Abstract
It has previously been shown that passage of Vibrio cholerae through the human intestine imparts a transient hyperinfectious phenotype that may contribute to the epidemic spread of cholera. The mechanism underlying this human-passaged hyperinfectivity is incompletely understood, in part due to inherent difficulties in recovering and studying organisms that are freshly passed in human stool. Here, we demonstrate that passage of V. cholerae through the infant mouse intestine leads to an equivalent degree of hyperinfectivity as passage through the human host. We have used this infant mouse model of host-passaged hyperinfectivity to characterize the timing and the anatomic location of the competitive advantage of mouse-passaged V. cholerae as well as the contribution of three type IV pili to the phenotype.
Vibrio cholerae exists primarily as a free-living organism in estuarine environments. Pathogenic strains, however, are able to colonize the human small intestine and release cholera toxin, resulting in a secretory diarrhea that can be fatal in the absence of proper treatment (15). V. cholerae is unique among the bacterial diarrheal pathogens in its ability to cause worldwide pandemics of disease. Accordingly, the factors that facilitate the organism's departure from the aquatic environment and promote the epidemic spread of human disease are of great interest.
Environmental and climactic factors have been shown to contribute to the epidemic spread of V. cholerae (22). In areas where it is endemic, cholera occurs in recurrent peaks at the end of the monsoon season; these peaks have been linked to water temperature changes and zooplankton blooms (17) as well as to the prevalence of vibriophages in the environment (8). The genetic composition of the bacteria itself may also play an important role in the transition from the aquatic environment to a cycle of epidemic disease. In particular, the acquisition of key virulence genes by horizontal transfer events is important in the evolution of pathogenic strains of V. cholerae (28). Such a genetic transfer event preceded the 1992 emergence of a novel serogroup of epidemic cholera, V. cholerae O139, which arose through the acquisition of the wbf gene cluster from a nontoxigenic V. cholerae isolate (26). Interestingly, recent studies of the transcriptional profile of organisms present in clinical samples suggest that the human upper intestine may be a particularly suitable niche for the horizontal gene transfer events that may give rise to V. cholerae strains with epidemic potential (16).
Passage of V. cholerae through the human host confers a transient phenotype of enhanced transmissibility that also may contribute to epidemic spread of the disease (20). The mechanism by which passage through the human intestine leads to V. cholerae hyperinfectivity is unknown, but the physiologic basis may be multifactorial and related to any of the key steps in V. cholerae infection, such as passage through the host gastric acid barrier, adherence to and colonization of the intestinal epithelium, or replication within the intestine. Alternatively, human-passaged organisms may express novel virulence factors that facilitate evasion of host immunity.
Some possible mechanisms of transient V. cholerae hyperinfectivity have been explored. For instance, acid-adapted strains are able to outcompete wild-type strains by approximately eightfold in the infant mouse model of cholera infection (18). Surprisingly, more rapid replication in the mouse intestine is responsible for this competitive advantage rather than enhanced survival of acid-adapted organisms during passage through the gastric barrier (1). Alterations in chemotaxis also affect the infectivity of V. cholerae (9). Paralogs of the chemotaxis genes cheW and cheR are repressed in V. cholerae in freshly passed human stool compared to in vitro-grown organisms, suggesting a role for altered chemotaxis in host-passaged hyperinfectivity (20). In an infant mouse model of V. cholerae infection, the ability of nonchemotactic mutants to outcompete wild-type in vitro-grown organisms is related to the direction of flagellar rotation and is associated with a broader distribution of organisms throughout the small intestine (4). The increased expression of essential colonization factors, such as the toxin-coregulated pilus (TCP), a type IV pilus, may also be associated with the hyperinfectious phenotype of host-passaged V. cholerae, since wild-type organisms greatly outcompete tcpA mutants in the infant mouse model of cholera (6, 19). Together, these results suggest that a number of potential mechanisms may contribute to host-passaged hyperinfectivity, although the specific mechanisms that mediate this phenomenon remain undefined.
To facilitate a better understanding of human-passaged hyperinfectivity, we examined whether the hyperinfectious phenotype previously documented in organisms recovered from fresh human stool could be reproduced using the infant mouse model of V. cholerae infection. Here, we show that passage of bacteria through the infant mouse confers hyperinfectivity equivalent to that of organisms found in human stool. We also characterize the intestinal population dynamics of hyperinfectious mouse-passaged bacteria and assess the contribution of three V. cholerae type IV pili, including TCP, mannose-sensitive hemagglutinin (MshA), and PilA, to hyperinfectivity using this model.
MATERIALS AND METHODS
Bacterial strains.
The strains used in this study are listed in Table 1 (10). All of the strains are derivatives of V. cholerae O1 El Tor N16961, the only fully sequenced clinical isolate of V. cholerae (12). Bacterial strains were grown in Luria-Bertani (LB) broth with aeration at 37°C for 16 to 18 h for stationary-phase bacteria and 3 to 4 h for exponential-phase bacteria. Antibiotics were used at the following concentrations: ampicillin, 50 μg/ml; streptomycin, 50 μg/ml. To induce in vitro expression of V. cholerae genes, AKI growth conditions were performed as described previously (14). Briefly, the strain was grown overnight in LB medium at 37°C on a roller shaker and then subcultured into 10 ml of medium containing 1.5% Bacto peptone, 0.4% yeast extract, 0.5% sodium chloride, and 0.3% sodium bicarbonate. Strains were then grown without shaking for 4 to 6 h at 37°C and transferred to 100-ml flasks for a subsequent 16 h of growth. Confirmation of expression of the B subunit of cholera toxin under AKI conditions was confirmed by ganglioside GM1 enzyme-linked immunosorbent assay (24).
TABLE 1.
Bacterial strains used in this study
| Strain | Relevant genotype or phenotypea | Reference |
|---|---|---|
| N16961 | El Tor biotype, wild type, Smr | Laboratory strain |
| KFV10 | N16961; Smr; ΔlacZ | 9 |
| KFV11 | N16961; Smr; ΔmshA | 9 |
| KFV6 | N16961; Smr; ΔpilA | 9 |
| KFV33 | N16961; Smr; ΔtcpA | 9 |
| MAV2 | KFV33; Smr, Ampr ΔtcpA lacZ::pGP704 | This study |
Amp, ampicillin; Sm, streptomycin.
Knockout of the lacZ gene in a tcpA mutant strain of V. cholerae (KFV33).
In order to conduct competition assays in a tcpA mutant background, Escherichia coli SM10λpir harboring a pGP704 vector carrying a 1,000-bp-long fragment of the amplified lacZ gene was mated with V. cholerae KFV33 (N16961ΔtcpA) (10) to create a lacZ tcpA double mutant strain. Exconjugants that were LacZ negative were selected as streptomycin- and ampicillin-resistant colonies. PCR analysis was performed to confirm correct integration of the plasmid.
Recovery of mouse-passaged and human stool-passaged V. cholerae.
Mouse-passaged bacteria were prepared by intragastric inoculation of 4- to 5-day-old suckling CD-1 mice or Swiss Webster mice with approximately 106 CFU of the appropriate strain. Organisms were recovered from the whole mouse gut by mechanical homogenization performed 20 to 24 h after inoculation. For recovery of V. cholerae from human stool, 100 ml of freshly passed rice water stool was collected from cholera patients admitted to the International Centre for Diarrheal Disease Research hospital in Dhaka, Bangladesh (ICDDR,B). Stool specimens that were positive for the characteristic darting movements of vibrios on dark-field microscopy were then tested for motility inhibition by a monoclonal antibody specific to V. cholerae O1 (23). Stool samples were centrifuged at 3,000 × g for 5 min to remove particulate matter, and then the bacteria were pelleted by high-speed centrifugation, resuspended in phosphate-buffered saline, and used immediately for competition assays. Confirmation of the infecting strain was performed by overnight culture on taurocholate-tellurite-gelatin agar and evaluation with V. cholerae O1 serotype-specific monoclonal antibodies (23). Human studies approval was obtained from the Research and Ethical Review Committees of the ICDDR,B and the Institutional Review Board of the Massachusetts General Hospital.
Competition assays.
Suckling CD-1 mice have been used extensively in V. cholerae competition assays (27). Germ-free Swiss and Swiss Webster mice models have been developed for evaluating vaccine strains of V. cholerae (5, 7), and suckling Swiss Webster mice have also been used for V. cholerae competition assays (20). Here, competition assays between different strains of V. cholerae were performed in 4- to 5-day-old suckling CD-1 mice (for competition assays using mouse-passaged organisms performed in the United States) or Swiss Webster mice (for competition assays using organisms freshly passed in human stool performed in Bangladesh) as previously described (20). Due to local availability, different strains of mice were used in the United States and in Bangladesh. For the competition with in vitro-grown organisms, human- or mouse-passaged bacteria were mixed with a 1:1,000 dilution of an in vitro-grown strain to achieve an approximate 1:1 input ratio. A total of 50 μl of the mixed strains, representing approximately 106 CFU, was administered intragastrically to suckling mice by gavage. After 20 to 24 h, the animals were euthanized and the small intestine was removed, homogenized, and plated on LB medium supplemented with 40 μg/ml of 5-bromo-4-chloro-3-indolyl-d-galactoside to allow for counting of the two competing strains, which were marked as LacZ+ and LacZ−. The ratio of the two strains in the intestine (output ratio) was corrected for the ratio of the two strains in the inoculum (input ratio), and results were expressed as the competitive index (CI) of the two strains examined. Experiments in which the input ratio differed by ≥10-fold from 1:1 were thrown out; no significant effect of <10-fold variability in the input ratio on the competitive index was observed. We also directly competed mouse-passaged against human-passaged organisms using the same technique described above.
Determination of intestinal population dynamics.
Following coinoculation of mouse-passaged and in vitro strains as described above, the small intestine was harvested at 1, 3, 5, 10, and 24 h after inoculation and plated to determine the ratios of LacZ+ and LacZ− colonies. To evaluate the distribution of individual strains in the small intestine, single strains were inoculated into suckling CD-1 mice by gavage, and mice were sacrificed at 5 and 24 h. The small intestine was removed and cut into five segments of equal length. Each of these five segments was mechanically homogenized and plated as described above to determine the percentage of total colonies in each individual segment.
Determination of ID50.
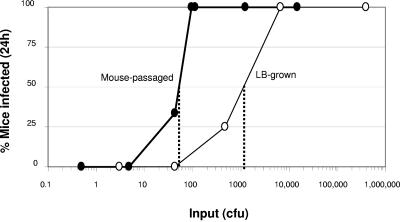
Four- to five-day-old suckling CD-1 mice were inoculated intragastrically with V. cholerae inocula of different strains, ranging from approximately 100 to 106 CFU. At 24 h postinoculation, the small intestines were removed and homogenized, and serial dilutions were plated to determine whether the inoculum had led to infection of the mouse. The lower limit of detection was approximately 10 CFU, and values equal to or greater than this were considered positive for infection. The infectious dose required to infect 50% of mice (ID50) was determined graphically by using the curves in Fig. 3.
FIG. 3.
Determination of ID50s of mouse-passaged V. cholerae (thick line with filled circles) and of V. cholerae grown to stationary phase in LB (thin line with open circles). Dotted lines represent the calculated ID50s. Each data point represents the mean result from at least three mice. The ID50 of mouse-passaged V. cholerae differed significantly from that of organisms grown to stationary phase (P < 0.001, chi-squared test).
Quantitative RT-PCR of tcpA mRNA.
RNA was extracted using Trizol (Life Technologies, Gaithersburg, MD). RNA samples were treated with DNase to remove contaminating DNA on an RNeasy column (QIAGEN, Valencia, CA), and reverse transcription (RT) was performed using the Reverse Transcription System (Promega, Madison, WI). Quantitative RT-PCRs were done using the TaqMan system (Applied Biosystems, Foster City, CA) and an Opticon 2 Continuous Fluorescence Detector (MJ Research, Waltham, MA). Primers and probes for tcpA and V. cholerae 16S RNA were designed using Primer Express (Applied Biosystems, Foster City, CA). Real-time PCRs were performed in a final volume of 25 μl containing 1× TaqMan Universal Master mix (Applied Biosystems, Foster City, CA), 900 nM forward and reverse TaqMan primers, and 250 nM TaqMan probe. Primers and probes were purchased from Applied Biosystems. Reactions were performed in MicroAmp Optical 96-well plates (Applied Biosystems, Foster City, CA) with the following cycle parameters: 50°C for 2 min, 95°C for 10 min, 95°C for 15 s, 60°C for 1 min. Validation experiments were performed for all TaqMan probe and primer sets, and these showed a linear relationship between the cycle threshold, Ct, and the logarithm of the template amount (genomic DNA), as expected. To control for genomic DNA contamination, reactions without reverse transcriptase were performed. Relative expression levels in the different samples were calculated by using the comparative Ct method with 16S RNA as the internal reference for normalization.
RESULTS
Passage through the infant mouse gut imparts a hyperinfectious phenotype in V. cholerae that is equivalent to human-passaged hyperinfectivity.
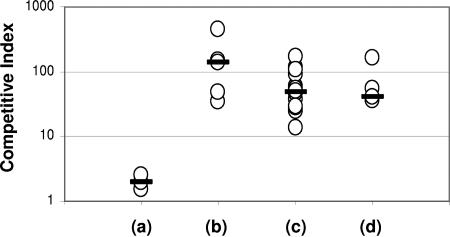
We used the growth of V. cholerae in vivo in an infant mouse to assess whether this could be used to model the hyperinfectious state that has been observed in organisms present in human stool. The sequenced wild-type reference strain of V. cholerae O1 El Tor N16961 (12) and a lacZ mutant of this strain were used in these experiments (Table 1). To ensure that there was no effect of the lacZ mutation itself on colonization, we first assessed the competitive index (CI) of the wild-type and lacZ mutant V. cholerae strains grown together in the CD-1 infant mouse intestine. No competitive advantage was observed for either strain (Fig. 1a). Next, we infected the wild-type strain into an infant mouse and recovered mouse-passaged organisms after growth overnight in vivo. These organisms were then competed with the lacZ mutant strain, grown overnight in LB broth in a second infant mouse, to determine the CI. As shown in Fig. 1b, the mouse-passaged strain outcompeted the in vitro-grown lacZ mutant strain by greater than 100-fold, consistent with the hyperinfectious phenotype previously seen in organisms recovered directly in human stool (20). We repeated this experiment using the lacZ mutant strain grown in the infant mouse and the wild-type strain grown in vitro. The mouse-passaged organisms retained an approximately 100-fold competitive advantage over the in vitro-grown organisms, demonstrating that the lacZ mutation had no intrinsic effect on the infectivity of V. cholerae (data not shown).
FIG. 1.
Competition assays in infant mice between LacZ+ V. cholerae N16961 and LacZ− V. cholerae KFV10 grown in various conditions prior to competition in CD-1 infant mice. (a) LacZ+ versus LacZ− V. cholerae, both grown to stationary phase in LB; (b) mouse-passaged LacZ+ V. cholerae versus LacZ− V. cholerae grown to stationary phase in LB; (c) mouse-passaged LacZ+ V. cholerae versus LacZ− V. cholerae grown to exponential phase in LB; (d) mouse-passaged LacZ+ V. cholerae versus LacZ− V. cholerae grown overnight in AKI conditions. Each data point represents the competitive index (CI) from a single animal. The flat bars indicate the median of the CIs for each experiment. By the unpaired t test, the results of experiments b to d each differed significantly from those for experiment a (P < 0.04).
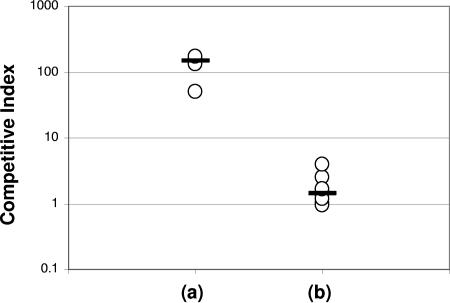
We next collected V. cholerae directly from freshly passed human stool and competed these organisms with V. cholerae grown to stationary phase in LB broth and with bacteria harvested from Swiss Webster mice. As previously observed (20), human-passaged organisms outcompeted in vitro-grown organisms by approximately 100-fold in Swiss Webster mice (Fig. 2a). Next, we directly compared the infectivity of mouse-passaged bacteria with that of organisms recovered from freshly passed human stool. The stool-harvested and mouse-passaged organisms demonstrated equivalent levels of competition (median CI, 1) (Fig. 2b). This finding supports our use of the infant mouse model for the study of host-induced hyperinfectivity.
FIG. 2.
Competition assays in Swiss Webster infant mice between wild-type V. cholerae freshly passed in human cholera stool and LacZ− N16961 V. cholerae, either (a) grown in LB broth to stationary phase or (b) harvested from infant mouse intestine. Each data point represents the competitive index (CI) from a single animal. The flat bars indicate the median of the CIs for each experiment. By the unpaired t test, the results of experiment b differed significantly from those for experiment a (P = 0.006).
Characterization of mouse-passaged hyperinfectivity.
Since the growth phase of host-passaged organisms is undefined, we assessed the CI of mouse-passaged organisms compared to organisms grown in vitro to exponential phase. Although we observed a slight decrease in the competitive advantage of the mouse-passaged organisms when competed with the organisms grown to exponential phase compared to those grown to stationary phase (Fig. 1c; median CI, 48), the hyperinfectious phenotype of the mouse-passaged organisms was preserved. This suggests that the hyperinfectious phenotype of the mouse-passaged bacteria is not dependent solely on growth phase.
We also assessed whether growth in in vitro conditions (AKI) that induce expression of V. cholerae virulence factors (14) would result in a hyperinfectious phenotype. We competed AKI-grown bacteria with mouse-passaged bacteria and with the organism grown to stationary phase in LB media. Although we observed that V. cholerae grown in AKI conditions outcompeted organisms grown to stationary phase in LB (median CI, 20), the mouse-passaged organisms still substantially outcompeted the organisms grown in AKI conditions (median CI, 41), as shown in Fig. 1d. This suggests that there are additional factors involved in the hyperinfectious phenotype following growth in the mouse intestine that are not induced during in vitro growth in AKI conditions.
We next verified that the hyperinfectious phenotype was transient and reversible following growth in the mouse intestine rather than the result of a heritable mutation selected in the mouse. Organisms were passaged in an infant mouse and then subsequently grown overnight in LB media and competed with organisms grown overnight in LB media; the hyperinfectious phenotype was lost after subsequent overnight growth in LB media (the mean CI was 1.4 for two mice).
We utilized a different assay of infectivity, determination of the ID50, to verify the hyperinfectivity of mouse-passaged bacteria compared to in vitro-grown bacteria (Fig. 3). The ID50 of the in vitro-grown bacteria was more than 1 log10 higher than that of the mouse-passaged bacteria, consistent with the results of the competition assays between host-passaged and in vitro-grown bacteria.
Intestinal survival and distribution of mouse-passaged bacteria compared to in vitro-grown organisms.
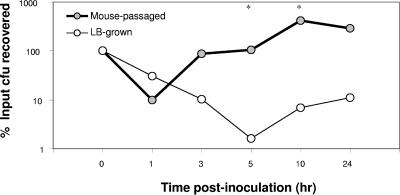
We assessed the intestinal survival of mouse-passaged and in vitro-grown V. cholerae at various time points after coinfection of the infant mouse (Fig. 4). In both cases, the majority of the inoculating organisms was killed in the first hour after infection. However, by 3 h postinfection, mouse-passaged bacteria had recovered to the starting inoculum. In contrast, replication of the in vitro-grown bacteria was not apparent in the mouse until 5 to 10 h after inoculation. This suggests that both strains were similarly susceptible to initial host bactericidal mechanisms but that the mouse-passaged bacteria were able to more rapidly replicate and establish successful colonization of the mouse intestine following infection.
FIG. 4.
Percent of input CFU recovered from the mouse small intestine at various time points following inoculation of either mouse-passaged V. cholerae or V. cholerae grown to stationary phase in LB media. Each data point represents the mean of the results from at least two mice. Data points marked with an asterisk differed significantly (P < 0.05, independent samples t test).
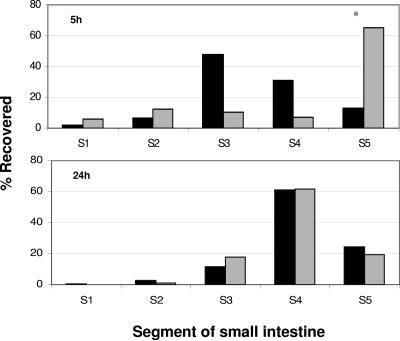
To characterize the location of the competitive advantage of mouse-passaged bacteria over in vitro-grown bacteria, we analyzed the distribution of organisms colonizing five different segments of the small intestine at 5 h and 24 h postinoculation in single-strain experiments (Fig. 5). At 5 h postinoculation, mouse-passaged bacteria were present in the highest numbers in the more proximal regions of the small intestine (segments 3 and 4) compared to in vitro-grown bacteria, which were primarily found in the most distal segment. By 24 h postinoculation, the distributions of the mouse-passaged and in vitro-grown bacteria were essentially indistinguishable, with the majority of the organisms localized to the most distal two segments. Previous work has shown that at 24 h postinfection, V. cholerae preferentially colonizes the distal segments of the small intestine (2). Our results suggest that mouse-passaged V. cholerae can both replicate more rapidly and preferentially colonize the more proximal segments of the small intestine in the early phases of infection.
FIG. 5.
Distribution of V. cholerae recovered from different intestinal segments 5 h and 24 h after inoculation, expressed as a percentage of total CFU recovered. The distribution of mouse-passaged V. cholerae is shown in dark bars, and the distribution of V. cholerae grown to exponential phase in LB is shown in gray bars. The total CFUs recovered were as follows: mouse passaged, 5 h (9.1 × 106); LB grown, 5 h (8.2 × 104); mouse passaged, 24 h (4.3 × 106); LB grown, 24 h (9.0 × 106). Each data point represents the mean of the results from at least two mice. Data points marked with an asterisk differed significantly (P < 0.05, independent samples t test).
Role of type IV pili in the hyperinfectious phenotype.
Given the importance of type IV pili as colonization factors in enteric pathogens (11, 13), we analyzed V. cholerae strains mutated in mshA and pilA (Table 1) in our infant mouse model of hyperinfectivity. We found that these mutants demonstrated a competitive advantage following mouse passage that was equivalent to the wild-type strain, suggesting that MshA and PilA are not necessary for the hyperinfectious phenotype (median CIs, 31 and 10, respectively).
Because TCP is an essential colonization factor, we also compared the ID50 of a mouse-passaged tcpA mutant strain with that of the same strain grown in vitro to determine if a tcpA mutant strain demonstrates host-passaged hyperinfectivity. We found no significant difference in the ID50 of a tcpA mutant strain grown in vitro or passaged through the mouse intestine, suggesting that mutation of tcpA reduced development of the hyperinfectious phenotype. The ID50 of the tcpA mutant, regardless of whether it was grown in vitro or was mouse passaged, was approximately 3 log10 greater than that of the wild-type strain.
To further analyze expression of tcpA in mouse-passaged compared to in vitro-grown organisms, we used quantitative RT-PCR specific for this transcript. The level of tcpA transcript was more than 100-fold higher in organisms recovered following passage through the mouse compared to in vitro growth to stationary phase in LB media. By 24 h after inoculation, however, expression of tcpA in vibrios recovered from mice infected with in vitro-grown organisms had increased to levels comparable to those in vibrios recovered from mice infected with the mouse-passaged strain.
DISCUSSION
Understanding the bacterial factors that contribute to the epidemic spread of cholera may lead to novel approaches for disease containment. Passage of V. cholerae through the environment of the human intestine imparts a transient hyperinfectivity for the next host. This hyperinfectivity is maintained for at least 5 h after passage into the environment (20), yet the mechanism of this phenomenon has not been completely defined. Here, we have demonstrated that passage through the infant mouse confers a hyperinfectious phenotype that is equivalent to that conferred by passage through the human intestine. Because of this, we propose that the infant mouse model of V. cholerae infection can be used to further characterize the mechanism of this transient hyperinfectious state.
Our use of the infant mouse model also provides insight into the mechanism of host-passaged hyperinfectivity. We have shown that growth phase alone does not account for the ability of host-passaged bacteria to outcompete in vitro-grown organisms, since host-passaged bacteria are able to substantially outcompete bacteria grown in vitro to both stationary and exponential phases. Furthermore, we have shown that hyperinfectivity is not due to a genetic mutation selected for during passage through the intestinal environment. Rather, hyperinfectivity is a transient phenotype that may relate to adaptation of the organism to substrates particular to the host (21, 25). The particular factors that mediate this transient phenotype merit further study. Our results suggest that enhanced tolerance for passage through the gastric acid barrier is unlikely to be one of these factors, since host-passaged bacteria are not more resistant to the initial killing events that occur in the earliest stage of infection (within 1 h). Our results do suggest that host-passaged bacteria preferentially colonize the more proximal regions of the small intestine and replicate more quickly in the intestine than in vitro-grown bacteria. Hence, factors relating to colonization and adaptation to characteristics of the intestinal environment, such as anaerobiasis or nutrient limitation (3, 20), may play a role in mediating hyperinfectivity.
Because our results suggested a possible contribution of colonization factors to the hyperinfectious phenotype, we evaluated the effect of mutations in three of the known type IV pili of V. cholerae in our infant mouse model of hyperinfectivity. MshA and PilA appeared to have no effect on the hyperinfectious phenotype; however, deletion of tcpA resulted in loss of the difference in ID50 between in vitro-grown and mouse-passaged organisms. This suggests that appropriate expression of tcpA may influence the hyperinfectious phenotype and is consistent with our observation that tcpA transcripts are >100-fold more highly expressed in host-passaged than in in vitro-grown organisms. Nevertheless, given the essential role of TCP as a colonization factor, it is difficult to conclude that altered expression of tcpA is solely responsible for the hyperinfectious phenotype observed in host-passaged bacteria. Because the tcpA mutant strain is severely attenuated in its ability to colonize the intestine, it is possible that additional factors that modulate the hyperinfectivity of host-passaged bacteria are not apparent in analysis of this mutant.
In summary, we have established that the infant mouse can be used to model human-passaged hyperinfectivity of V. cholerae and that TcpA may play a necessary role in this phenotype. Further exploration of the effect of mutations in chemotaxis, biofilm formation, nutrient acquisition, or novel virulence genes using this model system may enhance understanding of this phenotype.
Acknowledgments
Financial support was from the ICDDR,B, Centre for Health and Population Research, and NIH grants K01-TW07144 (R.C.L.), AI40725 (E.T.R.), and U01-AI58935 (S.B.C.). A.A. was supported by a training grant from the Fogarty International Center (D43-TW05572 to S.B.C.). J.B.H. is an NICHD fellow of the Pediatric Scientist Development Program (NICHD Grant Award K12-HD00850). R.C.L. is the recipient of an Ellison Medical Foundation Postdoctoral Fellowship in International Infectious Diseases from the Infectious Diseases Society of America.
Editor: W. A. Petri, Jr.
REFERENCES
- 1.Angelichio, M. J., D. S. Merrell, and A. Camilli. 2004. Spatiotemporal analysis of acid adaptation-mediated Vibrio cholerae hyperinfectivity. Infect. Immun. 72:2405-2407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Angelichio, M. J., J. Spector, M. K. Waldor, and A. Camilli. 1999. Vibrio cholerae intestinal population dynamics in the suckling mouse model of infection. Infect. Immun. 67:3733-3739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Bina, J., J. Zhu, M. Dziejman, S. Faruque, S. Calderwood, and J. Mekalanos. 2003. ToxR regulon of Vibrio cholerae and its expression in vibrios shed by cholera patients. Proc. Natl. Acad. Sci. USA 100:2801-2806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Butler, S. M., and A. Camilli. 2004. Both chemotaxis and net motility greatly influence the infectivity of Vibrio cholerae. Proc. Natl. Acad. Sci. USA 101:5018-5023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Butterton, J. R., E. T. Ryan, R. A. Shahin, and S. B. Calderwood. 1996. Development of a germfree mouse model of Vibrio cholerae infection. Infect. Immun. 64:4373-4377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Chiang, S. L., and J. J. Mekalanos. 1998. Use of signature-tagged transposon mutagenesis to identify Vibrio cholerae genes critical for colonization. Mol. Microbiol. 27:797-805. [DOI] [PubMed] [Google Scholar]
- 7.Crean, T. I., M. John, S. B. Calderwood, and E. T. Ryan. 2000. Optimizing the germfree mouse model for in vivo evaluation of oral Vibrio cholerae vaccine and vector strains. Infect. Immun. 68:977-981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Faruque, S. M., I. B. Naser, M. J. Islam, A. S. Faruque, A. N. Ghosh, G. B. Nair, D. A. Sack, and J. J. Mekalanos. 2005. Seasonal epidemics of cholera inversely correlate with the prevalence of environmental cholera phages. Proc. Natl. Acad. Sci. USA 102:1702-1707. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Freter, R., and P. C. O'Brien. 1981. Role of chemotaxis in the association of motile bacteria with intestinal mucosa: fitness and virulence of nonchemotactic Vibrio cholerae mutants in infant mice. Infect. Immun. 34:222-233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Fullner, K. J., and J. J. Mekalanos. 1999. Genetic characterization of a new type IV-A pilus gene cluster found in both classical and El Tor biotypes of Vibrio cholerae. Infect. Immun. 67:1393-1404. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gutierrez-Cazarez, Z., F. Qadri, M. J. Albert, and J. A. Giron. 2000. Identification of enterotoxigenic Escherichia coli harboring longus type IV pilus gene by DNA amplification. J. Clin. Microbiol. 38:1767-1771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Heidelberg, J. F., J. A. Eisen, W. C. Nelson, R. A. Clayton, M. L. Gwinn, R. J. Dodson, D. H. Haft, E. K. Hickey, J. D. Peterson, L. Umayam, S. R. Gill, K. E. Nelson, T. D. Read, H. Tettelin, D. Richardson, M. D. Ermolaeva, J. Vamathevan, S. Bass, H. Qin, I. Dragoi, P. Sellers, L. McDonald, T. Utterback, R. D. Fleishmann, W. C. Nierman, and O. White. 2000. DNA sequence of both chromosomes of the cholera pathogen Vibrio cholerae. Nature 406:477-483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Herrington, D. A., R. H. Hall, G. Losonsky, J. J. Mekalanos, R. K. Taylor, and M. M. Levine. 1988. Toxin, toxin-coregulated pili, and the toxR regulon are essential for Vibrio cholerae pathogenesis in humans. J. Exp. Med. 168:1487-1492. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Iwanaga, M., K. Yamamoto, N. Higa, Y. Ichinose, N. Nakasone, and M. Tanabe. 1986. Culture conditions for stimulating cholera toxin production by Vibrio cholerae O1 El Tor. Microbiol. Immunol. 30:1075-1083. [DOI] [PubMed] [Google Scholar]
- 15.Kaper, J. B., J. G. Morris, Jr., and M. M. Levine. 1995. Cholera. Clin. Microbiol. Rev. 8:48-86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.LaRocque, R. C., J. B. Harris, M. Dziejman, X. Li, A. I. Khan, A. S. G. Faruque, S. M. Faruque, G. B. Nair, E. T. Ryan, F. Qadri, J. J. Mekalanos, and S. B. Calderwood. 2005. Transcriptional profiling of Vibrio cholerae recovered directly from patient specimens during early and late stages of human infection. Infect. Immun. 73:4488-4493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Lobitz, B., L. Beck, A. Huq, B. Wood, G. Fuchs, A. S. Faruque, and R. Colwell. 2000. Climate and infectious disease: use of remote sensing for detection of Vibrio cholerae by indirect measurement. Proc. Natl. Acad. Sci. USA 97:1438-1443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Merrell, D. S., and A. Camilli. 1999. The cadA gene of Vibrio cholerae is induced during infection and plays a role in acid tolerance. Mol. Microbiol. 34:836-849. [DOI] [PubMed] [Google Scholar]
- 19.Merrell, D. S., D. L. Hava, and A. Camilli. 2002. Identification of novel factors involved in colonization and acid tolerance of Vibrio cholerae. Mol. Microbiol. 43:1471-1491. [DOI] [PubMed] [Google Scholar]
- 20.Merrell, D. S., S. M. Butler, F. Qadri, N. A. Dolganov, A. Alam, M. B. Cohen, S. B. Calderwood, G. K. Schoolnik, and A. Camilli. 2002. Host-induced epidemic spread of the cholera bacterium. Nature 417:642-645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Monod, J. 1966. From enzymatic adaptation to allosteric transitions. Science 154:475-483. [DOI] [PubMed] [Google Scholar]
- 22.Pascual, M., X. Rodo, S. P. Ellner, R. Colwell, and M. J. Bouma. 2000. Cholera dynamics and El Nino-southern oscillation. Science 289:1766-1769. [DOI] [PubMed] [Google Scholar]
- 23.Qadri, F., C. Wenneras, M. J. Albert, J. Hossain, K. Mannoor, Y. A. Begum, G. Mohi, M. A. Salam, R. B. Sack, and A. M. Svennerholm. 1997. Comparison of immune responses in patients infected with Vibrio cholerae O139 and O1. Infect. Immun. 65:3571-3576. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Sack, D. A., S. Huda, P. K. Neogi, R. R. Daniel, and W. M. Spira. 1980. Microtiter ganglioside enzyme-linked immunosorbent assay for vibrio and Escherichia coli heat-labile enterotoxins and antitoxin. J. Clin. Microbiol. 11:35-40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Stanier, R. Y. 1951. Enzymatic adaptation in bacteria. Annu. Rev. Microbiol. 5:35-56. [DOI] [PubMed] [Google Scholar]
- 26.Stroeher, U. H., K. E. Jedani, B. K. Dredge, R. Morona, M. H. Brown, L. E. Karageorgos, M. J. Albert, and P. A. Manning. 1995. Genetic rearrangements in the rfb regions of Vibrio cholerae O1 and O139. Proc. Natl. Acad. Sci. USA 92:10374-10378. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Taylor, R. K., V. L. Miller, D. B. Furlong, and J. J. Mekalanos. 1987. Use of phoA gene fusions to identify a pilus colonization factor coordinately regulated with cholera toxin. Proc. Natl. Acad. Sci. USA 84:2833-2837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Waldor, M. K., and J. J. Mekalanos. 1996. Lysogenic conversion by a filamentous phage encoding cholera toxin. Science 272:1910-1914. [DOI] [PubMed] [Google Scholar]