Abstract
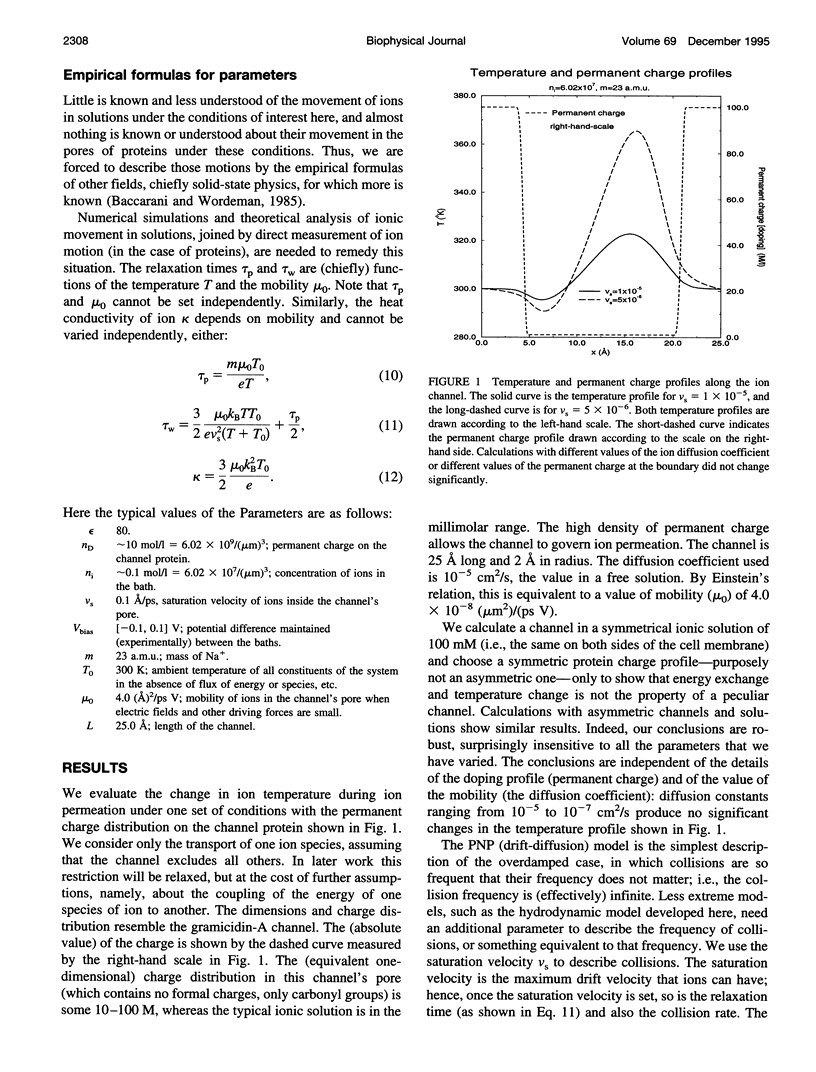
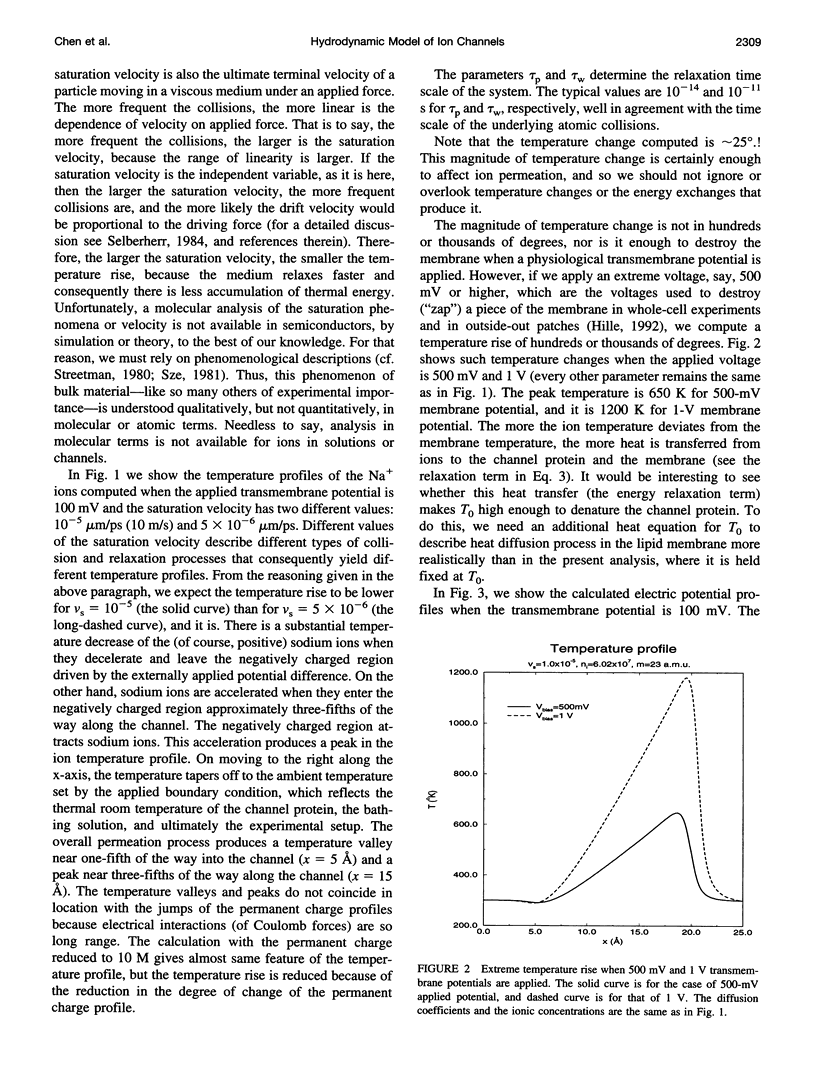
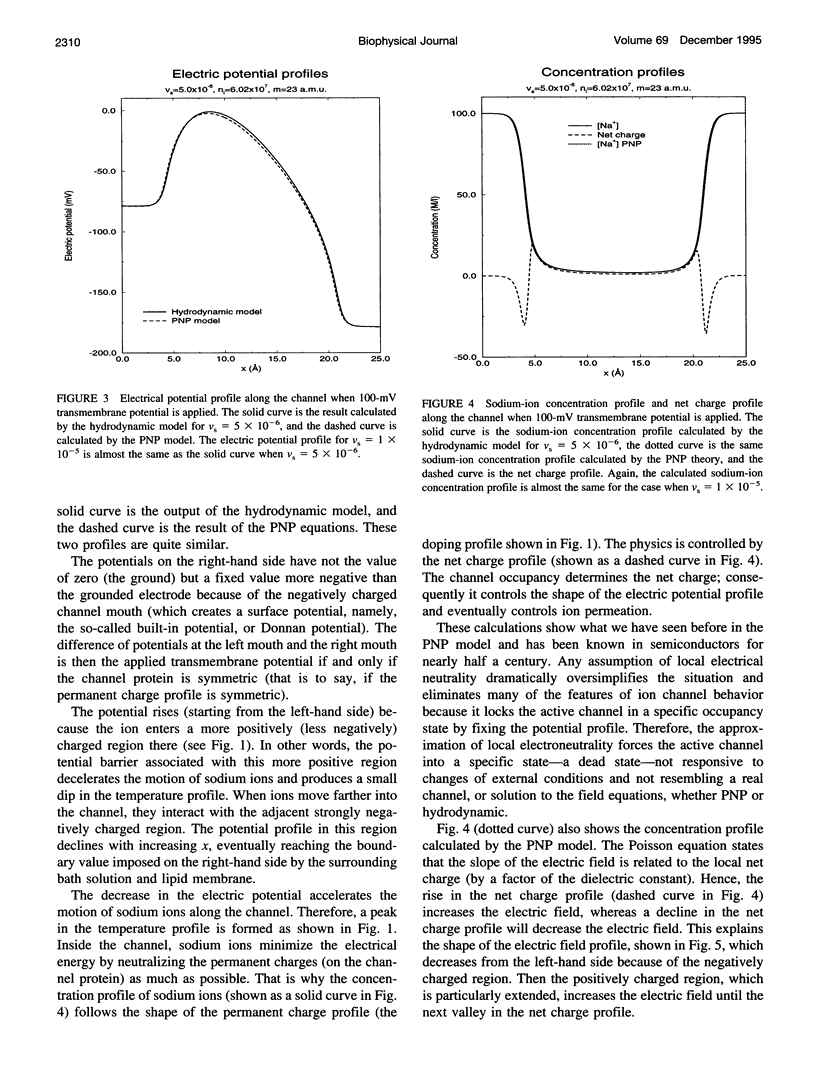
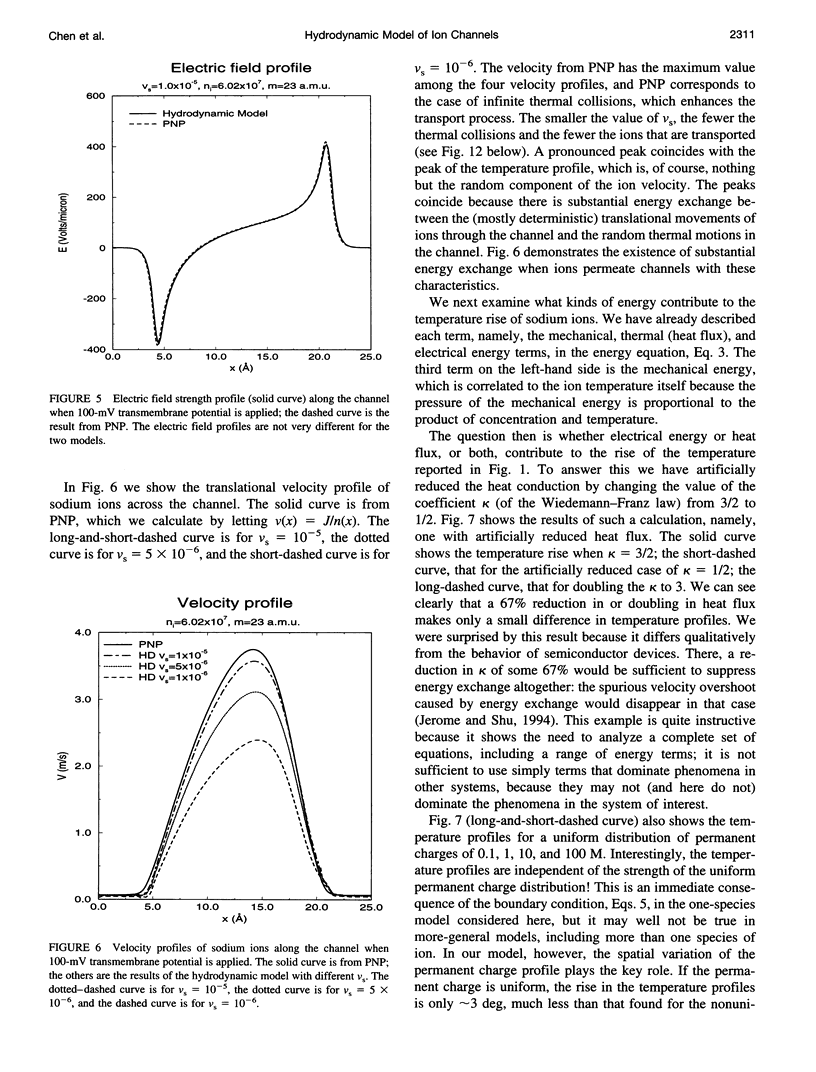
Most theories of open ionic channels ignore heat generated by current flow, but that heat is known to be significant when analogous currents flow in semiconductors, so a generalization of the Poisson-Nernst-Planck theory of channels, called the hydrodynamic model, is needed. The hydrodynamic theory is a combination of the Poisson and Euler field equations of electrostatics and fluid dynamics, conservation laws that describe diffusive and convective flow of mass, heat, and charge (i.e., current), and their coupling. That is to say, it is a kinetic theory of solute and solvent flow, allowing heat and current flow as well, taking into account density changes, temperature changes, and electrical potential gradients. We integrate the equations with an essentially nonoscillatory shock-capturing numerical scheme previously shown to be stable and accurate. Our calculations show that 1) a significant amount of electrical energy is exchanged with the permeating ions; 2) the local temperature of the ions rises some tens of degrees, and this temperature rise significantly alters for ionic flux in a channel 25 A long, such as gramicidin-A; and 3) a critical parameter, called the saturation velocity, determines whether ionic motion is overdamped (Poisson-Nernst-Planck theory), is an intermediate regime (called the adiabatic approximation in semiconductor theory), or is altogether unrestricted (requiring the full hydrodynamic model). It seems that significant temperature changes are likely to accompany current flow in the open ionic channel.
Full text
PDF










Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Cai D, Lai YC, Winslow RL. Complex dynamics in coupled cardiac pacemaker cells. Phys Rev Lett. 1993 Oct 11;71(15):2501–2504. doi: 10.1103/PhysRevLett.71.2501. [DOI] [PubMed] [Google Scholar]
- Chen D. P., Eisenberg R. S. Flux, coupling, and selectivity in ionic channels of one conformation. Biophys J. 1993 Aug;65(2):727–746. doi: 10.1016/S0006-3495(93)81099-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen D., Eisenberg R. Charges, currents, and potentials in ionic channels of one conformation. Biophys J. 1993 May;64(5):1405–1421. doi: 10.1016/S0006-3495(93)81507-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chiu S. W., Subramaniam S., Jakobsson E., McCammon J. A. Water and polypeptide conformations in the gramicidin channel. A molecular dynamics study. Biophys J. 1989 Aug;56(2):253–261. doi: 10.1016/S0006-3495(89)82671-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dani J. A., Levitt D. G. Water transport and ion-water interaction in the gramicidin channel. Biophys J. 1981 Aug;35(2):501–508. doi: 10.1016/S0006-3495(81)84805-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Elber R., Chen D. P., Rojewska D., Eisenberg R. Sodium in gramicidin: an example of a permion. Biophys J. 1995 Mar;68(3):906–924. doi: 10.1016/S0006-3495(95)80267-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finkelstein A., Andersen O. S. The gramicidin A channel: a review of its permeability characteristics with special reference to the single-file aspect of transport. J Membr Biol. 1981 Apr 30;59(3):155–171. doi: 10.1007/BF01875422. [DOI] [PubMed] [Google Scholar]
- Fischetti MV, Laux SE. Monte Carlo analysis of electron transport in small semiconductor devices including band-structure and space-charge effects. Phys Rev B Condens Matter. 1988 Nov 15;38(14):9721–9745. doi: 10.1103/physrevb.38.9721. [DOI] [PubMed] [Google Scholar]
- Fornili S. L., Vercauteren D. P., Clementi E. Water structure in the Gramicidin A transmembrane channel. Biochim Biophys Acta. 1984 Apr 11;771(2):151–164. doi: 10.1016/0005-2736(84)90527-3. [DOI] [PubMed] [Google Scholar]
- Green W. N., Andersen O. S. Surface charges and ion channel function. Annu Rev Physiol. 1991;53:341–359. doi: 10.1146/annurev.ph.53.030191.002013. [DOI] [PubMed] [Google Scholar]
- Howarth J. V. Heat production non-myelinated nerves. Philos Trans R Soc Lond B Biol Sci. 1975 Jun 10;270(908):425–432. doi: 10.1098/rstb.1975.0020. [DOI] [PubMed] [Google Scholar]
- Levitt D. G., Elias S. R., Hautman J. M. Number of water molecules coupled to the transport of sodium, potassium and hydrogen ions via gramicidin, nonactin or valinomycin. Biochim Biophys Acta. 1978 Sep 22;512(2):436–451. doi: 10.1016/0005-2736(78)90266-3. [DOI] [PubMed] [Google Scholar]
- Mackay D. H., Berens P. H., Wilson K. R., Hagler A. T. Structure and dynamics of ion transport through gramicidin A. Biophys J. 1984 Aug;46(2):229–248. doi: 10.1016/S0006-3495(84)84016-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rejto P. A., Bindewald E., Chandler D. Visualization of fast energy flow and solvent caging in unimolecular dynamics. Nature. 1995 May 11;375(6527):129–131. doi: 10.1038/375129a0. [DOI] [PubMed] [Google Scholar]
- Rosenberg P. A., Finkelstein A. Water permeability of gramicidin A-treated lipid bilayer membranes. J Gen Physiol. 1978 Sep;72(3):341–350. doi: 10.1085/jgp.72.3.341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roux B., Karplus M. Ion transport in a model gramicidin channel. Structure and thermodynamics. Biophys J. 1991 May;59(5):961–981. doi: 10.1016/S0006-3495(91)82311-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smart O. S., Goodfellow J. M., Wallace B. A. The pore dimensions of gramicidin A. Biophys J. 1993 Dec;65(6):2455–2460. doi: 10.1016/S0006-3495(93)81293-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace B. A. Gramicidin channels and pores. Annu Rev Biophys Biophys Chem. 1990;19:127–157. doi: 10.1146/annurev.bb.19.060190.001015. [DOI] [PubMed] [Google Scholar]


