Abstract
Background:
Presentation affects several surgical parameters and post-operative outcomes. This study compares emergency versus elective colon cancer outcomes and prognostic factors associated with postoperative complications.
Methods:
All colon cancer patients who underwent curative resection between July 2015 and December 2019 were retrospectively reviewed and analyzed. Patients were divided into emergency and elective groups based on their presentation.
Results:
A total of 215 patients were included; 70 patients (32.6%) in the emergency group. Of them, 61 patients presented with obstruction, and nine with free perforation. The elective group had a higher laparoscopic approach (82.8%) with primary anastomosis (90.3%) compared to the emergency group (57.1% and 68.6%, respectively, P < 0.001). Postoperative complications were clinically higher in the emergency group (30% vs. 20.7%, P = 0.171). ICU/HDU admission was higher in the emergency group (40% vs. 17.2%, P < 0.001), and had a longer hospital stay (15 vs. 10 days, P < 0.003). Regression analysis revealed increased risk of complications among patients with comorbidities (OR 3.21; 95% CI, P = 0.002), albumin levels <30g/dl (OR 2.20; 95% CI, P = 0.01), complete obstruction (OR 2.42; 95% CI, P = 0.017), perforation (OR 9.98; 95% CI, P < 0.001), and open surgery (OR 4.84; 95% CI, P < 0.001).
Conclusion:
Emergency presentation rate was high among our patients with less-favorable surgical outcomes. Open surgery and stoma creation were more prevalent in the emergency group. Complete obstruction, perforation, open surgery, hypo-albuminemia, and presence of comorbidities were independent risk factors for complications.
Keywords: Colon cancer, elective surgery, emergency surgery, surgical outcomes
INTRODUCTION
Colorectal cancer is one of the most prevalent and preventable cancers worldwide.[1] In Saudi Arabia, it is the second most common of all cancers in males, and the third most common in females.[2] Colon cancer may present early with an asymptomatic mass on screening colonoscopy, or late with a more complicated presentation.[3] It is believed that the introduction of screening colonoscopy enables early detection of colon polyps and cancer. Additionally, patients who are detected by colonoscopy are typically in earlier cancer stage, which enhances their prognosis.[4,5] The American Cancer Society recommends initiating screening for colon cancer at the age of 50 in all individuals except for African Americans, for whom they recommend screening at the age of 45.[1] Due to the lack of a properly structured screening program in Saudi Arabia, it is necessary to report the outcomes of various colon cancer presentations as well as the predicting factors that can result in unfavorable outcomes.
Symptoms of colon cancer could include abdominal pain, a change in bowel habits, hematochezia or melena, and weakness. Patients’ symptoms may differ because they are affected by various variables, which include the location, size, and stage of the malignancy. Moreover, colon cancer can present with emergency presentation such as bleeding, obstruction, and perforation.[3] The most common emergency presentation reported in the literature is obstruction, followed by perforation. Although bleeding can present in up to half of the initial presentations, it usually is low in volume and requires no emergency intervention.[6,7,8] Emergency presentation has been associated with higher postoperative morbidity and mortality rates compared to elective surgeries.[9,10]
There are apparent differences between elective and emergency patients in the severity of the disease and the type of surgery. We reported the oncological outcomes of emergency versus elective colon cancer presentations in our center in a previous paper.[11] The aim of this study is to compare the characteristics of elective and emergency colon cancer presentations, operative parameters, as well as the surgical outcomes. We also assessed the factors associated with higher post operative complications.
PATIENTS AND METHODS
This is a retrospective study conducted at a tertiary hospital in Riyadh, Saudi Arabia. A chart review was conducted on the electronic medical records of all patients with colon cancer, between July 2015 and December 2019. They were screened for our inclusion criteria, as only patients who underwent curative intent resection were included. The decision of curative versus palliative management was based on the Tumor board multidisciplinary teams’ discussion, especially for patients with stage 4 disease. Pediatric age younger than 16 years and patients with rectal cancer (within 12 cm from anal verge) and peritoneal carcinomatosis were excluded. Colon cancer was defined as any colon cancer from the cecum down to 12 cm above the anal verge.
Patients were allocated into two groups based on the presentation: elective and emergency groups.
Elective group included any patients previously screened or identified through non-emergent presentations. Emergency groups were defined according to the patient presentation, which typically is discovered through symptomatic presentations (obstruction or perforation) to the emergency department requiring immediate investigations and surgical operation. All perforated tumor cases were reviewed comprehensively to assess for the ability if curative resection. Perforated cancer with peritoneal dissemination or inability to perform complete en-bloc resection were excluded from the analysis.
Demographic and clinical data, disease characteristics and surgical parameters were collected. Postoperative complications and clinical outcomes were documented and analyzed. Intra-abdominal collections accounted for any intra-abdominal abscess after surgery; Patients with ascites, free or contained anastomotic leaks, were not considered intra-abdominal collections. Clavien–Dindo classification was used to assess the severity of post-operative complications.[12] Many factors were tested for their predictability toward postoperative complications.
All elective procedures were performed by qualified colorectal consultants. Emergency cases were handled by the general surgery on-call or acute care surgery teams, with the assistance of 24-hour available colorectal consultants on-call if required.
The decision for curative or palliative intent treatment for stage 4 colon cancer was based on multiple factors, including the tumor board’s multidisciplinary decision on primary tumor and metastatic lesions, including resection with R0 margins.
Ethical approval was obtained from the Institutional Review Board (IRB) at King Saud University.
Power analysis
The statistical power was calculated to compare between the two groups. In each group the sample was given a minimum value of 0.80, the post hoc test of power (1-β err prob) has a result of 0.928% or 92.8%, which was above the minimum limit. Hence, the achieved power for this particular statistical test with a parameter of 2-tailed and medium effect size with alpha 0.05, the sample size of emergency cases equaling 70, and the sample size of elective cases equaling 145 is sufficient.
Statistical analysis
Descriptive statistics were calculated to present numbers and percentages for all categorical variables, while mean and standard deviation were used to present all continuous variables. Between comparisons, the Chi-square test, Fisher’s Exact test, the independent sample t-test, and Mann–Whitney U-test were applied. Binary regression analyses were performed to determine the prognostic factors that influence the variable of interest. Normality tests were performed using the Shapiro-Wilk test as well as the Kolmogorov–Smirnov test. A P value of 0.05 was considered statistically significant. The data were analyzed using Statistical Packages for Social Sciences (SPSS) version 26 (Armonk, NY: IBM Corp, USA).
RESULTS
We reviewed the surgical outcomes of 215 patients who underwent curative surgery for colon cancer. Patients were divided into two groups, emergency group with 70 patients (32.6%) and 145 patients (67.4%) were allocated in elective group. In the emergency group, 61 patients presented with colonic obstruction, while 9 patients presented with perforation.
Age was comparable in both groups, with a mean of 59.6 (SD ± 13.4) years old, with nearly 60% being males, as shown in Table 1. The mean American Society of Anesthesiologists (ASA) score was higher in emergency group 2.41 (SD ± 0.75) compared to 2.18 (SD ± 0.69) in elective group (P = 0.031). Comorbidities and use of blood thinner medications were comparable in both groups (P = 0.322, P = 0.277, respectively).
Table 1.
Demographics and Baseline characteristics
| Factor | Emergency N (%) (n=70) | Elective N (%) (n=145) | P ¦ |
|---|---|---|---|
| Age in years (Mean±SD) | 58.9±14.2 | 56.9±13.1 | 0.300 |
| Gender | |||
| • Male | 38 (54.3%) | 90 (62.1%) | 0.276 |
| • Female | 32 (45.7%) | 55 (37.9%) | |
| BMI (kg/m2) (Mean±SD) | 27.9±5.87 | 27.7±5.31 | 0.718 |
| ASA score (Mean±SD) | 2.41±0.75 | 2.18±0.69 | 0.031** |
| Smoking | 4 (5.7%) | 16 (11%) | 0.316£ |
| Family history of cancer | 21 (30%) | 23 (15.9%) | 0.016** |
| Comorbidities | 38 (54.3%) | 89 (61.4%) | 0.322 |
| Blood thinner medications | 3 (4.3%) | 13 (9%) | 0.277£ |
| Hemoglobin (Mean±SD) | 11.9±2.46 | 11.3±2.81 | 0.118 |
| Albumin (Mean±SD) | 30.9±5.41 | 32.5±5.61 | 0.052 |
| Tumor site* | |||
| • Rectal sigmoid junction | 11 (15.7%) | 26 (17.9%) | 0.847 |
| • Sigmoid colon | 33 (47.1%) | 95 (65.5%) | 0.074 |
| • Descending colon | 15 (21.4%) | 24 (16.6%) | 0.385 |
| • Transverse colon | 4 (5.7%) | 11 (7.6%) | 0.778 |
| • Ascending colon | 4 (5.7%) | 22 (15.2%) | 0.048**£ |
| • Cecum | 5 (7.1%) | 22 (15.2%) | 0.096 |
| TNM Pathological Stage | |||
| • Stage 1 | 3 (4.3%) | 31 (21.4%) | 0.001**£ |
| • Stage 2 | 35 (50%) | 41 (28.3%) | |
| • Stage 3 | 25 (35.7%) | 56 (38.6%) | |
| • Stage 4 | 7 (10%) | 17 (11.7%) |
*Some patients have multiple tumor sites. §P-value has been calculated using the Chi-square test. £P-value has been calculated using Fisher’s Exact test. **Significant at P<0.05 level. BMI=Body mass index, ASA=American Society of Anesthesiologists, TNM=Tumor, node, and metastasis
Surgical intervention and operative parameters are shown in Table 2. The elective group had better perioperative outcomes than the emergency group. Laparoscopic surgery was performed significantly more in an elective group with 120 patients (82.8%) compared to 40 patients (57.1%) in the emergency group (P ≤ 0.001). Diverting stoma was used as initial intervention in 11.4%, and end ileostomy/colostomy (21.4%) were significantly more prevalent in the emergency group (P = 0.001 and P = 0.002, respectively), while primary anastomosis (90.3%) was higher in the elective group (P = 0.001).
Table 2.
Surgical approach and operative parameters
| Factor | Emergency N (%) (n=70) | Elective N (%) (n=145) | P ¦ |
|---|---|---|---|
| Surgical approach | |||
| Laparoscopic | 40 (57.1%) | 120 (82.8%) | <0.001** |
| Open | 30 (42.9%) | 25 (17.2%) | |
| Conversion to open | 10 (25%) | 13 (10.8%) | 0.237 |
| Type of Surgery* | |||
| Diverting stoma (as initial intervention) | 8 (11.4%) | 1 (0.70%) | 0.001**£ |
| Primary anastomosis | 48 (68.6%) | 131 (90.3%) | <0.001** |
| End ileostomy/colostomy | 15 (21.4%) | 9 (6.2%) | 0.002** |
| Anastomosis with diverting stoma | 11 (15.7%) | 6 (4.1%) | 0.006** |
| Operative parameters | |||
| Lymph node retrieval (mean±SD) | 20.8±8.97 | 22.1±10.7 | 0.357‡ |
| • <12 nodes | 6 (8.6%) | 7 (4.8%) | 0.280 |
| Radial margin | 2 (2.9%) | 1 (0.70%) | 0.248¥ |
| Preoperative blood transfusion | 7 (10%) | 13 (9%) | 0.807 |
| Postoperative blood transfusion | 3 (4.3%) | 3 (2.1%) | 0.355¥ |
| Operative time (minutes), median (min-max) | 197.5 (88 – 530) | 181 (54 – 397) | 0.086£ |
*Some patients received multiple surgeries. ‡P-value has been calculated using an independent sample t-test. £P-value has been calculated using Mann–Whitney U-test. §P-value has been calculated using the Chi-square test. ¥P-value has been calculated using Fisher’s Exact test. **Significant at P<0.05 level
Postoperative complications and clinical outcomes are presented in Table 3. Postoperative complications were documented in 21 patients (30%) of the emergency group compared to 30 patients (20.7%) in the elective group (P = 0.171). Intra-abdominal collection (12 patients, 17.1%) was the most common complication in the emergency group, compared to only 5 patients (3.4%) in the elective group P < 0.001). However, severe complications with Clavien–Dindo ≥ Grade III were only in 7.1% and 9% in emergency and elective groups, respectively (P = 0.757). Figure 1 represents the severity of the overall complications according to the Clavien–Dindo classification. Mild complications (Grade I and II) were 36/43 (84%) complications in the elective group, compared to 24/37 (65%) in the emergency group. Some patients have presented with multiple concomitant complications. Intensive care unit (ICU) and high dependency unit (HDU) admissions were reported to be high among patients in the emergency group, with 28 patients (40%) compared to 25 patients (17.2%) in the elective group (P < 0.001). Moreover, total hospital stay was significantly prolonged in the emergency group with a median of 15 days compared to 10 days in the elective group (P = 0.003). However, both groups showed similar average post-operative stay. The elective group length of stay included pre-operative stay for staging workup.
Table 3.
Surgical complications according to presentation
| Factor | Emergency N (%) (n=70) | Elective N (%) (n=145) | P ¦ |
|---|---|---|---|
| Postoperative complication (per patient)* | 21 (30%) | 30 (20.7%) | 0.171 |
| • Intra-abdominal collection | 12 (17.1%) | 5 (3.4%) | <0.001** |
| • Wound infection | 11 (15.7%) | 17 (11.7%) | 0.415 |
| • Urinary tract infection | 0 | 3 (2.1%) | 0.553¥ |
| • Respiratory complications | 4 (5.7%) | 4 (2.8%) | 0.442¥ |
| • Ileus | 2 (2.9%) | 6 (4.1%) | 1.000¥ |
| • Bleeding | 2 (2.9%) | 5 (3.4%) | 1.000¥ |
| • Anastomotic leak | 1 (1.4%) | 3 (2.1%) | 1.000¥ |
| • Others | 5 (7.1%) | 0 | 0.326¥ |
| Clavien-Dindo classification | |||
| • Grade I | 5 (7.1%) | 6 (4.1%) | 0.752¥ |
| • Grade II | 7 (10%) | 14 (9.7%) | |
| • Grade III | 0 | 2 (1.4%) | |
| • Grade IIIA | 1 (1.4%) | 4 (2.8%) | |
| • Grade IIIB | 2 (2.9%) | 6 (4.1%) | |
| • Grade IV | 2 (2.9%) | 1 (0.70%) | |
| Clavien dindo≥Grade III (per patient)* | 5 (7.1%) | 13 (9%) | 0.757 |
| Clinical outcomes | |||
| ICU/HDU admission | 28 (40%) | 25 (17.2%) | <0.001** |
| 30-Day reoperation | 6 (8.6%) | 8 (5.5%) | 0.395 |
| 30-Day readmission | 5 (7.1%) | 14 (9.7%) | 0.543 |
| 30-Day mortality | 2 (2.9%) | 0 | 0.105¥ |
| Total hospital stay (days), median (min-max) | 15 (5 – 113) | 10 (4 – 222) | 0.003**£ |
| Post operative stay (days), median (min-max) | 6 (2 – 89) | 5 (2 – 161) | 0.033**£ |
*Some patients have multiple complications. Each patient had at least one complication. £P-value has been calculated using Mann–Whitney U-test. §P-value has been calculated using the Chi-square test. ¥P-value has been calculated using Fisher’s Exact test. **Significant at P<0.05 level. ICU=Intensive Care Unit; HDU=High Dependency Unit
Figure 1.
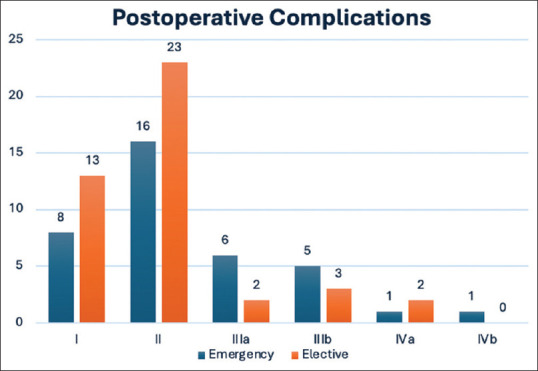
Severity of postoperative complications according to Clavien–Dindo classification. (N.B. Some patients had more than one complication)
A binary logistic regression analysis has been performed in Table 4 to determine the prognostic variables for surgical complications. Based on the results, it was observed that the increased risk for surgical complications was predicted among patients with associated comorbidities with 3.22-fold higher (OR = 3.218; 95% CI = 1.544 – 6.708; P = 0.002), 2.2 times higher among patients with albumin less than 30 (OR = 2.204; 95% CI = 1.144 – 4.246; P = 0.018), 2.43-fold higher among completely obstructed patients (OR = 2.426; 95% CI = 1.175 – 5.009; P = 0.017), 9.98-fold higher in perforation (OR = 9.984; 95% CI = 2.540 – 39.25; P = 0.001), 4.84 times higher among patients who underwent open surgery (OR = 4.841; 95% CI = 2.453 – 9.556; P < 0.001), 4.76-fold higher among patients who underwent left hemicolectomy (OR = 4.762; 95% CI = 1.005 – 22.56; P = 0.049), 12.5-fold higher among those who underwent total-subtotal colectomy (OR = 12.5; 95% CI = 2.127 – 73.47; P = 0.005) and 7.84 times higher among those who underwent Hartmann procedure (OR = 7.843; 95% CI = 1.708 – 36.01; P = 0.0005). However, regression estimates showed that increasing age (OR = 0.957; 95% CI = 0.932 – 0.982; P = 0.001), Body mass index (BMI) (OR = 0.943; 95% CI = 0.891 – 0.999; P = 0.044), ASA score (OR = 0.428; 95% CI = 0.263 – 0.697; P = 0.001) were associated with increased risk to surgical complications.
Table 4.
Prognostic variables toward complications (n=215)
| Factor | Complications N (%) (n=51) | No Complication N (%) (n=164) | OR (95% CI) | P |
|---|---|---|---|---|
| Comorbidities | 40 (78.4%) | 87 (53%) | 3.218 (1.544 – 6.708) | 0.002** |
| Albumin <30 | 22 (43.1%) | 42 (25.6%) | 2.204 (1.144 – 4.246) | 0.018** |
| Hemoglobin <10 | 17 (33.3%) | 44 (26.8%) | 1.364 (0.693 – 2.683) | 0.369 |
| Hemoglobin <7 | 1 (2%) | 4 (2.4%) | 0.800 (0.087 – 7.323) | 0.843 |
| Presentation | ||||
| • Elective | 30 (58.8%) | 115 (70.1%) | Ref | |
| • Emergency | 21 (41.2%) | 49 (29.9%) | 1.643 (0.857 – 3.148) | 0.135 |
| Complete obstruction | 16 (31.4%) | 26 (15.9%) | 2.426 (1.175 – 5.009) | 0.017** |
| Partial obstruction | 12 (23.5%) | 42 (25.6%) | 0.894 (0.428 – 1.866) | 0.765 |
| Perforation | 8 (15.7%) | 3 (1.8%) | 9.984 (2.540 – 39.25) | 0.001** |
| Type of operation | ||||
| • Laparoscopic | 25 (49%) | 135 (82.3%) | Ref | |
| • Open | 26 (51%) | 29 (17.7%) | 4.841 (2.453 – 9.556) | <0.001** |
| Conversion to open | 9 (17.6%) | 14 (8.5%) | 2.296 (0.929 – 5.673) | 0.072 |
| TNM Pathological Stage | ||||
| • Stage 1 | 6 (11.8%) | 28 (17.1%) | Ref | |
| • Stage 2 | 22 (43.1%) | 54 (32.9%) | 1.922 (0.553 – 6.679) | 0.304 |
| • Stage 3 | 16 (31.4%) | 65 (39.6%) | 1.011 (0.368 – 2.776) | 0.984 |
| • Stage 4 | 7 (13.7%) | 17 (10.4%) | 1.673 (0.593 – 4.715) | 0.331 |
| Preoperative transfusion | 6 (11.8%) | 14 (8.5%) | 1.429 (0.519 – 3.933) | 0.490 |
| Intraoperative transfusion | 2 (3.9%) | 4 (2.4%) | 1.633 (0.290 – 9.184) | 0.578 |
| Type of procedure | ||||
| • Right hemicolectomy | 14 (27.5%) | 40 (24.4%) | Ref | |
| • Left hemicolectomy | 4 (7.8%) | 30 (18.3%) | 4.762 (1.005 – 22.56) | 0.049** |
| • Total/sub-total colectomy | 11 (21.6%) | 11 (6.7%) | 12.50 (2.127 – 73.47) | 0.005** |
| • Anterior resection | 17 (33.3%) | 80 (48.8%) | 1.667 (0.318 – 8.743) | 0.546 |
| • Hartmann procedure | 5 (9.8%) | 3 (1.8%) | 7.843 (1.708 – 36.01) | 0.008** |
| Age in years, mean±SD | 63.8±12.9 | 55.8±12.9 | 0.957 (0.932 – 0.982) | 0.001** |
| BMI (kg/m2), mean±SD | 29.2±6.01 | 27.3±5.29 | 0.943 (0.891 – 0.999) | 0.044** |
| ASA score, mean±SD | 2.54±0.82 | 2.16±0.65 | 0.428 (0.263 – 0.697) | 0.001** |
| • ASA 1 | 6 (11.8%) | 23 (14%) | Ref | |
| • ASA 2 | 19 (37.3%) | 92 (56.1%) | 2.034 (0.736 – 5.621) | 0.171 |
| • ASA 3 and 4 | 26 (51%) | 49 (29.9%) | 2.569 (1.294 – 5.100) | 0.007** |
| Operative time, median (min-max) | 210.5 (89-395) | 210.5 (54-530) | 0.995 (0.991 – 0.999) | 0.021** |
*Some patients have multiple tumor sites. **Significant at P<0.05 level. OR=Odds ratio, CI=Confidence Interval, TNM=Tumor, Node, and metastasis, BMI=Body Mass Index, ASA=American Society of Anesthesiologists
The results of the multivariate regression analysis are shown in Table 5, for prognostic variables for complications in general. It was found that there is increased risk in patients with comorbidities (Adjusted Odds Ratio [AOR] = 4.527; 95% CI = 1.732-11.828; P = 0.002), blood albumin levels below 30 (AOR = 2.122; 95% CI = 1.081-4.163; P = 0.029), and perforated patients (AOR = 9.33; 95% CI = 2.312-37.66; P = 0.002). In addition, the type of surgery - open laporatomy (AOR = 4.985; 95% CI = 2.456-10.11; P < 0.001) and total-subtotal colectomy (AOR = 9.19; 95% CI = 1.47-57.41; P = 0.018) were associated with higher odds of complications. In addition, ASA scores 3 and 4 showed a higher likelihood of complications (AOR = 2.31; 95% CI = 1.12-4.75; P = 0.023). Finally, increased age, high BMI, and longer operation times were associated with higher risk of developing surgical complications (AOR = 0.96; 95% CI = 0.93-0.988; P = 0.006), (AOR = 0.92; 95% CI = 0.86-0.98; P = 0.011), (AOR = 0.99; 95% CI = 0.99-1.0; P = 0.043), respectively.
Table 5.
Multivariate regression analysis for prognostic variables for complications in general (n=215)
| Factor | AOR | 95% CI | P |
|---|---|---|---|
| Comorbidities | 4.527 | 1.732 – 11.828 | 0.002** |
| Albumin <30 | 2.122 | 1.081 – 4.163 | 0.029** |
| Complete Obstruction | 1.580 | 0.770 – 3.242 | 0.213 |
| Perforation | 9.333 | 2.312 – 37.667 | 0.002** |
| Type of operation | |||
| • Laparoscopic | Ref | ||
| • Open | 4.985 | 2.456 – 10.118 | <0.001** |
| Type of procedure | |||
| • Right hemicolectomy | Ref | ||
| • Left hemicolectomy | 3.517 | 0.699 – 17.691 | 0.127 |
| • Total-sub total colectomy | 9.194 | 1.472 – 57.419 | 0.018** |
| • Hartmann procedure | 0.888 | 0.154 – 5.138 | 0.895 |
| ASA | |||
| • ASA 1 | Ref | ||
| • ASA 2 | 1.817 | 0.585 – 5.646 | 0.302 |
| • ASA 3 and 4 | 2.312 | 1.124 – 4.754 | 0.023** |
| Age in years, mean±SD | 0.961 | 0.934 – 0.988 | 0.006** |
| BMI (kg/m2), mean±SD | 0.920 | 0.863 – 0.981 | 0.011** |
| Operative time, median (min-max) | 0.996 | 0.991 – 1.000 | 0.043** |
Adjusted with gender, smoking, family history of cancer and DM. AOR=Adjusted Odd Ratio, CI=Confidence Interval, ASA=American society of anesthesiologists, BMI=Body mass index. **Significant at P<0.05 level
DISCUSSION
Colon cancer emergency presentations are detrimental to patients’ outcome in numerous ways.[6,13,14,15] The incidence of emergency presentation in patients diagnosed with colon cancer has been approximately in the range of 8% to 33% based on several studies.[6,10,13,16,17] Our results revealed a high prevalence rate of 32.6% for emergency presentation among our patients. Lack of proper channels for healthcare access[2] can explain this finding, in addition to the potential implications for overall outcomes and quality of care at our center. Hence, this study aimed to assess the clinical outcomes of emergency versus elective presentation of colon cancer patients who underwent curative resection. Also, we investigated the prognostic factors toward postoperative complications.
Laparoscopic approach offers numerous advantages in colon surgery. Many studies reported early recovery of bowel function, shorter hospital stay, lower rate of morbidity and mortality in comparison to open surgery.[18,19,20] Moreover, no significant disparity was observed in terms of oncological outcomes between laparoscopic and open surgical approaches.[21,22] In our study, the laparoscopic approach was utilized significantly less in the emergency group (P < 0.001) with a lower conversion rate. Similarly, open colectomies were commonly prevalent in emergency surgeries.[23,24] This is most likely attributed to the increased complexity of laparoscopic surgery in cases including bowel distention, in the event of an obstruction, or abdominal contamination and inflammation. A recent meta-analysis of 29 international studies showed that a wide range of laparoscopic versus open approach was undertaken in such scenarios (3.3% - 66.7%), while it’s worthwhile to note that most of the centers with higher rates of laparoscopic surgery were centers with experienced colorectal surgeons.[25] In contrast to our findings, a local study reported only 24.4% of emergency surgeries being done using laparoscopy compared to 57.1% in our study.[26] Furthermore, some international studies found higher rates of 43.5%[27], and 16.3% to 35.4% over the past 7 years in the laparoscopic approach.[28] In our study, we observed that diverting stoma, end colostomy/ileostomy, and primary anastomosis with diversion were significantly higher in patients with an emergency presentation. Conversely, primary anastomosis was the most common approach performed in the elective group (P < 0.001). These findings are consistent with other studies that report higher stoma creation in the emergency groups.[29,30]
Morbidity and postoperative complications were reported to be higher among patients with emergency surgery in many studies.[30,31,32] In contrast, Amri et al. found the rates of complications among patients hospitalized after emergency presentation were not significantly different when compared to those who underwent elective colon cancer resections.[33] In our results, the post-operative complication rate was higher in the emergency group (30%) compared to the elective group (20.7%). However, it was not statistically significant (P = 0.171). The most common complications reported in our study were intra-abdominal collection and wound infection. Moreover, those who were admitted electively had a much lower rate of intra-abdominal collection (3.4%) compared to 17.1% in the emergency group (P < 0.001). In addition, our study found that the individuals who underwent emergency surgical procedures exhibited more ICU admissions and longer hospital stays than the elective group.
Many factors were correlated with higher postoperative complications in colon cancer, such as late stages of cancer, ASA score, increasing age, hypoalbuminemia, and emergency presentation.[9,15,34] Malnutrition is commonly encountered in patients with colon cancer, and albumin serves as one of the key markers used to evaluate their nutritional status.[34] Several studies have documented a potential correlation between hypoalbuminemia and unfavorable outcomes following gastrointestinal surgery.[35–37] However, it is worth noting that contradictory findings have also been reported in other investigations.[38,39] In our study, it was observed that a blood albumin level below 30 was significantly correlated with a twofold increase in postoperative complications. This is further supported by Truong et al. (2016) in their study of preoperative measurement of albumin and preoperative optimization, which led to improved overall outcomes in morbidity and mortality in colorectal surgery.[40] In terms of both short-term and long-term outcomes, the literature does not give any precise comparisons between obstruction and perforation. However, it is reported that perforation is associated with high postoperative complications.[41,42,43] Our investigation revealed that there is a much higher risk of complications associated with complete obstruction (2.5-fold increase) and perforation (10-fold increase). Nevertheless, patients presenting with partial obstruction have not shown a significant association. Moreover, many studies have shown that emergency cases were more likely to be open surgery rather than laparoscopic,[23,44] which corroborates our findings. Moreover, previous research has demonstrated a correlation between open surgeries and increased incidence of complications.[19] Similarly, our study revealed a higher occurrence of complications in open surgeries as compared to laparoscopic surgeries.
This study highlights the prevalence of emergent colonic cancer, while not necessarily contributing to a higher complication rate; It showed an overall higher length of stay, in addition to increased exposure to more invasive procedures. The study further identifies several independent risk factors for complications, including complete obstruction, perforation, conventional surgical approaches, hypoalbuminemia, and the presence of comorbidities. It posits that the future enhancement of patient outcomes may hinge on the implementation of systematic colorectal cancer screening programs, such as occult fecal blood tests and colonoscopies. In addition to more awareness campaigns, such programs could heighten public awareness, potentially reducing the incidence of emergency presentations and subsequent elective resections.
This study has some limitations. First, the overall number of participants is lower when compared to other studies, and with a small sample size, we could not fully adjust for all potential confounding factors. In addition, the retrospective nature could indicate possible bias. Finally, this is the experience of a single center; a multicentric approach is recommended to generalize these findings. However, this study does take place in a tertiary care center with a dedicated colorectal surgery department. In addition, this remains the largest local cohort assessing surgical outcomes in such patients within this region.
In conclusion, the emergency group showed overall worse outcomes; open surgery and stoma creation rates were more evident compared to the elective group. Complete obstruction, perforation, open surgery, hypoalbuminemia, and the presence of comorbidities were independent risk factors for complications. More efforts should be directed to improve public awareness and early detection of colon cancer.
Conflicts of interest
There are no conflicts of interest.
Funding Statement
Nil.
REFERENCES
- 1.Ahmed M. Colon cancer: A clinician's perspective in 2019. Gastroenterology Res. 2020;13:1–10. doi: 10.14740/gr1239. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Almatroudi A. The incidence rate of colorectal cancer in Saudi Arabia: An observational descriptive epidemiological analysis. Int J Gen Med. 2020;13:977–90. doi: 10.2147/IJGM.S277272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Cappell MS. Pathophysiology, clinical presentation, and management of colon cancer. Gastroenterol Clin North Am. 2008;37:1–24. doi: 10.1016/j.gtc.2007.12.002. [DOI] [PubMed] [Google Scholar]
- 4.Burt RW. Colon cancer screening. Gastroenterology. 2000;119:837–53. doi: 10.1053/gast.2000.16508. [DOI] [PubMed] [Google Scholar]
- 5.Menon G, Cagir B. StatPearls [Internet] Treasure Island (FL): StatPearls Publishing; 2025. [[Last accessed on 2025 Feb 27]]. Colon Cancer. Available from: https://www.ncbi.nlm.nih.gov/books/NBK470380/ [Google Scholar]
- 6.Baer C, Menon R, Bastawrous S, Bastawrous A. Emergency presentations of colorectal cancer. Surg Clin North Am. 2017;97:529–45. doi: 10.1016/j.suc.2017.01.004. [DOI] [PubMed] [Google Scholar]
- 7.Pisano M, Zorcolo L, Merli C, Cimbanassi S, Poiasina E, Ceresoli M, et al. 2017 WSES guidelines on colon and rectal cancer emergencies: Obstruction and perforation. World J Emerg Surg. 2018;13:36. doi: 10.1186/s13017-018-0192-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Chen TM, Huang YT, Wang GC. Outcome of colon cancer initially presenting as colon perforation and obstruction. World J Surg Oncol. 2017;15:164. doi: 10.1186/s12957-017-1228-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Sjo OH, Larsen S, Lunde OC, Nesbakken A. Short term outcome after emergency and elective surgery for colon cancer. Colorectal Dis. 2009;11:733–9. doi: 10.1111/j.1463-1318.2008.01613.x. [DOI] [PubMed] [Google Scholar]
- 10.Jestin P, Nilsson J, Heurgren M, Påhlman L, Glimelius B, Gunnarsson U. Emergency surgery for colonic cancer in a defined population. Br J Surg. 2005;92:94–100. doi: 10.1002/bjs.4780. [DOI] [PubMed] [Google Scholar]
- 11.Bin Traiki TA, AlShammari SA, AlRabah RN, AlZahrani AM, Alshenaifi ST, Alhassan NS, et al. Oncological outcomes of elective versus emergency surgery for colon cancer: A tertiary academic center experience. Saudi J Gastroenterol. 2023;29:316–22. doi: 10.4103/sjg.sjg_31_23. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Dindo D, Demartines N, Clavien PA. Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann Surg. 2004;240:205–13. doi: 10.1097/01.sla.0000133083.54934.ae. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Ghazi S, Berg E, Lindblom A, Lindforss U Low-Risk Colorectal Cancer Study Group. Clinicopathological analysis of colorectal cancer: A comparison between emergency and elective surgical cases. World J Surg Oncol. 2013;11:133. doi: 10.1186/1477-7819-11-133. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Mitchell AD, Inglis KM, Murdoch JM, Porter GA. Emergency room presentation of colorectal cancer: A consecutive cohort study. Ann Surg Oncol. 2007;14:1099–104. doi: 10.1245/s10434-006-9245-z. [DOI] [PubMed] [Google Scholar]
- 15.McArdle CS, Hole DJ. Emergency presentation of colorectal cancer is associated with poor 5-year survival. Br J Surg. 2004;91:605–9. doi: 10.1002/bjs.4456. [DOI] [PubMed] [Google Scholar]
- 16.Arnarson Ö, Syk I, Butt ST. Who should operate patients presenting with emergent colon cancer? A comparison of short- and long-term outcome depending on surgical sub-specialization. World J Emerg Surg. 2023;18:3. doi: 10.1186/s13017-023-00474-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Hogan J, Samaha G, Burke J, Chang KH, Condon E, Waldron D, et al. Emergency presenting colon cancer is an independent predictor of adverse disease-free survival. Int Surg. 2015;100:77–86. doi: 10.9738/INTSURG-D-13-00281.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Reza MM, Blasco JA, Andradas E, Cantero R, Mayol J. Systematic review of laparoscopic versus open surgery for colorectal cancer. Br J Surg. 2006;93:921–8. doi: 10.1002/bjs.5430. [DOI] [PubMed] [Google Scholar]
- 19.Papageorge CM, Zhao Q, Foley EF, Harms BA, Heise CP, Carchman EH, et al. Short-term outcomes of minimally invasive versus open colectomy for colon cancer. J Surg Res. 2016;204:83–93. doi: 10.1016/j.jss.2016.04.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Zwanenburg ES, Veld JV, Amelung FJ, Borstlap WA, Dekker JW, Hompes R, et al. On behalf of the Dutch Snapshot Research Group. Short- and Long-term outcomes after laparoscopic emergency resection of left-sided obstructive colon cancer: A nationwide propensity score-matched analysis. Dis Colon Rectum. 2023;66:774–84. doi: 10.1097/DCR.0000000000002364. [DOI] [PubMed] [Google Scholar]
- 21.Sammour T, Jones IT, Gibbs P, Chandra R, Steel MC, Shedda SM, et al. Comparing oncological outcomes of laparoscopic versus open surgery for colon cancer: Analysis of a large prospective clinical database. J Surg Oncol. 2015;111:891–8. doi: 10.1002/jso.23893. [DOI] [PubMed] [Google Scholar]
- 22.Jackson TD, Kaplan GG, Arena G, Page JH, Rogers SO., Jr Laparoscopic versus open resection for colorectal cancer: A metaanalysis of oncologic outcomes. J Am Coll Surg. 2007;204:439–46. doi: 10.1016/j.jamcollsurg.2006.12.008. [DOI] [PubMed] [Google Scholar]
- 23.Odermatt M, Miskovic D, Siddiqi N, Khan J, Parvaiz A. Short- and long-term outcomes after laparoscopic versus open emergency resection for colon cancer: An observational propensity score-matched study. World J Surg. 2013;37:2458–67. doi: 10.1007/s00268-013-2146-y. [DOI] [PubMed] [Google Scholar]
- 24.Ozhathil DK, Li Y, Smith JK, Witkowski E, Coyne ER, Alavi K, et al. Colectomy performance improvement within NSQIP 2005-2008. J Surg Res. 2011;171:e9–13. doi: 10.1016/j.jss.2011.06.052. [DOI] [PubMed] [Google Scholar]
- 25.Warps AK, Zwanenburg ES, Dekker JW, Tollenaar RA, Bemelman WA, Hompes R, et al. Laparoscopic versus open colorectal surgery in the emergency setting: A systematic review and meta-analysis. Ann Surg Open. 2021;2:e097. doi: 10.1097/AS9.0000000000000097. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Alselaim NA, Altoub HA, Alhassan MK, Alhussain RM, Alsubaie AA, Almomen FA, et al. The role of laparoscopy in emergency colorectal surgery. Saudi Med J. 2022;43:1333–40. doi: 10.15537/smj.2022.43.12.20220658. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hussain MI, Piozzi GN, Sakib N, Duhoky R, Carannante F, Khan JS. Laparoscopic versus Open Emergency Surgery for Right Colon Cancers. Diagnostics (Basel) 2024;14:407. doi: 10.3390/diagnostics14040407. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Smyth R, Darbyshire A, Mercer S, Khan J, Richardson J. Trends in emergency colorectal surgery: A 7-year retrospective single-centre cohort study. Surg Endosc. 2023;37:3911–20. doi: 10.1007/s00464-023-09876-0. [DOI] [PubMed] [Google Scholar]
- 29.Lavanchy JL, Vaisnora L, Haltmeier T, Zlobec I, Brügger LE, Candinas D, et al. Oncologic long-term outcomes of emergency versus elective resection for colorectal cancer. Int J Colorectal Dis. 2019;34:2091–9. doi: 10.1007/s00384-019-03426-8. [DOI] [PubMed] [Google Scholar]
- 30.Bayar B, Yılmaz KB, Akıncı M, Şahin A, Kulaçoğlu H. An evaluation of treatment results of emergency versus elective surgery in colorectal cancer patients. Ulus Cerrahi Derg. 2015;32:11–7. doi: 10.5152/UCD.2015.2969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wanis KN, Ott M, Van Koughnett JA, Colquhoun P, Brackstone M. Long-term oncological outcomes following emergency resection of colon cancer. Int J Colorectal Dis. 2018;33:1525–32. doi: 10.1007/s00384-018-3109-4. [DOI] [PubMed] [Google Scholar]
- 32.Acar N. Should we still doubt the success of emergency oncologic colorectal surgery?: A Retrospective Study. Turk J Trauma Emerg Surg. 2019. [[Last accessed on 2023 Sep 16]]. Available from: https://jag.journalagent.com/travma/pdfs/UTD-04043-CLINICAL_ARTICLE-ACAR.pdf . [DOI] [PubMed]
- 33.Amri R, Bordeianou LG, Sylla P, Berger DL. Colon cancer surgery following emergency presentation: Effects on admission and stage-adjusted outcomes. Am J Surg. 2015;209:246–53. doi: 10.1016/j.amjsurg.2014.07.014. [DOI] [PubMed] [Google Scholar]
- 34.Chiang JM, Chang CJ, Jiang SF, Yeh CY, You JF, Hsieh PS, et al. Pre-operative serum albumin level substantially predicts post-operative morbidity and mortality among patients with colorectal cancer who undergo elective colectomy. Eur J Cancer Care (Engl) 2017;26 doi: 10.1111/ecc.12403. doi: 10.1111/ecc.12403. [DOI] [PubMed] [Google Scholar]
- 35.Lai CC, You JF, Yeh CY, Chen JS, Tang R, Wang JY, et al. Low preoperative serum albumin in colon cancer: A risk factor for poor outcome. Int J Colorectal Dis. 2011;26:473–81. doi: 10.1007/s00384-010-1113-4. [DOI] [PubMed] [Google Scholar]
- 36.Buzby GP, Mullen JL, Matthews DC, Hobbs CL, Rosato EF. Prognostic nutritional index in gastrointestinal surgery. Am J Surg. 1980;139:160–7. doi: 10.1016/0002-9610(80)90246-9. [DOI] [PubMed] [Google Scholar]
- 37.Detsky AS, Baker JP, O’Rourke K, Johnston N, Whitwell J, Mendelson RA, et al. Predicting Nutrition-associated complications for patients undergoing gastrointestinal surgery. JPEN J Parenter Enteral Nutr. 1987;11:440–6. doi: 10.1177/0148607187011005440. [DOI] [PubMed] [Google Scholar]
- 38.Ryan JA, Taft DA. Preoperative nutritional assessment does not predict morbidity and mortality in abdominal operations. Surg Forum. 1980;31:96–8. [Google Scholar]
- 39.Nilsson E, Lamke LO, Liljedahl SO. Elfstrnot predict morbidity and mortality in abdomin for colorectal cancer? Acta Chir Scand. 1980;146:619–22. [PubMed] [Google Scholar]
- 40.Truong A, Hanna MH, Moghadamyeghaneh Z, Stamos MJ. Implications of preoperative hypoalbuminemia in colorectal surgery. World J Gastrointest Surg. 2016;8:353–62. doi: 10.4240/wjgs.v8.i5.353. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Fielding LP, Phillips RK, Hittinger R. Factors influencing mortality after curative resection for large bowel cancer in elderly patients. Lancet. 1989;1:595–7. doi: 10.1016/s0140-6736(89)91618-8. [DOI] [PubMed] [Google Scholar]
- 42.Runkel NS, Schlag P, Schwarz V, Herfarth C. Outcome after emergency surgery for cancer of the large intestine. Br J Surg. 1991;78:183–8. doi: 10.1002/bjs.1800780216. [DOI] [PubMed] [Google Scholar]
- 43.Alvarez JA, Baldonedo RF, Bear IG, Truán N, Pire G, Alvarez P. Presentation, treatment, and multivariate analysis of risk factors for obstructive and perforative colorectal carcinoma. Am J Surg. 2005;190:376–82. doi: 10.1016/j.amjsurg.2005.01.045. [DOI] [PubMed] [Google Scholar]
- 44.Osagiede O, Spaulding AC, Cochuyt JJ, Naessens JM, Merchea A, Crandall M, et al. Factors associated with minimally invasive surgery for colorectal cancer in emergency settings. J Surg Res. 2019;243:75–82. doi: 10.1016/j.jss.2019.04.089. [DOI] [PubMed] [Google Scholar]


