Abstract
Background:
Diabetes mellitus (DM) is a major public health problem that badly affects oral health and overall quality of life. This study aimed to examine oral health problems and associated factors of type 2 DM patients attending governmental primary health centers (PHCs) in the Gaza strip, Palestine.
Methods:
A cross-sectional study was applied to 376 patients with type 2 DM selected through systematic random sampling from five randomly selected governmental PHCs. The World Health Organization Decayed, Missing and Filling Tooth (DMFT) index and semistructured questionnaire were used to collect data accordingly. Descriptive and analytical analyses were applied using the using the IBM SPSS for windows version 23, IBM, USA software.
Results:
The mean DMFT index is 13.18 ± 6.39, and it is higher among patients having type 2 DM for more than 10 years and with a history of chronic diseases. The mean teeth with loss of attachment are 1.88 ± 0.76 and are higher among males, exsmokers, patients having type 2 DM for more than 10 years, and those with a history of chronic diseases. Gingival bleeding is higher among males and exsmokers. The majority of participants had no periodontitis (160/376; 42.6%). In return, 8.5% (32/376) presents with severe periodontitis. The mean of pocket is higher among patients having uncontrolled DM and type 2 DM <10 years.
Conclusions:
Patients with type 2 DM suffer from many oral health problems, especially dental caries, periodontitis and loss of attachment. The findings underscore the significance of early intervention and consistent dental care for people with type 2 DM in order to avert and handle oral health complications. Oral health education campaigns that are effective should be customized to meet the particular cultural, socioeconomic, and healthcare challenges.
Keywords: Dental carries, DMFT, loss of attachment, periodontitis, type 2 diabetes mellitus
Introduction
It is one of the most common noncommunicable diseases and a significant public health issue, diabetes mellitus (DM) affects around 8.5% of people globally and has serious repercussions.[1] It is a metabolic disease caused by either resistance to the effects of insulin or a lack of insulin production.[2] It is primarily divided into two types: type 1 and type 2, with the latter being the most common (>87% of cases), which is also the situation in Palestine. Before the development of effective treatments, diabetes was often fatal, but today, its long-term complications remain common.
According to reports, 50% of patients who go misdiagnosed run the danger of developing major issues that could impact their social,[3] psychological,[4] and physical well-being.[5] One example of these problems is oral diseases. There is proof that DM alters the mouth cavity in a harmful way. Although the list is lengthy, it is primarily caused by dental caries, xerostomia (dry mouth), fungal infection, bone loss, and periodontitis.[5] Although many oral alterations are thought to be early indicators of diabetes, they are not pathognomonic.[6] Since most DM patients go undetected, dentists can play a significant role in the early detection of the disease by identifying these symptoms.
There is a connection between DM and oral complications. It has been demonstrated that individuals with type 2 DM (T2DM) experience a variety of oral complications, particularly xerostomia, dental caries, tooth loss, and periodontitis, as well as severe fungal and bacterial infections. A number of studies confirmed the relationship between low quality of life and poor oral health status.[7] Bacterial colonization on tooth surfaces is what leads to dental caries. Patients with T2DM have a higher susceptibility to oral infections that cause tooth decay and loss. Bissong et al. found that the prevalence of dental caries was greater among individuals with T2DM.[8] The connection between diabetes and the development of dental caries remains uncertain. However, it is hypothesized that the glucose leaked into the oral cavity facilitates. The proliferation of aciduric and acidogenic bacteria led to the development of dental caries.[9] Diabetes patients exhibited a significantly elevated Decayed, Missing and Filling Tooth (DMFT) index.[10] Through the DMF-S, research by Ribeiro et al. found that individuals with T2DM exhibited dental caries rates that were significantly elevated (relative risk [RR] =1.37; 95% CI = 1.09–1.71).[11]
Periodontitis is a class of diseases that impacts the gingiva, periodontal ligaments, cementum, and alveolar bone, which together form the supportive structure for the teeth. If not treated, chronic periodontitis can lead to irreversible tooth and bone loss. A number of studies have investigated how DM affects periodontal tissue and have identified a connection between the two. There is a genuine two-way connection between diabetes and periodontitis. given the strong evidence that hyperglycemia adversely affects oral health via the overproduction of advanced glycation end products (AGE),[12] and that severe periodontitis can hinder glycemic control Dental caries are caused by the destruction of dental hard tissue from acidic and proteolytic substances, a process that is further compounded by diabetes mellitus.[9]
Research carried out by Preshaw et al. demonstrated that periodontitis influences glycemic status in patients with T2DM in both directions.[13] For diabetic patients with poor glycemic control, the risk of periodontitis rises.[14] It has also been reported that certain factors, including smoking, inadequate oral hygiene, and prolonged diabetes duration, increase the susceptibility of T2DM patients to periodontitis.[15] 68% of diabetic patients exhibited moderate periodontitis, as indicated by the Picture of Periodontal status index ≥4.[16] DM patients have a threefold increase in periodontitis risk compared to nondiabetics. However, this risk is significantly reduced among individuals with controlled T2DM.[17] Nascimento et al. discovered in their meta-analysis of prospective cohort studies that the risk of periodontitis advancement for patients with diabetes was 86% higher (RR = 1.86; 95% CI = 1.3–2.8).[18]
The prevalence of DM in the Gaza Strip is 6.3% in the general population aged 18 and 60, and the incidence is 149.4 per 100,000 inhabitants.[19] The Ministry of Health (MoH) and the United Nations Relief and Works Agency for Palestine Refugees (UNRWA) for registered refugees provide most of the primary healthcare in the Palestinian health system, including diabetes care, dental care, and school health. In the Gaza Strip, there are difficulties in accessing dental care, stemming from a lack of medical supplies, dentists, and modern equipment. Furthermore, the economic circumstances in the Gaza Strip are difficult; more than half of Gaza’s population lives below the poverty line, and a considerable portion of households find it difficult to satisfy their fundamental requirements, such as food and medical care, which leads them to depend on humanitarian assistance. This implies that oral health issues in diabetic patients might remain unaddressed or poorly managed, thereby exacerbating their overall health and diabetes regulation.
There aren’t many studies on type 2 diabetes’ oral health in the Gaza Strip.[20] This study, which concentrated on patients who visited UNRWA health centers, revealed that they had a high DMFT index and were ignorant of the consequences diabetes had on the mouth. There is no information on patients who visit MoH health centers. The ministry’s yearly reports contain the information that is currently available regarding dental care and patient volume; however, there is a lack of accurate and comprehensive data regarding the oral health status of patients with type 2 DM. Thus, the purpose of this study was to assess the oral health condition of individuals with type 2 DM who were attending primary health clinics (PHCs) of the MoH. This included gingival bleeding, loss of attachment, dental caries and missing, and periodontitis. It is crucial to study the oral health problems in the Gaza Strip to gain a better understanding of local health challenges, enhance healthcare interventions, prevent complications, and improve the quality of life for individuals affected by diabetes.
Materials and Methods
Study design and setting
Patients who attended governmental PHCs and had type 2 DM participated in a facility-based cross-sectional study. Fifty-two PHCs are distributed throughout five governorates in Gaza and are run by the MoH. Five PHCs offering dental services were chosen at random, with one PHC serving as a governorate representative. These PHCs are Beit Lahia Martyrs in the north, Al-Remal Martyrs Health Center in Gaza city, Der-Albalh Martyrs clinic in the middle, Khanyounes Martyrs Clinic and Rafah Martyrs Clinic in the south.
Study period and populations
Data for the study was gathered over the course of six months between 2021 and 2022. Patients with type 2 DM who visited government PHCs are included in the study population. MoH reported that there are 66,209 DM patients overall registered with MoH and UNRWA databases.[19] For patients over the ages of 18, 40, and 60, the prevalence is 6.3%, 17.6%, and 49%, respectively. The overall registered number of type 2 DM patients is 11088 patients in governmental PHCs distributed in the five governorates: 1389, 5949, 918, 2173, and 689 in the north, Gaza, middle area, Khanyounes, and Rafah governorates, respectively.
Inclusion criteria
Patients should have had a type 2 DM diagnosis for at least 5 years
Participants ought to be helpful
Participants must be older than forty
At least 20 teeth should be present in each participant’s oral cavity.
Exclusion criteria
People without any teeth in their mouths
Elderly people with Alzheimer’s disease or who are mute
Patients who experienced trauma or an accident and lost their teeth accordingly.
Sample size and sampling process
The online SurveyMonkey website, which can be accessed at https://www.surveymonkey.com/mp/sample-size-calculator/, was used to determine the sample size. The sample was determined with 95% confidence interval and 5% margin of error. We used the most recent MoH report from 2021, which indicated that the prevalence of type 2 diabetes is 17.6%, meaning that there were roughly 11088 people over the age of 40.[19] Three hundred seventy-two patients should be included, according to the sample calculation. A two-phase sampling strategy was followed. In the first phase, one PHC is chosen at random from each governorate out of five governorates. In the second phase, participants are chosen at random which comes after proportionating stratified systematic sampling. Numbers of T2DM patients registered in each governorate were determined and then were proportionally selected. With 48 from the north, 199 from Gaza, 31 from the middle zone, and 73 and 25 from Khanyounes and Rafah, respectively, the sample size was evenly split among the five governorates. The distribution percentages are 12.5%, 53.5%, 8.25%, 19.5%, and 6.2%, in that order. A list of the patients, both male and female, from each of the study’s participating health centers was then created. The Kth interval is 30.
Data collection tools
A semistructured questionnaire and a clinical examination were used to gather data during the day shift. Sociodemographic (age, marital status, income, education level, etc.) and clinical (duration of DM, presence of chronic diseases, presence of oral diseases, last HbA1c reading, etc.) variables made up the first two sections of the questionnaire. The clinical examiner was blinded from patients, and the selection of participants was made by the head nurse of the selected PHCs who was involved in the study to collect data on sociodemographic and clinical variables.
Patients were invited to sit in a dental chair for clinical examinations, and mirrors and probes were utilized accordingly. A mirror and a probe were used to find decaying, missing, and filled teeth using the DMFT index. The DFMT index sheet used for the dental examination has the following coding: Dental caries is represented by the letter “D” and has code [1]. “F” accepts code [2,3,6] and represents filling teeth. Taking code [4,5], “M” denotes missing teeth.
The bleeding on probing, probing pocket depth, and loss of attachment were measured using the WHO community periodontal index (CPI) to examine periodontitis. To determine whether a bleeding response was present or not, the tip of the WHO CPI probe was gently inserted between the gingiva and each tooth in the mouth. No more than 20g of sensing force was applied. The ball tip of the probe followed the tooth root’s surface anatomically when it was implanted. The entire extent of the gingival sulcus or pocket was examined after the probe tip was carefully placed. The probe was carefully advanced down the buccal sulcus/pocket to the mesial surface of the second molar, making brief upward and downward motions. Starting with the distolingual aspect of the second molar, a similar process was performed for lingual surfaces.
Each tooth’s probing pocket depth is scored on a 6-point scale, and the number of bleeding seconds after probing is recorded.
The CPI consists of measuring six sites on each tooth (usually the mesial, midline, and distal areas on both the buccal and lingual surfaces). For measurement purposes, at least two teeth from each quadrant of the mouth (upper right and left, lower right and left), one anterior and one posterior, are usually selected. Typically, the molar or first incisor is used. The WHO CPI probe is inserted gently into the gingival sulcus (the space between the tooth and the gum), making sure it adheres to the natural contour of the tooth.
The WHO CPI probe will show the pocket depth by the location of the colored bands:
0–3.5 mm: If the probe reaches the first colored band (3.5 mm), this is considered a healthy pocket (no sign of disease)
>3.5 mm: Measure deeper into the pocket, noting if the probe reaches 5.5 mm or 8.5 mm (which indicates periodontitis).
It is normal to experience loss of attachment if it measures 0–3 mm and 4–5 mm at the cemento-enamel junction (CEJ) within the black band. Considering that it measures 6–8 mm CEJ between the upper limit of the black band and the 8.5 mm ring, it could also be mild. The loss of attachment is classified as moderate or severe based on the CEJ measurements: it is 9–11 mm within the 8.5 mm to 11.5 mm ring and 12 mm or more beyond the 11.5 mm ring. The presence or absence of pockets and loss of attachment[21] was examined using teeth 16, 17, 11, 26, 27, 36, 37, 31, 46, and 47.
Ethical considerations
The Helsinki Committee of the Palestinian Health Research Council provided ethical approval (PHRC/HC/858/21). Additionally, the MoHs general office of human resources granted permission to carry out the study in its PHCs (No: 686506). Patients received information on the goals of the trial. Confidentiality, anonymity, and voluntary involvement were all guaranteed. Before completing the questionnaire, patients were requested to sign the attached consent form.
Data analysis
Data analysis was conducted using version 22 of the Statistical Package for Social Sciences. Data were examined for missing and extreme values before analysis. The descriptive analysis method was used. For the continuous variables of DMFT, periodontitis, and certain clinical parameters, mean ± standard deviation and median ± standard errors were used. Frequencies and percentages were used to display categorical variables. Because of its non-normal distribution, the DMFT was analyzed using the Mann–Whitney U test. ANOVA and t-test analysis were used to compare DMFT, periodontitis, loss of attachment, and gingival bleeding with independent variables (sociodemographic and clinical variables). The statistical significance was set at P < .05.
Results
Characteristics of participated patients
Three hundred seventy-six individuals with type 2 DM took part in the study. The participants’ mean age is 57.8 ± 8.76 years. Males make up 44.9% of the sample. Patients under the age of 55 and those over 65 make up at least one-fifth of each of the five age groups. Most participants (93.6%) completed formal education, with 17.3% and 27.7% attending preparatory and elementary schools, respectively. Nearly three-quarters do not smoke (77.7%), and 85.2% have at least six children. In return, 10.4% are exsmokers, and the average daily cigarette consumption is 11.45 ± 8.28 cigarettes. The majority of participants live below the severe poverty line (83.5%) and are unemployed (72.9%) at the time of data collection [Table 1].
Table 1.
Sociodemographic characteristics of the participants (n=376)
| Variables | Frequency | Percentage | Mean±SD |
|---|---|---|---|
| Age group (years) | 57.86±8.76 | ||
| 50 or less | 84 | 22.3 | |
| 51–55 | 69 | 18.4 | |
| 56–60 | 75 | 19.9 | |
| 61–65 | 70 | 18.6 | |
| More than 65 | 78 | 20.7 | |
| Residency | |||
| North Gaza | 48 | 12.8 | |
| Gaza city | 199 | 52.9 | |
| Middle zone | 31 | 8.2 | |
| Khan Younis | 73 | 19.4 | |
| Rafah | 25 | 6.6 | |
| Level of education | |||
| Illiterate | 24 | 6.4 | |
| Up to elementary school | 65 | 17.3 | |
| Up to preparatory school | 104 | 27.7 | |
| Up to secondary school | 113 | 30.1 | |
| University and above | 70 | 18.6 | |
| Marital status | |||
| Unmarried | 62 | 16.5 | |
| Married | 314 | 83.5 | |
| Income | |||
| Under poverty line (<1974 NIS) | 314 | 83.5 | |
| Above poverty line (≥1974 NIS) | 62 | 16.5 | |
| Number of family members | 1.71±0.76 | ||
| 5 or less | 102 | 27.1 | |
| 6–10 | 219 | 58.2 | |
| More than 10 | 55 | 14.6 | |
| Working status | |||
| Working | 102 | 27.1 | |
| Not working | 274 | 72.9 |
SD=Standard deviation
Type 2 DM mean duration is 10.99 ± 6.61 years. Nearly two-thirds (64.2%) have had diabetes for <10 years. Additionally, 12.8% use insulin, 17% use a combination of insulin injection and oral hypoglycemic medicines, and two thirds (65.4%) are receiving therapy with oral hypoglycemic agents. The mean HbA1c is 8.11% ±1.84%, and about one-fifth of people have controlled diabetes (HbA1c <7%). Yet, in addition to type 2 DM, two-thirds of individuals suffer from other chronic conditions, primarily hypertension (43.9%) and hypertension with heart disease (10.9%). Of those with type 2 DM, half (49.5%) have a family history of oral problems. In the past 12 months, the majority of individuals (79.8%) reported oral manifestations, and 56.6% attended dental clinics as a result. Participants frequently reported tooth decay (58.2%) and tooth loss (53.2%) as oral symptoms. Problems with teeth, gums, or mouths account for most visits to dental clinics (57.2%) [Table 2].
Table 2.
Clinical characteristics of the participants (n=376)
| Variables | Frequency | Percentage | Mean±SD |
|---|---|---|---|
| Having medical insurance | |||
| Yes | 367 | 97.6 | |
| No | 9 | 2.4 | |
| Duration of DM (years) | 10.99±6.61 | ||
| 10 or less | 241 | 64.1 | |
| More than 10 | 135 | 35.9 | |
| Receiving education about oral care | |||
| Yes | 298 | 79.3 | |
| No | 78 | 20.7 | |
| Last reading of FBS (mg/dl) | 185.9±61.33 | ||
| ≤140 | 156 | 41.5 | |
| >140 | 220 | 58.5 | |
| HbA1c (%) | 8.11%±1.84% | ||
| ≤7 | 169 | 38 | |
| ≥7 | 207 | 62 | |
| Presence of other chronic diseases | |||
| No | 236 | 62.8 | |
| Yes | 140 | 37.2 | |
| If yes, specify | |||
| Hypertension | 165 | 43.9 | |
| Hypertension, heart disease | 16 | 4.3 | |
| Heart disease | 4 | 1.1 | |
| Arthritis | 4 | 1.1 | |
| Hypertension, asthma | 1 | 0.3 | |
| Arthritis, hypertension | 1 | 0.3 | |
| Hypertension, cancer | 1 | 0.3 | |
| Hypertension, kidney disease | 2 | 0.5 | |
| Osteoporosis | 6 | 1.6 | |
| Suffering from oral diseases in the last year | |||
| Yes | 300 | 79.8 | |
| No | 76 | 20.2 | |
| Among yes reporting to have oral diseases in the last year | |||
| Tooth mobility | 86 | 22.9 | |
| Tooth loss | 290 | 77.1 | |
| Tooth decay | 200 | 53.2 | |
| Tooth sensitivity | 157 | 41.8 | |
| Gum bleeding | 112 | 29.8 | |
| Bacterial infection | 88 | 23.4 | |
| Fungal infection | 56 | 14.9 | |
| Mouth ulcers | 39 | 10.4 | |
| Visiting dental clinic last year | |||
| Yes | 213 | 56.6 | |
| No | 163 | 43.4 | |
| If yes, why | |||
| Consultation/advice | 42 | 11.2 | |
| Pain or trouble with teeth, gums or mouth | 135 | 35.9 | |
| Treatment, follow-up treatment | 35 | 9.3 | |
| Routine check-up/assessment | 21 | 5.6 | |
| Don’t know/don’t remember | 33 | 8.8 | |
| Total | 376 | 100.0 |
SD=Standard deviation, FBS=Fasting blood sugar, DM=Diabetes mellitus
Dental status
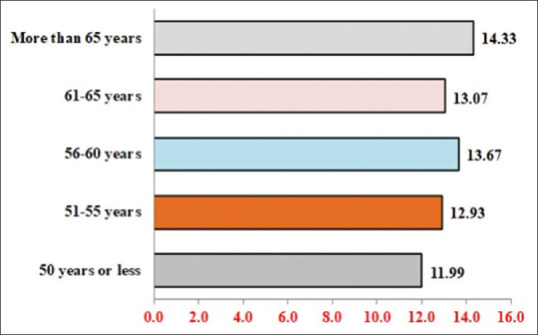
The mean of DMFT is 13.18 ± 6.39. The average number of decayed teeth is 5.74 ± 4.49, whereas the average number of teeth that have been lost because of caries is 5.22 ± 4.20, and the average number of teeth that have been lost because of various causes other than caries is 0.78 ± 2.12. Furthermore, it appears that there are very few caries-filled teeth on average (0.55 ± 1.15) [Table 3]. As people aged, the mean DFMT rose (P < .05) [Figure 1].
Table 3.
Distribution of the study participants according to dentition status (n=376)
| Crown | Sum | Mean | Median | SD |
|---|---|---|---|---|
| 0=Sound | 5719 | 15.21 | 15 | 6.63 |
| 1=Caries | 2158 | 5.74 | 5 | 4.49 |
| 2=Filled w/caries | 205 | 0.55 | 0 | 1.15 |
| 3=Filled, no caries | 340 | 0.9 | 0 | 1.47 |
| 4=Missing due to caries | 1959 | 5.22 | 4 | 4.2 |
| 5=Missing for any another reason | 295 | 0.78 | 0 | 2.12 |
| 6=Fissure sealant | 0 | 0 | 0 | 0 |
| 7=Fixed dental prosthesis/crown abutment, veneer, implant | 1118 | 2.97 | 0 | 4.66 |
| 8=Unerupted | 131 | 0.35 | 0 | 0.97 |
| 9=Not recorded | 107 | 0.28 | 0 | 0.94 |
SD=Standard deviation
Figure 1.
Distribution of decayed, missing, and filling tooth according to age
Regarding gingival bleeding, the median score for 82 (21.8%) patients was 5.12 ± 0.34. The median periodontal pocket size is 3.97 ± 0.28 in 58 out of 376 patients (15.4%). The median teeth that have loss of attachment is 1.88 ± 0.04. Of the subjects, 23.2% (87/376) exhibit severe loss of attachment, while 32.4% and 44.4% exhibit mild and moderate loss of attachment, respectively [Table 4].
Table 4.
Distribution of the study population according to periodontal status (n=376)
| Periodontal status | n | Percentage | Mean±SD |
|---|---|---|---|
| Gingival bleeding | |||
| Yes | 82 | 21.8 | 5.12±6.65 |
| No | 294 | 78.2 | |
| Yes | 58 | 15.4 | 3.97±5.43 |
| No | 318 | 84.6 | |
| Loss attachment | |||
| 0_3 mm | 122 | 32.4 | 1.88±0.76 |
| 4_5 mm | 167 | 44.4 | |
| 6_8 mm | 60 | 16.0 | |
| 9_11 mm | 18 | 4.8 | |
| 12 mm or more | 9 | 2.4 | |
| Total | 376 | 100.0 |
SD=Standard deviation
Many participants (160/376; 42.6%) do not have periodontitis. In return, mild periodontitis, moderate periodontitis, and severe periodontitis are present in 27.4%, 21.5%, and 8.5% of individuals, respectively.
Inferential analysis
Males and former smokers have greater mean DMFTs (P < .05). Although they are not statistically significant (P > 0.05), patients who are illiterate, have a family size greater than 10, and reside below the poverty line are found to have higher DMFT [Table 5].
Table 5.
The mean differences of dentition status decayed, missing, and filling related to sociodemographic data among study population (n=376)
| Sociodemographic data | (n=376) | DMF | (Maximum=32) Mean±SD DMFT | Statistical test | |
|---|---|---|---|---|---|
|
| |||||
| DMFT t/F | P | ||||
| Age group | |||||
| ≤50 | 84 | 1007 | 11.99±7.21 | 1.516 | 0.197 |
| 51–55 | 69 | 892 | 12.93±6 | ||
| 56–60 | 75 | 1025 | 13.67±5.46 | ||
| 61–65 | 70 | 915 | 13.07±5.99 | ||
| >65 | 78 | 1118 | 14.33±6.85 | ||
| Gender | |||||
| Male | 169 | 2368 | 14.01±6.58 | 2.268 | 0.024* |
| Female | 207 | 2590 | 12.51±6.16 | ||
| Residency | |||||
| North Gaza | 48 | 774 | 16.13±4.85 | 6.897 | 0.000** |
| Gaza city | 199 | 2663 | 13.38±6 | ||
| Middle zone | 31 | 383 | 12.35±5.89 | ||
| Khan Younis | 73 | 764 | 10.47±7.1 | ||
| Rafah | 25 | 373 | 14.92±7.47 | ||
| Smoker | |||||
| Yes | 45 | 637 | 14.16±6.25 | 4.844 | 0.008* |
| No | 292 | 3703 | 12.68±6.31 | ||
| Exsmoker | 39 | 617 | 15.82±6.5 | ||
| Level of education | |||||
| Illiterate | 24 | 383 | 15.96±4.26 | 1.961 | 0.100 |
| Up to elementary school | 65 | 841 | 12.94±6.31 | ||
| Up to prepar. school | 104 | 1422 | 13.67±5.64 | ||
| Up to secondary school | 113 | 1472 | 13.03±7.12 | ||
| University and above | 70 | 839 | 11.99±6.67 | ||
| Marital status | |||||
| Unmarried | 62 | 774 | 12.48±6.4 | −0.944 | 0.346 |
| Married | 314 | 4182 | 13.32±6.39 | ||
| Number of family member | |||||
| ≤5 | 102 | 1278 | 12.53±6.79 | 1.047 | 0.352 |
| 6–10 | 219 | 2906 | 13.27±6.23 | ||
| >10 | 55 | 772 | 14.04±6.22 | ||
| Working status | |||||
| Working | 102 | 1400 | 13.73±6.23 | 1.004 | 0.316 |
| Not working | 274 | 3556 | 12.98±6.45 | ||
| Income | |||||
| Under deep poverty line (<1974 NIS) | 314 | 1461 | 13.25±6.55 | 0.443 | 0.658 |
| Above deep poverty line (1974 or more NIS) | 62 | 797 | 12.85±5.52 | ||
*P<0.05, **P<0.001. SD=Standard deviation, DMFT=Decayed, Missing and Filling Tooth
Gender and smoking status are significantly correlated with gingival bleeding (P < .05). Males are more likely than females to have bleeding on average (6.22 ± 7.44 and 4.22 ± 5.79, respectively). Gingival bleeding is more common in patients who are illiterate, between the ages of 56 and 60, have a family size of fewer than five, and live below the poverty line; however, this difference is not statistically significant (P > .05).
Smoking status is significantly significant when it comes to pocket presence (P < .05). The mean pocket size is greater for exsmokers (7.92 ± 7.68). However, it is not statistically significant (P > .05) that patients who are male, between the ages of 61 and 65, unemployed, and below the poverty line had bigger mean pockets. Furthermore, there are notable distinctions in the prevalence of loss of attachment between smoking status, gender, and the number of family members (P < .05). Males (2.52 ± 2.13), smokers (2.44 ± 2.06), and families with fewer than five individuals (2.47 ± 2.23) have the highest mean loss of attachment. In return, illiterate patients over 65 and who are below the poverty line have higher rates of attachment loss than patients in other groups; nevertheless, the difference is not statistically significant (P > .05) [Table 6].
Table 6.
The mean differences in (periodontal status) bleeding, pockets, loss of attachment related to sociodemographic data among study population (n=376)
| Sociodemographic data | Bleeding on pocket | Loss of attachment | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
|
|
|
|
|||||||||
| (n=376) | Sum | (Maximum=32) Mean±SD | t/F | P | (Maximum=32) Mean±SD | t/F | P | (Maximum=6) Mean±SD | t/F | P | |
| Age group | |||||||||||
| ≤50 | 84 | 146 | 4.2±5.74 | 0.755 | 0.555 | 3.95±5.76 | 0.986 | 0.562 | 1.74±2.09 | 1.332 | 0.258 |
| 51–55 | 69 | 142 | 5.62±7.18 | 3.06±4.99 | 2.06±2.07 | ||||||
| 56–60 | 75 | 158 | 5.71±6.62 | 4.03±5.04 | 2.11±1.98 | ||||||
| 61–65 | 70 | 157 | 4.71±6.15 | 4.89±5.87 | 2.24±2.02 | ||||||
| >65 | 78 | 191 | 5.45±7.52 | 3.92±5.4 | 2.45±1.98 | ||||||
| Gender | |||||||||||
| Male | 169 | 426 | 6.22±7.44 | 2.933 | 0.004* | 4.41±5.48 | 1.413 | 0.159 | 2.52±2.13 | 3.576 | 0.000** |
| Female | 207 | 368 | 4.22±5.79 | 3.61±5.38 | 1.78±1.9 | ||||||
| Residency | |||||||||||
| North Gaza | 48 | 174 | 9.35±5.8 | 41.547 | 0.000** | 0.06±0.43 | 20.894 | 0.000** | 3.63±2.25 | 11.318 | 0.000** |
| Gaza city | 199 | 343 | 3.28±4.94 | 4.08±4.95 | 1.72±1.75 | ||||||
| Middle zone | 31 | 47 | 6.06±6.2 | 2.68±3.63 | 1.52±1.36 | ||||||
| Khan Younis | 73 | 156 | 3.03±5.79 | 4.4±6.1 | 2.14±2.33 | ||||||
| Rafah | 25 | 74 | 16.52±7.83 | 11±6.8 | 2.96±1.97 | ||||||
| Smoker | |||||||||||
| Yes | 45 | 110 | 6.67±7.78 | 2.993 | 0.050* | 3.58±4.31 | 12.202 | 0.000** | 2.44±2.06 | 4.448 | 0.012* |
| No | 292 | 571 | 4.45±5.96 | 3.5±5.02 | 1.96±2 | ||||||
| Exsmoker | 39 | 113 | 8.36±8.78 | 7.92±7.68 | 2.9±2.12 | ||||||
| Level of education | |||||||||||
| Illiterate | 24 | 66 | 8.13±7.79 | 1.404 | 0.232 | 3.92±4.87 | 1.584 | 0.178 | 2.75±2.31 | 1.000 | 0.407 |
| Up to elementary school | 65 | 126 | 4.78±6.37 | 3.86±5.32 | 1.94±2.02 | ||||||
| Up to preparatory school | 104 | 224 | 4.66±6.09 | 2.95±4.85 | 2.15±2.09 | ||||||
| Up to secondary school | 113 | 247 | 5.17±6.83 | 4.73±6.23 | 2.19±2.12 | ||||||
| University and above | 70 | 131 | 4.99±6.91 | 4.37±5.05 | 1.87±1.69 | ||||||
| Marital status | |||||||||||
| Unmarried | 62 | 136 | 3.74±5.7 | −1.787 | 0.075 | 4.02±5.82 | 0.072 | 0.943 | 2.19±2.12 | 0.346 | 0.729 |
| Married | 314 | 658 | 5.39±6.8 | 3.96±5.37 | 2.1±2.02 | ||||||
| Number of family member | |||||||||||
| ≤5 | 102 | 376 | 5.3±7.35 | 0.078 | 0.925 | 5.22±6.43 | 2.923 | 0.061 | 2.47±2.23 | 4.102 | 0.017* |
| 6–10 | 219 | 254 | 5±6.55 | 3.41±4.86 | 1.87±1.84 | ||||||
| >10 | 55 | 164 | 5.22±5.7 | 3.89±5.31 | 1.86±1.89 | ||||||
| Working status | |||||||||||
| Employed | 102 | 669 | 6.15±7.09 | 1.838 | 0.067 | 3.5±4.96 | −1.025 | 0.306 | 2.13±2.05 | 0.404 | 0.686 |
| Not employed | 274 | 125 | 4.73±6.45 | 4.15±5.6 | 2.02±1.98 | ||||||
| Income | |||||||||||
| Under deep poverty line (<1974 NIS) | 314 | 102 | 5.38±6.61 | 1.703 | 0.089 | 3.86±5.43 | −0.890 | 0.374 | 274 | 2.69±2.26 | 3.386 |
| Above deep poverty line (≥1974 NIS) | 62 | 274 | 3.81±6.73 | 4.53±5.45 | 520 | 1.9±1.9 | |||||
*P<0.05, **P<0.001. SD=Standard deviation
Patients who have had type 2 DM for more than 10 years have higher mean DMFT, gingival bleeding, and loss of attachment, while those who have had the disease for < 10 years have higher mean pocket (P < .05). Similar results are observed for controlled diabetes (HbA1c > 7%); nevertheless, gingival bleeding and loss of attachment are statistically significant (P < .05) [Table 7].
Table 7.
Distribution of decayed, missing, and filling tooth and periodontitis (bleeding, pocket, and loss of attachment) related to duration of DM and HbA1c among study population (n=376)
| Domains | Duration DM (years) | n | Sum | Mean | SD | t | P |
|---|---|---|---|---|---|---|---|
| DMFT (maximum=32) | 10 or less | 241 | 3148 | 13.06 | 6.39 | 0.491 | 0.623 |
| More than 10 | 135 | 1809 | 13.40 | 6.41 | |||
| Bleeding (maximum=32) | 10 or less | 241 | 1153 | 4.78 | 6.53 | 1.298 | 0.195 |
| More than 10 | 135 | 771 | 5.71 | 6.85 | |||
| Poket-32 (maximum=32) | 10 or less | 241 | 961 | 3.99 | 5.44 | −0.080 | 0.936 |
| More than 10 | 135 | 532 | 3.94 | 5.44 | |||
| Loss of attachment (maximum=6) | 10 or less | 241 | 496 | 2.06 | 2.03 | 0.682 | 0.496 |
| More than 10 | 135 | 298 | 2.21 | 2.04 | |||
|
| |||||||
| Domains | Last reading (HbA1c) | n | Sum | Mean | SD | t | P |
|
| |||||||
| DMFT (maximum=32) | ≤7% | 143 | 1863 | 13.03 | 6.30 | 0.369 | 0.712 |
| >7% | 233 | 3094 | 13.28 | 6.46 | |||
| Bleeding (maximum=32) | ≤7% | 143 | 596 | 4.17 | 5.92 | 2.179 | 0.030* |
| >7% | 233 | 1328 | 5.70 | 7.01 | |||
| Pocket (maximum=32) | ≤7% | 143 | 579 | 4.05 | 5.63 | −0.218 | 0.827 |
| >7% | 233 | 914 | 3.92 | 5.32 | |||
| Loss of attachment (maximum=6) | ≤7% | 143 | 263 | 1.84 | 1.92 | 2.043 | 0.042* |
| >7% | 233 | 531 | 2.28 | 2.09 | |||
*P<0.05. SD=Standard deviation, DMFT=Decayed, missing, and filling Tooth, DM=Diabetes mellitus
Although it is not significant, the mean DMFT is higher among individuals who reported having chronic conditions than those who did not (13.3 ± 6.2 vs. 12.9 ± 6.6). The mean of gingival bleeding, periodontal pocket, and loss of attachment is seen to be high among patients with no history of chronic diseases; however, it is not statistically significant (P < .05).
Discussion
Many studies examined the oral symptoms in relation to type 2 DM including dental carries, gingivitis, periodontitis, salivary dysfunction, changed taste, oral mucosal disorders, and infections.[9,20] In this study, we assessed DMFT, missing teeth, gingival bleeding, loss of attachment, and periodontitis in patients with type 2 DM attended governmental PHCs.
The WHO has recognized oral health as one of the most important public health concerns because of the impact it has on day-to-day living. The DMFT index has been regarded as the most significant index for evaluating oral and dental health globally for almost 70 years. In public health epidemiological investigations, this measure is also crucial. Patients with DM may exhibit oral symptoms, including xerostomia, dental carries, periodontitis, and delayed wound healing.[22]
We found that the mean DMFT index is high (13.18 ± 6.39). This outcome is higher than reports from the West Bank (9 ± 0.5)[23] but lower than the results of local research carried out at UNRWA PHCs in the Gaza Strip (18.6).[20] Global findings reveal that they are nearly identical to ours. Our findings revealed that dental carries and missing teeth are also high. Using the DMFS, Ribeiro et al. found significantly greater dental caries among type 2 DM patients (RR = 1.37; 95% CI = 1.09–1.71).[11] According to Simon et al., the mean number of missing teeth in the US was 10.1 ± 7.2.[16] Hintao et al. examined the number of decayed/filled root surfaces and root surface carries in patients with type 2 DM and those without the disease.[24] Individuals with diabetes had a greater and substantial prevalence (40.0% vs. 18.5%, respectively). In their study using panoramic radiography, Khan et al. revealed that patients with diabetes had a greater rate of missing teeth, while the nondiabetic group had more carious lesions and replaced teeth.[22]
There is debate over the existence of dental caries in diabetes individuals, and their relationship is complicated. Several researchers have proposed mechanisms that link DM to dental caries. These include metabolic control of diabetes, a low-carbohydrate diet, increased protein intake, increased salivary buffer capacity, delayed eruption of permanent teeth, and decreased oral tooth exposure time.[25,26] Generally, due to their obesity and consumption of meals high in calories and carbohydrates, people with type 2 DM are predicted to have more dental cavities. It has also been noted that decreased salivary flow raises the incidence of dental cavities.[26]
However, according to some research in the literature, there was no link between DM and dental cavities in studies comparing the DM and control groups.[27,28] In their study of 70 people with diabetes and 74 people without the disease, Blanco et al. examined the mean numbers of caries and found no difference between the two groups.[28] Patients without diabetes had a rate of 6.91, whereas those with diabetes had a rate of 7.9%. According to Bharateesh et al., the prevalence of dental caries was 13.6% in 300 patients with diabetes and 13.6% in 300 patients without the disease.[27]
The study found that a diet higher in protein and lower in fermentable carbs was responsible for the reduced occurrence of dental cavities in diabetic patients.[27] According to Buysschaert et al., the group of diabetes patients with well-managed blood sugar had a lower caries rate than the control group.[29]
We found males have a greater mean DFMT than females, although this difference is not statistically significant. The study carried out at UNRWA PHCs in the Gaza Strip is in line with this.[20] This, however, contradicts Bonev et al., which demonstrated a statistically significant correlation between the DMFT index and gender and that the DFMT index of females was higher than that of males.[30] According to Shaffer et al., sex differences in dental caries are seen in various populations, with females generally having more damaged teeth and a greater prevalence.[31] Factors such as variations in salivary composition and flow rate, hormonal changes, diet, genetic differences, and specific social roles within their family could play a role. Systemic diseases identified as linked to DMFT have also been found to be associated with gender differences. It has not been demonstrated that prolonged exposure to the oral cavity or a more cariogenic oral microflora contributes to a higher prevalence in women.[9,10,22,26,30] Further investigation of these domains could elucidate their role, or absence thereof, in the increased caries rate among men.
We found that DMFT was significant when education level was low, which is consistent with the findings of Alqedra and Aljeesh.[20] This is also in line with global studies.[32] People with less education may know less about food and oral hygiene habits, and they are less likely to have access to dental treatment. Furthermore, a high family size increases the risk of dental complications. The majority of participants lack access to preventive measures like dental care and a healthy diet because they are low-income and live below the poverty line. Higher family wealth and educational attainment were associated with less severe DMFT, according to Wang et al. Family income directly influences material resources, which can then influence clinical judgments and the capacity to pay for services.[33]
As people age, the severity of DMFT and periodontal disease tends to increase. Since “time” is a key component in the development of the disease, this also implies that as the duration of DM rises, the state of the teeth deteriorates, which is consistent with the usual pathogenesis of dental caries. This result is consistent with a study conducted in the Gaza Strip.[20] Age and the length of DM have indirect impacts on dental health, which are mostly influenced by human behavior in the areas of nutrition limitation, oral hygiene, diabetes management and control, and dental hygiene.
Periodontitis and type 2 DM have been linked in both directions; that is, periodontitis is linked to poor glucose control in patients with diabetes type 2, and diabetes type 2 is linked to an increase in the incidence and progression of periodontitis.[34] There was a substantial correlation between periodontitis and type 2 DM, according to cross-sectional research. Prospective research confirmed that periodontitis and type 2 DM increased each other’s occurrence and were associated with the severity of the condition. Our results are consistent with other research that found substantial differences between type 2 DM and type 1 DM in terms of probing depth and the presence of connection loss.[35] Our study found that 58% of participants had periodontitis. This is far less than the 91% prevalence rate of type 2 diabetes indicated in international reports.[36,37] On the other hand, because some studies only documented periodontitis when CAL surpassed 3 or 4 mm, they have reported a lower prevalence of periodontitis (13%) in exchange.[38] Our study found that 58% of participants had periodontitis.
Likewise, it was discovered that moderate and severe periodontitis were more common[36,37,39] unlike findings from India.[40] Discrepancy in prevalence could be caused by variations in the definition and diagnosis of periodontitis, participant ages, and sampling design techniques. To determine the true incidence of periodontitis in the diabetic population, the current study more accurately documented even the slightest attachment loss.
In general, DM significantly contributes to periodontal inflammation. It impairs the immune system’s ability to function effectively which weakens the ability to fight infections, including periodontal pathogens. It also leads to increased levels of inflammatory mediators such as interleukins (IL-1, IL-6), tumor necrosis factor-alpha (TNF-α), and prostaglandins in periodontal tissues. These proinflammatory cytokines can promote the destruction of periodontal tissues. The interaction of hyperglycemia with periodontal bacteria like Porphyromonas gingivalis enhances the release of these mediators. Moreover, it accelerates production of AGE-products which later accumulate in periodontal tissues, leading to the activation of inflammatory pathways.[41]
Gender and the prevalence of periodontitis in people with type 2 DM are significantly correlated. Our findings are in line to studies conducted in Saudi Arabia, Korea and India,[39,40] but they differ from those conducted in Nepal, and India.[42,43] Compared to women, men were 68% more likely to have periodontal disease.[44] Compared to females, males typically practice oral hygiene in a less-than-ideal manner. Compared to their female counterparts, male participants who practice poor oral hygiene are more likely to develop periodontal disease.[45] Other variables may account for the reason why men exhibit a greater incidence of periodontitis than their counterparts. To begin with, the female hormones estrogen and progesterone are thought to help protect against periodontitis. These hormones adjust the immune response, lowering inflammation and safeguarding the gums against infection. Loos and Papapanou discovered that women are less prone to developing severe periodontitis during periods of peak female hormone levels, like during pregnancy.[46] Secondly, with aging, men may encounter a buildup of risk factors for periodontitis, including the natural decline of immune function and the rise of chronic conditions such as diabetes. These age-related factors may exacerbate the impact of other risk factors such as smoking or an unhealthy diet.[47]
Among type 2 DM with low education, periodontitis was more common. Participants with no formal education had a six-fold higher risk of developing periodontal disease, according to Alahmari et al.[39] This also supports the findings of Masriadi et al., who found that low level of education was a risk factor for periodontitis.[48] People with lower levels of education are more likely to earn less income and be unable to pay for the typically costly periodontal treatments, which raises their risk of developing periodontitis. There have been reports linking socioeconomic characteristics, such as poor social class and primary education, to a higher frequency of periodontitis in adults.[49] This is in fact true given that 83.5% of the participants in our study are below the poverty level.
According to this study, smoking and the prevalence of periodontitis in people with type 2 DM are significantly correlated. According to AlAhmari et al., smokers are 2.1 times more likely than nonsmokers to develop periodontitis.[39] It could be explained by how smoking affects periodontal tissues both locally and systemically. In addition to previous studies conducted,[43,50] our is consistent with a study conducted in India.[40]
Destructive periodontitis results from uncontrolled diabetes’s most noticeable alterations, which include weakened defenses and heightened vulnerability to infections. People with diabetes have higher blood and gingival fluid glucose content than people without the disease. The elevated glucose levels in diabetic patients’ blood and gingival fluid may alter the microbiota’s habitat, causing qualitative alterations in bacteria that may exacerbate the severity of periodontitis seen in those with poorly managed diabetes. In patients with poorly controlled diabetes, the function of polymorphonuclear granulocytes and monocytes/macrophages is impaired, and as a result, the primary defense against periodontal pathogens is diminished, and bacterial proliferation is more likely to occur.[51]
Smoking is linked to an increased risk of DMFT and periodontitis. Although the exact relationship between diabetes and tooth loss is unclear, it is crucial to determine the potential contributions of additional variables, including orthodontic procedures, injuries, prosthetics, social, cultural, and economic issues.[52] According to reports, the following are the main factors that lead to the development of periodontitis in diabetic patients: (1) infections in diabetic patients as a result of immunological changes; (2) a nonenzymatic process that produces AGE products that increase the secretion of IL-1 and TNF-α; (3) the presence of gram-negative anaerobic bacteria (such as Actinobacilus actinomycetemcomitans); (4) changes in the microcirculatory blood flow in periodontal tissues; and (5) additional socioeconomic and clinical related factors actors like age, gender, income level, and not least metabolic control.[51,53]
The variability in periodontitis prevalence across studies is significantly influenced by sociodemographic factors, including age, gender, socioeconomic status, ethnicity, education level, occupation, and geographic location. Variations in these elements can result in inconsistencies in reported rates, as they affect oral health behaviors, access to care, and exposure to risk factors.
To sum up, those suffering from type 2 DM endure a variety of oral health issues, particularly dental caries, periodontitis, and loss of attachment. The findings underscore the significance of early intervention and consistent dental care for people with type 2 DM in order to avert and handle oral health complications. Oral health education campaigns that are effective should be customized to meet the particular cultural, socioeconomic, and healthcare challenges. Some potentially effective strategies include targeted awareness campaigns, screening programs for diabetes and oral health, educational workshops and seminars, behavioral change approaches, and interdisciplinary care.
There were certain limitations to this study. The cross-sectional study which limits the causal relationship between variables. The best design for this study would have been a case-control approach. In return, using the WHO assessment tool considers a strength for this study. The study employed randomization of the sample, and the study participants represented the whole geographical areas of the Gaza strip; therefore, findings could be generalized to patients with T2DM who meet the inclusion criteria.
In conclusion, Diabetes mellitus, particularly type 2, is a significant public health concern in the Gaza Strip, where it is closely linked to various oral health complications such as dental caries, periodontitis, and tooth loss. These complications not only affect patients’ oral well-being but also contribute to the worsening of glycemic control, creating a detrimental cycle of poor health outcomes.
Policy implications
Integrate oral health into diabetes management programs: oral health screening and preventive care should be a routine part of diabetes management protocols in primary health centers. Develop integrated care pathways that include both medical and dental check-ups.
Implement targeted oral Health education and awareness campaigns: launch culturally appropriate educational campaigns to raise awareness among diabetic patients about the link between diabetes and oral health. Use visual materials, workshops, and digital platforms tailored to different literacy levels.
Train primary healthcare providers on oral-systemic health connections: develop and include oral-systemic health modules in training curricula for doctors, nurses. Provide training on oral health risk assessments for diabetic patients.
Prioritize high-risk groups for intervention: develop screening and outreach programs targeting high-risk groups, such as mobile dental units or periodic dental camps in underserved areas
Strengthen health information systems to monitor oral health in diabetic patients: include oral health indicators in national health surveillance systems for diabetes. Encourage consistent data collection and reporting at the primary care level
Promote behavior change through community-based programs: implement behavior change interventions (e.g., smoking cessation programs, peer-led oral hygiene clubs) within communities and diabetes clinics.
Conflicts of interest
There are no conflicts of interest.
Acknowledgments
The authors would like to express their acknowledgment to the department of research and directorate of human resources development at the MoH for facilitating this research and data collection process.
Funding Statement
Nil.
References
- 1.World Health Organization (2016) World Health Day 2016: Beat diabetes in Palestine. Apr, 2016. [[Last accessed on 09 Mar 2021]]. Available from: http://www.emro.who.int/pse/palestine-news/world-health-day-2016-beat-diabetes-in-palestine-april-2016html .
- 2.American Diabetes Association (ADA) (2021) Introduction: Standards of Medical Care in Diabetes—2021. Diabetes Care. 2021;44((Supplement-1)):S1–2. [Google Scholar]
- 3.Elrayah-Eliadarous HA, Östenson CG, Eltom M, Johansson P, Sparring V, Wahlström R. Economic and social impact of diabetes mellitus in a low-income country: A case-control study in Sudan. J Diabetes. 2017;9:1082–90. doi: 10.1111/1753-0407.12540. [DOI] [PubMed] [Google Scholar]
- 4.Kalra S, Jena BN, Yeravdekar R. Emotional and psychological needs of people with diabetes. Indian J Endocrinol Metab. 2018;22:696–704. doi: 10.4103/ijem.IJEM_579_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Rohani B. Oral manifestations in patients with diabetes mellitus. World J Diabetes. 2019;10:485–9. doi: 10.4239/wjd.v10.i9.485. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Newman MG, Takei HH, Klokkevold PR, Carranza FA. Carranza's Clinical Periodontology. 12 th ed. St. Louis: Saunders Elsevier; 2014. [Google Scholar]
- 7.Irani FC, Wassall RR, Preshaw PM. Impact of periodontal status on oral health-related quality of life in patients with and without type 2 diabetes. J Dent. 2015;43:506–11. doi: 10.1016/j.jdent.2015.03.001. [DOI] [PubMed] [Google Scholar]
- 8.Bissong M, Azodo CC, Agbor MA, Nkuo-Akenji T, Fon PN. Oral health status of diabetes mellitus patients in Southwest Cameroon. Odontostomatol Trop. 2015;38:49–57. [PubMed] [Google Scholar]
- 9.Latti BR, Kalburge JV, Birajdar SB, Latti RG. Evaluation of relationship between dental caries, diabetes mellitus and oral microbiota in diabetics. J Oral Maxillofac Pathol. 2018;22:282–3. doi: 10.4103/jomfp.JOMFP_163_16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Jawed M, Shahid SM, Qader SA, Azhar A. Dental caries in diabetes mellitus: Role of salivary flow rate and minerals. J Diabetes Complications. 2011;25:183–6. doi: 10.1016/j.jdiacomp.2010.07.001. [DOI] [PubMed] [Google Scholar]
- 11.Ribeiro BA, Vieira Lima CP, Alves LS, Damé-Teixeira N. Impact of detection criteria on coronal and root caries estimates in adults with and without type 2 diabetes mellitus. Clin Oral Investig. 2022;26:3687–95. doi: 10.1007/s00784-021-04339-z. [DOI] [PubMed] [Google Scholar]
- 12.Kuo LC, Polson AM, Kang T. Associations between periodontal diseases and systemic diseases: A review of the inter-relationships and interactions with diabetes, respiratory diseases, cardiovascular diseases and osteoporosis. Public Health. 2008;122:417–33. doi: 10.1016/j.puhe.2007.07.004. [DOI] [PubMed] [Google Scholar]
- 13.Preshaw PM, Alba AL, Herrera D, Jepsen S, Konstantinidis A, Makrilakis K, Taylor R. Periodontitis and diabetes: A two-way relationship. Diabetologia. 2012;55:21–31. doi: 10.1007/s00125-011-2342-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Barrientos NA, Acosta ME, Espinoza GA. Periodontal health conditions in patients with diabetes in a family medicine unit. Int J Fam Commun Med. 2019;3:124. [Google Scholar]
- 15.Irwin C, Mullally B, Ziada H, Allen E, Byrne PJ. Periodontics:2. Risk factors and susceptibility in periodontitis. Dent Update. 2007;34:270–2. doi: 10.12968/denu.2007.34.5.270. 275-6. [DOI] [PubMed] [Google Scholar]
- 16.Simon LE, Karhade DS, Tobey ML. Oral health status of hospitalized patients with type 2 diabetes. Diabetes Spectr. 2020;33:58–65. doi: 10.2337/ds18-0089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Alpert PT. Oral health: The oral-systemic health connection. Home Health CareManag Pract. 2017;29:56–9. [Google Scholar]
- 18.Nascimento GG, Leite FR, Vestergaard P, Scheutz F, Lopez R. Does diabetes increase the risk of periodontitis?A systematic review and meta-regression analysis of longitudinal prospective studies. Acta Diabetol. 2018;55:653–67. doi: 10.1007/s00592-018-1120-4. [DOI] [PubMed] [Google Scholar]
- 19.Ministry of Health Unit of Health Information (2021) [[Last accessed on 18 Jun 2021]];Annual health Report. Available from:7%D9%88%D9%84.pdf. [Google Scholar]
- 20.Alqedra E, Aljeesh YI. DMFT Index of Type 2 diabetic patients attending UNRWA Health Centers in Gaza Governorates. Sci J Res Dentistry. 2020;4:007–14. [Google Scholar]
- 21.Ting GS. The Oral Health of Rest Home Residents with Dementia (Doctoral Dissertation, University of Otago) 1999 [Google Scholar]
- 22.Khan SQ, Khabeer A, Al-Thobity AM, Benrashed MA, Alyousef NI, AlMaimouni Y. Correlation between diabetes mellitus and number of restored, carious lesions and missing teeth: A retrospective radiographic evaluation. Saudi Dent J. 2021;33:131–6. doi: 10.1016/j.sdentj.2019.12.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Kateeb E, Sarhan M, Ghannam I. Oral health status among convenient sample of Palestinian adults. Int Dental J. 2015;65:55–85. [Google Scholar]
- 24.Hintao J, Teanpaisan R, Chongsuvivatwong V, Dahlen G, Rattarasarn C. Root surface and coronal caries in adults with type 2 diabetes mellitus. Community Dent Oral Epidemiol. 2007;35:302–9. doi: 10.1111/j.1600-0528.2007.00325.x. [DOI] [PubMed] [Google Scholar]
- 25.Taylor GW, Manz MC, Borgnakke WS. Diabetes, periodontal diseases, dental caries and tooth loss: A review of the literature. Compendium. 2004;25:179–88. [PubMed] [Google Scholar]
- 26.Miralles L, Silvestre FJ, Hernández-Mijares A, Bautista D, Llambes F, Grau D. Dental caries in type 1 diabetics: Influence of systemic factors of the disease upon the development of dental caries. Med Oral Patol Oral Cir Bucal. 2006;11:E256–60. [PubMed] [Google Scholar]
- 27.Bharateesh J, Ahmed M, Kokila G. Diabetes and oral health: A case control study. Int J Prev Med. 2012;3:806–9. [PMC free article] [PubMed] [Google Scholar]
- 28.Blanco JJ, Villar BB, Martinez EJ, Vallejo PS, Blanco FJ. Problemas bucodentales en pacientes con diabetes mellitus (I): Indice de placa y caries dental. Med Oral. 2003;8:97–109. [Google Scholar]
- 29.Buysschaert M, Buysschaert B, Jamart J. Dental caries and diabetes: A Belgian survey of patients with type 1 and type 2 diabetes. Diabetes Metab. 2020;46:248–9. doi: 10.1016/j.diabet.2018.06.002. [DOI] [PubMed] [Google Scholar]
- 30.Bonev B, Avramova N, Yaneva K, Filchev D. Dental caries and associated socio-demographic factors in adult people in Bulgaria. Balk J Dent Med. 2015;19:33–7. [Google Scholar]
- 31.Shaffer JR, Leslie EJ, Feingold E, Govil M, McNeil DW, Crout RJ, et al. Caries experience differs between females and males across age groups in Northern Appalachia. Int J Dent 2015. 2015:938213. doi: 10.1155/2015/938213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Mohammad T, Mirza AJ, Jahngir SM. Effects of educational attainment on dentition status in socioeconomically deprived population of Karachi. Pak Oral Dental J. 2014;34:105–8. [Google Scholar]
- 33.Wang L, Cheng L, Yuan B, Hong X, Hu T. Association between socio-economic status and dental caries in elderly people in Sichuan province, China: A cross-sectional study. BMJ Open. 2017;7:e016557. doi: 10.1136/bmjopen-2017-016557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Grossi SG, Genco RJ. Periodontal disease and diabetes mellitus: A two-way relationship. Ann Periodontol. 1998;3:51–61. doi: 10.1902/annals.1998.3.1.51. [DOI] [PubMed] [Google Scholar]
- 35.Campus G, Salem A, Uzzau S, Baldoni E, Tonolo G. Diabetes and periodontal disease: A case-control study. J Periodontol. 2005;76:418–25. doi: 10.1902/jop.2005.76.3.418. [DOI] [PubMed] [Google Scholar]
- 36.Singh M, Bains VK, Jhingran R, Srivastava R, Madan R, Maurya SC, et al. Prevalence of periodontal disease in type 2 diabetes mellitus patients: A cross-sectional study. Contemp Clin Dent. 2019;10:349–57. doi: 10.4103/ccd.ccd_652_18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Kumar A, Pandey MK, Singh A, Mittra P, Kumar P. Prevalence and severity of periodontal disease in type 2 diabetes mellitus of Bareilly region (India) Int J Med Sci Public Health. 2013;2:77–83. [Google Scholar]
- 38.Morton AA, Williams RW, Watts TL. Initial study of periodontal status in noninsulindependent diabetics in Mauritius. J Dent. 1995;23:343–5. doi: 10.1016/0300-5712(94)00001-v. [DOI] [PubMed] [Google Scholar]
- 39.Alahmari MM, AlShaiban HM, Mahmood SE. Prevalence and associated factors for periodontal disease among type I and II diabetes mellitus patients: A cross-sectional study. Healthcare (Basel) 2023;11:796. doi: 10.3390/healthcare11060796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Pathak AK, Shakya VK, Chandra A, Goel K. Association between diabetes mellitus and periodontal status in north Indian adults. Eur J Gen Dent. 2013;2:58–61. [Google Scholar]
- 41.Bolchis V, Jumanca D, Dumitrescu R, Balean O, Toderas NA, Popescu S, et al. Glycemic control, inflammatory mediators, and periodontal health: A cross-sectional study in patients with diabetes. J Clin Med. 2025;14:2847. doi: 10.3390/jcm14082847. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Shah A, Kaushik RM, Kandwal A, Kaushik R. Periodontal disease as a complication in type 2 Diabetes Mellitus. A hospital-based study in Uttarakhand, India. SRHU Med J. 2018;1:67–72. [Google Scholar]
- 43.Gupta S, Maharjan A, Dhami B, Amgain P, Katwal S, Adhikari B, et al. Status of tobacco smoking and diabetes with periodontal disease. J Nepal Med Assoc. 2018;56:818–24. doi: 10.31729/jnma.3610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Kiryowa HM, Munabi IG, Buwembo W, Rwenyonyi CM, Kaddumukasa M, Sabakaki EM. Prevalence and factors associated with periodontal disease in patients with diabetes mellitus attending Kiruddu National Referral Hospital, Uganda. Pan Afr Med J. 2022;43:202. doi: 10.11604/pamj.2022.43.202.35971. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Salim NA, Alamoush RA, Al-Abdallah MM, AlAsmar AA, Satterthwaite JD. Relationship between dental caries, oral hygiene and malocclusion among Syrian refugee children and adolescents: A cross-sectional study. BMC Oral Health. 2021;21:629. doi: 10.1186/s12903-021-01993-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Loos BG, Papapanou PN. Systemic manifestations of periodontitis: Cardiovascular disease, diabetes, and adverse pregnancy outcomes. Periodontol. 2005;39:82–95. [Google Scholar]
- 47.Baker SR, Bissada NF, Singhal S. Age-related changes in periodontal disease in men and women. J Periodontol. 2010;81:792–7. [Google Scholar]
- 48.Masriadi, Irawati E, Chotimah C, Pamewa K, Aslan S, Hamzah M. Risk factors for periodontal disease in diabetes mellitus type 2 patients at Padongko Health Center, Barru Regency (Study Epidemiology) Sys Rev Pharm. 2020;11:1501–05. [Google Scholar]
- 49.Bomfim RA, Frias AC, Pannuti CM, Zilbovicius C, Pereira AC. Socio-economic factors associated with periodontal conditions among Brazilian elderly people-Multilevel analysis of the SBSP15 study. PLoS One. 2018;13:e0206730. doi: 10.1371/journal.pone.0206730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Al-Abdaly MM, Alasmari AH, Asiri AK, Alqahtani SJ, Alzahrani AA, Alwadai JM, et al. The impact of severity of periodontal bone loss and the levels of glycated hemoglobin (HbA1c) on the periodontal clinical parameters of the 2017 world workshop among type 2 diabetic patients in Saudi Arabia. Int J Clin Med. 2021;12:570–91. [Google Scholar]
- 51.Iacopino AM. Periodontitis and diabetes interrelationships: Role of inflammation. Ann Periodontol. 2001;6:125–37. doi: 10.1902/annals.2001.6.1.125. [DOI] [PubMed] [Google Scholar]
- 52.Skamagas M, Breen TL, LeRoith D. Update on diabetes mellitus: Prevention, treatment, and association with oral diseases. Oral Dis. 2008;14:105–14. doi: 10.1111/j.1601-0825.2007.01425.x. [DOI] [PubMed] [Google Scholar]
- 53.Rodriguez-Martinez M, Patino-Marin N, Loyola-Rodriguez JP, Brito-Orta MD. Gingivitis and periodontitis as antagonistic modulators of gingival perfusion. J Periodontol. 2006;77:1643–50. doi: 10.1902/jop.2006.050311. [DOI] [PubMed] [Google Scholar]


