Abstract
Objective
This study explores the prognostic value of combining the PaO2/FiO2 ratio and nonthyroidal illness syndrome (NTIS) for predicting 28-day mortality in patients with sepsis.
Methods
A retrospective cohort study was conducted using data from adult sepsis patients admitted to the ICU in the Medical Information Mart for Intensive Care IV (MIMIC-IV) database. Patients were divided into four groups based on PaO2/FiO2 ratio levels and the presence of NTIS (Groups 1–4). Kaplan–Meier (K-M) analysis with the log-rank test was used to compare 28-day mortality among groups. Multivariate Cox regression was performed to evaluate the independent association between group classification and mortality. A restricted cubic spline (RCS) was used to explore the relationship between PaO2/FiO2 ratio and 28-day mortality.
Results
A total of 599 sepsis patients were included in the analysis. Among the four P/F–NTIS stratified groups, Group 4 (P/F ≤ 200 mmHg with NTIS) exhibited the highest 28-day mortality rate (37.11%), significantly higher than Groups 1, 2, and 3 (p < .001). K-M analysis revealed a significantly lower survival probability in Group 4 (log-rank test, p < .001). Univariate Cox regression analysis identified P/F–NTIS classification as significant predictors of 28-day mortality. Multivariate Cox regression analysis confirmed that Group 4 was independently associated with increased 28-day mortality (HR = 1.70, 95% CI [1.08–2.69], p = .022). RCS analysis showed a nonlinear but statistically significant association between P/F ratio and 28-day mortality (p for nonlinearity = .242), further supporting the prognostic relevance of oxygenation status.
Conclusion
The combined evaluation of the P/F and NTIS provides a more comprehensive risk stratification approach, allowing for improved 28-day mortality prediction in septic patients.
Keywords: Sepsis, oxygenation index, nonthyroidal illness syndrome, mortality, MIMIC-IV database
Introduction
Sepsis is a syndrome of multiple organ dysfunction caused by an abnormal immune response to infection. 1 Its core pathophysiological mechanism lies in the disruption of immune homeostasis, leading to an imbalance between excessive inflammation and compensatory antiinflammatory responses.2,3 This dysregulation ultimately results in tissue hypoxia, metabolic disturbances, and organ failure. Despite continuous advancements in medical treatment, the mortality rate of sepsis remains high at 19.7% of global mortality, 4 with particularly poor prognosis in patients with concomitant acute respiratory distress syndrome (ARDS) or malnutrition.
Currently, the Acute Physiology and Chronic Health Evaluation II (APACHE II), Simplified Acute Physiology Score II (SAPS II), and Sequential Organ Failure Assessment (SOFA) scores are widely used clinical tools for prognostic assessment. 5 While they evaluate organ dysfunction based on static physiological parameters, both have inherent limitations. The SOFA score is based on the physiological parameters of a patient at a single time point and primarily reflects the degree of organ dysfunction. Although a higher SOFA score is associated with increased mortality, its predictive performance is slightly inferior to that of more complex scoring systems, such as APACHE II and SAPS II. SAPS II provides a more comprehensive assessment of disease severity, however, its evaluation requires the assessment of 12 variables, making the process time-consuming. 6 Notably, neither scoring system adequately captures the critical roles of metabolic dysregulation and immune status in patient outcomes. Previous studies have shown that the P/F ratio, a component of both SOFA and SAPS II scores, 7 serves as an indicator of disease severity and has been identified as an independent risk factor for 28-day mortality in sepsis patients.7,8 However, its predictive performance alone is slightly inferior to that of traditional scoring systems. 9 On the other hand, NTIS is recognized as an integrated marker of metabolic-immune dysregulation in critically ill patients. It is characterized by reduced plasma triiodothyronine (T3) levels, normal or decreased thyroxine (T4) levels, and increased reverse T3 (rT3) in the presence of normal or low thyroid-stimulating hormone (TSH) levels.9–12 NTIS is closely associated with cytokine storms, immune exhaustion, and nutritional–metabolic imbalances. 9 Although NTIS itself is not an independent predictor of mortality in sepsis, 9 studies have shown that it correlates with poor outcomes in conditions such as sepsis, multiple trauma, and ARDS, significantly increasing the risk of death. 9
A low P/F ratio (≤200 mmHg) reflects impaired pulmonary oxygenation and consequent tissue hypoxia, while the presence of NTIS indicates metabolic reserve depletion and impaired immune recovery. The coexistence of these two conditions may exacerbate cellular energy crises and promote the progression of organ dysfunction in sepsis. Given these synergistic pathophysiological effects, an integrated assessment incorporating both oxygenation status and metabolic-immune function may overcome the limitations of conventional scoring systems, which often fail to capture immunological and nutritional-metabolic derangements. In this study, we aimed to investigate the prognostic value of the comorbidity of a low P/F ratio and NTIS in predicting 28-day mortality among septic patients. Utilizing data from the Medical Information Mart for Intensive Care IV (MIMIC-IV) database, we conducted a retrospective cohort analysis to evaluate whether this combined approach could improve risk stratification and guide personalized management strategies in sepsis.
Methods
Data source
This retrospective cohort study was based on health-related data extracted from the MIMIC-IV database (version 3.1), a large, publicly available critical care database maintained by the Laboratory for Computational Physiology at the Massachusetts Institute of Technology. The database includes over 90,000 high-quality electronic medical records from patients admitted to the intensive care units (ICUs) of Beth Israel Deaconess Medical Center (BIDMC). All personally identifiable information in the database has been fully deidentified to protect patient privacy, in compliance with the Health Insurance Portability and Accountability Act. The study protocol was approved through the PhysioNet credentialed data access process, and the requirement for individual informed consent was waived by the Institutional Review Board of BIDMC due to the retrospective nature of the study. This study was conducted in accordance with the ethical principles outlined in the Declaration of Helsinki (originally adopted in 1964 and revised in 1975 and 2024). The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology guidelines. 13
Study population
Adult critically ill patients diagnosed with sepsis were enrolled in this study. Sepsis was identified based on the Sepsis-3 criteria, requiring a suspected or confirmed infection along with an increase of ≥2 points in the total SOFA score. 14 A total of 599 patients were included in the final analysis. Figure 1 shows the selection process for patients. Initially, 41,296 ICU sepsis admission records were extracted from the MIMIC-IV database 3.1, and 31,911 patients with first ICU admission records were identified. Subsequently, patients were excluded based on seven criteria: ICU length of stay < 24 h (n = 11,568), age < 18 years (n = 0), ICU length of stay > 30 days (n = 3794), history of thyroid disease (n = 5727), history of cerebrovascular disease (n = 946), history of autoimmune diseases (n = 486), and missing data on thyroid function parameters or P/F ratio (n = 18,176). After sequential exclusions, the final cohort consisted of 599 patients, who were further stratified into four subgroups by combining the P/F ratio (with a cutoff at 200 mmHg) and the presence of NTIS: Group 1 (P/F > 200 mmHg and without NTIS, n = 191), Group 2 (P/F > 200 mmHg and with NTIS, n = 168), Group 3 (P/F ≤ 200 mmHg and without NTIS, n = 81), and Group 4 (P/F ≤ 200 mmHg and with NTIS, n = 159).
Figure 1.
Flow chart for inclusion of participants.
Data extraction and variables
Data extraction and preprocessing were performed using SQL in a PostgreSQL (v14) environment. Baseline characteristics and laboratory parameters were retrieved by querying item IDs stored in the MIMIC-IV database and linking them to selected patients via a unique subject identifier (subject_id).
The extracted variables included baseline demographic and clinical characteristics such as age, sex, height, weight, Glasgow Coma Scale score, SAPS II, SOFA score, and the use of mechanical ventilation and vasopressor therapy. Vital signs were also collected, including heart rate (HR), systolic blood pressure, diastolic blood pressure (DBP), mean arterial pressure, and respiratory rate (RR). Comorbidities included hypertension, diabetes mellitus, heart failure, myocardial infarction, coronary artery disease, and chronic obstructive pulmonary disease (COPD). Laboratory parameters extracted encompassed white blood cell (WBC) count, lymphocyte count, neutrophil count, hemoglobin, C-reactive protein, platelet count, international normalized ratio, prothrombin time, partial thromboplastin time, alanine aminotransferase, aspartate aminotransferase, alkaline phosphatase (ALP), creatinine, cholesterol, triglycerides, glucose, calcium, potassium, pH, and lactate levels. Thyroid function indicators, including T3, T4, and TSH levels, were also extracted. In addition, data on hospital and ICU admissions—including admission and discharge dates, ICU admission and discharge dates, and in-hospital mortality—were collected. All clinical and laboratory measurements used in this study were obtained from the first recorded assessment following the patient's initial ICU admission.
Data definitions
The oxygenation index (OI) is calculated as the ratio of arterial oxygen partial pressure (PaO2, mmHg) to the fraction of inspired oxygen (FiO2, %), expressed as OI = PaO2/FiO2. The normal P/F range is 400–500 mmHg. According to the Berlin definition of ARDS, P/F is categorized as mild (200 < P/F ≤ 300 mmHg), moderate (100 < P/F ≤ 200 mmHg), and severe (P/F ≤ 100 mmHg). 15 In this study, patients were classified based on their P/F ratio, with mild hypoxemia defined as P/F > 200 mmHg and moderate-to-severe hypoxemia as P/F ≤ 200 mmHg. NTIS also known as euthyroid sick syndrome, is a thyroid function abnormality that occurs in the setting of acute or chronic systemic illness, without intrinsic thyroid gland pathology. It is typically characterized by decreased T3 levels, with or without reduced T4, and a normal or slightly decreased TSH. 16 This condition reflects an adaptive metabolic response rather than a primary thyroid disorder.9–12 In this study, NTIS was defined as a T3 level below 80 ng/dL and/or a T4 level below 4.6 μg/dL, with normal or decreased TSH levels. Patients were divided into four groups based on their P/F and NTIS status: Group 1 included patients with a P/F > 200 mmHg without NTIS; Group 2 comprised those with a P/F > 200 mmHg with NTIS; Group 3 consisted of patients with a P/F ≤ 200 mmHg without NTIS; and Group 4 included patients with a P/F ≤ 200 mmHg with NTIS.
Outcomes
The primary outcome was 28-day all-cause mortality after ICU admission, determined based on follow-up death dates recorded in the MIMIC-IV 3.1 database.
Statistical analysis
Patients were stratified into four groups based on baseline P/F ratio and the presence or absence of NTIS. Continuous variables were reported as mean ± standard deviation (SD) or median with interquartile range, as appropriate. Categorical variables were expressed as counts and percentages. Group comparisons were performed using the chi-square test for categorical variables, one-way analysis of variance for normally distributed continuous variables, and the Kruskal–Wallis H test for nonnormally distributed variables. Patients with a P/F ≥ 200 mmHg and without NTIS were used as the reference group in subsequent analyses.
To identify predictors of 28-day mortality in sepsis, patients were stratified by survival status (nonsurvivors vs survivors), and baseline characteristics were compared. Variables with a p-value < .05 were included in univariate Cox regression analysis. Those remaining significant were entered into a multivariate Cox proportional hazards model to estimate hazard ratios (HRs) and 95% confidence intervals (CIs) for mortality across P/F–NTIS stratified groups. Model 1 was unadjusted; Model 2 was adjusted for age, SOFA score, and SAPS-II score; and Model 3 was further adjusted for age, SOFA, SAPS-II, HR, RR, WBC count, neutrophil count, ALP, creatinine, hemoglobin, use of mechanical ventilation, vasopressor use, and lactate levels. To avoid multicollinearity, variables with a variance inflation factor (VIF) > 5 were excluded from the final model. K–M analysis was conducted to assess survival differences among the four P/F–NTIS groups, with the log-rank test used to compare survival curves. Restricted cubic spline (RCS) analysis was also performed to explore the nonlinear association between P/F ratio and 28-day mortality.
Results
Baseline characteristics
A total of 599 patients were included in the final analysis. The patient selection process is illustrated in Figure 1. Baseline characteristics of survivors and nonsurvivors at 28 days are summarized in Table 1. Compared with survivors, nonsurvivors were significantly older and had higher SAPS II and SOFA scores (p < .05). Among laboratory parameters, the nonsurvivor group exhibited lower hemoglobin levels and higher levels of ALP and lactate (p < .05). In terms of therapeutic interventions, nonsurvivors had significantly higher rates of mechanical ventilation and vasopressor use, along with a more severe P/F–NTIS stratification profile. Additionally, initial vital signs, including HR and RR, were significantly elevated in nonsurvivors. Table 2 presents baseline characteristics stratified by P/F ratio and NTIS status. Notably, Group 4 (P/F ≤ 200 mmHg with NTIS) had the highest 28-day ICU mortality rate compared to the other groups (37.11% vs 30.86%, 22.62%, and 17.28%, respectively; p < .001).
Table 1.
The characteristics of included patients stratified by 28-day survival outcomes.
| Variable | Overall (n = 599) | 28-day survival (n = 445) | 28-day nonsurvival (n = 145) | p-value |
|---|---|---|---|---|
| Age, years | 65 (18–96) | 63 (18–96) | 68 (25–92) | .001 |
| Male | 347 (57.93) | 258 (57.98) | 89 (57.79) | .968 |
| Height, cm | 173 (144–203) | 173 (144–203) | 172 (144–203) | .670 |
| Weight, kg | 78 (39–287.60) | 77.20 (39–287.60) | 80.25 (46.30–171.20) | .835 |
| BMI, kg/m2 | 26.93(12.62–88.77) | 26.73(12.62–88.77) | 27.57(15.54–57.15) | .446 |
| Clinical severity | ||||
| SAPS-II | 48 (5–152) | 45 (5–152) | 58.50 (17–124) | <.001 |
| GCS | 15 (3–15) | 15 (3–15) | 15 (3–15) | .810 |
| SOFA | 3 (2–16) | 3 (2–14) | 4 (2–16) | <.001 |
| Vital signs | ||||
| Heart rate, (beats/min) | 89 (38–170) | 88 (38–154) | 93 (58–170) | .001 |
| SBP (mmHg) | 115 (36–210) | 118 (36–210) | 109 (67–190) | .107 |
| DBP (mmHg) | 66 (12–190) | 66 (20–190) | 63.50 (12–142) | .168 |
| MAP (mmHg) | 78 (25–197) | 79 (26–197) | 75 (25–146) | .283 |
| RR (beats/min) | 19 (0–45) | 18 (0–45) | 20 (7–40) | .037 |
| Comorbidities | ||||
| Hypertension (%) | 198 (33.06) | 150 (33.71) | 48 (31.17) | .564 |
| Heart failure (%) | 241 (40.23) | 169 (37.98) | 72 (46.75) | .056 |
| Myocardial infarction (%) | 61 (10.18) | 41 (9.21) | 20 (12.99) | .182 |
| Coronary artery disease (%) | 230 (38.40) | 169 (37.98) | 61 (39.61) | .720 |
| COPD (%) | 92 (15.36) | 65 (14.61) | 27 (17.53) | .385 |
| Diabetes (%) | 201 (33.56) | 142 (31.91) | 59 (38.31) | .147 |
| Laboratory tests | ||||
| WBC (K/UL) | 15.30 (0.20–395.20) | 15.10 (0.20–116.50) | 17 (0.30–395.20) | .053 |
| Lymphocytes (K/UL) | 1.25 (0–366.95) | 1.29 (0–8.25) | 1.12 (0.03–366.95) | .273 |
| Neutrophils (K/UL) | 10.53 (0–63.83) | 10.12 (0–63.83) | 11.85 (0–35.44) | .110 |
| Monocytes (K/UL) | 0.70 (0–10.60) | 0.70 (0–4.62) | 0.71 (0–10.60) | .350 |
| Hemoglobin (g/dL) | 10.10 (4.80–18) | 10.30 (4.80–18) | 9.60 (4.80–17.70) | <.001 |
| CRP (mg/L) | 26.20 (0.10–327.10) | 23.70 (0.10–327.10) | 32.85 (0.10–327.10) | .299 |
| Platelets (K/UL) | 199 (5–1092) | 203 (5–1039) | 187.50 (9–1092) | .239 |
| INR (s) | 1.20 (0.80–8.30) | 1.20 (0.80–8.30) | 1.20 (0.90–4.30) | .485 |
| PT (s) | 13.20 (9.20–81.30) | 13.10 (9.20–81.30) | 13.50 (9.40–46.40) | .507 |
| APTT (s) | 29.70 (19.20–150) | 29.40 (19.20–150) | 30.90 (21.20–150) | .136 |
| ALT (u/L) | 26 (3–12,680) | 24 (3–12,680) | 32 (8–1720) | .358 |
| AST (u/L) | 31 (9–11,220) | 29 (9–11,220) | 33.50 (9–4580) | .787 |
| ALP (u/L) | 84 (24–2840) | 79 (24–1518) | 92.50 (42–2840) | .033 |
| Creatinine (mg/dL) | 1 (0.20–16.10) | 1 (0.20–16.10) | 1.20 (0.50–6.20) | .053 |
| Chloride (mg/dL) | 102 (84–119) | 102 (85–119) | 101 (84–119) | .129 |
| Triglyceride (mg/dL) | 128 (28–8300) | 126 (28–8300) | 136 (28–3003) | .439 |
| Glucose (mg/dL) | 116 (58–722) | 115 (58–722) | 124 (69–702) | .150 |
| Albumin (g/dL) | 3.00 (1.20–5.00) | 3.00 (1.40–5.00) | 2.75 (1.20–4.50) | <.001 |
| Total protein (g/dL) | 5.30 (3.40–7.80) | 5.40 (3.40–7.80) | 5.00 (3.60–7.10) | .044 |
| Calcium (mmol/L) | 9 (4.10–17.70) | 9 (4.10–17.70) | 9 (4.10–17.70) | .811 |
| Sodium (mmol/L) | 139 (117–157) | 139 (117–153) | 139 (119–157) | .975 |
| Potassium (mmol/L) | 4.20 (2.30–8.70) | 4.20 (2.30–8.70) | 4.20 (2.70–7.80) | .498 |
| PH | 7.38 (6.53–7.60) | 7.38 (6.89–7.60) | 7.39 (6.53–7.58) | .504 |
| Lactate (mmol/L) | 1.60 (0.40–19) | 1.60 (0.40–13.50) | 2 (0.60–19) | .017 |
| Treatments | ||||
| Glucocorticoid (%) | 187.00(31.222) | 132.00(29.66) | 55.00(35.71) | .162 |
| Vasoactive drugs (%) | 271 (45.24) | 190 (42.70) | 81 (52.60) | .033 |
| Ventilation (%) | 415 (69.28) | 319 (71.69) | 96 (62.34) | .030 |
| P/F–NTIS stratification (%) | <.001 | |||
| 1 | 191.00 (31.89%) | 159.00 (35.73%) | 32.00 (20.78%) | |
| 2 | 168.00 (28.05%) | 130.00 (29.21%) | 38.00 (24.68%) | |
| 3 | 81.00 (13.52%) | 56.00 (12.58%) | 25.00 (16.23%) | |
| 4 | 159.00 (26.54%) | 100.00 (22.47%) | 59.00 (38.31%) | |
| Outcome | ||||
| 28-day mortality (%) | 155 (25.88) | 1 (0.22) | 154 (100) | <.001 |
| ICU-stay-time (h) | 117 (24.20–2299.08) | 110.90 (24.20–2299.08) | 141.31 (24.70–1820.30) | .278 |
Abbreviations: BMI: body mass index; SAPS-II: Simplified Acute Physiology Score II; GCS: Glasgow Coma Scale; SOFA: Sequential Organ Failure Assessment; SBP: systolic blood pressure; DBP: diastolic blood pressure; MAP: mean arterial pressure; RR: respiratory rate; COPD: chronic obstructive pulmonary disease; WBC: white blood cell count; CRP: C-reactive protein; INR: international normalized ratio; PT: prothrombin time; APTT: activated partial thromboplastin time; ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase, PH: potential of hydrogen; NTIS: nonthyroidal illness syndrome.
Table 2.
Baseline characteristics in sepsis patients based on combined P/F and NTIS stratification.
| Variable | Overall (n = 599) | Group 1 (n = 191) | Group 2 (n = 168) | Group 3 (n = 81) | Group 4 (n = 159) | p-value |
|---|---|---|---|---|---|---|
| Age, years | 65.00 (54.00, 75.00) | 62.00 (54.00, 71.00) | 69.00 (57.00, 77.00) | 64.00 (50.00, 76.00) | 65.00 (54.00, 77.00) | .015 |
| Male | 347.00 (57.93) | 102.00 (53.40) | 105.00 (62.50) | 36.00 (44.44) | 104.00 (65.41) | .005 |
| Height, cm | 173.00 (165.00, 178.00) | 173.00 (165.00, 178.00) | 170.00 (165.00, 178.00) | 170.00 (165.00, 178.00) | 171.00 (165.00, 178.00) | .080 |
| Weight, kg | 78.00 (65.00, 95.00) | 76.00 (63.40, 88.70) | 76.90 (63.60, 93.80) | 80.40 (65.00, 95.30) | 81.70 (70.20, 100.30) | .012 |
| BMI, kg/m2 | 26.93(12.62, 88.77) | 25.47 (12.62,45.00) | 26.43 (13.82,67.57) | 28.43 (15.78,56.76) | 28.43 (15.78,56.76) | <.001 |
| Clinical severity | ||||||
| SAPS-II | 48.00 (36.00, 62.00) | 44.00 (34.00, 57.00) | 47.00 (36.50, 61.50) | 51.00 (32.00, 67.00) | 52.00 (41.00, 69.00) | <.001 |
| GCS | 15.00 (14.00, 15.00) | 15.00 (14.00, 15.00) | 15.00 (13.50, 15.00) | 15.00 (14.00, 15.00) | 15.00 (14.00, 15.00) | .559 |
| SOFA | 3.00 (2.00, 5.00) | 3.00 (2.00, 4.00) | 3.00 (2.00, 4.00) | 3.00 (2.00, 5.00) | 4.00 (3.00, 6.00) | <.001 |
| Vital signs | ||||||
| Heart rate (beats/min) | 89.00 (76.00, 105.00) | 91.00 (78.00, 106.00) | 85.00 (72.00, 95.50) | 93.00 (81.00, 105.00) | 93.00 (77.00, 107.00) | <.001 |
| SBP (mmHg) | 115.00 (100.00, 135.00) | 117.00 (100.00, 136.00) | 114.00 (99.50, 134.00) | 116.00 (104.00, 142.00) | 111.50 (99.00, 133.00) | .277 |
| DBP (mmHg) | 66.00 (55.00, 77.00) | 67.00 (59.50, 77.00) | 64.50 (53.00, 75.50) | 64.00 (53.00, 78.00) | 65.00 (52.00, 77.00) | .129 |
| MAP (mmHg) | 78.00 (68.00, 90.00) | 79.00 (70.00, 89.00) | 77.00 (67.00, 88.00) | 78.50 (68.00, 94.00) | 78.50 (65.00, 91.00) | .395 |
| RR (beats/min) | 19.00 (16.00, 23.00) | 19.00 (15.00, 24.00) | 18.00 (15.00, 22.00) | 20.00 (17.00, 25.00) | 19.00 (16.00, 23.00) | .017 |
| Comorbidities | ||||||
| Hypertension (%) | 198.00 (33.06) | 66.00 (34.55) | 56.00 (33.33) | 29.00 (35.80) | 47.00 (29.56) | .716 |
| Heart failure (%) | 241.00 (40.23) | 70.00 (36.65) | 76.00 (45.24) | 24.00 (29.63) | 71.00 (44.65) | .049 |
| Myocardial infarction (%) | 61.00 (10.18) | 15.00 (7.85) | 18.00 (10.71) | 7.00 (8.64) | 21.00 (13.21) | .394 |
| Coronary artery disease (%) | 230.00 (38.40) | 66.00 (34.55) | 80.00 (47.62) | 20.00 (24.69) | 64.00 (40.25) | .003 |
| COPD | 92.00 (15.36) | 26.00 (13.61) | 26.00 (15.48) | 16.00 (19.75) | 24.00 (15.09) | .646 |
| Diabetes | 201.00 (33.56) | 56.00 (29.32) | 67.00 (39.88) | 26.00 (32.10) | 52.00 (32.70) | .197 |
| Vasoactive drug | 271 (45.24) | 77 (40.31) | 74 (44.05) | 38 (46.91) | 82 (51.57) | .201 |
| Ventilation | 415 (69.28) | 133 (69.63) | 116 (69.05) | 54 (66.67) | 112 (70.44) | .945 |
| 28-day mortality (%) | 155.00 (25.88) | 33.00 (17.28) | 38.00 (22.62) | 25.00 (30.86) | 59.00 (37.11) | <.001 |
| ICU-stay-time (h) | 117.00 (65.20–284.18) | 95.92 (54.12–257.48) | 146.28 (75.90–351.87) | 104.78 (52.02–201.85) | 124.58 (59.73–280.98) | .006 |
| Laboratory tests | ||||||
| WBC (K/UL) | 15.30 (11.10, 21.10) | 15.40 (11.00, 21.00) | 15.30 (11.55, 19.05) | 14.80 (10.30, 21.10) | 16.00 (11.30, 22.70) | .625 |
| Lymphocytes (K/UL) | 1.25 (0.80, 1.90) | 1.34 (0.88, 1.97) | 1.19 (0.80, 1.70) | 1.13 (0.67, 1.96) | 1.25 (0.71, 2.14) | .353 |
| Neutrophils (K/UL) | 10.53 (6.26, 15.90) | 10.87 (6.38, 15.92) | 9.98 (6.36, 15.35) | 9.89 (5.55, 15.21) | 10.90 (5.83, 16.34) | .764 |
| Monocytes (K/UL) | 0.70 (0.40, 1.07) | 0.73 (0.38, 1.10) | 0.66 (0.40, 0.90) | 0.71 (0.36, 1.20) | 0.74 (0.46, 1.07) | .355 |
| Hemoglobin (g/dL) | 10.10 (8.80, 11.80) | 10.60 (9.20, 12.20) | 10.20 (8.80, 11.60) | 9.70 (8.40, 11.20) | 9.80 (8.70, 11.20) | .006 |
| CRP (mg/L) | 26.20 (4.70, 85.90) | 16.00 (3.80, 66.70) | 40.45 (4.45, 97.85) | 29.80 (6.40, 80.40) | 30.00 (6.00, 91.50) | .024 |
| Platelets (K/UL) | 199.00 (133.00, 270.00) | 206.00 (141.00, 285.00) | 217.50 (138.00, 274.50) | 179.00 (133.00, 258.00) | 180.00 (115.00, 249.00) | .043 |
| INR (s) | 1.20 (1.10, 1.40) | 1.10 (1.10, 1.40) | 1.20 (1.10, 1.70) | 1.10 (1.00, 1.20) | 1.20 (1.10, 1.40) | <.001 |
| PT (s) | 13.20 (11.90, 15.50) | 13.00 (12.10, 15.30) | 13.75 (12.35, 18.25) | 12.40 (11.50, 13.70) | 13.10 (11.70, 15.30) | <.001 |
| APTT (s) | 29.70 (26.10, 34.40) | 30.00 (26.10, 34.90) | 29.10 (26.10, 34.05) | 29.60 (26.90, 33.30) | 29.60 (26.20, 35.40) | .963 |
| ALT (u/L) | 26.00 (17.00, 52.00) | 26.00 (17.00, 42.00) | 27.00 (17.00, 57.50) | 26.00 (16.00, 49.00) | 25.00 (17.00, 59.00) | .745 |
| AST (u/L) | 31.00 (20.00, 64.00) | 30.00 (20.00, 56.00) | 30.00 (20.50, 67.00) | 27.00 (20.00, 48.00) | 32.00 (21.00, 71.00) | .735 |
| ALP (u/L) | 84.00 (65.00, 115.00) | 85.00 (65.00, 123.00) | 80.00 (63.00, 113.50) | 87.00 (70.00, 114.00) | 80.00 (65.00, 109.00) | .375 |
| Creatinine (mg/dL) | 1.00 (0.80, 1.40) | 0.90 (0.70, 1.20) | 1.05 (0.80, 1.50) | 1.00 (0.90, 1.30) | 1.10 (0.90, 1.80) | <.001 |
| Chloride (mg/dL) | 102.00 (99.00, 105.00) | 101.00 (98.00, 105.00) | 102.00 (99.00, 105.00) | 103.00 (100.00, 105.00) | 103.00 (99.00, 106.00) | .137 |
| Triglyceride (mg/dL) | 128.00 (88.00, 194.00) | 117.00 (81.00, 178.00) | 127.00 (87.00, 174.00) | 129.00 (92.00, 165.00) | 144.00 (105.00, 239.00) | .002 |
| Glucose (mg/dL) | 116.00 (96.00, 149.00) | 109.00 (93.00, 145.00) | 115.00 (94.00, 144.00) | 124.00 (100.00, 166.00) | 124.00 (100.00, 165.00) | .006 |
| Albumin (g/dL) | 3.00(1.20, 5.00) | 3.00 (1.80,4.70) | 3.00 (1.40,5.00) | 2.90 (1.90,4.30) | 2.90 (1.20,4.40) | .180 |
| Total protein (g/dL) | 5.30 (3.40,7.80) | 5.70 (4.00, 7.00) | 5.40 (3.60, 7.80) | 5.95 (3.60,7.00) | 4.80 (3.40,7.30) | .005 |
| Calcium (mmol/L) | 9.00 (8.50, 9.50) | 9.10 (8.70, 9.60) | 9.00 (8.50, 9.40) | 9.10 (8.50, 9.50) | 8.90 (8.30, 9.40) | .003 |
| Sodium (mmol/L) | 139.00 (136.00, 141.00) | 139.00 (136.00, 141.00) | 139.00 (136.00, 141.00) | 139.00 (137.00, 142.00) | 139.00 (136.00, 142.00) | .413 |
| Potassium (mmol/L) | 4.20 (3.90, 4.60) | 4.10 (3.80, 4.50) | 4.20 (3.90, 4.50) | 4.30 (3.90, 4.90) | 4.40 (3.90, 4.90) | <.001 |
| PH | 7.38 (7.31, 7.43) | 7.39 (7.34, 7.44) | 7.39 (7.34, 7.44) | 7.35 (7.29, 7.41) | 7.36 (7.29, 7.42) | <.001 |
| Lactate (mmol/L) | 1.60 (1.20, 2.70) | 1.50 (1.00, 2.40) | 1.60 (1.10, 2.55) | 1.70 (1.30, 3.50) | 1.80 (1.20, 3.40) | .008 |
| Treatments | ||||||
| Glucocorticoid (%) | 187.00 (31.22) | 53.00 (27.75) | 52.00 (30.95) | 26.00 (32.10) | 56.00 (35.22) | .514 |
| Vasoactive drug (%) | 271 (45.24) | 77 (40.31) | 74 (44.05) | 38 (46.91) | 82 (51.57) | .201 |
| Ventilation (%) | 415 (69.28) | 133 (69.63) | 116 (69.05) | 54 (66.67) | 112 (70.44) | .945 |
| Outcome | ||||||
| 28-day mortality (%) | 155.00 (25.88) | 33.00 (17.28) | 38.00 (22.62) | 25.00 (30.86) | 59.00 (37.11) | <.001 |
| ICU-stay-time (h) | 117.00 (65.20–284.18) | 95.92 (54.12–257.48) | 146.28 (75.90–351.87) | 104.78 (52.02–201.85) | 124.58 (59.73–280.98) | .006 |
BMI: body mass index; SAPS-II: Simplified Acute Physiology Score II; GCS: Glasgow Coma Scale; SOFA: Sequential Organ Failure Assessment; SBP: systolic blood pressure; DBP: diastolic blood pressure; MAP: mean arterial pressure; RR: respiratory rate; COPD: chronic obstructive pulmonary disease; WBC: white blood cell count; CRP: C-reactive protein; INR: international normalized ratio; PT: prothrombin time; APTT: activated partial thromboplastin time; ALT: alanine aminotransferase; AST: aspartate aminotransferase; ALP: alkaline phosphatase; PH: potential of hydrogen; NTIS: nonthyroidal illness syndrome.
Univariate analysis of 28-day ICU mortality among sepsis patients
As shown in Table 3, univariate Cox regression analysis identified age, ALP, SAPS III score, SOFA score, initial HR, and RR as significant predictors of 28-day mortality. Compared with Group 1, mortality risk was significantly elevated in Groups 3 and 4, whereas no significant difference was observed for Group 2. Among laboratory parameters, hemoglobin was inversely associated with mortality risk, indicating a protective effect, while elevated lactate levels were positively correlated with increased risk. Regarding therapeutic interventions, vasopressor use and longer duration of mechanical ventilation were linked to higher mortality, whereas diabetes showed no significant association with 28-day mortality. To assess multicollinearity among variables, VIF analysis was conducted. As detailed in Table 4, all variables had VIF values between 1.06 and 1.12, well below the accepted threshold of 5, indicating no significant multicollinearity and supporting their inclusion in subsequent multivariate analyses.
Table 3.
Univariate cox regression analysis of variables associated with 28-day mortality.
| Variable | HR | [95% CI] | p-value |
|---|---|---|---|
| Age | 1.016 | [1.01–1.03] | .002 |
| SAPS-II | 1.02 | [1.01–1.03] | <.001 |
| SOFA | 1.148 | [1.09–1.21] | <.001 |
| Heart rate | 1.012 | [1.01–1.02] | .001 |
| RR | 1.024 | [1.00–1.05] | .05 |
| Diabetes | 1.227 | [0.89–1.70] | .216 |
| P/F–NTIS stratification | |||
| 1 | |||
| 2 | 1.34 | [0.841–2.14] | .218 |
| 3 | 1.976 | [1.18–3.32] | .01 |
| 4 | 2.391 | [1.56–3.66] | <.001 |
| Hemoglobin | 0.876 | [0.81–0.95] | .001 |
| Lactate | 1.098 | [1.03–1.17] | .003 |
| ALP | 1.001 | [1.00–1.001] | .001 |
| Albumin (g/dL) | 0.574 | [0.43–0.77] | <.001 |
| Total protein (g/dL) | 0.705 | [0.48–1.03] | .069 |
| Glucocorticoid | 1.269 | [0.91–1.76] | .155 |
| Vasociactive | 1.399 | [1.02–1.92] | .037 |
| Ventilation | 0.704 | [0.51–0.98] | .035 |
SAPS-II: Simplified Acute Physiology Score II; SOFA: Sequential Organ Failure Assessment; RR: respiratory rate; ALP: alkaline phosphatase; NTIS: nonthyroidal illness syndrome.
Table 4.
VIF analysis of independent variables.
| Variable | VIF | 1/VIF |
|---|---|---|
| Age | 1.11 | 0.90 |
| SAPS-II | 1.10 | 0.91 |
| SOFA | 1.09 | 0.92 |
| Heart rate | 1.09 | 0.92 |
| Hemoglobin | 1.06 | 0.94 |
| Lactate | 1.11 | 0.90 |
| ALP | 1.06 | 0.94 |
| Albumin | 1.16 | 0.86 |
| Vasoactive drug | 1.12 | 0.89 |
| Ventilation | 1.11 | 0.90 |
VIF: variance inflation factor; SAPS-II: Simplified Acute Physiology Score II; SOFA: Sequential Organ Failure Assessment; ALP: alkaline phosphatase.
Association between 28-day mortality and P/F–NTIS stratification
We performed a multivariate Cox regression analysis to evaluate the independent impact of P/F–NTIS stratification on 28-day mortality in ICU patients with sepsis, as detailed in Table 5. In the unadjusted Model 1, compared to Group 1, both Group 3 (HR = 1.98, 95% CI [1.18–3.32], p = .01) and Group 4 (HR = 2.40, 95% CI [1.56–3.66], p < .001) exhibited a significantly higher risk of adverse outcomes. However, Group 2 (HR = 1.34, 95% CI [0.84–2.14], p = .218) did not show a statistically significant association. After adjusting for age, SOFA and SAPS-II in Model 2, the HRs for Group 3 (HR = 1.77, 95% CI [1.05–2.97], p = .033) and Group 4 (HR = 1.70, 95% CI [1.03–2.64], p = .019) remained statistically significant, though slightly attenuated. In Model 3, which incorporated additional laboratory indicators and vital signs, the association for Group 3 was no longer statistically significant (HR = 1.55, 95% CI [0.92–2.66], p = .100), while Group 4 remained a significant risk factor (HR = 1.70, 95% CI [1.08–2.69], p = .022). Overall, these findings suggest that higher P/F–NTIS stratification categories, especially Group 4, are independently associated with an increased risk of 28-day mortality, even after adjusting for age, SAPS-II score, SOFA score, HR, hemoglobin level, lactate level, ALP, use of vasoactive drugs, and mechanical ventilation.
Table 5.
Cox proportional hazard ratios for 28-day mortality.
| Variable | Model 1 | Model 2 | Model 3 | |||
|---|---|---|---|---|---|---|
| HR [95% CI] | p-value | HR [95% CI] | p-value | HR [95% CI] | p-value | |
| P/F–NTIS stratification | ||||||
| Group 1 | (Reference) | (Reference) | (Reference) | |||
| Group 2 | 1.34 [0.84–2.14] | .218 | 1.10 [0.68–1.76] | .708 | 1.22 [0.75–1.99] | .42 |
| Group 3 | 1.98 [1.18–3.32] | .01 | 1.77 [1.05–2.97] | .033 | 1.55 [0.91–2.66] | .10 |
| Group 4 | 2.40 [1.56–3.66] | <.001 | 1.70 [1.03–2.64] | .019 | 1.70 [1.08–2.69] | .022 |
NTIS: nonthyroidal illness syndrome; SAPS-II: Simplified Acute Physiology Score II; SOFA: Sequential Organ Failure Assessment; RR: respiratory rate; ALP: alkaline phosphatase.
Note: Model 1 was unadjusted.
Model 2 was adjusted for age, SOFA, SAPS-II.
Model 3 was adjusted for age, SOFA, SAPS-II, heart rate, RR, WBC, ALP, neutrophils, creatinine, hemoglobin, lactate, albumin ventilation, vasoactive drug.
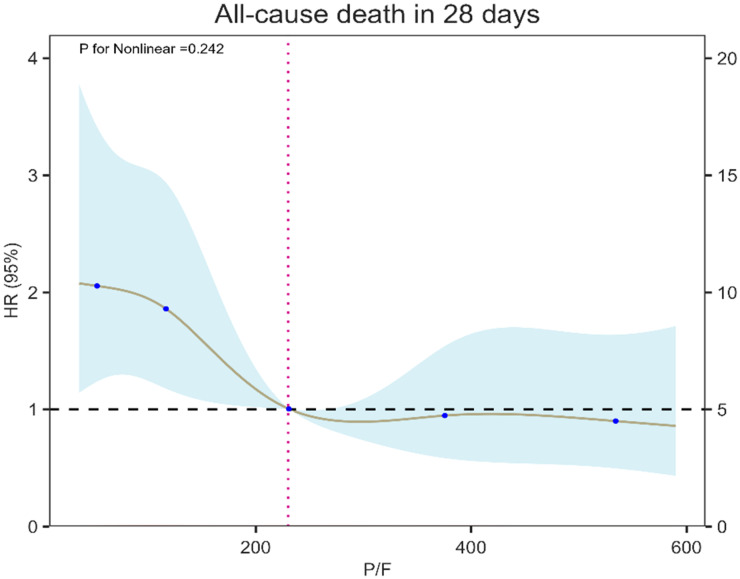
To explore the association between the P/F ratio and 28-day mortality, we performed an RCS analysis. The results, shown in Figure 2, revealed a nonlinear relationship between the P/F ratio and 28-day mortality (p for nonlinearity = .242). Specifically, the risk of death declined steeply as the P/F ratio increased up to approximately 200 mmHg, beyond which the curve flattened, indicating a plateau in mortality risk at higher P/F values.
Figure 2.
The RCS analysis for the nonlinear association between the P/F and 28-day mortality.
RCS: restricted cubic spline.
28-day mortality in different groups
As shown in Figure 3, the K–M survival analysis revealed significant differences among the four groups (log-rank test, p < .001).The survival probability showed a downward trend over time across all groups, with distinct separation between the curves. Group 1 exhibited the highest survival probability, while Groups 3 and 4 showed lower survival rates. The number at risk decreased over time in all groups, with Group 3 experiencing the steepest decline. These results suggest a statistically significant disparity in survival outcomes among the different groups.
Figure 3.
Kaplan–Meier survival analysis curves for 28-day mortality.
Discussion
This retrospective cohort study demonstrates that the combination of the P/F ratio and NTIS significantly enhances the prediction of 28-day mortality in patients with sepsis. By stratifying patients based on P/F levels and NTIS status, we identified a subgroup—those with P/F ≤ 200 mmHg and concurrent NTIS—that exhibited the highest mortality risk (37.11%). Even after adjusting for potential confounders, this subgroup remained an independent predictor of poor outcomes (HR = 1.70, 95% CI [1.08–2.69], p = .022), emphasizing the synergistic prognostic significance of impaired oxygenation and metabolic-endocrine dysfunction.
Our findings are consistent with prior research indicating that low P/F ratios are associated with increased sepsis-related mortality. Previous studies have shown that a lower P/F ratio at ICU admission is an independent risk factor for short-term mortality, with a threshold around 28.55 cmHg demonstrating prognostic value.17,18 Additionally, Bi et al. identified a U-shaped relationship between P/F and 28-day mortality in sepsis using the MIMIC-IV database, 19 suggesting that both hypoxemia and hyperoxia carry risks. In the present study, the RCS analysis revealed a nonlinear relationship between the P/F ratio and 28-day mortality. When the P/F ratio was below approximately 200 mmHg, the risk of death decreased rapidly with increasing P/F values, and this trend plateaued at higher levels. Although the nonlinearity test did not reach statistical significance (p = .242), the overall trend still highlighted that a lower P/F ratio was associated with a higher risk of 28-day mortality, reinforcing the prognostic relevance of impaired oxygenation in septic patients. This finding is consistent with previous studies that have demonstrated a strong association between reduced oxygenation and increased mortality in sepsis.
Furthermore, the results of the RCS analysis further support the rationale for stratifying based on the P/F ratio threshold of 200 mmHg, as this threshold effectively distinguishes between different mortality risk levels. However, as a standalone indicator, the P/F ratio has certain limitations. It primarily reflects pulmonary oxygenation but does not directly capture tissue oxygen delivery or microcirculatory status. 20 Moreover, its susceptibility to ventilator settings and individual variability may compromise its prognostic reliability. Integrating the P/F ratio with additional biomarkers may improve risk prediction and clinical decision-making in sepsis.
NTIS is commonly observed in critically ill patients, including those with sepsis, and is characterized by low T3 levels, variable TSH, and, in severe cases, elevated reverse T3 and reduced T4.21–23 These alterations are thought to represent an adaptive response to critical illness, aimed at conserving energy and mitigating catabolic processes.23–25 However, the clinical implications of NTIS remain controversial, as persistent thyroid dysfunction is often linked to worse outcomes. Currently, some studies support the use of exogenous thyroid hormone supplementation in septic patients with decreased T3 levels to maintain or restore normal thyroid function, which may have a positive effect on the development of the disease. 25 However, other studies have cautioned against the use of exogenous thyroid hormones. Whether patients with sepsis would benefit from the treatment of NTIS remains unknown. This question can only be answered through large-scale randomized controlled trials.
Interestingly, our study found that NTIS alone did not significantly increase mortality in patients with preserved oxygenation (P/F > 200 mmHg), suggesting that NTIS may function as a stress-adaptive response under less severe conditions. However, when NTIS coexisted with hypoxia, its prognostic impact was markedly amplified. This finding supports the hypothesis that the detrimental effects of NTIS are context-dependent, becoming more pronounced in the presence of impaired oxygenation.
From a physiological perspective, there may be a complex bidirectional regulatory relationship between NTIS and hypoxic conditions. On one hand, reduced T3 levels in NTIS can lead to respiratory muscle weakness, impairing ventilatory function and exacerbating hypoxia. 26 On the other hand, hypoxia can further suppress the hypothalamic–pituitary–thyroid axis, promote the release of inflammatory mediators such as IL-6, and induce oxidative stress, thereby inhibiting the peripheral conversion of T4 to T3 and facilitating the onset and progression of NTIS. 16 This mutually reinforcing mechanism may explain the significantly increased mortality observed when NTIS coexists with hypoxia.
The combined use of P/F ratio and NTIS offers a more comprehensive and robust tool for risk stratification in sepsis than either marker alone. The P/F ratio provides insights into cardiopulmonary function and oxygenation status, while NTIS reflects systemic metabolic and endocrine derangements. Together, they capture distinct but complementary aspects of sepsis pathophysiology, facilitating more accurate mortality risk prediction and potentially guiding early, individualized interventions.
We further explored potential confounders that may influence the observed associations. Nutritional status was assessed using total protein, albumin levels, and body mass index. Nonsurvivors had significantly lower albumin levels compared to survivors (median: 2.75 vs 3.00 g/dL, p < .001), and albumin was independently associated with reduced 28-day mortality (HR = 0.574, 95% CI [0.43–0.77], p < .001). This suggests that malnutrition may be an important mediator linking NTIS to adverse outcomes. Additionally, we evaluated the potential impact of glucocorticoid use, which may interfere with the hypothalamic–pituitary–thyroid axis. Although not statistically significant between groups (p = .162), this factor warrants consideration in clinical interpretation.
This study has several limitations. First, its retrospective design and reliance on the MIMIC-IV database may introduce potential biases and limit the ability to draw causal inferences. Second, the lack of standardized diagnostic criteria for NTIS may affect the reproducibility of our findings and comparability with other studies. Third, although multiple important confounders were adjusted for, residual confounding cannot be entirely excluded. Most importantly, a substantial number of patients were excluded due to missing thyroid function parameters and/or P/F ratio data. While these exclusions ensured the internal validity of the analyses and accurate patient stratification, they inevitably introduced selection bias, which may affect the representativeness of the study cohort and the generalizability of the results. Future multicenter prospective studies with more complete datasets are warranted to externally validate our findings and further strengthen their robustness and applicability.
Conclusions
The P/F–NTIS stratification effectively predicts the 28-day mortality in sepsis patients, especially in those with P/F ≤ 200 mmHg and NTIS, where the mortality risk is significantly higher. This stratification approach offers new insights for risk assessment and personalized interventions in sepsis. Future research should explore its application in different clinical contexts and potential intervention targets.
Acknowledgements
The author gratefully acknowledges Director Jian Wan for his support. Thanks are also extended to the MIT Laboratory for Computational Physiology for maintaining the MIMIC-IV database and providing public access to this valuable resource.
Footnotes
Ethical considerations: This study used deidentified data from the publicly available MIMIC-IV database. Ethical approval and individual patient consent were not required, as confirmed by the Institutional Review Board of the Beth Israel Deaconess Medical Center. Access to the database was approved after completion of the CITI “Data or Specimens Only Research” course (Record ID: 68950718).
Author contributions: Wei Song: conceptualization, study design, data acquisition/analysis/interpretation, manuscript drafting, visualization, project administration, final approval, and accountability; Jian Wan: supervision, funding acquisition, critical review/editing, project oversight, correspondence, final approval, and accountability; Conghui Fan, Song Chen, Qian Zhang, and Fei Zhong: investigation, data collection, methodology development, validation, formal analysis, resources provision, critical review, final approval, and accountability. These authors contributed equally with the first author; Tao Zhang: software implementation, data processing, visualization, final approval, and accountability; Ye Yuan: literature synthesis, figure design, editorial support, final approval, and accountability; Xueke Wu: technical implementation, data verification, final approval, and accountability; Yijiong Zhang: statistical modeling, results interpretation, final approval, and accountability; Zhen Han: clinical expertise, quality assurance, final approval, and accountability; Lixue Wu: data curation, ethical compliance, final approval, and accountability; Qingzhong Zhao: strategic supervision, clinical trial design, manuscript critique, final approval, and accountability.
Funding: This study was supported by the following programs: Project of Clinical Outstanding Discipline Construction in Shanghai Pudong New Area, Discipline Leader Training Program of the Pudong New Area Health System, Shanghai, Talent Development Program Plan of the Qihang Program at Pudong New District People's Hospital, Shanghai, The Project of Key Medical Discipline Group Construction in Shanghai Pudong New Area (Grant Nos. PWYgy2021-09, PWRd2024-12, PRYQH202302, and PWZxq2022-13).
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
References
- 1.Wasyluk W, Wasyluk M, Zwolak A. Sepsis as a pan-endocrine illness-endocrine disorders in septic patients. J Clin Med 2021; 10: 2075. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Cajander S, Kox M, Scicluna BP, et al. Profiling the dysregulated immune response in sepsis: overcoming challenges to achieve the goal of precision medicine. Lancet Respir Med 2024; 12: 305–322. [DOI] [PubMed] [Google Scholar]
- 3.Zhang M, Duan M, Zhi D, et al. Risk factors for 28-day mortality in patients with sepsis-related myocardial injury in intensive care units. J Int Med Res 2021; 49: 3000605211004759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Black RE, Cousens S, Johnson HL, et al. Global, regional, and national causes of child mortality in 2008: a systematic analysis. Lancet (London, England) 2010; 375: 1969–1987. [DOI] [PubMed] [Google Scholar]
- 5.Park JS, Kim SJ, Lee SW, et al. Initial low oxygen extraction ratio is related to severe organ dysfunction and high in-hospital mortality in severe sepsis and septic shock patients. J Emerg Med 2015; 49: 261–267. [DOI] [PubMed] [Google Scholar]
- 6.Seymour CW, Liu VX, Iwashyna TJ, et al. Assessment of clinical criteria for sepsis: for the third international consensus definitions for sepsis and septic shock (sepsis-3). Jama 2016; 315: 762–774. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lambden S, Laterre PF, Levy MMet al. et al. The SOFA score-development, utility and challenges of accurate assessment in clinical trials. Critical Care (London, England) 2019; 23: 74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Bi C, Zhu M, Ni C, et al. Predictive value of oxygenation index at intensive care unit admission for 30-day mortality in patients with sepsis. Zhonghua wei Zhong Bing ji jiu yi xue 2025; 37: 111–117. [DOI] [PubMed] [Google Scholar]
- 9.Cornu MG, Martinuzzi ALN, Roel P, et al. Incidence of low-triiodothyronine syndrome in patients with septic shock. Rev Bras Ter Intensiva 2020; 32: 514–520. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Ilera V, Delfino LC, Zunino A, et al. Correlation between inflammatory parameters and pituitary-thyroid axis in patients with COVID-19. Endocrine 2021; 74: 455–460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Langouche L, Lehmphul I, Perre SV, et al. Circulating 3-T1AM and 3,5-T2 in critically ill patients: a cross-sectional observational study. Thyroid: Official Journal of the American Thyroid Association 2016; 26: 1674–1680. [DOI] [PubMed] [Google Scholar]
- 12.Zhang X, Liu L, Ma X, et al. Clinical significance of non-thyroidal illness syndrome on disease activity and dyslipidemia in patients with SLE. PLoS One 2020; 15: e0231622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.von Elm E, Altman DG, Egger M, et al. The strengthening the reporting of observational studies in epidemiology (STROBE) statement: guidelines for reporting observational studies. J Clin Epidemiol 2008; 61: 344–349. [DOI] [PubMed] [Google Scholar]
- 14.Singer M, Deutschman CS, Seymour CW, et al. The third international consensus definitions for sepsis and septic shock (sepsis-3). Jama 2016; 315: 801–810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ranieri VM, Rubenfeld GD, Thompson BT, et al. Acute respiratory distress syndrome: the Berlin definition. Jama 2012; 307: 2526–2533. [DOI] [PubMed] [Google Scholar]
- 16.Ning N, Li J, Sun W, et al. Different subtypes of nonthyroidal illness syndrome on the prognosis of septic patients: a two-centered retrospective cohort study. Front Endocrinol (Lausanne) 2023; 14: 1227530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.de Jonge E, Peelen L, Keijzers PJ, et al. Association between administered oxygen, arterial partial oxygen pressure and mortality in mechanically ventilated intensive care unit patients. Critical Care (London, England) 2008; 12: R156. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ihle JF, Bernard S, Bailey MJ, et al. Hyperoxia in the intensive care unit and outcome after out-of-hospital ventricular fibrillation cardiac arrest. Crit Care Resusc 2013; 15: 186–190. [PubMed] [Google Scholar]
- 19.Bi H, Liu X, Chen C, et al. The PaO(2)/FiO(2) is independently associated with 28-day mortality in patients with sepsis: a retrospective analysis from MIMIC-IV database. BMC Pulm Med 2023; 23: 87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lee CU, Jo YH, Lee JH, et al. The index of oxygenation to respiratory rate as a prognostic factor for mortality in sepsis. Am J Emerg Med 2021; 45: 426–432. [DOI] [PubMed] [Google Scholar]
- 21.Angelousi AG, Karageorgopoulos DE, Kapaskelis AMet al. et al. Association between thyroid function tests at baseline and the outcome of patients with sepsis or septic shock: a systematic review. Eur J Endocrinol 2011; 164: 147–155. [DOI] [PubMed] [Google Scholar]
- 22.Castro I, Quisenberry L, Calvo RM, et al. Septic shock non-thyroidal illness syndrome causes hypothyroidism and conditions for reduced sensitivity to thyroid hormone. J Mol Endocrinol 2013; 50: 255–266. [DOI] [PubMed] [Google Scholar]
- 23.Siampa VN, Abadi S, Aman AM, et al. Association between severity of sepsis and thyroid function profile. Acta Bio-medica: Atenei Parmensis 2023; 94: e2023239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Contreras-Jurado C, Alonso-Merino E, Saiz-Ladera C, et al. The thyroid hormone receptors inhibit hepatic interleukin-6 signaling during endotoxemia. Sci Rep 2016; 6: 30990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wang Y, Sun F, Hong Get al. et al. Thyroid hormone levels as a predictor marker predict the prognosis of patients with sepsis. Am J Emerg Med 2021; 45: 42–47. [DOI] [PubMed] [Google Scholar]
- 26.Bloise FF, Santos AT, de Brito J, et al. Sepsis impairs thyroid hormone signaling and mitochondrial function in the mouse diaphragm. Thyroid: Official Journal of the American Thyroid Association 2020; 30: 1079–1090. [DOI] [PubMed] [Google Scholar]