Abstract
Background
Alzheimer’s disease (AD) is the predominant age-related neurodegenerative condition, characterised by the gradual and irreversible loss of neurons. Key pathological features include amyloid plaques and neurofibrillary tangles, which trigger a chronic inflammatory response in the brain, leading to microglial activation and proliferation.
Purpose
This study evaluated the neuroprotective potential of diosgenin (DGN) and pterostilbene, two phytoconstituents with known antioxidant and anti-inflammatory properties, in amyloid-β 1-42 exposed SH-SY5Y cells.
Methods
Human neuroblastoma cells were cultured and neurodifferentiated with retinoic acid, then exposed to amyloid-β 1-42 to simulate the AD model. Treatments included DGN (1.5 µM), pterostilbene (PTB) (1.5 µM), their combination (0.25 µM and 0.5 µM), and donepezil (1.2 µM) as a standard drug for comparison. The effects of treatments were assessed through cell viability, reactive oxygen species (ROS) levels, apoptosis, inflammatory cytokines, and BDNF levels using various assays, including flow cytometry, ELISA, Western blotting, and inhibitory assays for NOS, H2O2-mediated oxidative stress, DPPH, AChE, and β-secretase.
Results
DGN and PTB combination indicated increased cell viability, reduced microglial activation, decreased apoptosis, and lower ROS levels, with the maximum effect observed in the combination group (0.5 µM). Combination treatments also showed maximum inhibition in various assays and reduced levels of cytokines while upregulating BDNF, highlighting their neuroprotective, anti-inflammatory, and antioxidant activities.
Conclusion
The findings suggest that the combination of DGN and PTB may serve as an effective neuroinflammatory modulator in managing neurodegenerative diseases.
Keywords: Alzheimer’s disease, neuroinflammation, neuroprotection, diosgenin, pterostilbene
Neurodegenerative disorders are a major health concern in geriatrics, affecting nearly 15% of the global population. These are characterised by progressive neuronal damage with loss of neuronal function in the central nervous system (CNS), leading to cognitive decline. Abnormal protein aggregation activating inflammatory pathways is a characteristic feature observed in most prototypical neurodegenerative diseases, such as Alzheimer’s disease (AD), amylotrophic lateral sclerosis, Parkinson’s disease, etc. Astrocytes and microglial cells present in the CNS are equivalent to monocytes and microphages, protecting neurons against infection and toxic deposits. Inflammation is the first response to early neuronal degeneration, resulting in microglial activation that regulates repair of tissues. Microglial activation initially acts as a first line of defence, controlling tissue damage. However, an excessive activation over a prolonged period leads to chronic neuroinflammation progressing to neurodegeneration as seen in AD.1–7
According to the Alzheimer’s Association 2016, AD is the most prevalent neurological disorder affecting 50 million people throughout the world. Extracellular amyloid-β deposition and presence of hyperphosphorylated tau are the hallmarks of AD pathogenesis. Other associated features are increased production of inflammatory mediators, interleukins, cytokines, reactive oxygen species (ROS), Nitric oxide (NO), tumour necrosis factor-alpha (TNF-α), decreased acetylcholine levels due to cholionergic cell death. Their presence around the amyloid-β deposits and tau tangles implicates neuroinflammation and oxidative stress as primary mechanisms in AD pathogenesis.8–13
Despite considerable research, currently available treatments for AD are symptomatic. Approved therapies such as cholinesterase inhibitors merely counterbalance the fluctuating neurotransmitter levels.14 Therefore, focusing on multiple pathways, including neuroinflammation and employing antioxidant strategies, might prove beneficial in arresting the advancement of the disease. Extensive research revealed the anti-inflammatory nature of diosgenin (DGN) and pterostilbene in multiple models. Through up-regulation of antioxidant enzymes, they also demonstrated antioxidant activity.15, 16
Many modern pharmaceuticals trace their origin to plant-based sources, highlighting the significant contribution of herbal remedies to major pharmacopoeias. Ongoing research continues for herbal therapies to address numerous illnesses, particularly neurological disorders, where effective treatment options remain limited. Additionally, people consider herbal options due to their personal beliefs and perceived lesser toxic effects.17, 18 Natural products and their isolated compounds are a significant source of drug discovery in the pursuit of more effective treatment for AD. Galantamine, an often-used cholinesterase inhibitor, is a natural product, which signifies the potential of natural sources. Selectivity of isolated phytochemicals for the brain receptors and their anti-inflammatory and antioxidant nature establishes a scientific basis for the use of medicinal plants in both animal and human studies. 19 Moreover, the use of herbal medication has become widespread due to easy availability, safety, efficacy and cost-effectiveness. 20
DGN, a phytochemical found in Dioscorea species, Trigonella foenum gracecum and Smilax species. It is highly valued and investigated in neurological disorders because of its ability to cross the BBB. It is also a source from which numerous therapeutically active drugs are derived. Its hypoglycaemic, anti-inflammatory, hypolipidemic, anti-proliferative and potent antioxidant effects are demonstrated in several studies.21–27 Another reported phytocompound, pterostilbene (PTB), is a naturally occurring stilbenoid found in blueberries. Its high oral bioavailability and potent anti-inflammatory activity render it a probable drug candidate. PTB also demonstrated Nrf2 (nuclear erythroid related factor) mediated antioxidant response induced neuroprotection.28–31
In vivo studies utilising various animal models to investigate novel therapies are the most preferred form of research. Despite being a stronger research tool to study AD pathogenesis, they may fail to accurately simulate the human brain’s response and environment, due to the difference in the genetic and physical make-up of the human and rodent brains. Therefore, the human cell line-derived in vitro model may recapitulate the conditions of the human brain. Furthermore, the use of animals in research on a regular basis raises ethical issues and study costs. The approach also restricts the overuse of lab animals in the process of developing new drugs. Hence, these provide better translational values to animal data, making it a more reliable and effective approach in drug development.
Cell lines cultured from human brain astrocytes, neuronal cells from AD-affected rats and brain endothelial cells were used predominantly. Based on this, we explored the effect of individual and combination of DGN and PTB in various in vitro models. Amyloid β-exposed SH-SY5Y cells were given the phytocompounds treatment, and their effects in suppression of pro-inflammatory mediators production and neuroinflammation-mediated neurotoxicity were evaluated.
Materials and Methods
SH-SY5Y cell lines were outsourced from Pune’s National Centre for Cell Science. Aβ1-42, Retinoic acid (RA), phosphate buffered saline (PBS) (10X)-pH 7.2, TRIZOL and MTT were bought from Sigma Aldrich. Dulbecco’s Modified Essential Medium (DMEM) (AL066A) was obtained from HI Media. Fetal Calf Serum-qualified, Brazil (10270106), Flow Cytometry Annexin V containing Apoptosis Kits (V13242), 10,000 U/ml of Penicillin-Streptomycin - (15140122), CM-H2DCFDA (General Oxidative Stress Indicator) - (C6827) and 96, 6 and 24 well plates, Corning (Corning nunc) from Thermofisher. Human Interleukin-6 ELISA Kit (Catalogue #ab178013), Human IL-1beta Matched Antibody Pair Kit (Catalogue #ab217608) and Human TNF alpha ELISA Kit (Catalogue #ab181421) from Abcam (United States).
Cell Culture
The DMEM:F12 medium (HI media) supplemented with 2 mM L-glutamine, 20 mg/ml streptomycin, 20 units/ml penicillin and 10% (vol/vol) heat-inactivated foetal bovine serum was used to incubate the SH-SY5Y cell line. The incubator allowed the flask to be placed in valve position with 37 °C incubation and 5% CO2. Cells were cultivated at 10 5 cells/cm 2 in flasks coated with 0.05 mg/ml collagen. 10 gm all-trans-RA in DMEM:F12 and foetal bovine serum media were added one day after the cells were plated. After 11 days, the cells were rinsed three times and cultured in DMEM (F12 media supplemented with RA) with 50 ng/ml BDNF added and incubated for three more days. Following RA application, a cell cycle test was used, flow cytometric analysis (The CyFlow@ Cube6, Germany) to validate differentiation.32, 33
Toxicity Model
96-well plates were seeded with SH-SY5Y differentiated neuroblastoma cells at 10 5 cells per well. In order to determine the IC50 value during a 24-hour period, the cell cultures were administered β-amyloid 1-42 dissolved in 0.1% DMSO in various concentration ranges between 3.125 µM and 200 µM (to find the lowest concentration at which cell cultures remain toxic-free). After 24 hours of incubation with amyloid-β 1-42 peptides, its effects against toxicity were evaluated. Triton X was utilised as a reference standard, and each sample was prepared in triplicate for the various treatment groups. 34
Toxic Effects of DGN and PTB on SH-SY5Y Cells
The cellular toxicity of donepezil (DPZ), DGN and PTB was evaluated through cell viability by assessing their effect in the MTT assay and determining their IC50 values. SH-SY5Y cells were subjected to different concentrations of DPZ, DGN and PTB (250 & 500 nM, 1, 2, 5 & 10 µM) for 24 h. Then, each culture well was added MTT (20 µl, 0.5 mg/ml) and incubated at 37 ℃ for 4 h. The formazon product was dissolved by adding 150 µl of DMSO after the medium was carefully removed. Fluorescence in culture was measured at 570 nm with a spectroscopic plate reader.
Experimental Design
AD cell model was created using Aβ1-42 (10 µM). Aβ1-42 was dissolved in 0.1% DMSO-containing saline (0.22 nmol/µl). This was added to cells cultured in a water bath for seven days at 37 °C to yield an aggregated Aβ1-42 culture. These SH-SY5Y cells, growing logarithmically, were seeded in a 96-well plate, followed by a 24-hour incubation period. The cells were then divided into a control group (DMSO), Negative control group (Aβ1-42, 10 µM), Standard control group (DPZ, 1.2 µM), DGN (1.51 µM), PTB (1.51 µM), DGN and PTB (250 nM)—low dose combination group and DGN and PTB (500 nM)—high dose combination treatment group.
Cell Viability by MTT Assay
Using the MTT technique, the percentage of cell viability of DPZ (standard treatment), DGN and PTB treatments at various concentrations in Aβ1-42 treated SH-SY5Y cells was evaluated.35, 36 Treatments with compound DPZ, DGN, and PTB were carried out on fully differentiated cells exposed to 10 µM Aβ1-42 in normal media in 96-well plates. After 24 and 48 hours after treatment, MTT (20 µl, 0.5 mg/ml) with cultures was incubated at 37 °C for 2 hours and then lysed in 150 µl of DMSO, and the formazan produced was evaluated using a spectroscopic plate reader by measuring the absorbance at 570 nm.
MTT Data Correlation for Synergistic Effect of Combination
To test synergy for the combination of DGN and PTB, the MTT data correlation was carried out using the CompuSyn software. The computerised simulation allows the constant ratio combination to be represented as the median effect curves, dose-effect curves, CI plots and isobolograms. Statistical analysis was carried out to find out whether DGN acted synergistically with PTB. 37
Apoptosis Assay
The SH-SY5Y cells were seeded separately according to the experimental design for a 24-hour period in 6-well plates at 37 °C with 5% CO2 and 95% humidity. Cell death was induced using the live/dead apoptosis detection kit (Thermofisher/Annexin V kit) in conformity with the manufacturer’s guidelines. After collection, cultured cells were washed with ice-cold phosphate buffer three times, suspended in 100 µl of DNA-binding solution, and then incubated at 4 °C for 5 minutes. Subsequently, 5 µl of Annexin V and 10 µl of PI were added to the cells and stained for 10 minutes. The Cyto-FLEX flow cytometer was used to analyse apoptosis via flow cytometry data (Beckman Cutler, Inc., USA). 38
Measurement of ROS by Flow Cytometry
For the detection and measurement of ROS levels, the experimental groups were sampled individually. The SH-SY5Y cells in each group were treated with trypsin, washed with phosphate buffer, and then added 10 µM dichloro dihydro fluorescein diacetate (DCFH-DA) and incubated for 15 min. Penetration of the esterified fluorescent probe H2DCFDA into the intracellular matrix of cells generates fluorescent dichlorofluorescein (DCF) due to ROS interaction. The green fluorescence was recorded with UV filters at 460 nm wavelength. A fluorescent microscope (Nikon TS-100 OLYMPUS, Japan) analysed the fluorescence intensities and captured the pictures. The fluorescent signal was measured by the Cyto-FLEX flow cytometer via flow cytometry data (Beckman Cutler, Inc., USA), and values were expressed in percentage of the fluorescence (ROS levels) in comparison to control group. 39
Acridine Orange/Ethidium Bromide Staining
Examination of the morphological changes in differentiated SH-SY5Y cells was done using fluorescent probes that were permeable to acridine orange (AO) and non-permeable to ethidium bromide (EtBr). The RA-SH-SY5Y cell line cultures in each experimental group, incubated individually at 37 °C for 24 hours with 5% CO2. Following two washes with PBS, the treated cells were stained with 1 µl of AO/EtBr solution (stock concentration 100 µg/ml). The excess dye was removed using warm PBS, after incubating the cells at room temperature for 20minutes. A fluorescent microscope (Nikon TS-100 OLYMPUS, Japan) was used to view and capture the morphology of the cells. 40
TNF-α Levels
The fully differentiated SH-SY5Y cells (1.5 × 10 5 cells/ml) exposed to 10 µM Aβ1-42 in normal media in 96-well plates were treated as per the experimental design separately for 24 hours. Following treatment, using the TNF-α (MAb11) FITC monoclonal antibody kit (Catalogue # MA5-44101; Invitrogen), the cytokine TNF-α levels were estimated in each treatment group in accordance with the manufacturer’s instructions. Briefly, 100 µl of cell suspension was treated with 4 µl of staining reagent and left at 25 °C for 15 minutes. A BD FACSCanto II flow cytometer was used for the flow-cytometry analysis. 10,000 cells were employed for every flow cytometer run. 40
Interleukin-1 Beta (IL-1β) Levels
SH-SY5Y cells (1.5 × 10 5 cells/ml) exposed to 10 µM Aβ1-42 in normal media were treated as per the experimental design separately for 24 hours, to which 250 µl of pre-cooled PBS was added, and the freeze-thaw procedure was repeated multiple times until the cells were completely lysed. The supernatants were then collected and kept for assay after the centrifugation of cells for 10 minutes at 1500×g at 2 °C–8 °C. As per the manufacturer’s instructions, the IL-1β ELISA kit (Cat#ab197742, Abcam) was used to estimate the intensity of IL-1β in the various treatment groups. Briefly, 50 µl of homogenate was poured into each well of a 96-well plate, followed by the addition of 50 µl of the antibody cocktail. The plate was then sealed and incubated at room temperature on a plate shaker for an hour at 400 rpm. Then it was washed with 350 µl of 1× washing buffer and inverted to remove excess liquid from the well using blotting paper. After adding 100 µl of TMB development mix to each well, it is incubated in the dark for 10 minutes at 400 rpm on a plate shaker. Followed by 100 µl of stop solution to each well and again shaken for 1 minute to ensure even distribution of the reagent. The absorbance was then measured at 450 nm using a microplate reader. 40
IL-6 Levels
The fully differentiated SH-SY5Y cells (1.5 × 10 5 cells/ml)exposed to 10 µM Aβ1-42 in normal media in a 96-well plate were treated as per the experimental design separately for 24 hours. Using an IL-6 recombinant (4H16L21) FITC monoclonal antibody kit (Invitrogen; Catalogue # 701028), the levels of the cytokine IL-6 were estimated in each of the treatment groups, according to the manufacturer’s instructions. Briefly, 100 µl of cell suspension was mixed with 10 µl of staining reagent, and the mixture was incubated at 25 °C for 15 minutes. A BD FACSCanto II flow cytometer was used for the flow-cytometry analysis. 10,000 cells were employed for every flow cytometer run. 40
Nitric Oxide Synthase Assay
The SH-SY5Y cells (1.5 × 10 5 cells/ml) exposed to 10 µM Aβ1-42 in normal media were treated as per the experimental design separately for 24 hours. Cells were homogenised completely using 200 µl of NOS lysis buffer and centrifuged at 10,000g, at 4 °C for 10 minutes. After collecting the supernatant and keeping it on ice, the cell lysate (30–60 µl) was taken and put in a 96-well plate, and the volume was adjusted to 57.5 µl/well using NOS assay buffer. The Nitric Oxide Synthase (NOS) Activity Assay Kit (Catalogue :# K2094; Bio Vision) was used to detect the release of NOS in the desired samples according to the manufacturer’s guidelines. Briefly, the sample wells were filled with 40 µl of reaction mix and 2.5 µl of 10 mM NOS co-factor 1 and mixed carefully. Following a 30-minute incubation at 37 °C, each well received 95 µl of NOS assay buffer and 5 µl of enhancer and was incubated for an additional 10 minutes at room temperature. After that, each well received 5 µl of enhancer and 95 µl of NOS assay solution. Finally, 50 µl of Griess reagent was added, and the plate was incubated for a further 10 minutes. A microplate reader operating in endpoint mode measured the absorbance at OD 540 nm. 41
H2O2-mediated Oxidative Stress in SH-SY5Y
After culturing for 24 hours in 96-well plates at a density of 1 × 10 4 cells/well, SH-SY5Y cells were treated with various treatments as per the experimental design for 6 hours, followed by treatment with 200 µM H2O2 for 24 hours for inducing neurotoxicity (Figures 3c and 3d). The Cell Counting Kit-8 (CCK-8) (KeyGEN Biotechnology Corporation, Nanjing, China) was used to assess cell viability. A microplate reader (Bio-Rad in Hercules, California, USA) measured the absorbance at 420 nm after 10 µl of CCK-8 was incubated in each well for two hours at 37 °C. At least three replications of each assay were performed. Expressed as percentage cell viability relative to the non-treated control. 42
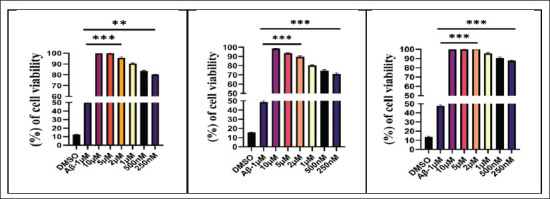
Figure 3. Represents Amyloid β Inhibitors DPZ, DGN and PTB, Reducing the Cytotoxicity Induced by Aβ1-42 in a Dose-dependent Manner, Compared to Control DMSO and Amyloid β-treated Group. Cytotoxicity was quantified using MTT dye reduction. Data were represented as a median ± SD standard deviation (n = 3; ns = not significant; **p < .01; ***p < .001 using one-way ANOVA Dunnett’s test.
Free Radical Scavenging Assay Using DPPH (2, 2-Diphenyl-1-Picrylhydrazyl)
To determine the free radical scavenging in the various selected treatment groups, DPPH in methanol solution (6 × 10−5 M) was prepared freshly and added individually in a 1:1 ratio to all groups, followed by overtaxing. It was carried out in the dark at room temperature. The standards used were Trolox and ascorbic acid. The absorbance of all the tubes was measured with a UV spectrophotometer at OD 517 nm.
The percentage inhibition for this assay was calculated using the equation as follows:
Inhibition (%) = [(A0 − A)/A0] × 100
where,
A0 - absorbance of DPPH without treatment.
A - absorbance of DPPH with treatment.
Percentage inhibition plots of sample solutions at different concentrations were used for estimation of IC50 values (The IC50 value is defined as the concentration required to scavenge 50% of the free radical). 43
Inhibition of Acetylcholinesterase (AChE) Activity
The fully differentiated SH-SY5Y cells (1.5 × 10 5 cells/ml) exposed to 10 µM Aβ1-42 in normal media in a 96-well plate, treated as per the experimental design separately, were resuspended in 100 µl of extraction buffer after being washed with cold PBS. Cells of each treatment group are homogenised individually in ice-cold PBS, pH 7.4, and centrifuged at 13,000 rpm, at 4 °C for 20 minutes. To make an AChE assay volume of 100 µl/well, each well of the treatment groups was added 50 µl of the acetylthiocholine reaction mixture. Protected from light, the incubation of the reaction mixtures lasted between 10 to 30 minutes. The absorbance was determined at 410 ± 5 nm using a microplate reader for absorbance. 44
Inhibition of β-secretase Activity
The fully differentiated SH-SY5Y cells (1.5 × 10 5 cells/ml) exposed to 10 µM Aβ1-42 in normal media in 96-well plates, treated as per the experimental design, are thawed on ice separately. Reconstituted in 100 µl of the extraction buffer after being washed with cold PBS. After a few up-and-down pipetting, the cells are rapidly homogenised and incubated on ice for 15 to 30 minutes. To remove insoluble debris, samples were centrifuged at 4 °C for 2–5 minutes using a cold microcentrifuge. After being collected, the supernatant was placed on ice in a clean tube. 50 µl of each treatment group was taken individually with 4 µl of reconstituted active β-secretase in the reaction wells, and added reaction buffer of 50 µl. To this, 2 µl of β-secretase substrate was added separately to all wells, covered, and incubated in the dark at 37 °C for an hour. Fluorescent microplate reader measured each plate at 335/495 nm. 45
Western Blotting Technique
The RIPA buffer cocktail extracted total protein from SH-SY5Y cells (1.5 × 10 5 cells/ml) separately from all treatment groups as per the experimental design. Total protein was extracted from SH-SY5Y cells (1.5 × 10 5 cells/ml) separately from all treatment groups as per the experimental design, using the RIPA buffer cocktail. Approximately 1 mg of sample was minced in the presence of disintegration buffer containing a protease concoction and stored at 4 °C for 30 min before centrifuging at 21,000 g for 10 min and collecting the extracted top-layer supernatant solution to estimate the total protein concentration using Bradford’s method. A total protein concentration of 20 g was utilised for sodium dodecyl, and those proteins were then converted onto a nitrocellulose membrane and incubated for 90 minutes in TBST buffer containing 3% bovine albumin (BSA). The obstructed membrane was left to incubate at 4 °C overnight with the reference proteins actin (1:000), BDNF (1:1000), Nf-kb (1:1000), and Nfr2 (1:1000). On day two, the membrane is washed three times at 10-minute intervals with TBST rinsing buffer, then the membranes is incubated at room temperature for 3 hours with a second antibody of goat anti-rabbit conjugates IgG-HRP (1:2000), and at last, the Blot was rinsed three times with TBST at 10-minute intervals; finally, TBST is washed, and 1 ml of Pierce ECL Western. The membrane had been covered with a blotting substrate (Cat No. 32106), and the protein bands were identified using the Box Syngene Gel Dock system, Frederick, MD, USA. 46
Statistical Analysis
The experiments were repeated three times, and each set of results was analysed individually using the ANOVA method by Prism (version 8.0.1) software. Data were represented as a median ± SD standard deviation (n = 3; ns = not significant; **p < .001; ***p < .001) using one-way ANOVA Dunnett’s test.
Results
SH-SY5Y Cell Neurodifferentiation by RA
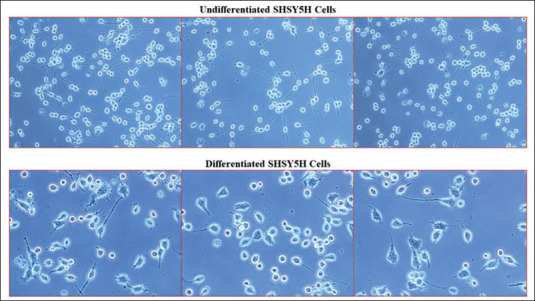
In comparison to control cells, SH-SY5Y neuroblastoma cells treated with 10 µM RA showed longer neurite development (Figure 1). Multiple neurite outgrowths and increased axon span were observed in the majority of the SH-SY5Y cells during morphological observation. Neurite connections between nearby cells were seen in more than 95% of the population of differentiated SH-SY5Y cells; in contrast, only 10% of the untreated control cells exhibited similar characteristics. The differentiated cells were therefore taken into consideration as an in vitro model for further experimentation to assess the neuroprotective effect.
Figure 1. Represents Retinoic Acid (RA) Induced Neuronal Differentiation and Morphological Changes in SH-SY5Y Cells. SH-SY5Y undifferentiated cells have shorter neurites. While neuronal-differentiated SH-SY5Y cells show RA-induced neurites with outgrowth.
Toxic Effects of DGN and PTB on SH-SY5Y Cells
Toxicity studies of DPZ, DGN and PTB on β-amyloid 1-42 exposed SH-SY5Y cells revealed concentrations up to 10 µM to be safe with no visible toxic effects or changes. The IC50 values for the treatments were calculated as represented in Figure 2 and Table 1.
Figure 2. Graphical Representation of the IC50 Values of the Amyloid β Inhibitors DPZ, DGN and PTB in Aβ1-42-treated SHSY5Y Cells.
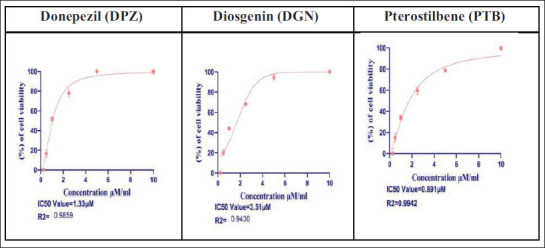
Table 1. IC50 Values of Amyloid Β Inhibitors DPZ, DGN and PTB.
| S.no | Treatments | IC50 Value |
| 1. | Donepezil (DPZ) | 1.33 ± 0.04 µM |
| 2. | Diosgenin (DGN) | 3.51 ± 0.26 µM |
| 3. | Pterostilbene (PTB) | 0.89 ± 0.81 µM |
Cell Viability by MTT Assay
This assay was carried out to assess the effect of DGN and PTB on SH-SY5Y cells’ viability and proliferation after Aβ 1-42 challenge. Our findings demonstrated that, 24 and 48 hours after treatment, cell viability was enhanced in a concentration-dependent way without appreciable alterations in cell shape (Figure 3 and Table 2).
Table 2. A, B and C Represent the Percentage Cell Viability for Donepezil (DPZ), Diosgenin (DGN) and Pterostilbene (PTB) Treatments at Various Concentrations in SHSY5H Cells, Respectively.
| a. Donepezil | ||
| Concentrations | Mean | SD |
| 10 µM | 100 | 0.45 |
| 5 | 100 | 0.34 |
| 2.5 | 95.64 | 0.78 |
| 1 | 90.45 | 0.41 |
| 0.5 | 83.45 | 0.78 |
| 0.25 | 80.14 | 0.21 |
| b. Diosgenin | ||
| Concentrations | Mean | SD |
| 10 | 100 | 0.45 |
| 5 | 93.65 | 0.49 |
| 2.5 | 89.65 | 0.93 |
| 1 | 80.41 | 0.54 |
| 0.5 | 74.65 | 0.94 |
| 0.25 | 71.05 | 0.65 |
| c. Pterostilbene | ||
| Concentrations | Mean | SD |
| 10 | 100 | 0.31 |
| 5 | 100 | 0.27 |
| 2.5 | 96.35 | 0.88 |
| 1 | 89.90 | 0.47 |
| 0.5 | 85.64 | 0.56 |
| 0.25 | 79.86 | 0.67 |
Combination Index-synergistic Effect of DGN and PTB
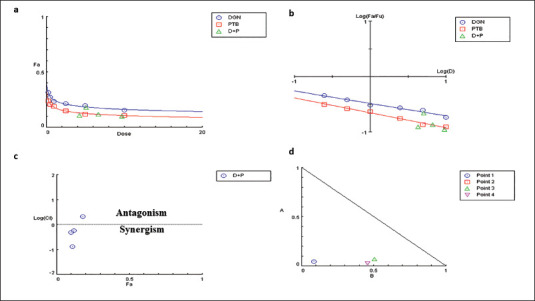
The MTT data correlation analysis using the CompuSyn software revealed that DGN and PTB were acting against SH-SY5Y cell lines in a concentration-dependent manner, both individually and in combination. The Dm (indicates the potency of the drug) of DGN in SH-SY5Y cells was 3.5 µM, and the Dm of PTB was 0.89 µM, respectively, in monotherapy, which was derived from the plots of the median effect (Figure 4a). The combination therapy of DGN and PTB achieved a Dm value of 0.00697 and 0.00354 for SH-SY5Y cells. Using the IC50 values, the interaction between DGN and PTB was determined as either antagonistic (CI > 1), additive (CI = 1), or synergistic (CI < 1). Chou and Talley defined CI as the total of the ratios between the individual and combinational doses of each medicine needed to achieve a specific Fa. It was derived from CI = 2.10611 for SH-SY5Y 24 hours with Fa = 0.5. Figure 4b represents the remaining SH-SY5Y cells’ CI vs. Fa data points. The curve displays a pattern of exponential decay. This revealed the potency of the synergism of DGN and PTB combination. The isobologram graphic additionally illustrates the shift in the combination of DGN and PTB doses in order to get Fa = 0.5, 0.75, and 0.9 (Figure 4c). By evaluating the slopes of the graphs, the effect of varying one drug’s dosage on another to maintain the Fa was determined. Their comparatively flat slopes (slopes close to 0) suggested that a small increase in the dose of DGN causes a significant drop in the PTB dose. CompuSyn was used to calculate all the curves and doses. DGN and PTB showed significant synergistic behaviour on SH-SY5Y cells at 24 hours, as per the combinational index study. Considering the 1:2 dilution of DGN with PTB, the DGN dose was around 3.5 µg and PTB 0.81 µg in SH-SY5Y cells, respectively. However, the findings show that the Dm value and IC50 of DGN and PTB were significantly reduced by ~4–8 fold in comparison with the single dose for SH-SY5Y cells, respectively. In addition, the exponential decay curve shows the potency of DGN, which is 20-fold and 28-fold higher than PTB, respectively. Furthermore, the exponential decay curve demonstrates the potency of DGN, which is 20 and 28 fold more potent than PTB, respectively (Figure 4d).
Figure 4. The CompuSyn Reports of Diosgenin (DGN) and (PTB) and Their Combinations at 24 Hours (a). The Relationship Between Fa and Dose in SHSY5Y Cells Is Demonstrated by the Dose-effect Curve. Dose-effect Curve Demonstrating Fa and Dose Relationship in SHSY5Y Cells (b), Median-effect (ME) Plot Illustrates Remapping of the Dosage Effect Curve to Get the IC50 Value. The Value Was Derived from the ME plot’s X-intercept. ‘Potency’ in SHSY5Y Cells Is Indicated by the ME Dosage at the IC50 Value. (c) Seven Combination Data Points Are Shown by the Combination Index Plot, of Which Two Depict an Additive Relationship and the Other Five Show a Synergistic Relationship (CI < 1). (d) Substantial Antagonism Was Evident in the Simulation at Low Fa. An isobologram with 50%, 75%, and 90% Inhibitions Is Displayed in SHSY5Y Cells. Diosgenin and Pterostilbene’s Data Points on the Diagonal Line Show Additive Effects, Synergistic Effects Are Shown on the Lower Left, and Antagonistic Effects Are Shown on the Upper Right. IC50, IC75, and IC90 in This Indicate Synergism.
Inhibitory Effect of DGN and PTB Combination in SH-SY5Y Cell Apoptosis Induced by Aβ1-42
In this investigation, the effect of apoptosis on Aβ1-42-treated SH-SY5Y cells following treatment with DGN, PTB, and their combinations was evaluated by flow cytometry and double staining with Annexin V-FITC/PI. Phosphatidylserine (PS) gets externalised in apoptotic cells, which gets stained and used as a marker of apoptosis. The apoptosis assay was performed in Aβ1-42 treated SH-SY5Y cells with seven different groups following treatment with DMSO, βamyloid, DPZ 1.2 µM, DGN 1.5 µM, PTB 1.5 µM, DGN+PTB 0.25 µM and DGN+PTB 0.5 µM. The effect of various selected treatment groups using flow cytometry to examine the effects of Aβ1-42 administration on SH-SY5Y cell cycle progression (Table 3). The control group had a percentage of 67.99% apoptosis with no viable cells, whereas the β amyloid group had the largest amount of apoptotic cells compared to the other treatment groups, with 87.32% of apoptotic cells with no viable cells. The group receiving PTB 1.5 µM/ml, DGN+PTB 0.25 µM/ml and DGN+PTB 0.5 µM/ml has significantly inhibited the apoptosis exhibiting neuroprotective activity, the apoptotic cells were found to be 23.71%, 22.98% and 3.17%, and viable cells were found to be 75.49%, 71.27% and 96.83%, respectively. The DPZ and DGN group showed lesser apoptosis compared to the β-amyloid group, percentage of apoptosis and viable cells were found as 35.15% & 62.95% and 61.31% & 24.04%, respectively (Figure 5). The apoptosis assay results of various selected treatment groups were tabulated as follows. The fluorescence intensity statistical analysis results revealed that the rate of apoptosis in SH-SY5Y cells treated with 10 µM Aβ1-42 for 24 hours was considerably higher (p < .01) than in the control group, with an apoptosis rate of 87.23%. The rate of apoptosis following treatment with 1.5 µM of PTB and DGN was 18.45% and 59.31%, respectively, and was statistically significant (p < .1) when compared to the disease model group. The apoptosis rate further decreased significantly (p < .01) when compared to the disease group in 0.25 µM and 0.5 µM DGN and PTB combination groups to 5.28% and 3.58%, respectively (Figure 6).
Table 3. Apoptosis Assay Results Using Flowcytometry Method by Annexin V-FITC/PI Detection Kit.
| Group | UL-Necrotised Cells Percentage | UR-Late Apoptotic Cells Percentage | LR-Early Apoptotic Cells Percentage | LL-Late Viable Cells Percentage |
| DMSO | 32.01 | 67.99 | 0 | 0 |
| β-Amyloid | 12.68 | 87.32 | 0 | 0 |
| DPZ 1.2 µM | 1.90 | 23.75 | 11.40 | 62.95 |
| DGN 1.5 µM | 14.65 | 57.83 | 3.48 | 24.04 |
| PTB 1.5 µM | 0.80 | 13.29 | 10.42 | 75.49 |
| DGN+PTB 250 nM | 5.76 | 3.21 | 19.77 | 71.27 |
| DGN+PTB 500 nM | 0 | 0.03 | 3.14 | 96.83 |
Figure 5. Representative Dot Plots for the Effect of Apoptosis on Aβ1-42 Treated SHSY5Y Cells Following Treatment with Diosgenin, Pterostilbene and Their Combinations. Flow cytometry and Annexin V-FITC/PI double staining were used to measure cell apoptosis.
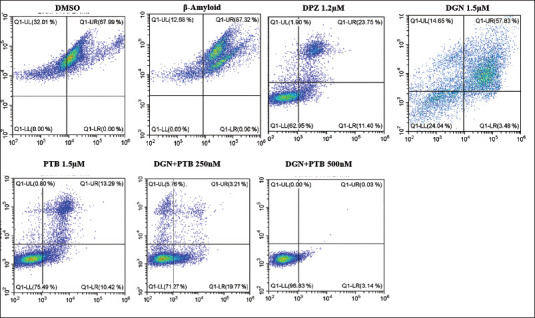
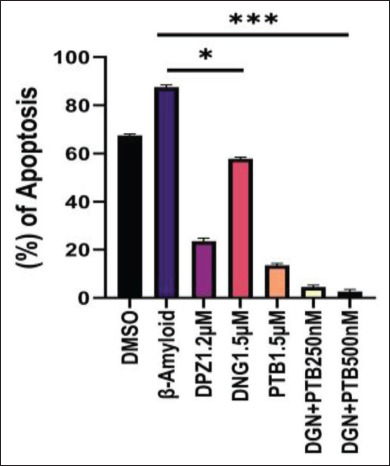
Figure 6. Represents Statistical Analysis of Flow Cytometry Results Quantification for Apoptosis Study in SHSY5Y Cells for the Following Groups: DMSO, Donepezil (DPZ), Diosgenin (DGN), Pterostilbene (PTB) and DGN+PTB Combination Groups. Data were represented as a median ± SD standard deviation (n = 3; ns = not significant; **p < .01; ***p < .001 using one-way ANOVA Dunnett’s test.
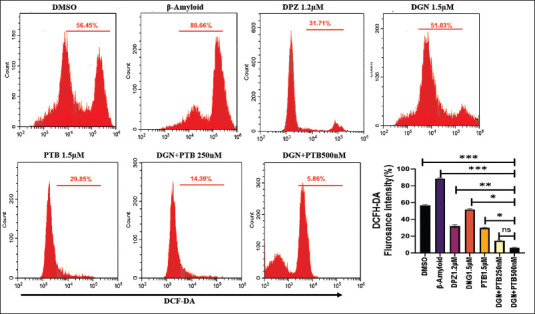
Flow Cytometric Analysis of ROS
The neuroprotective effects of the various treatment groups were evaluated by the measurement of the level of intracellular ROS determined by flow cytometry using the probe DCFH-DA (10−5 M). The results showed that the DCF probe’s fluorescence intensity in cells of the β-amyloid group was significantly greater than all the other groups, 88.66% with p < .01. The various treatments as per experimental design reduced ROS levels significantly in comparison to the control group. The ROS level in DGN (1.5 µM) was 51.83%, PTB (1.5 µM) was 29.85%, and 14.36% and 5.86% in DGN+ PTB (0.25 µM) and DGN+PTB (0.5 µM), respectively. The combination groups were found to be highly significant at ***p > .01 compared to standard treatment DPZ (1.2 µM) with ROS levels 31.76%. Therefore, the combination of DGN and PTB was proved to be the most effective in preventing the production of ROS, which in turn decreased the damage induced by amyloid-beta to neurons and protected the cells from degeneration (Figure 7).
Figure 7. Representative Images Showing ROS Expression in SH-SY5 Cells. The SHSY5H cells were dyed with H2DCFDA to detect ROS levels. SHSY5H cells from the control DMSO, Beta-amyloid, DPZ, DGN, PTB and DGN+PTB (0.25 and 0.5 µM)—supplemented IVM system. Comparative histograms showing a fold change relative to the background level of untreated cells [DC(−)], representing the mean fluorescence intensity of the DCF dye [DC(+)]. Data were represented as a median ± SD standard deviation (n = 3; ns = not significant; **p < .01; ***p < .001 using one-way ANOVA Dunnett’s test.
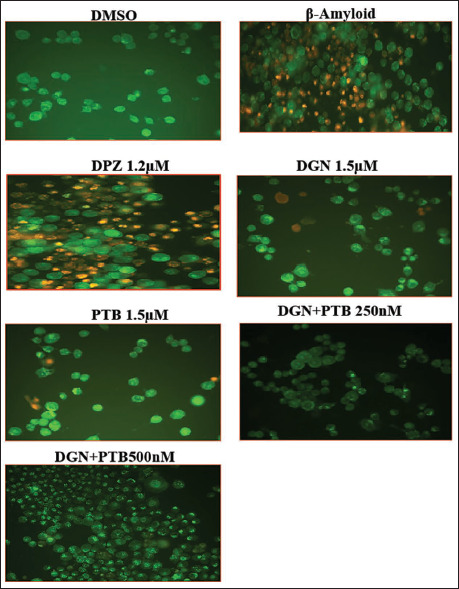
AO/EB Staining
To evaluate the treatment effect on morphological changes and cell apoptosis in SH-SY5Y cells, AO/EB staining was carried out in all the treatment groups as per the experimental design. In contrast to EtBr, which only stains live cells that have lost membrane integrity, AO stains both dead and living cells. Living cells thus appear green, while necrotic dead cells appear orange. After 24-hour treatment, the dead necrotised cells were found highly concentrated in the amyloid-beta group in comparison to all other groups. The two DGN+PTB combination groups, in a dose-dependent manner, showed highly concentrated green cells representing the decreased oxidative stress-mediated neuronal cell damage, signifying the neuroprotective effect of the novel combination group. The other treatment groups, DPZ, DGN and PTB, showed few early apoptotic cells with minimal morphological changes (Figure 8).
Figure 8. AO/EB Staining of SH-SY5Y Cell Culture Showing the Neuroprotective Effect of the Diosgenin and Pterostilbene Treatments by Decreasing the Amyloid-β Induced Glial Activation. (a) DMSO, (b) Amyloid-β, (c) Donepezil (DPZ), (d) Diosgenin (DGN), (e) Pterostilbene (PTB), (f) Diosgenin+Pterostilbene (DGN+PTB 250 nm), (g) Diosgenin+Pterostilbene (DGN+PTB 500 nm). Staining causes necrotic dead cells to appear orange and living cells to appear green.
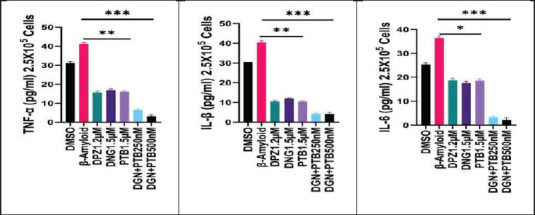
Effect of DGN and PTB Combination on Levels of TNF-α, IL-1β and IL-6
The effect of various treatments as per experimental design on amyloid-β pretreated SH-SY5Y cells was determined by ELISA. All treatment groups showed a significant decrease in cytokine levels when compared to the negative control group (the amyloid group). However, the reduction of various cytokines (TNF-α, IL-1β and IL-6) was maximum and highly significant (***p > .01) in the DGN+PTB combination groups, highlighting their anti-inflammatory neuroprotective nature (Figure 9).
Figure 9. Graphical Representation of the Effects of Diosgenin and Pterostilbene Treatments on Levels of Proinflammatory Mediators Determined by ELISA. Data were represented as a median ± SD standard deviation (n = 3; ns = not significant; **p < .01; ***p < .001 using one-way ANOVA Dunnett’s test.
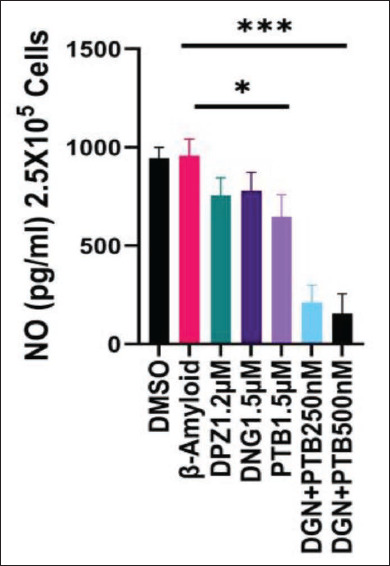
NOS Inhibitory Effect
The SH-SY5Y cells exposed to 10 µM Aβ1-42 were treated as per the experimental design separately for 24 hours, and when tested using the NOS Activity Assay Kit, revealed the NO inhibitory nature of the treatment groups. The inhibitory activity was highest in the DGN+PTB combination group, reported by low levels of NO, which was very significant (***p > .001) as shown in Figure 10.
Figure 10. Effect of Diosgenin and Pterostilbene Treatments on the Nitric Oxide Inhibitory Nature of the Treatment Groups. The inhibitory activity was highest in the diosgenin+pterostilbene combination group, reported by low levels of NO, which was very significant. Data were represented as a median ± SD standard deviation (n = 3; ns = not significant; **p < .01; ***p < .001 using one-way ANOVA Dunnett’s test.
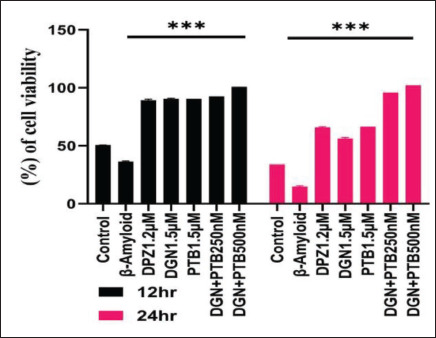
H2O2-mediated Oxidative Stress in SH-SY5Y
The H2O2-mediated oxidative stress assay was carried out to assess the effect of the DGN and PTB on SH-SY5Y cells’ viability and proliferation after Aβ 1-42 challenge after 6 hours and 24 hours. Our results highlighted that cell viability increased in PTB and the combination group significantly in comparison to the standard DPZ group (Figure 11).
Figure 11. Represents Percentage Cell Viability for DPZ, DGN and PTB Treatment in 10 µM Aβ1-42 Treated SHSY5Y Cells. The MTT dye reduction method was used to measure cytotoxicity. Data were represented as a median ± SD standard deviation (n = 3; ns = not significant; **p < .01; ***p < .001 using one-way ANOVA Dunnett’s test.
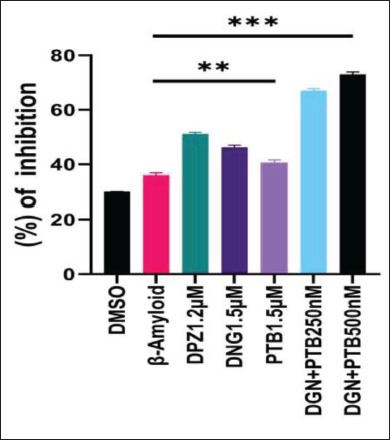
Antioxidant Activity by DPPH Assay
The DPPH radical scavenging activity was used to assess the antioxidant activity of the selected treatments. An effective way to evaluate the ability of natural antioxidants to scavenge free radicals is to employ DPPH, which is a stable free radical donor. Percentage inhibition in radical scavenging activity was significantly high in PTB and the combination group when compared to the standard DPZ group (Figure 12).
Figure 12. Percentage Inhibition of Oxidative Stress in 10 µM Aβ1-42 Treated SHSY5H Cells Given Diosgenin and Pterostilbene Treatment as per Experimental Groups. Data were represented as a median ± SD standard deviation (n = 3; ns = not significant; **p < .01; ***p < .001 using one-way ANOVA Dunnett’s test.
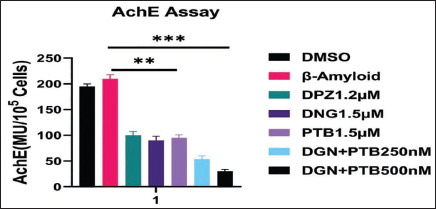
AChE Inhibitory Activity
Using Ellman’s method, the AChE inhibitory activity was assessed in SH-SY5Y cells exposed to Aβ1-42 and treated with test drugs as per the experimental design. All the tested compounds, DGN, PTB and combination groups, significantly inhibited the activity of AChE in the SH-SY5Y cell cultures (Figure 13). However, the maximum AChE inhibitory activity when compared with the control group was shown by the combination of diosgenin+pterostilbene 0.25 and 0.5 µM, which was very significant (***p > .01).
Figure 13. AChE Activity in SH-SY5Y Cells Treated as per Specified Treatment Groups Using Ellman’s Technique. Maximum significant inhibition in AChE activity was seen in the combination of diosgenin+pterostilbene 0.25 and 0.5 µM treatment groups in comparison to control (DMSO) and standard (Donepezil) treatment groups. Data were represented as a median ± SD standard deviation (n = 3; ns = not significant; **p < .01; ***p < .001 using one-way ANOVA Dunnett’s test.
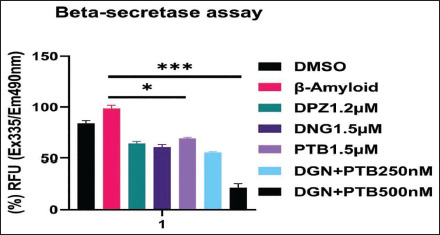
β-secretase Inhibitory Activity
The Aβ1-42 exposed SH-SY5Y cells pre-treated as per the experimental design separately for 24 hours, when tested with Beta-secretase Activity Assay Kit (ab65357), revealed a significant decrease in fluorescence intensity of the various treatment groups compared to the control group. The decrease in fluorescence intensity is representative of decreased β-secretase enzymatic activity in samples, indicating the reduction of amyloid β plaque formation. The decrease in fluorescence intensity was highly significant (***p > .001) in DGN+PTB (0.5 µM) combination group in comparison to the control group (Figure 14).
Figure 14. β-secretase Enzymatic Activity in SH-SY5Y Cells Treated as per Specified Treatment Groups Revealed a Significant Decrease in Fluorescence Intensity of the Treatment Groups Compared to Control Groups, Indicative of Decreased β-secretase Enzymatic Activity of Samples, Indicating the Reduction of Amyloid β Plaque Formation. The decrease in fluorescence intensity was highly significant ( ***p > .01) in the DGN+PTB (0.5 µM) combination group when compared to the control group. Data were represented as a median ± SD standard deviation (n = 3; ns = not significant; **p < .01; ***p < .001 using one-way ANOVA Dunnett’s test.
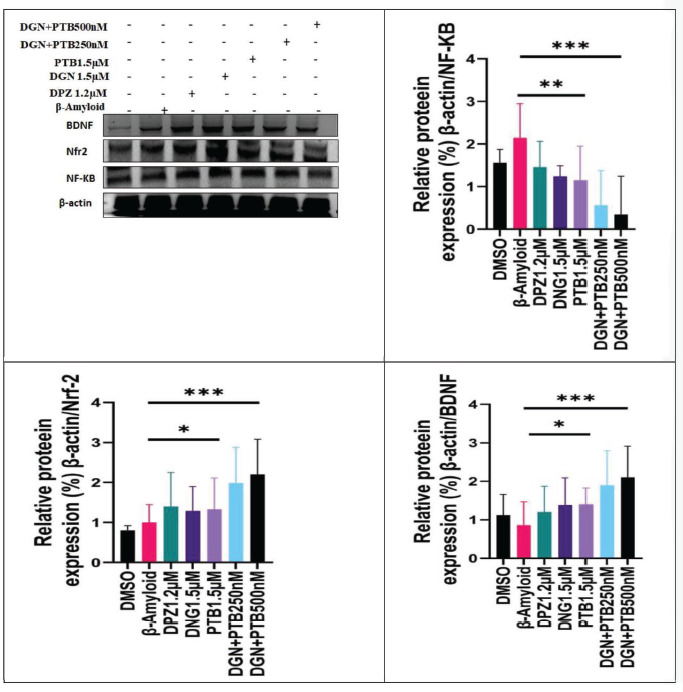
NF-KB, Nrf2 and BDNF levels by Western Blot in the SH-SY5Y Cells
The levels of proteins NF-KB, Nrf2 and BDNF were determined by western blotting. All treatment groups had a significant decrease in the level of NF-KB as compared to the control group, with a highly significant decline (p < .001) observed in DGN and PTB combination group. Whereas the levels of BDNF and Nrf2 had significantly increased, with very significantly high levels reported in combination treatment groups (DGN+PTB - 0.25 & 0.5 µM) as represented in Figure 15.
Figure 15. Representative Western Blots and Relative Protein Expressions of NF-KB, Nfr2 and BDNF in 10 µM Aβ1-42 Treated SH-SY5Y Cells Given Diosgenin and Pterostilbene Treatment as per Experimental Groups. Data were represented as a median ± SD standard deviation (n = 3; ns = not significant; **p < .01; ***p < .001 using one-way ANOVA Dunnett’s test.
Discussion
In vitro studies of DGN and PTB in SH-SY5Y cell cultures were carried out to investigate the neuroprotective mechanisms involved in preventing neurodegeneration. For research in CNS, numerous in vitro models have been created to understand the mechanisms underlying insults that result in neuronal death. And to identify improved therapeutic targets for neurodegenerative diseases linked to neuroinflammation. Neuronal cell cultures have been extensively utilised to create various in vitro models of neurodegenerative disorders, aiming to elucidate the mechanisms of neuroprotection and identify new therapeutic targets.
The buildup of neurofibrillary tangles and the aggregation of amyloid plaques in AD are common pathological features that impede neurotransmission, leading to decreased cognitive function. Consequently, amyloid β polymers trigger a chronic inflammatory response through the activation of astrocytes and microglia, causing overproduction of proinflammatory cytokines. 47 Progressive neurodegeneration results from these cytokines, increased exacerbation of neuroinflammation and glial activation. This hypothesis supports the application of anti-inflammatory agents to mitigate neurodegenerative pathology. Therefore, the study employed a combination of the two reported anti-inflammatory compounds, DGN and PTB, in the management of AD to offer neuroprotection.
In this study, various models were employed to evaluate the neuroprotective and anti-inflammatory potential of a combination of DGN and PTB in amyloid β-induced SH-SY5Y neuronal cell cultures. 48 Different concentrations of DGN, PTB, and the standard DPZ were tested to determine their IC50 values. All concentrations demonstrated neuroprotective effects by enhancing cell viability as shown by the MTT assay in a concentration-dependent manner. Additionally, AO/EtBr staining further affirmed the increased neuronal cell viability due to the treatments.
Anti-inflammatory compounds often regulate the altered morphology, activation, and proliferation of microglia by inhibiting nitric oxide synthesis and reducing the production of inflammatory mediators such TNF-α, IL-1β, and IL-6. 49 AD is strongly linked to excessive IL-1β secretion, as low levels promote neuronal growth while increased levels have been linked to neuronal toxicity, contributing to abnormal accumulation of amyloid β. Although TNF-α is normally found in microglia and astrocytes, its unnecessary increase is toxic to neurons. 50 The findings of the study confirmed decreased levels of TNF-α, IL-1β and IL-6 and inhibition of nitric oxide synthesis, which effectively suppressed neuroinflammation in amyloid β-induced SH-SY5Y cell cultures.
Similar to microglial activation, astrogliosis is observed in synaptic dysfunction and neuroinflammation following CNS insult. According to reports, proinflammatory cytokine production and enhanced astrocyte activity were observed in mice given an intracerebroventricular injection of amyloid beta (Aβ). 51 Our findings reaffirm the neuroprotective effect through the down-regulation of these protein levels and their mRNA expression.
Another key factor contributing to neurotoxicity is the release of ROS due to the formation of amyloid plaques and neurofibrillary tangles. 39 These amyloid aggregates also cause decreased cholinergic functions in the initial stages of neurodegeneration. Therefore, interfering with Aβ aggregation, suppressing ROS levels, and increasing acetylcholine levels are suitable strategies to combat neurotoxicity, restore synaptic plasticity, and improve cognition. 32 The study results suggested that DGN and PTB treatment inhibited the amyloidogenic process by decreasing β-secretase expression. The reduced aggregation of Aβ polymers contributed to lower ROS levels, leading to increased cell viability and improved cell function. Additionally, increased AChE inhibition contributed to elevated acetylcholine levels, enhancing memory as a consequence of improved neurotransmission.
Neuronal apoptosis is another feature associated with Aβ aggregation, leading to neuronal loss by arresting the cell cycle in the S phase. 52 Apoptosis assays and flow cytometric analysis of cell cultures receiving treatment in the study demonstrated alleviation of cell cycle arrest and improved neuronal cell viability. The decreased Aβ toxicity, coupled with apoptosis inhibition, was identified as a significant pathway contributing to the neuroprotective effect of the combination treatment.
Studies demonstrate that Nrf2 has a part in mediating antioxidant properties and preventing TLR4/NF-κB signalling pathways and the activation of the NLRP3 inflammasome, thereby reducing neuroinflammation.53, 54 Widely dispersed throughout the nervous system, NF-κB has a role in apoptosis, inflammation, and cell proliferation. While normal levels of NF-κB have a protective role for neurons, numerous inflammatory mediators are released as a result of excessive activation. 55
BDNF deregulation leads to cognitive impairment due to synaptic loss and neuronal dysfunction in AD. Elevated BDNF levels inhibit the PI3-K/Akt and MAPK signalling pathways’ activity, preventing the hyperphosphorylation of tau protein, which is crucial for neuronal regeneration. 56 Restoration of cognitive function and learning ability in 2xTg-AD mice was demonstrated through increased BDNF levels. 57 The critical role of BDNF in promoting neuronal development in AD pathology was further confirmed by increased BDNF levels in SH-SY5Y cell cultures.
It is hypothesised that Aβ accumulation may cause the formation of inflammasome complexes, resulting in increased inflammation, apoptosis, and ROS generation. 58 These contribute to microglial activation and astrogliosis, leading to the release of proinflammatory cytokines. Thus, neuroinflammation-mediated neurodegeneration is attenuated through the modulation of inflammatory responses, inhibition of apoptosis, suppression of nitric oxide synthesis, AChE inhibition, β-secretase inhibition, and elevated BDNF levels. These effects were observed in SH-SY5Y cell cultures treated with a combination of DGN and PTB following exposure to Aβ.
Conclusion
Studies reveal the pathophysiological role of β-amyloid and phosphorylated tau, causing oxidative stress and inducing inflammatory pathways through release of IL-6, TNF-alpha, IL-1β in Diabetes associated AD. 59 Despite failure of many drugs during clinical trials as inhibitors of GSK-3β and β-secretase, there is always a search for therapeutic strategies to introduce lead candidates with a newer mechanism to mitigate pathophysiological complications of the disease. DGN and PTB both significantly demonstrated neuroprotective effects against amyloid toxicity in differentiated SH-SY5Y cell culture. The models employed co-cultures of SH-SY5Y cells, which closely resembled the brain pathology of AD and paved the pathways for neuroprotection. 60 DGN and PTB individually and in combination significantly ameliorated the toxicity incurred by Aβ1-42 in SH-SY5Y cells by decreasing inflammatory mediators, ROS levels and apoptotic cell death. Further, the neuroprotective nature was demonstrated by increased cell viability and BDNF levels. The Combination index of DGN and PTB computed by MTT data correlation analysis using the CompuSyn software highlighted the combined synergistic effect of DGN and PTB, ascertaining it to be a very effective strategy against AD. Decreased levels of inflammatory mediators evidenced neuroinflammation and neurodegeneration as two critical interlinked pathways, and targeting these pathways could prove to be an effective strategy against AD. 61 Thus, the findings suggest the potential of the combination of DGN and PTB to be considered in the treatment of AD.
Author Contributions
Significant contributions were made by both the authors to the conception and design, data acquisition, analysis, and interpretation; both participated in the article’s drafting or critical revision for significant intellectual content; both agreed to submit the work to the current journal; gave final approval of the version to be published; and agree to be accountable for all aspects of the work. Both the authors are eligible to be an author as per the international committee of medical journal editors (ICMJE) requirements/guidelines.
Data Availability
This research article includes all of the generated and analysed data.
Statement of Ethics
There are no animal or human subjects involved in this study’s experiments.
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding: The authors received no financial support for the research, authorship and/or publication of this article.
ORCID iD: Syeda Jabeen Fatima  https://orcid.org/0009-0000-7844-6596
https://orcid.org/0009-0000-7844-6596
References
- 1.Feigin VL, Vos T, Nichols E, et al. The global burden of neurological disorders: Translating evidence into policy. Lancet Neurol 2019. Mar 1; 19: 255–265. DOI: 10.1016/S1474-4422 (19)30411-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Scheff SW, Price DA, Schmitt FA, et al. Synaptic alterations in CA1 in mild Alzheimer disease and mild cognitive impairment. Neurology 2007. May 1; 68: 1505–1508. DOI: 10.1212/01.wnl.0000260698.46517 [DOI] [PubMed] [Google Scholar]
- 3.Ross CA and Poirier MA.. Protein aggregation and neurodegenerative disease. Nat Med 2004; 10(Suppl): S10–S17. [DOI] [PubMed] [Google Scholar]
- 4.Emerit J, Edeas M and Bricaire F.. Neurodegenerative diseases and oxidative stress. Biomed Pharmacother 2004; 58(1): 39–46. DOI: 10.1016/j.biopha.2003.11.004 [DOI] [PubMed] [Google Scholar]
- 5.Streit WJ. Microglia and neuroprotection: implications for Alzheimer’s disease. Brain Res Bull 2005; 48(2): 234–239. DOI: 10.1016/j.brainresrev.2004.12.013 [DOI] [PubMed] [Google Scholar]
- 6.Heneka MT, Carson MJ, El Khoury J, et al. Neuroinflammation in Alzheimer’s disease. Lancet Neurol 2015; 14(4): 388–405. DOI: 10.1016/S1474-4422(15)70016-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Perry VH, Nicoll JA and Holmes C.. Microglia in neurodegenerative disease. Nat Rev Neurol 2010; 6(4): 193–201. [DOI] [PubMed] [Google Scholar]
- 8.Alzheimer’s Association. Alzheimer’s disease facts and figures. Alzheimers Dement 2016; 12: 459–509. DOI: 10.1016/j.jalz.2016.03.001 [DOI] [PubMed] [Google Scholar]
- 9.Chen Y, Liang Z, Blanchard J, et al. A non-transgenic mouse model (icv-STZ mouse) of Alzheimer’s disease: Similarities to and differences from the transgenic model (3xTg-AD mouse). Mol Neurobiol 2013; 47: 711–725. DOI: 10.1007/s12035- 012-8375-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sonkusare S, Srinivasan K, Kaul C, et al. Effect of donepezil and lercanidipine on memory impairment induced by intracerebroventricular streptozotocin in rats. Life Sci 2005; 77: 1–14. DOI: 10.1016/j.lfs.2004.10.036 [DOI] [PubMed] [Google Scholar]
- 11.Joo Y, Kim HS, Woo RS, et al. Mefenamic acid shows neuroprotective effects and improves cognitive impairment in in vitro and in vivo Alzheimer’s disease models. Mol Pharmacol 2006; 69(1): 76–84. DOI: 10.1124/mol.105.015206 [DOI] [PubMed] [Google Scholar]
- 12.Emerit J, Edeas M and Bricaire F.. Neurodegenerative diseases and oxidative stress. Biomed Pharmacother 2004; 58(1): 39–46. DOI: 10.1016/j.biopha.2003.11.004 [DOI] [PubMed] [Google Scholar]
- 13.Tiwari S, Atluri V, Kaushik A, et al. Alzheimer’s disease: Pathogenesis, diagnostics, and therapeutics. Int J Nanomedicine 2019; 14: 5541–5554. DOI: 10.2147/IJN.S200490 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Tönnies E and Trushina E.. Oxidative stress, synaptic dysfunction, and Alzheimer’s disease. J Alzheimers Dis 2017; 57(4): 1105–1121. DOI: 10.3233/JAD-161088 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Deture MA and Dickson DW.. The neuropathological diagnosis of Alzheimer’s disease. Mol Neurodegener 2019; 5(2): 1–18. DOI: 10.1186/s13024-019-0333-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Yiannopoulou KG and Papageorgiou SG.. Current and future treatments for Alzheimer’s disease. Ther Adv Neurol Disord 2013; 6(1): 19–33. DOI: 10.1177/1756285612461679 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dhami N. Trends in pharmacognosy: A modern science of natural medicines. J Herb Med 2013; 3(4): 123–131. DOI: 10.1016/j.hermed.2013.06.001 [Google Scholar]
- 18.Zahn R, Perry N, Perry E, et al. Use of herbal medicines: Pilot survey of UK users’ views. Complement Ther Med 2019; 44: 83–90. DOI: 10.1016/j.ctim.2019.02.007 [DOI] [PubMed] [Google Scholar]
- 19.Aglawe MM, Kale MB, Rahangdale SR, et al. Agmatine improves the behavioral and cognitive impairments associated with chronic gestational ethanol exposure in rats. Brain Res Bull 2021; 167: 37–47. DOI: 10.1016/j.brainresbull.2020.11.015 [DOI] [PubMed] [Google Scholar]
- 20.Sam S. Importance and effectiveness of herbal medicines. J Pharmacogn Phytochem 2019; 8(2): 354–357. [Google Scholar]
- 21.Chaudhary SA, Chaudhary PS, Syed BA, et al. Validation of a method for diosgenin extraction from fenugreek (Trigonella foenum-graecum L.). Acta Sci Pol Technol Aliment 2018; 17(4): 377–385. DOI: 10.17306/J.AFS.2018.0606 [DOI] [PubMed] [Google Scholar]
- 22.Manobharathi V and Mirunalini S.. Pharmacological characteristics of a phyto steroidal food saponin: Diosgenin. Afr J Biol Sci 2020; 2(2): 77–87. DOI: 10.33472/AFJBS.2.2.2020.77-87 [Google Scholar]
- 23.Kaur B, Khatun S and Suttee A.. Current highlights on biochemical and pharmacological profile of Dioscorea alata: A review. Plant Arch 2021; 21(1): 552–559. DOI: 10.51470/PLANTARCHIVES.2021.v21.S1.085 [Google Scholar]
- 24.He L, Jiang Y, Liu K, et al. Insights into the impact of a membrane-anchoring moiety on the biological activities of bivalent compounds as potential neuroprotectants for Alzheimer’s disease. J Med Chem 2018; 61(3): 777–790. DOI: 10.1021/acs.jmedchem.7b01284 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Koh EK, Yun WB, Kim JE, et al. Beneficial effect of diosgenin as a stimulator of NGF on the brain with neuronal damage induced by Aβ-42 accumulation and neurotoxicant injection. Lab Anim Res 2016; 32: 105–115. DOI: 10.5625/lar.2016.32.2.105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Li B, Xu P, Wu S, et al. Diosgenin attenuates lipopolysaccharide-induced Parkinson’s disease by inhibiting the TLR/NF-κB pathway. J Alzheimers Dis 2018; 64(3): 943–955. DOI: 10.3233/JAD-180330 [DOI] [PubMed] [Google Scholar]
- 27.Selim S and Al Jaouni S.. Anti-inflammatory, antioxidant and antiangiogenic activities of diosgenin isolated from traditional medicinal plant, Costus speciosus (Koen ex. Retz.) Sm. Nat Prod Res 2016; 30(16): 1830–1833. DOI: 10.1080/14786419.2015.1065493 [DOI] [PubMed] [Google Scholar]
- 28.Mondal S. Anticancer potential of biologically active diosgenin and its derivatives: An update. Curr Tradit Med 2024; 10(1): 67–80. DOI: 10.2174/2215083809666230202143136 [Google Scholar]
- 29.Lin WS, Leland JV, Ho CT, et al. Occurrence, bioavailability, anti-inflammatory, and anticancer effects of pterostilbene. J Agric Food Chem 2020; 68(46): 12788–12799. DOI: 10.1021/acs.jafc.9b07860 [DOI] [PubMed] [Google Scholar]
- 30.Nikhil K, Sharan S, Palla SR, et al. Understanding the mode of action of a pterostilbene derivative as anti-inflammatory agent. Int Immunopharmacol 2015; 28(1): 10–21. DOI: 10.1016/j.intimp.2015.05.003 [DOI] [PubMed] [Google Scholar]
- 31.Liu J, Fan C, Yu L, et al. Pterostilbene exerts an anti-inflammatory effect via regulating endoplasmic reticulum stress in endothelial cells. Cytokine 2016; 77: 88–97. DOI: 10.1016/j.cyto.2015.11.006 [DOI] [PubMed] [Google Scholar]
- 32.Wang B, Liu H, Yue L, et al. Neuroprotective effects of pterostilbene against oxidative stress injury: Involvement of nuclear factor erythroid 2-related factor 2 pathway. Brain Res 2016; 1643: 70–79. DOI: 10.1016/j.brainres.2016.04.048 [DOI] [PubMed] [Google Scholar]
- 33.Arslan ME, Türkez H and Mardinoğlu A.. In vitro neuroprotective effects of farnesene sesquiterpene on Alzheimer’s disease model of differentiated neuroblastoma cell line. Int J Neurosci 2021; 131(8): 745–754. DOI: 10.1080/00207454. 2020.1754211 [DOI] [PubMed] [Google Scholar]
- 34.Moujalled D, Grubman A, Acevedo K, et al. TDP-43 mutations causing amyotrophic lateral sclerosis are associated with altered expression of RNA-binding protein hnRNP K and affect the Nrf2 antioxidant pathway. Hum Mol Genet 2017; 26(9): 1732–1746. DOI: 10.1093/hmg/ddx093 [DOI] [PubMed] [Google Scholar]
- 35.Pulkrabkova L, Muckova L, Hrabinova M, et al. Differentiated SH-SY5Y neuroblastoma cells as a model for evaluation of nerve agent-associated neurotoxicity. Arch Toxicol 2023; 97(8): 2209–2217. DOI: 10.1007/s00204-023-03525-0 [DOI] [PubMed] [Google Scholar]
- 36.Wang H, Jiang T, Li W, et al. Resveratrol attenuates oxidative damage through activating mitophagy in an in vitro model of Alzheimer’s disease. Toxicol Lett 2018; 282: 100–108. DOI: 10.1016/j.toxlet.2017.10.021 [DOI] [PubMed] [Google Scholar]
- 37.Wang YJ, Zhou SM, Xu G, et al. Interference of phenylethanoid glycosides from Cistanche tubulosa with the MTT assay. Molecules 2015; 20(5): 8060–8071. DOI: 10.3390/molecules20058060 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Mutlu Altundağ E, Jannuzzi AT, Özbilenler C, et al. Synergistic role of thymoquinone and 5-fluorouracil in U-251MG glioblastoma cell line. Turk J Biochem 2024; 49(1): 82–89. DOI: 10.1515/tjb-2023-0150 [Google Scholar]
- 39.Segawa K and Nagata S.. An apoptotic ‘eat me’ signal: phosphatidylserine exposure. Trends Cell Biol 2015; 25(11): 639–650. DOI: 10.1016/j.tcb.2015.08.003 [DOI] [PubMed] [Google Scholar]
- 40.Lingappa S, Shivakumar MS, Manivasagam T, et al. Neuroprotective effect of epalrestat on hydrogen peroxide-induced neurodegeneration in SH-SY5Y cellular model. J Microbiol Biotechnol 2021; 31(6): 867–874. DOI: 10.4014/jmb.2101.01002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Jiang H, Yu Y, Liu S, et al. Proteomic study of a Parkinson’s disease model of undifferentiated SH-SY5Y cells induced by a proteasome inhibitor. Int J Med Sci 2019; 16(1): 84–92. DOI: 10.7150/ijms.28595 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Ma WW, Li CQ, Zhao L, et al. NF-κB-mediated inflammatory damage is differentially affected in SH-SY5Y and C6 cells treated with 27-hydroxycholesterol. Food Sci Nutr 2019; 7(5): 1685–1694. DOI: 10.1002/fsn3.1005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Castillo WO, Palomino NV, Takahashi CS, et al. Genistein and galantamine combinations decrease β-amyloid peptide (1–42)–induced genotoxicity and cell death in SH-SY5Y cell line: An in vitro and in silico approach for mimic of Alzheimer’s disease. Neurotox Res 2020; 38(3): 691–706. DOI: 10.1007/s12640-020- 00243-8 [DOI] [PubMed] [Google Scholar]
- 44.Kalyanaraman B, Darley-Usmar V, Davies KJ, et al. Measuring reactive oxygen and nitrogen species with fluorescent probes: Challenges and limitations. Free Radic Biol Med 2012; 52(1): 1–6. DOI: 10.1016/j.freeradbiomed.2011.09.030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Choi HY, Jhun EJ and Lim BO.. Application of flow injection-chemiluminescence to the study of radical scavenging activity in plants. Phytother Res 2000; 14(4): 250–253. DOI: 10.1002/1099-1573 [DOI] [PubMed] [Google Scholar]
- 46.Li Q, Wu D, Zhang L, et al. Effects of galantamine on β-amyloid release and beta-site cleaving enzyme 1 expression in differentiated human neuroblastoma SH-SY5Y cells. Exp Gerontol 2010; 45(11): 842–847. DOI: 10.1016/j.exger.2010.06.008 [DOI] [PubMed] [Google Scholar]
- 47.Tungalag T and Yang DK.. Sinapic acid protects SH-SY5Y human neuroblastoma cells against 6-hydroxydopamine-induced neurotoxicity. Biomedicines 2021; 9(3): 295. DOI: 10.3390/biomedicines9030295 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Dani M, Wood M, Mizoguchi R, et al. Microglial activation correlates in vivo with both tau and amyloid in Alzheimer’s disease. Brain 2018; 141(9): 2740–2754. DOI: 10.1093/brain/awy188 [DOI] [PubMed] [Google Scholar]
- 49.Forster JI, Köglsberger S, Trefois C, et al. Characterization of differentiated SH-SY5Y as neuronal screening model reveals increased oxidative vulnerability. J Biomol Screen 2016; 21(5): 496–509. DOI: 10.1177/1087057115625190 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Kirkley KS, Popichak KA, Afzali MF, et al. Microglia amplify inflammatory activation of astrocytes in manganese neurotoxicity. J Neuroinflamm 2017; 14: 1–8. DOI: 10.1186/s12974-017- 0871-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Reinsfelt B, Westerlind A, Blennow K, et al. Open-heart surgery increases cerebrospinal fluid levels of Alzheimer-associated amyloid β. Acta Anaesthesiol Scand 2013; 57(1): 82–88. DOI: 10.1111/j.1399-6576.2012.02769.x [DOI] [PubMed] [Google Scholar]
- 52.Bashir DJ, Manzoor S, Sarfaraj M, et al. Magnoflorine-loaded chitosan collagen nanocapsules ameliorate cognitive deficit in scopolamine-induced Alzheimer’s disease-like conditions in a rat model by downregulating IL-1β, IL-6, TNF-α, and oxidative stress and upregulating brain-derived neurotrophic factor and DCX expressions. ACS Omega 2023; 8(2): 2227–2236. DOI: 10.1021/acsomega.2c06467 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Moh C, Kubiak JZ, Bajic VP, et al. Cell cycle deregulation in the neurons of Alzheimer’s disease. In: Cell cycle in development. Berlin: Springer, 2011, pp.565–576. DOI: 10.1007/978-3- 642-19065-0_23 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Jin X, Liu MY, Zhang DF, et al. Baicalin mitigates cognitive impairment and protects neurons from microglia-mediated neuroinflammation via suppressing NLRP3 inflammasomes and TLR4/NF-κB signaling pathway. CNS Neurosci Ther 2019; 25(5): 575–590. DOI: 10.1111/cns.13086 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Elango B, Dornadula S, Paulmurugan R, et al. Pterostilbene ameliorates streptozotocin-induced diabetes through enhancing antioxidant signaling pathways mediated by Nrf2. Chem Res Toxicol 2016; 29(1): 47–57. DOI: 10.1021/acs.chemrestox.5b00378 [DOI] [PubMed] [Google Scholar]
- 56.Hogan KA, Ravindran A, Podolsky MA, et al. The TGFβ1 pathway is required for NFκB dependent gene expression in mouse keratinocytes. Cytokine 2013; 64(3): 652–659. DOI: 10.1016/j.cyto.2013.09.004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Rai SN, Dilnashin H, Birla H, et al. The role of PI3K/Akt and ERK in neurodegenerative disorders. Neurotox Res 2019; 35: 775–795. DOI: 10.1007/s12640-019-0003-y [DOI] [PubMed] [Google Scholar]
- 58.Nigam SM, Xu S, Kritikou JS, et al. Exercise and BDNF reduce Aβ production by enhancing α-secretase processing of APP. J Neurochem 2017; 142(2): 286–296. DOI: 10.1111/jnc.14034 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Bandaru LJ, Murumulla L, Devarakonda KP, et al. Exposure of combination of environmental pollutant, lead (Pb) and β-amyloid peptides causes mitochondrial dysfunction and oxidative stress in human neuronal cells. J Bioenerg Biomembr 2023; 55(1): 79–89. DOI: 10.1007/s10863-023-09956-9 [DOI] [PubMed] [Google Scholar]
- 60.Rad SK, Arya A, Karimian H, et al. Mechanism involved in insulin resistance via accumulation of β-amyloid and neurofibrillary tangles: link between type 2 diabetes and Alzheimer’s disease. Drug Des Devel Ther 2018; 12: 3999–4021. DOI: 10.2147/DDDT.S173970 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.de Medeiros LM, De Bastiani MA, Rico EP, et al. Cholinergic differentiation of human neuroblastoma SH-SY5Y cell line and its potential use as an in vitro model for Alzheimer’s disease studies. Mol Neurobiol 2019; 56: 7355–7367. DOI: 10.1007/s12035-019-1605-3 [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
This research article includes all of the generated and analysed data.


