Deliv. 2020;27:585–598. doi: 10.1080/10717544.2020.1748758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Zhang X., Zhang H., Gu J., Zhang J., Shi H., Qian H., et al. Engineered extracellular vesicles for cancer therapy. Adv Mater. 2021;33 doi: 10.1002/adma.202005709. [DOI] [PubMed] [Google Scholar]
- 10.Rong Y., Wang Z., Tang P., Wang J., Ji C., Chang J., et al. Engineered extracellular vesicles for delivery of siRNA promoting targeted repair of traumatic spinal cord injury. Bioact Mater. 2023;23:328–342. doi: 10.1016/j.bioactmat.2022.11.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Liu C., Wang Y., Li L., He D., Chi J., Li Q., et al. Engineered extracellular vesicles and their mimetics for cancer immunotherapy. J Control Release. 2022;349:679–698. doi: 10.1016/j.jconrel.2022.05.062. [DOI] [PubMed] [Google Scholar]
- 12.Zitvogel L., Regnault A., Lozier A., Wolfers J., Flament C., Tenza D., et al. Eradication of established murine tumors using a novel cell-free vaccine: dendritic cell-derived exosomes. Nat Med. 1998;4:594–600. doi: 10.1038/nm0598-594. [DOI] [PubMed] [Google Scholar]
- 13.Zhang L., Sun M., He Z., Sun J., Li H., Luo Q. Multi-functional extracellular vesicles: potentials in cancer immunotherapy. Cancer Lett. 2022;551 doi: 10.1016/j.canlet.2022.215934. [DOI] [PubMed] [Google Scholar]
- 14.Kalluri R., LeBleu V.S. The biology, function, and biomedical applications of exosomes. Science. 2020;367:eaau6977. doi: 10.1126/science.aau6977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Stahl P.D., Raposo G. Extracellular vesicles: exosomes and microvesicles, integrators of homeostasis. Physiology. 2019;34:169–177. doi: 10.1152/physiol.00045.2018. [DOI] [PubMed] [Google Scholar]
- 16.Santavanond J.P., Rutter S.F., Atkin-Smith G.K., Poon I.K.H. Apoptotic bodies: mechanism of formation, isolation and functional relevance. Subcell Biochem. 2021;97:61–88. doi: 10.1007/978-3-030-67171-6_4. [DOI] [PubMed] [Google Scholar]
- 17.Zhao D., Tao W., Li S., Chen Y., Sun Y., He Z., et al. Apoptotic body-mediated intercellular delivery for enhanced drug penetration and whole tumor destruction. Sci Adv. 2021;7:eabg0880. doi: 10.1126/sciadv.abg0880. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.van Niel G., D’Angelo G., Raposo G. Shedding light on the cell biology of extracellular vesicles. Nat Rev Mol Cell Biol. 2018;19:213–228. doi: 10.1038/nrm.2017.125. [DOI] [PubMed] [Google Scholar]
- 19.Peng X., Yang L., Ma Y., Li Y., Li H. Focus on the morphogenesis, fate and the role in tumor progression of multivesicular bodies. Cell Commun Signal. 2020;18:122. doi: 10.1186/s12964-020-00619-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Villarroya-Beltri C., Baixauli F., Mittelbrunn M., Fernández-Delgado I., Torralba D., Moreno-Gonzalo O., et al. ISGylation controls exosome secretion by promoting lysosomal degradation of MVB proteins. Nat Commun. 2016;7 doi: 10.1038/ncomms13588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Langemeyer L., Fröhlich F., Ungermann C. Rab GTPase function in endosome and lysosome biogenesis. Trends Cell Biol. 2018;28:957–970. doi: 10.1016/j.tcb.2018.06.007. [DOI] [PubMed] [Google Scholar]
- 22.Wang Q., Lu Q. Plasma membrane-derived extracellular microvesicles mediate non-canonical intercellular NOTCH signaling. Nat Commun. 2017;8:709. doi: 10.1038/s41467-017-00767-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wortzel I., Dror S., Kenific C.M., Lyden D. Exosome-mediated metastasis: communication from a distance. Dev Cell. 2019;49:347–360. doi: 10.1016/j.devcel.2019.04.011. [DOI] [PubMed] [Google Scholar]
- 24.Hermann D.M., Xin W., Bähr M., Giebel B., Doeppner T.R. Emerging roles of extracellular vesicle-associated non-coding RNAs in hypoxia: insights from cancer, myocardial infarction and ischemic stroke. Theranostics. 2022;12:5776–5802. doi: 10.7150/thno.73931. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Colombo M., Raposo G., Théry C. Biogenesis, secretion, and intercellular interactions of exosomes and other extracellular vesicles. Annu Rev Cell Dev Biol. 2014;30:255–289. doi: 10.1146/annurev-cellbio-101512-122326. [DOI] [PubMed] [Google Scholar]
- 26.Xie F., Zhou X., Fang M., Li H., Su P., Tu Y., et al. Extracellular vesicles in cancer immune microenvironment and cancer immunotherapy. Adv Sci (Weinh) 2019;6 doi: 10.1002/advs.201901779. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Banushi B., Joseph S.R., Lum B., Lee J.J., Simpson F. Endocytosis in cancer and cancer therapy. Nat Rev Cancer. 2023;23:450–473. doi: 10.1038/s41568-023-00574-6. [DOI] [PubMed] [Google Scholar]
- 28.Ju S.W., Mu J.Y., Dokland T., Zhuang X.Y., Wang Q.L., Jiang H., et al. Grape exosome-like nanoparticles induce intestinal stem cells and protect mice from DSS-induced colitis. Mol Ther. 2013;21:1345–1357. doi: 10.1038/mt.2013.64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Rahimi Ghiasi M., Rahimi E., Amirkhani Z., Salehi R. Leucine-rich repeat-containing G-protein coupled receptor 5 gene overexpression of the rat small intestinal progenitor cells in response to orally administered grape exosome-like nanovesicles. Adv Biomed Res. 2018;7:125. doi: 10.4103/abr.abr_114_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wang B.M., Zhuang X.Y., Deng Z.B., Jiang H., Mu J.Y., Wang Q.L., et al. Targeted drug delivery to intestinal macrophages by bioactive nanovesicles released from grapefruit. Mol Ther. 2014;22:522–534. doi: 10.1038/mt.2013.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhuang X.Y., Deng Z.B., Mu J.Y., Zhang L.F., Yan J., Miller D., et al. Ginger-derived nanoparticles protect against alcohol-induced liver damage. J Extracell Vesicles. 2015;4 doi: 10.3402/jev.v4.28713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kim J., Zhu Y., Chen S., Wang D., Zhang S., Xia J., et al. Anti-glioma effect of ginseng-derived exosomes-like nanoparticles by active blood‒brain-barrier penetration and tumor microenvironment modulation. J Nanobiotechnol. 2023;21:253. doi: 10.1186/s12951-023-02006-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Lam S.M., Zhang C., Wang Z., Ni Z., Zhang S., Yang S., et al. A multi-omics investigation of the composition and function of extracellular vesicles along the temporal trajectory of COVID-19. Nat Metab. 2021;3:909–922. doi: 10.1038/s42255-021-00425-4. [DOI] [PubMed] [Google Scholar]
- 34.Chen Q.B., Li Q., Liang Y.Q., Zu M.H., Chen N.X., Canup B.S.B., et al. Natural exosome-like nanovesicles from edible tea flowers suppress metastatic breast cancer via ROS generation and microbiota modulation. Acta Pharm Sin B. 2022;12:907–923. doi: 10.1016/j.apsb.2021.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Tomiyama E., Fujita K., Matsuzaki K., Narumi R., Yamamoto A., Uemura T., et al. EphA2 on urinary extracellular vesicles as a novel biomarker for bladder cancer diagnosis and its effect on the invasiveness of bladder cancer. Br J Cancer. 2022;127:1312–1323. doi: 10.1038/s41416-022-01860-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Tian F., Zhang S., Liu C., Han Z., Liu Y., Deng J., et al. Protein analysis of extracellular vesicles to monitor and predict therapeutic response in metastatic breast cancer. Nat Commun. 2021;12:2536. doi: 10.1038/s41467-021-22913-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Yang M., Luo Q.Q., Chen X., Chen F.X. Bitter melon derived extracellular vesicles enhance the therapeutic effects and reduce the drug resistance of 5-fluorouracil on oral squamous cell carcinoma. J Nanobiotechnol. 2021;19:259. doi: 10.1186/s12951-021-00995-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Fujita D., Arai T., Komori H., Shirasaki Y., Wakayama T., Nakanishi T., et al. Apple-derived nanoparticles modulate expression of organic-anion-transporting polypeptide (OATP) 2B1 in Caco-2 cells. Mol Pharm. 2018;15:5772–5780. doi: 10.1021/acs.molpharmaceut.8b00921. [DOI] [PubMed] [Google Scholar]
- 39.Timms K., Holder B., Day A., McLaughlin J., Forbes K.A., Westwood M. Watermelon-derived extracellular vesicles influence human ex vivo placental cell behavior by altering intestinal secretions. Mol Nutr Food Res. 2022;66 doi: 10.1002/mnfr.202200013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Aquilano K., Ceci V., Gismondi A., De Stefano S., Iacovelli F., Faraonio R., et al. Adipocyte metabolism is improved by TNF receptor-targeting small RNAs identified from dried nuts. Commun Biol. 2019;2:317. doi: 10.1038/s42003-019-0563-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Liu Y., Tan M.L., Zhu W.J., Cao Y.N., Peng L.X., Yan Z.Y., et al. In vitro effects of tartary buckwheat-derived nanovesicles on gut microbiota. J Agric Food Chem. 2022;70:2616–2629. doi: 10.1021/acs.jafc.1c07658. [DOI] [PubMed] [Google Scholar]
- 42.Li L., Zhang F., Zhang J., Shi X., Wu H., Chao X., et al. Identifying serum small extracellular vesicle microRNA as a noninvasive diagnostic and prognostic biomarker for ovarian cancer. ACS Nano. 2023;17:19197–19210. doi: 10.1021/acsnano.3c05694. [DOI] [PubMed] [Google Scholar]
- 43.You D.G., An J.Y., Um W., Jung J.M., Oh B.H., Nguyen V.Q., et al. Stem cell-derived extracellular vesicle-bearing dermal filler ameliorates the dermis microenvironment by supporting CD301b-expressing macrophages. ACS Nano. 2022;16:251–260. doi: 10.1021/acsnano.1c06096. [DOI] [PubMed] [Google Scholar]
- 44.Wang L., Wei X., He X., Xiao S., Shi Q., Chen P., et al. Osteoinductive dental pulp stem cell-derived extracellular vesicle-loaded multifunctional hydrogel for bone regeneration. ACS Nano. 2024;18:8777–8797. doi: 10.1021/acsnano.3c11542. [DOI] [PubMed] [Google Scholar]
- 45.Hsu Y.L., Hung J.Y., Chang W.A., Lin Y.S., Pan Y.C., Tsai P.H., et al. Hypoxic lung cancer-secreted exosomal miR-23a increased angiogenesis and vascular permeability by targeting prolyl hydroxylase and tight junction protein ZO-1. Oncogene. 2017;36:4929–4942. doi: 10.1038/onc.2017.105. [DOI] [PubMed] [Google Scholar]
- 46.Fan T.W.M., Zhang X., Wang C., Yang Y., Kang W.-Y., Arnold S., et al. Exosomal lipids for classifying early and late stage non-small cell lung cancer. Anal Chim Acta. 2018;1037:256–264. doi: 10.1016/j.aca.2018.02.051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Brzozowski J.S., Jankowski H., Bond D.R., McCague S.B., Munro B.R., Predebon M.J., et al. Lipidomic profiling of extracellular vesicles derived from prostate and prostate cancer cell lines. Lipids Health Dis. 2018;17:211. doi: 10.1186/s12944-018-0854-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Shao H., Im H., Castro C.M., Breakefield X., Weissleder R., Lee H. New technologies for analysis of extracellular vesicles. Chem Rev. 2018;118:1917–1950. doi: 10.1021/acs.chemrev.7b00534. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Buzas E.I., György B., Nagy G., Falus A., Gay S. Emerging role of extracellular vesicles in inflammatory diseases. Nat Rev Rheumatol. 2014;10:356–364. doi: 10.1038/nrrheum.2014.19. [DOI] [PubMed] [Google Scholar]
- 50.Yu Z., Yang Y., Fang W., Hu P., Liu Y., Shi J. Dual tumor exosome biomarker co-recognitions based nanoliquid biopsy for the accurate early diagnosis of pancreatic cancer. ACS Nano. 2023;17:11384–11395. doi: 10.1021/acsnano.3c00674. [DOI] [PubMed] [Google Scholar]
- 51.de Abreu R.C., Fernandes H., da Costa Martins P.A., Sahoo S., Emanueli C., Ferreira L. Native and bioengineered extracellular vesicles for cardiovascular therapeutics. Nat Rev Cardiol. 2020;17:685–697. doi: 10.1038/s41569-020-0389-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Qian F., Huang Z., Zhong H., Lei Q., Ai Y., Xie Z., et al. Analysis and biomedical applications of functional cargo in extracellular vesicles. ACS Nano. 2022;16:19980–20001. doi: 10.1021/acsnano.2c11298. [DOI] [PubMed] [Google Scholar]
- 53.Hao S.J., Wan Y., Xia Y.Q., Zou X., Zheng S.Y. Size-based separation methods of circulating tumor cells. Adv Drug Deliv Rev. 2018;125:3–20. doi: 10.1016/j.addr.2018.01.002. [DOI] [PubMed] [Google Scholar]
- 54.Yang F., Liao X., Tian Y., Li G. Exosome separation using microfluidic systems: size-based, immunoaffinity-based and dynamic methodologies. Biotechnol J. 2017;12:1600699. doi: 10.1002/biot.201600699. [DOI] [PubMed] [Google Scholar]
- 55.Shi N., Mohibullah M., Easley C.J. Active flow control and dynamic analysis in droplet microfluidics. Annu Rev Anal Chem. 2021;14:133–153. doi: 10.1146/annurev-anchem-122120-042627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Bobrie A., Colombo M., Krumeich S., Raposo G., Théry C. Diverse subpopulations of vesicles secreted by different intracellular mechanisms are present in exosome preparations obtained by differential ultracentrifugation. J Extracell Vesicles. 2012;1:18397. doi: 10.3402/jev.v1i0.18397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Lin S., Yu Z., Chen D., Wang Z., Miao J., Li Q., et al. Progress in microfluidics-based exosome separation and detection technologies for diagnostic applications. Small. 2020;16 doi: 10.1002/smll.201903916. [DOI] [PubMed] [Google Scholar]
- 58.Ding L., Yang X., Gao Z., Effah C.Y., Zhang X., Wu Y., et al. A holistic review of the state-of-the-art microfluidics for exosome separation: an overview of the current status, existing obstacles, and future outlook. Small. 2021;17 doi: 10.1002/smll.202007174. [DOI] [PubMed] [Google Scholar]
- 59.Chen M., Lin S., Zhou C., Cui D., Haick H., Tang N. From conventional to microfluidic: progress in extracellular vesicle separation and individual characterization. Adv Healthcare Mater. 2023;12 doi: 10.1002/adhm.202202437. [DOI] [PubMed] [Google Scholar]
- 60.Chen C., Skog J., Hsu C.H., Lessard R.T., Balaj L., Wurdinger T., et al. Microfluidic isolation and transcriptome analysis of serum microvesicles. Lab Chip. 2010;10:505–511. doi: 10.1039/b916199f. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Casadei L., Choudhury A., Sarchet P., Mohana Sundaram P., Lopez G., Braggio D., et al. Cross-flow microfiltration for isolation, selective capture and release of liposarcoma extracellular vesicles. J Extracell Vesicles. 2021;10 doi: 10.1002/jev2.12062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Soong W.J., Wang C.H., Chen C., Lee G.B. Nanoscale sorting of extracellular vesicles via optically-induced dielectrophoresis on an integrated microfluidic system. Lab Chip. 2024;24:1965–1976. doi: 10.1039/d3lc01007d. [DOI] [PubMed] [Google Scholar]
- 63.Zhang G., Wang Y., Zhou W., Lei Y., Lu J., Yin W., et al. A magnetically driven tandem chip enables rapid isolation and multiplexed profiling of extracellular vesicles. Angew Chem Int Ed Engl. 2023 doi: 10.1002/anie.202315113. [DOI] [PubMed] [Google Scholar]
- 64.Im H., Shao H., Park Y.I., Peterson V.M., Castro C.M., Weissleder R., et al. Label-free detection and molecular profiling of exosomes with a nano-plasmonic sensor. Nat Biotechnol. 2014;32:490–495. doi: 10.1038/nbt.2886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Bai J.J., Zhang X., Wei X., Wang Y., Du C., Wang Z.J., et al. Dean-flow-coupled elasto-inertial focusing accelerates exosome purification to facilitate single vesicle profiling. Anal Chem. 2023;95:2523–2531. doi: 10.1021/acs.analchem.2c04898. [DOI] [PubMed] [Google Scholar]
- 66.Pan W.L., Feng J.J., Luo T.T., Tan Y., Situ B., Nieuwland R., et al. Rapid and efficient isolation platform for plasma extracellular vesicles: EV-Fisher. J Extracell Vesicles. 2022;11 doi: 10.1002/jev2.12281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Zong Z., Liu X., Ye Z., Liu D. A double-switch pHLIP system enables selective enrichment of circulating tumor microenvironment-derived extracellular vesicles. Proc Natl Acad Sci U S A. 2023;120 doi: 10.1073/pnas.2214912120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Niu Q., Shu Y., Chen Y., Huang Z., Yao Z., Chen X., et al. A fluid multivalent magnetic interface for high-performance isolation and proteomic profiling of tumor-derived extracellular vesicles. Angew Chem Int Ed Engl. 2023;62 doi: 10.1002/anie.202215337. [DOI] [PubMed] [Google Scholar]
- 69.Preethi K.A., Selvakumar S.C., Ross K., Jayaraman S., Tusubira D., Sekar D. Liquid biopsy: exosomal microRNAs as novel diagnostic and prognostic biomarkers in cancer. Mol Cancer. 2022;21:54. doi: 10.1186/s12943-022-01525-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Sil S., Dagur R.S., Liao K., Peeples E.S., Hu G., Periyasamy P., et al. Strategies for the use of extracellular vesicles for the delivery of therapeutics. J Neuroimmune Pharmacol. 2020;15:422–442. doi: 10.1007/s11481-019-09873-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Keller S., Ridinger J., Rupp A.K., Janssen J.W., Altevogt P. Body fluid derived exosomes as a novel template for clinical diagnostics. J Transl Med. 2011;9:86. doi: 10.1186/1479-5876-9-86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Wei Z., Batagov A.O., Schinelli S., Wang J., Wang Y., El Fatimy R., et al. Coding and noncoding landscape of extracellular RNA released by human glioma stem cells. Nat Commun. 2017;8:1145. doi: 10.1038/s41467-017-01196-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Tao Y., Tang Y., Yang Z., Wu F., Wang L., Yang L., et al. Exploration of serum exosomal LncRNA TBILA and AGAP2-AS1 as promising biomarkers for diagnosis of non-small cell lung cancer. Int J Biol Sci. 2020;16:471–482. doi: 10.7150/ijbs.39123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Chen G., Huang A.C., Zhang W., Zhang G., Wu M., Xu W., et al. Exosomal PD-L1 contributes to immunosuppression and is associated with anti-PD-1 response. Nature. 2018;560:382–386. doi: 10.1038/s41586-018-0392-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Bijnsdorp I.V., Geldof A.A., Lavaei M., Piersma S.R., van Moorselaar R.J., Jimenez C.R. Exosomal ITGA3 interferes with non-cancerous prostate cell functions and is increased in urine exosomes of metastatic prostate cancer patients. J Extracell Vesicles. 2013;2:22097. doi: 10.3402/jev.v2i0.22097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Cordonnier M., Nardin C., Chanteloup G., Derangere V., Algros M.P., Arnould L., et al. Tracking the evolution of circulating exosomal-PD-L1 to monitor melanoma patients. J Extracell Vesicles. 2020;9 doi: 10.1080/20013078.2019.1710899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.van der Mijn J.C., Sol N., Mellema W., Jimenez C.R., Piersma S.R., Dekker H., et al. Analysis of AKT and ERK1/2 protein kinases in extracellular vesicles isolated from blood of patients with cancer. J Extracell Vesicles. 2014;3 doi: 10.3402/jev.v3.25657. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Shuen T.W.H., Alunni-Fabbroni M., Öcal E., Malfertheiner P., Wildgruber M., Schinner R., et al. Extracellular vesicles may predict response to radioembolization and sorafenib treatment in advanced hepatocellular carcinoma: an exploratory analysis from the SORAMIC trial. Clin Cancer Res. 2022;28:3890–3901. doi: 10.1158/1078-0432.CCR-22-0569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.You Y., Muraoka S., Jedrychowski M.P., Hu J., McQuade A.K., Young-Pearse T., et al. Human neural cell type-specific extracellular vesicle proteome defines disease-related molecules associated with activated astrocytes in Alzheimer’s disease brain. J Extracell Vesicles. 2022;11 doi: 10.1002/jev2.12183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Pietrowska M., Zebrowska A., Gawin M., Marczak L., Sharma P., Mondal S., et al. Proteomic profile of melanoma cell-derived small extracellular vesicles in patients’ plasma: a potential correlate of melanoma progression. J Extracell Vesicles. 2021;10 doi: 10.1002/jev2.12063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Zhang Z., Liu X., Yang X., Jiang Y., Li A., Cong J., et al. Identification of faecal extracellular vesicles as novel biomarkers for the non-invasive diagnosis and prognosis of colorectal cancer. J Extracell Vesicles. 2023;12 doi: 10.1002/jev2.12300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Daassi D., Mahoney K.M., Freeman G.J. The importance of exosomal PDL1 in tumour immune evasion. Nat Rev Immunol. 2020;20:209–215. doi: 10.1038/s41577-019-0264-y. [DOI] [PubMed] [Google Scholar]
- 83.von Felden J., Garcia-Lezana T., Dogra N., Gonzalez-Kozlova E., Ahsen M.E., Craig A., et al. Unannotated small RNA clusters associated with circulating extracellular vesicles detect early stage liver cancer. Gut. 2021;71:1935–1936. doi: 10.1136/gutjnl-2021-325036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Zhou L., Wang W., Wang F., Yang S., Hu J., Lu B., et al. Plasma-derived exosomal miR-15a-5p as a promising diagnostic biomarker for early detection of endometrial carcinoma. Mol Cancer. 2021;20:57. doi: 10.1186/s12943-021-01352-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Lucero R., Zappulli V., Sammarco A., Murillo O.D., Cheah P.S., Srinivasan S., et al. Glioma-derived miRNA-containing extracellular vesicles induce angiogenesis by reprogramming brain endothelial cells. Cell Rep. 2020;30 doi: 10.1016/j.celrep.2020.01.073. 2065–74.e4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Brinkman K., Meyer L., Bickel A., Enderle D., Berking C., Skog J., et al. Extracellular vesicles from plasma have higher tumour RNA fraction than platelets. J Extracell Vesicles. 2020;9 doi: 10.1080/20013078.2020.1741176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Hannafon B.N., Trigoso Y.D., Calloway C.L., Zhao Y.D., Lum D.H., Welm A.L., et al. Plasma exosome microRNAs are indicative of breast cancer. Breast Cancer Res. 2016;18:90. doi: 10.1186/s13058-016-0753-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Nguyen L.T.H., Zhang J., Rima X.Y., Wang X., Kwak K.J., Okimoto T., et al. An immunogold single extracellular vesicular RNA and protein (Au SERP) biochip to predict responses to immunotherapy in non-small cell lung cancer patients. J Extracell Vesicles. 2022;11 doi: 10.1002/jev2.12258. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Zhu L., Li J., Gong Y., Wu Q., Tan S., Sun D., et al. Exosomal tRNA-derived small RNA as a promising biomarker for cancer diagnosis. Mol Cancer. 2019;18:74. doi: 10.1186/s12943-019-1000-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 90.Ning T., Li J., He Y., Zhang H., Wang X., Deng T., et al. Exosomal miR-208b related with oxaliplatin resistance promotes Treg expansion in colorectal cancer. Mol Ther. 2021;29:2723–2736. doi: 10.1016/j.ymthe.2021.04.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Guo T., Tang X.H., Gao X.Y., Zhou Y., Jin B., Deng Z.Q., et al. A liquid biopsy signature of circulating exosome-derived mRNAs, miRNAs and lncRNAs predict therapeutic efficacy to neoadjuvant chemotherapy in patients with advanced gastric cancer. Mol Cancer. 2022;21:216. doi: 10.1186/s12943-022-01684-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Wang G., Liu W., Zou Y., Wang G., Deng Y., Luo J., et al. Three isoforms of exosomal circPTGR1 promote hepatocellular carcinoma metastasis via the miR449a-MET pathway. EBioMedicine. 2019;40:432–445. doi: 10.1016/j.ebiom.2018.12.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Xie M., Yu T., Jing X., Ma L., Fan Y., Yang F., et al. Exosomal circSHKBP1 promotes gastric cancer progression via regulating the miR-582-3p/HUR/VEGF axis and suppressing HSP90 degradation. Mol Cancer. 2020;19:112. doi: 10.1186/s12943-020-01208-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Zhao R., Zhang Y., Zhang X., Yang Y., Zheng X., Li X., et al. Exosomal long noncoding RNA HOTTIP as potential novel diagnostic and prognostic biomarker test for gastric cancer. Mol Cancer. 2018;17:68. doi: 10.1186/s12943-018-0817-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Li H., Chiang C.L., Kwak K.J., Wang X., Doddi S., Ramanathan L.V., et al. Extracellular vesicular analysis of glypican 1 mRNA and protein for pancreatic cancer diagnosis and prognosis. Adv Sci (Weinh) 2024 doi: 10.1002/advs.202306373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Marcuello M., Vymetalkova V., Neves R.P.L., Duran-Sanchon S., Vedeld H.M., Tham E., et al. Circulating biomarkers for early detection and clinical management of colorectal cancer. Mol Aspect Med. 2019;69:107–122. doi: 10.1016/j.mam.2019.06.002. [DOI] [PubMed] [Google Scholar]
- 97.Maire C.L., Fuh M.M., Kaulich K., Fita K.D., Stevic I., Heiland D.H., et al. Genome-wide methylation profiling of glioblastoma cell-derived extracellular vesicle DNA allows tumor classification. Neuro Oncol. 2021;23:1087–1099. doi: 10.1093/neuonc/noab012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Wan Y., Liu B., Lei H., Zhang B., Wang Y., Huang H., et al. Nanoscale extracellular vesicle-derived DNA is superior to circulating cell-free DNA for mutation detection in early-stage non-small-cell lung cancer. Ann Oncol. 2018;29:2379–2383. doi: 10.1093/annonc/mdy458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99.Cheng A.N., Cheng L.C., Kuo C.L., Lo Y.K., Chou H.Y., Chen C.H., et al. Mitochondrial Lon-induced mtDNA leakage contributes to PD-L1-mediated immunoescape via STING-IFN signaling and extracellular vesicles. J Immunother Cancer. 2020;8:e001372. doi: 10.1136/jitc-2020-001372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100.Campos-Carrillo A., Weitzel J.N., Sahoo P., Rockne R., Mokhnatkin J.V., Murtaza M., et al. Circulating tumor DNA as an early cancer detection tool. Pharmacol Ther. 2020;207 doi: 10.1016/j.pharmthera.2019.107458. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 101.Skotland T., Ekroos K., Kauhanen D., Simolin H., Seierstad T., Berge V., et al. Molecular lipid species in urinary exosomes as potential prostate cancer biomarkers. Eur J Cancer. 2017;70:122–132. doi: 10.1016/j.ejca.2016.10.011. [DOI] [PubMed] [Google Scholar]
- 102.Sharma R., Huang X., Brekken R.A., Schroit A.J. Detection of phosphatidylserine-positive exosomes for the diagnosis of early-stage malignancies. Br J Cancer. 2017;117:545–552. doi: 10.1038/bjc.2017.183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Clos-Garcia M., Loizaga-Iriarte A., Zuñiga-Garcia P., Sánchez-Mosquera P., Rosa Cortazar A., González E., et al. Metabolic alterations in urine extracellular vesicles are associated to prostate cancer pathogenesis and progression. J Extracell Vesicles. 2018;7 doi: 10.1080/20013078.2018.1470442. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Chen Y., Wu T., Zhu Z., Huang H., Zhang L., Goel A., et al. An integrated workflow for biomarker development using microRNAs in extracellular vesicles for cancer precision medicine. Semin Cancer Biol. 2021;74:134–155. doi: 10.1016/j.semcancer.2021.03.011. [DOI] [PubMed] [Google Scholar]
- 105.Hoshino A., Kim H.S., Bojmar L., Gyan K.E., Cioffi M., Hernandez J., et al. Extracellular vesicle and particle biomarkers define multiple human cancers. Cell. 2020;182 doi: 10.1016/j.cell.2020.07.009. 1044–61.e18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Nikanjam M., Kato S., Kurzrock R. Liquid biopsy: current technology and clinical applications. J Hematol Oncol. 2022;15:131. doi: 10.1186/s13045-022-01351-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Nieuwland R., Siljander P.R. A beginner’s guide to study extracellular vesicles in human blood plasma and serum. J Extracell Vesicles. 2024;13 doi: 10.1002/jev2.12400. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Palviainen M., Saraswat M., Varga Z., Kitka D., Neuvonen M., Puhka M., et al. Extracellular vesicles from human plasma and serum are carriers of extravesicular cargo-Implications for biomarker discovery. PLoS One. 2020;15 doi: 10.1371/journal.pone.0236439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Zhang X., Takeuchi T., Takeda A., Mochizuki H., Nagai Y. Comparison of serum and plasma as a source of blood extracellular vesicles: increased levels of platelet-derived particles in serum extracellular vesicle fractions alter content profiles from plasma extracellular vesicle fractions. PLoS One. 2022;17 doi: 10.1371/journal.pone.0270634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Karimi N., Dalirfardouei R., Dias T., Lötvall J., Lässer C. Tetraspanins distinguish separate extracellular vesicle subpopulations in human serum and plasma—contributions of platelet extracellular vesicles in plasma samples. J Extracell Vesicles. 2022;11 doi: 10.1002/jev2.12213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Li M., Fang F., Sun M., Zhang Y., Hu M., Zhang J. Extracellular vesicles as bioactive nanotherapeutics: an emerging paradigm for regenerative medicine. Theranostics. 2022;12:4879–4903. doi: 10.7150/thno.72812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Wiklander O.P., Nordin J.Z., O’Loughlin A., Gustafsson Y., Corso G., Mäger I., et al. Extracellular vesicle in vivo biodistribution is determined by cell source, route of administration and targeting. J Extracell Vesicles. 2015;4 doi: 10.3402/jev.v4.26316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Qiao L., Hu S., Huang K., Su T., Li Z., Vandergriff A., et al. Tumor cell-derived exosomes home to their cells of origin and can be used as Trojan horses to deliver cancer drugs. Theranostics. 2020;10:3474–3487. doi: 10.7150/thno.39434. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Headley M.B., Bins A., Nip A., Roberts E.W., Looney M.R., Gerard A., et al. Visualization of immediate immune responses to pioneer metastatic cells in the lung. Nature. 2016;531:513–517. doi: 10.1038/nature16985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Nakazawa Y., Nishiyama N., Koizumi H., Kanemaru K., Nakahashi-Oda C., Shibuya A. Tumor-derived extracellular vesicles regulate tumor-infiltrating regulatory T cells via the inhibitory immunoreceptor CD300a. Elife. 2021;10:e61999. doi: 10.7554/eLife.61999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Wang S., Sun J., Dastgheyb R.M., Li Z. Tumor-derived extracellular vesicles modulate innate immune responses to affect tumor progression. Front Immunol. 2022;13 doi: 10.3389/fimmu.2022.1045624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Ricklefs F.L., Alayo Q., Krenzlin H., Mahmoud A.B., Speranza M.C., Nakashima H., et al. Immune evasion mediated by PD-L1 on glioblastoma-derived extracellular vesicles. Sci Adv. 2018;4 doi: 10.1126/sciadv.aar2766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Li Q., Cai S., Li M., Salma K.I., Zhou X., Han F., et al. Tumor-derived extracellular vesicles: their role in immune cells and immunotherapy. Int J Nanomed. 2021;16:5395–5409. doi: 10.2147/IJN.S313912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.Tian X., Shen H., Li Z., Wang T., Wang S. Tumor-derived exosomes, myeloid-derived suppressor cells, and tumor microenvironment. J Hematol Oncol. 2019;12:84. doi: 10.1186/s13045-019-0772-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Gabrilovich D.I., Nagaraj S. Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol. 2009;9:162–174. doi: 10.1038/nri2506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Rodríguez-Fuentes D.E., Fernández-Garza L.E., Samia-Meza J.A., Barrera-Barrera S.A., Caplan A.I., Barrera-Saldaña H.A. Mesenchymal stem cells current clinical applications: a systematic review. Arch Med Res. 2021;52:93–101. doi: 10.1016/j.arcmed.2020.08.006. [DOI] [PubMed] [Google Scholar]
- 122.Wu R., Fan X., Wang Y., Shen M., Zheng Y., Zhao S., et al. Mesenchymal stem cell-derived extracellular vesicles in liver immunity and therapy. Front Immunol. 2022;13 doi: 10.3389/fimmu.2022.833878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 123.Kou M., Huang L., Yang J., Chiang Z., Chen S., Liu J., et al. Mesenchymal stem cell-derived extracellular vesicles for immunomodulation and regeneration: a next generation therapeutic tool? Cell Death Dis. 2022;13:580. doi: 10.1038/s41419-022-05034-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Rehman F.U., Liu Y., Yang Q., Yang H., Liu R., Zhang D., et al. Heme Oxygenase-1 targeting exosomes for temozolomide resistant glioblastoma synergistic therapy. J Control Release. 2022;345:696–708. doi: 10.1016/j.jconrel.2022.03.036. [DOI] [PubMed] [Google Scholar]
- 125.Katsuda T., Tsuchiya R., Kosaka N., Yoshioka Y., Takagaki K., Oki K., et al. Human adipose tissue-derived mesenchymal stem cells secrete functional neprilysin-bound exosomes. Sci Rep. 2013;3:1197. doi: 10.1038/srep01197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Witwer K.W., Van Balkom B.W.M., Bruno S., Choo A., Dominici M., Gimona M., et al. Defining mesenchymal stromal cell (MSC)-derived small extracellular vesicles for therapeutic applications. J Extracell Vesicles. 2019;8 doi: 10.1080/20013078.2019.1609206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Gu H., Ji R., Zhang X., Wang M., Zhu W., Qian H., et al. Exosomes derived from human mesenchymal stem cells promote gastric cancer cell growth and migration via the activation of the Akt pathway. Mol Med Rep. 2016;14:3452–3458. doi: 10.3892/mmr.2016.5625. [DOI] [PubMed] [Google Scholar]
- 128.Lu Z., Zuo B., Jing R., Gao X., Rao Q., Liu Z., et al. Dendritic cell-derived exosomes elicit tumor regression in autochthonous hepatocellular carcinoma mouse models. J Hepatol. 2017;67:739–748. doi: 10.1016/j.jhep.2017.05.019. [DOI] [PubMed] [Google Scholar]
- 129.Pitt J.M., André F., Amigorena S., Soria J.C., Eggermont A., Kroemer G., et al. Dendritic cell-derived exosomes for cancer therapy. J Clin Investig. 2016;126:1224–1232. doi: 10.1172/JCI81137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Ghorbaninezhad F., Alemohammad H., Najafzadeh B., Masoumi J., Shadbad M.A., Shahpouri M., et al. Dendritic cell-derived exosomes: a new horizon in personalized cancer immunotherapy? Cancer Lett. 2023;562 doi: 10.1016/j.canlet.2023.216168. [DOI] [PubMed] [Google Scholar]
- 131.Liu Q., Rojas-Canales D.M., Divito S.J., Shufesky W.J., Stolz D.B., Erdos G., et al. Donor dendritic cell-derived exosomes promote allograft-targeting immune response. J Clin Investig. 2016;126:2805–2820. doi: 10.1172/JCI84577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Näslund T.I., Gehrmann U., Qazi K.R., Karlsson M.C., Gabrielsson S. Dendritic cell-derived exosomes need to activate both T and B cells to induce antitumor immunity. J Immunol. 2013;190:2712–2719. doi: 10.4049/jimmunol.1203082. [DOI] [PubMed] [Google Scholar]
- 133.Kim S.H., Bianco N., Menon R., Lechman E.R., Shufesky W.J., Morelli A.E., et al. Exosomes derived from genetically modified DC expressing FasL are anti-inflammatory and immunosuppressive. Mol Ther. 2006;13:289–300. doi: 10.1016/j.ymthe.2005.09.015. [DOI] [PubMed] [Google Scholar]
- 134.Munich S., Sobo-Vujanovic A., Buchser W.J., Beer-Stolz D., Vujanovic N.L. Dendritic cell exosomes directly kill tumor cells and activate natural killer cells via TNF superfamily ligands. OncoImmunology. 2012;1:1074–1083. doi: 10.4161/onci.20897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 135.Li J., Li J., Peng Y., Du Y., Yang Z., Qi X. Dendritic cell derived exosomes loaded neoantigens for personalized cancer immunotherapies. J Control Release. 2023;353:423–433. doi: 10.1016/j.jconrel.2022.11.053. [DOI] [PubMed] [Google Scholar]
- 136.Wynn T.A., Vannella K.M. Macrophages in tissue repair, regeneration, and fibrosis. Immunity. 2016;44:450–462. doi: 10.1016/j.immuni.2016.02.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Barone A., d’Avanzo N., Cristiano M.C., Paolino D., Fresta M. Macrophage-derived extracellular vesicles: a promising tool for personalized cancer therapy. Biomedicines. 2022;10:1252. doi: 10.3390/biomedicines10061252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Gangadaran P., Rajendran R.L., Oh J.M., Hong C.M., Jeong S.Y., Lee S.W., et al. Extracellular vesicles derived from macrophage promote angiogenesis in vitro and accelerate new vasculature formation in vivo. Exp Cell Res. 2020;394 doi: 10.1016/j.yexcr.2020.112146. [DOI] [PubMed] [Google Scholar]
- 139.Zhang C., Li D., Hu H., Wang Z., An J., Gao Z., et al. Engineered extracellular vesicles derived from primary M2 macrophages with anti-inflammatory and neuroprotective properties for the treatment of spinal cord injury. J Nanobiotechnol. 2021;19:373. doi: 10.1186/s12951-021-01123-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Li L., Wang Z., Wang K., Fu S., Li D., Wang M., et al. Paintable bioactive extracellular vesicle ink for wound healing. ACS Appl Mater Interfaces. 2023;15:25427–25436. doi: 10.1021/acsami.3c03630. [DOI] [PubMed] [Google Scholar]
- 141.Fan L., Xu G., Zeng X. M2 macrophage-derived extracellular vesicles augment immune evasion and development of colorectal cancer via a circRNA_CCDC66/microRNA-342-3p/metadherin axis. Cytotechnology. 2023;75:293–308. doi: 10.1007/s10616-023-00577-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142.Wang X., Ye X., Chen Y., Lin J. Mechanism of M2 type macrophage-derived extracellular vesicles regulating PD-L1 expression via the MISP/IQGAP1 axis in hepatocellular carcinoma immunotherapy resistance. Int Immunopharmacol. 2023;124 doi: 10.1016/j.intimp.2023.110848. [DOI] [PubMed] [Google Scholar]
- 143.Ma X., Yao M., Gao Y., Yue Y., Li Y., Zhang T., et al. Functional immune cell-derived exosomes engineered for the trilogy of radiotherapy sensitization. Adv Sci (Weinh) 2022;9 doi: 10.1002/advs.202106031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Li A., Li D., Gu Y., Liu R., Tang X., Zhao Y., et al. Plant-derived nanovesicles: further exploration of biomedical function and application potential. Acta Pharm Sin B. 2023;13:3300–3320. doi: 10.1016/j.apsb.2022.12.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Cai Q., He B., Wang S., Fletcher S., Niu D., Mitter N., et al. Message in a bubble: shuttling small RNAs and proteins between cells and interacting organisms using extracellular vesicles. Annu Rev Plant Biol. 2021;72:497–524. doi: 10.1146/annurev-arplant-081720-010616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Bruns C., McCaffery J.M., Curwin A.J., Duran J.M., Malhotra V. Biogenesis of a novel compartment for autophagosome-mediated unconventional protein secretion. J Cell Biol. 2011;195:979–992. doi: 10.1083/jcb.201106098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Lian M.Q., Chng W.H., Liang J., Yeo H.Q., Lee C.K., Belaid M., et al. Plant-derived extracellular vesicles: recent advancements and current challenges on their use for biomedical applications. J Extracell Vesicles. 2022;11 doi: 10.1002/jev2.12283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Ly N.P., Han H.S., Kim M., Park J.H., Choi K.Y. Plant-derived nanovesicles: current understanding and applications for cancer therapy. Bioact Mater. 2023;22:365–383. doi: 10.1016/j.bioactmat.2022.10.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Zhuang X., Deng Z.B., Mu J., Zhang L., Yan J., Miller D., et al. Ginger-derived nanoparticles protect against alcohol-induced liver damage. J Extracell Vesicles. 2015;4 doi: 10.3402/jev.v4.28713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Ou X., Wang H., Tie H., Liao J., Luo Y., Huang W., et al. Novel plant-derived exosome-like nanovesicles from Catharanthus roseus: preparation, characterization, and immunostimulatory effect via TNF-α/NF-κB/PU.1 axis. J Nanobiotechnol. 2023;21:160. doi: 10.1186/s12951-023-01919-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 151.Chen Q., Li Q., Liang Y., Zu M., Chen N., Canup B.S.B., et al. Natural exosome-like nanovesicles from edible tea flowers suppress metastatic breast cancer via ROS generation and microbiota modulation. Acta Pharm Sin B. 2022;12:907–923. doi: 10.1016/j.apsb.2021.08.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152.Wang B., Zhuang X., Deng Z.B., Jiang H., Mu J., Wang Q., et al. Targeted drug delivery to intestinal macrophages by bioactive nanovesicles released from grapefruit. Mol Ther. 2014;22:522–534. doi: 10.1038/mt.2013.190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Dorado E., Doria M.L., Nagelkerke A., McKenzie J.S., Maneta-Stavrakaki S., Whittaker T.E., et al. Extracellular vesicles as a promising source of lipid biomarkers for breast cancer detection in blood plasma. J Extracell Vesicles. 2024;13 doi: 10.1002/jev2.12419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154.Krokidis M.G., Pucha K.A., Mustapic M., Exarchos T.P., Vlamos P., Kapogiannis D. Lipidomic analysis of plasma extracellular vesicles derived from Alzheimer’s disease patients. Cells. 2024;13:702. doi: 10.3390/cells13080702. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Nishida-Aoki N., Izumi Y., Takeda H., Takahashi M., Ochiya T., Bamba T. Lipidomic analysis of cells and extracellular vesicles from high- and low-metastatic triple-negative breast cancer. Metabolites. 2020;10:67. doi: 10.3390/metabo10020067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156.Cao M., Yan H., Han X., Weng L., Wei Q., Sun X., et al. Ginseng-derived nanoparticles alter macrophage polarization to inhibit melanoma growth. J Immunother Cancer. 2019;7:326. doi: 10.1186/s40425-019-0817-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157.Stanly C., Alfieri M., Ambrosone A., Leone A., Fiume I., Pocsfalvi G. Grapefruit-derived micro and nanovesicles show distinct metabolome profiles and anticancer activities in the A375 human melanoma cell line. Cells. 2020;9:2722. doi: 10.3390/cells9122722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Yang M., Liu X., Luo Q., Xu L., Chen F. An efficient method to isolate lemon derived extracellular vesicles for gastric cancer therapy. J Nanobiotechnol. 2020;18:100. doi: 10.1186/s12951-020-00656-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 159.Raimondo S., Naselli F., Fontana S., Monteleone F., Lo Dico A., Saieva L., et al. Citrus limon-derived nanovesicles inhibit cancer cell proliferation and suppress CML xenograft growth by inducing TRAIL-mediated cell death. Oncotarget. 2015;6:19514–19527. doi: 10.18632/oncotarget.4004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 160.Xiao Q., Zhao W., Wu C., Wang X., Chen J., Shi X., et al. Lemon-derived extracellular vesicles nanodrugs enable to efficiently overcome cancer multidrug resistance by endocytosis-triggered energy dissipation and energy production reduction. Adv Sci (Weinh) 2022;9 doi: 10.1002/advs.202105274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Zhang L., He F., Gao L., Cong M., Sun J., Xu J., et al. Engineering exosome-like nanovesicles derived from asparagus cochinchinensis can inhibit the proliferation of hepatocellular carcinoma cells with better safety profile. Int J Nanomed. 2021;16:1575–1586. doi: 10.2147/IJN.S293067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 162.Kim K., Yoo H.J., Jung J.H., Lee R., Hyun J.K., Park J.H., et al. Cytotoxic effects of plant sap-derived extracellular vesicles on various tumor cell types. J Funct Biomater. 2020;11:22. doi: 10.3390/jfb11020022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Zhang M., Viennois E., Prasad M., Zhang Y., Wang L., Zhang Z., et al. Edible ginger-derived nanoparticles: a novel therapeutic approach for the prevention and treatment of inflammatory bowel disease and colitis-associated cancer. Biomaterials. 2016;101:321–340. doi: 10.1016/j.biomaterials.2016.06.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Kimiz-Gebologlu I., Oncel S.S. Exosomes: large-scale production, isolation, drug loading efficiency, and biodistribution and uptake. J Control Release. 2022;347:533–543. doi: 10.1016/j.jconrel.2022.05.027. [DOI] [PubMed] [Google Scholar]
- 165.Rufino-Ramos D., Albuquerque P.R., Carmona V., Perfeito R., Nobre R.J., Pereira de Almeida L. Extracellular vesicles: novel promising delivery systems for therapy of brain diseases. J Control Release. 2017;262:247–258. doi: 10.1016/j.jconrel.2017.07.001. [DOI] [PubMed] [Google Scholar]
- 166.Pascucci L., Coccè V., Bonomi A., Ami D., Ceccarelli P., Ciusani E., et al. Paclitaxel is incorporated by mesenchymal stromal cells and released in exosomes that inhibit in vitro tumor growth: a new approach for drug delivery. J Control Release. 2014;192:262–270. doi: 10.1016/j.jconrel.2014.07.042. [DOI] [PubMed] [Google Scholar]
- 167.Mathiyalagan P., Sahoo S. Exosomes-based gene therapy for microRNA delivery. Methods Mol Biol. 2017;1521:139–152. doi: 10.1007/978-1-4939-6588-5_9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Haney M.J., Klyachko N.L., Zhao Y., Gupta R., Plotnikova E.G., He Z., et al. Exosomes as drug delivery vehicles for Parkinson’s disease therapy. J Control Release. 2015;207:18–30. doi: 10.1016/j.jconrel.2015.03.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Zhuang X., Xiang X., Grizzle W., Sun D., Zhang S., Axtell R.C., et al. Treatment of brain inflammatory diseases by delivering exosome encapsulated anti-inflammatory drugs from the nasal region to the brain. Mol Ther. 2011;19:1769–1779. doi: 10.1038/mt.2011.164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170.Zhang Y., Liu D., Chen X., Li J., Li L., Bian Z., et al. Secreted monocytic miR-150 enhances targeted endothelial cell migration. Mol Cell. 2010;39:133–144. doi: 10.1016/j.molcel.2010.06.010. [DOI] [PubMed] [Google Scholar]
- 171.Qiu Y., Sun J., Qiu J., Chen G., Wang X., Mu Y., et al. Antitumor activity of cabazitaxel and MSC-TRAIL derived extracellular vesicles in drug-resistant oral squamous cell carcinoma. Cancer Manag Res. 2020;12:10809–10820. doi: 10.2147/CMAR.S277324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Mizrak A., Bolukbasi M.F., Ozdener G.B., Brenner G.J., Madlener S., Erkan E.P., et al. Genetically engineered microvesicles carrying suicide mRNA/protein inhibit schwannoma tumor growth. Mol Ther. 2013;21:101–108. doi: 10.1038/mt.2012.161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Koppers-Lalic D., Hackenberg M., Bijnsdorp Irene V., van Eijndhoven Monique A.J., Sadek P., Sie D., et al. Nontemplated nucleotide additions distinguish the small RNA composition in cells from exosomes. Cell Rep. 2014;8:1649–1658. doi: 10.1016/j.celrep.2014.08.027. [DOI] [PubMed] [Google Scholar]
- 174.Alvarez-Erviti L., Seow Y., Yin H., Betts C., Lakhal S., Wood M.J. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat Biotechnol. 2011;29:341–345. doi: 10.1038/nbt.1807. [DOI] [PubMed] [Google Scholar]
- 175.Kim M.S., Haney M.J., Zhao Y., Mahajan V., Deygen I., Klyachko N.L., et al. Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomedicine. 2016;12:655–664. doi: 10.1016/j.nano.2015.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 176.Kim M.S., Haney M.J., Zhao Y., Mahajan V., Deygen I., Klyachko N.L., et al. Development of exosome-encapsulated paclitaxel to overcome MDR in cancer cells. Nanomed Nanotechnol Biol Med. 2016;12:655–664. doi: 10.1016/j.nano.2015.10.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Kim O.Y., Dinh N.T., Park H.T., Choi S.J., Hong K., Gho Y.S. Bacterial protoplast-derived nanovesicles for tumor targeted delivery of chemotherapeutics. Biomaterials. 2017;113:68–79. doi: 10.1016/j.biomaterials.2016.10.037. [DOI] [PubMed] [Google Scholar]
- 178.Wang Q., Ren Y., Mu J., Egilmez N.K., Zhuang X., Deng Z., et al. Grapefruit-derived nanovectors use an activated leukocyte trafficking pathway to deliver therapeutic agents to inflammatory tumor sites. Cancer Res. 2015;75:2520–2529. doi: 10.1158/0008-5472.CAN-14-3095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179.Du S., Guan Y., Xie A., Yan Z., Gao S., Li W., et al. Extracellular vesicles: a rising star for therapeutics and drug delivery. J Nanobiotechnol. 2023;21:231. doi: 10.1186/s12951-023-01973-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 180.Wang L., Wang D., Ye Z., Xu J. Engineering extracellular vesicles as delivery systems in therapeutic applications. Adv Sci (Weinh) 2023;10 doi: 10.1002/advs.202300552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181.Yong T., Wei Z., Gan L., Yang X. Extracellular-vesicle-based drug delivery systems for enhanced antitumor therapies through modulating the cancer-immunity cycle. Adv Mater. 2022;34 doi: 10.1002/adma.202201054. [DOI] [PubMed] [Google Scholar]
- 182.de Voogt W.S., Tanenbaum M.E., Vader P. Illuminating RNA trafficking and functional delivery by extracellular vesicles. Adv Drug Deliv Rev. 2021;174:250–264. doi: 10.1016/j.addr.2021.04.017. [DOI] [PubMed] [Google Scholar]
- 183.Rayamajhi S., Nguyen T.D.T., Marasini R., Aryal S. Macrophage-derived exosome-mimetic hybrid vesicles for tumor targeted drug delivery. Acta Biomater. 2019;94:482–494. doi: 10.1016/j.actbio.2019.05.054. [DOI] [PubMed] [Google Scholar]
- 184.Reck M., Remon J., Hellmann M.D. First-line immunotherapy for non-small-cell lung cancer. J Clin Oncol. 2022;40:586–597. doi: 10.1200/JCO.21.01497. [DOI] [PubMed] [Google Scholar]
- 185.Karachi A., Dastmalchi F., Mitchell D.A., Rahman M. Temozolomide for immunomodulation in the treatment of glioblastoma. Neuro Oncol. 2018;20:1566–1572. doi: 10.1093/neuonc/noy072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Cui J., Wang X., Li J., Zhu A., Du Y., Zeng W., et al. Immune exosomes loading self-assembled nanomicelles traverse the blood-brain barrier for chemo-immunotherapy against glioblastoma. ACS Nano. 2023;17:1464–1484. doi: 10.1021/acsnano.2c10219. [DOI] [PubMed] [Google Scholar]
- 187.Wang M., Lv C.Y., Li S.A., Wang J.K., Luo W.Z., Zhao P.C., et al. Near infrared light fluorescence imaging-guided biomimetic nanoparticles of extracellular vesicles deliver indocyanine green and paclitaxel for hyperthermia combined with chemotherapy against glioma. J Nanobiotechnol. 2021;19:210. doi: 10.1186/s12951-021-00907-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 188.Nguyen Cao T.G., Kang J.H., Kim W., Lim J., Kang S.J., You J.Y., et al. Engineered extracellular vesicle-based sonotheranostics for dual stimuli-sensitive drug release and photoacoustic imaging-guided chemo-sonodynamic cancer therapy. Theranostics. 2022;12:1247–1266. doi: 10.7150/thno.65516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 189.Tomar M.S., Kumar A., Srivastava C., Shrivastava A. Elucidating the mechanisms of Temozolomide resistance in gliomas and the strategies to overcome the resistance. Biochim Biophys Acta Rev Cancer. 2021;1876 doi: 10.1016/j.bbcan.2021.188616. [DOI] [PubMed] [Google Scholar]
- 190.Sun D., Zhuang X., Xiang X., Liu Y., Zhang S., Liu C., et al. A novel nanoparticle drug delivery system: the anti-inflammatory activity of curcumin is enhanced when encapsulated in exosomes. Mol Ther. 2010;18:1606–1614. doi: 10.1038/mt.2010.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 191.Hadla M., Palazzolo S., Corona G., Caligiuri I., Canzonieri V., Toffoli G., et al. Exosomes increase the therapeutic index of doxorubicin in breast and ovarian cancer mouse models. Nanomedicine. 2016;11:2431–2441. doi: 10.2217/nnm-2016-0154. [DOI] [PubMed] [Google Scholar]
- 192.Martins-Marques T., Pinho M.J., Zuzarte M., Oliveira C., Pereira P., Sluijter J.P., et al. Presence of Cx43 in extracellular vesicles reduces the cardiotoxicity of the anti-tumour therapeutic approach with doxorubicin. J Extracell Vesicles. 2016;5 doi: 10.3402/jev.v5.32538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 193.Niu W., Xiao Q., Wang X., Zhu J., Li J., Liang X., et al. A biomimetic drug delivery system by integrating grapefruit extracellular vesicles and doxorubicin-loaded heparin-based nanoparticles for glioma therapy. Nano Lett. 2021;21:1484–1492. doi: 10.1021/acs.nanolett.0c04753. [DOI] [PubMed] [Google Scholar]
- 194.Alshaer W., Zureigat H., Al Karaki A., Al-Kadash A., Gharaibeh L., Hatmal M.M., et al. siRNA: mechanism of action, challenges, and therapeutic approaches. Eur J Pharmacol. 2021;905 doi: 10.1016/j.ejphar.2021.174178. [DOI] [PubMed] [Google Scholar]
- 195.Guo S., Perets N., Betzer O., Ben-Shaul S., Sheinin A., Michaelevski I., et al. Intranasal delivery of mesenchymal stem cell derived exosomes loaded with phosphatase and tensin homolog siRNA repairs complete spinal cord injury. ACS Nano. 2019;13:10015–10028. doi: 10.1021/acsnano.9b01892. [DOI] [PubMed] [Google Scholar]
- 196.Pi F., Binzel D.W., Lee T.J., Li Z., Sun M., Rychahou P., et al. Nanoparticle orientation to control RNA loading and ligand display on extracellular vesicles for cancer regression. Nat Nanotechnol. 2018;13:82–89. doi: 10.1038/s41565-017-0012-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 197.Nieves W., Heang J., Asakrah S., Höner zu Bentrup K., Roy C.J., Morici L.A. Immunospecific responses to bacterial elongation factor Tu during Burkholderia infection and immunization. PLoS One. 2010;5 doi: 10.1371/journal.pone.0014361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Gujrati V., Kim S., Kim S.H., Min J.J., Choy H.E., Kim S.C., et al. Bioengineered bacterial outer membrane vesicles as cell-specific drug-delivery vehicles for cancer therapy. ACS Nano. 2014;8:1525–1537. doi: 10.1021/nn405724x. [DOI] [PubMed] [Google Scholar]
- 199.Katakowski M., Buller B., Zheng X., Lu Y., Rogers T., Osobamiro O., et al. Exosomes from marrow stromal cells expressing miR-146b inhibit glioma growth. Cancer Lett. 2013;335:201–204. doi: 10.1016/j.canlet.2013.02.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Bronisz A., Wang Y., Nowicki M.O., Peruzzi P., Ansari K., Ogawa D., et al. Extracellular vesicles modulate the glioblastoma microenvironment via a tumor suppression signaling network directed by miR-1. Cancer Res. 2014;74:738–750. doi: 10.1158/0008-5472.CAN-13-2650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 201.Viñas J.L., Burger D., Zimpelmann J., Haneef R., Knoll W., Campbell P., et al. Transfer of microRNA-486-5p from human endothelial colony forming cell-derived exosomes reduces ischemic kidney injury. Kidney Int. 2016;90:1238–1250. doi: 10.1016/j.kint.2016.07.015. [DOI] [PubMed] [Google Scholar]
- 202.Ohno S., Takanashi M., Sudo K., Ueda S., Ishikawa A., Matsuyama N., et al. Systemically injected exosomes targeted to EGFR deliver antitumor microRNA to breast cancer cells. Mol Ther. 2013;21:185–191. doi: 10.1038/mt.2012.180. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Liu M., Hu S., Yan N., Popowski K.D., Cheng K. Inhalable extracellular vesicle delivery of IL-12 mRNA to treat lung cancer and promote systemic immunity. Nat Nanotechnol. 2024;19:565–575. doi: 10.1038/s41565-023-01580-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 204.Nagelkerke A., Ojansivu M., van der Koog L., Whittaker T.E., Cunnane E.M., Silva A.M., et al. Extracellular vesicles for tissue repair and regeneration: evidence, challenges and opportunities. Adv Drug Deliv Rev. 2021;175 doi: 10.1016/j.addr.2021.04.013. [DOI] [PubMed] [Google Scholar]
- 205.Liao W., Du Y., Zhang C., Pan F., Yao Y., Zhang T., et al. Exosomes: the next generation of endogenous nanomaterials for advanced drug delivery and therapy. Acta Biomater. 2019;86:1–14. doi: 10.1016/j.actbio.2018.12.045. [DOI] [PubMed] [Google Scholar]
- 206.Zhang B., Wang M., Gong A., Zhang X., Wu X., Zhu Y., et al. HucMSC-exosome mediated-Wnt4 signaling is required for cutaneous wound healing. Stem Cell. 2015;33:2158–2168. doi: 10.1002/stem.1771. [DOI] [PubMed] [Google Scholar]
- 207.Pironti G., Strachan R.T., Abraham D., Mon-Wei Yu S., Chen M., Chen W., et al. Circulating exosomes induced by cardiac pressure overload contain functional angiotensin II type 1 receptors. Circulation. 2015;131:2120–2130. doi: 10.1161/CIRCULATIONAHA.115.015687. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 208.Mulcahy L.A., Pink R.C., Carter D.R. Routes and mechanisms of extracellular vesicle uptake. J Extracell Vesicles. 2014;3:24641. doi: 10.3402/jev.v3.24641. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 209.Poggio M., Hu T., Pai C.C., Chu B., Belair C.D., Chang A., et al. Suppression of exosomal PD-L1 induces systemic anti-tumor immunity and memory. Cell. 2019;177 doi: 10.1016/j.cell.2019.02.016. 414–27.e13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210.Chen S.W., Zhu S.Q., Pei X., Qiu B.Q., Xiong D., Long X., et al. Cancer cell-derived exosomal circUSP7 induces CD8(+) T cell dysfunction and anti-PD1 resistance by regulating the miR-934/SHP2 axis in NSCLC. Mol Cancer. 2021;20:144. doi: 10.1186/s12943-021-01448-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.Zhang P.F., Gao C., Huang X.Y., Lu J.C., Guo X.J., Shi G.M., et al. Cancer cell-derived exosomal circUHRF1 induces natural killer cell exhaustion and may cause resistance to anti-PD1 therapy in hepatocellular carcinoma. Mol Cancer. 2020;19:110. doi: 10.1186/s12943-020-01222-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Zhang W., Zhong W., Wang B., Yang J., Yang J., Yu Z., et al. ICAM-1-mediated adhesion is a prerequisite for exosome-induced T cell suppression. Dev Cell. 2022;57 doi: 10.1016/j.devcel.2022.01.002. 329–43.e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Zhou W., Zhou Y., Chen X., Ning T., Chen H., Guo Q., et al. Pancreatic cancer-targeting exosomes for enhancing immunotherapy and reprogramming tumor microenvironment. Biomaterials. 2021;268 doi: 10.1016/j.biomaterials.2020.120546. [DOI] [PubMed] [Google Scholar]
- 214.Fan M., Liu H., Yan H., Che R., Jin Y., Yang X., et al. A CAR T-inspiring platform based on antibody-engineered exosomes from antigen-feeding dendritic cells for precise solid tumor therapy. Biomaterials. 2022;282 doi: 10.1016/j.biomaterials.2022.121424. [DOI] [PubMed] [Google Scholar]
- 215.Hu S., Ma J., Su C., Chen Y., Shu Y., Qi Z., et al. Engineered exosome-like nanovesicles suppress tumor growth by reprogramming tumor microenvironment and promoting tumor ferroptosis. Acta Biomater. 2021;135:567–581. doi: 10.1016/j.actbio.2021.09.003. [DOI] [PubMed] [Google Scholar]
- 216.Wang R., Li J., Wang X., Zhang Y., Zhu A., Feng K., et al. Personalized nanovaccines enhance lymph node accumulation and reprogram the tumor microenvironment for improved photodynamic immunotherapy. Nano Lett. 2024;24:7432–7442. doi: 10.1021/acs.nanolett.4c01678. [DOI] [PubMed] [Google Scholar]
- 217.Wang R., Wang X., Zhao H., Li N., Li J., Zhang H., et al. Targeted delivery of hybrid nanovesicles for enhanced brain penetration to achieve synergistic therapy of glioma. J Control Release. 2024;365:331–347. doi: 10.1016/j.jconrel.2023.11.033. [DOI] [PubMed] [Google Scholar]
- 218.Zhang L., Yu D. Exosomes in cancer development, metastasis, and immunity. Biochim Biophys Acta Rev Cancer. 2019;1871:455–468. doi: 10.1016/j.bbcan.2019.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 219.Yin Z., Ma T., Huang B., Lin L., Zhou Y., Yan J., et al. Macrophage-derived exosomal microRNA-501-3p promotes progression of pancreatic ductal adenocarcinoma through the TGFBR3-mediated TGF-β signaling pathway. J Exp Clin Cancer Res. 2019;38:310. doi: 10.1186/s13046-019-1313-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 220.Lu Z., Zuo B., Jing R., Gao X., Rao Q., Liu Z., et al. Dendritic cell-derived exosomes elicit tumor regression in autochthonous hepatocellular carcinoma mouse models. J Hepatol. 2017;67:739–748. doi: 10.1016/j.jhep.2017.05.019. [DOI] [PubMed] [Google Scholar]
- 221.Yang P., Cao X., Cai H., Feng P., Chen X., Zhu Y., et al. The exosomes derived from CAR-T cell efficiently target mesothelin and reduce triple-negative breast cancer growth. Cell Immunol. 2021;360 doi: 10.1016/j.cellimm.2020.104262. [DOI] [PubMed] [Google Scholar]
- 222.Fan M., Liu H., Yan H., Che R., Jin Y., Yang X., et al. A CAR T-inspiring platform based on antibody-engineered exosomes from antigen-feeding dendritic cells for precise solid tumor therapy. Biomaterials. 2022;282 doi: 10.1016/j.biomaterials.2022.121424. [DOI] [PubMed] [Google Scholar]
- 223.Johnson L.R., Lee D.Y., Eacret J.S., Ye D., June C.H., Minn A.J. The immunostimulatory RNA RN7SL1 enables CAR-T cells to enhance autonomous and endogenous immune function. Cell. 2021;184 doi: 10.1016/j.cell.2021.08.004. 4981–95.e14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 224.Li J., Wang X., Guo Y., Zhang Y., Zhu A., Zeng W., et al. Ginsenoside Rg3-engineered exosomes as effective delivery platform for potentiated chemotherapy and photoimmunotherapy of glioblastoma. Chem Eng J. 2023;471 [Google Scholar]
- 225.Wang R., Li J., Wang X., Zhang Y., Zhu A., Feng K., et al. Personalized nanovaccines enhance lymph node accumulation and reprogram the tumor microenvironment for improved photodynamic immunotherapy. Nano Lett. 2024;24:7432–7442. doi: 10.1021/acs.nanolett.4c01678. [DOI] [PubMed] [Google Scholar]
- 226.Veerman R.E., Akpinar G.G., Offens A., Steiner L., Larssen P., Lundqvist A., et al. Antigen-loaded extracellular vesicles induce responsiveness to anti-PD-1 and anti-PD-L1 treatment in a checkpoint refractory melanoma model. Cancer Immunol Res. 2023;11:217–227. doi: 10.1158/2326-6066.CIR-22-0540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 227.Matsumoto A., Asuka M., Takahashi Y., Takakura Y. Antitumor immunity by small extracellular vesicles collected from activated dendritic cells through effective induction of cellular and humoral immune responses. Biomaterials. 2020;252 doi: 10.1016/j.biomaterials.2020.120112. [DOI] [PubMed] [Google Scholar]
- 228.Zuo B., Zhang Y., Zhao K., Wu L., Qi H., Yang R., et al. Universal immunotherapeutic strategy for hepatocellular carcinoma with exosome vaccines that engage adaptive and innate immune responses. J Hematol Oncol. 2022;15:46. doi: 10.1186/s13045-022-01266-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 229.Hansen A.S., Jensen L.S., Gammelgaard K.R., Ryttersgaard K.G., Krapp C., Just J., et al. T-cell derived extracellular vesicles prime macrophages for improved STING based cancer immunotherapy. J Extracell Vesicles. 2023;12 doi: 10.1002/jev2.12350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230.Shin S., Jung I., Jung D., Kim C.S., Kang S.M., Ryu S., et al. Novel antitumor therapeutic strategy using CD4(+) T cell-derived extracellular vesicles. Biomaterials. 2022;289 doi: 10.1016/j.biomaterials.2022.121765. [DOI] [PubMed] [Google Scholar]
- 231.Zuo B., Qi H., Lu Z., Chen L., Sun B., Yang R., et al. Alarmin-painted exosomes elicit persistent antitumor immunity in large established tumors in mice. Nat Commun. 2020;11:1790. doi: 10.1038/s41467-020-15569-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 232.Kim G.B., Nam G.H., Hong Y., Woo J., Cho Y., Kwon I.C., et al. Xenogenization of tumor cells by fusogenic exosomes in tumor microenvironment ignites and propagates antitumor immunity. Sci Adv. 2020;6:eaaz2083. doi: 10.1126/sciadv.aaz2083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 233.Cheng Q., Dai Z., Smbatyan G., Epstein A.L., Lenz H.J., Zhang Y. Eliciting anti-cancer immunity by genetically engineered multifunctional exosomes. Mol Ther. 2022;30:3066–3077. doi: 10.1016/j.ymthe.2022.06.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 234.Yu D., Li Y., Wang M., Gu J., Xu W., Cai H., et al. Exosomes as a new frontier of cancer liquid biopsy. Mol Cancer. 2022;21:56. doi: 10.1186/s12943-022-01509-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 235.Tominaga N., Yoshioka Y., Ochiya T. A novel platform for cancer therapy using extracellular vesicles. Adv Drug Deliv Rev. 2015;95:50–55. doi: 10.1016/j.addr.2015.10.002. [DOI] [PubMed] [Google Scholar]
- 236.Chen W., Tang C., Chen G., Li J., Li N., Zhang H., et al. Boosting checkpoint immunotherapy with biomimetic nanodrug delivery systems. Adv Healthcare Mater. 2024;13 doi: 10.1002/adhm.202304284. [DOI] [PubMed] [Google Scholar]
- 237.Ostrowski M., Carmo N.B., Krumeich S., Fanget I., Raposo G., Savina A., et al. Rab27a and Rab27b control different steps of the exosome secretion pathway. Nat Cell Biol. 2010;12:19–30. doi: 10.1038/ncb2000. sup pp. 1–13. [DOI] [PubMed] [Google Scholar]
- 238.Lane R.E., Korbie D., Hill M.M., Trau M. Extracellular vesicles as circulating cancer biomarkers: opportunities and challenges. Clin Transl Med. 2018;7:14. doi: 10.1186/s40169-018-0192-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 239.Medhin L.B., Beasley A.B., Warburton L., Amanuel B., Gray E.S. Extracellular vesicles as a liquid biopsy for melanoma: are we there yet? Semin Cancer Biol. 2023;89:92–98. doi: 10.1016/j.semcancer.2023.01.008. [DOI] [PubMed] [Google Scholar]
- 240.Kuypers S., Smisdom N., Pintelon I., Timmermans J.P., Ameloot M., Michiels L., et al. Unsupervised machine learning-based clustering of nanosized fluorescent extracellular vesicles. Small. 2021;17 doi: 10.1002/smll.202006786. [DOI] [PubMed] [Google Scholar]
- 241.Buzas E.I. The roles of extracellular vesicles in the immune system. Nat Rev Immunol. 2023;23:236–250. doi: 10.1038/s41577-022-00763-8. [DOI] [PMC free article] [PubMed] [Google Scholar]