Abstract
CD8+ T cells from HLA-A2.1-transgenic mice, but not wild-type mice, immunized with the amino-terminus region (aa 41–152) of dense granule protein 6 (GRA6Nt) of Toxoplasma gondii secreted large amounts of perforin and granzyme B in response to GRA6Nt through antigen presentation by HLA-A2.1 in vitro. When those CD8+ T cells were transferred into chronically infected HLA-A2.1-expressing NSG mice deficient in T cells, cerebral cyst burden of the recipients of HLA-A2.1-transgenic T cells, but not of WT T cells, became significantly less than that of control mice with no cell transfer. Furthermore, the significant reduction of the cyst burden by a transfer of the HLA-A2.1-transgenic CD8+ immune T cells required an expression of HLA-A2.1 in the recipient NSG mice. Thus, antigen presentation of GRA6Nt by human HLA-A2.1is able to activate anti-cyst CD8+ T cells that eliminate T. gondii cysts through antigen presentation by human HLA-A2.1.
Keywords: Toxoplasma, Tissue cysts, CD8+ T cells, Protective immunity, Dense granule protein 6, HLA-A2
Toxoplasma gondii establishes chronic infection by forming tissue cysts preferentially in the brains of infected hosts. This chronic infection is widespread in humans worldwide [1], and it can reactivate and cause potentially life-threatening toxoplasma encephalitis in immunocompromised individuals such as those with AIDS, cancers, and immunosuppresive treatments for organ transplants [1]. However, there are currently no drugs available to target T. gondii cysts for treatment of this chronic infection. Thus, it is crucial to explore a new approach to target T. gondii cysts for their elimination.
Our recent studies uncovered that CD8+ T cells of genetically resistant BALB/c mice have the capability to destroy T. gondii cysts for their elimination from the brain through their perforin-mediated activity in collaboration with phagocytes that accumulate to the T cell-attacked cysts [2–4]. We also identified the first T. gondii antigen, the amino-terminus region (amino acids 41–152) of dense granule protein 6 (GRA6Nt), capable of activating the anti-cyst cytotoxic CD8+ T cells in the mice [5]. Therefore, it is important to examine whether GRA6Nt is able to activate the anti-cyst CD8+ T cells in the human immune system.
CD8+ T cells recognize their target antigens presented by the major histocompatibility complex (MHC) class I molecules, and different MHC class I molecules can present different target antigens. Since there are three most common MHC class I molecules, HLA-A2, -A3, and-B7, in humans, in the present study we examined whether GRA6Nt of T. gondii is able to activate CD8+ T cells capable of removing T. gondii cysts through antigen presentation by human HLA-A2.1 molecule using transgenic mice expressing this human MHC class I molecule. We found that an immunization of the HLA-A2.1-transgenic mice with recombinant GRA6Nt (rGRA6Nt) activates CD8+ cytotoxic T cells that recognize this antigen presented by human HLA-A2.1, and that an adoptive transfer of the CD8+ immune T cells from the rGRA6Nt-immunized HLA-A2.1 transgenic mice is able to significantly reduce pre-existing cyst burden in the brains of infected immunodeficient NSG mice through antigen presentation by the HLA-A2.1.
1. Materials and methods
1.1. Mice
C57BL/6 (B6) wild-type (WT), C57BL/6 -background transgenic mice expressing human HLA-A2.1, NSG mice expressing HLA-A2.1 (HLA-A2.1-NSG), and control NSG mice not expressing the HLA-A2.1 were from the Jackson Laboratory (Bar Harbor, ME). The NSG mice are immunodeficient and lack both T and B cells and NK cells. Swiss-Webster mice were from Taconic (Germantown, NY). The studies were performed in accordance with approved protocols from the Institutional Animal Care and Use Committee. Female mice were used for all studies. There were 3–5 mice in each experimental group in each experiment.
1.2. Infection with T. gondii
HLA-A2.1-NSG mice and control NSG mice not expressing the HLA-A2.1were infected intraperitoneally with 10 cysts of the ME49 strain of T. gondii obtained from the brains of chronically infected Swiss-Webster mice. Mice were treated with sulfadiazine (400 mg/L) in their drinking water [2,3] beginning at 5 days after infection until the end of the experiments to control tachyzoite proliferation and maintain the parasite as tissue cysts in their brains.
1.3. Immunization with rGRA6Nt and culture of CD8+ T cells
WT and HLA-A2.1-transgenic mice were immunized intraperitoneally with 50 μg of recombinant GRA6Nt (rGRA6Nt) of the genotype II (the 76K strain) of T. gondii [5] twice or three times with 4-week intervals. Two weeks after the final immunization, CD8+ T cells were purified from their spleens as described previously [6,7] and cultured in 96 well-culture plates (3 × 105 cells/well) with or without rGRA6Nt (5 μg/ml) for 72 h in the presence of splenic innate immune cells (1.5 × 105 cells/well) from infected, sulfadiazine-treated HLA-A2.1-NSG mice and the control NSG mice not expressing the HLA-A2.1 as antigen-presenting cells (APCs). There were 5 wells in each experimental group. The concentrations of perforin and granzyme B (GzmB) in the culture supernatants were measured by ELISA using commercial kits from CUSABIO (Houston, TX) and BD Biosciences (San Jose, CA) [8], respectively by following the protocol indicated in their commercial inserts.
1.4. Adoptive transfer of CD8+ T cells
The CD8+ T cells purified from the spleens of the WT and HLA-A2.1-transgenic mice immunized with rGRA6Nt twice or three times described earlier were injected intravenously into infected HLA-2.1-NSG mice (2 × 106 cells/mouse) from a tail vein at 4 weeks after infection. An additional group of the HLA-A2.1-NSG mice did not receive any T cells as a control. In another experiment, CD8+ T cells from the rGRA6Nt-immunized HLA-A2–1-transgenic mice (2 × 106 cells/mouse) were injected intravenously into infected HLA-A2.1-NSG and control NSG mice not expressing the HLA-A2.1. Seven days later, their brains were applied for quantification of the cyst burden by real time RT-PCR.
1.5. Real time RT-PCR
RNA was isolated from half brain of each mouse and treated with DNase I (Invitrogen, Waltham, MA) as described previously [9,10]. cDNA was synthesized with 1 or 4 μg of the purified RNA. PCR reactions with the cDNA were performed using StepOnePlus real-time PCR system with Taqman reagents (Applied Biosystems, Branchburg, NJ) [11]. The sequences of the primers and probe for bradyzoite (cyst)-specific BAG1 were described previously [3,4,12]. The primers and probes for mouse β-actin, perforin, GzmB, and CD8β were ready-made products from Applied Biosystems.
1.6. Statistical analysis
Levels of significance in differences between experimental groups were determined by Student’s t, or Mann–Whitney U test using GraphPad Prism software 9.0. Differences that had p values < 0.05 were considered significant.
2. Results and discussion
CD8+ T cells purified from WT and HLA-2.1-transgenic B6 mice immunized with rGRA6Nt three times were cultured with and without rGRA6Nt in the presence of APCs expressing the HLA-A2.1 for 72 h. Amounts of perforin (Fig. 1A) and GzmB (Fig. 1B) in the culture supernatants were approximately 2 times and 6 times greater, respectively, in the cultures of the HLA-A2.1-transgenic CD8+ T cells stimulated with rGRA6Nt than in their control cultures without the antigen (P < 0.01 for perforin and P < 0.001 for GzmB). In contrast, amounts of perforin and GzmB in the cultures of WT CD8+ T cells did not significantly differ between the presence and absence of rGRA6Nt in the culture, although the amounts of these immune molecules tended to be slightly higher in the presence of the antigen in the culture (Fig. 1). In addition, the amounts of perforin and GzmB in the cultures of the HLA-A2.1-transgenic mouse T cells in the presence of rGRA6Nt were significantly greater than those of the cultures of WT T cells with the antigen (P < 0.05 for perforin and P < 0.001 for GzmB, Fig. 1A and B). In contrast, in the presence of APCs not expressing HLA-A2.1, amounts of perforin and GzmB in the culture supernatants of the HLA-A2.1-transgenic CD8+ T cells did not differ between the presence and absence of rGRA6Nt in the cultures (data not shown). These results indicate that human HLA-A2.1 molecules expressed in the HLA-A2.1-transgenic mice can efficiently present rGRA6Nt of T. gondii and potently activate the cytotoxic activity of CD8+ T cells that recognize this parasite antigen through presentation by the HLA-A2.1.
Fig. 1. Immunization with rGRA6Nt of T. gondii activates CD8+ cytotoxic T cells to this antigen through antigen presentation by human HLA-A2.1 molecule.
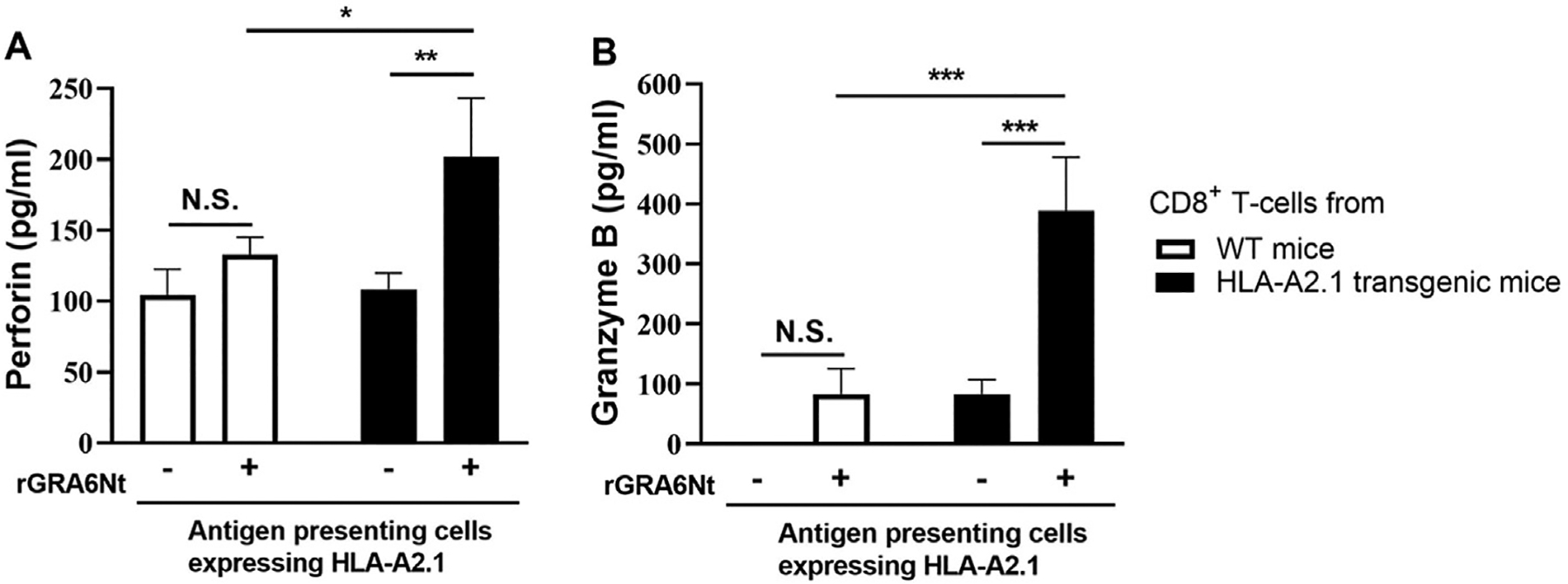
Transgenic mice expressing human HLA-A2.1 and WT mice were immunized intraperitoneally with 50 μg of rGRA6Nt three times with 4-week intervals. Two weeks after the third immunization, CD8+ T cells were purified from their spleens and cultured in 96 well-culture plates (3 × 105 cells/well) with or without rGRA6Nt (5 μg/ml) for 72 h in the presence of splenic innate immune cells (1.5 × 105 cells/well) from infected, sulfadiazine-treated immunodeficient NSG mice (deficient in T, B, and NK cells) expressing the HLA-A2.1 as APCs. CD8+ immune T cells purified from 3 mice were pooled for the cultures. There were 5 wells in each experimental group. The concentrations of perforin (A) and GzmB (B) in the culture supernatants were measured by ELISA. The results presented are from one experiment. *P < 0.05, **P < 0.01, ***P < 0.001.
We next examined whether CD8+ T cells of the HLA-A2.1-transgenic mice immunized with rGRA6Nt are able to remove T. gondii cysts through antigen-presentation by the HLA-A2.1 in infected mice. CD8+ T cells (2 × 106 cells) from the HLA-A2.1-transgenic and WT B6 mice immunized with this antigen three times were injected intravenously into infected and sulfadiazine-treated NSG mice (deficient in T, B, and NK cells) expressing the HLA-A2.1. As a negative control, a group of the infected HLA-A2.1-NSG mice received no T cells. Seven days after the cell transfer, the relative expression levels of mRNA for bradyzoite (cyst)-specific BAG1 in the brains of the recipients of the HLA-A2.1-transgenic CD8+ T cells were approximately a half of those of the control mice with no T cell transfer (P < 0.05, Fig. 2A). Amounts of BAG1 mRNA in the brains of WT CD8+ T cell recipients also tended to be lower than the control groups with no T cell transfer (Fig. 2A), but the difference between WT CD8+ T cell recipients and the control mice did not reach statistical significance (Fig. 2A). We previously demonstrated that CD8+ T cells destroy and eliminate T. gondii cysts using their perforin-mediated mechanism [2,3]. The results from this cell transfer study along with those of the cell cultures shown in Fig. 1 strongly suggest that CD8+ cytotoxic T cells activated by the immunization with rGRA6Nt through antigen-presentation by the HLA-A2.1 molecule are able to remove T. gondii cysts from the brains of infected mice that express this human HLA molecule.
Fig. 2. CD8+ immune T cells activated through antigen presentation of rGRA6Nt of T. gondii by human HLA-A2.1 molecule have the capability to remove tissue cysts of the parasite.

The HLA-A2.1-transgenic and WT mice were immunized intraperitoneally with 50 μg of rGRA6Nt twice (panel E) or three times (panels A-D) with 4-week intervals. Two weeks after the final immunization, CD8+ T cells were purified from their spleens and injected intravenously into infected HLA-2.1-NSG mice (2 × 106 cells/mouse) from a tail vein at 4 weeks after infection. An additional group of the HLA-A2.1-NSG mice did not receive any T cells as a control. These NSG mice were under treatment with sulfadiazine beginning at 5 days after infection to establish a chronic infection and maintain the parasite as the cysts until the end of the study. Seven days after the T cell transfer, the brains of the HLA-A2.1-NSG mice were applied for quantification of mRNA levels for bradyzoite (cyst)-specific BAG1 (A and E), CD8β (B), perforin (C), and GzmB (D) by real time RT-PCR. The results from three (panels A-D) or two (panel E) independent experiments are combined. Number of mice in each experimental group was 10–12 mice (panels A-D) and 7 or 8 mice (panel E). *P < 0.05, **P < 0.01, ***P < 0.001.
After a systemic transfer of WT and HLA-A2.1-transgenic CD8+ immune T cells to infected HLA-A2.1-NSG mice, the transferred T cells need to migrate into the brains of the recipients to display their anti-cyst activity. To address whether the HLA-A2.1-transgenic and WT CD8+ immune T cells both efficiently migrated into the brains of the recipients, we measured cerebral mRNA levels for CD8β in the recipients of those T cells. The CD8β mRNA levels in the brains of the control mice without any T cell transfer were almost undetectable as expected (Fig. 2B) because the NSG mice lack T cells. In contrast, markedly high CD8β mRNA levels were detected similarly in the brains of recipients of the HLA-A2.1-transgenic CD8+ T cells and those of WT CD8+ T cells (Fig. 2B), suggesting that the CD8+ T cells from both donor mouse groups migrated efficiently into the brain of the recipient HLA-A2.1-NSG mice. These results support that the difference in the reduction of BAG1 mRNA levels between the transfer of HLA-A2.1-transgenic and WT CD8+ immune T cells was not due to a difference in their efficiency in migrating into the brains of the recipient mice but due to the difference in their capability to remove T. gondii cysts after migrating into their brains.
After the migration of the transferred CD8+ T cells into the brains of the recipient HLA-A2.1-NSG mice, those T cells need to detect host cells harboring T. gondii cysts through antigen presentation by the MHC class I molecules expressed on those infected cells to activate their anti-cyst cytotoxic activity. Therefore, the activation of the transferred T cells depends on the frequencies of cyst-harboring cells in the brains of the recipients. Thus, for measuring the efficiency of activating the cytotoxic activity of the transferred CD8+ T cells in the recipients, we calculated a ratio of mRNA levels for perforin and GzmB to mRNA levels for BAG1 in the brains of the recipient mice. Markedly increased ratios of perforin mRNA to BAG1 mRNA levels were detected in the brains of the recipients of the HLA-A2.1-transgenic CD8+ T cells and those of WT CD8+ T cells when compared with the control mice with no T cell transfer (P < 0.001, Fig. 2C). In addition, notably the ratios of perforin mRNA levels to BAG1 mRNA levels in the recipients of the HLA-A2.1 transgenic T cells were significantly greater than those of the recipients of the WT T cells (P < 0.05, Fig. 2C). Highly increased GzmB mRNA levels were also detected in both recipients of the HLA-A2.1-transgenic and WT CD8+ T cells (Fig. 2D). The GzmB mRNA levels in the recipients of the HLA-A2.1 CD8+ T cells also tended to be greater than those of the WT T cell recipients, but the difference did not reach statistical significance (Fig. 2D). Since the process of eliminating T. gondii cysts by CD8+ cytotoxic T cells is initiated by perforin [2,3], greater expression of levels of perforin mRNA, rather than GzmB mRNA levels, could efficiently indicate the activation of anti-cyst cytotoxic activity of CD8+ T cells after their migration into the brains of the recipients HLA-A2-NSG mice.
In the studies described above, the donor mice were immunized with rGRA6Nt three times. We examined whether two immunizations with this T. gondii antigen is sufficient to activate the anti-cyst CD8+ T cells. Two weeks after the second immunization, CD8+ T cells from the immunized HLA-A2.1-transgenic and WT mice were transferred into infected, sulfadiazine-treated HLA-A2.1-NSG mice. Seven days after the T cell transfer, the relative BAG1 mRNA levels in the brains of the recipients of the HLA-A2.1-transgenic CD8+ T cells were more than 3 times less than those of the control mice with no T cell transfer (P < 0.05) (Fig. 2E). In contrast, BAG1 mRNA levels in the brains of the WT CD8+ T cell recipients were about a half of those of the control mice without the T cell transfer, and the difference between these two groups did not reach statistical significance (Fig. 2E). Notably, the relative BAG1 mRNA levels in the recipients of the HLA-A2.1-transgenic CD8+ T cells were significantly less than those in the recipients of the WT CD8+ T cells (P < 0.05, Fig. 2E). Thus, two immunizations with rGRA6Nt are sufficient to efficiently activate anti-cyst CD8+ T cells capable of removing T. gondii cysts through antigen presentation by human HLA-A2.1 molecule.
One unexpected difference was noticed between the results on the perforin and GzmB responses of CD8+ T cells from the rGRA6Nt-immunized HLA-A2–1-transgenic and WT mice in the cell cultures (Fig. 1) and in the cell transfer studies (Fig. 2). In the cultures, CD8+ T cells from the immunized HLA-A2.1-transgenic mice, but not those from the WT mice, displayed potent perforin and GzmB secretions in responses to the target antigen in the presence of APCs expressing HLA-A2.1. In contrast, in the cell transfer studies, CD8+ T cells from both of the immunized HLA-A2.1-transgenic and WT mice showed markedly increased perforin and GzmB responses in the recipient NSG mice expressing HLA-A2.1, although the perforin responses of the recipients of the HLA-A2.1-transgenic CD8+ T cells were significantly greater than those of the recipients of the WT CD8+ T cells. One possible reason for this difference is a difference in the efficiency of antigen presentation of GRA6Nt through the mouse MHC class I molecule(s) expressed in the HLA-A2.1-transgenic and WT mice in the in vitro and in vivo systems. Details in this regard is described below.
The HLA-A2.1-transgenic mice express not only human HLA-A2.1 molecule but also the mouse MHC class I molecules from their B6 (the H-2b haplotype)-background. NSG mice (the H-2g7 haplotype) express the H-2Db molecule that is also expressed in the HLA-A2.1-transgenic and WT B6 mice (see Supplemental Table 1). In infection with T. gondii and immunization with rGRA6Nt in vivo, antigen presentation of GRA6Nt by the H-2Db, in addition to that by the HLA-A2.1, may become efficient to activate CD8+ T cells when compared to that in the in vitro culture system shown in Fig. 1. This antigen presentation of GRA6Nt by the H-2Db could be a reason for the increased mRNA expressions for perforin and GzmB in the brains of the infected HLA-A2.1-NSG mice that had received the CD8+ T cells from WT B6 mice immunized with rGRA6Nt shown in Fig. 2.
Since adoptive transfer of CD8+ T cells from the immunized HLA-A2.1-transgenic mice, but not of the T cells from the WT mice, induced a significant reduction of cerebral cyst burden in infected HLA-A2.1-NSG mice, it is most likely that the CD8+ T cells from the transgenic mice recognize cyst-harboring cells in the recipients through antigen presentation by the HLA-A2.1 molecule on the surface of those infected cells. To obtain direct evidence in this regard, we performed an additional experiment, in which CD8+ T cells from rGRA6-immunized HLA-A2.1-transgenic mice were transferred to both infected HLA-A2.1-NSG mice and the NSG mice not expressing the HLA-A2.1. One week after a transfer of CD8+ T cells (2 × 106 cells) purified from the HLA-A2.1-transgenic mice immunized with rGRA6 twice, relative BAG1 mRNA levels in the brains of the HLA-A2.1-NSG mice that had received the T cells were about 3 times less than those of the control HLA-A2.1-NSG mice that had received no T cells (P < 0.05, Fig. 3A). In contrast, in the control NSG mice not expressing HLA-A2.1, relative BAG1 mRNA levels in the brains of the recipients of the HLA-A2.1-transgenic CD8+ immune T cells were a little greater than a half of the control group with no T cell transfer, and the difference did not reach statistical significance (Fig. 3A). Markedly increased mRNA levels for CD8β were detected in the brains of both NSG and HLA-A2.1-NSG mice with the T cell transfer (P < 0.001 for NSG recipients, and P < 0.0001 for HLA-A2.1-NSG recipients, Fig. 3D), strongly suggesting that the CD8+ T cells efficiently migrated into the brains of both of these two groups of the recipient mice. These results indicate that CD8+ T cells from the rGAR6-immunized HLA-A2.1-transgenic mice eliminate T. gondii cysts from the brains of the infected HLA-A2.1-NSG mice through recognition of cyst-harboring cells by antigen presentation by the HLA-A2.1 molecule.
Fig. 3. T. gondii cyst elimination by adoptive transfer of CD8+ T cells from rGRA6Nt-immunized HLA-A2.1-transgenic mice requires an expression of the HLA-A2.1 in the recipients.

HLA-A2.1-transgenic mice were immunized intraperitoneally with 50 μg of rGRA6Nt twice with a 4-week interval. Two weeks after the second immunization, CD8+ T cells (2 × 106 cells) purified from their spleens were injected intravenously into infected HLA-2.1-NSG mice and control NSG mice not expressing the HLA-A2.1 from a tail vein at 4 weeks after infection in the recipients. An additional group of the HLA-A2.1-NSG and control NSG mice did not receive any T cells as a control. These NSG mice were under treatment with sulfadiazine beginning at 5 days after infection to establish a chronic infection and maintain the parasite as the cysts until the end of the study. Seven days after the CD8+ T cell transfer, the brains of the recipient HLA-A2.1-NSG and control NSG mice were applied for quantification of mRNA levels for bradyzoite (cyst)-specific molecules, BAG1 (A), CD8β (B), Prf1 (C), and GzmB (D) by real time RT-PCR. Number of mice in each experimental group was 4 mice. *P < 0.05, **P < 0.01, ***P < 0.001.
Markedly increased levels of the ratios of mRNA for Prf1 and GzmB levels to BAG1 mRNA levels were detected similarly in the brains of both the HLA-A2.1 NSG and control NSG mice that had received the CD8+ cells from rGRA6Nt-immunized HLA-A2.1-transgenic mice (P < 0.001 for Prf1 and P < 0.01 for GzmB in both NSG and HLA-A2.1-NSG recipients, Fig. 3C and D). It is unclear why the relative expression levels of these effector molecules, especially Prf1, were not greater in the former than the latter as observed in the study shown in Fig. 2. One possible reason would be that mRNA levels for Prf1 and GzmB may not accurately reflect the actual cytotoxic activities operated by the CD8+ T cells existing in the brains in the HLA-A2.1-NSG and control NSG mice in this cell transfer study.
The present study shed light on the capability of rGRA6Nt of T. gondii to activate CD8+ cytotoxic T cells specific to this parasite antigen through antigen presentation by human HLA-A2.1 molecule in transgenic mice expressing this MHC class I molecule. Importantly, the present study also uncovered that these CD8+ T cells activated by the rGRA6Nt-immunization in the HLA-A2.1-transgenic mice are able to remove T. gondii cysts through their recognition of cyst-harboring cells through antigen presentation by the HLA-A2.1 molecules. Although there is scattered information on T. gondii antigens that can activate IFN-γ production by T cells through antigen presentation by human HLA molecules [13–15], T. gondii antigen that can activate CD8+ T cells capable of removing T. gondii cysts through antigen presentation by human HLA molecules has not been reported before. There are currently no drugs available to target T. gondii cysts for treating chronic infection of this parasite as mentioned earlier. Therefore, the results from the present study provide a valuable insight on a potential of an immunological intervention to target T. gondii cysts for curing and/or preventing chronic T. gondii infection in humans.
Supplementary Material
Appendix A. Supplementary data
Supplementary data to this article can be found online at https://doi.org/10.1016/j.micinf.2023.105182.
Acknowledgements
The studies are supported in part by the National Institutes of Health, the United States (NIH) grants (AI095032, AI152597, AI136821, and AI134323) (Y.S.). The authors appreciate the support from the Protein Core Laboratory of the University of Kentucky College of Medicine, United States for production and purification of rGRA6Nt.
Footnotes
Declaration of competing interest
All authors declare that there is no conflict of interest.
References
- [1].Montoya JG, Liesenfeld O. Toxoplasmosis. Lancet 2004;363(9425):1965–76. [DOI] [PubMed] [Google Scholar]
- [2].Suzuki Y, Wang X, Jortner BS, Payne L, Ni Y, Michie SA, et al. Removal of Toxoplasma gondii cysts from the brain by perforin-mediated activity of CD8+ T cells. Am J Pathol 2010;176(4):1607–13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].Tiwari A, Hannah R, Lutshumba J, Ochiai E, Weiss LM, Suzuki Y. Penetration of CD8+ cytotoxic T cells into large target, tissue cysts of Toxoplasma gondii, leads to its elimination. Am J Pathol 2019;189(8):1597–607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [4].Lutshumba J, Ochiai E, Sa Q, Anand N, Suzuki Y. Selective upregulation of transcripts for six molecules related to T cell costimulation and phagocyte recruitment and activation among 734 immunity-related genes in the brain during perforin-dependent, CD8(+) T cell-mediated elimination of toxoplasma gondii cysts. mSystems 2020;5(2). [DOI] [PMC free article] [PubMed] [Google Scholar]
- [5].Sa Q, Ochiai E, Tiwari A, Mullins J, Shastri N, Mercier C, et al. Determination of a key antigen for immunological intervention to target the latent stage of Toxoplasma gondii. J Immunol 2017;198(11):4425–34. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Mani R, Abdelaziz MH, Michelon A, Suzuki Y. Human MHC class I molecule, HLA-A2. 1, mediates activation of CD8+ T cell IFN-γ production and the T cell-dependent protection against reactivation of cerebral Toxoplasma infection. Front Immunol 2022;13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].Wang X, Kang H, Kikuchi T, Suzuki Y. Gamma interferon production, but not perforin-mediated cytolytic activity, of T cells is required for prevention of toxoplasmic encephalitis in BALB/c mice genetically resistant to the disease. Infect Immun 2004;72(8):4432–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [8].Miller R, Wen X, Dunford B, Wang X, Suzuki Y. Cytokine production of CD8+ immune T cells but not of CD4+ T cells from Toxoplasma gondii-infected mice is polarized to a type 1 response following stimulation with tachyzoite-infected macrophages. J Interferon Cytokine Res 2006;26(11):787–92. [DOI] [PubMed] [Google Scholar]
- [9].Singh J, Graniello C, Ni Y, Payne L, Sa Q, Hester J, et al. Toxoplasma IgG and IgA, but not IgM, antibody titers increase in sera of immunocompetent mice in association with proliferation of tachyzoites in the brain during the chronic stage of infection. Microb Infect 2010;12(14–15):1252–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [10].Hester J, Mullins J, Sa Q, Payne L, Mercier C, Cesbron-Delauw MF, et al. Toxoplasma gondii antigens recognized by IgG antibodies differ between mice with and without active proliferation of tachyzoites in the brain during the chronic stage of infection. Infect Immun 2012;80(10):3611–20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].Wen X, Kudo T, Payne L, Wang X, Rodgers L, Suzuki Y. Predominant interferon-gamma-mediated expression of CXCL9, CXCL10, and CCL5 proteins in the brain during chronic infection with Toxoplasma gondii in BALB/c mice resistant to development of toxoplasmic encephalitis. J Interferon Cytokine Res 2010;30(9):653–60. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [12].Sa Q, Ochiai E, Tiwari A, Perkins S, Mullins J, Gehman M, et al. Cutting edge: IFN-gamma produced by brain-resident cells is crucial to control cerebral infection with toxoplasma gondii. J Immunol 2015;195(3):796–800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Cong H, Mui EJ, Witola WH, Sisney J, Alexander J, Sette A, et al. Human immunome, bioinformatic analyses using HLA supermotifs and the parasite genome, binding assays, studies of human T cell responses, and immunization of HLA-A*1101 transgenic mice including novel adjuvants provide a foundation for HLA-A03 restricted CD8+T cell epitope based, adjuvanted vaccine protective against Toxoplasma gondii. Immunome Res 2011;6:12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Cardona NI, Moncada DM, Gomez-Marin JE. A rational approach to select immunogenic peptides that induce IFN-gamma response against Toxoplasma gondii in human leukocytes. Immunobiology 2015;220(12):1337–42. [DOI] [PubMed] [Google Scholar]
- [15].El Bissati K, Zhou Y, Paulillo SM, Raman SK, Karch CP, Reed S, et al. Engineering and characterization of a novel Self Assembling Protein for Toxoplasma peptide vaccine in HLA-A*11:01, HLA-A*02:01 and HLA-B*07:02 transgenic mice. Sci Rep 2020;10(1):16984. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


