Abstract
Vitamin D is a crucial element in bone metabolism and plays a role in innate immunity and inflammation suppression. Deficiency in this vitamin leads to disturbances in many biological functions, including the lipid profile. The decrease in HDL levels is associated with disruptions in its function and dynamic modifications in its components, such as apolipoproteins, including ApoM, ApoA-1, and ApoD. Consequently, the anti-inflammatory potential of HDL and HDL-associated proteins is reduced, resulting in heightened inflammation. However, the relationship between modifications in lipid profile, apolipoproteins, inflammation, and vitamin D remains unclear. This review highlights the connection between vitamin D status and its possible effects on the lipid profile, specifically HDL-associated proteins.
Keywords: Vitamin D, Inflammation, HDL, ApoM, ApoA-1, Immune modulation
Introduction and background
Vitamin D, a fat-soluble prohormone, plays a crucial role in maintaining bone health by regulating calcium and phosphorus homeostasis. It is obtained from dietary sources (e.g., salmon, egg yolk, mushrooms) or synthesized through exposure to UVB radiation. The native form of vitamin D converts in the liver to 25-hydroxyvitamin D [25(OH)D], a major circulatory vitamer. Subsequently, the active form of vitamin D is produced in the kidney through the conversion of 25(OH)D to 1,25-dihydroxyvitamin D (1,25(OH)2D) via 25-hydroxyvitamin D-1α-hydroxylase [1]. The native form of vitamin D has two analogs: D2 and D3. D2, also known as Ergocalciferol, is derived from plant sources, such as soybeans, while D3 (cholecalciferol) comes from animal sources, like fish. The differences in the structures of both forms affect their binding affinity to various proteins, including their carrier, vitamin D-binding protein (DBP), which impacts their plasma half-life. D2 has a shorter plasma half-life and lower affinity for binding DBP and the vitamin D receptor (VDR) [2].
VDR is a member of a superfamily of nuclear receptors working as a transcription regulator. The VDR gene is located on chromosome 12q13.11 [2]. VDR is usually allocated in the cytoplasm. Specifically, VDR is highly expressed in immune cells, modulating inflammatory responses [3]. Conformational changes occur upon interacting with VDR and 1,25(OH)2D3. Later, VDR dimers bind to retinoid X receptor (RXR) and transfer to the nucleus. The complex binds to the vitamin D response elements (VDREs) in the nucleus of vitamin D-responsive genes. Subsequently, this process activates or suppresses certain genes according to the target genes [4, 5].
Vitamin D: physiology and deficiency
Vitamin D is essential for maintaining bone health by regulating the absorption of calcium and phosphorus in the small intestine and promoting bone mineralization [1]. It also supports muscle function and systemic immune responses. Deficiency in vitamin D impairs calcium homeostasis, leading to conditions such as rickets in children and osteomalacia in adults, as well as an increased risk of osteoporosis due to reduced bone mineral density [1, 6]. Beyond bone health, vitamin D deficiency disrupts immune regulation, increasing susceptibility to infections and exacerbating chronic inflammatory conditions, including metabolic syndrome and dyslipidemia [7].
Vitamin D regulates numerous biological processes, with the vitamin D receptor (VDR) highly expressed in T lymphocytes and macrophages, thereby modulating immunity by reducing T lymphocyte activity and promoting the transcription of TGFβ-1 and IL-4 [8]. Interferon (IFN)-γ is another example; the ligand-bound VDR and VDRE form a complex that binds to the IFN-γ promoter region, resulting in a repressive effect. At the same time, it increases IL-10 production, IL-10 is an anti-inflammatory cytokine [9].
NF-κB is one of 1,25(OH)2D3 targets. Upon the activation of NF-κB, a downstream target nucleotide-binding oligomerization domain 2 (NOD2) will be stimulated. NOD2 is a pathogen-recognizing protein. Subsequently, human β-defensin 2 DEFB4 (skin-antimicrobial peptide 1 (SAP1)) will be activated by NOD2; this process is downregulated by the presence of 1,25(OH)2D3. Another antimicrobial peptide, cathelicidin AMP (CAMP)( LL-37 is another name for the human cathelicidin), is affected by vitamin D, where it has been noticed to be highly induced by 1,25(OH)2D3. The activity of 1,25(OH)2D3 in stimulating the antimicrobial peptides was seen against some pathogens, such as Pseudomonas aeruginosa. This effect has been reported in human immune cells, keratinocytes, monocytes, and neutrophils [10–12].
Additionally, 1,25(OH)2D3 has been demonstrated to activate macrophage autophagy, and autophagy-associated proteins Atg-5 and Beclin-1. Those proteins are also induced through the upregulation of cathelicidin. Furthermore, cathelicidin or LL-37 activation leads to the induction of p38, ERK, and C/EBPβ [13]. Autophagy is a catabolic biological process mediated by a lysosome, aiming to remove unwanted or dysfunctional components [14].
Mature T cells exhibit elevated VDR expression and CYP27B1, the enzyme converting vitamin D to active 1,25(OH)₂D₃, compared to naive T cells, highlighting 1,25(OH)₂D₃’s role in T cell regulation. It suppresses Th1 and Th17 cytokines (IL-2, IFN-γ, TNF-α, IL-17, IL-21, IL-22), promotes Th2 cytokines (IL-4, IL-5, IL-10), and induces FoxP3 for regulatory T cell differentiation [15–17].
Vitamin D deficiency represents a significant global public health issue. Reports are pointing to 1 billion people suffering from various degrees of vitamin D deficiency and insufficiency. The Institute of Medicine previously defined vitamin D deficiency as serum 25-hydroxyvitamin D (25(OH)D) concentrations below 12 ng/mL, insufficiency as 12–20 ng/mL, and normal levels above 20 ng/mL; however, current clinical guidelines define deficiency as below 20 ng/mL, insufficiency as 20–30 ng/mL, and sufficiency at or above 30 ng/mL [6, 18]. Vitamin D deficiency disrupts immune homeostasis by enhancing pro-inflammatory pathways, such as NF-κB and MAPK signaling, which are implicated in chronic diseases like metabolic syndrome and dyslipidemia [7]. For example, low 25(OH)D levels (< 20 ng/mL) increase circulating inflammatory markers, such as C-reactive protein (CRP) and interleukin-6 (IL-6), which contribute to insulin resistance and atherogenesis [7, 19]. These inflammatory changes are associated with altered lipid profiles, including reduced high-density lipoprotein (HDL) cholesterol and elevated triglycerides, which exacerbate cardiovascular risk [19]. Vitamin D also influences lipid metabolism, particularly high-density lipoprotein (HDL), a lipoprotein complex that facilitates reverse cholesterol transport and exhibits anti-inflammatory properties [20]. Low 25(OH)D levels are associated with reduced HDL cholesterol, which may exacerbate inflammation and cardiovascular risk [21]. As demonstrated in our previous publication, low 25(OH)D levels (< 20 ng/mL) in young, healthy adults were associated with an increased monocyte-to-HDL ratio (MHR), a biomarker of subclinical inflammation that reflects the balance between pro-inflammatory monocytes and anti-inflammatory HDL. This suggests that vitamin D deficiency may impair HDL’s protective functions, contributing to cardiovascular risk [19].
Vitamin D in cardiovascular health
The risk for cardiovascular diseases is known to be impacted by several lifestyle factors, such as smoking, drinking alcohol, and malnutrition, in addition to metabolic abnormalities, such as diabetes, insulin resistance, and obesity. Those factors are cross-linked with risk factors of vitamin D deficiency [22, 23]. A prospective study by Giovannucci et al. followed 18,225 male US health professionals (aged 40–75 years, predominantly non-obese) over 10 years and found that low 25(OH)D levels (≤ 15 ng/mL) were associated with a 2-fold increased risk of myocardial infarction (adjusted relative risk: 2.09, 95% CI: 1.24–3.54) compared to sufficient levels (≥ 30 ng/mL), after controlling for age, smoking, and other cardiovascular risk factors [24]. Similarly, research on coronary angiography patients over 7.7 years linked reduced 25(OH)D and 1,25(OH)₂D levels to increased cardiovascular mortality [25]. Furthermore, low 25(OH)D significantly elevates risks of heart failure and sudden cardiac death [26].
Vitamin D, in general, protects against cardiovascular diseases through multi-layer mechanisms. The first mechanism by which vitamin D helps relax vessels is by stimulating nitric oxide (NO) production from endothelial cells [27]. Moreover, vitamin D suppresses NFkappaB and p38, which are responsible for activating the inflammatory pathways leading to exacerbation of the atherosclerotic disorder [21].
Similarly, HDL possesses anti-inflammatory effects, and disturbance in HDL and lipid profile would contribute to inflammation.
Restoring the optimal level of serum 25(OH)D above 20ng/mL (50nmol/L)- 30ng/mL (75nmol/L), preferably between 40 and 60 ng/mL (100–150 nmol/L) has documented health benefits including improving the outcomes of chronic disease and reducing the risks of getting the disease in the first place.
In vitro studies have shown that vitamin D supplementation enhances immune response toward infection, including increasing phagocytes, suppressing inflammatory cytokine production, and inducing antimicrobial peptides such as cathelicidin expression [28].
Dyslipidemia and lipoproteins profile
Dyslipidemia involves a range of abnormalities in the metabolism of plasma lipids and lipoproteins, resulting in augmenting or reducing the concentrations of lipoproteins or changing the particle configuration [29]. Dyslipidemia triggers can be related to genetic factors, causing familial dyslipidemia, also called primary dyslipidemia. Conversely, environmental factors such as diseases, diet, and general life regime cause acquired dyslipidemia, also named secondary dyslipidemia [30]. Total cholesterol (TC), low-density lipoprotein cholesterol (LDL-c), or high-density lipoprotein cholesterol (HDL-c) concentrations deviate when suffering from dyslipidemia [31]. Dyslipidemia is defined by elevated total cholesterol (≥ 5.2 mmol/L), LDL-C (≥ 3.0 mmol/L), triglycerides (≥ 1.7 mmol/L), or reduced HDL-C (< 1.0 mmol/L in men, < 1.3 mmol/L in women), with stricter LDL-C targets (< 1.8 mmol/L) for high-risk patients [32, 33]. Variation in lipid metabolites marks dyslipidemia as a significant risk factor in several diseases, for instance, cardiovascular disease, diabetes, and stroke [34]. Research on dyslipidemia and related macromolecules has linked Vitamin D deficiency to increased risk, given that VDRs are present in multiple cell types, such as vascular endothelial cells and cardiomyocytes [29]. Dyslipidemia is a significant element in metabolic syndrome, which is characterized by a combination of several pathological conditions, mainly central obesity, hypertension, low levels of HDL, elevated LDL and TG, endothelial dysfunction, and diabetes [35]. Studies reported a strong correlation between metabolic syndrome and vitamin D deficiency, but the bidirectional nature of this association is still not explicit [36, 37] Lipoproteins, including chylomicrons, low-density lipoproteins (LDL), very-low-density lipoproteins (VLDL), and high-density lipoproteins (HDL), are the main players in lipid profile disruption; therefore, CVD [34, 38].
LDL particle
LDL is the primary transporter of cholesterol to peripheral tissues. It drives atherosclerosis plaque formation and cardiovascular disease [39]. LDL is formed via VLDL conversion by lipoproteins [40]. LDL contains apolipoprotein, Apo B-100, and lipid like linoleate [41].
Elevated LDL promotes atherosclerosis. When LDL enters the endothelial layer, free radicals oxidize the LDL particle, forming ox-LDL [42, 43], stimulating the endothelial cells to produce Monocyte Chemoattractant Protein-1 (MCP-1). This triggers monocytes to infiltrate and differentiate into macrophages. Those macrophages ingest ox-LDL, forming foam cells. The accumulation of foam cells drives plaque formation [31, 44, 45]. Vitamin D deficiency may exacerbate LDL-driven atherosclerosis by reducing VDR-mediated suppression of inflammatory pathways, such as NF-κB, which enhances the formation of ox-LDL and foam cell accumulation [25, 46, 47]. Low 25(OH)D levels (< 20 ng/mL) are associated with increased LDL cholesterol in observational studies [48], suggesting a role in dyslipidemia.
VLDL particle
VLDL, triglyceride-rich particles, are produced in the liver [49]. High concentrations of VLDL-TG were linked to an increased risk of cardiovascular disease [50]. Vitamin D deficiency is associated with elevated VLDL triglycerides, potentially due to impaired VDR signaling in hepatocytes, which disrupts triglyceride metabolism [51]. This contributes to dyslipidemia and heightened cardiovascular risk.
Chylomicrons
Chylomicrons are formed in the intestine from dietary lipids, transport triglycerides to peripheral tissue, and their remnants are cleared by ApoE-mediated endocytosis [52]. Although direct evidence linking vitamin D to chylomicron metabolism is limited, low 25(OH)D levels may indirectly influence triglyceride transport by altering ApoE expression, as VDR regulates lipid metabolism genes [53].
HDL particles
HDL, known for reverse cholesterol transport (RCT), mitigates atherosclerosis by shuttling cholesterol from tissues to the liver. HDL exhibits anti-inflammatory, anti-apoptotic, and antioxidant properties, contributing to its protective effects against CVD [54]. HDL-cholesterol (HDL-c) is a good indicator of the functionality of HDL and the ability to carry cholesterol from tissues more than HDL particle (HDL-P). However, HDL-P has been more studied in clinical studies in cardiovascular disorders in isolation from HDL-c [55]. Low HDL-c has a significant impact on disease prognosis or survival rate. For instance, in severe sepsis, it has been observed that patients who have low values of HDL upon the beginning of sepsis deteriorate very rapidly, and have the worst mortality and clinical outcomes [56]. High values of HDL-c in patients with reduced ejection fraction were associated with a lower risk of heart failure and a better prognosis and survival. In a similar study, patients with high HDL-c recorded a decline in inflammation biomarker C-reactive protein (CRP) values [52].
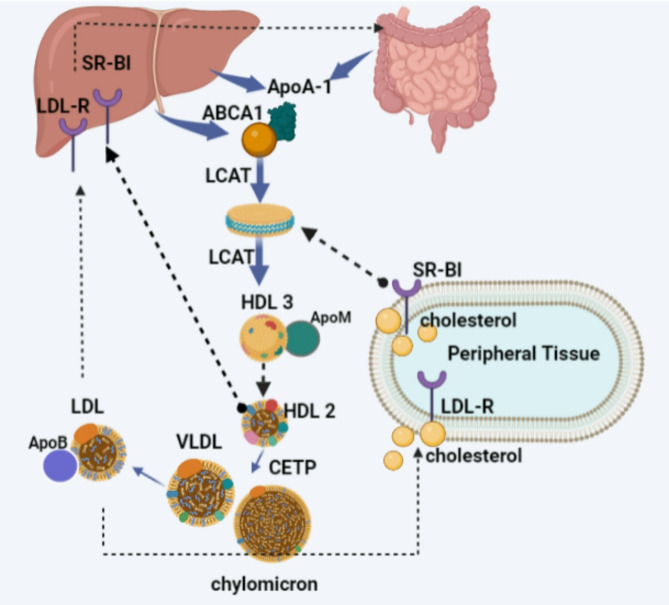
HDL is a very condensed, heterogeneous complex in comparison to other lipoproteins. HDL size ranges from 1.063 to 1.210 g/mL. It contains a high composition of proteins in comparison with other lipoproteins [57]. Proteins form up to 60% of HDL mass, and ApoA-I is the main component of those proteins [58]. The biogenesis of HDL starts mainly in the liver upon the interaction between ApoA-I and the cell membrane protein ATP-binding cassette transporter A1 (ABCA1) [59]. ABCA1 is found in the basolateral surface of the hepatocytes [60]. The mutation in ABCA1 proteins could result in severe malfunctioning in HDL. As a result, a large amount of cholesterol is deposited in tissue macrophages, leading to atherosclerotic formation [61]. Later, this lipidated ApoA-I is catalyzed by an enzyme called lecithin/cholesterol acyltransferase (LCAT) [62]. The first step of transforming the immature HDL into mature HDL occurs when scavenger receptor class B, type I (SR-BI) mediates the cholesterol efflux from macrophages and tissues into discoidal HDL [63].
The second step in transforming the discoidal hydrophobic core of ABCA1-derived HDL into spherical lipid-rich HDL is the interaction with LCAT for the second time [64]. A mutation in LCAT is also associated with cardiovascular diseases. Two disorders are associated with LCAT mutation: familial LCAT deficiency (FLD), which affects the esterification of cholesterol on HDL and LDL, resulting in the accumulation of discoidal HDL in the plasma. Fish eye disease (FED) affects the esterification of cholesterol on HDL only [65].
The cholesteryl ester transfer protein (CETP) pathway follows the RCT process. In this step, CETP facilitates the exchange of cholesteryl esters from HDL particles with the triglycerides in low-density lipoprotein (LDL), very-low-density lipoprotein (VLDL), and Chylomicrons. The liver takes up the cholesterol via the LDL receptor, which is present in VLDL and LDL [66] (Fig. 1).
Fig. 1.
The biogenesis of HDL and RCT. Created by Hanaa Mousa using the BioRender website. Upon the interaction of ApoA-1 and ABCA1, the resulting complex is catalyzed by the LCAT enzyme, forming a discoidal HDL. SR-B1 mediates cholesterol efflux from macrophages and tissues into discoidal high-density lipoprotein (HDL). Another interaction with LCAT formed the mature HDL. CETP facilitates the exchange of cholesteryl esters from HDL particles with the triglycerides in LDL, VLDL, and Chylomicrons. The liver takes the cholesterol via the LDL receptor. Note: Figures 1, and 3 Created with BioRender.com
HDL is a dynamic particle, that undergoes continuous remodeling due to the instant interaction of cell receptors and other proteins and cofactors particularly serum amyloid A (SAA), hepatic lipase (HL), endothelial lipase (EL), cholesteryl ester transfer protein (CETP), phospholipid transfer protein (PLTP), scavenger receptor class B type I (SR-BI), ATP-binding cassette transporter G1 (ABCG1), the F1 subunit of ATPase (Ecto F1-ATPase), and apolipoprotein M (ApoM) [67, 68]. Furthermore, the HDL-associated apolipoproteins, which are bound to HDL, interact with numerous cellular receptors, e.g., ATP-binding cassette A1 or G1. This interaction enables cholesterol efflux from cells to the HDL particle [69]. Consequently, HDL particles continuously remodel their physical structure and metabolite composition [70]. Inflammation impairs the reverse cholesterol transport (RCT) pathway, hindering cholesterol efflux from macrophage foam cells to the liver and resulting in dysfunctional high-density lipoprotein (HDL) particles [71].
HDL anti-inflammatory properties
The apolipoproteins on the HDL surface, such as ApoA-1, ApoM, and ApoD, which are mainly associated with HDL particles, more than the other lipoproteins [72, 73], enabling HDL to exhibit anti-inflammatory and anti-oxidative properties [58]. HDL suppresses the sphingosine kinase in the endothelial cell, an enzyme derived from sphingomyelins and considered essential in the activation of nuclear factor κB (NF-κB), which induces adhesion molecules [57]. In addition, HDL can suppress those adhesion molecules directly. An in vitro study in human umbilical vein endothelial cells (HUVECs) demonstrated that HDL suppressed cytokine-induced adhesion molecules, including VCAM-1, ICAM-1, and E-selectin, in a concentration-dependent manner [74]. Therefore, HDL suppresses the adhesion molecules produced by endothelial cells, which is crucial for the cellular adhesion of circulating leukocytes during atherogenesis formation [75].
Van der Vorst et al. study. In vitro experiments using murine and human primary macrophages demonstrated that HDL triggers a pro-inflammatory response through passive cholesterol depletion. The result was confirmed using peritoneal macrophages from ApoA-1 transgenic mice, which have elevated HDL levels. Pseudomonas aeruginosa bacteria were used to induce infectious inflammatory conditions. HDL activated the TLR in general, particularly TLR4, through the PKC-NF-kB/STAT1-IRF1 pathway, followed by an elevation of inflammatory cytokine expression IL-12 and TNF-α and down-regulation in the anti-inflammatory cytokine IL-10 [76].
HDL possesses some Antioxidant properties due to its association with antioxidant molecules, e.g., paraoxonase/arylesterase 1 (PON1). PON1 enables the HDL to prevent LDL glycation and lipid peroxidation, thus protecting the cardiovascular system from the bad effects of glycated LDL particles [77].
Vitamin D enhances HDL’s anti-inflammatory properties by upregulating ApoA-1 and ApoM expression in some contexts, though 1,25(OH)₂D₃ may suppress ApoA-1 transcription in hepatocytes [78, 79]. For example, supplementation with vitamin D (1000 mg capsule) daily for one month in healthy children increased HDL cholesterol level, potentially improving its anti-inflammatory capacity [80].
Clinical aspects of HDL
A low serum concentration of HDL, Less than 40 mg/dL (1.0 mmol/L) in men and Less than 50 mg/dL (1.3 mmol/L) in women, is associated with a high risk of developing cardiovascular diseases [66]. Hyperalphalipoproteinemia is a genetic disorder characterized by a deficiency in HDL levels in the blood, accompanied by a mild elevation in triglycerides. Hyperalphalipoproteinemia affects components responsible for HDL biogenesis, ApoA-1, ABCA1, and LCAT. The Functional mutation in ABCA1 (Tangier disease) impairs the ABCA1’s ability to secrete the newly released ApoA-1 from the liver and intestines. This causes rapid catabolism and clearance of ApoA-1, resulting in low HDL levels. In in vivo trials, ABCA1 knockout in the liver induced an 80% reduction in HDL concentration while ABCA1 knockout in the intestines caused a 30% reduction in HDL. When HDL cholesterol and ApoA-1 levels are low, as is the case in Fish Eye Disease, more small HDL particles are produced, resulting in decreased overall HDL levels. The opposite scenario occurs in CETP deficiency, where high levels of HDL cholesterol are associated with large HDL particles. Here, the deficient CETP will be unable to exchange triglycerides for cholesterol between HDL, VLDL, and LDL, resulting in the accumulation of large, malformed HDL particles [81, 82].
Other than the hereditary causes of low HDL, factors like smoking, obesity particularly central obesity, alcohol, poor diet habits, lack of physical activity, metabolic disorders e.g., insulin resistance, and medications e.g. steroids, niacin, statins, etc. can lead to a decreased level of HDL [83, 84]. Low HDL was linked to several illnesses. A recent study of stable angina patients revealed that low HDL-c and high triglyceride levels were correlated with a higher prevalence of coronary atherosclerotic disease events. Accordingly, the ratio of HDL-c to triglycerides could predict the outcome of coronary atherosclerotic disease independently of LDL-c [85]. Nevertheless, a meta-analysis study of 108 randomized trials, including 299,310 participants, demonstrated that raising HDL alone could not modify the progression of cardiovascular diseases or the resulting mortality. However, the study suggested that lowering the LDL as a primary goal could be a good alternative [86]. The LDL-c/HDL-c ratio was found to be a more accurate indicator of the severity of coronary artery disease in STEMI patients than HDL or LDL [87]. Overall, both LDL and HDL are important in reflecting cardiovascular health.
The role of HDL in reducing inflammation by endotoxin neutralization
Inflammation is induced by pathogen-derived molecular patterns (PAMPs), such as endotoxin, or by tissue damage and the release of danger-associated molecular patterns (DAMPs). Sepsis is a severe inflammatory response that usually occurs due to bacterial endotoxin and overproduction of cytokines released from inflammatory cells, particularly Tumor necrosis factor-alpha (TNF-α) [88]. Sepsis incidence has increased over the past decade, where a study conducted from 1979 to 2016 estimated 31.5 million sepsis cases with 5.3 million annual deaths [89].
Toll-like receptor 4 (TLR4), a transmembrane glycoprotein highly expressed on macrophages and other immune cells, recognizes approximately 5% of circulating LPS [90]. TLR4 recognizes endotoxin (LPS) via its co-receptor MD-2. The binding of LPS in the calyx of MD-2 leads to TLR4 dimerization and the initiation of a signaling cascade that activates the NF-κB complex (nuclear factor kappa-light-chain-enhancer of activated B cells) and other signaling pathways, ultimately resulting in inflammation and cytokine production [91–93]. NF-κB was found to have an adverse effect on ApoA-1 and HDL-C by suppressing peroxisome proliferator-activated receptor alpha (PPARα), a key regulator of lipid metabolism in the liver [94]. Experimental evidence revealed the downregulation of TLR4 following an increase in HDL-C after transferring ApoA-1 genes in mice. The decrease of TLR4 expression occurred after 2 weeks of transferring the ApoA-1 gene, and the fold change was about 8.4. The decrease of TLR4 continues even after the administration of LPS in those mice, and the survival rate was noticed to be improved [95]. Another study on mice demonstrated that the infusion of reconstituted HDL reduced inflammation, improved survival, decreased bacterial count, and prevented the organ damage that occurs usually as a consequence of sepsis [96].
More studies have also shown that structural and functional modifications occur in HDL particles during sepsis. For instance, serum amyloid A (SAA1) is one of the acute-phase proteins associated with high-density lipoprotein (HDL). During sepsis, SAA-1 is overexpressed, replacing ApoA-1 in HDL. SAA-1 binds directly to LPS and shuttles it with HDL to promote clearance. Clinical evidence has shown that the displacement of HDL contents leads to HDL particle remodeling, which affects the anti-inflammatory and antioxidant functions [97, 98]. SAA-1-enriched HDL enlarges the HDL size and becomes denser; as a consequence, the mobility of HDL becomes slower [99]. Moreover, the concentration of total HDL in serum is decreased due to the pathophysiological changes associated with sepsis. Several mechanisms contribute to the decrease in HDL, including human secretory phospholipase A2 (sPLA2). This sPLA2 is an acute-phase protein that is upregulated in the presence of LPS, which stimulates SAA1, leading to modifications in HDL particle size and composition [100]. The interaction between LPS and HDL is mediated by several proteins, including CETP, PLTP, and LPS-binding protein (LBP). It is worth noting that HDL and associated proteins have a high affinity for binding and eliminating LPS compared to other lipoproteins [101]. HDL evolved to become an important component of the innate immune system, protecting against endotoxins and immune system overstimulation [102]. However, the exact mechanism by which HDL neutralizes endotoxin and confers anti-inflammatory activity remains unclear. These anti-inflammatory and endotoxin-neutralizing functions of HDL are mediated by its associated apolipoproteins, which are explored in detail in the following section. Vitamin D enhances HDL’s endotoxin-neutralizing capacity by inducing cathelicidin (LL-37), a peptide associated with HDL that inhibits LPS-TLR4 interactions and reduces TNF-α release [103, 104]. The LL-37 gene contains a VDRE, making it highly responsive to 1,25(OH)₂D₃ [105].
Apolipoproteins
Apolipoproteins are a group of proteins considered key elements in lipoproteins composition. They are responsible mainly for lipid binding and transportation and are classified into apolipoprotein A (ApoA-1, ApoA-2, ApoA-4, and apolipoprotein A-V (ApoA5), apolipoprotein B (Apo B48 and Apo B100), apolipoprotein C (Apo C-I, Apo C-II, Apo C-III, and Apo C-IV), apolipoprotein D, E, F, H, L, and M [106]. The four classes of apolipoproteins, ApoA-I, ApoAII, ApoCs, and ApoE, are found in all lipoproteins [107].
HDL-associated apolipoproteins
Apolipoproteins facilitate lipid transport in HDL, with ApoA-I, ApoM, and ApoD being central to its anti-inflammatory roles. The table below summarizes their functions (Table 1).
Table 1.
Apolipoproteins associated with HDL and their functions, and the reference paper
| Apolipoprotein | Function | Reference |
|---|---|---|
| ApoA-1 | Reverse cholesterol transportation | [108] |
| ApoA-II | cholesterol binding | [109] |
| ApoC-I | Inhibitors of lipoprotein binding to the LDL receptor | [110] |
| ApoC-II | Triglyceride hydrolysis of VLDL and CM for energy regulation | [111] |
| ApoC3-III | Antiatherogenic properties and Triglyceride homeostasis. | [112, 113] |
| ApoD | Molecule transportation, e.g., Retinoic acid, sphingomyelins. | [114] |
| ApoE | Lipid transportation | [115] |
| ApoM | A ligand of S1P, anti-inflammatory, antioxidant effects | [116, 117] |
| ApoL 1 | Lipid transportation | [118] |
| ApoJ | Extracellular chaperone | [119, 120] |
Apolipoproteins A-1 (ApoA-1)
Apolipoprotein A-1 is the major component of HDL, located on chromosome 11 and composed of 3 helical chains with a 45 kDa molecular weight, produced in the liver and intestine [121]. ApoA-1 production and regulation depend on the following factors: peroxisome proliferator-activated receptor-γ (PPARγ), the hepatocyte nuclear factor 4 (HNF4), Liver Receptor Homologue 1 (LRH1), and the ApoA-I Regulatory Protein 1 (ARP1/NR2F2) [122]. The construction of HDL starts with the assembly of two to four molecules of ApoA-I with ABCA1. ApoA-1 helps stabilize the ABCA1 protein in hepatocytes and enterocytes, allowing for the synthesis of lipid-poor apoA-1 HDL particles by mediating the efflux of cellular phospholipids and free cholesterol [123]. Most of the ApoA-1 in serum was found associated with HDL, and about 8% of the total ApoA-1 was found lipid-free [108]. In addition, ApoA-1 mediated the cholesterol that effluxes from macrophages in ABCA1-dependent and -independent mechanisms [124].
ApoA-1 modulates innate immunity and adaptive immunity. For instance, ApoA-1 represses both NF-κB and phosphatidylinositol 3-kinase (PI3K). Add to that stimulating the Activator of Transcription Factor 3 (ATF3) (Toll-like receptor down-regulator), which prevents of activation the pro-inflammatory chemokines release e.g. CCL2, CCL5, and CX [3]CL1 [125, 126]. In vivo, Iqbal et al. demonstrated that acute ApoA-I administration reduces monocyte chemotaxis by suppressing the expression of CCL2 and CCL5 [127]. The aforementioned action of ApoA-1 protects against atherosclerosis, and for this reason, some studies consider ApoA-1 a biomarker for predicting cardiovascular diseases [128]. However, a recent meta-analysis of 15 randomized controlled trials has shown that HDL/apoA-1 mimetics are ineffective in reducing atheroma size in the arteries of patients with acute coronary syndrome. Vitamin D modulates ApoA-1 expression, with in vitro studies showing that 1,25(OH)₂D₃ suppresses ApoA-1 gene transcription in HepG2 cells via VDR-VDRE interactions, resulting in a reduction of ApoA-1 mRNA by up to 50% [78]. However, human supplementation studies (e.g., 50,000 IU/week for 12 weeks) suggest vitamin D increases ApoA-1 and HDL cholesterol levels, indicating context-dependent effects [80, 129].
Apolipoprotein M (ApoM)
ApoM is a 23–25 kDa protein, with its human gene located in the major histocompatibility complex (MHC) class III locus on chromosome 6 [130]. ApoM belongs to the lipocalin family (a group of proteins that transport small hydrophobic molecules) and is produced mainly by the liver and kidneys (Fig. 2). Several important proteins belong to this group, such as retinol-binding protein (RBP), apolipoprotein D, and MD-2, the co-receptor for toll-like receptor 4 (TLR4) [131].
Fig. 3.
illustrates the effect of vitamin D on the different HDL-associated proteins, eventually suppressing inflammation. Note: Figures 1, and 3 Created with BioRender.com
Fig. 2.
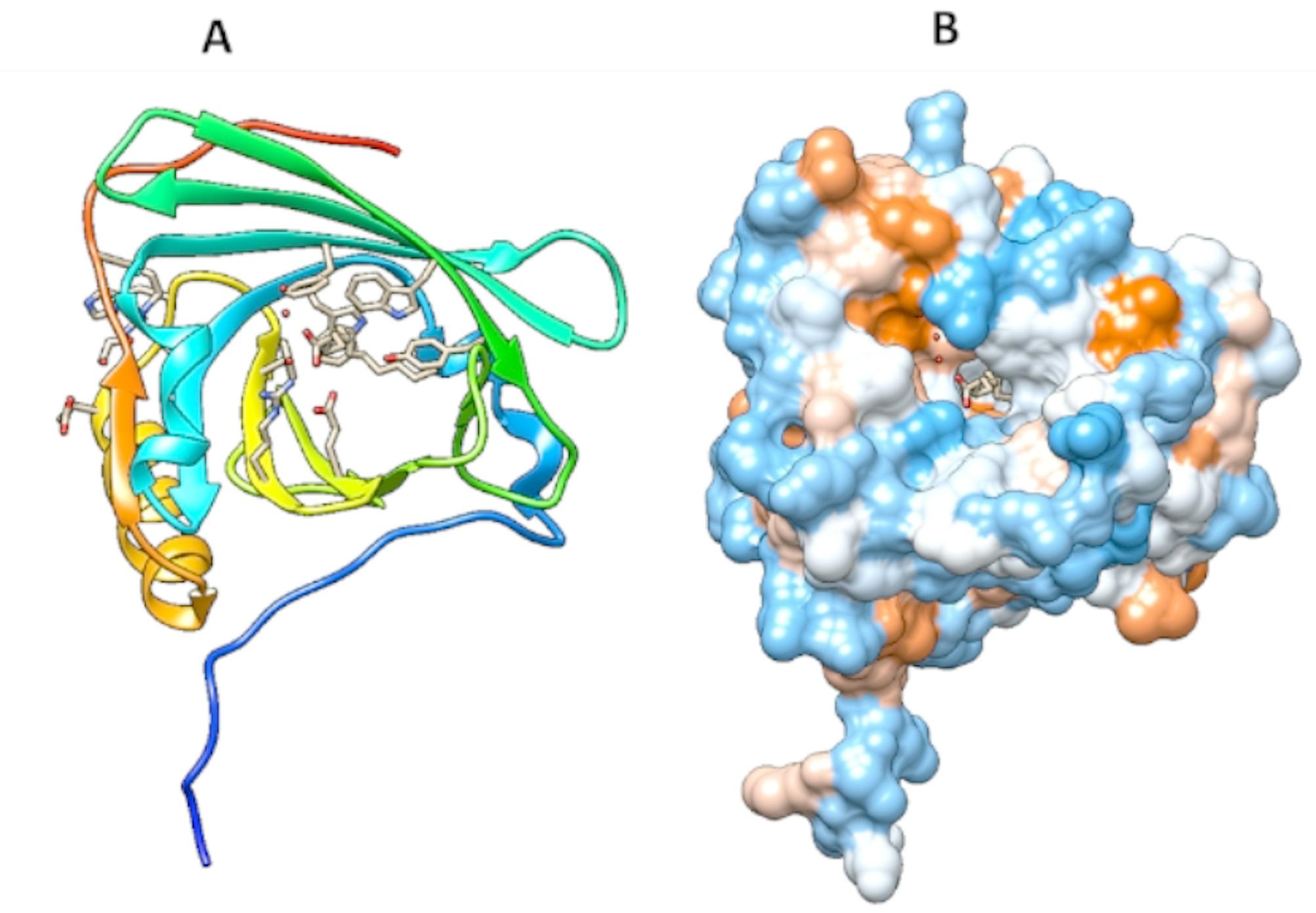
3D structure of ApoM PDB file 2WEW as a lipid carrier generated using Chimera. The Lipocalin fold, consisting of eight β-strands forming a β-barrel and an α-helix with the calyix carrying myristic acid ligand, is shown. A: 3D structure with Ribon presentation. B: 3D structure with surface presentation. The signaling peptide tail is not cleaved, as shown at the bottom of the 3D figure. (Figure 2 generated using Chimera)
ApoM is required for HDL biogenesis, thereby accounting for approximately 95% of plasma HDL. ApoM is found in association with HDL, where it functions as the primary carrier of sphingosine-1-phosphate (S1P). This bioactive lipid mediator plays a key role in modulating vascular inflammation, and research indicates that ApoM-bound S1P is essential for HDL’s documented antiatherogenic and anti-inflammatory properties [116]. Several transcription factors regulate ApoM expression, such as liver receptor homolog (LRH)-1, hepatic nuclear factor (HNF)-1α, and Forkhead box A2 (Foxa2). Any mutation in those factors could lead to ApoM dysregulation, thereby causing HDL malfunction [132, 133]. However, it is unknown whether ApoM is a vitamin D-responsive gene, i.e., whether it contains a VDRE binding site. Structurally, ApoM has a typical lipocalin fold that consists of eight β-strands forming a β-barrel (pocket-like or calyx) and an α-helix [134]. The distinctive structure of ApoM bears some resemblance to the MD2 unit within TLR4. MD2 itself is characterized by two β-strands that collectively form a cup-like architecture [135]. The substantial similarity in the structure of ApoM and MD2 suggests an analogous function for both, which may result in an affinity for binding endotoxins.
ApoM is found to be associated mainly with HDL and, to a lesser extent, with LDL [136]. The terminal signal peptide of ApoM doesn’t cleave from prior release to plasma, as is the case in the other proteins. This character helps ApoM anchor HDL particles and prevents dissociation between the two [137].
As demonstrated in our previous publication, ApoM is more than a vehicle for S1P1 and other lipids. It was observed that ApoM bound to LPS with high affinity using Isothermal Titration Calorimetry (ITC) and computational molecular dynamics modeling. A poM exhibited a high affinity for E. coli LPS, forming 1:1 complexes with dissociation constants (Kd) below 1 µM. The binding process is strongly exothermic and driven by enthalpy (ΔrH = − 36.5 kJ/mol), suggesting the formed complex of ApoM and LPS is highly stable. Computational simulations also predicted high-affinity binding between ApoM and E. coli LPS, showing that E. coli LPS docks near the calyx of ApoM without obstructing the pocket. In vitro studies confirmed the neutralizing capability of HDL for E. coli LPS, as evidenced by the suppression of TNF-α release from human THP-1 cells. This direct interaction facilitates endotoxin neutralization and clearance by HDL, which involves shuttling LPS to the liver for removal [138]. This process of endotoxin neutralization reduces the exacerbation of the immune response towards bacterial endotoxins, aiming to suppress inflammation and restore hemostasis in the body.
Functionally, ApoM is known as a carrier of sphingosine-1-phosphate (S1P) [139]. Some studies have documented that the complex of ApoM/S1P is important for the antiatherogenic and anti-inflammatory effects of HDL [116]. Ruiz et al. reported that the ApoM/S1P complex plays a crucial role in suppressing the surface abundance of vascular adhesion molecule-1 (VCAM-1) and E-selectin during inflammation. Whereas ApoM alone and HDL alone were not successful in conferring this adhesion suppression [116]. The association of HDL/ApoM/S1P may also contribute to HDL’s vasoprotective and antiatherogenic effects [140, 141].
ApoM/S1P was also observed to be upregulated during the inflammation [142]. However, A study by Winkler et al. observed a decline in S1P levels in septic shock patients. They proposed that this significant decrease in S1P was attributable to the drastic reduction in HDL levels commonly seen during septic shock [143]. S1P also interacts with the endothelial scavenger receptor class B type 1 (SR-BI). This SR-BI is also known as the cholesterol-sensing receptor. The importance of S1P is due to its role in facilitating the uptake of cholesterol from tissues by HDL particles [144]. Briefly, the associated S1P with HDL interacts with S1P1 to generate some proximity with SR-BI. Later, intracellular calcium is released, and cholesterol efflux and downstream signaling events occur [145].
Certain disorders have been associated with the disturbance in ApoM levels and function; for example, plasma ApoM concentration was noticed to be decreased in type 2 diabetes patients [146] and heart failure [147]. Furthermore, the complications of diseases were found to be positively associated with changes in ApoM level and, consequently, HDL function, such as diabetes, e.g., diabetic nephropathy [148]. Similarly, changes in ApoM activity due to genetic polymorphisms, for example, are associated with increased susceptibility to impaired HDL and cholesterol metabolism, and a higher risk of developing coronary artery disease [149].
However, most studies have linked ApoM and S1P to a positive outcome in human health. Christoffersen et. al.‘s research linked the overstimulation of the S1P1 receptor to the decrease in triglyceride uptake and brown adipose formation. In this study, it was revealed that ApoM-deficient mice exhibited an increase in stimulating brown adipose tissue formation, which in turn increased triglyceride uptake, thereby protecting against diet-induced obesity [150]. Hajny et al.‘s study also supports this claim; they examined the triglyceride turnover rate in the human ApoM transgenic mouse model. This model is designed to simulate higher concentrations of ApoM and S1P levels compared to normal. Upon comparing ApoM transgenic with wild-type mice, researchers noticed a reduction in overall triglyceride turnover rate. Additionally, the rate of fatty acid uptake in subcutaneous adipocytes was lower than usual. This could be explained by the decrease in both the plasma level of lipase and the plasma level of fibroblast growth factor 21 (FGF21), as a response to the dramatic increase of ApoM and S1P levels. Both lipase and FGF21 are very crucial for triglyceride clearance from plasma. The disturbance in their level impacts causes an elevation in plasma triglyceride levels after administering a high-fat diet to ApoM transgenic mice [151]. Vitamin D may influence ApoM expression indirectly, as ApoM overexpression in colorectal cancer cells upregulates VDR mRNA, suggesting a bidirectional regulatory relationship [152]. Proteomic analyses also show reduced ApoM levels in vitamin D-deficient individuals with dyslipidemia, correlating with enhanced inflammatory pathways like MAPK and JAK-STAT [79].
Apolipoprotein D (ApoD)
ApoD is another member of the lipocalin family associated with HDL and with the enzyme lecithin-cholesterol acyltransferase (LCAT) that converts cholesterol to cholesteryl ester. ApoD is secreted into the plasma and enriched in the brain, testes, breast, and B cells. ApoD has a molecular weight of 33 kDa and is structurally similar to ApoM [153]. In contrast to other HDL-associated proteins like ApoA-1, which adopt a helix structure, ApoM and ApoD are lipocalins characterized by the typical β-sheet fold with calyx, and act as carrier proteins. ApoD binds hydrophobic ligands such as progesterone, retinoic acid, sphingomyelin, pregnenolone, and arachidonic acid [114]. ApoD plays a significant role in the neurological system, especially in the regeneration and healing of nerves. In vivo studies, ApoD was noticed to elevate tremendously during the healing process of sciatic nerve injury [53]. An increase in ApoD expression has been shown in some neurological diseases such as schizophrenia, bipolar disorder, and Multiple sclerosis [154]. Alzheimer’s disease has been linked to the elevation of ApoD level in CSF and cortex [155, 156].
Moreover, in vivo studies exhibit suppression of the innate immunity system by ApoD during the acute encephalitis induced by coronavirus OC43 in mice with restricted phospholipase A2 activity. Additionally, it has been demonstrated that overexpressing human ApoD in a mouse model of viral infection decreases T-cell infiltration into the CNS, lowers the production of pro-inflammatory cytokines, including IL-1ß and TNF, and downregulates the activity of phospholipase A2 (PLA2) [157]. Vitamin D upregulates ApoD expression, with 1,25(OH)₂D₃ increasing ApoD mRNA up to 5-fold in breast and prostate cancer cells, potentially enhancing HDL’s anti-inflammatory capacity [158, 159]. Proteomic data confirm reduced ApoD in vitamin D-deficient, dyslipidemic individuals, linked to heightened inflammation [79].
Vitamin D impact on HDL-associated proteins ApoA-1, apom, apod, and LL-37
In vitro studies on HepG2 cells (hepatocellular carcinoma) demonstrated that 1,25(OH)₂D₃ suppresses ApoA-1 gene expression at the transcriptional level through VDR-mediated binding to vitamin D response elements (VDREs) in the ApoA-1 promoter [78]. This suppression reduces ApoA-1 mRNA and protein secretion in a dose-dependent manner, potentially impacting HDL biogenesis [78, 160]. For example, Wehmeier et al. [78]showed that 1,25(OH)₂D₃ at concentrations of 10–100 nM inhibited ApoA-1 promoter activity by up to 50%, suggesting a direct regulatory role of vitamin D in HDL composition.
Upon comparing the wild-type mice with VDR knockout ones, experiments illustrated increased HDL-c in general and ApoA-1 expression in the liver. In addition to an increase in total cholesterol levels. However, some differences in the serum concentration of HDL-c between female and male VDR knockout mice have been reported, with a greater increase in males. Consequently, ApoA-1 mRNA was also increased by 49.2% in males [160]. This indicates a more substantial effect of vitamin D in suppressing HDL and ApoA-1 in both genders, particularly in males. Due to species-specific differences, this result could be debated, as rodents and humans do not share similar regulatory mechanisms for HDL-c, ApoA-1, and VDR [161]. From another perspective, Vitamin D supplementation in human studies has been shown to positively affect HDL-c levels [80]. Radkhah et al.‘s systematic review suggested that vitamin D supplementation for ≤ 12 weeks increases Apo-A1 levels; however, the data were not conclusive, and the link was not well elucidated [129].
An in vitro study using colorectal cancer cell lines demonstrated that ApoM overexpression increases VDR mRNA expression, indicating a regulatory relationship [152]. However, the effect of vitamin D on ApoM expression remains unknown. The study by Yu et al. on ApoM in Hepatocellular carcinoma cell lines, using the CRISPR/Cas9 technique, revealed an interesting result. Knocking out ApoM genes in SMMC7721 cell lines was observed to inhibit tumor cell death and induce tumorigenic characteristics, such as increased proliferation rates. A considerable reduction in VDR expression followed this. Further investigations demonstrated that VDR overexpression could reverse the previous effect and suppress the tumorigenicity. Therefore, ApoM can suppress the tumorigenicity characteristics in hepatocellular carcinoma cell lines via enhancing the expression of VDR [162]. In some diseases, such as Systemic lupus erythematosus (SLE), ApoM is inversely correlated with disease activity [163]. Similarly, ApoM was inversely related to the risk of type 2 diabetes [164]. ApoD expression is affected in 1,25(OH)2 D3 presence. ApoD increased up to 5-fold in the presence of D3, more than the increase from other steroid hormones (ApoD ligands). This increase is associated with an inhibitory effect on certain cancers, including breast and prostate cancer cells [158, 165]. These data indicate a direct relationship between ApoA-1, ApoM, ApoD, and vitamin D, although the nature of this association is unclear.
Mousa et al. proteomic analysis of 274 participants revealed reduced HDL-associated ApoM and ApoD in individuals with vitamin D deficiency and dyslipidemia. Conversely, those participants had increased levels of proteins (SAA1 and SOD1), which are associated with the activation of inflammatory pathways. Furthermore, pathway enrichment analysis revealed increased inflammatory activity in individuals with vitamin D deficiency and dyslipidemia, with significant enrichment in pathways such as MAPK, JAK-STAT, Ras signaling, cytokine-cytokine receptor interaction, AGE-RAGE, ErbB signaling, and cancer pathways. Meanwhile, participants with vitamin D deficiency and normal lipid profiles had enrichment in inflammation pathways, such as cytokine signaling pathways [79].
Vitamin D is a potent inducer of cathelicidins (LL-37/ hCAP18), small cationic peptides with antimicrobial properties towards gram-negative and gram-positive bacteria [103]. Cathelicidin (LL-37/hCAP18) plays a crucial role in innate immunity by inhibiting the interaction between LPS and LPS-binding protein, thereby preventing TLR4 activation and resulting in decreased TNF-α release [104, 166, 167]. Furthermore, LL-37 suppresses inflammation and cell death by inhibiting IL-1β expression and caspase-1 activation [168]. Proteomics studies have documented that LL-37 is associated with lipoproteins, including HDL, VLDL, and LDL [169]. Further, the LL-37 gene has a vitamin D response element (VDRE) in its promoter. Thus, it is highly induced by vitamin D [105].
Recent studies have elucidated the association between vitamin D deficiency and dyslipidemia. Sharba et al. found that low vitamin D levels have impacted the lipid profile, including cholesterol, triglycerides, HDL, and LDL [170]. Consistent with our prior findings, the combination of dyslipidemia and vitamin D deficiency modifies the proteomic profiles of healthy individuals. In addition to enhancing inflammatory pathways, a decrease in HDL-associated apolipoproteins, particularly ApoM and ApoD, has been noticed [79]. Vitamin D supplementation has been found to impact dyslipidemia and help alleviate inflammation [171] (Fig. 3).
Strengths and limitations
This review offers a focused and comprehensive analysis of how Vitamin D modulates immune function through its effects on HDL and associated proteins. It provides mechanistic insights into these complex interactions, covering Vitamin D’s influence on lipid metabolism, HDL biogenesis mediated by ApoM, and anti-inflammatory pathways. The detailed discussion of key apolipoproteins (ApoA-1, ApoD, and ApoM) and their roles in lipid transport and anti-inflammatory processes establishes a robust scientific framework. Furthermore, the review effectively highlights the clinical relevance of these molecular interactions in cardiovascular health and chronic inflammatory conditions.
As a comprehensive narrative review, this work synthesizes existing literature without presenting new experimental findings. While it integrates diverse observations, the review could further reconcile seemingly contradictory data on Vitamin D’s impact on apolipoprotein expression and function. Additionally, it highlights a broader limitation in the current research, where studies comprehensively elucidating Vitamin D’s relationship with the full spectrum of apolipoproteins remain limited.
Conclusion and future directions
Vitamin D modulates HDL functionality through its effects on apolipoproteins (ApoA-1, ApoM, ApoD) and cathelicidin (LL-37), influencing lipid metabolism and inflammation [78–80, 103, 158]. While in vitro studies show that 1,25(OH)₂D₃ suppresses ApoA-1 transcription [78], human supplementation increases HDL cholesterol and ApoA-1 [80, 129], suggesting context-dependent effects. ApoM and ApoD levels are reduced in vitamin D-deficient, dyslipidemic individuals, correlating with the activation of enhanced inflammatory pathways [79]. LL-37, induced by vitamin D via VDRE, enhances HDL’s endotoxin-neutralizing capacity [105].
Longitudinal studies and preclinical models (e.g., VDR-knockout mice) are needed to establish causality between vitamin D status, HDL protein composition, and cardiovascular outcomes. Clinical trials evaluating the impact of vitamin D supplementation on ApoM, ApoD, and LL-37 levels could inform therapeutic strategies for dyslipidemia and inflammation-related diseases.
Author contributions
H.M. wrote the main manuscript text of the review as part of her thesis work, which has not been published previously. S.Z. edited and revised the manuscript for clarity and scientific accuracy. Both H.M. and S.Z. served as corresponding authors, managing communication and submission processes. All authors reviewed and approved the final manuscript.
Funding
This review was not supported by any grant funding; however, the Article Processing Charges (APC) were covered by Qatar University.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Competing interests
The authors declare no competing interests.
Ethical approval
Not applicable.
Conflict of interest
The authors declare no conflicts of interest.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Holick MF. Vitamin D deficiency. N Engl J Med. 2007;357(3):266–81. [DOI] [PubMed] [Google Scholar]
- 2.Szpirer J, Szpirer C, Riviere M, Levan G, Marynen P, Cassiman J-J, et al. The Sp1 transcription factor gene (SP1) and the 1,25-dihydroxyvitamin D3 receptor gene (VDR) are colocalized on human chromosome arm 12q and rat chromosome 7. Genomics. 1991;11(1):168–73. [DOI] [PubMed] [Google Scholar]
- 3.Bikle D. Nonclassic actions of vitamin D. J Clin Endocrinol Metab. 2009;94(1):26–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Nagpal S, Na S, Rathnachalam R. Noncalcemic actions of vitamin D receptor ligands. Endocr Rev. 2005;26(5):662–87. [DOI] [PubMed] [Google Scholar]
- 5.Pike JW, Meyer MB, Bishop KA. Regulation of target gene expression by the vitamin D receptor: an update on mechanisms. Reviews Endocr Metabolic Disorders. 2012;13(1):45–55. [DOI] [PubMed] [Google Scholar]
- 6.Holick MF, Binkley NC, Bischoff-Ferrari HA, Gordon CM, Hanley DA, Heaney RP, et al. Evaluation, treatment, and prevention of vitamin D deficiency: an endocrine society clinical practice guideline. J Clin Endocrinol Metab. 2011;96(7):1911–30. [DOI] [PubMed] [Google Scholar]
- 7.Aranow C. Vitamin D and the immune system. J Investig Med. 2011;59(6):881–6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Cantorna MT, Woodward WD, Hayes CE, DeLuca HF. 1,25-dihydroxyvitamin D3 is a positive regulator for the two anti-encephalitogenic cytokines TGF-beta 1 and IL-4. J Immunol. 1998;160(11):5314–9. [PubMed] [Google Scholar]
- 9.Ragab D, Soliman D, Samaha D, Yassin A. Vitamin D status and its modulatory effect on interferon gamma and interleukin-10 production by peripheral blood mononuclear cells in culture. Cytokine. 2016;85:5–10. [DOI] [PubMed] [Google Scholar]
- 10.Verway M, Behr MA, White JH, Vitamin D. NOD2, autophagy and crohn’s disease. Expert Rev Clin Immunol. 2010;6(4):505–8. [DOI] [PubMed] [Google Scholar]
- 11.Wang TT, Nestel FP, Bourdeau V, Nagai Y, Wang Q, Liao J, et al. Cutting edge: 1,25-dihydroxyvitamin D3 is a direct inducer of antimicrobial peptide gene expression. J Immunol. 2004;173(5):2909–12. [DOI] [PubMed] [Google Scholar]
- 12.Chung C, Silwal P, Kim I, Modlin RL, Jo EK. Vitamin D-Cathelicidin axis: at the crossroads between protective immunity and pathological inflammation during infection. Immune Netw. 2020;20(2):e12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Yuk JM, Shin DM, Lee HM, Yang CS, Jin HS, Kim KK, et al. Vitamin D3 induces autophagy in human monocytes/macrophages via Cathelicidin. Cell Host Microbe. 2009;6(3):231–43. [DOI] [PubMed] [Google Scholar]
- 14.Park JM, Jung CH, Seo M, Otto NM, Grunwald D, Kim KH, et al. The ULK1 complex mediates MTORC1 signaling to the autophagy initiation machinery via binding and phosphorylating ATG14. Autophagy. 2016;12(3):547–64. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Ikeda U, Wakita D, Ohkuri T, Chamoto K, Kitamura H, Iwakura Y, et al. 1α,25-Dihydroxyvitamin D3 and all-trans retinoic acid synergistically inhibit the differentiation and expansion of Th17 cells. Immunol Lett. 2010;134(1):7–16. [DOI] [PubMed] [Google Scholar]
- 16.Joshi S, Pantalena LC, Liu XK, Gaffen SL, Liu H, Rohowsky-Kochan C, et al. 1,25-dihydroxyvitamin D(3) ameliorates Th17 autoimmunity via transcriptional modulation of interleukin-17A. Mol Cell Biol. 2011;31(17):3653–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Urry Z, Chambers ES, Xystrakis E, Dimeloe S, Richards DF, Gabryšová L, et al. The role of 1α,25-dihydroxyvitamin D3 and cytokines in the promotion of distinct Foxp3 + and IL-10 + CD4 + T cells. Eur J Immunol. 2012;42(10):2697–708. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Institute of Medicine (US) Committee to Review Dietary Reference Intakes for Vitamin D and Calcium; Ross AC, Taylor CL, Yaktine AL, et al., editors. Dietary Reference Intakes for Calcium and Vitamin D. Washington (DC): National Academies Press (US) et al. 2011. Available from: https://www.ncbi.nlm.nih.gov/books/NBK56070/doi:10.17226/13050 [PubMed]
- 19.Mousa H, Islam N, Ganji V, Zughaier SM. Serum 25-Hydroxyvitamin D is inversely associated with monocyte percentage to HDL cholesterol ratio among young healthy adults in Qatar. Nutrients [Internet]. 2021; 13(1). [DOI] [PMC free article] [PubMed]
- 20.Surdu AM, Pînzariu O, Ciobanu D-M, Negru A-G, Căinap S-S, Lazea C et al. Vitamin D and its role in the lipid metabolism and the development of atherosclerosis. Biomedicines [Internet] 2021; 9(2). [DOI] [PMC free article] [PubMed]
- 21.Talmor Y, Bernheim J, Klein O, Green J, Rashid G. Calcitriol blunts pro-atherosclerotic parameters through NFkappaB and p38 in vitro. Eur J Clin Invest. 2008;38(8):548–54. [DOI] [PubMed] [Google Scholar]
- 22.Wimalawansa SJ. Vitamin D and cardiovascular diseases: causality. J Steroid Biochem Mol Biol. 2018;175:29–43. [DOI] [PubMed] [Google Scholar]
- 23.Parva NR, Tadepalli S, Singh P, Qian A, Joshi R, Kandala H, et al. Prevalence of vitamin D deficiency and associated risk factors in the US population (2011–2012). Cureus. 2018;10(6):e2741. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Giovannucci E, Liu Y, Hollis BW, Rimm EB. 25-hydroxyvitamin D and risk of myocardial infarction in men: a prospective study. Arch Intern Med. 2008;168(11):1174–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Dobnig H, Pilz S, Scharnagl H, Renner W, Seelhorst U, Wellnitz B, et al. Independent association of low serum 25-hydroxyvitamin d and 1,25-dihydroxyvitamin d levels with all-cause and cardiovascular mortality. Arch Intern Med. 2008;168(12):1340–9. [DOI] [PubMed] [Google Scholar]
- 26.Pilz S, März W, Wellnitz B, Seelhorst U, Fahrleitner-Pammer A, Dimai HP, et al. Association of vitamin D deficiency with heart failure and sudden cardiac death in a large cross-sectional study of patients referred for coronary angiography. J Clin Endocrinol Metab. 2008;93(10):3927–35. [DOI] [PubMed] [Google Scholar]
- 27.Borges AC, Feres T, Vianna LM, Paiva TB. Effect of cholecalciferol treatment on the relaxant responses of spontaneously hypertensive rat arteries to acetylcholine. Hypertension. 1999;34(4 Pt 2):897–901. [DOI] [PubMed] [Google Scholar]
- 28.Lei GS, Zhang C, Cheng BH, Lee CH. Mechanisms of action of vitamin D as supplemental therapy for Pneumocystis pneumonia. Antimicrob Agents Chemother. 2017;61(10). [DOI] [PMC free article] [PubMed]
- 29.Bozic M, Álvarez Á, de Pablo C, Sanchez-Niño MD, Ortiz A, Dolcet X, et al. Impaired vitamin D signaling in endothelial cell leads to an enhanced Leukocyte-Endothelium interplay: implications for atherosclerosis development. PLoS ONE. 2015;10(8):e0136863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Dixon DL, Riche DM. Dyslipidemia. In: DiPiro JT, Yee GC, Posey LM, Haines ST, Nolin TD, Ellingrod V, editors. Pharmacotherapy: A pathophysiologic approach, 11e. New York, NY: McGraw-Hill Education; 2020. [Google Scholar]
- 31.Hedayatnia M, Asadi Z, Zare-Feyzabadi R, Yaghooti-Khorasani M, Ghazizadeh H, Ghaffarian-Zirak R, et al. Dyslipidemia and cardiovascular disease risk among the MASHAD study population. Lipids Health Dis. 2020;19(1):42. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Pearson GJ, Thanassoulis G, Anderson TJ, Barry AR, Couture P, Dayan N, et al. 2021 Canadian cardiovascular society guidelines for the management of dyslipidemia for the prevention of cardiovascular disease in adults. Can J Cardiol. 2021;37(8):1129–50. [DOI] [PubMed] [Google Scholar]
- 33.Mach F, Baigent C, Catapano AL, Koskinas KC, Casula M, Badimon L, et al. 2019 ESC/EAS guidelines for the management of dyslipidaemias: lipid modification to reduce cardiovascular risk. Eur Heart J. 2020;41(1):111–88. [DOI] [PubMed] [Google Scholar]
- 34.Qi Q, Qi L. Lipoprotein(a) and cardiovascular disease in diabetic patients. Clin Lipidol. 2012;7(4):397–407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kaur J. A comprehensive review on metabolic syndrome. Cardiol Res Pract. 2014;2014:943162. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 36.Schmitt EB, Nahas-Neto J, Bueloni-Dias F, Poloni PF, Orsatti CL, Petri Nahas EA. Vitamin D deficiency is associated with metabolic syndrome in postmenopausal women. Maturitas. 2018;107:97–102. [DOI] [PubMed] [Google Scholar]
- 37.Pan GT, Guo JF, Mei SL, Zhang MX, Hu ZY, Zhong CK, et al. Vitamin D deficiency in relation to the risk of metabolic syndrome in Middle-Aged and elderly patients with type 2 diabetes mellitus. J Nutr Sci Vitaminol (Tokyo). 2016;62(4):213–9. [DOI] [PubMed] [Google Scholar]
- 38.Lund-Katz S, Liu L, Thuahnai ST, Phillips MC. High density lipoprotein structure. Front Biosci. 2003;8:d1044–54. [DOI] [PubMed] [Google Scholar]
- 39.Pirahanchi Y, Sinawe H, Dimri M, Biochemistry. LDL cholesterol. StatPearls. Treasure Island (FL): StatPearls publishing copyright © 2022. StatPearls Publishing LLC.; 2022. [PubMed]
- 40.Cox RA, García-Palmieri MR. Cholesterol, triglycerides, and associated lipoproteins. In: Walker HK, Hall WD, Hurst JW, editors. Clinical methods: the history, physical, and laboratory examinations. Boston: Butterworths copyright © 1990. Butterworth Publishers, a division of Reed Publishing.; 1990.
- 41.Hevonoja T, Pentikäinen MO, Hyvönen MT, Kovanen PT, Ala-Korpela M. Structure of low density lipoprotein (LDL) particles: basis for Understanding molecular changes in modified LDL. Biochim Biophys Acta. 2000;1488(3):189–210. [DOI] [PubMed] [Google Scholar]
- 42.Staprans I, Rapp JH, Pan XM, Feingold KR. Oxidized lipids in the diet are incorporated by the liver into very low density lipoprotein in rats. J Lipid Res. 1996;37(2):420–30. [PubMed] [Google Scholar]
- 43.Ahotupa M. Oxidized lipoprotein lipids and atherosclerosis. Free Radic Res. 2017;51(4):439–47. [DOI] [PubMed] [Google Scholar]
- 44.Hao W, Friedman A. The LDL-HDL profile determines the risk of atherosclerosis: a mathematical model. PLoS ONE. 2014;9(3):e90497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Randolph GJ. Mechanisms that regulate macrophage burden in atherosclerosis. Circ Res. 2014;114(11):1757–71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Fenercioglu AK. The Anti-Inflammatory roles of vitamin D for improving human health. Curr Issues Mol Biol. 2024;46(12):13514–25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zhu C, Chen W, Cui H, Huang Z, Ding R, Li N, et al. TRIM64 promotes ox-LDL-induced foam cell formation, pyroptosis, and inflammation in THP-1-derived macrophages by activating a feedback loop with NF-κB via IκBα ubiquitination. Cell Biol Toxicol. 2023;39(3):607–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Ezzat Y, Sayed S, Gaber W, Mohey AM, Kassem TW. 25-Hydroxy vitamin D levels and its relation to disease activity and cardiovascular risk factors in women with systemic lupus erythematosus. Egypt Rheumatologist. 2011;33(4):195–201. [Google Scholar]
- 49.Feingold KR. Introduction to lipids and lipoproteins. In: Feingold KR, Anawalt B, Boyce A, Chrousos G, de Herder WW, Dhatariya K, et al. editors. Endotext. South Dartmouth (MA): mdtext.com, inc. Copyright © 2000–2022. MDText.com, Inc.; 2000.
- 50.Mittendorfer B, Yoshino M, Patterson BW, Klein S. VLDL triglyceride kinetics in lean, overweight, and obese men and women. J Clin Endocrinol Metab. 2016;101(11):4151–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Lupton JR, Faridi KF, Martin SS, Sharma S, Kulkarni K, Jones SR, et al. Deficient serum 25-hydroxyvitamin D is associated with an atherogenic lipid profile: the very large database of lipids (VLDL-3) study. J Clin Lipidol. 2016;10(1):72–e811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Cai A, Li X, Zhong Q, Li M, Wang R, Liang Y, et al. Associations of high HDL cholesterol level with all-cause mortality in patients with heart failure complicating coronary heart disease. Med (Baltim). 2016;95(28):e3974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Boyles JK, Notterpek LM, Anderson LJ. Accumulation of Apolipoproteins in the regenerating and remyelinating mammalian peripheral nerve. Identification of Apolipoprotein D, Apolipoprotein A-IV, Apolipoprotein E, and Apolipoprotein A-I. J Biol Chem. 1990;265(29):17805–15. [PubMed] [Google Scholar]
- 54.Dai L, Datta G, Zhang Z, Gupta H, Patel R, Honavar J, et al. The Apolipoprotein A-I mimetic peptide 4F prevents defects in vascular function in endotoxemic rats. J Lipid Res. 2010;51(9):2695–705. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Hutchins PM, Ronsein GE, Monette JS, Pamir N, Wimberger J, He Y, et al. Quantification of HDL particle concentration by calibrated ion mobility analysis. Clin Chem. 2014;60(11):1393–401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Chien JY, Jerng JS, Yu CJ, Yang PC. Low serum level of high-density lipoprotein cholesterol is a poor prognostic factor for severe sepsis. Crit Care Med. 2005;33(8):1688–93. [DOI] [PubMed] [Google Scholar]
- 57.Schonfeld G, Pfleger B. The structure of human high density lipoprotein and the levels of Apolipoprotein A-I in plasma as determined by radioimmunoassay. J Clin Invest. 1974;54(2):236–46. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Karan S, Mohapatra A, Sahoo PK, Garg LC, Dixit A. Structural-functional characterization of Recombinant Apolipoprotein A-I from Labeo rohita demonstrates heat-resistant antimicrobial activity. Appl Microbiol Biotechnol. 2019. [DOI] [PubMed]
- 59.Zannis VI, Cole FS, Jackson CL, Kurnit DM, Karathanasis SK. Distribution of Apolipoprotein A-I, C-II, C-III, and E mRNA in fetal human tissues. Time-dependent induction of Apolipoprotein E mRNA by cultures of human monocyte-macrophages. Biochemistry. 1985;24(16):4450–5. [DOI] [PubMed] [Google Scholar]
- 60.Neufeld EB, Demosky SJ Jr., Stonik JA, Combs C, Remaley AT, Duverger N, et al. The ABCA1 transporter functions on the basolateral surface of hepatocytes. Biochem Biophys Res Commun. 2002;297(4):974–9. [DOI] [PubMed] [Google Scholar]
- 61.Oram JF, Heinecke JW. ATP-binding cassette transporter A1: a cell cholesterol exporter that protects against cardiovascular disease. Physiol Rev. 2005;85(4):1343–72. [DOI] [PubMed] [Google Scholar]
- 62.Curtiss LK, Valenta DT, Hime NJ, Rye KA. What is so special about Apolipoprotein AI in reverse cholesterol transport? Arterioscler Thromb Vasc Biol. 2006;26(1):12–9. [DOI] [PubMed] [Google Scholar]
- 63.Soltani S, Boozari M, Cicero AFG, Jamialahmadi T, Sahebkar A. Effects of phytochemicals on macrophage cholesterol efflux capacity: impact on atherosclerosis. Phytother Res. 2021;35(6):2854–78. [DOI] [PubMed] [Google Scholar]
- 64.Zannis VI, Fotakis P, Koukos G, Kardassis D, Ehnholm C, Jauhiainen M, et al. HDL biogenesis, remodeling, and catabolism. Handb Exp Pharmacol. 2015;224:53–111. [DOI] [PubMed] [Google Scholar]
- 65.Elkhalil L, Majd Z, Bakir R, Perez-Mendez O, Castro G, Poulain P, et al. Fish-eye disease: structural and in vivo metabolic abnormalities of high-density lipoproteins. Metabolism. 1997;46(5):474–83. [DOI] [PubMed] [Google Scholar]
- 66.Bailey A, Mohiuddin SS, Biochemistry. High density lipoprotein. StatPearls. Treasure Island (FL): StatPearls publishing copyright © 2022. StatPearls Publishing LLC.; 2022. [PubMed]
- 67.Jahangiri A, de Beer MC, Noffsinger V, Tannock LR, Ramaiah C, Webb NR, et al. HDL remodeling during the acute phase response. Arterioscler Thromb Vasc Biol. 2009;29(2):261–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Zannis VI, Fotakis P, Koukos G, Kardassis D, Ehnholm C, Jauhiainen M, et al. HDL biogenesis, remodeling, and catabolism. High density lipoproteins: from biological Understanding to clinical exploitation. Cham: Springer International Publishing; 2015. pp. 53–111. [DOI] [PubMed] [Google Scholar]
- 69.Ye D, Lammers B, Zhao Y, Meurs I, Van Berkel TJ, Van Eck M. ATP-binding cassette transporters A1 and G1, HDL metabolism, cholesterol efflux, and inflammation: important targets for the treatment of atherosclerosis. Curr Drug Targets. 2011;12(5):647–60. [DOI] [PubMed] [Google Scholar]
- 70.Kontush A, Chapman MJ. Functionally defective high-density lipoprotein: a new therapeutic target at the crossroads of dyslipidemia, inflammation, and atherosclerosis. Pharmacol Rev. 2006;58(3):342–74. [DOI] [PubMed] [Google Scholar]
- 71.Smith Jonathan D. Dysfunctional HDL as a diagnostic and therapeutic target. Arterioscler Thromb Vasc Biol. 2010;30(2):151–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Xu N, Dahlback B. A novel human Apolipoprotein (apoM). J Biol Chem. 1999;274(44):31286–90. [DOI] [PubMed] [Google Scholar]
- 73.Bittilo Bon G, Cazzolato G, Avogaro P. Preparative isotachophoresis of human plasma high density lipoproteins HDL2 and HDL3. J Lipid Res. 1981;22(6):998–1002. [PubMed] [Google Scholar]
- 74.Cockerill GW, Rye K-A, Gamble JR, Vadas MA, Barter PJ, Arteriosclerosis. Thromb Vascular Biology. 1995;15(11):1987–94. [DOI] [PubMed]
- 75.Blankenberg S, Barbaux S, Tiret L. Adhesion molecules and atherosclerosis. Atherosclerosis. 2003;170(2):191–203. [DOI] [PubMed] [Google Scholar]
- 76.van der Vorst EPC, Theodorou K, Wu Y, Hoeksema MA, Goossens P, Bursill CA, et al. High-Density lipoproteins exert Pro-inflammatory effects on macrophages via passive cholesterol depletion and PKC-NF-κB/STAT1-IRF1 signaling. Cell Metab. 2017;25(1):197–207. [DOI] [PubMed] [Google Scholar]
- 77.Younis NN, Soran H, Charlton-Menys V, Sharma R, Hama S, Pemberton P, et al. High-density lipoprotein impedes glycation of low-density lipoprotein. Diab Vasc Dis Res. 2013;10(2):152–60. [DOI] [PubMed] [Google Scholar]
- 78.Wehmeier K, Beers A, Haas MJ, Wong NC, Steinmeyer A, Zugel U, et al. Inhibition of Apolipoprotein AI gene expression by 1, 25-dihydroxyvitamin D3. Biochim Biophys Acta. 2005;1737(1):16–26. [DOI] [PubMed] [Google Scholar]
- 79.Mousa H, Al Saei A, Razali RM, Zughaier SM. Vitamin D status affects proteomic profile of HDL-associated proteins and inflammatory mediators in dyslipidemia. J Nutr Biochem. 2024;123:109472. [DOI] [PubMed] [Google Scholar]
- 80.Tavakoli F, Namakin K, Zardast M, Vitamin D. Supplementation and High-Density lipoprotein cholesterol: A study in healthy school children. Iran J Pediatr. 2016;26(4):e3311. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Schumacher T, Benndorf RA. ABC transport proteins in cardiovascular Disease-A brief summary. Molecules. 2017;22(4). [DOI] [PMC free article] [PubMed]
- 82.Mabuchi H, Nohara A, Inazu A. Cholesteryl ester transfer protein (CETP) deficiency and CETP inhibitors. Mol Cells. 2014;37(11):777–84. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Singh IM, Shishehbor MH, Ansell BJ. High-density lipoprotein as a therapeutic target: a systematic review. JAMA. 2007;298(7):786–98. [DOI] [PubMed] [Google Scholar]
- 84.de Backer G, de Bacquer D, Kornitzer M. Epidemiological aspects of high density lipoprotein cholesterol. Atherosclerosis. 1998;137(Suppl):S1–6. [DOI] [PubMed] [Google Scholar]
- 85.Caselli C, De Caterina R, Smit JM, Campolo J, El Mahdiui M, Ragusa R, et al. Triglycerides and low HDL cholesterol predict coronary heart disease risk in patients with stable angina. Sci Rep. 2021;11(1):20714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Briel M, Ferreira-Gonzalez I, You JJ, Karanicolas PJ, Akl EA, Wu P, et al. Association between change in high density lipoprotein cholesterol and cardiovascular disease morbidity and mortality: systematic review and meta-regression analysis. BMJ. 2009;338:b92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Gao P, Wen X, Ou Q, Zhang J. Which one of LDL-C /HDL-C ratio and non-HDL-C can better predict the severity of coronary artery disease in STEMI patients. BMC Cardiovasc Disord. 2022;22(1):318. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Beutler B, Cerami A. Tumor necrosis, cachexia, shock, and inflammation: a common mediator. Annu Rev Biochem. 1988;57:505–18. [DOI] [PubMed] [Google Scholar]
- 89.Fleischmann C, Scherag A, Adhikari NK, Hartog CS, Tsaganos T, Schlattmann P, et al. Assessment of global incidence and mortality of Hospital-treated sepsis. Current estimates and limitations. Am J Respir Crit Care Med. 2016;193(3):259–72. [DOI] [PubMed] [Google Scholar]
- 90.Brubaker SW, Bonham KS, Zanoni I, Kagan JC. Innate immune pattern recognition: a cell biological perspective. Annu Rev Immunol. 2015;33:257–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Vaure C, Liu Y. A comparative review of toll-like receptor 4 expression and functionality in different animal species. Front Immunol. 2014;5:316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 92.Ohashi K, Burkart V, Flohe S, Kolb H. Cutting edge: heat shock protein 60 is a putative endogenous ligand of the toll-like receptor-4 complex. J Immunol. 2000;164(2):558–61. [DOI] [PubMed] [Google Scholar]
- 93.Beutler B, Endotoxin. Toll-like receptor 4, and the afferent limb of innate immunity. Curr Opin Microbiol. 2000;3(1):23–8. [DOI] [PubMed] [Google Scholar]
- 94.Morishima A, Ohkubo N, Maeda N, Miki T, Mitsuda N. NFkappaB regulates plasma Apolipoprotein A-I and high density lipoprotein cholesterol through Inhibition of peroxisome proliferator-activated receptor alpha. J Biol Chem. 2003;278(40):38188–93. [DOI] [PubMed] [Google Scholar]
- 95.Van Linthout S, Spillmann F, Graiani G, Miteva K, Peng J, Van Craeyveld E, et al. Down-regulation of endothelial TLR4 signalling after Apo A-I gene transfer contributes to improved survival in an experimental model of lipopolysaccharide-induced inflammation. J Mol Med (Berl). 2011;89(2):151–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Tanaka S, Genève C, Zappella N, Yong-Sang J, Planesse C, Louedec L, et al. Reconstituted High-density lipoprotein therapy improves survival in mouse models of Sepsis. Anesthesiology. 2020;132(4):825–38. [DOI] [PubMed] [Google Scholar]
- 97.Cheng N, Liang Y, Du X, Ye RD. Serum amyloid A promotes LPS clearance and suppresses LPS-induced inflammation and tissue injury. EMBO Rep. 2018;19(10). [DOI] [PMC free article] [PubMed]
- 98.Sato M, Ohkawa R, Yoshimoto A, Yano K, Ichimura N, Nishimori M et al. Effects of serum amyloid A on the structure and antioxidant ability of high-density lipoprotein. Biosci Rep. 2016;36(4). [DOI] [PMC free article] [PubMed]
- 99.Cabana VG, Siegel JN, Sabesin SM. Effects of the acute phase response on the concentration and density distribution of plasma lipids and apolipoproteins. J Lipid Res. 1989;30(1):39–49. [PubMed] [Google Scholar]
- 100.Tietge UJ, Maugeais C, Lund-Katz S, Grass D, deBeer FC, Rader DJ. Human secretory phospholipase A2 mediates decreased plasma levels of HDL cholesterol and apoA-I in response to inflammation in human apoA-I Transgenic mice. Arterioscler Thromb Vasc Biol. 2002;22(7):1213–8. [DOI] [PubMed] [Google Scholar]
- 101.Tanaka S, Couret D, Tran-Dinh A, Duranteau J, Montravers P, Schwendeman A, et al. High-density lipoproteins during sepsis: from bench to bedside. Crit Care. 2020;24(1):134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Nazir S, Jankowski V, Bender G, Zewinger S, Rye K-A, van der Vorst EPC. Interaction between high-density lipoproteins and inflammation: function matters more than concentration! Adv Drug Deliv Rev. 2020;159:94–119. [DOI] [PubMed] [Google Scholar]
- 103.Kosciuczuk EM, Lisowski P, Jarczak J, Strzalkowska N, Jozwik A, Horbanczuk J, et al. Cathelicidins: family of antimicrobial peptides. A review. Mol Biol Rep. 2012;39(12):10957–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Zughaier SM, Svoboda P, Pohl J, Stephens DS, Shafer WM. The human host defense peptide LL-37 interacts with Neisseria meningitidis capsular polysaccharides and inhibits inflammatory mediators release. PLoS ONE. 2010;5(10):e13627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Zasloff M. Fighting infections with vitamin D. Nat Med. 2006;12(4):388–90. [DOI] [PubMed] [Google Scholar]
- 106.Alaupovic P. The concept of apolipoprotein-defined lipoprotein families and its clinical significance. Curr Atheroscler Rep. 2003;5(6):459–67. [DOI] [PubMed] [Google Scholar]
- 107.Saito H, Lund-Katz S, Phillips MC. Contributions of domain structure and lipid interaction to the functionality of exchangeable human apolipoproteins. Prog Lipid Res. 2004;43(4):350–80. [DOI] [PubMed] [Google Scholar]
- 108.Neary RH, Gowland E. Stability of free Apolipoprotein A-1 concentration in serum, and its measurement in normal and hyperlipidemic subjects. Clin Chem. 1987;33(7):1163–9. [PubMed] [Google Scholar]
- 109.Ritter MC, Scanu AM. Apolipoprotein A-II and structure of human serum high density lipoproteins. An approach by reassembly techniques. J Biol Chem. 1979;254(7):2517–25. [PubMed] [Google Scholar]
- 110.Westerterp M, Berbée JF, Delsing DJ, Jong MC, Gijbels MJ, Dahlmans VE, et al. Apolipoprotein C-I binds free fatty acids and reduces their intracellular esterification. J Lipid Res. 2007;48(6):1353–61. [DOI] [PubMed] [Google Scholar]
- 111.Wolska A, Dunbar RL, Freeman LA, Ueda M, Amar MJ, Sviridov DO, et al. Apolipoprotein C-II: new findings related to genetics, biochemistry, and role in triglyceride metabolism. Atherosclerosis. 2017;267:49–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Jensen MK, Aroner SA, Mukamal KJ, Furtado JD, Post WS, Tsai MY, et al. High-Density lipoprotein subspecies defined by presence of Apolipoprotein C-III and incident coronary heart disease in four cohorts. Circulation. 2018;137(13):1364–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Chan DC, Chen MM, Ooi EM, Watts GF. An ABC of Apolipoprotein C-III: a clinically useful new cardiovascular risk factor? Int J Clin Pract. 2008;62(5):799–809. [DOI] [PubMed] [Google Scholar]
- 114.Rassart E, Bedirian A, Do Carmo S, Guinard O, Sirois J, Terrisse L, et al. Apolipoprotein D Biochim Biophys Acta. 2000;1482(1–2):185–98. [DOI] [PubMed]
- 115.Marcel YL, Vezina C, Milne RW. Cholesteryl ester and Apolipoprotein E transfer between human high density lipoproteins and chylomicrons. Biochim Biophys Acta. 1983;750(2):411–7. [DOI] [PubMed] [Google Scholar]
- 116.Ruiz M, Frej C, Holmér A, Guo Li J, Tran S, Dahlbäck B. High-Density Lipoprotein–Associated Apolipoprotein M limits endothelial inflammation by delivering Sphingosine-1-Phosphate to the Sphingosine-1-Phosphate receptor 1. Arterioscler Thromb Vasc Biol. 2017;37(1):118–29. [DOI] [PubMed] [Google Scholar]
- 117.Elsøe S, Ahnström J, Christoffersen C, Hoofnagle AN, Plomgaard P, Heinecke JW, et al. Apolipoprotein M binds oxidized phospholipids and increases the antioxidant effect of HDL. Atherosclerosis. 2012;221(1):91–7. [DOI] [PubMed] [Google Scholar]
- 118.Duchateau PN, Pullinger CR, Orellana RE, Kunitake ST, Naya-Vigne J, O’Connor PM, et al. Apolipoprotein L, a new human high density lipoprotein Apolipoprotein expressed by the pancreas. Identification, cloning, characterization, and plasma distribution of Apolipoprotein L. J Biol Chem. 1997;272(41):25576–82. [DOI] [PubMed] [Google Scholar]
- 119.Nuutinen T, Suuronen T, Kauppinen A, Salminen A, Clusterin. A forgotten player in alzheimer’s disease. Brain Res Rev. 2009;61(2):89–104. [DOI] [PubMed] [Google Scholar]
- 120.Fernández-de-Retana S, Cano-Sarabia M, Marazuela P, Sánchez-Quesada JL, Garcia-Leon A, Montañola A, et al. Characterization of ApoJ-reconstituted high-density lipoprotein (rHDL) nanodisc for the potential treatment of cerebral β-amyloidosis. Sci Rep. 2017;7(1):14637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Wu Z, Gogonea V, Lee X, May RP, Pipich V, Wagner MA, et al. The low resolution structure of ApoA1 in spherical high density lipoprotein revealed by small angle neutron scattering. J Biol Chem. 2011;286(14):12495–508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Kardassis D, Mosialou I, Kanaki M, Tiniakou I, Thymiakou E. Metabolism of HDL and its regulation. Curr Med Chem. 2014;21(25):2864–80. [DOI] [PubMed] [Google Scholar]
- 123.Duong PT, Weibel GL, Lund-Katz S, Rothblat GH, Phillips MC. Characterization and properties of pre beta-HDL particles formed by ABCA1-mediated cellular lipid efflux to apoA-I. J Lipid Res. 2008;49(5):1006–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Chen W, Wu Y, Lu Q, Wang S, Xing D. Endogenous ApoA-I expression in macrophages: A potential target for protection against atherosclerosis. Clin Chim Acta. 2020;505:55–9. [DOI] [PubMed] [Google Scholar]
- 125.Bursill CA, Castro ML, Beattie DT, Nakhla S, van der Vorst E, Heather AK, et al. High-density lipoproteins suppress chemokines and chemokine receptors in vitro and in vivo. Arterioscler Thromb Vasc Biol. 2010;30(9):1773–8. [DOI] [PubMed] [Google Scholar]
- 126.De Nardo D, Labzin LI, Kono H, Seki R, Schmidt SV, Beyer M, et al. High-density lipoprotein mediates anti-inflammatory reprogramming of macrophages via the transcriptional regulator ATF3. Nat Immunol. 2014;15(2):152–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Iqbal AJ, Barrett TJ, Taylor L, McNeill E, Manmadhan A, Recio C et al. Acute exposure to Apolipoprotein A1 inhibits macrophage chemotaxis in vitro and monocyte recruitment in vivo. Elife. 2016;5. [DOI] [PMC free article] [PubMed]
- 128.Cromwell WC, Otvos JD, Keyes MJ, Pencina MJ, Sullivan L, Vasan RS, et al. LDL particle number and risk of future cardiovascular disease in the Framingham offspring Study - Implications for LDL management. J Clin Lipidol. 2007;1(6):583–92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Radkhah N, Shabbidar S, Zarezadeh M, Safaeiyan A, Barzegar A. Effects of vitamin D supplementation on Apolipoprotein A1 and B100 levels in adults: systematic review and meta-analysis of controlled clinical trials. J Cardiovasc Thorac Res. 2021;13(3):190–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Luo G, Xu N, Apolipoprotein M. Research progress and clinical perspective. Adv Exp Med Biol. 2020;1276:85–103. [DOI] [PubMed] [Google Scholar]
- 131.Flower DR. The Lipocalin protein family: structure and function. Biochem J. 1996;318(Pt 1):1–14. (Pt 1)(. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Venteclef N, Smith JC, Goodwin B, Delerive P. Liver receptor homolog 1 is a negative regulator of the hepatic acute-phase response. Mol Cell Biol. 2006;26(18):6799–807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133.Nielsen LB, Christoffersen C, Ahnstrom J, Dahlback B. ApoM: gene regulation and effects on HDL metabolism. Trends Endocrinol Metab. 2009;20(2):66–71. [DOI] [PubMed] [Google Scholar]
- 134.Sevvana M, Ahnstrom J, Egerer-Sieber C, Lange HA, Dahlback B, Muller YA. Serendipitous fatty acid binding reveals the structural determinants for ligand recognition in Apolipoprotein M. J Mol Biol. 2009;393(4):920–36. [DOI] [PubMed] [Google Scholar]
- 135.Kim HM, Park BS, Kim J-I, Kim SE, Lee J, Oh SC, et al. Crystal structure of the TLR4-MD-2 complex with bound endotoxin antagonist Eritoran. Cell. 2007;130(5):906–17. [DOI] [PubMed] [Google Scholar]
- 136.Huang LZ, Gao JL, Pu C, Zhang PH, Wang LZ, Feng G, et al. Apolipoprotein M: research progress, regulation and metabolic functions (Review). Mol Med Rep. 2015;12(2):1617–24. [DOI] [PubMed] [Google Scholar]
- 137.Christoffersen C, Ahnström J, Axler O, Christensen EI, Dahlbäck B, Nielsen LB. The signal peptide anchors Apolipoprotein M in plasma lipoproteins and prevents rapid clearance of Apolipoprotein M from plasma*. J Biol Chem. 2008;283(27):18765–72. [DOI] [PubMed] [Google Scholar]
- 138.Mousa H, Thanassoulas A, Zughaier SM. ApoM binds endotoxin contributing to neutralization and clearance by high density lipoprotein. Biochem Biophys Rep. 2023;34:101445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Xiong Y, Hla T. S1P control of endothelial integrity. In: Oldstone MBA, Rosen H, editors. Sphingosine-1-Phosphate signaling in immunology and infectious diseases. Cham: Springer International Publishing; 2014. pp. 85–105. [Google Scholar]
- 140.Tran-Dinh A, Diallo D, Delbosc S, Varela-Perez LM, Dang QB, Lapergue B, et al. HDL and endothelial protection. Br J Pharmacol. 2013;169(3):493–511. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Potì F, Simoni M, Nofer JR. Atheroprotective role of high-density lipoprotein (HDL)-associated sphingosine-1-phosphate (S1P). Cardiovasc Res. 2014;103(3):395–404. [DOI] [PubMed] [Google Scholar]
- 142.Zheng Z, Zeng Y, Zhu X, Tan Y, Li Y, Li Q, et al. ApoM-S1P modulates Ox-LDL-Induced inflammation through the PI3K/Akt signaling pathway in HUVECs. Inflammation. 2019;42(2):606–17. [DOI] [PubMed] [Google Scholar]
- 143.Winkler MS, Märtz KB, Nierhaus A, Daum G, Schwedhelm E, Kluge S, et al. Loss of sphingosine 1-phosphate (S1P) in septic shock is predominantly caused by decreased levels of high-density lipoproteins (HDL). J Intensive Care. 2019;7(1):23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Saddar S, Carriere V, Lee W-R, Tanigaki K, Yuhanna IS, Parathath S, et al. Scavenger receptor class B type I is a plasma membrane cholesterol sensor. Circul Res. 2013;112(1):140–51. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 145.Yao Mattisson I, Christoffersen C. Apolipoprotein M and its impact on endothelial dysfunction and inflammation in the cardiovascular system. Atherosclerosis. 2021;334:76–84. [DOI] [PubMed] [Google Scholar]
- 146.Zhang P, Gao J, Pu C, Feng G, Wang L, Huang L, et al. Effects of hyperlipidaemia on plasma Apolipoprotein M levels in patients with type 2 diabetes mellitus: an independent case-control study. Lipids Health Dis. 2016;15(1):158. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 147.Chirinos JA, Zhao L, Jia Y, Frej C, Adamo L, Mann D, et al. Reduced Apolipoprotein M and adverse outcomes across the spectrum of human heart failure. Circulation. 2020;141(18):1463–76. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Baker NL, Hammad SM, Hunt KJ, Semler A, Klein RL, Lopes-Virella MF. Plasma ApoM levels and progression to kidney dysfunction in patients with type 1 diabetes. Diabetes. 2022;71(8):1795–9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Zheng L, Luo G, Zhang J, Mu Q, Shi Y, Berggren-Söderlund M, et al. Decreased activities of Apolipoprotein m promoter are associated with the susceptibility to coronary artery diseases. Int J Med Sci. 2014;11(4):365–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150.Christoffersen C, Federspiel CK, Borup A, Christensen PM, Madsen AN, Heine M, et al. The Apolipoprotein M/S1P Axis controls triglyceride metabolism and brown fat activity. Cell Rep. 2018;22(1):175–88. [DOI] [PubMed] [Google Scholar]
- 151.Hajny S, Borup A, Elsøe S, Christoffersen C. Increased plasma ApoM levels impair triglyceride turnover in mice. Biochim Et Biophys Acta (BBA) - Mol Cell Biology Lipids. 2021;1866(9):158969. [DOI] [PubMed] [Google Scholar]
- 152.Yu MM, Yao S, Luo KM, Mu QF, Yu Y, Luo GH, et al. Apolipoprotein M increases the expression of vitamin D receptor mRNA in colorectal cancer cells detected with duplex fluorescence reverse transcription-quantitative polymerase chain reaction. Mol Med Rep. 2017;16(2):1167–72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153.Eichinger A, Nasreen A, Kim HJ, Skerra A. Structural insight into the dual ligand specificity and mode of high density lipoprotein association of Apolipoprotein D. J Biol Chem. 2007;282(42):31068–75. [DOI] [PubMed] [Google Scholar]
- 154.Muffat J, Walker DW, Apolipoprotein D. An overview of its role in aging and age-related diseases. Cell Cycle. 2010;9(2):269–73. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155.Terrisse L, Poirier J, Bertrand P, Merched A, Visvikis S, Siest G, et al. Increased levels of Apolipoprotein D in cerebrospinal fluid and hippocampus of alzheimer’s patients. J Neurochem. 1998;71(4):1643–50. [DOI] [PubMed] [Google Scholar]
- 156.Thomas EA, Laws SM, Sutcliffe JG, Harper C, Dean B, McClean C, et al. Apolipoprotein D levels are elevated in prefrontal cortex of subjects with alzheimer’s disease: no relation to Apolipoprotein E expression or genotype. Biol Psychiatry. 2003;54(2):136–41. [DOI] [PubMed] [Google Scholar]
- 157.Do Carmo S, Jacomy H, Talbot PJ, Rassart E. Neuroprotective effect of Apolipoprotein D against human coronavirus OC43-induced encephalitis in mice. J Neurosci. 2008;28(41):10330–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Lopez-Boado YS, Puente XS, Alvarez S, Tolivia J, Binderup L, Lopez-Otin C. Growth Inhibition of human breast cancer cells by 1,25-dihydroxyvitamin D3 is accompanied by induction of Apolipoprotein D expression. Cancer Res. 1997;57(18):4091–7. [PubMed] [Google Scholar]
- 159.Darwish NM, Al-Hail MK, Mohamed Y, Al Saady R, Mohsen S, Zar A et al. The role of apolipoproteins in the commonest cancers: A review. Cancers (Basel). 2023;15(23). [DOI] [PMC free article] [PubMed]
- 160.Wang J-H, Keisala T, Solakivi T, Minasyan A, Kalueff AV, Tuohimaa P. Serum cholesterol and expression of apoai, LXRβ and SREBP2 in vitamin D receptor knock-out mice. J Steroid Biochem Mol Biol. 2009;113(3):222–6. [DOI] [PubMed] [Google Scholar]
- 161.Jaimungal S, Wehmeier K, Mooradian AD, Haas MJ. The emerging evidence for vitamin D–mediated regulation of Apolipoprotein A-I synthesis. Nutr Res. 2011;31(11):805–12. [DOI] [PubMed] [Google Scholar]
- 162.Yu M, Lili P, Chen S, Qinfeng M, Lu Z, Guanghua L, et al. Apolipoprotein M could inhibit growth and metastasis of SMMC7721 cells via vitamin D receptor signaling. Cancer Manage Res. 2019;11(null):3691–701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 163.Du W, Shen T, Li H, Liu Y, He L, Tan L, et al. Low Apolipoprotein M serum levels correlate with systemic lupus erythematosus disease activity and Apolipoprotein M gene polymorphisms with lupus. Lipids Health Dis. 2017;16(1):88. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Hajny S, Christoffersen M, Dalila N, Nielsen LB, Tybjærg-Hansen A, Christoffersen C. Apolipoprotein M and risk of type 2 diabetes. J Clin Endocrinol Metab. 2020;105(9). [DOI] [PubMed]
- 165.Simard J, Veilleux R, de Launoit Y, Haagensen DE, Labrie F. Stimulation of Apolipoprotein D secretion by steroids coincides with Inhibition of cell proliferation in human LNCaP prostate cancer cells. Cancer Res. 1991;51(16):4336–41. [PubMed] [Google Scholar]
- 166.Nagaoka I, Hirota S, Niyonsaba F, Hirata M, Adachi Y, Tamura H, et al. Augmentation of the lipopolysaccharide-neutralizing activities of human Cathelicidin CAP18/LL-37-derived antimicrobial peptides by replacement with hydrophobic and cationic amino acid residues. Clin Diagn Lab Immunol. 2002;9(5):972–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167.Coorens M, Schneider VAF, de Groot AM, van Dijk A, Meijerink M, Wells JM, et al. Cathelicidins inhibit Escherichia coli-Induced TLR2 and TLR4 activation in a Viability-Dependent manner. J Immunol. 2017;199(4):1418–28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Hu Z, Murakami T, Suzuki K, Tamura H, Kuwahara-Arai K, Iba T, et al. Antimicrobial Cathelicidin peptide LL-37 inhibits the LPS/ATP-induced pyroptosis of macrophages by dual mechanism. PLoS ONE. 2014;9(1):e85765. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169.Sorensen O, Bratt T, Johnsen AH, Madsen MT, Borregaard N. The human antibacterial cathelicidin, hCAP-18, is bound to lipoproteins in plasma. J Biol Chem. 1999;274(32):22445–51. [DOI] [PubMed] [Google Scholar]
- 170.Sharba ZF, Shareef RH, Abd BA, Hameed EN. Association between dyslipidemia and vitamin D deficiency: a Cross-Sectional study. Folia Med (Plovdiv). 2021;63(6):965–9. [DOI] [PubMed] [Google Scholar]
- 171.MacGirlley R, Phoswa WN, Mokgalaboni K. Modulatory properties of vitamin D in type 2 diabetic patients: A focus on inflammation and dyslipidemia. Nutrients. 2023;15:21. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
No datasets were generated or analysed during the current study.