Abstract
Introduction
Mild cognitive impairment (MCI) presents a significant risk, with a 75–80% likelihood of progressing to dementia. Despite this high risk, there is currently no straightforward and effective treatment strategy to halt or reverse this progression. Olfactory dysfunction, in conjunction with subjective cognitive decline, offers a crucial opportunity for early intervention in older adults at risk of MCI. Conventional olfactory training (COT) has demonstrated potential in enhancing neuroplasticity, which is vital for cognitive health. Initial studies indicate that modified olfactory training (MOT) may yield superior outcomes compared to COT. Thus, this study aims to evaluate the efficacy of MOT in delaying and preventing the progression of MCI in high-risk individuals.
Methods and analysis
This randomised, controlled, multicentre, prospective and open-label trial will be conducted at Peking University Third Hospital and enrol 114 participants, who will be randomised 1:1:1 into three groups: MOT, COT and a control group. Standardised assessments will be conducted at baseline, 3 months, 6 months, 12 months and 24 months to measure cognitive and olfactory outcomes. The primary outcomes will be the change in Montreal Cognitive Assessment score, neuroimaging assessments and the Sniffin’ Sticks test score. The secondary outcomes will include olfactory bulb volume and several neuroimaging tests.
Ethics and dissemination
This study protocol has been registered with ClinicalTrials.gov and has received approval from the Peking University Third Hospital Medical Science Research Ethics Committee (2023-347-01). The results will be disseminated through publication in scientific peer-reviewed journals.
Trial registration number
Keywords: Dementia, Parkinson-s disease, Adult otolaryngology, Physical Therapy Modalities
Strengths and limitations of this study.
This study innovatively defines the mild cognitive impairment (MCI) high-risk population as older adults presenting with both olfactory dysfunction and subjective cognitive decline, enabling precise and timely intervention.
We employ a modified olfactory training device that incorporates bidirectional airflow and positive pressure, which has independent intellectual property rights, to intervene in MCI progression.
The study uses comprehensive neuroimaging techniques and cognitive function-related questionnaires to detect changes in cognitive abilities following the modified olfactory training, thereby providing robust evidence for its therapeutic efficacy.
As a limitation, the inclusion of a no-intervention control group may introduce potential bias due to variations in participants’ psychological states.
Additionally, the relatively small sample size and specific inclusion criteria limit the generalisability of findings, making it difficult to determine whether olfactory training would be effective for MCI high-risk populations without olfactory dysfunction.
Introduction
Background
Cognitive impairment is a major global public health concern, exacerbated by ageing populations and its substantial socioeconomic burden. In China alone, approximately 38.77 million individuals over the age of 60 suffer from mild cognitive impairment (MCI), while 15.07 million have progressed to dementia.1 The transition from MCI to dementia occurs in approximately 20–40% of cases over 1–6 years follow-ups, with annual conversion rates ranging 10–15%,2 which emphasises the urgent need for early intervention strategies.3 MCI represents an intermediary stage between normal ageing and dementia, with amnestic MCI often progressing to Alzheimer’s Disease (AD) and non-amnestic MCI being linked to vascular and frontotemporal dementia.4 Despite its high prevalence, there is currently no established treatment capable of halting or reversing cognitive decline.5 6
Olfactory dysfunction has emerged as a significant early biomarker of neurodegenerative diseases, with studies indicating that 85–90% of early AD and Parkinson’s disease cases exhibit olfactory impairments.7 8 The prodromal period of these neurodegenerative disorders spans several years, during which early symptoms such as hyposmia (reduced sense of smell) often precede detectable cognitive deficits. This phenomenon is believed to be associated with the accumulation of amyloid-β, tau and α-synuclein proteins within the olfactory system, particularly in the anterior olfactory nucleus.9,13 Structural changes in olfactory regions have been demonstrated using MRI and volumetric analyses, supporting the hypothesis that olfactory decline is an early indicator of neurodegenerative processes.13,15 Moreover, olfactory dysfunction has been significantly correlated with the risk of developing MCI, highlighting its potential utility as a screening tool. A study conducted in rural China found that olfactory identification deficits were significantly associated with both amnestic and non-amnestic MCI.7 Furthermore, a 3-year longitudinal study demonstrated that combining olfactory function assessments with cerebrospinal fluid biomarkers of AD significantly improved the predictive accuracy for cognitive decline.16 This growing body of evidence underscores the necessity of incorporating olfactory assessments into routine cognitive impairment screenings.
Subjective cognitive decline (SCD) has also been identified as a prodromal stage of MCI and dementia. SCD is characterised by self-reported cognitive deficits without objective impairments on neuropsychological tests, yet it significantly increases the risk of future cognitive decline. A meta-analysis of longitudinal studies found that individuals with SCD have a 2.48-fold higher risk of developing dementia (HR=1.90, 95% CI 1.52 to 2.36; OR=2.48, 95% CI 1.97 to 3.14, p=0.006) and a 1.83-fold risk of progressing to MCI (HR=1.73, 95% CI 1.18 to 2.52; OR=1.83, 95% CI 1.56 to 2.16, p=0.007).17 Additionally, long-term observational studies suggest that SCD symptoms can manifest up to 10 years before a formal AD diagnosis, reinforcing its role as an early marker of neurodegeneration.18 Neuroimaging analyses have further revealed that individuals with SCD exhibit structural alterations in brain regions associated with memory and executive function, including the hippocampus and entorhinal cortex.19
Olfactory training (OT) has been recognised as a promising intervention that concurrently enhances olfactory function and cognitive performance. This correlation is underscored by findings that improvements in olfactory function, particularly in odour identification,20 21 are strongly positively correlated with enhancements in cognitive abilities, as evidenced by a correlation coefficient of 0.73 (p<0.001) with the total olfactory score on the Montreal Cognitive Assessment (MoCA)-Japanese assessment.22 The underlying neural mechanisms through which OT may facilitate cognitive improvements are multifaceted. Research indicates a direct neural pathway between the olfactory system and the hippocampus.23 Olfactory signals project directly to the amygdala-hippocampal complex, establishing a direct link that renders OT capable of influencing both psychological and cognitive functions.24 Continuous olfactory stimulation may promote hippocampal grey matter thickening and increase the grey matter density of the hippocampus by enhancing the plasticity of hippocampal neurons, dendritic growth and synaptic connections.25 26 Furthermore, OT has been shown to improve specific cognitive domains, including verbal fluency, working memory and executive functions.27 Olfactory information, originating from the olfactory bulb, undergoes processing and integration through multiple brain regions, including the primary olfactory cortex, orbitofrontal cortex, amygdala, hippocampus and thalamus.28 OT may enhance the processing capabilities of chemosensory information by optimising the efficiency of information transfer through the regulation of synchronous activity among these brain regions.29
Conventional OT methods face several obstacles, including low participant adherence, lack of standardised protocols and suboptimal odourant delivery mechanisms. Current OT devices primarily rely on passive odour diffusion, which can be compromised by nasal airflow obstruction, leading to inadequate odourant deposition in the olfactory epithelium.30 31 In response to these challenges, our research team has developed a modified OT (MOT) device designed to enhance odourant delivery efficiency. Preliminary clinical studies have demonstrated that this modified device increases olfactory cleft odourant deposition rates from 5% to 25% (p<0.001), improves the treatment efficacy for olfactory dysfunction by 30% (p=0.015) and yields significant cognitive improvements32 (figure 1).
Figure 1. Description of modified olfactory training device. During exhalation, the soft palate completely seals off the nasal cavity (A). Exhaled air, now carrying odourants, enters the nose and exits through the opposite nasal passage. This creates a bidirectional airflow through both the left and right nasal cavities (B). This bidirectional delivery method significantly improved odour deposition in the olfactory region while significantly reducing deposition in the anterior nasal cavity.
In patients with MCI, results from OT studies have demonstrated that OT can significantly affect hippocampal thickness and improve cognitive functions. Notably, these improvements are significantly correlated with the compliance brought about by memory enhancements.33 34 Patients with SCD often report subjective cognitive concerns that typically do not correspond to objective deficits on neuropsychological tests. This contrasts with patients with MCI who exhibit measurable cognitive impairments.35 A study validating the Ten-Word Immediate Recall Test found that patients with MCI scored significantly lower than those with SCD (p=0.001).36 These findings suggest that individuals with idiopathic olfactory impairments and SCD, who have not yet progressed to MCI, may exhibit higher compliance and responsiveness to OT. This approach not only leverages the relatively preserved cognitive and neural functions in SCD compared with MCI but also maximises the potential for neuroplastic adaptations in response to sensory training. Thus, initiating OT during SCD could potentially delay or mitigate the progression to more severe cognitive impairments, offering a proactive strategy in managing cognitive decline. Based on these findings, we hypothesise that older adults with idiopathic olfactory dysfunction and SCD, who have not yet developed MCI, will exhibit greater compliance and responsiveness to OT. This randomised controlled trial aims to determine whether modified OT can prevent or delay the progression to MCI, thereby improving quality of life and reducing societal burdens. Specifically, this study will assess the efficacy of modified OT in improving olfactory and cognitive function in high-risk individuals and compare the effectiveness of modified OT devices to conventional devices in enhancing neuroplasticity. Our aims are to investigate the neural mechanisms underlying the cognitive benefits of OT using neuroimaging analyses and provide empirical evidence supporting the integration of olfactory assessments and interventions in dementia prevention strategies. By addressing these research objectives, this study has the potential to contribute to early dementia prevention strategies, ultimately benefiting public health by mitigating the societal and economic burden associated with cognitive disorders.
Objectives
Primary objective
This study aims to determine the extent to which an MOT device prevents progression to MCI in older adults with idiopathic olfactory dysfunction and SCD.
Secondary objectives
This study aims to compare the effectiveness of MOT with conventional OT (COT), and to observe changes in the progression from SCD to MCI through neurological biomarker testing and neurological assessments.
Methods and analysis
Study design
This open-label randomised controlled trial will be conducted at the Department of Otorhinolaryngology, Peking University Third Hospital, and the Neurology Clinic, Peking University Sixth Hospital. Older adults with idiopathic olfactory dysfunction and subjective cognitive decline were recruited starting in March 2025 and assigned evenly to one of the three study arms (24-month traditional OT, 24-month MOT or a blank control) by randomised blinding, based on an a priori randomisation list generated by an independent member of the clinic using the Excel-generated allocation sequence of the chunking randomisation method. At 3, 6, 12 and 24 months after the start of the therapeutic intervention, participants in the three arms will be tested for olfactory function and cognitive function by examiners who are unaware of the subgroups. The olfactory function and cognitive function tests will be completed on the same day. The trial will conclude in October 2027, after the last participant has completed the examination, followed by data analysis. The design overview chart is shown in figure 2, and the details and timeframe of the trial are presented in online supplemental table 1. The SPIRIT (Standard Protocol Items: Recommendations for Interventional Trials) checklist (online supplemental material 1) guided our protocol (2025.7.24 V.1.2) development.
Figure 2. Flow diagram illustrating the study design. COT, conventional olfactory training; MOT, modified olfactory training.
Participants
Eligibility criteria
Inclusion criteria
The participants will be eligible for this study if they meet all of the following criteria:
Age above 60 years.
Diagnosis of idiopathic olfactory dysfunction: Olfactory function was tested using the ‘Sniffin’ Sticks’ test battery. The combined odour threshold, odour discrimination and odour identification score (TDI score) was used to identify olfactory dysfunction (TDI<30.75). Idiopathic olfactory loss was diagnosed if a participant had no history of brain injury, olfactory loss did not coincide with an upper respiratory infection, medication usage or environmental factors, and neurological evaluations revealed no indications of neurodegenerative or neurovascular diseases.
Diagnosis of subjective cognitive decline: (1) self-experienced decline in cognitive functioning, compared with a previously normal cognitive status, which is unrelated to an acute event and (2) normal performance on standardised tests used to classify MCI. Unimpaired cognition was defined as MoCA score >25 for ages 60–79, >24 for ages 80–89 and >23 for ages ≥90.
Consent: Willingness to sign an informed consent form.
Exclusion criteria
The participants will not be eligible for this study if they meet any of the following criteria:
Smoking (current or within 6 months).
Pregnancy.
Acute or chronic sinonasal diseases.
Comorbid chronic diseases including diabetes, bronchopneumonia or chronic obstructive pulmonary disease.
Recent (within 4 weeks) use of psychotropics, corticosteroids, antimicrobials, leukotriene antagonists or antihistamines.
Inability to comply with study requirements: Based on the researcher’s judgement, individuals who are unable to tolerate olfactory function tests and treatment, complete the study or comply with its requirements due to memory or behavioural abnormalities, severe alcohol consumption or previous non-compliance.
Withdrawal criteria
Participants will be free to withdraw their informed consent and withdraw from the study. The investigator may withdraw the participant from the study under any of the following conditions:
Participants withdraw consent.
Serious changes in clinical or laboratory examination results during the study that would be detrimental to the subject’s healthcare as observed by the investigator.
Participants who exhibit poor compliance, refuse to undergo all required tests and cannot adhere to the study plan.
Participants who are unable to attend the follow-up or drop out.
Any participants the investigator deems not clinically able to continue to participate in this study.
Patient and public involvement
Participants or the public will not be involved in the design, conduct, reporting or dissemination plans of this study.
Recruitment
All eligible participants will be recruited through hospital-based screening. The primary recruitment approach involves identifying potential individuals from hospitals directly in the clinic or via an electronic medical record search. After a comprehensive explanation of study objectives, methodologies, potential benefits/risks and data handling protocols, written informed consent will be obtained from all willing participants through investigator-administered documentation. Consented individuals will then undergo a structured screening visit comprising: (1) systematic documentation of demographic characteristics (age, sex, education years), medical/surgical history, current medications and comorbidities; (2) otolaryngologist-conducted olfactory validation using Sniffin’ Sticks testing, nasal endoscopy and paranasal sinus CT to confirm idiopathic olfactory loss by excluding sinonasal/inflammatory causes; and (3) neurologist-administered cognitive prescreening via MoCA to exclude mild cognitive impairment or dementia while verifying subjective cognitive decline through standardised interviews. Participants failing inclusion criteria will be withdrawn, while eligible candidates proceed to baseline assessments.
Randomisation
114 participants will be divided into three equal parts through random allocation. Randomisation will be performed by a computer-generated random number list prepared by independent statisticians with no clinical involvement in the trial. The block randomisation sequence is created using the Excel-generated allocation sequence of the chunking randomisation method. There will be 38 participants in each block. The block sizes will not be disclosed to ensure concealment. After the physicians responsible for recruitment obtain eligible participant consent, the research nurse will contact an online, central randomisation service by a secure computer for allocation consignment. The research nurse will then provide information about the treatment allocation to the intervention physicians.
Interventions
Participants will be allocated at a 1:1:1 ratio into three groups: the MOT group, the COT group (COT) and the control group. The intervention process will be carried out in the Peking University Third Hospital Drug Clinical Trial Center. In the design of our OT intervention, we used three types of odours commonly referenced in classical olfactory dysfunction literature. Notably, we substituted eucalyptus with menthol. Since the scent of menthol is more familiar to the Chinese population and familiarity with the odour can influence the participant’s engagement and response to the intervention. Moreover, menthol has the added advantage of activating not only the olfactory system but also the trigeminal nerve system.37 This dual activation facilitates mixed mechanisms of sensory adaptation and compensation in the interactions between the olfactory and trigeminal systems.38 We also adhered to the guidelines for the treatment of olfactory disorders by administering topical corticosteroids to each group to enhance therapeutic efficacy.39,41
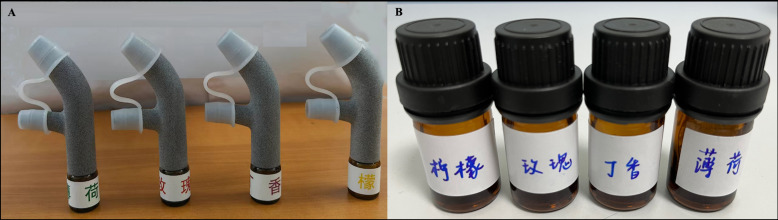
For the MOT Group (figure 3A), participants will use a MOT device based on expiratory pressure. Four odours will be used: rose, mint, lemon and clove. Each odourant is sniffed for 10 s per session, with a 10-s interval between odours. Each training session lasts 5 min and is conducted two times a day, before breakfast and at bedtime, for a duration of 2 years.
Figure 3. The modified olfactory training device (A) and conventional olfactory training device (B).
For the COT group (figure 3B), participants received four brown glass jars (total volume 5 mL) with one of the four odours in each (1 mL each, soaked in cotton pads to prevent spilling). The following odours were used: phenyl ethyl alcohol: rose, menthol: mint, citronellal: lemon and eugenol: cloves. All jars were labelled with the odour name. Participants had to sniff one odour for approximately 10 s and turn to another after a rest of 10 s. Each training session lasts 5 min and is conducted two times a day, before breakfast and at bedtime, for a duration of 2 years.23 42 43
For the COT group, participants will not receive any OT and will only be provided with topical corticosteroids to treat olfactory dysfunction.
Moreover, an ‘Olfactory Training Diary’ was used after the training to assess the participant’s overall olfactory ability and evaluate whether OT was performed regularly. In addition, participants will be contacted regularly via text messages or phone calls to improve adherence. If the participant is diagnosed with mild cognitive impairment in a certain examination, the participant will be treated with relevant treatment and the participant will be asked to withdraw from the experiment.
Outcomes
All participants underwent olfactory testing, neuropsychological testing, blood tests and neuroimaging at baseline (Day 0) and at months 3, 6, 12 and 24 (online supplemental table 1). A trained research assistant, blinded to group assignment and serving as an independent rater, conducted olfactory and neuropsychological assessments.
Olfactory examination
Olfactory function is assessed using the Sniffin’ Sticks test, an internationally established subjective method. It comprises three components: odour threshold (T), odour discrimination (D) and odour identification (I). Odour threshold (T): Tested using n-butanol or phenylethyl alcohol as the odourant. 16 triplets of pens are presented in ascending order of concentration. Each triplet contains two blanks and one odourant pen. Scoring ranges from 16 (detection at the lowest concentration) to 1 (no detection at the highest concentration); Odour discrimination (D): 16 triplets of pens are used. Within each triplet, the subject must identify the pen with a different odour from the other two (two odourless pens and one scented pen, or two pens with the same scent and one with a different scent). Scoring is from 16 (all correct) to 0 (none correct); Odour identification (I): 16 pens, each with a distinct odour, are presented. After smelling each pen, the subject selects the correct odour name from four verbal descriptors. One point is awarded for each correct identification.
The sum of the threshold (T), discrimination (D) and identification (I) scores yields the TDI score, with a maximum of 48 points. The TDI score assesses overall olfactory function. A score≤30.75 indicates olfactory dysfunction.44
Neuropsychological tests
The MoCA is a screening test for detecting MCI. Results≥26/30 are considered normal. The test is standardised for participants with≤12 education years. It was performed to exclude participants with MCI and to detect the influence of OT on global cognitive function.45 46 The Auditory Verbal Learning Test was used to measure memory, with higher scores reflecting better verbal learning and recall abilities.47 Attention and working memory were measured by the Digit Span Test.48 Executive function was tested through the Trail Making Test, with shorter completion times representing better cognitive flexibility and set-shifting ability.49 Visuospatial function was examined by the Clock Drawing Test, where higher accuracy scores denote better visuospatial organisation.50 Language function was assessed using the Boston Naming Test51 52 and Verbal Fluency Test.53 To evaluate functional independence among all participants, the Barthel ADL Index was utilised to assess activities of daily living (ADL) and the Lawton Instrumental Activities of Daily Living Scale was employed to evaluate instrumental ADL.54,56 Neuropsychiatric symptoms were quantified by the Neuropsychiatric Inventory,57 Hamilton Anxiety Scale58 and Hamilton Depression Scale59 (table 1). SCD questionnaire 21 was developed to detect SCD concerns at risk of MCI60 (online supplemental material 2).
Table 1. Cognitive domains and assessment scales.
| Cognitive domain | Assessment scale(s) |
|---|---|
| Overall cognitive screening | Montreal Cognitive Assessment |
| Memory | Auditory Verbal Learning Test |
| Attention and working memory | Digit Span Test |
| Executive function | Trail Making Test |
| Visuospatial function | Clock Drawing Test |
| Language function | Boston Naming Test Verbal Fluency Test |
| Activities of daily living | Activities of Daily Living Scale Instrumental Activities of Daily Living |
| Neuropsychiatric symptoms | Neuropsychiatric Inventory Hamilton Anxiety Scale Hamilton Depression Scale |
Neurology biomarker tests
Levels of University of Gothenburg (UGOT) p-tau217,61 62 Cerebrospinal Fluid (CSF) Aβ42,63 p-tau181,64 65 total tau66 and the ratio of amyloid β42 to total tau67 will be quantitatively assessed. These biomarkers are crucial for monitoring the biochemical progression of cognitive impairment and are indicative of Alzheimer’s disease pathology.
Neuroimaging examination
Structural MRI will focus on the temporal lobes and other relevant brain regions to assess neurodegeneration. Parameters such as hippocampal volume, cortical thickness and white matter hyperintensity volumes will be quantified.68 In addition, amyloid PET scans will measure amyloid burden and regional tau deposition. The standardised uptake value ratio (SUVr) will be used as a primary quantitative metric to evaluate the presence and extent of amyloid plaques and tau tangles in the brain.69,71
Sample size
This study is a randomised controlled trial with three groups: control, COT and MOT groups. The sample size is calculated based on the efficacy rates of OT, using the formula:
Where α=0.05 (significance level) and power (1−β)=0.9. Assuming efficacy rates of 17% (p=0.004) for the control group, 37% (p=0.008) for the COT group,72 and 60% for the MOT group from preliminary data, the total sample size needed is 90, adjusting for a 20% dropout rate to total 114 participants, with 38 in each group.
Blinding
This research employs an open-label approach. In this study, neither participants nor physicians could remain unaware of the study conditions. However, the responsible physicians conducting telephone follow-ups at various intervals post-procedure will not be informed of the participants’ allocation status. Data entry will be performed by personnel not involved in the research team, while statisticians, who will also remain blinded to the allocation details, will complete the data analysis.
Data collection and management
Data for this study will be collected at multiple time points: baseline, mid-intervention and post-intervention. Olfactory function will be assessed using standardised olfactory tests,73 while cognitive function will be evaluated through a series of neuropsychological assessments. Additionally, biological samples for biomarker analysis (plasma and CSF) will be collected, and neuroimaging scans (MRI and PET) will be conducted.74 All assessments will be performed by trained personnel to ensure consistency and reliability across all data points.
On collection, all data will be coded and entered into a secure database management system. Each participant will be assigned a unique identifier to maintain confidentiality. The database will be password protected and accessible only to authorised personnel involved in the study.
To ensure the accuracy and integrity of the data, quality control measures will be implemented. These include double data entry, where data entered into the database will be checked by a second individual for any discrepancies. Any inconsistencies found will be resolved by referring back to the original data source. Regular audits of the database will also be conducted to ensure that all data are entered correctly and securely stored.
All physical data forms, such as consent forms and paper-based tests, will be stored in locked filing cabinets in secure locations. Electronic data, including digital forms, images and results from automated tests, will be backed up regularly on encrypted hard drives. Long-term storage of biological samples will be in biosecure, temperature-controlled facilities to ensure their preservation and integrity for future analyses.75
Data analysis will be conducted using SPSS (V.25.0, IBM) and R. This software will facilitate the comprehensive analysis of complex datasets, allowing for the application of advanced statistical models as outlined in the study protocol.
The study will adhere to ethical guidelines as approved by the Institutional Review Board. Participants will provide informed consent before participation, and all data will be de-identified to protect participant privacy. On completion of the study, de-identified data may be shared with the broader scientific community through a data-sharing platform, under conditions that comply with data protection regulations.
A data monitoring committee (DMC) will be established to oversee the conduct of the trial and ensure the safety and rights of participants are protected. The DMC will review data periodically and can recommend modifications to the trial or halt the trial if safety concerns arise.
Statistical methods
The statistical analyses for this study on the effects of olfactory training on cognitive function will be performed using SPSS software (V.25.0, IBM). Statistical significance for all tests will be determined at a p value threshold of<0.05 (two-sided). The analysis plan is structured to comprehensively assess the impact of olfactory training across various cognitive and biological metrics. Shedding participants were included in the analysis using intention to treat analysis.
All collected data will be tested for normality. Continuous variables that follow a normal distribution will be expressed as mean±SD, whereas non-normally distributed variables will be presented using medians and IQRs. Categorical variables will be summarised by frequency and percentage. Baseline characteristics between the experimental and control groups will be compared using one-way analysis of variance (ANOVA) to ensure homogeneity at the start of the intervention.
The primary analysis will involve repeated measures ANOVA, with ‘time’ as a within-subject factor and ‘treatment type’ (OT vs control) as a between-subject factor. This will assess the main and interaction effects of the intervention over time. For scales assessing memory, attention, executive function, visuospatial skills, language and mood, separate analyses will be conducted to ascertain the specific contributions of OT to these domains. Post-hoc analysis with Bonferroni corrections will be applied for pairwise comparisons of pre-intervention and post-intervention scores within each group to control for type I errors.
Partial correlation analysis will be used to explore the relationships between changes in olfactory function and cognitive scores across different domains, such as memory, attention, executive function, visuospatial skills, language and mood.76
Logistic regression will be conducted to identify risk factors for MCI, using variables such as gender, age, disease duration, aetiology and baseline scores on specific cognitive and olfactory tests. This analysis will be performed in both univariate and multivariate frameworks to adjust for potential confounders.2
Plasma and CSF biomarkers will be quantitatively analysed using high-sensitivity immunoassay platforms, such as the single-molecule array technology. Changes in biomarker levels from baseline to post-intervention will be assessed using paired t-tests or Wilcoxon signed-rank tests, depending on their distribution.77
MRI and PET scan data will be processed to measure structural and functional changes in the brain. Techniques such as volumetric analysis for MRI and SUVr calculations for PET scans will be employed. Mixed-effects models will be used to analyse these changes, accounting for intra-individual variability over time.69
Longitudinal statistical models will integrate both biomarker levels and neuroimaging metrics to evaluate their temporal changes and associations with cognitive outcomes. Covariates such as age, sex and Apolipoprotein E (APOE) ε4 genotype will be included to address.78
The Amyloid, Tau and Neurodegeneration framework will be applied to classify and interpret the comprehensive biomarker profiles, aiding in understanding the disease progression and its linkage to cognitive decline.79
Correlation analyses will be conducted to assess the relationships between changes in biomarkers, neuroimaging metrics and cognitive test scores. Further, multivariable regression analysis will determine the predictive value of these biomarkers and imaging findings on the cognitive outcomes of interest.
Ethics and dissemination
Ethical approval has been obtained from the Peking University Third Hospital Medical Science Research Ethics Committee (2023-347-01). There are no interim analyses or extra auditing planned for this study beyond the continuous monitoring by the investigators. However, all protocol amendments will be submitted to the ethical committees. Any modifications will be updated at ClinicalTrials.gov by the principal investigator. The study’s results will be published in scientific peer-reviewed journals with open access.
This study will be monitored by the DMC, which is established by the foundation sponsor. The DMC is independent of the research group and consists of one to two statistical experts and one to two clinical experts.23 The DMC does not usually have executive power; rather, it communicates the outcome of its deliberations to the sponsor. The DMC is responsible for verifying the protocol modification, project process, serious adverse events, informed consent forms, etc, and checking the traceability of all data. The DMC will conduct monitoring one to two times during the period of recruitment and provide monitoring reports.
The original data from this study will be maintained in a traceable, real-time, easily interpretable and accurate format. All research data generated during the trial will be securely stored and appropriately managed by the principal investigators. In cases where data corrections are required, the reason for the correction, the individual making the amendment and the date of modification will be documented, ensuring that all changes are traceable. Electronic data will be safeguarded with password-protected local server backups, and any modifications will require prior authorisation. Quality control and quality assurance mechanisms will be implemented in strict compliance with the study protocol and applicable legal and regulatory standards. Clinical research team members will undergo comprehensive training on the trial protocol at the study’s onset. Investigators are expected to adhere to the Standard Operating Procedures and trial protocol, performing their tasks meticulously and documenting their work promptly, thoroughly and in a standardised format. Confidentiality of all personal information and assessment data from both potential and enrolled participants will be strictly upheld. Participant names and addresses will not be included in the database, and no files will store information containing identifiable participant details. The research team will ensure that participant identities remain confidential. Blood samples will be obtained from all participants at the designated clinical centre, with plasma and serum analysed the same day or stored at −80°C for future analysis in line with the study objectives. A validated methodology will be employed for sample analysis, and the detailed procedures and outcomes will be documented in the method validation and sample analysis reports.
Discussion
This study uses a MOT device based on bidirectional airflow and positive pressure, boasting its own intellectual property rights.32 This research targets patients with high-risk MCI and compares MOT with COT and a control group to more readily discern the benefits of the modified method. However, the study has a few limitations. The control group only undergoes pharmacological treatment for olfactory disorders without any OT, which may potentially affect the study results due to the emotional state of the patients.80 Moreover, the study does not include healthy older adults, making it impossible to compare the MCI high-risk group’s biomarker concentrations with a normal baseline derived from the natural ageing of the olfactory system and cognitive function decline in healthy older adults.81 Additionally, in the olfactory function tests, we employed the Sniffin’ Sticks method, the identification part of which uses 16 scents including 4 that are also used in OT. Although these training scents do not affect the detection of the threshold (T) and discrimination (D) scores, and testers were not informed about the correctness of their scent identification when assessing the identification (I) scores to minimise bias, the observed improvement could still be partially attributed to a learning effect rather than a true training effect. Furthermore, although the sample size is statistically sound, the participants are only recruited from two hospitals in Beijing, limiting the generalisability of the study’s conclusions.82 A larger group could enhance the applicability of the study’s findings to a broader population.
Despite MCI being an early manifestation of AD and potentially causing a significant societal burden,83 84 there is still no simple yet effective treatment.85 This study explores a relatively simple, low-cost treatment method and introduces a new key window for the treatment of MCI and dementia by determining a high-risk group for MCI using olfactory disorders, SCD and age. The baseline cognitive functions of participants are thoroughly examined using cognitive-related biomarker detection, neuroimaging examinations and cognitive function test questionnaires. The data collected during the trial will not only help determine the intervention effects of olfactory training on cognitive and olfactory functions but also provide data support for future research on the potential mechanisms of olfactory training interventions on cognitive and olfactory functions.
Supplementary material
Footnotes
Funding: The study is financially funded by the Key clinical projects of Peking University Third Hospital (BYSYZD2023029), Natural Science Foundation of China (82000954), Beijing Science and Technology Nova Program (Z201100006820086), Beijing Hospitals Authority Youth Program (QML20190617) and Beijing Hospitals Authority Clinical Medicine Development of Special Funding (XMLX202136).
Prepub: Prepublication history and additional supplemental material for this paper are available online. To view these files, please visit the journal online (https://doi.org/10.1136/bmjopen-2025-100880).
Provenance and peer review: Not commissioned; externally peer reviewed.
Patient consent for publication: Not applicable.
Patient and public involvement: Patients and/or the public were not involved in the design, or conduct, or reporting, or dissemination plans of this research.
References
- 1.Jia L, Du Y, Chu L, et al. Prevalence, risk factors, and management of dementia and mild cognitive impairment in adults aged 60 years or older in China: a cross-sectional study. Lancet Public Health. 2020;5:e661–71. doi: 10.1016/S2468-2667(20)30185-7. [DOI] [PubMed] [Google Scholar]
- 2.Roberts R, Knopman DS. Classification and epidemiology of MCI. Clin Geriatr Med. 2013;29:753–72. doi: 10.1016/j.cger.2013.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.De Vito AN, Kunicki ZJ, Joyce HE, et al. Parallel changes in cognition, neuropsychiatric symptoms, and amyloid in cognitively unimpaired older adults and those with mild cognitive impairment. Alzheimers Dement. 2025;21:e14568. doi: 10.1002/alz.14568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Knopman DS, Amieva H, Petersen RC, et al. Alzheimer disease. Nat Rev Dis Primers. 2021;7:33. doi: 10.1038/s41572-021-00269-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Hanseeuw BJ, Betensky RA, Jacobs HIL, et al. Association of Amyloid and Tau With Cognition in Preclinical Alzheimer Disease: A Longitudinal Study. JAMA Neurol. 2019;76:915–24. doi: 10.1001/jamaneurol.2019.1424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Ströhle A, Schmidt DK, Schultz F, et al. Drug and Exercise Treatment of Alzheimer Disease and Mild Cognitive Impairment: A Systematic Review and Meta-Analysis of Effects on Cognition in Randomized Controlled Trials. Am J Geriatr Psychiatry. 2015;23:1234–49. doi: 10.1016/j.jagp.2015.07.007. [DOI] [PubMed] [Google Scholar]
- 7.Fatuzzo I, Niccolini GF, Zoccali F, et al. Neurons, Nose, and Neurodegenerative Diseases: Olfactory Function and Cognitive Impairment. Int J Mol Sci. 2023;24:2117. doi: 10.3390/ijms24032117. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Ubeda-Bañon I, Saiz-Sanchez D, Flores-Cuadrado A, et al. The human olfactory system in two proteinopathies: Alzheimer’s and Parkinson’s diseases. Transl Neurodegener. 2020;9:22. doi: 10.1186/s40035-020-00200-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Carnemolla SE, Kumfor F, Liang CT, et al. Olfactory Bulb Integrity in Frontotemporal Dementia and Alzheimer’s Disease. J Alzheimers Dis. 2022;89:51–66. doi: 10.3233/JAD-220080. [DOI] [PubMed] [Google Scholar]
- 10.Dan X, Wechter N, Gray S, et al. Olfactory dysfunction in aging and neurodegenerative diseases. Ageing Res Rev. 2021;70:101416. doi: 10.1016/j.arr.2021.101416. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Kim M, Lee JY, Kim YH, et al. Normative Parameters of Olfactory Bulbs Based on Magnetic Resonance Imaging and Olfactory Function. J Korean Med Sci. 2024;39:e321. doi: 10.3346/jkms.2024.39.e321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Nasserzare S, Lehrner J. Olfaction and thyroid hormones in patients with subjective cognitive decline, non-amnestic and amnestic mild cognitive impairment. Wien Klin Wochenschr. 2025;137:172–81. doi: 10.1007/s00508-024-02431-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Liang X, Ding D, Zhao Q, et al. Association between olfactory identification and cognitive function in community-dwelling elderly: the Shanghai aging study. BMC Neurol. 2016;16:199. doi: 10.1186/s12883-016-0725-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Georgiopoulos C, Witt ST, Haller S, et al. A study of neural activity and functional connectivity within the olfactory brain network in Parkinson’s disease. Neuroimage Clin. 2019;23:101946. doi: 10.1016/j.nicl.2019.101946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bohnen NI, Müller MLTM. In vivo neurochemical imaging of olfactory dysfunction in Parkinson’s disease. J Neural Transm (Vienna) 2013;120:571–6. doi: 10.1007/s00702-012-0956-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Manan HA, Yahya N, Han P, et al. A systematic review of olfactory-related brain structural changes in patients with congenital or acquired anosmia. Brain Struct Funct. 2022;227:177–202. doi: 10.1007/s00429-021-02397-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pike KE, Cavuoto MG, Li L, et al. Subjective Cognitive Decline: Level of Risk for Future Dementia and Mild Cognitive Impairment, a Meta-Analysis of Longitudinal Studies. Neuropsychol Rev. 2022;32:703–35. doi: 10.1007/s11065-021-09522-3. [DOI] [PubMed] [Google Scholar]
- 18.Wang X-T, Wang Z-T, Hu H-Y, et al. Association of Subjective Cognitive Decline with Risk of Cognitive Impairment and Dementia: A Systematic Review and Meta-Analysis of Prospective Longitudinal Studies. J Prev Alzheimers Dis. 2021;8:277–85. doi: 10.14283/jpad.2021.27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Parker AF, Ohlhauser L, Scarapicchia V, et al. A Systematic Review of Neuroimaging Studies Comparing Individuals with Subjective Cognitive Decline to Healthy Controls. J Alzheimers Dis. 2022;86:1545–67. doi: 10.3233/JAD-215249. [DOI] [PubMed] [Google Scholar]
- 20.Roalf DR, Moberg MJ, Turetsky BI, et al. A quantitative meta-analysis of olfactory dysfunction in mild cognitive impairment. J Neurol Neurosurg Psychiatry. 2017;88:226–32. doi: 10.1136/jnnp-2016-314638. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ekström I, Josefsson M, Larsson M, et al. Subjective Olfactory Loss in Older Adults Concurs with Long-Term Odor Identification Decline. Chem Senses. 2019;44:105–12. doi: 10.1093/chemse/bjy079. [DOI] [PubMed] [Google Scholar]
- 22.Fukumoto T, Ezaki T, Urakami K. Verification of the association between cognitive decline and olfactory dysfunction using a DEmentia screening kit in subjects with Alzheimer’s dementia, mild cognitive impairment, and normal cognitive function (DESK study): A multicenter, open-label, interventional study. eNeurologicalSci. 2022;29:100439. doi: 10.1016/j.ensci.2022.100439. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pieniak M, Oleszkiewicz A, Avaro V, et al. Olfactory training – Thirteen years of research reviewed. Neuroscience & Biobehavioral Reviews. 2022;141:104853. doi: 10.1016/j.neubiorev.2022.104853. [DOI] [PubMed] [Google Scholar]
- 24.Oleszkiewicz A, Abriat A, Doelz G, et al. Beyond olfaction: Beneficial effects of olfactory training extend to aging-related cognitive decline. Behav Neurosci. 2021;135:732–40. doi: 10.1037/bne0000478. [DOI] [PubMed] [Google Scholar]
- 25.Vance DE, Del Bene VA, Kamath V, et al. Does Olfactory Training Improve Brain Function and Cognition? A Systematic Review. Neuropsychol Rev. 2024;34:155–91. doi: 10.1007/s11065-022-09573-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Han P, Musch M, Abolmaali N, et al. Improved Odor Identification Ability and Increased Regional Gray Matter Volume After Olfactory Training in Patients With Idiopathic Olfactory Loss. Iperception. 2021;12 doi: 10.1177/20416695211005811. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Birte-Antina W, Ilona C, Antje H, et al. Olfactory training with older people. Int J Geriatr Psychiatry. 2018;33:212–20. doi: 10.1002/gps.4725. [DOI] [PubMed] [Google Scholar]
- 28.Slabik D, Garaschuk O. Olfactory dysfunction as a common biomarker for neurodegenerative and neuropsychiatric disorders. Neural Regen Res. 2023;18:1029–30. doi: 10.4103/1673-5374.355756. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Xie B, Yang S, Hao Y, et al. Impaired olfactory identification in dementia-free individuals is associated with the functional abnormality of the precuneus. Neurobiol Dis. 2024;194:106483. doi: 10.1016/j.nbd.2024.106483. [DOI] [PubMed] [Google Scholar]
- 30.Djupesland PG. Nasal drug delivery devices: characteristics and performance in a clinical perspective-a review. Drug Deliv Transl Res. 2013;3:42–62. doi: 10.1007/s13346-012-0108-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Xi J, Wang Z, Nevorski D, et al. Nasal and Olfactory Deposition with Normal and Bidirectional Intranasal Delivery Techniques: In Vitro Tests and Numerical Simulations. J Aerosol Med Pulm Drug Deliv. 2017;30:118–31. doi: 10.1089/jamp.2016.1295. [DOI] [PubMed] [Google Scholar]
- 32.Liu Y, Wu D. Bi-directional nasal drug delivery systems: A scoping review of nasal particle deposition patterns and clinical application. Laryngoscope Investig Otolaryngol. 2023;8:1484–99. doi: 10.1002/lio2.1190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Chen B, Espin M, Haussmann R, et al. The Effect of Olfactory Training on Olfaction, Cognition, and Brain Function in Patients with Mild Cognitive Impairment. J Alzheimers Dis. 2022;85:745–54. doi: 10.3233/JAD-215257. [DOI] [PubMed] [Google Scholar]
- 34.Haehner A, Chen B, Espin M, et al. Training with Odors Impacts Hippocampal Thickness in Patients with Mild Cognitive Impairment. J Alzheimers Dis. 2022;88:743–55. doi: 10.3233/JAD-220248. [DOI] [PubMed] [Google Scholar]
- 35.Wang Z, Xu L, Wang Y. Validation of the Ten‐word Test for Immediate Memory in Middle‐Aged and Older Population with Subjective Cognitive Decline and Mild Cognitive Impairment. Alzheimer’s &Amp; Dementia. 2023;19:e079481. doi: 10.1002/alz.079481. [DOI] [Google Scholar]
- 36.Chan KY, Lee S, Ju CH, et al. 34 Association Between Subjective Cognitive Decline and Mental Wellbeing in Normal Cognition and MCI Older Adults. J Int Neuropsychol Soc. 2023;29:344–5. doi: 10.1017/S1355617723004708. [DOI] [Google Scholar]
- 37.Renner B, Schreiber K. Olfactory and trigeminal interaction of menthol and nicotine in humans. Exp Brain Res. 2012;219:13–26. doi: 10.1007/s00221-012-3063-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Frasnelli J, Schuster B, Hummel T. Interactions between olfaction and the trigeminal system: what can be learned from olfactory loss. Cereb Cortex. 2007;17:2268–75. doi: 10.1093/cercor/bhl135. [DOI] [PubMed] [Google Scholar]
- 39.Kronenbuerger M, Pilgramm M. StatPearls. Treasure Island (FL): StatPearls Publishing; 2025. Olfactory training. [Google Scholar]
- 40.Hellings PW, Rombaux P. Medical therapy and smell dysfunction. B-ENT. 2009;5 Suppl 13:71–5. [PubMed] [Google Scholar]
- 41.Ozaki F, Ueno T, Kondo S, et al. Effects of Corticosteroid Treatment on Olfactory Dysfunction in LATY136F Knock-In Mice. Cureus. 2024;16:e65791. doi: 10.7759/cureus.65791. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Mahmut MK, Musch M, Han P, et al. The effect of olfactory training on olfactory bulb volumes in patients with idiopathic olfactory loss. Rhinology. 2020;58:410–2. doi: 10.4193/Rhin20.223. [DOI] [PubMed] [Google Scholar]
- 43.Besser G, Oswald MM, Liu DT, et al. Flavor education and training in olfactory dysfunction: a pilot study. Eur Arch Otorhinolaryngol. 2020;277:1987–94. doi: 10.1007/s00405-020-05950-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hummel T, Sekinger B, Wolf SR, et al. “Sniffin” sticks’: olfactory performance assessed by the combined testing of odor identification, odor discrimination and olfactory threshold. Chem Senses. 1997;22:39–52. doi: 10.1093/chemse/22.1.39. [DOI] [PubMed] [Google Scholar]
- 45.Nasreddine ZS, Phillips NA, Bédirian V, et al. The Montreal Cognitive Assessment, MoCA: a brief screening tool for mild cognitive impairment. J Am Geriatr Soc. 2005;53:695–9. doi: 10.1111/j.1532-5415.2005.53221.x. [DOI] [PubMed] [Google Scholar]
- 46.Chen K-L, Xu Y, Chu A-Q, et al. Validation of the Chinese Version of Montreal Cognitive Assessment Basic for Screening Mild Cognitive Impairment. J Am Geriatr Soc. 2016;64:e285–90. doi: 10.1111/jgs.14530. [DOI] [PubMed] [Google Scholar]
- 47.Brugnolo A, Morbelli S, Arnaldi D, et al. Metabolic correlates of Rey auditory verbal learning test in elderly subjects with memory complaints. J Alzheimers Dis. 2014;39:103–13. doi: 10.3233/JAD-121684. [DOI] [PubMed] [Google Scholar]
- 48.BLACKBURN HL, BENTON AL. Revised administration and scoring of the digit span test. J Consult Psychol. 1957;21:139–43. doi: 10.1037/h0047235. [DOI] [PubMed] [Google Scholar]
- 49.Tombaugh TN. Trail Making Test A and B: normative data stratified by age and education. Arch Clin Neuropsychol. 2004;19:203–14. doi: 10.1016/S0887-6177(03)00039-8. [DOI] [PubMed] [Google Scholar]
- 50.Hazan E, Frankenburg F, Brenkel M, et al. The test of time: a history of clock drawing. Int J Geriatr Psychiatry. 2018;33:e22–30. doi: 10.1002/gps.4731. [DOI] [PubMed] [Google Scholar]
- 51.Roth C. Encyclopedia of Clinical Neuropsychology. New York, NY: Springer; 2011. Boston naming test; pp. 430–3. [Google Scholar]
- 52.Kharkhurin AV. A Preliminary Version of an Internet-Based Picture Naming Test. OJML. 2012;02:34–41. doi: 10.4236/ojml.2012.21005. [DOI] [Google Scholar]
- 53.Levin HS. A Compendium of Neuropsychological Tests: Administration, Norms, and Commentary. Arch Neurol. 1993;50:451. doi: 10.1001/archneur.1993.00540050007005. [DOI] [Google Scholar]
- 54.KATZ S, FORD AB, MOSKOWITZ RW, et al. STUDIES OF ILLNESS IN THE AGED. THE INDEX OF ADL: A STANDARDIZED MEASURE OF BIOLOGICAL AND PSYCHOSOCIAL FUNCTION. JAMA. 1963;185:914–9. doi: 10.1001/jama.1963.03060120024016. [DOI] [PubMed] [Google Scholar]
- 55.Graf C. The Lawton Instrumental Activities of Daily Living Scale. AJN, American Journal of Nursing. 2008;108:52–62. doi: 10.1097/01.NAJ.0000314810.46029.74. [DOI] [PubMed] [Google Scholar]
- 56.Collin C, Wade DT, Davies S, et al. The Barthel ADL Index: a reliability study. Int Disabil Stud. 1988;10:61–3. doi: 10.3109/09638288809164103. [DOI] [PubMed] [Google Scholar]
- 57.Cummings JL, Mega M, Gray K, et al. The Neuropsychiatric Inventory: comprehensive assessment of psychopathology in dementia. Neurology (ECronicon) 1994;44:2308–14. doi: 10.1212/wnl.44.12.2308. [DOI] [PubMed] [Google Scholar]
- 58.Maier W, Buller R, Philipp M, et al. The Hamilton Anxiety Scale: reliability, validity and sensitivity to change in anxiety and depressive disorders. J Affect Disord. 1988;14:61–8. doi: 10.1016/0165-0327(88)90072-9. [DOI] [PubMed] [Google Scholar]
- 59.Hamilton M. A rating scale for depression. J Neurol Neurosurg Psychiatry. 1960;23:56–62. doi: 10.1136/jnnp.23.1.56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Hao L, Jia J, Xing Y, et al. The reliability and validity test of subjective cognitive decline questionnaire 21 with population in a Chinese community. Brain Behav. 2022;12:e2709. doi: 10.1002/brb3.2709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Janelidze S, Berron D, Smith R, et al. Associations of Plasma Phospho-Tau217 Levels With Tau Positron Emission Tomography in Early Alzheimer Disease. JAMA Neurol. 2021;78:149–56. doi: 10.1001/jamaneurol.2020.4201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Gonzalez-Ortiz F, Ferreira PCL, González-Escalante A, et al. A novel ultrasensitive assay for plasma p-tau217: Performance in individuals with subjective cognitive decline and early Alzheimer’s disease. Alzheimers Dement. 2024;20:1239–49. doi: 10.1002/alz.13525. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Bruno D, Gleason CE, Koscik RL, et al. The recency ratio is related to CSF amyloid beta 1-42 levels in MCI-AD. Int J Geriatr Psychiatry. 2019;34:415–9. doi: 10.1002/gps.5029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Karikari TK, Pascoal TA, Ashton NJ, et al. Blood phosphorylated tau 181 as a biomarker for Alzheimer’s disease: a diagnostic performance and prediction modelling study using data from four prospective cohorts. Lancet Neurol. 2020;19:422–33. doi: 10.1016/S1474-4422(20)30071-5. [DOI] [PubMed] [Google Scholar]
- 65.Kwon HS, Kim JY, Koh S-H, et al. Predicting cognitive stage transition using p-tau181, Centiloid, and other measures. Alzheimers Dement. 2023;19:4641–50. doi: 10.1002/alz.13054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66.Sperling R, Johnson K. Biomarkers of Alzheimer disease: current and future applications to diagnostic criteria. Continuum (Minneap Minn) 2013;19:325–38. doi: 10.1212/01.CON.0000429181.60095.99. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Pérez-Grijalba V, Romero J, Pesini P, et al. Plasma Aβ42/40 Ratio Detects Early Stages of Alzheimer’s Disease and Correlates with CSF and Neuroimaging Biomarkers in the AB255 Study. J Prev Alzheimers Dis. 2019;6:34–41. doi: 10.14283/jpad.2018.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Sundar U, Mukhopadhyay A, Raghavan S, et al. Evaluation of “Normal” Cognitive Functions and Correlation With MRI Volumetry: Towards a Definition of Vascular Cognitive Impairment. Cureus. 2023;15:e49461. doi: 10.7759/cureus.49461. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 69.Zhao Q, Liu M, Ha L, et al. Quantitative 18F-AV1451 Brain Tau PET Imaging in Cognitively Normal Older Adults, Mild Cognitive Impairment, and Alzheimer’s Disease Patients. Front Neurol. 2019;10:486. doi: 10.3389/fneur.2019.00486. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Ewers M, Insel P, Jagust WJ, et al. CSF biomarker and PIB-PET-derived beta-amyloid signature predicts metabolic, gray matter, and cognitive changes in nondemented subjects. Cereb Cortex. 2012;22:1993–2004. doi: 10.1093/cercor/bhr271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71.Kung M-P, Weng C-C, Lin K-J, et al. Amyloid plaque imaging from IMPY/SPECT to AV-45/PET. Chang Gung Med J. 2012;35:211–8. doi: 10.4103/2319-4170.106151. [DOI] [PubMed] [Google Scholar]
- 72.Pekala K, Chandra RK, Turner JH. Efficacy of olfactory training in patients with olfactory loss: a systematic review and meta-analysis. Int Forum Allergy Rhinol. 2016;6:299–307. doi: 10.1002/alr.21669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Yang X, Si J, Lin S, et al. Olfactory Function, Genetic Predisposition, and Cognitive Performance in Chinese Adults. Curr Alzheimer Res. 2021;18:1093–103. doi: 10.2174/1567205019666211222151851. [DOI] [PubMed] [Google Scholar]
- 74.Heredia L, Mateo D, Carrión N, et al. Dataset on neuropsychological profile and microbiota composition in cognitively unimpaired elderly and Alzheimer’s patients. Data Brief. 2025;61:111778. doi: 10.1016/j.dib.2025.111778. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Cunha-Oliveira T, Ioannidis JPA, Oliveira PJ. Best practices for data management and sharing in experimental biomedical research. Physiol Rev. 2024;104:1387–408. doi: 10.1152/physrev.00043.2023. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Pinto JM, Schumm LP, Wroblewski KE, et al. Racial disparities in olfactory loss among older adults in the United States. J Gerontol A Biol Sci Med Sci. 2014;69:323–9. doi: 10.1093/gerona/glt063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Chimthanawala NMA, Haria A, Sathaye S. Non-invasive Biomarkers for Early Detection of Alzheimer’s Disease: a New-Age Perspective. Mol Neurobiol. 2024;61:212–23. doi: 10.1007/s12035-023-03578-3. [DOI] [PubMed] [Google Scholar]
- 78.Mandecka M, Budziszewska M, Barczak A, et al. Association between Cerebrospinal Fluid Biomarkers for Alzheimer’s Disease, APOE Genotypes and Auditory Verbal Learning Task in Subjective Cognitive Decline, Mild Cognitive Impairment, and Alzheimer’s Disease. J Alzheimers Dis. 2016;54:157–68. doi: 10.3233/JAD-160176. [DOI] [PubMed] [Google Scholar]
- 79.Rezaeyan A, Asadi S, Kamrava SK, et al. Reorganizing brain structure through olfactory training in post-traumatic smell impairment: An MRI study. J Neuroradiol. 2022;49:333–42. doi: 10.1016/j.neurad.2021.04.035. [DOI] [PubMed] [Google Scholar]
- 80.Dal Bò E, Gentili C, Castellani A, et al. Olfactory meta-cognition in individuals with depressive and anxiety symptoms: The differential role of common and social odors. J Affect Disord. 2022;308:259–67. doi: 10.1016/j.jad.2022.04.071. [DOI] [PubMed] [Google Scholar]
- 81.Bothwell AR, Resnick SM, Ferrucci L, et al. Associations of olfactory function with brain structural and functional outcomes. A systematic review. Ageing Res Rev. 2023;92:102095. doi: 10.1016/j.arr.2023.102095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Serdar CC, Cihan M, Yücel D, et al. Sample size, power and effect size revisited: simplified and practical approaches in pre-clinical, clinical and laboratory studies. Biochem med (Online) 2021;31:27–53. doi: 10.11613/BM.2021.010502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 83.Ritchie C, Trepel D, Edwards S, et al. The impact of mild cognitive impairment on healthcare utilization and costs: A UK Biobank study. Alz & Dem Diag Ass & Dis Mo. 2025;17:e70065. doi: 10.1002/dad2.70065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Takechi H, Yoshino H. Health Care Resource Use and Social Costs in Mild Cognitive Impairment and Mild Alzheimer’s Disease. J Alzheimers Dis Rep. 2023;7:731–8. doi: 10.3233/ADR-230032. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Tahami Monfared AA, Tafazzoli A, Ye W, et al. Long-Term Health Outcomes of Lecanemab in Patients with Early Alzheimer’s Disease Using Simulation Modeling. Neurol Ther. 2022;11:863–80. doi: 10.1007/s40120-022-00350-y. [DOI] [PMC free article] [PubMed] [Google Scholar]