Abstract
During sporulation in Saccharomyces cerevisiae, vesicles transported to the vicinity of spindle pole bodies are fused to each other to generate bilayered prospore membranes (PSMs). PSMs encapsulate the haploid nuclei that arise from the meiotic divisions and serve as platforms for spore wall deposition. Membrane trafficking plays an important role in supplying vesicles for these processes. The endocytosis-deficient mutant, end3Δ, sporulated poorly and the spores produced lost resistance to ether vapor, suggesting that END3-mediated endocytosis is important for sporulation. End3p-GFP localized to cell and spore peripheries in vegetative and sporulating cells and colocalized with actin structures. Correspondingly, the actin cytoskeleton appeared aberrant during sporulation in end3Δ. Analysis of meiosis in end3Δ mutants revealed that the meiotic divisions occurred with wild-type kinetics. Furthermore, PSMs were assembled normally. However, the levels of proteins required for spore wall synthesis and components of the spore wall layers at spores were reduced, indicating that end3Δ mutants are defective in spore wall synthesis. Thus, END3-mediated endocytosis is important for spore wall formation. Additionally, cytological analyses suggest that trafficking between the plasma membrane and PSMs is important earlier during sporulation.
IN Saccharomyces cerevisiae, the initiation of the sporulation program in MATa/MATα diploid cells is triggered by deprivation of nitrogen in the presence of a nonfermentable carbon source (Kupiec et al. 1997). During sporulation, two major processes, meiosis, which reduces the ploidy of the cell, and spore formation are tightly coordinated to ensure the production of viable progeny (Kupiec et al. 1997). During the second meiotic division, novel bilayer membranous structures termed prospore membranes (PSMs) are formed by the fusion of vesicles at the cytoplasmic face of spindle pole bodies (SPBs). PSMs then grow and engulf each of the four haploid nuclei (Neiman 1998; Moreno-Borchart and Knop 2003). Once formed, PSMs serve as a platform for the deposition of the multi-layered spore wall that is laid down between the inner and outer lipid bilayer (Lynn and Magee 1970; Smits et al. 2001; Moreno-Borchart and Knop 2003). The molecular mechanisms underlying the formation and coordination of PSMs and spore walls are not well understood. However, a combination of classical genetics and genomic approaches have identified a number of molecules essential for these processes (Engebrecht et al. 1998; Rabitsch et al. 2001; Briza et al. 2002; Enyenihi and Saunders 2003; Coluccio et al. 2004)
SPO14 encodes the single yeast phosphatidylcholine (PC)-specific phospholipase D (PLD), which hydrolyzes PC, producing phosphatidic acid (PA) and choline (Rose et al. 1995; Rudge and Engebrecht 1999). Spo14p localizes to, and is essential for, PSM formation (Rudge et al. 1998, 2004). Furthermore, the enzymatic activity of Spo14p is required for PSM formation at the SPBs (Rudge et al. 1998). Another gene required for PSM formation is SSO1, a syntaxin, which functions in secretory vesicle fusion at the plasma membrane (Aalto et al. 1993). Sso1p is also essential for sporulation (Jäntti et al. 2002; Enyenihi and Saunders 2003; Öyen et al. 2004). Recently, cytological and biochemical analyses revealed that deletion of SPO14 or SSO1 results in the accumulation of vesicles in the vicinity of SPBs in sporulating cells, indicating that these proteins function specifically in vesicle fusion for PSM formation (H. Nakanishi, personal communication; M. Morishita, unpublished data).
During spore formation, vesicles are continuously transported to PSMs to bring and deposit spore wall materials (Lynn and Magee 1970; Smits et al. 2001). The spore wall consists of four discrete layers: mannan, glucan, chitosan, and dityrosine. The first two layers are similar to vegetative cell walls, while the outer layers are unique to spores and ensure their resistance to environmental stresses and harmful chemicals such as ethyl ether (Briza et al. 1994; Smits et al. 2001). The spore wall layers are laid down in a specific order in which the mannan, glucan, chitosan, and dityrosine layers overlay outward in a step-wise manner (Tachikawa et al. 2001; Coluccio et al. 2004). Many proteins and enzymes required for spore wall formation are transported to PSMs through vesicle trafficking. Dtr1p, a transporter for dityrosine (Felder et al. 2002), is transported from the Golgi to PSMs (M. Morishita, unpublished data), indicating that vesicle trafficking between the Golgi and PSMs is essential for spore formation (Neiman 1998; Moreno-Borchart and Knop 2003). Since Fks2p/Gsc2p, hereafter referred to as Gsc2p, a glucanase required for glucan synthesis (Mazur et al. 1995), and Chs3p, a chitin synthase required for the synthesis of the chitosan layer (Pammer et al. 1992), are present at the plasma membrane in vegetative cells, recycling of these proteins from the plasma membrane of the mother cell to PSMs may be necessary for spore wall formation. Consistent with this idea, Gsc2p and Chs3p mislocalize in mutants defective for the sporulation-specific SPS1 gene, which encodes a putative protein kinase (Friesen et al. 1994; Iwamoto et al. 2005). Sps1p has been proposed to regulate the movement of these enzymes to build the spore wall (Iwamoto et al. 2005). However, the pathway of this protein movement has not been elucidated.
Endocytosis, the uptake of membrane proteins and lipids, extracellular ligands, and soluble molecules from the cell surface, is important for membrane trafficking between the plasma membrane and internal compartments. Endocytosis is essential for the maintenance of plasma membrane functions such as cell growth regulation, cell polarity establishment, nutrient uptake, and ion homeostasis by control of the protein and lipid composition of the plasma membrane, regulation of signaling pathways, and uptake of nutrients and pathogens (Geli and Riezman 1998). In S. cerevisiae, a number of genes have been identified in screens for mutants defective in the internalization of pheromone receptor or the fluid-phase endocytic marker, lucifer yellow (Raths et al. 1993; Munn et al. 1995). Two of these mutants, end3 and end4, show clear defects in internalization, temperature-sensitive growth defects, and defects in organization of the actin cytoskeleton (Raths et al. 1993; Bénédetti et al. 1994). End4p/Sla2p is a phosphatidylinositide 4,5 bisphosphate binding protein and has been proposed to recruit Sla1p, which is required for assembly of the endocytic complex at the plasma membrane, and to regulate actin-dependent endocytic internalization (Wesp et al. 1997; Gourlay et al. 2003; Sun et al. 2005). END3 encodes a component of the endocytic complex, which is essential for the internalization step of endocytosis and regulation of actin organization (Tang et al. 2000). Additionally, the actin cytoskeleton is essential for endocytosis (Qualmann et al. 2000; Engqvist-Goldstein and Drubin 2003; Kaksonen et al. 2003; Merrifield 2004). Interestingly, analysis of a large number of actin mutations revealed a strong correlation between defects in endocytosis and sporulation, suggesting that endocytosis is important for sporulation (Whitacre et al. 2001). However, the precise defect in sporulation in these mutants was not examined.
To investigate the role of endocytosis in sporulation, we characterized the end3Δ mutant. Here we show that the end3Δ mutant shows defects in both endocytosis and actin organization during sporulation. The end3Δ mutant exhibits normal progression through meiosis and PSM assembly; however, the formation of the spore wall is aberrant. Thus, the END3-mediated endocytosis pathway is important in spore wall formation. Additionally, cytological analyses suggest that trafficking between the plasma membrane and PSMs is important for PSM assembly.
MATERIALS AND METHODS
Strains, media, and plasmids:
Routine growth and manipulation of S. cerevisiae strains were performed as described (Rose et al. 1990). Strains used in this study are all derived from the sporulation-proficient SK-1 strain background and are listed in Table 1. DNA-mediated transformation of yeast cells was performed using the lithium acetate procedure (Ito et al. 1983). Gene replacements were performed by the one-step method (Rothstein 1983). The end3::KanMX deletion mutant, which replaces the entire open reading frame with the marker kanMX4 (Wach et al. 1994), was made by PCR amplification of regions ∼300 bp upstream and downstream of the disrupted open reading frame from previously constructed knockout strains purchased from Research Genetics (Huntsville, AL). GFP chromosomal fusions were constructed using reagents FKS2-GFP and CHS3-GFP (Longtine et al. 1998) and the reagent END3-GFP (Sheff and Thorn 2004). All integrants were confirmed using the PCR and appropriate synthetic oligonucleotide primers. yEP-GFP-SPO14 is described in detail elsewhere (Sciorra et al. 2002). SPR28-GFP was expressed from a low-copy-number (CEN) URA3 plasmid (pSB19) (De Virgilio et al. 1996).
TABLE 1.
Strain list
| Strain | Genotype | Source |
|---|---|---|
| AN120 | MATa/MATα ura3/ura3 his3/his3 leu2/leu2 trp1/trp1 arg4/ARG4 rme::LEU2/RME | Neiman et al. (2000) |
| Y4733 | AN120, plus end3::KanMX/end3::KanMX | This laboratory |
| Y4734 | AN120, plus end3::KanMX/end3::KanMX | This laboratory |
| Y592 | MATa/MATα ho::LYS2/ho::LYS2 trp1-hisG/trp1-hisG ura3/ura3 leu2-k/leu2::hisG sps1::TRP1/sps1::TRP1 | Iwamoto et al. (2005) |
| Y4931 | AN120, plus GSC2:GFP(TRP1)/GSC2:GFP(TRP1) | This laboratory |
| Y4962 | MATa/MATα ura3/ura3 his3/his3 leu2/leu2 trp1/trp1 rme::LEU2/rme::LEU2 end3::KanMX/end3::KanMX GSC2:GFP(TRP1)/GSC2:GFP(TRP1) | This laboratory |
| Y4535 | AN120, plus CHS3:GFP(TRP1)/CHS3:GFP(TRP1) | This laboratory |
| Y4823 | Y4733, plus CHS3:GFP(TRP1)/CHS3:GFP(TRP1) | This laboratory |
| Y5095 | AN120, plus END3:GFP(HIS5)/end3::KanMX | This laboratory |
Analysis of sporulation:
Cells were grown on yeast extract peptone (YP)-glycerol medium overnight, transferred to YPAD (YPD + 10 mm adenine) medium, and incubated overnight. Cells were replica plated onto SPO (2% potassium acetate, 0.1% dextrose, 0.125% yeast extract, 0.05% amino acid dropout powder) medium and incubated at 24°, 30°, or 33° for 2 days; sporulation was monitored by differential interference contrast (DIC) microscopy. For liquid cultures, cells were grown on YPAD medium for 2 days after overnight incubation on YP-glycerol medium and inoculated into 2 ml YPAD liquid medium for 24 hr, and then 135 μl of saturated culture was inoculated into 10 ml YP-acetate medium. Cells were grown in YP-acetate medium for 16 hr, collected by centrifugation, and resuspended in an equal volume of SPM (1% potassium acetate, 0.02% raffinose, 0.1% amino acid drop-out powder) to induce sporulation. For analysis of GFP fusion proteins, live cells were examined by fluorescein-5-isothiocyanate on a Zeiss Axioskop 2 fluorescence microscope at various times after induction of sporulation. To monitor progression in sporulation and to ensure that cells were at the appropriate stage, parallel cultures were fixed and stained with 4′,6-diamidino-2-phenylindole (DAPI).
Ether sensitivity assay:
Cells were sporulated on SPO medium at 30° for 2 days, replica plated to YPAD medium, immediately exposed to 0.6 ml ether vapor for 15 min, and then exposed to an additional 2 ml of ether for 15 min. The plates were incubated at 30° overnight.
Vacuolar marker staining:
Cells were induced to sporulate in liquid as described above. FM4-64, N-(3-triethylammoniumpropyl)-4-(p-diethylaminophenylhexatrienyl) pyridinium dibromide (Molecular Probes, Eugene, OR) (Vida and Emr 1995) was added at a final concentration of 2 μg/ml (1 mg/ml stock solution in DMSO) and incubated on ice for 5 or 10 min. The cells were collected by centrifugation, inoculated into fresh SPM, and incubated with shaking at 30°. At various times, 15 mm NaN3 was added and cells were observed by fluorescence microscopy.
Actin staining with rhodamin-phalloidin:
Sporulating or mitotically dividing cells were fixed in PM buffer (1 m potassium phosphate pH 6.8, 1 m MgSO4) with 3% formaldehyde at 30° for 1 hr. After washing with PM buffer three times, fixed cells were resuspended in 1 ml PM containing 1% Triton X-100 and incubated at room temperature for 3 min for cell permeabilization. Permeabilized cells were washed with PM buffer, resuspended in 20 μl rhodamine-phalloidin (Molecular Probes)/PM buffer, and incubated with shaking at 30° for 1 hr. Cells were mounted on a glass slide and 8 μl of a 300-μm DAPI solution was added to visualize DNA.
In vivo BODIPY-PC analysis:
Cells were sporulated in liquid as described above and 2-decanoyl-1{O-[11-(4,4-difluoro-5,7-dimethyl-4-bora-3a,4a-diaza-s-indacene-3-propionyl)amino]undecyl}-sn-glycero-3-phosphocholine (BODIPY-PC) (4 μm final concentration) was added directly to the culture after a 4-hr incubation at 30°. Cells were harvested at 1.5 hr after the addition of BODIPY-PC. Lipids were extracted and analyzed by thin-layer chromatography as described (Rudge et al. 2001). All assays were performed in triplicate. The percentage of conversion of BODIPY-PC to -PA was determined from the pixel intensities obtained from the image using AlphaEase FC4.0 imager soft (Alpha Innotech).
Calcofluor White staining:
Cells were sporulated in liquid as described above. One milliliter of culture was harvested, washed once with water, and resuspended in 1 mg/ml Calcofluor White (CW) solution. Cells were incubated at room temperature for ∼5 min, washed three times with water, and resuspended in water for fluorescence microscopy using DAPI optics.
RESULTS
The end3Δ mutant is defective in sporulation:
END3 encodes a component of the endocytic machinery required for the internalization step of endocytosis (Bénédetti et al. 1994). To investigate the role of endocytosis in sporulation, we constructed an end3 deletion (end3::KanMX) in the SK-1 strain background, which sporulates rapidly and efficiently (Fast 1973). Contrary to a previous report of temperature-sensitive growth of the end3 deletion mutant, in which the N-terminal three-fourths was deleted (Bénédetti et al. 1994), the end3Δ SK-1 strain grew similarly to wild type at all temperatures examined (data not shown). The end3Δ mutation, however, conferred a sporulation-deficient phenotype (Figure 1; Table 2). DIC microscopy revealed that end3Δ sporulated poorly and that the spores produced appeared aberrant (Figure 1A). A large percentage of the sporulating end3Δ cells contained one-, two-, and three-spored asci, which were rarely seen in wild type, although a small population in end3Δ contained complete tetrads at 30° (3.5% end3Δ vs. 65.6% END3) and at 33° (3.3% end3Δ vs. 70.6% END3). This phenotype was less severe at 24° (25.8% end3Δ vs. 68.7% END3) (Table 2). We used 30° for all subsequent experiments.
Figure 1.—
end3Δ is defective in spore formation. (A) Sporulation in wild-type (AN120) and end3Δ (Y4733) cells. DIC microscopy of cultures induced in sporulation showed end3Δ-produced aberrant spores. (B) Ether vapor sensitivity test. Wild-type (AN120), end3Δ (Y4733 and Y4734), and sps1Δ (Y592) cells were incubated on sporulation medium for 2 days, replica plated onto rich medium, and then immediately exposed to ether vapor. The photo of the plate was taken after overnight incubation at 30°.
TABLE 2.
Sporulation efficiency
| No. of spores/cell
|
|||||||
|---|---|---|---|---|---|---|---|
| Strain | Relevant genotype |
Temperature | 4 | 3a | 2 | 1 | 0 |
| AN120 | END3/END3 | 24° | 68.7 (9.3) | 20.3 (4.2) | 5.4 (2.7) | 0.3 (0.2) | 5.2 (3.2) |
| 30o | 65.6 (2.7) | 19.2 (3.9) | 1.2 (0.5) | 0.2 (0.2) | 13.7 (4.9) | ||
| 33° | 70.6 (3.7) | 26.6 (3.8) | 0.0 (0.0) | 0.2 (0.4) | 2.7 (0.6) | ||
| Y4733 | end3Δ/end3Δ | 24° | 25.8 (2.8) | 28.1 (2.4) | 16.8 (2.9) | 9.8 (3.3) | 19.6 (8.3) |
| 30° | 3.5 (0.5) | 11.8 (3.5) | 11.5 (4.1) | 17.3 (3.1) | 55.9 (10.0) | ||
| 33° | 3.3 (1.2) | 8.0 (1.8) | 14.3 (2.3) | 26.0 (3.5) | 48.3 (6.1) | ||
Frequencies of spore formation are shown as percentages.
Values may be overrepresented since tetrads and three-spored asci are indistinguishable in wild-type cells, while three-spored asci in end3Δ mutant cells are clearly distinguished from tetrads. Standard deviation for each value is shown in parentheses. n = 300.
The outermost layer of the spore wall consists of dityrosine, which fluoresces when exposed to ultraviolet (UV) light. end3Δ spores emitted only very weak fluorescence under UV light (data not shown), suggesting that dityrosine molecules are not efficiently incorporated into end3Δ spores. Consistent with this result, end3Δ spores were sensitive to ether vapor, similar to the sps1Δ mutant, which is defective in the deposition of spore wall components (Friesen et al. 1994; Iwamoto et al. 2005), at a concentration that had no effect on wild-type spores (Figure 1B). Taken together, these results indicate that the end3Δ mutant fails to produce mature spores.
Endocytosis is impaired in end3Δ during sporulation:
To examine endocytosis, we monitored uptake of the vacuole membrane lipophilic dye, FM4-64, in wild type and in the end3Δ mutant during vegetative growth and sporulation (Figure 2, Tables 3 and 4 ). Wild-type and end3Δ mutant cells were incubated in rich or sporulation medium for 5 hr and labeled with FM4-64 (as described in materials and methods). During vegetative growth, wild-type cells rapidly internalized FM4-64 and displayed strong vacuolar staining by 20 min after removal of dye (Figure 2A; Table 3). In end3Δ mutants, the majority of the cells displayed very weak or no signal; strong vacuolar staining was observed in only a small number of cells after 60 min (Figure 2A; Table 3), indicating that end3Δ is defective in uptake of this dye during vegetative growth.
Figure 2.—
end3Δ is defective in the uptake of FM4-64. (A) Vegetative and (B) sporulating wild-type (AN120) and end3Δ mutant (Y4733) cells were stained with the lipophilic styryl marker FM4-64, as described in materials and methods. Fluorescence (bottom) and DIC (top) images of the same cells are shown. In wild-type cells, FM4-64 was observed as punctate signals in the cytoplasm and eventually at vacuoles by 60 min after removal of the dye during vegetative growth. In addition to vacuole staining of the mother cells, FM4-64 was detected at spores during sporulation. In end3Δ, only weak or no signals for FM4-64 were detected at vacuoles during vegetative growth, and plasma membrane staining persisted for 60 min after removal of dye during sporulation.
TABLE 3.
Quantification of FM4-64 uptake: vegetative cells
| Vacuoles
|
||||
|---|---|---|---|---|
| Time (min) after removal of FM4-64 |
Punctate | Strong | Weak | Faint cytoplasm or no signal |
| END3: % of cells labeled with FM4-64 | ||||
| 0 | 98.7 | 0.0 | 0.0 | 1.3 |
| 10 | 85.3 | 0.7 | 6.7 | 7.3 |
| 20 | 22.1 | 60.3 | 11.4 | 6.2 |
| 30 | 10.0 | 65.3 | 23.0 | 1.7 |
| 60 | 0.0 | 62.3 | 36.0 | 1.7 |
| end3Δ: % of cells labeled with FM4-64 | ||||
| 0 | 0.0 | 0.0 | 1.0 | 99.0 |
| 10 | 17.7 | 0.7 | 3.7 | 77.9 |
| 20 | 2.3 | 2.0 | 25.0 | 70.7 |
| 30 | 0.7 | 6.0 | 53.3 | 40.0 |
| 60 | 0.7 | 35.3 | 56.7 | 7.3 |
Five hundred cells were counted for vegetative wild-type and end3Δ strains.
TABLE 4.
Quantification of FM4-64 uptake: sporulating cells
| Time (min) after removal of FM4-64 |
Plasma membrane |
Punctate | Vacuoles | Prospore membranes |
Faint cytoplasm or no signal |
|---|---|---|---|---|---|
| END3: % of cells labeled with FM4-64 | |||||
| 0 | 30.6 | 46.6 | 21.4 | 0.6 | 0.8 |
| 10 | 17.4 | 51.0 | 26.2 | 3.6 | 1.8 |
| 20 | 6.6 | 63.2 | 24.2 | 4.8 | 1.2 |
| 30 | 7.2 | 31.9 | 45.3 | 12.2 | 3.4 |
| 60 | 10.0 | 2.2 | 66.6a | 18.6 | 2.6 |
| end3Δ: % of cells labeled with FM4-64 | |||||
| 0 | 90.8 | 1.2 | 0.0 | 3.4 | 4.6 |
| 10 | 66.8 | 16.8 | 0.2 | 2.8 | 13.4 |
| 20 | 58.8 | 25.1 | 9.8 | 2.2 | 4.1 |
| 30 | 29.9 | 31.3 | 21.0 | 11.4 | 6.4 |
| 60 | 16.4 | 5.8 | 56.0a | 14.8 | 7.0 |
Five hundred cells were counted for sporulating wild-type and end3Δ strains.
After 60 min 85% of wild-type and 51% of end3Δ cells displayed strong vacuolar staining.
In wild-type sporulating cells, FM4-64 was observed at the plasma membrane, punctate structures, and vacuoles (Figure 2B; Table 4). Greater than 80% of the cells no longer showed plasma membrane staining 10 min after removal from the dye and vacuolar staining increased steadily throughout the time course. In addition, some PSMs were stained by FM4-64. In contrast, end3Δ mutants had persistent plasma membrane staining; the majority of cells lost plasma membrane staining only after 60 min after removal from dye. In addition, significant vacuolar staining was observed only after 60 min, although not to the extent of wild type (Figure 2B; Table 4). Similar to wild type, some cells contained PSMs stained with FM4-64. Thus, endocytosis in the end3Δ mutant is also impaired during sporulation.
end3Δ exhibits defects in the actin cytoskeleton during sporulation:
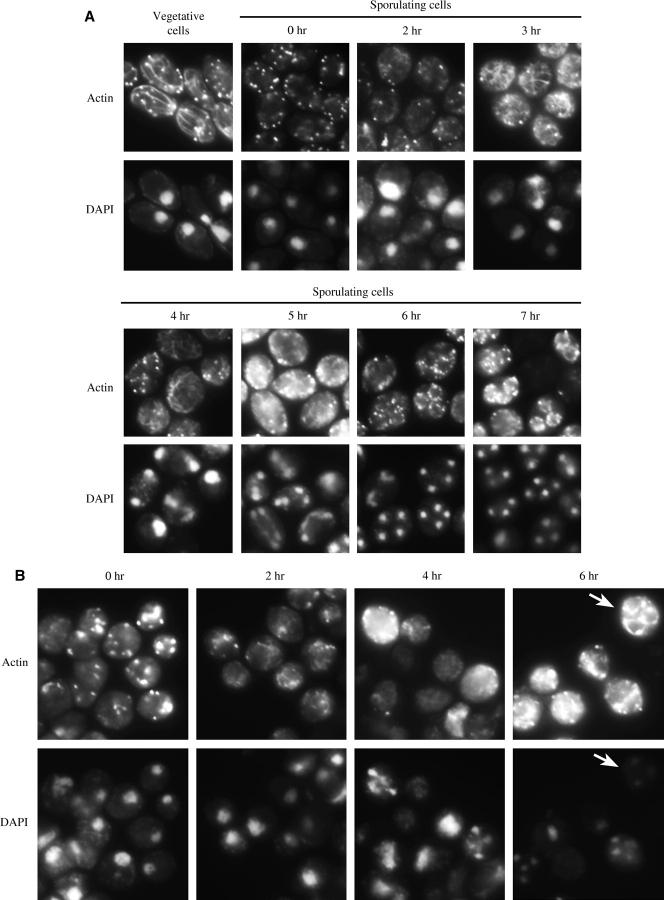
Endocytosis is tightly coordinated with the organization and movement of actin (Engqvist-Goldstein and Drubin 2003). Endocytosis-deficient mutants show defects in the actin cytoskeleton and, vice versa, mutants defective in actin or actin-related proteins have defects in endocytosis (Kübler and Riezman 1993; Raths et al. 1993; Munn et al. 1995). It has been previously demonstrated that actin mutants are defective in sporulation (Whitacre et al. 2001) and that actin patches are seen in spores (Doyle and Botstein 1996). To examine the actin cytoskeleton during sporulation, fixed cells were labeled with rhodamine-conjugated phalloidin to visualize F-actin at various times throughout sporulation (Figure 3, A and B). In wild type, actin was observed as an extensive array of patches and cables in mitotically dividing cells (Figure 3A, vegetative cells). Cells arrested in G1 for entry into meiosis almost exclusively exhibited actin patches (Figure 3A, 0 hr). Interestingly, in addition to patches, network-like actin filaments appeared in cells undergoing the first and second meiotic divisions (Figure 3A, 2–5 hr). The actin filaments were maintained until meiosis was completed and then actin patches were observed around and/or inside the individual spores (Figure 3A, 6 and 7 hr). In end3Δ cells, large actin clumps were observed in the cytoplasm of sporulating cells (Figure 3B, 0, 2, and 4 hr) and huge actin aggregates were seen around spores (Figure 3B, 6 hr; marked by arrow), which were not seen in wild-type cells. Additionally, actin filaments were observed in some end3Δ cells (Figure 3B, 2 and 4 hr), although not to the extent of wild type. Thus, the actin cytoskeleton is aberrant in the end3Δ mutant.
Figure 3.—
Actin organization in wild type and end3Δ during sporulation. (A) Wild-type (AN120) and (B) end3Δ (Y4733) cells cultured in sporulation medium were harvested at indicated times, fixed, and then stained with rhodamine-phalloidin and DAPI as described in materials and methods. (A) In wild type, vegetative cells showed actin patches and cables throughout the cells (vegetative cells), while arresting cells contained primarily actin patches (0 hr). During meiosis, network-like actin filaments appeared (2–4 hr) and as sporulation proceeded actin patches where observed around the maturing spore peripheries (6 and 7 hr). (B) end3Δ cells showed accumulation of large actin clumps in the cytoplasm and around the spore peripheries. Arrows show cell that completed meiosis and displayed actin clumps around spores.
Meiosis and PSM assembly are normal in the end3Δ mutant:
To determine at what stage of sporulation End3p-mediated endocytosis functions, we examined several molecular and cytological markers of sporulation in the end3Δ mutant. The expression of sporulation-specific genes (i.e., HOP1, Hollingsworth and Byers 1989; NDT80, Xu et al. 1995; and DIT1, Briza et al. 1994) was not affected in the end3Δ mutant (data not shown). Analysis of progression through the meiotic divisions was analyzed by DAPI staining in a time course of sporulation (Figure 4). The meiotic divisions occurred with normal kinetics; however, the efficiency of the second meiotic division was slightly reduced in end3Δ mutants (71.8% ± 4.2 end3Δ vs. 91.8% ± 0.6 END3; Figure 4). Thus, end3Δ mutants induce the sporulation program and progress through meiosis similarly to wild type.
Figure 4.—

Meiotic kinetics in end3Δ is similar to that in wild type. At indicated times after induction of sporulation, cells were stained with DAPI. Mono-, bi-, and tetranucleate cells were counted (n = 300). end3Δ underwent meiosis with a kinetics similar to that of wild type, although the efficiency of completion of the second meiotic division was slightly lower in end3Δ.
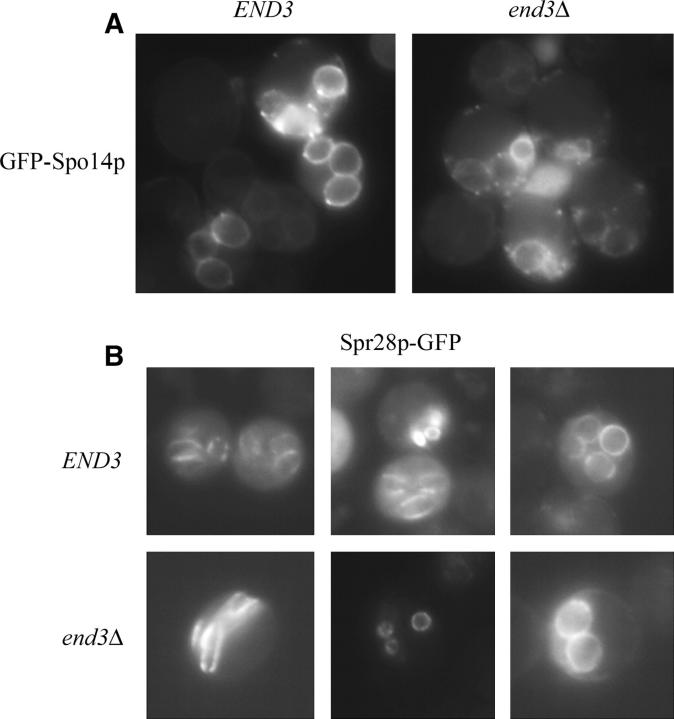
We next examined whether PSM assembly, an early event in spore formation, is perturbed in the end3Δ mutant. To that end, we examined GFP-Spo14p, the major yeast PLD, which is essential for and marks PSMs (Rudge et al. 1998, 2004) in live cells induced to sporulate. The end3Δ mutant showed clear GFP-Spo14p signals at PSMs analogous to the pattern observed in wild-type cells (Figure 5A). To determine whether inactivation of End3p had any effect on Spo14p function, we monitored Spo14p PLD activity by measuring conversion of internalized BODIPY-PC to BODIPY-PA. As previously reported (Rudge et al. 2001), hydrolysis of BODIPY-PC occurs almost exclusively by the action of Spo14p, as spo14Δ strains did not generate any appreciable BODIPY-PA (Table 5). BODIPY-PC can be internalized by endocytosis and flip-flop action of lipids at the plasma membrane (Rudge et al. 2001). BODIPY-PC was efficiently internalized in end3Δ mutants under the conditions used. Furthermore, the conversion rate of BODIPY-PA/BODIPY-PC revealed that end3Δ retained the equivalent efficiency of Spo14p PLD activity (Table 5). Therefore, we conclude that PSM assembly and PA production by Spo14p PLD activity are normal in the end3Δ mutant.
Figure 5.—
PSM formation and closure are normal in end3Δ. Localization of (A) GFP-Spo14 and (B) Spr28p-GFP in sporulating wild type (AN120) and end3Δ (Y4733). Both wild-type and end3Δ cells showed a PSM staining pattern of GFP fluorescence for GFP-Spo14p and an analogous pattern of Spr28-GFP signals, bar structures, and PSMs.
TABLE 5.
PLD-catalyzed hydrolysis of internalized BODIPY-PC
| END3 | spo14Δ | end3Δ | |
|---|---|---|---|
| % BODIPY-PAa | 0.82 ± 0.11 | 0.02 ± 0.02 | 0.85 ± 0.20 |
The percentage conversion of intracellular BODIPY-PC to BODIPY-PA was determined as described in materials and methods. The mean values ±SD from three independent experiments are shown.
After PSM closure, spore wall components are deposited in the lumen between the double membranes (Lynn and Magee 1970). Septins are organized at the leading edge of the PSM and are believed to facilitate spore wall deposition (Fares et al. 1996). Septin localization is altered in mutants where PSM closure is not sensed or has failed (Tachikawa et al. 2001). To determine if PSM closure occurs in end3Δ, Spr28p-GFP, a sporulation-specific septin (De Virgilio et al. 1996), was examined in cells induced in sporulation. Septins form parallel bar structures and then eventually surround the spores. Analogous localization patterns were observed in both wild-type and end3Δ cells (Figure 5B), suggesting that septins organize properly in the absence of End3p. Thus, PSMs are assembled and undergo closure in end3Δ mutants.
end3Δ is defective in spore wall synthesis:
Next we examined spore walls in the end3Δ mutant. The spore wall contains four distinct layers: mannan, glucan, chitosan, and dityrosine from the inner to the outer layer, which are formed outwardly step-by-step. Proteins required for the formation of the spore wall layers are deposited at PSMs; Gsc2p and Chs3p are responsible for synthesis of the glucan and chitosan layer, respectively, and both localize to PSMs (Iwamoto et al. 2005). We constructed GSC2-GFP or CHS3-GFP in wild type and the end3Δ mutant and observed their localization during sporulation (Figure 6). Weak but detectable Gsc2p-GFP staining was observed at spores and in the cytoplasm in end3Δ cells (Figure 6A). However, unlike wild type, Chs3p-GFP was detected primarily in the cytoplasm, although a limited number of cells showed faint fluorescence at spores (Figure 6B). These results indicate that both Gsc2p and Chs3p are not efficiently localized in end3Δ mutants.
Figure 6.—

Spore wall maturation in end3Δ. Localization of (A) Gsc2p-GFP and (B) Chs3p-GFP in end3Δ cells. Gsc2p-GFP and Chs3p-GFP localize to PSMs in wild-type cells (top). end3Δ cells displayed Gsc2p-GFP and Chs3p-GFP fluorescence in the cytoplasm and only very weak fluorescence at PSMs. Strains used here were GSC2:GFP END3, Y4931; GSC2:GFP end3Δ, Y4962; CHS3:GFP END3, Y4535; and CHS3:GFP end3Δ, Y4823. (C) CW staining in end3Δ cells. Wild-type AN120) and end3Δ (Y4733) cells were incubated at 30° for 24 hr and stained with CW, which binds to chitin at bud scars and the chitosan layer of spores. In wild type (left), the majority of the spores were mature and not stained, although some immature spores showed bright CW fluorescence. While in end3Δ (right), many spores were stained with a lower intensity of CW fluorescence compared with that in wild type, suggesting that end3Δ has defects in the formation of the outer layers of spores, chitosan, and dityrosine.
We also stained sporulating cells with CW, which binds to chitin at bud scars and chitosan in the spore wall. CW, however, cannot access the chitosan layer once the outermost dityrosine layer is formed. In wild type, the majority of the cells contained mature spores and thus were not stained by CW, although some immature spores were present as observed by intense CW staining (Figure 6C). Consistent with the aberrant Chs3p-GFP localization, the intensity of CW staining in end3Δ spores was much lower than that in the immature wild-type spores (Figure 6C). Furthermore, a much larger proportion of sporulating end3Δ cells stained with CW (Figure 6C), suggesting that dityrosine is not incorporated into the end3Δ spores. This is consistent with the weak dityrosine fluorescence observed under UV light. Taken together, these results indicate that the end3Δ mutation is impaired in spore wall synthesis.
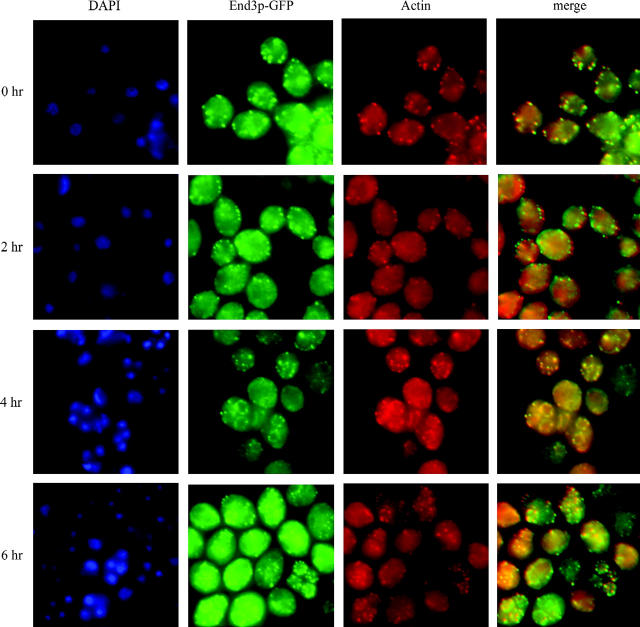
End3p-GFP localizes to actin patches:
In the endocytic complex, End3p interacts with Pan1p, which has been reported to associate with actin patches (Tang et al. 2000). To examine whether End3p also associates with actin patches and how End3p behaves during sporulation, the END3 gene was fused with GFP and End3p-GFP fluorescence was monitored in sporulating cells. The End3p-GFP fusion was functional as assessed by sporulation efficiency (data not shown). End3p-GFP localized to the peripheries of vegetative cells (data not shown) and in sporulating cells (Figure 7). End3p-GFP patches showed complete colocalization with actin stained with rhodamine-conjugated phallodin at the surface and/or inside of the spores in addition to the periphery of the mother cell (Figure 7). Thus, End3p associates with the actin cytoskeleton at cell peripheries in both vegetative and sporulating cells.
Figure 7.—
End3p-GFP associates with the actin cytoskeleton during sporulation. END3-GFP cells (Y5095) incubated in sporulation medium were harvested at indicated times, fixed, and then stained with rhodamine-phalloidin and DAPI. Photos show DAPI, End3p-GFP rhodamine-phalloidin, and merged images of End3p-GFP and rhodamine-phalloidin (merge). End3p-GFP was detected as bright patches at the cell periphery (0 and 2 hr) and around spore walls (4 and 6 hr). End3p-GFP colocalized to actin patches at all the stages of sporulation (merge).
FM4-64 is transported to PSMs during sporulation:
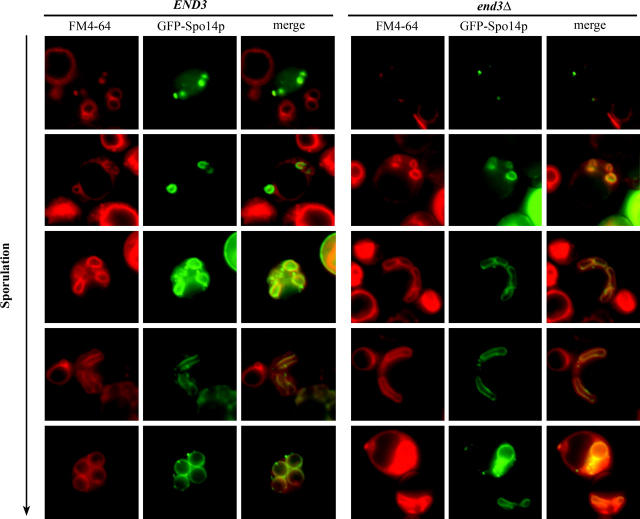
The end3Δ mutant is defective in sporulation, resulting from defects in the formation of the spore wall (Figure 6). However, FM4-64 and BODIPY-PC are internalized in end3Δ mutants, although not as efficiently as in wild type. Thus, it appears that these molecules can be internalized independently of End3p. To examine if this End3p-independent pathway of internalization is important at earlier stages of sporulation, we conducted FM4-64 staining in sporulating cells expressing the PSM marker GFP-Spo14p and examined the cells after 1 hr of incubation, at a time when there is significant FM4-64 staining in both wild-type and end3Δ cells (Figure 2B; Table 4). Figure 8 shows different stages of PSM formation, such as initiation, development, engulfment, and shaping of PSMs in cells labeled with FM4-64 and GFP-Spo14p. The FM4-64 signal was detected at vacuoles and at PSMs at all stages of PSM formation (Figure 8, END3). FM4-64 also labeled PSMs in the end3Δ mutant (Figure 8, end3Δ). Therefore, some FM4-64 molecules at the plasma membrane are internalized and delivered to PSMs during sporulation even in end3Δ mutant cells. Thus, it is likely that multiple pathways allow for communication between the plasma membrane and PSMs and that trafficking between these compartments is important throughout sporulation.
Figure 8.—
FM4-64 is internalized and transported to PSMs in wild type and end3Δ during sporulation. Wild-type (AN120) and end3Δ (Y4733) cells expressing GFP-Spo14p incubated for 5 hr after the induction of sporulation were stained with FM4-64. Photos of cells at different stages of sporulation were taken. FM4-64 was internalized and transported to vacuoles, and PSMs, marked by GFP-Spo14p, in end3Δ at all stages of sporulation were examined, indicating that there may be alternative pathway(s) independent of End3p-mediated endocytosis to transport FM4-64 during sporulation.
DISCUSSION
Spore morphogenesis requires the de novo synthesis of a complicated, stratified structure within the mother cell. Previous work has shown that vesicles derived from the Golgi carry lipids and proteins important for spore formation. However, the contributions of other membrane compartments, such as the plasma membrane, vacuoles, and endoplasmic reticulum, have not been investigated. The plasma membrane syntaxin Sso1p is required for sporulation (Jäntti et al. 2002; Öyen et al. 2004), in particular, in the process of vesicle fusion at SPBs (H. Nakanishi, personal communication). Thus, the endocytic pathway may play a role in recycling plasma membrane proteins and lipids to PSMs for spore morphogenesis. Furthermore, the fact that the spore wall contains mannoproteins and β-1,3-glucan, which are also components of the vegetative cell wall, suggests that there is communication between these different organelles.
To determine whether trafficking between the plasma membrane and the PSM is important for sporulation, we constructed and characterized mutants harboring a deletion of the endocytic complex gene, END3, during sporulation. This was possible because, unlike the previously reported deletion mutant (Bénédetti et al. 1994), the end3Δ mutant in SK-1 exhibited no growth defect; presumably this discrepancy is due to differences in strain background. The end3Δ mutant showed significant defects in internalization of the dye FM4-64, during both vegetative growth and sporulation; however, measurable uptake did occur. This is consistent with previous studies in which FM4-64 is transported to the vacuole even in mutants with strong endocytic defects (Vida and Emr 1995). Thus, it is likely that there are alternative pathways independent of End3p-mediated endocytosis for uptake of FM4-64 and other molecules. The end3Δ mutant did produce aberrant spores consistent with either a partial block in endocytosis or the existence of additional pathways. The aberrant end3Δ spores appear immature, containing little or no dityrosine, indicating that the End3p-mediated endocytosis pathway plays an important role in the process of spore formation.
The end3Δ mutants underwent meiosis with wild-type kinetics, suggesting that endocytosis is not required for the early stages of sporulation. However, there was a small reduction in the number of cells that completed the second meiotic division. This may be due to the inability to observe discrete DAPI-staining bodies in a mutant that is aberrant in producing mature spores. Alternatively, endocytosis may play a subtle role during meiosis.
Spo14p PLD activity is essential for PSM formation (Rudge et al. 1998). Fluorescence microscopy analysis revealed that GFP-Spo14p localized properly to PSMs in end3Δ cells and an in vivo biochemical assay showed that end3Δ retained wild-type Spo14p PLD enzymatic activity. These results indicate that deletion of END3 does not affect the localization and function of Spo14p at PSMs. Consistent with this result, end3Δ shows identical kinetics of PSM formation, including vesicle fusion and growth of PSMs, to those of wild type. Furthermore, spetins are properly organized in the end3Δ mutant, indicating that closure occurs. Thus, End3p-mediated endocytosis is not essential for PSM assembly. However, the finding that FM4-64 is internalized and present at PSMs even in the absence of End3p, suggests that trafficking between the plasma membrane and PSMs is important for PSM formation. Whether FM4-64 is transported directly to PSMs or via a plasma membrane-vacuole-PSM route is not clear from these experiments and remains an interesting question for future study.
Once PSM closure occurs, proteins required for spore wall synthesis, including Gsc2p and Chs3p, are deposited at spores. Neither Gsc2p-GFP nor Chs3p localized normally to spores in end3Δ mutants. In vegetative end3 cells, chitin on the cell surface is delocalized, suggesting that Chs3p fails to undergo endocytosis and thus is not properly targeted to the bud neck (Bénédetti et al. 1994). Similarly, during sporulation Chs3p and Gsc2p are endocytosed from the plasma membrane of the mother cell and redirected to PSMs (Iwamoto et al. 2005); in the absence of END3, plasma membrane Chs3p and Gcs2p are inefficiently internalized and redirected to PSMs. Since Dtr1p-GFP is delivered to the PSMs from the Golgi (M. Morishita, unpublished data) and late-acting SEC genes are essential for PSM formation (Neiman 1998), Golgi-to-PSM trafficking clearly plays a critical role in the early stages of spore formation.
End3p is a component of the endocytic complex essential for the internalization step of endocytosis, which includes, Pan1p, Ent1/2p, and Sla1p (Tang et al. 2000). During endocytosis, internalization, vesicle formation, vesicle scission, and vesicle transport require a functional actin cytoskeleton. Many mutants defective in endocytosis have mutations in actin or actin-related genes (i.e., ACT1, Munn et al. 1995; SLA2, Wesp et al. 1997; SAC6, Kübler and Riezman 1993; ARP2/3, Moreau et al. 1997; PRK1/ARK1, Smythe and Ayscough 2003; and MYO5, Jonsdottir and Li 2004). Interestingly, several alleles of the essential ACT1 gene have been reported to confer a sporulation-deficient phenotype (Whitacre et al. 2001); the severity of the sporulation defect correlates well with the severity of the endocytic defects observed, suggesting that endocytosis is important for sporulation. In addition, actin patches have previously been observed in spores (Doyle and Botstein 1996). Taken together with this study, it appears that the actin cytoskeleton plays an important role during yeast sporulation in large part due to the role it plays in endocytosis. Detailed analysis of actin reveals that the actin cytoskeleton undergoes changes throughout sporulation, forming patches and network-like filaments. It is noteworthy that this is the first observation of filamentous actin in sporulating cells. It is possible that this filamentous actin is important for the cell cycle changes that occur during meiosis. At late stages of sporulation, actin patches are observed around the peripheries of each of the four spores, raising the possibility that endocytosis plays an important role even after the spore is constructed (Kono et al. 2005). Consistent with what has been observed in vegetative cells, end3Δ mutant cells exhibit defects in actin organization during sporulation (Figure 3B). Furthermore, End3p localizes to actin patches (Figure 7), indicating that, similar to vegetative cells, endocytosis and actin organization are tightly coordinated for the process of sporulation.
In conclusion, our results provide direct evidence that endocytosis is important for sporulation and contributes to vesicle trafficking of plasma membrane proteins required for spore formation. Future work will elucidate the specific route that internalized proteins and lipids follow and determine the contribution of End3p-independent pathways for trafficking from the plasma membrane of the mother cell to the new plasma membrane of the developing spore.
Acknowledgments
We thank Aaron M. Neiman for the generous gift of the yeast strain and Jaime Connolly, Aimee Jaramillo-Lambert, and Stephen Fairclough for useful advice and helpful discussion. This work was supported by National Institutes of Health research grant GM66124 to J.E.
References
- Aalto, M. K., L. Ruohonen, K. Hosono and S. Keränen, 1993. Yeast syntaxins Sso1p and Sso2p belong to a family of related membrane proteins that function in vesicular transport. EMBO J. 12: 4095–4104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bénédetti, H., S. Raths, F. Crausaz and H. Riezman, 1994. The END3 gene encodes a protein that is required for the internalization step of endocytosis and for actin cytoskeleton organization in yeast. Mol. Biol. Cell 5: 1023–1037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briza, P., M. Eckerstorfer and M. Breitenbach, 1994. The sporulation-specific enzymes encoded by the DIT1 and DIT2 genes catalyze a two-step reaction leading to a soluble LL-dityrosine-containing precursor of the yeast spore wall. Proc. Natl. Acad. Sci. USA 91: 4524–4528. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Briza, P., E. Bogengruber, A. Thür, M. Rützler, M. Münsterkötter et al., 2002. Systematic analysis of sporulation phenotypes in 624 non-lethal homozygous deletion strains of Saccharomyces cerevisiae. Yeast 19: 403–422. [DOI] [PubMed] [Google Scholar]
- Coluccio, A., E. Bogengruber, M. N. Conrad, M. E. Dresser, P. Briza et al., 2004. Morphogenetic pathway of spore wall assembly in Saccharomyces cerevisiae. Eukaryot. Cell 3: 1464–1475. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Virgilio, C., D. J. DeMarini and J. R. Pringle, 1996. SPR28, a sixth member of the septin gene family in Saccharomyces cerevisiae that is expressed specifically in sporulating cells. Microbiology 142: 2897–2905. [DOI] [PubMed] [Google Scholar]
- Doyle, T., and D. Botstein, 1996. Movement of yeast cortical actin cytoskeleton visualized in vivo. Proc. Natl. Acad. Sci. USA 93: 3886–3891. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engebrecht, J., S. Masse, L. Davis, K. Rose and T. Kessel, 1998. Yeast meiotic mutants proficient for the induction of ectopic recombination. Genetics 148: 581–598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Engqvist-Goldstein, Å. E. Y., and D. G. Drubin, 2003. Actin assembly and endocytosis: from yeast to mammals. Annu. Rev. Cell Dev. Biol. 19: 287–332. [DOI] [PubMed] [Google Scholar]
- Enyenihi, A. H., and W. S. Saunders, 2003. Large-scale functional genomic analysis of sporulation and meiosis in Saccharomyces cerevisiae. Genetics 163: 47–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fares, H., L. Goetsch and J. R. Pringle, 1996. Identification of a developmentally regulated septin and involvement of the septins in spore formation in Saccharomyces cerevisiae. J. Cell Biol. 132: 399–411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fast, D., 1973. Sporulation synchrony of Saccharomyces cerevisiae grown in various carbon sources. J. Bacteriol. 116: 925–930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Felder, T., E. Bogengruber, S. Tenreiro, A. Ellinger, I. Sá-Correia et al., 2002. Dtr1p, a multidrug resistance transporter of the major facilitator superfamily, plays an essential role in spore wall maturation in Saccharomyces cerevisiae. Eukaryot. Cell 1: 799–810. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Friesen, H., R. Lunz, S. Doyle and J. Segall, 1994. Mutation of the SPS1-encoded protein kinase of Saccharomyces cerevisiae leads to defects in transcription and morphology during spore formation. Genes Dev. 8: 2162–2175. [DOI] [PubMed] [Google Scholar]
- Geli, M. I., and H. Riezman, 1998. Endocytic internalization in yeast and animal cells: similar and different. J. Cell Sci. 111: 1031–1037. [DOI] [PubMed] [Google Scholar]
- Gourlay, C. W., H. Dewar, D. R. Warren, R. Costa, N. Satish et al., 2003. An interaction between Sla1p and Sla2p plays a role in regulating actin dynamics and endocytosis in budding yeast. J. Cell Sci. 116: 2551–2564. [DOI] [PubMed] [Google Scholar]
- Hollingsworth, N. M., and B. Byers, 1989. HOP1: a yeast meiotic pairing gene. Genetics 121: 445–462. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ito, H., Y. Fukada, K. Murata and A. Kimura, 1983. Transformation of intact yeast cells treated with alkali cations. J. Bacteriol. 153: 163–168. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iwamoto, M., S. Fairclough, S. Rudge and J. Engebrecht, 2005. Saccharomyces cerevisiae Sps1p regulates trafficking of enzymes required for spore wall synthesis. Eukaryot. Cell 4: 536–544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jäntti, J., M. K. Aalto, M. Öyen, L. Sundqvist, S. Karänen et al., 2002. Characterization of temperature-sensitive mutations in the yeast syntaxin 1 homologues Sso1p and Sso2p and evidence of a distinct function for Sso1p in sporulation. J. Cell Sci. 115: 409–420. [DOI] [PubMed] [Google Scholar]
- Jonsdottir, G. A., and R. Li, 2004. Dynamics of yeast myosin I: evidence for a possible role in scission of endocytic vesicles. Curr. Biol. 14: 1604–1609. [DOI] [PubMed] [Google Scholar]
- Kaksonen, M., Y. Sun and D. G. Drubin, 2003. A pathway for association of receptors, adaptors, and actin during endocytic internalization. Cell 115: 475–487. [DOI] [PubMed] [Google Scholar]
- Kono, K., R. Matsunaga, A. Hirata, G. Suzuki, M. Abe et al., 2005. Involvement of actin and polarisome in morphological change during spore germination of Saccharomyces cerevisiae. Yeast 22: 129–139. [DOI] [PubMed] [Google Scholar]
- Kübler, E., and H. Riezman, 1993. Actin and fimbrin are required for the internalization step of endocytosis in yeast. EMBO J. 12: 2855–2862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kupiec, M., B. Byers, R. E. Esposito and A. P. Mitchell, 1997 Meiosis and sporulation in Saccharomyces cerevisiae, pp. 889–1036 in The Molecular and Cellular Biology of the Yeast Saccharomyces: Cell Cycle and Cell Biology, edited by E. W. Jones. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY.
- Longtine, M. S., A. McKenzie, III, D. J. Demarini, N. G. Shah, A. Wach et al., 1998. Additional modules for versatile and economical PCR-based gene deletion and modification in Saccharomyces cerevisiae. Yeast 14: 953–961. [DOI] [PubMed] [Google Scholar]
- Lynn, R. R., and P. T. Magee, 1970. Development of the spore wall during ascospore formation in Saccharomyces cerevisiae. J. Cell Biol. 44: 688–692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mazur, P., N. Morin, W. Baginsky, M. el-Sherbeini, J. A. Clemas et al., 1995. Differential expression and function of two homologous subunits of yeast 1,3-beta-D-glucan synthase. Mol. Cell. Biol. 15: 5671–5681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Merrifield, C. J., 2004. Seeing is believing: imaging actin dynamics at single sites of endocytosis. Trends Cell Biol. 14: 352–358. [DOI] [PubMed] [Google Scholar]
- Moreau, V., J.-M. Galan, G. Devilliers, R. Haguenauer-Tsapis and B. Winsor, 1997. The yeast actin-related protein Arp2p is required for the internalization step of endocytosis. Mol. Biol. Cell 8: 1361–1375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Moreno-Borchart, A. C., and M. Knop, 2003. Prospore membrane formation: how budding yeast gets shaped in meiosis. Microbiol. Res. 158: 83–90. [DOI] [PubMed] [Google Scholar]
- Munn, A. L., B. J. Stevenson, M. I. Geli and H. Riezman, 1995. end5, end6, and end7: mutations that cause actin delocalization and block the internalization step of endocytosis in Saccharomyces cerevisiae. Mol. Biol. Cell 6: 1721–1742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neiman, A. M., 1998. Prospore membrane formation defines a developmentally regulated branch of the secretory pathway in yeast. J. Cell Biol. 140: 29–37. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Neiman, A. M., L. Katz and P. J. Brennwald, 2000. Identification of domains required for developmentally regulated SNARE function in Saccharomyces cerevisiae. Genetics 155: 1643–1655. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Öyen, M., J. Jäntti, S. Keränen and H. Ronne, 2004. Mapping of sporulation-specific functions in the yeast syntaxin gene SSO1. Curr. Genet. 45: 76–82. [DOI] [PubMed] [Google Scholar]
- Pammer, M., P. Briza, A. Ellinger, T. Schuster, R. Stucka et al., 1992. DIT101 (CSD2, CAL1), a cell cycle-regulated yeast gene required for synthesis of chitin in cell walls and chitosan in spore walls. Yeast 8: 1089–1099. [DOI] [PubMed] [Google Scholar]
- Qualmann, B., M. M. Kessels and R. B. Kelly, 2000. Molecular links between endocytosis and the actin cytoskeleton. J. Cell Biol. 150: F111–F116. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rabitsch, K. P., A. Toth, M. Galova, A. Schleiffer, G. Schaffner et al., 2001. A screen for genes required for meiosis and spore formation based on whole-genome expression. Curr. Biol. 11: 1001–1009. [DOI] [PubMed] [Google Scholar]
- Raths, S., J. Rohrer, F. Crausaz and H. Riezman, 1993. end3 and end4: two mutants defective in receptor-mediated and fluid-phase endocytosis in Saccharomyces cerevisiae. J. Cell Biol. 120: 55–65. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose, K., S. A. Rudge, M. A. Frohman, A. J. Morris and J. Engebrecht, 1995. Phospholipase D signaling is essential for meiosis. Proc. Natl. Acad. Sci. USA 92: 12151–12155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rose, M. D., F. Winston and P. Hieter, 1990 Methods in Yeast Genetics: A Laboratory Course Manual. Cold Spring Harbor Laboratory Press, Cold Spring Harbor, NY.
- Rothstein, R., 1983. One step gene disruption in yeast. Methods Enzymol. 101: 202–211. [DOI] [PubMed] [Google Scholar]
- Rudge, S. A., and J. Engebrecht, 1999. Regulation and function of PLDs in yeast. Biochim. Biophys. Acta 1439: 167–174. [DOI] [PubMed] [Google Scholar]
- Rudge, S. A., A. J. Morris and J. Engebrecht, 1998. Relocalization of phospholipase D activity mediates membrane formation during meiosis. J. Cell Biol. 140: 81–90. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudge, S. A., T. R. Pettitt, C. Zhou, M. J. Wakelam and J. Engebrecht, 2001. SPO14 separation-of-function mutations define unique roles for phospholipase D in secretion and cellular differentiation in Saccharomyces cerevisiae. Genetics 158: 1431–1444. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rudge, S. A., V. A. Sciorra, M. Iwamoto, C. Zhou, T. Strahl et al., 2004. Roles of phosphoinositides and of Spo14p (phospholipase D)-generated phosphatidic acid during yeast sporulation. Mol. Biol. Cell 15: 207–218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sciorra, V. A., S. A. Rudge, J. Wang, S. McLaughlin, J. Engebrecht et al., 2002. Dual role of phosphoinositides in regulation of yeast and mammalian phospholipase D enzymes. J. Cell Biol. 159: 1039–1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheff, M. A., and K. S. Thorn, 2004. Optimized cassettes for fluorescent protein tagging in Saccharomyces cerevisiae. Yeast 21: 661–670. [DOI] [PubMed] [Google Scholar]
- Smits, G. J., H. van den Ende and F. M. Klis, 2001. Differential regulation of cell wall biogenesis during growth and development in yeast. Microbiology 147: 781–794. [DOI] [PubMed] [Google Scholar]
- Smythe, E., and K. R. Ayscough, 2003. The Ark1/Prk1 family of protein kinases: regulators of endocytosis and the actin cytoskeleton. EMBO Rep. 4: 246–251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sun, Y., M. Kaksonen, D. T. Madden, R. Schekman and D. G. Drubin, 2005. Interaction of Sla2p's ANTH doman with PtdIns(4,5)P2 is important for actin-dependent endocytic internalization. Mol. Biol. Cell 16: 717–730. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tachikawa, H., A. Bloecher, K. Tatchell and A. M. Neiman, 2001. A Gip1p-Glc7p phosphatase complex regulates septin organization and spore wall formation. J. Cell Biol. 155: 797–808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tang, H.-Y., J. Xu and M. Cai, 2000. Pan1p, End3p, and Sla1p, three yeast proteins required for normal cortical actin cytoskeleton organization, associate with each other and play essential roles in cell wall morphogenesis. Mol. Cell. Biol. 20: 12–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vida, T. A., and S. D. Emr, 1995. A new vital stain for visualizing vacuolar membrane dynamics and endocytosis in yeast. J. Cell Biol. 128: 779–792. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wach, A., A. Brachat, R. Pohlmann and P. Philippsen, 1994. New heterologous modules for classical or PCR-based gene disruptions in Saccharomyces cerevisiae. Yeast 10: 1793–1808. [DOI] [PubMed] [Google Scholar]
- Wesp, A., L. Hicke, J. Palecek, R. Lombardi, T. Aust et al., 1997. End4p/Sla2p interacts with actin-associated proteins for endocytosis in Saccharomyces cerevisiae. Mol. Biol. Cell 8: 2291–2306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whitacre, J. L., D. A. Davis, K. A. Toenjes, S. M. Brower and A. E. M. Adams, 2001. Generation of an isogenic collection of yeast actin mutants and identification of three interrelated phenotypes. Genetics 157: 533–543. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu, L., M. Ajimura, R. Padmore, C. Klein and N. Kleckner, 1995. NDT80, a meiosis-specific gene required for exit from pachytene in Saccharomyces cerevisiae. Mol. Cell. Biol. 15: 6572–6581. [DOI] [PMC free article] [PubMed] [Google Scholar]