Abstract
Background
Numerous types of arthroplasties may be used in the surgical treatment of a hip fracture (proximal femoral fracture). The main differences between the implants are in the design of the stems, whether the stem is cemented or uncemented, whether a second articulating joint is included within the prosthesis (bipolar prosthesis), or whether a partial (hemiarthroplasty) or total whole hip replacement is used.
Objectives
To review all randomised controlled trials comparing different arthroplasties for the treatment of hip fractures in adults.
Search methods
We searched the Cochrane Bone, Joint and Muscle Trauma Group Specialised Register (September 2009), CENTRAL (The Cochrane Library 2009, Issue 3), MEDLINE, EMBASE and trial registers (all to September 2009), and reference lists of articles.
Selection criteria
All randomised and quasi‐randomised controlled trials comparing different arthroplasties and their insertion with or without cement, for the treatment of hip fractures.
Data collection and analysis
Two review authors independently assessed trial quality, by use of a 10‐item checklist, and extracted data.
Main results
Twenty‐three trials involving 2861 older and mainly female patients with proximal femoral fractures are included. Cemented prostheses, when compared with uncemented prostheses (6 trials, 899 participants) were associated with a less pain at a year or later and improved mobility. No significant difference in surgical complications was found. One trial of 220 participants compared a hydroxyapatite coated hemiarthroplasty with a cemented prosthesis and reported no notable differences between the two prosthesis. Comparison of unipolar hemiarthroplasty with bipolar hemiarthroplasty (7 trials, 857 participants, 863 fractures) showed no significant differences between the two types of implant. Seven trials involving 734 participants compared hemiarthroplasty with a total hip replacement (THR). Most studies involved cemented implants. Dislocation of the prosthesis was more common with the THR but there was a general trend within these studies to better functional outcome scores for those treated with the THR.
Authors' conclusions
There is good evidence that cementing the prostheses in place will reduce post‐operative pain and lead to better mobility. From the trials to date there is no evidence of any difference in outcome between bipolar and unipolar prosthesis. There is some evidence that a total hip replacement leads to better functional outcome than a hemiarthroplasty. Further well‐conducted randomised trials are required.
Keywords: Adult; Female; Humans; Male; Bone Cements; Arthroplasty, Replacement, Hip; Arthroplasty, Replacement, Hip/methods; Femoral Neck Fractures; Femoral Neck Fractures/surgery; Hip Fractures; Hip Fractures/surgery; Prosthesis Design; Randomized Controlled Trials as Topic; Treatment Outcome
Plain language summary
Comparison of different types of artificial hip joints that may be used for treating fractures of the hip
Many different types of artificial hip joints (arthroplasties) may be used to treat hip fractures, which are breaks (fractures) in the thigh bone (femur) near the hip joint. Differences in these artificial joints include different shapes of the stem set into the bone; the incorporation of a secondary joint (bipolar joint); joints that replace only the ball part of the ball and socket hip joint (hemiarthroplasty) and those that also involve replacing the socket part of the hip joint (total hip replacement). In addition an arthroplasty may be of the press fit type or secured in place within the bone using a filler (bone cement).
Twenty‐three trials involving 2861 older and mainly female patients with hip fractures are included in this review. The findings from the three main comparisons are summarised here. Six studies involving 899 participants compared a press fit arthroplasty with one that was secured in place with bone cement. Those joints that were cemented in place resulted in less pain and better mobility than those that were of the press fit type. Seven trials involving 857 participants compared those implants which have a second joint built into them (bipolar hemiarthroplasties) with those without this additional joint (unipolar hemiarthroplasties). No notable differences between these two types of implant were demonstrated. Seven studies of 734 participants compared different types of hemiarthroplasty with a total hip replacement. Most implants had been cemented in place. There was a trend to better functional outcomes after total hip replacement, but firm conclusions could not be made because of the lack of patient numbers.
There is good evidence that people with arthroplasties that are cemented in place have less pain and better mobility after the operation than those, which are inserted as a press fit. There is limited evidence that a total hip replacement leads to better functional outcome than a hemiarthroplasty.
Background
Description of the condition
Arthroplasty of the hip refers to replacement of all or part of the hip joint with a prosthetic implant. Numerous different types of arthroplasty exist for the hip. Those that involve replacement of the femoral head can be divided into two groups: hemiarthroplasty and total hip replacement (THR). Hemiarthroplasty involves replacing the femoral head with a prosthesis whilst retaining the natural acetabulum and acetabular cartilage. The type of hemiarthroplasty can be divided into two groups: unipolar and bipolar. Total hip replacement involves the replacement of the acetabulum in addition to the femoral head. The acetabular component is usually made of a combination of high density polyethylene and metal, and is often cemented into place. The femoral stems may be either held in place with cement or inserted as a 'press fit', without cement.
Description of the intervention
Unipolar hemiarthroplasty
The best known of the early hemiarthroplasty designs are the Moore prosthesis (Moore 1952) and the FR Thompson Hip Prosthesis (Thompson 1954). These are both one‐piece all‐metal implants and remain, 40 years after their introduction, two of the most frequently used hemiarthroplasty prostheses. Both these prostheses were designed before the development of polymethylmethacrylate bone cement and were therefore originally inserted as a 'press fit'. The Moore prosthesis has a femoral stem, which is fenestrated and also has a square stem with a shoulder to enable stabilisation within the femur, which resists rotation within the femoral canal. It is generally used without cement and, in the long term, bone in‐growth into the fenestrations frequently occurs. The Thompson prosthesis has a smaller stem without fenestrations and nowadays is often used in conjunction with cement. Numerous other designs of unipolar hemiarthroplasties exist based on stems that have been used for total hip replacements. Depending on the design of the stem they may be used either with or without cement.
Bipolar prostheses
These prostheses are designed to allow movement to occur, not only between the acetabulum and the prosthesis, but also at a joint within the prosthesis itself. The object of the second joint is to reduce acetabular wear. This type of prosthesis has a spherical inner metal head with a size between 22 to 36 millimetres in diameter. This fits into a polyethylene shell, which in turn is enclosed by a metal cap. There are a number of different types of prostheses with different stem designs. Examples of bipolar prostheses are the Charnley‐Hastings, Bateman, Giliberty and the Monk prostheses, but many other types with different stem designs exist. The bipolar concept has been further developed by the incorporation of a modular joint into some of the prostheses. This is a tapered joint in which a socket in the head of the prosthesis fits onto a tapered projection from the stem. The modular joint enables a variety of different stems to be used with or without cement fixation. A range of different heads are available and the options include a bipolar jointed head, ceramic head or a simple unipolar head.
Total hip replacement
This arthroplasty consists of both a femoral component and an acetabular component. Numerous designs of THR exist. Either one or both components may be held in place with cement.
Hydroxyapatite coating
A prosthesis may be coated with a substance such as hydroxyapatite which promotes bone growth around the prosthesis, thus forming a strong bond between the prosthesis and the bone. Implants coated with this are therefore inserted as a press fit rather than with cement.
Bone cement
Polymethylmethacrylate bone cement may be inserted at the time of surgery. It sets hard to form a solid bond between the prosthesis and the femoral bone at the time of surgery. Without cement a firm bond between the prosthesis and femur is dependent upon bony in‐growth and osseous integration. Potential advantages of cement are a reduced degree of post‐operative pain, as the prosthesis is more firmly fixed within the femur and a reduced long‐term revision rate from loosening of the prosthesis. Major side effects of cement are cardiac arrhythmias and cardio‐respiratory collapse, which occasionally occur following its insertion. These complications may be fatal. The cause of this is either embolism from marrow contents forced into the circulation (Christie 1994) or a direct toxic effect of the cement. Another consideration with cement is that it may make revision arthroplasty more difficult.
Potential complications of arthroplasty of the hip include wound infection (superficial and deep), dislocation of the prosthesis, loosening of the prosthesis stem, loosening of the acetabular component (THR only), acetabular wear (hemiarthroplasty only), breakage of the implant, disassembly of the implant, and fracture below the implant.
Objectives
We aimed to compare the relative effects (benefits and harms) of:
prostheses inserted with cement versus without cement;
different types of unipolar hemiarthroplasties;
different types of bipolar hemiarthroplasties;
unipolar hemiarthroplasty versus bipolar hemiarthroplasty;
uncemented hemiarthroplasty versus total hip replacement;
cemented hemiarthroplasty versus total hip replacement;
different types of total hip replacement.
Methods
Criteria for considering studies for this review
Types of studies
All randomised controlled trials comparing different types of arthroplasty and the use of arthroplasty with or without cement for the treatment of fractures of the proximal femur. Quasi‐randomised trials and trials in which the treatment allocation was inadequately concealed were considered for inclusion.
Types of participants
Skeletally mature patients with a proximal femoral fracture. The types of fractures studied were all intracapsular hip fractures, but had any study been found on extracapsular fractures they would have been be included.
Types of interventions
Prostheses (unipolar hemiarthroplasty, bipolar hemiarthroplasty, or THR), as described in the Background, applied with or without cement.
Types of outcome measures
Data for the following outcomes were sought:
1. Operative details
length of surgery (in minutes)
hypotension during surgery
operative fracture of the femur
operative blood loss (in millilitres)
post‐operative blood transfusion (in units)
number of patients transfused
2. Implant related complications
dislocation of the prosthesis
loosening of the prosthesis
acetabular wear
breakage of the implant
disassembly of the implant
fracture below or around the implant
complications related to cement insertion
other surgical complications of fixation (as detailed in each study)
reoperation and revision rate (within the follow‐up period of the study). For the analyses these are separated into 'minor' reoperations such as closed reduction of a dislocation or attention to the wound or surrounding tissues, and 'major' reoperations such as revision or removal of the implant or open reduction of a dislocation.
superficial wound infection (infection of the wound in which there is no evidence that the infection extends to the site of the implant)
deep wound infection (infection around the implant)
3. Post‐operative complications
pneumonia
thromboembolic complications (deep vein thrombosis or pulmonary embolism)
any medical complication (as detailed in each individual study)
4. Hospital stay and use of resources
length of hospital stay (in days)
cost of treatment
5. Anatomical restoration
leg shortening
range of hip movements
6. Final outcome measures
mortality (within the follow‐up period of the study, both early and late)
pain (persistent pain at the final follow‐up assessment)
residence at final follow up (return to living at home, discharge location)
mobility (use of walking aids, return of mobility)
other functional outcomes as listed in each study
health related quality of life measures
The final outcome measures (category 6) should be considered the most important outcomes.
Search methods for identification of studies
Electronic searches
We searched the Cochrane Bone, Joint and Muscle Trauma Group Specialised Register (September 2009), the Cochrane Central Register of Controlled Trials (The Cochrane Library 2009, Issue 3), MEDLINE (1950 to September 2009), MEDLINE In‐process (18 September 2009), EMBASE (1988 to 2009 week 38), CINAHL (to January 2007), and our own reference databases. No language restriction was applied.
In MEDLINE we combined a subject‐specific search strategy with the Cochrane Highly Sensitive Search Strategy for identifying randomised trials in MEDLINE: sensitivity‐ and precision‐maximizing version (Lefebvre 2008). SeeAppendix 1 for search strategies used in this update.
Former search strategies for MEDLINE and EMBASE are shown in Appendix 2, along with the CINAHL strategy not used for this update.
We searched the WHO International Clinical Trials Registry Platform Search Portal, Current Controlled Trials, and the UK National Research Register (NRR) Archive (all to 21 September 2009) to identify ongoing and recently completed trials.
We also scrutinised weekly downloads of "Fracture" articles in new issues of 17 journals (Acta Orthopaedica Scandinavica (to December 2004); American Journal of Orthopedics; Archives of Orthopaedic and Trauma Surgery; Clinical Journal of Sport Medicine; Clinical Orthopaedics and Related Research; Emergency Medicine Clinics of North America; Foot & Ankle International; Injury; Journal of Accident & Emergency Medicine (to November 2000); Journal of the American Academy of Orthopaedic Surgeons; Journal of Arthroplasty; The Journal of Bone and Joint Surgery ‐ American; The Journal of Bone and Joint Surgery ‐ British; Journal of Foot & Ankle Surgery; Journal of Orthopaedic Trauma; Journal of Trauma; Orthopedics) from Amedeo.
Searching other resources
We undertook a general perusal of locally accessible conference proceedings: for example, British Orthopaedic Association Congress 2000, 2001, 2002 and 2003. We also searched reference lists of articles.
Data collection and analysis
Two review authors independently extracted data for the outcomes listed above and the methodological quality of each trial was assessed independently without masking of journal name and author details (seeTable 1). Differences were resolved by discussion. The main assessment of methodology was the quality of allocation concealment. A further nine aspects of methodology were evaluated.
1. Methodological quality assessment scheme.
| 1. Was there clear concealment of allocation? | Score 3 (and code A) if allocation clearly concealed (e.g. numbered sealed opaque envelopes drawn consecutively). Score 2 (and code B) if there was a possible chance of disclosure before allocation. Score 1 (and code B) if the method of allocation concealment or randomisation was not stated or was unclear. Score 0 (and code C) if allocation concealment was clearly not concealed such as those using quasi‐randomisation (e.g. even or odd date of birth). |
| 2. Were the inclusion and exclusion criteria clearly defined? | Score 1 if text states type of fracture and which patients were included and excluded. Otherwise score 0. |
| 3. Were the outcomes of participants who withdrew or excluded after allocation described and included in an intention‐to‐treat analysis? | Score 1 if yes or text states that no withdrawals occurred or data are presented clearly showing 'participant flow' which allows this to be inferred. Otherwise score 0. |
| 4. Were the treatment and control groups adequately described at entry and if so were the groups well matched, or an appropriate co‐variate adjustment made? | Score 1 if at least four admission details given (e.g. age, sex, mobility, function score, mental test score) with either no important difference between groups or an appropriate adjustment made. Otherwise score 0. |
| 5. Were the surgeons experienced at both operations prior to commencement of the trial? | Score 1 if text states there was an introductory period or all surgeons were experienced in both operations. Otherwise score 0. |
| 6. Were the care programmes other than the trial options identical? | Score 1 if text states they were or this can be inferred. Otherwise score 0. |
| 7. Were all the outcome measures clearly defined in the text with a definition of any ambiguous terms encountered? | Score 1 if yes. Otherwise score 0. |
| 8. Were the outcome assessors blind to assignment status? | Score 1 if assessors of anatomical restoration, pain and function at follow up were blinded to treatment outcome. Otherwise score 0. |
| 9. Was the timing of outcome measures appropriate? | A minimum of 12 months follow up for all surviving participants with active review of participants at set time periods. Score 1 if yes. Otherwise score 0. |
| 10. Was loss to follow up reported and if so were less than five per cent of surviving participants lost to follow up? | Score 1 if yes. Otherwise score 0. |
Wherever necessary, and for all studies reported as only a conference abstract, the authors of studies were contacted for additional details of methodology and trial results. The authors of this review would welcome any additional information from trialists.
Data analysis
For each study, risk ratios and 95% confidence intervals were calculated for dichotomous outcomes, and mean differences and 95% confidence intervals were calculated for continuous outcomes. Where appropriate, results of comparable groups of trials were pooled using both the fixed‐effect and the random‐effects models. Heterogeneity between comparable trials was tested using a standard chi‐squared test with additional consideration of the I² statistic (Higgins 2003). Generally, the results for the random‐effects model are presented when there is significant heterogeneity (P < 0.10; I² = 50% or more) in the results of individual trials. There were insufficient data to perform subgroup analyses or sensitivity analyses.
Results
Description of studies
Twenty‐three randomised controlled trials were identified and included in this review. One quasi‐randomised trial (Dorr 1986) compared a cemented bipolar hemiarthroplasty with an uncemented bipolar hemiarthroplasty with a cemented total hip replacement in 89 patients. However, on reappraisal of the study methods, it became clear that the two bipolar hemiarthroplasty groups were not concurrent. This resulted in the removal of Dorr 1986 from the cemented versus uncemented comparison.
Six studies compared a cemented with an uncemented prosthesis. Sonne‐Holm 1982 compared an Austin Moore prosthesis with or without cement in 112 patients. Two studies compared a cemented Thompson prosthesis with an uncemented Thompson prosthesis: Branfoot 2000 (91 participants), and Harper 1994 (137 participants). Emery 1991 compared a cemented bipolar Thompson prosthesis with an uncemented bipolar Moore prosthesis in 53 patients. Santini 2005 compared a cemented bipolar hemiarthroplasty with an uncemented bipolar hemiarthroplasty in 106 patients. Parker 2009 compared an uncemented Austin Moore hemiarthroplasty with a cemented Thompson prosthesis in 400 patients.
Figved 2009 compared a cemented Bipolar Spectron hemiarthroplasty with a hydroxyapatite coated Corail bipolar prosthesis for 220 participants. Livesley 1993 compared an uncemented Moore stem bipolar prosthesis with a hydroxyapatite coated Furlong prosthesis in 82 patients.
One study (Stock 1997) compared a ceramic head Thompson prosthesis with a conventional metal Thompson prosthesis in 69 patients. It was not stated if cement was used.
Seven studies compared different unipolar hemiarthroplasties with various bipolar hemiarthroplasties. Three studies compared an Austin Moore hemiarthroplasty with the Bateman bipolar hemiarthroplasty (Malhotra 1995, 68 patients; Rosen 1992, 96 patients with 102 fractures and Van Thiel 1988, 93 patients). One study of 48 patients compared a cemented modular unipolar hemiarthroplasty with a cemented modular bipolar hemiarthroplasty (Cornell 1988). Raia 2003 also compared a cemented unipolar prosthesis with a cemented bipolar prosthesis in 115 patients. Two studies (Calder 1996; Davison 2001) compared a cemented Thompson prosthesis with a cemented Monk bipolar prosthesis in 437 patients.
Seven studies compared THRs with different types of hemiarthroplasty. The STARS 2006 trial compared a cemented bipolar hemiarthroplasty with a cemented THR in 138 patients. Baker 2006 compared a cemented unipolar hemiarthroplasty with a cemented THR in 81 patients. Blomfeldt 2007 compared a cemented Exeter stem bipolar hemiarthroplasty with a cemented Exeter stem total hip replacement in 120 participants. Two studies did not specify on the use of cement although it is probable that most of the arthroplasties were cemented in place. Mouzopoulos 2008 compared a Merete hemiarthroplasty with a total hip replacement in 86 patients. This study did not specify if cement was used although it is most likely from the prosthesis used that they were cemented in place. Macaulay 2008 compared a hemiarthroplasty with a THR in 40 patients: a variety of different types of hemiarthroplasty and THR were used in this study and it was not stated if cement was used. Dorr 1986 compared a cemented bipolar hemiarthroplasty or an uncemented bipolar hemiarthroplasty with a cemented THR in 89 patients. Skinner 1989 compared an uncemented Austin Moore hemiarthroplasty with a cemented THR in 180 patients.
Further details of these trials are given in the Characteristics of included studies. Five ongoing studies were identified. Bonke 1999 compares hemiarthroplasty with a total hip replacement, Moroni 2002 compares an uncemented with a cemented arthroplasty, Taylor compares a cemented with an uncemented hemiarthroplasty, Kavcic 2006 compares a bipolar hemiarthroplasty with a total hip replacement and Patel 2008 compares a unipolar with a bipolar hemiarthroplasty. Twelve studies were excluded for reasons given in the Characteristics of excluded studies.
Risk of bias in included studies
Twelve studies reported using an appropriate method of randomisation. STARS 2006 used a computerised telephone randomisation service. Baker 2006 and Parker 2009 used sealed opaque numbered envelopes that were independently prepared. Figved 2009 and Macaulay 2008 used sealed envelopes containing a computer generated randomised code. Cornell 1988 used sealed envelopes opened in the operating theatre. Calder 1996, Davison 2001 and Raia 2003 used computer generated random numbers. Branfoot 2000, Blomfeldt 2007 and Emery 1991 used sealed envelopes. Six studies (Malhotra 1995; Mouzopoulos 2008; Rosen 1992; Sonne‐Holm 1982; Stock 1997; Van Thiel 1988) did not state their method of randomisation. Five studies were quasi‐randomised. Dorr 1986 and Harper 1994 used the patient's hospital number; Skinner 1989, the day of admission; Santini 2005 alternated days; and Livesley 1993, the week of admission.
For studies comparing different arthroplasties, ideally follow up should be for at least five years. No study achieved this; details of the different follow‐up periods are given in the Characteristics of included studies. Cornell 1988, Davison 2001, Figved 2009, Sonne‐Holm 1982 and Parker 2009 were the only studies to have a blinded assessment of outcome.
Assessment of methodological quality
Please see Table 2 for the methodological quality scores of the individual trials, as rated using the 10‐item assessment scheme shown in Table 1.
2. Methodological quality assessment scores.
| Cemented versus uncemented prosthesis | |||||||
| Study ID | Branfoot 2000 | Emery 1991 | Harper 1994 | Parker 2009 | Santini 2005 | Sonne‐Holm 1982 | |
| 1 | 2 | 2 | 0 | 3 | 0 | 1 | |
| 2 | 1 | 1 | 1 | 1 | 1 | 1 | |
| 3 | 1 | 1 | 0 | 1 | 0 | 0 | |
| 4 | 1 | 1 | 0 | 1 | 1 | 0 | |
| 5 | 0 | 0 | 0 | 1 | 0 | 0 | |
| 6 | 1 | 1 | 1 | 1 | 1 | 0 | |
| 7 | 1 | 1 | 0 | 1 | 0 | 1 | |
| 8 | 0 | 0 | 0 | 1 | 0 | 1 | |
| 9 | 0 | 1 | 1 | 1 | 1 | 1 | |
| 10 | 1 | 0 | 0 | 1 | 0 | 1 | |
| Hemiarthroplasty versus hydroxyapatite hemiarthroplasty | |||||||
| Study ID | Figved 2009 | Livesley 1993 | |||||
| 1 | 3 | 0 | |||||
| 2 | 1 | 0 | |||||
| 3 | 0 | 0 | |||||
| 4 | 1 | 1 | |||||
| 5 | 0 | 0 | |||||
| 6 | 1 | 1 | |||||
| 7 | 1 | 1 | |||||
| 8 | 1 | 0 | |||||
| 9 | 1 | 1 | |||||
| 10 | 1 | 1 | |||||
| Hemiarthroplasty versus hemiarthroplasty (differing head materials) | |||||||
| Study ID | Stock 1997 | ||||||
| 1 | 1 | ||||||
| 2 | 0 | ||||||
| 3 | 0 | ||||||
| 4 | 0 | ||||||
| 5 | 0 | ||||||
| 6 | 0 | ||||||
| 7 | 0 | ||||||
| 8 | 0 | ||||||
| 9 | 0 | ||||||
| 10 | 0 | ||||||
| Unipolar hemiarthroplasty versus bipolar hemiarthroplasty | |||||||
| Study ID | Calder 1996 | Cornell 1988 | Davison 2001 | Malhotra 1995 | Raia 2003 | Rosen 1992 | Van Thiel 1988 |
| 1 | 2 | 3 | 2 | 1 | 2 | 1 | 1 |
| 2 | 1 | 1 | 1 | 0 | 1 | 0 | 0 |
| 3 | 1 | 0 | 0 | 0 | 0 | 0 | 0 |
| 4 | 1 | 1 | 1 | 0 | 1 | 0 | 0 |
| 5 | 1 | 0 | 0 | 1 | 0 | 0 | 0 |
| 6 | 1 | 1 | 1 | 1 | 0 | 0 | 0 |
| 7 | 1 | 1 | 0 | 1 | 1 | 0 | 0 |
| 8 | 0 | 1 | 1 | 0 | 0 | 0 | 0 |
| 9 | 1 | 0 | 1 | 1 | 1 | 1 | 0 |
| 10 | 0 | 0 | 0 | 1 | 0 | 0 | 0 |
| Hemiarthroplasty versus total hip replacement | |||||||
| Study ID | Baker 2006 | Blomfeldt 2007 | Dorr 1986 | Macaulay 2008 | Mouzopoulos 2008 | Skinner 1989 | STARS 2006 |
| 1 | 3 | 2 | 0 | 3 | 1 | 0 | 3 |
| 2 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 3 | 1 | 1 | 0 | 0 | 0 | 0 | 1 |
| 4 | 1 | 1 | 1 | 1 | 1 | 0 | 0 |
| 5 | 0 | 1 | 0 | 0 | 0 | 0 | 0 |
| 6 | 1 | 1 | 1 | 0 | 1 | 1 | 0 |
| 7 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 8 | 0 | 0 | 0 | 0 | 0 | 0 | 0 |
| 9 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| 10 | 1 | 1 | 1 | 0 | 1 | 0 | 1 |
Effects of interventions
Cemented hemiarthroplasty versus uncemented hemiarthroplasty
This comparison was considered in the following six studies involving 899 participants.
Branfoot 2000: Cemented Thompson prosthesis versus an uncemented Thompson prosthesis (91 participants). Emery 1991: Cemented bipolar Thompson prosthesis versus an uncemented bipolar Moore prosthesis (53 participants). Harper 1994: Cemented Thompson prosthesis versus an uncemented Thompson prosthesis (137 participants). Parker 2009: Cemented Thompson prosthesis versus an uncemented Austin Moore prosthesis (400 participants). Santini 2005: Cemented bipolar prosthesis versus an uncemented bipolar prosthesis (106 participants). Sonne‐Holm 1982: Cemented Austin Moore prosthesis versus an uncemented Austin Moore prosthesis (112 participants).
Figved 2009, which compared a cemented hemiarthroplasty with an uncemented hydroxyapatite coated prosthesis, is considered separately in the section on hydroxyapatite coated prosthesis.
Where available the data from these studies are shown in Analysis 1.1 to Analysis 1.27. These are subgrouped according to the basic types of implants used in the studies. A summary of the main findings, achieving statistically significant differences between cemented and uncemented prostheses, is given below.
1.1. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 1 Length of surgery (minutes).
1.27. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 27 Failure to return home.
1. Longer operative time for cemented prosthesis (data from three trials: mean difference 7.24 minutes, 95% confidence interval (CI) 4.75 to 9.73 minutes) (seeAnalysis 1.1).
2. Reduced risk of operative fracture of the femur for the cemented prosthesis (0/291 versus 17/306 (5.6%), risk ratio (RR) 0.09, 95% CI 0.02 to 0.44) (seeAnalysis 1.2).
1.2. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 2 Operative fracture of the femur.
3. Lower reduction of a mobility score (signifying less loss of mobility) for those treated with a cemented prosthesis (RR ‐0.80, 95% CI ‐1.23 to ‐0.37) (seeAnalysis 1.23).
1.23. Analysis.
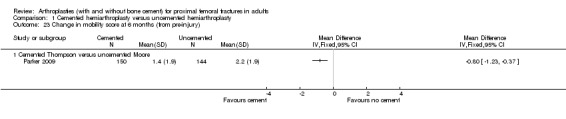
Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 23 Change in mobility score at 6 months (from pre‐injury).
4. Fewer patients with residual pain in the hip at three months (67/192 (35%) versus 84/183 (46%), RR 0.77, 95% CI 0.60 to 0.98; seeAnalysis 1.24 ), and at longer‐term follow up (seeAnalysis 1.25); also, lower pain scores (signifying less pain) for those treated with a cemented prosthesis (seeAnalysis 1.26).
1.24. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 24 Pain at early follow up (3 months).
1.25. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 25 Pain at 1 to 2 years.
1.26. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 26 Pain score at 6 months (0 to 10: worst).
In addition there was a trend to an increased operative blood loss (seeAnalysis 1.3), need for blood transfusion (seeAnalysis 1.4), occurrence of medical complications (seeAnalysis 1.15 and Analysis 1.16), a longer hospital stay (seeAnalysis 1.17), a lower proportion of patients returning to their original residence (seeAnalysis 1.27) and a greater proportion of patients who failed to regain their pre‐fracture mobility (seeAnalysis 1.22) for those treated with the uncemented prosthesis, although none of these results reached statistical significance.
1.3. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 3 Operative blood loss (ml).
1.4. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 4 Mean units blood transfused.
1.15. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 15 Medical complications.
1.16. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 16 All reported medical complications.
1.17. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 17 Length of hospital stay (days).
1.22. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 22 Failure to regain mobility.
No statistically significant difference in mortality between cemented and uncemented prosthesis was seen at any of the time intervals (See Analysis 1.18, Analysis 1.19, Analysis 1.20, and Analysis 1.21) during follow up. The results at one to three months after surgery were 49/359 (14%) versus 49/349 (14%), RR 0.98, 95% CI 0.68 to 1.41 (seeAnalysis 1.19).
1.18. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 18 Mortality before 1 month.
1.19. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 19 Mortality at 1 to 3 months.
1.20. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 20 Mortality at 1 year.
1.21. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 21 Mortality at 3 years.
Summaries, additional trial results and comments on specific aspects of the six trials are given below.
Sonne‐Holm 1982, which compared a cemented Austin Moore hemiarthroplasty with an uncemented Austin Moore hemiarthroplasty inserted via a posterior approach in 112 people, provided no operative results. Mortality was reported only at six weeks from operation: there was no difference between the two groups (seeAnalysis 1.19). Functional outcome, recorded in terms of hip movements, pain and mobility, was assessed at six weeks, and three, six and 12 months post‐operatively. While no difference between groups was reported for hip mobility at final follow up (seeAnalysis 1.22), significantly better walking was reported at three and six months for those treated with a cemented prosthesis. Pain was assessed using a scale of one (most pain) to six (no pain). Significantly more patients had residual pain at six months for the uncemented group. There was a trend to more pain for the uncemented group at six weeks, three months and one year, however the difference between groups was not statistically significant (seeAnalysis 1.24).
Harper 1994 compared a cemented Thompson prosthesis with an uncemented Thompson prosthesis in 137 people. Clinical follow up at 8 to 12 weeks after surgery was achieved in only 60 patients (29 cemented, 31 uncemented). There were inconsistencies between the full report (in a thesis) and a conference abstract: one intra‐operative death, which occurred in the cemented group, was reported in the thesis, but two deaths in the theatre recovery room were reported in the abstract, which also reported a study population of 140 patients rather than 137. Differences between the two groups in mortality at three months and one year (seeAnalysis 1.19 and Analysis 1.20) were not statistically significant. Follow‐up assessment of pain at eight to 12 weeks after surgery was for fewer than 60 participants (exact number not stated). Pain was reported as being significantly reduced (P < 0.001) in participants with a cemented prosthesis.
Branfoot 2000 also compared a cemented Thompson prosthesis with an uncemented Thompson prosthesis in 91 patients. The only operative complications reported were one dislocation and one fracture of the femur, both in the uncemented group. There was no significant difference in mortality, assessed at a mean follow up of 16 months (seeAnalysis 1.20). Pain was graded on a scale of zero (no pain) to three (night pain). The mean pain scores for the 70 surviving participants (0.42 versus 0.24) were reported to be not statistically significantly different. Other assessments using the Harris Hip Score were for limp, mobility, use of walking aids, and activities: none of the Harris Hip Scores showed any statistical difference between the two groups.
Emery 1991 compared a cemented bipolar on a Thompson stem with an uncemented bipolar Thompson stem in 53 patients. Neither of the differences in length of surgery (seeAnalysis 1.1) nor operative blood loss (seeAnalysis 1.3) were statistically significant. There was no significant difference between the two groups in length of hospital stay (seeAnalysis 1.17). Mortality was reported at two weeks, three months and 17 months, with no difference between groups (seeAnalysis 1.18, Analysis 1.19 and Analysis 1.20). Pain at 17 months (mean follow up) was significantly less in the cemented group (seeAnalysis 1.25). In addition, significantly fewer people in this group were more dependent on walking aids (seeAnalysis 1.22). Similar numbers were unable to return to their pre‐fracture residence (seeAnalysis 1.27).
Santini 2005 also compared a cemented bipolar hemiarthroplasty with an uncemented bipolar hemiarthroplasty in 106 patients. Length of surgery was significantly longer in the cemented group (seeAnalysis 1.1). There was no significant difference in units of blood transfused (seeAnalysis 1.4). Surgical complications reported were one dislocation in the cemented group and two cases of operative fracture of the femur in the uncemented group. There was no statistically significant difference between the two groups in deep wound infection (seeAnalysis 1.10) or specific medical complications (seeAnalysis 1.15). The difference between the two groups in the mean costs (3090 Euros versus 4008 Euros) was entirely due to the difference in cost of the prostheses. One‐year mortality was similar between the two groups (seeAnalysis 1.20). Function was assessed using a scoring system for walking, personal activities, daily activities and living conditions. None of the scores, either overall or for the separate items, showed any statistically significant difference between the two groups.
1.10. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 10 Deep sepsis.
Parker 2009 compared a cemented Thompson hemiarthroplasty with an uncemented Austin Moore hemiarthroplasty for 400 patients. Length of surgery was significantly longer in the cemented group (seeAnalysis 1.1). There was no significant difference in use of blood transfusion between groups (seeAnalysis 1.4 and Analysis 1.5). Surgical complications reported were two dislocations in the cemented group and one in the uncemented group. In addition there were 14 cases of operative fracture of the femur in the uncemented group and two patients in the cemented group had cement retained in the acetabulum. There was a tendency to reduced medical complications in the cemented group, although the difference was not statistically significant (seeAnalysis 1.15 and Analysis 1.16). Hospital stay was on average four days shorter with the cemented prosthesis (seeAnalysis 1.17). This difference was not statistically significant using the fixed effect method but using a non‐parametric test (Mann‐Whitney U test) in the published article the difference was reported as being statistically significant (difference in total hospital stay 20.3 days versus 24.7 days, P value = 0.004). There was no difference in mortality between groups at any time period after surgery (seeAnalysis 1.18 to Analysis 1.21). Residual pain was assessed by a blinded observer using a visual analogue scale at six weeks after discharge and using a six point pain scale at three, six, nine, 12, 24, 36, 48 and 60 months after surgery. Pain was consistently less for the cemented prosthesis with the result being statistically significant for all time periods up to and including two years from surgery. Regain of mobility was also assessed by the blinded observer using a nine point mobility score and showed a statistically significant better regain of mobility at six, nine and 12 months after surgery. Also reported was the regain of 16 activities of daily living. There was a tendency for these to be regained quicker in the cemented group but only in three activities did this reach statistical significance. These were the ability to bend down and pick up an object from the floor (mean 15.7 weeks versus 23.6 weeks, P value reported as 0.0045), the ability to get in and out of a car (mean 9.1 weeks versus 15.9 weeks (reported P = 0.006) and ability to go shopping without assistance (mean 16.6 weeks versus 27.0 weeks, reported P = 0.0002).
1.5. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 5 Number of patients transfused.
Hemiarthroplasty (with or without cement) versus hydroxyapatite coated hemiarthroplasty
This comparison was considered in two studies, one of which used a cemented hemiarthroplasty and the other an uncemented hemiarthroplasty to compare with a hydroxyapatite coated hemiarthroplasty. Data from these two very different trials are not pooled.
Figved 2009 compared a cemented bipolar hemiarthroplasty with an uncemented hydroxyapatite coated bipolar hemiarthroplasty for 220 patients. Outcomes reported included the length of surgery, operative blood loss and total blood loss all of which were less for the hydroxyapatite coated implant (seeAnalysis 2.1 to Analysis 2.3). Other reported outcomes were requirement for blood transfusion, dislocation, operative and later fracture of the femur, reoperation, wound sepsis, medical complications and length of hospital stay (seeAnalysis 2.4 to Analysis 2.15). None of the other reported outcomes were shown to have any statistically significant difference between groups. One intra‐operative cardiac arrest was reported in the cemented group with the patient dying within 72 hours and another patient had hypotension on cement insertion and died 24 hours later. Mortality was reported at 7, 30, 90 days and one and two years. There was no statistically significant difference between groups, but a tendency to a lower mortality at one year from surgery for the cemented group (see Analysis 2.16). Other outcomes reported by a blinded assessor, at three and 12 months, were the number of patients with residual pain requiring medication, number of patients who still needed to use walking aids, Harris hip score, Barthel index and a health related quality of life score (ED‐5D). None of these outcomes showed any statistical significance difference between the two groups (seeAnalysis 2.17 to Analysis 2.22).
2.1. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 1 Length of surgery (minutes).
2.3. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 3 Total blood loss (ml).
2.4. Analysis.
Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 4 Number of patients transfused.
2.15. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 15 Length of hospital stay (days).
2.16. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 16 Mortality.
2.17. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 17 Residual pain.
2.22. Analysis.
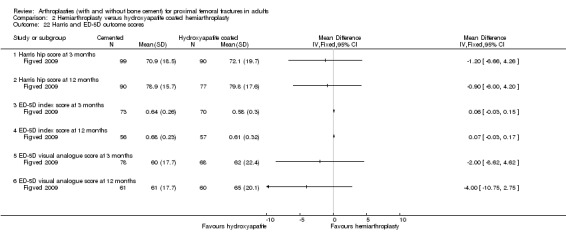
Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 22 Harris and ED‐5D outcome scores.
Livesley 1993 compared an uncemented Moore stem bipolar prosthesis with a hydroxyapatite coated Furlong stem bipolar prosthesis for 82 patients The various outcome measures recorded are presented in the analyses. None of the differences between the two groups in the outcomes shown in the analyses reached statistical significance. There were, however, seven cases of operative fracture of the femur in the Furlong prosthesis group compared with none in the Moore group, a difference that approaches statistical significance (seeAnalysis 2.5). The single reoperation in the Moore group was for persistent thigh pain. The four reoperations in the Furlong group were for dislocation (twice in one patient), deep infection and fracture at the tip of the prosthesis. At follow up, 30 of the Moore prosthesis group were reported to have radiographic evidence of loosening and a further five to have acetabular wear. Conversely, only one Furlong case was reported to have acetabular wear with no radiographic loosening of the prosthesis. There was no statistically significant difference between the two groups in mortality (seeAnalysis 2.16) or the number back at home or able to shop at one year (seeAnalysis 2.19 and Analysis 2.20). The study reported without data, that at one year from injury fewer patients in the Furlong group used walking aids (reported P = 0.0001). Some of the other outcomes assessed using a functional assessment scale were reported to show a tendency for better results in the Furlong group; these were rest pain (reported P = 0.05), pain on rising from the chair (P = 0.003), activity pain (P = 0.005), ability to climb stairs (P = 0.05) and the patient's opinion of their hip (P = 0.03). Factors showing a non statistically significant difference were assistance in walking (P = 0.08) and walking distance (P = 0.08).
2.5. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 5 Operative fracture of femur.
2.19. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 19 Failure to return back home at 1 year.
2.20. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 20 Failure to be able to shop at 1 year.
Unipolar hemiarthroplasty versus unipolar hemiarthroplasty (differing head materials)
The only identified study (Stock 1997) compared a Thompson prosthesis with a ceramic head with a conventional metal Thompson prosthesis in 69 patients. It was not mentioned whether cement was used. The only outcome measure reported was the Harris Hip Score for 56 of the participants. The time interval from surgery to assessment was not stated. The mean hip score was 74.5 in the ceramic group and 69.3 in the conventional, a difference that was reported as not being statistically significant (P = 0.177).
Unipolar hemiarthroplasty versus bipolar hemiarthroplasty
This was considered in seven studies (Calder 1996; Cornell 1988; Davison 2001; Malhotra 1995; Raia 2003; Rosen 1992; Van Thiel 1988). Three studies (Malhotra 1995; Rosen 1992; Van Thiel 1988) compared an Austin Moore hemiarthroplasty with a Bateman bipolar hemiarthroplasty. Only Malhotra 1995 stated that a posterior approach was used and all implants were used without cement. The other two studies which looked at this comparison did not state the surgical approach or if cement was used. Cornell 1988 and Raia 2003 both compared a cemented unipolar hemiarthroplasty with a cemented bipolar hemiarthroplasty. Calder 1996 compared a cemented unipolar Thompson prosthesis with a cemented Monk bipolar prosthesis for patients aged over 80 years and Davison 2001 compared the same implants in those aged 65 to 79 years.
Summation of the data available from these studies is given in Analysis 3.1 to Analysis 3.12. These indicate no statistically significant differences between the two types of prostheses for the outcomes of dislocation, acetabular erosion, deep wound sepsis, reoperations, deep vein thrombosis, mortality or mobility.
3.1. Analysis.

Comparison 3 Unipolar hemiarthroplasty versus bipolar hemiarthroplasty, Outcome 1 Dislocation.
3.12. Analysis.

Comparison 3 Unipolar hemiarthroplasty versus bipolar hemiarthroplasty, Outcome 12 Failure to regain mobility.
Summaries and comments on specific aspects of the seven trials are given below.
The 68 participants of Malhotra 1995 were much younger (mean age 66 years) and more likely to be male (56%) than in the other studies. Length of surgery was equal in both groups. The difference in mean blood loss (350 ml versus 400 ml) was reported as not statistically significant. Dislocation occurred in one participant in each group; however, the dislocation in the bipolar group could not be reduced closed and required an open reduction. Both cases of deep wound infection, which occurred in the unipolar group, were treated by a Girdlestone arthroplasty. In addition, two participants of the unipolar group were later revised to a THR for acetabular protrusion. The mean hospital stay was similar in both groups (18.1 versus 17.2 days). No deaths were reported during the two year (average) follow‐up period. Malhotra 1995 also stated, without data, that patients treated with the bipolar mobilised more quickly and had less post‐operative pain. Follow‐up assessment was by the use of a scoring system, which included pain, walking ability and range of hip movement. A greater proportion of 'excellent results' were noted in the bipolar group (24/32 (75%) versus 17/36 (47%)).
Rosen 1992, which included 96 participants, was only reported as a conference abstract with no data. The length of surgery and operative blood loss were stated to be significantly greater for the bipolar hemiarthroplasty. Reoperations were equal in both groups but significantly more post‐operative complications occurred in the bipolar group. Mortality was reported as not significantly different between groups. Of only 27 participants followed up, pain was reported to be slightly less in the bipolar group and mobility better in the unipolar group.
Van Thiel 1988, which included 93 participants, was only reported as a conference abstract with no data. The only clinical outcome measure was the Harris Hip Score, which was reported as showing no significant difference between groups. Radiographic parameters of loosening, peri‐articular ossification and protrusion acetabuli also showed no significant difference between groups.
Cornell 1988 compared a cemented modular unipolar hemiarthroplasty with a cemented bipolar hemiarthroplasty in 48 patients. All the stems used were identical and all the prostheses were inserted via a posterior approach. Follow up was only six months. Length of surgery and operative blood loss were stated without data as showing no significant difference between the two groups. Dislocation occurred in one person in each group. The only other complication reported was one case of deep vein thrombosis in the bipolar group. Mean length of hospital stay was reported as not being significantly shorter in the unipolar group (10.3 versus 13.4 days). There was no significant difference in mortality at six months (1/15 versus 2/33; RR 1.10, 95% CI 0.11 to 11.21) (seeAnalysis 3.10). Overall hip function was assessed using the Johansen Hip Scoring system, which showed no difference between the two groups (64.9 versus 63.2 points). However, hip movements were stated as being "greater" after the bipolar prosthesis (flexion: 110 versus 104 degrees; rotation: 50 versus 36.6 degrees; abduction: 38 versus 22 degrees). Walking speed was also reported to be better for the bipolar (2.67 versus 1.93 feet/sec, reported P < 0.03) as was the 'get up and go test' (27.3 versus 33.1 seconds), although the last difference was not statistically significant.
3.10. Analysis.

Comparison 3 Unipolar hemiarthroplasty versus bipolar hemiarthroplasty, Outcome 10 Mortality at 6 months.
Raia 2003 compared a cemented unipolar hemiarthroplasty with a cemented bipolar in 115 people. All prostheses stems were identical and inserted by a posterolateral approach. Follow up was for one year. Mean operative blood loss was similar between the two groups (252 ml versus 237 ml) and there was no statistically significant difference between the two groups in the numbers of participants receiving blood transfusion (15/55 versus 18/60; analysis not shown). One person in the unipolar group had three dislocations and one person in the bipolar group had a single dislocation. All dislocations were treated by closed reduction. The one case of deep sepsis, reported in the unipolar group, required debridement and later implant removal. No other surgical complications were reported. Length of stay on the orthopaedic ward was similar in the two groups (mean 5.5 days versus 5.2 days). Mortality and failure to regain mobility at one year were similar in the two groups as shown in Analysis 3.11 and Analysis 3.12 respectively. Seventy‐eight participants were assessed at one year from surgery using the Short Form 36 questionnaire and the Musculoskeletal Functional Assessment score. No significant differences or trends were noted between the two groups.
3.11. Analysis.

Comparison 3 Unipolar hemiarthroplasty versus bipolar hemiarthroplasty, Outcome 11 Mortality at 1 to 2 years.
Calder 1996 compared a Thompson prosthesis with a Monk bipolar prosthesis in 250 patients aged over 80 years of age. All prostheses were cemented in place, had identical stems and were inserted via an antero‐lateral Hardinge approach. Dislocation occurred twice in the unipolar group and once in the bipolar group (seeAnalysis 3.1). Radiographic evidence of acetabular erosion was seen in three cases, all with unipolar prosthesis; none of these had needed revision surgery by the time of final follow up (seeAnalysis 3.3 and Analysis 3.4). Deep wound infection occurred in five unipolar group participants and four bipolar group participants (seeAnalysis 3.5). The length of hospital stay was similar in the two groups (medians: 18 versus 17 days). There was no statistically significant difference between the two groups in mortality at six months (seeAnalysis 3.10) or one year (seeAnalysis 3.11). Other results for this trial were reported as percentages without actual numbers. The proportions of patients with no or mild pain at follow up were reported similar in the two groups (53% versus 55%), as were the proportions returning to their pre‐operative place of residence (41% versus 42%). Calder 1996 reported that, after adjustment for confounding factors, the proportion of patients returning to their pre‐injury subjective status was significantly greater in the unipolar group (P = 0.041). This statement conflicted with the proportions given in the text (29% versus 40%). There was no statistical difference between the two groups in the proportions of patients who reported a limp (23% versus 25%) or who were satisfied with their operation (67% versus 63%). Similarly, there was no significant difference in the mean Harris Hip Scores of the two groups (70 versus 72). The Nottingham Health Profile, which was conducted on a subgroup of 128 participants, showed no statistically significant difference between implant groups.
3.3. Analysis.
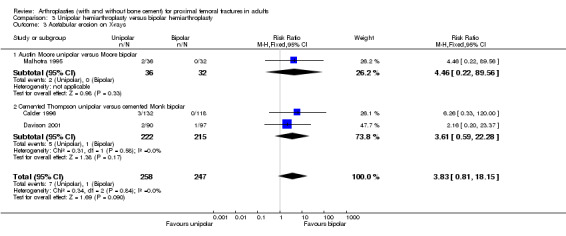
Comparison 3 Unipolar hemiarthroplasty versus bipolar hemiarthroplasty, Outcome 3 Acetabular erosion on X‐rays.
3.4. Analysis.

Comparison 3 Unipolar hemiarthroplasty versus bipolar hemiarthroplasty, Outcome 4 Acetabular erosion requiring revision arthroplasty.
3.5. Analysis.

Comparison 3 Unipolar hemiarthroplasty versus bipolar hemiarthroplasty, Outcome 5 Deep sepsis.
Davison 2001 compared a cemented unipolar Thompson prosthesis with a cemented Monk bipolar prosthesis involved 187 participants aged between 65 and 79 years, with a minimum of follow up of two years. Dislocation occurred twice in one unipolar group participant and in two participants of the bipolar group (seeAnalysis 3.1). Open reduction was required for one of the bipolar dislocations (seeAnalysis 3.2). The sole deep wound infection occurred in one patient in the bipolar group (seeAnalysis 3.5). Acetabular erosion requiring revision was required twice in the unipolar group and once in the bipolar group (seeAnalysis 3.4). Other reoperations, all in the bipolar group, were one Girdlestone for sepsis and one biopsy and excision of heterotopic ossification. Other outcomes, which were reported as showing no significant difference between the two groups during the follow‐up period of up to five years, were the mean Harris Hip Score, Barthel Index, patient satisfaction, return to pre‐injury state and mortality. The Nottingham Health Profile, assessed in 73 participants, was reported as showing a non‐statistically significant trend to lower physical mobility for the unipolar group.
3.2. Analysis.

Comparison 3 Unipolar hemiarthroplasty versus bipolar hemiarthroplasty, Outcome 2 Dislocation requiring open reduction.
Hemiarthroplasty (various types) versus total hip replacement (THR)
The seven trials testing this comparison are grouped according to the use of cement. Dorr 1986, which compared cemented hemiarthroplasty or uncemented hemiarthroplasty versusTHR is grouped together with the two trials that did not specify whether cement was used. The studies were as follows:
Cemented implants
Baker 2006: Cemented unipolar hemiarthroplasty versus a cemented THR (81 participants). Blomfeldt 2007: Cemented Exeter stem bipolar hemiarthroplasty versus a cemented Exeter THR (120 participants). STARS 2006: Cemented bipolar hemiarthroplasty with a cemented THR (138 participants).
Cement use not specified or a mixed population
Dorr 1986: Cemented (39) or uncemented (13) bipolar hemiarthroplasty versus a cemented THR (37) (89 participants). Mouzopoulos 2008: Merete hemiarthroplasty versus THR (86 participants). Macaulay 2008: Different hemiarthroplasties versus various THR's (40 participants).
Uncemented hemiarthroplasties
Skinner 1989: Uncemented Austin Moore hemiarthroplasty versus a cemented THR (180 participants).
Where possible, results are presented in the analyses (seeAnalysis 4.1 to Analysis 4.20). These are subgrouped according to the basic types of implants used in the studies. A summary of the main findings, achieving statistically significant differences between hemiarthroplasty and THR, is given below.
4.1. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 1 Length of surgery (minutes).
4.20. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 20 Final outcomes for survivors (continuous outcomes for which a higher score is advantageous).
1. Surgical time was significantly less for the hemiarthroplasty (MD ‐18.53 minutes, 95% CI ‐23.74 to ‐13.32 minutes) (seeAnalysis 4.1).
2. Increased risk of dislocation for THR (15/343 (4.3%) versus 24/30 (7.9%); RR 0.53, 95% CI 0.29 to 0.97) (seeAnalysis 4.4).
4.4. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 4 Dislocation.
3. More 'minor' reoperations for the THR group (13/274 (4.7%) versus 22/236 (9.3%); RR 0.50, 95% CI 0.26 to 0.95) (seeAnalysis 4.10).
4.10. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 10 Reoperations: minor.
4. More 'major' reoperations for the hemiarthroplasty group (25/317 (7.9%) versus 9/279 (3.2%); RR 2.22, 95% CI 1.09 to 4.51) (seeAnalysis 4.11). This difference was mainly accounted for by the increased reoperation rate for the uncemented hemiarthroplasties.
4.11. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 11 Reoperations: major.
5. Less residual pain at one year for THR in comparison with uncemented hemiarthroplasties (seeAnalysis 4.16).
4.16. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 16 Residual pain at 1 year.
6. A number of the final functional outcomes scores showed a statistically significant difference in favour of better function for those treated with the THR. These included the Oxford hip score, Harris hip score, Barthel score and EuroQol score (seeAnalysis 4.19 and Analysis 4.20).
4.19. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 19 Final outcomes for survivors (continuous outcomes for which a lower score is advantageous).
There were no notable differences between the two groups in any of the other outcomes, including mortality.
Summaries, additional trial results and comments on specific aspects of the seven trials are given below.
Cemented implants
STARS 2006 compared a cemented bipolar hemiarthroplasty with a cemented total hip replacement in 138 patients. The mean surgical time was significantly less in those treated with a cemented hemiarthroplasty (mean difference ‐21.20 minutes, 95% CI ‐29.09 to ‐13.31 minutes) (seeAnalysis 4.1). At two‐year follow up there were no significant differences between the two groups in the number of dislocations (2 versus 3) or of superficial (2 versus 2) or deep (1 versus 1) wound infections (seeAnalysis 4.4). There were also no significant differences between the two groups in the number of reoperations (5/69 versus 6/69; RR 0.83, 95% CI 0.27 to 2.60) (seeAnalysis 4.12) or medical complications (seeAnalysis 4.13). The mean cost for those treated with a hemiarthroplasty was £15,263 compared with a mean cost of £12,253 for those treated with a THR (reported 95% confidence interval for the difference was £1400 to £7420). The study reported no statistically significant differences between the two groups in mortality (seeAnalysis 4.15) or in residual pain or discomfort at 12 months (seeAnalysis 4.16). Patient‐reported outcomes were obtained at four months, and one and two years using a Hip Rating Questionnaire (modified Johansen hip scoring system), which assessed impact of the hip problem (global), pain, walking, and function, and the EuroQol (EQ‐5D) questionnaire to assess general health status. The P values for these are summarised in Table 3. Those outcomes for walking at four months, one and two years, and function and overall hip‐related functioning at two years were better for patients allocated a THR (seeAnalysis 4.20).
4.12. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 12 Reoperations: any.
4.13. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 13 Medical complications.
4.15. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 15 Mortality.
3. STARS 2006: reported P values (unadjusted data).
| Outcome | 4 months | 1 year | 2 year |
| Pain | 0.90 | 0.47 | 0.53 |
| Walking | 0.06 | 0.05 | 0.02 |
| Function | 0.26 | 0.32 | 0.03 |
| Health related quality of life | 0.20 | 0.42 | 0.05 |
| EuroQol (EQ‐5D) general health outcome measure | 0.10 | 0.43 | 0.005 |
Baker 2006 compared a cemented unipolar hemiarthroplasty with a cemented total hip replacement in 81 patients. The mean surgical time was less for hemiarthroplasty (78 versus 93 minutes, reported P = < 0.001). There were three dislocations in the THR group; one of these was recurrent (seeAnalysis 4.10 to Analysis 4.12). Three patients in the hemiarthroplasty group required revision surgery: two for acetabular wear and one for fracture around the prosthesis. One patient of the THR group required revision for loosening and one wound required debridement in the same group. Radiographic changes of acetabular wear were also reported for 21 out of 32 cases in the hemiarthroplasty group. Medical complications were reported for 15 patients as detailed in Analysis 4.13. Mortality at three years is given in Analysis 4.15, with no statistically significant difference between groups. Functional outcome, based on the Oxford hip score, significantly favoured the THR (mean scores 22.3 versus 18.8; reported P = 0.033, seeAnalysis 4.19). The short form 36 physical and mental scores were reported as not being statistically significantly different between the two groups (38.1 versus 40.5, P = 0.36; and 55.3 versus 52.0, P = 0.35). The self reported walking distance was significantly shorter (1.9 km versus 3.6 km; reported P = 0.039) (seeAnalysis 4.20).
Blomfeldt 2007 compared a cemented bipolar hemiarthroplasty with a cemented total hip replacement in 120 patients. The mean surgical time was less for hemiarthroplasty (78 versus 102 minutes, reported P = < 0.001). Surgical complications were one later fracture below the implant in the THR group, which was treated by plating of the femur, and one deep wound infection in the THR group requiring three debridements and then closure of the wound. There were also two superficial wound infections in each group. Medical complications reported in for this study are presented in Analysis 4.13. There was no significant difference between the two groups in mortality at one year (seeAnalysis 4.15). Functional outcome, based on the Harris hip score, was statistically significant better for the THR (mean scores 79.4 versus 87.2; reported P = < 0.001). The components of pain and function of the Harris hip score both favoured THR, whilst there was no difference between implants for deformity or range of movement. The EuroQol (EQ‐5D) assessment was reported to favour THR (mean scores: 0.63 versus 0.68), but the difference the two groups did not reach statistical significance (reported P = 0.636).
Cement use not specified or a mixed population
Dorr 1986 compared bipolar hemiarthroplasty, either cemented (37 cases) or uncemented (13 cases), with a cemented total hip replacement (39 cases). Surgical complications were two dislocations in the hemiarthroplasty group, both involved a cemented arthroplasty, and seven dislocations in the THR group. No wound infections were reported. Three participants with a cemented hemiarthroplasty required revision for removal of cement in the acetabulum, heterotopic ossification and repeated dislocations. One participant with an uncemented hemiarthroplasty group required revision for recurrent dislocation and loosening. Two of the THR cases required revision for recurrent dislocation and loosening. No difference in the length of hospital stay was noted. Trial participants were assessed at three, 12 and 24 months post‐operatively. Mortality was only reported as 'no difference between groups'. Mean pain scores on a scale of zero to six (with the most severe pain score of 0) were 5.4, 5.2 and 5.1 for participants with a cemented hemiarthroplasty, 3.7, 3.6 and 3.0 for participants with an uncemented hemiarthroplasty and 4.9, 5.5 and 5.5 for participants with a THR.This difference was reported to be statistically significant for participants with an uncemented hemiarthroplasty. Mobility was assessed on a scale of 0 to 6 (with worst mobility at 0). Mean mobility at three, 12 and 24 months after surgery was 4.0, 4.2 and 4.0 for participants with a cemented hemiarthroplasty and 3.7, 3.6 and 3.0 for those with an uncemented hemiarthroplasty versus 4.1, 4.1 and 5.5 for the those with a THR. The reduced mobility for participants with an uncemented hemiarthroplasty was reported as being statistically significant. More participants in the hemiarthroplasty group used walking aids compared with the THR group (15/50 (30%) versus 7/39 (18%)). However, the excess in the use of walking aids was in those treated with an uncemented hemiarthroplasty (9/13 (69%)) compared with those with a cemented hemiarthroplasty (6/37 (16%)). Gait analysis of walking velocity and single limb stance showed no difference between the cemented prostheses but a greater degree of impaired gait for participants with an uncemented hemiarthroplasty.
Mouzopoulos 2008 compared a Merete hemiarthroplasty with a THR in 86 patients. There was only limited reporting of outcomes with five revision operations reported in the hemiarthroplasty group versus one in the THR group. Mortality at one and four years showed no difference between groups (seeAnalysis 4.15). The walking speed was reported without data to be faster for those treated with a hemiarthroplasty (P < 0.05). Both the Harris hip score and the Barthel score favoured the THR at both one and four years from injury (seeAnalysis 4.20).
Macaulay 2008 compared a hemiarthroplasty with a THR in 40 patients. There was only limited data available because of incomplete reporting of denominators. Surgical complications were one dislocation in the THR group and one wound infection in the hemiarthroplasty group. There was no difference in mortality between groups (seeAnalysis 4.15). Final outcomes were assessed using the Harris hip score, timed get up and go score, WOMAC score, and SF‐36 scale at 12 and 24 months. Numbers of patients assessed at these timed intervals were not given. The only differences that were reported to be statistically significant were more bodily pain for the hemiarthroplasty group at 12 months (P value reported as 0.02) and 24 months (P = 0.03) and improved function on the WOMAC score at 24 months (P = 0.03).
Uncemented hemiarthroplasties
Skinner 1989 compared an uncemented Moore hemiarthroplasty with a cemented Howse THR in 180 patients. All prostheses were inserted with a posterior‐lateral approach. The main implant‐related complication was dislocation in 11 hemiarthroplasties and 10 THRs. The only case of deep sepsis occurred in a THR patient. Revision surgery was required in 13 hemiarthroplasties for recurrent dislocation, loosening, fracture around the prosthesis or ectopic calcification. Three of the THRs were revised for recurrent dislocation. There was no statistically significant difference between the two groups in mortality at 2 or 12 months follow up (seeAnalysis 4.15). Pain was more prevalent in those treated with hemiarthroplasty. Mobility, graded on a scale of one to four, was reported to be significantly better for the THR. However, there was no significant difference in the numbers failing to regain their former mobility (seeAnalysis 4.17). A later conference abstract (Ravikumar 1998), reported a 13‐year follow up. The mean survival was 3.17 years for the hemiarthroplasty group and 4.27 years for the THR group, with no significant difference found. The number of surviving patients assessed was not stated but would be small given the approximately 85% overall mortality. The mean Harris Hip Score for survivors treated by THR was significantly better than that for those treated by hemiarthroplasty (mean score 80 versus 55). Pain was also reported to be less for THR group survivors, and this group was also stated to be more mobile. The long‐term revision rate was significantly higher for those treated with a hemiarthroplasty (13/100 (13.0%) versus 3/80 (3.8%); RR 3.47, 95% CI 1.02 to 11.75 (seeAnalysis 4.11).
4.17. Analysis.
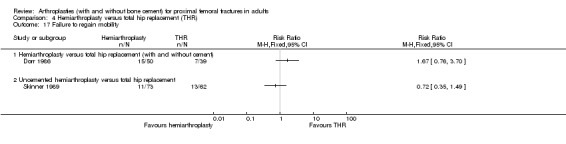
Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 17 Failure to regain mobility.
Discussion
Many of the trial reports indicated a poor level of methodological rigour, in particular regarding concealment of allocation, assessor blinding and intention‐to‐treat analysis. Inadequate trial methodology may allow a patient being randomised to a more extensive surgical procedure such as use of cement or a THR, then being excluded when they are felt to be unfit for the more extensive procedure at the time of surgery. Such events will bias results in favour of the more extensive procedure. Furthermore, many of the studies involved small numbers of patients and had limitations, including inadequate length of follow up, in their assessment and reporting of outcome. In addition some of the studies were only reported as conference abstracts with a failure to provide additional information. However in some cases this apparent poor methodology may reflect poor reporting of the trial rather than poor trial methodology. The variation in characteristics of the prostheses used in the individual trials hampers interpretation of their findings. For instance, in the comparison of cemented versus uncemented prostheses, the two prostheses under test sometimes differed in other important ways as well as in the use of cement when comparing a hemiarthroplasty with THR. For some of the findings, it may not be possible to attribute an outcome to the intervention under test (cement) or to the other differences in the implants. While remaining a source of heterogeneity, pooling of data from several trials with this comparison should help to address the basic question (whether to use cement of not). The presentation of the results of this review, which subgroups the different combinations of implants under test, also should help to get the answers to the basic questions and explore the differences. So far, the limited numbers of patients studied preclude the drawing of definite conclusions for many of the comparisons under review.
Cemented versus uncemented prostheses
The six included studies investigating this comparison involved a total of 899 patients. The largest study was that of Parker 2009 with 400 participants. This study, which had the strongest methodology including blinded assessment of outcome, found strongly in favour for the use of cement. The study of Harper 1994 with 137 patients, was of poor methodology with very limited reporting of outcomes and inadequate follow up of patients: therefore no conclusions can be made from this study. Santini 2005 had an inadequate method or randomisation and limited reporting of outcomes. Sonne‐Holm 1982 excluded the results of 37 patients who had either died, moved away or had the wrong prosthesis implanted. Branfoot 2000 had limited reporting of results for 91 patients. Despite the methodology limitations of many of the studies, most reported similar findings.
Most studies reported increased pain for uncemented prostheses (Emery 1991; Harper 1994; Parker 2009; Sonne‐Holm 1982). This is particularly relevant in Parker 2009 and Sonne‐Holm 1982 both of which used blinded outcome assessors. Branfoot 2000 also reported less pain for the cemented group although the results did not reach statistical significance. Three studies reported better mobility in the cemented group ( Emery 1991; Parker 2009; Sonne‐Holm 1982), however Santini 2005 found no difference in walking ability. Regarding hospital stay and post‐operative medical complications, there was a tendency for these to be less with the cemented prosthesis, although the differences were not statistically significant. Very few major adverse effects of cement were reported. There was one intra‐operative death from 77 patients treated with a cemented prosthesis in Harper 1994 and one intra‐operative cardiac arrest from 200 cemented prosthesis in Parker 2009. The other studies did not report any intra‐operative adverse events associated with bone cement. No difference in mortality was reported between groups.
It is notable that in two quasi‐randomised studies (Dorr 1986; Skinner 1989) comparing hemiarthroplasty with a cemented THR, some (Dorr 1986) or all (Skinner 1989) patients in the hemiarthroplasty group had an uncemented implant. Both studies found superior results for the THR but it is not possible to determine if these improved outcomes were due to the use of cement or the differences in type of implant.
Potential biases in the results of the studies may occur if frailer participants are omitted from the cemented group. This is more likely to occur if the method of randomisation was inadequate, which is particularly relevant for Harper 1994 who used an open method of randomisation.
In summary, there is reasonable evidence from randomised studies to date that cementing a prosthesis in place will reduce the amount of residual pain in the hip and also allow better regain of mobility. There is no evidence of any significant adverse effects of cement on mortality or other complications encountered apart from the very occasional adverse reaction to cement during surgery.
Hemiarthroplasties (with and without cement) versus hydroxyapatite hemiarthroplasty
Only one study to date has addressed the issue as to whether a modern uncemented hydroxyapatite coated arthroplasty will produce comparable results to that of a cemented arthroplasty (Figved 2009). This study of 220 participants essentially found comparable outcomes between the two groups for all the outcome measures studied. However further studies with increased patient numbers with a longer follow up are required before definite conclusions regarding similar outcome measures can be made.
Livesley 1993 compared two very different prostheses: a standard Austin Moore with its single size fenestrated stem and collar versus a solid collarless hydroxyapatite coated Furlong stem of different sizes. Both prostheses were used with a bipolar head. Any difference in the results, for the two implants may be due to either the hydroxyapatite, the collar, the stem shape or the availability of different stem sizes. The results from this small quasi‐randomised trial showed a tendency to better final outcome results with the Furlong prosthesis. However, the limitations in the available evidence from this small study means that definite conclusion on the relative effects of the two implants cannot be made.
Unipolar hemiarthroplasty versus unipolar hemiarthroplasty (differing head materials)
The one trial included for this comparison (Stock 1997) was reported only as a conference abstract without any details of the trial methodology and only one outcome measure, the Harris Hip Score. Thus, no conclusions can be drawn on the relative effects of a ceramic and metal‐headed Thompson prosthesis.
Unipolar hemiarthroplasty versus bipolar hemiarthroplasty
The limited data available from the seven studies testing this comparison showed no statistically significant differences between the groups. Some significant differences were reported for specific outcomes in individual studies, but confirmatory data were rarely provided. Notably, two of the three studies that compared an Austin Moore hemiarthroplasty with a Bateman bipolar hemiarthroplasty (Rosen 1992; Van Thiel 1988) were only reported as conference abstracts and provided minimal information. The third study (Malhotra 1995) gave more information but the characteristics of the trial participants were markedly different to those of the general population of people with hip fractures. The mean age of the participants was 66 years (as opposed to 75 to 80 years for most other studies), 56% were male (as opposed to 25% to 35% in other studies), and there were no deaths by two years follow up. This as well as the small sample size (68 participants), means that the results of this study, which tended to favour bipolar hemiarthroplasty, may not anyway apply to the general hip fracture population.
Raia 2003 compared cemented unipolar and cemented bipolar arthroplasties in 115 people followed up for one year. No differences in outcomes were noted between groups. Cornell 1988 undertook a similar study with only 48 participants. There appeared to be a greater range of movement and improved gait for those treated with the bipolar prosthesis but there was no difference in the hip scores between the two groups. Because the number of participants in this study was small, no definite conclusions can be made. The larger study of Calder 1996 for those aged 80 years and over with a more complete reporting of outcomes showed no advantages for the bipolar prosthesis in this age group. In the study involving those aged less than 80 years of age (Davison 2001), again no advantage was demonstrated for the bipolar implant.
Hemiarthroplasty versus total hip replacement
Three studies (Baker 2006; Blomfeldt 2007; STARS 2006) involving 339 patients compared a cemented hemiarthroplasty with a cemented THR. These found little differences between prostheses except for a definite trend towards better long term hip function and mobility in the THR group. Two further (Macaulay 2008; Mouzopoulos 2008) involving 126 patients did not specify the use of cement although it seems likely that most implants were cemented in place. Both studies again reported a tendency to better functional outcomes with a THR. One dated and flawed study (Dorr 1986), in which three‐quarters of the patients in the hemiarthroplasty group had a cemented prosthesis, failed to find any differences between the prostheses.
Two quasi‐randomised studies compared an uncemented hemiarthroplasty with a cemented THR (Dorr 1986; Skinner 1989). In Dorr 1986, 13 of the 52 participants in the hemiarthroplasty group had an uncemented prosthesis. Both studies reported increased pain for the uncemented prosthesis. Skinner 1989, which included 180 participants, reported better mobility and a lower long‐term revision rate for those treated with a THR. It is not possible to be certain if the improved results in the THR group were due to the use of cement or the acetabular replacement as part of the THR.
Authors' conclusions
Implications for practice.
There is reasonable evidence to indicate that cementing a prosthesis in place for a hemiarthroplasty will reduce pain in the limb and result in improved mobility. It is possible that these differences may not exist for a hydroxyapatite coated uncemented prosthesis. There is limited evidence that a THR results in less residual pain and better function than an hemiarthroplasty. There is inadequate evidence to support or refute the use of a bipolar prosthesis.
Implications for research.
Further well conducted randomised trials with full reporting of outcomes, blinding of outcome assessors, adequate length of follow up and correct methodology are required to determine the optimum prosthesis for a hip fracture. Of particularly relevance is THR versus hemiarthroplasty and also bipolar versus unipolar hemiarthroplasty. Trials of older hemiarthroplasties such as the Austin Moore or Thompson with or without cement are of less relevance now, but there is still a place for trials of a contemporary uncemented hydroxyapatite coated stem with a cemented stem hemiarthroplasty.
What's new
| Date | Event | Description |
|---|---|---|
| 8 January 2010 | New citation required and conclusions have changed | For the fourth update (Issue 6, 2010), the following changes were made 1. Search was updated to September 2009. 2. New studies of Figved 2009, Macaulay 2008, Mouzopoulos 2008 and Parker 2009 were included. 3. Ongoing studies of Kavcic 2006 and Patel 2008 were identified. 4. Study of Georgescu 2004 now excluded (previously awaiting classification). 5. Extra reference for the excluded study of Karpman 1992 added. 6. Reappraisal of Dorr 1986 led to its exclusion from the cemented versus uncemented hemiarthroplasty comparison. Substantial changes made to the conclusions of the review. |
History
Protocol first published: Issue 3, 1999 Review first published: Issue 3, 2001
| Date | Event | Description |
|---|---|---|
| 9 May 2008 | Amended | Converted to new review format. |
| 19 February 2008 | New search has been performed | For the third update (Issue 2, 2008), the following changes were made: 1. Search was updated to February 2007. 2. New studies of Baker 2006 and Blomfeldt 2007 were included. 3. Functional outcome data for Stars 2006 presented in the analyses There were no substantial changes made to the conclusions of the review. |
| 19 May 2006 | New search has been performed | For the second update (Issue 3, 2006) the following changes were made: 1. Search updated to December 2005; 2. New studies of STARS 2005 and Santini 2005 included; 3. Ongoing study of Georgescu 2004 identified; 4. Study of Field 2005 excluded; 5. Comparisons adjusted to split hemiarthroplasty versus total hip replacement into those with and without cement. There were no changes to the conclusions of the review. |
| 18 February 2004 | New search has been performed | For the first update (Issue 2, 2004) the following changes were made: 1. New studies of Branfoot 2000 and Raia 2003 included; 2. Studies of Clark 2001, Faraj 1999, Gierer 2002, Graf 2000, Johnson 2001, Leidinger 2002, and Sadr 1977 added to excluded studies; 3. Study of Bonke 1999 and Moroni 2002 added to ongoing studies. There were no changes to the conclusions of the review. |
Acknowledgements
Daniel Rajan was co‐author for the first version of this review. We are indebted to Prof William Gillespie, Dr Helen Handoll and Mrs Lesley Gillespie for their help. We also thank the following for useful comments at editorial review: Prof James Hutchison, Prof Harley Gray, Prof Rajan Madhok, Prof Gordon Murray, Prof Marc Swiontkowski, Prof Peter Herbison and Dr Janet Wale.
Appendices
Appendix 1. Search strategies
The Cochrane Library (Wiley InterScience interface)
#1 MeSH descriptor Hip Fractures explode all trees #2 ((hip* or proximal or neck) NEAR (femur* or femoral*) NEAR fracture*):ti,ab #3 (#1 OR #2) #4 MeSH descriptor Arthroplasty, this term only #5 MeSH descriptor Arthroplasty, Replacement, Hip, this term only #6 (hemiarthroplast* or arthroplast* or prosthes*) #7 (#4 OR #5 OR #6) #8 (#3 AND #7) (229 records)
MEDLINE (Ovid interface)
1 exp Hip Fractures/ 2 ((hip$ or ((femur$ or femoral$) adj3 (neck or proximal or intracapsul$))) adj4 fracture$).tw. 3 or/1‐2 4 Arthroplasty/ or Arthroplasty, Replacement,Hip/ 5 (arthroplast$ or hemiarthroplast$ or hemi‐arthroplast$ or prosthes$ or implant$ or bipolar or unipolar).tw. 6 or/4‐5 7 and/3,6 8 Randomized Controlled Trial.pt. 9 Controlled Clinical Trial.pt. 10 randomized.ab. 11 placebo.ab. 12 Clinical Trials as Topic/ 13 randomly.ab. 14 trial.ti. 15 8 or 9 or 10 or 11 or 12 or 13 or 14 16 exp Animals/ not Humans/ 17 15 not 16 18 17 and 7 (224 records)
EMBASE (Ovid interface)
1 exp Hip Fracture/ 2 ((hip$ or ((femur$ or femoral$) adj3 (neck or proximal or intracapsul$))) adj4 fracture$).tw. 3 or/1‐2 4 Arthroplasty/ or Hip Arthroplasty/ or Hip Hemiarthroplasty/ 5 (arthroplast$ or hemiarthroplast$ or hemi‐arthroplast$ or prosthes$ or implant$ or bipolar or unipolar).tw. 6 or/4‐5 7 and/3,6 8 exp Randomized Controlled Trial/ 9 exp Double Blind Procedure/ 10 exp Single Blind Procedure/ 11 exp Crossover Procedure/ 12 Controlled Study/ 13 or/8‐12 14 ((clinical or controlled or comparative or placebo or prospective$ or randomi#ed) adj3 (trial or study)).tw. 15 (random$ adj7 (allocat$ or allot$ or assign$ or basis$ or divid$ or order$)).tw. 16 ((singl$ or doubl$ or trebl$ or tripl$) adj7 (blind$ or mask$)).tw. 17 (cross?over$ or (cross adj1 over$)).tw. 18 ((allocat$ or allot$ or assign$ or divid$) adj3 (condition$ or experiment$ or intervention$ or treatment$ or therap$ or control$ or group$)).tw. 19 or/14‐18 20 or/13,19 21 limit 20 to human 22 and/7,21 (816 records)
Appendix 2. Search strategies used in 2001, 2004 and 2006 versions
MEDLINE (OVID WEB)
1. exp Hip Fractures/ 2. hip$ or femur$ or femoral$ or trochant$ or pertrochant$ or intertrochant$ or subtrochant$ or intracapsular$ or extracapsular$) adj4 fracture$).tw. 3. or/1‐2 4. (pin$1 or nail$ or screw$1 or plate$1 or arthroplast$ or fix$ or prosthes$).tw. 5. Internal Fixators/ or Bone Screws/ or Fracture Fixation, Internal/ or Bone Plates/ or Bone Nails/ 6. Arthroplasty/ or Arthroplasty, Replacement, Hip/ 7. or/4‐6 8. and/3,7
Line 8 was combined with all three stages of the Cochrane optimal trial search strategy (Higgins 2006)
EMBASE (OVID WEB)
1. exp Hip Fracture/ 2. ((hip$ or ((femur$ or femoral$) adj3 (neck or proximal))) adj4 fracture$).tw. 3. or/1‐2 4. exp Randomized Controlled trial/ 5. exp Double Blind Procedure/ 6. exp Single Blind Procedure/ 7. exp Crossover Procedure/ 8. Controlled Study/ 9. or/4‐8 10. ((clinical or controlled or comparative or placebo or prospective$ or randomi#ed) adj3 (trial or study)).tw. 11. (random$ adj7 (allocat$ or allot$ or assign$ or basis$ or divid$ or order$)).tw. 12. ((singl$ or doubl$ or trebl$ or tripl$) adj7 (blind$ or mask$)).tw. 13. (cross?over$ or (cross adj1 over$)).tw. 14. ((allocat$ or allot$ or assign$ or divid$) adj3 (condition$ or experiment$ or intervention$ or treatment$ or therap$ or control$ or group$)).tw. 15. or/10‐14 16. or/9,15 17. limit 16 to human 18. and/3,17
CINAHL (OVID WEB)
1. exp Hip Fractures/ 2. ((hip$ or ((femur$ or femoral$) adj3 (neck or proximal))) adj4 fracture$).tw. 3. or/1‐2 4. exp Clinical Trials/ 5. exp Evaluation Research/ 6. exp Comparative Studies/ 7. exp Crossover Design/ 8. clinical trial.pt. 9. or/4‐8 10. ((clinical or controlled or comparative or placebo or prospective or randomi#ed) adj3 (trial or study)).tw. 11. (random$ adj7 (allocat$ or allot$ or assign$ or basis$ or divid$ or order$)).tw. 12. ((singl$ or doubl$ or trebl$ or tripl$) adj7 (blind$ or mask$)).tw. 13. (cross?over$ or (cross adj1 over$)).tw. 14. ((allocat$ or allot$ or assign$ or divid$) adj3 (condition$ or experiment$ or intervention$ or treatment$ or therap$ or control$ or group$)).tw. 15. or/10‐14 16. or/9,15 17. and/3,16
Data and analyses
Comparison 1. Cemented hemiarthroplasty versus uncemented hemiarthroplasty.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Length of surgery (minutes) | 3 | 559 | Mean Difference (IV, Fixed, 95% CI) | 7.24 [4.75, 9.73] |
| 1.1 Cemented Thompson versus uncemented Moore | 1 | 400 | Mean Difference (IV, Fixed, 95% CI) | 6.90 [4.29, 9.51] |
| 1.2 Cemented Thompson bipolar versus uncemented Moore bipolar | 1 | 53 | Mean Difference (IV, Fixed, 95% CI) | 8.0 [‐1.82, 17.82] |
| 1.3 Cemented bipolar versus uncemented bipolar hemiarthroplasty | 1 | 106 | Mean Difference (IV, Fixed, 95% CI) | 18.02 [2.03, 34.01] |
| 2 Operative fracture of the femur | 3 | 597 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.09 [0.02, 0.44] |
| 2.1 Cemented Thompson versus uncemented Thompson | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.03 [0.00, 0.57] |
| 2.2 Cemented Thompson versus uncemented Thompson | 1 | 91 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.46 [0.02, 11.03] |
| 2.3 Cemented bipolar versus uncemented bipolar hemiarthroplasty | 1 | 106 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.2 [0.01, 4.07] |
| 3 Operative blood loss (ml) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 3.1 Cemented Thompson bipolar versus uncemented Moore bipolar | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Mean units blood transfused | 2 | 506 | Mean Difference (IV, Fixed, 95% CI) | 0.12 [‐0.04, 0.27] |
| 4.1 Cemented Thompson versus uncemented Moore | 1 | 400 | Mean Difference (IV, Fixed, 95% CI) | 0.12 [‐0.04, 0.28] |
| 4.2 Cemented bipolar versus uncemented bipolar hemiarthroplasty | 1 | 106 | Mean Difference (IV, Fixed, 95% CI) | 0.05 [‐0.71, 0.81] |
| 5 Number of patients transfused | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 5.1 Cemented Thompson versus uncemented Moore | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Peri‐operative death | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 6.1 Cemented Thompson versus uncemented Moore | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6.2 Cemented Thompson versus uncemented Thompson | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Later fracture of the femur | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 7.1 Cemented Thompson versus uncemented Moore | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 Dislocation | 4 | 740 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.03 [0.59, 6.93] |
| 8.1 Cemented Thompson versus uncemented Moore | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.0 [0.18, 21.88] |
| 8.2 Cemented Thompson versus uncemented Thompson | 2 | 234 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.83 [0.36, 9.20] |
| 8.3 Cemented bipolar versus uncemented bipolar hemiarthroplasty | 1 | 106 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.12, 72.02] |
| 9 Superficial wound infection | 4 | 708 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.08 [0.41, 2.83] |
| 9.1 Cemented Thompson versus uncemented Moore | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.33 [0.30, 5.88] |
| 9.2 Cemented Thompson versus uncemented Thompson | 1 | 143 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.57 [0.10, 3.32] |
| 9.3 Cemented Moore versus uncemented Moore | 1 | 112 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.07, 16.16] |
| 9.4 Cemented Thompson bipolar versus uncemented Moore bipolar | 1 | 53 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.89 [0.12, 67.96] |
| 10 Deep sepsis | 4 | 761 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.25 [0.48, 3.24] |
| 10.1 Cemented Thompson versus uncemented Moore | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.2 [0.37, 3.87] |
| 10.2 Cemented Thompson versus uncemented Thompson | 1 | 143 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.58 [0.11, 62.21] |
| 10.3 Cemented Moore versus uncemented Moore | 1 | 112 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.35 [0.01, 8.30] |
| 10.4 Cemented bipolar versus uncemented bipolar hemiarthroplasty | 1 | 106 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.12, 72.02] |
| 11 Wound haematoma | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 11.1 Cemented Thompson versus uncemented Moore | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 12 Reoperations: minor | 2 | 491 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.41 [0.10, 1.75] |
| 12.1 Cemented Thompson versus uncemented Moore | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.4 [0.08, 2.04] |
| 12.2 Cemented Thompson versus uncemented Thompson | 1 | 91 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.46 [0.02, 11.03] |
| 13 Reoperations: major | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 13.1 Cemented Thompson versus uncemented Moore | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 14 Reoperations: any | 2 | 491 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.55 [0.27, 1.14] |
| 14.1 Cemented Thompson versus uncemented Moore | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.56 [0.26, 1.17] |
| 14.2 Cemented Thompson versus uncemented Thompson | 1 | 91 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.46 [0.02, 11.03] |
| 15 Medical complications | 3 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 15.1 Pneumonia | 2 | 454 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.11, 1.01] |
| 15.2 Pulmonary embolism | 2 | 453 | Risk Ratio (M‐H, Fixed, 95% CI) | 4.91 [0.58, 41.25] |
| 15.3 Deep vein thrombosis | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.14, 7.03] |
| 15.4 Pulmonary embolism or pneumonia | 2 | 506 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.58 [0.23, 1.46] |
| 15.5 Myocardical infarction | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.5 [0.05, 5.47] |
| 15.6 Acute cardiac arrhythmia | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.01, 8.13] |
| 15.7 Myocardial infarction or cardiac arrhythmia | 2 | 506 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.30, 3.38] |
| 15.8 Congestive c ardiac failu re | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.67 [0.19, 2.33] |
| 15.9 Cerebrovascular accident | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.0 [0.18, 21.88] |
| 15.10 R uptured aortic aneurysm | 1 | 54 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.01, 7.84] |
| 15.11 Acute renal failure | 2 | 454 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.14, 6.99] |
| 15.12 Gastrointestinal haemor r hage | 2 | 454 | Risk Ratio (M‐H, Fixed, 95% CI) | 6.0 [0.73, 49.27] |
| 15.13 Intestinal obstruction | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.01, 8.13] |
| 15.14 Peritonitis | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.0 [0.12, 73.20] |
| 15.15 Clostridian diarrhoea | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.01, 8.13] |
| 15.16 Gastric disease | 1 | 106 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.06, 15.57] |
| 15.17 Urinary tract infection | 2 | 160 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.81 [0.35, 1.87] |
| 15.18 Acute confusion sta te | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.14, 7.03] |
| 15.19 Pressure sores | 3 | 559 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.51 [0.23, 1.16] |
| 16 All reported medical complications | 3 | 559 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.59, 1.13] |
| 16.1 Cemented Thompson versus uncemented Moore | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.63 [0.39, 1.01] |
| 16.2 Cemented Thompson bipolar versus uncemented Moore bipolar | 1 | 53 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.54 [0.58, 4.10] |
| 16.3 Cemented bipolar versus uncemented bipolar hemiarthroplasty | 1 | 106 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.60, 1.66] |
| 17 Length of hospital stay (days) | 4 | 696 | Mean Difference (IV, Fixed, 95% CI) | ‐1.42 [‐3.15, 0.32] |
| 17.1 Cemented Thompson versus uncemented Moore | 1 | 400 | Mean Difference (IV, Fixed, 95% CI) | ‐4.40 [‐9.13, 0.33] |
| 17.2 Cemented Thompson versus uncemented Thompson | 1 | 143 | Mean Difference (IV, Fixed, 95% CI) | ‐2.18 [‐4.80, 0.44] |
| 17.3 Cemented Thompson bipolar versus uncemented Moore bipolar | 1 | 47 | Mean Difference (IV, Fixed, 95% CI) | 2.30 [‐3.50, 8.10] |
| 17.4 Cemented bipolar versus uncemented bipolar hemiarthroplasty | 1 | 106 | Mean Difference (IV, Fixed, 95% CI) | ‐0.23 [‐3.21, 2.75] |
| 18 Mortality before 1 month | 2 | 453 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.84 [0.38, 1.84] |
| 18.1 Cemented Thompson versus uncemented Moore | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.82 [0.35, 1.93] |
| 18.2 Cemented Thompson bipolar versus uncemented Moore bipolar | 1 | 53 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.96 [0.15, 6.34] |
| 19 Mortality at 1 to 3 months | 4 | 708 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.98 [0.68, 1.41] |
| 19.1 Cemented Thompson versus uncemented Moore | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.76 [0.45, 1.27] |
| 19.2 Cemented Thompson versus uncemented Thompson | 1 | 143 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.71 [0.68, 4.32] |
| 19.3 Cemented Moore versus uncemented Moore | 1 | 112 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.04 [0.49, 2.19] |
| 19.4 Cemented Thompson bipolar versus uncemented Moore bipolar | 1 | 53 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.28 [0.32, 5.19] |
| 20 Mortality at 1 year | 5 | 793 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.71, 1.13] |
| 20.1 Cemented Thompson versus uncemented Moore | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.85 [0.63, 1.17] |
| 20.2 Cemented Thompson versus uncemented Thompson | 2 | 234 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.56, 1.40] |
| 20.3 Cemented Thompson bipolar versus uncemented Moore bipolar | 1 | 53 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.28 [0.52, 3.19] |
| 20.4 Cemented bipolar versus uncemented bipolar hemiarthroplasty | 1 | 106 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.93 [0.48, 1.78] |
| 21 Mortality at 3 years | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 21.1 Cemented Thompson versus uncemented Moore | 1 | Odds Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 22 Failure to regain mobility | 3 | 378 | Risk Ratio (M‐H, Random, 95% CI) | 0.84 [0.64, 1.11] |
| 22.1 Cemented Thompson versus uncemented Moore | 1 | 281 | Risk Ratio (M‐H, Random, 95% CI) | 0.92 [0.78, 1.09] |
| 22.2 Cemented Moore versus uncemented Moore | 1 | 58 | Risk Ratio (M‐H, Random, 95% CI) | 0.96 [0.62, 1.48] |
| 22.3 Cemented Thompson bipolar versus uncemented Moore bipolar | 1 | 39 | Risk Ratio (M‐H, Random, 95% CI) | 0.53 [0.30, 0.93] |
| 23 Change in mobility score at 6 months (from pre‐injury) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 23.1 Cemented Thompson versus uncemented Moore | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 24 Pain at early follow up (3 months) | 2 | 375 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.77 [0.60, 0.98] |
| 24.1 Cemented Thompson versus uncemented Moore | 1 | 324 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.80 [0.62, 1.04] |
| 24.2 Cemented Moore versus uncemented Moore | 1 | 51 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.53 [0.24, 1.17] |
| 25 Pain at 1 to 2 years | 3 | 369 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.55 [0.40, 0.75] |
| 25.1 Cemented Thompson versus uncemented Moore | 1 | 272 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.58 [0.38, 0.87] |
| 25.2 Cemented Moore versus uncemented Moore | 1 | 58 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.63 [0.33, 1.22] |
| 25.3 Cemented Thompson bipolar versus uncemented Moore bipolar | 1 | 39 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.39 [0.20, 0.79] |
| 26 Pain score at 6 months (0 to 10: worst) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 26.1 Cemented Thompson versus uncemented Moore | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 27 Failure to return home | 2 | 439 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.62 [0.34, 1.12] |
| 27.1 Cemented Thompson versus uncemented Moore | 1 | 400 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.62 [0.32, 1.20] |
| 27.2 Cemented Thompson bipolar versus uncemented Moore bipolar | 1 | 39 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.63 [0.17, 2.29] |
1.6. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 6 Peri‐operative death.
1.7. Analysis.
Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 7 Later fracture of the femur.
1.8. Analysis.
Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 8 Dislocation.
1.9. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 9 Superficial wound infection.
1.11. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 11 Wound haematoma.
1.12. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 12 Reoperations: minor.
1.13. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 13 Reoperations: major.
1.14. Analysis.

Comparison 1 Cemented hemiarthroplasty versus uncemented hemiarthroplasty, Outcome 14 Reoperations: any.
Comparison 2. Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Length of surgery (minutes) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 1.1 Cemented bipolar versus hydroxyapatite coated bipolar hemiarthroplasty | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 2 Operative blood loss (ml) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 Cemented bipolar versus hydroxyapatite coated bipolar | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Total blood loss (ml) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 3.1 Cemented bipolar versus hydroxyapatite coated bipolar | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Number of patients transfused | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 4.1 Cemented bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5 Operative fracture of femur | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 5.1 Cemented bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 5.2 Uncemented Moore bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 6 Later fracture of the femur | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 6.1 Cemented bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Dislocation | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 7.1 Cemented bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7.2 Uncemented Moore bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 8 Superficial wound sepsis | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 8.1 Cemented bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 9 Deep wound sepsis | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 9.1 Cemented bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 10 Any wound infection | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 10.1 Cemented bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 10.2 Uncemented Moore bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 11 Reoperations: minor | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 11.1 Cemented bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 11.2 Uncemented Moore bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 12 Reoperations: major | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 12.1 Cemented bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 12.2 Uncemented Moore bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13 Reoperations: any | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 13.1 Cemented bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 13.2 Uncemented Moore bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 14 Medical complications | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 14.1 Pneumonia | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 14.2 Deep vein thrombosis | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 14.3 Pulmonary embolism | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 14.4 Myocardial infarction | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 14.5 Pressure sores | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 14.6 All medical complications | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 15 Length of hospital stay (days) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 15.1 Cemented bipolar versus hydroxyapatite coated bipolar | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 16 Mortality | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 16.1 7 days | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 16.2 30 days | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 16.3 90 days | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 16.4 12 months | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 16.5 24 months | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 17 Residual pain | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 17.1 Need for pain medication at 3 months | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 17.2 Need for pain medication at 12 months | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 18 Need to use walking aids | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 18.1 Unable to walk without aids at 3 months | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 18.2 Unable to walk without aids at 12 months | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 19 Failure to return back home at 1 year | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 19.1 Uncemented Moore bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 20 Failure to be able to shop at 1 year | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 20.1 Uncemented Moore bipolar versus hydroxyapatite coated bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 21 Barthel index | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 21.1 Barthel score less than 19 at 3 months | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 21.2 Barthel score less than 19 at 12 months | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 22 Harris and ED‐5D outcome scores | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 22.1 Harris hip score at 3 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 22.2 Harris hip score at 12 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 22.3 ED‐5D index score at 3 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 22.4 ED‐5D index score at 12 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 22.5 ED‐5D visual analogue score at 3 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 22.6 ED‐5D visual analogue score at 12 months | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
2.2. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 2 Operative blood loss (ml).
2.6. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 6 Later fracture of the femur.
2.7. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 7 Dislocation.
2.8. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 8 Superficial wound sepsis.
2.9. Analysis.
Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 9 Deep wound sepsis.
2.10. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 10 Any wound infection.
2.11. Analysis.
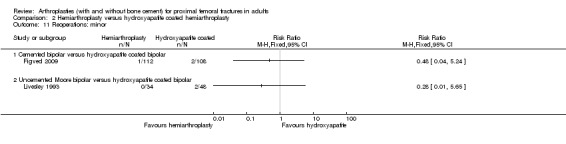
Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 11 Reoperations: minor.
2.12. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 12 Reoperations: major.
2.13. Analysis.
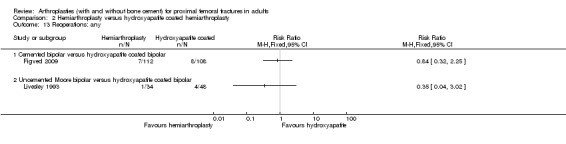
Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 13 Reoperations: any.
2.14. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 14 Medical complications.
2.18. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 18 Need to use walking aids.
2.21. Analysis.

Comparison 2 Hemiarthroplasty versus hydroxyapatite coated hemiarthroplasty, Outcome 21 Barthel index.
Comparison 3. Unipolar hemiarthroplasty versus bipolar hemiarthroplasty.
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Dislocation | 5 | 668 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.09 [0.36, 3.31] |
| 1.1 Austin Moore unipolar versus Moore bipolar | 1 | 68 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.89 [0.06, 13.64] |
| 1.2 Cemented unipolar versus cemented bipolar | 2 | 163 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.40 [0.21, 9.20] |
| 1.3 Cemented Thompson unipolar versus cemented Monk bipolar | 2 | 437 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.98 [0.20, 4.93] |
| 2 Dislocation requiring open reduction | 4 | 418 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.32 [0.03, 3.07] |
| 2.1 Austin Moore unipolar versus Moore bipolar | 1 | 68 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.30 [0.01, 7.05] |
| 2.2 Cemented unipolar versus cemented bipolar | 2 | 162 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 2.3 Cemented Thompson unipolar versus cemented Monk bipolar | 1 | 188 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.36 [0.01, 8.61] |
| 3 Acetabular erosion on X‐rays | 3 | 505 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.83 [0.81, 18.15] |
| 3.1 Austin Moore unipolar versus Moore bipolar | 1 | 68 | Risk Ratio (M‐H, Fixed, 95% CI) | 4.46 [0.22, 89.56] |
| 3.2 Cemented Thompson unipolar versus cemented Monk bipolar | 2 | 437 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.61 [0.59, 22.28] |
| 4 Acetabular erosion requiring revision arthroplasty | 3 | 505 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.97 [0.47, 18.85] |
| 4.1 Austin Moore unipolar versus Moore bipolar | 1 | 68 | Risk Ratio (M‐H, Fixed, 95% CI) | 4.46 [0.22, 89.56] |
| 4.2 Cemented Thompson unipolar versus cemented Monk bipolar | 2 | 437 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.16 [0.20, 23.37] |
| 5 Deep sepsis | 4 | 620 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.34 [0.50, 3.62] |
| 5.1 Austin Moore unipolar versus Moore bipolar | 1 | 68 | Risk Ratio (M‐H, Fixed, 95% CI) | 4.46 [0.22, 89.56] |
| 5.2 Cemented unipolar versus cemented bipolar | 1 | 115 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.75 [0.11, 66.23] |
| 5.3 Cemented Thompson unipolar versus cemented Monk bipolar | 2 | 437 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.92 [0.29, 2.99] |
| 6 Reoperations: minor | 3 | 370 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.97 [0.22, 4.31] |
| 6.1 Austin Moore unipolar versus Moore bipolar | 1 | 68 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.68 [0.11, 63.45] |
| 6.2 Cemented unipolar versus cemented bipolar | 1 | 115 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.92 [0.06, 14.30] |
| 6.3 Cemented Thompson unipolar versus cemented Monk bipolar | 1 | 187 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.54 [0.05, 5.84] |
| 7 Reoperations: major | 3 | 370 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.63 [0.51, 5.25] |
| 7.1 Austin Moore unipolar versus Moore bipolar | 1 | 68 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.56 [0.42, 30.19] |
| 7.2 Cemented unipolar versus cemented bipolar | 1 | 115 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.75 [0.11, 66.23] |
| 7.3 Cemented Thompson unipolar versus cemented Monk bipolar | 1 | 187 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.72 [0.12, 4.20] |
| 8 Reoperations: any | 3 | 370 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.41 [0.54, 3.69] |
| 8.1 Austin Moore unipolar versus Moore bipolar | 1 | 68 | Risk Ratio (M‐H, Fixed, 95% CI) | 4.44 [0.55, 36.06] |
| 8.2 Cemented unipolar versus cemented bipolar | 1 | 115 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.83 [0.17, 19.66] |
| 8.3 Cemented Thompson unipolar versus cemented Monk bipolar | 1 | 187 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.65 [0.16, 2.63] |
| 9 Deep vein thrombosis | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 9.1 Cemented unipolar versus cemented bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 10 Mortality at 6 months | 3 | 366 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.73, 1.76] |
| 10.1 Austin Moore unipolar versus Moore bipolar | 1 | 68 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 10.2 Cemented unipolar versus cemented bipolar | 1 | 48 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.1 [0.11, 11.21] |
| 10.3 Cemented Thompson unipolar versus cemented Monk bipolar | 1 | 250 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.13 [0.72, 1.78] |
| 11 Mortality at 1 to 2 years | 3 | 433 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.64, 1.26] |
| 11.1 Austin Moore unipolar versus Moore bipolar | 1 | 68 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 11.2 Cemented Thompson unipolar versus cemented Monk bipolar | 2 | 365 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.90 [0.64, 1.26] |
| 12 Failure to regain mobility | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 12.1 Cemented unipolar versus cemented bipolar | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
3.6. Analysis.
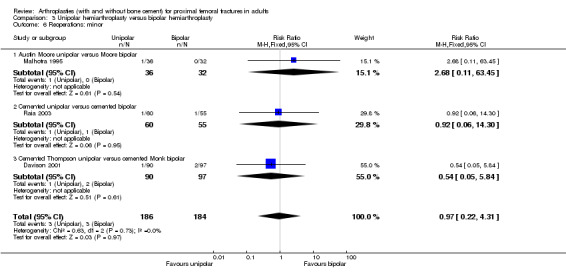
Comparison 3 Unipolar hemiarthroplasty versus bipolar hemiarthroplasty, Outcome 6 Reoperations: minor.
3.7. Analysis.

Comparison 3 Unipolar hemiarthroplasty versus bipolar hemiarthroplasty, Outcome 7 Reoperations: major.
3.8. Analysis.

Comparison 3 Unipolar hemiarthroplasty versus bipolar hemiarthroplasty, Outcome 8 Reoperations: any.
3.9. Analysis.

Comparison 3 Unipolar hemiarthroplasty versus bipolar hemiarthroplasty, Outcome 9 Deep vein thrombosis.
Comparison 4. Hemiarthroplasty versus total hip replacement (THR).
| Outcome or subgroup title | No. of studies | No. of participants | Statistical method | Effect size |
|---|---|---|---|---|
| 1 Length of surgery (minutes) | 4 | 379 | Mean Difference (IV, Fixed, 95% CI) | ‐18.53 [‐23.74, ‐13.32] |
| 1.1 Cemented hemiarthroplasty versus cemented THR | 3 | 339 | Mean Difference (IV, Fixed, 95% CI) | ‐19.20 [‐24.55, ‐13.84] |
| 1.2 Hemiarthroplasty versus THR (with or without cement) | 1 | 40 | Mean Difference (IV, Fixed, 95% CI) | ‐7.0 [‐29.30, 15.30] |
| 2 Total blood loss (ml) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 2.1 Cemented hemiarthroplasty versus cemented THR | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 3 Mean volume blood transfused (ml) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 3.1 Cemented hemiarthroplasty versus cemented THR | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 4 Dislocation | 6 | 648 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.53 [0.29, 0.97] |
| 4.1 Cemented hemiarthroplasty versus total hip replacement | 3 | 339 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.38 [0.09, 1.60] |
| 4.2 Hemiarthroplasty versus total hip replacement (with and without cement) | 2 | 129 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.23 [0.06, 0.89] |
| 4.3 Uncemented hemiarthroplasty versus total hip replacement | 1 | 180 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.39, 1.97] |
| 5 Fracture around implant at surgery | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 5.1 Cemented hemiarthroplasty versus total hip replacement | 2 | 219 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.93 [0.12, 69.83] |
| 6 Later fracture around implant | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 6.1 Cemented hemiarthroplasty versus total hip replacement | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 7 Superficial wound infection | 4 | 428 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.23, 2.19] |
| 7.1 Cemented hemiarthroplasty versus total hip replacement | 3 | 339 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.71 [0.23, 2.19] |
| 7.2 Hemiarthroplasty versus total hip replacement (with and without cement) | 1 | 89 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8 Deep wound infection | 5 | 608 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.47 [0.09, 2.49] |
| 8.1 Cemented hemiarthroplasty versus total hip replacement | 3 | 339 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.6 [0.08, 4.48] |
| 8.2 Hemiarthroplasty versus total hip replacement (with and without cement) | 1 | 89 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] |
| 8.3 Uncemented hemiarthroplasty versus total hip replacement | 1 | 180 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.27 [0.01, 6.47] |
| 9 Any wound infection | 6 | 648 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.68 [0.28, 1.68] |
| 9.1 Cemented hemiarthroplasty versus total hip replacement | 3 | 339 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.66 [0.24, 1.82] |
| 9.2 Hemiarthroplasty versus total hip replacement (with and without cement) | 2 | 129 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.25 [0.10, 52.07] |
| 9.3 Uncemented hemiarthroplasty versus total hip replacement | 1 | 180 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.27 [0.01, 6.47] |
| 10 Reoperations: minor | 5 | 510 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.50 [0.26, 0.95] |
| 10.1 Cemented hemiarthroplasty versus total hip replacement | 2 | 201 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.16 [0.02, 1.33] |
| 10.2 Hemiarthroplasty versus total hip replacement (with and without cement) | 2 | 129 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.22 [0.05, 1.01] |
| 10.3 Uncemented hemiarthroplasty versus total hip replacement | 1 | 180 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.88 [0.39, 1.97] |
| 11 Reoperations: major | 6 | 596 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.22 [1.09, 4.51] |
| 11.1 Cemented hemiarthroplasty versus total hip replacement | 2 | 201 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.38 [0.28, 6.85] |
| 11.2 Hemiarthroplasty versus total hip replacement (with and without cement) | 3 | 215 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.80 [0.62, 5.25] |
| 11.3 Uncemented hemiarthroplasty versus total hip replacement | 1 | 180 | Risk Ratio (M‐H, Fixed, 95% CI) | 3.47 [1.02, 11.75] |
| 12 Reoperations: any | 7 | 734 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.99 [0.66, 1.48] |
| 12.1 Cemented hemiarthroplasty versus total hip replacement | 3 | 339 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.62 [0.27, 1.42] |
| 12.2 Hemiarthroplasty versus total hip replacement (with and without cement) | 3 | 215 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.83 [0.39, 1.76] |
| 12.3 Uncemented hemiarthroplasty versus total hip replacement | 1 | 180 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.48 [0.80, 2.71] |
| 13 Medical complications | 4 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 13.1 Deep vein thrombosis | 3 | 339 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.26 [0.07, 1.04] |
| 13.2 Pulmonary embolism | 3 | 259 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.45 [0.69, 8.73] |
| 13.3 Pneumonia | 3 | 241 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.07 [0.32, 3.55] |
| 13.4 Pressure sores | 2 | 201 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.03, 3.11] |
| 13.5 Myocardial infarction | 3 | 298 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.63 [0.20, 1.98] |
| 13.6 Cerebrovascular accident | 1 | 138 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.0 [0.14, 6.90] |
| 13.7 Gastrointestinal bleed | 2 | 121 | Risk Ratio (M‐H, Fixed, 95% CI) | 2.57 [0.28, 23.83] |
| 13.8 Cardiac arrhythmia | 3 | 241 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.16 [0.28, 4.81] |
| 13.9 Congestive cardiac failure | 1 | 120 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.01, 8.02] |
| 13.10 Hyponatraemia | 1 | 81 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.33 [0.01, 7.76] |
| 13.11 Paralytic ileus | 1 | 40 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.15 [0.01, 2.94] |
| 13.12 All medical complications | 4 | 379 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.74 [0.47, 1.16] |
| 14 Hospital stay (days) | 3 | 264 | Mean Difference (IV, Fixed, 95% CI) | ‐0.42 [‐1.90, 1.06] |
| 14.1 Cemented hemiarthroplasty versus total hip replacement | 1 | 138 | Mean Difference (IV, Fixed, 95% CI) | ‐0.80 [‐3.82, 2.22] |
| 14.2 Hemiarthroplasty versus total hip replacement (with and without cement) | 2 | 126 | Mean Difference (IV, Fixed, 95% CI) | ‐0.30 [0.00, 1.40] |
| 15 Mortality | 6 | Risk Ratio (M‐H, Fixed, 95% CI) | Subtotals only | |
| 15.1 Mortality at 3 to 6 months | 3 | 358 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.88 [0.96, 3.68] |
| 15.2 Mortality at 1 year | 4 | 524 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.15 [0.76, 1.74] |
| 15.3 Mortality at 2 years | 1 | 138 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.5 [0.56, 3.99] |
| 15.4 Mortality at 3 to 4 years | 3 | 207 | Risk Ratio (M‐H, Fixed, 95% CI) | 1.16 [0.73, 1.85] |
| 16 Residual pain at 1 year | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 16.1 Cemented hemiarthroplasty versus total hip replacement | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 16.2 Uncemented hemiarthroplasty versus total hip replacement | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 17 Failure to regain mobility | 2 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 17.1 Hemiarthroplasty versus total hip replacement (with and without cement) | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 17.2 Uncemented hemiarthroplasty versus total hip replacement | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 18 Poor comorbidity index | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | Totals not selected | |
| 18.1 Cemented hemiarthroplasty versus total hip replacement | 1 | Risk Ratio (M‐H, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 19 Final outcomes for survivors (continuous outcomes for which a lower score is advantageous) | 1 | Mean Difference (IV, Fixed, 95% CI) | Totals not selected | |
| 19.1 Oxford Hip Score | 1 | Mean Difference (IV, Fixed, 95% CI) | 0.0 [0.0, 0.0] | |
| 20 Final outcomes for survivors (continuous outcomes for which a higher score is advantageous) | 4 | Mean Difference (IV, Fixed, 95% CI) | Subtotals only | |
| 20.1 Hip rating questionnaire | 1 | 106 | Mean Difference (IV, Fixed, 95% CI) | ‐6.10 [‐12.38, 0.18] |
| 20.2 Self reported walking distance (kilometres) | 1 | 69 | Mean Difference (IV, Fixed, 95% CI) | ‐1.7 [‐3.28, ‐0.12] |
| 20.3 Short form 36 physical score | 1 | 69 | Mean Difference (IV, Fixed, 95% CI) | ‐2.43 [‐7.56, 2.70] |
| 20.4 Short form 36 mental score | 1 | 69 | Mean Difference (IV, Fixed, 95% CI) | 3.30 [‐3.52, 10.12] |
| 20.5 EuroQol (EQ‐5d) questionnaire | 1 | 131 | Mean Difference (IV, Fixed, 95% CI) | ‐0.16 [‐0.28, ‐0.04] |
| 20.6 Harris Hip Score for pain | 1 | 111 | Mean Difference (IV, Fixed, 95% CI) | ‐4.0 [‐6.33, ‐1.67] |
| 20.7 Harris Hip Score for function | 1 | 111 | Mean Difference (IV, Fixed, 95% CI) | ‐3.70 [‐7.13, ‐0.27] |
| 20.8 Harris Hip Score at one year: total score | 2 | 174 | Mean Difference (IV, Fixed, 95% CI) | ‐5.47 [‐8.39, ‐2.55] |
| 20.9 Harris Hip Score at 4 years: total score | 1 | 43 | Mean Difference (IV, Fixed, 95% CI) | ‐4.20 [‐7.66, ‐0.74] |
| 20.10 Barthel score at 1 year | 1 | 63 | Mean Difference (IV, Fixed, 95% CI) | ‐8.0 [‐13.61, ‐2.39] |
| 20.11 Barthel score at 4 years | 1 | 43 | Mean Difference (IV, Fixed, 95% CI) | ‐5.70 [‐11.19, ‐0.21] |
4.2. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 2 Total blood loss (ml).
4.3. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 3 Mean volume blood transfused (ml).
4.5. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 5 Fracture around implant at surgery.
4.6. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 6 Later fracture around implant.
4.7. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 7 Superficial wound infection.
4.8. Analysis.
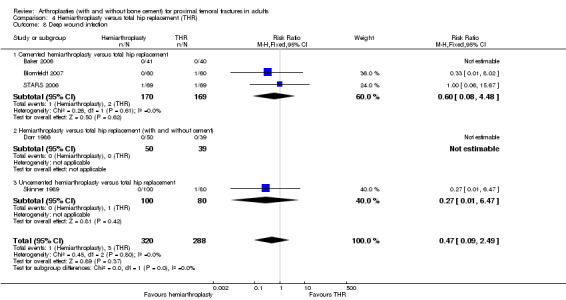
Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 8 Deep wound infection.
4.9. Analysis.
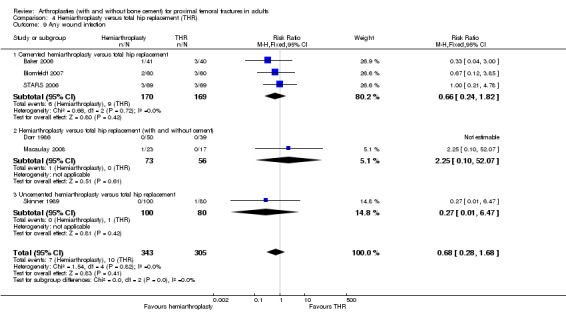
Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 9 Any wound infection.
4.14. Analysis.

Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 14 Hospital stay (days).
4.18. Analysis.
Comparison 4 Hemiarthroplasty versus total hip replacement (THR), Outcome 18 Poor comorbidity index.
Characteristics of studies
Characteristics of included studies [ordered by study ID]
Baker 2006.
| Methods | Method of randomisation: sealed opaque numbered envelopes. | |
| Participants | Orthopaedic unit in Bristol, UK. 81 people with an intracapsular fracture. Mean age: 75 years (range 66‐86). Percentage male: 21%. Follow up for 8 to 20 months. Loss to follow up: 3%. | |
| Interventions | Cemented CPT collarless hemiarthroplasty versus cemented CPT collarless total hip replacement. Surgical transgluteal approach for all cases. | |
| Outcomes | Length of surgery Dislocation Fracture around implant Reoperation Radiological signs of acetabular wear Radiological signs of cement wear Superficial wound infection Deep wound infection Mortality Pulmonary embolism Deep vein thrombosis Pneumonia Pressure sores Hematemesis Other medical complications Oxford hip score Self reported walking distance Short form 36 physical and mental score | |
| Notes | Additional information on trial methodology provided by the authors. The standard deviations for the outcomes of length of surgery, Oxford Hip Score, walking distance and short form 36 were estimated from the P values quoted, using the formula in the Cochrane Handbook. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Low risk | A ‐ Adequate |
Blomfeldt 2007.
| Methods | Method of randomisation: sealed envelopes | |
| Participants | Orthopaedic unit in Stockholm, Sweden. 120 patients with an intracapsular fracture. Mean age: 81 years (range 70‐90). Percentage male: 16%. Follow up for 12 months. Loss to follow up: none. | |
| Interventions | Cemented Exeter bipolar hemiarthroplasty versus Exeter stem and OGEE cup total hip arthroplasty. All prosthesis inserted with cement using an anterolateral approach. | |
| Outcomes | Length of surgery Operative blood loss Mean volume of blood transfused Dislocation Later fracture of the femur Superficial wound infection Deep wound infection Reoperation Deep vein thrombosis Pneumonia Pressure sores Atrial fibrillation Myocardial infarction Congestive cardiac failure Mortality at 1 year Harris hip score (total, pain, function, deformity, movement) Ceder disability index Health related quality of life | |
| Notes | Standard deviations for continuous outcomes measures were calculated from reported P‐values. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
Branfoot 2000.
| Methods | Method of randomisation: sealed envelopes. | |
| Participants | Orthopaedic unit in Yorkshire, UK. 91 people with an intracapsular fracture. Mean age: 83 years (range 63‐97). Percentage male: 11%. Follow up for 8 to 20 months. Loss to follow up: none. | |
| Interventions | Cemented Thompson prosthesis versus uncemented Thompson prosthesis. Surgical approach anterolateral for all cases. | |
| Outcomes | Dislocation Operative fracture femur Mortality Pain at follow up Limp Use of walking aids Activities Mobility | |
| Notes | Study reported as a conference abstract with additional information provided by the authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
Calder 1996.
| Methods | Method of randomisation: computer generated random numbers. | |
| Participants | Orthopaedic unit in Leicester, UK. 250 people with an intracapsular fracture. Mean age: not stated. Median age: 85 years (range not stated). Interquartile range: 82 to 88 years. All over 80 years. Percentage male: 14%. Follow up for a median of 594/694 days (mean not stated). Loss to follow up: not stated. | |
| Interventions | Cemented Thompson prosthesis versus cemented Monk prosthesis. Surgical approach not stated. | |
| Outcomes | Dislocation Acetabular erosion Deep wound infection Length of hospital stay Mortality Pain at follow up Failure to return to same residence Satisfaction with operation Limp Harris Hip Score Nottingham Health Profile | |
| Notes | See Calder SJ et al. J Bone Joint Surg Br 1995;77(3):494‐6 for methods. Patients aged less than 80 years randomised to three arms (internal fixation v unipolar prosthesis v bipolar prosthesis and those aged more than 80 years to two arms (unipolar prosthesis v bipolar prosthesis). Calder 1996 reports results for over 80 year olds. Results for under 80 yr olds are reported in Davison 2001 which has been included as a separate study. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
Cornell 1988.
| Methods | Method of randomisation: sealed envelopes containing random generated numbers. Envelopes opened in the operating room. | |
| Participants | Orthopaedic unit in New York, USA. 48 people with an intracapsular fracture. Mean age: 78 years (range 62 to 97). Percentage male: 25%. Follow up for six months. Loss to follow up: not stated. | |
| Interventions | Cemented modular unipolar hemiarthroplasty versus cemented modular bipolar hemiarthroplasty (all implant stems identical) All prosthesis inserted via posterior approach. | |
| Outcomes | Operative blood loss Superficial wound infection Dislocation Deep vein thrombosis Length of hospital stay Mortality Hip movements Gait analysis Hip score assessment | |
| Notes | Paper states that the results presented are a preliminary report of the first patients entered in the study with a short follow up. Further information requested from the authors. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Low risk | A ‐ Adequate |
Davison 2001.
| Methods | Method of randomisation: computer generated random numbers. | |
| Participants | Orthopaedic unit in Leicester, UK. 187 patients with a intracapsular fracture. Mean age: not stated (range not stated). Median ages 75 and 76 years. All aged between 65 to 79 years. Percentage male: 24%. Follow up for minimum two years. Loss to follow up: 18%. | |
| Interventions | Cemented Thompson prosthesis versus cemented Monk prosthesis. Both prosthesis had identical stems. All inserted via a Hardinge direct lateral approach. | |
| Outcomes | Acetabular erosion Dislocation Deep wound sepsis Reoperation Mortality Nottingham Health Profile Harris Hip Score Bartel Score Return to pre‐injury state Patient satisfaction | |
| Notes | See Calder SJ et al. J Bone Joint Surg Br 1995;77(3):494‐6 for methods. Davison 2001 reports on participants less than 80 years of age randomised to three arms (internal fixation versus unipolar prosthesis versus bipolar prosthesis). Participants who had internal fixation are not included in this review but included in the Cochrane review "Internal fixation versus arthroplasty for intracapsular proximal femoral fractures in adults". | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
Dorr 1986.
| Methods | Method of randomisation: odd or even hospital record number. | |
| Participants | Orthopaedic unit in California, USA. 89 patients with a intracapsular fracture. Mean age: 69 years (range 41 to 89 years) . Percentage male: 35%. Follow up for a range of two to four years. Loss to follow up: text suggests none. | |
| Interventions | Cemented bipolar hemiarthroplasty (first year of recruitment) or uncemented bipolar hemiarthroplasty (second year) versus cemented total hip replacement. Exact type of prosthetic stem not stated. All prosthesis inserted via posterior approach. | |
| Outcomes | Operative blood loss Superficial wound infection Dislocation Superficial wound infection Deep wound infection Reoperation Length of hospital stay Mortality Pain at follow up Failure to return to same residence Use of walking aids X‐ray findings at follow up Gait analysis | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | High risk | C ‐ Inadequate |
Emery 1991.
| Methods | Method of randomisation: sealed envelope opened at the time of operation. | |
| Participants | Orthopaedic unit in Essex, UK. 53 patients with a intracapsular fracture. Mean age: 79 years (range 61 to 96). Percentage male: 13%. Follow up for a mean of 17 months. Loss to follow up: no patients lost to follow up other than by death in 14 cases. | |
| Interventions | Cemented bipolar Thompson prosthesis versus uncemented Moore bipolar. Surgical approach not stated. | |
| Outcomes | Length of operation Operative blood loss Superficial wound infection Pulmonary embolism Pressure sores Pneumonia Other medical complications Mortality at 2 weeks, 3 and 17 months Hospital stay Pain at follow up Failure to return to same residence Use of walking aids | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
Figved 2009.
| Methods | Method of randomisation: sealed envelopes containing computer generated random numbers created in blocks of five. | |
| Participants | Two orthopaedic units in Norway. 220 patients with a displaced intracapsular fracture. Mean age: 83 years (range not stated). Percentage male: 24%. Follow up for a mean of 12 months. Loss to follow up: no patients lost to follow up other than by death. | |
| Interventions | Uncemented Corail hydroxyapatite coated bipolar prosthesis versus cemented Spectron bipolar hemiarthroplasty. Surgical approach posterior. | |
| Outcomes | Length of surgery Operative blood loss Total blood loss Transfusion requirements Number of patients transfused Dislocation Operative fracture of the femur Later fracture of femur Reoperation Superficial sepsis Deep sepsis Deep vein thrombosis Pulmonary embolism Pneumonia Myocardial infarction Length of hospital stay Mortality Need for pain medication Failure to be able to live at own home Harris hip score Barthel index of less than 19 Health related quality of life score (ED‐5D) Health related quality of life visual analogue scale ((ED‐5D) Inability to walk without aids | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Low risk | A ‐ Adequate |
Harper 1994.
| Methods | Method of randomisation: even or odd number of medical record. | |
| Participants | Orthopaedic unit in Leicester, UK. 137 patients with a intracapsular fracture. Mean age: 83 years (range 60 to 100). Percentage male: 26%. Follow up for a mean of 12 months. Loss to follow up: not stated | |
| Interventions | Cemented Thompson prosthesis versus Thompson prosthesis without cement. Surgical approach direct lateral without osteotomy of the trochanter. | |
| Outcomes | Dislocation Superficial wound sepsis Deep wound sepsis Intra‐operative deaths Length of orthopaedic ward stay Mortality Pain at follow up | |
| Notes | Most results for the study from the thesis of Harper. The number of trial participants was stated as 140 in the abstracts but as 137 in the thesis. Also abstract states there were two deaths in the recovery room in the cemented group. Thesis mentions only one intra‐operative death. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | High risk | C ‐ Inadequate |
Livesley 1993.
| Methods | Method of randomisation: week of admission. | |
| Participants | Orthopaedic unit in Mansfield, UK. 82 patients with an intracapsular fracture. Mean age: 81 years (range not stated). Percentage male: not stated. Follow up for 12 months. Loss to follow up: none | |
| Interventions | Uncemented Austin Moore stem bipolar hemiarthroplasty versus hydroxyapatite coated Furlong stem bipolar hemiarthroplasty. Surgical approach not stated. | |
| Outcomes | Dislocation Operative fracture of the femur Later fracture of the femur Wound infection Acetabular wear Reoperation Pressure sores Other medical complications Hospital stay Pain at follow up Mobility Use of walking aids Hip flexion | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | High risk | C ‐ Inadequate |
Macaulay 2008.
| Methods | Method of randomisation: sealed opaque envelopes containing blocked randomisation scheme. | |
| Participants | Five orthopaedic units in the USA. 40 patients with a displaced intracapsular fracture. Mean age: 79 years (range not stated). Percentage male: 48%. Follow up for 24 months. Loss to follow up: not clearly stated ? none. | |
| Interventions | Unipolar or bipolar hemiarthroplasty versus any type of total hip replacement with a 28mm head. Prosthesis could be cemented or uncemented. Surgical approach was choice of surgeon. | |
| Outcomes | Length of surgery Dislocation Wound sepsis Reoperation Deep vein thrombosis Pulmonary embolism Pneumonia Myocardial infarction Cardiac arrhythmia Other medical complications Length of hospital stay Mortality Inability to walk independently Level of activities of daily living SF36 score Womac score Timed get up and go score Harris hip score | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Low risk | A ‐ Adequate |
Malhotra 1995.
| Methods | Method of randomisation: not stated. | |
| Participants | Orthopaedic unit in New Delhi, India. 68 patients with an intracapsular fracture. Mean age: 67 years (range not stated). Percentage male: 56%. Follow up for a mean of 23/26 months. Range 9‐47 months. Loss to follow up: none. | |
| Interventions | Uncemented Austin Moore prosthesis versus uncemented Bateman bipolar prosthesis (Moore stem). Surgical approach not stated. | |
| Outcomes | Length of operation Operative blood loss Dislocation Deep wound infection Acetabular wear Range of hip movement Reoperation Hospital stay Pain at follow up Mobility Grading of outcome | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
Mouzopoulos 2008.
| Methods | Method of randomisation: not stated. | |
| Participants | Orthopaedic unit in Athens, Greece. 86 patients with an displaced intracapsular fracture. Mean age: 74 years (range not stated). Percentage male: 28%. Follow up for a mean of 48 months. Loss to follow up: 2(4%). Inclusion criteria: age over 70 years, good cognitive function, moderately dependent, displaced intracapsular fracture Exclusion criteria: Fracture due to Padgets disease or tumour, rheumatoid arthritis | |
| Interventions | Merte hemiarthroplasty versus De Puy total hip replacement. It was not stated if the prosthesis were cemented in place | |
| Outcomes | Reoperation Length of hospital stay Mortality Range of movement Barthel score (1 and 3 years) Harris hip score (1 and 3 years) Walking speed | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
Parker 2009.
| Methods | Method of randomisation: sealed opaque envelopes independently prepared. | |
| Participants | Patients aged over 60 years with a displaced intracapsular fracture of the hip Orthopaedic unit in Peterborough, United Kingdom. 400 people with an intracapsular fracture Mean age: 83 years (range 61 to 104) Percentage male:23%. Follow up for 2‐5 years. Loss to follow up: 4(1%). | |
| Interventions | Cemented Thompson prosthesis versus uncemented Austin Moore | |
| Outcomes | Length of surgery Length of anaesthesia Transfusion requirements Number of patients transfused Dislocation Operative fracture of the femur Later fracture of femur Other operative complications Reoperation Superficial sepsis Deep sepsis Wound haematoma Deep vein thrombosis Pulmonary embolism Pneumonia Pressure sores All medical complications Length of hospital stay Shortening of the limb Loss of limb flexion Mortality Pain score Number of patients with residual pain (pain level of pain when starting walking and occasional analgesia or higher) Failure to return to same residential Failure to regain mobility (patients with the same or an improved mobility score at one year) Regain of activities of daily living | |
| Notes | Additional trial details provided by M Parker | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Low risk | A ‐ Adequate |
Raia 2003.
| Methods | Method of randomisation: computer generated random numbers. | |
| Participants | Orthopaedic unit in New York, USA. 115 patients with a intracapsular fracture. Mean age: 82 years (range 65 to101). Percentage male: 28%. Follow up: one year. Loss to follow up: 13 (11%). | |
| Interventions | Cemented Unitrax unipolar prosthesis versus cemented Centrax bipolar prosthesis (Howmedica Rutherford, NJ). All inserted via a posterior‐lateral surgical approach. | |
| Outcomes | Operative blood loss Number of patients transfused Dislocation Deep wound infection Reoperation Length of acute hospital stay Mortality Failure to regain mobility Short form 36 (SF36) assessment score Musculoskeletal functional assessment score | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
Rosen 1992.
| Methods | Method of randomisation: not stated. | |
| Participants | Orthopaedic unit in Montreal, Canada. 96 patients with 102 intracapsular fractures. Mean age: not stated (range not stated). Percentage male: not stated. Follow up for two years. Loss to follow up: four patients lost to follow up but by two years only 25 patients with 27 arthroplasties were reviewed due to deaths and loss to follow up. | |
| Interventions | Austin Moore prosthesis versus Bateman prosthesis. Not stated if with or without cement or which surgical approach used. | |
| Outcomes | Length of operation Operative blood loss Intra‐operative complications Post‐operative complications Reoperation Mortality Pain at follow up Mobility | |
| Notes | Trial only reported as a conference abstract with limited information. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
Santini 2005.
| Methods | Method of randomisation: alternate days. | |
| Participants | Orthopaedic unit in Vicenza, Italy. 106 patients with an intracapsular fracture. Mean age: 81 years. Percentage male: 23%. Follow up for 12 months. Loss to follow up: not stated. | |
| Interventions | Cemented bipolar hemiarthroplasty versus uncemented bipolar hemiarthroplasty. Surgical approach lateral for all cases. | |
| Outcomes | Dislocation Operative fracture femur Units of blood transfused Deep wound infection Cardiac complications Gastric complications Pulmonary embolism/pneumonia Pressure sores Length of hospital stay Cost of treatment Mortality at one year Functional score based on walking, personal and daily activities and living conditions | |
| Notes | ||
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | High risk | C ‐ Inadequate |
Skinner 1989.
| Methods | Method of randomisation: day of week when admitted. | |
| Participants | Orthopaedic unit in Croydon, UK. 180 people with an acute intracapsular fracture. Mean age: 81 years (range: not stated). Percentage male: 10%. Follow up for 12 months. Loss to follow up: 7% were lost to follow up by 13 years. Loss to follow up at one year not stated. | |
| Interventions | Uncemented Austin Moore prosthesis versus cemented Howes II total hip replacement inserted. All prosthesis inserted via a posterior‐lateral approach. | |
| Outcomes | Dislocation Deep wound infection Incidence of second anaesthetic Reoperation Mortality Pain at follow up Mobility Harris Hip Score | |
| Notes | Trial also included a third group which was internal fixation. These patients have been considered in a separate Cochrane review "Internal fixation versus arthroplasty for intracapsular proximal femoral fractures in adults". The report of Ravikumar 1998 is a long term follow‐up report of patients. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | High risk | C ‐ Inadequate |
Sonne‐Holm 1982.
| Methods | Method of randomisation: not stated. | |
| Participants | Orthopaedic unit in Hellerup, Denmark. 112 patients with an acute intracapsular fracture. Mean age: 76 years (study criteria was patients aged over 70 years but range was 62 to 95 years). Percentage male: 33%. Follow up for 12 months. The results for 37 participants were excluded as they had either died, moved away, had the wrong implant inserted or refused to attend. | |
| Interventions | Cemented Moore prosthesis versus Moore prosthesis without cement. All prosthesis inserted using a posterior approach. | |
| Outcomes | Superficial wound infection Deep wound infection Mortality at 6 weeks Pain Hip movement Mobility Bone scintimetry | |
| Notes | The bone scintimetry was for a subgroup of 29 cases and reported as a separate article (Sonne‐Holm 1993). | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
STARS 2006.
| Methods | Randomisation by: computer telephone randomisation service. | |
| Participants | 11 hospitals in Scotland, UK. 138 patients with an acute intracapsular fracture. Mean age: 75 years (range 60 to 93). Male: 32 (23%). Follow up for 24 months. Loss to follow up: 0.7% Inclusion criteria: mobile, mental text score equal to or more than 7/10, age 60 and above, no serious concomitant disease, displaced intracapsular fracture Exclusion criteria: those not satisfying the above criteria | |
| Interventions | Cemented bipolar hemiarthroplasty versus cemented total hip replacement. | |
| Outcomes | Length of surgery in minutes Dislocation Reoperation Superficial wound infection Deep sepsis Deep vein thrombosis (DVT) Pulmonary embolism Cerebrovascular accident Septicaemia Other medical complications Mortality at 4, 12, 24 months Pain at 4, 12, 24 months Walking score at 4,12, 24 months Function score at 4,12, 24 months EQ5D score at 4,12, 24 months Estimated cost for the trial procedures | |
| Notes | Additional information supplied by trialists. Results of comparison with internal fixation were considered in the Cochrane review "Internal fixation versus arthroplasty for intracapsular proximal femoral fractures in adults". Four of the hemiarthroplasty cases were unipolar rather than bipolar. Approximately 10% of those allocated to THR received a bipolar hemiarthroplasty and a further 6% received a unipolar hemiarthroplasty. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Low risk | A ‐ Adequate |
Stock 1997.
| Methods | Method of randomisation: not stated. | |
| Participants | Orthopaedic unit in Western Australia. 69 patients with a hip fracture. Mean age: not stated (range not stated). Percentage male: not stated. Follow up and loss to follow up: not stated. | |
| Interventions | Ceramic head Thompson hemiarthroplasty versus metal head Thompson. Not stated if with or without cement or which surgical approach used. | |
| Outcomes | Harris Hip Score | |
| Notes | Trial only reported as a conference abstract with limited information. | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
Van Thiel 1988.
| Methods | Method of randomisation: not stated. | |
| Participants | Orthopaedic unit in Amsterdam, Holland. 93 patients with an intracapsular fracture. Mean age: not stated (range not stated). Percentage male: not stated. Follow up for three years. Loss to follow up: not stated. | |
| Interventions | Austin Moore prosthesis versus Bateman prosthesis. Not stated if with or without cement or which surgical approach used. | |
| Outcomes | Harris Hip Score Radiographic findings | |
| Notes | Trial only reported as a conference abstract with limited information | |
| Risk of bias | ||
| Bias | Authors' judgement | Support for judgement |
| Allocation concealment? | Unclear risk | B ‐ Unclear |
Characteristics of excluded studies [ordered by study ID]
| Study | Reason for exclusion |
|---|---|
| Christie 1994 | This randomised trial compared 10 patients allocated to an uncemented Austin Moore hemiarthroplasty with 10 to a cemented Hastings hemiarthroplasty. The outcome measures were emboli within the heart chambers as seen on trans‐oesophageal echocardiography and fat in blood samples taken from the right atrium. Cemented arthroplasty produced greater and more prolonged embolic cascades, pulmonary hypertension, diminished oxygen tension and saturation. This biometric study was excluded due to the absence of clinical outcome measures. |
| Clark 2001 | This was a prospective controlled study for 20 patients, treated with either a cemented or an uncemented hemiarthroplasty. Cardiac output was measured using a transoesophageal Doppler probe. For those treated using cement there was a transient but significant reduction in cardiac output and stroke volume. The study was excluded as there was no randomisation of patients. |
| Faraj 1999 | This study involved 101 patients with an intracapsular femoral neck fracture treated by Thompson's prosthesis. The prosthesis was fixed in the femoral shaft with cement in 23 people and was inserted uncemented in 78 people. There was no statistically significant difference between groups for the Harris Hip Scores or any of the individual components of this score. The study was excluded as there was no randomisation of patients; the use of cement was at the operating surgeons' preference. |
| Field 2005 | This was a randomised study of 24 patients with a displaced intracapsular fracture treated with a cemented Thompson hemiarthroplasty and a hydroxyapatite coated Cambridge acetabular cup against 26 treated with the same implants but the acetabular cup was not coated with hydroxyapatite. Five revision operations were required in the group without the hydroxyapatite coating against none in the other group. The study was excluded because of the limited number of cases using what is at present an experimental new cup. |
| Georgescu 2004 | This study was reported only as a conference abstract as a randomised trial comparing a unipolar with a bipolar prosthesis for 122 participants. The mean age of patient was 62 years. Twenty four patients died during the follow‐up period and 32 were lost to follow‐up. For the 66 patients assessed at a minimum of 18 months after surgery there was no reported difference in functional status, incidence of complications or need for revision. The study was excluded because of a lack of reported results within the conference abstract. |
| Gierer 2002 | This report compared 35 patients treated with a cemented hip prosthesis with 37 patients with a cementless modular hip prosthesis. Both groups had no difference in their Harris Hip Score at follow up. The study was excluded because the decision on the use of cement was at the surgeon's preference and there was no randomisation of patients. |
| Graf 2000 | This was a comparison of 45 cemented prostheses with 44 uncemented prostheses. The study was excluded as it was not a randomised study. |
| Johnson 2001 | This study reported a series of 80 patients with a femoral neck fracture with the first 40 treated with a uncemented unipolar prosthesis and the next 40 with a cemented unipolar prosthesis. The study was excluded as it was not a randomised trial. |
| Karpman 1992 | This study was only reported in the conference proceedings of the American Academy of Orthopaedic Surgeons. The study involved 125 patients randomised to either an Austin Moore prosthesis, cemented bipolar or uncemented bipolar. Follow up of patients ranged from two to five years (mean 3.7 years). Results were presented without data and stated that there was "no significant difference between any of the groups with regard to functional outcome." The study was excluded, as there was inadequate reporting of the trial. Attempts were made to contact the trialists for further information, without success. |
| Leidinger 2002 | This was a randomised study of 72 patients. In the control group of 36 patients, surgical repair was performed with palacos mixed conventionally. In the second group (vacuum group) of 36 patients, surgical repair was performed with palacos mixed in vacuum. Invasive haemodynamic monitoring and transoesophageal echocardiography was performed in all cases. Those in the vacuum group had a lower incidence of echocardiographical changes (86% versus 14%) and clinical complications (53% versus 11%). Mortality in the control group was 14% compared with 3% in the vacuum group. The study was excluded, as it was not within the remit of this review which is choice of arthroplasty or use of cement. Variations of cementing technique are not included. |
| Pitto 2000 | This was a randomised study of 40 patients in which, for 20 patients, the femoral component was cemented using a contemporary technique. For the other 20 patients, a drainage hole was placed in the trochanteric region, along the prolongation of the linea aspera and a distal hole was placed 2 cm below the tip of the femoral component. The medullary cavity was then aspirated during cement insertion. Embolic phenomena were documented intraoperatively by continuous transoesophageal echocardiographic imaging of the right atrium and ventricle. Patients in the control group showed more severe and longer‐lasting episodes of embolism than patients of the bone vacuum group. No other outcome measures were given. The study was excluded because of the small numbers and lack of other outcome measures. |
| Sadr 1977 | This was a comparison of 20 patients with an intracapsular fracture treated using Thompson prosthesis coated with Proplast, compared with 20 patients treated using a cemented Thompson prosthesis. The study stated patients were allocated to one of the groups by a "random selection", but it remained unclear as to the exact method of randomisation. Radiographic loosening occurred in one case of the cemented group versus nine in the Proplast group. Two cases of ectopic calcification and one dislocation were also reported in the Proplast group. Mortality at one year was four in the Proplast group and nine in the uncemented group. Functional outcome for the survivors (excluding the two patients lost to follow up) showed no difference between groups. The study was excluded for the following reasons. 1. It was not possible to be confident that this was a randomised study rather than a case comparison study. 2. The use of Proplast coated prosthesis is no longer prevalent. 3. The total number of patients in the study is small (40) with limited reporting of outcomes for the 25 patients assessed at follow up. |
Characteristics of ongoing studies [ordered by study ID]
Bonke 1999.
| Trial name or title | Hemiarthroplasty or total hip replacement for femoral neck fractures. |
| Methods | |
| Participants | Patients aged 70 or over with a displaced intracapsular fracture of the hip. Target sample size 400 participants. |
| Interventions | Hemiarthroplasty versus total hip replacement |
| Outcomes | Annual follow up for five years |
| Starting date | March 1995 |
| Contact information | |
| Notes | English abstract |
Kavcic 2006.
| Trial name or title | Hemiarthroplasty versus total hip arthroplasty for femoral neck fractures |
| Methods | Randomly selected patients |
| Participants | Active elderly patients with a displaced intracapsular fracture |
| Interventions | Bipolar hemiarthroplasty versus total hip arthroplasty |
| Outcomes | Harris hip score, revision surgery |
| Starting date | 1.1.2003 |
| Contact information | |
| Notes | Reported only as conference abstract with no contact details |
Moroni 2002.
| Trial name or title | Cemented vs uncemented fixation of femoral neck fractures in osteoporotic patients. A prospective randomised study |
| Methods | |
| Participants | Elderly patients with a displaced femoral neck fracture |
| Interventions | Cemented arthroplasty versus HA coated uncemented implant |
| Outcomes | Harris Hip score Mortality Dislocation Revision surgery |
| Starting date | |
| Contact information | |
| Notes |
Patel 2008.
| Trial name or title | Bipolar versus unipolar for displaced intracapsular fractures |
| Methods | Randomised study |
| Participants | Patients aged over 70 years |
| Interventions | Bipolar versus Thompson prosthesis |
| Outcomes | Mortality, hospital stay, pain |
| Starting date | |
| Contact information | |
| Notes |
Taylor.
| Trial name or title | A randomised trial of cemented vs uncemented hemiarthroplasty in elderly patients |
| Methods | Randomised trial |
| Participants | Patients with a displaced subcapital fracture |
| Interventions | Cemented versus uncemented hemiarthroplasty |
| Outcomes | Pain, mobility |
| Starting date | |
| Contact information | |
| Notes |
Contributions of authors
Martyn Parker initiated and designed the review, extracted data, contacted trialists for further information and compiled the review. For the fourth update, Kurinchiselvan Gurusamy and Shin Azegami independently extracted data, checked the text and assisted in writing the update. Martyn Parker is the guarantor of the review.
Sources of support
Internal sources
Peterborough and Stamford Hospitals NHS Foundation Trust, UK.
External sources
No sources of support supplied
Declarations of interest
The lead review author, Dr Martyn Parker, is the principal investigator of one of the trials included in this review (Parker 2009). Data for this review and its assessment was undertaken by the other two authors for this review. In addition Martyn Parker has received payment and reimbursement from a number of commercial companies for attending meetings to give lectures or advising on different aspects of hip fracture treatment. Martyn Parker has also received royalties for the design and development of an implant for the internal fixation of intracapsular hip fractures from the company BBraun.
Edited (conclusions changed)
References
References to studies included in this review
Baker 2006 {published and unpublished data}
- Baker RP, Squires B, Gargan MF, Bannister GC. A randomised controlled comparison of total hip arthroplasty and hemiarthroplasty in mobile independent patients with displaced intracapsular femoral neck fracture [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 2006;88‐B(Supp III):431‐2. [DOI] [PubMed] [Google Scholar]
- Baker RP, Squires B, Gargan MF, Bannister GC. A randomised controlled comparison of total hip arthroplasty and hemiarthroplasty in mobile independent patients with displaced intracapsular femoral neck fracture [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 2008;90‐B(Suppl II):301‐2. [Google Scholar]
- Baker RP, Squires B, Gargan MF, Bannister GC. Total hip arthroplasty and hemiarthroplasty in mobile, independent patients with a displaced fracture of the femoral neck; a randomized, controlled trial. Journal of Bone and Joint Surgery ‐ American Volume 2006;88:2583‐9. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
Blomfeldt 2007 {published data only}
- Blomfeldt R, Tornkvist H, Eriksson K, Soderqvist A, Ponzer S, Tidermark J. A randomised trial comparing bipolar hemiarthroplasty with total hip replacement for displaced intracapsular fractures of the femoral neck in elderly patients. Journal of Bone and Joint Surgery ‐ British Volume 2007;89‐B(2):160‐5. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
- Blomfeldt R, Tornkvist H, Eriksson K, Soderqvist A, Ponzer S, Tidermark J. Bipolar hemiarthroplasty compared with total hip replacement for displaced femoral neck fractures in the elderly. A randomised controlled trial [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 2009;91‐B(Suppl I):169. [Google Scholar]
Branfoot 2000 {published and unpublished data}
- Branfoot T, Faraj AA, Porter P. Cemented versus uncemented Thompson's prosthesis: a randomised prospective functional outcome study [abstract]. Injury 2000;31:280‐1. [Google Scholar]
Calder 1996 {published data only}
- Calder SJ, Anderson GH, Gregg PJ. Bipolar hemiarthroplasty for displaced intracapsular hip fractures: a randomised prospective trial [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1995;77(Suppl II):214. [PubMed] [Google Scholar]
- Calder SJ, Anderson GH, Harper WM, Gregg PJ. The place of bipolar hemiarthroplasty in displaced intracapsular hip fracture [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1995;77(Suppl I):6. [Google Scholar]
- Calder SJ, Anderson GH, Harper WM, Gregg PJ. The use of bipolar hemi‐arthroplasty in intracapsular hip fracture ‐ a prospective randomised trial [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1995;77(Suppl I):103. [Google Scholar]
- Calder SJ, Anderson GH, Harper WM, Gregg PJ. The use of the Nottingham Health Profile in a randomised trial of treatment of displaced intracapsular hip fractures [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1994;76(Suppl II & III):150. [Google Scholar]
- Calder SJ, Anderson GH, Harper WM, Jagger C, Gregg PJ. A subjective health indicator for follow‐up: A randomised trial after treatment of displaced intracapsular hip fractures. Journal of Bone and Joint Surgery ‐ British Volume 1995;77‐B(3):494‐6. [PubMed] [Google Scholar]
- Calder SJ, Anderson GH, Jagger C, Harper WM, Gregg PJ. Unipolar or bipolar prosthesis for displaced intracapsular hip fracture in octogenarians. A randomised prospective study. Journal of Bone and Joint Surgery ‐ British Volume 1996;78(3):391‐4. [MEDLINE: ] [PubMed] [Google Scholar]
- Warwick D, Learmonth ID, Whitehouse S. Unipolar or bipolar prosthesis for displaced intracapsular hip fracture. Journal of Bone and Joint Surgery ‐ British Volume 1997;79(4):694‐5. [PubMed] [Google Scholar]
Cornell 1988 {published data only}
- Cornell CN, Levine D, Odoherty J, Lyden J. Unipolar versus bipolar hemiarthroplasty for the treatment of femoral neck fractures in the elderly. Clinical Orthopaedics and Related Research 1998;(348):67‐71. [MEDLINE: ] [PubMed] [Google Scholar]
Davison 2001 {published data only}
- Calder SJ, Anderson GH, Gregg PJ. Bipolar hemiarthroplasty for displaced intracapsular hip fractures: a randomised prospective trial [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1995;77(Suppl II):214. [PubMed] [Google Scholar]
- Calder SJ, Anderson GH, Harper WM, Gregg PJ. The place of bipolar hemiarthroplasty in displaced intracapsular hip fracture [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1995;77(Suppl I):6. [Google Scholar]
- Calder SJ, Anderson GH, Harper WM, Gregg PJ. The use of bipolar hemi‐arthroplasty in intracapsular hip fracture ‐ a prospective randomised trial [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1995;77‐B(Suppl I):103. [Google Scholar]
- Calder SJ, Anderson GH, Harper WM, Gregg PJ. The use of the Nottingham Health Profile in a randomised trial of treatment of displaced intracapsular hip fractures [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1994;76(Suppl II & III):150. [Google Scholar]
- Calder SJ, Anderson GH, Harper WM, Jagger C, Gregg PJ. A subjective health indicator for follow‐up: A randomised trial after treatment of displaced intracapsular hip fractures. Journal of Bone and Joint Surgery ‐ British Volume 1995;77(3):494‐6. [PubMed] [Google Scholar]
- Davison J, Harper WM, Gregg PJ. Which treatment for the displaced fractures of the femoral neck? A prospective randomised comparison of three surgical procedures [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1997;79(Suppl II):243‐4. [Google Scholar]
- Davison JN, Calder SJ, Anderson GH, Ward G, Jagger C, Harper WM, et al. Treatment for displaced intracapsular fracture of the proximal femur: a prospective, randomised trial in patients aged 65 to 79 years. Journal of Bone and Joint Surgery ‐ British Volume 2001;83(2):206‐12. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
- Davison JNS, Harper WM, Gregg PJ. Treatment for displaced intracapsular fracture of the proximal femur. A prospective randomised trial in patients aged from 65 to 79 years [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1999;81(Suppl II):232. [DOI] [PubMed] [Google Scholar]
Dorr 1986 {published data only}
- Dorr LD, Glousman R, Hoy AL, Vanis R, Chandler R. Treatment of femoral neck fractures with total hip replacement versus cemented and noncemented hemiarthroplasty. Journal of Arthroplasty 1986;1(1):21‐8. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
Emery 1991 {published data only}
- Emery RJ, Broughton NS, Desai K, Bulstrode CJ, Thomas TL. Bipolar hemiarthroplasty for subcapital fracture of the femoral neck. A prospective randomised trial of cemented Thompson and uncemented Moore stems. Journal of Bone and Joint Surgery ‐ British Volume 1991;73(2):322‐4. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
Figved 2009 {published data only}
- Figved W, Opland V, Frihagen F, Jervidalo T, Madsen JE, Nordsletten L. Cemented versus uncemented hemiarthroplasty for displaced femoral neck fractures. Clinical Orthopaedics and Related Research 2009;467(9):2426‐35. [MEDLINE: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Harper 1994 {published data only}
- Anderson GH, Dias JJ, Hoskinson J, Harper WM. A randomized study of the use of bone cement with Thompson's prosthesis in the treatment of intracapsular fractures of the femoral neck [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1992;74‐B(Suppl II):132‐3. [Google Scholar]
- Harper WM. Treatment of intracapsular proximal femoral fractures. A prospective randomised trial comparing cemented and uncemented Thompson hemiarthroplasty in the treatment of displaced intracapsular proximal femoral fractures [dissertation]. Leicester (UK): Univ. of Leicester, 1994. [Google Scholar]
- Harper WM, Gregg PJ. The treatment of intracapsular proximal femoral fractures: A randomized prospective trial [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1992;74(Suppl III):282. [Google Scholar]
Livesley 1993 {published data only}
- Livesley PJ, Srivastiva VM, Needoff M, Prince HG, Moulton AM. Use of a hydroxyapatite‐coated hemiarthroplasty in the management of subcapital fractures of the femur. Injury 1993;24(4):236‐40. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
Macaulay 2008 {published data only}
- Macaulay W, Nellans KW, Garvin KL, Iorio R, Healy WL, Rosenwasser MP, et al. Prospective randomized clinical trial comparing hemiarthroplasty to total hip arthroplasty in the treatment of displaced femoral neck fractures: winner of the Dorr Award. Journal of Arthroplasty 2008;23(6 Suppl I):2‐8. [DOI] [PubMed] [Google Scholar]
Malhotra 1995 {published data only}
- Malhotra R, Arya R, Bhan S. Bipolar hemiarthroplasty in femoral neck fractures. Archives of Orthopaedic & Trauma Surgery 1995;114(2):79‐82. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
Mouzopoulos 2008 {published data only}
- Mouzopoulos G, Stamatakos M, Arabatzi H, Vasiliadis G, Batanis G, Tsembeli A, et al. The four‐year function results after a displaced subcapital fracture treated with three different surgical options. International Orthopaedics 2008;32(3):367‐73. [MEDLINE: ] [DOI] [PMC free article] [PubMed] [Google Scholar]
Parker 2009 {published and unpublished data}
- Haleem S, Pryor GA, Parker MJ. Randomised controlled trial of cemented versus uncemented hemiarthroplasty for displaced intracapsular fractures [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 2008;90‐B(Suppl III):535. [Google Scholar]
- Haleem S, Pryor GA, Parker MJ. Randomised controlled trial of cemented versus uncemented hemiarthroplasty for displaced intracapsular fractures [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 2009;91‐B(Suppl II):254. [Google Scholar]
- Parker M. Trial of cemented versus uncemented arthroplasty for intracapsular hip fractures. National Research Register (NRR) Archive. Available at https://portal.nihr.ac.uk/Profiles/NRR.aspx?Publication_ID=N0181123527 (accessed 16 September 2009).
Raia 2003 {published data only}
- Chapman CB, Schweppe MW, Raia FJ, Rosenwasser MP. Bipolar versus unipolar prosthesis for displaced femoral neck fractures: is there at difference in quality of life and functional outcome?. American Academy of Orthopaedic Surgeons 68th Annual Meeting; 2001 Feb 28‐March 4;San Francisco (CA). 2001:http://www.aaos.org/wordhtml/anmt2001/poster/pe054.htm (accessed 30/11/03).
- Raia FJ, Chapman CB, Herrera MF, Schweppe MW, Michelsen CB, Rosenwasser MP. Unipolar or bipolar hemiarthroplasty for femoral neck fractures in the elderly?. Clinical Orthopaedics and Related Research 2003;(414):259‐65. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
- Schweppe MW, Vitale MG, Yaron I, Herrera MF, Sollano JA, Michelsen CB, et al. Randomized evaluation of bipolar versus unipolar hemiarthroplasty for displaced femoral neck fractures in the elderly: preliminary results [abstract]. Trauma Association Annual Meeting;1998 Oct 8‐10; Vancouver (BC). 1998:http://www.ota.org/education/amabstracts.htm (accessed 25/11/03).
Rosen 1992 {published data only}
- Rosen LL, Miller BJ, Dupuis PR, Jarzem P, Hadjipavlou A. A prospective randomized study comparing bipolar hip arthroplasty and hemiarthroplasty in elderly patients with subcapital fractures [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1992;74(Suppl III):282. [Google Scholar]
Santini 2005 {published data only}
- Santini S, Rebeccato A, Bolgan I, Turi G. Hip fractures in elderly patients treated with bipolar hemiarthroplasty: comparison between cemented and cementless implants. Journal of Orthopaedics & Traumatology 2005;6(2):80‐7. [EMBASE: 2005334475] [Google Scholar]
Skinner 1989 {published data only}
- Ravikumar KJ, Marsh G, Jairaj P. Internal fixation vs hemiarthroplasty vs total hip replacement for displaced subcapital fractures of the femur ‐ 13 year results of a prospective randomised study [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1998;80(Suppl 1):50‐1. [Google Scholar]
- Skinner FW, Ellory J, Riley D, Beaumont A. Displaced subcapital fractures of the femur: which operation? [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1988;70(4):677. [Google Scholar]
- Skinner P, Riley D, Ellery J, Beaumont A, Coumine R, Shafighian B. Displaced subcapital fractures of the femur: a prospective randomized comparison of internal fixation, hemiarthroplasty and total hip replacement. Injury 1989;20(5):291‐3. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
Sonne‐Holm 1982 {published data only}
- Sonne‐Holm S, Dyrbye M, Walter S, Jensen JS. Bone scintigraphy in Moore hemiarthroplasty with and without cement following femoral neck fractures. A controlled study. Acta Orthopaedica Scandinavica 1983;54(2):194‐7. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
- Sonne‐Holm S, Walter S, Jensen JS. Moore hemi‐arthroplasty with and without bone cement in femoral neck fractures. A clinical controlled trial. Acta Orthopaedica Scandinavica 1982;53(6):953‐6. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
STARS 2006 {published data only}
- Keating JF. Scottish trial of arthroplasty or reduction for subcapital fractures (STARS). controlled‐trials.com/ISRCTN32884890 (accessed 20 September 2009).
- Keating JF, Grant A, Masson M, Scott NW, Forbes JF. Displaced intracapsular hip fractures in fit, older people: a randomised comparison of reduction and fixation, bipolar hemiarthroplasty and total hip arthroplasty. Health Technology Assessment 2005; Vol. 9, issue 41:iii‐iv, ix‐x, 1‐65. Available from http://www.ncchta.org/execsumm/summ941.htm. [MEDLINE: ] [DOI] [PubMed]
- Keating JF, Grant A, Masson M, Scott NW, Forbes JF. Randomized comparison of reduction and fixation, bipolar hemiarthroplasty, and total hip arthroplasty. Treatment of displaced intracapsular hip fractures in healthy older patients. Journal of Bone and Joint Surgery ‐ American Volume 2006;88(2):249‐60. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
- Keating JF, Masson MA, Forbes JF, Scott NW, Grant A. Randomized trial of reduction and fixation versus bipolar hemiarthroplasty versus total hip arthroplasty for displaced subcapital fractures in the fit older patient [abstract]. Orthopaedic Trauma Association Annual Meeting; 2002 October 11‐13; Toronto (Ontario). www.hwbf.org/ota/am/ota02/otapa/OTA02641.htm (accessed 17 June 2004).
Stock 1997 {published data only}
- Stock M, Halliday M, Wood D, Brennon J, Smith A. A randomised prospective trial comparing ceramic and Thompsons hemiarthroplasty in subcapital femoral fractures [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1997;79(Suppl IV):386. [Google Scholar]
Van Thiel 1988 {published data only}
- Thiel PH, Snellen JP, Jansen WBJ, Slikke W. Moore prosthesis versus bipolar Bateman prosthesis: a prospective randomised clinical study [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 1988;70(4):677. [Google Scholar]
References to studies excluded from this review
Christie 1994 {published data only}
- Christie J, Burnett R, Potts HR, Pell AC. Echocardiography of transatrial embolism during cemented and uncemented hemiarthroplasty of the hip. Journal of Bone and Joint Surgery ‐ British Volume 1994;76(3):409‐12. [MEDLINE: ] [PubMed] [Google Scholar]
Clark 2001 {published data only}
- Clark DI, Ahmed AB, Baxendale BR, Moran CG. Cardiac output during hemiarthroplasty of the hip. A prospective, controlled trial of cemented and uncemented prostheses. Journal of Bone and Joint Surgery ‐ British Volume 2001;83(3):414‐8. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
Faraj 1999 {published data only}
- Faraj AA, Branfoot T. Cemented versus uncemented Thompson's prostheses: a functional outcome study. Injury 1999;30(10):671‐5. [EMBASE: 1999401247] [DOI] [PubMed] [Google Scholar]
Field 2005 {published data only}
- Field RE, Rushton N. Five‐year clinical, radiological and post‐mortem results of the Cambridge Cup in patients with displaced fractures of the neck of the femur. Journal of Bone and Joint Surgery ‐ British Volume 2005;87‐B(10):1344‐51. [EMBASE: 2005481155] [DOI] [PubMed] [Google Scholar]
Georgescu 2004 {published data only}
- Georgescu N, Iancu C, Trandabat C, Sirbu P, Alexa O. A prospective randomised study comparing two types of hemiarthroplasty for displaced fractures of the femoral neck in elderly patients [abstract]. Journal of Bone and Joint Surgery ‐ British Volume 2004;86‐B(Suppl III):338. [Google Scholar]
Gierer 2002 {published data only}
- Gierer P, Landes J, Grubwinkler M, Gradl G, Lob G, Andress H‐J. The femoral neck fracture in the elderly patient ‐ Cemented or cementless hip arthroplasty? [Die Schenkelhalsfraktur des alten menschenzementierte oder unzementierte endoprothetische versorgung?]. Zentralblatt fur Chirurgie 2002;127(6):514‐8. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
Graf 2000 {published data only}
- Graf C, Scherer MA, Gumppenberg S. Cemented vs. cement‐free endoprothetics in the traumatology of older people [Zementierte vs. zementfreie endoprothetik in der traumatologie des alten menschen]. Hefte zur der Unfallchirurg 2000;282:343. [Google Scholar]
Johnson 2001 {published data only}
- Johnson MM, Thompson B, Hartford JM. Complications of cemented versus noncemented hemiarthroplasty for femoral neck fractures [abstract]. Mid‐America Orthopaedic Association 19th annual meeting; 2001 April 18‐22; Amelia Island (FL). http://www.jbjs.org/abstracts/maoa01/abs_001.htm (accessed 24/11/03).
Karpman 1992 {published data only}
- Karpman RR, Lee TJ, Moore BM. Austin‐Moore versus bipolar hemiarthroplasty for displaced femoral neck fractures: a randomized prospective study [abstract]. Annual Meeting of the American Academy of Orthopaedic Surgeons; 1992 Feb 20‐25; Orlando (FL). http://www.aaos.org/wordhtml/anmeet92/scipro/ppr418.htm (accessed 23 February 2001).
- Karpman RR, Lee TK, Moore BM. Austin‐Moore versus bipolar hemi‐arthroplasty for displaced femoral neck fractures: a randomized prospective study [abstract]. Orthopaedic Transactions 1992;16:749‐50. [Google Scholar]
Leidinger 2002 {published data only}
- Leidinger W, Hoffmann G, Meierhofer JN, Wolfel R. Reduction of severe cardiac complications during implantation of cemented total hip endoprostheses in femoral neck fractures [Verminderung von schweren kardialen Komplikationen wahrend der Implantation von zementierten Hufttotalendoprothesen bei Oberschenkelhalsfrakturen]. Unfallchirurg 2002;105(8):675‐9. [DOI] [PubMed] [Google Scholar]
Pitto 2000 {published data only}
- Pitto RP, Blunk J, Kossler M. Transesophageal echocardiography and clinical features of fat embolism during cemented total hip arthroplasty. A randomized study in patients with a femoral neck fracture. Archives of Orthopaedic and Trauma Surgery 2000;120(1‐2):53‐8. [DOI] [PubMed] [Google Scholar]
Sadr 1977 {published data only}
- Sadr B, Arden GP. A comparison of the stability of Proplast‐coated and cemented Thompson prostheses in the treatment of subcapital femoral fractures. Injury 1977;8(3):234‐7. [MEDLINE: ] [DOI] [PubMed] [Google Scholar]
References to ongoing studies
Bonke 1999 {published data only}
- Bonke H, Schnater J, Kleijnen J, Raaymakersk E. Hemiarthroplasty or total hip replacement for femoral neck fractures. A preliminary report of a randomized trial [abstract]. Hefte zur der Unfallchirurg 1999;272:176‐7. [Google Scholar]
Kavcic 2006 {published data only}
- Kavcic G, Hudoklin P, Mikek M, Hussein M. Hemiarthroplasty versus total arthroplasty for treatment of femoral neck fractures. European Journal of Trauma 2006;32(Supplement I):24. [Google Scholar]
Moroni 2002 {published data only}
- Moroni A, Pegreffi F, Romagnoli M, Giannini S. Cemented vs uncemented fixation of femoral neck fractures in osteoporotic patients. A prospective randomized study [abstract]. Hip International 2002;12(2):245‐6. [Google Scholar]
- Moroni A, Pegreffi F, Romagnoli M, Hoang‐Kim A, Tesei F, Giannini S. Results in osteoporotic femoral neck fractures treated with cemented vs uncemented hip arthroplasty. Journal of Bone and Joint Surgery. British Volume 2009;91‐B(Suppl I):167. [Google Scholar]
Patel 2008 {published data only}
- Patel N, Brijlall S. Bipolar versus unipolar for displaced intracapsular hip fractures ‐ a preliminary report. Journal of Bone and Joint Surgery ‐ British Volume 2008;90‐B(Suppl III):473. [Google Scholar]
Taylor {published data only}
- Taylor F. A randomised clinical trial of pain and mobility following cemented vs uncemented hemiarthroplasty in elderly patients with displaced subcapital neck of femur fracture. Australian New Zealand Clinical Trials Registry. www.anzctr.org.au/ACTRN12609000367246.aspx (accessed 21 September 2009).
Additional references
Higgins 2003
- Higgins JPT, Thompson SG, Deeks JJ, Altman DG. Measuring inconsistency in meta‐analyses. BMJ 2003;327:557‐60. [DOI] [PMC free article] [PubMed] [Google Scholar]
Higgins 2006
- Higgins JPT, Green S, editors. Appendix 5b. Highly sensitive search strategies for identifying reports of randomized controlled trials in MEDLINE. Cochrane Handbook for Systematic Reviews of Interventions 4.2.6 [updated September 2006]. The Cochrane Collaboration, 2006. Available from www.cochrane.org/resources/handbook/hbook.htm.
Lefebvre 2008
- Lefebvre C, Manheimer E, Glanville J. Chapter 6: Searching for studies. 6.4.11: Search filters. In: Higgins JPT, Green S (editors).Cochrane Handbook for Systematic Reviews of Interventions Version 5.0.1 [updated September 2008]. The Cochrane Collaboration, 2008. Available from www.cochrane‐handbook.org.
Moore 1952
- Moore AT. Metal hip joint: a new self‐locking vitallium prosthesis. Southern Medical Journal 1952;45:1015‐9. [DOI] [PubMed] [Google Scholar]
Ravikumar 1998
- Ravikumar KJ, Marsh G, Jairaj P. Internal fixation vs hemiarthroplasty vs total hip replacement for displaced subcapital fractures of the femur ‐ 13 year results of a prospective randomised study [Abstract]. Journal of Bone and Joint Surgery. British Volume 1998;80(Suppl I):50‐1. [Google Scholar]
Thompson 1954
- Thompson FR. Two and a half years' experience with a vitallium intramedullary hip prosthesis. Journal of Bone and Joint Surgery. American Volume 1954;36:489‐500. [PubMed] [Google Scholar]
References to other published versions of this review
Parker 2001
- Parker MJ, Gurusamy K. Arthroplasties (with and without bone cement) for proximal femoral fractures in adults. Cochrane Database of Systematic Reviews 2001, Issue 3. [DOI: 10.1002/14651858.CD001706.pub1] [DOI] [PubMed] [Google Scholar]
Parker 2004
- Parker MJ, Gurusamy K. Arthroplasties (with and without bone cement) for proximal femoral fractures in adults. Cochrane Database of Systematic Reviews 2004, Issue 2. [DOI: 10.1002/14651858.CD001706.pub2] [DOI] [PubMed] [Google Scholar]
Parker 2006
- Parker MJ, Gurusamy K. Arthroplasties (with and without bone cement) for proximal femoral fractures in adults. Cochrane Database of Systematic Reviews 2006, Issue 3. [DOI: 10.1002/14651858.CD001706.pub3] [DOI] [PubMed] [Google Scholar]


