Abstract
Background
Despite the low incidence of coronavirus disease 2019 (COVID-19) vaccine–induced allergic reactions reported to date, concerns of such reactions have been reported in the literature among individuals with and without a history of allergic disease.
Objectives
Herein, we provide an update to a previous scoping review published by our group, focusing on COVID-19 vaccine hesitancy in relation to allergy and the incidence of anaphylactic reactions to COVID-19 vaccines.
Methods
The current review follows an a priori protocol drafted in accordance with Arksey and O’Malley’s framework for methodological reviews. A comprehensive search was conducted on 4 scientific databases (CINAHL, PsycINFO, MEDLINE, Embase) to identify eligible studies published since our initial review. Eligible articles included those published between February 12, 2022, and November 10, 2023, and were retrieved using an established search process developed by content and methodological experts. Among the 2,099 unique citations, 45 articles (2.1%) were included.
Results
Consistent with previously reviewed literature, COVID-19 vaccine–induced anaphylaxis remains rare among both those with allergies and the general population. Despite the rarity of anaphylaxis, hesitancy persists among individuals with and without allergies.
Conclusions
To prepare for future pandemics, it is evident that more efforts are needed to address concerns regarding the potential for allergic reactions following vaccination. As part of this process, it is important to ensure medical professionals are updated as new information becomes available and that evidence-based risk communication is accurate, accessible, and culturally appropriate.
Key words: Allergy, anaphylaxis, COVID-19, vaccine hesitancy, scoping review
Despite the success of vaccination in reducing the global burden of disease, many people remain vaccine hesitant.1 The abundance of both valid and invalid information contributed to what has been described as the “infodemic” by the World Health Organization (WHO) and has played a pivotal role in amplifying doubts and fueling vaccine hesitancy, particularly in the context of coronavirus disease 2019 (COVID-19).2,3 Concerns about vaccine hesitancy are not new, but the outbreak of the COVID-19 pandemic has arguably intensified these concerns by laying bare the impact of hesitancy on efforts to control the pandemic. While the causes of hesitancy are multifaceted, confidence in vaccine safety has been identified as a primary concern influencing vaccine uptake.4 In light of the important role played by safety in influencing vaccine confidence, it stands to reason that individuals with a history of allergy to food, drugs, or insects may be more hesitant to receive various vaccinations, including the COVID-19 vaccine, given that some vaccine excipients do contain allergens known to cause reactions in sensitive individuals.5 While reactions following vaccination are rare, extensive media coverage and misinformation surrounding vaccine-induced reactions hold the potential to undermine the confidence of some individuals with a history of allergic disease who may perceive themselves as vulnerable to such reactions.5 Furthermore, the emergence of anaphylaxis cases prompted cautionary advice discouraging individuals with allergies from getting vaccinated.6, 7, 8 The inconsistent communication from both governmental and nongovernmental entities regarding the suitability of COVID-19 vaccines for those with allergic conditions potentially exacerbated skepticism and uncertainty among individuals with a prior history of severe allergies.6
In light of this, we previously conducted a scoping review9 (published online in February 2023) aimed at better understanding the incidence of allergic reactions following COVID-19 vaccination and whether fear of allergic reactions contributes to vaccine hesitancy among both the general public and those with allergic disease. This review was based on literature published following the outbreak of the pandemic, but prior to February 11, 2022.9 Through this work, we found that rates of allergic reactions and anaphylaxis following vaccination for COVID-19 were relatively low (median incidence of anaphylaxis = 5 cases per million doses; median incidence of allergic reactions = 528 cases per million doses).9 Results also revealed that a history of allergic disease appeared to increase an individual’s risk of reaction; however, the incidence of adverse events following vaccination was low even among those with allergies.9 Despite the rarity of reactions, we found that fear of allergic reactions was a significant concern in both individuals living with allergy and those without a history of allergic disease. Not surprisingly, these concerns were also found to undermine vaccine confidence among those with a history of allergy.9
While these findings are important in demonstrating an initial link between fear of reactions and COVID-19 vaccine hesitancy, many studies have been published on the topic since our initial review of the literature that may help better establish the impact of reaction-related fears on vaccine hesitancy. The objective of this current study is to provide an updated synthesis of the recently published literature that investigates the incidence of anaphylaxis following COVID-19 vaccination and explores the relationship between such reactions and vaccine confidence. By critically examining the newly published studies, this review will contribute to closing the existing gaps and resolving inconsistencies, and serve as a valuable resource for informing and guiding future pandemic responses.
Methods
This review serves as an update to our original review 9 which was conducted in February 2022, and focuses on peer-reviewed literature published in the same 4 databases included in our previous review (ie, MEDLINE [Ovid], CINAHL [EBSCO], Embase [Ovid], and PsycINFO [Ovid]). Eligible articles included those published between February 12, 2022, and November 10, 2023 (the date of our final search), that provided original data on rates of anaphylaxis following vaccination for COVID-19. We also included articles that examined the relationship between allergic reactions or allergic disease and hesitancy surrounding the COVID-19 vaccine. For the purposes of the current study, hesitancy was defined as negative feelings toward the COVID-19 vaccine, delays in acceptance of the vaccine, or vaccine refusal. Any studies that were published in February 2022 and identified in both our original and updated search were included in the original scoping review only. The methodology, including search terms, study design, and data charting processes, closely mirrors that of our original review.9, 10, 11 The exclusion of gray literature further ensures that our synthesis is based on high-quality, peer-reviewed studies.
Shift in focus to anaphylaxis
In contrast to our original review, which encompassed both allergic reactions and anaphylaxis, the focus of this update is primarily on anaphylaxis. The initial review’s emphasis on both outcomes was prompted by limited data on anaphylaxis during the acute phase of the COVID-19 pandemic. Given the evolving COVID-19 landscape and the accumulation of more data on anaphylaxis, our current goal is to underscore the incidence of anaphylaxis. Given that there is no consensus definition of anaphylaxis, we did not restrict articles based on how anaphylaxis was defined as long as the definition did not differ markedly from commonly cited guidelines.
Of note, papers that investigated adverse reactions to the COVID-19 vaccine in individuals with mastocytosis were excluded from the review. It was believed that including these articles may unduly bias estimates of the incidence of allergic reactions following vaccination as the condition places individuals at higher risk of anaphylaxis.12 However, papers examining postvaccination reactions and hesitancy in individuals with other chronic conditions were eligible for the current review.
Incidence calculation
To determine the incidence of anaphylaxis within the included studies, we employed an approach that involved calculating the proportion of reported anaphylaxis cases in relation to the total administered doses.
The formula used to calculate the incidence is as follows:
where represents the incidence of anaphylaxis, represents the number of reported anaphylaxis cases, and represents the total number of administered COVID-19 doses.
Results
Literature search results
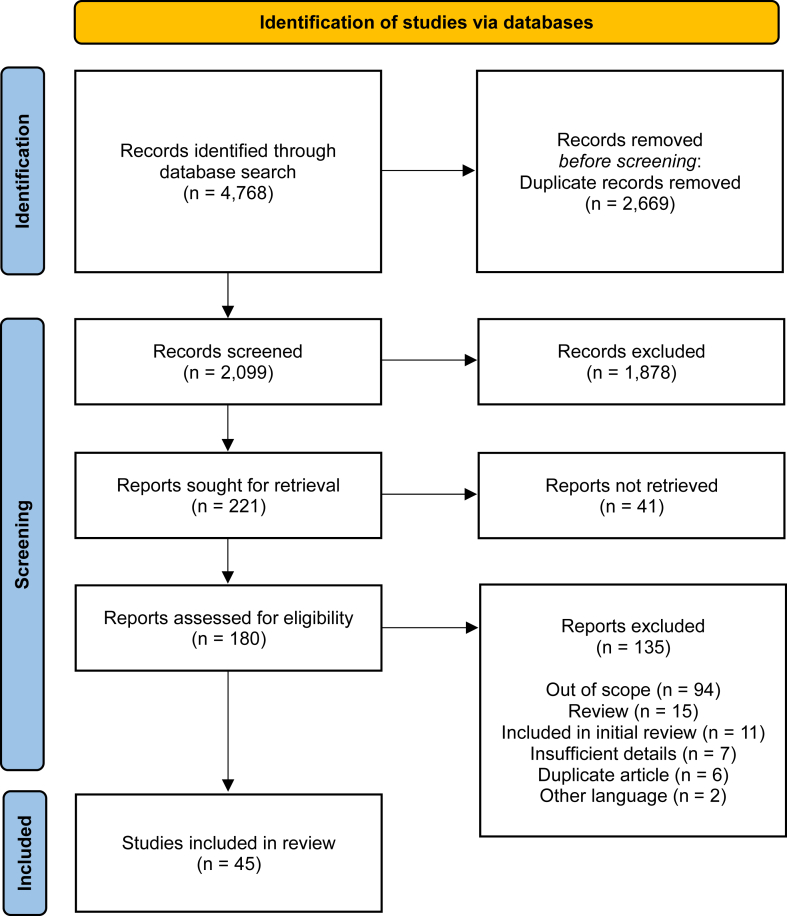
The academic literature search yielded 4768 articles (1429 from MEDLINE [Ovid], 3114 from Embase [Ovid], 28 from PsycINFO [Ovid], and 197 from CINAHL [EBSCO]). After removing duplicate papers, 2099 academic articles remained, which were then screened at the title and abstract level. Of these, 221 were assessed for eligibility at the full-text level, resulting in the inclusion of 45 academic articles (see Fig 1 for an overview of the screening process and Table I for an overview of the included papers).
Fig 1.
Preferred Reporting Items for Systematic Reviews and Meta-Analyses flow diagram demonstrating selection process of articles for the review update.
Table I.
Summary table of the included studies
| First author | Year | Title | Country | Aim | Sample size | Methodology | Outcome measures | Key findings |
|---|---|---|---|---|---|---|---|---|
| Aberumand | 2022 | Understanding attitudes and obstacles to vaccination against COVID-19 in patients with primary immunodeficiency | Canada | To understand the perceptions regarding COVID-19 vaccination among patients with primary immunodeficiency and to identify factors related to vaccine hesitancy | 449 | Cross-sectional study | Ad hoc questionnaire on vaccine hesitancy | Reasons for vaccine hesitancy encompass concerns regarding potential long-term side effects (50.4%) and fear of reaction to the vaccine (32.2%) |
| Ahsanuddin | 2024 | Otolaryngologic side effects after COVID-19 vaccination | USA | To investigate otolaryngologic adverse events of COVID-19 vaccines reported to the VAERS and their clinical relevance | 1,280,950 | Retrospective study | Assessment of otolaryngologic adverse events following COVID-19 vaccination using the Food and Drug Administration's VAERS database | Anaphylaxis: n = 1,323 (PRR: 0.37 [95% CI: 0.26-0.54]), (ROR: 0.37 [95% CI: 0.26-0.53]) |
| Al-Obaydi | 2022 | Hesitancy and reactogenicity to mRNA-based COVID-19 vaccines—early experience with vaccine rollout in a multi-site healthcare system | USA | To understand factors associated with mRNA-based COVID-19 vaccines hesitancy and reactogenicity | 5,709 | Cross-sectional study | Ad hoc questionnaire exploring factors for declining vaccination (nonvaccine receivers) and factors associated with occurrence and severity of local and systemic reactions after the first and second vaccine doses (vaccine receivers) | Anaphylaxis: n = 3 Allergy to other vaccines was associated with vaccine hesitancy |
| Amarasinghe | 2023 | COVID-19 vaccine-related adverse events following immunization in the WHO Western Pacific Region, 2021-2022 | WHO Western Pacific Region Countries | To understand the landscape of COVID-19 vaccine-related adverse events following immunization in the Western Pacific Region | 732,000,000 | Retrospective observational study | Weekly reports provided by WHO Western Pacific Region country offices and countries | Anaphylaxis: n = 7,566 |
| Cebeci Kahraman | 2022 | Cutaneous reactions after COVID-19 vaccination in Turkey: a multicenter study | Turkey | To investigate cutaneous and systemic adverse reactions following COVID-19 vaccination with Sinovac/CoronaVac (inactivated SARS-CoV-2) and Pfizer/BioNTech (BNT162b2) vaccines | 2,290 | Prospective cross-sectional study | Systemic, local injection site, and nonlocal cutaneous reactions after vaccination were identified, and their rates were determined | Anaphylaxis: n = 3, first dose (n = 1 for CoronaVac; n = 2 for BioNTech); second dose n = 1 for BioNTech |
| Connolly | 2022 | Disease flare and reactogenicity in patients with rheumatic and musculoskeletal diseases following two-dose SARS-CoV-2 messenger RNA vaccination | USA | To investigate disease flare and postvaccination reactions (reactogenicity) in patients with rheumatic and musculoskeletal diseases following 2-dose SARS-CoV-2 mRNA vaccination | 1,377 | Prospective observational study | Ad hoc questionnaire detailing local and systemic adverse events | Anaphylaxis: n = 0 Frequent reactions: injection site pain (87% after dose 1, 86% after dose 2) and fatigue (60% after dose 1, 80% after dose 2) |
| Crăciun | 2022 | mRNA COVID-19 vaccine reactogenicity among healthcare workers: results from an active survey in a pediatric hospital from Bucharest, January-February 2021 | Romania | To investigate the adverse events following mRNA Pfizer-BioNTech vaccine immunization among health care workers at a Bucharest pediatric hospital, vaccinated during the first phase of the national vaccination campaign in Romania | 574 | Cross-sectional study | Ad hoc questionnaire on adverse events | Anaphylaxis: n = 0 Most frequent adverse event: injection site pain (80.3% after dose 1; 75.1% after dose 2) |
| Cserep | 2022 | The effect of a single dose of BNT162b2 vaccine on the incidence of severe COVID-19 infection in patients on chronic hemodialysis: a single-centre study | United Kingdom | To understand the 8-wk safety and efficiency of a single dose of the BNT162b2 vaccine in patients on chronic hemodialysis | 83 | Retrospective observational study | Local and systemic reactions in the 7-days following vaccination | Anaphylaxis: n = 0 No life-threatening allergic reactions or side effects observed |
| Du | 2022 | Safety of an inactivated COVID-19 vaccine in patients with wheat-dependent exercise-induced anaphylaxis | China | To investigate the comparison of adverse events associated with an inactivated COVID-19 vaccine between WDEIA patients and healthy controls | n = 72 WDEIA; n = 730 healthy matched controls | Cross-sectional study | Ad hoc questionnaire on adverse reactions after vaccination | Anaphylaxis: n = 0 Similar rates of adverse events in case and control groups |
| Filon | 2022 | Allergic reactions to COVID-19 vaccination in high-risk allergic patients: the experience of Trieste University Hospital (North-Eastern Italy) | Italy | To understand the risk of reactions in high-risk allergic patients vaccinated for COVID-19 at the University Health Agency Giuliano-Isontina of Trieste | 269 | Cross-sectional study | Structured survey questionnaire investigating the signs and symptoms developed after any COVID-19 vaccine dose | Anaphylaxis: n = 0 No anaphylactic events were observed |
| Gamez | 2022 | Safety of mRNA COVID-19 vaccines in patients with well-controlled myasthenia gravis | Spain | To investigate the safety of mRNA-based 2-dose vaccination in a cohort of patients with myasthenia gravis | 91 | Prospective observational study | Local and systemic reactogenicity after injection was monitored for each dose administered | Anaphylaxis: n = 0 No anaphylactic reactions reported |
| Goda | 2022 | Association between adverse reactions for the first and second doses of COVID-19 vaccine | Japan | To understand the frequency of adverse reactions to COVID-19 vaccines in Japan and the impact of first-dose adverse reactions on second-dose adverse reactions | 671 | Prospective cohort study | Ad hoc self-administered questionnaire on adverse vaccine events and general health | Anaphylaxis: n = 0 No anaphylaxis cases were reported |
| Guarnieri | 2022 | Adverse reactions to BNT162B2 vaccine in health care workers from an Italian Tertiary Care Hospital | Italy | To investigate adverse reactions and possible hypersensitivity reactions during the vaccination campaign of health care workers at Meyer Children's University Hospital in Florence (Italy), conducted with the mRNA Pfizer vaccine from January 1 to March 2, 2021 | 1,293 | Prospective cohort study | Notifications by hospital pharmacy and face-to-face declarations with medical doctors | Anaphylaxis: n = 0 Following dose 1, individuals with a history of allergic disease were more likely to have had a suspected hypersensitivity reaction; after dose 2, this relationship did not reach statistical significance |
| Holzmann-Littig | 2022 | COVID-19 vaccines: fear of side effects among German health care workers | Germany | To investigate which side effects concern health care workers and how these are associated with vaccine hesitancy | 4,500 | Cross-sectional exploratory study | Ad hoc questionnaire about adverse reactions and fears of side effects | 153 of 4,500 respondents (3.4%) feared an allergic reaction; concerns about serious side effects were associated with vaccination refusal |
| Ieven | 2022 | COVID-19 vaccination safety and tolerability in patients allegedly at high risk for immediate hypersensitivity reactions | Belgium | To investigate the allergist-observed safety and tolerability of COVID-19 vaccines in patients deemed at higher risk of COVID-19 vaccine-induced immediate hypersensitivity reactions, vaccinated in-hospital under medical supervision | 196 | Retrospective cohort study | On-site assessment by allergist and telephone interviews after 3-7 days | Anaphylaxis: n = 0 None fulfilled criteria for anaphylaxis |
| Imai | 2022 | Incidence and risk factors of immediate hypersensitivity reactions and immunization stress-related responses with COVID-19 mRNA vaccine | Japan | To investigate the incidence and risk factors for immediate hypersensitivity reactions and immunization stress-related responses with the Moderna COVID-19 vaccine | 614,151 | Nested case-control study | Relevant clinical findings of adverse events following immunization were collected via medical records in first-aid rooms and classified by trained physicians | Anaphylaxis: n = 2 Of the 318 immediate hypersensitivity reaction events, 2 were classified as anaphylaxis based on the Brighton criteria |
| Iwayama | 2022 | Early immunological responses to the mRNA SARS-CoV-2 vaccine in patients with neuromuscular disorders | Japan | To investigate the safety and efficacy of BNT16b2 in patients with NMDs with muscular atrophy compared to that in healthy subjects | 11 patients with NMDs; 346 healthy patients | Observational cohort study | Ad hoc questionnaire on adverse reactions | Anaphylaxis: n = 0 No anaphylaxis occurred in any of the participants |
| Kara | 2023 | Self-reported allergic adverse events following inactivated SARS-CoV-2 vaccine (TURKOVAC) among general and high-risk population | Turkey | To investigate the distribution and severity of allergic adverse events following the administration of the TURKOVAC whole-virion inactivated COVID-19 vaccine as either the primary or the booster dose in 15 provinces in Turkey | 29,584 | Prospective cohort study | Ad hoc self-report structured questionnaire | Anaphylaxis: n = 0 |
| Lewin | 2023 | COVID-19 vaccine concerns of health care providers and ancillary staff | USA | To understand COVID-19 concerns among a range of health care providers and ancillary staff, this study assessed reported vaccination of their adolescent children and intent to vaccinate younger children as part of an effort to improve pediatric COVID-19 vaccine uptake | 1,437 | Prospective cohort study | Ad hoc web-based survey to assess intent to vaccinate children against COVID-19 and concerns regarding vaccination | Nurses and other personnel had more vaccine concerns than physicians and pharmacists, especially about allergic reactions (concerns about allergic reactions: 15% of physicians vs 36% of nurses) |
| Lim | 2022 | Role of occupational health services in planning and implementing of staff COVID-19 vaccination clinic: a tertiary hospital experience in Singapore | Singapore | To investigate ways to address and overcome vaccine hesitancy among health care workers in a tertiary hospital in Singapore and ensure maximal uptake of the Pfizer-BioNTech COVID-19 vaccine | 7,671 | Interventional study | Patients were monitored for 30 min following vaccination, with adverse events recorded, and staff were also encouraged to report adverse events to their hospital’s occupational health clinic | Anaphylaxis: n = 0 |
| Low | 2022 | Breastfeeding mother and child clinical outcomes after COVID-19 vaccination | Singapore | To investigate solicited adverse effects unique to lactating individuals, including axillary lymphadenopathy, mastitis, and breast engorgement, as well as to examine the systemic and local adverse effects of the Pfizer/BioNTech mRNA COVID-19 vaccine (BNT162b2) on lactating health care workers | 88 | Prospective cohort study | Ad hoc structured questionnaire | Anaphylaxis: n = 0 |
| Lyons | 2022 | Risk stratification through allergy history: single-centre experience of specialized COVID-19 vaccine clinic | Ireland | To investigate the establishment of a specialized COVID-19 vaccine allergy clinic aimed at addressing the COVID-19 vaccination needs of individuals aged ≥16 y | n = 206 | Observational study | Observations of adverse reactions | Anaphylaxis: n = 0 No cases of anaphylaxis |
| Matson | 2023 | Paediatric safety assessment of BNT162b2 vaccination in a multistate hospital-based electronic health record system in the USA: a retrospective analysis | USA | To investigate the adverse events associated with 2-dose BNT162b2 vaccination in the pediatric population (children aged 5-17 y) | 56,436 | Retrospective study | Multistate hospital-based electronic health records | Anaphylaxis: n = 6; n = 3 after first dose; n = 3 after second dose |
| Milito | 2023 | Safety of mRNA COVID-19 vaccines in patients with inborn errors of immunity: an Italian multicentric study | Italy | To investigate the reactogenicity and adverse event profile following 2, 3, and 4 doses of mRNA vaccines in patients with inborn errors of immunity | 342 | Observational study | Ad hoc self-report questionnaire | Anaphylaxis: n = 0 |
| Najjar | 2023 | COVID-19 vaccination reported side effects and hesitancy among the Syrian population: a cross-sectional study | Syria | To investigate postvaccination side effects and identify factors associated with both general side effects and severe side effects | 1,672 | Cross-sectional study | Ad hoc questionnaire designed to collect specific and targeted information from participants regarding their experiences and perceptions related to COVID-19 vaccination | Anaphylaxis: n = 1 |
| Niekrens | 2022 | Homologous COVID-19 BNT162b2 vaccination at a German tertiary care university hospital: a survey-based analysis of reactogenicity, safety, and inability to work among healthcare workers | Germany | To investigate the age- and sex-dependent reactogenicity and safety of BNT162b2 in a real-life setting | 2,372 | Retrospective study | Ad hoc questionnaire on adverse effects after first or second vaccine dose | Anaphylaxis: n = 4 |
| Njoga | 2022 | COVID-19 vaccine hesitancy and determinants of acceptance among healthcare workers, academics and tertiary students in Nigeria | Nigeria | To investigate factors associated with COVID-19 vaccine hesitancy among Nigerian health care workers, academics, and tertiary students | 1,525 | Cross-sectional study | Ad hoc online questionnaire | 24.6% (307 of 1,079) of participants expressed fear of an allergic reaction (307 of 1,079) |
| Odejinmi | 2022 | COVID-19 vaccine hesitancy: a midwifery survey into attitudes towards the COVID-19 vaccine | England | To understand midwifery uptake of and attitudes toward the COVID-19 vaccine in 2 ethnically diverse areas | 378 | Mixed methods | Ad hoc questionnaire on vaccine uptake and factors influencing vaccine hesitancy | 22% of participants expressed fear of having an allergic reaction |
| Oka | 2022 | Awareness, barriers and concerns of adolescents toward the COVID-19 vaccine: a cross-sectional study in Singapore | Singapore | To investigate the awareness, concerns, and barriers of adolescents toward the COVID-19 vaccine | 460 | Cross-sectional study | Ad hoc web-based questionnaire on vaccine literacy and hesitancy | 73.2% of participants had concerns about anaphylaxis |
| Ou | 2021 | Safety and reactogenicity of 2 doses of SARS-CoV-2 vaccination in solid organ transplant recipients | USA | To investigate the safety and reactogenicity of SARS-CoV-2 mRNA vaccines in solid organ transplant recipients | 741 | Prospective cohort study | Ad hoc questionnaire assessing local symptoms and systemic adverse reactions after each vaccine dose | Anaphylaxis: n = 0 |
| Rayamajhi | 2022 | Adverse events following immunization with ChAdOx1 nCoV-19 and BBIBP-CorV vaccine: a comparative study among healthcare professionals of Nepal | Nepal | To investigate the local and systemic adverse events following vaccination with Vaxzevria (ie, ChAdOx1 nCoV-19) and BIBP (ie, BBIBP-CorV; Sinopharm, Beijing, China) among health care professionals of Nepal | 606 | Cross-sectional study | Ad hoc self-administered online survey tool on adverse events | Anaphylaxis: n = 0 No serious and life-threatening adverse events like anaphylaxis were reported |
| Rosada | 2023 | Safety of mRNA COVID-19 vaccinations in patients with allergic diseases | Poland | To investigate the degree of safety and assess the possible risk of acute allergic reactions following mRNA COVID-19 vaccination among a group of patients predisposed to allergic diseases | 52 | Prospective observational study | Ad hoc questionnaire aimed to gather information about participants' experiences of various symptoms after receiving the COVID-19 vaccine | Anaphylaxis: n = 0 |
| Sa | 2022 | The safety of mRNA-1273, BNT162b2 and JNJ-7836735 COVID-19 vaccines: safety monitoring for adverse events using real-world data | USA | To understand the adverse effects and provide accurate postvaccination safety information | 481,172 | Retrospective study | VAERS database was searched from December 14, 2020 to September 30, 2021, to analyze and characterize postvaccination adverse events | Anaphylaxis: mRNA-1273 (ie, Moderna; n = 79), BNT162b2 (ie, Pfizer; n = 79), JNJ-78436735 (ie, Johnson & Johnson; n = 13) mRNA vaccines had lower risk for anaphylaxis in univariate analysis, but higher risk in multivariate analysis |
| Sarca Çevk | 2022 | Attitudes of family medicine residents in Ankara towards COVID-19 vaccines administered in Turkey: a descriptive, cross-sectional study | Turkey | To investigate the attitudes of family medicine residents in Ankara about the inactivated and mRNA COVID-19 vaccines being administered in Turkey | 139 | Cross-sectional study | Ad hoc survey on attitudes of residents toward COVID-19 vaccines | 5% of residents said they would not recommend vaccination if the patient had a history of allergic reactions or anaphylaxis. Between 43.2% and 49.6% would not recommend a second dose of the COVID-19 vaccine after an anaphylactic reaction to the first |
| Sayaca | 2023 | The percentage of hesitation and factors associated with acceptance or refusal for COVID-19 vaccination: does education about vaccines by allergist affect personal decision? | Turkey | To investigate the vaccination perspective of patients and learn how many patients were persuaded to get vaccinated with the effect of the education provided by allergists | 200 | Cross-sectional study | Antivaccination scale form and questionnaire form developed by allergists | A similar proportion of patients with history of anaphylaxis (55.3%) indicated they planned to be vaccinated compared to those without such a history (61.7%); 88.2% of patients with allergies who indicated they were not interested in being vaccinated before the education session indicated that they ultimately received the vaccination after attending the educational talk |
| Sen | 2022 | COVID-19 vaccination-related adverse events among autoimmune disease patients: results from the COVAD study | United Kingdom; 94 countries | To investigate adverse events in patients with systemic autoimmune and inflammatory disorders and healthy controls 7 days postvaccination | 10,900 | Cross-sectional study | Ad hoc online survey of adverse events | Anaphylaxis: systemic autoimmune and inflammatory disorders group = 0.2% (11 of 5,867); healthy control = 0.4% (5 of 5,033) |
| Shivarev | 2023 | Adverse event reports of anaphylaxis after Comirnaty and Vaxzevria COVID-19 vaccinations, Western Australia, 22 February to 30 June 2021 | Australia | To investigate anaphylaxis rates, characteristics, and outcomes of reported cases of anaphylaxis to COVID-19 vaccines in Western Australia | 727,750 | Retrospective observational study | Clinician reports of adverse events following immunization to the Western Australian vaccine safety surveillance system | Anaphylaxis: n = 12, with 8 cases associated with Vaxzevria and 4 cases with Comirnaty, resulting in rates of 15.9 and 17.7 per million doses administered for Vaxzevria and Comirnaty, respectively |
| Stehlin | 2022 | Intradermal testing with COVID-19 mRNA vaccines predicts tolerance | Switzerland | To investigate the management of individuals with a history of polyethylene glycol allergy or suspected allergic reactions after receiving the COVID-19 mRNA vaccine | 604,267 | Observational cohort study | A standardized evaluation involving people responding to questions either online or through a hotline | Anaphylaxis: n = 18 |
| Sturkenboom | 2022 | Cohort monitoring of 29 adverse events of special interest prior to and after COVID-19 vaccination in four large European electronic healthcare data sources | Italy, The Netherlands, United Kingdom, Spain | To investigate the use of COVID-19 vaccines and monitor the incidence rates of prespecified adverse events of special interest prior to and after COVID-19 vaccination | 25,720,158 | Retrospective cohort study | PHARMO Database Network (The Netherlands), BIFAP database, ARS (Italian) database, Clinical Practice Research Datalink (United Kingdom) | Results revealed an anaphylaxis incidence rate of 11.7 per 100,000 person-years |
| Vanijcharoenkarn | 2021 | Immediate reactions after the first dose of SARS-CoV-2 mRNA vaccines do not preclude second-dose administration | USA | To investigate the institutional experience with immediate reactions to the COVID-19 vaccine, and to understand the protocol for graded or full-dose vaccine readministration for immediate reactors at low risk for true anaphylaxis | 20,657 | Retrospective study | Review of occupational health records, skin testing and observation | Likely anaphylactic episodes from dose 1 (Brighton level 2): 0.03% (n = 20657). Brighton level 3 reactions: 0.39% |
| Wang et al | 2023 | Characteristics of immune response profile in patients with immediate allergic and autoimmune urticarial reactions induced by SARS-CoV-2 vaccines | Taiwan | To investigate the risk factors and immune mechanisms associated with patients who develop immediate allergic reactions and chronic urticaria following SARS-COV-2 vaccination | 129 | Prospective observational study | Naranjo algorithm; Brighton Collaboration criteria | Of the 129 participants who reported immediate reactions, 5 experienced anaphylaxis (3.9%) |
| Wentrys | 2021 | COVID-19 vaccination including 198 patients with history of severe anaphylactic reaction—own observations | Poland | To understand the outcomes of patients with a history of severe anaphylactic reaction who were vaccinated against COVID-19 | 403 | Prospective observational study | 60-min observation; telephone interview after 1 wk to collect data about late allergic reaction | Incidence of anaphylaxis among participants with a history of anaphylaxis: n = 3 (1.5%); incidence of anaphylaxis among participants with no history of anaphylaxis: n = 6 (3%) |
| Wijesinghe | 2021 | Acceptance of COVID-19 vaccine in Sri Lanka: applying the health belief model to an online survey | Sri Lanka | To understand health and illness-related behaviors related to vaccines | 895 | Cross-sectional study | Questionnaire based on health belief model | Sample had concerns about vaccine brand, side effects, allergies, and protection duration, which influenced vaccine acceptance and beliefs about vaccine protection and future infection |
| Wong | 2022 | Adverse events of special interest and mortality following vaccination with mRNA (BNT162b2) and inactivated (CoronaVac) SARS-CoV-2 vaccines in Hong Kong: a retrospective study | China | To investigate and compare the incidence of various adverse events of special interest and all-cause mortality between CoronaVac (inactivated vaccine) and BNT162b2 (ie, the Pfizer mRNA-based vaccine) | 1,938,237 | Retrospective population-based cohort study | Electronic medical records of the Hong Kong Hospital Authority | Cumulative incidence of anaphylaxis after the first dose: BNT162b2, 27 per 100,000 person-years; CoronaVac, 7 per 100,000 person-years Crude incidence rate after the first dose: BNT162b2, 376 per 100,000 person-years, CoronaVac, 134 per 100,000 person-years Incidence rate ratio for first dose anaphylaxis: 0.34 (95% CI: 0.14–0.79, P = .01), indicating a lower risk with BNT162b2 Cumulative incidence of anaphylaxis after the second dose: BNT162b2, 13 per 100,000 person-years, CoronaVac, 3 per 100,000 person-years Crude incidence rate after the second dose: BNT162b2, 212 per 100,000 person-years, CoronaVac, 67 per 100,000 person-years |
| Yilmazbas | 2023 | Allergic children's parents' hesitancy about COVID-19 vaccination | Turkey | To investigate the COVID-19 vaccination rates in allergic children aged 12-19 y and evaluate contributing factors to vaccine hesitation | 261 | Cross-sectional study | Ad hoc questionnaire covering sociodemographic details of the child and parents, child's allergic disease type, COVID-19 vaccination status for both child and parents, reasons for vaccine acceptance or refusal, and the parent's perceived stress scale | 4 of 124 individuals (3.22%) who were not vaccinated cited concerns about allergic reactions as a reason for the hesitancy; history of anaphylaxis did not significantly (P = .342) impact whether child received the vaccine or not |
ARS, Agenzia regionale di sanità della Toscana; BIFAP, Base de Datos para la Investigación Farmacoepidemiológica en Atención Primaria; NMDs, neuromuscular disorders; PRR, proportional reporting ratio; ROR, reporting odds ratio; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2; VAERS, Vaccine Adverse Event Reporting System; WDEIA, wheat-dependent exercise-induced anaphylaxis.
Evidence supporting findings from the original review
Incidence of anaphylaxis to COVID-19 vaccines
Our initial review of the literature from January 2020 to February 2022 found a low incidence of COVID-19 vaccine–induced anaphylaxis and allergic reactions (mean and median incidence for anaphylaxis = 12.4 cases per million doses and 5 cases per million doses, respectively; mean and median incidence for allergic reactions = 489.0 cases per million doses and 528.0 cases per million doses, respectively).9 In reviewing the literature published since that time, we have identified more support for this finding, as detailed below.
Among the included studies, 33 (73.3%) reported on the incidence of anaphylaxis following vaccination for COVID-19.13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45 Across these studies, anaphylaxis was found to range from 0.0% to 0.5% of doses. In 18 of these studies (54.5%), no cases of anaphylaxis were reported.16, 17, 18, 19, 20, 21, 22, 23, 24,26, 27, 28, 29, 30,32,35, 36, 37
In the United States, 5 studies investigated the frequency of postvaccination reactions, all of which consistently reported a low incidence of anaphylaxis.13,16,31,35,43 Al-Obaydi et al13 reported an incidence of 0.06% (3 of 5,340; 3 of 5,202) for anaphylaxis following both the first and second vaccine doses. Matson et al31 observed an incidence of 0.005% (6 of 106,872) for anaphylaxis among children aged 5-17 years old, with 3 cases occurring after the first dose and 3 following the second dose. Vanijcharoenkarn et al43 noted that 0.4% (88 of 20,657) of doses resulted in allergic reactions, with 8% (0.032% of the total number of doses) likely representing anaphylaxis within this group. In their examination of adverse events following COVID-19 vaccination, Ahsanuddin et al46 found 0.10% (1,323 of 1,280,950) of adverse events reported to the Vaccine Adverse Events Reporting System were coded as anaphylaxis. The remaining 2 American studies, by Connolly et al16 and Ou et al, 35 reported no cases of anaphylaxis.
Thirteen European studies also reported on anaphylaxis incidence.17,18,20,21,23,24,30,32,34,37,41,42,44 In Germany, Niekrens et al34 reported 4 cases of anaphylaxis, resulting in an incidence of 0.024% (4 of 16,704). In Switzerland, Stehlin et al41 reported an incidence of anaphylaxis at 0.003% (18 of 604,267). In a multinational study spanning Italy, The Netherlands, the United Kingdom, and Spain, Sturkenboom et al42 reported an incidence rate of 11.7 per 100,000 person-years for anaphylaxis. In Poland, Wentrys and Stopyra44 reported an anaphylaxis incidence rate of 0.5% (4 of 800) among a mixed sample of adults with a history of severe anaphylactic reactions (57% of the sample) and those without a history of reactions (43% of the sample). Nine European studies documented no cases of anaphylaxis.17,18,20,21,23,24,30,32,37
Thirteen studies conducted in Asia, primarily focused on exploring the safety profile and incidence of anaphylaxis, consistently reported low rates of anaphylaxis incidence.14,15,19,22,25, 26, 27, 28, 29,33,36,38,45 Three Chinese studies, which investigated various aspects of COVID-19 vaccine safety, reported a low incidence of anaphylaxis.19,45,47 Wong et al45 found that the incidence rate of anaphylaxis was 0.002% (27 of 1,308,820) for the first doses of the Pfizer-BioNTech Comirnaty vaccine (Pfizer, New York, NY; BioNTech, Mainz, Germany) and 0.0007% (7 of 955,859) for the Sinovac-CoronaVac vaccine (Sinovac Biotech, Beijing, China). After the second dose, the rates were 0.001% (13 of 1,116,677) and 0.0004% (3 of 821,560) for the Pfizer-BioNTech and Sinovac-CoronaVac vaccines, respectively. In a separate Chinese study involving patients with a history of wheat-dependent exercise-induced anaphylaxis, there were no reported cases of anaphylaxis.19 In a study of individuals who experienced allergic or urticarial reactions following vaccination for COVID-19, Wang et al47 discovered that 3.9% (5 of 129) of patients who experienced reactions had symptoms consistent with anaphylaxis.
A Korean study reported an incidence of anaphylaxis to mRNA vaccines ranging from 0.03 to 0.05 cases per 100,000 doses, while for viral vaccines, the incidence rate was slightly higher at 0.09 cases per 100,000 doses.38 In Japan, Imai et al25 conducted a focused study on immediate hypersensitivity reactions following COVID-19 vaccination. Imai et al25 reported an anaphylaxis incidence of 0.0002% (2 of 1,201,688). Two additional Japanese studies examining adverse reactions to the COVID-19 vaccine reported 0 cases of anaphylaxis.22,26 In Turkey, 2 studies investigating adverse reactions found low anaphylaxis incidence.15,27 Cebeci Kahraman et al15 reported an incidence of 0.13% (3 of 2290) for first-dose anaphylaxis and 0.05% (1 of 2189) for second-dose anaphylaxis, while Kara et al27 reported no anaphylaxis cases. Similarly, 2 Singaporean studies by Lim et al28 and Low et al29 also reported no anaphylaxis cases. Furthermore, a Nepalese study investigating adverse events following immunization reported no cases of anaphylaxis.36 In Syria, Najjar et al33 reported an incidence of 0.06% (1 of 1,672).
A study conducted by Amarasinghe et al,14 which incorporated data from 31 countries in the WHO’s Western Pacific Region revealed varying reporting rates for anaphylaxis by vaccine type, ranging from 0.3 cases per 1 million doses administered for Sinopharm (Beijing, China) to 13.7 cases per 1 million doses for Pfizer-BioNTech. In Oceania, Shivarev et al40 reported an incidence of 0.0016% (12 of 727,750), with 8 cases linked to Vaxzevria (AstraZeneca, Cambridge, UK) and 4 cases attributed to Comirnaty. This led to rates of 15.9 and 17.7 per million doses administered for Vaxzevria and Comirnaty, respectively.
Potential factors associated with anaphylaxis
Vaccine brand/type
In total, 5 of the 45 reviewed studies (11.1%) inferentially examined differences in the incidence of anaphylaxis across vaccine brands or types.15,38,40,42,45 The findings from this collection of studies were somewhat equivocal with 3 studies providing evidence of significant differences in anaphylaxis across various COVID-19 vaccines.38,42,45 Results were also mixed in regard to whether the incidence of allergic reactions significantly differed across vaccine type (ie, mRNA, viral vector, inactivated). More specifically, 2 of the 3 studies that found significant differences across vaccines provided evidence that anaphylaxis was more common following vaccination with a mRNA-based vaccine,38,45 while 1 study found higher rates of reactions with viral vector or inactivated vaccines.42 Notably, a WHO study in the Western Pacific region showed that the Pfizer-BioNTech vaccine exhibited the highest anaphylaxis reporting rate of the examined vaccines at 13.7 cases per 1 million doses, followed by AstraZeneca (9.9 cases per 1 million doses), Moderna (Cambridge, Mass; 8.9 per 1 million doses), Johnson & Johnson (New Brunswick, NJ; 5.6 cases per 1 million doses), Gamaleya (Moscow, Russia) (1.6 cases per 1 million doses), Sinovac (1.5 cases per 1 million doses), and Sinopharm (0.3 cases per 1 million doses).14 However, it is important to note that the study did not inferentially analyze these differences.
Hesitancy in relation to the COVID-19 vaccines
Review of the included studies revealed fear of allergic reactions to the COVID-19 vaccine was common. Although the proportion of participants expressing this concern varied considerably across papers (ie, 1.5%-73.2%), it was found to exceed 10% in 7 of the 9 reviewed studies that focused on vaccination-related concerns.48, 49, 50, 51, 52, 53, 54, 55, 56 A Singaporean study highlighted that 73.2% of respondents were concerned about anaphylaxis.53 Not surprisingly, the majority of these papers also provided evidence linking fears of reactions to vaccine hesitancy.48,49,51,54 For instance, Sayaca et al54 found that 39% of individuals who did not consider getting vaccinated expressed concerns about the potential for allergic reactions, with similar findings reported by a Canadian study of patients with primary immunodeficiency.48 Similarly, a Nigerian study on COVID-19 vaccine hesitancy reported that 28.5% of unvaccinated respondents cited allergic reactions as a reason preventing them from getting vaccinated.51 A German study assessing health care workers found that a significantly greater number of vaccine-hesitant participants reported concerns about allergic reactions, compared to individuals who accepted the vaccine.49 In contrast to the previous findings, a Turkish study investigating vaccine hesitancy among the parents of children with allergic disease found that only 3% of parents whose children were not vaccinated expressed concerns about the potential for allergic reactions.56 Taken as a whole, however, findings from the reviewed studies suggest that concerns about allergic reactions are often linked to vaccine hesitancy.
Emerging insights since the original review
COVID-19 vaccine hesitancy among health care professionals
Interestingly, results from the current review suggest that concerns regarding allergic reactions affect the vaccination decisions of not only the general population, but also health care professionals. A Turkish study that examined the attitudes of family medicine residents toward the COVID-19 vaccine found that 5% of residents would not recommend vaccination to patients with a history of allergic reaction/anaphylaxis.57 Results also revealed that 43.2% of participants would not recommend further vaccination to patients who had a history of anaphylaxis after the first dose of the Pfizer-BioNTech vaccine. By comparison, 49.6% of the residents would not recommend further vaccination to patients who had anaphylaxis after the first dose of the Sinovac-CoronaVac vaccine.
In addition, some studies have demonstrated vaccine hesitancy among health care professionals and ancillary staff (ie, administrative personnel, laboratory scientists/technologists, and imaging personnel) due to fears of adverse reactions.49, 50, 51, 52 For instance, an American study discovered the proportion of nurses, advanced practice providers, and ancillary health care professionals who expressed concern (nurses: 36%; advanced care providers: 44%; ancillary staff: 39%) regarding allergic reactions was over double that of physicians (15%) and pharmacists (13%).50 Similarly, 3 additional studies reported a notable proportion of health care professionals expressing hesitancy toward vaccination, citing concerns about postvaccination side effects and allergic reactions.49,51,52
Discussion
Consistent with the original scoping review, the findings from the current review demonstrate anaphylaxis following COVID-19 vaccination continues to be rare. Despite the rarity of anaphylaxis, the literature indicates that a portion of the general population remains vaccine hesitant. Consistent with the literature included in the original review, many of the more recent studies have found that vaccine hesitancy associated with the potential for allergic reactions and anaphylaxis persists, which may indicate that risk communication surrounding such reactions to the COVID-19 vaccine has minimally, if at all, improved vaccine hesitancy.
Effective risk communication plays a crucial role in addressing vaccine hesitancy and promoting vaccine uptake. Qualitative research has provided valuable insights into the concerns and preferences of individuals living with allergy, enabling communicators to tailor their messages accordingly. One important finding from recent research is the need for tailored messages that directly address the concerns of individuals with allergy.58 This suggests that a one-size-fits-all approach may not be effective in addressing vaccine hesitancy. Instead, communication efforts should be nuanced and customized to address the diverse range of concerns expressed by different individuals, patient populations, and communities.
Research also suggests that the source of the message plays a role in how well it is received.58 Perhaps not surprisingly, health care providers, and public health agencies, have been cited as some of the most trusted sources of vaccine-related information.59,60 In light of this, it appears as though allergists and immunologists have a particularly influential role to play in addressing the vaccination concerns of individuals with allergic disease and other immunological conditions. While addressing hesitancy is certainly no easy task, simply emphasizing the exceptionally rare nature of serious reactions following vaccination for COVID-19 may go some ways in reducing the concerns of patients.61 More generally, research has shown that adopting an assertive conversational style when discussing vaccinations, by using presumptive statements like: “Your son is due for his 4-month vaccines. We will give him these before you leave” is more effective in promoting acceptance than a participatory style (eg, “What do you want to do about your son’s vaccinations?”).62 For individuals who are especially entrenched in their hesitancy, follow-up visits reaffirming the importance of vaccination and motivational interviewing techniques have been shown to be effective in helping some individuals move toward acceptance.62
A lack of access to health care professionals has also been reported to be a significant barrier to risk communication.58 In such cases, alternative channels for information dissemination become essential. Pharmacists, community health workers, and designated personnel in administrative roles can serve as important intermediaries to deliver accurate vaccine information to individuals who may not have direct access to health care professionals. As we move past the acute phase of the pandemic, lessons like these should be considered and taken to heart well in advance of future outbreaks to ensure the necessary public health measures can be put into place as quickly as possible.
The current review expands on the previous findings by examining the attitudes and levels of vaccine acceptance among specific populations. By incorporating studies from different countries, exploring diverse ethnic groups, age groups, and occupational backgrounds, this review offers a broad perspective on the impact of anaphylaxis and vaccine hesitancy. For example, the current review has provided more insight into the attitudes of health care professionals toward allergic reactions, anaphylaxis, and vaccines. The concerns surrounding allergic reactions and anaphylaxis among health care providers underscore the critical role of timely, tailored, and evidence-based knowledge translation efforts. In the context of anaphylaxis, successful knowledge translation ensures that all members of the health care team are equipped with up-to-date information about anaphylaxis, minimizing knowledge gaps and ultimately promoting vaccine confidence.
Several limitations of the current review update warrant mention. This update, along with the original, used a narrative synthesis technique, as opposed to a meta-analysis. As a qualitative technique, narrative syntheses may be more likely to be affected by the biases of the reviewers, given that included studies are not synthesized quantitatively as they are in a meta-analysis. Also, it is important to highlight that although less severe reactions were frequently observed, the literature does not always explicitly specify whether these adverse events were allergic in nature. Defining severe allergic reactions, including anaphylaxis, can be challenging, which may introduce biases in the reported rates of such reactions. For instance, Hourihane et al63 underscored the potential for ascertainment bias in reporting severe allergic reactions associated with COVID-19 vaccines. The study emphasized the importance of using appropriate diagnostic criteria and adequate clinical information when classifying postvaccination anaphylaxis to prevent overestimations of its incidence. Lastly, due to the brevity of this update, a comprehensive review of the gray literature was not included in the analysis.
Conclusion
As the severity of the COVID-19 pandemic continues to diminish, the literature describes the ways in which our response to the pandemic has been successful and provides suggestions for improvement in future outbreaks. The original review, along with the current update, highlight the rarity of anaphylaxis to all types of COVID-19 vaccines, including mRNA vaccines. Additionally, inconsistencies in defining anaphylaxis, may contribute to the overestimation of anaphylaxis risk and increased vaccine hesitancy. Despite the rarity of anaphylaxis, a sizeable proportion of individuals, both with and without allergic disease report being concerned about the potential for reactions. To prepare for future pandemics, it is evident that more efforts are needed to address these concerns. As part of this process, it is also important to ensure medical professionals are updated as new information becomes available and that evidence-based risk communication is accurate, accessible, and culturally appropriate for the patient population. Arguably, equipping health professionals with the tools to address hesitancy may provide one way to help ensure we are better prepared to face the next pandemic.
Disclosure statement
Funding for this study was provided by a COVID-19 Vaccine Confidence Operating Grant provided by the Canadian Institutes of Health Research, awarded to J.L.P. The funding body had no influence on the study design, data collection, analysis and interpretation, or manuscript writing.
Disclosure of potential conflict of interest: A. L. R. Batac serves on the Manitoba and Saskatchewan Steering Committee for ImmUnity Canada. P. Bégin is a member of Food Allergy Canada’s Healthcare Advisory Board and reports personal fees from ALK-Abelló, Aralez Pharmaceuticals, AstraZeneca, Bausch Health, Novartis, Pfizer, Sanofi-Genzyme, and Valeo Pharma; and has received grants from DBV Technologies, Novartis, Sanofi, and Regeneron Pharmaceuticals. M. Ben-Shoshan serves on several advisory boards, including Food Allergy Canada, Novartis, Sanofi, and Stallergenes Greer; reports personal fees from Bausch Health, Novartis, Sanofi, and Stallergenes Greer; has received grants from Stallergenes Greer; and has participated in clinical trials conducted by Aimmune Therapeutics, Novartis, and Sanofi. E. Ladouceur is the co-founder and director of Science for All Audiences; and reports consultancy for Novartis. J. L. Protudjer is the Section Head for Allied Health, Co-Lead of the Research Pillar for the Canadian Society of Allergy and Clinical Immunology; a member of the steering committee for Canada's National Food Allergy Action Plan; and reports consultancy work for Ajinomoto Cambrooke, ALK-Abelló, Novartis, and Nutricia. The rest of the authors declare that they have no relevant conflicts of interest.
References
- 1.Sallam M. COVID-19 vaccine hesitancy worldwide: a concise systematic review of vaccine acceptance rates. Vaccines (Basel) 2021;9:160. doi: 10.3390/vaccines9020160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Managing the COVID-19 infodemic: promoting healthy behaviours and mitigating the harm from misinformation and disinformation [Internet]. World Health Organization. https://www.who.int/news/item/23-09-2020-managing-the-covid-19-infodemic-promoting-healthy-behaviours-and-mitigating-the-harm-from-misinformation-and-disinformation Available at: . Accessed October 7, 2023.
- 3.Puri N., Coomes E.A., Haghbayan H., Gunaratne K. Social media and vaccine hesitancy: new updates for the era of COVID-19 and globalized infectious diseases. Hum Vaccin Immunother. 2020;16:2586–2593. doi: 10.1080/21645515.2020.1780846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Betsch C., Schmid P., Heinemeier D., Korn L., Holtmann C., Böhm R. Beyond confidence: development of a measure assessing the 5C psychological antecedents of vaccination. PLoS One. 2018;13 doi: 10.1371/journal.pone.0208601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Nilsson L., Brockow K., Alm J., Cardona V., Caubet J.C., Gomes E., et al. Vaccination and allergy: EAACI position paper, practical aspects. Pediatr Allergy Immunol. 2017;28:628–640. doi: 10.1111/pai.12762. [DOI] [PubMed] [Google Scholar]
- 6.[Withdrawn] Confirmation of guidance to vaccination centres on managing allergic reactions following COVID-19 vaccination with the Pfizer/BioNTech vaccine. GOV.UK. 2021. https://www.gov.uk/government/news/confirmation-of-guidance-to-vaccination-centres-on-managing-allergic-reactions-following-covid-19-vaccination-with-the-pfizer-biontech-vaccine Available at:
- 7.Mahase E. Covid-19: people with history of significant allergic reactions should not receive Pfizer vaccine, says regulator. BMJ. 2020;371 doi: 10.1136/bmj.m4780. [DOI] [PubMed] [Google Scholar]
- 8.Banerji A., Wolfson A.R., Wickner P.G., Cogan A.S., McMahon A.E., Saff R., et al. COVID-19 vaccination in patients with reported allergic reactions: updated evidence and suggested approach. J Allergy Clin Immunol Pract. 2021;9:2135–2138. doi: 10.1016/j.jaip.2021.03.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Batac A.L.R., Merrill K.A., Askin N., Golding M.A., Abrams E.M., Bégin P., et al. Vaccine confidence among those living with allergy during the COVID pandemic (ACCORD): a scoping review. J Allergy Clin Immunol Glob. 2023;2 doi: 10.1016/j.jacig.2023.100079. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Golding M.A., Askin N., Batac A.L.R., Merrill K.A., Abrams E.M., Bégin P., et al. vACcine COnfidence amongst those living with alleRgy during the COVID pandemic (ACCORD): a scoping review protocol. Allergy Asthma Clin Immunol. 2022;18:83. doi: 10.1186/s13223-022-00723-w. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Tricco A.C., Lillie E., Zarin W., O’Brien K.K., Colquhoun H., Levac D., et al. PRISMA Extension for Scoping Reviews (PRISMA-ScR): checklist and explanation. Ann Intern Med. 2018;169:467–473. doi: 10.7326/M18-0850. [DOI] [PubMed] [Google Scholar]
- 12.Rama T.A., Castells M. Triggers of anaphylaxis in mastocytosis patients: evidence of the current drug-avoidance recommendation. Curr Treat Options Allergy. 2023;10:442–457. [Google Scholar]
- 13.Al-Obaydi S., Hennrikus E., Mohammad N., Lehman E.B., Thakur A., Al-Shaikhly T. Hesitancy and reactogenicity to mRNA-based COVID-19 vaccines—early experience with vaccine rollout in a multi-site healthcare system. PLoS One. 2022;17 doi: 10.1371/journal.pone.0272691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Amarasinghe A., Cho H., Katalbas E.R., Takashima Y. COVID-19 vaccine-related adverse events following immunization in the WHO Western Pacific Region, 2021-2022. Western Pac Surveill Response J. 2023;14:1–11. doi: 10.5365/wpsar.2023.14.2.1046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Cebeci Kahraman F., Savaş Erdoğan S., Aktaş N.D., Albayrak H., Türkmen D., Borlu M., et al. Cutaneous reactions after COVID-19 vaccination in Turkey: a multicenter study. J Cosmet Dermatol. 2022;21:3692–3703. doi: 10.1111/jocd.15209. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Connolly C.M., Ruddy J.A., Boyarsky B.J., Barbur I., Werbel W.A., Geetha D., et al. Disease flare and reactogenicity in patients with rheumatic and musculoskeletal diseases following two-dose SARS-CoV-2 messenger RNA vaccination. Arthritis Rheumatol. 2022;74:28–32. doi: 10.1002/art.41924. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Crăciun M.D., Nițescu G.V., Golumbeanu M., Tănase A.A., Pițigoi D., Săndulescu O., et al. mRNA COVID-19 vaccine reactogenicity among healthcare workers: results from an active survey in a pediatric hospital from Bucharest, January-February 2021. Vaccines (Basel) 2022;10:836. doi: 10.3390/vaccines10060836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Cserep G., Morrow D., Latchford K., Jesset R., Dosa A., Kirmizis D. The effect of a single dose of BNT162b2 vaccine on the incidence of severe COVID-19 infection in patients on chronic hemodialysis: a single-centre study. Clin Exp Nephrol. 2022;26:54–58. doi: 10.1007/s10157-021-02118-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Du Z., Yin J. Safety of an inactivated COVID-19 vaccine in patients with wheat-dependent exercise-induced anaphylaxis. Allergol Immunopathol (Madr) 2022;50:132–137. doi: 10.1016/j.iac.2014.04.004. [DOI] [PubMed] [Google Scholar]
- 20.Filon F.L., Lazzarato I., Patriarca E., Iavernig T., Peratoner A., Perri G., et al. Allergic reactions to COVID-19 vaccination in high-risk allergic patients: the experience of Trieste University Hospital (North-Eastern Italy) Vaccines (Basel) 2022;10:1616. doi: 10.3390/vaccines10101616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Gamez J., Gamez A., Carmona F. Safety of mRNA COVID-19 vaccines in patients with well-controlled myasthenia gravis. Muscle Nerve. 2022;66:612–617. doi: 10.1002/mus.27703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Goda K., Kenzaka T., Yahata S., Okayama M., Nishisaki H. Association between adverse reactions to the first and second doses of COVID-19 vaccine. Vaccines (Basel) 2022;10:1232. doi: 10.3390/vaccines10081232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Guarnieri V., Barni S., Giovannini M., Liccioli G., Sarti L., Ricci S., et al. Adverse reactions to BNT162B2 vaccine in health care workers from an Italian Tertiary Care Hospital. Clin Exp Allergy. 2022;52:911–915. doi: 10.1111/cea.14155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Ieven T., Vandebotermet M., Nuyttens L., Devolder D., Vandenberghe P., Bullens D., et al. COVID-19 vaccination safety and tolerability in patients allegedly at high risk for immediate hypersensitivity reactions. Vaccines (Basel) 2022;10:286. doi: 10.3390/vaccines10020286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Imai K., Tanaka F., Kawano S., Esaki K., Arakawa J., Nishiyama T., et al. Incidence and risk factors of immediate hypersensitivity reactions and immunization stress-related responses with COVID-19 mRNA vaccine. J Allergy Clin Immunol Pract. 2022;10:2667–2676.e10. doi: 10.1016/j.jaip.2022.07.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Iwayama H., Ishihara N., Kawahara K., Madokoro Y., Togawa Y., Muramatsu K., et al. Early immunological responses to the mRNA SARS-CoV-2 vaccine in patients with neuromuscular disorders. Front Immunol. 2022;13 doi: 10.3389/fimmu.2022.996134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Kara A., Coskun A., Temel F., Özelci P., Topal S., Ates İ. Self-reported allergic adverse events following inactivated SARS-CoV-2 vaccine (TURKOVACTM) among general and high-risk population. Vaccines (Basel) 2023;11:437. doi: 10.3390/vaccines11020437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Lim S.M., Chan H.C., Santosa A., Quek S.C., Liu E.H.C., Somani J. Role of occupational health services in planning and implementing of staff COVID-19 vaccination clinic: a tertiary hospital experience in Singapore. Int J Environ Res Public Health. 2022;19 doi: 10.3390/ijerph192114217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Low J.M., Lee L.Y., Ng Y.P.M., Zhong Y., Amin Z. Breastfeeding mother and child clinical outcomes after COVID-19 vaccination. J Hum Lact. 2022;38:37–42. doi: 10.1177/08903344211056522. [DOI] [PubMed] [Google Scholar]
- 30.Lyons D., Murray C., Hannigan S., Sui J., Alamin S., Conlon N., et al. Risk stratification through allergy history: single-centre experience of specialized COVID-19 vaccine clinic. Clin Exp Immunol. 2022;209:182–187. doi: 10.1093/cei/uxac064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Matson R.P., Niesen M.J.M., Levy E.R., Opp D.N., Lenehan P.J., Donadio G., et al. Paediatric safety assessment of BNT162b2 vaccination in a multistate hospital-based electronic health record system in the USA: a retrospective analysis. Lancet Digit Health. 2023;5:e206–e216. doi: 10.1016/S2589-7500(22)00253-9. [DOI] [PubMed] [Google Scholar]
- 32.Milito C., Cinetto F., Garzi G., Palladino A., Puca M., Brambilla E., et al. Safety of mRNA COVID-19 vaccines in patients with inborn errors of immunity: an Italian multicentric study. J Clin Immunol. 2023;43:299–307. doi: 10.1007/s10875-022-01402-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Najjar M., Albuaini S., Fadel M., Mohsen F., Group D.C. Covid-19 vaccination reported side effects and hesitancy among the Syrian population: a cross-sectional study. Ann Med. 2023;55 doi: 10.1080/07853890.2023.2241351. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Niekrens V., Esse J., Held J., Knobloch C.S., Steininger P., Kunz B., et al. Homologous COVID-19 BNT162b2 mRNA vaccination at a German tertiary care university hospital: a survey-based analysis of reactogenicity, safety, and inability to work among healthcare workers. Vaccines (Basel) 2022;10:650. doi: 10.3390/vaccines10050650. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ou M.T., Boyarsky B.J., Motter J.D., Greenberg R.S., Teles A.T., Ruddy J.A., et al. Safety and reactogenicity of 2 doses of SARS-CoV-2 vaccination in solid organ transplant recipients. transplantation. 2021;105:2170–2174. doi: 10.1097/TP.0000000000003780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rayamajhi S., Rafi M.A., Tripathi N., Dongol A.S., Pandey M., Rayamajhi S., et al. Adverse events following immunization with ChAdOx1 nCoV-19 and BBIBP-CorV vaccine: a comparative study among healthcare professionals of Nepal. PLoS One. 2022;17 doi: 10.1371/journal.pone.0272729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Rosada T., Napiórkowska-Baran K., Cała Ł., Alska E., Paulina K., Czarnowska M., et al. Safety of mRNA COVID-19 vaccinations in patients with allergic diseases. Public Health Pract (Oxf) 2023;5 doi: 10.1016/j.puhip.2022.100354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Sa S., Lee C.W., Shim S.R., Yoo H., Choi J., Kim J.H., et al. The safety of mRNA-1273, BNT162b2 and JNJ-78436735 COVID-19 vaccines: safety monitoring for adverse events using real-world data. Vaccines (Basel) 2022;10:320. doi: 10.3390/vaccines10020320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Sen P., Ravichandran N., Nune A., Lilleker J.B., Agarwal V., Kardes S., et al. COVID-19 vaccination-related adverse events among autoimmune disease patients: results from the COVAD study. Rheumatology (Oxford) 2022;62:65–76. doi: 10.1093/rheumatology/keac305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Shivarev A., Phillips A., Brophy-Williams S., Ford T., Richmond P., Effler P., et al. Adverse event reports of anaphylaxis after Comirnaty and Vaxzevria COVID-19 vaccinations, Western Australia, 22 February to 30 June 2021. Intern Med J. 2023;53:275–279. doi: 10.1111/imj.16001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Stehlin F., Mahdi-Aljedani R., Canton L., Monzambani-Banderet V., Miauton A., Girard C., et al. Intradermal testing with COVID-19 mRNA vaccines predicts tolerance. Front Allergy. 2022;3 doi: 10.3389/falgy.2022.818049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Sturkenboom M., Messina D., Paoletti O., de Burgos-Gonzalez A., García-Poza P., Huerta C., et al. Cohort monitoring of 29 adverse events of special interest prior to and after COVID-19 vaccination in four large European electronic healthcare data sources. medRxiv. August 2022. http://medrxiv.org/lookup/doi/10.1101/2022.08.17.22278894 Available at:
- 43.Vanijcharoenkarn K., Lee F.E.H., Martin L., Shih J., Sexton M.E., Kuruvilla M.E. Immediate reactions after the first dose of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) messenger RNA vaccines do not preclude second-dose administration. Clin Infect Dis. 2021;73:2108–2111. doi: 10.1093/cid/ciab448. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Wentrys Ł., Stopyra L. COVID-19 vaccination including 198 patients with history of severe anaphylactic reaction—own observations. Przegl Epidemiol. 2021;75:315–325. doi: 10.32394/pe.75.28. [DOI] [PubMed] [Google Scholar]
- 45.Wong C.K.H., Lau K.T.K., Xiong X., Au I.C.H., Lai F.T.T., Wan E.Y.F., et al. Adverse events of special interest and mortality following vaccination with mRNA (BNT162b2) and inactivated (CoronaVac) SARS-CoV-2 vaccines in Hong Kong: a retrospective study. PLoS Med. 2022;19 doi: 10.1371/journal.pmed.1004018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Ahsanuddin S., Jin R., Dhanda A.K., Georges K., Baredes S., Eloy J.A., et al. Otolaryngologic side effects after COVID-19 vaccination. Laryngoscope. 2024;134:1163–1168. doi: 10.1002/lary.30923. [DOI] [PubMed] [Google Scholar]
- 47.Wang C.W., Chen C.B., Lu C.W., Chen W.T., Hui R.C.Y., Chiu T.M., et al. Characteristics of immune response profile in patients with immediate allergic and autoimmune urticarial reactions induced by SARS-CoV-2 vaccines. J Autoimmun. 2023;138 doi: 10.1016/j.jaut.2023.103054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Aberumand B., Ayoub Goulstone W., Betschel S. Understanding attitudes and obstacles to vaccination against COVID-19 in patients with primary immunodeficiency. Allergy Asthma Clin Immunol. 2022;18:38. doi: 10.1186/s13223-022-00679-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Holzmann-Littig C., Frank T., Schmaderer C., Braunisch M.C., Renders L., Kranke P., et al. COVID-19 vaccines: fear of side effects among German health care workers. Vaccines (Basel) 2022;10:689. doi: 10.3390/vaccines10050689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Lewin B.J., Bronstein D., Tubert J.E., Chang J., Luo Y.X., Choi K.R., et al. COVID-19 vaccine concerns of health care providers and ancillary staff. Pediatr Res. 2023;93:460–462. doi: 10.1038/s41390-022-02171-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Njoga E.O., Mshelbwala P.P., Abah K.O., Awoyomi O.J., Wangdi K., Pewan S.B., et al. COVID-19 vaccine hesitancy and determinants of acceptance among healthcare workers, academics and tertiary students in Nigeria. Vaccines (Basel) 2022;10:626. doi: 10.3390/vaccines10040626. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Odejinmi F., Mallick R., Neophytou C., Mondeh K., Hall M., Scrivener C., et al. COVID-19 vaccine hesitancy: a midwifery survey into attitudes towards the COVID-19 vaccine. BMC Public Health. 2022;22:1219. doi: 10.1186/s12889-022-13540-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Oka P., Thia B.W.Q., Gunalan S.Z., Kwan J.R.Y., Ng D.X., Aau W.K., et al. Awareness, barriers and concerns of adolescents toward the COVID-19 vaccine: a cross-sectional study in Singapore. Front Public Health. 2022;10 doi: 10.3389/fpubh.2022.903152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Sayaca N., Aşık Cansız K., Yıldırım E., Öztürk B., Kırmaz C. The percentage of hesitation and factors associated with acceptance or refusal for COVID-19 vaccine: does training about vaccines by allergist affect personal decision? Rev Fr Allergol (2009) 2023;63 doi: 10.1016/j.reval.2022.09.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Wijesinghe M.S.D., Weerasinghe W.M.P.C., Gunawardana I., Perera S.N.S., Karunapema R.P.P. Acceptance of COVID-19 vaccine in Sri Lanka: applying the health belief model to an online survey. Asia Pac J Public Health. 2021;33:598–602. doi: 10.1177/10105395211014975. [DOI] [PubMed] [Google Scholar]
- 56.Yilmazbas P., Ozceker D. Allergic children’s parents’ hesitancy about COVID-19 vaccination. Asthma Allergy Immunol. 2023;21:18–24. [Google Scholar]
- 57.Sarca Çevk H., Baykan R., Peker A., Tekner S. Attitudes of family medicine residents in Ankara towards COVID-19 vaccines administered in Turkey: a descriptive, cross-sectional study. Family Med Primary Care Rev. 2022;24:156–161. [Google Scholar]
- 58.Batac A.L., Merrill K.A., Golding M.A., Abrams E.M., Bégin P., Ben-Shoshan M., et al. A qualitative investigation into vaccine hesitancy and confidence among people managing allergy. Ann Allergy Asthma Immunol. 2023;131:775–777. doi: 10.1016/j.anai.2023.08.600. [DOI] [PubMed] [Google Scholar]
- 59.National Academies of Sciences, Engineering, and Medicine, Health and Medicine Division, Board of Global Health, Forum on Microbial Threats . In: The critical public health value of vaccines: tackling issues of access and hesitancy: proceedings of a workshop. Nicholson A., Minicucci C., Liao J., editors. The National Academies Press; Washington, DC: 2021. https://nap.nationalacademies.org/catalog/26134/the-critical-public-health-value-of-vaccines-tackling-issues-of Available at: [PubMed] [Google Scholar]
- 60.Centers for Disease Control and Prevention Acceptability of adolescent COVID-19 vaccination among adolescents and parents of adolescents—United States, April 15-23, 2021. Weekly. 2021;70:997–1003. doi: 10.15585/mmwr.mm7028e1. https://www.cdc.gov/mmwr/volumes/70/wr/mm7028e1.htm?s_cid=mm7028e1_w Available at: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Abrams E.M., Shaker M., Sinha I., Greenhawt M. COVID-19 vaccines: addressing hesitancy in young people with allergies. Lancet Respir Med. 2021;9:1090–1092. doi: 10.1016/S2213-2600(21)00370-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Turner P.J., Larson H., Dubé È., Fisher A. Vaccine hesitancy: drivers and how the allergy community can help. J Allergy Clin Immunol Pract. 2021;9:3568–3574. doi: 10.1016/j.jaip.2021.06.035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Hourihane J.O., Byrne A.M., Blümchen K., Turner P.J., Greenhawt M. Ascertainment bias in anaphylaxis safety data of COVID-19 vaccines. J Allergy Clin Immunol Pract. 2021;9:2562–2566. doi: 10.1016/j.jaip.2021.04.025. [DOI] [PMC free article] [PubMed] [Google Scholar]