Abstract
Background
Robot-assisted pancreaticoduodenectomy (RPD) has been gaining attention for its potential benefits in short-term surgical outcomes compared with open pancreaticoduodenectomy (OPD) in pancreatic cancer. However, the evidence of its long-term oncological efficacy is limited.
Methods
This retrospective study compared the long-term outcomes of RPD and OPD in patients with pancreatic cancer at Seoul National University Hospital between January 2015 and October 2023. Patients with stage III or IV disease and those who underwent open surgery were excluded. Propensity score matching (PSM) at a 1:2 ratio was performed based on sex, age, the American Society of Anesthesiologists (ASA) class, and initial resectability. The primary outcomes were overall survival (OS) and disease-free survival (DFS), and the secondary outcomes were postoperative recovery and complication rates.
Results
In total, 522 patients (82 RPD and 440 OPD) were reviewed, and 82 RPD and 164 matched OPD patients were analyzed after PSM. The mean age was 64.8 years in the RPD and 65.2 years in the matched OPD group; there were 51.2% male patients in the RPD and 52.4% in the OPD groups. After PSM, patients with RPDs showed better OS and DFS (5-year OS 58.2% vs. 32.3%, P = 0.001; 5-year DFS: 44.6% vs. 24.7%, P = 0.005). The R0 resection rates and harvested lymph node (LN) number were also comparable (RPD versus OPD: R0 resection rate 92.7% versus 90.9%, P = 0.809; harvested LNs 20.0 ± 7.3 versus 23.1 ± 11.5, P = 0.090). Additionally, patients with RPD had a shorter postoperative recovery time and relatively higher adjuvant therapy completion rate, although the difference was only marginally significant (91.6% vs. 75.4%, P = 0.062).
Conclusion
RPD is a feasible alternative to OPD, with potential advantages of early recovery without compromising long-term outcomes after PSM. However, further prospective studies are required to confirm these findings.
Graphical abstract
Keywords: Robot-assisted pancreaticoduodenectomy, Open pancreaticoduodenectomy, Pancreatic cancer, Long-term outcomes, Adjuvant therapy
Robot-assisted surgery is an emerging field of surgery and its applicability is expanding exponentially, driven by the evolution of robotic ergonomics and instruments. A meta-analysis reported that robotic abdominopelvic surgery offers several advantages over open surgery [1]. However, the application of robotic platforms to pancreaticoduodenectomy (PD) remains restricted owing to technical challenges. Moreover, in pancreatic cancer, the presence of pancreatitis, fibrosis, and vessel invasion significantly increases the surgical complexity. Consequently, robot-assisted PD (RPD) has primarily been performed in high-volume centers [2, 3].
However, several studies have reported an increasing number of robotic pancreatic surgeries from various centers and a growing body of postoperative outcome data with favorable results compared to open methods [2, 4–15]. Additionally, with advances in neoadjuvant therapy, the demand for pancreatic cancer surgery and expectations for minimally invasive techniques are growing.
Despite the merits of robotic surgery, there is still an ongoing controversy among oncologists regarding the feasibility of minimally invasive surgery, including robotic surgery, for cancer [16]. In line with this, current studies addressing RPD outcomes often include diverse inclusion rates for pancreatic cancer, ranging from 24–81%, lacking data focused exclusively on pancreatic cancer [5, 7, 10, 11, 15].
Most studies have focused on the short-term outcomes of RPDs, primarily addressing technical and perioperative recovery. While short-term outcomes of RPD and open surgery are comparable, data on long-term effects remain limited. This study aimed to compare the long-term oncological outcomes of RPD with those of open PD (OPD) for pancreatic cancer.
Materials and methods
Patients and selection criteria
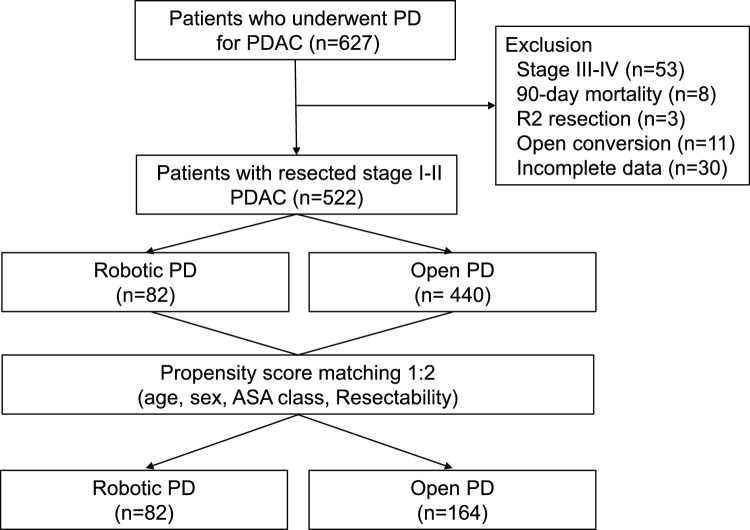
All patients who were diagnosed of pancreatic ductal adenocarcinoma (PDAC) and underwent RPD or OPD at Seoul National University Hospital (SNUH), Seoul, South Korea, between January 2015 and October 2023 were retrospectively reviewed in this study. To minimize selection bias, patients with pathological stage III or IV PDAC were excluded as surgeons might be more likely to prefer open surgery for advanced cancers, potentially confounding the comparison of survival outcomes. To clarify the classification of the operation method, 11 patients who were converted to open surgery during the operation were excluded. Also, 11 patients who underwent incomplete (R2) resection (n = 3), or within 90-day mortality (n = 8) were excluded. Thirty patients with incomplete or unavailable clinical data were excluded (Fig. 1). These patients either became lost to follow-up or transferred to other hospitals immediately after surgery, making it impossible to collect reliable information on recurrence or survival outcomes. This study was approved by the Institutional Review Board, which waived the requirement for obtaining informed consent from patients (IRB No.: 2407-114-1553).
Fig. 1.
Flow chart of patient selection
Data collection
Clinical data were collected through a retrospective review of electronic medical records. Baseline demographics, clinical and pathological data including tumor marker levels, tumor size, clinical stage classified by initial tumor resectability (resectable, borderline resectable, locally advanced, or metastatic), and pathological stage were collected. Other oncological treatment history data, including neoadjuvant or adjuvant therapy data, were also retrieved. The primary oncological outcomes were assessed based on the overall survival (OS) and disease-free survival (DFS). OS was defined as the time from the date of surgery to the date of death from any cause, whereas DFS was defined as the time from the date of surgery to the date of the first documented recurrence or death during the follow-up period. Secondary outcomes were assessed based on perioperative and pathological outcomes, including the operation time, blood loss, number of retrieved lymph nodes, resection margin status, complication rate, and length of postoperative hospital stay. The completion rate of adjuvant therapy and the time from the date of surgery to the initiation of adjuvant therapy were also assessed. The time to first passage of flatus, removal of the surgical drain, discontinuation of fluid infusion, and removal of intravenous patient-controlled analgesia (IV PCA) were measured as postoperative functional recovery indicators.
Surgical procedures
All RPD and OPD were performed by the same team at SNUH using the same protocol among the surgeons. The detailed protocol was described in a previously published report from the same institution [17].
Statistical analysis
Propensity score matching (PSM) was performed at a 1:2 ratio for the RPD and OPD groups based on the sex, age, the American Society of Anesthesiologists (ASA) class, and initial resectability. Categorical variables were compared using the chi-square test or Fisher’s exact test, while continuous variables were analyzed using the Student's t-test or Mann–Whitney U test, as appropriate. Survival outcomes, including OS and DFS, were assessed using the survival analysis method with the Kaplan–Meier curve and compared using the log-rank test. Survival rates are presented with 95% confidence intervals (CI). Cox proportional hazards regression analysis was used to identify factors associated with survival, presenting hazard ratios (HR) with 95% confidence intervals (CI). Statistical significance was set at P < 0.05. All statistical analyses were performed using R software, version 4.4.1 (R Foundation for Statistical Computing).
Results
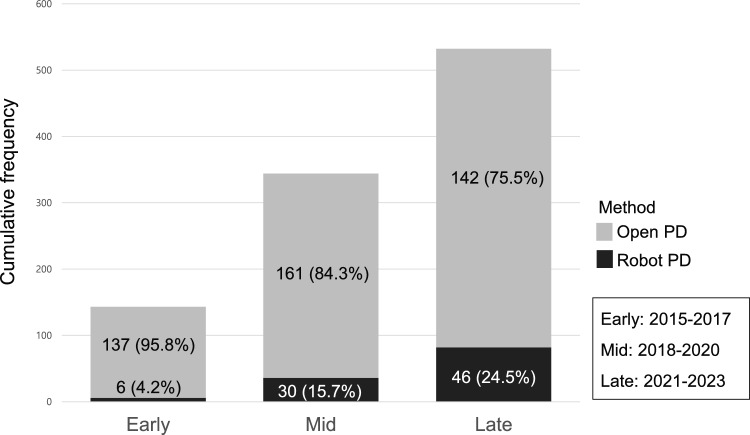
Of total 522 patients with PDAC who underwent either RPD or OPD, 82 underwent RPD and 440 underwent OPD. The two groups were matched in a 1:2 ratio, resulting in 82 RPD patients and 164 OPD patients for the final analysis (Fig. 1). The overall trends in the cumulative number of RPD and OPD performed over time at SNUH are shown in Fig. 2. The entire period was divided into three intervals of three years each: early period (2015–2017), mid-period (2018–2020), and late period (2021–2023). During this period, the number of RPDs performed was estimated, showing an increase in proportion from 4.2% in the early period to 24.5% in the late period.
Fig. 2.
Trends in the number of RPD and OPD cases for PDAC over time in SNUH. RPD Robot-assisted pancreaticoduodenectomy, OPD open pancreaticoduodenectomy, PDAC Pancreatic ductal adenocarcinoma, SNUH Seoul National University Hospital
Baseline characteristics
The baseline characteristics of the patients who underwent RPD and OPD, both before and after PSM, are summarized in Table 1. Mean age of RPD and OPD groups were 64.8 ± 10.9 years old and 64.6 ± 9.2 years old respectively, and male patients were 51.2% of RPD group and 55.9% in OPD group before PSM. The RPD and total OPD groups showed significant differences in ASA class distribution and initial carbohydrate antigen (CA) 19–9 (U/L, median [interquartile range], RPD 58.0 [9.0–299.0] versus OPD 110.7 [19.0–619.5]). Differences between the two groups before PSM were eliminated. The initial clinical stages differed between the two groups had different proportions (RPD versus OPD (before PSM), resectable 89.0% versus 30.5%, borderline resectable 8.5% versus 41.4%, locally advanced 2.4% versus 15.5%, and metastatic 0% versus 12.7%) (P < 0.001). After PSM, the proportions of clinical stages were matched between the two groups: resectable, 89.0% versus 81.7%; borderline resectable, 8.5% versus 15.9%; locally advanced, 2.4% versus 2.4%; and metastatic, 0.0% versus 0.0% (P = 0.316). In addition, pathological TNM stages were comparable after matching (P = 0.707). The OPD group had more patients who underwent neoadjuvant therapy (RPD 14.6% versus OPD (before PSM) 48.9%; (after PSM) 32.3%) and combined vessel resection during surgery (RPD 6.1% vs. OPD (before PSM) 29.5%; (after PSM) 25.0%). The proportions of cases per time period was 56.1% for RPD and 39.0% for OPD in the period of 2021–2023, and 43.9% for RPD and 61% for OPD in the period of 2015–2020 in the matched group.
Table 1.
Baseline demographics and clinicoathologic characteristics of RPD and OPD, before and after propensity score matching
| Before PSM | After PSM | |||||
|---|---|---|---|---|---|---|
| Variables | RPD (n = 82) | OPD (n = 440) | P-value | OPD (n = 164) | P-value | |
| Age (years), mean ± SD | 64.8 ± 10.9 | 64.6 ± 9.2 | 0.895 | 65.2 ± 9.6 | 0.958 | |
| Sex (male), n (%) | 42 (51.2) | 246 (55.9) | 0.507 | 86 (52.4) | 0.964 | |
| BMI (kg/m2) | 23.3 ± 7.1 | 22.9 ± 6.4 | 0.617 | 22.2 ± 6.1 | 0.351 | |
| ASA class, n (%) | I | 7 (8.5) | 49 (11.1) | 0.047* | 18 (11.0) | 0.317 |
| II | 66 (80.5) | 322(73.2) | 122 (74.4) | |||
| III | 9 (11.0) | 69 (15.7) | 24 (14.6) | |||
| Initial CA 19–9 (U/L), median [IQR] | 58.0 [9.0–299.0] | 110.7 [19.0–619.5] | 0.025* | 89.5 [21.0–575.0] | 0.083 | |
| Initial clinical stage, n (%) | R | 73 (89.0) | 134 (30.5) | < 0.001* | 134 (81.7) | 0.316 |
| BR | 7 (8.5) | 182 (41.4) | 26 (15.9) | |||
| LA | 2 (2.4) | 68 (15.5) | 4 (2.4) | |||
| M | 0 (0.0) | 56 (12.7) | 0 (0.0) | |||
| Tumor size (cm), mean ± SD | 2.5 ± 0.9 | 2.6 ± 0.8 | 0.106 | 2.6 ± 1.0 | 0.570 | |
| Pathologic stage, n (%) | 0 | 2 (2.4) | 18 (4.1) | 0.750 | 5 (3.0) | 0.707 |
| I | 38 (46.3) | 194 (44.1) | 66 (40.2) | |||
| II | 42 (51.2) | 228 (51.8) | 93 (56.7) | |||
| T classification, n (%) | T0 | 1 (1.2) | 17 (3.9) | 0.510 | 4 (2.4) | 0.871 |
| T1 | 21 (25.6) | 120 (27.3) | 41 (25.0) | |||
| T2 | 55 (67.1) | 267 (60.7) | 106 (64.6) | |||
| T3 | 5 (6.1) | 36 (8.0) | 13 (7.9) | |||
| N classification, n (%) | N0 | 43 (52.4) | 216 (49.1) | 0.202 | 68 (41.5) | 0.135 |
| N1 | 37 (45.1) | 189 (43.0) | 73 (44.5) | |||
| N2 | 2 (2.4) | 35 (18.0) | 23 (14.0) | |||
| Vessel resection, n (%) | 5 (6.1) | 130 (29.5) | < 0.001* | 41 (25.0) | < 0.001* | |
| Neoadjuvant therapy, n (%) | 12 (14.6) | 215 (48.9) | < 0.001* | 53 (32.3) | 0.005* | |
| Adjuvant therapy, n (%) | 72 (87.8) | 403 (91.6) | 0.208 | 130 (79.3) | 0.141 | |
ASA American Society of Anesthesiologists physical status classification system, CA 19–9 Carbohydrate antigen 19–9, R resectable, BR borderline resectable, LA locally advanced, M metastatic
*P-values in statistical significance (P < 0.05)
Survival analysis
Overall survival
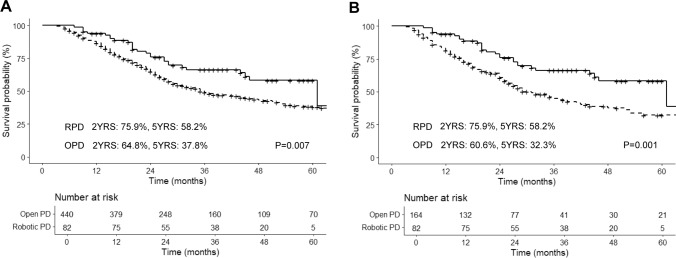
OS between the RPD and OPD group showed significant difference (RPD versus OPD, median OS 61 months versus 35 months; 5-year OS 58.2% (95% CI, 46.5–72.8%) versus 37.8% (95% CI, 32.8–43.5%), P = 0.007) (Fig. 3A). After PSM (Fig. 3B), the RPD group still showed a significantly better OS than the OPD group (RPD versus OPD, median OS 61 months versus 31 months; 5-year OS 58.2% (95% CI, 46.5–72.8%) versus 32.3% (95% CI, 24.3–42.9%), P = 0.001) (Fig. 3B).
Fig. 3.
Kaplan–Meier curves of overall survival for RPD and OPD before PSM (A), after PSM (B). PSM propensity score matching, RPD robot-assisted pancreaticoduodenectomy, OPD open pancreaticoduodenectomy
Disease-free survival
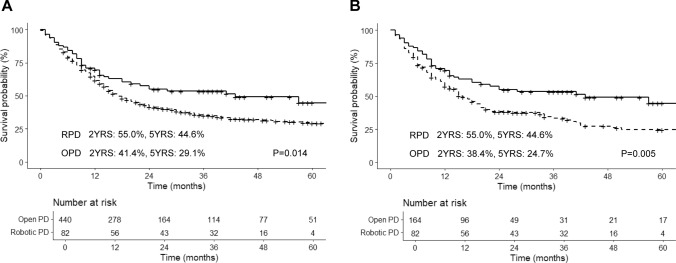
The RPD group also demonstrated a favorable DFS rate, with a median survival of 43 months (5-year DFS rate of 44.6% (95% CI, 32.9–60.4%). The OPD group showed a significant difference from the RPD group before PSM (median DFS 17 months; 5-year DFS 29.1% (95% CI, 24.7–34.4%); P = 0.014) (Fig. 4A). Similarly, after PSM (Fig. 4B), the RPD group showed better DFS compared to the OPD group (RPD versus OPD, median DFS 43 months versus 16 months; 5-year DFS 44.6% (95% CI, 32.9–60.4%) versus 24.7% (95% CI, 17.8–34.2%), P = 0.005) (Fig. 4B).
Fig. 4.
Kaplan–Meier curves of disease-free survival for RPD and OPD before PSM (A), after PSM (B). RPD robot-assisted pancreaticoduodenectomy, OPD open pancreaticoduodenectomy, PSM propensity score matching
Perioperative outcomes
Short-term perioperative outcomes were comparable between the RPD and OPD groups (Table 2). Before PSM, RPD had a longer operation time (RPD 331.9 ± 79.2 min versus OPD 295.6 ± 81.3 min, P < 0.001), and this difference persisted after PSM (331.9 ± 79.2 min versus 298.3 ± 86.2 min, P = 0.001). The number of retrieved lymph nodes was fewer in the RPD group than the OPD group before PSM (mean, RPD 20.0 ± 7.3 versus OPD 22.0 ± 10.6, P = 0.039), but this difference was no longer significant after PSM (RPD 20.0 ± 7.3 versus OPD 23.1 ± 11.5, P = 0.090). Blood loss and R0 resection rates were comparable between the two groups, both before and after PSM. Furthermore, the overall complication rate (RPD 41.5% versus OPD (before PSM) 41.6%, P = 1.000; (after PSM) 40.2%, P = 0.963), and severe complication rate, defined as Clavien-Dindo classification ≥ 3 (RPD 14.6% versus OPD (before PSM) 13.2%, P = 0.859; (after PSM) 11.6%, P = 0.634), showed no significant differences. This finding was consistent across major complications, including clinically relevant postoperative pancreatic fistula, delayed gastric emptying, post-pancreatectomy hemorrhage, biliary fistula, and wound complications.
Table 2.
Perioperative outcomes of RPD and OPD, before and after propensity score matching
| Before PSM | After PSM | |||||
|---|---|---|---|---|---|---|
| Variables | RPD (n = 82) | OPD (n = 440) | P-value | OPD (n = 164) | P-value | |
| Operation time (min), mean ± SD | 331.9 ± 79.2 | 295.6 ± 81.3 | < 0.001* | 298.3 ± 86.2 | 0.001* | |
| Blood loss (mL), mean ± SD | 557.8 ± 587.2 | 587.4 ± 631.9 | 0.625 | 484.1 ± 488.6 | 0.761 | |
| Retrieved lymph node, mean ± SD | 20.0 ± 7.3 | 22.0 ± 10.6 | 0.039* | 23.1 ± 11.5 | 0.090 | |
| Resection margin, n (%) | R0 | 76 (92.7) | 386 (87.7) | 0.270 | 149 (90.9) | 0.809 |
| R1 | 6 (7.3) | 54 (12.3) | 15 (9.1) | |||
| Recurrence, n (%) | 36 (43.9) | 267 (60.7) | 0.007* | 102 (62.2) | 0.010* | |
| Overall complication, n (%) | 34 (41.5) | 183 (41.6) | 1.000 | 66 (40.2) | 0.963 | |
| Severe complication (CD ≥ 3), n (%) | 12 (14.6) | 58 (13.2) | 0.859 | 19 (11.6) | 0.634 | |
| CR-POPF, n (%) | 5 (6.1) | 20 (4.5) | 0.763 | 6 (3.7) | 0.725 | |
| DGE, n (%) | 4 (4.9) | 17 (3.9) | 0.902 | 6 (3.7) | 0.735 | |
| PPH, n (%) | 2 (2.4) | 7 (1.6) | 0.937 | 2 (1.2) | 0.603 | |
| Biliary fistula, n (%) | 2 (2.4) | 7 (1.6) | 0.937 | 4 (2.4) | 1.000 | |
| Wound, n (%) | 0 (0.0) | 14 (3.2) | 0.260 | 3 (1.8) | 0.553 | |
| Time to flatus (day) | 3.8 ± 1.4 | 4.0 ± 1.6 | 0.441 | 4.2 ± 1.8 | 0.099 | |
| Time to drain removal (day) | 7.4 ± 6.2 | 9.1 ± 5.4 | 0.013* | 10.2 ± 7.2 | < 0.001* | |
| Time to no fluid infusion (day) | 6.3 ± 2.9 | 8.3 ± 6.4 | < 0.001* | 9.0 ± 5.1 | < 0.001* | |
| Time to IV PCA removal (day) | 4.5 ± 1.7 | 5.5 ± 17.7 | 0.260 | 4.7 ± 2.2 | 0.608 | |
| Hospital stays (day) | 9.6 ± 7.0 | 13.2 ± 10.6 | < 0.001* | 14.2 ± 11.5 | < 0.001* | |
| Adjuvant therapy completion, n (%) | 63 (87.5) | 290 (78.0) | 0.094 | 98 (75.4) | 0.062 | |
| Adjuvant therapy cessation, n (%) | 9 (12.5) | 82 (22.0) | 0.094 | 32 (24.6) | 0.062 | |
| Patient tolerance | 4 (5.6) | 51 (13.7) | 0.084 | 21 (16.2) | 0.049* | |
| Early disease progression | 5 (6.9) | 31 (8.3) | 0.873 | 11 (8.5) | 0.912 | |
| Op to adjuvant therapy (day) | 46.7 ± 16.2 | 50.4 ± 34.9 | 0.171 | 49.1 ± 15.8 | 0.099 | |
CD Clavien-Dindo classification grade, CR-POPF clinically relevant postoperative pancreatic fistula, DGE delayed gastric emptying, PPH postoperative pancreatic hemorrhage, IV PCA intravenous patient-controlled analgesia
*P-values in statistical significance (P < 0.05)
Postoperative functional recovery
For functional recovery indicators, the RPD showed shorter postoperative recovery time, in the time to drain removal (RPD 7.4 ± 6.2 days versus OPD (before PSM) 9.1 ± 5.4 days, P = 0.013; (after PSM) 10.2 ± 7.2 days, P < 0.001), the time to discontinuation of fluid infusion (RPD 6.3 ± 2.9 days versus OPD (before PSM) 8.3 ± 6.4 days, P < 0.001; (after PSM) 9.0 ± 5.1 days, P < 0.001, and length of hospital stay (RPD 9.6 ± 7.0 days versus OPD (before PSM) 13.2 ± 10.6 days, P < 0.001; (after PSM) 14.2 ± 11.5 days, P < 0.001), both before and after PSM.
Adjuvant therapy tolerance
After surgery, a comparable number of patients underwent adjuvant therapy in both groups (RPD 87.8% versus OPD (before PSM) 91.6%, P = 0.208; (after PSM) 79.3%, P = 0.141). In terms of the completion of adjuvant therapy, the RPD group had a completion rate of 87.5% compared with 75.4% in the OPD group after PSM (P = 0.062). The patients who could not complete adjuvant therapy were recategorized by the cause of cessation. A comparable proportion of patients with early disease progression during adjuvant therapy were converted to palliative settings (RPD 6.9% versus matched OPD 8.5%, P = 0.912). Notably, the number of patients who ceased adjuvant therapy due to deterioration in patient tolerance was estimated, and the RPD group showed significantly lower adjuvant therapy cessation than the matched OPD group (5.6% versus 16.2%, P = 0.049), despite no difference in the initial ASA status between the two groups. The time from surgery to initiation of adjuvant therapy was relatively short in both groups and showed no significant differences between the groups. These findings should affect the survival gap between RPD and OPD.
Prognostic factors for survival
Cox proportional hazards regression analysis was performed to identify prognostic factors for OS (Table 3). In the univariable analysis, elevated initial CA19-9 level (≥ 150 U/L) (HR 1.441 (95% CI 1.001–2.076, P = 0.049), positive nodal status (versus N0, HR 1.815 (95% CI 1.226–2.689, P = 0.003)), completion of adjuvant therapy (HR 0.381 (95% CI 0.242–0.601, P < 0.001)), and operation modality, whether robot-assisted or open (HR 0.503 (95% CI 0.328–0.771, P = 0.002) were associated with OS. In multivariate analysis, positive nodal status (versus N0, HR 2.003 (95% CI 1.221–3.286, P = 0.006)) and completion of adjuvant therapy (HR 0.415 (95% CI 0.261–0.660, P < 0.001)) were identified as significant prognostic factors for OS. The surgical modality was not significantly associated with OS in the multivariate analysis, with an HR of 0.750 (95% CI 0.461–1.220, P = 0.247).
Table 3.
Cox proportional hazards regression analysis for overall survival
| Univariable analysis | Multivariable analysis | ||||||
|---|---|---|---|---|---|---|---|
| Variables | HR | 95% CI | P-value | HR | 95% CI | P-value | |
| Age (year) | ≥ 70 | 1.267 | 0.860–1.865 | 0.231 | – | – | – |
| Sex | Female | 0.956 | 0.665–1.374 | 0.807 | – | – | – |
| Male | Reference | ||||||
| Initial CA 19–9, U/L | ≥ 150 | 1.441 | 1.001–2.076 | 0.049* | 1.159 | 0.750–1.791 | 0.507 |
| Clinical stage | LAPC | 2.701 | 0.991–7.365 | 0.052 | 1.255 | 0.170–9.251 | 0.824 |
| R/BR | Reference | ||||||
| T stage | T3,4 T1,2 | 1.369 Reference | 0.690–2.717 | 0.370 | – | – | – |
| N stage | N + N0 | 1.815 Reference | 1.226–2.689 | 0.003* | 2.003 | 1.221–3.286 | 0.006* |
| Resection margin | R1 R0 | 1.031 Reference | 0.519–2.045 | 0.931 | – | – | – |
| Harvested LNs number | ≥ 15 | 1.552 | 0.958–2.515 | 0.074 | 1.601 | 0.868–2.952 | 0.132 |
| Neoadjuvant therapy | Yes | 0.886 | 0.555–1.413 | 0.611 | – | – | – |
| Adjuvant completion | Yes | 0.381 | 0.242–0.601 | < 0.001* | 0.415 | 0.261–0.660 | < 0.001* |
| Operation modality |
Robot Open |
0.503 Reference |
0.328–0.771 | 0.002* | 0.750 | 0.461–1.220 | 0.247 |
CA 19–9 Carbohydrate antigen 19–9, LAPC locally advanced pancreatic cancer, R resectable, BR borderline resectable, LN lymph node
*P-values in statistical significance (P < 0.05)
Discussion
This study compared long-term oncological outcomes of RPD and OPD in patients diagnosed with pancreatic cancer. The RPD group demonstrated superior OS and DFS rates as compared to the open approach group for pancreatic cancer patients after stratification by age, sex, the ASA class, and clinical resectability. In addition, there were no significant differences between the two groups in terms of R0 resection, harvested LN numbers, and overall complication rates after PSM.
Our findings are consistent with those of previous studies that reported similar trends in survival outcomes between RPD and OPD. Wang SE et al. (2018) found a 2-year survival rate of 64.8% for RPD and 40.0% for OPD, while Shyr YM et al. (2021) reported a median survival of 15.3 months for RPD and 12.3 months for OPD in PDAC patients [18–20]. On the other hand, our study demonstrated generally better survival outcomes for both RPD and OPD groups compared to these reports. This discrepancy could be attributed to the more recent patient population in this study, the exclusion of advanced stages in the analysis, and the increased use of neoadjuvant and adjuvant chemotherapy protocols, which collectively improved the pancreatic cancer survival rates over time. Using the National Cancer Database (NCDB), Nassour et al. (2021) analyzed the outcomes of RPD and OPD in patients with pancreatic cancer who underwent neoadjuvant chemotherapy. They also reported similar rates of the positive margin status (RPD 21% versus OPD 16%), and a shorter length of hospital stay in RPD group (RPD 8 ± 5 versus OPD 10 ± 7, P < 0.001), findings that are consistent with our study. However, in survival analysis, they found no significant difference in the OS rates (median OS: RPD 25.6 months versus OPD 27.5 months, P = 0.879). This discrepancy should be interpreted with caution, considering the nature of the NCDB data, which aggregates information from diverse facilities. Notably, the proportion of patients treated at large-volume facilities, including academic/research programs and integrated network cancer programs, differed between the RPD (95%) and OPD (85%) groups. These variations in institutional composition may have influenced both surgical outcomes and OS results [21].
In the Cox regression analysis for OS, the surgical modality was not a relevant factor (HR 0.750, 95% CI 0.461–1.220). This implies that the robotic technique itself is not the actual effective variable for prognosis; rather, the sequentially affected variables are the true determinants of survival. Another independent prognostic factor for survival was the completion of adjuvant therapy (HR 0.415, 95% CI 0.261–0.660). The rate of discontinuation of adjuvant therapy owing to a deteriorating condition was lower in the RPD group (5.6% vs. 16.2%, P = 0.049). DePeralta et al. [22] reported that completion of adjuvant therapy was an independent prognostic factor for both recurrence-free survival (HR 0.52, 95% CI 0.359–0.755) and overall survival (HR 0.410, 95% CI 0.274–0.614) in pancreatic ductal adenocarcinoma, which is comparable to our analysis [22].
The major reasons for failure to initiate or continue adjuvant therapy were drug toxicity and poor functional status of the patients. In the RPD group, 4 patients did not initiate or discontinued adjuvant chemotherapy, due to poor performance status (n = 2) and patient preference (n = 2). In the OPD group, 52 patients withheld adjuvant chemotherapy for similar reasons, including poor performance status (n = 32) and patient preference (n = 20). Adverse effects leading to discontinuation of adjuvant therapy were more frequently reported in the OPD group.
Indicators of recovery from surgery, such as the time to drain removal, fluid infusion discontinuation, and hospital stay, were significantly shorter in the RPD group. Recently, two randomized trials comparing the short-term outcomes of RPD and OPD reported conflicting results [23, 24]. The trend of our study was consistent with findings from Liu et al. (2024). They reported that the RPD group had a shorter hospital stay (11.0 [9.0–20.0] versus 14.0 [12.0–21.5] days) and a faster postoperative recovery, including earlier time to first flatus (2.0 [1.0–2.0] versus 2.0 [2.0–3.0] days), and a shorter duration of analgesic use (6.0 [5.0–8.0] versus 8.0 [6.0–10.0] days) [23]. These may denote that robotic surgery may be associated with reduced surgical stress or mitigate immediate postoperative decline, potentially enabling patients to achieve tolerable conditions for adjuvant chemotherapy more quickly. The time to initiation of adjuvant therapy after surgery did not differ significantly between the two groups (RPD 46.7 vs. OPD 47.8 days, P = 0.878). Notably, previous studies have reported that the delayed initiation of adjuvant therapy is not associated with OS in patients with pancreatic cancer [25, 26]. The actual assessment for progression of postoperative physical decline should be further investigated in future studies to validate this hypothesis.
Conversely, another recent randomized study Klots et al. [24] reported that the RPD group was associated with a longer hospital stay and a higher incidence of major complications than the OPD group. However, they also observed a relatively high rate of overall complications (58.6%) and a clinically relevant POPF incidence (37.9%) in the RPD group [24]. Given these findings, it is important to consider the possible impact of the learning curve in the RPD when interpreting these results.
Besides, possible bias in initial patient selection should be carefully considered. Surgeons often tend to select patients with better health status and fewer comorbidities for robotic surgeries. Although the surgical groups were matched for sex, age, ASA class, and resectability, excluding stage III and IV cancers in this study, it remains challenging to retrospectively assess the baseline performance status at the time of surgery to evaluate bias. Also, the rate of combined vessel resection was varied between two groups, implying possible discrepancy in the clinical resectability between two groups. In addition, the RPD and OPD groups showed different proportions of neoadjuvant therapy (14.6% versus 48.9%, after matching 32.3%). This finding can affect the survival gap between groups. However, additional subgroup analysis of the neoadjuvant and non-neoadjuvant subgroups showed no specific differences in long-term survival outcomes between the RPD and OPD groups. As the proportion of RPD candidates receiving neoadjuvant therapy gradually increases over time, future studies should include more balanced population groups for analysis.
Another potential source of bias is the difference in perioperative management strategies. In South Korea, patients who undergo robotic surgery are often of higher socioeconomic status due to the substantial cost difference between robotic and conventional surgical approaches. This socioeconomic disparity may influence postoperative recovery and access to adjuvant treatment.
Furthermore, the time distribution between the two groups should be taken into account. A higher proportion of patients in the RPD group underwent surgery in more recent years (56.1% vs. 39.0%), which may reflect evolving treatment strategies, including increased use of neoadjuvant or adjuvant chemotherapy. These temporal differences could have contributed to improved survival outcomes in the RPD group.A limitation of our study is the potential patient selection bias, as the assignment of robotic surgery was influenced by the surgeon or the patients themselves, as discussed above. Additionally, the exclusion of patients with advanced-stage disease and intraoperative open conversion to minimize bias restricts the comparison of oncologic efficacy to early stage and clinically resectable pancreatic cancer. The limitations of this retrospective study should be considered. These limitations should be considered further and analyzed in prospective settings in future studies.
A potential limitation is residual confounding from unmatched factors such as preoperative CA19-9 and nodal stage. These were not included in the matching process, as no significant between-group differences were observed after matching. However, their clinical importance is acknowledged, and future studies should consider incorporating such variables for more robust adjustment.
Conclusion
In conclusion, this study suggests that RPD is a feasible alternative to OPD, offering certain short-term postoperative advantages without compromising long-term oncological outcomes in pancreatic cancer. The observed trend towards better outcomes in the RPD group, coupled with its higher adjuvant therapy completion rate, underscores the potential oncological benefits of this minimally invasive approach. Further prospective multicenter studies with balanced populations are needed to validate these findings and optimize patient selection criteria for robot-assisted pancreatic surgery for pancreatic cancer.
Funding
Open Access funding enabled and organized by Seoul National University. This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute IKHIDI), funded by the Ministry of Health & Wel-fare, Republic of Korea (RS-2023-KH140186).
Data availability
All data analyzed during this study are included in this article (and its supplementary material) as references to published articles or are available from the corresponding authors on reasonable request.
Declarations
Disclosures
Dr. Seo, Dr. Jung, Mrs.Han, Dr. Lee, Dr. Choi, Dr. Chae, Dr. Yun, Dr. Cho, Prof. Kwon, Prof. Park, Prof. Jang have no conflicts of interest or financial ties to disclose.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Younsoo Seo and Hye-Sol Jung have contributed equally to this study.
References
- 1.Rivero-Moreno Y, Echevarria S, Vidal-Valderrama C, Pianetti L, Cordova-Guilarte J, Navarro-Gonzalez J, Acevedo-Rodríguez J, Dorado-Avila G, Osorio-Romero L, Chavez-Campos C, Acero-Alvarracín K (2023) Robotic surgery: a comprehensive review of the literature and current trends. Cureus 15:e42370 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Shyr BS, Shyr YM, Chen SC, Wang SE, Shyr BU (2024) Reappraisal of surgical and survival outcomes of 500 consecutive cases of robotic pancreaticoduodenectomy. J Hepatobiliary Pancreat Sci 31:99–109 [DOI] [PubMed] [Google Scholar]
- 3.Napoli N, Kauffmann EF, Vistoli F, Amorese G, Boggi U (2021) State of the art of robotic pancreatoduodenectomy. Updates Surg 73:873–880 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baimas-George M, Watson M, Murphy KJ, Iannitti D, Baker E, Ocuin L, Vrochides D, Martinie JB (2020) Robotic pancreaticoduodenectomy may offer improved oncologic outcomes over open surgery: a propensity-matched single-institution study. Surg Endosc 34:3644–3649 [DOI] [PubMed] [Google Scholar]
- 5.Baker EH, Ross SW, Seshadri R, Swan RZ, Iannitti DA, Vrochides D, Martinie JB (2016) Robotic pancreaticoduodenectomy: comparison of complications and cost to the open approach. Int J Med Robot 12:554–560 [DOI] [PubMed] [Google Scholar]
- 6.Bao PQ, Mazirka PO, Watkins KT (2014) Retrospective comparison of robot-assisted minimally invasive versus open pancreaticoduodenectomy for periampullary neoplasms. J Gastrointest Surg 18:682–689 [DOI] [PubMed] [Google Scholar]
- 7.Buchs NC, Addeo P, Bianco FM, Ayloo S, Benedetti E, Giulianotti PC (2011) Robotic versus open pancreaticoduodenectomy: a comparative study at a single institution. World J Surg 35:2739–2746 [DOI] [PubMed] [Google Scholar]
- 8.Kostakis ID, Sran H, Uwechue R, Chandak P, Olsburgh J, Mamode N, Loukopoulos I, Kessaris N (2019) Comparison between robotic and laparoscopic or open anastomoses: a systematic review and meta-analysis. Robot Surg 6:27–40 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.McCarron FN, Vrochides D, Martinie JB (2023) Current progress in robotic hepatobiliary and pancreatic surgery at a high-volume center. Ann Gastroenterol Surg 7:863–870 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.McMillan MT, Zureikat AH, Hogg ME, Kowalsky SJ, Zeh HJ, Sprys MH, Vollmer CM Jr (2017) A propensity score-matched analysis of robotic vs open pancreatoduodenectomy on incidence of pancreatic fistula. JAMA Surg 152:327–335 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Park SE, Choi HJ, You YK, Hong TH (2021) Effectiveness and stability of robot-assisted anastomosis in minimally invasive pancreaticoduodenectomy. Ann Surg Treat Res 100:329–337 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ritschl PV, Miller HK, Hillebrandt K, Timmermann L, Felsenstein M, Benzing C, Globke B, Öllinger R, Schöning W, Schmelzle M, Pratschke J, Malinka T (2022) Feasibility of robotic-assisted pancreatic resection in patients with previous minor abdominal surgeries: a single-center experience of the first three years. BMC Surg 22:86 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Shyr YM, Wang SE, Chen SC, Shyr BU, Shyr BS (2021) Robotic pancreaticoduodenectomy for pancreatic head cancer and periampullary lesions. Ann Gastroenterol Surg 5:589–596 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Varley PR, Zenati MS, Klobuka A, Tobler J, Hamad A, Hogg ME, Zeh HJ 3rd, Borhani AA, Zureikat AH (2019) Does robotic pancreaticoduodenectomy improve outcomes in patients with high risk morphometric features compared to the open approach. HPB 21:695–701 [DOI] [PubMed] [Google Scholar]
- 15.Wang SE, Shyr BU, Chen SC, Shyr YM (2018) Comparison between robotic and open pancreaticoduodenectomy with modified Blumgart pancreaticojejunostomy: a propensity score-matched study. Surgery 164:1162–1167 [DOI] [PubMed] [Google Scholar]
- 16.Huo Q, Yang Q, Gorczynski RM, Li T (2019) Oncological minimally invasive surgery. J Oncol 2019:1903297 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kim HS, Han Y, Kang JS, Kim H, Kim JR, Kwon W, Kim SW, Jang JY (2018) Comparison of surgical outcomes between open and robot-assisted minimally invasive pancreaticoduodenectomy. J Hepatobiliary Pancreat Sci 25:142–149 [DOI] [PubMed] [Google Scholar]
- 18.Uesaka K, Boku N, Fukutomi A, Okamura Y, Konishi M, Matsumoto I, Kaneoka Y, Shimizu Y, Nakamori S, Sakamoto H, Morinaga S, Kainuma O, Imai K, Sata N, Hishinuma S, Ojima H, Yamaguchi R, Hirano S, Sudo T, Ohashi Y (2016) Adjuvant chemotherapy of S-1 versus gemcitabine for resected pancreatic cancer: a phase 3, open-label, randomised, non-inferiority trial (JASPAC 01). Lancet 388:248–257 [DOI] [PubMed] [Google Scholar]
- 19.Conroy T, Hammel P, Hebbar M, Ben Abdelghani M, Wei AC, Raoul JL, Choné L, Francois E, Artru P, Biagi JJ, Lecomte T, Assenat E, Faroux R, Ychou M, Volet J, Sauvanet A, Breysacher G, Di Fiore F, Cripps C, Kavan P, Texereau P, Bouhier-Leporrier K, Khemissa-Akouz F, Legoux JL, Juzyna B, Gourgou S, O’Callaghan CJ, Jouffroy-Zeller C, Rat P, Malka D, Castan F, Bachet JB (2018) FOLFIRINOX or gemcitabine as adjuvant therapy for pancreatic cancer. N Engl J Med 379:2395–2406 [DOI] [PubMed] [Google Scholar]
- 20.Versteijne E, Suker M, Groothuis K, Akkermans-Vogelaar JM, Besselink MG, Bonsing BA, Buijsen J, Busch OR, Creemers GM, van Dam RM, Eskens F, Festen S, de Groot JWB, Groot Koerkamp B, de Hingh IH, Homs MYV, van Hooft JE, Kerver ED, Luelmo SAC, Neelis KJ, Nuyttens J, Paardekooper G, Patijn GA, van der Sangen MJC, de Vos-Geelen J, Wilmink JW, Zwinderman AH, Punt CJ, van Eijck CH, van Tienhoven G (2020) Preoperative chemoradiotherapy versus immediate surgery for resectable and borderline resectable pancreatic cancer: results of the dutch randomized phase III PREOPANC trial. J Clin Oncol 38:1763–1773 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Nassour I, Tohme S, Hoehn R, Adam MA, Zureikat AH, Alessandro P (2021) Safety and oncologic efficacy of robotic compared to open pancreaticoduodenectomy after neoadjuvant chemotherapy for pancreatic cancer. Surg Endosc 35:2248–2254 [DOI] [PubMed] [Google Scholar]
- 22.DePeralta DK, Ogami T, Zhou JM, Schell MJ, Powers BD, Hodul PJ, Malafa MP, Fleming JB (2020) Completion of adjuvant therapy in patients with resected pancreatic cancer. HPB 22:241–248 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Liu Q, Li M, Gao Y, Jiang T, Han B, Zhao G, Lin C, Lau WY, Zhao Z, Liu R (2024) Effect of robotic versus open pancreaticoduodenectomy on postoperative length of hospital stay and complications for pancreatic head or periampullary tumours: a multicentre, open-label randomised controlled trial. Lancet Gastroenterol Hepatol 9:428–437 [DOI] [PubMed] [Google Scholar]
- 24.Klotz R, Mihaljevic AL, Kulu Y, Sander A, Klose C, Behnisch R, Joos MC, Kalkum E, Nickel F, Knebel P, Pianka F, Diener MK, Büchler MW, Hackert T (2024) Robotic versus open partial pancreatoduodenectomy (EUROPA): a randomised controlled stage 2b trial. Lancet Reg Health Eur 39:100864 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Valle JW, Palmer D, Jackson R, Cox T, Neoptolemos JP, Ghaneh P, Rawcliffe CL, Bassi C, Stocken DD, Cunningham D, O’Reilly D, Goldstein D, Robinson BA, Karapetis C, Scarfe A, Lacaine F, Sand J, Izbicki JR, Mayerle J, Dervenis C, Oláh A, Butturini G, Lind PA, Middleton MR, Anthoney A, Sumpter K, Carter R, Büchler MW (2014) Optimal duration and timing of adjuvant chemotherapy after definitive surgery for ductal adenocarcinoma of the pancreas: ongoing lessons from the ESPAC-3 study. J Clin Oncol 32:504–512 [DOI] [PubMed] [Google Scholar]
- 26.Xia BT, Ahmad SA, Al Humaidi AH, Hanseman DJ, Ethun CG, Maithel SK, Kooby DA, Salem A, Cho CS, Weber SM, Stocker SJ, Talamonti MS, Bentrem DJ, Abbott DE (2017) Time to initiation of adjuvant chemotherapy in pancreas cancer: a multi-institutional experience. Ann Surg Oncol 24:2770–2776 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data analyzed during this study are included in this article (and its supplementary material) as references to published articles or are available from the corresponding authors on reasonable request.