ABSTRACT
Background
Geographical specific data is required to guide empirical antimicrobial selection in equine neonates.
Objectives
Evaluate antibiograms and survival in foals from a United Kingdom (UK)‐based hospital to guide antimicrobial selection.
Animals
Blood and synovial fluid samples from 208 foals ≤ 30 days old admitted to Rossdales Equine Hospital from 2018 to 2023.
Methods
Retrospective cohort study. Antimicrobial susceptibility was assessed by disc diffusion method. Bacterial culture and susceptibility and foal survival were recorded. The effects of the presence of positive culture or multidrug resistance (MDR) isolates on survival were evaluated using univariable mixed effects logistic regression.
Results
Ninety‐one isolates were identified from 381 samples from 208 foals. Predominantly gram‐positive (75%, 68/91; 95% confidence interval [CI]: 65%–83%) isolates were identified, and Enterococcus (26%, 24/91; 95% CI: 18%–37%) was the most commonly isolated bacteria. MDR was identified in 21% of isolates (19/91; 95% CI: 13%–31%). Enterococcus was the most frequent MDR isolate (7/19). The combination of ampicillin and amikacin showed in vitro susceptibility in 90% (81/90; 95% CI: 82%–95%) of aerobic isolates. In total, 87% of foals were discharged from the hospital (180/208; 95% CI: 81%–91%). No association was identified between survival and the presence of positive culture or MDR isolates.
Main Limitations
Retrospective design; missing data for prior antimicrobial treatment, reason for admission and admission variables.
Conclusions and Clinical Importance
Ampicillin and amikacin are appropriate combination first‐line antimicrobial treatments in this population. Many Gram‐positive isolates were identified, most notably Enterococcus. Culture and susceptibility guided antimicrobial choices remain crucial, especially given the unpredictable susceptibility of Enterococcus and the frequency of MDR Enterococcus isolates identified.
Keywords: antimicrobials, culture, foal, resistance, susceptibility
Abbreviations
- AST
antimicrobial susceptibility testing
- CLSI
Clinical & Laboratory Standards Institute
- EUCAST
European Committee on Antimicrobial Susceptibility Testing
- MDR
multidrug resistance
1. Introduction
Sepsis remains an important cause of mortality in neonatal foals, and bacterial culture is the only method available to identify a causative agent [1]. Improved outcomes for survival have been shown when empirically selected antimicrobial regimens include an antimicrobial to which isolated bacteria are susceptible [2]. In foals with signs of sepsis, it is rarely appropriate to wait for culture and susceptibility results before starting antimicrobial treatment, and therefore empirical selection of antimicrobials is necessary [2, 3, 4]. However, making the correct empirical antimicrobial selection can be challenging. In a recent study of United Kingdom (UK) and European veterinary surgeons, third‐ or fourth‐generation cephalosporins were commonly used for treating equine neonates [5]. This practice is despite the World Health Organization (WHO) listing these drugs as highest priority, critically important antimicrobials [6], meaning they should not be used for first line treatment. The use of third‐ or fourth‐generation cephalosporins in foals is appealing because of their broad spectrum of activity, ease of administration, and lack of adverse side effects [3]. However, evidence for whether these are still appropriate for treatment of foals in the United Kingdom is lacking. Changes have occurred recently in the bacterial profiles of foals diagnosed with sepsis, from predominantly gram‐negative infections towards increasingly frequent gram‐positive infections [7, 8, 9, 10, 11]. Evidence also exists that the susceptibility of Enterobacteriaceae to cephalosporins is becoming less predictable, with multidrug resistant (MDR) bacteria also reported in foals [4, 7, 8, 11]. To further complicate appropriate empirical antimicrobial selection, geographical differences exist in bacterial antibiograms, which should be considered when making empirical antimicrobial selections [4, 7, 11, 12]. To our knowledge, no studies have evaluated antibiograms in foals in the United Kingdom, and current guidelines are extrapolated from studies in other countries [3]. Given the importance of protecting highest priority critically important antimicrobials and developing trends in antimicrobial resistance, selection of appropriate antimicrobials is crucial for clinical outcomes as well as minimizing antimicrobial resistance. However, blood culture has poor sensitivity, with low positive culture rates on blood cultures from foals with concurrent signs of sepsis (23.4%–49% of cultures yielding positive results) [1, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21].
The aim of our retrospective cohort study was to evaluate antibiograms from foals admitted to a UK‐based hospital. Specific study objectives were to: (a) determine the prevalence of different bacterial species isolated from blood and synovial fluid samples collected from hospitalized foals ≤ 30 days of age at Rossdales Equine Hospital and submitted to Rossdales Laboratories over a 6‐year period; (b) document antimicrobial susceptibility results to guide rational empirical antimicrobial choices for practitioners; and (c) determine if mortality was associated with positive culture or growth of MDR isolates from one or more samples.
2. Materials and Methods
2.1. Study Design and Case Selection
A retrospective review of laboratory records was performed to identify all samples submitted to Rossdales Laboratories for culture and susceptibility testing between 2018 and 2023. The following information was collected from Rossdales Equine Hospital's medical records: age, breed, sex, and survival, defined as survival to hospital discharge, of foals that had samples submitted. To be included, blood or synovial fluid samples, or both, had to be collected from foals admitted to Rossdales Equine Hospital, and that were ≤ 30 days old at the time of sample collection. Data collected on samples included type of sample submitted, bacterial isolates detected (including aerobic and anaerobic), qualitative assessment of the extent of bacterial growth (no growth, few colonies, moderate growth, profuse growth), and antimicrobial susceptibility (sensitive, intermediate, resistant) [22]. Data for admission variables, reason for referral, and if antimicrobials had been administered before referral was not available. Samples from other sites such as IV catheters, umbilical tissue, peritoneal fluid, and tracheal wash fluid were not included. A positive culture was considered to reflect a contaminant (false positive) if only scant growth was obtained in the direct culture.
2.2. Sample Collection
All blood culture samples were taken in a sterile manner through an IV catheter at the time of placement, or from a sterile blood collection (where the site was aseptically prepared before venipuncture). The first time collection was performed, the sample was referred to as “first blood culture.” If sequential blood cultures were taken (referred to as second, third, fourth blood culture), they were collected via sterile blood collection 24–48 h apart. All blood samples (5–10 mL) were inoculated immediately at the time of collection into a fluid culture medium bottle (2018–2022 “Bloodgrow” dual culture MW 900, Medical Wire & Equipment Co. Ltd.; from 2023 BD BACTEC Plus Aerobic [aerobic only], BD). Synovial fluid was collected aseptically and placed into a fluid culture medium bottle (2018–2022 “Bloodgrow” dual culture MW900, Medical Wire & Equipment co Ltd.; from 2023 BD BACTEC Plus Aerobic [aerobic only], BD) either at the time of sampling, or into a sterile container before being inoculated into a fluid culture medium bottle at Rossdales Laboratories. If the sample could not be immediately submitted to Rossdales Laboratories (e.g., if collected out of hours), samples were incubated at 37°C before submission. Samples were submitted for aerobic culture with or without anaerobic culture, depending on clinician preference and availability of fluid culture medium bottles.
2.3. Bacterial Isolation, Identification, and Classification
Samples were incubated in fluid culture medium bottles for 24 h before plating for colony identification. Samples were inoculated and incubated using standardized techniques for both aerobic and anaerobic culture (excluding 2023 when only aerobic culture was available) [23]. If multiple bacterial colony types were detected, these were subcultured onto separate plates. Bacterial colonies were identified based on colony morphology, microscopy, and biochemical testing.
2.4. Antimicrobial Testing
Antimicrobial susceptibility testing (AST) was carried out on all isolates using the disk diffusion method [23]. Susceptibility testing was performed for the following routinely used antibiotics: amikacin, ampicillin, amoxicillin, azithromycin, cefotaxime, ceftiofur, clarithromycin, doxycycline, erythromycin, gentamicin, marbofloxacin, oxytetracycline, penicillin, rifampicin, and trimethoprim‐sulfonamide. Susceptibility testing also was performed for cefquinome, ceftriaxone, piperacillin and tazobactam, ticarcillin and clavulanic acid. Susceptibility also was recorded for combinations of commonly used antimicrobials including ampicillin/amikacin, ampicillin/gentamicin, penicillin/gentamicin, and penicillin/amikacin. Susceptibility for chloramphenicol, sodium fuscidate, enrofloxacin, and neomycin was not included because of their infrequent use. Susceptibility to cefquinome was tested routinely from 2018 to 2020, after which it was only included on request by the clinician. Susceptibility for ticarcillin and clavulanic acid, erythromycin, and amoxicillin was not tested after 2020.
Antimicrobial susceptibility was based on zone diameter breakpoints using guidelines established by the European Committee on Antimicrobial Susceptibility Testing (EUCAST) and Clinical & Laboratory Standards Institute (CLSI) [24, 25]. Interpretation of zone sizes on isolates was performed using the Sirscan 2000 automatic AST reader or by measuring zones using calipers. As per EUCAST 2019 guidelines, only resistant (and not intermediate) results were included when defining bacteria as MDR [22]. The MDR isolates were defined as isolates that were not susceptible to ≥ 1 antimicrobial in three or more classes [26]. Intrinsic resistance was not included when defining bacteria as MDR.
2.5. Data Analysis
Sample size calculations indicated that information from 209 foals was required to identify an odds ratio ≥ 2.9 when the percentage of non‐surviving foals with a negative culture was between 12% and 70%, assuming 1.35 positive cultures per negative culture, a 95% confidence interval (CI) and 80% power [1]. Data analysis was performed using Excel version 16.77.1 (Microsoft) and SPSS version 24 (IBM). Numerical data was assessed for normality using the Shapiro–Wilk test and, if found to be normally distributed, presented as mean ± SD. If the data was not normally distributed, median ± interquartile range (IQR) was used. Because it was possible for multiple samples to be collected from the same foal, an interception‐only mixed effects logistic regression, with foal as a random effect, was used to evaluate clustering. The model showed evidence of clustering (estimate, 3.12; p < 0.001). Therefore, relationships between the binary outcome of mortality and a positive culture or presence of a MDR isolate were analyzed using univariable mixed effects logistic regression, with foal included as a random effect in all models. Statistical significance was defined as p < 0.05.
3. Results
Over the period 2018–2023, 381 samples from 208 different foals fulfilled the inclusion criteria (Figure 1). Foals were predominantly Thoroughbreds (75.9%, 158/208; 95% CI: 70%–82%), with the remaining foals predominantly Warmbloods (8.2%, 17/208; 95% CI: 5%–13%) and Irish Sports Horses (3.8%, 8/208; 95% CI: 2%–7%). Breed was not recorded in four foals. The median age of the foals at sample submission was 2 [1–7] days (median [IQR]) but varied between sample types: 2 [1–5] days for first blood culture and 19 [8–23] days for synovial samples. Samples were predominantly blood cultures (85.0%, 324/381; 95% CI: 81%–88%) and were most commonly first (50.1%, 191/381; 95% CI: 45%–55%), rather than subsequent, blood cultures. Between 2018 and 2022, when anaerobic fluid culture medium was available, 273/331 (82%) samples were tested for both aerobic and anaerobic growth.
FIGURE 1.
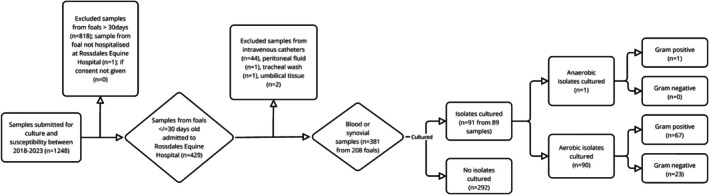
Flowchart of enrolled samples and results of bacterial culture for aerobic or anaerobic followed by Gram status.
3.1. Positive Culture Frequency
Only two samples grew > 1 isolate (up to two isolates) and only one sample grew an anaerobic isolate. More than one sample was submitted for 103 foals (median one sample per foal [IQR: 1–3], maximum seven samples in one foal). Positive cultures were obtained in 23.4% (89/381, 95% CI: 19%–28%) of samples and most frequently from synovial fluid (37%, 21/57; 95% CI: 24%–51%; Table 1).
TABLE 1.
Percentage of isolates cultured from different fluid types and Gram status of those isolates from 381 samples from 208 foals.
| Type of fluid sample | Number of isolates/number of samples (percentage, 95% CI) | Number of Gram‐positive isolates/total number isolates (percentage, 95% CI) | Number of Gram‐negative isolates/total number isolates (percentage, 95% CI) |
|---|---|---|---|
| First blood culture | 53/191 (28%, 22%–35%) | 43/53 (81%, 68%–91%) | 10/53 (19%, 9%–32%) |
| Second blood culture | 11/82 (13%, 7%–23%) | 9/11 (82%, 48%–98%) | 2/11 (18%, 2%–52%) |
| Third blood culture | 4/47 (9%, 2%–20%) | 3/4 (75%, 19%–99%) | 1/4 (25%, 1%–81%) |
| Fourth blood culture | 2/4 (50%, 7%–93%) | 2/2 (100%, 16%–100%) | 0/2 (0%, 0%–84%) |
| All blood cultures | 70/324 (22%, 17%–26%) | 57/70 (81%, 70%–90%) | 13/70 (19%, 10%–30%) |
| Synovial fluid | 21/57 (37%, 24%–51%) | 11/21 (52%, 30%–74%) | 10/21 (48%, 26%–70%) |
3.2. Isolate Frequency
Of the 91 isolates identified, 75% were Gram‐positive bacteria (68/91; 95% CI: 65%–83%), and 25% (23/91; 95% CI: 17%–35%) were Gram‐negative bacteria (Figure 2). Gram status varied between fluid sample types (Table 1). The most commonly isolated bacteria were Enterococcus, followed by Staphylococcus and Streptococcus (Table 2). Ten foals had blood and synovial fluid sampled concurrently at admission: three foals had no growth from either sample; two foals had profuse growth of the same bacteria from each fluid source (Klebsiella; Streptococcus); one foal had profuse growth of Streptococcus from blood culture and profuse growth of Actinobacillus from a synovial sample; three foals had single positive growth from synovial fluid and not from blood culture (Actinobacillus; Enterobacter; Streptococcus); and one foal grew Escherichia on blood culture but had no growth from synovial fluid.
FIGURE 2.

Bar chart showing the proportion of samples tested in each year of the study that yielded Gram‐positive or Gram‐negative isolates or without bacterial growth identified in each year. The numbers reported within each column are the absolute number of samples.
TABLE 2.
Percentage of bacterial isolates detected, and types of fluid sample submitted from 381 samples from 208 foals.
| Bacteria isolated | Number of isolate/total number of isolates (and percentage, 95% CI) | Fluid submission type |
|---|---|---|
| Enterococcus | 24/91 (26%, 18%–37%) | Blood (n = 18), synovial (n = 6) |
| Staphylococcus | 18/91 (20%, 12%–30%) | blood (n = 17), synovial (n = 1) |
| Streptococcus | 18/91 (20%, 12%–30%) | Blood (n = 14), synovial (n = 4) |
| Escherichia | 12/91 (13%, 7%–22%) | Blood (n = 9), synovial (n = 3) |
| Bacillus | 7/91 (8%, 3%–15%) | Blood (n = 7) |
| Klebsiella | 5/91 (5%, 2%–12%) | Blood (n = 3), synovial (n = 2) |
| Actinobacillus | 4/91 (4%, 1%–11%) | Synovial (n = 4) |
| Enterobacter | 1/91 (1%, 0.03%–6%) | Synovial (n = 1) |
| Pasteurella | 1/91 (1%, 0.03%–6%) | Blood (n = 1) |
| Clostridium | 1/91 (1%, 0.03%–6%) | Blood (n = 1) |
3.3. Antimicrobial Susceptibility
Susceptibility results were available for all samples, but antimicrobials tested varied among samples based on disc availability and clinician preference. A maximum of 21 antimicrobials were tested against any single isolate, with a median of 15 (IQR: 15–20; minimum, 13) antimicrobials per isolate. Antibiograms were created for all aerobic, Gram‐negative and Gram‐positive bacteria as well as isolates that were identified in > 10% of all samples (Tables 3 and 4).
TABLE 3.
Antibiograms for isolates cultured for individual antimicrobials for foals < 30 days from blood and synovial fluid. Enterococcus, Staphylococcus, Streptococcus, and Escherichia were the most commonly identified isolates.
| Bacteria isolated from sample | Aminoglycosides | Ansamycin | Penicillins | Cephalosporins | Anti‐pseudo penicillin + B‐lactamase inhibitors | Fluoroquinolones | Folate pathway inhibitor | Macrolides | Tetracyclines | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Amikacin | Gentamicin | Rifampicin | Amoxicillin | Ampicillin | Penicillin | Cefotaxime | Cefquinome | Ceftiofur | Ceftriaxone | Piperacillin and tazobactam | Ticarcillin and clavulanic acid | Marbofloxacin | Trimethoprim sulphonamide | Azithromycin | Clarithromycin | Erythromycin | Doxycycline | Oxytetracycline | |
| All aerobes | 45/85 | 54/90 | 57/90 | 31/36 | 61/87 | 36/90 | 73/74 | 31/36 | 76/90 | 72/87 | 81/88 | 31/33 | 72/90 | 51/90 | 64/87 | 63/88 | 21/33 | 71/90 | 51/89 |
| 53% | 60% | 63% | 86% | 70% | 40% | 99% | 86% | 84% | 83% | 92% | 94% | 80% | 57% | 74% | 72% | 64% | 79% | 57% | |
| Gram positive | 26/62 | 39/67 | 55/67 | 25/26 | 52/64 | 36/67 | 53/54 | 21/25 | 55/67 | 52/65 | 60/66 | 23/24 | 50/67 | 44/67 | 54/66 | 59/66 | 19/24 | 56/67 | 44/66 |
| 42% | 58% | 82% | 96% | 81% | 54% | 98% | 84% | 82% | 80% | 91% | 96% | 75% | 66% | 82% | 89% | 79% | 84% | 67% | |
| Gram negative | 19/23 | 15/23 | 2/23 | 6/10 | 9/23 | 0/23 | 20/20 | 10/11 | 21/23 | 20/22 | 21/22 | 8/9 | 22/23 | 7/23 | 10/21 | 4/22 | 2/9 | 15/23 | 7/23 |
| 83% | 65% | 9% | 60% | 39% | 0% | 100% | 91% | 91% | 91% | 95% | 89% | 96% | 30% | 48% | 18% | 22% | 65% | 30% | |
| Enterococcus | 3/24 | 7/24 | 15/24 | 9/10 | 20/24 | 6/24 | 12/12 | 5/9 | 14/24 | 12/23 | 17/23 | 7/8 | 11/24 | 9/24 | 15/23 | 20/23 | 6/8 | 15/24 | 11/24 |
| 13% | 29% | 63% | 90% | 83% | 25% | 100% | 56% | 58% | 52% | 74% | 88% | 46% | 38% | 65% | 87% | 75% | 63% | 46% | |
| Staphylococcus | 13/14 | 18/18 | 18/18 | 9/9 | 13/18 | 12/18 | 18/18 | 9/9 | 18/18 | 17/17 | 18/18 | 9/9 | 17/18 | 16/18 | 16/18 | 17/18 | 7/9 | 17/18 | 15/18 |
| 93% | 100% | 100% | 100% | 72% | 67% | 100% | 100% | 100% | 100% | 100% | 100% | 94% | 89% | 89% | 94% | 78% | 94% | 83% | |
| Streptococcus | 4/18 | 7/18 | 18/18 | 4/4 | 15/15 | 16/18 | 18/18 | 4/4 | 18/18 | 18/18 | 18/18 | 4/4 | 15/18 | 12/18 | 17/18 | 17/18 | 4/4 | 17/18 | 13/17 |
| 22% | 39% | 100% | 100% | 100% | 89% | 100% | 100% | 100% | 100% | 100% | 100% | 83% | 67% | 94% | 94% | 100% | 94% | 76% | |
| Escherichia | 12/12 | 10/12 | 0/12 | 4/8 | 4/12 | 0/12 | 11/11 | 8/9 | 12/12 | 11/11 | 11/11 | 6/7 | 12/12 | 4/12 | 3/10 | 1/11 | 0/7 | 6/12 | 3/12 |
| 100% | 83% | 0% | 50% | 33% | 0% | 100% | 89% | 100% | 100% | 100% | 86% | 100% | 33% | 30% | 9% | 0% | 50% | 25% | |
TABLE 4.
Antibiogram for combinations of commonly used antimicrobials. Number sensitive/total number tested and percentage (and 95% CI). Enterococcus, Staphylococcus, Streptococcus, and Escherichia were the most commonly identified isolates.
| Isolate(s) | Ampicillin + amikacin | Penicillin + amikacin | Penicillin + gentamicin | Ampicillin + gentamicin |
|---|---|---|---|---|
| All aerobes | 81/90 | 65/90 | 66/90 | 79/90 |
| 90% (82%–95%) | 72% (62%–81%) | 73% (63%–82%) | 88% (79%–94%) | |
| Gram positive | 59/67 | 46/67 | 51/67 | 60/67 |
| 88% (78%–95%) | 69% (56%–80%) | 76% (64%–86%) | 90% (80%–96%) | |
| Gram negative | 22/23 | 19/23 | 15/23 | 19/23 |
| 96% (78%–100%) | 83% (61%–95%) | 65% (43%–84%) | 83% (61%–95%) | |
| Enterococcus | 20/24 | 7/24 | 9/24 | 20/24 |
| 83% (63%–95%) | 29% (13%–51%) | 38% (19%–59%) | 83% (63%–95%) | |
| Staphylococcus | 15/18 | 17/18 | 18/18 | 18/18 |
| 83% (59%–96%) | 94% (73%–99%) | 100% (82%–100%) | 100% (82%–100%) | |
| Streptococcus | 15/18 | 17/19 | 16/18 | 15/18 |
| 83% (59%–96%) | 89% (67%–99%) | 89% (65%–99%) | 83% (59%–96%) | |
| Escherichia | 12/12 | 12/12 | 10/12 | 10/12 |
| 100% (74%–100%) | 100% (74%–100%) | 83% (52%–98%) | 83% (52%–98%) |
Of the 91 isolates, 21% (19/91; 95% CI: 13%–31%) were classified as MDR from 18 foals (9%, 18/208; 95% CI: 5%–13%). One foal had an MDR Escherichia identified on both the first and third blood cultures. The MDR bacteria were most commonly identified from first blood cultures (9/19; 47%; 95% CI: 24%–71%). Enterococcus was the most common isolate that was MDR (7/19, 37%; 95% CI: 16%–62%; Table 5).
TABLE 5.
Number of multidrug resistant (MDR) bacterial isolates detected, and types of fluid sample submitted from 381 fluid samples from 208 foals.
| MDR bacteria isolated | Number of isolate/total number of isolates (percentage and 95% CI) | Fluid submission type |
|---|---|---|
| Enterococcus | 7/19 (37%, 16%–62%) | Blood (n = 7; first culture [n = 4], second culture [n = 1], third culture [n = 1], fourth culture [n = 1]) |
| Escherichia | 5/19 (26%, 9%–51%) | Blood (n = 4; first culture [n = 3], third culture [n = 1]), synovial (n = 1) |
| Staphylococcus | 2/19 (11%, 1%–33%) | Blood—first culture (n = 2) |
| Actinobacillus | 2/19 (11%, 1%–33%) | Synovial (n = 2) |
| Bacillus | 1/19 (5%, 0.1%–26%) | Blood—fourth culture (n = 1) |
| Klebsiella | 1/19 (5%, 0.1%–26%) | Blood—second culture (n = 1) |
| Enterobacter | 1/19 (5%, 0.1%–26%) | Synovial (n = 1) |
3.4. Survival of Foals
Survival data was available for all foals and 86.5% (180/208; 95% CI: 81%–91%) survived to discharge from hospital. Of those that survived to discharge, 43.9% (79/180; 95% CI: 37%–52%) had at least one bacterial isolate cultured from at least one fluid sample, and 8.9% (16/180; 95% CI: 5%–14%) of foals had an MDR isolate cultured on any given sample. Of the 13.5% of foals (28/208; 95% CI: 9%–19%) that died or were euthanized, 36% (10/28; 95% CI: 19%–56%) had at least one bacterial isolate cultured from at least one fluid sample, and 7% (2/28; 95% CI: 1%–24%) foals had an MDR isolate cultured on any given sample. No significant associations were identified on univariable mixed effects logistic regression between mortality and the presence of an MDR isolate or mortality and a positive culture (Table 6).
TABLE 6.
Univariable generalized mixed effect logistic regression model of risk factors associated with mortality in foals admitted to a hospital with blood or synovial samples or both submitted for culture and susceptibility.
| Variable | Odds ratio | Lower 95% CI | Upper 95% CI | p |
|---|---|---|---|---|
| Positive culture | 0.9 | 0.3 | 2.3 | 0.8 |
| MDR identified | 0.7 | 0.1 | 5.9 | 0.7 |
4. Discussion
4.1. Positive Culture Frequency
A positive culture was obtained in 23.4% of the samples obtained in our study. This result is at the lower end of what is reported in other studies (23.4%–49%) [11, 27]. This difference may be related to prior antimicrobial administration, which was not recorded in our study. However, conflicting evidence exists in foals regarding prior antimicrobial administration and lower positive culture frequency [1, 16, 18, 19]. In people, prior antimicrobial treatment can decrease positive blood culture by > 50% when given 2–72 h before blood culture, with the largest decrease (up to 9.1%) seen in the first 5 h [28]. The percentage of patients with a positive culture before IV antimicrobials, however, was only 25% [28]. Another hypothesis for lower positive culture rates was that fewer cases had sepsis. Reasons for admission and taking bacterial cultures, and variables at admission were not recorded in our study, and thus it is not possible to determine if signs of sepsis were present in these neonates [9]. It is also possible that bacterial culture techniques resulted in false negative results and therefore a lower positive culture frequency. In one study, blood culture failed to detect isolates in 40% of septic foals [29]. Currently, no standardized technique exists for collection of blood culture samples from foals. In our study, blood culture medium bottles were filled with 5–10 mL blood as previously described [11, 13, 15, 17, 18, 19]. Evidence in adult humans indicates that repeated sampling and increasing the volume from 10 to 30 mL increased the likelihood of a positive culture yield by 61% [30]. A potentially interesting observation was that a higher proportion (47%, 16/34) of samples was positive on culture in 2023 when a different type of culture medium bottle with resins was used. Evidence indicates that using resins in blood culture medium binds antimicrobials and can increase the frequency of positive culture rates of Escherichia coli in horses [31]. Further research is needed to determine if increasing the volume of blood collected and the type of culture medium bottle used increases the sensitivity of blood culture in bacteremic foals.
4.2. Isolate Frequency
The frequency of Gram‐positive isolates (75%) follows and exceeds current trends towards predominantly Gram‐positive infections in foals [7, 8, 10, 11, 17, 19, 20]. A previous study also identified 75% gram‐positive isolates, but that study only examined blood culture results [19]. The location of sampling potentially can affect the detection of gram‐negative bacteria, with an under‐diagnosis of gram‐negative infections when using blood culture alone [29]. Synovial samples in our study had a higher frequency of Gram‐negative (48%) isolates compared with blood cultures. Both Gram‐positive and Gram‐negative bacteria are identified in sepsis arising from synovial infections in horses, with a reported increasing predominance of gram‐positive isolates since 2008 [32, 33]. Only 2/10 foals that had concurrent blood and synovial samples taken had the same isolate identified on both samples. These results further emphasize the importance of sampling location, especially if localizing signs of infection are present.
Anaerobic bacteria only were identified in one sample, compared to reports of 1.2%–6% [9, 13, 15, 17, 18, 29, 33, 34, 35] with one study reporting as high as 13% [10]. Although the difference in our study could be attributed to the lack of anaerobic samples tested in 2023, 82% of the other 331 samples were tested, and therefore this factor is unlikely to have been relevant. A consensus is lacking in humans and equids regarding the routine use of both aerobic and anaerobic (dual) cultures [27, 36]. Facultative anaerobes may only be identified in anaerobic cultures compared to aerobic, supporting the use of dual cultures in people [36, 37, 38]. Where collection volume is limited, such as in blood samples or synovial fluid samples in neonates, it may be beneficial to submit a single aerobic culture to increase the volume in a single sample and improve detection rates [27, 30]. Our results do not support an emerging trend in anaerobic culture as hypothesized previously [10]. However, more research is needed to determine if anaerobic infections affect outcome in equine neonates and therefore whether routine inclusion of anaerobic cultures is necessary given their low incidence.
Enterococcus was the most commonly isolated bacteria in our study. Enterococcus spp. was identified as the most common cause of bacteremia in 29% of foals with diarrhea in one study [16]. The indication for performing fluid culture was not recorded in our study, and thus it is possible that this finding is due to the underlying disease process or selection due to prior antimicrobial treatment. Enterococcus has emerged in recent years as an increasingly important cause of sepsis in humans and foals, and the high prevalence in our study is concerning [7, 11, 39]. In England, there also has been a recent trend of Enterococcus faecium being more frequently identified than Enterococcus faecalis in humans [40]. In cases of sepsis in humans, E. faecium has been associated with higher mortality and antimicrobial resistance compared with E. faecalis [40]. Two previous studies reported a higher prevalence of E. faecium than E. faecalis in foals [7, 41]. Increased antimicrobial resistance was reported previously in foals with E. faecium compared with E. faecalis, and foals that cultured Enterococcus were significantly less likely to survive than foals that cultured other bacteria [41]. It was not possible to determine Enterococcus species in our study, but given the emerging trends, ongoing surveillance of enterococcal species in horses, along with restrictive use of antimicrobial drugs, is crucial to prevent further resistance [42].
4.3. Antimicrobial Susceptibility
Some of the antimicrobials evaluated in our study are classified as critically important antimicrobials by the WHO [6]. These drugs should be reserved for cases in which no other alternatives are effective and only after appropriate susceptibility testing or when evidence for their use in certain diseases is compelling [43].
Our study is in agreement with other research that the combined use of ampicillin and amikacin in foals is an appropriate choice for both Gram‐negative and positive isolates [2, 10]. Ampicillin also showed good in vitro efficacy against Enterococcus, further supporting its use in this setting [41, 44]. Although it has been suggested, given increased resistance of E. faecium to ampicillin, that chloramphenicol would be a more appropriate choice [41]. Chloramphenicol was not included in our study because it was infrequently tested during the study period, and enterococcal species identification was not performed routinely. Chloramphenicol has substantial drawbacks to its use in neonates, including limited Gram‐negative enteric spectrum, bacteriostatic nature, lack of IV formulation, and risk of toxicity in humans [44]. Chloramphenicol may be an appropriate choice for older foals with localized Enterococcus infections, such as those of the lower urogenital tract, but its use should be guided by culture and susceptibility testing with consideration of its pharmacokinetics [41, 44]. The pharmacokinetics of chloramphenicol in equine neonates changes significantly in the first 6 weeks [45, 46]. Therefore, the dose and frequency of dosing must be appropriate for the age of the foal. Even at higher doses, the use of chloramphenicol may be limited to bacteria with a minimum inhibitory concentration (MIC) < 2 μg/L because of inadequate serum concentrations needed for therapeutic efficacy [47, 48].
Our study supports the possible use of cephalosporins for foals in which aminoglycosides are not an option (e.g., those with concurrent azotemia) [3]. Breakpoints for cephalosporins have not been established in foals, but the labeled dosage of ceftiofur for adults is considered to be effective against most Gram‐positive respiratory tract pathogens [49]. The higher dosages of ceftiofur used in neonates could provide sufficient in vivo susceptibility for bacteria with higher in vitro breakpoints, such as Gram‐negative bacteria, but are unlikely to be effective for Enterococcus because of intrinsic resistance [49, 50]. Cefotaxime was the most effective antimicrobial in vitro for all aerobic isolates (99%, 73/74; 95% CI: 93%–100%). However, these include Enterococcus, which has intrinsic resistance to cephalosporins and has unpredictable in vivo susceptibility patterns to other antimicrobials [7, 17, 41]. Consequently, susceptibility results are even more important if Enterococcus is identified. If Enterococcus is removed from the antibiogram, cefotaxime and ceftiofur are still effective in 98% (61/63) and 94% (62/66) of all aerobes, respectively. However, despite the popular use of ceftiofur for foals in the UK, because of the prevalence of Enterococcus, cephalosporins may not be considered a good empirical antimicrobial choice in this population [5, 44].
Other beta‐lactam antimicrobials that were effective in vitro were ticarcillin‐clavulanic acid and piperacillin‐tazobactam. The use of these antipseudomonal penicillin antimicrobials is reported infrequently in veterinary literature and both antimicrobials were classified in 2018 under WHO guidelines for “human use only” [6, 51, 52, 53].
4.4. MDR Isolates
Few studies have reported the frequency of MDR isolates in fluid cultures from foals, with a reported range of 13%–44.3% for all isolates [8, 10, 11, 17, 41]. In our study, Enterococcus was the most commonly isolated bacteria and the most likely to be MDR (29% [7/24] of all Enterococcus isolated were MDR). Enterococcus spp. have intrinsic in vivo resistance to cephalosporins, aminoglycosides, and potentiated sulfonamides, and these groups were not included when defining MDR. Exclusion of intrinsically resistant antimicrobials when reporting MDR isolates is not consistently performed across studies, making comparison among studies difficult. In one study, 59% of Enterococcus were MDR and associated with a higher mortality rate in foals compared to non‐MDR Enterococcus [41]. Too few samples were available to investigate an association in our study for MDR compared with non‐MDR Enterococcus. However, it is concerning that 21% of all isolates tested were MDR, further emphasizing the importance of performing susceptibility testing in foals to ensure appropriate antimicrobial selection, especially for MDR isolates [10].
4.5. Survival of Foals
Survival of hospitalized, critically ill neonatal foals since 2007 is reported to be 52%–80.6% [1, 9, 10, 11, 16, 17, 18, 20, 34, 35, 54]. Because sepsis scores and variables at admission were not evaluated, the higher survival rate (87%) in our study could be related to case selection bias, less severe disease, or lower sepsis scores in our population. Caution is required when comparing survival results between studies due to the differences in defining the diagnosis of sepsis and inclusion criteria. However, the proximity of referrals is an important factor that could influence survival in this population. Most referrals are within 60 min of the hospital, and decreased duration of disease before referral has been associated with better outcomes in sepsis in foals [55]. Our results are in agreement with those of other studies in that survival was not associated with the presence of a positive culture [11, 16, 17, 18, 20, 54, 55]. In agreement with other studies, foals identified with a MDR bacteria were not less likely to survive compared to foals when no MDR isolates were detected [10, 11]. We did not evaluate whether correct selection of antimicrobials impacted survival, but this factor has been discussed in other studies [2, 10].
4.6. Limitations
The most important limitation of our study was its retrospective design and missing data regarding admission variables, reason for referral, and whether or not antimicrobials had been administered before referral. Case selection bias was present because a single hospital was used with a mostly Thoroughbred population. This choice was made to minimize inconsistency in sampling technique and sample handling. Antibiograms vary geographically and therefore extrapolation of our results to other populations should be made cautiously [56]. Antibiograms ideally are created and used by individual hospitals or groups of hospitals because the source population will be most applicable to the patient [57]. Therefore, development of regional or local antibiograms is recommended as part of antimicrobial stewardship in veterinary medicine [56, 57, 58]. Because of the duration of the study, inconsistencies existed with the type of blood culture medium bottle and availability of discs for AST. The disc diffusion technique has inherent limitations compared with other methods such as antimicrobial gradient or broth dilution methods [59]. Disc diffusion is a well‐standardized technique with zone diameter breakpoints, but fewer veterinary breakpoints are available compared with MIC breakpoints [25]. Disc diffusion is a widely used AST method in clinical laboratories, and the results are relevant to practitioners [23]. Newer methods for faster isolate identification such as matrix‐assisted laser desorption ionization‐time‐of‐flight (MALDI‐TOF) mass spectrometry are reported in veterinary species, with some commercial laboratories in the United Kingdom starting to use this method for isolate identification [60]. However, for all methods used, it is important to remember that in vitro does not equate to in vivo susceptibility, especially when considering the in vivo synergistic effects of some antimicrobial combinations [44]. This factor should be considered when interpreting susceptibility results in clinical cases.
5. Conclusion
Our results support the use of ampicillin and amikacin in combination in this population of equine neonates as a first‐line antimicrobial. A high frequency of gram‐positive isolates was found, most notably Enterococcus. The use of culture and susceptibility to guide antimicrobial choices remains critically important. Given the unpredictable susceptibility of Enterococcus and frequency of MDR Enterococcus isolates in our study, the value of culture and susceptibility has been further emphasized to ensure appropriate antimicrobial choices.
Disclosure
This retrospective paper discusses the susceptibility testing of amoxicillin, piperacillin and tazobactam, clarithromycin, ceftriaxone, amikacin, rifampicin, doxycycline, ampicillin, ticarcillin and clavulanic acid, erythromycin, cefotaxime, azithromycin, cefquinome, and marbofloxacin but not used in the study.
Ethics Statement
Research ethics committee oversight not required for retrospective study of clinical records. Owners signed consent forms that explained that data from the medical records might be used for research in general. Authors declare human ethics approval was not needed.
Conflicts of Interest
The authors declare no conflicts of interest.
Acknowledgments
Acknowledgments to Bianca Tella, Kevin Grimes, Susan Yarrow, and Alastair Foote for their assistance in data collection and interpretation, and Jack Graham for assistance with Microsoft Excel.
Graham A. E., Colgate V. A., and Floyd E. F., “Antibiograms of Bacterial Cultures From Equine Neonates at a United Kingdom Hospital: 381 Samples (2018–2023),” Journal of Veterinary Internal Medicine 39, no. 5 (2025): e70198, 10.1111/jvim.70198.
Funding: The authors received no specific funding for this work.
References
- 1. Giguère S., Weber E. J., and Sanchez L. C., “Factors Associated With Outcome and Gradual Improvement in Survival Over Time in 1065 Equine Neonates Admitted to an Intensive Care Unit,” Equine Veterinary Journal 49, no. 1 (2017): 45–50, 10.1111/evj.12536. [DOI] [PubMed] [Google Scholar]
- 2. Theelen M. J. P., Wilson W. D., Byrne B. A., Edman J. M., Kass P. H., and Magdesian K. G., “Initial Antimicrobial Treatment of Foals With Sepsis: Do Our Choices Make a Difference?,” Veterinary Journal 243 (2019): 74–76, 10.1016/j.tvjl.2018.11.012. [DOI] [PubMed] [Google Scholar]
- 3. Floyd E. F., Easton‐Jones C. A., and Theelen M. J. P., “Systemic Antimicrobial Therapy in Foals,” Equine Veterinary Education 34, no. 1 (2022): 49–56, 10.1111/eve.13467. [DOI] [Google Scholar]
- 4. Theelen M. J. P., Wilson W. D., Byrne B. A., et al., “Differences in Isolation Rate and Antimicrobial Susceptibility of Bacteria Isolated From Foals With Sepsis at Admission and After ≥48 Hours of Hospitalization,” Journal of Veterinary Internal Medicine 34, no. 2 (2020): 955–963, 10.1111/jvim.15692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Wilson A., Mair T., Williams N., McGowan C., and Pinchbeck G., “Antimicrobial Prescribing and Antimicrobial Resistance Surveillance in Equine Practice,” Equine Veterinary Journal 55, no. 3 (2023): 494–505, 10.1111/evj.13587. [DOI] [PubMed] [Google Scholar]
- 6. World Health Organisation, WHO Advisory Group on Integrated Surveillance of Antimicrobial Resistance (AGISAR) , Criticall Important Antimicrobials for Human Medicine, 6th Revision (World Health Organization, 2018), https://www.who.int/publications/i/item/9789241515528. [Google Scholar]
- 7. Theelen M. J., Wilson W. D., Edman J. M., Magdesian K. G., and Kass P. H., “Temporal Trends in Prevalence of Bacteria Isolated From Foals With Sepsis: 1979‐2010,” Equine Veterinary Journal 46, no. 2 (2014): 169–173, 10.1111/evj.12131. [DOI] [PubMed] [Google Scholar]
- 8. Toombs‐Ruane L. J., Riley C. B., Kendall A. T., Hill K. E., Benschop J., and Rosanowski S. M., “Antimicrobial Susceptibility of Bacteria Isolated From Neonatal Foal Samples Submitted to a New Zealand Veterinary Pathology Laboratory (2004 to 2013),” New Zealand Veterinary Journal 64, no. 2 (2016): 107–111, 10.1080/00480169.2015.1109006. [DOI] [PubMed] [Google Scholar]
- 9. Wong D. M., Ruby R. E., Dembek K. A., et al., “Evaluation of Updated Sepsis Scoring Systems and Systemic Inflammatory Response Syndrome Criteria and Their Association With Sepsis in Equine Neonates,” Journal of Veterinary Internal Medicine 32, no. 3 (2018): 1185–1193, 10.1111/jvim.15087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Bookbinder L. C., Mani R., and Carr E. A., “Antibiograms of Field and Hospital Acquired Equine Neonatal Bacterial Fluid Cultures in the Midwestern United States: 149 Samples (2007‐2018),” Journal of Veterinary Internal Medicine 37, no. 3 (2023): 1193–1200, 10.1111/jvim.16671. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Flood J. A., Collins N. M., Russell C. M., Cuming R. S., Carrick J. B., and Cudmore L. A., “Blood Culture Isolates and Antimicrobial Sensitivities From 1621 Critically Ill Neonatal Foals (2005‐2022),” Australian Veterinary Journal 103, no. 4 (2025): 163–170, 10.1111/avj.13423. [DOI] [PubMed] [Google Scholar]
- 12. Hritcu O., Schmidt V. M., Salem S. E., et al., “Geographical Variations in Virulence Factors and Antimicrobial Resistance Amongst Staphylococci Isolated From Dogs From the United Kingdom and Romania. Frontiers in Veterinary,” Science 7 (2020): 414, 10.3389/fvets.2020.00414. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Koterba A. M., Brewer B. D., and Tarplee F. A., “Clinical and Clinicopathological Characteristics of the Septicaemic Neonatal Foal: Review of 38 Cases,” Equine Veterinary Journal 16, no. 4 (1984): 376–382, 10.1111/j.2042-3306.1984.tb01950.x. [DOI] [PubMed] [Google Scholar]
- 14. Hoffman A. M., Staempfli H. R., and Willan A., “Prognostic Variables for Survival of Neonatal Foals Under Intensive Care,” Journal of Veterinary Internal Medicine 6, no. 2 (1992): 89–95, 10.1111/j.1939-1676.1992.tb03157.x. [DOI] [PubMed] [Google Scholar]
- 15. Marsh P. S. and Palmer J. E., “Bacterial Isolates From Blood and Their Susceptibility Patterns in Critically Ill Foals: 543 Cases (1991‐1998),” Journal of the American Veterinary Medical Association 218, no. 1610 (2001): 1608, 10.2460/javma.2001.218.1608. [DOI] [PubMed] [Google Scholar]
- 16. Hollis A. R., Wilkins P. A., Palmer J. E., and Boston R. C., “Bacteremia in Equine Neonatal Diarrhea: A Retrospective Study (1990‐2007),” Journal of Veterinary Internal Medicine 22, no. 5 (2008): 1203–1209, 10.1111/j.1939-1676.2008.0152.x. [DOI] [PubMed] [Google Scholar]
- 17. Russell C. M., Axon J. E., Blishen A., and Begg A. P., “Blood Culture Isolates and Antimicrobial Sensitivities From 427 Critically Ill Neonatal Foals,” Australian Veterinary Journal 86, no. 7 (2008): 266–271, 10.1111/j.1751-0813.2008.00311.x. [DOI] [PubMed] [Google Scholar]
- 18. Hytychová T. and Bezděková B., “Retrospective Evaluation of Blood Culture Isolates and Sepsis Survival Rate in Foals in The Czech Republic: 50 Cases (2011‐2013),” Journal of Veterinary Emergency and Critical Care 25, no. 5 (2015): 660–666, 10.1111/vec.12348. [DOI] [PubMed] [Google Scholar]
- 19. Fouché N., Gerber V., Thomann A., and Perreten V., “Antimicrobial Susceptibility Patterns of Blood Culture Isolates From Foals in Switzerland,” Schweizer Archiv für Tierheilkunde 160, no. 11 (2018): 665–671, 10.17236/sat00184. [DOI] [PubMed] [Google Scholar]
- 20. Furr M. and H. McKenzie, III , “Factors Associated With the Risk of Positive Blood Culture in Neonatal Foals Presented to a Referral Center (2000‐2014),” Journal of Veterinary Internal Medicine 34, no. 6 (2020): 2738–2750, 10.1111/jvim.15923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21. Fernandez T., Robin M., Baillie S., and Ubert B., “Blood Culture Results and Antimicrobial Sensitivity Patterns in Foals From a Veterinary Hospital in Doha, Qatar,” Equine Veterinary Journal 56, no. S59 (2024): 25, 10.1111/evj.14176. [DOI] [Google Scholar]
- 22. The European Committee on Antimicrobial Susceptibility Testing , Definitions of S, I and R (EUCAST, 2019), https://www.eucast.org/newsiandr. [Google Scholar]
- 23. The European Committee on Antimicrobial Susceptibility Testing , Antimicrobial Susceptibility Testing: EUCAST Disk Diffusion Method Manual, Version 12.0 (EUCAST, 2024), https://www.eucast.org/fileadmin/src/media/PDFs/EUCAST_files/Disk_test_documents/2024_manuals/Manual_v_12.0_EUCAST_Disk_Test_2024.pdf. [Google Scholar]
- 24. The European Committee on Antimicrobial Susceptibility Testing , Breakpoint Tables for Interpretation of MICs and Zone Diameters, Version 14.0 (EUCAST, 2024), https://www.eucast.org/clinical_breakpoints. [Google Scholar]
- 25. Clinical & Laboratory Standards Institute , CLSI M100 Performance Standards for Antimicrobial Susceptibility Testing, 30th ed. (CLSI, 2020), https://clsi.org/standards/products/microbiology/documents/m100/. [Google Scholar]
- 26. Magiorakos A. P., Srinivasan A., Carey R. B., et al., “Multidrug‐Resistant, Extensively Drug‐Resistant and Pandrug‐Resistant Bacteria: An International Expert Proposal for Interim Standard Definitions for Acquired Resistance,” Clinical Microbiology and Infection 18, no. 3 (2012): 268–281, 10.1111/j.1469-0691.2011.03570.x. [DOI] [PubMed] [Google Scholar]
- 27. Giancola S. and Hart K. A., “Equine Blood Cultures: Can We Do Better?,” Equine Veterinary Journal 55, no. 4 (2023): 584–592, 10.1111/evj.13891. [DOI] [PubMed] [Google Scholar]
- 28. Rand K. H., Beal S. G., Rivera K., Allen B., Payton T., and Lipori G. P., “Hourly Effect of Pretreatment With IV Antibiotics on Blood Culture Positivity Rate in Emergency Department Patients,” Open Forum Infectious Diseases 6, no. 5 (2019): ofz179, 10.1093/ofid/ofz179. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Wilson W. D. and Madigan J. E., “Comparison of Bacteriologic Culture of Blood and Necropsy Specimens for Determining the Cause of Foal Septicemia: 47 Cases (1978‐1987),” Journal of the American Veterinary Medical Association 195, no. 12 (1989): 1759–1763. [PubMed] [Google Scholar]
- 30. Schelonka R. L., Chai M. K., Yoder B. A., Hensley D., Brockett R. M., and Ascher D. P., “Volume of Blood Required to Detect Common Neonatal Pathogens,” Journal of Pediatrics 129, no. 2 (1996): 275–278, 10.1016/s0022-3476(96)70254-8. [DOI] [PubMed] [Google Scholar]
- 31. Lorenzo‐Figueras M., Pusterla N., Byrne B. A., and Samitz E. M., “In Vitro Evaluation of Three Bacterial Culture Systems for the Recovery of Escherichia coli From Equine Blood,” American Journal of Veterinary Research 67, no. 12 (2006): 2025–2029, 10.2460/ajvr.67.12.2025. [DOI] [PubMed] [Google Scholar]
- 32. Pearson G. B., Papa B., Mosaddegh A., et al., “Equine Synovial Sepsis Laboratory Submissions Yield a Low Rate of Positive Bacterial Culture and a High Prevalence of Antimicrobial Resistance,” American Journal of Veterinary Research 84, no. 8 (2023): 1–10, 10.2460/ajvr.23.05.0085. [DOI] [PubMed] [Google Scholar]
- 33. Vos N. J. and Ducharme N. G., “Analysis of Factors Influencing Prognosis in Foals With Septic Arthritis,” Irish Veterinary Journal 61, no. 2 (2008): 102–106, 10.1186/2046-0481-61-2-102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Corley K. T., Pearce G., Magdesian K. G., and Wilson W. D., “Bacteraemia in Neonatal Foals: Clinicopathological Differences Between Gram‐Positive and Gram‐Negative Infections, and Single Organism and Mixed Infections,” Equine Veterinary Journal 39, no. 1 (2007): 84–89, 10.2746/042516407x157585. [DOI] [PubMed] [Google Scholar]
- 35. Sanchez L. C., Giguère S., and Lester G. D., “Factors Associated With Survival of Neonatal Foals With Bacteremia and Racing Performance of Surviving Thoroughbreds: 423 Cases (1982‐2007),” Journal of the American Veterinary Medical Association 233, no. 9 (2008): 1446–1452, 10.2460/javma.233.9.1446. [DOI] [PubMed] [Google Scholar]
- 36. Noh G. Y., Park Y. S., Kim S. H., Song S. A., and Shin J. H., “Clinical Usefulness of Anaerobic Blood Culture in Pediatric Patients With Bacteremia,” Anaerobe 84 (2023): 102804, 10.1016/j.anaerobe.2023.102804. [DOI] [PubMed] [Google Scholar]
- 37. Gottschalk A., Coggins S., Dhudasia M. B., et al., “Utility of Anaerobic Blood Cultures in Neonatal Sepsis Evaluation,” Journal of the Pediatric Infectious Diseases Society 13, no. 8 (2024): 406–412, 10.1093/jpids/piae056. [DOI] [PubMed] [Google Scholar]
- 38. Messbarger N. and Neemann K., “Role of Anaerobic Blood Cultures in Neonatal Bacteremia,” Journal of the Pediatric Infectious Diseases Society 7, no. 3 (2018): e65–e69, 10.1093/jpids/pix088. [DOI] [PubMed] [Google Scholar]
- 39. Horner C., Mushtaq S., Allen M., et al., “Replacement of Enterococcus faecalis by Enterococcus faecium as the Predominant Enterococcus in UK Bacteraemias,” JAC‐Antimicrobial Resistance 3, no. 4 (2021): dlab185, 10.1093/jacamr/dlab185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40. UK Health Security Agency , “Laboratory Surveillance of Enterococcus spp. Bacteraemia (England): 2021,” updated 2023, https://www.gov.uk/government/publications/enterococcus‐spp‐bacteraemia‐voluntary‐surveillance‐2021/laboratory‐surveillance‐of‐enterococcus‐spp‐bacteraemia‐england‐2021#acknowledgements.
- 41. Willis A. T., Magdesian K. G., Byrne B. A., and Edman J. M., “ Enterococcus Infections in Foals,” Veterinary Journal 248 (2019): 242–247, 10.1016/j.tvjl.2019.04.005. [DOI] [PubMed] [Google Scholar]
- 42. Wei Y., Palacios Araya D., and Palmer K. L., “ Enterococcus faecium : Evolution, Adaptation, Pathogenesis and Emerging Therapeutics,” Nature Reviews Microbiology 22, no. 11 (2024): 705–721, 10.1038/s41579-024-01058-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Bowen M., “Antimicrobial Stewardship: Time for Change,” Equine Veterinary Journal 45, no. 2 (2013): 127–129, 10.1111/evj.12041. [DOI] [PubMed] [Google Scholar]
- 44. Magdesian K. G., “Antimicrobial Pharmacology for the Neonatal Foal,” Veterinary Clinics of North America. Equine Practice 33, no. 1 (2017): 47–65, 10.1016/j.cveq.2016.12.004. [DOI] [PubMed] [Google Scholar]
- 45. Adamson P. J., Wilson W. D., Baggot J. D., Hietala S. K., and Mihalyi J. E., “Influence of Age on the Disposition Kinetics of Chloramphenicol in Equine Neonates,” American Journal of Veterinary Research 52, no. 3 (1991): 426–431. [PubMed] [Google Scholar]
- 46. Brumbaugh G. W., Martens R. J., Knight H. D., and Martin M. T., “Pharmacokinetics of Chloramphenicol in the Neonatal Horse,” Journal of Veterinary Pharmacology and Therapeutics 6, no. 3 (1983): 219–227, 10.1111/j.1365-2885.1983.tb00467.x. [DOI] [PubMed] [Google Scholar]
- 47. Patel T., Magdesian K. G., Estell K. E., Edman J. M., and Knych H. K., “Pharmacokinetics of Chloramphenicol Base in Horses and Comparison to Compounded Formulations,” Journal of Veterinary Pharmacology and Therapeutics 42, no. 6 (2019): 609–616, 10.1111/jvp.12777. [DOI] [PubMed] [Google Scholar]
- 48. Estell K. E., Knych H. K., Patel T., Edman J. M., and Magdesian K. G., “Pharmacokinetics of Multiple Doses of Chloramphenicol in Fed Adult Horses,” Veterinary Journal 257 (2020): 105446, 10.1016/j.tvjl.2020.105446. [DOI] [PubMed] [Google Scholar]
- 49. Ryan C. A., McNeal C. D., and Credille B. C., “Ceftiofur Use and Antimicrobial Stewardship in the Horse,” Equine Veterinary Journal 55, no. 6 (2023): 944–961, 10.1111/evj.13930. [DOI] [PubMed] [Google Scholar]
- 50. Meyer S., Giguère S., Rodriguez R., Zielinski R. J., Grover G. S., and Brown S. A., “Pharmacokinetics of Intravenous Ceftiofur Sodium and Concentration in Body Fluids of Foals,” Journal of Veterinary Pharmacology and Therapeutics 32, no. 4 (2009): 309–316, 10.1111/j.1365-2885.2008.01041.x. [DOI] [PubMed] [Google Scholar]
- 51. Lappin M. R., Blondeau J., Boothe D., et al., “Antimicrobial Use Guidelines for Treatment of Respiratory Tract Disease in Dogs and Cats: Antimicrobial Guidelines Working Group of the International Society for Companion Animal Infectious Diseases,” Journal of Veterinary Internal Medicine 31, no. 2 (2017): 279–294, 10.1111/jvim.14627. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Prescott J. F., “Other Beta‐Lactam Antibiotics,” in Antimicrobial Therapy in Veterinary Medicine, ed. Giguère S., Prescott J. F., and Dowling P. M. (Wiley, 2013), 175–187, 10.1002/9781118675014.ch10. [DOI] [Google Scholar]
- 53. Sweeney R. W., Beech J., and Simmons R. D., “Pharmacokinetics of Intravenously and Intramuscularly Administered Ticarcillin and Clavulanic Acid in Foals,” American Journal of Veterinary Research 49, no. 1 (1988): 23–26. [PubMed] [Google Scholar]
- 54. Hepworth‐Warren K. L., Wong D. M., Fulkerson C. V., Wang C., and Sun Y., “Bacterial Isolates, Antimicrobial Susceptibility Patterns, and Factors Associated With Infection and Outcome in Foals With Septic Arthritis: 83 Cases (1998‐2013),” Journal of the American Veterinary Medical Association 246, no. 7 (2015): 785–793, 10.2460/javma.246.7.785. [DOI] [PubMed] [Google Scholar]
- 55. Gayle J. M., Cohen N. D., and Chaffin M. K., “Factors Associated With Survival in Septicemic Foals: 65 Cases (1988‐1995),” Journal of Veterinary Internal Medicine 12, no. 3 (1998): 140–146, 10.1111/j.1939-1676.1998.tb02109.x. [DOI] [PubMed] [Google Scholar]
- 56. Burbick C. R., Fajt V. R., Frey E., et al., “Benefits and Challenges of Creating Veterinary Antibiograms for Empiric Antimicrobial Selection in Support of Antimicrobial Stewardship and Advancement of One‐Health Goals,” American Journal of Veterinary Research 84, no. 9 (2023): 1–8, 10.2460/ajvr.23.05.0086. [DOI] [PubMed] [Google Scholar]
- 57. Lorenz C., Silva M., Frey E., Fritz H., and Marshall E., “Antibiograms as One‐Health Tools for Antimicrobial Stewardship: California's Experience With Livestock Antibiogram Development,” Journal of the American Veterinary Medical Association 261, no. 9 (2023): 1272–1280, 10.2460/javma.23.05.0228. [DOI] [PubMed] [Google Scholar]
- 58. Ekakoro J. E., Guptill L., Hendrix K., Anderson M., and Ruple A., “Approaches Used to Construct Antibiograms for Dogs in a Veterinary Teaching Hospital in the United States,” Antibiotics 12, no. 6 (2023): 1034, 10.3390/antibiotics12061034. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59. Jorgensen J. H. and Ferraro M. J., “Antimicrobial Susceptibility Testing: A Review of General Principles and Contemporary Practices,” Clinical Infectious Diseases 49, no. 11 (2009): 1749–1755, 10.1086/647952. [DOI] [PubMed] [Google Scholar]
- 60. Thompson J. E., “Matrix‐Assisted Laser Desorption Ionization‐Time‐of‐Flight Mass Spectrometry in Veterinary Medicine: Recent Advances (2019‐Present),” Veterinary World 15, no. 11 (2022): 2623–2657, 10.14202/vetworld.2022.2623-2657. [DOI] [PMC free article] [PubMed] [Google Scholar]


