Abstract
Introduction
Pregnancy-related AKI (PR-AKI), has profound maternal and fetal implications, including high mortality and long-term risks such as the development of chronic kidney disease (CKD). This systematic review aims to evaluate the burden of CKD owing to PR-AKI cases during follow-up in developing countries, particularly India.
Methods
A systematic search of PubMed, Embase, and Cochrane databases was performed for Indian studies published between 2000 and June 2024. We included cross-sectional, retrospective, and prospective cohort studies that reported the incidence of PR-AKI, subsequent CKD, and dialysis dependency in Indian cohorts during follow-up. Details of etiology of PRAKI, and adverse fetal and maternal outcomes were also recorded. Only studies that provided follow-up kidney outcomes were considered.
Results
A total of 25 studies comprising 2,306 participants were included in the analysis. The incidence of PR-AKI ranged from 1 to 12% across different studies. Sepsis was the most common cause of PR-AKI, accounting for up to 78% of cases, followed by hypertensive disorders, obstetric haemorrhage, and tropical etiologies. Hemodialysis was required in 20–85% of patients. CKD development during follow-up was observed in 12.8–35% of cases, with up to 30% remaining dialysis-dependent. Maternal mortality ranged from 2.5 to 34%, while perinatal mortality reached as high as 67.3%. Pre-term delivery rates varied between 13.9% and 58%.
Conclusions
Up to one-third of PR-AKI patients may develop CKD and remain dialysis-dependent during follow-up. PR-AKI significantly impacts both maternal and fetal morbidity and mortality. Early prevention and prompt management by healthcare professionals are critical to improving outcomes in PR-AKI.
Summary at a Glance
Pregnancy-related acute kidney injury (PR-AKI) significantly affects maternal and fetal health, leading to high mortality and long-term complications such as chronic kidney disease (CKD). This systematic review, focusing on developing countries like India, evaluated the burden of CKD due to PR-AKI patients. The review analyzed Indian studies published between 2000 and June 2024, including 25 studies with 2,306 participants. PR-AKI incidence ranged from 1 to 12%, with sepsis being the leading cause in up to 78% of cases, followed by hypertensive disorders, obstetric hemorrhage, and tropical fevers. RRT was needed in 20–85% of patients, and 12.8–35% developed CKD during follow-up, with up to 30% remaining dialysis-dependent. Maternal mortality varied from 2.5 to 34%, while perinatal mortality reached 67.3%. The study emphasizes the critical need for early prevention timely intervention and need for long-term follow-up to reduce the high morbidity and mortality rates associated with PR-AKI.
Supplementary Information
The online version contains supplementary material available at 10.1186/s40748-025-00224-9.
Keywords: Pregnancy-related acute kidney injury, Chronic kidney disease (CKD) hemolysis, Elevated liver enzymes and low platelets (HELLP), Pre-eclampsia Thrombotic microangiopathy and Dialysis
Introduction
Pregnancy-related acute kidney injury (PRAKI) represents a critical cause of maternal and fetal morbidity and mortality, particularly in developing countries. While its epidemiology varies globally, the burden remains disproportionately high in low- and middle-income regions, where healthcare access and resources are limited [1]. The maternal mortality in PRAKI is as high as 25% [2]. Despite recent improvements in maternal care, India still grapples with a significant incidence of PRAKI, which contributes to long-term complications such as chronic kidney disease (CKD and end stage kidney disease (ESKD). PRAKI not only threatens immediate maternal outcomes but also serves as a hidden driver of CKD, adding to the existing healthcare burden in a country already witnessing an epidemic of CKD [3]. Moreover, the long-term repercussions of PRAKI are profound, as up to 30% of survivors develop CKD, and 20% may remain dialysis-dependent [4]. In India, the incidence of PRAKI has shown a steady decline over the past decades—from 22% in the 1960s to 3–7% in the 2000s—primarily due to enhanced antenatal care, a reduction in septic abortions, and better management of obstetric complications such as anti and postpartum hemorrhage [5]. Recent data from the Indian Society of Nephrology (ISN) registry indicate that obstetric complications contribute to 5% of all acute kidney injury (AKI) cases, underscoring a persistent and significant public health concern in India [6] The evolving landscape of PRAKI in India shows a shift from traditional causes like septic abortion to emerging drivers such as hypertensive disorders, preeclampsia, and thrombotic microangiopathies (TMA). These complications, which arise predominantly in the later stages of pregnancy, continue to pose diagnostic and therapeutic challenges [7]. Conditions such as acute fatty liver of pregnancy (AFLP), hemolysis, elevated liver enzymes, low platelets (HELLP syndrome), and puerperal sepsis also contribute to the risk of AKI, complicating both maternal and fetal outcomes. The significant incidence of PRAKI, along with a substantial number of women developing CKD highlights the significance of understanding PRAKI’s contribution to the rising CKD epidemic among women in developing countries such as India.
Given the significant variability in PRAKI incidence and outcomes across different regions in India, there is a critical need for a systematic review of Indian studies. By analyzing the evolving etiological patterns, risk factors, and long-term consequences, more precise strategies for early diagnosis and intervention can be developed. Furthermore, as India progresses towards adopting advanced diagnostic tools such as genetic analysis, identifying complications like TMA, preeclampsia, and HELLP syndrome can lead to better-targeted therapies, ultimately improving maternal and fetal health outcomes.
Methodology
Inclusion criteria
We included studies that reported kidney outcomes in women following an episode of AKI during pregnancy in India. Only studies published as full-text articles and providing data on kidney outcomes were included. Eligible studies included cohort studies, randomized control trials, case-control studies, and cross-sectional studies, except for in vitro studies, letters to the editor, review articles, editorials, and short communications. Articles published in languages other than English and outside India were also excluded.
The study adhered to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) statement guidelines [8]. The systematic review protocol is registered in PROSPERO (registration number: CRD42024576213; http://www.crd.york.ac.uk/PROSPERO). Prisma checklist of the systematic analysis is provided in supplementary material 2.
Data search
A comprehensive literature search was conducted across EMBASE, PubMed/MEDLINE, and the Cochrane CENTRAL databases from January 2000 to July 30, 2024. The search strategy utilized MeSH terms and relevant keywords, including “pregnancy,” “obstetrics,” “pregnant,” “acute kidney injury,” “acute renal injury,” “acute renal insufficiency,” “acute renal failure,” and “India.” A detailed search strategy is provided in Supplementary Annexure 1.
Article selection
The selection process for articles is illustrated in the PRISMA flow chart (Fig. 1). After removing duplicate records, the titles and abstracts were independently reviewed by two reviewers (Priti Meena and Vinant Bhargava) to assess eligibility. Full-text articles were screened for inclusion, with discrepancies resolved through consensus with a third author (Sandip Panda). For studies with multiple publications, the most comprehensive or latest dataset was selected. Articles deemed unsuitable based on title and/or abstract consensus were excluded. Full-text articles underwent independent screening by two authors (Paromita Das and Kritika Mohan), with mutual agreement required for inclusion. Disagreements were addressed by consulting a third author. Data extraction was performed by four authors using a pre-designed form, and verification was conducted by two additional independent authors.
Fig. 1.
Prisma flowchart
Data extraction
Data extracted from each included article encompassed the following: Author, year of the study, site of the study in India, the total number of included patients, number of patients with PRAKI, the definition criteria of PRAKI, information about parity and timing of AKI occurrence in correspondence to the stage of pregnancy. The etiologies of PR-AKI were recorded in detail.
Outcomes
Pregnancy outcomes are categorized into maternal and fetal outcomes. Maternal outcomes included maternal survival and the need for intensive care unit (ICU) admission. Fetal outcomes included premature delivery and fetal mortality. Kidney outcomes included the need for dialysis, renal recovery, and non-recovery (progression to CKD or remaining dialysis-dependent). Information about renal biopsies was also included in provided in the study.
Quality assessment
The quality of observational studies was assessed using the Newcastle-Ottawa Quality Assessment Scale [9]. A comprehensive evaluation of each study is provided in the online Supplementary Annexure 2, Table 1, detailing strengths and limitations identified through these quality appraisal methods. This assessment was conducted independently by two reviewers (PD and SP), with discrepancies resolved through discussion. The certainty of evidence for each outcome was determined using the “Grading of Recommendations Assessment, Development, and Evaluation” (GRADE) methodology.
Table 1.
Baseline characteristics of the included studies AKI: acute kidney injury, PRAKI: Pregnancy-related acute kidney injury,
| Sl, number | First author | Year of publication | Study design | Location in India | Patients number (n) | Incidence of PRAKI | Inclusion criterion | AKI Criterion | Mean Age | Gravida | Timing of AKI |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Goplani KR et al, [10] | 2008 | Prospective study | Gujrat | 70 | 9.06% | Urine output < 400 ml/day and azotemia serum creatinine > 2 mg/dl | Not defined | 25.6yrs |
Primigravida: 22(31.4%) Multigravida: 48(68.57%) multi |
Early pregnancy:14(20%) 5(7.14%) late and peupeural: 51(72.85%) |
| 2 | Najar MS et al., [11] | 2008 | Prospective observational | Kashmir | 40 | 7.02% | Oliguria(urine output < 400 ml/day) or anuria with serum creatinine > 1.5 mg/dl | Not defined | 28.94 ± 5.93 |
12 (30%)- primigravida 28 (70%)- multigravida |
15 (75%)- first trimester 5 (25%)- second trimester |
| 3 | Arora N et al., [12] | 2010 | Prospective, Observational | West Bengal | 57 | 6.5% of all AKI patients requiring dialysis) | sudden oliguria (urine output less than 300 mL over 24 h), or anuria with a sudden increase in serum creatinine to more than 1.5 mg/dL or an increase in serum creatinine of more than 0.5 mg/dL per day from baseline | RIFLE | 25.8 ± 4.5 |
primigravida − 26(45.6%) Gravida 2–9 (15.8%) Gravida ≥ 3–22 (38.6%) |
11 (19.3%) cases in the first half of pregnancy (< 20 weeks of gestation) 6 (10.5%) cases in the second half of pregnancy 40 (70.2%) during puerperium |
| 4 | Sivakumar V et al., [13] | 2011 | Retrospective | Andhra Pradesh | 59 | 4.36% | All PRAKI cases during study period | Not defined | 25.6 | NR | Postpartum 43(74.57%), Third trimester 10 (16.94%), Second trimester 4(6.77%), First trimester 2(1.69%) |
| 5 | Patel ML et al. [14] | 2013 | Prospective | Uttar Pradesh | 60 | 11.5% | Oliguria(urine output < 300 ml/day) or anuria with serum creatinine > 1.5 mg/dl or increase in serum creatinine > 0.5 mg/dl per day from the baseline | NR | 28(median age) |
Primigravida 32 (53.4%)- Multiparous: 28 (46.7%)- |
32 (53.3%)- early part 28 (46.7%)- later half of pregnancy and puerperium |
| 6 | Pahwa N et al., [15] | 2014. | Retrospective | Madhyapradesh | 27 | 3.59% |
All the post-partum patients with AKI |
RIFLE | 26.7 ± 4.2 | NR | Only postpartum AKI |
| 7 | Godara SM et al., [16] | 2010 | Prospective | Gujarat | 57 | 9.82% | Oliguria (urine output < 400 ml/d)and/or mounting azotemia(serum creatinine > 2 mg/dL) | NR | 26.4 ± 5.1 | 32 (56.14%) - multi- gravida 25 (43.86%) -primigravida |
- Early pregnancy 10 patients (17.54%) Late pregnancy: 13 patients (22.80%) Post partum: 34; 59.64%) |
| 8 | Krishna A et al., [17] | 2015 | Retrospective | Uttar pradesh | 98 (only dialysis requiring patient including) | 3.39% | PRAKI cases requiring dialysis | - | 28 ± 5 |
primigravida − 20(20.4%) Multi gravida 78(79.60%) |
Is trimester − 15(15.3%), IInd trimester − 28(28.57%),IIIrd trimester − 55(56.12%) |
| 9 | Gopalakrishnan N et al., [18] | 2015 | Prospective, Observational | Chennai | 130 | 7.8% | Increase in serum creatinine about 1.5 times from the baseline or when the urine output decreased to less than 400 mL for more than 6 h or both | KDIGO | 25.4 ± 4.7 | 62(47.69%) Multigravida |
Is trimester– 5 (4%) IInd trimester − 11 (9%) IIIrd trimester − 24(19%) Post partum- 88(68%) |
| 10 | Gullipali et al., [19] | 2015 | Prospective observational | Vishakhapatnam | 48 | 10.5% | Postpartum AKI cases | AKIN criterion | 24.75 ± 4.31 years | Primigravida39 (79.16%), multigravida: 12 (20.83) % | Postpartum AKI: 100% |
| 11 | Eswarappa et al., [20] | 2016 | Retrospective, observational | Karnataka | 99 | NR | Postpartum AKI cases | RIFLE criteria | 23 |
Primigravida: 52% Multigravida: 48% |
Postpartum AKI: 100% |
| 12 | Mishra V et al., [21] | 2016 | Prospective observational | Gujrat | 52 | 9.12% | Oliguria(urine output < 400 ml/day) and azotemia (serum creatinine > 2 mg/dl) | NR | 26.2 |
Primigravida 23 (44.23%)- Multigravida: 29 (55.77%)- |
Early pregnancy : 3 (5.76%) Late pregnancy 49 (94.23%)- |
| 13 | Prakash J et al., [22] | 2016 | Retrospective | Pradesh | 259 | 8.35% (of all AKI) |
(1) Serum creatinine > 1 mg/dl, (2) oOiguria/anuria > 12 h duration, and (3) Need for dialysis (Any 1 out of 3) |
None | 29.8 years | NR |
Early pregnancy (1st and second trimester):152 (58.68%) Late pregnancy (3rd trimester + Post partum): 107 (41.31% |
| 14 | Prakash J et al., [23] | 2017 | Observational, prospective | Uttar pradesh | 132 |
(2.78%) [132 out of 4741 pregnant woman– 2.78% had AKI] |
(1) sudden elevation of serum creatinine ≥ 1 mg/dl; (2) oligoanuria for > 12 h; and (3) need for dialysis. | NR | 26.8 ± 4.8 |
Primigravida 56 (42.4%) Multigravida 76 (57.8%) |
Ist trimester − 8 (6.1%) IInd trimester − 3 (1.5.%) IIIrd trimester: 88 (66.6%) Post partam-33 (25%) |
| 15 | Mir M et al., [24] | 2017 | Retrospective | Srinagar | 28 | 4.27% Of all AKI | All PRAKI cases during study period | KDIGO | 26.10 ± 4.3 | Multi gravida 34 (75%) Primi-gravida: 12 (43%) | Only post partum AKI patients were included. |
| 16 | Tanwar et al., [25] | 2018 | Prospective, observational | Rajasthan | 60 | 68 (12.8%) had postpartum AKI. | Postpartum AKI requiring dialysis delivery | Standard indications for dialysis | 26.5 |
Primipara: 25 (41.7%) Multi gravida: 35 (58.3%) |
Postpartum (100%) |
| 17 | Saini et al., [26] | 2020 | Prospective, observational | Kolkata, West Bengal | 69 |
15% incidence of dialysis requiring PRAKI of all dialysis requiring AKI of the institute, one out of 470 pregnant women |
All PRAKI cases during study period | KDIGO criteria | 23.64 | Primigravida: 31 (46%), Multigravida:38 (54%) | First trimester 4 (4.9%), Second trimester 5 (6.17%), Third trimester and postpartum 72 (88.88%) |
| 18 | Chowdhary PK, [27] | 2021 | Retrospective, observational | Raipur | 107 |
3.26% (Only post partum AKI included) |
All postpartum AKI. | KDIGO AKI criteria | 27.3 ± 4.77 years |
Primipara-58 (54.2%) Multipara- 49 (45.8%) |
All postpartum AKI |
| 19 | Banerjee A et al., [28] | 2020 | Prospective study, | Uttar Pradesh | 36 | NR |
• 1) Two rimes increase in serum creatinine from baseline. • 2) Urine output < 0.6 mL/kg in the last12 hrs; or • 3)Urea > twofold from the baseline |
RIFLE criteria. |
Patients divided in two froups Groupt 1: early RRT: 13 (27.34%) Group two: 23 (28.96%) |
NR | NR |
| 20 | Sahay M et al., [29] | 2022 | Retrospective, observational | Hyderabad | 395 | 8.1% | • All PRAKI cases during study period | KDIGO | 27 ± 3 years | Multigravida- 231 (58.6%) |
Postpartam- 288 (72.9%) Antepartam– 107 (27.08%) Before or within 7 days of delivery- 207(52.4%) Between 7–14 days of delivery- 129 (32.6%) ≥ 14 days: 57 (14.4%) |
| 21 | Sachan R et al., [30] | 2022 | Prospective observational | Uttar pradesh | 150 | Out of 14,750 obstetrics pt, 150 (1%), PRAKI | • All PRAKI cases during study period | KDIGO | 20–35 years | - | - |
| 22 | Yadav S et al., [31] | 2022 | Prospective, Observational | Haryana | 51 | NR | • Increase in serum creatinine by ≥ 26.5 µmol/L within 48 h, increase to more than 1.5 times baseline, or reduced urine volume < 0.5 mL/kg/h for six hours. | RIFLE | 25.9 |
Primigravida: 21(41.2%) Multigravida 30(58.8%) |
Third trimester- 21 (50.9%) |
| 23 | Sandilya S, [32] | 2023 | Prospective observational | New Delhi | 50 | NR | • All PRAKI cases during study period | KDIGO | 25.18 ± 3.8 | NR | Postpartum– 12 (24%) |
| 24 | Kharkongor D et al., [33] | 2023 | Prospective observational | Uttar pradesh | 110 | 1.08% | • All PRAKI cases during study period | KDIGO | 27.95 ± 4.85 | Primigravida 34 (30.9%), Multiparous 76(69.1%) | NR |
| 25 | Saxena D et al., [34] | 2024 | Prospective observational | Rajasthan | 62 | 11.8% | • PRAKI patients requiring dialysis | KDIGO | 28 ± 3 | Primigravida 24 (38.7%), Multigravida: 38 (61.3%) | Postpartum: 53 (85.5%) |
Data synthesis
The Review Manager 5.4 (RevMan) Meta-Analysis software, was used for all statistical analyses. Proportions and percentages were calculated for each outcome in the context of PRAKI cases, using the total number of PRAKI patients as the denominator. Trends in maternal and kidney outcomes were assessed across studies, focusing on variations in PRAKI definitions and geographic differences in outcomes. Additionally, narrative synthesis was employed to describe the range of kidney outcomes. No meta-analysis was performed due to heterogeneity in study designs, lack of RCTs and outcome reporting across studies.
Results
Study selection
The initial search yielded 3608 articles, after removing duplicates (n = 32), 3576 studies underwent title and abstract screening. The full texts of 500 articles were retrieved for eligibility assessment, and 474 articles were excluded for the following reasons: outside India (n = 198), not reporting kidney outcome (n = 36), case reports (n = 110), review articles (n = 88), a systematic review from another continent (n = 1), a systematic review on cardiovascular outcomes and aetiology (n = 2), study reporting outcomes only in pre-eclampsia cases (n = 1) and conference abstracts (n = 40). Finally, a total of 2306 patients across 25 studies were included in the final analysis [10–34]., the PRISMA flowchart is provided in Fig. 1.
Baseline characteristics
The incidence of PRAKI in Indian studies has varied, ranging from 1.08 to 12% of AKI cases reported by different centres. Jaiprakash et al. and Sachan et al. reported AKI incidence rates of 2.73% and 1%, respectively, among all pregnancies during their study periods [23, 30]. The baseline characteristics of the included studies are provided in Table 1.
The patient population in the included studies varied from 27 to 395 individuals, with the majority being conducted in government hospitals. However, one study was conducted at a private tertiary care centre [16]. All the studies were single-centre and 8 out of 25 studies were retrospective in design [13, 16, 17, 20, 22, 24, 27, 29]. The third trimester or postpartum period was identified as the most common phase for the presentation of PRAKI with up to 80% of cases occurring during this time. Notably, Najar et al. reported a significant number of early-trimester AKI cases (75%) [11]. There was a general predominance of multigravida patients in the reviewed studies. The criteria for defining AKI were not uniform across the studies; only 9 studies applied the KDIGO AKI criteria for staging [18, 24, 26, 27, 29, 30, 32–34]. In contrast, other studies defined AKI based on oliguria or an increase in serum creatinine levels. Additionally, six studies focused exclusively on postpartum AKI cases [16, 19, 20, 24, 25, 27].
Etiology of PRAKI
Sepsis was found to be the most frequent cause of PRAKI, ranging from 13.9 to 78.8% across different cohorts. (See Table 2) Puerperal sepsis was commonly reported, constituting the primary cause of sepsis in many studies, while post-abortal sepsis also contributed significantly in some. [10, 11, 16, 17], Najar et al. reported that septic abortion was the leading cause of PRAKI accounting for 20 cases (50%), with 15 cases (75%) occurring in the first trimester and 5 cases (25%) in the second trimester. Of 20 patients with septic abortion, 18 (90%) were from rural areas and their abortions had been conducted by untrained midwives [11]. Pre-eclampsia and eclampsia, another significant contributor to PRAKI, appeared frequently in multiple studies, with incidences ranging from 14 to 46.9%. Gopalakrishnan et al. reported a 21% incidence of pre-eclampsia/eclampsia [18], while Prakash et al. found it in 46.9% of cases [21]. In addition, HELLP syndrome was a contributing factor in certain cases, with incidences as high as 44.45% reported by Kharkongor et al. [33] Obstetric hemorrhages such as antepartum haemorrhage (APH) and postpartum haemorrhage (PPH) were also prevalent in many of the studies. Krishna et al. found that 23.4% of patients experienced APH and PPH [17], while Sahay et al. reported a combined incidence of 44.5% [29]. Other studies, such as those by Sandilya et al. and Kharkongor et al., reported significant occurrences of hemorrhage-related complications, which were often associated with sepsis or pre-eclampsia ([32–33]). Glomerular diseases contributed to 0.7 to 5.8% of PRAKI cases. The most common aetiology was lupus nephritis. Several other etiologies were noted across the reviewed studies, including malaria, snake bite, acute gastroenteritis, hemolytic uremic syndrome (HUS), and acute fatty liver of pregnancy (AFLP). Out of the total of 2306 cases of PRAKI, AFLP was only due to 18 cases. Only 3 cases were contributed by obstructive etiology.
Table 2.
Etiologies of pregnancy associated acute kidney injury
| Sl, number | First author | Sepsis (n%) | Sepsis Etiology | APH (n%) | Pre-eclampsia/Eclampsia (n%) | PPH (n%) | Glomerular Disease (n%) | HELLP (n%) | AFLP (n%) | Others with Etiology |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | Goplani KR et al, [10] | 57 (71%) | Puerperal sepsis − 43 and post abortal-14 | 10 (14.28%) | 20 (28.57%) (includes HELLP) | 17 (24.28%) | NR | NR | NR | Malaria (3) (4.28%) |
| 2 | Najar MS et al., [11] | 20 (50%) |
Abortion, Acute pyelonephritis-2 (5%), |
6 (15%) | 6 (15%) | 2 (5%) | NR | NR | NR | AGE-3 (7.5%) |
| 3 | Arora N et al., [12] | 19 (33.3%) | Puerperal sepsis (100%) | - | PE/other hypertensive disorder-7 (12.3%), Eclampsia-8 (14.0%) | 16 (28.1) | NR | NR | NR | Snake bite-2 (3.5%), Perioperative hypotension-1 (1.7%), HUS-1 (1.7%), Unknown-3 (5.3%) |
| 4 | Sivakumar V et al., [13] | 28 (47.41%) | Puerperal sepsis: 28 (47.41%) | 5 (8.47%) | 17 (30.50%) | 6 (10.16%) | NR | NR | NR | Miscellaneous (18.64%) |
| 5 | Patel ML et al. [14] | 25 (41.7%) | Post partum sepsis | 4 (6.66%) | 20 (33.3%) | 4 (6.66%) | - | 1 (1.7%) | - | Abortion-5 (8.3%), DIC-1 (1.7%) |
| 6 | Pahwa N et al., [15] | 19 (70.3%) |
Puerperal sepsis: 9 (29.7%) UTI: 5 (18.5%) |
2 (10%) | 2 (10%) | 1 (6%) | NR | 2 (8.4%) | NA | HUS-2 (9%) |
| 7 | Godara SM et al., [16] | N = 44 (78.8%) | Puerperal sepsis: 44 (63.1%), post-abortion sepsis: (4) 15.7% | 7 (14%) | 14 (33.33%) | 2 (8.7%) | FSGS − 1(1.75%), MPGN-1 (1.75%) | NR | NR | Malaria 3(5.2%), |
| 8 | Krishna A et al., [17] | 55 (56.1%) | Post abortal | PPH + APH-23 (23.4%) | 14 (14.2%) | NR | NR | 4 (4.08%) | HUS-2 (2.04%) | NR |
| 9 | Gopalakrishnan N et al., [18] | 51 (39%) | Septic abortion-4 (7.84%), Puerperal sepsis − 47 (92.15%) | 13 (10%) | 27 (21%) | 3 (2%) | 4 (3%) | 5 (3.84%) | NR | Acute Diarrhea-13 (10%), TMA-11 (9%), Others-3 (2.30%) |
| 10 | Gullipali et al., [19] | 16 (33.33%) | Puerperal sepsis in all cases of sepsis | 2(4.16%) | 12(25%) |
6 (12.5%), |
5 (10.41%) | 12(25%) with pre eclampsia | NR | Complicated malaria: 3(6.25%) |
| 11 | Eswarappa et al., [20] | 74 (75%) |
Puerperal sepsis: 60.6%(n = 61) UTI: 6 (6%) Septic thrombophlebiitis 1 (1%) Necrotizing fascitis 1 (1%) Mucormycosis 1 (1%) |
NR | NR | 12% (n = 14) | LN-1 (1%), | n = 10 (8%) along with preeclampsia | None |
HUS-1 (1%),Transfusion reaction − 1(1%), Isolated postpartum PIH-1 (1%) H1N1: 1 (1%) |
| 12 | Mishra V et al., [21] | 17 (32.6%) |
Post abortal-3 (5.77%) Peurperal-8 (15.38%), Combined hemorrhage and sepsis: 9 (17.30%). |
9 (17.30%) | PE-31 (59.6%), Eclampsia-4 (7.69%) | 11 (21.15%) | - | 1 (1.92%) | 1 (1.92%) | Malaria, IUD with sepsis |
| 13 | Prakash J et al., [22] | 39 (15%) | Puerperal sepsis: 39 (15%) | (APH + PPH) 25 (9.65%) | PE + HELLP: 41 (15.8%) | NR | LN: 2 (0.07%) | NR | 1 (0.06%) | 152 (58.68%) Post abortal, HUS: 1 (0.06%) |
| 14 | Prakash J et al., [23] | 42 (31.8%) | Puerperal sepsis-34 (25.8%), Post abortal sepsis-8 (6.1) | 11 (8.3%) | 62 (46.9%) | 28 (21.2%) | 1 (0.75%)-LN | 9 (6.8%) | 5 (3.8%) | Fulminant hepatitis-4 (3.03%), Acute gastroentritis-3 (2.27%), UTI-2 (1.5%), Complicated malaria-1 (0.75%) |
| 15 | Mir M et al., [24] | 11 (39.28%) |
Puerperal sepsis: 11 (39.28%) Acute pyelonephritis-3 (11%) |
NR | 4 (14%) | 7 (25%) | ANCA-1 (4%), | NR | NR | HUS-1 (4%) |
| 16 | Tanwar et al., [25] | 31(61.6%) | Puerperal sepsis: 31(61.6%) | 14 (23.3%) | 21 (35%) | 7 (11.7%) | NR | 3 (5%) | 4(6.7%) |
Tropical infections, malaria- 02 (5%), TMA − 2 (3.3%), Hemorrhage in early pregnancy (8.7%) Dengue hemorrhagic fever, N = 1 |
| 17 | Saini et al., [26] | 42 (49.4%) | Puerperal sepsis: 41 (49%) | NR | 13 (12.3%) | 12 (16%)) | NR | 3 (4.9%) | 2 (2.4%) |
Malaria (1.2%), Unexplained (1.2%) P-aHUS: 14 (17.28%) |
| 18 | Chowdhary PK, [27] | 35 (32.71%) | Puerperal sepsis, RPOC-6 (17.14%), UTI-22 (62.85%), Pneumonia-6 (17.14%), Abdominal wound-5 (14.28%), Episiotomy wound-3 (8.57%) | NR | 8 (7.47%) | 4 (3.73%) | 2 (1.86%) | NR | 1 (0.93%) | HUS, N = 1 |
| 19 | Banerjee A et al., [28] | 5 (13.9%) |
Bacterial sepsis: 5 (13.39) Viral sepsis: 3 (3.33%) |
NR | 13 (36.11%) | Total 4 (11.11%) hemorrhage cases | None | 1 (2.72%) | 2 (5.56%) |
Hyperemesis gravidarum: 2 (5.56%), Malaria: 4 (11%), Dengue:1 (2.72%), Snake bite: 1 (2.72%) |
| 20 | Sahay M et al., [29] | 132 (33.41%) | NR | 31 (8%) | 176 (44.5%) | 46 (12%) | 11 (2.7%) | GN 11 (10.6%) | NR |
9 (8.7%)-HUS Obstruction: n = 2 |
| 21 | Sachan R et al., [30] | 68 (45.3%) |
Puerperal sepsis, n = 64 (42.6) Septic abortion, 4 (2.6%) |
15 (10%) |
HDP: 72 (48%) PE-30 (20%) / EC-42 (28%) |
39 (26%) | NR | NR | NR |
AGE: n = 2, Liver dysfunction n = 11, Heart diseases, n = 2 Caogulopathy, n = 25 |
| 22 | Yadav S et al., [31] | 21(18.1%) | Puerperal Sepsis | - | PE- 14 (39%) Eclampsia– 8(13%) | NR | NR | 6 (16.6%) | NR | AGE − 01 (3%) |
| 23 | Sandilya S, [32] | 12 (24%) |
Puerperal sepsis: 12 (24%) Pyelonephritis: 2 (4%) |
Abruption:7(14%) | 14 (28%), | 10 (20%) | NR | NR | NR | TMA: 1 (2%) |
| 24 | Kharkongor D et al., [32] | 31 (28.18%) | Puerperal + postabortal + sepsis: 31 (28.18%) | APH + PPH + post abortal: 39 (35.45%) | PE/Eclampsia/HELLP: 49 (44.45%) | APH + PPH 49 (44.54%) | NR | PE/Eclampsia/HELLP: 49 (44.45%) | 12 (10.91%) | Others: 3 (2.72) |
| 25 | Saxena D et al., [34] | 20 (29.4%) | NR | 1 (1.4%) | 9 (13.2%) | 18 (26.4%) | 4 (5.8%) | 2 (2.9%) | NR | Obstructive uropathy, n = 1 (1.4%) |
AFLP: Acute fatty liver of pregnancy, HUS: hemolytic uremic syndrome, APH: Antepartum haemorrhage, PPH: Antepartum haemorrhage, HELLP: Hemolysis, Elevated Liver enzymes and Low Platelets, RPOCs: retained products of conception, PE: Pre-eclampsia, TMA: Thrombotic microangiopathy, AGE: Acute Gastroenteritis, UTI: Urinary tract infection, HDP: Hypertensive disorders of pregnancy
Renal outcome
Renal outcomes in PRAKI varied considerably across studies, with a significant proportion of patients requiring renal replacement therapy (RRT) up to 85%, including hemodialysis (HD) and peritoneal dialysis (PD), as well as long-term impacts such as chronic kidney disease (CKD) and dialysis dependency. (Table 3). In the majority of the studies, hemodialysis modality was used, in only 3 studies use of PD was reported [18, 22, 23]. For instance, in the cohort by Goplani et al., 97.14% of patients underwent HD, and 85.9% achieved full recovery, though 12.85% progressed to CKD, and 14.28% became dialysis dependent [10]. Similarly, Najar et al. found that 40% of their patients required dialysis, with 72.5% achieving full recovery, while only 5% developed CKD [11].
Table 3.
Renal outcomes HD: hemodialysis, ATN: acute tubular necrosis, ATIN: acute tubulointerstitial nephritis, ACN: acute cortical necrosis, TMA: thrombotic microangiopathy, CRRT: continuous renal replacement therapy, FSGS: focal and segmental glomerulosclerosis, MPGN: membranoproliferative glomerulonephritis, MGN: membranous glomerulonephritis, PD: peritoneal dialysis, LN: lupus nephritis, GN: glomerulonephritis, CVVHDF: continuous venovenous hemodiafiltration,
| Sl, number | First author | Renal biopsy | RRT modality | Dialysis requirement | Full revovery | CKD development | Dialysis dependant |
|---|---|---|---|---|---|---|---|
| 1 | Goplani KR et al, [10] |
Performed in 11 (N = 11)patients ACN: 10 Glomerular endotheliosis-1(associated with preeclampsia) |
89, (97.14)% -HD 8 (8.6%) patients received PD initially and later shifted to HD. |
49 (85.9%) | 38 (54.28%) | 9 (12.85%) | 10(14.28%) |
| 2 | Najar MS et al., [11] | NR | HD: 13 (32.5%) PD: 6(15%) and both modalities to 5 (12.5%) | 24(40%) | 29 (72.5%) | 2 (5%) | 1 (2.5%) |
| 3 | Arora N et al., [12] |
Renal Biopsy in patients ATN: 2, ACN: 3, subacute interstitial nephritis:1 Inconclusive 1 |
100% HD | 49 (6.9%) | 24 (50) | 5 (8) | 3(5.2) |
| 4 | Sivakumar V et al., [13] | NR | HD 29 (49.15%), CVVHD 4 (6.77%), PD 3 (3.38%) | 35 (59.3%) | 31 (54.23%) | 6 (10.16%) | NR |
| 5 | Patel ML et al. [14] | NR | Hemodialysis- 23 (38.33%) | 23(38.33%) | 45 (75%) | 1 (1.7%) | 5 (8.3%) |
| 6 | Pahwa N et al., [15] |
N = 11 (18.5%) ACN 5, |
All HD | 25 (92%) | 11(40. 7%) | 6(22.2%) | 2(7.4%) |
| 7 | Godara SM et al., [16] | N = 21 (36.84%) ACN: n = 13 (61.9%), ATN n = 4, Glomerular disease (n = 3,) FSGS-1. MPGN-1 MGN-1 ATIN-1 | HD: 50 (87.7%), PD:3 (5.2%) | 50 (87.7%) | 50 (52.64%) | 12 (21.05%) | 15 (26.31%) |
| 8 | Krishna A et al., [17] |
N = 16 ACN- 8 ACN(patchy) + TMA − 6 ATN − 2 |
HD: 98 (100%) |
24 (24.49%)1 session HD 74 (75.5%)-multiple sessions |
53 (54.28%) | 13 (12.85%) | 14 (14.28%) |
| 9 | Gopalakrishnan N et al., [18] |
N = 46 (35.3%), ACN: 16 (34.7%), TMA: 11 (23.9%), ATN: 9 (19.5), ATIN: 1 Glomerular disease- 9 (Lupus nephritis 4 IgA nephropathy 1 Diffuse mesangial proliferation 1 Focal segmental glomerulosclerosis 1 Pauci-immune necrotising GN 1 Infection-related GN- 1) |
All HD | 96 (74%) | 73 (56%) | 46 (35%) | 1(7.69%) |
| 10 | Gullipali et al., [19] |
N = 15 (31.2%) ACN: 6 (12.5) LN: 4 (8.3) TMA: 3 (6.24%) FSGS: 1 (2.08%) ATN: 1 (2.08%) |
HD in all patients | 23(47%) | 38 (79.4%) | 2 (4.1%) | 4(8.3%) |
| 11 | Eswarappa et al., [20] | NR | HD: 29 (29%), PD:6(6%), CRRT: 4 (4%) | 39 (39%) | 70 (70%) | 5 (5%) | 3 (3%) |
| 12 | Mishra V et al., [21] |
N = 10 ACN- 8 ATN- 2 |
All HD | 43 (84.62%) | 29 (55.76%) | 19 (50%) | 4(7.69%) |
| 13 | Prakash J et al., [22] |
N = 18 Diffuse ACN 8 Patchy ACN 7 Glomerular disease-2, both LN Changes of CKD- 1 |
All HD | 223 (86.1%) | 199 (76.83%) | 28 /259 (10.8%) | 7 (2.7%) |
| 14 | Prakash J et al., [23] |
N = 4 ACN: N = 3, LN: N = 1 |
All HD | 62 (47%) | 118 (89.4%) | 8(6.06%) | 6 (4.6%) |
| 15 | Mir M et al., [24] |
N = 9 (14.7) ACN n = 4(44.44). ATN: n = 3 (33.33) Glomerular disease: 1 (pauci-immune GN)(11.11), TMA: 1 (11.11) |
9 (33%) HD and 4 (14%)-PD |
13(46%) | 15(54%) | 6(21%) | 4(14%) |
| 16 | Tanwar et al., [25] |
N = 11 (18.3%) ACN: 6, ATN: 2 TMA: 2 Inconclusive: 1 |
HD:54 (90%), PD: 25(41.7%), | 100% | NR | 31 (51.3%) | 2(5.4%) |
| 17 | Saini et al., [26] |
N-24(30%) TMA: 13 (54%) ACN:4 (17) ATN: 6(25) Diffuse Glomeruloscleorsis: 1 (4%) |
HD: 68 (84%) | 68 (84%) | 27 (39%) | 23 (33%) | 11 (16%) |
| 18 | Chowdhary PK, [27] |
N = 7(6.54%) patients ATN: n = 3 ACN: N = 3, and TMA: n = 1 |
All HD | 73 (68.22%) | 75 (88.2%) | 6(7.05%) | 4(4.7%) |
| 19 | Banerjee A et al., [28] | NR | All HD | Only dialysis patients included | NR | NR | Total n = 3 (8.3%) Early RRT group-2(15.38%), Standard RRT group-1(4.35%) |
| 20 | Sahay M et al., [29] |
N = 103 Patchy cortical necrosis 25 (22.3%), Diffuse ACN 23 (20.3%), ATN 20 (19.4%), AIN 10 (9.7%), TMA 9 (8.7%), GN 11 (10.6%), IgA n = 4, MPGN, n = 2, FSGS n = 2, LN n = 3, endotheliosis, n = 2 |
All HD | 290(73.6%) | 285 (76%) | 59 (15.7%) | 31 (8.3%) |
| 21 | Sachan R et al., [30] | NR | HD | 98(65.33%) | 41(27.3%) | 46(31.3%) | 5(3.39%) |
| 22 | Yadav S et al., [31] | NR | NR | 14 (27.5%) | 33 (64.7%) | 6 (12%) | NR |
| 23 | Sandilya S, [32] | NR | HD | 23 (46%) | 40 (80%) | 10 (20%) | NR |
| 24 | Kharkongor D et al., [32] | NR | HD | 29 (26.4%) | 45(40.9%) | 25 (22.7%) | 10 (9.1%) |
| 25 | Saxena D et al., [34] |
N = 33 Diffuse ACN-14 Patchy ACN − 6 ATN − 9 Glomerular disease − 4 |
HD | 62 (72.1) | 21 (38.2%) | 15 (30%) | 16 (32%) |
Outcomes also varied significantly regarding CKD development and dialysis dependency. For example, Prakash et al. found that 76.83% of patients fully recovered, though 10.8% progressed to CKD [22]. Gopalakrishnan et al. identified 35% of patients developing CKD, with 7.69% becoming dialysis dependent [18]. Mir et al. noted a CKD development rate of 21% [24], while Pahwa et al. reported a 22.2% progression to CKD among their patients [16]. Tanwar et al. found that 51.3% of patients progressed to CKD. Saxena D et al. also reported a very high rate of CKD development in 15 (30%), and 16 (32%) remaining dialysis dependant [35].
In terms of non-recovery of renal function, patients who did not recover renal function by six weeks exhibited a substantially elevated risk of prolonged non-recovery, with an RR of 24.7 (95% CI: 3.4–179.5) [17]. Saini et al., in their multivariate analysis, identified TMA as a significant predictor of non-recovery, with an odds ratio (OR) of 14.00 (95% CI: 1.13–172.58, P = 0.039). Other important but non-significant predictors included obstetric haemorrhage (OR: 7.50, 95% CI: 0.62–90.61, P = 0.11) and sepsis (OR: 4.28, 95% CI: 0.44–41.35, P = 0.20) [24]. Sahay et al. further highlighted that clinical variables such as anuria, sepsis, TMA, and findings of cortical necrosis on renal biopsy were crucial predictors of non-recovery of renal function in PRAKI patients [20].
Renal biopsy data
Renal biopsy was reported in 346 (15%) of patients. (Fig. 2). The most common finding was acute cortical necrosis (ACN) in 172 (49.71%) followed by acute tubular necrosis in 63 (18.2%), TMA in 47 (13.5%), GN in 36 (10.4%) and ATIN in 13 (3.75%). The most common GN was LN (N = 14), followed by IGAN (N = 5), FSGS (N = 5), MPGN (N = 3), membranous nephropathy (N = 1), ANCA vasculitis (N = 2) and infections related GN (N = 1). Changes in endotheliosis were reported in 3 patients.
Fig. 2.
Kidney biopsy findings in cases of Pregnancy-related acute kidney injury
Fetomaternal outcome
The fetomaternal outcomes in PRAKI in India demonstrate significant variability across 25 different studies. (Table 4). Maternal mortality ranged from 2.5 to 34%. Larger studies, such as Sachan et al. reported a maternal mortality rate of 34% [30] Patel et al. observed a 15% maternal mortality rate [14], with Pahwa et al. reported 18.51% [16]. Several other studies also reflected similar mortality rates. Perinatal outcomes were also significantly impacted. For example, Krishna et al. reported a perinatal mortality rate of 26.19% [17], Mishra et al. observed fetal mortality as high as 67.3% [21]. The adverse fetal outcomes reported by Gopalakrishnan et al., which included intrauterine death (IUD) and stillbirths, were 54% [16]. Pre-term delivery was reported in several studies, with rates ranging from 13.9 to 58%. For example, Sandilya S and Eswarappa reported very high rates of premature deliveries with rates of 40%, and 58% respectively [18, 30]. Krishna et al. [17]. reported several significant risk factors associated with increased mortality in patients with PRAKI. The risk of mortality was markedly higher in individuals with oliguria, sepsis, central nervous system involvement, hepatic dysfunction, and low platelet counts. Additionally, patients requiring multiple dialysis sessions had a significantly higher risk, with a relative risk (RR) of 2.96 (95% CI: 1.75–4.99, P = 0.001).
Table 4.
Maternal and fetal outcomes
| Sl, number | First author | Maternal mortality (N%) | Requirement of ICU (N%) | Perinatal mortality (N%) | IUD (N%) | Pre-mature deliveries, (N%) |
|---|---|---|---|---|---|---|
| 1 | Goplani KR et al, [10] | 13 (18.57%) | NR | NR | NR | NR |
| 2 | Najar MS et al., [11] | 8 (20%) | NR | NR | NR | NR |
| 3 | Arora N et al., [12] | 16 (28.1%) | NR | NR | NR | NR |
| 4 | Sivakumar V et al., [13] | 14 (23.72%) | NR | NR | NR | NR |
| 5 | Patel ML et al. [14] | 9(15%) | NR | NR | NR | 25 (41.7%) |
| 6 | Pahwa N et al., [15] | 5 (18.51%) | NR | NR | NR | NR |
| 7 | Godara SM et al., [16] | 9 (15.78%) | NR | 28 (49.12%) | 3 (5%) | |
| 8 | Krishna A et al., [17] | 18 (18.36%) | 13 (13.8%) | 25 (26.19%) | NR | NR |
| 9 | Gopalakrishnan N et al., [18] | 10 (7.69%) | NR | Adverse fetal outcome (IUD + stillbirth) 70 (54%) | 29 (23%) | NR |
| 10 | Gullipali et al., [19] | 4 (8.3%) | NR | 7 (14.7%) | 11 (22.9%) | |
| 11 | Eswarappa et al., [20] | 18 (19%) | 99 (100%) | 14 (22%) | NR | NR |
| 12 | Mishra V et al., [21] | 17(32.69%) | 15 (28.85%) | Fetal mortality − 67.3% | 17 (32.69%) | NR |
| 13 | Prakash J et al., [22] | 25 (9.60%) | NA | 117 (45.17%) | NR | NR |
| 14 | Prakash J et al., [23] | 8 (6.1%) | NR | 30 (23%) | 23 (17.5%) | 54 (40.9%) |
| 15 | Mir M et al., [24] | 3 (11%) | NR | 3 (11%) | 2 (7.15%) | NR |
| 16 | Tanwar et al., [25] | 17 (28.3%) | 17 (31.7%) | NR | 14 (23.3%) | NR |
| 17 | Saini et al., [26] | 20 (25%) | NR | 19 (23.5%) | NR | NR |
| 18 | Chowdhary PK, [27] | 22 (20.56%) | NR | 15 (14.01%) | NR | NR |
| 19 | Banerjee A et al., [28] | 1 (2.7%) | 36 (all in ICU) | Total n = 14 (38.8%), Early RRT: 2 (14.29%), Standard: 12 (85.71%) | NR |
18 (50%) , Early 8 (44.44%), Standard 10 (55.56%) |
| 20 | Sahay M et al., [29] | 20 (5%) | NR | 30 (7.5%) | 42 (10.6%) | 45 (13.9%) |
| 21 | Sachan R et al., [30] | 51 (34%) | 149 (96.1%) | 56 (38.4%) (IUD + Still births) | 56 (38.4%) | 34 (23.3%) |
| 22 | Yadav S et al., [31] | 14 (23.5%) | 18 (33.3%) | 19 (37.3%) | 20 (39.2%) | 8 (15.7%) |
| 23 | Sandilya S, [32] | 7 (14%) | 50 (100%) | 6 (16%) | 12 (24%) | 20 (40%) |
| 24 | Kharkongor D et al., [32] | 22 (20%) | NR | NR | NR | NR |
| 25 | Saxena D et al., [34] | 9 (14.5%) | NR | NR | 16 (25.8%) | 12 (19.4%) |
IUD: Intrauterine deaths, ICU: Intensive care unit, RRT: Renal replacement therapy, NR: Not reported/not clear
Figure 3 shows the visual graphical summary of the findings of the analysis.
Fig. 3.
Summary of the article
Discussion
The present review of 25 Indian studies, comprising over 2306 patients, highlights significant findings concerning PRAKI with incidence varying from 1 to 12% at different centres. The third trimester and postpartum period were identified as the most common times for the onset of PRAKI, accounting for up to 80% of cases. Septic abortion in the early trimester, often linked to care by untrained midwives, is a significant yet preventable cause of PRAKI [11].
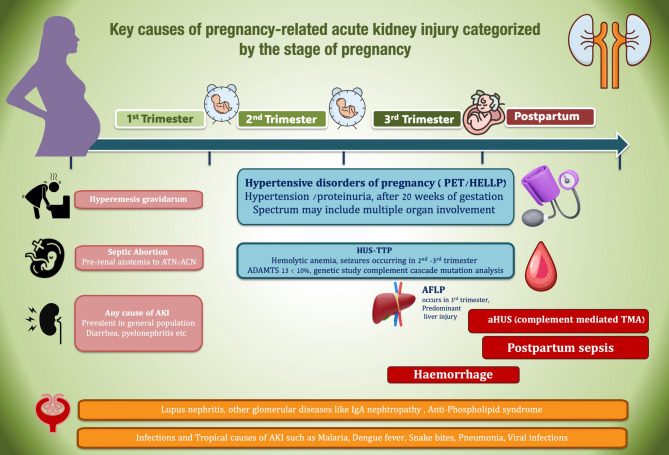
Sepsis emerged as the most frequent cause of PRAKI, with an incidence ranging from 13.9 to 78.8% across various cohorts. Puerperal sepsis was often cited as the leading cause, though post-abortal sepsis also contributed significantly, particularly in rural areas. The decline in septic abortions over recent years is likely due to improved policies and healthcare access, yet a significant proportion of sepsis cases were still associated with unsafe abortions carried out by traditional birth attendants. Pre-eclampsia and eclampsia were other significant contributors to PRAKI, with reported incidences ranging from 14 to 46.9%. Obstetric hemorrhages, were also the common cause of PRAKI. While the most common causes of PRAKI—sepsis, hypertensive disorders of pregnancy (HDP), and hemorrhage—account for more than 50% of cases, less common causes such as glomerulonephritis, HELLP syndrome, AFLP and HUS were also noted. None of the studies reported thrombotic thrombocytopenic purpura (TTP) or complement-mediated disorders as causes of PRAKI nor did they provide any genetic information related to these conditions. This suggests a potential underdiagnosis of such disorders, likely due to financial constraints and limited access to advanced diagnostic tools. Figure 4 presents common etiologies of PRAKI in developing countries. Figure 5 provides key causes of PRAKI categorized by the stage of pregnancy.
Fig. 4.
Etiology of pregnancy-associated acute kidney injury
Fig. 5.
Key causes of pregnancy-related acute kidney injury categorized by the stage of pregnancy. ATN: Acute tubular necrosis, ACN Acute cortical necrosis, AKI; Acute kidney injury, Acute cortical necrosis, PE: Preeclamptic toxemia, HELLP: Hemolysis, elevated liver enzymes, low platelets; AFLP: Acute fatty liver of pregnancy; APS: Antiphospholipid antibody syndrome, TMA: Thrombotic microangiopathy, TTP Thrombotic thrombocytopenic purpura, aHUS Atypical hemolytic uremic syndrome, POC: Product of conception
Renal outcomes varied significantly. Up to 85% of PRAKI patients required RRT. Some studies like by Mir et al., Pahwa et al. and Tanwar et al. reported very high rates of CKD development in 25 to 50% of PRAKI cases [16, 24, 25] Saxena D et al. also reported a very high rate of CKD development in 15 (30%), and 16 (32%) remaining dialysis dependant [34].
This highlights the need for long-term follow-up to monitor renal function and manage the progression to ESKD. Maternal outcomes were similarly concerning, with mortality rates ranging from 2.5 to 34%. Larger studies, such as those by Sachan et al. and Patel et al., reported maternal mortality rates of 34% and 15%, respectively [14, 30]. Pre-term deliveries are common, with incidences ranging from 13.9 to 58%.
The incidence of PRAKI in pregnancy in India has shown a decline over the years, with older studies reporting rates as high as 12%, while more recent data, such as the 2023 study by Kharkongor et al., report an incidence of only 1.08% [33]. This reflects improvements in antenatal care and healthcare awareness among pregnant women. Despite these advances, sepsis remains the predominant cause of PRAKI in many developing regions. The shift from post-abortal sepsis as the leading cause to puerperal sepsis reflects improvements in antenatal care but also highlights the persistent risk of sepsis following childbirth.
The low socioeconomic status of the majority of patients, particularly in rural areas (most of the included centres cater to low socioeconomic groups and rural populations), remains a major barrier to adequate prenatal care and timely treatment. Some studies reported that a substantial percentage of patients had not received any antenatal care, and many deliveries were attended by untrained traditional birth attendants. For instance, Patel et al. found that 53.3% of patients had no antenatal care, with the majority delivering at home [14]. The lack of antenatal care significantly contributes to the burden of PRAKI, as early detection and management of complications such as sepsis, haemorrhage, and hypertensive disorders are critical for improving outcomes.
The high rates of CKD progression show the long-term renal damage that can follow an AKI episode during pregnancy, particularly in cases complicated by sepsis, preeclampsia, or obstetric haemorrhage. The progression to CKD is concerning not only because of its immediate health implications for women but also due to its contribution to the broader CKD burden in India. The country is already burdened with an escalating CKD epidemic, driven by factors such as diabetes, hypertension, newly recognized hot spots for CKD of unknown etiology (CKDu) and poor access to healthcare. PRAKI, which affects women of lower socioeconomic status—often those with limited access to healthcare access adds another dimension to this epidemic. Reports indicate an age-adjusted incidence of ESKD in India as 229 per million population, more than double the previously estimated rate of 100 per million. India now has over 130,000 patients receiving dialysis, with the number steadily rising [35]. The findings from this review suggest that PRAKI is a critical, yet underrecognized, factor in this epidemic, contributing both to the growing number of CKD cases and to the increasing demand for RRT. The strain on healthcare resources due to PRAKI-associated CKD is likely to grow as more women survive acute episodes but remain at risk for long-term renal impairment. Without proper follow-up and management, these women may require lifelong dialysis or kidney transplantation, both of which are limited for socioeconomically challenged groups.
A systematic review from African countries identified preeclampsia, obstetric haemorrhage, and sepsis as the primary causes of PRAKI with maternal mortality ranging between 0% and 34.4%. While most women required ICU admission and HD, renal recovery was achieved in 53.1–90% of cases. Perinatal mortality was also significant, ranging from 1.5 to 60.5% [36].
Studies from other countries report that the prevalence of PRAKI ranges from 2 to 8% in hypertensive disorder cohorts and from 5 to 18% in preeclampsia cases. In severe cases of eclampsia that require intensive care, PRAKI incidences can reach as high as 67%. Preeclampsia complicated by PRAKI significantly increases maternal mortality rates (3%) compared to preeclampsia without PRAKI (0.5%) [3]. However, in India, sepsis remains the predominant cause of AKI, although there has been a shift towards hypertensive disorders of pregnancy (HDP) and hemolytic uremic syndrome (HUS) as major causes.
One of the key strengths of our systematic analysis is the comprehensive inclusion of 25 studies from across India, involving more than 2300 patients, which provides a broad representation of the burden of PRAKI in a developing country context. This review draws attention to the significant variability in PRAKI aetiology, clinical presentation, renal biopsy findings and outcomes. By synthesizing data across diverse cohorts, our analysis emphasizes the long-term impact of PRAKI on the CKD epidemic, particularly its contribution to CKD progression and dialysis dependency. Moreover, our review identifies critical gaps in PRAKI diagnosis and follow-up, highlighting the need for standardized diagnostic criteria and long-term monitoring. The focus on socioeconomic and healthcare disparities provides a valuable lens for understanding the disproportionate impact of PRAKI in low-resource settings, further strengthening the relevance of our findings to public health and policy interventions.
While this systematic review sheds light on PRAKI in India, it has limitations. The heterogeneity of the studies, single-centre analysis, and some retrospective studies limit the robustness of the findings. Furthermore, the absence of data from private sector hospitals and urban health facilities likely skews the understanding of PRAKI incidence and outcomes. The lack of meta-analysis due to study heterogeneity limits the ability to draw conclusive generalizations from the data. A major limitation of the reviewed studies was the heterogeneity in defining PRAKI. Only 9 studies used the Kidney Disease: Improving Global Outcomes (KDIGO) criteria, while others relied on less specific measures such as serum creatinine levels or oliguria or previously used AKIN/RIFFLE criterion. This inconsistency in diagnostic criteria makes cross-study comparisons challenging and may lead to underreporting or misclassification of PRAKI. The reported incidence rates of pregnancy-related PRAKI in India, ranging from 1–12%, lack scenario-specific contextualization, as the data predominantly originate from academic tertiary centres. This sampling introduces an ascertainment bias, as lower-risk pregnancies managed in smaller facilities or community settings, particularly in rural areas, are underrepresented or missing. Consequently, these reported incidence rates are descriptive and not broadly generalizable. Moreover, the definition of PR-AKI is not standardized, with no international consensus on creatinine-based diagnostic criteria, given the physiological fluctuations in serum creatinine levels throughout pregnancy. The absence of uniform definitions also raises concerns about the accuracy and comparability of outcomes, highlighting the need for standardized diagnostic protocols in PR-AKI research. Most patients lack baseline pre-pregnancy creatinine measurements or consistent serial data throughout uncomplicated pregnancies, further complicating accurate diagnosis. As a result, there is a risk that a portion of patients classified as having ‘PR-AKI’ may have undiagnosed CKD, especially amid the rising CKD burden in India. Additionally, cases of mild PR-AKI may go undetected due to infrequent creatinine monitoring, leading to underestimation in analyses.”
Early detection and timely interventions are crucial to reducing these fatal outcomes, but the current healthcare infrastructure and resources in many developing countries are insufficient to meet this need. The burden of PRAKI, particularly in developing countries like India, necessitates urgent attention from healthcare authorities and professionals. The management of PRAKI requires a multidisciplinary approach. Specialized obstetric nephrology clinics could provide early diagnosis and long-term management for both mothers and their infants. Public awareness campaigns, improved access to prenatal care, safer abortion services, and delivery by skilled healthcare professionals will be instrumental in reducing the incidence of PR-AKI and its related complications. Additionally, strengthening communication and referral systems in rural areas, and establishing more obstetric critical care units at tertiary centres, may reduce mortality associated with PR-AKI, particularly in resource-poor settings.
Our review also highlights the critical need for community awareness programs to educate women about the importance of antenatal care, safe delivery practices, and the potential long-term consequences of PRAKI. Improved access to healthcare resources, including well-trained personnel and modern diagnostic tools, is essential to managing high-risk pregnancies effectively.
Conclusion: While the incidence of PRAKI in India has decreased due to better healthcare access and awareness, the condition continues to pose a significant risk to maternal and fetal health. CKD following an AKI episode during pregnancy poses a significant burden to the rising number of CKD patients in the health care system. Further research, particularly large multicenter studies, is needed to better understand the risk factors and outcomes of PRAKI and to improve strategies for early detection, management, and prevention.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgements
none.
Abbreviations
- ATN
Acute tubular necrosis
- PRAKI
Pregnancy-associated Acute kidney injury
- ACN
Acute cortical necrosis
- AKI
Acute kidney injury
- CKD
Chronic kidney disease
- ESKD
End-stage kidney disease
- KDIGO
Kidney Disease: Improving Global Outcomes
- LM
Light microscopy
- AFLP
Acute fatty liver of pregnancy
- HUS
hemolytic uremic syndrome
- APH
Antepartum haemorrhage
- PPH
Antepartum haemorrhage
- HELLP
Hemolysis, Elevated Liver enzymes and Low Platelets
- RPOCs
retained products of conception
- PE
Pre-eclampsia
- TMA
Thrombotic microangiopathy
- HDP
Hypertensive disorders of pregnancy
- HD
Haemodialysis
- RRT
Renal replacement therapy
- TMA
Thrombotic microangiopathy
Author contributions
PM conceptualised the idea and drafted the manuscript. VS, PR, UK and AM collected the data. SP and VB verified the data. KM and AA reviewed and edited the manuscript. PM interpreted the data and edited the manuscript. All authors reviewed the manuscript.
Funding
None.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Davidson B, Bajpai D, Shah S, Jones E, Okyere P, Wearne N, Gumber R, Saxena N, Osafo C. Pregnancy-Associated acute kidney injury in Low-Resource settings: progress over the last decade. Semin Nephrol. 2022;42(5):151317. 10.1016/j.semnephrol.2023.151317. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Gautam M, Ahmed A, Mishra P, Azim A, Ahmad A, Dandu H, Agrawal, Avinash, Atam, Virendra, Jaiswar. Shyam. Maternal mortality due to Pregnancy-Related acute kidney injury (PRAKI); A study of the epidemiological factors and possible solutions. J Obstet Gynecol India. 2024;21(1):112. 10.1007/s13224-024-01942-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Liu Y, Ma X, Zheng J, Liu X, Yan T. Pregnancy outcomes in patients with acute kidney injury during pregnancy: a systematic review and meta-analysis. BMC Pregnancy Childbirth. 2017;17(1):235. 10.1186/s12884-017-1402-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Trakarnvanich T, Ngamvichchukorn T, Susantitaphong P. Incidence of acute kidney injury during pregnancy and its prognostic value for adverse clinical outcomes: A systematic review and meta-analysis. Med (Baltim). 2022;101(30):e29563. 10.1097/MD.0000000000029563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Prakash J, Prakash S, Ganiger VC. Changing epidemiology of acute kidney injury in pregnancy: A journey of four decades from a developing country. Saudi J Kidney Dis Transpl 2019 Sep-Oct;30(5):1118–30. 10.4103/1319-2442.270268 [DOI] [PubMed]
- 6.Prasad N, Jaiswal A, Meyyappan J, Gopalakrishnan N, Chaudhary AR, Fernando E, Rathi M, Singh S, Rajapurkar M, Jeloka T, Kishun J, Lobo V. Community-acquired acute kidney injury in india: data from ISN-acute kidney injury registry. Lancet Reg Health Southeast Asia. 2024;21:100359. 10.1016/j.lansea.2024.100359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Rao S, Jim B. Acute kidney injury in pregnancy: the changing landscape for the 21st century. Kidney Int Rep. 2018;3(2):247–57. 10.1016/j.ekir.2018.01.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, Mulrow CD, Shamseer L, Tetzlaff JM, Akl EA, Brennan SE, Chou R, Glanville J, Grimshaw JM, Hróbjartsson A, Lalu MM, Li T, Loder EW, Mayo-Wilson E, McDonald S, McGuinness LA, Stewart LA, Thomas J, Tricco AC, Welch VA, Whiting P, Moher D. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews. BMJ. 2021;372:n71. 10.1136/bmj.n71. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wells GA, Shea B, O’Connell D et al. The Newcastle-Ottawa Scale (NOS) for assessing the quality of nonrandomized studies in meta-analyses. http://www.ohri.ca/programs/clinical_epidemiology/oxford.asp. Accessed January 11, 2016.
- 10.Goplani KR, Shah PR, Gera DN, Gumber M, Dabhi M, Feroz A, Kanodia K, Suresh S, Vanikar AV, Trivedi HL. Pregnancy-related acute renal failure: A single-center experience. Indian J Nephrol. 2008;18(1):17–21. 10.4103/0971-4065.41283. PMID: 20368915; PMCID: PMC2847724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Najar MS, Shah AR, Wani IA, Reshi AR, Banday KA, Bhat MA, Saldanha CL. Pregnancy related acute kidney injury: A single center experience from the Kashmir Valley. Indian J Nephrol. 2008;18(4):159–61. 10.4103/0971-4065.45291..PMID: 20142928; PMCID: PMC2813539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Arora N, Mahajan K, Jana N, Taraphder A. Pregnancy-related acute renal failure in Eastern India. Int J Gynaecol Obstet. 2010;111(3):213–6. [DOI] [PubMed] [Google Scholar]
- 13.Sivakumar V, Sivaramakrishna G, Sainaresh VV, Sriramnaveen P, Kishore CK, Rani CS, Jagadeesh K. Pregnancy-related acute renal failure: a ten-year experience. Saudi J Kidney Dis Transpl. 2011;22(2):352-3. PMID: 21422644. [PubMed]
- 14.Patel ML, Sachan R, Radheshyam, Sachan P. Acute renal failure in pregnancy: tertiary centre experience from North Indian population. Niger Med J. 2013 May;54(3):191–5. 10.4103/0300-1652.114586. PMID: 23900700; PMCID: PMC3719246. [DOI] [PMC free article] [PubMed]
- 15.Pahwa N, Bharani R,,Kumar R. Post-Partum acute kidney injury. Saudi J Kidney Dis Transpl. 2014;25(6):1244–7. [DOI] [PubMed] [Google Scholar]
- 16.Godara SM, Kute VB, Trivedi HL, Vanikar AV, Shah PR, Gumber MR, Patel HV, Gumber VM. Clinical profile and outcome of acute kidney injury related to pregnancy in developing countries: a single-center study from India. Saudi J Kidney Dis Transpl. 2014;25(4):906– 11. 10.4103/1319-2442.135215. PMID: 24969215. [DOI] [PubMed]
- 17.Krishna A, Singh R, Prasad N, Gupta A, Bhadauria D, Kaul A, Sharma RK, Kapoor D. Maternal, fetal and renal outcomes of pregnancy-associated acute kidney injury requiring dialysis. Indian J Nephrol. 2015 Mar-Apr;25(2):77–81. PMID: 25838643; PMCID: PMC4379629. [DOI] [PMC free article] [PubMed]
- 18.Gopalakrishnan N, Dhanapriya J, Muthukumar P, Sakthirajan R, Dineshkumar T, Thirumurugan S, Balasubramaniyan T. Acute kidney injury in pregnancy–a single center experience. Ren Fail. 2015;37(9):1476-80. doi: 10.3109/0886022X.2015.1074493. Epub 2015 Sep 3. PMID: 26338215. [DOI] [PubMed]
- 19.Gullipalli P, Srinivasulu N. Spectrum of postpartum kidney injury– A tertiary care center experience in a developing Nation. IOSR J Dent Med Sci. 2015;14:92–5. [Google Scholar]
- 20.Eswarappa M, Madhyastha PR, Puri S, Varma V, Bhandari A, Chennabassappa G. Postpartum acute kidney injury: a review of 99 cases. Ren Fail. 2016;38(6):889–doi. 10.3109/0886022X.2016.1164015. Epub 2016 Jun 20. PMID: 27319810. [DOI] [PubMed] [Google Scholar]
- 21.Mishra V, Goyal Preeti A, Aggarwal Rohina S, Choudhary S, Tanvir T, Dharaiya Nisarg D, Gaddagi Rashmi A. A Single-Centre Experience of Obstetric Acute Kidney Injury. J Obstet Gynaecol India. 2016;66(Suppl 1):207– 11. doi: 10.1007/s13224-016-0839-5. Epub 2016 Mar 3. PMID: 27651605; PMCID: PMC5016445. [DOI] [PMC free article] [PubMed]
- 22.Prakash J, Pant P, Prakash S, Sivasankar M, Vohra R, Doley PK, Pandey LK, Singh U. Changing picture of acute kidney injury in pregnancy: study of 259 cases over a period of 33 years. Indian J Nephrol. 2016 Jul-Aug;26(4):262–7. 10.4103/0971-4065.161018. PMID: 27512298; PMCID: PMC4964686. [DOI] [PMC free article] [PubMed]
- 23.Prakash J, Ganiger VC, Prakash S, Iqbal M, Kar DP, Singh U, Verma A. Acute kidney injury in pregnancy with special reference to pregnancy-specific disorders: a hospital based study (2014–2016). J Nephrol. 2018;31(1):79–85. 10.1007/s40620-017-0466-y. Epub 2018 Jan 4. PMID: 29302904. [DOI] [PubMed]
- 24.Mir MM, Najar MS, Chaudary AM, Azad H, Reshi AR, Banday KA, et al. Postpartum acute kidney injury: experience of a tertiary care center. Indian J Nephrol. 2017;27:181–4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tanwar RS, Agarwal D, Gupta RK, Rathore V, Beniwal P, Joshi P. Malhotra Characteristics and outcome of postpartum acute kidney injury requiring dialysis: A single-center experience from North India. Saudi J Kidney Dis Transpl. 2018 Jul- Aug;29(4):837–845. 10.4103/1319-2442.239663. PMID: 30152420. [DOI] [PubMed]
- 26.Saini S, Chaudhury AR, Divyaveer S, Maurya P, Sircar D, Dasgupta S, Sen D, Bandyopadhyay S, Pandey R. The changing face of pregnancy-related acute kidney injury from eastern part of India: A hospital-based, prospective, observational study. Saudi J Kidney Dis Transpl. 2020 Mar-Apr;31(2):493–502. 10.4103/13192442.284025. PMID: 32394923. [DOI] [PubMed]
- 27.Chowdhary PK, Tibrewal A, Kale SA. Postpartum Acute Kidney Injury in Tertiary Care Center: Single-Center Experience from Central India. Saudi J Kidney Dis Transpl. 2021 Jul-Aug;32(4):1111–1117. 10.4103/1319-2442.338284. PMID: 35229810. [DOI] [PubMed]
- 28.Banerjee A, Mehrotra G. Comparison of standard Conservative treatment and early initiation of renal replacement therapy in Pregnancy-related acute kidney injury: A Single-center prospective study. Indian J Crit Care Med. 2020;24(8):688–94. 10.5005/jp-journals-10071-23537. PMID: 33024376; PMCID: PMC7519595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Sahay M, Priyashree, Dogra L, Ismal K, Vali S. Pregnancy-related Acute Kidney Injury in Public Hospital in South India: Changing Trends. J Assoc Physicians India. 2022;70(8):11–12. 10.5005/japi-11001-0064. PMID: 36082728. [DOI] [PubMed]
- 30.Sachan R, Shukla S, Shyam R, Patel ML, Verma ML. Role of renal replacement therapy in pregnancy related acute kidney injury and its outcome. J Family Med PrimCare. 2022;11(5):2155–61. 10.4103/jfmpc.jfmpc_1019_21. Epub 2022 May 14. PMID: 35800491; PMCID: PMC9254767. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Yadav S, Chauhan M, Jain D, Aggarwal HK, Yadav RK. Renal outcomes of Pregnancy-Related acute kidney injury: a single centre experience in India Maedica. (Bucur). 2022;17(1):80–7. 10.26574/maedica.2022.17.1.80. PMID: 35733733; PMCID: PMC9168580. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Sandilya S, Rani KU, Kumar R. Risk factors and fetomaternal outcome in pregnancy-related acute kidney injury. J Family Med Prim Care. 2023;12(12):3346–50. 10.4103/jfmpc.jfmpc_924_23. Epub 2023 Dec 21. PMID: 38361835; PMCID:PMC10866243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Kharkongor D, Mehrotra S, Solanki V, Singh U, Patel ML, Agarwal S. Acute kidney injury in obstetrics: Hospital-based cohort study. Trop Doct. 2023;53(3):356–61. Epub 2023 Apr 20. PMID: 37078137. [DOI] [PubMed] [Google Scholar]
- 34.Saxena D, Kumar T, Malhotra V, Yadav M, Sharma S, Beniwal P, et al. Pregnancy related acute kidney injury: an exigent cause of chronic kidney disease in developing countries. J Obstet Gynaecol India. 2024;74(6):541–546. 10.1007/s13224-023-01929-7 [DOI] [PMC free article] [PubMed]
- 35.Modi GK, Jha V. The incidence of end-stage renal disease in india: A population-based study. Kidney Int. 2006;70:2131–3. [DOI] [PubMed] [Google Scholar]
- 36.Shalaby AS, Shemies RS. Pregnancy-related acute kidney injury in the African continent: where do we stand? A systematic review. J Nephrol. 2022;35(9):2175–89. 10.1007/s40620-022-01349-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
No datasets were generated or analysed during the current study.