Abstract
Background
Children with Down Syndrome (DS) are more likely to have multi-system comorbidities leading to more frequent hospitalizations than the general population. We aim to evaluate whether racial differences contribute to hospitalization outcomes and mortality among children with DS.
Methods
Hospital discharge records were obtained for children (< 21 y) with DS hospitalized between 2006 and 2019 from the Kid’s Inpatient Database. The primary exposure was the Black race. Primary outcomes were invasive mechanical ventilation (IMV) and mortality. Secondary outcomes were non-invasive mechanical ventilation (NIMV), length of hospital stay (LOS), and inflation-adjusted cost of hospitalization (IACH). Multivariable logistic regression models were used to ascertain associations between Black race and outcomes.
Results
Among 163,870 hospitalizations in children with DS, 16,208 (9.89%) were Black children. Compared with non-Black children, Black children were younger, of lower household incomes, more likely to have public insurance, more likely to have asthma, OSA, obesity, prematurity, congenital heart disease, pulmonary hypertension, congenital airway anomalies, neuromuscular weakness, and dysphagia. Descriptive analyses indicated that Black race was associated with higher risks of mortality, IMV, NIMV, longer LOS, and greater IACH. After multivariable adjustment, Black race remained independently associated with mortality (OR:1.35, 95%-CI:1.15–1.59, p < 0.0001), IMV (OR:1.34, 95%-CI:1.23–1.45, p < 0.0001), NIMV (OR:1.41, 95%-CI:1.26–1.59, p < 0.0001) and increased LOS (IRR:1.08, 95%-CI:1.04–1.13, p < 0.0001), but not IACH.
Conclusions
Hospitalized Black children with DS are more likely to be younger, of lower household incomes, with public insurance, and with other underlying comorbidities. Black children had increased risks of mortality and IMV and increased LOS.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12887-025-05899-9.
Keywords: Down syndrome, Black, Children
Background
Down syndrome (DS) is a common chromosomal anomaly observed in children, occurring at a rate of approximately 1 per 800 live births in the United States [1]. Children with DS are at increased risk for a variety of health conditions, including but not limited to dysmorphic features, congenital heart disease, sleep-disordered breathing, gastrointestinal disorders, endocrine disorders, hematologic disorders, and other medical conditions [2]. The DS-associated comorbidities predispose children with DS to increased morbidities and mortality as compared to children without DS, particularly congenital heart diseases, congenital anomalies, leukemia, and low birth weight [3–7]. The American Academy of Pediatrics (AAP) has published comprehensive guidelines for clinicians to streamline the care of children with DS since 1994, with the most recent updates in 2022 [2].
The life expectancy of individuals with DS has significantly increased over the years [3, 8]. This is partially related to improved protocolized medical care and increased awareness of DS-associated comorbidities among the general public. However, evidence suggests that Black children or children of minoritized background with DS experience higher mortality than their White counterparts [3–5, 9]. Previous studies accounting for DS-associated comorbidities (e.g., congenital heart disease) did not fully explain racial disparity in mortality [4, 10]. It was hypothesized that the mortality racial disparity could be partially driven by a myriad of social and environmental exposures, such as neighborhood disadvantage, lower SES, access to medical care, parental education status, medical literacy [11]. We, therefore, planned this study utilizing the Kids’ Inpatient Database to characterize the hospitalization outcomes of children with DS. Specifically, we aimed to characterize the main causes and outcomes of hospitalization among children with DS, and to determine if Black children with DS had different patterns of morbidity and mortality compared to non-Black children.
Methods
This study utilized hospital discharge data from children under 21 years of age, covering the years 2000, 2003, 2006, 2009, 2012, 2016, and 2019, sourced from the US Kids’ Inpatient Database (KID) [12]. The KID is an administrative database that includes a stratified sample of pediatric discharges from all short-term, nonfederal, general, and specialty hospitals involved in the Healthcare Cost and Utilization Project (HCUP). It contains sampling weights to produce national estimates of annual pediatric hospitalizations, outcomes, and healthcare costs [12]. KID data are released every three years, each sample containing about 3 million pediatric discharges from participating states [12]. The KID contains event-level data for each admission, including diagnostic and procedural codes; however, the absence of unique identifiers prevents the tracking of repeated admissions for the same individuals. Since all data collected were deidentified, IRB submission and approval were not required [13].
Children with DS were identified using the International Classification of Diseases, 9th and 10th revisions, Clinical Modification (ICD-9/10-CM) codes 758.0 and Q90.0-Q90.9.
A detailed description of the KID database, study design, and statistical analyses were reported in our previous studies [14–19].
Measurements of variables
The analyses included the following patient characteristics: age, sex, race, type of insurance (e.g., Medicare/Medicaid), and median household income quartiles based on counties of residence. In accordance with KID’s reporting method, Hispanics are categorized as a race rather than an ethnicity [12]. The analyses also considered hospital characteristics, such as hospital region, teaching status, and categorical hospital size based on the hospital’s location. Using previously validated diagnostic codes, DS-associated comorbidities were identified, including prematurity, OSA, asthma, pulmonary hypertension, congenital heart diseases, arrhythmia, congenital airway anomalies, laryngeal, glottic, and subglottic stenosis (LGSS), which were grouped under the same ICD-9/10 codes of 478.74 and J38.6, as well as hypothyroidism, leukemia, Hirschsprung’s disease, dysphagia, and gastroesophageal reflux disease (GERD) [17, 20–24].
Definition of outcomes
The primary outcome was mortality, while the co-primary outcome was invasive mechanical ventilation, a binary variable identified by the Clinical Classifications Software (CCS) code 216 (Respiratory intubation and mechanical ventilation) [25]. This methodology, created by AHRQ, categorizes ICD-9-CM codes into clinically relevant and mutually exclusive groups. Secondary outcomes included length of hospital stay (LOS), and inflation-adjusted cost of hospitalization (IACH).
Summary of diagnostic categories
The Clinical Classifications Software (CCS) is a validated tool used to summarize common diagnoses in children with DS, based on primary ICD-9 and ICD-10 diagnostic and procedural codes [25].
CCS tools also facilitated the grouping of diagnoses into 18 broader, mutually exclusive categories (e.g., infectious disease, respiratory illness), offering greater insights into specific groupings of codes [25].
Statistical analysis
Children with diagnosis of DS were summarized and stratified by Black race (Black vs. non-Black). Categorical variables were reported as counts and proportions, while continuous variables were presented as means with standard deviations (SD). Descriptive statistics were used to examine differences between groups. A Jonckheere–Terpstra test for trend was conducted to evaluate linear trends in mortality rates among all children with DS, as well as among Black, White and non-Black children with DS over the study period [15].
Multivariable logistic regression analyses were conducted to examine the associations between Black race and primary outcomes in children with DS, adjusting for patient characteristics, hospital characteristics, and patients’ comorbidities. Two models were used to adjust for different sets of factors: (1) The base model (model 1) adjusted for age categories, sex, teaching hospital status, prematurity, congenital heart diseases, congenital airway anomalies, asthma, obesity, LGSS, neuromuscular weakness, GERD, leukemia, and hypothyroidism. (2) The extended model (model 2) further adjusted for insurance status and lower household income levels, defined as 1–25% of the median household income. Insurance status and household income level were included in model 2 as it is considered a metric for socioeconomic disadvantage rather than demographics alone.
To analyze LOS, a multivariable negative binomial model was used to examine its association with Black race. The effect measure of this model is the incidence rate ratio (IRR). All charges were adjusted to 2019 US dollars using the average Consumer Price Index to enable direct comparisons of hospital charges across years while accounting for inflation. The IACH was also log-transformed to address the data’s skewness. A multivariable linear regression model was then employed to assess the relationship between log-transformed IACH and Black race.
Recognizing that non-Black children represent a heterogeneous reference group, we performed sensitivity analyses, including: (1) descriptive statistics stratified by each original KID racial category, and (2) multivariable models directly comparing Black children with White children only.
In the multivariable analyses, sensitivity analyses were performed with the inclusion of CCS codes. The rationale was that the severity of diseases could be directly related to our outcomes. Adjustment of primary diagnostic disease leading to hospitalization would thus allow for better assessment of the association between race and outcomes. Additionally, to explore whether Black were more likely to be affected by social determinants of health factors, we introduced interaction terms between Black race, insurance status and low-income status.
All the analyses accounted for sampling weights provided in the KID and were consistent with HCUP reporting methodology [12]. All tests were two-sided, and a p-value of less than 0.05 was considered statistically significant. Statistical analysis was conducted using Stata 14.0 (Stata Corp, College Station, TX).
Results
Demographics and clinical characteristics of hospitalized children with DS
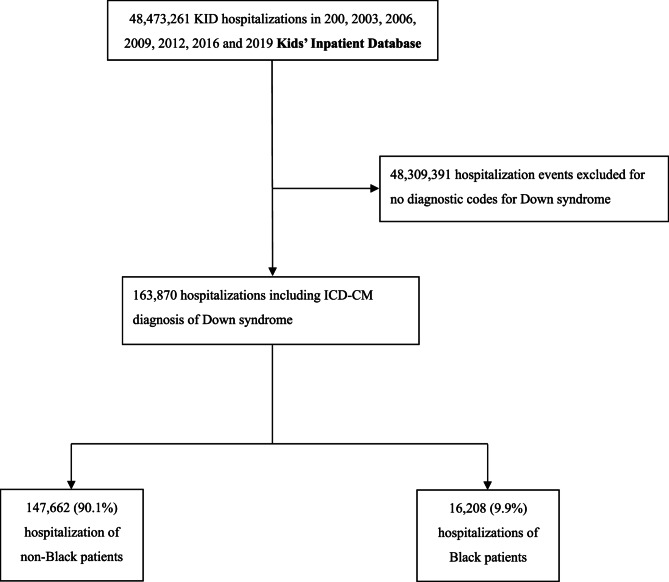
Out of 48,473,261 hospitalizations during the study period, 163,870 (0.34%) involved a diagnosis of DS. Figure 1 provides details on how the study cohort was assembled. The average age of the patients was 3.4 years (SD 0.03), with 44.9% being female and 9.9% being Black children. Black children were generally younger, from lower-income households, more likely to have public insurance, and had higher rates of asthma, OSA, obesity, prematurity, congenital heart disease, pulmonary hypertension, congenital airway anomalies, neuromuscular weakness, epilepsy, and GERD compared to non-Black children. Table 1 offers a detailed comparison between Black and non-Black children. The comparison between Black and other groups was provided in Supplemental Table 1.
Fig. 1.
Construction of the study cohort. Schematic illustration of the construction of the study cohort using the Kids Inpatient Database (KID)
Table 1.
Characteristics of patients with Down syndrome hospitalized during 2000–2019
| Total (n=163,870) | Non-Black (n=147,662) | Black (n=16,208) | p value | |
|---|---|---|---|---|
| Demographic characteristics | ||||
| Age (y), mean (SD) | 3.40 (0.03) | 3.41 (0.03) | 3.37 (0.06) | <.0001 |
| 15-21 years, n (%) | 12076 (7.37) | 10861 (7.36) | 1215 (7.49) | 0.1092 |
| 10-14 years, n (%) | 11824 (7.22) | 10675 (7.23) | 1149 (7.09) | |
| 5-9 years, n (%) | 17463 (10.66) | 15846 (10.73) | 1617 (9.98) | |
| 2-4 years, n (%) | 27097 (16.54) | 24500 (57.82) | 2597 (16.02) | |
| 0–1 year, n (%) | 95293 (58.15) | 85665 (58.01) | 9628 (59.40) | |
| Sex | ||||
| Female, n (%) | 73522 (44.87) | 66289 (44.89) | 7233 (44.63) | 0.0687 |
| Race/Ethnicity | ||||
| White, n (%) | 69765 (42.57) | N/A | N/A | N/A |
| Black, n (%) | 16208 (9.89) | N/A | N/A | |
| Asian, n (%) | 4540 (2.77) | N/A | N/A | |
| Hispanic, n (%) | 39482 (24.09) | N/A | N/A | |
| Median household income | ||||
| 0-25 %, n (%) | 41537 (25.35) | 34660 (83.44) | 6877 (42.43) | <.0001 |
| 26-50 %, n (%) | 41212 (25.15) | 37225 (25.21) | 3987 (24.60) | |
| 51-75 %, n (%) | 41516 (25.33) | 38608 (26.15) | 2908 (17.94) | |
| 76-100 %, n (%) | 36844 (22.48) | 34741 (23.53) | 2103 (12.98) | |
| Insurance | ||||
| Public, n (%) | 77025 (47.00) | 66571 (45.08) | 10454 (64.50) | <.0001 |
| Private, n (%) | 66757 (40.74) | 63002 (42.67) | 3755 (23.17) | |
| Teaching Hospital, n (%) | 126688 (77.31) | 17914 (12.13) | 1980 (12.22) | <.0001 |
| Clinical Characteristics | ||||
| OSA, n (%) | 13453 (8.26) | 11600 (7.86) | 1942 (11.98) | <.0001 |
| Asthma, n (%) | 4265 (2.60) | 3748 (2.54) | 517 (3.19) | 0.0010 |
| Obesity, n (%) | 2695 (1.64) | 2194 (1.49) | 501 (3.09) | <.0001 |
| Prematurity, n (%) | 11303 (6.90) | 10052 (6.81) | 1251 (7.72) | 0.0016 |
| Arrhythmia, n (%) | 5405 (3.30) | 4751 (3.22) | 654 (4.04) | 0.0001 |
| Congenital cardiac anomalies, n (%) | 68789 (41.98) | 61603 (41.72) | 7185 (44.33) | 0.0001 |
| Pulmonary hypertension, n (%) | 8840 (5.39) | 7745 (5.25) | 1095 (6.76) | 0.0002 |
| Laryngeal stenosis, n (%) | 4250 (2.59) | 3726 (2.52) | 524 (3.23) | 0.0010 |
| Congenital craniofacial anomalies, n (%) | 173 (0.11) | 158 (0.11) | 14 (0.09) | 0.5869 |
| Congenital airway anomalies, n (%) | 6725 (4.10) | 5875 (3.98) | 850 (5.24) | <.0001 |
| Neuromuscular weakness, n (%) | 3142 (1.92) | 2745 (1.86) | 397 (2.45) | 0.0093 |
| Epilepsy, n (%) | 8124 (4.96) | 7093 (4.80) | 1030 (6.36) | <.0001 |
| Hirschsprung’s disease, n (%) | 3419 (2.09) | 3052 (2.07) | 366 (2.26) | 0.2985 |
| SGA, n (%) | 2432 (1.48) | 2150 (1.46) | 282 (1.74) | 0.0189 |
| Hypothyroidism, n (%) | 17754 (10.83) | 16326 (11.06) | 1428 (8.81) | <.0001 |
| Leukemia, n (%) | 6733 (4.11) | 6335 (4.29) | 398 (2.45) | <.0001 |
| Dysphagia, n (%) | 6967 (4.25) | 6134 (4.15) | 833 (5.14) | 0.0011 |
| GERD, n (%) | 18078 (11.03) | 16276 (11.02) | 1802 (11.12) | 0.8388 |
| Treatment and outcomes | ||||
| AT, n (%) | 3409 (2.08) | 2939 (1.99) | 471 (2.90) | <.0001 |
| Gastrostomy, n (%) | 4549 (2.78) | 4045 (2.74) | 504 (3.11) | 0.0480 |
| IMV, n (%) | 12963 (7.91) | 11205 (7.59) | 1758 (10.85) | <.0001 |
| NIMV, n (%) | 6260 (3.82) | 5360 (3.63) | 899 (5.55) | <.0001 |
| Mortality, n (%) | 2031 (1.24) | 1748 (1.18) | 283 (1.75) | <.0001 |
| Cost of hospitalization, mean (SD)† | 75906 (1689) | 74725 (1708) | 86704 (3206) | <.0001 |
| Length of hospitalization (d), mean (SD) | 8.05 (0.07) | 7.92 (0.07) | 9.18 (0.19) | <.0001 |
Abbreviation: OSA Obstructive sleep apnea, IMV Invasive mechanical ventilation, NIMV Non-invasive mechanical ventilation, SGA Small for gestational age, T Tonsillectomy, A Adenoidectomy, SD Standard deviation, n sample size, d day, GERD Gastroesophageal reflux disease, N/A Not available
†Costs were adjusted to 2019 USD using average consumer price index
Most common diagnostic and procedural codes for hospitalization
Among the 163,870 hospitalizations with a diagnosis of DS, following the diagnostic code of DS, the most frequent primary diagnoses were: (1) liveborn (19.95%), (2) pneumonia (11.6%), and (3) cardiac and circulatory congenital anomalies (9.38%). The most common procedures were: (1) other OR heart procedures (5.88%), (2) respiratory intubation and mechanical ventilation (5.55%), and (3) other therapeutic procedures (4.06%) as shown in Table 2. For non-Black children (n = 147,662), the top primary diagnoses were: (1) liveborn (19.97%), (2) pneumonia (11.53%), and (3) cardiac and circulatory congenital anomalies (9.32%). The most common procedures included: (1) other OR heart procedures (5.86%), (2) respiratory intubation and mechanical ventilation (5.29%), and (3) other therapeutic procedures (4.12%). For Black children (n = 16,208), the most frequent primary diagnoses were: (1) liveborn (19.81%), (2) cardiac and circulatory congenital anomalies (9.89%), and (3) pneumonia (9.85%). The most common procedures were: (1) respiratory intubation and mechanical ventilation (7.90%), (2) other OR heart procedures (6.06%), and (3) other therapeutic procedures (3.55%), as detailed in Table 2.
Table 2.
Common primary diagnostic and procedural codes of patients with Down syndrome hospitalized during 2000–2019
| Total (n=163,870) | Non-Black (n=147,662) | Black (n=16,208) | ||||
|---|---|---|---|---|---|---|
| Most common diagnostic codes | n (%) | Most common diagnostic codes | n (%) | Most common diagnostic codes | n (%) | |
| 1 | Liveborn | 32693 (19.95) | Liveborn | 29482 (19.97) | Liveborn | 3211 (19.81) |
| 2 | Pneumonia (except that caused by tuberculosis or sexually transmitted disease) | 18624 (11.36) | Pneumonia (except that caused by tuberculosis or sexually transmitted disease) | 17028 (11.53) | Cardiac and circulatory congenital anomalies | 1603 (9.89) |
| 3 | Cardiac and circulatory congenital anomalies | 15365 (9.38) | Cardiac and circulatory congenital anomalies | 13763 (9.32) | Pneumonia (except that caused by tuberculosis or sexually transmitted disease) | 1596 (9.85) |
| 4 | Acute bronchitis | 9428 (5.75) | Acute bronchitis | 8583 (5.81) | Acute bronchitis | 844 (5.21) |
| 5 | Other upper respiratory infections | 4028 (2.46) | Other perinatal conditions | 3593 (2.43) | Respiratory failure; insufficiency; arrest | 474 (2.93) |
| 6 | Other perinatal conditions | 3914 (2.39) | Other upper respiratory infections | 3572 (2.42) | Other upper respiratory infections | 456 (2.82) |
| 7 | Maintenance chemotherapy; radiotherapy | 3865 (2.36) | Maintenance chemotherapy; radiotherapy | 3531 (2.39) | Asthma | 367 (2.26) |
| 8 | Fluid and electrolyte disorders | 3644 (2.22) | Fluid and electrolyte disorders | 3170 (2.15) | Acute and chronic tonsillitis | 329 (2.03) |
| 9 | Respiratory failure; insufficiency; arrest | 3139 (1.92) | Respiratory failure; insufficiency; arrest | 2927 (1.98) | Maintenance chemotherapy; radiotherapy | 321 (1.98) |
| 10 | Digestive congenital anomalies | 3137 (1.91) | Digestive congenital anomalies | 2894 (1.96) | Other perinatal conditions | 321 (1.98) |
| Most common procedural codes | n (%) | Most common procedural codes | n (%) | Most common procedural codes | n (%) | |
| 1 | Other OR heart procedures | 9632 (5.88) | Other OR heart procedures | 8649 (5.86) | Respiratory intubation and mechanical ventilation | 1280 (7.90) |
| 2 | Respiratory intubation and mechanical ventilation | 9090 (5.55) | Respiratory intubation and mechanical ventilation | 7810 (5.29) | Other OR heart procedures | 983 (6.06) |
| 3 | Other therapeutic procedures | 6654 (4.06) | Other therapeutic procedures | 6078 (4.12) | Other therapeutic procedures | 576 (3.55) |
| 4 | Circumcision | 4674 (2.85) | Circumcision | 4111 (2.78) | Circumcision | 563 (3.47) |
| 5 | Prophylactic vaccinations and inoculations | 4335 (2.65) | Prophylactic vaccinations and inoculations | 3776 (2.56) | Prophylactic vaccinations and inoculations | 559 (3.45) |
| 6 | Other vascular catheterization; not heart | 3876 (2.37) | Other vascular catheterization; not heart | 3499 (2.37) | Tonsillectomy and/or adenoidectomy | 424 (2.62) |
| 7 | Enteral and parenteral nutrition | 3770 (2.30) | Enteral and parenteral nutrition | 3369 (2.28) | Enteral and parenteral nutrition | 401 (2.47) |
| 8 | Cancer chemotherapy | 3419 (2.09) | Cancer chemotherapy | 3161 (2.14) | Other vascular catheterization; not heart | 377 (2.32) |
| 9 | Tonsillectomy and/or adenoidectomy | 3029 (1.85) | Tonsillectomy and/or adenoidectomy | 2605 (1.76) | Cancer chemotherapy | 259 (1.60) |
| 10 | Heart valve procedures | 2238 (1.37) | Heart valve procedures | 2014 (1.36) | Heart valve procedures | 224 (1.38) |
Abbreviation: n sample size, OR Operation room
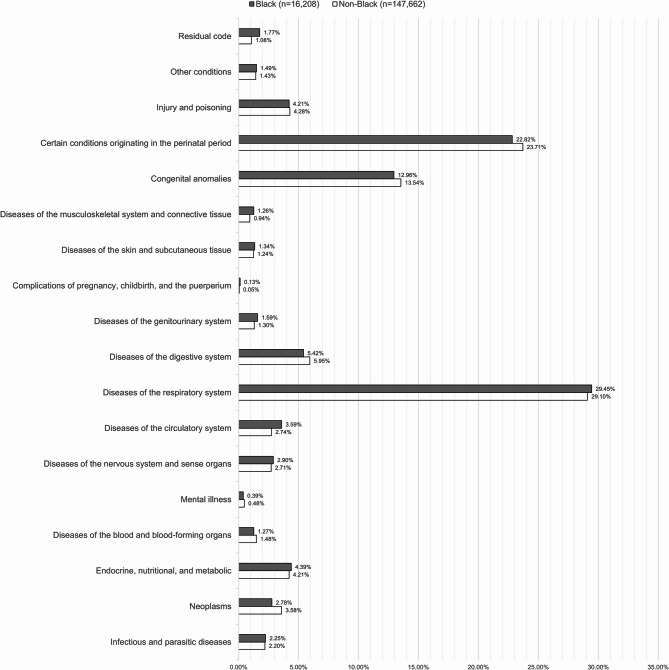
Using CCS Diagnostic Categories to classify primary diagnostic codes into 18 mutually exclusive groups, the most common diagnostic categories for all hospitalizations (n = 163,870) were: (1) diseases of the respiratory system (29.13%), (2) certain conditions originating in the perinatal period (23.62%), and (3) congenital anomalies (13.48%) (Fig. 2). For non-Black children (n = 147,662), the most frequent diagnostic categories were: (1) diseases of the respiratory system (29.10%), (2) certain conditions originating in the perinatal period (23.71%), and (3) congenital anomalies (13.54%). For Black children (n = 16,208), the top diagnostic categories were: (1) diseases of the respiratory system (29.45%), (2) certain conditions originating in the perinatal period (22.82%), and (3) congenital anomalies (12.96%) (Fig. 2).
Fig. 2.
Primary diagnostic categories for hospitalizations, expressed as a percentage, based on Clinical Classifications Software (CCS) for children with Down syndrome, stratified by Black race from year 2000 to year 2019. Diagnostic categories expressed as a percentage using the Clinical Classifications Software (CCS) for children with Down syndrome, stratified by Black Children (n=16,208), and non-Black Children (n=147,662)
Primary outcomes
Among the 163,870 hospitalizations with DS, 12,963 (7.91%) received IMV, and 2,031 (1.24%) had mortality (Table 1). Multivariable adjusted regression models (model 1) showed that Black race was associated with a higher risk of IMV (adjusted OR 1.36 [95% CI 1.26–1.48], p < 0.0001) and mortality (adjusted OR 1.37 [95% CI 1.17–1.61], p < 0.0001). Even after further adjusting for low household income and insurance status (model 2), Black race remained significantly associated with IMV (adjusted OR 1.34 [95% CI 1.23–1.45], p < 0.0001) and mortality (adjusted OR 1.35 [95% CI 1.15–1.59], p < 0.0001), with no notable attenuation in these associations (0.29% decrease for IMV and 1.69% decrease for mortality) compared to model 1 (Table 3). Multivariable-adjusted regression showed that Black race remained an independent predictor for increased mortality and risk of IMV as compared to White children (Supplemental Table 2).
Table 3.
Multivariable analyses of Black vs. non-Black race with invasive mechanical ventilation and mortality among children with Down syndrome hospitalized
| Predictors | Invasive mechanical ventilation | Mortality | ||||||
|---|---|---|---|---|---|---|---|---|
| Model 1* | Model 2* | Model 1* | Model 2* | |||||
| aOR (95% CI) | P value | aOR (95% CI) | P value | aOR (95% CI) | P value | aOR (95% CI) | P value | |
| Black† | 1.36 (1.26 - 1.48) | <.0001 | 1.34 (1.23 - 1.45) | <.0001 | 1.37 (1.17 - 1.61) | <.0001 | 1.35 (1.15 - 1.59) | <.0001 |
| Infancy | 1.37 (1.27 - 1.47) | <.0001 | 1.36 (1.27 - 1.46) | <.0001 | 1.18 (1.01 - 1.38) | 0.035 | 1.16 (0.99 - 1.36) | 0.059 |
| Female | 0.98 (0.93 - 1.02) | 0.329 | 0.98 (0.93 - 1.02) | 0.334 | 1.01 (0.90 - 1.13) | 0.846 | 1.02 (0.91 - 1.15) | 0.702 |
| Insurance | ||||||||
| Public | N/A | N/A | Ref | N/A | N/A | Ref | ||
| Private | N/A | N/A | 0.96 (0.90 - 1.02) | 0.174 | N/A | N/A | 0.86 (0.75 - 0.98) | 0.025 |
| Others | N/A | N/A | 1.03 (0.92 - 1.17) | 0.584 | N/A | N/A | 1.29 (1.05 - 1.58) | 0.016 |
| Low household income (1-25%) | N/A | N/A | 1.07 (1.00 - 1.13) | 0.046 | N/A | N/A | 0.93 (0.82 - 1.06) | 0.016 |
| Teaching Hospital | 2.05 (1.83 - 2.30) | <.0001 | 2.03 (1.81 - 2.28) | <.0001 | 2.13 (1.76 - 2.59) | <.0001 | 2.10 (1.73 - 2.55) | <.0001 |
| Prematurity | 2.18 (2.01 - 2.36) | <.0001 | 2.19 (2.02 - 2.37) | <.0001 | 5.53 (4.75 - 6.45) | <.0001 | 5.69 (4.88 - 6.64) | <.0001 |
| Asthma | 0.55 (0.42 - 0.72) | <.0001 | 0.55 (0.41 - 0.72) | <.0001 | 0.76 (0.48 - 1.21) | 0.192 | 0.74 (0. - 6.64) | 0.246 |
| Obesity | 2.30 (1.93 - 2.73) | <.0001 | 2.30 (1.93 - 2.74) | <.0001 | 1.96 (1.31 - 2.93) | 0.001 | 1.99 (1.33 - 2.98) | 0.001 |
| Congenital airway anomalies | 2.26 (2.05 - 2.49) | <.0001 | 2.27 (2.05 - 2.50) | <.0001 | 2.47 (2.00 - 3.04) | <.0001 | 2.40 (1.93 - 2.97) | <.0001 |
| Congenital cardiac anomalies | 2.09 (1.95 - 2.24) | <.0001 | 2.09 (1.96 - 2.24) | <.0001 | 1.24 (1.07 - 1.43) | 0.003 | 1.24 (1.07 - 1.43) | 0.003 |
| GERD | 1.18 (1.09 - 1.28) | <.0001 | 1.18 (1.09 - 1.28) | <.0001 | 0.51 (0.40 - 0.64) | <.0001 | 0.52 (0.42 - 0.66) | <.0001 |
| Leukemia | 0.39 (0.31 - 0.51) | <.0001 | 0.38 (0.30 - 0.49) | <.0001 | 1.39 (1.01 - 1.90) | 0.042 | 1.36 (0.99 - 1.86) | 0.056 |
| Hypothyroidism | 1.23 (1.13 - 1.33) | <.0001 | 1.23 (1.13 - 1.33) | <.0001 | 0.76 (0.61 - 0.94) | 0.012 | 0.74 (0.60 - 0.92) | 0.007 |
Abbreviation: CI Confidence interval, OR Odds ratio, GERD Gastroesophageal reflux disease, aOR adjusted odds ratio
*Variables adjusted for in model 1: infancy (0-1 year), sex, Black race, teaching hospital status, prematurity, asthma, obesity, congenital airway anomalies, congenital heart disease, GERD, leukemia, and hypothyroidism; Variables adjusted for in the model 2: infancy (0-1 year), sex, Black race, teaching hospital status, prematurity, asthma, obesity, congenital airway anomalies, congenital heart disease, GERD, leukemia, hypothyroidism, insurance status, and low household income status
†Reference group is non-Black children
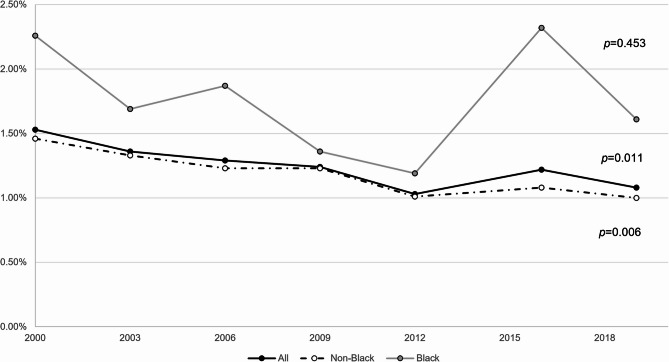
Trend analysis revealed that the overall mortality rate for children with DS steadily declined from 1.53% in 2000 to 1.08% in 2019 (p = 0.011). For non-Black children, mortality fell from 1.46 to 1.00% (p = 0.006); for Black children, it decreased from 2.26 to 1.69% (p = 0.453) (Fig. 3). Among White children, mortality dropped from 1.39 to 0.89% (p = 0.006) over the same period (Figure E1).
Fig. 3.
The trend of the mortality rate of Black and non-Black children with Down syndrome hospitalized from year 2000 to year 2019. Schematic illustration of the changes in rates of mortality among hospitalized children with Down syndrome from year 2000 to year 2019. Jonckheere–Terpstra test showed significant changes in trends for all the patients group (p=0.011) and non-Black children (p=0.006), while a non-significant change in trend in mortality for Black children (p=0.453)
Secondary outcomes: non-invasive mechanical ventilation, hospital utilization and healthcare cost
Among 163,870 hospitalizations with DS, 6,360 (3.82%) had NIMV. Children with DS had a mean IACH of $75,906 (SD $1,689) and a mean LOS of 8.05 days (SD 0.07). Univariable descriptive statistics showed that, compared to non-Black children, Black children were more likely to receive NIMV, and had higher IACH and longer LOS (Table 1).
In multivariable-adjusted logistic regression, Black race was associated with a higher risk of NIMV (adjusted OR 1.41 [95% CI 1.26–1.59], p < 0.0001). The negative binomial regression model indicated that Black race was a significant predictor of increased LOS (adjusted IRR 1.08 [95% CI 1.04–1.13], p < 0.0001). However, in the multivariable linear regression, Black race was not significantly associated with IACH (adjusted β −0.01 [95% CI −0.06–0.03], p = 0.585) (Table 4). Multivariable-adjusted regression demonstrated that Black race remained an independent predictor of increased risk for NIMV, longer LOS, and higher IACH compared to White children (Supplemental Tables 4 and 5).
Table 4.
Multivariable analyses of Black vs. non-Black race with secondary outcomes among children with Down syndrome hospitalized
| Predictors | Non-Invasive mechanical ventilation | Length of hospital stay (d) | Log-transformed cost of hospitalization (USD) | |||
|---|---|---|---|---|---|---|
| OR (95% CI) | P value | IRR (95% CI) | P value | β (95% CI) | P value | |
| Black† | 1.41 (1.26 - 1.59) | <.0001 | 1.08 (1.04 - 1.13) | <.0001 | -0.01 (-0.06 - 0.03) | 0.585 |
| Infancy | 1.36 (1.23 - 1.52) | <.0001 | 1.34 (1.30 - 1.37) | <.0001 | -0.09 (-0.12 - -0.06) | <.0001 |
| Female | 1.00 (0.94 - 1.08) | 0.895 | 1.01 (0.99 - 1.03) | 0.533 | -0.01 (-0.02 - 0.01) | 0.616 |
| Insurance | ||||||
| Public | Ref | Ref | Ref | |||
| Private | 1.02 (0.93 - 1.12) | 0.686 | 0.90 (0.87 - 0.92) | <.0001 | -0.13 (-0.16 - -0.10) | <.0001 |
| Others | 1.08 (0.86 - 1.35) | 0.529 | 0.93 (0.89 - 0.98) | 0.008 | -0.18 (-0.25 - -0.11) | <.0001 |
| Low household income (1-25%) | 1.00 (0.91 - 1.10) | 0.996 | 1.05 (1.02 - 1.07) | <.0001 | 0.05 (0.02 - 0.09) | 0.001 |
| Teaching Hospital | 2.20 (1.88 - 2.56) | <.0001 | 1.42 (1.37 - 1.47) | <.0001 | 0.76 (0.69 - 0.82) | <.0001 |
| Prematurity | 3.51 (3.18 - 3.88) | <.0001 | 2.55 (2.47 - 2.64) | <.0001 | 0.69 (0.64 - 0.74) | <.0001 |
| Asthma | 1.85 (1.48 - 2.31) | <.0001 | 0.86 (0.82 - 0.91) | <.0001 | -0.09 (-0.15 - -0.04) | 0.001 |
| Obesity | 4.48 (3.71 - 5.41) | <.0001 | 1.51 (1.38 - 1.64) | <.0001 | 0.57 (0.50 - 0.63) | <.0001 |
| Congenital airway anomalies | 1.60 (1.41 - 1.82) | <.0001 | 1.58 (1.50 - 1.67) | <.0001 | 0.46 (0.41 - 0.51) | <.0001 |
| Congenital cardiac anomalies | 1.74 (1.60 - 1.88) | <.0001 | 1.76 (1.71 - 1.80) | <.0001 | 0.86 (0.83 - 0.89) | <.0001 |
| GERD | 1.05 (0.92 - 1.19) | <.0001 | 1.22 (1.18 - 1.27) | <.0001 | 0.25 (0.21 - 0.28) | <.0001 |
| Leukemia | 0.24 (0.15 - 0.37) | <.0001 | 2.04 (1.93 - 2.16) | <.0001 | 0.57 (0.49 - 0.64) | <.0001 |
| Hypothyroidism | 1.30 (1.17 - 1.44) | <.0001 | 1.15 (1.11 - 1.19) | <.0001 | 0.24 (0.21 - 0.28) | <.0001 |
Abbreviation: IRR Incidence rate ratio, CI Confidence interval, USD US Dollars, d days, NIMV Non-invasive mechanical ventilation, GERD Gastroesophageal reflux, OR Odds ratio
†Reference group is non-Black children
Sensitivity analyses
Sensitivity analyses including underlying major diagnostic categories did not change the results on multivariable analyses for primary and secondary outcomes. Exploratory analyses with inclusion of interaction terms between Black and insurance type and low-income status did not show significant interaction for mortality, and mechanical ventilation (Supplemental Table 2). Interestingly, for the length of stay, there was a significant interaction between Black and low-income status (IRR = 0.92 (0.85–1.00), p = 0.042) (Supplemental Table 3). For inflation-adjusted cost of hospitalization, there was a significant interaction between Black and private insurance (β = 0.15 (0.07–0.23), p < 0.001) (Supplemental Table3).
Discussion
This study demonstrated that Black children with DS have higher mortality, increased risk of IMV and NIMV, and longer LOS compared to non-Black children while accounting for DS-associated comorbidities. Respiratory diseases and congenital anomalies were the major contributors to hospitalizations for all children with DS. Racial disparity in mortality, respiratory failure, and LOS suggest that efforts are needed to identify and intervene with drivers to improve the care of Black children with DS.
Our findings are aligned with previous literature demonstrating that children with DS have higher mortality, and increased likelihood of having not only DS-associated comorbidities, such as congenital heart diseases, pHTN, and oropharyngeal dysphagia but also conditions that disproportionately affect among Black children, such as asthma and obesity [26–28]. These findings resonated with AAP practice guidelines to manage DS-associated health conditions [2]. Furthermore, Black children also have higher mortality, increased likelihood of respiratory failure needing intubation and mechanical ventilation, and longer LOS, independent of the effects from DS-comorbidities (e.g., congenital heart diseases, immune dysregulation). Our findings of racial disparity in mortality and healthcare resources utilization among patients with DS are aligned with previous studies [29, 30]. While the literature has suggested the possibility of mediating effects of adverse exposures associated with living in disadvantaged neighborhoods and lower socioeconomic status on mortalities and DS-associated morbidity [11], we did not see significant attenuation of the associations between Black children and mortality after accounting for insurance status and household income. We hypothesized these null findings are due to the fact that household income and insurance status are not comprehensive metrics able to capture the adverse exposures associated with living in disadvantaged neighborhood and lower socioeconomic status. Limited by the nature of this administrative database, we were not able to use a more comprehensive metric such as the Childhood Opportunity Index (COI)) [31, 32].
Our sensitivity analyses of the race-by-socioeconomic interactions yielded two complementary insights. First, although Black race and low-income status were each independently associated with longer hospitalization, their joint effect on length of stay was sub-multiplicative (interaction IRR = 0.92). This finding implies that racial disparities in LOS are most pronounced among higher-income children and are attenuated within low-income strata, perhaps because Medicaid’s uniform discharge incentives and shared access barriers constrain LOS similarly across low-income groups. Second, the interaction between Black race and private insurance was positive (interaction β = 0.15), showing that the cost savings typically observed with private coverage are largely absent for Black children; within the privately insured cohort, Black patients incurred higher inpatient charges than their non-Black peers. These cost disparities may reflect differences in hospital case-mix, billing intensity, or payer-specific negotiated rates rather than LOS alone. Further research is needed to elucidate the mechanisms through which race and socioeconomic factors intersect to influence resource use and clinical outcomes.
Notably, this study showed that Black children with DS had a lower likelihood of hypothyroidism. We hypothesized that this finding could be partially related to accessibility to healthcare resources. While the AAP guidelines recommend newborn screening for thyroid function and annual thyroid function labs [2], inaccessibility to healthcare screening might preclude Black children from timely diagnosis and treatment. Additionally, other contributing factors include selection bias (hospitalized patients only) and misclassification bias (under-coding). Future prospective studies are needed to delineate this observation.
Respiratory and congenital heart diseases are the most common diagnoses for hospitalizations for all children with DS. Respiratory diseases are the most common diagnostic category in children with DS, with pneumonia, acute bronchitis, and URI being the most frequent codes. Respiratory diseases in children with DS could result from a combination of factors, including ineffective airway mucus clearance from decreased muscle tone, oropharyngeal dysphagia, immunologic impairment in both innate and adaptive systems, upper and/or lower airway anomalies, and interstitial lung disease, although rare [33–35]. Other complicating factors may also lead to decreased pulmonary reserve and hence more severe manifestations of respiratory infection, including congenital heart diseases, gastroesophageal reflux, and dysphagia. Previous studies have demonstrated children with DS are at higher risk of morbidity with RSV bronchiolitis [36]. These emphasize the importance of routine vaccination of children with DS, including pneumococcal and influenza vaccine. Additionally, children with DS will benefit from passive immunoprophylaxis (e.g., Nirsevimab) and maternal RSV vaccine during pregnancy [37]. For children with DS under 8 months of age, if the mother did not receive an RSV vaccination at least 14 days prior to delivery, clinicians should consider Nirsevimab, which provides 5-month protection during RSV season. For children with DS aged 8 through 19 months who are at increased risk for severe RSV (e.g., those with chronic lung disease or cystic fibrosis), a second dose of Nirsevimab upon entering their second RSV season is also recommended. In the event that Nirsevimab is not available, Palivizumab, which requires monthly injections, should still be considered for high-risk patients (e.g., those with congenital heart disease or bronchopulmonary dysplasia) [38]. For subgroups of patients at risk of recurrent respiratory infection or with inadequate titer response due to less specific switched memory B cells, pneumococcal booster vaccine could be considered.
Congenital heart diseases were also a major contributor to the hospitalization of our study population. Literature has suggested congenital heart disease was a major contributor not only to mortality for all children with DS but also to racial disparity in mortality [3, 39]. In a longitudinal analysis of children with DS, Santoro et al. have also shown an increased incidence of congenital heart disease and referral for Black children with DS compared with White children [30]. These suggest timely screening for congenital heart disease during pregnancy and prior to discharge from the nursery, and high clinical suspicion for underdiagnosed heart diseases should be exerted when evaluating patients with DS.
We noticed a trend of decreasing the mortality rate of our study sample throughout the study period. Although the downward trend for Black children did not reach statistical significance, the absolute reduction (from 2.26 to 1.69%) was comparable to that seen in non-Black and White children. The absence of significance could be due to limited sample size and loss of statistical power rather than a qualitatively different trajectory. Importantly, Black children still remained at higher risk for in-hospital mortality even after adjusting for DS-associated comorbidities, underscoring persistent racial disparities. Future studies are needed to clarify the drivers of this gap and to inform targeted, multimodal strategies that improve outcomes for Black children with DS.
Our study has a few limitations. First, readers should be aware of misclassification bias from miscoding, a known limitation inherent to research using administrative databases. Diagnoses that were less relevant to change management during the hospital course might not be accurately documented. For instance, hypothyroidism is commonly seen among children with DS, and if untreated, it may have a significant impact on metabolic and neurodevelopmental outcomes. However, as hypothyroidism rarely leads to acute morbidity and mortality, it might not be reflected in the diagnostic codes. Secondly, we couldn’t distinguish distinct individuals across the study years in KID. This limitation is significant for patients at risk of recurrent hospitalization for respiratory infection or complications as a result of congenital heart diseases or other congenital anomalies. Thus, readers should be aware that an unknown subset of patients may have been admitted more than once, potentially introducing bias. Third, the hospitalization records from the KID database may not accurately reflect the actual racial and ethnic structure of the US population. For instance, the proportion of study population identified as Black is lower than that reported by the US Census (9.9% vs. 13.6%) [40]. This discrepancy could result from selection bias of an inpatient population, the structure of the database (Hispanic was coded as one category of race), and potential disparities in healthcare resource utilization. Lastly, using non-Black children as the comparison group in the analyses could introduce heterogeneity and complicate interpretation. Therefore, we performed additional sensitivity analyses restricting the comparison group to White children. Results from these analyses indicated that Black race remained a significant predictor of both primary and secondary outcomes in the multivariable-adjusted models.
Conclusion
Hospitalized Black children with DS are more likely to be younger, of lower household incomes, with public insurance, and with other underlying comorbidities. Black children had increased risks of mortality and IMV, and increased LOS. Future studies should investigate the underlying factors contributing to these disparities and develop strategies to improve outcomes for children with Down syndrome.
Supplementary Information
Supplementary Material 2: Figure E1. The trend of the mortality rate of Black and White children with Down syndrome hospitalized from year 2000 to year 2019. Schematic illustration of the changes in rates of mortality among hospitalized children with Down syndrome from year 2000 to year 2019. Jonckheere–Terpstra test showed significant changes in trends for all the patients group (p=0.011) and White children (p=0.006), while a non-significant change in trend in mortality for Black children (p=0.453)
Acknowledgements
We thank Dr. Pei-Lun Kuo for his contribution to data and additional support.
Abbreviations
- DS
Down syndrome
- BPD
Bronchopulmonary dysplasia
- IMV
Invasive mechanical ventilation
- NIMV
Non-invasive mechanical ventilation
- LOS
Length of stay
- IACH
Inflation-adjusted cost of hospitalization
- PHTN
Pulmonary hypertension
- CHD
Congenital heart diseases
- SGA
Small of gestational age
- GERD
Gastroesophageal reflux disease
- RSV
Respiratory Syncytial Virus
- OR
Odds ratio
- CI
Confidence interval
- IRR
Incidence rate ratio
Authors’ contributions
Drs. Po-Yang Tsou, had full access to all the data in the study and take responsibility for the integrity of the data and the accuracy of the data analysis. Concept and design: Po-Yang Tsou, and Ignacio Tapia. Drafting of the manuscript: Po-Yang Tsou, Yu-Hsun Wang and Ignacio Tapia. Critical revision of the manuscript for important intellectual content: Ignacio Tapia. Statistical analysis: Po-Yang Tsou. Supervision: Ignacio Tapia.
Funding
Po-Yang Tsou received funding from the American Thoracic Society (ATS) Aspire Fellowship.
Data availability
The data that support the findings of this study are available from the Healthcare Cost and Utilization Project (HCUP) (https://www.hcup-us.ahrq.gov/nisoverview.jsp), but restriction apply to the availability of these data, which were used under licnese for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of HCUP.
Declarations
Ethics approval and consent to participate
KID includes event-level data presented for each admission, and includes diagnostic and procedural codes; the lack of unique identifiers does not allow for identification of repeated admission of the same patients. As all data obtained were deidentified, IRB approval was exempt.
Consent for publication
See response in “Ethics approval and consent to participate”.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Po-Yang Tsou and Yu-Hsun Wang contributed equally as first authors.
References
- 1.Canfield MA, Honein MA, Yuskiv N, Xing J, Mai CT, Collins JS, Devine O, Petrini J, Ramadhani TA, Hobbs CA, et al. National estimates and race/ethnic-specific variation of selected birth defects in the united states, 1999–2001. Birth Defects Res Clin Mol Teratol. 2006;76(11):747–56. [DOI] [PubMed] [Google Scholar]
- 2.Bull MJ, Trotter T, Santoro SL, Christensen C, Grout RW. Genetics TCO: health supervision for children and adolescents with down syndrome. Pediatrics. 2022;149(5). [DOI] [PubMed]
- 3.Leonard S, Bower C, Petterson B, Leonard H. Survival of infants born with down’s syndrome: 1980-96. Paediatr Perinat Epidemiol. 2000;14(2):163–71. [DOI] [PubMed] [Google Scholar]
- 4.Day SM, Strauss DJ, Shavelle RM, Reynolds RJ. Mortality and causes of death in persons with Down syndrome in California. Dev Med Child Neurol. 2005;47(3):171–6. [DOI] [PubMed] [Google Scholar]
- 5.Rasmussen SA, Wong LY, Correa A, Gambrell D, Friedman JM. Survival in infants with Down syndrome, metropolitan Atlanta, 1979–1998. J Pediatr. 2006;148(6):806–12. [DOI] [PubMed] [Google Scholar]
- 6.Yang Q, Rasmussen SA, Friedman JM. Mortality associated with down’s syndrome in the USA from 1983 to 1997: a population-based study. Lancet. 2002;359(9311):1019–25. [DOI] [PubMed] [Google Scholar]
- 7.Boghossian NS, Hansen NI, Bell EF, Stoll BJ, Murray JC, Laptook AR, Shankaran S, Walsh MC, Das A, Higgins RD, et al. Survival and morbidity outcomes for very low birth weight infants with down syndrome. Pediatrics. 2010;126(6):1132–40. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Irving C, Basu A, Richmond S, Burn J, Wren C. Twenty-year trends in prevalence and survival of down syndrome. Eur J Hum Genet. 2008;16(11):1336–40. [DOI] [PubMed] [Google Scholar]
- 9.Shin M, Kucik JE, Correa A. Causes of death and case fatality rates among infants with Down syndrome in metropolitan Atlanta. Birth Defects Res A Clin Mol Teratol. 2007;79(11):775–80. [DOI] [PubMed] [Google Scholar]
- 10.Kucik JE, Shin M, Siffel C, Marengo L, Correa A, Congenital Anomaly Multistate P, Survival C. Trends in survival among children with down syndrome in 10 regions of the united States. Pediatrics. 2013;131(1):e27–36. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hodapp RM, Urbano RC. Demographics of African-American vs. European-heritage mothers of newborns with down syndrome. J Policy Pract Intellect Disabil. 2008;5(3):187–93. [Google Scholar]
- 12.HCUP Kids’ Inpatient Database (KID). Healthcare Cost and Utilization Project (HCUP). 2000, 2003, 2006, 2009, and 2012. Agency for Healthcare Research and Quality, Rockville, MD. www.hcup-us.ahrq.gov/kidoverview.jsp.
- 13.Healthcare Cost and Utilization Project Data Use Agreement Course. Agency for Healthcare Research and Quality, Rockville, MD. https://www.hcup-us.ahrq.gov/DUA/dua_508/DUA508version.jsp.
- 14.Bull MJ. Genetics tCo: health supervision for children with down syndrome. Pediatrics. 2011;128(2):393–406. [DOI] [PubMed] [Google Scholar]
- 15.Tsou PY, Tapia IE. Tracheostomy and inpatient outcomes among children with congenital central hypoventilation syndrome: a kids’ inpatient database study. Pediatr Pulmonol. 2024;59(7):1932–43. [DOI] [PubMed] [Google Scholar]
- 16.Tsou PY, Cielo CM, Xanthopoulos MS, Wang YH, Kuo PL, Tapia IE. The burden of obstructive sleep apnea in pediatric sickle cell disease: a Kids’ inpatient database study. Sleep. 2021. 10.1093/sleep/zsaa157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tsou PY, Hayden LP. Obstructive sleep apnea is associated with use of assisted ventilation among children with bronchopulmonary dysplasia hospitalized with respiratory illness: a nationwide inpatient cohort. Sleep Med. 2023;109:181–9. [DOI] [PubMed] [Google Scholar]
- 18.Tsou PY, Cielo C, Xanthopoulos MS, Wang YH, Kuo PL, Tapia IE. Impact of obstructive sleep apnea on assisted ventilation in children with asthma exacerbation. Pediatr Pulmonol. 2021;56(5):1103–13. [DOI] [PubMed] [Google Scholar]
- 19.Tsou PY, Ruiz-Gutierrez M, Alomari A, Kuo PL, Klouda T. Epidemiology of complications among hospitalized children with hereditary hemorrhagic telangiectasia: a kids’ inpatient database study. Pediatr Pulmonol. 2024;59(5):1507–10. [DOI] [PubMed] [Google Scholar]
- 20.Lewis CW, Carron JD, Perkins JA, Sie KC, Feudtner C. Tracheotomy in pediatric patients: a national perspective. Arch Otolaryngol Head Neck Surg. 2003;129(5):523–9. [DOI] [PubMed] [Google Scholar]
- 21.Perez JM, Melvin PR, Berry JG, Mullen MP, Graham RJ. Outcomes for children with pulmonary hypertension undergoing tracheostomy placement: a multi-institutional analysis. Pediatr Crit Care Med. 2022;23(9):717–26. [DOI] [PubMed] [Google Scholar]
- 22.Qasim A, Dam T, Kim JJ, Valdes SO, Howard T, Diaz MA, Morris SA, Miyake CY. Trends in hospitalization and factors associated with in-hospital death among pediatric admissions with implantable cardioverter defibrillators. J Cardiovasc Electrophysiol. 2022;33(3):502–9. [DOI] [PubMed] [Google Scholar]
- 23.Quiroz HJ, Perez EA, Franklin KN, Willobee BA, Ferrantella AR, Parreco JP, Lasko DS, Thorson CM, Sola JE. Pull-through procedure in children with Hirschsprung disease: a nationwide analysis on postoperative outcomes. J Pediatr Surg. 2020;55(5):899–903. [DOI] [PubMed] [Google Scholar]
- 24.Muir CS, Percy C. Cancer registration: principles and methods. Classification and coding of neoplasms. IARC Sci Publ. 1991;(95):64–81. [PubMed]
- 25.Healthcare Cost and Utilization Project (HCUP). Clinical Classifications Software (CCS) for ICD-9-CM. http://www.hcup-us.ahrq.gov/toolssoftware/ccs/ccs.jsp.
- 26.Johnson DA, Ohanele C, Alcantara C, Jackson CL. The need for social and environmental determinants of health research to understand and intervene on racial/ethnic disparities in obstructive sleep apnea. Clin Chest Med. 2022;43(2):199–216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Forno E, Celedon JC. Health disparities in asthma. Am J Respir Crit Care Med. 2012;185(10):1033–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Isong IA, Rao SR, Bind MA, Avendano M, Kawachi I, Richmond TK. Racial and ethnic disparities in early childhood obesity. Pediatrics. 2018. 10.1542/peds.2017-0865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Derrington TM, Kotelchuck M, Plummer K, Cabral H, Lin AE, Belanoff C, Shin M, Correa A, Grosse SD. Racial/ethnic differences in hospital use and cost among a statewide population of children with down syndrome. Res Dev Disabil. 2013;34(10):3276–87. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Santoro SL, Esbensen AJ, Hopkin RJ, Hendershot L, Hickey F, Patterson B. Contributions to racial disparity in mortality among children with Down syndrome. J Pediatr. 2016;174:240-e246241. [DOI] [PubMed] [Google Scholar]
- 31.Acevedo-Garcia D, Noelke C, McArdle N, Sofer N, Hardy EF, Weiner M, Baek M, Huntington N, Huber R, Reece J. Racial and ethnic inequities in children’s neighborhoods: evidence from the new child opportunity index 2.0. Health Aff (Millwood). 2020;39(10):1693–701. [DOI] [PubMed] [Google Scholar]
- 32.Acevedo-Garcia D, McArdle N, Hardy EF, Crisan UI, Romano B, Norris D, Baek M, Reece J. The child opportunity index: improving collaboration between community development and public health. Health Aff (Millwood). 2014;33(11):1948–57. [DOI] [PubMed] [Google Scholar]
- 33.Ghezzi M, Garancini N, De Santis R, Gianolio L, Zirpoli S, Mandelli A, Farolfi A, D’Auria E, Zuccotti GV. Recurrent respiratory infections in children with Down syndrome: a review. Children (Basel). 2024;11(2): 246. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Santoro SL, Chicoine B, Jasien JM, Kim JL, Stephens M, Bulova P, Capone G. Pneumonia and respiratory infections in down syndrome: a scoping review of the literature. Am J Med Genet A. 2021;185(1):286–99. [DOI] [PubMed] [Google Scholar]
- 35.Alsubie HS, Rosen D. The evaluation and management of respiratory disease in children with Down syndrome (DS). Paediatr Respir Rev. 2018;26:49–54. [DOI] [PubMed] [Google Scholar]
- 36.Beckhaus AA, Castro-Rodriguez JA. Down syndrome and the risk of severe RSV infection: a meta-analysis. Pediatrics. 2018;142(3):e20180225. [DOI] [PubMed] [Google Scholar]
- 37.AAP Recommendations for the Prevention of RSV Disease in Infants and, Children. https://publications.aap.org/redbook/resources/25379/AAP-Recommendations-for-the-Prevention-of-RSV?autologincheck=redirected.
- 38.Caserta MT, O’Leary ST, Munoz FM, Ralston SL, Committee On Infectious D. Palivizumab Prophylaxis in infants and young children at increased risk of hospitalization for respiratory syncytial virus infection. Pediatrics. 2023;152(1). [DOI] [PubMed]
- 39.Frid C, Drott P, Lundell B, Rasmussen F, Anneren G. Mortality in Down’s syndrome in relation to congenital malformations. J Intellect Disabil Res. 1999;43(Pt 3):234–41. [DOI] [PubMed] [Google Scholar]
- 40.Table and US Census Bureau. https://www.census.gov/quickfacts/fact/table/US/PST045222#PST045222.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Material 2: Figure E1. The trend of the mortality rate of Black and White children with Down syndrome hospitalized from year 2000 to year 2019. Schematic illustration of the changes in rates of mortality among hospitalized children with Down syndrome from year 2000 to year 2019. Jonckheere–Terpstra test showed significant changes in trends for all the patients group (p=0.011) and White children (p=0.006), while a non-significant change in trend in mortality for Black children (p=0.453)
Data Availability Statement
The data that support the findings of this study are available from the Healthcare Cost and Utilization Project (HCUP) (https://www.hcup-us.ahrq.gov/nisoverview.jsp), but restriction apply to the availability of these data, which were used under licnese for the current study, and so are not publicly available. Data are however available from the authors upon reasonable request and with permission of HCUP.