Abstract
Background
Stereotactic body radiotherapy (SBRT) is crucial for lung tumor treatment, but facing challenges like intra-fractional anatomical changes and organ risks. Magnetic resonance-guided online adaptive SBRT (MRg-SBRT) is an innovative technique promising safer delivery of ablative doses and protect organ at risk (OAR). This study aimed to investigate the feasibility of the whole process of MRg-SBRT for non-small cell lung cancer (NSCLC) patients.
Methods
Physical parameters of radiotherapy for 23 cases of NSCLC who underwent MRg-SBRT were retrospectively analyzed. This included patients’ treatment course, planned target volume (PTV), target area coverage, and OAR recipient volume. The focus was on inter- and intra-fraction MR real-time monitoring to correct off-target. Local control and adverse event outcomes of patients’ SBRT treatment were also retrospectively analyzed.
Results
All 23 patients completed treatment on time without any treatment interruptions or pauses due to the MR. Tumor movement was predominantly in the superior-inferior (SI) directions through Elekta Unity real-time online monitoring. The baseline plan was altered in four patients, and an adaptive plan was used to correct inter- and intra-fraction off-targeting in a timely manner. The prescribed dose coverage for PTV was 95.3%, with a median bilateral lung volume 20 (V20) GY of 6.3%, and a maximal dose to the spinal cord of 117.3 cGy. The response results showed that the disease control rate (DCR) was 100% with an objective response rate (ORR) of 82.6%. Follow-up results showed an acute grade one to two pneumonia incidence of 82.6% and grade three pneumonias in one patient.
Conclusions
MRg-SBRT can guide treatment plans for the clinical needs of SBRT for lung cancer patients, especially for old patients, proving the feasibility of SBRT for lung lesions with MRg-SBRT, and online real-time monitoring reduces intra- and inter-fraction off-targeting, and guarantees the treatment efficiency of patients without significantly increasing the incidence of serious adverse events.
Keywords: Magnetic resonance-guided radiotherapy (MR-guided radiotherapy), stereotactic body radiotherapy (SBRT), non-small cell lung cancer (NSCLC)
Highlight box.
Key findings
• Magnetic resonance-guided stereotactic body radiotherapy (MRg-SBRT) for non-small cell lung cancer (NSCLC) demonstrated high feasibility, with real-time adaptive planning correcting intra-/inter-fraction motion (4/23 cases). Treatment was completed without MR-related interruptions.
• Excellent disease control (disease control rate was 100% with an objective response rate of 82.6%) and acceptable toxicity (grade 1–2 pneumonitis: 82.6%, grade 3: 4.3%) were achieved.
What is known and what is new?
• SBRT is standard for early-stage NSCLC but limited by tumor motion and organ at risk (OAR) proximity. Conventional imaging lacks real-time soft-tissue tracking.
• This is the first clinical evidence supporting MRg-SBRT’s feasibility for NSCLC, highlighting its ability to mitigate motion errors via adaptive planning and real-time monitoring, without increasing severe toxicity.
What is the implication, and what should change now?
• MRg-SBRT should be prioritized for NSCLC cases with significant motion or high OAR risks. Prospective trials comparing MRg-SBRT vs. conventional SBRT are needed to validate survival/toxicity benefits. Centers with MR-linacs should adopt this approach to enhance precision, while cost-effectiveness studies should guide broader implementation.
Introduction
Lung cancer stereotactic body radiotherapy (SBRT) is a standard treatment for patients with early-stage lung cancer who are inoperable or refuse surgery (1). Hypo-fractionated radiotherapy technology shortens the treatment time, improves patient compliance, and achieves more effective killing of tumor cells (1). During the implementation of hypo-fractionated radiotherapy, the movement of tumors and organs is an important issue that must be considered and solved.
An important technological advancement in the field of radiation therapy has been the introduction of magnetic resonance guided radiotherapy (MRgRT) in recent years (2). Magnetic resonance imaging (MRI) is a popular clinical imaging technique, which obtains proton density layer images by measuring the changes in the energy state of hydrogen atoms before and after the application of radiofrequency pulses in a magnetic field environment. MRI has the advantages of safety and no radiation, high resolution in soft tissues, and multifunctional imaging with multiple sequences, which has a high value of application in diagnosis, treatment monitoring, and prognosis prediction of malignant lesions (2,3). It can guide and optimize radiotherapy through the whole process, online and in real time, at all stages of treatment (before, during and after treatment), so as to realize individualized and precise treatment, which is of great clinical value for SBRT treatment with high fractionated dose (4,5).
However, the presence of magnetic field affects the dose distribution, because of the special characteristics of lung tumors, the presence of more air cavities is more likely to generate scattered electrons at the interfaces of the tissues, which are more susceptible to the influence of magnetic field (6).
Initial study has demonstrated the feasibility and safety of adaptive stereotactic MRgRT for lung cancers and have reported dosimetric and clinical benefits including improved target coverage, organ at risk (OAR) sparing, low toxicity, and promising local control (LC) and progression-free survival (PFS) (7). However, previous study often comprise relatively small and inhomogeneous cohorts (8). In Asia, there are no reports of a large number of cases of well-established SBRT radiotherapy for lung cancer patients treated with Elekta Unity.
Clinical trials of Elekta Unity, which integrates a 1.5T medical diagnostic-grade magnetic resonance device into a 7 MV linear gas pedal, began in 2019 at Shandong Cancer Hospital and Institute, Shandong First Medical University and Shandong Academy of Medical Sciences. Initial enrollment of 3 SBRT patients showed that the dose requirements for SBRT could be met even with magnetic field imaging, but the overall process time was long. Therefore, based on previous studies and the experience of clinical practice in our oncology center, we have summarized the data of patients who underwent SBRT for lung cancer in our radiotherapy department Elekta Unity. From real-time monitoring to reduce inter- and intra-fraction off-targeting, optimization of online processes as well as patient efficacy and radiotherapy-related adverse events were analyzed and summarized in detail in this article to provide theoretical and practical basis for the subsequent realization of MRgRT SBRT individualized radiotherapy for lung cancer patients. We present this article in accordance with the STROBE reporting checklist (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-686/rc).
Methods
Patients enrollment
Retrospective collection of data on patients with non-small cell lung cancer (NSCLC) who received SBRT radiotherapy with Elekta Unity at the Shandong Cancer Hospital and Institute, Shandong First Medical University and Shandong Academy of Medical Sciences from 2021 to 2023.
The inclusion criteria included: (I) patients 18 years or older; (II) patients with histologically confirmed NSCLC; (III) ability to receive radiotherapy within the indications for SBRT treatment; (IV) the patient’s imaging, hematology, and medical records related to SBRT before and after treatment were complete; (V) we enrolled patients with peripheral lung tumors.
Exclusion criteria: (I) metastatic lung cancer diagnosis; (II) patients who underwent routine segmentation guided by MR; (III) lack of imaging data before and after treatment; (IV) consideration of patients with MRI contraindications or those unable to provide written informed consent. This study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the ethics committee of Shandong Cancer Hospital and Institute, Shandong First Medical University and Shandong Academy of Medical Sciences (No. SDTHEC202505047) and individual consent for this retrospective analysis was waived.
Stereotactic MRgRT workflow/delivery of online MR-guided SBRT
Simulation of computed tomography (CT) and MR
The patients were positioned supine, with their hands crossed and securely fixed using a vacuum negative pressure bag or a thermoplastic grid body membrane combined with abdominal compression immobilizer (with pressure scale). Routine CT scanning was performed with a layer thickness of three mm and no interlayer septum, covering the area from the upper edge of the cricothyroid membrane to the middle abdomen. MR simulation: After the patient had completed CT simulation, MR scanning was performed on the MR gas pedal in the same position fixation scheme. (I) Coronal half-Fourier acquisition single-layer excitation fast spin-echo sequence scan, TR/TE =2,000 ms/93 ms; flip angle 150°: FOV =400 mm × 400 mm; 256×320 matrix; three mm layer thickness: 10–30% layer spacing. (II) Conventional transverse 2D-FLASHTW transverse bit scan, TR/TE =7.6 ms/4.5 ms; Acquisition bandwidth: 432.7 HZ; Flip angle 30°; FOV =(360 mm × 360 mm) to (400 mm × 400 mm); 184×256 matrix: AVERAGE two; three mm layer thickness; 20% layer spacing. (III) 2D-TSET2WI transverse axial scan, TR/TE =2,100 ms/206 ms; acquisition bandwidth 1,220.7 HZ: 90° flip angle: FOV =350 mm × 350 mm; 160×224 matrix: AVERAGE1; 3 mm layer thickness: 20% layer spacing. The above images are transmitted to the Monaco planning system for target area and organ at risk mapping. GE advantage four-dimension (4D) software (GE Healthcare, Waukesha, WI, USA) sorted the reconstructed 4D-CT images into ten respiratory phases labelled as 0–90% based on these tags, with 0% corresponding to end inspiration and 50% corresponding to end expiration.
Target delineation and radiotherapy planning
Alignment of MR images and reference CT simulation images acquired in the free-breathing state was performed using the mutual information-based alignment algorithm that comes with the Monaco program system. The alignment scheme used was a rigid alignment based on the bony structure, which was outlined by the same physician in order to avoid inter-investigator variability. In order to ensure the consistency of image contrast, the same window widths and positions were used for all images. The gross tumor volume (GTV) was reconstructed as the internal target volume (ITV) by superimposing the GTVs sketched in 10 phases, and the planning target volume (PTV) was formed by flaring the ITV by 5 mm in the 3D direction.LR, CC, and AP represent the displacement of the center of the GTVs in the inter-subfractionation period. LR stands for the horizontal direction, CC stands for the head and foot direction, and AP stands for the dorsal-abdominal direction.
Treatment planning: fixed-field static intensity-modulated radiation therapy (IMRT) was developed for each patient, utilizing Monaco 5.11.01 to develop a plan based on the Elekta Precise gas pedal data model, while ensuring consistent target area and critical organ volumes. After aligning the MR images acquired on the MR gas pedal with the reference plan CT at each treatment session, the planned PTV and critical organ volumes within each fraction were read off based on the dose distribution of the online plan. Evaluate the SBRT radiation treatment plan with reference to the OAR limits recommended by the consensus definition of SBRT by the American College of Radiology (ACR)/American Society for Radiation Oncology (ASTRO) (9), the related practice guidelines for SBRT, and the guidance from Task Group 101 of the American Association of Physicists in Medicine (AAPM) (10).
Prescribed dose limits
In general, the dose distribution should be normalized so that 95% of the PTV receives at least 100% of the prescribed dose, and for lungs 99% of the PTV should receive at least 90% of the prescribed dose. The maximum dose within the target volume is generally between 110% and 140% of the prescribed dose, the point of maximum dose must be within the PTV, and the volume outside the PTV that exceeds 105% of the prescribed dose is no more than 15% of the PTV volume.
Treatment process
(I) The patients were scanned with MR images before treatment, and the sequence of 3D T2 was selected, which was determined by mapping based on the simulated positioning in the pre-treatment period, and could be used for the display of lung tumors very well. (II) In order to simplify the treatment process and improve the treatment efficiency, the patients were treated in the adaptive position mode, and the online MR images were aligned with the reference CT, and the physicists and radiotherapy doctor jointly decided whether the alignment results met the criteria for clinical application: Each of our patients requires MR-guided real-time positional verification before and during each radiation therapy session. Pre-treatment MR image guidance automatically corrects for posing errors, and after observing 10 complete respiratory time-phases, radiation therapy is initiated if the tumor motion is all within the PTV. (III) Based on the above alignment results, the reference plan was re-optimized by selecting “optimize weight and shape”, using the reference CT electron density for dose calculation, recording the plan optimization time, and having the clinician decide whether the plan meets the clinical needs. If the plan meets the clinical need, online physical quality control of the plan will be performed using the online 2-time dose calculation tool. (IV) During the treatment process, the tumor is observed for off-targeting by real-time MR dynamic sequence images. If the tumor motion of MR real-time images is found to exceed the PTV in any of the three-dimensional directions, radiotherapy is suspended online correction of the target area is performed, the radiotherapy plan is created online, evaluated, and it is verified again that the tumor will not be off-target, and radiotherapy is continued.
All patients were operated on with adapt to position (ATP). If insufficient target coverage and/or violations of OAR constraints, the treatment plan adaptation was performed as online plan adaptation. Online adaptive plans can be (I) optimized based on a baseline plan; and (II) re-optimized and crafted for a new plan. The dosage requirements and OAR limits for the online adaptive program are the same as the initial program requirements and are also subject to quality assurance (QA).
Dosimetric characteristics analysis of stereotactic MRgRT
The statistical dosimetric parameters mainly included the conformity index (CI) of the PTV, the affected lungs and bilateral lungs volume 5 (V5) GY, volume 20 (V20) GY, and Dmean, spinal cord maximal point dose, heart Dmean, and rib maximal point; Dx% denotes the dose corresponding to x% volume; Vy denotes the volume corresponding to receiving Y dose; and Dmean denotes the mean dose.
CI represents the extent to which the prescribed dose wraps around the target area and is defined as follows:
| [1] |
where VCTV, ref denotes the target zone volume of the prescription dose package, VCTV denotes the volume of the target zone, Vref denotes the volume included in the prescription dose, and the larger the CI value the closer it is to 1, indicating the better the dose conformity of the target zone.
Statistical analysis
The primary endpoints of this study were to assess the treatment’s feasibility through off-target and toxicity reports. Secondary endpoints included evaluating LC and PFS. The assessment of treatment response followed the Response Evaluation Criteria in Solid Tumors (RECIST) standard v1.1 criteria (11), categorizing outcomes as complete remission (CR), partial remission (PR), disease stability (SD), and disease progression (PD). Statistical analysis was performed using SPSS 26.0 software.
Results
Patient characteristics
From December 2021 to September 2023, a total of 23 patients diagnosed with NSCLC underwent MR-guided SBRT. The patient characteristics are summarized in Table 1. The median age of all the patients was 73 years, and the median Karnofsky Performance Status (KPS) was 80. All patients had pathologically diagnosed confirmed NSCLC with stage I staging. Four patients had combined epidermal growth factor receptor (EGFR) or anaplastic lymphoma kinase (ALK) gene mutations. Two patients had undergone surgical treatment for lung cancer. All enrolled patients were peripheral lung cancer patients. And there were more comorbidities, mainly diabetes mellitus, coronary heart disease, hypertension and anemia. The Charlson Comorbidity Index (CCI) was relatively high.
Table 1. Patients’ characteristics.
| Characteristics | Value |
|---|---|
| Sex | |
| Male | 17 (73.9) |
| Female | 6 (26.1) |
| Age (years) | 73 [56–85] |
| KPS | |
| <90 | 14 (60.9) |
| ≥90 | 9 (39.1) |
| Previous lung resection | 2 (8.7) |
| Smoking history | |
| Former/current | 15 (65.2) |
| Never | 8 (34.8) |
| EGFR mutation | 2 (8.7) |
| ALK mutation | 2 (8.7) |
| Pathological diagnosis | |
| Squamous | 8 (34.8) |
| Adenocarcinoma | 15 (65.2) |
| Tumor location | |
| Right upper lobe | 9 (39.1) |
| Right middle lobe | 0 (0) |
| Right lower lobe | 5 (21.7) |
| Left upper lobe | 8 (34.8) |
| Left lower lobe | 1 (4.35) |
| T staging | |
| T1 | 17 (73.9) |
| T2 | 6 (26.1) |
| CCI | 5 [4–8] |
| Fraction of SBRT | 5–10 |
| BED of SBRT | 82.5–120 Gy |
Data are presented as n (%), range or median [range]. BED, biologically effective dose; CCI, Charlson Comorbidity Index; KPS, Karnofsky Performance Status; SBRT, stereotactic body radiotherapy; T, tumor.
Radiotherapy parameters
All patients received a total of 186 fractions of SBRT with the biologically effective dose (BED) range from 82.5 to 120 Gy. All patients were treated with fixed-field IMRT. The median of PTV prescription dose coverage volume was 95.3% with the range 95.0–97.8%, with a median PTV of 31.5 cm3 (range: 5.6–115.8 cm3). The average lung dose, bilateral lung V20 GY, average heart dose, and maximum spinal cord dose are provided in Table 2.
Table 2. Physical parameters and OAR of SBRT.
| Dosimetric characteristics | Median | Range | ∆rate after adaptive plan (mean %) |
|---|---|---|---|
| PTV volume, cm3 | 31.5 | 5.6–115.8 | 36.3 |
| V5 of bilateral lungs, % | 19.5 | 8.0–33.2 | 13.1 |
| V10 of bilateral lungs, % | 11.3 | 4.9–22.7 | 12.4 |
| V20 of bilateral lungs, % | 6.3 | 2.6–14.2 | 13.3 |
| V30 of bilateral lungs, % | 3.9 | 1.6–10.9 | 11.8 |
| Mean heart dose, cGy | 94 | 15.7–347 | 1.3 |
| Maximum spinal cord dose, cGy | 1,172.9 | 492.3–3,370.8 | 4.5 |
| CI | 0.65 | 0.61–0.7 | NA |
∆rate after adaptive plan (mean %): the percentage change in physical parameters after the adaptive plan as a percentage of the baseline plan was averaged. CI, conformity index; NA, not applicable; OAR, organ at risk; PTV, planning volume target; SBRT, stereotactic body radiotherapy.
Therapeutic efficiency
The median treatment time of 23 patients was 32 min, which was composed of treatment positioning time and patient preparation time. The MR scan time of the patients was fixed 4 min, the median online planning time was 14.5 min, and the median treatment time with X-ray on was 16 min.
Self-adaptation
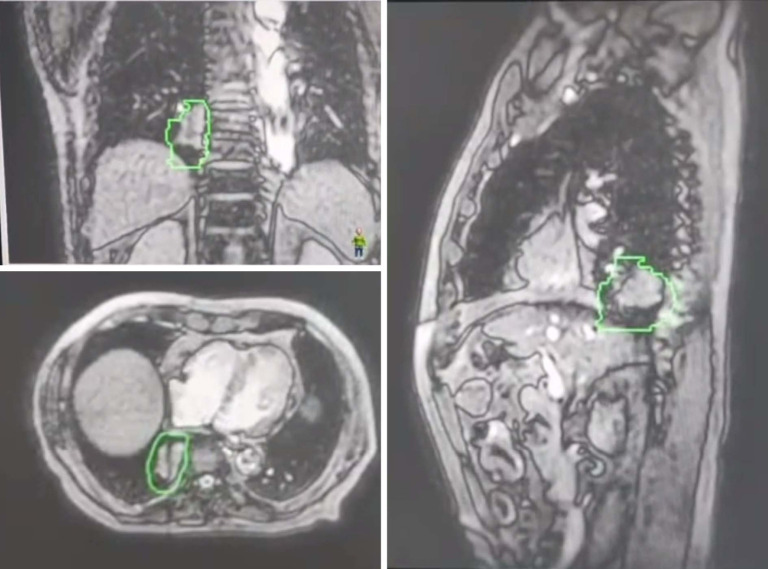
Throughout the treatment course, the adaptability of MR-guided large-segmented radiotherapy allows for adjustments in response to changes in tumor volume, respiratory motion, and other factors, which may deviate from the initial baseline plan. Among the 23 patients, four cases experienced modifications to the baseline plan. Four cases experienced an increase in PTV due to inhomogeneity of respiratory movements between and within radiation therapy fractions in the cranio-caudal direction, aiming to prevent off-target effects. Figure 1 shows a real-time image of MR. At the time of the first radiotherapy in this patient, MR real-time (Figure 1) monitoring identified a small detachment of the target area in the superior-inferior (SI) direction due to uneven respiratory motion in the SI direction, and the tumor border was already a small portion out of the PTV range, which led to an immediate interruption of the treatment and an online adaptive outlining of the target area. After the adaptive planning, MR was used again to perform guidance and found that the tumors were all located within the PTV. The changes of radiotherapy parameters after adaptive plan are summarized in Table 2.
Figure 1.
Real time MR before the first radiation therapy. The green line is the PTV. MR, magnetic resonance; PTV, planning volume target.
Objective response
All the patients completed the treatment successfully, and the short-term curative efficacy was assessed through chest CT within three months after treatment. A short-term efficacy analysis was conducted on all patients, patients of CR: five, PR fourteen; SD four. The ORR of our research is 82.6% and DCR is 100% of three months after SBRT.
Survival
As of the follow-up time of July 15, 2024, we analyzed and counted the recurrence and metastasis of a total of 23 enrolled patients to further analyze the PFS.
A total of three patients experienced disease progression during the follow-up period, including two distant metastases and one localized progression. The median PFS is still not reached. All patients who progressed received further systemic therapy, including immunotherapy and chemotherapy. We also showed the PFS curve in Figure 2.
Figure 2.

Progression free survival of all patients.
Toxic and side effects
Toxicity assessment was conducted following the Common Terminology Criteria for Adverse Events (CTCAE) 5.0 (12). Events within 90 days from the initiation of radiotherapy were classified as acute, while those occurring more than 90 days after the completion of radiotherapy were considered late events. This study presents the observed toxic side effects after MR-guided hypo-fractionated radiotherapy for NSCLC patients. The most common acute adverse event observed through follow-up remained pneumonia, with an 82.6% (19/23) incidence of grade one to two pneumonia and one patient experiencing grade three pneumonia, which recovered with treatment. There was radiotherapy-related chest wall pain in one patient, radiation acute esophagitis in one patient, and no radiotherapy-related gastrointestinal reactions. The highest incidence of long-term adverse events was pulmonary fibrosis, with 16 patients in grade one, four patients in grade two, and no grade three or higher long-term pulmonary fibrosis occurring (Table 3).
Table 3. Acute and late toxicity of SBRT.
| Grade | Acute toxicity | Late toxicity | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Radiation pneumonitis (n) | Radiation esophagitis (n) | Myelosuppression (n) | Gastrointestinal reactions (n) | Radiation dermatitis (n) | Pulmonary fibrosis (n) | Chest pain (n) | Radiation-induced heart disease (n) | Radiation myelitis (n) | ||
| G0 | 3 | 0 | 0 | 0 | 0 | 3 | 0 | 0 | 0 | |
| G1 | 12 | 1 | 1 | 0 | 2 | 16 | 1 | 0 | 0 | |
| G2 | 7 | 0 | 0 | 0 | 0 | 4 | 0 | 0 | 0 | |
| G3 | 1 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
| G4 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | 0 | |
SBRT, stereotactic body radiotherapy.
Discussion
Thoracic cancer is arguably one of the most important treatment sites for MRgRT, as the moving target within a large low-density lung tissue can be more clearly seen and tracked by MRI (13). MRgRT may also enable the potential for daily plan adaptation and margin reduction, which could lead to improved OAR dose sparing (14). Daily plan adaptation could account for anatomical and physiological changes throughout the course of radiotherapy and thereby has the potential to improved osimetric accuracy (15). The “beam-on” capabilities of MRgRT systems permit real-time monitoring during radiotherapy treatment. This may allow for motion mitigation by gating or tracking and therefore again may facilitate the use of smaller margins. However, the thorax is also one of the most challenging sites for MR-linac dose calculation, where the magnetic field significantly influences charged particle transportation in the inhomogeneous lung anatomy (15). To date, most MRgRT clinical experience in thoracic cancer treatments is from SBRT for lung cancer or thoracic oligo-metastases with small samples enrolled or focuses on physical parameters of the radiotherapy program (16).
In this study, we enrolled 23 patients, and confirmed the feasibility of MR-guided SBRT for NSCLC patients. According to the requirements of the SBRT guidelines for lung tumors, MRgRT is able to meet the planning requirements for SBRT of lung lesions through plan optimization, selection of penetration fields, and other techniques. the electron return effect (ERE) and electron stream effect (ESE) effects can be overcome, and the radiotherapy plan does not exceed the limitations for key OARs such as lungs.
Real-time image guidance contributes to more accurate tumor localization and tracking, thereby enhancing treatment precision. This is particularly crucial for tumors like NSCLC that are located relatively deep or challenging to precisely target. In our study, during 4D-CT positioning, it could be observed that there were patients who were very tense, and even through psychological reassurance the patient’s tension could not be relieved. The same tension may be carried over to the first radiotherapy placement, and the tense state of the patient’s body can cause great errors in precise radiotherapy, especially in SBRT treatment. As a result, the PTV error and the tumor target area in the real-time MR monitoring between radiotherapy sessions did not completely coincide, and there were even patients with a very large off-target range, which cannot be solved by ATP. Through the tripartite positioning of physicians, physicists and technicians, we can re-sketch the target area, create and evaluate the online plan, and restart radiotherapy for the patient after passing the test. Meanwhile, during the radiotherapy, as long as the patient can accept the noise of MR, we tried to monitor the whole process of MR in real time. Surprisingly, the respiratory motion within fractions remained inhomogeneous, and this inhomogeneity varied greatly on an individualized basis from patient to patient, especially for lesions located in the lower lobes of both lungs. Real-time monitoring by MR suggests precision and quality control of the whole process of radiotherapy 4D-CT, ITV determination, radiotherapy positioning, and so on, for our patient. Through MR guidance and real-time monitoring, the off-target rate of SBRT in our lung lesions was reduced to zero, which is significant for SBRT radiotherapy with only three to five fractions (one fractional off-target, even if partial, is devastating to the patient’s treatment). The above advantages are especially significant for patients undergoing pulmonary SBRT who are mostly elderly or who cannot tolerate the procedure with their own comorbid cardiopulmonary disease, as these patients may have unattainable requirements for respiratory gating.
Our study showed that almost all patients had acute pneumonia, which may be related to the higher CCI of the patients themselves, suggesting that the patients had more co-morbidities. However, most of them had grade one to two pneumonia and only one patient has grade three acute pneumonia and also recovered from the treatment. For late pulmonary fibrosis also most of them were grade one to two.
In the future, the use of MR-guided hypo-fractionated radiotherapy for NSCLC will present challenges related to equipment costs, technical complexities, and treatment plan development. Consequently, further research is crucial. Firstly, additional optimization is necessary in the research of MR-guided hypo-fractionated radiotherapy plans, including personalized dose adjustments and the integration of new imaging technologies. Secondly, the current cost of MR-guided hypo-fractionated radiotherapy exceeds that of traditional treatments, emphasizing the need for personalized treatment plan selection and formulation as a direction for future research. Our group intends to follow up the advantages and shortcomings of MR-guided SBRT compared with conventional cone beam CT (CBCT), and further enroll centralized patients to enrich clinical experience. In summary, a comparative cohort study between MR-guided hypo-fractionated radiotherapy and traditional radiotherapy methods is essential to further explore their respective advantages and disadvantages in treating NSCLC.
Conclusions
In conclusion, the incorporation of real-time MR imaging for patients, coupled with adaptive planning tools, facilitates immediate treatment decision-making both before and during radiotherapy (inter and intra fraction). MRgRT can fulfill the full process requirements of SBRT with acceptable treatment period. Through the observation of the recent treatment efficacy and side effects in patients, this provides further evidence supporting the feasibility of MR-guided SBRT in the treatment of NSCLC patients.
Supplementary
The article’s supplementary files as
Acknowledgments
We would like to thank Dr. Xin Wang, Dr. Liyang Jiang, Dr. Kaikai Zhao [Department of Radiation Oncology, Shandong Cancer Hospital and Institute, Shandong First Medical University and Shandong Academy of Medical Sciences, Shandong, China] and our nurses’ group for the help of treatment for patients.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. This study was conducted in accordance with the Declaration of Helsinki and its subsequent amendments. The study was approved by the ethics committee of Shandong Cancer Hospital and Institute, Shandong First Medical University and Shandong Academy of Medical Sciences (No. SDTHEC202505047) and individual consent for this retrospective analysis was waived.
Footnotes
Reporting Checklist: The authors have completed the STROBE reporting checklist. Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-686/rc
Funding: The project was supported by China Postdoctoral Science Foundation (grant No. 2022M721446), Shandong Provincial Natural Science Foundation (grant No. ZR2022MH288), Shandong Postdoctoral Science Foundation (grant No. SDCX-ZG-202303064), Wu Jieping Medical Foundation (320.6750.2023-17-26), Xinrui Cancer Foundation (FB2023031673), Shandong Medical Association Clinical Research Foundation (grant No. YXH2022ZX02151), Bethune Translational Research Foundation of Radiation Oncology (grant No. flzh202125), Healthy Science Development Plan of Shandong, China (grant No. 202009030673), Shandong First Medical University Research Project on Education and Teaching (XM2022163), Medical and Health Science and Technology Development Project of Shandong Province (202315021029), Joint TCM Science&Technology Projects of National Demonstration Zones for Comprehensive TCM Reform (GZY-KJS-SD-2023-073), Shandong Traditional Chinese Medicine Science and Technology Program (Z-2023092).
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-686/coif). The authors have no conflicts of interest to declare.
Data Sharing Statement
Available at https://jtd.amegroups.com/article/view/10.21037/jtd-24-686/dss
References
- 1.Shinde A, Li R, Kim J, et al. Stereotactic body radiation therapy (SBRT) for early-stage lung cancer in the elderly. Semin Oncol 2018;45:210-9. 10.1053/j.seminoncol.2018.06.002 [DOI] [PubMed] [Google Scholar]
- 2.Chetty IJ, Doemer AJ, Dolan JL, et al. MRI-guided Radiotherapy (MRgRT) for Treatment of Oligometastases: Review of Clinical Applications and Challenges. Int J Radiat Oncol Biol Phys 2022;114:950-67. 10.1016/j.ijrobp.2022.07.027 [DOI] [PubMed] [Google Scholar]
- 3.Psoroulas S, Paunoiu A, Corradini S, et al. MR-linac: role of artificial intelligence and automation. Strahlenther Onkol 2025;201:298-305. 10.1007/s00066-024-02358-9 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Bryant JM, Weygand J, Keit E, et al. Stereotactic Magnetic Resonance-Guided Adaptive and Non-Adaptive Radiotherapy on Combination MR-Linear Accelerators: Current Practice and Future Directions. Cancers (Basel) 2023;15:2081. 10.3390/cancers15072081 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.van Overeem Felter M, Møller PK, Josipovic M, et al. 1-year efficacy results after MR-guided risk-adapted stereotactic radiotherapy of infra-diaphragmatic oligometastases in a multicenter phase II trial. Radiother Oncol 2025;205:110748. 10.1016/j.radonc.2025.110748 [DOI] [PubMed] [Google Scholar]
- 6.Nardini M, Meffe G, Galetto M, et al. Why we should care about gas pockets in online adaptive MRgRT: a dosimetric evaluation. Front Oncol 2023;13:1280836. 10.3389/fonc.2023.1280836 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Park JM, Wu HG, Kim HJ, et al. Comparison of treatment plans between IMRT with MR-linac and VMAT for lung SABR. Radiat Oncol 2019;14:105. 10.1186/s13014-019-1314-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Regnery S, Katsigiannopulos E, Lau H, et al. How to protect the proximal bronchial tree during stereotactic radiotherapy of ultracentral lung tumors: Lessons from MR-guided treatment. Clin Transl Radiat Oncol 2025;51:100899. 10.1016/j.ctro.2024.100899 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Schneider BJ, Daly ME, Kennedy EB, et al. Stereotactic Body Radiotherapy for Early-Stage Non-Small-Cell Lung Cancer: American Society of Clinical Oncology Endorsement of the American Society for Radiation Oncology Evidence-Based Guideline. J Clin Oncol 2018;36:710-9. 10.1200/JCO.2017.74.9671 [DOI] [PubMed] [Google Scholar]
- 10.Benedict SH, Yenice KM, Followill D, et al. Stereotactic body radiation therapy: the report of AAPM Task Group 101. Med Phys 2010;37:4078-101. 10.1118/1.3438081 [DOI] [PubMed] [Google Scholar]
- 11.Eisenhauer EA, Therasse P, Bogaerts J, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1). Eur J Cancer 2009;45:228-47. 10.1016/j.ejca.2008.10.026 [DOI] [PubMed] [Google Scholar]
- 12.Common Terminology Criteria for Adverse Events (CTCAE) . Available online: https://ctep.cancer.gov/protocolDevelopment/electronic_applications/ctc.htm
- 13.Padgett KR, Simpson GN, Llorente R, et al. Feasibility of Adaptive MR-guided Stereotactic Body Radiotherapy (SBRT) of Lung Tumors. Cureus 2018;10:e2423. 10.7759/cureus.2423 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Finazzi T, Palacios MA, Haasbeek CJA, et al. Stereotactic MR-guided adaptive radiation therapy for peripheral lung tumors. Radiother Oncol 2020;144:46-52. 10.1016/j.radonc.2019.10.013 [DOI] [PubMed] [Google Scholar]
- 15.Ding S, Liu H, Wang B, et al. Impact of field number and beam angle on ERE for lung stereotactic body radiotherapy with 1.5T MR-Linac. Cancer Radiother 2021;25:366-72. 10.1016/j.canrad.2021.01.006 [DOI] [PubMed] [Google Scholar]
- 16.Hering S, Nieto A, Marschner S, et al. The role of online MR-guided multi-fraction stereotactic ablative radiotherapy in lung tumours. Clin Transl Radiat Oncol 2024;45:100736. 10.1016/j.ctro.2024.100736 [DOI] [PMC free article] [PubMed] [Google Scholar]