Abstract
Background
Because frailty is a major health concern among older patients, identifying frailty-related biomarkers will help in the early detection and prevention of frailty. Thus, we aimed to determine the association between circulating levels of silent mating-type information regulation 2 homolog 1 (SIRT1) and frailty.
Methods
We assessed circulating SIRT1 levels in 16 robust, 74 prefrail, and 40 frail older adults. Frailty was diagnosed based on the Fried phenotype. Circulating cytokine and adipokine (e.g., vaspin, adiponectin, and leptin) levels were assessed. Differences in SIRT1 levels among the three subject groups were compared; correlations of SIRT1 levels with physical function and adipokine and cytokine levels were analyzed.
Results
Serum SIRT1 levels were significantly higher among frail older adults than among robust ones. Older adults with slowness or weight loss had high SIRT1 levels. Serum SIRT1 levels negatively correlated with gait speed, even after adjustment for age and sex; age; and insulin, vaspin, and leptin levels; they correlated negatively with phospholipase A2 levels.
Conclusions
High SIRT1 levels were observed in frail elderly patients and were correlated with decreased physical function. Insulin and adipokine levels might be the link between SIRT1 and frailty, whereas inflammation may not be involved in this process.
Key words: SIRT1, frailty, adipokine, older adult, insulin
Abbreviations
- SIRT1
silent mating-type information regulation 2 homolog 1
- CR
calorie restriction
- BMI
body mass index
- CRP
C-reactive protein
- IIS
inflammation index score
Introduction
Aging is a lifelong process characterized by a gradual, cumulative decline in physiological functional capacity. Unlike normal age-related changes and other age-related diseases, frailty is a geriatric syndrome that is defined as a state of reduced physiological reserve and increased vulnerability to stressors. The most commonly used criteria for frailty was proposed by Fried et al., in which individuals exhibited 3 or more of the 5 following components: slowness, exhaustion, weight loss, weakness, and low activity, with a biological underpinning (1). Other definitions of frailty involved a model of accumulated health deficits (2). Regardless of different definitions, frail individuals are extremely vulnerable to adverse outcomes such as disability and death, leading to increased health care costs (1, 3). However, the underlying pathophysiology of frailty is complex and poorly understood. Early intervention is important to reverse frailty, but diagnosing frailty at an early stage is difficult. Because frailty is highly prevalent and associated with adverse outcomes, identifying frailty-related biomarkers would be useful for the early detection and prevention of frailty. However, little is known regarding frailty-related biomarkers in Chinese older adults.
Silent mating-type information regulation 2 homolog 1 (SIRT1) is an NAD+-dependent deacetylase with roles in several key pathways such as those involving energy metabolism, cell differentiation, cell aging, tumor suppression, and anti-inflammation (4, 5, 6. Increased SIRT1 activity is associated with delayed aging and delayed onset of metabolic disorders in animal models (5, 7, 8). Because sirtuins play an important role in metabolic regulation pathways in mammals and lead to the beneficial effects of calorie restriction (CR), they may be helpful in assessing the process of aging and treatment of aging-related diseases (9). We previously demonstrated that CR could delay cognitive decline by increasing SIRT1 levels in mice (10). As a key regulator of metabolism and lifespan, circulating SIRT1 levels have been implicated in preventing aging-related diseases. Resveratrol, an agonist of SIRT1, delayed aging in various laboratory models such as yeast, Caenorhabditis elegans, Drosophila, and mice (5, 7). Therefore, clarifying the mechanisms underlying frailty is important to predict longevity and devise therapeutic strategies for frail older adults. SIRT1 has been found to be a promising biomarker of age-related diseases in animal models; therefore, considering its significance in aging, researchers have focused on using SIRT1 in clinical settings, such as for patients with asthma (11), rheumatoid arthritis (12), multiple sclerosis (13), and Alzheimer’s disease (14). However, recent studies on SIRT1 levels and frailty have yielded controversial results. Kumar et al. found that low sirtuin levels were significantly associated with frailty (15), whereas the Concord Health and Ageing in Men Project study suggested a paradoxical association between low serum SIRT1 levels and robustness (16). Therefore, whether circulating SIRT1 levels can be used as a potential marker for frailty remains unclear. Thus, here, we aimed to determine SIRT1 levels in frail individuals and assess its association with inflammation and adipokine levels in Chinese older adults.
Methods
Study design and participants
Data were from cardiovascular health, cognition and aging study (CHCAS). This cross-sectional study included 130 inpatients aged ≥60 years (16 robust, 74 prefrail, and 40 frail) from Xuanwu Hospital, Capital Medical University. The average ages of subjects in the robust, prefrail, and frail groups were 68.44±7.29, 71.41±7.81 and 77.13±8.95 years, respectively (frail vs. robust, p=0.001). Comprehensive medical histories and frailty measurements of all patients were obtained by trained staff. Individuals having acute illnesses or severe diseases or those receiving corticosteroids, methotrexate, nonsteroidal anti-inflammatory drugs or other immunemodulating agents were excluded. The study protocol was approved by the institutional review board of Xuanwu Hospital, Capital Medical University, and informed consent was obtained from all participants prior to participation.
Frailty assessment
To explore the relationship between age-related frailty and the underlying processes that influenced changes in health, we used the frailty phenotype to define frailty. Frailty was defined according to the Fried phenotype that includes slowness, exhaustion, weight loss, weakness, and low activity (1). The participants were classified as frail (≥3 components), prefrail (1-2 components), and robust (0 component).
Grip strength was defined as the maximum value of the three times in the dominant hand in the standing position. The cutoff value for weak grip strength was determined as previously reported (17), after adjusting for sex and body mass index (BMI).
Walking speed was measured by a 20-m walk test. Participants were encouraged to walk 20 m at their usual pace. The cutoff value for slow walking speed was determined as in our unpublished paper[18], after adjustment for sex and height.
Exhaustion was indicated by self-response to questions from the Geriatric Depression Scale; a response of “I felt everything I did was an effort” was considered a positive answer and that of “I am full of energy” was considered a negative answer.
Weight loss was defined as a self-reported unintentional weight loss of 2 kg in the past 6 months or as having a BMI <18.5 kg/m2.
Low activity was defined as self-reported exercise for <3 h/week over the past 12 months (19).
Blood test
Peripheral blood samples were collected after overnight fasting; serum samples were isolated and were stored at −80°C before use. SIRT1, vaspin, leptin, adiponectin, phospholipase A2, interleukin-6, and tumor necrosis factor receptor 1 levels were measured using enzyme-linked immunosorbent assays. C-reactive protein (CRP) levels were measured using immunoturbidimetry. An inflammation index score (IIS) that included interleukin-6 and tumor necrosis factor receptor 1 levels was calculated; IIS was identified as the best predictor of 10-year mortality in the Cardiovascular Health Study (20).
Statistical analysis
Data were presented as mean ± standard deviation or mean rank or number and percentage. The difference in the characteristics between the two groups was evaluated by chisquare test for categorical variables, independent t test for continuous variables and Kruskal-Wallis comparisons for the abnormally distributed continuous variables. Pearson correlation coefficients were calculated for the relationship of physical function with circulating cytokines and adipokines. All these statistical analyses were performed with SPSS (Chicago, IL, USA, version 11.0) or GraphPad Prism 7.0 software (GraphPad Software Inc., CA, USA). P-value was significant at P <0.05.
Results
Frail participants had higher SIRT1 levels than robust participants (frail, (4.66±1.64) ng/ml; prefrail, (4.24±1.62) ng/ml; robust, (3.68±1.68) ng/ml, frail vs robust, p = 0.046). To determine whether differences existed among the five components measured in our study, we compared serum SIRT1 levels between participants with or without each component. Of the 130 older adults, 51 were designated as having slowness. SIRT1 levels were higher in the slowness groups than in the normal walking speed group ((5.03±1.58) ng/ml vs (3.85±1.53) ng/ml, p < 0.001). Although the number of participants having weight loss was small, SIRT1 levels was higher in the weight loss group than that in the no weight loss group ((5.75±1.08) ng/ml vs (4.24±1.64) ng/ml, p = 0.017). As for the other 3 components of frailty, there was no significant difference between the two groups (with and without the component) (Fig. 1).
Figure 1.
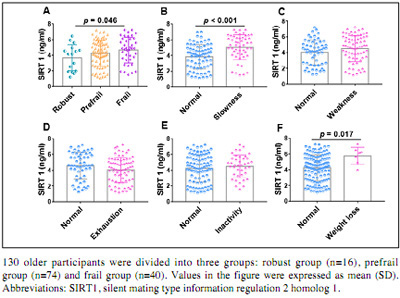
Comparison of SIRT1 levels in different groups
To confirm our finding that serum SIRT1 levels increased with frailty, we analyzed the correlation between serum SIRT1 levels and physical functions such as grip strength and walking speed. Circulating SIRT1 levels were negatively correlated with gait speed (r=-0.278, p<0.001), while there was no correlation with grip strength (Fig. 2). A positive correlation was observed between circulating SIRT1 levels and age (r=0.288, p=0.001), but not between circulating SIRT1 levels and BMI (Fig. 2). To avoid the effect of age on the correlation between SIRT1 levels and walking speed, we adjusted for age and sex; however, the negative correlation remained unchanged (r=-0.188, p=0.039).
Figure 2.
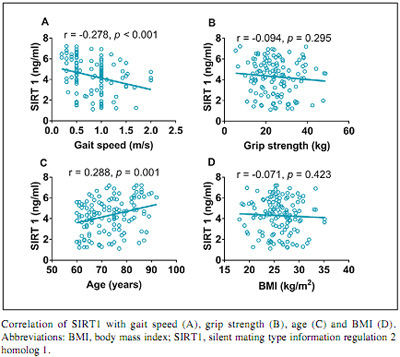
Correlation of SIRT1 levels with physical function, age, and BMI
As chronic inflammation is a major factor in the process of frailty, we explored whether circulating SIRT1 levels were correlated with inflammation. As the IIS, including interleukin-6 and tumor necrosis factor receptor 1, could better capture the effect of chronic inflammation on mortality (20), we used the IIS and CRP levels as inflammatory markers. Our results showed no correlation between serum SIRT1 levels and IIS or CRP levels. Since the Concord Health and Ageing in Men Project study revealed that serum SIRT1 expression might reflect the nutritional status of frail individuals, we further determined whether circulating SIRT1 levels were correlated with insulin and adipokine levels. Our results showed that SIRT1 levels were negatively correlated with phospholipase A2 levels (r=-0.318, p<0.001) and positively correlated with vaspin (r=0.233, p=0.008), leptin (r=0.220, p=0.012) and insulin (r=0.216, p=0.014) levels. There were no correlations between SIRT1 levels and glucose and adiponectin levels (Fig. 3). This indicates that insulin and adipokines, such as vaspin and leptin, might be involved in the process of frailty.
Figure 3.
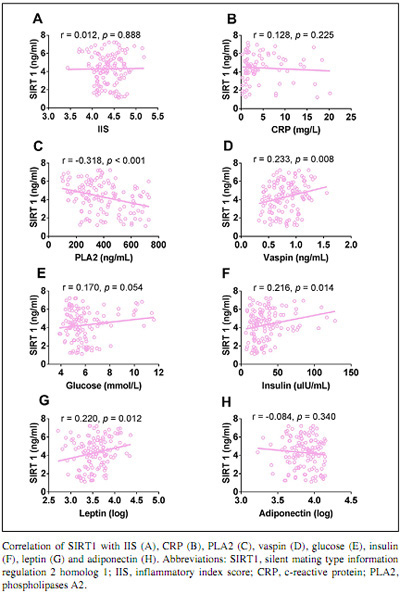
Correlation of SIRT1 levels with inflammatory markers and levels of insulin and adipokines.
Discussion
Our results demonstrated that frail patients have high SIRT1 levels. SIRT1 levels were higher in the slowness group than in the normal walking speed group; similar results were obtained for weight loss. Circulating SIRT1 levels were negatively correlated with walking speed and positively correlated with age; no correlation was observed with grip strength or BMI.
SIRT1 is a key regulator of aging-related metabolic changes and aging processes. Zhong et al. found that older adults had significantly lower serum SIRT1 levels than younger adults (21). Another study showed that an aging-related correlation between serum SIRT1 levels and basal metabolic rate was observed in women; SIRT1 levels was positively correlated with triglyceride (TG) in men and with waist circumference, blood pressure, and TG in women; and women in their thirties had highest SIRT1 levels, indicating that serum SIRT1 levels may be utilized as a marker of aging (22). Serum SIRT1 levels declined with age, and low SIRT1 levels were found in patients with Alzheimer’s disease and mild cognitive impairment (14). A significant association between SIRT1 single-nucleotide polymorphisms and longevity has been reported (23); however, another study showed no association between SIRT1 variants and longevity (24). In addition, most studies revealed that SIRT1 levels decreased during various diseases such as fatty liver infiltration (25); however, other studies reported opposing results, e.g., SIRT1 levels increased in patients with asthma (11).
To our knowledge, this is the first study to demonstrate a correlation between circulating SIRT1 levels and insulin and adipokine levels. SIRT1 levels were negatively correlated with phospholipase A2 levels and positively correlated with insulin and adipokine levels, such as those of vaspin and leptin. No correlation was observed with IIS and CRP, glucose, and adiponectin levels in this study. After adjustment for age and sex, SIRT1 levels were still negatively correlated with walking speed. Although many studies have suggested that SIRT1 plays a role in inflammation, we did not observe any correlation between SIRT1 levels and inflammation in this study. There is no literature on SIRT1 and adipokines. There have been several studies on the association between serum SIRT1 levels and frailty with inconsistent results. One study found that SIRT1 single-nucleotide polymorphisms and serum SIRT1 levels in older men were possibly more closely associated with nutrition and body composition than with aging and age-related conditions (26). Another study found no association between frailty and serum SIRT1 levels, although post hoc analysis suggested a paradoxical association between low serum SIRT1 levels and robustness (16).
Our study showed that SIRT1 levels increased in frail individuals. Several hypotheses might explain this increase. First, SIRT1 plays a critical role in regulating metabolism and aging in mammals (27). CR and resveratrol significantly increased circulating SIRT1 levels (28). Because weight loss is a component of frailty, lower BMI might mimic the effects of CR, resulting in increased SIRT1 levels. Second, Le Couteur et al. found that low serum SIRT1 levels were associated with less frail and high total lean body weight, indicating that serum SIRT1 levels might reflect the nutritional status of an individual (16). Third, different tissue matters. Lung SIRT1 levels decreased, whereas serum SIRT1 levels increased in the setting of asthma (11); furthermore, in old rats, SIRT1 levels decreased in the heart but not in the adipose tissue (29). Finally, the different downstream targets of SIRT1 might be responsible for this increase. Overexpression or knockout of SIRT1 can inhibit peroxisome-proliferator-activated receptor coactivator, further decreasing mitochondrial activity (30). Forkhead box O1 and peroxisome proliferator-activated receptor gamma are downstream targets of SIRT1, and they differed by types of target tissues and stimulus (31).
In the present study, we observed elevated SIRT1 levels in frail individuals and a correlation between serum SIRT1 levels and adipokine levels; however, the cross-sectional study design could not explain the causal relationship between circulating SIRT1 levels and frailty, and therefore, further analyses based on longitudinal data are needed.
Levels of SIRT1, a class III histone deacetylase with both anti-inflammatory and anti-aging effects, were elevated in frail older adults. To our knowledge, this is the first study to demonstrate that circulating SIRT1 levels were correlated with adipokine levels. Our study indicated that higher serum SIRT1 levels were a biological characteristic of frailty. Furthermore, insulin and adipokine levels might be the link between SIRT1 and frailty, whereas inflammation may not be involved in this process.
This study has important implications for clinical work as it will help clinicians in the early detection and prevention of frailty in older adults, thereby reducing frailty-related adverse outcomes such as disability and death. Prospective studies will be necessary to examine the value of circulating SIRT1 levels as a biomarker for frailty and determine its clinical significance.
Acknowledgments
We acknowledge all the people who participated in the study.
Competing interest
None.
Funding
This work was supported by the Beijing Municipal Administration of Hospitals Clinical Medicine Development of Special Funding Support (ZYLX201706), Milstein Medical Asian American Partnership Foundation Project Award in Geriatrics (2018), and Beijing Municipal Health Bureau Research Fund (Jing 17-12).
Ethical standards
The present study followed the ethical guideline of the declaration of Helsinki and was approved by the Ethics Committees.
References
- 1.Fried LP, Tangen CM, Walston J, Newman AB, Hirsch C, Gottdiener J, et al. Frailty in Older Adults: Evidence for a Phenotype. Journals Gerontol Ser A Biol Sci Med Sci. 2001;56:M146–57. doi: 10.1093/gerona/56.3.m146. 10.1093/gerona/56.3.M146 [DOI] [PubMed] [Google Scholar]
- 2.Rockwood K, Mitnitski A. Frailty in relation to the accumulation of deficits. J Gerontol A Biol Sci Med Sci. 2007;62:722–727. doi: 10.1093/gerona/62.7.722. 10.1093/gerona/62.7.722 PubMed PMID: 17634318. [DOI] [PubMed] [Google Scholar]
- 3.Ma L, Zhang L, Tang Z, Sun F, Diao L, Wang J, et al. Use of the frailty index in evaluating the prognosis of older people in Beijing: A cohort study with an 8-year follow-up. Arch Gerontol Geriatr. 2016;64:172–177. doi: 10.1016/j.archger.2015.11.002. 10.1016/j.archger.2015.11.002 PubMed PMID: 26778493. [DOI] [PubMed] [Google Scholar]
- 4.Ma L, Li Y. SIRT1: Role in cardiovascular biology. Clin Chim Acta. 2015;440:8–15. doi: 10.1016/j.cca.2014.10.036. 10.1016/j.cca.2014.10.036 PubMed PMID: 25444742. [DOI] [PubMed] [Google Scholar]
- 5.Baur JA, Ungvari Z, Minor R L, Couteur D d, Cabo R. Are sirtuins viable targets for improving healthspan and lifespan. Nat Rev Drug Discov. 2012;11:443–461. doi: 10.1038/nrd3738. 10.1038/nrd3738 PubMed PMID: 22653216, PMCID 4684642. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sinclair DA, Guarente L. Small-Molecule Allosteric Activators of Sirtuins. Annu Rev Pharmacol Toxicol. 2014;54:363–380. doi: 10.1146/annurev-pharmtox-010611-134657. 10.1146/annurev-pharmtox-010611-134657 PubMed PMID: 24160699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Baur JA, Pearson KJ, Price NL, Jamieson HA, Lerin C, Kalra A, et al. Resveratrol improves health and survival of mice on a high-calorie diet. Nature. 2006;444:337–342. doi: 10.1038/nature05354. 10.1038/nature05354 PubMed PMID: 17086191, PMCID 4990206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Curcio C-L-L, Henao G-M, Gomez F, Yang Y, Lee L, Rodríguez-Mañas L, et al. Frailty among rural elderly adults. BMC Geriatr. 2014;14:2. doi: 10.1186/1471-2318-14-2. 10.1186/1471-2318-14-2 PubMed PMID: 24405584, PMCID 3898393. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Qadir MI, Anwar S. Sirtuins in Brain Aging and Neurological Disorders. Crit Rev Eukaryot Gene Expr. 2017;27(4):321–329. doi: 10.1615/CritRevEukaryotGeneExpr.2017019532. 10.1615/CritRevEukaryotGeneExpr.2017019532 PubMed PMID: 29283326. [DOI] [PubMed] [Google Scholar]
- 10.Ma L, Dong W, Wang R, Li Y, Xu B, Zhang J, et al. Effect of caloric restriction on the SIRT1/mTOR signaling pathways in senile mice. Brain Res Bull. 2015;116:67–72. doi: 10.1016/j.brainresbull.2015.06.004. 10.1016/j.brainresbull.2015.06.004 PubMed PMID: 26135885. [DOI] [PubMed] [Google Scholar]
- 11.Wang Y, Li D, Ma G, Li W, Wu J, Lai T, et al. Increases in peripheral SIRT1: a new biological characteristic of asthma. Respirology. 2015;20:1066–1072. doi: 10.1111/resp.12558. 10.1111/resp.12558 PubMed PMID: 26040995. [DOI] [PubMed] [Google Scholar]
- 12.Wendling D, Abbas W, Godfrin-Valnet M, Kumar A, Guillot X, Khan KA, et al. Dysregulated Serum IL-23 and SIRT1 Activity in Peripheral Blood Mononuclear Cells of Patients with Rheumatoid Arthritis. PLoS One. 2015;10:e0119981. doi: 10.1371/journal.pone.0119981. 10.1371/journal.pone.0119981 PubMed PMID: 25799392, PMCID 4370395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Li R, Sun X, Shu Y, Wang Y, Xiao L, Wang Z, et al. Serum CCL20 and its association with SIRT1 activity in multiple sclerosis patients. J Neuroimmunol. 2017;313:56–60. doi: 10.1016/j.jneuroim.2017.10.013. 10.1016/j.jneuroim.2017.10.013 PubMed PMID: 29153609. [DOI] [PubMed] [Google Scholar]
- 14.Kumar R, Chaterjee P, Sharma PK, Singh AK, Gupta A, Gill K, et al. Sirtuin1: A Promising Serum Protein Marker for Early Detection of Alzheimer’s Disease. PLoS One. 2013;8:e61560. doi: 10.1371/journal.pone.0061560. 10.1371/journal.pone.0061560 PubMed PMID: 23613875, PMCID 3628714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kumar R, Mohan N, Upadhyay AD, Singh AP, Sahu V, Dwivedi S, et al. Identification of serum sirtuins as novel noninvasive protein markers for frailty. Aging Cell. 2014;13:975–980. doi: 10.1111/acel.12260. 10.1111/acel.12260 PubMed PMID: 25100619, PMCID 4326933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Le Couteur DG, Benson VL, McMahon AC, Blyth F, Handelsman DJ, Seibel MJ, et al. Determinants of Serum-Induced SIRT1 Expression in Older Men: The CHAMP Study. Journals Gerontol Ser A Biol Sci Med Sci. 2011;66A:3–8. doi: 10.1093/gerona/glq158. 10.1093/gerona/glq158 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Wu, C, Smit, E, Xue, Q-L, Odden, MC, Prevalence and Correlates of Frailty among Community-Dwelling Chinese Older Adults: The China Health and Retirement Longitudinal Study, J Gerontol A Biol Sci Med Sci, 2017 [DOI] [PMC free article] [PubMed]
- 18.Ma, L, Cognitive Function in Prefrail and Frail Community-dwelling Older Adults in China, 2018 [DOI] [PMC free article] [PubMed]
- 19.Ma, L, Tang, Z, Chan, P WJ, Novel Frailty Screening Questionnaire (FSQ) Predicts 8-year Mortality in Older Adults in China, J Frailty Aging, 2018 [DOI] [PMC free article] [PubMed]
- 20.Varadhan R, Yao W, Matteini A, Beamer BA, Xue QL, Yang H, et al. Simple biologically informed infammatory index of two serum cytokines predicts 10 year all-cause mortality in older adults. Journals Gerontol -Ser A Biol Sci Med Sci. 2014;69A:165–173. doi: 10.1093/gerona/glt023. 10.1093/gerona/glt023 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Zhong Y, Chen AF, Zhao J, Gu Y-J, Fu G-X. Serum levels of cathepsin D, sirtuin1, and endothelial nitric oxide synthase are correlatively reduced in elderly healthy people. Aging Clin Exp Res. 2016;28:641–645. doi: 10.1007/s40520-015-0472-7. 10.1007/s40520-015-0472-7 PubMed PMID: 26462844. [DOI] [PubMed] [Google Scholar]
- 22.Lee HJ, Yang SJ. Aging-Related Correlation between Serum Sirtuin 1 Activities and Basal Metabolic Rate in Women, but not in Men. Clin Nutr Res. 2017;6:18. doi: 10.7762/cnr.2017.6.1.18. 10.7762/cnr.2017.6.1.18 PubMed PMID: 28168178, PMCID 5288549. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kim S, Bi X, Czarny-Ratajczak M, Dai J, Welsh DA, Myers L, et al. Telomere maintenance genes SIRT1 and XRCC6 impact age-related decline in telomere length but only SIRT1 is associated with human longevity. Biogerontology. 2012;13:119–131. doi: 10.1007/s10522-011-9360-5. 10.1007/s10522-011-9360-5 PubMed PMID: 21972126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Lin R, Yan D, Zhang Y, Liao X, Gong G, Hu J, et al. Common variants in SIRT1 and human longevity in a Chinese population. BMC Med Genet. 2016;17:31. doi: 10.1186/s12881-016-0293-3. 10.1186/s12881-016-0293-3 PubMed PMID: 27089876, PMCID 4836161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Mariani S, Fiore D, Basciani S, Persichetti A, Contini S, Lubrano C, et al. Plasma levels of SIRT1 associate with non-alcoholic fatty liver disease in obese patients. Endocrine. 2015;49:711–716. doi: 10.1007/s12020-014-0465-x. 10.1007/s12020-014-0465-x PubMed PMID: 25358448. [DOI] [PubMed] [Google Scholar]
- 26.Razi S, Cogger VC, Kennerson M, Benson VL, McMahon AC, Blyth FM, et al. SIRT1 Polymorphisms and Serum-Induced SIRT1 Protein Expression in Aging and Frailty: The CHAMP Study. Journals Gerontol Ser A. 2017;72:870–876. doi: 10.1093/gerona/glx018. 10.1093/gerona/glx018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Imai S. The NAD World: A New Systemic Regulatory Network for Metabolism and Aging—Sirt1, Systemic NAD Biosynthesis, and Their Importance. Cell Biochem Biophys. 2009;53:65–74. doi: 10.1007/s12013-008-9041-4. 10.1007/s12013-008-9041-4 PubMed PMID: 19130305, PMCID 2734380. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mansur AP, Roggerio A, Goes MFS, Avakian SD, Leal DP, Maranhão RC, et al. Serum concentrations and gene expression of sirtuin 1 in healthy and slightly overweight subjects after caloric restriction or resveratrol supplementation: A randomized trial. Int J Cardiol. 2017;227:788–794. doi: 10.1016/j.ijcard.2016.10.058. 10.1016/j.ijcard.2016.10.058 PubMed PMID: 28029409. [DOI] [PubMed] [Google Scholar]
- 29.Ferrara N, Rinaldi B, Corbi G, Conti V, Stiuso P, Boccuti S, et al. Exercise Training Promotes SIRT1 Activity in Aged Rats. Rejuvenation Res. 2008;11:139–150. doi: 10.1089/rej.2007.0576. 10.1089/rej.2007.0576 PubMed PMID: 18069916. [DOI] [PubMed] [Google Scholar]
- 30.Nemoto S, Fergusson MM, Finkel T. SIRT1 Functionally Interacts with the Metabolic Regulator and Transcriptional Coactivator PGC-1a. J Biol Chem. 2005;280:16456–16460. doi: 10.1074/jbc.M501485200. 10.1074/jbc.M501485200 PubMed PMID: 15716268. [DOI] [PubMed] [Google Scholar]
- 31.Guarente L. Calorie restriction and sirtuins revisited. Genes Dev. 2013;27:2072–2085. doi: 10.1101/gad.227439.113. 10.1101/gad.227439.113 PubMed PMID: 24115767, PMCID 3850092. [DOI] [PMC free article] [PubMed] [Google Scholar]


