Abstract
This case series explores 4 unique presentations of vaginal cuff lesions, highlighting their diverse etiologies and imaging characteristics. The cases include a vaginal cuff meshoma, benign vaginal cuff cystic lesions, and metastatic endometroid adenocarcinoma. The series emphasizes the importance of a multifaceted approach utilizing patient history, clinical examination, and various imaging modalities (transvaginal ultrasound, MRI, PET-CT) for accurate diagnosis and optimal patient management.
Keywords: Vaginal cuff lesions, Meshoma, Inclusion cyst, Gartner duct cyst, Metastases
Introduction
The vaginal cuff is the residual tissue remaining after a hysterectomy. In cases where the hysterectomy was performed for malignancies like cervical or endometrial carcinoma, the vaginal cuff may be assessed for tumor recurrence, typically using ultrasound or MRI. Vaginal cuff lesions may also include a wide spectrum of pathologies that pose a diagnostic challenge for clinicians. Common etiologies include benign lesions and metastatic lesions. Postsurgical complications include mesh erosion with meshoma. Understanding these lesions diverse presentations, underlying causes, and imaging findings is crucial for accurate diagnosis and patient management.
Case presentation
Case 1
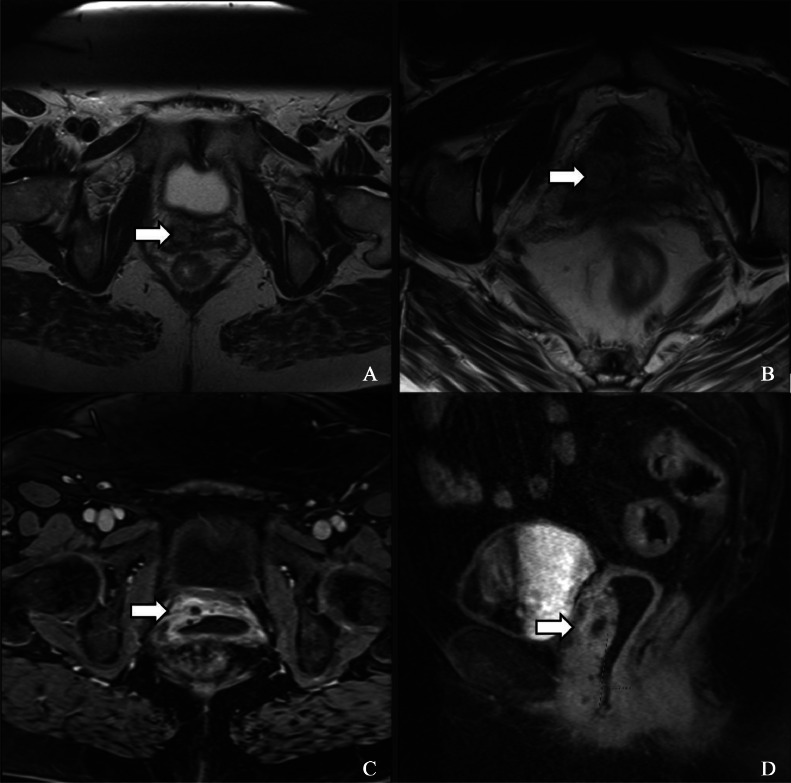
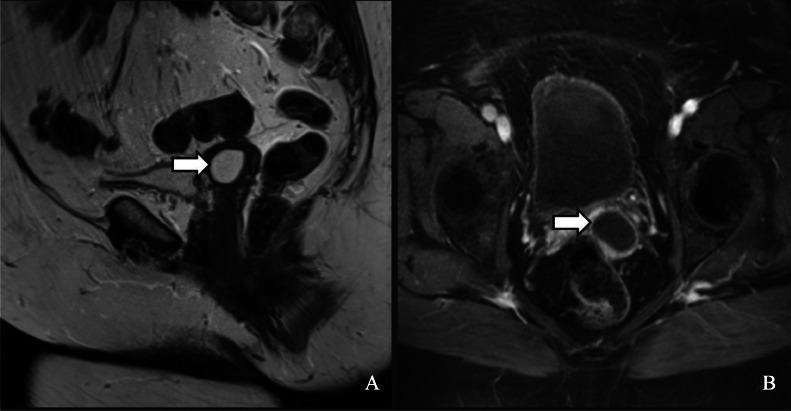
A 49-year-old woman with a history of total vaginal hysterectomy, abdominal perineal repair, and trans obturator tape (TOT) suburethral sling placement for pelvic organ prolapse presented with recurrent vaginal discharge and vaginal pain. MRI showed part of the mesh embedded into the anterior vaginal wall (Fig. 1A and B). MRI postcontrast imaging showed peripheral enhancing soft tissue around the mesh in the anterior vagina (Fig. 1C and D). This was indicated to most likely represent a granuloma versus phlegmon or evolving abscess.
Fig. 1.
(A and B) T2-weighted MRI image showing mesh embedded in the anterior vaginal wall (white arrow). (C and D) MRI postcontrast image showing a peripheral enhancing soft tissue mass seen along the anterior vaginal cuff in the paramedian location (white arrow).
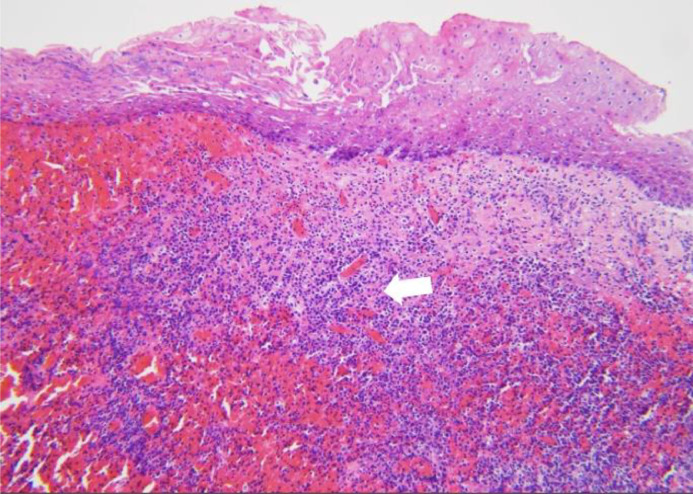
The patient underwent successful laparoscopic excision of the mesh and surrounding tissue with histopathological confirmation of meshoma formation. Cystoscopy was performed and there was found to be no mesh erosion into the bladder. Pathology reported the presence of synthetic mesh with adherent fibrous tissue and necro-inflammatory debris (Fig. 2).
Fig. 2.
Histopathology section of the attached tissue to excised vaginal mesh demonstrating squamous mucosa with submucosal hemorrhage, acute and chronic inflammation (marked with white arrow), and necrosis (H&E stain, 100x magnification).
Case 2
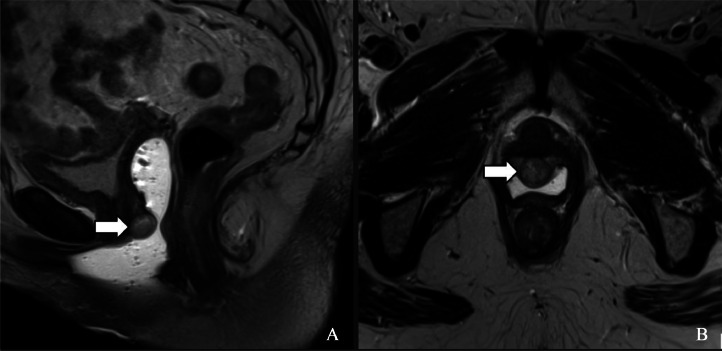
A 62-year-old woman with a history of transvaginal hysterectomy and bilateral salpingo-oophorectomy for uterine prolapse and history of benign ovarian tumors, presented with pelvic pain. Transvaginal ultrasound identified a well-defined, predominantly hypoechoic lesion with hyperechoic foci within the vaginal cuff, findings suggestive of benign cystic lesion (Fig. 3A and B). MRI evaluation identified a well-defined lesion along the anterior vaginal wall, consistent with a benign inclusion cyst (white arrows). The lesion demonstrates hyperintense signal on both T1-weighted noncontrast, nonfat-suppressed (Fig. 4A) and fat-suppressed (Fig. 4B) sequences. Persistent hyperintensity on fat suppression images suggests the lesion is not composed of fat.
Fig. 3.
Sagittal (A) and coronal (B) transvaginal ultrasound showing a predominantly hypoechoic lesion (marked with white arrow), measuring 1.6 × 1.3 × 1.4 cm, with multiple punctate hyperechoic foci within.
Fig. 4.
MRI T1 weighted noncontrast nonfat suppressed (A) and fat-suppressed (B) images showing the well-defined anterior vaginal wall inclusion cyst ( marked with white arrows). It appears hyperintense in both images and is not suppressed on fat-suppressed imaging.
The patient underwent marsupialization of the vaginal cuff wall cyst and findings were suggestive of a vaginal inclusion cyst.
Case 3
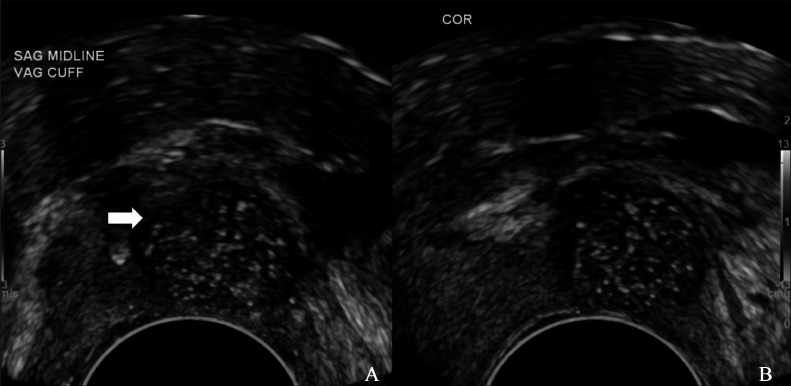
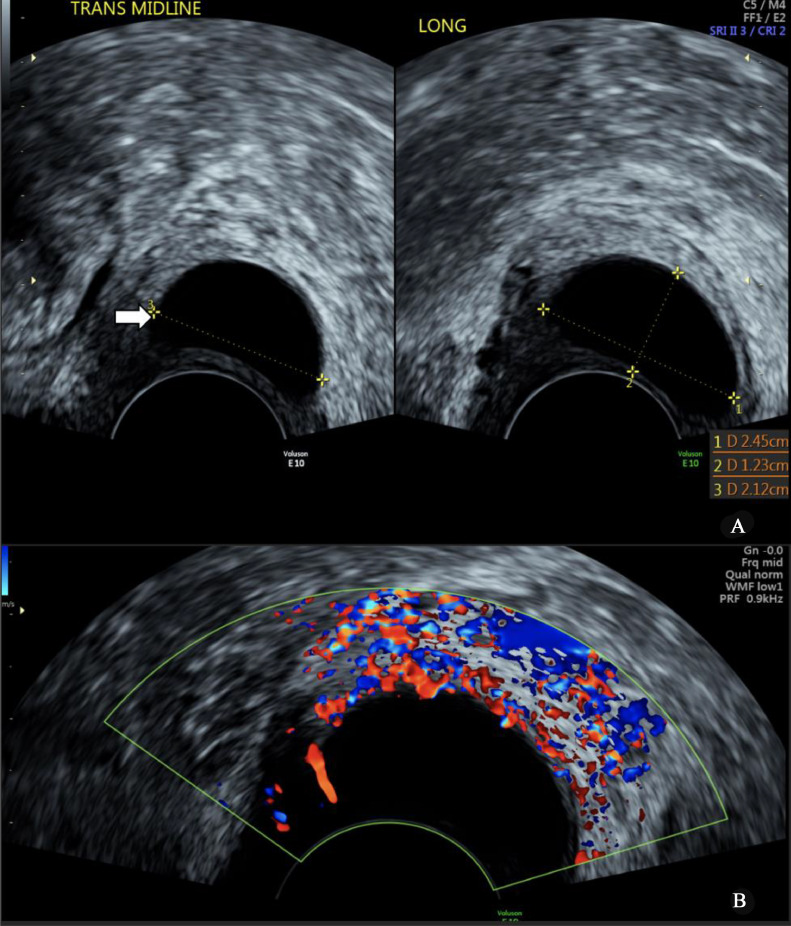
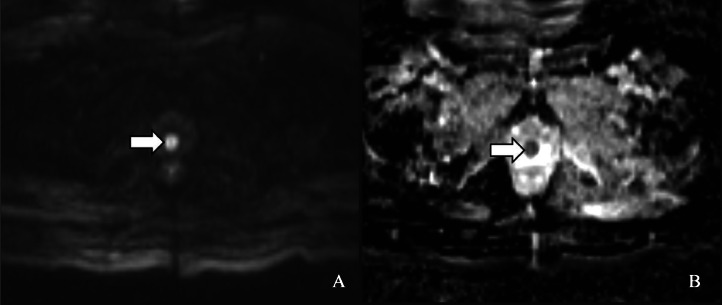
A 58-year-old female underwent a total abdominal hysterectomy 30 years ago for abnormal uterine bleeding. She presented with a complaint of pelvic pressure. Transvaginal ultrasound revealed a well-defined anechoic cyst within the vaginal cuff with peripheral vascularity (Fig. 5). Subsequent MRI confirmed a well-defined hyperintense cystic lesion with mild peripheral enhancement (Fig. 6A and B). These findings and the location of the cyst being well above the pubic symphysis were suggestive of a Gartner cyst.
Fig. 5.
Transvaginal ultrasound image (A) displaying a well-defined anechoic cyst (white arrow) with peripheral vascularity within the vaginal cuff (B).
Fig. 6.
T2-weighted MRI image (A) showing a hyperintense lesion (white arrow) with mild peripheral enhancement (white arrow) on T1 contrast MRI image (B).
Operative findings revealed a 2 × 2 cm thin-walled cyst arising from the left aspect of the vaginal cuff filled with clear fluid. The cyst was successfully excised, and revealed the presence of a squamous-lined fibroconnective tissue with chronic inflammation but no malignancy. The findings were suggestive of a benign Gartner duct cyst.
Case 4
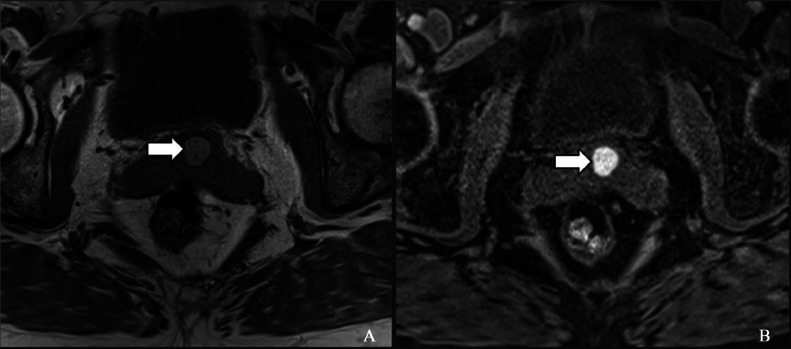
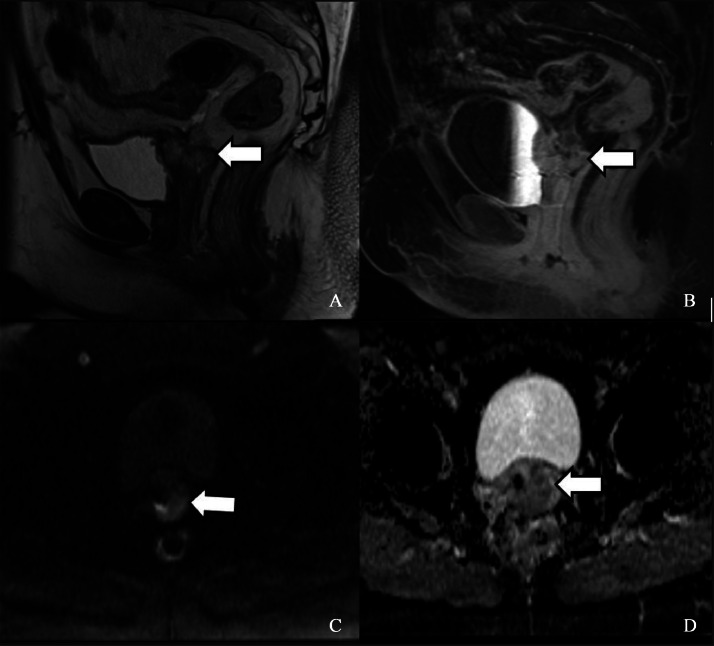
A 57-year-old woman with a history of endometroid adenocarcinoma (FIGO stage IIIB) treated with hysterectomy and bilateral salpingo-oophorectomy presented for a follow-up MRI 3 months later. The scan revealed a new, well-defined lesion along the anterior vaginal cuff (Fig. 7A and B). The lesion demonstrated predominant peripheral enhancement with subtle nodular areas of internal enhancement. Diffusion-weighted imaging (DWI) showed diffusion restriction, appearing hyperintense on DWI and hypointense on ADC maps. A PET scan was also performed; however, the lesion could not be visualized due to tracer excretion into the urinary bladder, which obscured the small focal lesion at the vaginal cuff (Fig. 8A and B).
Fig. 7.
Sagittal (A) and axial (B) T2 weighted MRI images showing the new metastatic vaginal lesion (white arrows). It is a well-defined heterogenous lesion along the anterior wall of the vaginal cuff, just posterior to the urethral meatus.
Fig. 8.
Axial MRI images, showing diffusion restriction with hyperintensity on DWI (A) and hypointensity on ADC image (B).
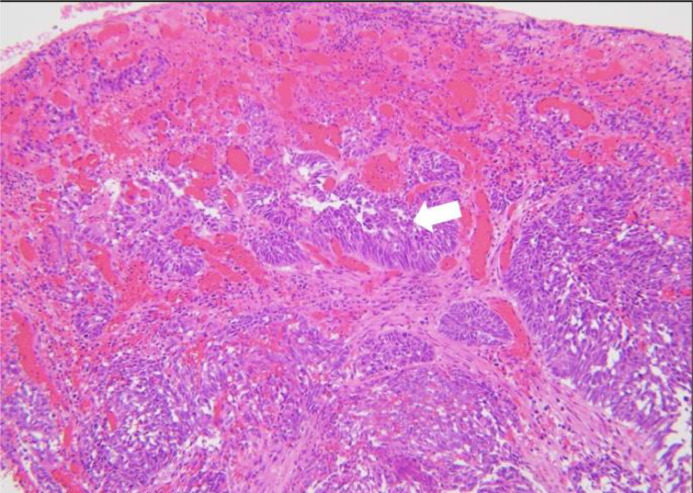
The patient underwent a distal partial anterior vaginectomy to remove the lesion. Pathology showed it to be a high-grade differentiated carcinoma with scant cytoplasm and enlarged nuclei (Fig. 9). These pathological findings confirmed the lesion to be a high-grade metastatic carcinoma consistent with the patient's primary malignancy (Fig. 10).
Fig. 9.
Histopathological image showing infiltrating malignant glands (marked with white arrow) throughout the vaginal mucosa. The malignant glands are morphologically identical to those observed in the prior hysterectomy specimen, which showed endometrioid adenocarcinoma. The findings indicate metastatic involvement of the vagina by the patient's primary endometrial cancer. (H&E stain, 100x magnification).
Fig. 10.
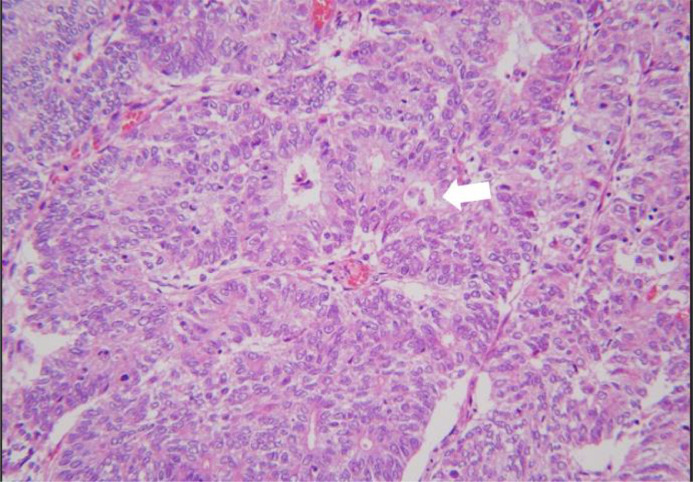
Histology of hysterectomy specimen showing infiltrating malignant glands (marked with white arrow) in Endometrial carcinoma, endometrioid type, FIGO Grade 3 with residual 20% myometrial invasion (H&E stain, 200× magnification).
We present a companion case of vaginal cuff metastasis in a patient with a history of subtotal colectomy for synchronous ascending and descending colon cancers. Histopathology revealed a mucinous carcinoma. One year postsurgery, rising CEA levels were noted, prompting further evaluation. Follow-up MRI of the pelvis revealed lesions at the vaginal cuff, characterized by T2-intermediate signal intensity and heterogeneous nodular thickening at the apex of the vaginal cuff (Fig. 11A). Postcontrast images demonstrated enhancing soft tissue nodularity (Fig. 11B) with mild diffusion restriction (Fig. 11C and D). PET-CT showed intense FDG avidity in the vaginal cuff region (Fig. 12A and B). These findings were consistent with vaginal cuff metastasis. The patient subsequently underwent chemotherapy.
Fig. 11.
MRI Pelvis images showing intermediate signal heterogenous nodular thickening involving top of vaginal cuff on T2W sequence (A) and contrast enhancement is seen in sagittal T1 contrast image (B). Axial MRI images showing mild diffusion restriction with mild hyperintensity on DWI (C) and hypointensity on ADC sequence (D) (marked with white arrow).
Fig. 12.
FDG PET Coronal (A) and Axial (B) images show intense FDG avidity in vaginal cuff lesions (marked with white arrow).
Discussion
The vaginal cuff is the uppermost part of the vagina, formed by suturing the vaginal walls together following a hysterectomy. It is a common site for recurrent gynecologic malignancies, particularly cervical, endometrial, and ovarian cancers—with cervical cancer being the most common and ovarian cancer the rarest. In addition to malignancies, a variety of benign and infectious lesions have also been reported at the vaginal cuff [1]. Trans vaginal Ultrasound (USG) is typically the first-line imaging modality, while MRI serves as the gold standard for the radiological evaluation of vaginal cuff lesions due to its superior soft tissue contrast. Restriction pattern on DWI sequence of MRI overlaps between benign and malignant lesions, making it difficult to differentiate between the 2. Vaginal cuff lesions are categorized as based on their etiology and nature which is represented in flowchart below:

The 4 above presented cases including both benign and malignant lesions of vaginal cuff are discussed in detail below.
Meshoma
The incidence of vaginal vault prolapse is around 12% after hysterectomy [2]. Sacrocolpopexy (SCP) with synthetic mesh is considered the gold standard procedure for treating vaginal vault prolapse [3]. Mesh repair of vaginal vault prolapse is associated with the complication of mesh erosion into the vagina, occurring in 4.2% of patients [4]. A meshoma is a clinical condition in which the mesh folds and wrinkles into a ball, causing chronic pelvic pain. It was first described by Amid after the mesh repair of the abdominal wall hernia. Causes include nonfixation, insufficient fixation, or insufficient dissection to make adequate space for the prosthesis [5]. Meshoma formation can be difficult to diagnose clinically, particularly when presenting late or with nonspecific symptoms such as discharge, dyspareunia, or pelvic pain. In our case, on MRI imaging- abnormal soft tissue mass in the anterior vaginal wall with an embedded mesh component was seen. The peripheral enhancement observed on postcontrast sequences raised suspicion for a granulomatous reaction, phlegmon, or early abscess formation—all of which are consistent with chronic inflammatory processes typically associated with mesh-related complications. MRI is the imaging modality of choice for soft tissue characterization and provides superior delineation of mesh location and associated complication like abscess formation. On CT and MRI, the imaging findings can mimic nonspecific postsurgical findings, inflammatory phlegmon, reactive lymph nodes, or indeterminate densities. However, a high index of suspicion based on clinical presentation, and effective communication with the operating gynecologist helps clinch the diagnosis. In our case, the MRI findings were crucial in guiding the surgical approach. Surgical excision remains the mainstay of treatment for symptomatic meshoma. Histopathology confirmed the diagnosis, revealing synthetic mesh with adherent fibrous tissue and necro-inflammatory debris - findings that underscore the chronic inflammatory nature of meshoma.
Benign vaginal cysts
Benign cysts like vaginal inclusion cysts and Gartner duct cysts have been known to occur in the vaginal vault, particularly in women with history of surgery. Although, these cysts are generally asymptomatic and detected incidentally or they sometimes present with symptoms such as pelvic pain, pressure, dyspareunia or urinary disturbances, prompting further evaluation through imaging.
Posthysterectomy vaginal inclusion cyst occurs where the opposed vaginal vaults were sutured for surgery. These occur due to entrapment of stratified squamous epithelium at the time of hysterectomy [6]. Imaging appearance is that of a benign cystic lesion- well circumscribed, unilocular and thin walled. On USG, they present as hypoechoic or anechoic lesions and occasionally show internal echoes due to hemorrhagic or proteinaceous content. MRI is particularly valuable for characterizing these lesions, with cysts typically showing hyperintensity on T2-weighted images and variable intensity on T1, depending on the presence of hemorrhagic or proteinaceous material. Persistent T1 hyperintensity on fat-suppressed sequences, as seen in our case, suggests nonfat content, ruling out dermoid cysts and pointing toward inclusion cysts with hemorrhagic or proteinaceous content. It can become challenging in cases of superimposed infection, wherein it can mimic an abscess. A hemorrhagic or proteinaceous cyst can mimic an endometriotic implant on imaging.
Gartner duct cyst arises at the anterolateral vaginal vault at or above the inferior margin of the pubic symphysis. Embryologically, it arises from the Wolffian ducts and may be associated with renal anomalies like agenesis due to their shared embryologic origin from mesonephric duct system [7]. Larger cysts can cause mass effects and lead to dyspareunia and urethral symptoms. Gartner duct cysts appear as well-defined, thin-walled, anechoic cysts on ultrasound with no internal vascularity. MRI confirms these findings, with high T2 signal intensity, low T1 signal, and mild peripheral enhancement after contrast administration as seen in our case. Their location relative to the pubic symphysis - being well above it, is a useful distinguishing feature. Surgical management is generally reserved for symptomatic cases.
Vaginal vault recurrence
The vaginal vault is one of the most common sites for recurrence in patients with endometrial cancer [3]. Imaging plays an essential role at the time of vaginal cuff recurrence. Pelvic MRI is the imaging modality of choice in describing the tumor recurrence, assessing local invasion, and distinguishing between recurrence and treatment-related changes. The MR imaging features of vaginal metastasis mimic the MR imaging features of the primary tumor, and T2-weighted MR imaging best delineates the extent of disease within and beyond the vagina [8]. MR imaging features that have been described include a mass with intermediate T2 signal intensity in the vaginal vault with diffusion restriction and may also appear as complex cystic lesions, diffuse plaque-like formations, and distal vaginal lesions [9]. Diffusion restriction is useful in detecting small recurrent disease at vaginal cuff because small deposit is usually covered by serosal surface of intestinal loops and solid viscera which are difficult to detect with CT or conventional MRI because similar attenuation or signal intensity of adjacent structure mask the lesion [10]. CT and PET-CT complement MRI by assessing nodal and distant metastases. However, PET-CT may have limitations in the pelvis, particularly near the bladder, due to physiological FDG excretion, which can obscure small or subtle lesions at the vaginal cuff as seen in our case.
Conclusion
This case series demonstrates the diverse spectrum of vaginal cuff lesions, ranging from benign entities like inclusion cysts and Gartner duct cysts to malignant processes like metastatic carcinomas and rare postoperative complication of vaginal cuff like meshoma. Distinguishing between benign and malignant lesions is paramount in guiding appropriate management strategies. Transvaginal ultrasound offers a readily available and cost-effective tool for initial evaluation, allowing visualization of lesions and assessment of blood flow. MRI provides superior soft tissue contrast resolution, enabling detailed characterization of the lesion's size, location, and relationship to surrounding structures. In specific cases, PET-CT scans can help identify distant metastases, particularly when the primary malignancy is unknown.
IRB approval
This has been submitted to institutional IRB and was approved and acknowledged by institutional IRB.
Patient consent
This has been submitted to the Institutional IRB and was approved and acknowledged by institutional IRB. As per our institutional IRB, we were not required to obtain consent as no patient identifiers were used.
Footnotes
Competing Interests: The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
References
- 1.Stein M.W., Grishina A., Shaw R.J., Roberts J.H., Ricci Z.J., Adachi A., et al. Gray-scale and color doppler sonographic features of the vaginal cuff and cervical remnant after hysterectomy. AJR Am J Roentgenol. 2006;187(5):1372–1376. doi: 10.2214/AJR.05.0453. [DOI] [PubMed] [Google Scholar]
- 2.Maher C., Feiner B., Baessler K., Christmann-Schmid C., Haya N., Brown J. Surgery for women with apical vaginal prolapse. Cochrane Database Syst Rev. 2016;10(10) doi: 10.1002/14651858.CD012376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Mikos T., Theodoulidis I., Kioussis G., Karalis T., Papaioannou S., Grimbizis G.F. Cuff meshoma post-laparoscopic sacrocolpopexy: vaginal-endoscopic mesh excision. Int Urogynecol J. 2023;34(10):2623–2625. doi: 10.1007/s00192-023-05591-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Visco A.G., Weidner A.C., Barber M.D., Myers E.R., Cundiff G.W., Bump R.C., et al. Vaginal mesh erosion after abdominal sacral colpopexy. Am J Obstet Gynecol. 2001;184(3):297–302. doi: 10.1067/mob.2001.109654. [DOI] [PubMed] [Google Scholar]
- 5.Amid P.K. Radiologic images of meshoma: a new phenomenon causing chronic pain after prosthetic repair of abdominal wall hernias. Arch Surg. 2004;139(12):1297–1298. doi: 10.1001/archsurg.139.12.1297. [DOI] [PubMed] [Google Scholar]
- 6.Davis M.C., Fishman E.K., Cameron J.L., Magid D., Siegelman S.S. Computed tomography of vaginal cuff cyst: a late complication of hysterectomy. J Comput Assist Tomogr. 1986;10(2):354–356. doi: 10.1097/00004728-198603000-00042. [DOI] [PubMed] [Google Scholar]
- 7.Zulfiqar M., Shetty A., Yano M., McGettigan M., Itani M., Naeem M., et al. Imaging of the vagina: spectrum of disease with emphasis on MRI appearance. Radiographics. 2021;41(5):1549–1568. doi: 10.1148/rg.2021210018. [DOI] [PubMed] [Google Scholar]
- 8.Parikh J.H., Barton D.P., Ind T.E., Sohaib S.A. MR imaging features of vaginal malignancies. Radiographics. 2008;28(1):49–63. doi: 10.1148/rg.281075065. quiz 322. [DOI] [PubMed] [Google Scholar]
- 9.Steiner A., Alban G., Cheng T., Kapur T., Bay C., McLaughlin P.Y., et al. Vaginal recurrence of endometrial cancer: MRI characteristics and correlation with patient outcome after salvage radiation therapy. Abdom Radiol (NY) 2020;45(4):1122–1131. doi: 10.1007/s00261-020-02453-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Motoshima S., Irie H., Nakazono T., Kamura T., Kudo S. Diffusion-weighted MR imaging in gynecologic cancers. J Gynecol Oncol. 2011;22(4):275–287. doi: 10.3802/jgo.2011.22.4.275. [DOI] [PMC free article] [PubMed] [Google Scholar]