Abstract
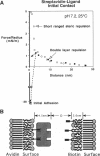
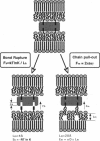
The strength of receptor-mediated cell adhesion is directly controlled by the mechanism of cohesive failure between the cell surface and underlying substrate. Unbinding can occur either at the locus of the specific bond or within the bilayer, which results in tearing the hydrophobic anchors from the membrane interior. In this work, the surface force apparatus has been used to investigate the relationship between the receptor-ligand bond affinities and the dominant mechanism of receptor-coupled membrane detachment. The receptors and ligands used in this study were membrane-bound streptavidin and biotin analogs, respectively, with solution affinities ranging over 10 orders of magnitude. With the optical technique of the surface force apparatus, the occurrence of membrane rupture was directly visualized in situ. The latter observations together with measurements of the corresponding intermembrane adhesive strengths were used to identify the dominant failure pathway for each streptavidin-analog pair. Even in cases where the membrane pull-out energy exceeded the equilibrium bond energy, cohesive failure occurred within the membrane interior at nearly all bond affinities considered. These results are consistent with previous findings and provide direct support for the commonly held view that, under nonequilibrium conditions of applied external stress, the gradient of the bond energy, not the equilibrium bond energy alone, determines the adhesive strength. Furthermore, our findings directly demonstrate that, in the presence of competing failure mechanisms, the preferred detachment mechanism- hence, the adhesive strength-will be determined by the bond that exhibits the weakest tensile strength. Because the tensile strength is determined by the gradient of the unbinding energy, the critical detachment force will be determined by both the bond energy and the effective bond length.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bell G. I. Models for the specific adhesion of cells to cells. Science. 1978 May 12;200(4342):618–627. doi: 10.1126/science.347575. [DOI] [PubMed] [Google Scholar]
- Blankenburg R., Meller P., Ringsdorf H., Salesse C. Interaction between biotin lipids and streptavidin in monolayers: formation of oriented two-dimensional protein domains induced by surface recognition. Biochemistry. 1989 Oct 3;28(20):8214–8221. doi: 10.1021/bi00446a037. [DOI] [PubMed] [Google Scholar]
- Chiruvolu S., Walker S., Israelachvili J., Schmitt F. J., Leckband D., Zasadzinski J. A. Higher order self-assembly of vesicles by site-specific binding. Science. 1994 Jun 17;264(5166):1753–1756. doi: 10.1126/science.8209255. [DOI] [PubMed] [Google Scholar]
- Darst S. A., Ahlers M., Meller P. H., Kubalek E. W., Blankenburg R., Ribi H. O., Ringsdorf H., Kornberg R. D. Two-dimensional crystals of streptavidin on biotinylated lipid layers and their interactions with biotinylated macromolecules. Biophys J. 1991 Feb;59(2):387–396. doi: 10.1016/S0006-3495(91)82232-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dembo M., Torney D. C., Saxman K., Hammer D. The reaction-limited kinetics of membrane-to-surface adhesion and detachment. Proc R Soc Lond B Biol Sci. 1988 Jun 22;234(1274):55–83. doi: 10.1098/rspb.1988.0038. [DOI] [PubMed] [Google Scholar]
- Evans E. A. Detailed mechanics of membrane-membrane adhesion and separation. II. Discrete kinetically trapped molecular cross-bridges. Biophys J. 1985 Jul;48(1):185–192. doi: 10.1016/S0006-3495(85)83771-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans E., Berk D., Leung A. Detachment of agglutinin-bonded red blood cells. I. Forces to rupture molecular-point attachments. Biophys J. 1991 Apr;59(4):838–848. doi: 10.1016/S0006-3495(91)82296-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Florin E. L., Moy V. T., Gaub H. E. Adhesion forces between individual ligand-receptor pairs. Science. 1994 Apr 15;264(5157):415–417. doi: 10.1126/science.8153628. [DOI] [PubMed] [Google Scholar]
- Green N. M. Avidin. Adv Protein Chem. 1975;29:85–133. doi: 10.1016/s0065-3233(08)60411-8. [DOI] [PubMed] [Google Scholar]
- Helm C. A., Knoll W., Israelachvili J. N. Measurement of ligand-receptor interactions. Proc Natl Acad Sci U S A. 1991 Sep 15;88(18):8169–8173. doi: 10.1073/pnas.88.18.8169. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaplanski G., Farnarier C., Tissot O., Pierres A., Benoliel A. M., Alessi M. C., Kaplanski S., Bongrand P. Granulocyte-endothelium initial adhesion. Analysis of transient binding events mediated by E-selectin in a laminar shear flow. Biophys J. 1993 Jun;64(6):1922–1933. doi: 10.1016/S0006-3495(93)81563-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuo S. C., Lauffenburger D. A. Relationship between receptor/ligand binding affinity and adhesion strength. Biophys J. 1993 Nov;65(5):2191–2200. doi: 10.1016/S0006-3495(93)81277-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Leckband D. E., Israelachvili J. N., Schmitt F. J., Knoll W. Long-range attraction and molecular rearrangements in receptor-ligand interactions. Science. 1992 Mar 13;255(5050):1419–1421. doi: 10.1126/science.1542789. [DOI] [PubMed] [Google Scholar]
- Leckband D. E., Schmitt F. J., Israelachvili J. N., Knoll W. Direct force measurements of specific and nonspecific protein interactions. Biochemistry. 1994 Apr 19;33(15):4611–4624. doi: 10.1021/bi00181a023. [DOI] [PubMed] [Google Scholar]
- Marra J., Israelachvili J. Direct measurements of forces between phosphatidylcholine and phosphatidylethanolamine bilayers in aqueous electrolyte solutions. Biochemistry. 1985 Aug 13;24(17):4608–4618. doi: 10.1021/bi00338a020. [DOI] [PubMed] [Google Scholar]
- Moy V. T., Florin E. L., Gaub H. E. Intermolecular forces and energies between ligands and receptors. Science. 1994 Oct 14;266(5183):257–259. doi: 10.1126/science.7939660. [DOI] [PubMed] [Google Scholar]
- Schmidt A., Spinke J., Bayerl T., Sackmann E., Knoll W. Streptavidin binding to biotinylated lipid layers on solid supports. A neutron reflection and surface plasmon optical study. Biophys J. 1992 Nov;63(5):1385–1392. doi: 10.1016/S0006-3495(92)81715-0. [DOI] [PMC free article] [PubMed] [Google Scholar]