Abstract
Perivascular epithelioid cell tumors (PEComas), rare mesenchymal neoplasms with heterogeneous behavior, are molecularly characterized by TSC2 inactivation driving mammalian target of rapamycin (mTOR) pathway activation. We present a typical case of a 63-year-old female with metastatic high-grade PEComa featuring a TSC2 mutation (68.57% VAF) and elevated tumor mutational burden (19.7 mut/Mb), manifesting as peritoneal carcinomatosis and pulmonary metastases. Everolimus therapy following multidisciplinary assessment induced a radiologically confirmed partial response within 4.5 months with sustained clinical benefit. This outcome validates mTOR inhibition in TSC2-mutated PEComas and underscores the imperative of molecular profiling in mesenchymal tumor management. The significant mutational burden suggests potential immunotherapy responsiveness, informing future combination strategies. These findings emphasize molecularly guided precision approaches in rare malignancies and warrant systematic exploration of therapeutic sequencing and resistance mechanisms in mTOR-driven tumors.
Keywords: PEComa, mTOR inhibitor, Everolimus, TSC2 mutation, Case report
Introduction
Perivascular epithelioid cell tumors (PEComas) represent a rare family of mesenchymal neoplasms characterized by co-expression of melanocytic and smooth muscle markers, including HMB45, MelanA, and SMA [1, 2]. These tumors exhibit a broad clinical spectrum, ranging from benign lesions to aggressive malignancies with metastatic potential, most frequently involving the abdomen, retroperitoneum, and lungs [3, 4]. While surgical resection remains the mainstay for localized disease, advanced or metastatic PEComas pose significant therapeutic challenges due to their resistance to conventional chemotherapy and lack of standardized treatment plan [5, 6].
The molecular pathogenesis of PEComas has been increasingly elucidated, with inactivating mutations in TSC1 or TSC2 genes identified in 60–80% of cases [7]. These mutations disrupt the TSC1/TSC2 complex, leading to constitutive activation of the mammalian target of rapamycin complex 1 (mTORC1) pathway, which drives uncontrolled cell proliferation and survival [8, 9]. This mechanistic insight has propelled mTOR inhibitors—such as everolimus and sirolimus—to the forefront of targeted therapy for advanced PEComas [10]. Early clinical studies, including the landmark registrational AMPECT trial by Wagner et al. [11]. —which demonstrated a 39% objective response rate (ORR) to nab-sirolimus in advanced malignant PEComa and led to its FDA approval—support mTOR inhibition.
Despite these advances, challenges persist in predicting tumor behavior and optimizing treatment sequencing. Emerging data suggest that co-occurring genetic alterations (e.g., KRAS mutations) and tumor microenvironment features may modulate therapeutic responses [12]. Herein, we report a diagnostically complex case of metastatic PEComa harboring concurrent TSC2 and KRAS mutations, achieving partial response to everolimus. This case reinforces the therapeutic potential of mTOR inhibition in molecularly selected patients and underscores the need for comprehensive molecular profiling in rare sarcomas.
Case report
Clinical presentation and diagnosis
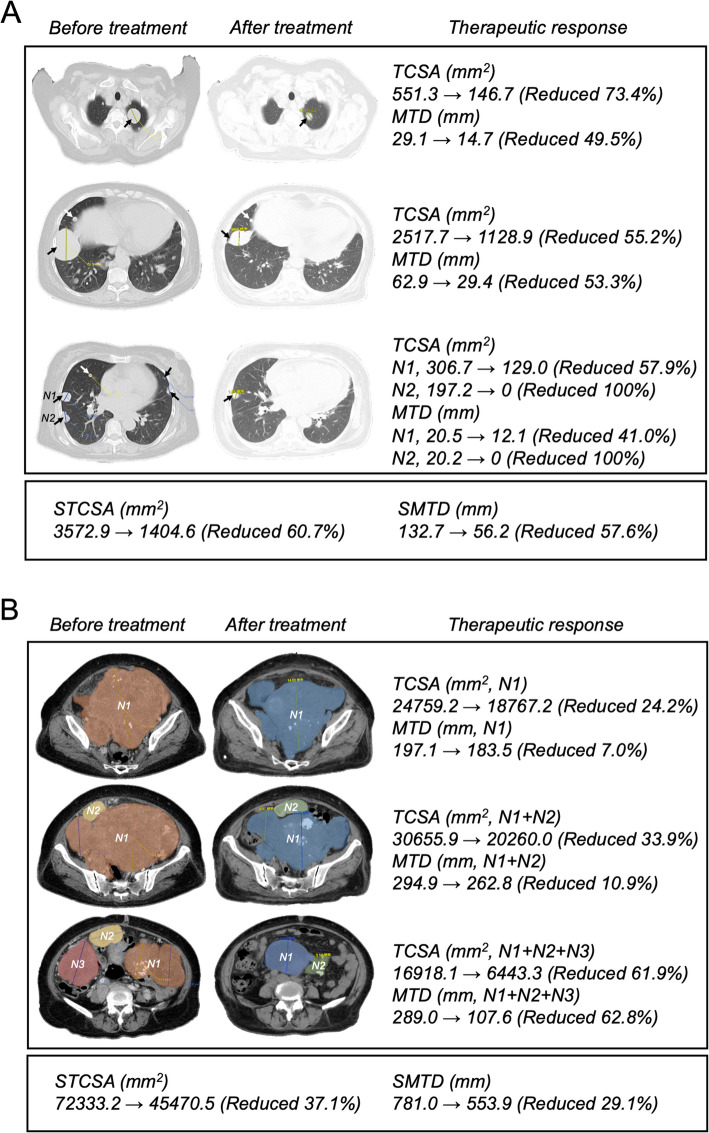
A 63-year-old female patient presented to our hospital with a six-month history of progressive abdominal distension and a two-week history of abdominal pain. On physical examination, abdominal distension was noted, but there were no signs of bowel sounds or peristaltic waves. The abdominal wall was soft, with no tenderness or rebound tenderness. A large palpable mass was detected below the umbilicus, with liver dullness present and shifting dullness not detectable. Bowel sounds were diminished, occurring at a rate of approximately one per minute. Laboratory investigations revealed an elevated CA125 level of 70.8 U/mL and increased lactate dehydrogenase (LDH) at 556 U/L. Enhanced chest computed tomography (CT) scan demonstrated multiple diffuse pulmonary nodules of varying sizes, with the largest located in the anterior basal segment of the right lower lung lobe, measuring approximately 63 × 53 mm (Fig. 1A). Enhanced abdominal and pelvic CT scans revealed multiple soft tissue masses of varying sizes and numerous enlarged lymph nodes in the retroperitoneum (Fig. 1B). These findings were suggestive of a malignant tumor with peritoneal carcinomatosis and pulmonary metastasis. Given the well-established predilection for uterine origin in PEComas, a focused gynecologic history was obtained and pelvic imaging was specifically re-evaluated. The patient unequivocally denied any prior history of gynecologic symptoms, including abnormal uterine bleeding, menorrhagia, or postmenopausal bleeding. Retrospective analysis of the pelvic CT imaging, performed as part of the comprehensive metastatic workup, confirmed the absence of uterine masses, endometrial abnormalities, or adnexal lesions. The documented soft-tissue masses and lymphadenopathy were exclusively retroperitoneal in location.
Fig. 1.
Diagnosis and therapeutic response in this PEcoma patient. (A) Chest CT imaging findings before and after treatment in this PEcoma patient; (B) Abdominal and pelvic CT imaging findings before and after treatment in this PEcoma patient. TCSA, Tumor cross-sectional area; MTD, Maximum tumor diameter; STCSA, Sum of tumor cross-sectional area of tumor; SMTD, Sum of the maximum tumor diameters
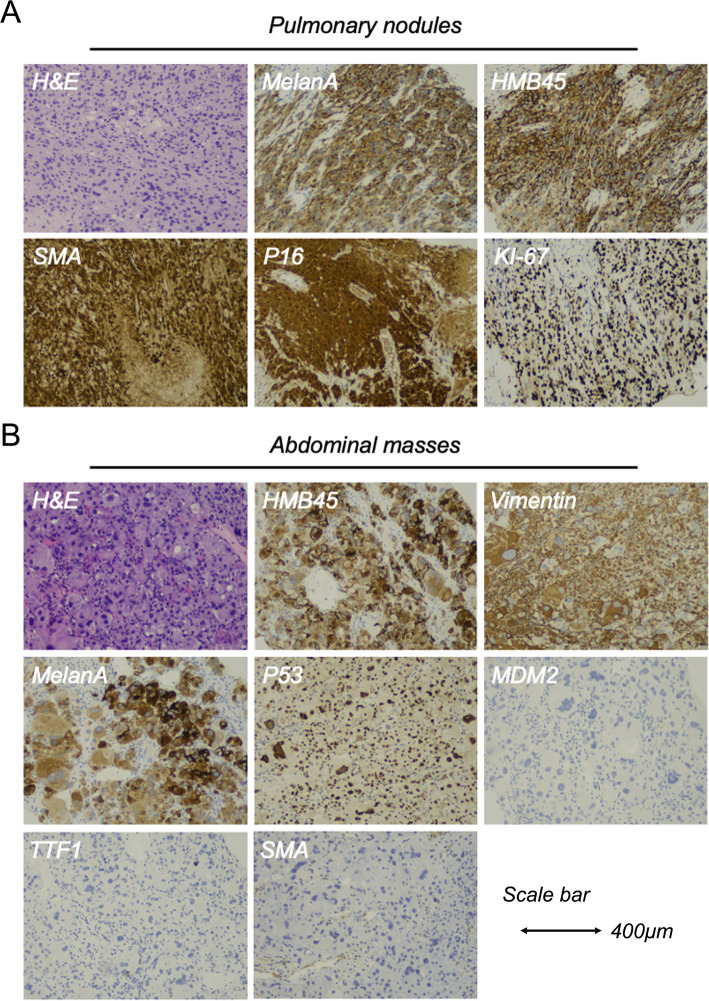
A CT-guided biopsy of the pulmonary nodules was performed, confirming the diagnosis of an interstitial-source malignant tumor. Histopathological examination revealed a neoplasm composed of both spindled and epithelioid cells, exhibiting significant cellular pleomorphism. Numerous mitotic figures were readily identified (averaging 8–10 per 10 high-power fields). Notably, immunohistochemistry revealed strong, diffuse nuclear p53 overexpression within the epithelioid tumor cells (> 70% of cells). This aberrant overexpression pattern is highly indicative of underlying TP53 gene mutation. The presence of this mutant p53 expression pattern, particularly in association with predominant epithelioid morphology, is a recognized adverse prognostic factor in PEComas, correlating with a significantly higher risk of aggressive clinical behavior, including local recurrence, metastasis, and disease-related mortality [13]. Immunohistochemical analysis also showed positivity for markers including Vimentin, HMB45, MelanA, TFE3, P16, and BRG1, with a Ki-67 proliferation index of 50% and partial positivity for CDK4. Markers such as Desmin, SMA, S-100, MDM2, CK7, CK20, TTF1, PAX8, SF1, beta-HCG, and p40 were negative (Fig. 2A). An ultrasound-guided biopsy of the abdominal and pelvic masses also indicated an interstitial-source malignant tumor. Immunohistochemical staining demonstrated positivity for SMA, HMB45, MelanA, P16, and CDK4, with a Ki-67 index of 80%, and isolated positivity for MDM2. The tumor was negative for markers such as S-100, CD34, CD117, DOG-1, and MUC4 (Fig. 2B).
Fig. 2.
Hematoxylin and eosin (HE) and immunohistochemical (IHC) staining in this PEcoma patient. (C) HE and IHC staining of pulmonary nodules in this PEcoma patient; (D) HE and IHC staining of abdominal mass in this PEcoma patient
To facilitate personalized treatment, the patient underwent next-generation sequencing (NGS) molecular testing. The results revealed a KRAS mutation (35.48%, Class II variant) and a TSC2 mutation (68.57%, Class III variant). The tumor proportion score (TPS) was 5%, and the combined positive score (CPS) was 10%. The tumor mutational burden (TMB) was measured at 19.7 mutations per megabase (mut/Mb). No mutations were detected in mismatch repair (MMR) genes, and there was a loss of heterozygosity in HLA. The tumor classified as microsatellite stable (MSS). Following a multidisciplinary discussion, a diagnosis of PEComa was established. Due to the presence of multiple metastases and the large size of the tumor, which precluded curative surgical intervention, palliative systemic therapy was recommended.
Effective disease control after everolimus treatment
Surgical resection deemed infeasible due to multifocal peritoneal/pulmonary involvement. First-line everolimus was recommended based on ESMO guidelines [14]. The patient commenced oral everolimus at a dosage of 5 mg twice daily. This regimen was maintained for five months. The follow-up CT scan demonstrated a partial response with a 57.6% reduction in pulmonary lesions and a 29.1% reduction in abdominal/pelvic lesions (Fig. 1A-B). Tumor marker CA125 levels remained relatively stable, showing no significant change from baseline levels. Throughout the treatment period, the patient experienced marked improvement in abdominal distension and pain, with no new adverse symptoms reported.
Discussions
PEComas represent a paradigm of mTOR-driven oncogenesis, with their biological complexity underscored by the interplay of molecular alterations and tumor microenvironment dynamics [15]. Our case of a metastatic TSC2/KRAS co-mutated PEComa with partial response to everolimus provides critical insights into three key areas: mechanistic foundations of mTOR dependency, biological implications of genetic heterogeneity, and therapeutic challenges in advanced disease.
Molecular drivers and therapeutic targeting
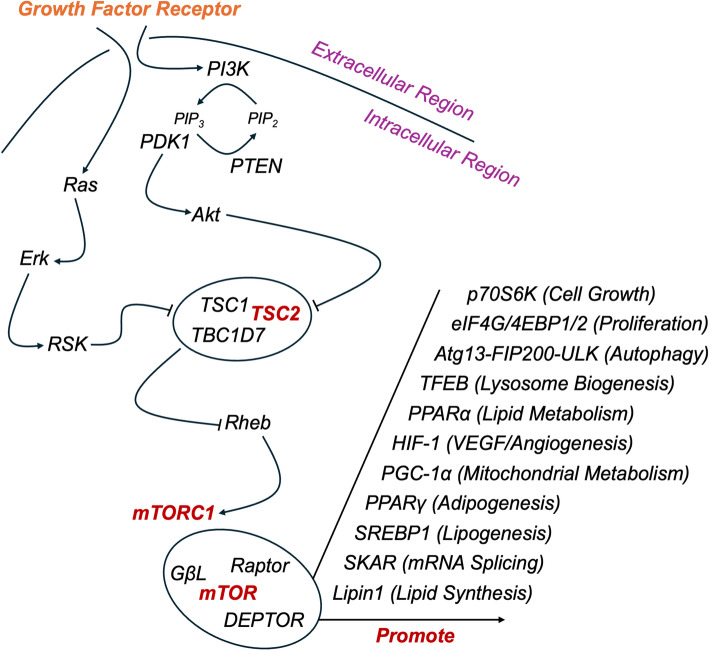
The discovery of mechanistic link between TSC1/2 inactivation and mTOR hyperactivation has significantly advanced the management of PEComa. The TSC complex serves as a critical negative regulator of mTORC1 by hydrolyzing Rheb-GTP, and its loss results in dysregulated protein synthesis and uncontrolled cell proliferation [16, 17] (Fig. 3). In this case, the identified TSC2 mutation (68.57% VAF) is consistent with genomic studies demonstrating TSC1/2 alterations in 60–80% of malignant PEComas, which are associated with increased sensitivity to mTOR inhibitor [7]. The pivotal EXIST-2 trial—which evaluated everolimus in TSC-associated angiomyolipomas, a PEComa-family neoplasm—established the clinical rationale for mTOR inhibition, reporting significant response rates (42% vs. 0%) and prolonged progression-free survival (HR = 0.08, P < 0.0001) [18]. Expanding upon this, real-world evidence from larger PEComa cohorts reinforces mTOR inhibition efficacy. Sanfilippo et al.. documented an ORR of 41% and disease stabilization in 36% of advanced PEComa patients treated with mTOR inhibitors (sirolimus/everolimus), further validating their therapeutic role across primary site [19]. Similarly, Dickson et al.. reported sustained clinical benefit in 80% of malignant PEComa patients receiving mTOR inhibitors [20]. While nab-sirolimus is FDA-approved for malignant PEComa based on the AMPECT trial (ORR: 39%, median duration of response: 10.6 months) [11], everolimus was selected here due to: (i) Global accessibility and extensive real-world safety data in TSC-driven diseases; (ii) Institutional formulary support; (iii) Uncertainty regarding nab-sirolimus efficacy in KRAS-co-mutated tumors. However, emerging evidence suggests molecular heterogeneity may influence outcomes. Argani et al.. noted inferior responses to mTOR inhibitors in TFE3-rearranged PEComas versus conventional subtypes [21], underscoring the relevance of TFE3-negative/TSC2-mutant profile in this case.
Fig. 3.
Schematic representation of the TSC1/2-mTOR signaling pathway
Our case exhibited a more pronounced tumor reduction (57.6% pulmonary, 29.1% abdominopelvic) than anticipated within 4.5 months. This aligns with reports of hyperresponsiveness in TSC2-mutant tumors, such as Testa et al.. where TSC2-inactivated PEComas showed deeper regression kinetics to mTOR inhibition than TSC1-altered counterparts [22]. This partial response may be linked to the elevated TMB (19.7 mut/Mb), as emerging evidence suggests that hypermutated tumors display increased mTOR dependency due to proteotoxic stress caused by misfolded proteins [23]. Although TMB is traditionally associated with immunotherapy response, its potential role in predicting mTOR inhibitor efficacy requires further prospective validation, particularly in light of the MSS status and low PD-L1 expression (CPS 10).
The co-occurrence of a KRAS G12D mutation (35.48% VAF) adds biological complexity to this case. While KRAS mutations are rare in PEComas, they may influence therapeutic outcomes through crosstalk between the MAPK and mTOR pathways [24]. Preclinical models indicate that KRAS-driven ERK activation can phosphorylate and destabilize TSC2, paradoxically suppressing mTORC1 activity while concurrently activating PI3K/AKT signaling [25, 26]. This dual regulatory effect may explain the sustained sensitivity to everolimus observed here, while also raising concerns about potential resistance, as KRAS-mutant tumors are known to exhibit reduced durability of mTOR-targeted therapies [27–29]. Importantly, longitudinal ctDNA monitoring, as recommended in recent guidelines, may enable the early detection of resistant clones and inform timely therapeutic adjustments [30].
Genetic heterogeneity and clinical implications
PEComas demonstrate substantial genetic heterogeneity extending beyond TSC1/2 alterations. Agaram et al.. reported recurrent TP53 mutations (63%) and chromatin rearrangement events involving TFE3 (23%) and RAD51B (8%) [7], which collectively underlie the spectrum of clinical behaviors observed in these tumors, ranging from indolent lesions to aggressive metastatic disease. The elevated Ki-67 proliferation index (80%) and rapid progression observed in this case align with genomic evidence linking TSC2 loss to increased mitotic activity and metastatic propensity [31]. Furthermore, the co-occurrence of KRAS and TSC2 mutations challenges conventional diagnostic frameworks. Although PEComas are histologically defined by melanocytic (HMB45/MelanA) and smooth muscle (SMA) markers, molecular profiling has become indispensable for differential diagnosis, particularly in tumors with atypical immunohistochemical features or aberrant metastatic patterns.
Notably, while immunohistochemical analysis revealed TFE3 positivity in this case, comprehensive molecular profiling did not detect a TFE3 gene fusion. This discordance underscores a critical diagnostic nuance: TFE3 immunostaining alone is not pathognomonic for TFE3-rearranged PEComa. Non-specific cytoplasmic or weakly nuclear TFE3 reactivity may occur in conventional PEComas and other mesenchymal neoplasms due to non-fusion-related mechanisms, including mTOR pathway hyperactivation or epigenetic dysregulation [15, 32]. In this context, the definitive exclusion of a TFE3 rearrangement by molecular analysis—coupled with the presence of a pathogenic TSC2 mutation, co-expression of melanocytic (HMB45/MelanA) and smooth muscle (SMA) markers, and absence of TFE3 fusion transcripts—robustly supports classification as a conventional PEComa rather than a TFE3-rearranged variant. This distinction carries clinical implications, as TFE3-rearranged tumors may exhibit distinct metastatic patterns and differential therapeutic responses.
Immunohistochemical pitfalls and diagnostic clarification
The immunohistochemical profile in this case warrants specific discussion regarding potential diagnostic mimics. Focal CDK4 positivity in the abdominal mass and isolated MDM2 overexpression in retroperitoneal lesions could theoretically raise consideration of dedifferentiated liposarcoma (DDLPS), particularly given CDK4/MDM2 co-amplification is a hallmark of DDLPS [33]. However, three key clinicopathological and molecular features definitively refute this differential: (i) NGS revealed a pathogenic TSC2 mutation (68.57% VAF), which is a genomic signature of PEComas but absent in DDLPS. DDLPS is instead characterized by MDM2 amplification (12q14-15), which was conspicuously absent in our NGS analysis. (ii) Both primary and metastatic lesions demonstrated strong, diffuse co-expression of melanocytic markers (HMB45, MelanA) and SMA. DDLPS lacks this immunoprofile, typically showing negative melanocytic markers and variable SMA expression. (iii) Pulmonary nodules were unequivocally MDM2-negative by IHC, inconsistent with DDLPS. CDK4 expression was focal (not diffuse) and occurred without concomitant MDM2 amplification—invalidating its specificity for DDLPS. Aberrant CDK4 expression is recognized in 15–20% of sarcomas, including PEComas, often reflecting cell cycle dysregulation rather than 12q amplification [34, 35]. This integrated diagnostic approach—prioritizing TSC2 mutational status, lineage-specific IHC, and stringent interpretation of “soft tissue sarcoma” markers—robustly confirms PEComa and eliminates DDLPS from consideration.
Unmet needs and future directions
Despite therapeutic advancements, PEComa management continues to present significant clinical challenges due to high recurrence rates and limited therapeutic options for refractory disease. While surgical resection remains curative for localized tumors, it is often not viable in metastatic cases such as ours. Conventional chemotherapy provides limited efficacy, with doxorubicin-based regimens demonstrating low ORR (13–20%) [19]. Targeted combination therapies represent a promising approach to circumvent resistance mechanisms: (i) Dual mTORC1/2 inhibitors (e.g., sapanisertib) have shown a 23% clinical benefit rate in everolimus-refractory advanced solid tumors, suggesting that comprehensive mTOR pathway inhibition may improve therapeutic outcomes [36]; (ii) MEK inhibitors (e.g., trametinib) exhibit preclinical efficacy in KRAS-mutant models by disrupting PD-1/PD-L1-mediated survival pathways, providing a biologically rational combinatorial strategy [37]; (iii) Autophagy inhibitors (e.g., hydroxychloroquine) synergized with everolimus in a phase I/II trial, achieving a 67% disease control rate (DCR), with 45% of DCR patients demonstrating progression-free survival exceeding 6 months [38].
This case highlights the critical need for adaptive trial designs incorporating real-time molecular monitoring. The durable response observed at 4.5 months suggests the potential for sustained clinical benefit; however, the absence of consensus guidelines for treatment duration or adjuvant therapy following initial response remains a major barrier. Emerging technologies such as patient-derived organoids (PDOs) hold substantial potential for advancing personalized therapy. Recent studies have demonstrated that PDO-based drug sensitivity testing predicts clinical responses with high predictive accuracy, enabling the optimization of tailored combination therapies for solid tumors [39, 40]. Furthermore, although the patient exhibited a favorable early treatment response, the short follow-up duration (5 months) precludes assessment of long-term outcomes. We will therefore maintain vigilant surveillance of the patient under continued everolimus therapy, including regular monitoring of tumor status, emergence of resistance, and potential serious complications. Should resistance develop, therapeutic transition to a dual mTORC1/2 inhibitor will be considered.
Conclusions
This case exemplifies the paradigm-shifting potential of molecularly guided therapy in PEComa management. While everolimus continues to serve as the therapeutic cornerstone for systemic treatment, the integration of longitudinal genomic surveillance and adaptive trial frameworks is imperative to counteract evolving resistance mechanisms. Our identification of elevated TMB in a clinically responsive patient provides a compelling rationale for expanding biomarker discovery efforts in this malignancy. Multidisciplinary collaboration, encompassing molecular pathology, clinical oncology, and translational research, is ultimately critical to refining therapeutic algorithms and improving outcomes for these rare neoplasms.
Acknowledgements
None.
Author contributions
Conception/design: M. X. and X. L.; Provision of study material or patients: X. S., L. Z. and X. Z.; Collection and/or assembly of data: R. D. and X. L.; Manuscript writing: X. S. and R. D.; Manuscript revised X. S. and X. L.; All authors reviewed the manuscript.
Funding
This study was supported by grants from the Young Elite Scientists Sponsorship Program (2023QNRC001). The study sponsors had no role in the design and preparation of this manuscript.
Data availability
All data supporting the findings of this study are available within the paper.
Declarations
Ethics approval and consent to participate
The study was approved by the Ethics Committee of Peking University International Hospital, and the study was conducted in accordance with the WMA declaration of Helsinki.
Consent for publication
Written informed consent for publication of this case report and any accompanying images was obtained from the patient’s legal guardian.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Xiaopeng Suo and Ruihan Dong contributed equally to this work.
Contributor Information
Xiangji Li, Email: scdxhxyxzx@163.com.
Mengmeng Xiao, Email: xiaomengmeng@pku.edu.cn.
References
- 1.Schoolmeester JK, Howitt BE, Hirsch MS, Dal Cin P, Quade BJ, Nucci MR. Perivascular epithelioid cell neoplasm (PEComa) of the gynecologic tract: clinicopathologic and immunohistochemical characterization of 16 cases. Am J Surg Pathol. 2014;38(2):176–88. [DOI] [PubMed] [Google Scholar]
- 2.Amante MF. Hepatic perivascular epithelioid cell tumors: benign, malignant, and uncertain malignant potential. World J Gastroenterol. 2024;30(18):2374–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Xie S, Lu J, Dong J, Shen Z. A case report of a perivascular epithelioid cell tumor in the liver. Asian J Surg. 2023;46(11):5092–3. [DOI] [PubMed] [Google Scholar]
- 4.Italiano A, Delcambre C, Hostein I, et al. Treatment with the mTOR inhibitor Temsirolimus in patients with malignant pecoma. Ann Oncol. 2010;21(5):1135–7. [DOI] [PubMed] [Google Scholar]
- 5.Wagner AJ, Malinowska-Kolodziej I, Morgan JA, et al. Clinical activity of mTOR Inhibition with sirolimus in malignant perivascular epithelioid cell tumors: targeting the pathogenic activation of mTORC1 in tumors. J Clin Oncol. 2010;28(5):835–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Musella A, De Felice F, Kyriacou AK, et al. Perivascular epithelioid cell neoplasm (PEComa) of the uterus: A systematic review. Int J Surg. 2015;19:1–5. [DOI] [PubMed] [Google Scholar]
- 7.Agaram NP, Sung YS, Zhang L, et al. Dichotomy of genetic abnormalities in PEComas with therapeutic implications. Am J Surg Pathol. 2015;39(6):813–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Charli-Joseph Y, Saggini A, Vemula S, Weier J, Mirza S, LeBoit PE. Primary cutaneous perivascular epithelioid cell tumor: a clinicopathological and molecular reappraisal. J Am Acad Dermatol. 2014;71(6):1127–36. [DOI] [PubMed] [Google Scholar]
- 9.Chiang S, Vasudevaraja V, Serrano J, et al. TSC2-mutant uterine sarcomas with JAZF1-SUZ12 fusions demonstrate hybrid features of endometrial stromal sarcoma and pecoma and are responsive to mTOR Inhibition. Mod Pathol. 2022;35(1):117–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wagner AJ, Ravi V, Riedel RF, et al. nab-Sirolimus for patients with malignant perivascular epithelioid cell tumors. J Clin Oncol. 2021;39(33):3660–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wagner AJ, Ravi V, Riedel RF, et al. Phase II Trial of nab-Sirolimus in Patients With Advanced Malignant Perivascular Epithelioid Cell Tumors (AMPECT): Long-Term Efficacy and Safety Update. J Clin Oncol. 2024;42(13):1472–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Skoulidis F, Goldberg ME, Greenawalt DM, et al. STK11/LKB1 mutations and PD-1 inhibitor resistance in KRAS-Mutant lung adenocarcinoma. Cancer Discov. 2018;8(7):822–35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yoo Y, Kim J, Song IH. Risk prediction criteria for the primary hepatic perivascular epithelioid cell tumour family, including angiomyolipoma: analysis of 132 cases with a literature review. Histopathology. 2025;86(6):979–92. [DOI] [PubMed] [Google Scholar]
- 14.Gronchi A, Miah AB, Dei Tos AP, et al. Soft tissue and visceral sarcomas: ESMO-EURACAN-GENTURIS clinical practice guidelines for diagnosis, treatment and follow-up☆. Ann Oncol. 2021;32(11):1348–65. [DOI] [PubMed] [Google Scholar]
- 15.Schmiester M, Dolnik A, Kornak U, et al. TFE3 activation in a TSC1-altered malignant pecoma: challenging the dichotomy of the underlying pathogenic mechanisms. J Pathol Clin Res. 2021;7(1):3–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Xie Y, Zhao Y, Shi L, et al. Gut epithelial TSC1/mTOR controls RIPK3-dependent necroptosis in intestinal inflammation and cancer. J Clin Invest. 2020;130(4):2111–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kwiatkowski DJ, Choueiri TK, Fay AP, et al. Mutations in TSC1, TSC2, and MTOR are associated with response to Rapalogs in patients with metastatic renal cell carcinoma. Clin Cancer Res. 2016;22(10):2445–52. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Bissler JJ, Kingswood JC, Radzikowska E, et al. Everolimus for Angiomyolipoma associated with tuberous sclerosis complex or sporadic Lymphangioleiomyomatosis (EXIST-2): a multicentre, randomised, double-blind, placebo-controlled trial. Lancet. 2013;381(9869):817–24. [DOI] [PubMed] [Google Scholar]
- 19.Sanfilippo R, Jones RL, Blay JY, et al. Role of chemotherapy, VEGFR inhibitors, and mTOR inhibitors in advanced perivascular epithelioid cell tumors (PEComas). Clin Cancer Res. 2019;25(17):5295–300. [DOI] [PubMed] [Google Scholar]
- 20.Dickson MA, Schwartz GK, Antonescu CR, Kwiatkowski DJ, Malinowska IA. Extrarenal perivascular epithelioid cell tumors (PEComas) respond to mTOR inhibition: clinical and molecular correlates. Int J Cancer. 2013;132(7):1711–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Argani P, Gross JM, Baraban E, et al. TFE3 -Rearranged PEComa/PEComa-like neoplasms: report of 25 new cases expanding the clinicopathologic spectrum and highlighting its association with prior exposure to chemotherapy. Am J Surg Pathol. 2024;48(7):777–89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Testa S, Bui NQ, Ganjoo KN. Systemic treatments and molecular biomarkers for perivascular epithelioid cell tumors: A Single-institution retrospective analysis. Cancer Res Commun. 2023;3(7):1212–23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Wang Z, Wang X, Xu Y, et al. Mutations of PI3K-AKT-mTOR pathway as predictors for immune cell infiltration and immunotherapy efficacy in dMMR/MSI-H gastric adenocarcinoma. BMC Med. 2022;20(1):133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Luo YD, Liu XY, Fang L, et al. Mutant Kras and mTOR crosstalk drives hepatocellular carcinoma development via PEG3/STAT3/BEX2 signaling. Theranostics. 2022;12(18):7903–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.De Luca A, Maiello MR, D’Alessio A, Pergameno M, Normanno N. The RAS/RAF/MEK/ERK and the PI3K/AKT signalling pathways: role in cancer pathogenesis and implications for therapeutic approaches. Expert Opin Ther Targets. 2012;16(Suppl 2):S17–27. [DOI] [PubMed] [Google Scholar]
- 26.Shimizu T, Tolcher AW, Papadopoulos KP, et al. The clinical effect of the dual-targeting strategy involving PI3K/AKT/mTOR and RAS/MEK/ERK pathways in patients with advanced cancer. Clin Cancer Res. 2012;18(8):2316–25. [DOI] [PubMed] [Google Scholar]
- 27.Isermann T, Sers C, Der CJ, Papke B. KRAS inhibitors: resistance drivers and combinatorial strategies. Trends Cancer. 2025;11(2):91–116. [DOI] [PubMed] [Google Scholar]
- 28.Knight JRP, Alexandrou C, Skalka GL, et al. MNK Inhibition sensitizes KRAS-Mutant colorectal cancer to mTORC1 Inhibition by reducing eIF4E phosphorylation and c-MYC expression. Cancer Discov. 2021;11(5):1228–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Mohseni M, Park BH. PIK3CA and KRAS mutations predict for response to everolimus therapy: now that’s RAD001. J Clin Invest. 2010;120(8):2655–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dumbrava EE, Call SG, Huang HJ, et al. PIK3CA mutations in plasma Circulating tumor DNA predict survival and treatment outcomes in patients with advanced cancers. ESMO Open. 2021;6(5):100230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hsieh HJ, Zhang W, Lin SH, et al. Systems biology approach reveals a link between mTORC1 and G2/M DNA damage checkpoint recovery. Nat Commun. 2018;9(1):3982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Agaimy A, Acosta AM, Cheng L, et al. TFE3-rearranged nonmelanotic renal pecoma: a case series expanding their phenotypic and fusion landscape. Histopathology. 2024;85(5):783–93. [DOI] [PubMed] [Google Scholar]
- 33.Tyler R, Wanigasooriya K, Taniere P, et al. A review of retroperitoneal liposarcoma genomics. Cancer Treat Rev. 2020;86:102013. [DOI] [PubMed] [Google Scholar]
- 34.Binh MB, Sastre-Garau X, Guillou L, et al. MDM2 and CDK4 immunostainings are useful adjuncts in diagnosing well-differentiated and dedifferentiated liposarcoma subtypes: a comparative analysis of 559 soft tissue neoplasms with genetic data. Am J Surg Pathol. 2005;29(10):1340–7. [DOI] [PubMed] [Google Scholar]
- 35.Schaefer IM, Fletcher CD, Hornick JL. Loss of H3K27 trimethylation distinguishes malignant peripheral nerve sheath tumors from histologic mimics. Mod Pathol. 2016;29(1):4–13. [DOI] [PubMed] [Google Scholar]
- 36.Lim B, Potter DA, Salkeni MA, et al. Sapanisertib plus exemestane or fulvestrant in women with hormone Receptor-Positive/HER2-Negative advanced or metastatic breast cancer. Clin Cancer Res. 2021;27(12):3329–38. [DOI] [PubMed] [Google Scholar]
- 37.Puyalto A, Rodríguez-Remírez M, López I, et al. Trametinib sensitizes KRAS-mutant lung adenocarcinoma tumors to PD-1/PD-L1 axis Blockade via Id1 downregulation. Mol Cancer. 2024;23(1):78. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Haas NB, Appleman LJ, Stein M, et al. Autophagy Inhibition to augment mTOR inhibition: a phase I/II trial of everolimus and hydroxychloroquine in patients with previously treated renal cell carcinoma. Clin Cancer Res. 2019;25(7):2080–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Farin HF, Mosa MH, Ndreshkjana B, et al. Colorectal cancer Organoid-Stroma biobank allows Subtype-Specific assessment of individualized therapy responses. Cancer Discov. 2023;13(10):2192–211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Fang X, Shu L, Chen T, et al. Organoids derived from patients provide a new opportunity for research and individualized treatment of malignant peritoneal mesothelioma. Mol Cancer. 2024;23(1):12. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
All data supporting the findings of this study are available within the paper.