Abstract
Background
Primary insomnia (PI) is a prevalent sleep disorder that significantly impacts quality of life. While pharmacological treatments are common, concerns about side effects and dependency have led to increased interest in non-pharmacological alternatives. This study systematically evaluates the efficacy and safety of various non-pharmacological therapies for adult PI through a network meta-analysis, providing evidence-based guidance for clinicians.
Methods
We analyzed 53 randomized controlled trials (RCTs) involving 4,181 adults with PI. The included studies assessed 11 non-pharmacological interventions, such as acupuncture, acupressure, cupping therapy, and cognitive behavioral therapy (CBT), alongside control groups (e.g., placebo, waitlist, and pharmacological comparators). Primary outcomes included the Pittsburgh Sleep Quality Index (PSQI), total sleep time (TST), sleep efficiency (SE), and sleep latency (SL). Data synthesis was performed using STATA 17 software with a random-effects model, and evidence quality was appraised using the GRADE framework.
Results
Pooled analyses revealed that all seven non-pharmacological therapies significantly improved PI outcomes compared to controls. Acupuncture reduced PSQI scores by −2.71 points (95% confidence interval (CI): −4.94 to −0.49) versus waitlist, while acupuncture showed a − 1.81 point reduction (95% CI: −2.93 to −0.68). For SE, acupressure and CBT increased SE by 1.48% (95% CI: 0.56–2.39) and 1.34% (95% CI: 0.70–1.98), respectively, compared to SH. Notably, CBT and acupressure shortened SL by approximately 10 min (e.g., CBT: −10.15 min, 95% CI: −11.79 to −8.52 vs. benzodiazepines), while acupressure extended TST by 2.07 h (95% CI: 0.46–3.68). SUCRA rankings identified CBT as the most effective for reducing SL (85.8% probability) and improving SE (89.2%), whereas acupuncture excelled in increasing TST (84.8%). Adverse events were infrequent and mild, primarily limited to transient localized reactions in acupuncture studies.
Conclusion
This study demonstrates that non-pharmacological therapies are effective and safe in managing PI, with CBT, acupuncture, and acupressure emerging as optimal choices for specific sleep parameters. These findings advocate non-pharmacological interventions into clinical practice and offer clinicians valuable insights for selecting appropriate treatment modalities for PI management. However, study limitations like heterogeneity and small sample sizes highlight the need for larger, well-designed RCTs. Future studies should use standardized measures for more specific insomnia assessment.
Keywords: primary insomnia, cognitive behavioral therapy, acupuncture, acupressure, cupping therapy
1. Introduction
Primary insomnia (PI) is a common sleep disorder characterized by difficulty falling asleep, maintaining sleep, or early morning awakenings, occurring at least three times per week for a minimum of 3 months. These symptoms are frequently accompanied by clinical significant distress and daytime functional impairments, including irritability, fatigue, and decreased productivity. With a global prevalence reaching 22.1% (1), PI substantially compromises health-related quality of life worldwide (2). Although earlier guidelines recommended pharmacological like benzodiazepine (3, 4), their long-term use is limited by adverse effects such as memory impairment, psychological disturbances, and elevated risk of depression/anxiety. Consequently, non-pharmacological interventions like cognitive behavioral therapy (CBT) were historically regarded as adjunctive options.
Recent paradigm shifts are evident in updated guidelines. The European Sleep Research Society now designates non-pharmacological therapies (particularly CBT) as first-line PI treatments, reserving short-term pharmacotherapy only for refractory cases (5). This reflects growing recognition of non-drug approaches as primary management strategies.
Among these interventions, CBT demonstrates robust efficacy in improving psychological outcomes and sleep efficiency (6). Acupuncture, another evidence-based modalities, exerts therapeutic effects through neuroendocrine modulation [e.g., melatonin regulation (7), and autonomic nervous system regulation sympathetic suppression/parasympathetic enhancement (8)]. Clinical study confirms acupuncture’s particular effectiveness in prolonging sleep duration (9). Notably, insomnia severity positively correlates with patients’ pursuit of physical therapies (10). However, existing randomized controlled trials (RCTs) on non-pharmacological treatments frequently exhibit methodological limitations—including small samples and placebo effects—compromising conclusions about their efficacy and safety profiles.
Epidemiological trends reveal increasing PI prevalence among younger populations (11). Adolescents, for instance, average merely 6.25 h of weekday sleep, with 65% experiencing >30-min sleep latency (SL). Despite this demographic shift, most meta-analyses disproportionately focus on elderly cohorts, neglecting younger age groups (12).
To address these gaps, this study conducts a network meta-analysis of RCTs evaluating non-pharmacological PI therapies. By systematically comparing efficacy and safety of various non-pharmacological treatments in adult, this study aims to establish evidence-based clinical recommendations for optimal therapeutic selection.
2. Materials and methods
2.1. Literature search
Two researchers independently performed systematic searches across eight databases (China National Knowledge Infrastructure, VIP, Wanfang, SinoMed, PubMed, EMBASE, Cochrane Library, and Web of Science) from inception through December 24, 2023. The search strategy combined subject terms and free-text keywords using the following terms: (primary insomnia) AND (acupuncture OR electroacupuncture OR acupressure OR cupping OR physical therapy OR cognitive behavioral therapy) AND (randomized controlled trial). The detailed search strategy is presented in Supplementary Tables S1–S8. Manual searches of reference lists from eligible RCTs and meta-analyses supplemented electronic retrieval.
2.2. Inclusion criteria
(1) Study design: Peer-reviewed RCTs published in Chinese or English.
(2) Participants: Adults (≥18 years) meeting DSM-IV-TR criteria (American Psychiatric Association) for PI (13), supplemented by the Chinese guidelines for adult insomnia diagnosis (14). Although DSM-5 represents the current standard, we retained DSM-IV-TR criteria because: (a) 77% of included trials used this framework; (b) core PI diagnostic features remained consistent across editions; and (c) this maintained methodological alignment with most included studies. ICSD-3 criteria (American Academy of Sleep Medicine, 2014) were additionally extracted when reported.
(3) Interventions: Eleven comparators were evaluate: acupuncture, acupressure, cupping therapy (CUP), CBT, sleep hygiene (SH), repetitive transcranial magnetic stimulation (rTMS), relaxation therapy (RT), placebo (sham acupuncture or sham treatment) (PLA), nenzodiazepines (BZD), non-benzodiazepines (NBZD), and waiting list control (Wait List).
(4) Outcome measures: Primary outcomes included Pittsburgh Sleep Quality Index (PSQI), total sleep time (TST), sleep efficiency (SE), and sleep latency (SL), selected based on: (1) consistent reporting in most included studies, and (2) validated psychometric properties in insomnia research. The PSQI is a well-validated self-report questionnaire designed to assess sleep quality over a one-month period, comprising seven components (each scored 0–3; total range: 0–21), with higher scores indicating poorer sleep quality (15). Key sleep parameters, including TST, SE, and SL, were derived from objective measures—either polysomnography (the gold standard for sleep assessment) or actigraphy (a wearable-based method estimating sleep parameters via movement detection)—where available. In cases where objective data were lacking, validated sleep diaries served as an alternative for self-reported TST, SE, and SL. SE was defined as the ratio of actual sleep duration to time spent in bed, while SL represented the duration from lights-out to sleep onset, and TST referred to the total sleep duration per night from sleep initiation to awakening. Although wake after sleep onset (WASO) and early morning awakening are clinically relevant to insomnia, these metrics were excluded from the primary analysis due to inconsistent reporting (available in fewer than 40% of studies) and significant methodological variability across trials.
While the PSQI encompasses a broad range of sleep-related domains—including environmental disturbances and physical symptoms (e.g., nocturnal breathing irregularities)—it remains a widely accepted tool in insomnia research, demonstrating strong correlations with insomnia severity. However, to improve diagnostic specificity, future studies may consider supplementing the PSQI with the Insomnia Severity Index (ISI), which specifically targets core insomnia symptoms, such as difficulties in sleep initiation, maintenance, and associated daytime impairment, thereby enhancing clinical sensitivity and precision in outcome assessment.
2.3. Exclusion criteria
(1) Study types: Excluded studies included case reports, retrospective studies, animal experiments, reviews, cell experiments, and experience summaries.
(2) Participants: Studies were excluded if (a) the diagnostic criteria were absent or unclear, or (b) the participants had concomitant psychiatric disorders or other organic diseases.
(3) Interventions: Studies comparing known effective treatments with experimental therapies were excluded.
(4) Outcome measures: Studies were excluded if (a) they lacked outcome measures or (b) the outcome data could not be obtained, and the authors could not be contacted.
2.4. Literature screening and data extraction
Two researchers independently screened all retrieved records from electronic databases, including clinical trials and the references of relevant systematic reviews or meta-analyses, based on predefined eligibility criteria. Duplicate records and studies available only as abstracts were excluded. Disagreements between researchers were resolved through discussion or, if necessary, by consultation with a third independent researcher.
2.5. Risk of Bias assessment
To assess methodological quality, two researchers independently evaluated the included studies using the Cochrane Risk of Bias (ROB) 2.0 tool (16). This updated version addresses limitations in assessing study design and baseline group comparability by refining evaluation criteria and eliminating ambiguous terminology. ROB 2.0 examines multiple bias domains, including randomization, allocation concealment, baseline imbalances, deviations from intended interventions, missing data, outcome measurement, and selective reporting. Each bias is rated as “low,” “high,” or “unclear.” If disagreements occurred, a third researcher would be consulted to make the final decision.
2.6. Statistical analysis
All analyses were performed using STATA 17 software, with results visualized using relationship plots. For safety outcomes, random-effects models were applied to estimate relative risk (RR) with 95% confidence interval (CI). Treatment effects were quantified as mean differences (MD) or standardized MD, depending on data compatibility. Heterogeneity was evaluated using the I2 statistic, categorized as low (I2 < 30%), moderate (I2, 30–50%), or high (I2 > 50%). Sensitivity analyses were conducted to test result robustness.
For primary outcomes, a network meta-analysis was performed to map associations between interventions. Node sizes reflected participant numbers per intervention, while connecting line widths represented the number of trials comparing paired interventions. Direct and indirect evidence were synthesized to assess comparative effectiveness and safety, expressed as RR with 95% CI. Intervention rankings were derived using the Surface under the Cumulative Ranking (SUCRA) curve, where higher SUCRA values indicated superior efficacy. Ranking probabilities were computed cumulatively to generate a hierarchy of interventions. All tests were two-tailed, with statistical significance set at p < 0.05.
3. Results
3.1. Literature search results
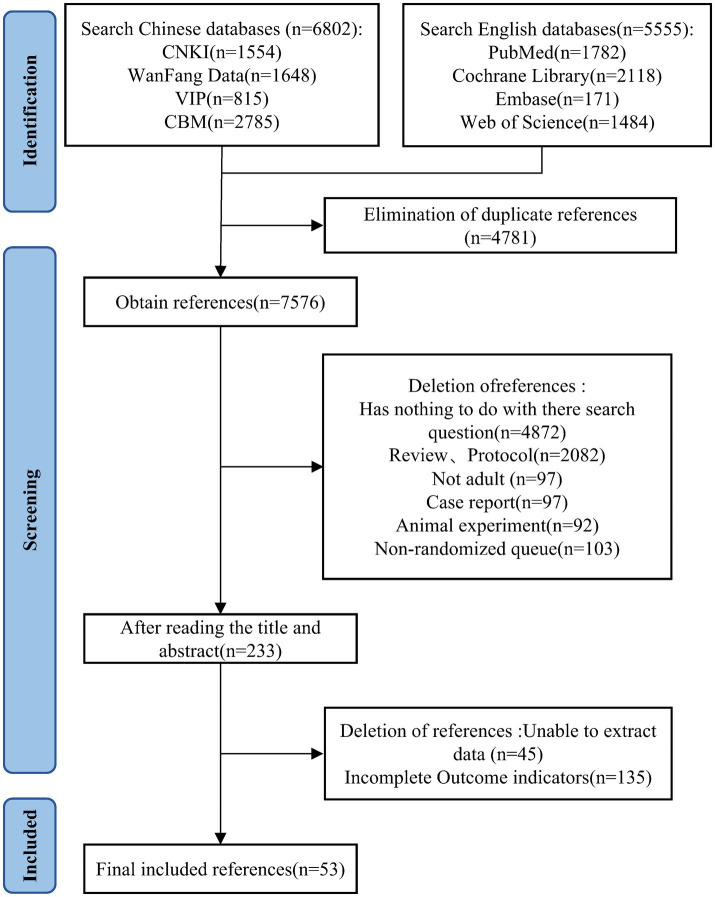
A total of 12,357 studies were systematic retrieved during the literature search. After removing duplicates using Endnote 20, 7,576 records remained. By screening titles and abstracts, 7,343 articles were excluded. A further 180 articles were excluded after full-text evaluation, resulting in the final inclusion of 53 studies (17–69). The detailed literature search process is illustrated in Figure 1.
Figure 1.
Flowchart of study selection.
3.2. Basic characteristics of the included studies
The final analysis included 53 RCTs (total N = 4,181 participants), comprising 46 two-arm and 7 three-arm trials (17–69). These studies evaluated 11 interventions: acupuncture, acupressure, CBT, RT, CUP, SH, rTMS, BZD, NBZD, PLA, and Wait List. Detailed study characteristics are presented in Table 1.
Table 1.
Characteristics of included studies.
| Study | Sample size (T/C) | Age (year, mean ±SD; or range) | Gender, Female (%) | Treatment | Control | Follow-up (w) |
Outcome | PI duration (year) |
|---|---|---|---|---|---|---|---|---|
| Cao et al. (17) | 36/36 | 38.5 ± 14 | 0.56 | AC | PLA | 4w | SE, TST | 1.2 |
| Gao and Luo (18) | 22/18 | 44.67 ± 11.26 | 0.55 | AC | BZD | 4w | PSQI | 5.3 |
| Li et al. (19) | 30/30 | 18–70 | NA | AC | BZD | 4w | PSQI | NA |
| Zhang et al. (20) | 33/33 | 51.7 ± 2.3 | 0.59 | CBT | NBZD | 6 M | PSQI | NA |
| Zhao et al. (21) | 45/45 | 43.97 ± 9.32 | 0.6 | AC | BZD | 4w | PSQI | 1 |
| Xuan et al. (22) | 24/22 | 48.96 ± 11.96 | 0.54 | AC | BZD | 4w | PSQI, TST | 5.31 |
| Li et al. (23) | 20/20 | 50.0 ± 3.49 | 0.55 | AC | PLA | 4w | PSQI | 0.72 |
| 20/20 | 50.0 ± 3.00 | 0.58 | BZD | PLA | 4w | PSQI | 0.72 | |
| 20/20 | 50.0 ± 3.49 | 0.58 | AC | BZD | 4w | PSQI | 0.72 | |
| Jiang (24) | 60/60 | 18–75 | NA | RT | NBZD | 4w | PSQI | NA |
| Feng et al. (25) | 43/37 | 53.57 ± 16.56 | 0.68 | rTMS | PLA | 1w | PSQI | 0.69 |
| Tu et al. (26) | 19/14 | 44.56 ± 8.05 | 0.55 | AC | PLA | 4w | PSQI | NA |
| Jespersen et al. (27) | 19/19 | 51.13 ± 9.6 | 0.79 | RT | PLA | 4w | PSQI | NA |
| Jacobs et al. (28) | 15/15 | 46.85 ± 8.9 | 0.70 | RT | PLA | 8w | SL, TST, SE | 9.55 |
| 15/15 | 46.00 ± 9.56 | 0.73 | NBZD | PLA | 8w | SL, TST, SE | 9.35 | |
| 15/15 | 46.25 ± 8.61 | 0.70 | RT | NBZD | 8w | SL, TST, SE | 10 | |
| Edinger et al. (29) | 25/25 | 55.75 ± 10.77 | 0.80 | CBT | PLA | 6 W | TST, SE | 13.9 |
| 25/25 | 55.1 ± 9.77 | 0.80 | RT | PLA | 7 W | TST, SE | 14 | |
| 25/25 | 55.15 ± 11.1 | 0.73 | CBT | RT | 8 W | TST, SE | 13.1 | |
| Siu et al. (30) | 105/110 | 67.3 ± 6.8 | 0.8 | RT | PLA | 12 W | PSQI | 10.37 |
| Yin et al. (31) | 36/36 | 38.5 ± 14.0 | 0.56 | AC | PLA | 4 W | TST | NA |
| Lee et al. (32) | 46/45 | 51.94 ± 8.8 | 0.75 | AC | PLA | 12 W | PSQI, TST, SE, SL | 6.31 |
| Ritterband et al. (33) | 22/22 | 44.86 ± 11.03 | 0.77 | CBT | PLA | 9 W | TST, SE, SL | 10.59 |
| Yeung et al. (34) | 30/30 | 48.0 ± 9.0 | 0.77 | AC | PLA | 3 W | PSQI, TST, SE, SL | 9.3 |
| Vanstraten et al. (35) | 59/59 | 49.4 ± 12.9 | 0.70 | CBT | PLA | 6 W | SE, SL | 11.8 |
| Scharf et al. (36) | 221/214 | 45.42 ± 11.28 | 0.61 | BZD | PLA | 3 M | TST, SL | NA |
| Feng et al. (37) | 17/17 | 38.15 ± 10.83 | 0.65 | CUP | PLA | 4 W | PSQI | 3.97 |
| 16/17 | 41.15 ± 10.8 | 0.58 | BZD | PLA | 4 W | PSQI | 3.97 | |
| 17/16 | 39.49 ± 10.92 | 0.58 | CUP | BZD | 4 W | PSQI | 3.97 | |
| Yeung et al. (38) | 70/70 | 42.1 ± 13.0 | 0.80 | AC | PLA | 5 W | TST, SE, SL | 5.3 |
| Khalsa and Goldstein (39) | 20/20 | 42.15 ± 10.55 | 0.63 | RT | SH | 8 W | PSQI, TST, SE, SL | NA |
| Riemann et al. (40) | 18/18 | 47.02 ± 10.73 | 0.42 | BZD | PLA | 4 W | PSQI, TST, SE, SL | NA |
| Passos et al. (41) | 12/12 | 44.4 ± 8 | 0.79 | RT | PLA | 24H | TST, SE, SL | 9.1 |
| Lo et al. (42) | 14/13 | 56.9 ± 4.66 | 1.00 | AC | PLA | 3 W | PSQI, TST, SE, SL | 16.8 |
| Morin et al. (43) | 18/18 | 64.65 ± 7.20 | 0.69 | CBT | PLA | 8 W | TST, SE | 16.6 |
| 17/18 | 64.51 ± 6.68 | 0.71 | NBZD | PLA | 8 W | TST, SE | 17 | |
| 18/17 | 64.25 ± 6.89 | 0.63 | CBT | NBZD | 8 W | TST, SE | 15.8 | |
| Wu et al. (44) | 19/17 | 38.8 ± 12.1 | 0.53 | CBT | PLA | 8 W | TST, SE, SL | 0.5 |
| 17/17 | 38.8 ± 12.1 | 0.53 | NBZD | PLA | 8 W | TST, SE, SL | 0.5 | |
| 19/17 | 38.8 ± 12.1 | 0.53 | CBT | NBZD | 8 W | TST, SE, SL | 0.5 | |
| Fu et al. (45) | 37/37 | 52.5 ± 5.56 | 1.00 | AC | PLA | 3 W | TST, SE, SL | 0.25 |
| Espie et al. (46) | 107/94 | 54.26 ± 14.9 | 0.68 | CBT | PLA | 2 W | TST, SE, SL | 0.5 |
| Abedian et al. (47) | 37/36 | 51 ± 4.44 | 1.00 | AP | PLA | 4 W | PSQI, SE, SL | 1 |
| Huang et al. (48) | 18/18 | 45.08 ± 11.09 | 0.50 | PT | PLA | 10d | PSQI | 0.25 |
| Tang et al. (49) | 38/38 | 45 ± 9.98 | 0.66 | AP | BZD | 1 M | PSQI | 1.67 |
| Wang and Zhou (50) | 35/35 | 53.98 ± 4.81 | 0.57 | AP | AC | 4 W | PSQI | 4.31 |
| Lin et al. (51) | 46/44 | 67.05 ± 6.28 | 0.50 | AC | CBT | 8 W | PSQI | 4.8 |
| Wang et al. (52) | 33/30 | 73 ± 6 | 0.59 | AC | BZD | 4 W | PSQI | 1.2 |
| Zhang et al. (53) | 30/30 | 78.07 ± 2.98 | 0.42 | RT | Wait List | 8 W | PSQI | 5.43 |
| Xu et al. (54) | 27/27 | 68.15 ± 7.25 | 0.44 | CBT | BZD | 4 W | PSQI | 6.51 |
| Liang (55) | 35/35 | 68 ± 6 | 0.57 | AC | BZD | 1 M | PSQI | 5.41 |
| Yu and Gao (56) | 28/28 | 71.8 ± 5.25 | 0.52 | AC | BZD | 4 W | PSQI | 4.71 |
| Christina et al. (57) | 11/9 | 77.2 ± 8 | 0.65 | CBT | SH | 1 W | TST, SE, SL | 10.6 |
| Edinger et al. (58) | 20/20 | 54.2 ± 13.7 | 0.125 | CBT | SH | 2 W | TST, SE, SL | 10 |
| Espie et al. (59) | 55/55 | 49 ± 13.5 | 0.73 | CBT | PLA | 6 W | TST, SE, SL | 5.62 |
| Jernelov et al. (60) | 44/43 | 47.9 ± 13.9 | 0.82 | CBT | Wait List | 1 W | TST, SE, SL | 11.8 |
| Kaldo et al. (61) | 73/75 | 48.01 ± 15.38 | 0.78 | CBT | PLA | 8 W | TST, SE, SL | 10.5 |
| Lichstein et al. (62) | 27/23 | 68.08 ± 6.98 | 0.72 | RT | PLA | 2 W | TST, SE, SL | 9.17 |
| Lovato et al. (63) | 86/32 | 63.76 ± 6.45 | 0.5 | CBT | Wait List | 4 W | TST, SE, SL | 5 |
| Sivertsen et al. (64) | 9/8 | 60.51 ± 5.54 | 0.38 | CBT | NBZD | 2 W | TST, SE, SL | 14.81 |
| 9/6 | 60.6 ± 4.53 | 0.53 | CBT | PLA | 2 W | TST, SE, SL | 14.28 | |
| 8/6 | 61.51 ± 5.94 | 0.54 | NBZD | PLA | 2 W | TST, SE, SL | 12.97 | |
| Wei et al. (65) | 40/40 | 47.53 ± 12.71 | 0.65 | AP | BZD | 3 W | PSQI | 2.1 |
| Wei et al. (66) | 40/40 | 40.15 ± 5.19 | 0.69 | AP | BZD | 10D | PSQI | 0.17 |
| Yang et al. (67) | 31/32 | 46.51 ± 12.43 | 0.63 | AP | BZD | 2 W | PSQI | 5.8 |
| Zhuang (68) | 50/50 | 48.49 ± 2.26 | 0.45 | AP | BZD | 3 W | PSQI | 4.03 |
| Wei et al. (69) | 30/30 | 47.23 ± 12.58 | 0.53 | AP | BZD | 12D | PSQI | 2.2 |
T, treatment group; C, control group; w, week; PSQI, Pittsburgh Sleep Quality Index; TST, Total Sleep Time; SE, Sleep Efficiency; SL, Sleep Latency; AC, Acupuncture; CBT, Cognitive Behavioral Therapy; AP, Acupressure; RT, Relaxation Therapy; CUP, Cupping Therapy; SH, Sleep Hygiene; rTMS, Repetitive Transcranial Magnetic Stimulation; BZD, Benzodiazepines; NBZD, Non-benzodiazepine Drugs; PLA, Placebo (sham acupuncture or sham treatment); Wait List, waiting list. ICD, Diagnosis Systems: International Classification of Diseases; DSM, Diagnostic and Statistical Manual of Mental Disorders; NA, Not Available.
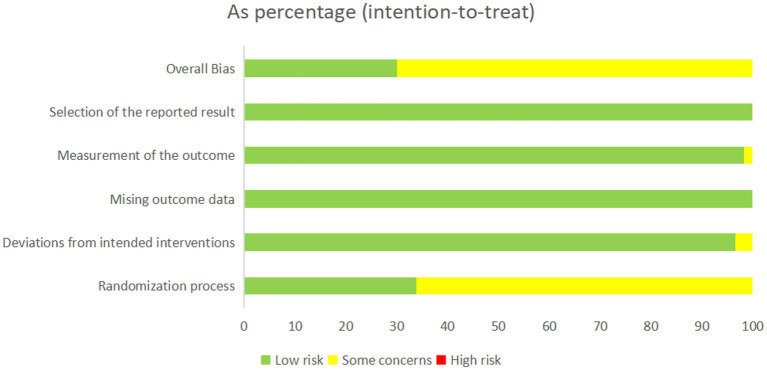
3.3. Risk of Bias assessment using ROB 2.0
Study quality was evaluated using the Cochrane ROB 2.0 tool. In the randomization domain, 18 studies (33.9%) demonstrated low risk of bias, whereas 35 studies (66.1%) lacked clearly allocation concealment descriptions. All trials showed high adherence to protocol (low risk of deviation bias). Complete outcome data were available for all 53 studies, with appropriate measurement methods for primary outcomes using both subject and objective measures. No studies exhibited high risk of measurement bias. Selective reporting bias was minimal (16 studies, 30.1% low risk; 30 studies, 69.9% potential risk), as detailed in Figure 2 and Supplementary Table S9.
Figure 2.
Risk of bias assessment using ROB 2.0 tool.
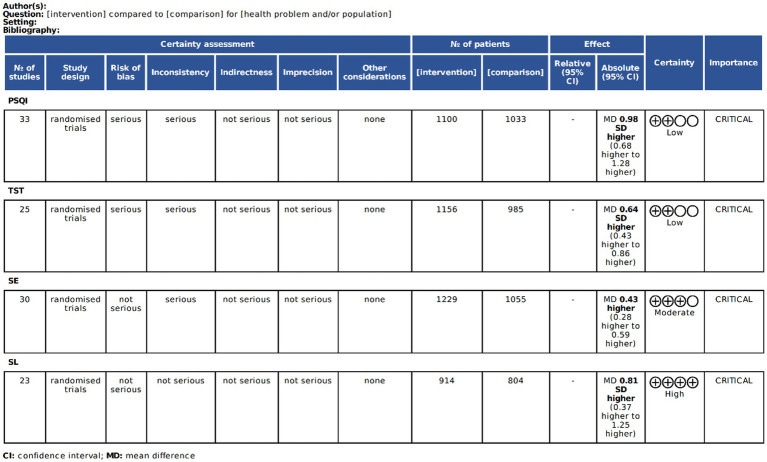
3.4. Evidence quality assessment
The GRADE system was applied to evaluate evidence quality for all primary outcomes. Final rating reflect comprehensive quality assessments across included studies (17–69) (Figure 3).
Figure 3.
Results of evidence assessment using GRADE quality.
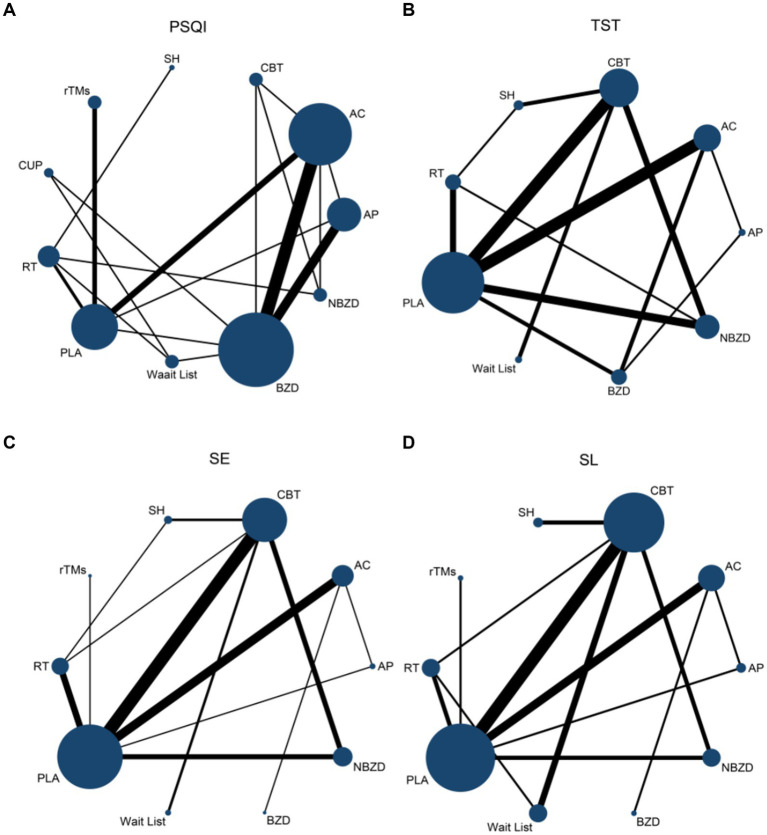
3.5. Pairwise meta-analysis results
The pairwise meta-analysis results for PSQI, TST, SE, and SL are presented in the Supplementary Figures S1–S4, and Figure 4. Compared to PLA and the Wait List, both BZD (MD = −1.25, 95% CI: −2.48 to −0.02; MD = −1.90, 95% CI: −3.97 to 0.16) and NBZD (MD = −1.06, 95% CI: −2.95 to 0.83; MD = −1.71, 95% CI: −4.20 to 0.78) significantly reduced PSQI scores. Additionally, significant SE improvements were also observed for acupressure (MD = 1.27, 95% CI: 0.37 to 2.18; MD = 1.74, 95% CI: 0.69 to 2.80), acupuncture (MD = 0.88, 95% CI: 0.15 to 0.60; MD = 1.35, 95% CI: 0.51 to 2.20), CBT (MD = 1.14, 95% CI: 0.55 to 1.73; MD = 1.61, 95% CI: 0.67 to 2.55), and BZD (MD = 0.85, 95% CI: 0.10 to 1.59; MD = 1.32, 95% CI: 0.31 to 2.33), with CBT additionally demonstrating significant SL reduction (MD = −0.58, 95% CI: −0.85 to −0.31; MD = −0.63, 95% CI: −0.98 to −0.68).
Figure 4.
Network evidence map of pairwise comparison on PSQI (A), TST (B), SE (C), and SL (D). PSQI, Pittsburgh Sleep Quality Index; TST, Total Sleep Time; SE, sleep efficiency; SL, sleep latency; AC, Acupuncture; CBT, Cognitive Behavioral Therapy; AP, Acupressure/Massage; RT, Relaxation Therapy; CUP, Cupping Therapy: SH, Sleep Hygiene; rTMS, Repetitive Transcranial Magnetic Stimulation; BZD, Benzodiazepines; NBZD, Non-Benzodiazepines; PLA, Placebo (sham acupuncture or sham treatment); Wait List, Waiting List.
Compared to SH, acupressure (MD = 1.48, 95% CI: 0.56 to 2.39; MD = −1.31, 95% CI: −2.20 to −0.42), CBT (MD = 1.34, 95% CI: 0.70 to 1.98; MD = −1.34, 95% CI: −2.02 to −0.66), CUP (MD = 0.82, 95% CI: 0.15 to 1.50; MD = −1.19, 95% CI: −2.00 to −0.37), and BZD (MD = 1.05, 95% CI: 0.28 to 1.59; MD = −1.32, 95% CI: −2.18 to −0.45) were all effective in improving SE and reducing SL. When compared to RT, acupressure (MD = 0.86, 95% CI: 0.23 to 1.49; MD = −0.55, 95% CI: −1.07 to −0.03) improved SE and shortened SL, while acupuncture showed a significant improvement in SE (MD = 0.47, 95% CI: 0.15 to 0.79), and CBT effectively shortened SL (MD = −0.58, 95% CI: −0.85 to −0.31).
Compared to the Wait List, CUP showed a significant improvement in SE (MD = 1.09, 95% CI: 0.11 to 2.07). Additionally, acupuncture (MD = −9.58, 95% CI: −11.15 to −8.00), acupressure (MD = −10.12, 95% CI: −11.78 to −8.46), CUP (MD = −10.00, 95% CI: −11.67 to −8.33), rTMS (MD = −9.65, 95% CI: −11.39 to −7.90), BZD (MD = −10.13, 95% CI: −11.83 to −8.43), PLA (MD = −9.52, 95% CI: −11.19 to −7.85), and RT (MD = −9.57, 95% CI: −11.19 to −7.56) were all effective in reducing SL. Moreover, acupuncture significantly extended TST compared to PLA, and shortened SL compared to acupressure and CBT.
3.6. Network meta-analysis findings
The network meta-analysis evaluated four efficacy indicators combining objective and subjective sleep measures. Thirty-two studies (n = 2,114) assessed PSQI scores, 25 studies (n = 2,451) measured TST, while SE and SL were evaluated in 2,284 and 1,718 patients, respectively (Table 2, panel A; Table 3, panel B).
Table 2.
Results of network meta-analysis (Panel A).
| AP | 0.63 (−2.41,3.66) |
1.35 (−2.10,4.80) |
1.63 (−2.12,5.38) |
– | – | 1.55 (−2.06,5.15) |
3.17 (−0.30,6.64) |
1.16 (−2.42,4.74) |
1.83 (−1.41,5.08) |
1.61 (−2.45,5.67) |
|---|---|---|---|---|---|---|---|---|---|---|
| −0.26 (−1.39,0.87) |
AC | 0.73 (−0.91,2.36) |
1.00 (−1.20,3.21) |
– | – | 0.92 (−1.02,2.87) |
2.54
(0.86,4.22) |
0.53 (−1.37,2.43) |
1.21
(0.05,2.36) |
0.99 (−1.71,3.68) |
| −0.39 (−2.26,1.48) |
−0.14 (−1.78,1.51) |
CBT | 0.28 (−1.55,2.11) |
– | – | 0.20 (−1.60,2.00) |
1.82 (−0.20,3.84) |
−0.19 (−1.70,1.32) |
0.48 (−0.68,1.64) |
0.26 (−1.88,2.40) |
| −1.79 (−5.02,1.44) |
−1.53 (−4.66,1.59) |
−1.40 (−4.80,2.00) |
SH | – | – | −0.08 (−2.13,1.97) |
1.54 (−0.96,4.04) |
−0.47 (−2.67,1.73) |
0.21 (−1.67,2.08) |
−0.02 (−2.83,2.80) |
| −1.40 (−3.44,0.65) |
−1.14 (−3.02,0.74) |
−1.00 (−3.43,1.42) |
0.39 (−3.01,3.80) |
rTMs | – | – | – | – | – | – |
| −0.36 (−3.02,2.31) |
−0.10 (−2.70,2.50) |
0.03 (−2.93,3.00) |
1.43 (−2.41,5.27) |
1.04 (−2.05,4.13) |
CUP | – | – | – | – | – |
| −1.38 (−3.23,0.48) |
−1.12 (−2.79,0.55) |
−0.98 (−3.12,1.15) |
0.41 (−2.23,3.06) |
0.02 (−2.13,2.17) |
−1.02 (−3.80,1.77) |
RT | 1.62 (−0.66,3.89) |
−0.39 (−2.34,1.56) |
0.29 (−1.28,1.85) |
0.06 (−2.73,2.86) |
| −0.81 (−1.76,0.13) |
−0.56 (−1.37,0.26) |
−0.42 (−2.09,1.25) |
0.98 (−2.17,4.12) |
0.58 (−1.36,2.53) |
−0.46 (−2.96,2.05) |
0.56 (−1.14,2.27) |
BZD | −2.01 (−4.25,0.23) |
−1.33 (−2.98,0.32) |
−1.56 (−4.50,1.39) |
| −1.00 (−2.98,0.98) |
−0.75 (−2.48,0.99) |
−0.61 (−2.51,1.29) |
0.79 (−2.46,4.04) |
0.39 (−2.02,2.81) |
−0.64 (−3.63,2.34) |
0.37 (−1.52,2.27) |
−0.19 (−2.01,1.63) |
NBZD | 0.68 (−0.83,2.18) |
0.45 (−2.17,3.07) |
|
−2.06
(−3.45,-0.68) |
−1.81
(−2.93,-0.68) |
−1.67 (−3.57,0.22) |
−0.27 (−3.32,2.78) |
−0.67 (−2.17,0.84) |
−1.71 (−4.41,0.99) |
−0.69 (−2.22,0.84) |
−1.25
(−2.48,-0.02) |
−1.06 (−2.95,0.83) |
PLA | −0.22 ( −2.66,2.21) |
| −2.71 (−4.94,-0.49) |
−2.46 (−4.58,-0.34) |
−2.32 (−4.87,0.22) |
−0.92 (−4.26,2.41) |
−1.32 (−3.96,1.32) |
−2.36 (−4.90,0.18) |
−1.34 (−3.38,0.70) |
−1.90 (−3.97,0.16) |
−1.71 (−4.20,0.78) |
−0.65 (−2.82,1.52) |
Wait List |
In panel A, the lower left corner shows the Pittsburgh Sleep Quality Index (PSQI), the upper right corner displays Total Sleep Time (TST); AC, Acupuncture; CBT, Cognitive Behavioral Therapy; AP, Acupressure/Massage; RT, Relaxation Therapy; CUP, Cupping Therapy; SH, Sleep Hygiene; rTMS, Repetitive Transcranial Magnetic Stimulation; BZD, Benzodiazepines; NBZD, Non-Benzodiazepines; PLA, Placebo (sham acupuncture or sham treatment); Wait List, Waiting List. Bold values in tables indicate statistically significant results (p < 0.05).
Table 3.
Results of network meta-analysis (Panel B).
| AP | −0.52 (−1.09,0.06) |
0.06 (−0.58,0.70) |
−1.01
(−1.86,-0.17) |
−0.51 (−1.44,0.42) |
−0.40 (−1.15,0.35) |
−10.09
(−11.80,-8.39) |
0.01 (−0.80,0.82) |
−0.58
(−1.15,-0.01) |
−0.62 (−1.36,0.12) |
|---|---|---|---|---|---|---|---|---|---|
| −0.34 (−1.37,0.68) |
AC |
0.58
(0.06,1.10) |
−0.50 (−1.26,0.26) |
0.01 (−0.84,0.86) |
0.12 (−0.53,0.77) |
−9.58
(−11.18,-7.98) |
0.52 (−0.19,1.24) |
−0.07 (−0.50,0.37) |
−0.10 (−0.74,0.54) |
| −0.70 (−1.72,0.31) |
−0.36 (−0.83,0.11) |
CBT |
−1.08
(−1.67,-0.49) |
−0.57 (−1.36,0.22) |
−0.46 (−0.97,0.05) |
−10.16
(−11.84,-8.47) |
−0.05 (−0.63,0.52) |
−0.64
(−0.94,-0.35) |
−0.68
(−1.06,-0.29) |
| 0.64 (−0.57,1.85) |
0.99
(0.18,1.80) |
1.35
(0.65,2.05) |
SH | 0.51 (−0.46,1.47) |
0.62 (−0.02,1.25) |
−9.08
(−10.85,-7.31) |
1.02
(0.21,1.83) |
0.43 (−0.19,1.06) |
0.40 (−0.29,1.08) |
| −0.15 (−1.51,1.22) |
0.20 (−0.83,1.22) |
0.55 (−0.46,1.57) |
−0.79 (−2.00,0.41) |
rTMs | 0.11 (−0.77,0.99) |
−9.58
(−11.40,-7.77) |
0.52 (−0.41,1.45) |
−0.07 (−0.80,0.66) |
−0.11 (−0.97,0.76) |
| −0.18 (−1.24,0.88) |
0.16 (−0.39,0.72) |
0.52
(0.03,1.01) |
−0.82
(−1.57,-0.08) |
−0.03 (−1.09,1.02) |
RT | −9.70 (−11.43,-7.97) |
0.41 (−0.33,1.14) |
−0.18 (−0.67,0.30) |
−0.22 (−0.80,0.36) |
| 1.01 (−0.40,2.42) |
1.35
(0.39,2.32) |
1.71
(0.64,2.79) |
0.36 (−0.90,1.62) |
1.16 (−0.25,2.57) |
1.19
(0.07,2.30) |
BZD |
10.10
(8.35,11.86) |
9.51
(7.85,11.17) |
9.48
(7.75,11.20) |
| −0.41 (−1.50,0.68) |
−0.06 (−0.68,0.55) |
0.29 (−0.21,0.79) |
−1.05
(−1.90,-0.21) |
−0.26 (−1.35,0.83) |
−0.23 (−0.87,0.42) |
−1.42 (−2.56,-0.27) |
NBZD |
−0.59
(−1.17,-0.01) |
−0.63 (−1.31,0.06) |
| 0.02 (−0.94,0.99) |
0.37
(0.02,0.72) |
0.73
(0.42,1.03) |
−0.62 (−1.35,0.11) |
0.17 (−0.79,1.13) |
0.20 (−0.23,0.63) |
−0.99 (−2.02,0.04) |
0.43 (−0.07,0.93) |
PLA | −0.04 (−0.50,0.43) |
| 0.44 (−0.79,1.66) |
0.78 (−0.04,1.61) |
1.14
(0.46,1.82) |
−0.21 (−1.18,0.77) |
0.58 (−0.64,1.80) |
0.62 (−0.22,1.45) |
−0.57 (−1.84,0.70) |
0.84
(0.00,1.69) |
0.41 (−0.33,1.16) |
Wait List |
In panel B, the lower left corner indicates Sleep Efficiency (SE), while the upper right corner represents Sleep Latency (SL); AC, Acupuncture; CBT, Cognitive Behavioral Therapy; AP, Acupressure/Massage; RT, Relaxation Therapy; CUP, Cupping Therapy; SH, Sleep Hygiene; rTMS, Repetitive Transcranial Magnetic Stimulation; BZD, Benzodiazepines; NBZD, Non-Benzodiazepines; PLA, Placebo (sham acupuncture or sham treatment); Wait List, Waiting List. Bold values in tables indicate statistically significant results (p < 0.05).
Compared to PLA and the Wait List, both acupressure (MD = -2.06, 95% CI -3.45 to −0.68; MD = -2.71, 95% CI -4.94 to −0.49) and acupuncture (MD = -1.81, 95% CI -2.93 to −0.68; MD = -2.46, 95% CI -4.58 to −0.34) significantly decreased PSQI scores. BZD also showed PSQI reduction versus PLA (MD = -1.25, 95% CI -2.48 to −0.02). For TST, acupuncture outperformed BZD (MD = 2.07, 95% CI 0.46–3.68).
For SE improvement, network meta-analysis revealed significant benefits across multiple interventions. Acupressure showed superior efficacy versus PLA (MD = 0.86, 95% CI: 0.23 to 1.49), Wait List (MD = 1.27, 95% CI: 0.37 to 2.18), SH (MD = 1.48, 95% CI: 0.56 to 2.39), and BZD (MD = 1.47, 95% CI: 0.69 to 2.80). CBT demonstrated comparable effects versus PLA (MD = 0.72, 95% CI: 0.45 to 1.00), Wait List (MD = 1.14, 95% CI: 0.55 to 1.73), SH (MD = 1.34, 95% CI: 0.70 to 1.98), and BZD (MD = 1.67, 95% CI: 0.61 to 2.55), as did acupuncture versus PLA (MD = 0.47, 95% CI: 0.15 to 0.79), Wait List (MD = 0.88, 95% CI: 0.15 to 1.60), SH (MD = 1.08, 95% CI: 0.35 to 1.82), and BZD (MD = 1.35, 95% CI: 0.51 to 2.20).
NBZD also significantly improved SE versus Wait List (MD = 0.85, 95% CI: 0.10 to 1.59), SH (MD = 1.05, 95% CI: 0.28 to 1.82), and BZD (MD = 1.32, 95% CI: 0.31 to 2.33). Furthermore, CBT outperformed RT (MD = 0.52, 95% CI: 0.08 to 0.95), while RT itself showed advantages over SH (MD = 0.82, 95% CI: 0.15 to 1.50) and BZD (MD = 1.09, 95% CI: 0.11 to 2.07).
For SL reduction, multiple interventions demonstrated significant efficacy versus comparators. CBT (MD = −1.34, 95% CI: −2.02 to −0.66; MD = −10.15, 95% CI: −11.79 to −8.52), acupressure (MD = −1.31, 95% CI: −2.20 to −0.42; MD = −10.12, 95% CI: −11.78 to −8.46), NBZD (MD = −1.32, 95% CI: −2.18 to −0.45; MD = −10.13, 95% CI: −11.83 to −8.43), RT (MD = −1.19, 95% CI: −2.00 to −0.37; MD = −10.00, 95% CI: −11.67 to −8.33), and PLA (MD = −0.76, 95% CI: −1.49 to −0.03; MD = −9.57, 95% CI: −11.19 to −7.96) all outperformed both SH and BZD.
Compared to acupuncture and PLA, CBT (MD = −0.58, 95% CI: −1.01 to −0.14; MD = −0.58, 95% CI: −0.85 to −0.31) and acupressure (MD = −0.54, 95% CI: −1.06 to −0.02; MD = −0.55, 95% CI: −1.07 to −0.03) showed greater SL reduction. Furthermore, rTMS (MD = −9.65, 95% CI: −11.39 to −7.90), acupuncture (MD = −9.58, 95% CI: −11.15 to −8.00), Wait List (MD = −9.25, 95% CI: −11.19 to −7.85), and SH (MD = −8.81, 95% CI: −10.58 to −7.05) were all superior to BZD for SL reduction. CBT also exceeded Wait List (MD = −0.63, 95% CI: −0.98 to −0.28), while NBZD surpassed PLA (MD = −0.56, 95% CI: −1.10 to −0.01) in shortening SL.
3.7. Inconsistency and heterogeneity analysis
Global inconsistency tests revealed significant heterogeneity across all outcomes (PSQI, TST, SE, and SL; all p < 0.05). Node-splitting analysis confirmed local inconsistencies (p < 0.05) in six triangular loops for PSQI (Supplementary Figure S5). The acupressure-PLA-BZD loop showed the highest inconsistency factor (IF = 5.327, 95% CI: 2.80, 7.86), followed by acupressure-acupuncture-PLA (IF = 3.594, 95% CI: 0.00, 10.62) and acupuncture-CBT-NBZD (IF = 3.392, 95% CI: 2.36, 4.42). Other loops demonstrated lower inconsistency: acupuncture-PLA-BZD (IF = 3.208, 95% CI: 0.00, 7.42), acupressure-acupuncture-BZD (IF = 1.476, 95% CI: 0.00, 3.92), and acupuncture-CBT-BZD (IF = 0.611, 95% CI: 0.00, 4.20). Inconsistency was confirmed in two loops (95% CI excluding 0), while four showed potential consistency (95% CI excluding 0), indicating variability between direct and indirect evidence.
For TST analysis, four triangular loops were formed, showing varying inconsistency levels. The acupuncture-PLA-BZD loop exhibited moderate inconsistency (IF = 4.878, 95% CI: 0.65, 9.11), followed by acupressure-acupuncture -BZD (IF = 3.912, 95% CI: 3.00, 4.82). Lower inconsistency appeared in CBT-PLA-NBZD (IF = 0.187, 95% CI: 0.00, 1.58) and RT-PLA-NBZD (IF = 0.154, 95% CI: 0.00, 2.46). Two loops demonstrated significant inconsistency (95% CI excluding 0), suggesting discordance between direct/indirect comparisons that warrants further investigation.
For SE analysis, four triangular loops were analyzed with distinct inconsistency patterns. The acupressure-acupuncture-RT loop showed the highest inconsistency (IF = 1.868, 95% CI: 0.59, 3.15). Three loops displayed minimal inconsistency: CBT-SH-CUP (IF = 0.451, 95% CI: 0.00, 1.09), CBT-RT-BZD (IF = 0.56, 95% CI: 0.00, 0.85), and CBT-CUP-RT (IF = 0.451, 95% CI: 0.00, 1.09). Two loops exhibited significant inconsistency (95% CI excluding 0), indicating potential divergence between evidence sources.
For SL analysis, four triangular loops were assessed. The acupressure-acupuncture-RT loop showed mild inconsistency (IF = 1.401, 95% CI: 0.21, 2.59), while three loops demonstrated minimal inconsistency: CBT-CUP-RT (IF = 0.654, 95% CI: 0.00, 1.48), CBT-RT-BZD (IF = 0.180, 95% CI: 0.00, 1.09), and CBT-CUP-PLA (IF = 0.062, 95% CI: 0.00, 1.16). Only one loop exhibited significant inconsistency (95% CI excluding 0), suggesting generally consistent direct and indirect comparisons.
Substantial heterogeneity was observed across all outcomes: PSQI (I2 = 91.8%), TST (I2 = 88.3%), SE (I2 = 69.3%), and SL (I2 = 96.6%). Visual inspection of funnel plots revealed symmetrical distributions for all outcomes, indicating low risk of publication bias (Figure 4).
3.8. SUCRA rankings of interventions
The average SUCRA values for each intervention are presented in the Supplementary Figures S6–S9. CBT demonstrated the highest efficacy for both reducing PSQI scores (84.4% probability) and improving SE (91.5%), while ranking first for shortening SL (85.8%). For TST extension, acupuncture showed the highest probability (84.8%), followed by NBZD (62.8%). NBZD also ranked second for SL reduction (82.5% probability). CBT maintained strong performance across all outcomes, ranking third for PSQI reduction (69.9%) and TST extension (54.6%), and second for SE improvement (89.2%). Acupuncture additionally ranked third for SE improvement (70.5% probability).
3.9. Sensitivity and subgroup analyses
The leave-one-out sensitivity analysis confirmed that while excluding individual studies slightly altered effect sizes and p-values (Supplementary Figure S10), the pooled effect direction and statistical significance remained consistent. This consistency indicates robust meta-analytic findings.
Subgroup analyses by treatment duration (Supplementary Figure S11) and disease course (Supplementary Figure S12) revealed substantial heterogeneity across studies (I2 ≥ 69%, p < 0.0001), suggesting significant variability in outcomes. This heterogeneity likely arises from differences in study design (e.g., inclusion criteria, intervention protocols, control groups) or patient characteristics (e.g., disease severity, comorbidities). Between-subgroup heterogeneity was statistically significant for TST and SL (p < 0.05), indicating that treatment duration influences these outcomes. Short-term treatment (0–5 weeks) worsened PSQI and SL scores, whereas long-term treatment (>10 weeks) showed more stable effects, particularly for SE, which exhibited low heterogeneity. Disease duration significantly affected PSQI, TST, and SL (p < 0.05). Shorter disease duration was associated with increased PSQI scores (worsened sleep quality), suggesting greater challenges in improving sleep during early disease stages. TST and SL varied across subgroups, with shorter disease duration linked to reduced TST or prolonged SL, highlighting prominent sleep initiation difficulties. In contrast, SE did not differ significantly between subgroups, implying that disease duration has a weaker impact on sleep efficiency than on other metrics, though the overall trend remained unfavorable.
3.10. Safety analysis
Among the 53 included studies, 23 (43.4%) reported adverse events. Medication-related adverse effects (6 studies) included hepatorenal dysfunction, cardiovascular events, xerostomia, and daytime somnolence. Acupuncture-related adverse events (2 studies) comprised transient local pain, bruising, and hand paresthesia, all resolving with continued treatment. One study reported sleep restriction in the CBT group. Four studies documented unspecified adverse events, while 10 studies reported no safety concerns. Overall, adverse event incidence was low, with acupuncture demonstrating particularly favorable safety due to its transient effects.
4. Discussion
PI remains a significant global public health challenge. This systematic review of 53 RCTs demonstrates that seven non-pharmacological interventions—compared against sham controls or pharmacotherapies—significantly improved core sleep parameters, including PSQI scores, TST, SE, and SL, supporting their role as first-line treatments for PI. These findings, while compelling, are tempered by the low-to-moderate certainty of evidence, primarily due to methodological limitations in the included studies.
Pairwise meta-analyses demonstrated that acupressure, acupuncture, and CBT were particularly effective in improving specific sleep parameters associated with PI, including PSQI scores, SE, SL, and TST. Among these, CBT showed the most pronounced effects in reducing PSQI scores and SL while enhancing SE, whereas acupuncture yield superior outcomes in extending TST. These results are consistent with previous findings (70), reinforcing CBT’s status as the benchmark non-pharmacological intervention for the treatment of PI. While PSQI, TST, SE, and SL are validated measures of sleep architecture, they do not assess key insomnia domains like cognitive hyperarousal or daytime impairment—a limitation inherent to our focus on sleep-specific outcomes. The PSQI’s inclusion of non-specific factors (e.g., sleep environment) may reduce its sensitivity for pure insomnia assessment compared to the ISI (71), which directly measures core symptoms. We recommend future trials adopt ISI to standardize insomnia-specific evaluation while maintaining PSQI for broader sleep quality assessment.
In contrast, BZDs showed limited efficacy, performing worse than waitlist controls in most sleep outcomes, with the exception of PSQI scores. This inferior performance likely reflects the well-documented risks of BZDs, such as tolerance, dependence, and withdrawal symptoms (72). Although NBZDs were effective in increasing TST and shortening SL, concerns remain regarding their adverse effect profile. Notably, non-pharmacological interventions—particularly CBT—were linked to significantly fewer side effects and demonstrated better safety and long-term tolerability, making them more appropriate for chronic PI, where pharmacological treatments may exacerbate cognitive impairment, mood disturbances, or rebound insomnia.
Subgroup and sensitivity analyses further validated the robustness of the findings. Specifically, patients with long-standing PI (duration >10 years) and those undergoing prolonged treatment exhibited greater therapeutic benefits alongside reduced heterogeneity, suggesting that non-pharmacological interventions may offer superior efficacy in chronic PI management. These results support a paradigm shift in clinical practice, advocating for the reclassification of non-pharmacological therapies—traditionally regarded as adjunctive—to first-line treatment status in evidence-based guidelines.
Nevertheless, this review focused solely on monotherapy approaches. Future studies should investigate synergistic effects of combined interventions (e.g., CBT plus acupuncture) to optimize clinical outcomes. Additionally, pragmatic trials focusing on implementation are needed to evaluate real-world barriers like cost, availability, and provider training—that may influence accessibility and adherence. These studies will be essential for developing practical, scalable care models for PI.
4.1. Strengths and limitations
The strengths of this study include the use of guideline-recommended therapeutic approaches as the basis for literature searches, offering a strong basis for clinical decision-making when selecting appropriate treatment options. Sensitivity analyses confirmed the robustness of the results, as no directional changes were observed, supporting the reliability of the findings. However, substantial heterogeneity remained despite performing subgroup analyses based on treatment duration and disease course.
Variability in study design, inclusion and exclusion criteria, baseline characteristics (e.g., average age and geographic location), and trial duration likely contributed to these inconsistencies. Furthermore, the use of different scoring systems across the included RCTs limited our ability to fully resolve the observed heterogeneity.
While network meta-analysis provides a powerful framework for comparing multiple interventions indirectly, its validity depends critically on the quality and methodological consistency of included studies. Several limitations should be acknowledged. First, the analysis included 53 eligible RCTs, limiting statistical power and generalizability. Many trials had small sample sizes and exhibited methodological heterogeneity, particularly in intervention protocols, blinding procedures, and allocation concealment. Although most studies employed adequate randomization, the inherent challenges of blinding in non-pharmacological research—coupled with a lack of validation regarding its success—may have introduced performance and detection biases, potentially compromising outcome objectivity. Further limitations arise from inconsistencies in outcome measurement and reporting. Critical metrics of sleep maintenance, such as WASO and early morning awakening, were excluded due to heterogeneous definitions and insufficient standardized data across studies. The PSQI, while a widely accepted tool, does not specifically quantify WASO, thereby limiting its sensitivity in detecting sleep fragmentation. These discrepancies underscore the pressing need for uniform outcome measures in insomnia research to facilitate cross-study comparisons and enhance clinical relevance. To address these gaps, future investigations should prioritize large-scale, rigorously RCTs featuring robust blinding procedures, transparent reporting standards, and harmonized outcome assessments. Such efforts would strengthen the evidence base and yield more definitive guidance for clinical practice.
5. Conclusion
Non-pharmacological interventions demonstrated superior efficacy compared with pharmacological approaches for improving multiple sleep parameters in patients with PI. These therapies significantly increased TST, enhanced SE, reduced SL, and lowered PSQI scores. Additionally, they exhibited excellent safety profiles and high patient acceptability. Among all modalities examined, CBT, acupuncture, and acupressure emerged as the most effective options and should be considered first-line treatments.
Despite the promising results, several limitations should be acknowledged that may affect the generalizability and interpretation of the findings. Most of the included trials used the PSQI as the primary outcome measure. Although widely used, the PSQI assesses a broad range of sleep-related domains and may lack specificity for core insomnia symptoms. Notably, the PSQI does not directly evaluate key features such as cognitive hyperarousal or daytime impairment. In addition, other clinically relevant indicators—such as WASO and early morning awakening—were excluded due to inconsistent reporting and methodological heterogeneity. These limitations may reduce both the interpretability and generalizability of the results.
To enhance measurement precision and comparability across studies, future trials should employ more insomnia-specific instruments, such as the ISI, which directly assesses the severity of insomnia symptoms and their impact on daytime functioning. Moreover, large-scale, rigorously designed RCTs using standardized outcome measures are needed to strengthen the evidence base and support future guideline development.
Acknowledgments
We thank the China-Swiss TCM Centre for their support of the publication fee.
Glossary
Glossary
- PI
Primary insomnia
- RCTs
randomized controlled trials
- CBT
cognitive behavioral therapy
- PSQI
Pittsburgh Sleep Quality Index
- TST
total sleep time
- SE
sleep efficiency
- SL
sleep latency
- ROB
Cochrane Risk of Bias
- RR
relative risk
- CI
confidence interval
- MD
Mean difference
- SUCRA
Surface under the Cumulative Ranking
- RT
Relaxation Therapy
- CUP
Cupping Therapy
- SH
Sleep Hygiene
- rTMS
Repetitive Transcranial Magnetic Stimulation
- BZD
Benzodiazepines
- NBZD
Non-Benzodiazepines
- PLA
Placebo (sham acupuncture or sham treatment)
- Wait List
Waiting List
- IF
inconsistency factor
Funding Statement
The author(s) declare that financial support was received for the research and/or publication of this article. This study was supported by the Scientific Research Projects of Medical and Health Institutions of Longhua District, Shenzhen (2022010). The funder was not involved in the study design, collection, analysis, interpretation of data, the writing of this article or the decision to submit it for publication.
Data availability statement
The original contributions presented in the study are included in the article/Supplementary material, further inquiries can be directed to the corresponding author/s.
Author contributions
Q-hZ: Conceptualization, Data curation, Methodology, Validation, Visualization, Writing – original draft, Writing – review & editing, Funding acquisition, Resources. Y-jL: Conceptualization, Data curation, Methodology, Resources, Software, Validation, Visualization, Writing – original draft, Writing – review & editing. GY: Conceptualization, Methodology, Resources, Validation, Visualization, Writing – original draft, Writing – review & editing. M-yZ: Conceptualization, Methodology, Resources, Software, Validation, Visualization, Writing – original draft, Writing – review & editing. Jia-hY: Data curation, Formal analysis, Methodology, Resources, Software, Validation, Visualization, Writing – original draft, Writing – review & editing. LL: Data curation, Methodology, Resources, Validation, Visualization, Writing – original draft, Writing – review & editing. X-mY: Data curation, Resources, Validation, Visualization, Writing – original draft, Writing – review & editing. Q-lL: Conceptualization, Resources, Validation, Visualization, Writing – original draft, Writing – review & editing. Jin-hY: Conceptualization, Data curation, Resources, Validation, Visualization, Writing – original draft, Writing – review & editing. X-lL: Conceptualization, Resources, Validation, Visualization, Writing – original draft, Writing – review & editing. Y-mL: Conceptualization, Resources, Validation, Visualization, Writing – original draft, Writing – review & editing. T-cX: Resources, Validation, Visualization, Writing – original draft, Writing – review & editing. FJ: Conceptualization, Data curation, Investigation, Project administration, Supervision, Validation, Visualization, Writing – original draft, Writing – review & editing.
Conflict of interest
Q-hZ and J-hY were employed by Shenzhen Frontiers in Chinese Medicine Research Co., Ltd.
The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Generative AI statement
The authors declare that no Gen AI was used in the creation of this manuscript.
Publisher’s note
All claims expressed in this article are solely those of the authors and do not necessarily represent those of their affiliated organizations, or those of the publisher, the editors and the reviewers. Any product that may be evaluated in this article, or claim that may be made by its manufacturer, is not guaranteed or endorsed by the publisher.
Supplementary material
The Supplementary material for this article can be found online at: https://www.frontiersin.org/articles/10.3389/fneur.2025.1607903/full#supplementary-material
References
- 1.Roth T, Coulouvrat C, Hajak G, Lakoma MD, Sampson NA, Shahly V, et al. Prevalence and perceived health associated with insomnia based on DSM-IV-TR; ICD-10; and research diagnostic criteria/ICSD-2 criteria: results from the American insomnia survey. Biol Psychiatry. (2011) 69:592–600. doi: 10.1016/j.biopsych.2010.10.023, PMID: [DOI] [PubMed] [Google Scholar]
- 2.Aernout E, Benradia I, Hazo JB, Sy A, Askevis-Leherpeux F, Sebbane D, et al. International study of the prevalence and factors associated with insomnia in the general population. Sleep Med. (2021) 82:186–92. doi: 10.1016/j.sleep.2021.03.028, PMID: [DOI] [PubMed] [Google Scholar]
- 3.Qaseem A, Kansagara D, Forciea MA, Cooke M, Denberg TD, for the Clinical Guidelines Committee of the American College of Physicians* . Management of chronic insomnia disorder in adults: a clinical practice guideline from the American College of Physicians. Ann Intern Med. (2016) 165:125–33. doi: 10.7326/M15-2175, PMID: [DOI] [PubMed] [Google Scholar]
- 4.Wilt TJ, Macdonald R, Brasure M, Olson CM, Carlyle M, Fuchs E, et al. Pharmacologic treatment of insomnia disorder: an evidence report for a clinical practice guideline by the American College of Physicians. Ann Intern Med. (2016) 165:103–12. doi: 10.7326/M15-1781, PMID: [DOI] [PubMed] [Google Scholar]
- 5.Riemann D, Espie CA, Altena E, Arnardottir ES, Baglioni C, Bassetti CLA, et al. The European insomnia guideline: an update on the diagnosis and treatment of insomnia 2023. J Sleep Res. (2023) 32:e14035. doi: 10.1111/jsr.14035, PMID: [DOI] [PubMed] [Google Scholar]
- 6.Hertenstein E, Trinca E, Wunderlin M, Schneider CL, Züst MA, Fehér KD, et al. Cognitive behavioral therapy for insomnia in patients with mental disorders and comorbid insomnia: a systematic review and meta-analysis. Sleep Med Rev. (2022) 62:101597. doi: 10.1016/j.smrv.2022.101597, PMID: [DOI] [PubMed] [Google Scholar]
- 7.Spence DW, Kayumov L, Chen A, Lowe A, Jain U, Katzman MA, et al. Acupuncture increases nocturnal melatonin secretion and reduces insomnia and anxiety: a preliminary report. J Neuropsychiatry Clin Neurosci. (2004) 16:19–28. doi: 10.1176/jnp.16.1.19, PMID: [DOI] [PubMed] [Google Scholar]
- 8.Li Z, Jiao K, Chen M, Wang C. Effect of magnitopuncture on sympathetic and parasympathetic nerve activities in healthy drivers – assessment by power spectrum analysis of heart rate variability. Eur J Appl Physiol. (2003) 88:404–10. doi: 10.1007/s00421-002-0747-5, PMID: [DOI] [PubMed] [Google Scholar]
- 9.Wang SC, Jiang YM, Lai DD, Liu LM, Cao YN. Clinical observation on the effect of acupuncture at Shenmen and Sishencong on polysomnography in 150 patients with primary insomnia. Chin J Integr Med. (2018) 33:3203–5. [Google Scholar]
- 10.Pfeiffer AM, Triplett C, Siengsukon CF. Examining the prevalence of sleep disturbances in patients seeking physical therapy services. Physiother Theory Pract. (2024) 40:556–64. doi: 10.1080/09593985.2022.2134754, PMID: [DOI] [PubMed] [Google Scholar]
- 11.Hysing M, Pallesen S, Stormark KM, Lundervold AJ, Sivertsen B. Sleep patterns and insomnia among adolescents: a population-based study. J Sleep Res. (2013) 22:549–56. doi: 10.1111/jsr.12055, PMID: [DOI] [PubMed] [Google Scholar]
- 12.Kwon CY, Lee B, Cheong MJ, Kim TH, Jang BH, Chung SY, et al. Non-pharmacological treatment for elderly individuals with insomnia: a systematic review and network meta-analysis. Front Psych. (2021) 11:608896. doi: 10.3389/fpsyt.2020.608896, PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.American Psychiatric Association . Diagnostic and statistical manual of mental disorders. Washington, DC: American Psychiatric Association; (2013). [Google Scholar]
- 14.Zhao ZX, Zhang P, Huang LQ. Guidelines for the diagnosis and treatment of insomnia in Chinese adults. Chin J Neurol. (2012) 45:11. doi: 10.3760/cma.j.issn.1006-7876.2012.07.022 [DOI] [Google Scholar]
- 15.Buysse DJ, Reynolds CF, Monk TH, Berman SR, Kupfer DJ. The Pittsburgh sleep quality index: a new instrument for psychiatric practice and research. Psychiatry Res. (1989) 28:193–213. doi: 10.1016/0165-1781(89)90047-4, PMID: [DOI] [PubMed] [Google Scholar]
- 16.Zhirong Y, Feng S, Siyan Z. Risk of bias assessment: (2) revised Cochrane risk of bias tool for individually randomized, parallel group trials (RoB 2.0). Chin J Endemiol. (2017) 38:1285–91. [DOI] [PubMed] [Google Scholar]
- 17.Cao Y, Yin X, Yue HY, Li SS, Xu SF. Effects of Acupuncture Based on Regulating Du Meridian and Calming Mind on Sleep Quality in Patients with Primary Insomnia Liaoning Zhongyi Zazhi. (2020) 47:157–60. doi: 10.13192/j.issn.1000-1719.2020.12.046 [DOI] [Google Scholar]
- 18.Gao SF, Luo YJ. Clinical observation on the intervention of auricular acupuncture for primary insomnia. Zhongyiyao Daobao. (2016) 22:94–5. doi: 10.13862/j.cnki.cn43-1446/r.2016.02.037 [DOI] [Google Scholar]
- 19.Li YJ, Xie M, Ruan Q, Feng CY, Tao HS. Evaluation of clinical efficacy of abdominal acupuncture for chronic primary insomnia with heart-kidney disharmony and its effect on plasma 5-HT. Liaoning Zhongyi Zazhi. (2019) 46:2412–5. doi: 10.13192/j.issn.1000-1719.2019.11.048 [DOI] [Google Scholar]
- 20.Zhang HJ, Yao Y, Zhang JW. A controlled study of cognitive-behavioral therapy and combined pharmacotherapy for primary insomnia. Chin J Pract Neurol. (2010) 13:6–9. [Google Scholar]
- 21.Zhao J, Hong QY, Yang Y, Yao J, Zhou XP, Wang GL. Effects of He’s three connections method on sleep-wake patterns and sleep quality in patients with primary insomnia. Hebei Zhongyi. (2020) 42:1241–4. [Google Scholar]
- 22.Xuan YB, Guo J, Wang LP, Wu X. The effect of acupuncture on sleep quality in patients with primary insomnia: a randomized controlled study. Zhongguo Zhen Jiu. (2007) 12:886–8. [PubMed] [Google Scholar]
- 23.Li L, Liu R, Zhang T, Guo J, Chen H. Effects of acupuncture on sleep quality and excessive wakefulness in patients with primary insomnia. Shanghai Zhenjiu Zazhi. (2019) 38:973–7. doi: 10.13460/j.issn.1005-0957.2019.09.0973 [DOI] [Google Scholar]
- 24.Jiang L. Clinical study on the treatment of primary insomnia with traditional Chinese medicine pillow combined with five-tone therapy. Sichuan Zhongyi. (2020) 38:210–2. [Google Scholar]
- 25.Feng XJ, Gai HJ, Wang XY. Observational study on the efficacy of repetitive transcranial magnetic stimulation for primary insomnia. Clin Psychiatr J. (2017) 27:415–7. [Google Scholar]
- 26.Tu JH, Chung WC, Yang CY, Tzeng DS. A comparison between acupuncture versus zolpidem in the treatment of primary insomnia. Asian J Psychiatry. (2012) 5:231–5. doi: 10.1016/j.ajp.2011.12.003, PMID: [DOI] [PubMed] [Google Scholar]
- 27.Jespersen KV, Otto M, Kringelbach M, van Someren E, Vuust P. A randomized controlled trial of bedtime music for insomnia disorder. J Sleep Res. (2019) 28:e12817. doi: 10.1111/jsr.12817, PMID: [DOI] [PubMed] [Google Scholar]
- 28.Jacobs GD, Pace-Schott EF, Stickgold R, Otto MW. Cognitive behavior therapy and pharmacotherapy for insomnia: a randomized controlled trial and direct comparison. Arch Intern Med. (2004) 164:1888–96. doi: 10.1001/archinte.164.17.1888 [DOI] [PubMed] [Google Scholar]
- 29.Edinger JD, Wohlgemuth WK, Radtke RA, Marsh GR, Quillian RE. Cognitive behavioral therapy for treatment of chronic primary insomnia: a randomized controlled trial. JAMA. (2001) 285:1856–64. doi: 10.1001/jama.285.14.1856, PMID: [DOI] [PubMed] [Google Scholar]
- 30.Siu PM, Yu AP, Tam BT, Chin EC, Yu DS, Chung KF, et al. Effects of tai chi or exercise on sleep in older adults with insomnia: a randomized clinical trial. JAMA Netw Open. (2021) 4:e2037199. doi: 10.1001/jamanetworkopen.2020.37199, PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yin X, Gou M, Xu J, Dong B, Yin P, Masquelin F, et al. Efficacy and safety of acupuncture treatment on primary insomnia: a randomized controlled trial. Sleep Med. (2017) 37:193–200. doi: 10.1016/j.sleep.2017.02.012, PMID: [DOI] [PubMed] [Google Scholar]
- 32.Lee MRG, Breitstein J, Hoyt T, Stolee J, Baxter T, Kwon H, et al. Cognitive behavioral therapy for insomnia among active duty military personnel. Psychol Serv. (2021) 18:42–50. doi: 10.1037/ser0000340, PMID: [DOI] [PubMed] [Google Scholar]
- 33.Ritterband LM, Thorndike FP, Gonder-Frederick LA, Magee JC, Bailey ET, Saylor DK, et al. Efficacy of an internet-based behavioral intervention for adults with insomnia. Arch Gen Psychiatry. (2009) 66:692–8. doi: 10.1001/archgenpsychiatry.2009.66, PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Yeung W, Chung K, Zhang S, Yeung WF, Chung KF, Zhang SP, et al. Electroacupuncture for primary insomnia: a randomized controlled trial. Sleep. (2009) 32:1039–47. doi: 10.1093/sleep/32.8.1039, PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Van Straten A, Emmelkamp J, De Wit J, Lancee J, Andersson G, van Someren EJ, et al. Guided internet-delivered cognitive behavioural treatment for insomnia: a randomized trial. Psychol Med. (2014) 44:1521–32. doi: 10.1017/S0033291713002249, PMID: [DOI] [PubMed] [Google Scholar]
- 36.Scharf MB, Black J, Hull S, Landin R, Farber R. Long-term nightly treatment with indiplon in adults with primary insomnia: results of a double-blind, placebo-controlled, 3-month study. Sleep. (2007) 30:743–52. doi: 10.1093/sleep/30.6.743, PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Schramm PJ, Zobel I, Mönch K, Schramm E, Michalak J. Sleep quality changes in chronically depressed patients treated with mindfulness-based cognitive therapy or the cognitive behavioral analysis system of psychotherapy: a pilot study. Sleep Med. (2016) 17:57–63. doi: 10.1016/j.sleep.2015.09.022, PMID: [DOI] [PubMed] [Google Scholar]
- 38.Yeung WF, Yu BYM, Yuen JWM, Ho JYS, Chung KF, Zhang ZJ, et al. Semi-individualized acupuncture for insomnia disorder and oxidative stress: a randomized, double-blind, sham-controlled trial. Nat Sci Sleep. (2021) 13:1195–207. doi: 10.2147/NSS.S318874, PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Khalsa SBS, Goldstein MR. Treatment of chronic primary sleep onset insomnia with kundalini yoga: a randomized controlled trial with active sleep hygiene comparison. J Clin Sleep Med. (2021) 17:1841–52. doi: 10.5664/jcsm.9320, PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Riemann D, Voderholzer U, Cohrs S, Rodenbeck A, Hajak G, Rüther E, et al. Trimipramine in primary insomnia: results of a polysomnographic double-blind controlled study. Pharmacopsychiatry. (2002) 35:165–74. doi: 10.1055/s-2002-34119, PMID: [DOI] [PubMed] [Google Scholar]
- 41.Passos GS, Poyares D, Santana MG, Garbuio SA, Tufik S, Mello MT. Effect of acute physical exercise on patients with chronic primary insomnia. J Clin Sleep Med. (2010) 6:270–5. doi: 10.5664/jcsm.27825 PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Lo C, Liao WC, Liaw JJ, Hang LW, Lin JG. The stimulation effect of auricular magnetic press pellets on older female adults with sleep disturbance undergoing polysomnographic evaluation. Evid Based Complement Alternat Med. (2013) 2013:1–8. doi: 10.1155/2013/530438, PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Morin CM, Colecchi C, Stone J, Sood R, Brink D. Behavioral and pharmacological therapies for late-life insomnia. JAMA. (1999) 281:991. doi: 10.1001/jama.281.11.991, PMID: [DOI] [PubMed] [Google Scholar]
- 44.Wu R, Bao J, Zhang C, Deng J, Long C. Comparison of sleep condition and sleep-related psychological activity after cognitive-behavior and pharmacological therapy for chronic insomnia. Psychother Psychosom. (2006) 75:220–8. doi: 10.1159/000092892, PMID: [DOI] [PubMed] [Google Scholar]
- 45.Fu C, Zhao N, Liu Z, Yuan LH, Xie C, Yang WJ, et al. Acupuncture improves peri-menopausal insomnia: A randomized controlled trial. Sleep. (2017) 40:153. doi: 10.1093/sleep/zsx153, PMID: [DOI] [PubMed] [Google Scholar]
- 46.Espie CA, MacMahon KMA, Kelly HL, MacMahon KM, Broomfield NM, Douglas NJ, et al. Randomized clinical effectiveness trial of nurse-administered small-group cognitive behavior therapy for persistent insomnia in general practice. Sleep. (2007) 30:574–84. doi: 10.1093/sleep/30.5.574, PMID: [DOI] [PubMed] [Google Scholar]
- 47.Abedian Z, Eskandari L, Abdi H, Ebrahimzadeh S. The effect of acupressure on sleep quality in menopausal women: a randomized control trial. Complement Ther Clin Pract. (2015) 40:328–34. PMID: [PMC free article] [PubMed] [Google Scholar]
- 48.Huang Z, Li Y, Bianchi MT, Zhan S, Jiang F, Li N, et al. Repetitive transcranial magnetic stimulation of the right parietal cortex for comorbid generalized anxiety disorder and insomnia: a randomized, double-blind, sham-controlled pilot study. Brain Stimul. (2018) 11:1103–9. doi: 10.1016/j.brs.2018.05.016 [DOI] [PubMed] [Google Scholar]
- 49.Tang HL, Chen Z, Pang J, Mo Q. Shujing tuina therapy for insomnia: a randomized controlled study. Zhongguo Zhen Jiu. (2015) 35:816–8. doi: 10.13703/j.0255-2930.2015.08.017 PMID: [DOI] [PubMed] [Google Scholar]
- 50.Wang N, Zhou XY. Effects of the three-part Tuina technique on monoamine neurotransmitters and sleep quality in patients with primary insomnia. Zhongyi Xuebao. (2024) 39:11. doi: 10.16368/j.issn.1674-8999.2024.03.112 [DOI] [Google Scholar]
- 51.Lin JZ, Zhang ML, Qu LM, Wang JC, Ye RF. Long-term efficacy of acupuncture combined with biofeedback relaxation therapy for chronic insomnia in elderly patients. Chin J Integr Med. (2012) 27:2222–4. [Google Scholar]
- 52.Wang JP, Wang JB, Wang LC, Zhang Y. Combined acupuncture and medication therapy for senile insomnia: a randomized controlled study. Zhongguo Zhen Jiu. (2015) 35:544–8. doi: 10.13703/j.0255-2930.2015.06.004 PMID: [DOI] [PubMed] [Google Scholar]
- 53.Zhang J, Liu X, Xie X, Zhao D, Shan MS, Zhang XL, et al. Mindfulness-based stress reduction for chronic insomnia in adults older than 75 years: a randomized, controlled, single-blind clinical trial. Explore (NY). (2015) 11:180–5. doi: 10.1016/j.explore.2015.02.005, PMID: [DOI] [PubMed] [Google Scholar]
- 54.Xu P, Ji WD, Pan YS. Effects of cognitive behavioral therapy combined with medication on serum cytokines and cortisol in elderly patients with sleep disorders. Pract Geriatr Med. (2015) 29:137–41. [Google Scholar]
- 55.Liang XM. Clinical observation on auricular acupressure treatment for primary insomnia in elderly patients. Shanghai Zhenjiu Zazhi. (2017) 36:719–22. doi: 10.13460/j.issn.1005-0957.2017.06.0719 [DOI] [Google Scholar]
- 56.Yu XP, Gao QC. Clinical study on the effects of acupuncture on sleep quality and cognitive function in elderly patients with primary insomnia. Jiangsu Zhongyi Yao. (2019) 51:62–4. [Google Scholar]
- 57.McCrae CS, McGovern R, Lukefahr R, Stripling AM. Research evaluating brief behavioral sleep treatments for rural elderly (RESTORE): a preliminary examination of effectiveness. Am J Geriatr Psychiatry. (2007) 15:979–82. doi: 10.1097/JGP.0b013e31813547e6, PMID: [DOI] [PubMed] [Google Scholar]
- 58.Edinger JD, Olsen MK, Stechuchak KM, Means MK, Lineberger MD, Kirby A, et al. Cognitive behavioral therapy for patients with primary insomnia or insomnia associated predominantly with mixed psychiatric disorders: a randomized clinical trial. Sleep. (2009) 32:499–510. doi: 10.1093/sleep/32.4.499, PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Espie CA, Kyle SD, Williams C, Ong JC, Douglas NJ, Hames P, et al. A randomized, placebo-controlled trial of online cognitive behavioral therapy for chronic insomnia disorder delivered via an automated media-rich web application. Sleep. (2012) 35:769–81. doi: 10.5665/sleep.1872, PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Jernelöv S, Lekander M, Blom K, Rydh S, Ljótsson B, Axelsson J, et al. Efficacy of a behavioral self-help treatment with or without therapist guidance for co-morbid and primary insomnia: a randomized controlled trial. BMC Psychiatry. (2012) 12:5. doi: 10.1186/1471-244X-12-5, PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Kaldo V, Jernelöv S, Blom K, Ljótsson B, Brodin M, Jörgensen M, et al. Guided internet cognitive behavioral therapy for insomnia compared to a control treatment – a randomized trial. Behav Res Ther. (2015) 71:90–100. doi: 10.1016/j.brat.2015.06.001, PMID: [DOI] [PubMed] [Google Scholar]
- 62.Lichstein KL, Riedel BW, Wilson NM, Lester KW, Aguillard RN. Relaxation and sleep compression for late-life insomnia: a placebo-controlled trial. J Consult Clin Psychol. (2001) 69:227–39. doi: 10.1037/0022-006X.69.2.227, PMID: [DOI] [PubMed] [Google Scholar]
- 63.Lovato N, Lack L, Wright H, Kennaway DJ. Evaluation of a brief treatment program of cognitive behavior therapy for insomnia in older adults. Sleep. (2014) 37:117–26. doi: 10.5665/sleep.3320, PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Sivertsen B, Omvik S, Pallesen S, Bjorvatn B, Havik OE, Kvale G, et al. Cognitive behavioral therapy vs Zopiclone for treatment of chronic primary insomnia in older adults. JAMA. (2006) 295:2851–8. doi: 10.1001/jama.295.24.2851, PMID: [DOI] [PubMed] [Google Scholar]
- 65.Wei M, Cao RF, Gu F, Gu F, Lv Q. Observation of therapeutic effect of neigong Yi Zhi Chan Tuina for insomnia. Shanghai J Tradit Chin Med. (2013) 47:60–1. [Google Scholar]
- 66.Wei DM, Liu JZ, Li Y, Chu R, Liu YL, Wang Z. Clinical observation on the efficacy of adult and pediatric Tuina combined with acupoint pressing for primary insomnia. Hebei Zhongyi. (2022) 44:460–3. [Google Scholar]
- 67.Yang HR, Wang YX, Zhou B, Lei Y, Yang T, Wang JM, et al. Randomized controlled study of the three-part Tuina technique for insomnia based on the theory of regulating Qi. Chin J Integr Med. (2024) 39:10. [Google Scholar]
- 68.Zhuang QX. Clinical efficacy and safety of traditional Chinese Tuina therapy for insomnia. Clin Rational Drug Use J. (2020) 13:11. doi: 10.15887/j.cnki.13-1389/r.2020.22.051 [DOI] [Google Scholar]
- 69.Wei M, Cao RF, Gu F, Lv Q. Clinical observation of the efficacy of Cao Renfa’s three-step Tuina method for insomnia. Shanghai J Tradit Chin Med. (2017) 51:61–3. doi: 10.16305/j.1007-1334.2017.01.018 [DOI] [Google Scholar]
- 70.Gao L, Kong DZ, Wang SJ, Zhang Z. Network meta-analysis of the efficacy of external TCM therapies in improving sleep quality for insomnia. World Chin Med. (2022) 17:2753–8. [Google Scholar]
- 71.Bastien CH, Vallières A, Morin CM. Validation of the insomnia severity index as an outcome measure for insomnia research. Sleep Med. (2001) 2:297–307. doi: 10.1016/S1389-9457(00)00065-4 [DOI] [PubMed] [Google Scholar]
- 72.Xiang XJ, Liu TB, Wang CY, Liu TQ, Zhou YF, Ruan CJ. Adverse reactions and management of benzodiazepines. Chin J Drug Abuse Prev Treat. (2017) 23:256–60. doi: 10.15900/j.cnki.zylf1995.2017.05.003 [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The original contributions presented in the study are included in the article/Supplementary material, further inquiries can be directed to the corresponding author/s.