Abstract
By integrating photothermal and chemodynamic properties, Ru-based nanomaterials have emerged as promising agents for tumor therapy. However, their clinical translation is hindered by high systemic toxicity, suboptimal therapeutic efficacy, and compromised chemodynamic performance caused by tumor antioxidant defense mechanisms. A multifunctional therapeutic platform (Ru-PC-PEITC-ALG) was developed through the coordination-driven self-assembly of ruthenium ions with procyanidins (PCs) to form Ru-PC nanoparticles, followed by coencapsulation with phenethyl isothiocyanate (PEITC) in a sodium alginate hydrogel. The Ru-PC complex demonstrated exceptional photothermal conversion efficiency, enabling rapid intratumoral temperature elevation under 808 nm laser irradiation to achieve localized thermal ablation. Simultaneously, Ru-PC exhibited tumor microenvironment-responsive catalytic activity, catalyzing the conversion of hydrogen peroxide (H2O2) into highly toxic hydroxyl radicals (·OH) via Fenton-like reactions. This ROS generation was substantially amplified through synergistic photothermal acceleration of reaction kinetics and PEITC-mediated glutathione (GSH) depletion, which effectively disabled the antioxidant defense system. Systematic evaluations, including in vitro cytotoxicity assays, transcriptomic sequencing, and murine xenograft models, confirmed the platform’s superior tumor suppression ability and favorable biosafety profile. Mechanistic studies revealed that combination therapy induced mitochondrial dysfunction and activated the apoptosis/ferroptosis pathways. This work presents a “precision disruption” strategy against tumor antioxidant armor, advancing the rational design of metal‒polyphenol-coordinated nanomaterials for enhanced oncotherapy.
Graphical Abstract
Supplementary Information
The online version contains supplementary material available at 10.1186/s12951-025-03626-1.
Introduction
In recent years, ruthenium-based nanomaterials have garnered significant attention in tumor therapy due to their unique physicochemical properties, including efficient photothermal conversion in the near-infrared (NIR) window and pH-universal Fenton-like catalytic activity [1–3]. In particular, ruthenium‒polyphenol coordination nanomaterials engineered through synergistic metal‒ligand interactions exhibit optimized photothermal performance with enhanced NIR absorption and superior photothermal conversion efficiency [4, 5]. Compared with traditional iron-based counterparts, the multivalent transition capability of ruthenium enables sustained reactive oxygen species (ROS) generation across a broad pH range (4.0–7.0), ensuring tumor microenvironment-specific therapeutic efficacy [6–8]. However, the clinical translation of current ruthenium coordination systems is limited by insufficient biosafety due to off-target nanoparticle accumulation and imprecise in vivo delivery, as well as compromised therapeutic outcomes caused by robust intracellular antioxidant defense mechanisms in tumor cells [9–11]. To address these limitations, the rational design of advanced nanoplatforms with improved photothermal efficiency, catalytic robustness, and enhanced biocompatibility has become a pivotal research frontier.
Procyanidin (PC), a class of naturally occurring polyphenolic compounds formed through C‒C bond-mediated condensation of catechin and epicatechin monomers, is abundantly distributed in botanical sources such as grape seeds, blueberries, and apples [12, 13]. These compounds have garnered significant research interest because of their distinctive anticancer properties. Mechanistic studies revealed that PC exerts antitumor effects through multifaceted regulation of key oncogenic signaling pathways, including nuclear factor kappa-B (NF-κB), mitogen-activated protein kinase (MAPK), and phosphatidylinositol 3-kinase/protein kinase B (PI3K/AKT) cascades, thereby suppressing malignant proliferation and promoting apoptotic cell death [14–16]. Notably, PC directly interacts with the catalytic domain of the AKT protein, effectively inhibiting its phosphorylation and subsequent activation. This molecular intervention disrupts downstream survival signaling pathways, resulting in significant growth suppression of hepatocellular carcinoma cells [14]. In ovarian cancer models, PC administration has been shown to induce apoptotic cell death through ROS overproduction, mitochondrial membrane potential collapse, and sequential activation of caspase cascades, as demonstrated in both in vitro and xenograft studies [17]. In addition to exerting direct tumoricidal effects, PC exhibits remarkable antiangiogenic activity that disrupts tumor neovascularization, thereby creating a nutrient-deprived microenvironment detrimental to cancer progression [18]. Despite these promising therapeutic potentials, the clinical translation of PC faces significant pharmacological challenges, particularly its inherent limitations in bioavailability and rapid systemic clearance, which substantially compromise monotherapy outcomes. Recent advances highlight that the polyphenolic architecture of PC enables stable coordination complex formation with various metal ions [19, 20]. This chemical modification not only enhances compound stability but also improves pharmacokinetic profiles. Consequently, the rational design of metal-coordinated PC nanoparticles represents a promising strategy to increase their therapeutic efficacy while overcoming inherent pharmacological limitations.
Glutathione (GSH), the most abundant antioxidant in tumor cells, directly neutralizes the ROS generated during chemodynamic therapy (CDT), significantly compromising its oxidative damage efficacy [21–23]. This fundamental limitation constitutes a critical barrier to CDT effectiveness. Consequently, the selective depletion of intracellular GSH has emerged as a pivotal strategy for enhancing CDT outcomes. Phenethyl isothiocyanate (PEITC), a naturally occurring small-molecule compound, can scavenge GSH through two mechanisms: its electrophilic group directly conjugates with the sulfhydryl groups of GSH to form redox-inactive GSH‒PEITC adducts while simultaneously suppressing glutamate‒cysteine ligase (GCL) activity to disrupt GSH biosynthesis [24, 25]. Experimental evidence indicates that PEITC-mediated GSH depletion enhances tumor cell sensitivity to ROS by 5–8-fold, creating crucial opportunities for therapeutic amplification in CDT [26, 27]. Furthermore, PEITC exerts additional antitumor effects through the inhibition of the PI3K/Akt and NF-κB signaling pathways, coupled with the release of damage-associated molecular patterns (DAMPs) to induce immunogenic cell death [28, 29]. Despite these multimodal therapeutic advantages, the clinical translation of PEITC faces substantial challenges because of its extremely low aqueous solubility (< 0.1 mg/mL) and inefficient tumor-targeted delivery, which collectively limits its bioavailability and therapeutic applicability [30].
To address the aforementioned challenges, an integrated antitumor platform (Ru-PC-PEITC-ALG) was developed through the coordination of ruthenium ions with PC, resulting in the formation of photoresponsive Ru-PC nanoparticles, followed by coencapsulation with PEITC in a sodium alginate (ALG) hydrogel matrix. The ALG hydrogel undergoes in situ gelation triggered by Ca2+ ions within the tumor microenvironment, enabling localized and sustained delivery of Ru-PC and PEITC to maximize therapeutic precision. Under 808 nm near-infrared (NIR) irradiation, Ru-PC demonstrates exceptional photothermal conversion efficiency, achieving rapid temperature elevation for effective photothermal ablation. The synergistic GSH depletion effect of Ru-PC and PEITC disrupted the antioxidant defense system of tumors and amplified the peroxidase (POD)-like catalytic activity of Ru-PC. This dual mechanism of action takes advantage of the elevated H2O2 levels and acidic pH in the tumor microenvironment to promote the generation of tumor-specific ROS via a Fenton-like reaction, thereby establishing a self-reinforcing “oxidative storm” cascade. Ru-PC-PEITC-ALG demonstrated good multienzyme activity and photothermal effects on the conversion efficiency of the characterization experiments, and transcriptome analysis revealed that Ru-PC-PEITC-ALG triggered mitochondrial damage and apoptosis and disrupted the cellular structure via photothermal effects under near-infrared light irradiation. In addition, the antitumor efficiency and biosafety of Ru-PC-PEITC-ALG were verified in animal experiments. This “precise delivery and antioxidant demethylation” strategy overcomes the limitations of monotherapy by spatiotemporally coordinating the photothermal and chemodynamic effects. This study establishes a paradigm for the application of metal‒polyphenol nanoplatforms in the treatment of solid tumors, with the dual advantages of microenvironmental adaptability and biosafety.
Materials and methods
Materials
Ruthenium chloride (RuCl3), proanthocyanidins, and GSH were purchased from Macklin Co., Ltd. (China). 3,3’,5,5’-Tetramethylbenzidine (TMB), 1,2-diaminobenzene (OPD), 5,5’-dithiobis-(2-nitrobenzoic acid) (DTNB), and polyvinylpyrrolidone (PVP) were purchased from Aladdin Co., Ltd. (China). Phosphate-buffered saline (PBS), RPMI 1640 medium, and trypsin-EDTA solution were purchased from Biosharp Biologics Co., Ltd. (China). Fetal bovine serum (FBS) was purchased from Gibco Co. (USA). The CCK-8 kits were obtained from SparkJade Co., Ltd. (China). ThiolTracker™ Violet was purchased from Thermo Fisher Scientific, Inc. (USA). Calcein-AM/PI and Annexin/PI kits were purchased from Bestbio Co., Ltd. (China). 2,7-Dichlorodihydrofluorescein diacetate (DCFH-DA), JC-1, DAPI, and GSH kits were purchased from Beyotime Co., Ltd. (China).
Preparation of Ru-PC
The Ru-PC nanocomplex was synthesized through a facile coordination-mediated self-assembly approach [19]. Briefly, 33 mg of polyvinylpyrrolidone (PVP) was completely dissolved in 5 mL of methanol under magnetic stirring. Subsequently, 1 mL of methanolic solution containing 20 mg of RuCl3 was introduced dropwise into the PVP solution, followed by continuous stirring in 120r/min for 5 min. Meanwhile, 10 mg of PC was separately dissolved in methanol and then incorporated into the above mixture. The reaction system was maintained under vigorous stirring for 3 h at ambient temperature, during which the color of the solution changed to dark green. The resulting mixture was subjected to overnight dialysis via membranes with molecular weight cutoffs ranging from 8,000 to 14,000 kDa to remove unreacted precursors. Following dialysis, the product was centrifuged at 10,000 rpm, washed three times with ultrapure water, and lyophilized to obtain grayish-black Ru-PC nanoparticles.
Ru-PC characterization
A projected electron microscope (JEM-1400 Plus, Tokyo, Japan) was used to study the morphology and elemental distribution of the samples, and the chemical composition was determined via X-ray photoelectron spectroscopy (XPS) (Thermo Scientific K-Alpha, Beijing, China). A UVEVIS spectrophotometer (Thermo Scientific™ GENESYS™ 50, USA) was used to measure the ultraviolet (UV) absorption spectrum of the substance.
Ru-PC-like enzyme activity and PEITC bioactivity
The validation experiments of POD-like enzymes were performed with TMB and OPD probes. Briefly, different concentrations of Ru-PC were mixed with H2O2, ROS generation was detected by the addition of 1 µmol/L TMB, and the absorbance curves and the change in absorbance at 652 nm of the supernatant after centrifugation were detected via a UV spectrophotometer. Similarly, the absorbance curve and the change in absorbance at 442 nm were detected by replacing TMB with OPD. Glutathione peroxidase-like enzyme activity was verified via the colorimetric reaction of DTNB with GSH. Different concentrations of Ru-PC were mixed and reacted with GSH. DTNB was added to the reaction system after a period of time, and the supernatant was centrifuged to measure the absorbance at 412 nm via a UV spectrophotometer (Thermo Scientific™ GENESYS™ 50, USA).
Characterization and properties of Ru-PC-PEITC-ALG
The nanotherapeutic platform Ru-PC-PEITC-ALG was prepared by mixing Ru-PC or PEITC with 5 mg/mL of the suggested ALG solution and evaluated for its characterization and properties in terms of gel-forming and loading capacity [31, 32]. High-resolution scanning electron microscopy (SEM) images of Ru-PC-PEITC-ALG were obtained by scanning electron microscopy (Tescan -MIRA LMS, Czech Republic). To measure the fluidic properties of Ru-PC-PEITC-ALG, the rheological properties of the hydrogels were evaluated via a rotational rheometer (MCR 302e, Austria). The manufactured ALG, Ru-PC and Ru-PC-PEITC-ALG were formed into gels and then freeze-dried, and the remaining solids were ground and supplemented with potassium bromide. Fourier transform infrared (FTIR) spectra were obtained via a Fourier transform infrared spectrometer (IRTracer 100, Japan). A simulated tumor microenvironment was prepared by adjusting the Ca2+ concentration to 1.8 mm in PBS, and the amount of Ru ions released from Ru-PC and Ru-PC-PEITC-ALG was measured via inductively coupled plasma‒mass spectrometry (ICP) in the simulated tumor microenvironment (TME) environment for 72 h.
Photothermal performance of Ru-PC-PEITC-ALG
To evaluate the photothermal properties of Ru-PC, the temperature changes in a 200 µg/mL Ru-PC solution under NIR irradiation (808 nm) at various power densities were measured over 4 min via an infrared thermal camera. Similarly, the temperature variations of Ru-PC solutions at different concentrations were monitored for 4 min under 808 nm NIR laser irradiation at 1.5 W/cm². The thermal stability of Ru-PC was validated through multiple NIR irradiation cycles. Additionally, the stability of Ru-PC was assessed by comparing its ultraviolet‒visible (UV‒Vis) absorption spectra before and after light irradiation via UV‒Vis spectrophotometry.
Cell line
Human umbilical vein endothelial cells (HUVECs) and murine breast cancer 4T1 cells were obtained from Procell Life Science & Technology Co., Ltd. (Wuhan, China). HUVECs were cultured in DMEM supplemented with 10% fetal bovine serum (FBS), 100 U/mL penicillin, and 100 µg/mL streptomycin. 4T1 cells were maintained in RPMI 1640 medium supplemented with 10% FBS, 100 U/mL penicillin, and 100 µg/mL streptomycin. All the cells were incubated at 37 °C in a humidified atmosphere with 5% CO2.
In vitro cytotoxicity of Ru-PC-PEITC-ALG
The cytotoxicity of Ru-PC-PEITC-ALG on HUVECs and 4T1 cells was evaluated via a CCK-8 assay kit. The cells were seeded into 96-well plates and allowed to adhere and reach 80% confluency. The cells were subsequently treated with different concentrations of Ru-PC-PEITC-ALG and coincubated for 6 h. For the groups designated for photothermal treatment, the cells were exposed to 808 nm near-infrared (NIR) irradiation at an intensity of 1.5 W/cm² for 4 min. After irradiation, the supernatant was removed, and the cells were incubated with 10% CCK-8 solution in fresh medium at 37 °C for 2 h to allow color development. The absorbance at 450 nm was measured via a microplate reader (SpectraMax iD3, Molecular Devices, Japan), and the cell viability was calculated on the basis of the optical density values.
The cytotoxic effect of Ru-PC-PEITC-ALG was further evaluated via calcein-AM/PI live/dead staining. 4T1 cells were seeded into confocal dishes and allowed to grow to 80–90% confluency. After treatment with different concentrations of Ru-PC-PEITC-ALG and 6 h of coincubation, the cells designated for PTT were irradiated with an 808 nm NIR laser (1.5 W/cm², 4 min). Following irradiation, the supernatant was removed, and the cells were washed with PBS. The cells were subsequently stained with calcein-AM and PI sequentially at 37 °C for 20 min in the dark to distinguish live (green fluorescence) and dead (red fluorescence) cells. Imaging was performed via a fluorescence confocal microscope (CSIM-130, Sunny Technology, China), and representative images were captured for qualitative analysis of cell viability.
Ru-PC-PEITC-ALG induces apoptosis in 4T1 cells
The apoptotic effect of Ru-PC-PEITC-ALG on 4T1 cells was evaluated via Annexin V-FITC/PI staining combined with flow cytometry. The cells were seeded into 6-well plates, treated with different concentrations of Ru-PC-PEITC-ALG, and coincubated for 6 h. For photothermal treatment, the cells were irradiated with an 808 nm NIR laser (1.5 W/cm², 4 min). After irradiation, both the supernatant and adherent cells were collected, centrifuged, washed with PBS, and stained with Annexin V-FITC and propidium iodide (PI) at 4 °C in the dark. Apoptotic rates were quantified via a flow cytometer (BD FACSCalibur, USA).
To assess intracellular reactive oxygen species (ROS) levels, DCFH-DA staining was performed. 4T1 cells were seeded into confocal dishes, treated with Ru-PC-PEITC-ALG, and coincubated for 6 h followed by NIR irradiation. The supernatant was discarded, and the cells were incubated with 10 µM DCFH-DA at 37 °C for 30 min in the dark. Nuclei were counterstained with DAPI (1 µg/mL) for 5 min. Fluorescence images were captured via a confocal microscope (CSIM-130, Sunny Technology, China), and semiquantitative analysis of the ROS fluorescence intensity was conducted via ImageJ software.
Exploring the synergies of the PEITC
To explore the synergistic role of PEITC, changes in the mitochondrial membrane potential were analyzed via JC-1 staining. 4T1 cells were seeded into confocal dishes and incubated to the appropriate confluency. After treatment with Ru-PC-PEITC-ALG and incubation for 6 h, the cells were irradiated with an 808 nm NIR laser (1.5 W/cm², 4 min). The supernatant was removed, and the cells were washed thoroughly with PBS, followed by staining with JC-1 dye (5 µg/mL) at 37 °C for 20 min in the dark. The cells were then washed three times with precooled PBS, and the nuclei were counterstained with DAPI (1 µg/mL) for 5 min. All procedures were performed on ice to minimize nonspecific enzymatic activity. Fluorescence images were captured via a confocal microscope (CSIM-130, Sunny Technology, China), with JC-1 aggregates (red fluorescence) and monomers (green fluorescence) visualized to assess mitochondrial damage.
Additionally, the depletion of intracellular GSH by PEITC was evaluated via ThiolTracker™ Violet. After treatment with Ru-PC-PEITC-ALG and NIR irradiation, the 4T1 cells were stained with 20 µmol/L ThiolTracker™ Violet at room temperature for 30 min in the dark. Fluorescence images were acquired via confocal microscopy, and semiquantitative analysis of GSH levels was performed via measurement of fluorescence intensity via ImageJ software. The reduced ThiolTracker™ Violet signal (green fluorescence) indicated GSH consumption, reflecting PEITC-mediated redox modulation.
Transcriptomic and metabolomic profiling of Ru-PC-PEITC-ALG mechanisms
To elucidate the antitumor mechanism of Ru-PC-PEITC-ALG, transcriptomic sequencing and metabolomic analyses were performed on 143b cells. Cells from the control and treatment groups (Ru-PC-PEITC-ALG treatment for 6 h followed by 808 nm NIR irradiation at 1.5 W/cm² for 4 min) were lysed via TRIzol reagent (Invitrogen, USA) for total RNA extraction. mRNA quantification, purification, reverse transcription, and sequencing were conducted by Shanghai Personal Biotechnology Co., Ltd. (Shanghai, China). Gene expression levels were normalized as fragments per kilobase of transcript per million mapped reads (FPKM). Differentially expressed genes (DEGs) were identified using a fold-change threshold of ≥ 2 and a false discovery rate (FDR) < 0.05. Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway and Gene Ontology (GO) enrichment analyses were performed to annotate functional pathways associated with the DEGs.
In vivo antitumor experiments
Animal experiments were performed according to the guidelines established by the Institutional Animal Care and Use Committee of Anhui Medical University (approved by LLSC20220731). The backs of 4-week-old BALB/c mice were shaved, and 1 × 106 4T1 cells were inoculated subcutaneously. After the tumor volume reached 80–100 cm3 after 7 days, the mice were randomly divided into 7 groups, each consisting of 5 individuals. The treatments used were as follows: (1) control; (2) ALG + laser; (3) Ru-PC-ALG; (4) PEITC-ALG; (5) Ru-PC-PEITC-ALG; (6) Ru-PC- ALG + laser; and (7) Ru-PC-PEITC-ALG + laser. All reagents were used at the following concentrations: ALG, 5 mg/mL; Ru-PC, 200 µg/mL; and PEITC, 20 µmol/L. Twelve hours after the injection, the tumor tissues in groups (2), (6), and (7) were treated with near-infrared (NIR) light at 808 nm NIR, 1.5 W/cm2 for 4 min. Tumor length and width and animal weight were measured every 2 days after treatment, and tumor volume was calculated as (tumor length) × (tumor width)2/2. At the end of the measurements on day 10, the mice were euthanized, and bluntly isolated tumor tissues were removed for imaging and fixation. The collected tumor tissues were subjected to H&E staining, immunohistochemical staining for Ki67 and Caspase 3, and immunofluorescence staining for TUNEL and ROS.
Biosafety assessment
Hemolysis Assay: BALB/c mice were sacrificed, and blood was collected from the eyeballs. Intact erythrocytes are centrifuged for hemolysis. PBS and different concentrations of NPs were added to the erythrocyte suspension, and the mixture was incubated for 6 h. The supernatant was collected after centrifugation, and the absorbance was measured at 577 nm via a UV‒visible spectrophotometer (Thermo Scientific Biomate 160, USA). NP-DSF-ALG solution was injected into the right lower abdomen of BALB/c nude mice (female, 6 weeks old, 18–20 g).
Hematological analysis: Mice were killed before injection and on days 4, 7 and 14 postinjection. Blood and biochemical parameters, including red blood cell (RBC) count, white blood cell (WBC) count, hemoglobin (HGB) level, hematocrit (HCT) level, neutrophil (NEU) count, lymphocyte (LYM) count, platelet (PLT) count, hemoglobin (HGB) level, hematocrit (HCT) level, and platelet (PLT) count, were also measured. The PLT, alanine aminotransferase (ALT), aspartate aminotransferase (AST), alkaline phosphatase (ALP), creatinine (CRE) and blood urea nitrogen (BUN) levels were also measured.
Evaluation of major organs: Mice were killed before injection and on days 4, 7 and 14 postinjection. Histological examination of major organs, including the heart, liver, spleen, lung and kidney, was performed via H&E staining.
Data processing
All the experiments were conducted at least twice or in triplicate. The findings presented in this report are representative. The mean and standard deviation (SD) were used to express quantitative data. Differences between groups were assessed via the standardized t test and were considered statistically significant when the p value was less than 0.05. NC: negative control; *, p < 0.05; **, p < 0.01; ***, p < 0.001.
Results
Characterization of Ru-PC
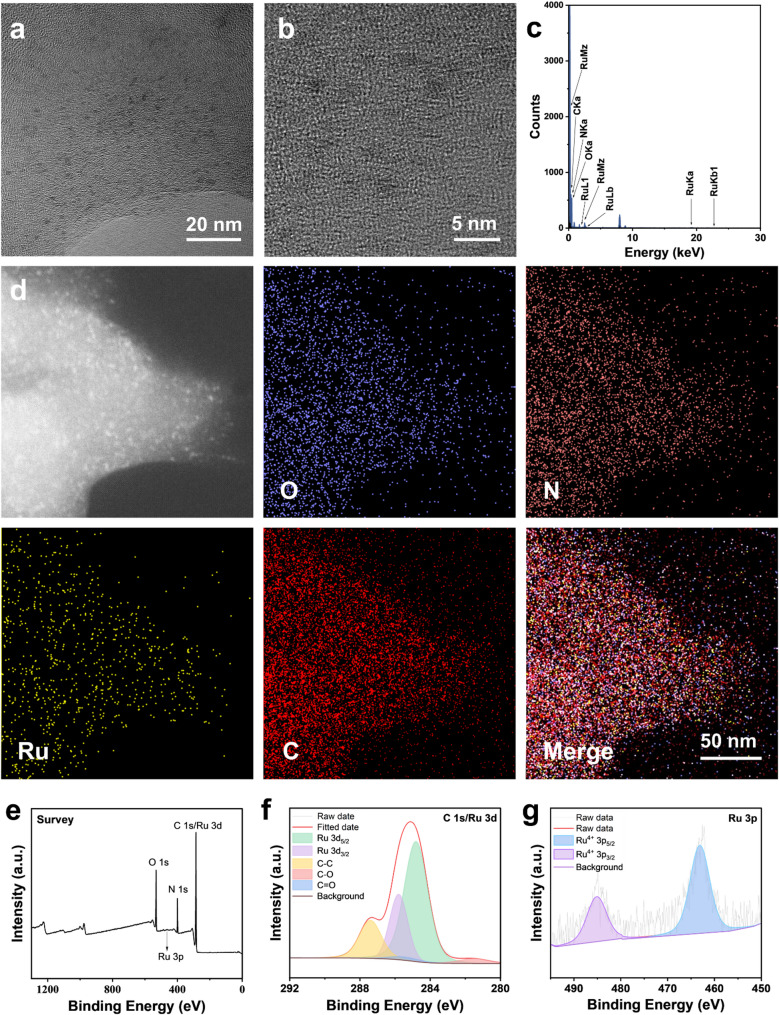
Ru-PC was prepared by mixing RuCl3, PC and PVP in a methanol solution with stirring. The solution after mixing gradually changed from yellowish brown to black and produced a precipitate, indicating the ligand binding of Ru ions to PC. As shown by the TEM images, Ru-PC formed nanoparticles during the encapsulation of PVP and had a smaller particle size of 2–5 nm (Fig. 1a-b), which was considered to have better delivery ability. EDS energy spectroscopy and elemental localization revealed that Ru-PC contained Ru, C, H, O, and N and was uniformly distributed inside the nanoparticles (Fig. 1c-d). In addition, DLS measurements were performed on Ru-PC. The results showed that the zeta potential of Ru-PC was − 17.75 eV, confirming its stability (Fig. S1). Furthermore, to explore the material composition of Ru-PC, XPS was performed. The results showed that Ru exists as Ru4+ within Ru-PC, and the presence of C-C, C-O, and C = O bonds was also confirmed in the energy spectrum of C 1 S (Fig. 1e-g). These results indicate that Ru-PC was successfully synthesized with high stability.
Fig. 1.
Characterization of Ru-PC. (a-b) High-resolution TEM images of Ru-PC. (c) EDS energy spectra of Ru-PC. (d) TEM elemental mapping images of Ru-PC. (e-g) Energy spectra of Ru-PC detected by XPS
Multienzymatic activity of Ru-PC and bioactivity of PEITC
The element Ru, as a transition metal, can be transformed between the valence states Ru(II), Ru(III) and Ru(IV) inside the cell, which results in multiple types of enzyme activities, such as POD and GPx (Fig. 2a). To test this theory, its POD-like enzyme activity was verified by probing. TMB can specifically bind to ROS to generate the compound ox-TMB, which has a distinctive absorbance peak. When different concentrations of Ru-PC are mixed with TMB, the absorbance at 652 nm is positively correlated with the concentration of Ru-PC, and at a fixed concentration of Ru-PC, the characteristic absorbance peaks of TMB also depend on the H2O2 concentration (Fig. 2b-c). Similarly, a validation was carried out using another probe, OPD, which showed that the absorbance values at 442 nm were dependent on the Ru-PC and H2O2 concentrations (Fig. 2d-e). These results demonstrated that Ru-PC possesses POD-like enzymatic activity, i.e., the ability to generate ROS via highly expressed H2O2 inside the tumor microenvironment.
Fig. 2.
Multiple enzyme activities of Ru-PC and the biological activity of PEITC. (a) Schematic diagram of multiple enzyme activity assays for Ru-PC and PEITC. (b-c) Changes in the TMB absorbance curves after treatment with different concentrations of Ru-PC or H2O2. (d-e) Changes in OPD absorbance curves after treatment with different concentrations of Ru-PC or H2O2. (f) Changes in the absorbance curves of DTNB with time after the reaction of 200 µg/mL Ru-PC with GSH. (g) Rate of GSH consumption by different concentrations of Ru-PC. (h) Changes in the color development of DNTB after the reaction of GSH with 1 µmol/L PEITC
The multivalent transformation of Ru ions has also been suggested to have a GPx enzyme-like ability to deplete GSH, a protective reducing molecule that is highly expressed in tumor cells and can react with strong oxidizing agents such as reactive oxygen species (ROS) for its cytotoxicity. In cells, Ru can deplete intracellular GSH through valence changes, thereby increasing intracellular oxidative stress. DTNB was employed to verify the depletion of GSH by Ru-PC, which was attributed to the fact that DTNB can bind to GSH and exhibit a characteristic absorption peak at 420 nm. The results revealed that the content of GSH gradually decreased with time in a solution with a concentration of 200 µg/mL Ru-PC and was completely depleted at approximately 5 h (Fig. 2f). On the other hand, experiments with different concentrations of Ru-PC verified that the rate of GSH depletion was positively correlated with the concentration of Ru-PC (Fig. 2g). These experiments confirmed that Ru-PC has dual POD-like and GPx-like enzyme activities, which demonstrated the excellent multienzyme activity of Ru-PC and its ability to increase the ROS level inside tumor tissues.
This finding was also validated considering that PEITC also has a unique ability to bind GSH. Compared with those of GSH, the characteristic peaks of DNTB disappeared significantly after mixing with 10 µmol/L PEITC, indicating that PEITC bound to GSH and thus lost its reducing properties (Fig. 2h).
Physicochemical properties of Ru-PC-PEITC-ALG
Ru-PC-PEITC-ALG was prepared by dissolving Ru-PC and PEITC in aqueous ALG solution with a sufficient mixture. In a previous report, we reported that a 5 mg/mL ALG solution had the best gel-forming and physicochemical properties inside the TME, so we still chose this concentration to configure the hydrogel in this study. According to the SEM images, the prepared Ru-PC-PEITC-ALG has a loose and porous structure, which means that it has a large surface area and can be loaded with more materials and drugs (Fig. 3a). The physical properties of Ru-PC-PEITC-ALG were examined via rotational rheology experiments. The results showed that the the rheological properties of Ru-PC-PEITC-ALG did not change dramatically compared with those of ALG, which proved that the addition of RU-PHS and PPI did not change the original properties of ALG (Fig. 3b-e). Among these, rheological viscosity and viscoelasticity demonstrated the excellent shaping ability of Ru-PC-PEITC-ALG after gelation, proving that it can exist in a stable form in the interstitial space of tumor tissue. The linear viscoelastic region (LVR) indicates that the structure can remain intact and undamaged within the stress range, while structural damage and recovery properties demonstrate that Ru-PC-PEITC-ALG has a certain degree of structural and character recovery ability. Furthermore, the molecular structure of Ru-PC-PEITC-ALG was explored via FT-IR. FT-IR patterns revealed that the characteristic peaks of ALG and Ru-PC were retained in Ru-PC-PEITC-ALG, which implied that the reactions between ALG and Ru-PC and PEITC did not occur and that structural integrity was maintained (Fig. S2). The characteristic peaks of PEITC could not be measured by FT-IR because of its volatile nature. The release of the loaded contents is also a key indicator of the properties of the hydrogel, which determines its effect after in situ gelation in tumor tissues. The release of Ru ions from Ru-PC and Ru-PC-PEITC-ALG in solutions simulating the TME environment (1.8 mmol/mL Ca2+, pH = 6.5) was measured by ICP at the same time. The results showed that approximately 40% of the Ru ions were released from Ru-PC-PEITC-ALG after 72 h, similar to Ru-PC alone, implying that Ru-PC-PEITC-ALG did not impede the diffusion of Ru-PC in the tissue (Fig. 3f). These results indicate that Ru-PC-PEITC-ALG has good loading capacity and could be used as an excellent nanocarrier platform for the precise delivery of Ru-PC and PEITC inside tumor tissues.
Fig. 3.
Characterization and photothermal properties of Ru-PC-PEITC-ALG. (a) SEM images of Ru-PC-PEITC-ALG. (b-e) Rheological properties, including rheological viscosity, viscoelasticity, linear viscoelastic region, structural damage and recovery properties, of Ru-PC-PEITC-ALG compared with those of ALG. (f) Release rates of Ru from Ru-PC and Ru-PC-PEITC-ALG over 72 h in a simulated TEM environment. (g-h) Infrared thermography images and (i-j) warming curves of Ru-PC-PEITC-ALG under 808 nm NIR irradiation at different concentrations and light intensities. (k) Stability curves of the PC-PEITC-ALG photothermal cycle
Photothermal properties of Ru-PC-PEITC-ALG
In view of the excellent photothermal properties of ruthenium-based compounds in general, the photothermal properties of Ru-PC-PEITC-ALG were measured. Ru-PC-PEITC-ALG was positively correlated with Ru-PC when it was irradiated with 808 nm near-infrared light at 1.5 W/cm2. The IR imager showed that Ru-PC-PEITC-ALG containing 200 µg/mL Ru-PC could be warmed from 25 °C to 55 °C in 4 min (Fig. 3g-i). Correspondingly, the final temperature of Ru-PC-PEITC-ALG at a fixed Ru-PC concentration (200 µg/mL) was dependent on the light intensity of the near-infrared light, which corresponded to the favorable photothermal properties of Ru-PC-PEITC-ALG (Fig. 3h-j). Thermal stability is also critical in photothermal therapy. The temperature profiles revealed no attenuation of the warming rate of Ru-PC-PEITC-ALG after three heating‒cooling cycles (Figs. 3k, S3). In addition, the UV absorption spectra of the hydrogel before and after receiving laser irradiation did not change significantly (Fig. S4). These experimental results indicate that Ru-PC-PEITC-ALG has good warming efficiency and photothermal stability and thus has good potential for photothermal therapy.
In vitro antitumor properties of Ru-PC-PEITC-ALG
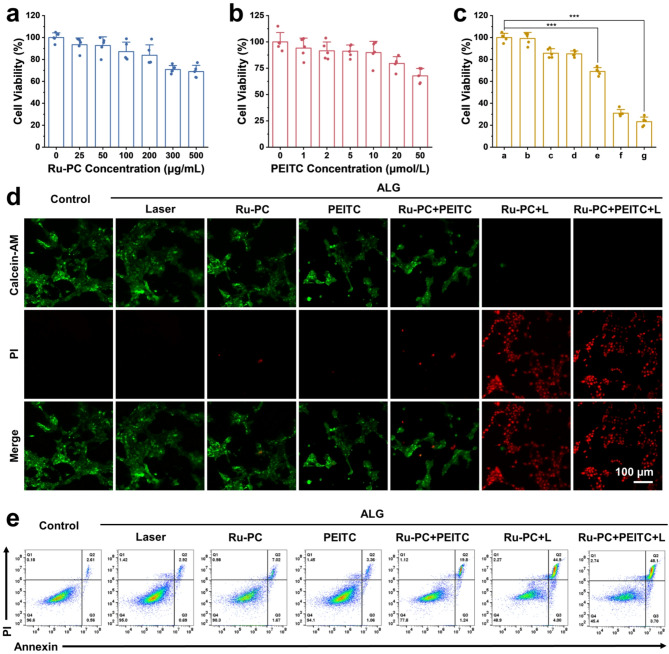
Encouraged by the Ru-PC-like enzyme activity and excellent physicochemical properties of Ru-PC-PEITC-ALG, Ru-PC-PEITC-ALG was introduced into in vitro cellular assays to further explore its antitumor properties. The toxicity of Ru-PC and PEITC on tumor cells and normal cells was evaluated via a CCK-8 assay. First, HUVECs, which are normal human tissue cells, had more than 80% viability at a Ru-PC concentration of less than 200 µg/mL in Ru-PC-ALG (Fig. S5a), whereas the corresponding concentration for PEITC was 20 µmol/L (Fig. S5b). Therefore, the above concentration was determined to be safe for subsequent experiments. In the CCK8 experiments with 4T1 cells, the viability of the corresponding concentrations was less than that of the corresponding concentrations of 4T1 cells, which indicated that both Ru-PC and PEITC had specific killing effects on 4T1 cells (Fig. 4a-b). By treating the cells in different ways, the introduction of 808 nm NIR illumination alone did not cause significant damage to the 4T1 cells, but the addition of Ru-PC reduced the cell viability to 30% of the original value, confirming the photothermal effect of Ru-PC. In addition, Ru-PC and PEITC significantly killed 4T1 cells, but the viability of 4T1 cells was reduced to 60% after the simultaneous addition of Ru-PC and PEITC. 4T1 viability was further reduced in the Ru-PC-PEITC-ALG + laser group compared with the Ru-PC-ALG + laser group. This evidence proved that Ru-PC and PEITC had a combined killing effect on 4T1 cells (Fig. 4c). To demonstrate the therapeutic effects of different subgroups more intuitively, the cells were fluorescently stained with a cell viability staining reagent (calcein-AM/PI), and the experimental results were essentially consistent with those of the CCK-8 assay (Fig. 4d), which confirmed the excellent antitumor performance of Ru-PC-PEITC-ALG under photothermal conditions (Fig. 4d).
Fig. 4.
In vitro antitumor ability of Ru-PC-PEITC-ALG. (a-b) CCK-8 assay for the independent inhibition of 4T1 cell activity by different concentrations of Ru-PC and PEITC. (c) CCK-8 assay for determining the activity of 4T1 cells in different groups. The groupings of a-g are as follows: control; ALG + laser; Ru-PC-ALG; PEITC-ALG; Ru-PC-PEITC-ALG; Ru-PC-ALG + laser; and Ru-PC-PEITC-ALG + laser. (d) CLSM images of different groups of live‒dead cells. (e) Flow cytometric analysis of different subgroups of apoptotic cells
To further explore the antitumor mechanism of Ru-PC-PEITC-ALG, different subgroups of cells were analyzed via Annexin/PI apoptosis staining and flow cytometry (Fig. 4e). The results showed that when only Ru-PC or PEITC was added, both could induce apoptosis in only 9.67% or 5.87% of 4T1 cells, which increased to 50% after the combination of the two, which also proved that there was a cascade-amplified crude apoptotic effect of Ru-PC and PEITC. Interestingly, the percentage of apoptosis in the Ru-PC-ALG and Ru-PC-PEITC-ALG groups also appeared to be elevated upon the addition of NIR illumination, which could be attributed to the fact that the photothermal effect enhances the reaction rate of POD-like enzymes of Ru-PC and therefore exacerbates the intracellular oxidative stress. Therefore, changes in the intracellular ROS levels were further observed via DCFH-DA staining (Fig. 5a). Fluorescence staining revealed that Ru-PC could produce ROS in tumor cells via POD-like enzyme activity, but the effect was weak, whereas PEITC was unable to induce ROS production in the cells. However, when both act together in 4T1 cells, the ROS level was significantly elevated, which was speculated to be related to the unique ability of PEITC to deplete protective GSH inside the cells. In addition, the introduction of NIR illumination also elevated ROS levels to some extent. The semiquantitative fluorescence statistics of the images also supported these conclusions (Fig. 5d).
Fig. 5.
Antitumor mechanism of Ru-PC-PEITC-ALG. (a) ROS fluorescence staining (DCFH-DA) CLSM image. (b) Mitochondrial membrane potential (JC-1) CLSM image. (c) CLSM image of intracellular GSH staining. (d) Semiquantitative analysis of ROS fluorescence staining and fluorescence. The groupings of a-g are as follows: control; ALG + laser; Ru-PC-ALG; Ru-PC-ALG + laser; PEITC-ALG; Ru-PC-PEITC-ALG; and Ru-PC-PEITC-ALG + laser. (e) Fluorescence semiquantitative analysis of intracellular GSH staining
Several reports have indicated that PEITC can hamper the normal energy metabolism of tumor cells by damaging their mitochondrial membranes [33, 34]. Therefore, JC-1 staining was performed to explore its effect on the mitochondrial membrane (Fig. 5b). After the addition of PEITC, the mitochondrial membrane potential significantly changed, suggesting that the mitochondrial membrane structure was damaged and that Ru-PC further amplified this effect. In addition, PEITC is believed to deplete intracellular GSH to increase oxidative stress-induced apoptosis in cells, which is a major factor in its ability to fight tumors. The results of intracellular GSH staining and semiquantitative fluorescence analysis revealed that PEITC effectively reduced the level of GSH in 4T1 cells, which was in agreement with previous findings (Fig. 5c, e). In addition, Ru-PC also depleted GSH to a certain extent because of its GPx-like enzyme activity. In summary, Ru-PC-PEITC-ALG can amplify intracellular oxidative stress through the joint action of Ru-PC and PEITC and combine with excellent photothermal properties to achieve precise and efficient killing of tumor cells in vitro.
In vitro cellular transcriptome sequencing
The excellent antitumor ability of Ru-PC-PEITC-ALG in in vitro cellular experiments inspired further exploration of its therapeutic mechanism (Fig. 6a). The differences in gene expression between the 4T1 cell control group and the Ru-PC-PEITC-ALG light treatment group were compared via transcriptome sequencing. Differential gene (DEG) analysis between the two groups was performed to obtain a gene heatmap (Fig. 6b), which revealed significant differences in gene expression between the two groups before and after treatment. The volcano plot of DEGs more intuitively revealed differences between the groups: relative to the control group, there were 3,592 upregulated genes and 3,111 downregulated genes in the treatment group (Fig. 6c). Among these, the downregulation of mitochondrial function-related genes (such as mt-Nd1, mt-Nd2, mt-Co1, and mt-Co2) is observed. mt-Nd1 and mt-Nd2 are involved in the first step of the electron transport chain (ETC), catalyzing the transfer of electrons from NADH to ubiquinone (UQ), driving proton pumping, and establishing the mitochondrial membrane potential. mt-Co1 and mt-Co2 are catalytic subunits of cytochrome c oxidase (complex IV), which transfers electrons from cytochrome c to oxygen. The downregulation of these gene expressions confirms the damaging effect of PEITC on the mitochondria of 4T1 cells [35]. On the other hand, the upregulation of the Ndrg gene may be related to heat stress caused by photothermal effects, while the upregulation of the Gzmm and Cldn4 genes indicates damage to the extracellular matrix induced by photothermal effects, which in turn affects normal cellular physiological activities [36]. On this basis, GSEA enrichment analysis was performed on DEGs, and significant differences in the enrichment pathways related to apoptosis and oxidative stress, such as the calcium signaling pathway and the PI3K-Akt signaling pathway, were found, which demonstrated that Ru-PC-PEITC-ALG could efficiently induce 4T1 cells to undergo apoptosis (Fig. 6d). In addition, the differences in enriched pathways, such as ECM-receptor interactions and cell adhesion molecules, indicated that the structure of the 4T1 cells was subjected to thermogenic damage [37].
Fig. 6.
Transcriptome sequencing. (a) Schematic diagram of the molecular mechanism of Ru-PC-PEITC-ALG intracellular antitumor therapy. (b) Heatmap of DEGs in the treatment and control groups. (c) Volcano plot of the DEGs. (d) GSEA enrichment analysis of DEGs. (e) Top 30 results of the KEGG enrichment analysis. (f) Top 30 terms from the GO enrichment analysis
Furthermore, the DEGs of the 4T1 cells before and after treatment were analyzed via Kyoto Encyclopedia of Genes and Genomes (KEGG) and Gene Ontology (GO) analyses. The results of the GO enrichment analysis revealed that after Ru-PC-PEITC-ALG light treatment, there was an enrichment of the entries of NADH/NAD(P)H; electron transfer activity, aerobic respiration and regulation of membrane potential were enriched in entries related to mitochondrial energy metabolism and redox, which represent the occurrence of mitochondrial damage and oxidative stress, whereas metal ion transmembrane transporter activity, on the other hand, may be associated with elevated intracellular Ru ions. In addition, a variety of entries related to the extracellular matrix (ECM) and membrane integrity were also enriched, which explains the cellular damage caused by the photothermal effect (Fig. 6e) [38, 39]. Similarly, KEGG enrichment revealed that pathways such as apoptosis, calcium signaling, PI3K-Akt and oxidative phosphorylation were activated and accompanied by ferroptosis-like pathways, whereas pathways such as ECM-receptor interaction, focal adhesion and mismatch repair represented the cellular damage caused by photothermal heat (Fig. 6f). Intriguingly, the expression of genes related to the heat shock protein family, which are theoretically produced during photothermal therapy, was not significantly upregulated, possibly because of two points: (1) The excellent photothermal properties of Ru-PC-PEITC-ALG allow rapid warming of the inside of the tumor tissues to kill the tumor cells, which prevents the production of heat shock proteins; [40] (2) PEITC has been reported to inhibit HSF1 phosphorylation by suppressing the inhibition of HSF1 phosphorylation to block the expression of heat shock proteins [41]. In view of the results of the transcriptome experiments, the mechanism of tumor killing by Ru-PC-PEITC-ALG through combined photothermal and chemodynamic therapy was demonstrated.
In vivo animal experiments
After summarizing the properties demonstrated by Ru-PC-PEITC-ALG in characterization experiments and in vitro experiments, the in vivo therapeutic effect was verified by constructing a subcutaneous 4T1 mammary cancer model in BALB/c mice. After successful construction of the hormonal mouse model, the corresponding drug mixture was injected in situ, and light treatment was performed 12 h later (Fig. 7a). The animals were continuously observed during the treatment period and sacrificed for dissection on day 10. There was no significant change in the body weight of the animals in each subgroup during the treatment (Fig. 7b). The digital photographs of the dissections revealed that neither light, Ru-PC, nor PEITC independently were able to produce effective tumor inhibition compared with that in the control group (Fig. 7c). However, this trend was partially alleviated by the combined application of Ru-PC and PEITC, and the best therapeutic effect was achieved by the reintroduction of NIR light. The summary statistics of tumor volume changes in each subgroup showed that tumor tissue ablation occurred immediately after phototherapy in the Ru-PC-ALG + laser group and the Ru-PC-PEITC-ALG + laser group. Furthermore, it was found that in the Ru-PC-ALG + laser group, a portion of the mice exhibited partial tumor volume rebound after day 7, whereas the Ru-PC-PEITC-ALG + laser group did not exhibit this phenomenon, and tumor volume continued to decrease. This suggests that PEITC enhances the therapeutic efficacy of Ru-PC (Fig. 7d-e). Furthermore, the exfoliated tumor tissues were subjected to fixed sectioning, and the internal physiological changes were explored via immunohistochemistry and immunofluorescence staining (Fig. 7f). H&E staining revealed that, compared with those in the control group, the tumor tissues in the Ru-PC-PEITC-ALG group presented a certain degree of apoptotic signs, and the tumor tissues in the two groups that underwent photothermal therapy presented obvious liquefaction and necrosis inside the tissues. Ki67 is a cell proliferation marker. Immunohistochemical staining revealed that the expression of Ki67 was significantly downregulated in both the PEITC-ALG group and the Ru-PC-PEITC-ALG group, and this trend was even more pronounced after the addition of light. Similarly, TUNEL fluorescence staining verified that Ru-PC-PEITC-ALG + laser could induce DNA damage. To explore the chemodynamic effect of Ru-PC-PEITC-ALG + laser, staining for ROS and Caspase-3 was used, and the results were consistent with those of previous cellular experiments showing that PEITC and photothermia could amplify the oxidative stress and apoptosis induced by Ru-PC. All of the above experimental results confirmed that Ru-PC-PEITC-ALG could achieve effective antitumor activity through a combined photothermal and chemodynamic strategy.
Fig. 7.
In vitro antitumor properties of Ru-PC-PEITC-ALG. (a) Schematic of the animal model construction and treatment. (b) Curve of body weight changes in each animal subgroup during treatment. (c) Digital photographs of isolated tumor tissues. (d-e) Tumor volume curves during treatment for each subgroup. (f) Micrographs of H&E, Ki67, TUNEL, ROS and Caspase 3 staining of tumor tissue sections from each subgroup
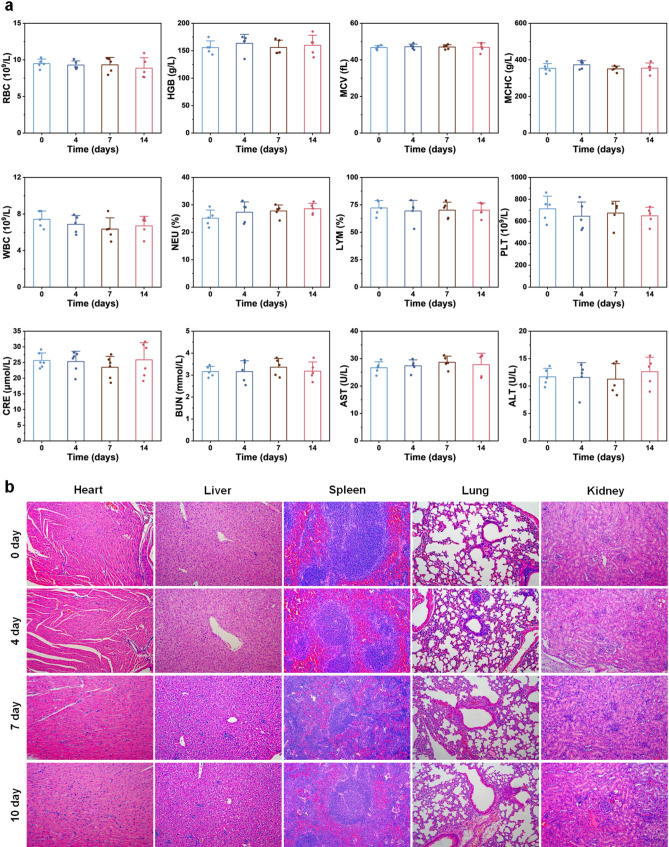
After the antitumor properties of Ru-PC-PEITC-ALG were verified, its biosafety was verified in several ways. First, the hemolysis assay revealed that this method was highly safe for erythrocytes, and no obvious hemolytic reaction was observed even when the concentration of the solution exceeded 400 µg/mL (Fig. S5). In addition, the hematological indices of the rats treated with Ru-PC-PEITC-ALG + laser were examined on days 0, 4, 7, and 14, and histological changes were observed in the sections of important organs (Fig. 8). The data revealed that after treatment, the blood parameters and blood biochemistry of the mice in each group did not change beyond the normal range, and important organs, such as the heart, liver, spleen, lung, and kidney, did not show any obvious accumulation or lesions. Therefore, Ru-PC-PEITC-ALG has excellent biological safety (Fig. 9).
Fig. 8.
Ru-PC-PEITC-ALG biosafety. (a) Changes in routine blood and blood biochemical indices in animals within 14 days after the injection of Ru-PC-PEITC-ALG. (b) H&E staining images of major organs within 14 days after Ru-PC-PEITC-ALG injection
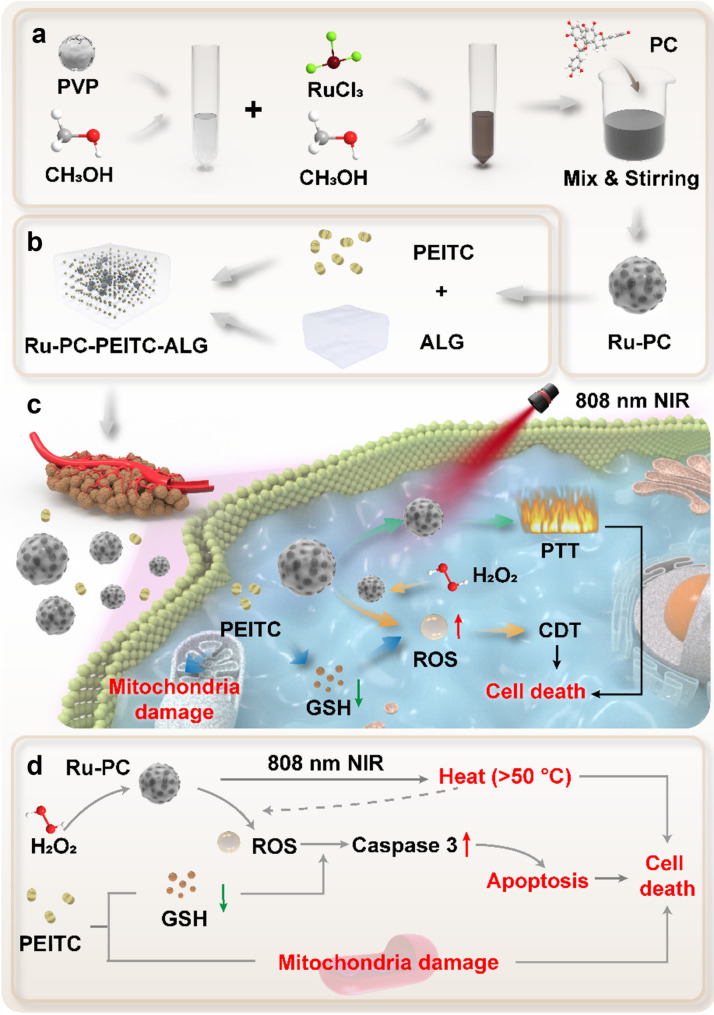
Fig. 9.
Schematic representation of the synthesis and antitumor mechanism of Ru-PC-PEITC-ALG. (a) Synthesis process of Ru-PC. (b) Synthesis flow of Ru-PC-PEITC-ALG. (c-d) Process and molecular biological mechanism of the intracellular antitumor effect of Ru-PC-PEITC-ALG
Conclusions
In conclusion, this study innovatively constructed ruthenium-procyanidin-liganded Ru-PC nanoparticles and coloaded them with PEITC in a Ca2+-crosslinked sodium alginate hydrogel to develop a light/microenvironmentally dual-responsive multimodal therapeutic platform, Ru-PC-PEITC-ALG. In the ALG hydrogel loading capacity achieved by Ru-PC and PEITC in tumor tissue after the precise delivery and release of Ru-PC and PEITC in tumor tissues, a large amount of ROS is generated through the multienzyme activity of Ru-PC, and the excellent photothermal performance of Ru-PC-PEITC-ALG achieves self-enhanced synergistic killing of tumor cells by CDT-PTT, which is mediated by 808 nm near-infrared light. In addition, the dual GSH depletion mediated by Ru-PC and PEITC lifted the tumor antioxidant defense and amplified the oxidative damage caused by Ru-PC. Compared with traditional photodynamic therapy combined with chemotherapy, this strategy can significantly inhibit tumor growth through the synergistic interaction between chemodynamics and photothermal effects. This mode of “precise delivery + disarming of antioxidant armor” for synergistic chemodynamic combined photothermal therapy provides a new paradigm for the design of metal–natural product hybrid systems.
Supplementary Information
Acknowledgements
The authors thank the Center for Scientific Research of Anhui Medical University for their valuable help with our experiments. The authors would like to thank the Shiyan laboratory (www.shiyanjia.com) for their help in language polishing.
Author contributions
L.Y. and Y.Q. conceived and designed the experiments. L.Y., Y.Q., P.W., W.L. and Y.Z. performed the experiments. L.Y., Y.Z., Z.Y. and X.W. discussed the results. L.Y. and Y.Z. wrote and revised the manuscript. All the authors discussed the results and commented on the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (No. 52202343, 82472933 and 82473406) and Heilongjiang Provincial Natural Science Foundation (Grant Number PL2024H180).
Data availability
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
The animal studies have been approved by the Ethics Committee of Anhui Medical University (No. LLSC20220731) and all handling of mice was performed in accordance with the institutional regulations.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Yang Qiao and Peisan Wang contributed equally to this work.
Change history
9/2/2025
The original online version of this article has been revised”: Affiliation has been updated.
Contributor Information
Xianwen Wang, Email: xianwenwang@ahmu.edu.cn.
Yiqun Zhang, Email: YiqunZhang0614@163.com.
Zhennan Yuan, Email: yuanzhennan123@hrbmu.edu.cn.
References
- 1.Lu X, Sun W, Zheng X, Yang L, Feng T, Deng X, et al. Ruthenium-doped carbon dots with three-in-one chemodynamic, photodynamic, and photothermal activity induce panoptosis for tumor therapy. Chem Eng J. 2025;509: 161355. [Google Scholar]
- 2.Chen W, Feng H, Mo Y, Pan Z, Ji S, Liang H, et al. Hyaluronic acid-functionalized ruthenium photothermal nanoenzyme for enhancing osteosarcoma chemotherapy: cascade targeting and bidirectional modulation of drug resistance. Carbohydr Polym. 2025;349: 122945. [DOI] [PubMed] [Google Scholar]
- 3.Liang JH, Zheng Y, Wu XW, Tan CP, Ji LN, Mao ZW. A tailored multifunctional anticancer nanodelivery system for Ruthenium-Based photosensitizers: tumor microenvironment adaption and remodeling. Adv Sci (Weinh). 2020;7(1):1901992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wang MF, Yang R, Tang SJ, Deng YA, Li GK, Zhang D et al. In vivo realization of dual photodynamic and photothermal therapy for melanoma by mitochondria targeting dinuclear ruthenium complexes under civil infrared Low-power laser. Angewandte Chemie-International Ed. 2022;61(38):e202208721. [DOI] [PubMed]
- 5.Xu G, Li C, Chi C, Wu L, Sun Y, Zhao J, et al. A supramolecular photosensitizer derived from an arene-Ru(II) complex self-assembly for NIR activated photodynamic and photothermal therapy. Nat Commun. 2022;13(1):3064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Meng C, Li S, Ma Y, Yu H, Song J, Zhi J, et al. Assembling ruthenium complexes to form ruthenosome unleashing Ferritinophagy-Mediated tumor suppression. ACS Nano. 2025;19(10):10207–19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lu Y, Zhu D, Gui L, Li Y, Wang W, Liu J, et al. A dual-targeting ruthenium nanodrug that inhibits primary tumor growth and lung metastasis via the PARP/ATM pathway. J Nanobiotechnol. 2021;19(1):115. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Małecka M, Skoczyńska A, Goodman DM, Hartinger CG, Budzisz E. Biological properties of ruthenium(II)/(III) complexes with flavonoids as ligands. Coord Chem Rev. 2021;436: 213849. [Google Scholar]
- 9.Qi X, Jha SK, Jha NK, Dewanjee S, Dey A, Deka R, et al. Antioxidants in brain tumors: current therapeutic significance and future prospects. Mol Cancer. 2022;21(1):204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nathan CF, Arrick BA, Murray HW, DeSantis NM, Cohn ZA. Tumor cell anti-oxidant defenses. Inhibition of the glutathione redox cycle enhances macrophage-mediated cytolysis. J Exp Med. 1981;153(4):766–82. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Lammers T. Nanomedicine tumor targeting. Adv Mater. 2024;36(26): e2312169. [DOI] [PubMed] [Google Scholar]
- 12.Tie S, Su W, Chen Y, Wu S, Wu H, Song Y, et al. Dual targeting procyanidin nanoparticles with glutathione response for colitis treatment. Chem Eng J. 2022;441: 136095. [Google Scholar]
- 13.Bystrom LM, Bezerra DP, Hsu HT, Zong H, Lara-Martínez LA, De Leon JP, et al. Cranberry A-type proanthocyanidins selectively target acute myeloid leukemia cells. Blood Adv. 2019;3(21):3261–5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Liu G, Shi A, Wang N, Li M, He X, Yin C, et al. Polyphenolic Proanthocyanidin-B2 suppresses proliferation of liver cancer cells and hepatocellular carcinogenesis through directly binding and inhibiting AKT activity. Redox Biol. 2020;37:101701. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lv J-l, Tan Y-j, Ren Y-s, Ma R, Wang X, Wang S-y, et al. Procyanidin C1 inhibits tumor growth and metastasis in colon cancer via modulating miR-501-3p/HIGD1A axis. J Adv Res. 2024;60:215–31. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhang Y, Weh K, Howard C, Lagisetty K, McEwen D, Lin J, et al. Proanthocyanidins enhance chemotherapy-induced esophageal adenocarcinoma cell death. Cancer Res. 2021;81(13Supplement):1418. [Google Scholar]
- 17.Taparia SS, Khanna A. Procyanidin-rich extract of natural cocoa powder causes ROS-mediated caspase-3 dependent apoptosis and reduction of pro-MMP-2 in epithelial ovarian carcinoma cell lines. Biomed Pharmacother. 2016;83:130–40. [DOI] [PubMed] [Google Scholar]
- 18.Zhai WY, Jia CP, Zhao H, Xu YS. Procyanidins inhibit tumor angiogenesis by crosslinking extracellular matrix. Chin J Cancer Res. 2011;23(2):99–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Shan J, Jin X, Zhang C, Huang MC, Xing JH, Li QR, et al. Metal natural product complex Ru-procyanidins with quadruple enzymatic activity combat infections from drug-resistant bacteria. Acta Pharm Sinica B. 2024;14(5):2298–316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wang H, Yang Y, Yu H, Ma L, Qi X, Qu J et al. Self-Cascade API nanozyme for synergistic Anti-Inflammatory, antioxidant, and ferroptosis modulation in the treatment of corneal neovascularization. Small. 2025;21(17): e2407751. [DOI] [PubMed]
- 21.Dong SM, Dong YS, Jia T, Liu SK, Liu J, Yang D, et al. GSH-depleted nanozymes with hyperthermia-enhanced dual enzyme-mimic activities for tumor nanocatalytic therapy. Adv Mater. 2020;32(42):e2002439. 10.1002/adma.202002439. [DOI] [PubMed] [Google Scholar]
- 22.Xiong YX, Xiao C, Li ZF, Yang XL. Engineering nanomedicine for glutathione depletion-augmented cancer therapy. Chem Soc Rev. 2021;50(10):6013–41. [DOI] [PubMed] [Google Scholar]
- 23.Cramer SL, Saha A, Liu J, Tadi S, Tiziani S, Yan W, et al. Systemic depletion of l-cyst(e)ine with cyst(e)inase increases reactive oxygen species and suppresses tumor growth. Nat Med. 2017;23(1):120–7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Niu B, Liao K, Zhou Y, Wen T, Quan G, Pan X, et al. Application of glutathione depletion in cancer therapy: enhanced ROS-based therapy, ferroptosis, and chemotherapy. Biomaterials. 2021;277:121110. [DOI] [PubMed] [Google Scholar]
- 25.Hu H, Chen JT, Yang H, Huang XT, Wu HL, Wu YX, et al. Potentiating photodynamic therapy of ICG-loaded nanoparticles by depleting GSH with PEITC. Nanoscale. 2019;11(13):6384–93. [DOI] [PubMed] [Google Scholar]
- 26.Trachootham D, Zhang H, Zhang W, Feng L, Du M, Zhou Y, et al. Effective elimination of fludarabine-resistant CLL cells by PEITC through a redox-mediated mechanism. Blood. 2008;112(5):1912–22. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Ju HQ, Lu YX, Chen DL, Tian T, Mo HY, Wei XL, et al. Redox regulation of stem-like cells though the CD44v-xCT axis in colorectal cancer: mechanisms and therapeutic implications. Theranostics. 2016;6(8):1160–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Aggarwal M, Saxena R, Sinclair E, Fu Y, Jacobs A, Dyba M, et al. Reactivation of mutant p53 by a dietary-related compound phenethyl isothiocyanate inhibits tumor growth. Cell Death Differ. 2016;23(10):1615–27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Chikara S, Nagaprashantha LD, Singhal J, Horne D, Awasthi S, Singhal SS. Oxidative stress and dietary phytochemicals: role in cancer chemoprevention and treatment. Cancer Lett. 2018;413:122–34. [DOI] [PubMed] [Google Scholar]
- 30.Jakubikova J, Sedlak J, Klippel S, Leiba M, Tai Y-T, Hideshima T, et al. Sulforaphane and PEITC augment activity of conventional and novel Anti-Myeloma drugs. Blood. 2008;112(11):916. [Google Scholar]
- 31.Xing J, Yang Y, Zhang W, Yan J, Qian H, Hao J, et al. Injectable hydrogel containing TiO nanosheets for synergistic photothermal/thermodynamic therapy. ACS Appl Mater Interfaces. 2023;15(29):34436–50. [DOI] [PubMed] [Google Scholar]
- 32.Zhang Y, Zhang N, Xing J, Sun Y, Jin X, Shen C, et al. In situ hydrogel based on Cu–Fe. Nanoclusters Exploits Oxidative Stress Ferroptosis/cuproptosis Pathw Chemodynamic Therapy Biomaterials. 2024;311:122675. [DOI] [PubMed] [Google Scholar]
- 33.Zhen C, Li J, Liu J, Lyu Y, Xie L, Lv H. Phenethyl isothiocyanate induces oxidative cell death in osteosarcoma cells with regulation on mitochondrial network, function and metabolism. Biochimica et Biophysica Acta (BBA). 2023;1869(6): 166740. [DOI] [PubMed] [Google Scholar]
- 34.Xiao D, Singh SV. p66 < SUP > Shc Is indispensable for phenethyl Isothiocyanate-Induced apoptosis in human prostate cancer cells. Cancer Res. 2010;70(8):3150–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Martins WK, Santos NF, Rocha CS, Bacellar IOL, Tsubone TM, Viotto AC, et al. Parallel damage in mitochondria and lysosomes is an efficient way to photoinduce cell death. Autophagy. 2019;15(2):259–79. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Richman TR, Spåhr H, Ermer JA, Davies SM, Viola HM, Bates KA, et al. Loss of the RNA-binding protein TACO1 causes late-onset mitochondrial dysfunction in mice. Nat Commun. 2016;7: 11884. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Tan T, Hu H, Wang H, Li J, Wang Z, Wang J, et al. Bioinspired lipoproteins-mediated photothermia remodels tumor stroma to improve cancer cell accessibility of second nanoparticles. Nat Commun. 2019;10(1):3322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Chen P-L, Huang P-Y, Chen J-Y, Shi Q-Y, Zhu Y-Y, Chen Y, et al. A self-delivery chimeric peptide for high efficient cell membrane-targeting low-temperature photothermal/photodynamic combinational therapy and metastasis suppression of tumor. Biomaterials. 2022;286:121593. [DOI] [PubMed] [Google Scholar]
- 39.Liu X, Wang C, Ma H, Yu F, Hu F, Yuan H. Water-Responsive hybrid nanoparticles codelivering ICG and DOX effectively treat breast cancer via Hyperthermia-aided DOX functionality and drug penetration. Adv Healthc Mater. 2019;8(8):e1801486. [DOI] [PubMed] [Google Scholar]
- 40.Sauvage F, Messaoudi S, Fattal E, Barratt G, Vergnaud-Gauduchon J. Heat shock proteins and cancer: how can nanomedicine be harnessed? J Controlled Release. 2017;248:133–43. [DOI] [PubMed] [Google Scholar]
- 41.Franceschelli S, Bruno AP, Festa M, Falco A, Gionti E, d’Avenia M, et al. BAG3 protein is involved in endothelial cell response to phenethyl isothiocyanate. Oxid Med Cell Longev. 2018;2018:5967890. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
No datasets were generated or analysed during the current study.