Abstract
Background:
Currently, exercise-based cardiac rehabilitation (CR) has been receiving increasing interest for its potentially beneficial effects on the health related quality of life (HRQoL) and outcomes of patients with coronary heart disease (CHD). The aim of this study was to evaluate the effect of exercise-based CR on patients after coronary artery bypass graft (CABG) and percutaneous coronary interventions (PCI).
Methods:
We searched PubMed, Embase, Cochrane Library, and Web of Science from inception to 1 December 2023 for relevant studies that evaluated the effect of exercise-based CR on patients after CABG and PCI. Our primary outcomes included mortality, complications, hospital admissions, and HRQoL between patients receiving exercise-based CR and usual care. All statistical analyses were performed using the standard statistical procedures provided in Review Manager 5.2 and Stata 12.0.
Results:
We finally indicated and included 25 randomized controlled trials (RCTs) with 4106 participants for the present analysis. Our pooled results indicated that, compared to usual care, exercise-based CR did not increase the all-cause (relative risk, RR: 0.84; 95% confidence interval, CI: 0.54–1.31) and cardiovascular (RR: 0.98; 95% CI: 0.38–2.54) mortality for patients after coronary artery operation. Similarly, exercise-based CR had an equal effect on coronary artery complications for patients after coronary artery surgery, including CABG (RR: 0.60; 95% CI: 0.32 ‒ 1.15) and PCI (RR: 0.92; 95% CI: 0.55–1.54). It was indicated that exercise-based CR significantly reduced the incidence of myocardial infarction (MI) by half with an RR of 0.50 (95% CI: 0.28–0.90). In addition, exercise-based CR also significantly reduced all-cause hospital admissions with an RR of 0.74 (95% CI: 0.62–0.88). Compared to usual care, exercise-based CR obviously improved HRQoL of patients after coronary artery operation evaluated with SF-36 summary scores (standardized mean difference, SMD: 0.24; 95% CI: 0.11–0.38) and SF-36 8 domains (SMD: 0.35; 95% CI: 0.24–0.46).
Conclusions:
Our analysis indicated that exercise-based CR had a significant effect on the improvement of HRQoL in patients after coronary artery surgeries without increasing mortality or the incidence of re-intervention with operations.
Keywords: cardiac rehabilitation, percutaneous coronary interventions, quality of life
Introduction
Though the French MONICA registries by Meirhaeghe, et al indicated that coronary heart disease (CHD) incidence still decreased between 2006 and 2014 in France[1], Roth, et al updated the GBD 2019 study and indicated that cardiovascular diseases (CVDs), especially coronary artery disease (CAD) remained the leading cause of disease burden in the world[2]. According to WHO 2018[3], coronary heart disease (CHD) was the single most common cause of death globally, with 7.46 million deaths in 2016, accounting for one-third of all deaths. At present, coronary artery bypass graft (CABG) and percutaneous coronary interventions (PCI) are still the standard of care for patients with CHD[4–6]. The postoperative recovery and long-term outcomes, such as quality of life for these patients, have become the most concerning issues.
Exercise-based cardiac rehabilitation (CR) was defined as a supervised or unsupervised inpatient, outpatient, community- or home-based intervention that includes some form of exercise training applied to a cardiac patient population[7]. The intervention could be exercise training alone or exercise training in addition to psychosocial or educational interventions, or both. At present, several organizations, including the American Heart Association (AHA), the American Association of Cardiovascular and Pulmonary Rehabilitation (AACVPR), and the Agency for Health Care Policy and Research, have agreed that a comprehensive CR program, especially exercise-based CR, has benefits in cardiovascular risk reduction, reducing disability, encouraging active and healthy lifestyle changes, and helping maintain those healthy habits. However, no studies have explored the effect of exercise-based CR on patients after coronary artery operations specifically.
Participation in multidisciplinary CR improves the prognosis and quality of life of patients with cardiovascular disease and has received class IA recommendations in international guidelines for the treatment of patients with chronic coronary syndromes. Moreover, participation in CR is cost-effective compared with non-participation and increased utilization of CR results in societal cost-savings[8–9]. Over the past decades, CR has evolved from a one-dimensional exercise-based program to a multidimensional intervention that includes risk factor modification, education, and treatment by psychologists, dietitians, and/or social workers. For patients with CAD, participation in CR reduces all-cause mortality (by 32–35%[10–12]) and cardiovascular mortality (by 26%[12]), major adverse cardiac events, and all-cause hospitalizations (by 23%[12]), and improves quality of life (QoL)[12]. However, previous studies included both non-operative and operative patients with CAD, which had confounding factors and may lead to bias in the results[10–13]. To clarify the effects of exercise-based CR after coronary artery operations, we comprehensively searched and identified studies that evaluated the effect of exercise-based CR on patients receiving CABG and PCI. We explored the effect of exercise-based CR on mortality, complications, and QoL by comparing it to usual care programs. In addition, we explored the effect of exercise-based CR according to subgroups such as operation types, rehabilitation duration, follow-up time, and CR components.
Methods
Search strategy and study selection
We searched PubMed, Embase, Cochrane Library, and Web of Science from inception to 1 December 2023 for relevant studies using a search strategy developed by a medical information specialist that involved controlled vocabulary and keywords relating to “myocardial ischemia, coronary artery bypass, percutaneous coronary intervention, acute coronary syndrom*, ACS, cardiac rehabilitation, exercise therapy, sports, physical exertion, exercise, rehabilitation, physical education, physical training, quality of life, outcome, survival, death.” The search strategy was limited to English-language articles. All references were imported into EndNote, version X9 (Clarivate), for the removal of duplicates. Manual screening of the references in the included articles was also conducted for a more comprehensive search. Two assessors independently screened the titles and abstracts of each study. When a relevant study was identified, its full text was obtained for further evaluation. The full text of related references was also obtained for review.
Criteria for considering studies
We included studies if they met the following PICOS criteria:
(P) patients with CHD received CABG or PCI;
exercise-based CR was considered as intervention;
(C) usual care was considered as controlling;
(O) outcomes, including cardiovascular events, survival outcomes, or other indicators, were assessed;
(S) studies were designed as RCTs.
Studies were excluded if they met the following criteria:
1.experimental trial on animals or a non-human study, non-RCTs;
2.study population included patients with other diseases that would affect outcomes;
3.reported in the form of an abstract, letter, editorial, expert opinion, review, or case report;
4.lack of sufficient data or failure to meet the inclusion criteria.
Data extraction and quality assessment
Two reviewers independently extracted the data from each study using a standardized extraction form. Baseline characteristics and outcomes from the included studies were extracted. Key study characteristics including country, study period, sample size, mean age, CR duration, CR frequency, interval time and follow-up time were extracted. Inconsistencies or any disagreements between reviewers were handled through discussion. A third reviewer was consulted when necessary. In addition, data were examined for accuracy and completeness by a third reviewer.
Two reviewers independently assessed the quality of each study using the validated five-point Jadad scale[14]. The two reviewers were trained according to a predetermined plan and independently reviewed each included study based on the Jadad scale items. The total scores were calculated and used for the final assessment of the quality of each study. Any inconsistency in quality assessment between the reviewers was addressed by a third evaluator. In addition, the risk of bias for each study and the risk of bias across all studies were evaluated and illustrated with figures generated by RevMan 5.2 software[15].
Data synthesis, statistical methods and definitions
The data of comparable outcomes were combined and analyzed using the standard statistical procedures provided in RevMan 5.2[15]. In this meta-analysis, the standardized mean difference (SMD) and relative risk (RR) were used as the treatment effect, with a 95% confidence interval (CI). The heterogeneity between studies was evaluated by the chi-square-based Q statistical test[16], with Ph value and I2 statistic, ranging from 0% to 100%, to quantify the effect of heterogeneity. Ph ≤ 0.10 was deemed to represent significant heterogeneit[17], and pooled estimates were estimated using a random-effects model (the DerSimonian and Laird method[18]). On the contrary, if statistical study heterogeneity was not observed (Ph > 0.10), a fixed-effects model (the Mantel–Haenszel method[19]) was used. In addition, to address the significant heterogeneity and to explore the source of heterogeneity, we also conducted subgroup analysis according to different study designs, study quality, characteristics of participants, and treatment methods. The effects of outcome measures were considered to be statistically significant if pooled SMD with its 95% CI did not overlap with 0 or RR with its 95% CI did not overlap with 1.
Finally, funnel plots were prepared to detect publication bias. If the shape of the funnel plot revealed no obvious evidence of asymmetry, we considered that there was no obvious publication bias. In addition, in order to estimate if the publication bias was significant, we performed Begg’s test and displayed the p-value in the funnel plots. If the p-value of the funnel plot is greater than 0.05, it will be considered no significant publication bias. All statistical analyses were performed using standard statistical procedures provided in RevMan 5.2 and Stata 12.0 (Stata Corp., College Station, TX, USA).
This work has been reported in line with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA)[20] and Assessing the Methodological Quality of Systematic Reviews (AMSTAR, http://links.lww.com/JS9/D882) Guidelines[21].
Health-related quality of life
We assessed the health-related quality of life (HRQoL) using a 36-item short-form (SF-36). The Medical Outcomes Study Short-Form 36 Health Survey (SF-36) is a standard instrument for the assessment of general health-related QoL. This questionnaire was used to quantify HRQoL in patients, independent of their actual state of health or their age. The questionnaire consists of 36 items subdivided into eight dimensions of subjective health: physical functioning, role limitations because of physical problems, bodily pain, general health perceptions, vitality, social functioning, role limitations because of emotional problems, and emotional well-being. All items can be combined to form two summary scales: the physical component score and the mental component score. Component scores were generated by adding the item responses and including given loadings for the different dimensions. Subscale and component scores ranged from 0 (“worst possible functioning”) to 100 (“best possible functioning”).
Results
Included studies, study characteristics, and quality assessment
At the beginning of the search, a total of 4991 records of citations were obtained; 4764 records were reviewed further after duplicates were removed. By screening the titles and abstracts, 2525 studies were excluded preliminarily, and then 143 studies were chosen to get full texts for further evaluation. After reading the full texts, 118 studies were excluded further, as displayed in Fig. 1. Eventually, 25 randomized controlled trials (RCTs)[22–46] with 4106 participants were identified and included in this systematic review and meta-analysis. Of these studies, the majority were single-center RCTs, except three, which were multi-center RCTs[26,35,37]. Three studies were from Russia, three were from the USA, and seven were from China. The other studies were from Sweden, Brazil, Finland, Egypt, Australia, Norway, the Kingdom of Saudi Arabia, Denmark, Japan, Europe, and Bangladesh. Six studies included CABG patients, 11 studies included patients after PCI, and the other studies included both CABG and PCI patients. The detailed search process and summary of studies are shown in the study flow diagram (Fig. 1). The other characteristics of each study are shown in Table 1.
Figure 1.
Flow diagram of literature search and the selection of included studies for meta-analysis.
Table 1.
The characteristics of each included studies for meta-analysis
| Study ID | Study design | Country | Age (mean ± sd), range | Operation | Sample size | Follow-up time | Total duration | Male | Intervention components |
|---|---|---|---|---|---|---|---|---|---|
| Aronov, et al (2019) | Single-center RCT | Russia | I: 58.6 ± 7.0 | CABG | 36 | 12 months | 10 months | 100% | Exercise + Education |
| C: 55.9 ± 7.0 | |||||||||
| Bubnova and Aronov (2020) | Single-center RCT | Russia | I: 51.9 ± 7.9 | PCI | 312 | 12 months | 1 year | 93.6% | Exercise |
| C: 51.7 ± 6.8 | |||||||||
| Carlsson (1998) | Single-center RCT | Sweden | I: 62.7 ± 4.8 | CABG | 235 | 1 year | 2 to 3 months | NR | Exercise + Education |
| C: 59.8 ± 4.8 | PCI | ||||||||
| Chaves, et al (2019) | Single-center RCT | Brazil | I: 59 ± 9.9 | PCI | 115 | 12 months | 6 months | 71.80% | Exercise + Education |
| C: 60.7 ± 8.8 | CABG | ||||||||
| Dorje, et al (2019) | Single-center RCT | China | I: 61.9 ± 8.7 | PCI | 312 | 12 months | 6 months | 81% | Exercise + Education |
| C: 59.1 ± 9.4 | |||||||||
| Engblom, et al (1996) | Single-center RCT | Finland | I: 54.1 ± 5.9 | CABG | 228 | 5 years | 3 weeks | 88% | Exercise + Education |
| C: 54.3 ± 6.2 | |||||||||
| Fletcher, et al (1994) | Single-center RCT | USA | I: 62 ± 8 | CABG | 88 | 6 months | 6 months | 100% | Exercise + Education |
| C: 63 ± 7 | PCI | ||||||||
| Haskell, et al (1994) | Multicenter RCT | USA | I: 58.3 ± 9.2 | CABG | 300 | 4 years | NR | 86% | Exercise + Education |
| C: 56.2 ± 8.2 | PCI | ||||||||
| Hassan and Nahas (2016) | Single-center RCT | Egypt | I: 52.6 ± 5 | PCI | 60 | 12 months | 6 months | 70% | Exercise + Education |
| C: 53.8 ± 5 | |||||||||
| He, et al (2020) | Single-center RCT | China | I: 60.6 ± 12.7 | PCI | 524 | 3 years | 3 years | 45.8% | Exercise |
| C: 60.9 ± 12.9 | |||||||||
| Higgins, et al (2001) | Single-center RCT | Australia | I: 48 (31 ‒ 63) | PCI | 105 | 51 weeks | NR | 83% | Exercise + Psychological + Education |
| C: 47(26‒ 63) | |||||||||
| Ma, et al (2020) | Single-center RCT | China | I: 63.1 ± 9.7 | CABG | 300 | 36 months | 12 months | 80.70% | Exercise + Education + Psychological nursing |
| C: 62.8 ± 10.7 | |||||||||
| Munk, et al (2009) | Single-center RCT | Norway | I: 57 ± 14 | PCI | 40 | 6 months | 6 months | 21% | Exercise |
| C: 61 ± 10 | |||||||||
| Mutwalli, et al (2012) | Single-center RCT | Kingdom of Saudi Arabia | I: 56.8 (53.6 ‒ 59.8) | CABG | 49 | 6 months | 6 months | 100% | Exercise + Education |
| C: 57.22 (54.4 ‒ 60.2) | |||||||||
| Oerkild, et al (2012) | Single-center RCT | Denmark | I: 77.3 ± 6.0 | PCI | 40 | 12 months | 12 months | 63.20% | Exercise + Education |
| C: 76.5 ± 7.7 | CABG | ||||||||
| Pomeshkina, et al (2019) | Single-center RCT | Russia | I: 56.9 ± 4.7 | CABG | 114 | 12 months | NR | 100% | Exercise |
| C: 57.1 ± 4.8 | |||||||||
| Reid, et al (2012) | Multi-center RCT | Canada | I: 56.7 ± 9.0 | PCI | 223 | 12 months | 20 weeks | 82.60% | Exercise + Psychological support |
| C: 56.0 ± 9.0 | |||||||||
| Seki, et al (2003) | Single-center RCT | Japan | I: 69.3 ± 2.9 | PCI | 38 | 6 months | 6 months | 100% | Exercise + Education |
| C: 70.1 ± 3.7 | CABG | ||||||||
| Snoek, et al (2020) | Multi-center RCT | Europe | I: 72.4 ± 5.4 | PCI | 179 | 12 months | 6 months | 78% | Exercise + Education |
| C: 73.6 ± 5.5 | CABG | ||||||||
| Toobert, et al (2000) | Single-center RCT | USA | I: 64 ± 10 | PCI | 25 | 24 months | 24 months | 0% | Exercise + Education + Psychological support |
| C: 63 ± 11 | CABG | ||||||||
| Uddin, et al (2020) | Single-center RCT | Bangladesh | I: 54 ± 6 | CABG | 142 | 12 months | 12 months | 93% | Exercise + Education |
| C: 55 ± 6 | |||||||||
| Xu, et al (2017) | Single-center RCT | China | I: 56.4 ± 8.1 | PCI | 130 | 6 months | 3 months | 79% | Exercise |
| C: 58.6 ± 8 | |||||||||
| Yu, et al (2003) | Single-center RCT | China | I: 62.3 ± 11.2 | PCI | 112 | 2 years | 8 months | 82% | Exercise + Education |
| C: 61.2 ± 10.2 | |||||||||
| Yu, et al (2004) | Single-center RCT | China | I: 64 ± 11 | PCI | 269 | 2 years | 8 months | 76% | Exercise + Education |
| C: 64 ± 11 | |||||||||
| Zhang, et al (2018) | Single-center RCT | China | I: 70.3 ± 10.7 | PCI | 130 | 6 months | 12 months | 90.8% | Exercise |
| C: 69.8 ± 10.4 |
CABG, coronary artery bypass graft; C, controlling, I, intervention; NR, no report; PCI, percutaneous coronary interventions; sd, standard deviation.
All of our included studies demonstrated good quality. The risk of bias for each study was presented as percentages across all included studies, and the risk-of-bias item for each included study was displayed in Figures S1 and S2, http://links.lww.com/JS9/D881. According to our assessment, the risk-of-bias graphs indicated a generally low risk of selection, performance, and detection bias. A high risk of bias was mainly observed in attrition bias and other biases. Unclear risk of bias was distributed across items and was mainly observed in attrition bias and other biases.
Exercise-based CR for mortality
(1) All-cause mortality
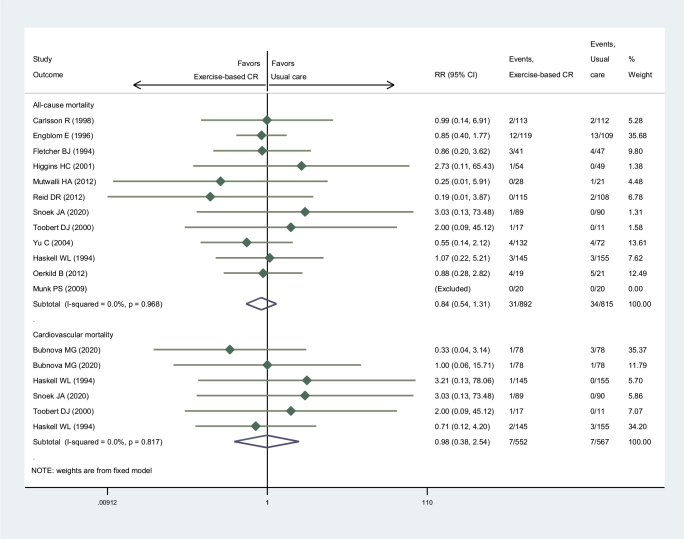
A total of 12 RCTs compared the effect of exercise-based CR for patients after coronary artery surgery on all-cause mortality. Our pooled results indicated that, compared to usual care, exercise-based CR did not increase the all-cause mortality (RR: 0.84; 95% CI: 0.54–1.31) (Fig. 2).
Figure 2.
Forest plot of the exercise-based cardiac rehabilitation effect on all-cause and cardiovascular mortality of patients after coronary artery surgeries. Horizontal lines after each study indicates the range of its 95% CI. Small diamond in each horizontal line indicates the effect size for each study. The large diamond frame indicates the pooled results of each group. CI, confidence interval; CR, cardiac rehabilitation; RR, relative risk.
In addition, we performed subgroup analysis according to the operation type patients received, follow-up time, total intervention duration, male proportion, and CR components. As a result, no significant difference was observed in operation type, follow-up time, total duration, male proportion, and CR components (Table 2).
Table 2.
The pooled results of subgroup analysis of the exercise-based cardiac rehabilitation effect on all-cause mortality for patients after PCI or CABG
| Subgroups | Sample size | Pooled results | Heterogeneity | |||
|---|---|---|---|---|---|---|
| RR [95% CI] | p-value | I2 | Analytical effect model | |||
| Operation | ||||||
| CABG | 277 | 0.78 [0.38, 1.59] | 0.49 | 1% | Fixed-effect model | |
| PCI | 570 | 0.57 [0.19, 1.69] | 0.31 | 0% | Fixed-effect model | |
| PCI + CABG | 860 | 1.05 [0.53, 2.09] | 0.89 | 3% | Fixed-effect model | |
| Follow-up time | ||||||
| 6 to 12 months | 1135 | 0.83 [0.48, 1.46] | 0.53 | 0% | Fixed-effect model | |
| 12 to 36 months | 232 | 0.70 [0.21, 2.33] | 0.56 | 0% | Fixed-effect model | |
| Longer than 3 years | 340 | 0.95 [0.37, 2.44] | 0.92 | 0% | Fixed-effect model | |
| Total duration | ||||||
| ≤6 months | 809 | 0.87 [0.48, 1.56] | 0.63 | 0% | Fixed-effect model | |
| 6 to 12 months | 244 | 0.71 [0.29, 1.70] | 0.44 | 11% | Fixed-effect model | |
| Longer than 12 months | 28 | 2.00 [0.09, 45.12] | 0.66 | 1% | Fixed-effect model | |
| Male | ||||||
| >80% | 991 | 0.80 [0.46, 1.41] | 0.45 | 2% | Fixed-effect model | |
| <80% | 491 | 0.88 [0.40, 1.96] | 0.76 | 7% | Fixed-effect model | |
| CR components | ||||||
| Exercise + Education | 1313 | 0.84 [0.52, 1.34] | 0.46 | 0% | Fixed-effect model | |
| Exercise + Education + Psychological support | 354 | 0.84 [0.19, 3.77] | 0.82 | 0% | Fixed-effect model | |
CABG, coronary artery bypass graft; CI, confidence interval; CR, cardiac rehabilitation; PCI, percutaneous coronary interventions; RR, relative risk; .
(2) Cardiovascular mortality
We further explored the effect of exercise-based CR for patients after coronary artery surgery on cardiovascular mortality, and finally 5, RCTs were identified for pooled analysis. As shown in Fig. 2, the cardiovascular mortality of exercise-based CR was equal to usual care, with a pooled RR of 0.98 (95% CI: 0.38–2.54). We also performed subgroup analysis according to the subgroups mentioned above similarly. As a result, a similar incidence of cardiovascular mortality was also found in each subgroup: operation type, follow-up time, total duration, male proportion and CR components (Table 3).
Table 3.
The pooled results of subgroup analysis of the exercise-based cardiac rehabilitation effect on cardiovascular mortality for patients after PCI or CABG
| Subgroups | Sample size | Pooled results | Heterogeneity | |||
|---|---|---|---|---|---|---|
| RR [95% CI] | p-value | I2 | Analytical effect model | |||
| Operation | ||||||
| PCI | 312 | 0.50 [0.09, 2.69] | 0.42 | 1% | Fixed-effect model | |
| PCI + CABG | 807 | 1.41 [0.42, 4.71] | 0.57 | 3% | Fixed-effect model | |
| Follow-up time | ||||||
| 6 to 12 months | 791 | 1.02 [0.30, 3.46] | 0.98 | 0% | Fixed-effect model | |
| 12 to 36 months | 28 | 2.00 [0.09, 45.12] | 0.66 | 0% | Fixed-effect model | |
| Longer than 3 years | 300 | 0.71 [0.12, 4.20] | 0.71 | 0% | Fixed-effect model | |
| Total duration | ||||||
| ≤6 months | 179 | 3.03 [0.13, 73.48] | 0.5 | 1% | Fixed-effect model | |
| 6 to 12 months | 312 | 0.50 [0.09, 2.69] | 0.42 | 0% | Fixed-effect model | |
| Longer than 12 months | 28 | 2.00 [0.09, 45.12] | 0.66 | 1% | Fixed-effect model | |
| Male | ||||||
| >80% | 912 | 0.76 [0.26, 2.26] | 0.62 | 1% | Fixed-effect model | |
| <80% | 207 | 2.47 [0.27, 22.67] | 0.42 | 0% | Fixed-effect model | |
| CR components | ||||||
| Exercise | 312 | 0.50 [0.09, 2.69] | 0.42 | 3% | Fixed-effect model | |
| Exercise + education | 479 | 3.12 [0.33, 29.75] | 0.32 | 1% | Fixed-effect model | |
| Exercise + education + psychological support | 28 | 2.00 [0.09, 45.12] | 0.66 | 0% | Fixed-effect model | |
CABG: coronary artery bypass graſt; CI, confidence interval; CR: cardiac rehabilitation; PCI: percutaneous coronary interventions; RR, relative risk; .
Exercise-based CR for coronary artery complications
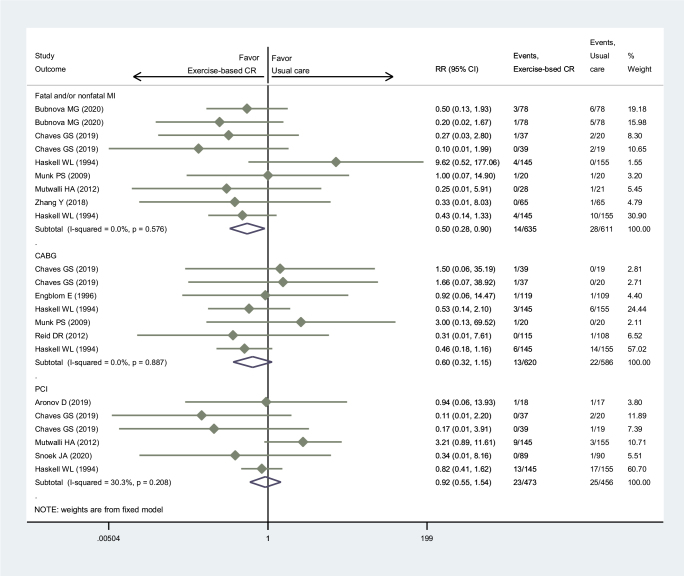
We compared and evaluated the effect of coronary artery complications between exercise-based CR and usual care for patients after coronary artery surgery. As a result, it was indicated that exercise-based CR reduced the incidence of fatal and/or non-fatal MI by half (RR: 0.50; 95% CI: 0.28–0.90) and had an equal effect on re-intervention with both CABG (RR: 0.60; 95% CI: 0.32 ‒ 1.15) and PCI (RR: 0.92; 95% CI: 0.55–1.54) compared to usual care for patients after coronary artery surgery (Fig. 3).
Figure 3.
Forest plot of the exercise-based cardiac rehabilitation effect on coronary artery complications (including MI, CABG, and PCI) for patients after coronary artery surgeries. Horizontal lines after each study indicates the range of its 95% CI. Small diamond in each horizontal line indicates th effect size for each study. The large diamond frame indicates the pooled results of each group. CI, confidence interval; CR, cardiac rehabilitation; RR, relative risk; MI, myocardial infarction; CABG, coronary artery bypass graft; PCI, percutaneous coronary intervention
We also performed a subgroup analysis for the effect of exercise-based CR on fatal and/or non-fatal MI and found an equal effect in subgroups, including operation type, follow-up time, total duration, male proportion, and CR components (Table 4). However, we found a lower incidence of fatal and/or non-fatal MI in exercise-based CR in the subgroup of exercise (RR: 0.39; 95% CI: 0.16–0.94) (Table 4).
Table 4.
The pooled results of subgroup analysis of the exercise-based cardiac rehabilitation effect on the incidence of fatal and/or non-fatal MI for patients after PCI or CABG
| Subgroups | Sample size | Pooled results | Heterogeneity | |||
|---|---|---|---|---|---|---|
| RR [95% CI] | p-value | I2 | Analytical effect model | |||
| Operation | ||||||
| CABG | 49 | 0.25 [0.01, 5.91] | 0.39 | 1% | Fixed-effect model | |
| PCI | 352 | 0.42 [0.15, 1.16] | 0.09 | 0% | Fixed-effect model | |
| PCI + CABG | 715 | 0.61 [0.29, 1.30] | 0.20 | 0% | Fixed-effect model | |
| Follow-up time | ||||||
| 6 to 12 months | 946 | 0.54 [0.27, 1.06] | 0.07 | 0% | Fixed-effect model | |
| Longer than 3 years | 300 | 0.43 [0.14, 1.33] | 0.14 | 0% | Fixed-effect model | |
| Total duration | ||||||
| ≤6 months | 204 | 0.29 [0.08, 1.02] | 0.05 | 0% | Fixed-effect model | |
| 6 to 12 months | 312 | 0.36 [0.12, 1.12] | 0.08 | 1% | Fixed-effect model | |
| Male | ||||||
| >80% | 1091 | 0.56 [0.29, 1.08] | 0.08 | 0% | Fixed-effect model | |
| <80% | 155 | 0.29 [0.07, 1.18] | 0.08 | 1% | Fixed-effect model | |
| CR components | ||||||
| Exercise | 539 | 0.39 [0.16, 0.94] | 0.04 | 1% | Fixed-effect model | |
| Exercise + Education | 707 | 0.63 [0.29, 1.37] | 0.24 | 0% | Fixed-effect model | |
CABG: coronary artery bypass graſt; CI, confidence interval; CR: cardiac rehabilitation; PCI: percutaneous coronary interventions; RR, relative risk; .
Exercise-based CR for all-cause hospital admissions
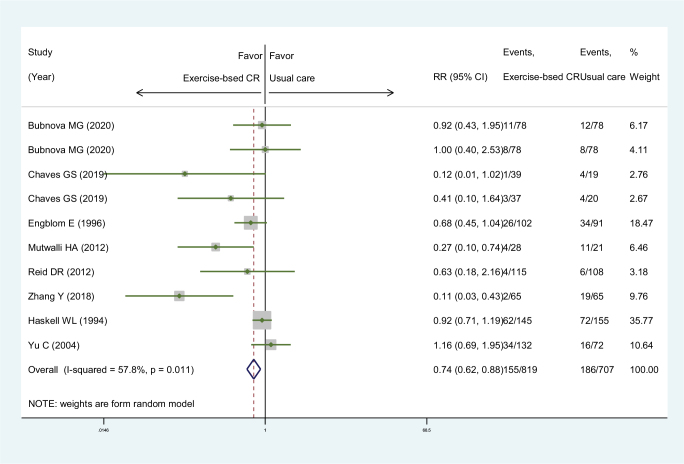
We also compared the incidence of all-cause hospital admissions of patients after coronary artery surgery between exercise-based CR and usual care. Eight RCTs were identified for the pooled analysis, and our pooled result showed that patients who received exercise-based CR had a lower incidence of all-cause hospital admissions (RR: 0.74; 95% CI: 0.52–0.88) (Fig. 4). Subgroup analysis found that patients who received exercise-based CR had a lower incidence of all-cause hospital admissions in the subgroup of follow-up with 6 to 12 months (RR: 0.50; 95% CI: 0.31–0.82) and total duration less than 6 months (RR: 0.52; 95% CI: 0.36–0.75) (Table 5). Similar results were also observed in subgroups of operation type, follow-up time with 12 to 36 months, total duration with 6 to 12 months, male proportion, and CR components (Table 5).
Figure 4.
Forest plot of the exercise-based cardiac rehabilitation effect on all-cause hospital admissions for patients after coronary artery surgeries. Horizontal lines after each study indicates the range of its 95% CI. Small diamond in each horizontal line indicates the effect size for each study. The large diamond frame indicates the pooled results of each group. CI, confidence interval; CR, cardiac rehabilitation; RR, relative risk.
Table 5.
The pooled results of subgroup analysis of the exercise-based cardiac rehabilitation effect on all-cause hospital admissions for patients after PCI or CABG
| Subgroups | Sample size | Pooled results | Heterogeneity | |||
|---|---|---|---|---|---|---|
| RR [95% CI] | P-value | I2 | Analytical effect model | |||
| Operation | ||||||
| CABG | 242 | 0.48 [0.20, 1.16] | 0.10 | 0% | Fixed-effect model | |
| PCI | 869 | 0.71 [0.36, 1.37] | 0.30 | 11% | Fixed-effect model | |
| PCI + CABG | 415 | 0.52 [0.18, 1.48] | 0.22 | 7% | Fixed-effect model | |
| Follow-up time | ||||||
| 6 to 12 months | 1022 | 0.50 [0.31, 0.82] | 0.006 | 50% | Random-effect model | |
| 12 to 36 months | 504 | 0.98 [0.77, 1.23] | 0.83 | 0% | Fixed-effect model | |
| Total duration | ||||||
| ≤6 months | 357 | 0.52 [0.36, 0.75] | 0.0004 | 0% | Fixed-effect model | |
| 6 to 12 months | 442 | 0.52 [0.16, 1.74] | 0.29 | 44% | Random-effect model | |
| Male | ||||||
| >80% | 984 | 0.63 [0.40, 1.00] | 0.05 | 61% | Random-effect model | |
| <80% | 319 | 0.86 [0.54, 1.35] | 0.58 | 0% | Fixed-effect model | |
| CR components | ||||||
| Exercise | 500 | 0.41 [0.13, 1.27] | 0.12 | 45% | Random-effect model | |
| Exercise + education | 599 | 0.65 [0.41, 1.02] | 0.06 | 50% | Random-effect model | |
| Exercise + education + psychological support | 223 | 0.63 [0.18, 2.16] | 0.46 | 0% | Fixed-effect model | |
CABG, coronary artery bypass graft; PCI, percutaneous coronary interventions; CR: cardiac rehabilitation; RR, relative risk; CI, confidence interval.
Exercise-based CR for health-related QoL
(1) SF-36 scale‒summary scores
In addition, we also assessed the effect of exercise-based CR on health-related QoL (HRQoL) with SF-36 scale‒summary scores. Compared with usual care, exercise-based CR significantly improved SF-36 scale‒summary scores of patients after coronary artery operation (SMD: 0.24; 95% CI: 0.11–0.38). Similar result was also observed in subgroup of physical component score (SMD: 0.29; 95% CI: 0.13–0.45). However, no difference was observed in subgroup of mental component score (SMD 0.20; 95% CI −0.03 ‒ 0.42) (Fig. 5).
Figure 5.
Forest plot of the exercise-based cardiac rehabilitation effect on HRQoL SF-36 scale – summary scores for patients after coronary artery surgeries.Horizontal lines after each study indicates the range of its 95% CI. Small diamond in each horizontal line indicates the effect size for each study. The large diamond frame indicates the pooled results of each group. SMD, standardized mean difference; CI, confidence interval; CR, cardiac rehabilitation; HRQoL, health-related quality of life.
(2) SF-36 scale‒8 domains
We evaluated the effect of exercise-based CR on HRQoL using the SF-36 scale ‒ which includes 8 domains, for patients after coronary artery operations. The pooled results showed a significant improvement in the SF-36 scale for patients receiving exercise-based CR compared to usual care (SMD 0.35; 95% CI 0.24 ‒ 0.46). Subgroup analysis also showed higher scores in patients receiving exercise-based CR compared to usual care in physical functioning (SMD: 0.54; 95% CI: 0.38–0.70), physical performance (SMD: 0.21; 95% CI: 0.03–0.38), general health (SMD: 0.32; 95% CI: 0.16–0.48), and vitality (SMD: 0.57; 95% CI: 0.27–0.88) (Fig. 6). However, no significant difference was observed in the subgroups of bodily pain, social functioning, emotional performance, and mental health (Fig. 6).
Figure 6.
Forest plot of the exercise-based cardiac rehabilitation effect on HRQoL SF-36 scale-8 domains for patients after coronary artery surgeries. Horizontal lines after each study indicates the range of its 95% CI. Small diamond in each horizontal line indicates the effect size for each study. The large diamond frame indicates the pooled results of each group. SMD, standardized mean difference; CI, confidence interval; CR, cardiac rehabilitation; HRQoL, health-related quality of life.
Publication bias
Funnel plot analysis was conducted to assess the publication bias of the included literature, and we could evaluate the publication bias by observing whether the shape displayed any obvious asymmetry. According to Fig. 7, the funnel plot with pseudo 95% confidence limits showed no clear evidence of publication bias for mortality (A), coronary artery complications (B), and all-cause hospital admissions (C).
Figure 7.
Funnel plot with pseudo 95% confidence limits showing publication bias of mortality (A), coronary artery complications, (B) and all-cause hospital admissions (C). Dotted lines on both sides indicate pseudo 95% CI. Dots indicate observed studies. RR, relative risk; se, standard error; CI, confidence interval.
Discussion
CVD is one of the leading causes of death worldwide and is the leading cause of death in the United States. CR is a complex, interprofessional intervention customized to individual patients with various CVDs, such as ischemic heart disease, heart failure, and myocardial infarctions, or patients who have undergone cardiovascular interventions, including coronary angioplasty or CABG. CR programs aim to limit the psychological and physiological stresses of CVD, reduce the risk of mortality secondary to CVD, and improve cardiovascular function to help patients achieve the highest QoL possible. Accomplishing these goals is the result of improving overall cardiac function and capacity, halting or reversing the progression of atherosclerotic disease, and increasing the patient’s self-confidence through gradual conditioning.
At present, several organizations, including AHA, AACVPR, and the Agency for Health Care Policy and Research, have agreed that a comprehensive CR program, especially exercise-based CR, has benefits in cardiovascular risk reduction, reducing disability, encouraging active and healthy lifestyle changes, and helping maintain those healthy habits. In addition, Anderson, et al (2016) and Dibben, et al (2021) also demonstrated that exercise-based CR provided important benefits to people with CHD, including reduced risk of MI, a likely small reduction in all-cause mortality, a large reduction in all-cause hospitalization, along with associated healthcare costs, and improved HRQoL up to 12 months’ follow-up[47,48]. Zhong and Liu also demonstrated the longer-term effects of cardiac telerehabilitation on patients with CADs[49]. However, no studies explored the effect of exercise-based CR on patients after coronary artery operations specifically. Thus, we identified 25 RCTs with 4106 participants in this analysis aiming to clarify this issue.
Exercise training has been shown to have direct benefits on the heart and coronary vasculature, including myocardial oxygen demand, endothelial function, autonomic tone, coagulation and clotting factors, inflammatory markers, and the development of coronary collateral vessels[11]. Findings of a previous review of exercise-based CR for CHD supported the hypothesis that reductions in mortality may also be mediated via the indirect effects of exercise through improvements in the risk factors for atherosclerotic disease (i.e., lipids, smoking, and blood pressure)[11–13]. In the present analysis, it was indicated that, compared to usual care, exercise-based CR had an equal effect on all-cause and cardiovascular mortality for patients after coronary artery operations, without increasing the incidence of re-intervention with operations, including CABG and PCI. Further, it was indicated that exercise-based CR significantly reduced all-cause hospital admissions and the incidence of MI by half when compared to usual care. In addition, compared to usual care, exercise-based CR obviously improved the HRQoL of patients after coronary artery operations evaluated with both SF-36 summary scores and SF-36 8 domains. Our results provide initial evidence supporting the use of exercise-based CR as part of the comprehensive rehabilitation of patients after coronary artery operations. By extending the benefits of cardiorespiratory effectiveness, exercise-based CR can promote patients’ long-term awareness of rehabilitation, thereby maximizing the prognosis for each patient.
Before our analysis, Anderson, et al demonstrated that, compared with no exercise control, exercise-based CR reduced the risk of cardiovascular mortality or hospitalization, but not total mortality, MI or revascularization[47]. They failed to assess the value of exercise-based CR in HRQoL for patients with CHD. However, different from their results, our results showed that, compared to usual care, exercise-based CR had an equal effect on all-cause and cardiovascular mortality for patients after coronary artery operation, without increasing the incidence of re-intervention with operations, including CABG and PCI. In addition, Dibben, et al supported the conclusion that exercise-based CR provided important benefits to people with CHD, including reduced risk of MI, a likely small reduction in all-cause mortality, and a large reduction in all-cause hospitalization, along with associated healthcare costs, and improved HRQoL up to 12 months’ follow-up. Similarly, we also found the same results that exercise-based CR significantly reduced all-cause hospital admissions and the incidence of MI by half when compared to usual care. Meanwhile, we also agree with the conclusion of Dibben, et al that compared with usual care, exercise-based CR obviously improved HRQoL of patients after coronary artery operation[48].
However, there existed several limitations in the present analysis. First, due to the lack of patient-level data, we could not perform additional subgroup analyses according to other characteristics such as age, smoking history and economic level. This limitation may lead to a risk of bias in our results, and the applicable population could not be well-identified for practice according to this conclusion. Second, we failed to evaluate the influence of the interval time between post-operation and the beginning of CR. All studies we included failed to provide available information about the optimal starting time of exercise-based CR programs. Thus, we could not determine the optimum time or when exercise-based CR should begin for patients after an operation. Finally, the follow-up time of our included studies ranged from 6 months to 3 years. Thus, the long-term effect of CR on patients after an operation could not be evaluated. In addition, Anderson, et al and Dibben, et al also demonstrated that exercise-based CR provided important benefits to people with CHD, with a large reduction in all-cause hospitalization[47,48]. However, we failed to explore this result. Future studies should be designed to clarify the effectiveness of exercise-based CR.
With the consideration of our analysis results and limitations, there are several suggestions for future research: First, well-designed, adequately-reported RCTs of exercise-based CR in patients with post-operative CHD, more representative of usual clinical practice, are still needed. Trials should explicitly report clinical outcomes, including mortality and hospital admissions, and include validated HRQoL outcome measures, especially over longer-term follow-up, and assess costs and cost-effectiveness. Second, for a long enough follow-up time and large enough sample size, retrospective, propensity score-matched (PSM) studies are also needed to demonstrate the efficacy of exercise-based CR for patients after coronary artery operations. Third, future studies should clarify the opportunity of CR beginning for different populations. Fourth, sufficiently detailed evaluation of variables, such as frailty, nutritional status, depression, or musculoskeletal status, may provide further insight into the benefits of the different components of the rehabilitation programs, which should be the focus of future prospective studies. Fifth, more detailed grouping or subgroup analysis according to different characteristics of patients is needed to clarify which patients may benefit more from exercise-based CR. Sixth, exercise-based CR programs may not only have an effect on the functional and psychocognitive recovery as well as QoL improvement, but also benefit the survival of patients after operations. Future studies should observe these patients with longer follow-up times to clarify their long-term survival status.
Conclusions
Exercise-based CR proved to be more effective in improving the HRQoL of patients after coronary artery operations without increasing mortality or the incidence of re-intervention with operations. Our study highlighted the vital role of exercise-based CR in reducing all-cause hospital admissions and the incidence of MI for patients after coronary artery operation.
Footnotes
Published online 04 February 2025
Contributor Information
Yan Shi, Email: 1246672619@qq.com.
Jige Dong, Email: dongjg2024@163.com.
Ethical approval
Not applicable.
Consent
Written informed consent was obtained from the patient for publication of this case report and accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal on request.
Sources of funding
No funding.
Author’s contribution
The authors of this paper all participated in the study design. All authors read, critiqued and approved the manuscript revisions as well as the final version of the manuscript. Additionally, all authors participated in a session to discuss the results and consider strategies for analysis and interpretation of the data before the final data analysis was performed and the manuscript was written. All authors have the appropriate permissions and rights to the reported data.
Conflicts of interest disclosure
The authors declare no relevant conflict of interest.
Research registration unique identifying number (UIN)
Research registry (UNI: reviewregistry1880).
Guarantor
Jige Dong.
Data availability statement
Not applicable.
Provenance and peer review
Not commissioned, externally peer-reviewed.
References
- [1].Meirhaeghe A, Montaye M, Biasch K, et al. Coronary heart disease incidence still decreased between 2006 and 2014 in France, except in young age groups: results from the French MONICA registries. Eur J Prev Cardiol 2020;27:1178–86. [DOI] [PubMed] [Google Scholar]
- [2].Roth GA, Mensah GA, Johnson CO, et al. Global burden of cardiovascular diseases and risk factors, 1990-2019: update from the GBD 2019 study. J Am Coll Cardiol 2020;76:2982–3021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [3].World Health Organization. Global health estimates 2016: deaths by cause, age, sex, by country and by region, 2000-2016. www.who.int/healthinfo/global_burden_disease/estimates/en/ (accessed prior to 12 October 2021).
- [4].Hannan EL, Wu Y, Harik L, et al. Coronary artery bypass surgery vs. percutaneous interventions for women with multivessel coronary artery disease. J Thorac Cardiovasc Surg 2024;168:863–872.e8. [DOI] [PubMed] [Google Scholar]
- [5].Kirov H, Caldonazo T, Riedel LL, et al. Comparing outcomes between coronary artery bypass grafting and percutaneous coronary intervention in octogenarians with left main or multivessel disease. Sci Rep 2023;13:22323. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [6].Madsen JM, Obling LER, Rytoft L, et al. Pre-hospital pulse glucocorticoid therapy in patients with ST-segment elevation myocardial infarction transferred for primary percutaneous coronary intervention: a randomized controlled trial (PULSE-MI). Trials 2023;24:808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [7].British Association for Cardiovascular Prevention and Rehabilitation. The BACPR standards and core components for cardiovascular disease prevention and rehabilitation 2017. 3rd ed. Available at www.bacpr.com/resources/BACPR_Standards_and_Core_Components_2017.pdf. Accessed 12 January 2021. [Google Scholar]
- [8].Shields GE, et al. Cost-effectiveness of cardiac rehabilitation: a systematic review. Heart 2018;104:1403–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [9].Frederix I, Vandijck D, Hens N. Economic and social impact of increased cardiac rehabilitation uptake and cardiac telerehabilitation in Belgium – a cost-benefit analysis. Acta Cardiol 2018;73:222–29. [DOI] [PubMed] [Google Scholar]
- [10].Salzwedel A, et al. Effectiveness of comprehensive cardiac rehabilitation in coronary artery disease patients treated according to contemporary evidence based medicine: update of the Cardiac Rehabilitation Outcome Study (CROS-II). Eur J Prev Cardiol 2020;27:1756–74. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [11].de Vries H, et al. Cardiac rehabilitation and survival in a large representative community cohort of Dutch patients. Eur Heart J 2015;36:1519–28. [DOI] [PubMed] [Google Scholar]
- [12].Eijsvogels TMH, et al. Association of cardiac rehabilitation with all-cause mortality among patients with cardiovascular disease in the Netherlands. JAMA Network Open 2020;3:e2011686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [13].Dibben GO, et al. Exercise-based cardiac rehabilitation for coronary heart disease: a meta-analysis. Eur Heart J 2023;44:452–69. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [14].Clark HD, Huët C, McAlister FA, Salmi LR, Fergusson D, Laupacis A. Assessing the quality of randomized trials: reliability of the Jadad scale. Control Clin Trials 1999;20:448–52. [DOI] [PubMed] [Google Scholar]
- [15].Review Manager (Revman) [Computer Program]. Version 5.2. Copenhagen: The Nordic Cochrane Centre, The Cochrane Collaboration: 2012. [Google Scholar]
- [16].Lau J, Ioannidis JP, Schmid CH. Quantitative synthesis in systematic reviews. Ann Intern Med 1997;127:820–26. [DOI] [PubMed] [Google Scholar]
- [17].University of York Centre for Reviews and Dissemination. Systematic Reviews: CRD’s Guidance for Undertaking Reviews in Health Care. York: CRD, University of York; 2009. [Google Scholar]
- [18].DerSimonian R, Laird N. Meta-analysis in clinical trials revisited. Contemp Clin Trials 2015;45:139–45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [19].Mantel N, Haenszel W. Statistical aspects of the analysis of data from retrospective studies of disease. J Natl Cancer Inst 1959;22:719–48. [PubMed] [Google Scholar]
- [20].Page MJ, McKenzie J, Bossuyt PM, et al. The PRISMA 2020 statement: an updated guideline for reporting systematic reviews, Int J Surg 2021;88:105906. [DOI] [PubMed] [Google Scholar]
- [21].Shea BJ, Reeves BC, Wells G, et al. AMSTAR 2: a critical appraisal tool for systematic reviews that include randomised or non-randomised studies of healthcare interventions, or both. BMJ 2017;21:358:j4008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [22].Aronov D, et al. Clinical efficacy of а medical centre- and home-based cardiac rehabilitation program for patients with coronary heart disease after coronary bypass graft surgery. Arch Med Res 2019;50:122–32. [DOI] [PubMed] [Google Scholar]
- [23].Bubnova MG, Aronov DM. Physical rehabilitation aLer acute myocardial infarction: focus on body weight. Russ J Cardiol 2020;25:3867. [Google Scholar]
- [24].Carlsson R. Serum cholesterol, lifestyle, working capacity and quality of life in patients with coronary artery disease. Experiences from a hospital-based secondary prevention programme. Scand Cardiovasc J Suppl 1998;50:1–20. [DOI] [PubMed] [Google Scholar]
- [25].Chaves GSS, et al. Maintenance of gains, morbidity, and mortality at 1 year following cardiac rehabilitation in a middle-income country: a wait-list control crossover trial. J Am Heart Assoc 2019;8:e011228. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [26].Dorje T, et al. Smartphone and social media-based cardiac rehabilitation and secondary prevention in China (SMART-CR/SP): a parallel-group, single-blind, randomised controlled trial. Lancet Digit Health 2019;1:e363–e374. [DOI] [PubMed] [Google Scholar]
- [27].Engblom E, et al. Effects of five years of cardiac rehabilitation after coronary artery bypass grafting on coronary risk factors. Am J Cardiol 1996;78:1428–31. [DOI] [PubMed] [Google Scholar]
- [28].Fletcher BJ, et al. Exercise testing and training in physically disabled men with clinical evidence of coronary artery disease. Am J Cardiol 1994;73:170–74. [DOI] [PubMed] [Google Scholar]
- [29].Haskell WL, et al. Effects of intensive multiple risk factor reduction on coronary atherosclerosis and clinical cardiac events in men and women with coronary artery disease. the Stanford Coronary Risk Intervention Project (SCRIP). Circulation 1994;89:975–90. [DOI] [PubMed] [Google Scholar]
- [30].Hassan AM, El Nahas NG. Efficacy of cardiac rehabilitation after percutaneous coronary intervention. Int J PharmTech Res 2016;9:134–41. [Google Scholar]
- [31].He CJ, et al. Effect of exercise-based cardiac rehabilitation on clinical outcomes in patients with myocardial infarction in the absence of obstructive coronary artery disease (MINOCA). Int J Cardiol 2020;315:9–14. [DOI] [PubMed] [Google Scholar]
- [32].Higgins HC, Hayes RL, McKenna KT. Rehabilitation outcomes following percutaneous coronary interventions (PCI). Patient Educ Couns 2001;43:219–30. [DOI] [PubMed] [Google Scholar]
- [33].Ma L, Deng L, Yu H. The effects of a comprehensive rehabilitation and intensive education program on anxiety, depression, quality of life, and major adverse cardiac and cerebrovascular events in unprotected left main coronary artery disease patients who underwent coronary artery bypass grafting. Ir J Med Sci 2020;189:477–88. [DOI] [PubMed] [Google Scholar]
- [34].Munk PS, et al. High-intensity interval training may reduce in-stent restenosis following percutaneous coronary intervention with stent implantation A randomized controlled trial evaluating the relationship to endothelial function and inflammation. Am Heart J 2009;158:734–41. [DOI] [PubMed] [Google Scholar]
- [35].Mutwalli HA, et al. Randomized controlled evaluation shows the effectiveness of a home-based cardiac rehabilitation program. Saudi Med J 2012;33:152–59. [PubMed] [Google Scholar]
- [36].Oerkild B, et al. Home-based cardiac rehabilitation is an attractive alternative to no cardiac rehabilitation for elderly patients with coronary heart disease: results from a randomised clinical trial. BMJ Open 2012;2:e001820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [37].Pomeshkina SA, Barbarash OL, Pomeshkin EV. [Exercise training and erectile dysfunction in patients after coronary artery bypass grafting]. Ter Arkh 2019;91:16–20. [DOI] [PubMed] [Google Scholar]
- [38].Reid RD, et al. Randomized trial of an internet-based computer-tailored expert system for physical activity in patients with heart disease. Eur J Prev Cardiol 2012;19:1357–64. [DOI] [PubMed] [Google Scholar]
- [39].Seki E, et al. Effects of phase III cardiac rehabilitation programs on health-related quality of life in elderly patients with coronary artery disease: Juntendo Cardiac Rehabilitation Program (J-CARP). Circ J 2003;67:73–77. [DOI] [PubMed] [Google Scholar]
- [40].Snoek JA, et al. Effectiveness of home-based mobile guided cardiac rehabilitation as alternative strategy for nonparticipation in clinic-based cardiac rehabilitation among elderly patients in Europe: a randomized clinical trial. JAMA Cardiol 2021;6:463–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [41].Toobert DJ, Glasgow RE, Radcliffe JL. Physiologic and related behavioral outcomes from the women’s lifestyle heart trial. Ann Behav Med 2000;22:1–9. [DOI] [PubMed] [Google Scholar]
- [42].Uddin J, et al. Effect of home-based cardiac rehabilitation in a lower-middle income country: results from a controlled trial. J Cardiopulm Rehabil Prev 2020;40:29–34. [DOI] [PubMed] [Google Scholar]
- [43].Xu Y, Feng Y, Su P, Li Y, Li C, Qiao J. Impact of exercise rehabilitation on cardiac function in coronary artery disease patients after percutaneous coronary intervention. Chin Circ J 2017;32:326–30. [Google Scholar]
- [44].Yu CM, et al. A short course of cardiac rehabilitation program is highly cost effective in improving long-term quality of life in patients with recent myocardial infarction or percutaneous coronary intervention. Arch Phys Med Rehabil 2004;85:1915–22. [DOI] [PubMed] [Google Scholar]
- [45].Yu CM, et al. Long-term changes in exercise capacity, quality of life, body anthropometry, and lipid profiles after a cardiac rehabilitation program in obese patients with coronary heart disease. Am J Cardiol 2003;91:321–25. [DOI] [PubMed] [Google Scholar]
- [46].Zhang Y, et al. Cardiac rehabilitation in acute myocardial infarction patients after percutaneous coronary intervention: a community-based study. Medicine (Baltimore) 2018;97:e9785. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [47].Anderson L, et al. Exercise-based cardiac rehabilitation for coronary heart disease. Cochrane Database Syst Rev 2016;2016:Cd001800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [48].Dibben G, et al. Exercise-based cardiac rehabilitation for coronary heart disease. Cochrane Database Syst Rev 2021;11:Cd001800. [DOI] [PMC free article] [PubMed] [Google Scholar]
- [49].Zhong W, Liu R. Longer-term effects of cardiac telerehabilitation on patients with coronary artery disease: systematic review and meta-analysis. JMIR Mhealth Uhealth 2023;11:e46359. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.