Abstract
Background
Extracellular vesicles (EVs) are membrane-bound particles crucial for intercellular communication and serve as promising biomarkers for diseases, including cancer. Isolating and characterizing specific EV subpopulations, particularly those in plasma/serum, enhances biomarker precision and supports targeted therapies. Cancer-derived EVs often express unique surface markers, enabling distinction from other EVs. Accurate sorting of tumor-associated EVs provides insights into cancer progression, metastasis, and treatment response.
Results
This study presents a robust method for isolating and sorting CD9 + plasma EVs as a proof-of-concept for broader EV subpopulation analyses. Plasma EVs were isolated via sucrose cushion ultracentrifugation, optimizing purity and yield. Flow cytometry with fluorescence threshold triggering was fine-tuned to detect and sort CD9 + EVs, with instrument calibration and parameter adjustments mitigating swarming and improving sorting accuracy. Size exclusion chromatography further enhanced efficiency by reducing background noise. Sorted CD9 + EVs retained size and marker expression, including Syntenin, Alix, Flotillin-1, and CD9, which were enriched post-sorting.
Conclusions
These advancements enable high-purity EV subpopulation isolation, facilitating applications such as identifying cancer biomarkers and developing EV-based targeted therapies.
Supplementary Information
The online version contains supplementary material available at 10.1186/s12575-025-00293-2.
Keywords: Extracellular Vesicles, Flow Cytometry, Sorting, Tumor Biomarkers, Cancer
Introduction
Extracellular vesicles (EVs), membrane-bound particles released by all cell types, have gained significant attention in recent years due to their roles in intercellular communication and modulation in various (patho)physiological processes [1]. EVs encapsulate a varied array of proteins, lipids, and nucleic acids, rendering them a valuable reservoir of biomarkers for pathological conditions like cancer, cardiovascular diseases, and neurodegenerative disorders [2]. As the field of EV research continues to expand, there is now a pressing need to complement bulk methods, which analyze the general population in a sample, with assays that focus on individual EVs or distinct EV subsets. The possibility of analyzing these specific EV groups is of paramount importance, especially when considering plasma as a source of EVs, as this could play a critical role in advancing our understanding of their function(s) in health and disease [3]. Indeed, the development of accurate and standardized assays to assess the complex profiles of distinct plasma-EV subpopulations could revolutionize disease diagnosis and monitoring by increasing biomarker precision and advancing targeted therapeutic options [4].
Flow cytometry-based EV analysis and sorting require fine-tuning of several critical parameters to ensure the reliability and reproducibility of results. The “threshold” defines the minimum signal level that triggers event detection and is crucial to distinguish true EV events from background noise or electronic artifacts. The nozzle size influences the hydrodynamic focusing of particles and the overall resolution of the system, particularly important when working with nanoscale vesicles. Sheath and sample pressures must also be carefully regulated, as they affect the fluidics and stability of the sorting stream. Optimizing these parameters is essential for reducing false positives and minimizing coincident particle detection, also known as swarm detection, which may compromise the purity and accuracy of sorted EV populations [5].
The application of the widely used flow cytometry methodology, employing multiple labels to identify and separate various EV types, has been investigated by several research groups primarily focusing on vesicles released by different cell populations in vitro [6–10]. Our study aims at establishing a robust and reproducible method for characterizing and sorting plasma-derived EVs. We have addressed the two major limiting flow cytometry-associated challenges that can impact the analysis and subsequent sorting of plasma EVs. These factors include the instrument’s detection limit and the potential presence of artifacts that may arise during the collection and processing of such a complex biofluid.
Herein we describe a well-validated method to isolate plasma EVs based on sucrose cushion ultracentrifugation, that represents the the best-trade-off between purity and yield [11, 12]. A flow cytometry strategy based on an already published fluorescence threshold triggering [13, 14] was initially applied to assess the percentage of CD9-positive EVs and subsequently to sort them. CD9 is one of the most highly expressed tetraspanins by plasma-derived EVs [15] and was selected here as a proof-of-concept antigen to describe our standardized sorting protocol. Although the sorting of CD9 + plasma EVs has already been reported [16], the isolation workflow and the instrumentation employed in that study significantly differ from the approach proposed here. To determine the minimal prerequisites for EV detection and sorting, instrument setup in terms of sheath and sample pressure, as well as particle concentration, were thoroughly considered. Indeed, the effect on event rates per second, side light scattering, and fluorescent signals obtained during the analysis of different EV numbers was assessed using various nozzles, demonstrating that particle concentration can significantly affect their accurate quantification and characterization. Analyzing an excessive number of EVs leads to the coincident detection of nano-sized vesicles, resulting in an unwanted swarming effect. Besides, we confirmed previous published data [13] reporting that the removal of unbound reagents (dyes, antibodies) after EV staining by size exclusion chromatography (SEC) has a significant impact on the percentage of CFSE + events. It’s important to highlight that the proportion of CD9 + events within the CFSE-positive population remains unaffected. Notably, the increase in CFSE + events following SEC improves the efficiency of CD9 + EV sorting, enhancing both the event rate (events/second) and the volume of material collected.
Methods
Blood collection and processing
Blood samples were provided by the Institutional Transfusion Center of IRCCS Ospedale Policlinico San Martino, Genova, Italy. Blood was collected from healthy donors in K2E EDTA tubes. Plasma and platelets were isolated from blood by serial centrifugation steps, performed at 4 °C: 120 × g 20 min, 120 × g 5 min, 360 × g 20 min and 3000 × g 10 min. The resulting plasma and the enriched platelet fraction were stored at −80 °C until the use. Each sorting experiment was performed using a plasma pool collected on the same day from a minimum of 10 different healthy donors.
Plasma EV isolation
Sucrose cushion ultracentrifugation was selected for isolating plasma EVs [12]. Plasma (2.3 ml) diluted in 0.22 μm filtered PBS-EDTA to a final volume of 11.5 ml were loaded on top of 2 ml 20% sucrose and centrifuged for 135 min at 100,000 × g in a SW41Ti swinging rotor. The supernatant was discarded, and the obtained EV pellet was resuspended in 1 ml of PBS supplemented with 2 mM EDTA (PBS-EDTA), centrifuged for 10 min at 3000 × g to remove any co-isolated contaminants (platelets, proteins, and lipoprotein aggregates) and concentrated for 2 h at 100,000 × g (4 °C) (SN985 swinging rotor, Optima TL, Beckman). The EV pellet was resuspended in 50 µl of PBS-EDTA. All the analyses and applications of plasma-EVs were performed on freshly isolated EVs. Six independent experimental replicates have been analyzed.
Size exclusion chromatography (SEC)
SEC was performed using IZON smart columns qEV/70. According to manufacturer’s instructions, 500 µl of plasma was loaded on the column and eluted with PBS-EDTA to overcome EV aggregation. Twenty eluted fractions (F) of 500 µl were collected in protein low binding tubes, and EVs were collected in fractions 7–9. Samples were further diluted depending on the intended analysis.
EV staining
Each EV preparation was stained with 1 µM of CFDA-SE (Vybrant™ CFDA SE Cell Tracer Kit, Thermo Fisher Scientific) at 4 °C as a control to verify CFDA-SE specificity and at room temperature (RT), for 30 min in PBS-2mM EDTA, to visualize intact EVs and set the correct dimensional gate. A mixture of fluorescent beads of varying diameters (Megamix-Plus FSC and Megamix-Plus SSC, Biocytex) was used following the manufacturer’s instructions to optimize the cytometer settings and define the detection parameters. These beads allowed us to set appropriate thresholds and identify the region of interest for EV detection based on side scatter and fluorescence signals. The expression of the markers CD9 (APC Mouse Anti-Human CD9, Clone HI9a, 312108; BioLegend), CD63 (PE-Cy7 Mouse Anti-Human CD63, Clone H5C6, 561982; BD Biosciences) and CD42a (CD42a antibody, anti-human, APC, REAfinity ™, Clone: REA209, 130-123-831; Miltenyi Biotec) was evaluated within the CFDA-SE-positive events and compared to the corresponding isotype controls. The antibodies have been incubated for 30 min at RT. Six independent experimental replicates have been stained and sorted.
Flow cytometer preparation and calibration
All EV sorting experiments (n = 6) were conducted using a BD FACS Symphony S6 Cell Sorter equipped with five lasers (305 nm, 405 nm, 488 nm, 561 nm, and 640 nm). For the purpose of this study, only the blue laser (488 nm, set to 200 mW) and the red laser (640 nm, set to 140 mW) were used. Flow cytometer has been meticulously prepared before performing analysis and sorting. The entire fluidic system has been thoroughly washed, and when necessary, further washing cycles have been repeated during the acquisitions, particularly if inconsistencies in the definition of gates with previous analysis were observed. The instrument’s calibration performance was checked and verified for each new experiment using the calibration beads (BD FACSDiva CS&T Research Beads #655051) according to the manufacturer’s instructions. If the report was not fully satisfactory, all washes have been repeated until the desired results were achieved. Only then is the machine ready and operational to first acquire ultrapure MilliQ water to observe the optical limit of the machine on the main plot, which should always be restricted to the intersection of the axes in the lower left corner. The next step for optimal negative control setting is the acquisition of the final buffer in which the EVs are resuspended, namely PBS-EDTA. This must also remain confined within the lower left gate. In this methodological work, as mentioned earlier, we use CFDA-SE to label whole EVs. To implement the unconventional flow cytometry protocol, the physical parameter used for size is SSC-A with the electronic threshold set on the blue laser − 530/30 channel. The threshold is set at 300 to effectively visualize both the labeled EVs and, in sufficient quantity to exclude the background. Rosetta Calibration Beads (Exometry, The Netherlands) have been used to estimate the size of sorted EV populations. Flow rate has been quantified in standard units using BD Trucount Tubes (Cat. No. 663028, BD Biosciences, Lot number: 47500). SPHERO Rainbow Calibration Particles (RCPs) (Spherotech Inc., Lake Forest IL) have been used to relate the arbitrary units of fluorescence to absolute units.
Nanoparticle tracking analysis (NTA)
All samples were diluted in filtered PBS-EDTA to a final volume of 1 ml. Optimal measurement concentrations were found by pre-testing the ideal particle per frame value (20–100 particles/frame). Camera level was increased until all particles were distinctly visible not exceeding a particle signal saturation over 20%. The ideal detection threshold was determined to include as many particles as possible with the restrictions that 10–100 red crosses were counted while only < 10% were not associated with distinct particles. Blue cross count was limited to 5. Autofocus was adjusted so that indistinct particles were avoided. For each measurement, five 1-min videos were captured under the following conditions: cell temperature: 25 °C; syringe speed: 40 µl/s. After capture, the videos have been analysed by NanoSight Software NTA 3.1 Build 3.1.46 with a detection threshold of 5. Hardware: embedded laser: 45 mW at 488 nm; camera: sCMOS. The number of completed tracks in NTA measurements was always greater than the proposed minimum of 1000 in order to minimise data skewing based on single large particles [17].
Transmission electron microscopy (TEM) imaging of EVs
Sorted EVs (7.0e7) were analyzed to evaluate their structure, as previously described [18]. Briefly, EVs were resuspended in 20 µl dPBS and properly diluted, then adsorbed on 300 mesh carbon-coated copper grids (Electron Microscopy Sciences, Hatfield, Pennsylvania, United States of America) for 10 min; after being fixed for 10 min in glutaraldehyde (2% in PBS) and washed 3 times with MilliQ water, EVs were negative stained using a 2% solution of pH 7 phosphotungstic acid. The observation was carried out on a Philips CM 100 TEM 80 kV transmission electron microscope.
Western blot analysis
EVs were resuspended in RIPA buffer (1% NONIDET p-40, 0.1% SDS, 0.1% Sodium deoxycholate, protease inhibitor cocktail 1x, in PBS pH7.5) and protein content was quantified by Bicinchoninic acid (BCA) assay (Thermo Fisher Scientific). Five µg of proteins for each sample were loaded on 4-12% NuPAGE Bis-Tris gel (Thermo Fisher Scientific). Electrophoresis was performed at 160 V and proteins were blotted on a polyvinylidene fluoride membrane for 1 h at 30 V with an XCell Blot Module (Invitrogen). After blocking nonspecific sites with 5% non-fat dry milk (EuroClone, Italy) in Tris Buffered Saline with Tween 20 (TTBS, 20 mM Tris pH 7.5, 500 mM NaCl, 0.05% Tween 20), the membrane was incubated overnight at 4 °C with specific primary antibodies for: Flotillin-1 (dilution 1:10 000, ab133497, Abcam), Syntenin (dilution 1:1000, ab13326, Abcam), Alix (dilution 1:1000, ab186429, Abcam), and CD9 (dilution 1:1000, ab236620, Abcam) diluted in 2.5% non-fat dry milk/TTBS. After rinsing three times with TTBS, membranes were incubated with specific HRP-conjugated secondary antibodies (Mouse IgG HRP Linked Whole Ab, GENA931 or Rabbit IgG HRP Linked Whole Ab, GENA934). Positivity was highlighted by providing the substrates for the chemiluminescence reaction of HRP (Amersham ECL Prime Western Blotting Detection Reagent, GE Healthcare, Chicago, Illinois, USA) and impressing a photographic sheet by autoradiography (GE Healthcare).
A dedicated SDS-PAGE electrophoresis was also performed under reducing conditions to assess potential lipoprotein contamination in both pre- and post-sorted EVs, together with a plasma sample depleted of albumin and IgG (Depleted Plasma) using PureProteome™ Albumin/IgG Depletion Kit (Merck, #LSKMAGD12-1), as a positive control for apolipoproteins. Protein quantification was carried out using the Qubit Protein Assay Kit (Invitrogen). A total of 40 µg of protein from EV-naïve preparations, sorted EVs, and depleted plasma, along with 10 µg of BSA protein, were loaded onto 4–20% Mini-PROTEAN® TGX Stain-Free™ gels (Bio-Rad, Hercules, CA, USA). SDS-PAGE was run at 120 V for 90 min. After electrophoresis, the gel was activated for total protein visualization using the ChemiDoc™ Imaging System (Bio-Rad) for 5 min. Proteins were then transferred onto a PVDF membrane using the Trans-Blot® Turbo Transfer System (Bio-Rad) for 10 min at 2.5 A and up to 25 V. Membranes were blocked with 5% BSA in TBST for 1 h at room temperature and incubated overnight at 4 °C with the following primary antibodies: ApoB diluted 1:30,000 in BSA5% (Abcam, #Ab 139401), ApoA1 1:1,000 in BSA5% (Cell signaling, #), and Flotillin1 1:10,000 in BSA5% (Abcam, #Ab 133497).
miRNA analysis
Total RNA, including small RNAs, was isolated from sorted CD42a-positive EVs and the corresponding platelets using the exoRNeasy Kit (Qiagen, Hilden, Germany). Briefly, the pellet from CD42a-positive EVs and the isolated platelets from the same plasma samples were resuspended in 700 µl of Qiazol including the UniSP6 spike-in (Qiagen) and processed according to the manufacturer’s instructions. The RNA was then eluted in 19 µl of RNase-free water, and the miRNA enrichment was assessed by the Qubit fluorometer using the Qubit microRNA Assay Kit (Thermo Fisher Scientific Inc., Waltham, MA US). To evaluate the expression levels of a panel of 16 miRNAs (i.e., let-7b-5p, let-7f-5p, let-7i-5p, miR-103a-3p, miR-1260a, miR-1260b, miR-1-3p, miR-150-5p, miR-30b-5p, miR-361-3p, miR-370-3p, miR-411-5p, miR-543, miR-574-3p, miR-574-5p, miR656-3p) previously described in human platelets (PMID: 35946052), 2 ng of RNA was reverse-transcribed with the miRCURY LNA RT Kit (Qiagen). Diluted cDNA (1:20) was amplified using iTaq Universal SYBR Green Supermix (Bio-Rad) with specific assays. The relative concentration of different miRNAs was calculated as dcT normalized on UniSP6 internal control (Ct-Mean).
Statistical analysis
The data were analyzed using: i) Two-way ANOVA and Tukey’s multiple comparison test (to compare the percentage of events falling within the BN and Unbound gates when unstained EVs were analyzed with the instrument threshold set to 300 or 200 and the percentage of events within the BN, Unbound, or Pos EVs gates with or without SEC); ii) Kruskal-Wallis and Dunn’s multiple comparisons test (to evaluate the percentage of CD9-positive events obtained by analyzing the same plasma-EV samples using four different nozzles); iii) Unpaired t test (to evaluate the size distribution by NTA analysis of purified plasma- EVs before and after sorting). Data are presented as mean ± SD considering at least three independent replicates for each assay and analyzed by GraphPad Prism 9.0.0. (Graph Pad Software, Inc.). For all analyses p < 0.05 was considered statistically significant. In all cases: ****p < 0.0001, ***p < 0.001, **p < 0.01, *p < 0.05.
Results
Optimization of flow cytometry calibration and sorting parameters for high-purity CD9-Positive plasma-EVs
An accurate flow cytometer calibration along with the implementation of unconventional cytometry settings and a systematic standardization of acquisition parameters are fundamental prerequisites for both detection and sorting of EVs. To obtain a significant yield of EVs, it is necessary to plan for lengthy sorting sessions, which may pose the risk of bacterial contamination. To mitigate this concern, the flow cytometer has been prepared performing accurate washing procedures and set for aseptic sorting. Additionally, all procedures involved in EV sample preparation have been managed under sterile conditions.
To establish a standardized methodological protocol, we used the default sheath pressure parameters associated with the different nozzles (nozzle 70 mm: 70 PSI; nozzle 85 mm: 45 PSI; nozzle 100 mm: 20PSI; nozzle 130 mm: 10 PSI) and we applied the minimum sample pressure [8, 19]. The window extension was set to 2.0 µs for all parameters, and the area scaling factor was set to 0.6. The optimal instrument performance to sort EVs has been achieved using a very low flow rate (flow rate = 1), with a “4-way purity” mask, commonly used for downstream molecular biology applications. This approach was employed to adhere to the theoretical principle that EVs should pass through the laser beam as slowly as possible [20].
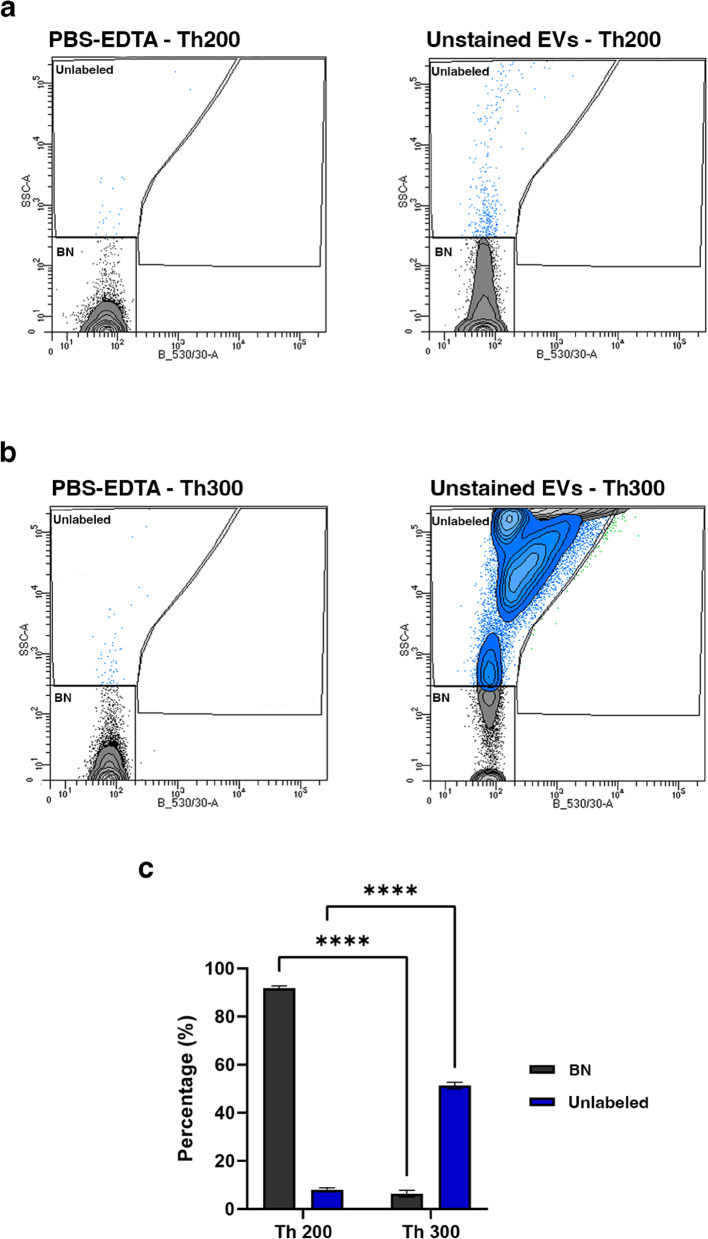
In our technical protocol, the threshold applied on FL1 (blue laser at 488 nm with B530/30 filter) was set to 300, as preliminary experiments indicated that a lower threshold (200) captured excessive background noise (BN) (Fig. 1). Although the percentage of BN remains nearly constant with the threshold set at 200 and 300 (99.2% and 98.9%, respectively) when analyzing the vehicle solution (PBS-EDTA) (Fig. 1a and b, left panels), this percentage changes significantly when unstained naïve EVs are examined, ranging from 6.4% ± 1.4 to 91.87% ± 0.92 (p < 0.0001) (Fig. 1a and b, right panels and Fig. 1c). Consequently, the percentage of events falling within the “Unlabeled” gate changes significantly depending on the applied threshold, rising from 51.3 ± 1.4 at a threshold of 300 to 7.9 ± 0.9 when the same sample is evaluated with a threshold of 200 (Fig. 1a and b, right panels and Fig. 1c).
Fig. 1.
Definition of the optimal instrumental threshold. Representative dot plots of PBS-EDTA or unstained plasma EVs analyzed by light scattering and fluorescence. Background noise (BN), shown in the lower corner of each plot, represents random sampling of scattered light from the laser. The“Unlabeled” gate indicates the events corresponding to unstained EVs. The analysis was performed with the threshold set at 300 (a) or 200 (b). c Histogram representing the percentage of events falling within the BN and Unlabeled gates when unstained EVs are analyzed with the instrument threshold set to 300 or 200. Data are presented as mean ± standard deviation (SD) from six independent biological replicates (n=6). **** p<0.0001 (Two-way ANOVA and Tukey’s multiple comparison test)
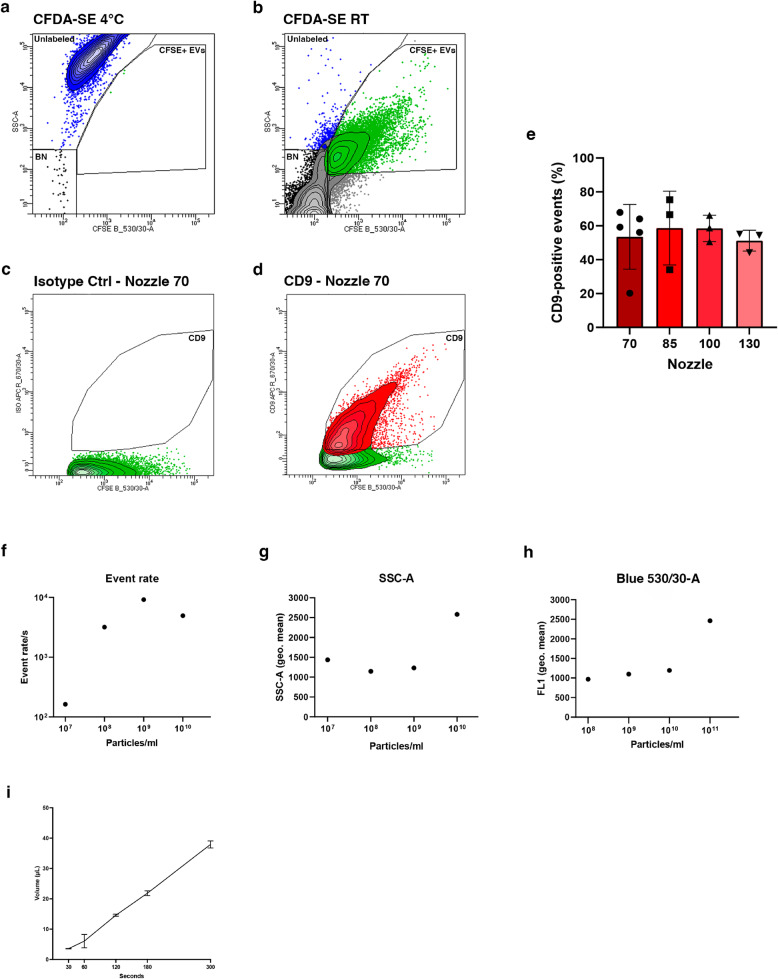
Accordingly, we selected the threshold of 300, given its greater detection robustness, to sort, as a proof of principle, CD9 + plasma EVs. Following a previously published protocol [14], we tested the volumes of CD9 + EVs obtained using the aforementioned parameters with different nozzles. This protocol utilized commercially available fluorescent beads, detected in the FL1 channel, and of various sizes to adjust the SSC voltage (Supplementary Fig. 1a).
Plasma-EVs were first stained with 1 µM CFDA-SE, incubated at 4 °C, and then at RT (Fig. 2a and b). This enabled us to define the gate for intact EVs with active enzymatic activity, identified as CFDA-SE positive (gate “CFSE + EVs”). Considering only the events falling within this gate, we performed labeling with an anti-CD9 antibody or the corresponding isotype control. This approach has been adopted using the 4 different nozzles (Fig. 2c and d and Supplementary Fig. 1b). Although the percentage of CD9 + EVs did not vary significantly with different nozzles (Fig. 2e), we observed that the volume of sorted CD9 + EVs differed substantially. Indeed, the volume required to sort the same number of CD9 + events (50,000 events) varied significantly when using the four different nozzles, with the 70 μm nozzle allowing sorting in a smaller volume (60 µl) compared to the others (120 and 150 µl when using the 85 and 100 μm nozzles, respectively, and reaching approximately 400 µl when switching to the 130 μm nozzle). This is a crucial factor to consider, as downstream analyses of sorted EVs typically require small volumes. Sorting excessive volumes would necessitate further manipulation to concentrate the sample, which could lead to the loss of EVs. Therefore, the analyses presented henceforth were conducted using the 70 μm nozzle.
Fig. 2.
Flow cytometry gating strategy and EV concentration assessment. Representative dot plots for EVs stained with 1mM CFDA-SE at 4°C (a) and at RT (b). Representative contour plots of CFSE+ EVs stained with either APC-conjugated CD9 antibody (d) or corresponding non-reactive immunoglobulin of the same isotype (c). These analyses have been performed using the 70 mm nozzle. (e) Histogram showing the percentage of CD9-positive events obtained by analyzing the same plasma-EV samples using four different nozzles (70, 85, 100, and 130 µm). Data are presented as mean ± SD from five independent biological replicates for nozzle 70 µm and three independent replicates for nozzles 85, 100 and 130 µm. (Kruskal-Wallis and Dunn’s multiple comparisons test). f-h Serial dilutions of a highly concentrated EV suspension (stained with CFDA-SE) were prepared and analyzed using a 70 µm nozzle and default sheath fluid pressure. EVs were analyzed at various concentrations, ranging from 1.0e7 to 1.0e10. f Analysis of the number of events per second, (g) SSC-A geometrical mean, and (h) fluorescence (FL1 geometrical mean). (i) Acquisition of three independent replicates of TruCount Tubes at specific time intervals using the 70 µm sort setup with default settings and minimum default sample pressure, in order to calculate the flow rate in standard units
Serial dilutions of plasma EVs were examined to investigate the impact of particle concentration on event rate, side scatter-area (SSC-A), and CFDA-SE fluorescent intensity (FL1). Analysis of the highest EV concentration (1.0e10) showed a lower-than-expected event rate (Fig. 2f). Diluting this sample to 1.0e9 resulted in an increased event rate, reaching 9,180 events per second. Additional dilutions beyond this number led to a decrease in event rates, reaching 163 events per second when 1.0e7 EVs were analyzed (Fig. 2f). These data indicate that higher event rates up to 9,180 per second are unreliable and the lack of linearity observed at higher event rates may be attributed to the coincident detection of EVs. This is supported by the observed decrease in event rate at the highest EV number (Fig. 2f). Coincident detection of EVs is also expected to result in increased SSC-A and FL1 values due to simultaneously detected vesicles. As EV numbers increase beyond the linear detection range (> 1.0e9), a steady rise in SSC-A (Fig. 2g) and fluorescent intensity (Fig. 2h) values are observed. When the EV count is below 1.0e9, these values remain relatively unchanged. However, examining EVs at a higher concentration leads to alterations in scatter profiles, attributed to coincidence and swarm detection (Fig. 2g and h). The decrease in event rate, coupled with the increase in SSC-A and FL1 values, suggests that coincident EV detection occurs at the highest EV concentrations. Taken together, these data suggest that the optimal concentration for sorting EVs in our system, while avoiding the swarming effect and maintaining a consistent flow rate over time, is approximately 1.0e8 particles/ml. At this concentration, the flow rate remains stable (approximately 2 K events/s). The flow rate was quantified in standard units using BD Trucount Tubes by running the beads for a known amount of time and applying the following calculation: (mean number of Trucount bead events acquired at different time points × 100)/total bead count per tube (Fig. 2i).
Enhanced sorting efficiency by a SEC-based removal of unbound reagents
The data obtained so far indicate that achieving a high yield of sorted EVs while adhering to the parameters mentioned above requires excessively long sorting times. This is due to the substantial amount of background noise and unbound reagents. The vehicle solution (PBS-EDTA) was stained with the same concentrations of CFDA-SE and anti-CD9 antibody, either individually or in combination, in order to assess signal contributions from potential aggregates [21]. Based on the gating strategy established for sorting, we acquired five replicates for each of the seven experimental groups (unstained vehicle solution; vehicle solution stained with CFDA-SE alone, anti-CD9 antibody alone, or both in combination; each with or without SEC). For each group, we recorded and tabulated the mean and standard deviation of event counts over 180 s for the following parameters: all events detected in the B530/30 channel (All B530/30), all events detected in the R670/30 channel (All R670/30), and all events falling within the Background Noise and Unbound Reagents gates (BN + UB). This analysis revealed that SEC led to a statistically significant reduction, in all the considered parameters (All B530/30: CFDA-SE PRE-SEC vs. CFDA-SE POST-SEC, p = 0.0159; CD9 PRE-SEC vs. CD9 POST-SEC, p < 0.0001; CFDA-SE + CD9 PRE-SEC vs. CFDA-SE + CD9 POST-SEC, p < 0.0001. All R670/30: CFDA-SE PRE-SEC vs. CFDA-SE POST-SEC, p < 0.0001; CD9 PRE-SEC vs. CD9 POST-SEC, p = 0.0179; CFDA-SE + CD9 PRE-SEC vs. CFDA-SE + CD9 POST-SEC, p < 0.0001. BN + UB: CFDA-SE PRE-SEC vs. CFDA-SE POST-SEC, p < 0.0001; CD9 PRE-SEC vs. CD9 POST-SEC, p = 0.0020; CFDA-SE + CD9 PRE-SEC vs. CFDA-SE + CD9 POST-SEC, p < 0.0001), in event counts derived from aggregates formed upon staining PBS-EDTA with either the dye alone, the antibody alone, or their combination, thereby highlighting differences in aggregate content between samples processed with and without SEC (Table 1).
Table 1.
Mean Fluorescence Intensities (MFI ± SD) of EV populations measured in the B530/30 and R670/30 channels under the following labeling conditions: PBS-EDTA (control), CFDA-SE, CD9, and CFDA-SE + CD9, before (PRE SEC) and after (POST SEC) size exclusion chromatography (SEC). BN + UB = background noise plus unbound reagents gates
| PBS-EDTA | PRE SEC | POST SEC | ||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| CFDA-SE | CD9 | CFDA-SE + CD9 | CFDA-SE | CD9 | CFDA-SE + CD9 | |||||||||
| Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | Mean | SD | |
| All B530/30 | 3.2 | 1.1 | 2140.2 | 24.5 | 337 | 54.4 | 5947.8 | 584.5 | 348 | 16.7 | 7.4 | 2.7 | 1548.4 | 64.0 |
| All R670/30 | 0 | 0 | 335 | 9.1 | 96.3 | 7.2 | 101.6 | 12.5 | 2 | 1.2 | 0.6 | 0.5 | 254.8 | 15.0 |
| BN + UB | 362.2 | 7.3 | 14889.7 | 1548.1 | 584.3 | 108.5 | 311542.8 | 19429.7 | 625.8 | 10.2 | 339.4 | 19.7 | 1550.6 | 47.8 |
Consequently, an EV washing step was introduced using SEC after staining with CFDA-SE and anti-CD9 antibody. This approach effectively removed most unbound reagents, drastically reducing the background and leading to a significant increase in the percentage of CFSE + EVs. When compared to the total number of events, the percentage of CD9 + EVs remains approximately constant (46% and 52%, respectively) (Fig. 3a and b, right panels). What is indeed affected by SEC washing is the percentage of CFSE + events falling within the Pos EVs gate, due to a significant reduction in background signal (Fig. 3a and b, left panels). Specifically, the percentage of events falling within the BN gate decreased significantly when the same sample of CFSE- and CD9-stained plasma EVs was washed using Sect. (44.2% ± 6.5 vs. 14.9% ± 3.4, p < 0.0001) (Fig. 3c). Consequently, there was a significant increase in the percentage of events within the CFSE + EVs gate, rising from 40.2% ± 5.7 when EVs were not subjected to SEC to 75.2% ± 3.5 after SEC (Fig. 3c). The SEC-based removal of BN noise allowed us to achieve a consistently acceptable experimental flow rate (15 K events/s), significantly higher than the 2 K events/s required without the SEC step, without generating swarming effects. This improvement is particularly important when separating a large number of EVs for subsequent downstream applications and analysis, enabling the sorting of 7.0e7 CD9 + EVs in approximately 75 min.
Fig. 3.
SEC removal of unbound dyes increases the signal to noise ratio of labeled EVs. (a, b) Representative dot plots of EVs stained with CFDA-SE and APC-conjugated anti-CD9 antibody, either without (a) or with (b) SEC following EV labeling. (c) Histogram showing the percentage of events within the BN, Unlabeled, or CFSE+ EVs gates, observed when CFDA-SE- and CD9-stained EVs either underwent SEC or skipped this step after labeling. Data are presented as mean ± SD from three independent biological replicates (Pre-SEC) and six independent biological replicates (Post-SEC). **** p>0.0001. (Two-way ANOVA and Tukey’s multiple comparison test)
To quantify the Molecules of Equivalent Fluorochrome (MEF) applied to each experiment and to relate the arbitrary units of fluorescence to absolute units, the SPHERO Rainbow Calibration Particles (RCPs) have been used according to manufacturer’s instructions. Specifically, RCPs must be acquired under identical cytometer settings as those used for the fluorescently labeled EVs. As a result, all of the peaks should be within the scale as shown Supplementary Fig. 2a and 2b, left panels. Calibration and normalization template, provided by the manufacturer and specific to the lot of beads used, enable the conversion of mean fluorescence intensity (MFI) values into absolute fluorescence units (MEF) across all photomultiplier channels employed in the EV sorting experiment. By generating linear regression curves from the calibration data (Supplementary Fig. 2a and 2b, right panels), it is thus possible to extrapolate the mean absolute fluorescence values of EV samples (Supplementary Fig. 2c) by inputting the corresponding channel numbers associated with gated subpopulations of interest.
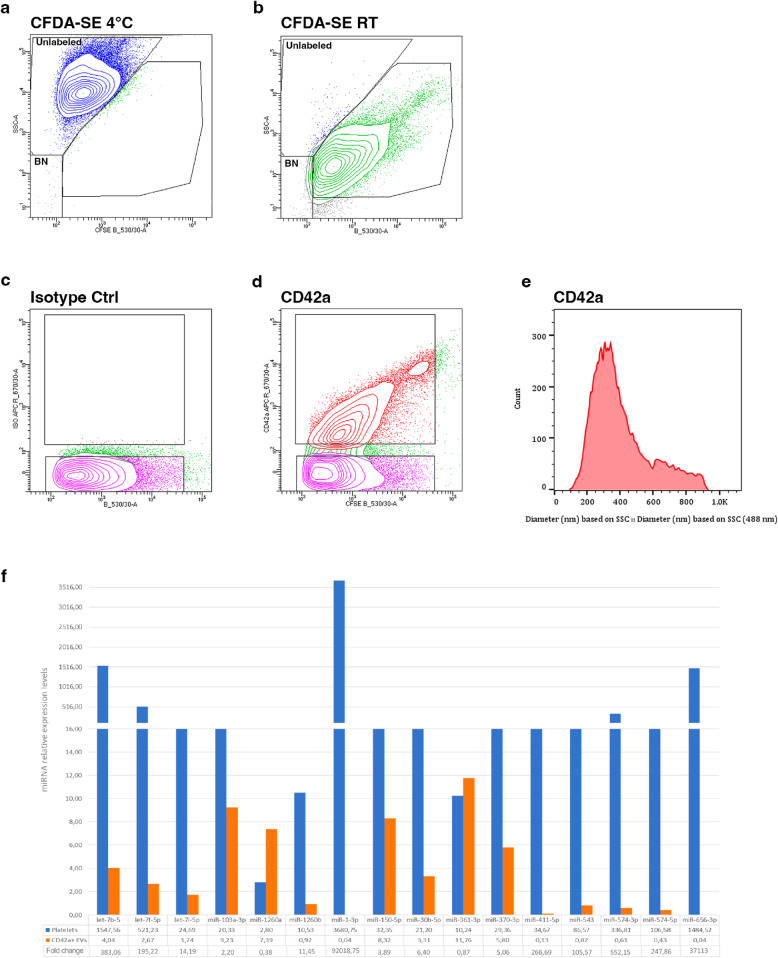
To further assess the efficiency and versatility of our EV-sorting strategy, we performed an additional experiment using a less commonly employed marker. We focused on CD42a, that is typically expressed by platelets [22]. Platelets are a key component of the tumor microenvironment and can engage in dynamic crosstalk with tumor cells. Platelet-derived EVs are emerging as potential mediators of this interaction, raising new questions about the mechanisms underlying this regulatory network [23]. Plasma EVs were processed exactly as described for CD9 + EVs: they were stained with CFDA-SE and an anti-CD42a antibody, washed by SEC to remove unbound reagents, and then analyzed and sorted. CD42a + events accounted for approximately 40% of th total CFSE + EV population (Fig. 6a–d). To avoid the size-estimation bias introduced by polystyrene beads (which have a different refractive index), we used Rosetta Calibration beads to convert SSC-A arbitrary units into true particle diameters in nanometers. This approach enabled us to determine the actual size distribution of the sorted CD42a + EVs (Fig. 6e). A total of 5.0 × 10^7 CD42a + EVs were successfully sorted.
Fig. 6.
Characterization of CD42a+ EVs. a Representative dot plots for EVs stained with 1mM CFDA-SE at 4°C (a) and at RT (b). Representative contour plots of CFSE+ EVs stained with either APC-conjugated CD42a antibody d or corresponding non-reactive immunoglobulin of the same isotype (c). These analyses have been performed using the 70 mm nozzle.e Histograms showing the normalized diameter (nm), as determined using Rosetta Calibration beads, for pre-sorted CD42a+ events. f Histogram representing the relative expression levels of 16 miRNAs analyzed on both CD42a+ (orange bars) sorted EVs and platelets (blue bars) derived from the same donors
Characterization of sorted EVs
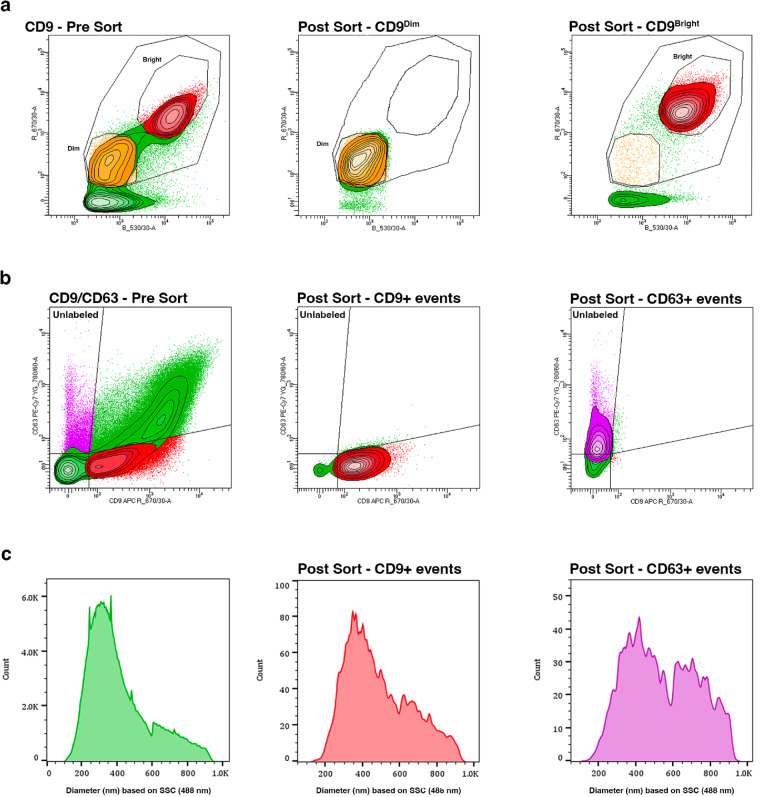
Figure 4 presents two representative examples of EV sorting using the anti-CD9 antibody either alone or combined with anti-CD63, another well-known vesicle marker belonging to the tetraspanin family. In some plasma samples, it was possible to distinguish CD9Dim from CD9Bright EVs (Fig. 4a, left panel). An efficient sorting of both CD9Dim and CD9Bright events was achieved, and re-analysis of the sorted CD9Dim and CD9Bright EVs with the same parameters revealed enrichment levels of 97.4% ± 5.1 and 95.3% ± 3.8, respectively (Fig. 4a, middle and right panels). Our approach can also be used to efficiently sort two distinct subpopulations from the same sample, here represented, as a proof of principle, by CD9 + and CD63 + EVs (Fig. 4b, left panel). As expected, the percentage of CD63 + EVs is lower than that of CD9 + EVs but sorting of both subpopulations resulted in substantial enrichment (CD63+: 74.5% ± 8.6; CD9+: 93.4% ± 3.9) (Fig. 4b, middle and right panels). Also in this case, Rosetta Calibration beads were used to convert SSC-A arbitrary units into true particle diameters in nanometers. This approach enabled us to determine the actual size distribution of CFSE + EVs (pre-sorting) (Fig. 4c, left panel), sorted CD9 + EVs (Fig. 4c, middle panel), and sorted CD63 + EVs (Fig. 4c, right panel).
Fig. 4.
Sorting of EVs based on the differential expression of CD9 alone or in combination with CD63. a Representative contour plot showing CFSE+ EVs (green) stained with APC-conjugated anti-CD9 antibody. A CD9Brightsubpopulation (red), distinguishable from CD9Dim (yellow), is observed depending on the plasma sample analyzed (left panel). Middle and right panels show the sorted EVs, back-gated in a contour plot, to confirm that the CD9Dim (middle panel) and CD9Bright (right panel) subpopulations correctly fall within their respective gates. b Representative contour plot showing CFSE+ EVs stained with both APC-conjugated anti-CD9 (red) and PE-Cy7-conjugated anti-CD63 (violet) antibodies (left panel). Middle and right panels show the sorted EVs, back-gated in a contour plot, to verify that the CD9-positive (middle panel) and CD63-positive (right panel) populations correctly fall within their respective gates. c Histograms showing the normalized diameter (nm), as determined using Rosetta Calibration beads, for pre-sorted CFSE+ events (left panel), post-sorted CD9+ events (middle panel), and post-sorted CD63+ events (right panel)
Sorted CD9 + EVs were characterized and compared with the corresponding pre-sorted EVs. To achieve this, EVs were ultracentrifuged after sorting to concentrate them and remove the excess of sheath fluid. Notably, the NTA analysis showed no significant differences in size between pre- and post-sorted EVs, with a median size of 181.5 ± 12.7 nm and 164.8 ± 9.7 nm, respectively (Fig. 5a). TEM analysis also confirmed the presence of vesicular structures, rounded and enclosed in a lipid membrane, both in pre- and post-sorted samples (Fig. 5b). Interestingly, the expression of the typical EV markers Alix, Flotillin-1, Syntenin and CD9 was maintained after sorting (Fig. 5c). We have performed additional western blot analysis on sorted EVs evaluating, together with the typical vesicle marker (Flotillin-1), the potential lipoprotein contamination analyzing the presence of vesicles expressing ApoB100 (found in LDL, IDL, and VLDL) and ApoA1 (found in HDL). The expression of these markers has been evaluated on pre-sorted EVs, sorted CD9 + EVs and a plasma sample depleted of albumin and IgG (Depleted Plasma), as a positive control for apolipoproteins. While ApoB expression has been revealed only on depleted plasma, the expression of ApoA1 has been observed also on both pre- and post-sorting EVs, even if at lower extent in comparison to depleted plasma, indicating that a minimal contamination of HDL lipoproteins in isolated EVs (Fig. 5d).
Fig. 5.
Characterization of plasma EVs before and after sorting. a Representative histograms showing the size distribution by NTA analysis of purified plasma- EVs before and after sorting. The graph on the right shows the calculated mean± SD from six independent biological replicates (n=6) of the median values associated with the NTA analysis (Unpaired t test). b Representative transmission electron microscopy (TEM) images of plasma-EVs before and after sorting. c Western blot analysis of plasma-EVs before and after sorting. Sorting does not affect the protein expression of typical vesicle markers (Alix, Flotillin-1, Syntenin, and CD9), whose expression is present in EVs both before and after sorting. d Western blot analysis on plasma-EVs before and after sorting and plasma depleted of albumin and IgG (depleted Plasma). Typical lipoprotein markers (ApoB100 and ApoA1) and Flotillin-1 have been evaluated in all the considered samples
This could be due to the removal of non-EV particles or debris during the sorting process, which results in a higher concentration of vesicles expressing these markers, thereby enhancing their relative abundance.
In order to demonstrate not only the sorting efficiency but also the functional applicability of our approach on downstream assessments, we applied it to the sorting of EVs expressing CD42a and performed a molecular evaluation on the sorted CD42a + EVs. Specifically, we aimed to go beyond conventional phenotypic characterization and assess the biological relevance of the isolated EV population. From the sorted EVs, it was possible to successfully extract miRNAs (5 ng), confirming the integrity of the EVs and their suitability for subsequent functional investigations. Furthermore, miRNA enrichment in CD42a-positive EVs was validated by analyzing a panel of 16 miRNAs previously identified in human platelets [24], using the corresponding platelets as a reference control. All 16 miRNAs were detected in the CD42a + EVs, with expression levels varying in a miRNA-dependent manner, ranging from 0.1% to over 100% (miR-361-3p) relative to the platelet counterparts (Fig. 6f). These results underscore the potential of our method to selectively enrich EV subpopulations based on specific surface markers that are not correlated with EV biogenesis, thus allowing our understanding of the molecular content of selected EVs that could be applied in different translational research contexts.
Discussion
In this study, we have developed and validated a robust methodology for sorting and characterizing plasma-derived EVs. Flow cytometry remains a cornerstone in the analysis of EVs, offering high-throughput and multiparametric capabilities [19]. However, the detection and sorting of EVs, particularly from complex matrices like plasma, present significant technical challenges [25, 26]. Our study highlights the importance of optimizing flow cytometry parameters to enhance EV analysis. First, CD9 + EVs were selected as a prototype to validate our approach, since CD9 is a well-characterized marker of plasma-derived EVs [27]. However, the methodological advancements demonstrated here can be adapted to sort and analyze a wide range of EV subpopulations based on various markers, including those associated with disease states such as cancer. Indeed, the ability to isolate EVs based on specific biomarkers is particularly relevant in oncology, as it could be pivotal for advancing early detection, monitoring disease progression, and evaluating therapeutic responses [28]. Beyond oncological applications, sorted EV populations could be exploited for functional studies aimed at investigating their role in intercellular communication, biomarker discovery, as well as therapeutic delivery. For instance, EVs isolated from specific cell types or disease contexts may be used to explore their cargo composition (e.g., RNA, proteins, lipids), assess their biological effects in vitro or in vivo, or evaluate their potential as carriers for targeted drug delivery. The optimization of flow cytometry parameters, including fluorescence threshold settings, was critical for enhancing the accuracy and reliability of EV analysis. Unlike sorting of cells, where larger size and distinct optical properties facilitate detection, sorting EVs requires meticulous adjustment of these parameters due to their nanoscale size [18]. This refinement is essential to achieve high-purity sorting, particularly in complex biological samples such as plasma. In our setup, setting the fluorescence threshold at 300 effectively reduced background noise, improving the detection of CD9-positive EVs. This approach aligns with recent findings emphasizing the importance of stringent parameter selection for accurate EV characterization [8, 28]. Although we did not systematically test variations in flow rate or sample pressure, we used a combination of nozzle size and associated default pressure settings recommended for small particle sorting. Thus, rather than independently optimizing each parameter, we relied on coordinated instrument settings to achieve a balance between purity and yield. In particular, our results suggest that a low flow rate and a 70 μm nozzle allow the collection of sorted EVs in a lower volume compared to the other nozzles, avoiding the need for condensing the volume for downstream analysis. In line with current best practices in the EV flow cytometry field, our study adheres to the MIFlowCyt-EV framework proposed by Welsh et al. [29], which provides standardized guidelines for reporting EV flow cytometry experiments. Specifically, we report critical experimental parameters such as the type of flow cytometer, nozzle size, sheath pressure, threshold triggering (fluorescence-based), fluorochromes used, and gating strategy. Moreover, we included details on the sample preparation method (sucrose cushion ultracentrifugation), staining procedures, and the post-staining purification step via size exclusion chromatography. These aspects are essential to ensure transparency, reproducibility, and comparability of results across studies. Nonetheless, some limitations in fully meeting the MIFlowCyt-EV guidelines remain. For example, while we provide particle concentrations and describe swarming avoidance strategies, we did not perform a side-by-side comparison of stained versus unstained samples with detergent lysis to demonstrate signal specificity. Furthermore, instrument calibration using reference beads or standardized small particle controls was not included in this initial methodological study but will be considered in future work to further align with the framework.
Addressing the challenge of background noise and artifacts in EV analysis, particularly from complex plasma samples, has been a focus of recent research [19]. Integrating a SEC-based washing step after staining with CFDA-SE and anti-CD9 antibody significantly improved the purity of CFSE + EVs. This approach aligns with recent advancements in EV purification techniques that emphasize minimizing contamination and enhancing sorting efficiency [9]. Notably, the same strategy was successfully applied to sort EVs expressing a less common marker, such as CD42a, a platelet-specific antigen. The effectiveness of this approach was further demonstrated by the ability to perform downstream molecular analyses, specifically, miRNA profiling, on the sorted vesicle population.
Our method allows for high-speed sorting (15 K events/s) while maintaining a constant flow rate and avoiding the swarming effect, which is crucial for large-scale studies. CFDA-SE is a membrane-permeable dye that, once inside vesicles, is converted into a fluorescent compound through esterase activity, enabling stable and long-lasting labeling [30]. This feature allows the identification and tracking of EVs without significantly altering their size or surface properties [31]. In addition, CFDA-SE labeling is relatively straightforward and does not require complex protocols or harsh conditions that could compromise vesicle integrity. However, some limitations should also be considered. Variability in esterase activity among different EV populations may affect labeling efficiency, potentially introducing bias in downstream analyses CFDA-SE can potentially label non-vesicular particles or aggregates, leading to false-positive events if not carefully controlled [32]. Moreover, unbound dye or dye aggregates may contribute to background noise if not adequately removed. Our use of SEC post-labeling addresses this limitation by reducing background signal, yet future studies could compare CFDA-SE to alternative labeling strategies, such as lipophilic dyes (e.g., PKH26, DiI) or protein-based approaches (e.g., fluorescent antibody labeling), to better assess labeling efficiency, specificity, and impact on EV integrity [33].
Our flow cytometry approach enabled the precise sorting and characterization of distinct EV subpopulations, such as CD9Bright and CD9Dim. This capability to differentiate EVs based on varying marker expression levels is vital for understanding the heterogeneity of plasma-derived EVs and their biological implications [10]. The ability to enrich EV populations based on varying marker expression levels highlights the method’s effectiveness in profiling EVs with specific biological characteristics. This approach is not limited to CD9-positive EVs but can be extended to analyze other markers (e.g. CD63, CD42a), thus serving as a versatile tool for a broad range of research applications. The comparison of pre- and post-sorted EVs revealed that our sorting protocol maintains the expression of typical EV markers, such as Alix, Flotillin-1, Syntenin, and CD9. While our sorting approach resulted in a clear enrichment of CD9+/CD63+/CD42a + particles and improved sample purity, we cannot exclude the presence of smaller EV subpopulations (e.g., < 200 nm) that may have escaped detection by flow cytometry and were not fully resolved by our gating strategy. Further studies using complementary approaches would be needed to fully assess these potential residual subpopulations. In addition, we validated the robustness and functional relevance of our sorting strategy by successfully applying it to a less commonly investigated marker, CD42a, a platelet-derived marker. Sorting for CD42a + EVs enabled the efficient isolation of vesicles of platelet origin, as confirmed by subsequent RNA extraction and analysis. Specifically, the expression of 16 putative platelet-associated miRNAs was assessed by quantitative PCR and compared with that of the corresponding parental platelets. The efficient and selective enrichment of platelet-derived miRNAs in the sorted EV fraction further supports the effectiveness of our approach in isolating functionally relevant EV subpopulations.
While our study provides a robust framework for EV sorting and characterization, several limitations and future research directions should be considered. Variations in flow cytometry instruments and settings may impact the reproducibility of our findings, highlighting the need for further optimization across different platforms [34]. Future research should also explore the applicability of our method to other biological fluids and contexts. Investigating the method’s performance with different sample types, such as urine or cerebrospinal fluid, could expand its utility and provide new insights into EV profiles associated with various pathological conditions.
Expanding the ability to isolate and characterize defined EV subsets across diverse biological fluids would not only deepen our understanding of EV biology but also open new avenues for mechanistic studies and the development of EV-based diagnostics and therapeutics.
Supplementary Information
Supplementary Material 1: Supplementary Fig. 1. (a) Representative bidimensional dot plot (530/30-A vs. SSC-A, in logarithmic scale) representing the fluorescent Megamix bead suspension (bead dimensions: 100, 160, 200, 240, 300, 500, and 900 nm) (left panel). The right panel reports the visualization as SSC-A histogram peaks of the various Megamix beads. (b, d) Representative contour plots of EVs falling within the CFSE + EVs gate (left panels) and stained with APC-conjugated anti-CD9 antibody (right panels), using 85 mm (b), 100 mm (c), or 130 mm (d) nozzle.
Supplementary Material 2: Supplementary Fig. 2. (a, c) Histograms representing RCP bead-associated peaks acquired in B530/30 (green) and R670/30 (red) channels, respectively. (b, d) Calibration graphs obtained upon conversion of Relative Channel Number to the relative values of the Mean Channel Number of FITC- (b) and APC (d)-channels through the use of the free available template (Spherotech, Inc). The values corresponding to the calibration curves are shown on the right side of each panel. (e) Histograms representing the CFSE + EVs (left panel) and CFSE + CD9 + EVs (right panel). Inserts within histograms indicate the mean of the relative channel number (CH#) and the corresponding Molecules of Equivalent Fluorescein (Calc. MEFL) or Allophycocyanin (Calc. MEAP), calculated through the use of the calibration graphs.
Acknowledgements
We gratefully acknowledge Giusy Capuano (BD Biosciences, Italy) for her valuable advice and insightful contributions.
Authors' contributions
D.R., M.C.C. and R.T. wrote the manuscript and prepared Figs. 1, 2, 3 and 4 and Supplementary Figure 1. D.R., N.R., A.C. and R.T. performed the flow cytometry experiments. G.P., I.G. and V.D. performed the transmission electron microscopy analysis and prepared figure 5. S.S, S.C. and M.C. performed the miRNA extraction and analysis and prepared figure 6. All authors read and approved the final manuscript.
Funding
This work was supported by Worldwide Cancer Research under Grant (Reference 24–0042) and Gilead Fellowship Program 2022.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Daniele Reverberi and Maria Chiara Ciferri contributed equally to this study.
References
- 1.Loconte L, Arguedas D, El R, Zhou A, Chipont A, Guyonnet L, et al. Detection of the interactions of tumour derived extracellular vesicles with immune cells is dependent on EV-labelling methods. J Extracell Vesicles. 2023;12:e12384. 10.1002/jev2.12384. [DOI] [PMC free article] [PubMed]
- 2.Kumar MA, Baba SK, Sadida HQ, Marzooqi S, Al, Jerobin J, Altemani FH et al. Extracellular vesicles as tools and targets in therapy for diseases. Signal Transduction and Targeted Therapy 2024 9:1 [Internet]. 2024 [cited 2024 Sep 19];9:1–41. Available from: https://www.nature.com/articles/s41392-024-01735-1 [DOI] [PMC free article] [PubMed]
- 3.Arraud N, Linares R, Tan S, Gounou C, Pasquet JM, Mornet S, et al. Extracellular vesicles from blood plasma: determination of their morphology, size, phenotype and concentration. J Thromb Haemost. 2014;12:614–27. [DOI] [PubMed] [Google Scholar]
- 4.Liang Y, Lehrich BM, Zheng S, Lu M. Emerging methods in biomarker identification for extracellular vesicle-based liquid biopsy. J Extracell Vesicles [Internet]. 2021 [cited 2024 Sep 19];10. Available from: http://pmc/articles/PMC8114032/. [DOI] [PMC free article] [PubMed]
- 5.Buntsma NC, Shahsavari M, Gąsecka A, Nieuwland R, van Leeuwen TG, van der Pol E. Preventing swarm detection in extracellular vesicle flow cytometry: a clinically applicable procedure. Res Pract Thromb Haemost [Internet]. 2023 [cited 2025 May 30];7:100171. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC10239693/ [DOI] [PMC free article] [PubMed]
- 6.Morales-Kastresana A, Musich TA, Welsh JA, Telford W, Demberg T, Wood JCS et al. High-fidelity detection and sorting of nanoscale vesicles in viral disease and cancer. J Extracell Vesicles [Internet]. 2019 [cited 2025 Apr 1];8. Available from: https://pubmed.ncbi.nlm.nih.gov/31258878/ [DOI] [PMC free article] [PubMed]
- 7.Bonifay A, Robert S, Champagne B, Petit PR, Eugène A, Chareyre C et al. A new strategy to count and sort neutrophil-derived extracellular vesicles: Validation in infectious disorders. J Extracell Vesicles [Internet]. 2022 [cited 2025 Apr 1];11. Available from: https://pubmed.ncbi.nlm.nih.gov/35362257/ [DOI] [PMC free article] [PubMed]
- 8.Kormelink TG, Ger †, Arkesteijn JA, Nauwelaers FA, Van Den Engh G, Nolte-’t Hoen ENM et al. Prerequisites for the analysis and sorting of extracellular vesicle subpopulations by high-resolution flow cytometry. Wiley Online LibraryTG Kormelink, GJA Arkesteijn, FA Nauwelaers, G van den Engh, ENM Nolte‐’t HoenCytometry Part A, 2016•Wiley Online Library [Internet]. 2015 [cited 2024 Sep 19];89:135–47. Available from: https://onlinelibrary.wiley.com/doi/abs/10.1002/cyto.a.22644 [DOI] [PubMed]
- 9.Maia J, Batista S, Couto N, Gregório AC, Bodo C, Elzanowska J et al. Employing flow cytometry to extracellular vesicles sample microvolume analysis and quality control. Front Cell Dev Biol [Internet]. 2020 [cited 2024 Sep 19];8:593750. Available from: www.frontiersin.org. [DOI] [PMC free article] [PubMed]
- 10.Morales-Kastresana A, Jones JC. Flow Cytometric Analysis of Extracellular Vesicles. Methods in Molecular Biology [Internet]. 2017 [cited 2024 Sep 20];1545:215–25. Available from: https://link.springer.com/protocol/10.1007/978-1-4939-6728-5_16 [DOI] [PMC free article] [PubMed]
- 11.Ciferri MC, Bruno S, Rosenwasser N, Gorgun C, Reverberi D, Gagliani MC et al. Correction to Standardized Method to Functionalize Plasma-Extracellular Vesicles via Copper-Free Click Chemistry for Targeted Drug Delivery Strategies. ACS Appl Bio Mater [Internet]. 2024 [cited 2024 Sep 23];7. Available from: https://pubmed.ncbi.nlm.nih.gov/38993165/ [DOI] [PubMed]
- 12.Holcar M, Ferdin J, Sitar S, Tušek-Žnidarič M, Dolžan V, Plemenitaš A et al. Enrichment of plasma extracellular vesicles for reliable quantification of their size and concentration for biomarker discovery. Sci Rep [Internet]. 2020 [cited 2024 Sep 23];10. Available from: https://pubmed.ncbi.nlm.nih.gov/33288809/ [DOI] [PMC free article] [PubMed]
- 13.Morales-Kastresana A, Welsh JA, Jones JC. Detection and Sorting of Extracellular Vesicles and Viruses Using nanoFACS. Curr Protoc Cytom [Internet]. 2020 [cited 2025 Apr 7];95:e81. Available from: https://onlinelibrary.wiley.com/doi/full/10.1002/cpcy.81 [DOI] [PMC free article] [PubMed]
- 14.Gorgun C, Reverberi D, Rotta G, Villa F, Quarto R, Tasso R. Isolation and Flow Cytometry Characterization of Extracellular-Vesicle Subpopulations Derived from Human Mesenchymal Stromal Cells. Curr Protoc Stem Cell Biol [Internet]. 2019 [cited 2024 Sep 23];48. Available from: https://pubmed.ncbi.nlm.nih.gov/30624011/ [DOI] [PubMed]
- 15.Karimi N, Dalirfardouei R, Dias T, Lötvall J, Lässer C. Tetraspanins distinguish separate extracellular vesicle subpopulations in human serum and plasma– Contributions of platelet extracellular vesicles in plasma samples. J Extracell Vesicles [Internet]. 2022 [cited 2025 Apr 8];11:e12213. Available from: https://pmc.ncbi.nlm.nih.gov/articles/PMC9077141/ [DOI] [PMC free article] [PubMed]
- 16.Khanna K, Salmond N, Halvaei S, Johnson A, Williams KC. Separation and isolation of CD9-positive extracellular vesicles from plasma using flow cytometry. Nanoscale Adv [Internet]. 2023 [cited 2025 Apr 7];5:4435–46. Available from: https://pubs.rsc.org/en/content/articlehtml/2023/na/d3na00081h [DOI] [PMC free article] [PubMed]
- 17.Gardiner C, Ferreira YJ, Dragovic RA, Redman CWG, Sargent IL. Extracellular vesicle sizing and enumeration by nanoparticle tracking analysis. J Extracell Vesicles [Internet]. 2013 [cited 2024 Sep 23];2. Available from: https://www.tandfonline.com/doi/abs/10.3402/jev.v2i0.19671 [DOI] [PMC free article] [PubMed]
- 18.Zavatti M, Beretti F, Giusti I, Poppa G, D’ascenzo S, Esposito L et al. Cancer Three-Dimensional Spheroids Mimic In Vivo Tumor Features, Displaying Inner Extracellular Vesicles and Vasculogenic Mimicry. 2022 [cited 2025 Apr 15]; Available from: 10.3390/ijms231911782 [DOI] [PMC free article] [PubMed]
- 19.Welsh JA, Arkesteijn GJA, Bremer M, Cimorelli M, Dignat-George F, Giebel B et al. A compendium of single extracellular vesicle flow cytometry. J Extracell Vesicles [Internet]. 2023 [cited 2024 Sep 23];12:e12299. Available from: https://onlinelibrary.wiley.com/doi/full/10.1002/jev2.12299 [DOI] [PMC free article] [PubMed]
- 20.Poncelet P, Robert S, Bailly N, Garnache-Ottou F, Bouriche T, Devalet B, et al. Tips and tricks for flow cytometry-based analysis and counting of microparticles. Transfus Apheres Sci. 2015;53:110–26. [DOI] [PubMed] [Google Scholar]
- 21.Rasmussen RW, Botha J, Prip F, Sanden M, Nielsen MH, Handberg A. Zoom in on Antibody Aggregates: A Potential Pitfall in the Search of Rare EV Populations. Biomedicines 2021, Vol 9, Page 206 [Internet]. 2021 [cited 2025 May 28];9:206. Available from: https://www.mdpi.com/2227-9059/9/2/206/htm [DOI] [PMC free article] [PubMed]
- 22.Keller Cecconello D, Spagnol F, Alegretti AP, Pilger DA, Farias MG. Flow cytometry immunophenotyping of healthy platelets and hospitalized patients with suspected platelet dysfunction: Challenges for establishing a cutoff value. Hematol Transfus Cell Ther [Internet]. 2023 [cited 2025 May 30];46 Suppl 5. Available from: https://pubmed.ncbi.nlm.nih.gov/37652805/ [DOI] [PMC free article] [PubMed]
- 23.Tasso R, Marconi S, Rossi G, Genova C, Coco S. Platelets and their derived extracellular vesicles: The new generation of markers in non-small cell lung cancer management. Drug Discov Today [Internet]. 2023 [cited 2025 May 30];28. Available from: https://pubmed.ncbi.nlm.nih.gov/37196761/ [DOI] [PubMed]
- 24.Bordin A, Chirivì M, Pagano F, Milan M, Iuliano M, Scaccia E et al. Human platelet lysate-derived extracellular vesicles enhance angiogenesis through miR-126. Cell Prolif [Internet]. 2022 [cited 2025 May 28];55:e13312. Available from: 10.1111/cpr.13312 [DOI] [PMC free article] [PubMed]
- 25.Min L, Wang B, Bao H, Li X, Zhao L, Meng J et al. Advanced Nanotechnologies for Extracellular Vesicle-Based Liquid Biopsy. Advanced Science [Internet]. 2021 [cited 2024 Sep 23];8:2102789. Available from: https://onlinelibrary.wiley.com/doi/full/10.1002/advs.202102789 [DOI] [PMC free article] [PubMed]
- 26.Welsh JA, Holloway JA, Wilkinson JS, Englyst NA. Extracellular vesicle flow cytometry analysis and standardization. Front Cell Dev Biol [Internet]. 2017;5:269952. Available from: www.frontiersin.org. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Hoshino A, Kim HS, Bojmar L, Gyan KE, Cioffi M, Hernandez J, et al. Extracellular vesicle and particle biomarkers define multiple human cancers. Cell. 2020;182:1044–e106118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pink RC, Beaman EM, Samuel P, Brooks SA, Carter DRF. Utilising extracellular vesicles for early cancer diagnostics: benefits, challenges and recommendations for the future. British Journal of Cancer 2021 126:3 [Internet]. 2022 [cited 2024 Sep 23];126:323–30. Available from: https://www.nature.com/articles/s41416-021-01668-4 [DOI] [PMC free article] [PubMed]
- 29.Welsh JA, Van Der Pol E, Arkesteijn GJA, Bremer M, Brisson A, Coumans F et al. MIFlowCyt-EV: a framework for standardized reporting of extracellular vesicle flow cytometry experiments. J Extracell Vesicles [Internet]. 2020 [cited 2025 Apr 15];9. Available from: https://pubmed.ncbi.nlm.nih.gov/32128070/ [DOI] [PMC free article] [PubMed]
- 30.Wang XQ, Duan XM, Liu LH, Fang YQ, Tan Y. Carboxyfluorescein diacetate succinimidyl ester fluorescent dye for cell labeling. Acta Biochim Biophys Sin (Shanghai) [Internet]. 2005 [cited 2025 May 28];37:379–85. Available from: https://pubmed.ncbi.nlm.nih.gov/15944752/ [DOI] [PubMed]
- 31.Morales-Kastresana A, Telford B, Musich TA, Mckinnon K, Clayborne C, Braig Z et al. Labeling extracellular vesicles for nanoscale flow cytometry. nature.comA Morales-Kastresana, B Telford, TA Musich, K McKinnon, C Clayborne, Z Braig, A RosnerScientific reports, 2017•nature.com [Internet]. 2017 [cited 2024 Sep 23];7. Available from: https://www.nature.com/articles/s41598-017-01731-2 [DOI] [PMC free article] [PubMed]
- 32.Mastoridis S, Bertolino GM, Whitehouse G, Dazzi F, Sanchez-Fueyo A, Martinez-Llordella M. Multiparametric analysis of Circulating exosomes and other small extracellular vesicles by advanced imaging flow cytometry. Front Immunol [Internet]. 2018 [cited 2025 May 28];9:358079. Available from: www.frontiersin.org. [DOI] [PMC free article] [PubMed]
- 33.Dlugolecka M, Czystowska-Kuzmicz M. Factors to consider before choosing EV labeling method for fluorescence-based techniques. Front Bioeng Biotechnol. 2024;12:1479516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Kalina T. Reproducibility of Flow Cytometry Through Standardization: Opportunities and Challenges. Cytometry Part A [Internet]. 2020 [cited 2024 Sep 23];97:137–47. Available from: https://onlinelibrary.wiley.com/doi/full/10.1002/cyto.a.23901 [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Supplementary Material 1: Supplementary Fig. 1. (a) Representative bidimensional dot plot (530/30-A vs. SSC-A, in logarithmic scale) representing the fluorescent Megamix bead suspension (bead dimensions: 100, 160, 200, 240, 300, 500, and 900 nm) (left panel). The right panel reports the visualization as SSC-A histogram peaks of the various Megamix beads. (b, d) Representative contour plots of EVs falling within the CFSE + EVs gate (left panels) and stained with APC-conjugated anti-CD9 antibody (right panels), using 85 mm (b), 100 mm (c), or 130 mm (d) nozzle.
Supplementary Material 2: Supplementary Fig. 2. (a, c) Histograms representing RCP bead-associated peaks acquired in B530/30 (green) and R670/30 (red) channels, respectively. (b, d) Calibration graphs obtained upon conversion of Relative Channel Number to the relative values of the Mean Channel Number of FITC- (b) and APC (d)-channels through the use of the free available template (Spherotech, Inc). The values corresponding to the calibration curves are shown on the right side of each panel. (e) Histograms representing the CFSE + EVs (left panel) and CFSE + CD9 + EVs (right panel). Inserts within histograms indicate the mean of the relative channel number (CH#) and the corresponding Molecules of Equivalent Fluorescein (Calc. MEFL) or Allophycocyanin (Calc. MEAP), calculated through the use of the calibration graphs.
Data Availability Statement
No datasets were generated or analysed during the current study.