Abstract
The brood-parasitic brown-headed cowbird (Molothrus ater) has one of the shortest incubation periods of any bird. Brown-headed cowbird eggs, and those of other avian brood parasites, tend to be more spherical due to their greater relative width. The traditional explanation for this egg shape is that it, combined with the thicker eggshells, resists host puncture-ejection. However, very few North American hosts of the brown-headed cowbird actually engage in puncture-ejection and therefore wider eggs may instead provide greater contact with a host’s brood patch during incubation, especially in large host nests. We tested whether greater egg width increased mean temperature and reduced temperature variation in brown-headed cowbirds by inserting temperature probes into brown-headed cowbird and house sparrow (Passer domesticus) eggs and placing them into red-winged blackbird (Agelaius phoeniceus) nests. House sparrow eggs are similar in appearance and in length to cowbird eggs, but are not as wide. We found no significant relationship between brown-headed cowbird egg width and mean incubation temperature. However, brown-headed cowbird eggs experienced less temperature variation than house sparrow eggs, and within brown-headed cowbird eggs, more spherical eggs experienced less temperature variation when accounting for differences in width. These results suggest that brown-headed cowbirds may have short incubation periods in part because their eggs exhibit less temperature variation over the course of incubation. The brown-headed cowbird’s egg shape may contribute to its accelerated embryonic development rate relative to host eggs of similar size, which explains its ability to hatch in a variety of host nests.
Keywords: brood parasitism, brown-headed cowbird, development, egg shape, incubation, Molothrus ater, temperature, thermoregulation
Obligate avian brood parasites such as the brown-headed cowbird (Molothrus ater) lay their eggs in the nests of other birds, thereby relying on these hosts to incubate the eggs and care for their offspring. Brown-headed cowbirds are host generalists having parasitized 248 host species and their young have fledged from the nests of > 170 species (Lowther 2024). As a result, their eggs and developing embryos are exposed to a multitude of host nest environments that vary in microclimate, clutch size, egg size, incubation period, and incubation routines. Their embryos must not only withstand this wide range of conditions, but excel in them. One of the most important adaptations possessed by parasitic cowbirds (Molothrus spp) is their short incubation period that allows them to hatch sooner than their hosts providing them with a developmental head-start outside the confines of the egg (Briskie and Sealy 1990).
The mechanism by which cowbirds hatch sooner than their hosts is unclear and several hypotheses have been proposed. Shiny cowbird (M. bonariensis) eggs have lower energy content, which may force them to hatch sooner with relatively small young (Kattan 1995; but see Cao et al. 2018). Brown-headed cowbird embryos develop faster relative to hosts, the presence of their eggs in a nest prolongs the incubation period of host eggs, and their embryos may also be stimulated by the activity of host embryos to hatch sooner (McMaster and Sealy 1998). Brown-headed cowbird eggshells are also more porous than hosts’ eggs, which accelerates their development relative to hosts, including the closely related red-winged blackbird (Agelaius phoeniceus; Jaeckle et al. 2012). This suggests that increased porosity has evolved as an adaptation for brood parasitism rather than being a trait common to all blackbirds (Jaeckle et al. 2012; see also Hargitai et al. 2010; Portugal et al. 2014). Finally, female brown-headed cowbirds remove host eggs in part to ensure efficient incubation of their own eggs (Peer and Bollinger 2000) and this is particularly important in the nests of larger host species (Peer and Bollinger 1997, 2000).
In this study, we examined the effect of egg shape on incubation temperatures because brown-headed cowbirds, similar to other brood parasites, have relatively wide eggs that give them a rounded shape (Picman 1989; Davies 2000; Peer and Bollinger 2000; Sealy et al. 2002) and a more recent analysis showed that brown-headed cowbird eggs are more symmetrical than those of their hosts (Lopez et al. 2021). Spherical shape, in addition to the thick eggshells of brood parasite eggs, may enhance their structural strength (Picman 1989). The traditional explanation for this combination of traits is that it decreases the likelihood of parasite eggs being punctured and removed by hosts (Picman 1989; Picman and Pribil 1997; Igic et al. 2011; Jaeckle et al. 2012; Antonov et al. 2013). However, Lopez et al. (2018) astutely pointed out that very few cowbird hosts in the New World actually puncture-eject cowbird eggs. Indeed, only a single North American host is known to be deterred by the thick-shelled egg of the brown-headed cowbird during egg ejection attempts (Peer et al. 2018). Instead, Lopez et al. (2018) found that the greater strength of brown-headed and shiny cowbird eggs prevents damage to cowbird eggs when they are laid onto host eggs and they also damage host eggs during the laying process (see also Ellison et al. 2019), and it also benefits shiny and screaming cowbird eggs (M. rufoaxillaris) by resisting damage in multiply parasitized nests (Lopez et al. 2021). This spherical shape might secondarily benefit brown-headed cowbird eggs by increasing contact with the host’s brood patch (Peer and Bollinger 1997, 2000) and by increasing heat retention, especially when hosts are not incubating the egg (Lack 1968; Drent 1975; Barta and Székely 1997; Johnsgard 1997). While there are exceptions (Boersma 1982), maintaining a relatively constant temperature is critical for the development of most avian embryos (Deeming 2002). If a brown-headed cowbird egg acquires heat more effectively from the host’s brood patch and is able to retain it, this could hasten the rate of embryonic development relative to hosts and provide the cowbird with an advantage by hatching sooner.
We investigated whether the spherical shape of brown-headed cowbird eggs increased heat acquisition and resulted in less temperature variation compared to the eggs of the non-parasitic house sparrow (Passer domesticus) when placed in red-winged blackbird nests. Because brown-headed cowbird eggs are wider, and more spherical relative to house sparrow eggs, we predicted that cowbird eggs should maintain higher and more constant temperatures compared to sparrow eggs.
Materials and Methods
Data collection
The study was conducted outdoors in McDonough County, Illinois (40°25ʹN, −90°36ʹW and 40°26ʹN, −90°40ʹW) and Scott County, Iowa (41°40ʹN, −90°27ʹW) from April through July of 2008 and 2009. We recorded egg temperatures of either a single brown-headed cowbird egg or house sparrow egg inside a red-winged blackbird nest. In addition to examining whether variation in egg width within brown-headed cowbirds played a role in heat acquisition and temperature variation, house sparrow eggs were specifically chosen because they resemble the appearance of cowbird eggs (Figure 1A,B) and are similar in length to cowbird eggs, but cowbird eggs are wider than sparrow eggs (21.45 × 16.42 mm, 21.64 × 15.55 mm, respectively; Lowther 2020; Lowther and Cink 2020; see below). This similarity in length allowed us to ascertain the importance of the brown-headed cowbird’s greater relative egg width, while the similar appearance was key because differently colored eggs can have varying thermal properties (Wisocki et al. 2020).
Figure 1.
Photographs showing (A) a clutch of house sparrow eggs and (B) a parasitized red-winged blackbird nest containing 3 host eggs and 1 cowbird egg, which demonstrate the similar spotting patterns between cowbird and sparrow eggs.
In addition to using naturally parasitized red-winged blackbird nests, brown-headed cowbird eggs were collected from other host nests and from cowbirds in outdoor aviaries. Brown-headed cowbird and house sparrow egg lengths and widths were measured with digital calipers. Small, precise holes, approximately 3–5 mm, were created in the sides of eggs using a Dremel® Multipro model 275 without causing further observable damage to the eggshells. Thermal Micro Control temperature probes were inserted inside the egg and sealed with quick-drying Gorilla® Super Glue. Probes were connected to HOBO® XT Temperature Loggers (Onset Computer Corp., Bourne, Massachusetts). Before being placed into red-winged blackbird nests, HOBOs were started using the Onset program (BoxCar Pro, Onset Computer Corp., Bourne, Massachusetts). Each temperature logger recorded the egg’s internal temperature to the nearest ± 0.7 °C every 2.5 min for 3 days and all data recorded during this interval were included in the analysis. The insertion of the temperature probes prevented the eggs from being turned by the red-winged blackbirds, and nests were occasionally deserted after the birds broke the eggs while apparently attempting to rotate them (n < 5). These nests were excluded from the analysis and because this occurred so infrequently, it was unlikely to have affected the results.
Red-winged blackbird nests were monitored daily until they contained three eggs, at which time one fresh “parasitic” egg was added. Parasitic eggs were assigned randomly to a nest except in cases in which a brown-headed cowbird egg was already present in a nest. If a host laid an additional egg, the egg was removed to maintain the clutch size at three. The most common parasitized clutch size at our sites was three red-winged blackbird eggs plus one brown-headed cowbird egg (unpublished data). Eggs with temperature probes were placed into the nest and the cord was connected to data loggers through the bottom of the nest where they were sealed in a watertight bag. Temperatures were recorded at the same stage of incubation for all nests (after the third egg was laid) and nests were checked daily until the data loggers were removed.
Data analysis
Sixteen a priori models were chosen to assess the effects of different combinations of eggshell dimensions on incubation properties (temperature mean and variation) as well as model stability. The 16 a priori models were developed and duplicated in 2 separate model sets: 1 set for temperature variation (SE (standard error) of temperatures recorded during the 3-day period) and 1 set for mean temperature. Both model sets investigated the effects of species, egg width, egg shape (width divided by length; Biggins et al. 2018; Nagy et al. 2019), egg volume (0.51 × length × width2; Nagy et al., 2019), and their additive and interactive effects on either dependent variable. Egg shape varies between 0 and 1, and the more spherical the egg is, the closer the shape approaches 1. All model sets (temperature variation a priori models and mean temperature a priori models) were adjusted for average daily ambient temperature range gathered from the National Weather Service’s NOAA Online Weather Data located at the nearest weather stations in Monmouth, IL and Moline, IL (www.weather.gov). The temperature variation a priori models were adjusted with the egg's mean temperature. Each model set included a global model (Models 16 and 32; Table 1) and a base model incorporating average daily ambient temperature range (Models 4 and 17; Table 1). Interactive models were included in order to compare differences in egg shape, volume, and width within species (Models 2, 5, 9, 12, 16, 25, 27, 29, 31, and 32; Table 1). To further evaluate significant interaction terms within species, the data were analyzed by stratifying the models for each species resulting in 4 separate model sets consisting of 6 a priori models each: 1 set for brown-headed cowbird temperature variation, 1 set for house sparrow temperature variation, 1 set for brown-headed cowbird mean temperature, and 1 set for house sparrow mean temperature (Table 2).
Table 1.
A priori models for temperature variation (Models 1–16) and mean temperature (Models 17–32) of brown-headed cowbird and house sparrow eggs. The number of parameters (K), sample size (N Obs), natural log likelihood (LNL), Akaike information criteria adjusted for small sample size (AICc), change in AICc from the smallest AICc value (∆ AICc), Akaike weight (wi), VIF, and model significance (Sig) are displayed for each model. The variables species, width, and shape relate to brown-headed cowbird versus house sparrow eggs, width of the eggs (millimeters), and egg shape, respectively
| K | N | LNL | AICc | ∆AICc | wi | VIF | Sig | |
|---|---|---|---|---|---|---|---|---|
| 1: Species | 4 | 41 | 139.112 | 147.760 | 0.000 | 0.231 | 1.01 | 0.106 |
| 2: Species × Shape | 6 | 41 | 135.512 | 149.226 | 1.466 | 0.111 | 2.03 | 0.168 |
| 3: Species + Width | 5 | 41 | 138.171 | 149.282 | 1.522 | 0.108 | 1.32 | 0.195 |
| 4: Null | 3 | 41 | 143.248 | 149.564 | 1.804 | 0.094 | NA | NA |
| 5: Species × (Shape + Width) | 8 | 41 | 130.424 | 149.818 | 2.058 | 0.083 | 2.87 | 0.120 |
| 6: Species + Volume | 5 | 41 | 138.763 | 149.874 | 2.113 | 0.080 | 1.40 | 0.242 |
| 7: Species + Shape | 5 | 41 | 139.111 | 150.222 | 2.462 | 0.068 | 1.28 | 0.279 |
| 8: Shape | 4 | 41 | 142.624 | 151.273 | 3.512 | 0.040 | 1.02 | 0.577 |
| 9: Species × Volume | 6 | 41 | 137.735 | 151.449 | 3.688 | 0.037 | 2.11 | 0.317 |
| 10: Volume | 4 | 41 | 142.955 | 151.603 | 3.843 | 0.034 | 1.06 | 0.702 |
| 11: Species + Shape + Width | 6 | 41 | 137.905 | 151.619 | 3.859 | 0.034 | 1.71 | 0.338 |
| 12: Species × Width | 6 | 41 | 137.951 | 151.665 | 3.904 | 0.033 | 2.03 | 0.347 |
| 13: Width | 4 | 41 | 143.218 | 151.867 | 4.106 | 0.030 | 1.08 | 0.903 |
| 14: Species + Shape + Width + Volume | 7 | 41 | 137.592 | 154.063 | 6.302 | 0.010 | 1,359.33 | 0.476 |
| 15: Shape + Width + Volume | 6 | 41 | 141.894 | 155.608 | 7.848 | 0.005 | 1,358.10 | 0.883 |
| 16: Species × (Shape + Width + Volume) | 10 | 41 | 129.844 | 155.651 | 7.890 | 0.004 | 3,792.83 | 0.268 |
| 17: Null | 3 | 41 | 197.885 | 204.201 | 0.000 | 0.360 | NA | NA |
| 18: Shape | 4 | 41 | 197.743 | 206.392 | 2.191 | 0.120 | 1.02 | 0.716 |
| 19: Volume | 4 | 41 | 197.846 | 206.495 | 2.294 | 0.114 | 1.06 | 0.849 |
| 20: Species | 4 | 41 | 197.881 | 206.529 | 2.328 | 0.112 | 1.01 | 0.948 |
| 21: Width | 4 | 41 | 197.885 | 206.534 | 2.333 | 0.112 | 1.08 | 0.999 |
| 22: Species + Shape | 5 | 41 | 197.671 | 208.782 | 4.581 | 0.036 | 1.28 | 0.907 |
| 23: Species + Volume | 5 | 41 | 197.810 | 208.921 | 4.720 | 0.034 | 1.40 | 0.967 |
| 24: Species + Width | 5 | 41 | 197.880 | 208.991 | 4.790 | 0.033 | 1.32 | 0.998 |
| 25: Species × Shape | 6 | 41 | 196.039 | 209.754 | 5.553 | 0.022 | 2.03 | 0.646 |
| 26: Volume + Shape + Width | 6 | 41 | 196.581 | 210.295 | 6.094 | 0.017 | 1,358.10 | 0.762 |
| 27: Species × Volume | 6 | 41 | 197.410 | 211.124 | 6.923 | 0.011 | 2.11 | 0.936 |
| 28: Species + Shape + Width | 6 | 41 | 197.624 | 211.339 | 7.138 | 0.010 | 1.71 | 0.973 |
| 29: Species × Width | 6 | 41 | 197.763 | 211.477 | 7.276 | 0.009 | 2.03 | 0.991 |
| 30: Species + Volume + Shape + Width | 7 | 41 | 196.518 | 212.989 | 8.788 | 0.004 | 1,359.33 | 0.880 |
| 31: Species × (Shape + Width) | 8 | 41 | 194.790 | 214.184 | 9.983 | 0.002 | 2.87 | 0.751 |
| 32: Species × (Volume + Shape + Width) | 10 | 41 | 192.134 | 217.940 | 13.739 | 0.000 | 3,792.83 | 0.682 |
Table 2.
A priori models for temperature variation (Models 1–12) and mean temperature (Models 13–24) stratified by brown-headed cowbird (CB; Models 1–6 and 13–18) and house sparrow eggs (HS; Models 7–12 and 19–24). The number of parameters (K), sample size (N Obs), natural log likelihood (LNL), Akaike information criteria adjusted for small sample size (AICc), change in AICc from the smallest AICc value (∆ AICc), Akaike weight (wi), VIF, and model significance (Sig) are displayed for each model. The variables species, width, and shape relate to brown-headed cowbird versus house sparrow eggs, width of the eggs (millimeters), and egg shape, respectively
| K | N | LNL | AICc | ∆AICc | wi | VIF | Sig | |
|---|---|---|---|---|---|---|---|---|
| 1 Null | 3 | 26 | 70.839 | 77.361 | 5.947 | 0.037 | NA | NA |
| 2 Width | 4 | 26 | 67.777 | 76.868 | 5.453 | 0.048 | 1.087 | 0.118 |
| 3 Shape | 4 | 26 | 69.385 | 78.476 | 7.061 | 0.021 | 1.000 | 0.334 |
| 4 Volume | 4 | 26 | 70.574 | 79.665 | 8.251 | 0.012 | 1.032 | 0.671 |
| 5 Width + Shape | 5 | 26 | 59.510 | 71.414 | 0.000 | 0.727 | 1.629 | 0.004 |
| 6 Width + Shape + Volume | 6 | 26 | 59.510 | 74.510 | 3.095 | 0.155 | 1,012.400 | 0.015 |
| 7 Null | 3 | 15 | 61.609 | 68.609 | 0.000 | 0.516 | NA | NA |
| 8 Width | 4 | 15 | 61.330 | 71.512 | 2.903 | 0.121 | 1.023 | 0.719 |
| 9 Shape | 4 | 15 | 60.458 | 70.639 | 2.031 | 0.187 | 1.115 | 0.439 |
| 10 Volume | 4 | 15 | 61.190 | 71.372 | 2.763 | 0.130 | 1.111 | 0.654 |
| 11 Width + Shape | 5 | 15 | 59.643 | 73.643 | 5.034 | 0.042 | 1.201 | 0.601 |
| 12 Width + Shape + Volume | 6 | 15 | 59.168 | 77.834 | 9.226 | 0.005 | 1,994.735 | 0.750 |
| 13 Null | 3 | 26 | 125.036 | 131.558 | 0.000 | 0.401 | NA | NA |
| 14 Width | 4 | 26 | 125.008 | 134.098 | 2.541 | 0.113 | 1.087 | 0.874 |
| 15 Shape | 4 | 26 | 123.772 | 132.863 | 1.305 | 0.209 | 1.000 | 0.284 |
| 16 Volume | 4 | 26 | 124.805 | 133.896 | 2.338 | 0.125 | 1.032 | 0.650 |
| 17 Width + Shape | 5 | 26 | 122.719 | 134.624 | 3.066 | 0.087 | 1.629 | 0.359 |
| 18 Width + Shape + Volume | 6 | 26 | 120.179 | 135.179 | 3.621 | 0.066 | 1,012.400 | 0.230 |
| 19 Null | 3 | 15 | 72.552 | 79.552 | 0.000 | 0.576 | NA | NA |
| 20 Width | 4 | 15 | 72.386 | 82.568 | 3.016 | 0.128 | 1.023 | 0.715 |
| 21 Shape | 4 | 15 | 72.145 | 82.327 | 2.775 | 0.144 | 1.115 | 0.566 |
| 22 Volume | 4 | 15 | 72.451 | 82.633 | 3.081 | 0.123 | 1.111 | 0.776 |
| 23 Width + Shape | 5 | 15 | 71.773 | 85.773 | 6.222 | 0.026 | 1.201 | 0.746 |
| 24 Width + Shape + Volume | 6 | 15 | 71.436 | 90.103 | 10.551 | 0.003 | 1,994.735 | 0.856 |
R 4.3.1 (R Core Team 2023) was used to fit gamma regression models with a log link function for temperature variation and linear regression models for mean temperature. Due to the variability of the house sparrow data with heavy tails (Figure 2), they were described with a gamma model instead of a linear model. Egg width, volume, and shape were standardized to prevent inflated standard errors. The variance inflation factor (VIF) was computed to assess multicollinearity among the independent variables. A VIF of 10 or more indicates multicollinearity. Models were evaluated with an information theoretic approach using Akaike’s information criterion corrected for small sample size to determine model fit (AICc; Simonoff 2003). Models with the lowest ∆ AICc were considered to best explain the data and models within 2 ∆ AICc units of the lowest model were considered as best competing models. Significance of the models and the predictor terms within the models were measured at an α of 0.05. Brown-headed cowbird and house sparrow eggshell dimensions were compared using 2-tailed independent t-tests.
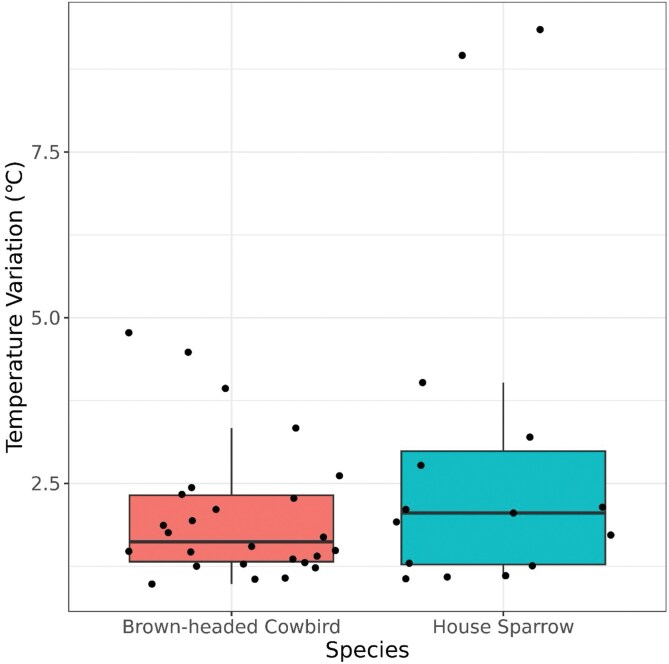
Figure 2.
Two box plots with a jitter plot overlayed displaying the central tendency and variability of temperature variation in brown-headed cowbird eggs (n = 26) and house sparrow eggs (n = 15). The plot indicates that cowbirds have a lower variability in temperature variation compared to sparrows.
Results
Brown-headed cowbird eggs were similar in length to house sparrow eggs (21.04 mm ± 0.2 [n = 26], 21.31 mm ± 0.3 [n = 15], respectively; t-test, t = −0.73, df = 38, P = 0.47), but significantly wider than sparrow eggs (16.24 mm ± 0.2, 15.48 mm ± 0.1, respectively; t-test, t = 2.94, df = 38, P = 0.006). Brown-headed cowbird eggs also had significantly larger volumes compared to house sparrow eggs (6.40 ± 0.6 [n = 26], 5.75 ± 0.4 [n = 15], respectively; t-test, t = 4.04, df = 38, P = 0.0002). Red-winged blackbird egg measurements average 24.7 × 17.8 mm (Yasukawa and Searcy 2020). The average brown-headed cowbird egg temperature variation (2.02 °C ± 0.20 SE) was less than that of the house sparrow eggs (2.94 °C ± 0.69 SE; Figure 2). Mean brown-headed cowbird egg temperature was 32.98 °C ± 0.60 SE and the mean house sparrow egg temperature was 32.79 °C ± 0.74 SE.
In the first model set, Model 1 (species) best explained egg temperature variation (AICc = 147.76, Table 1). However, this model was not significant (P = 0.106) nor was the predictor within the model (β = 0.417, P = 0.12). Model 2 (species × shape) and Model 3 (species + width) produced ∆ AICc values ≤ 2 units from Model 1, but neither was significant (P = 0.168, P = 0.195, respectively). Model 5 (species × [shape + width]) contained a significant interaction term between species and shape (β = 0.763, P = 0.02), while Model 2 (species × shape) did not have the same interaction effect (β = 0.514, P = 0.09) because width is necessary to demonstrate the effect between species and shape. In other words, Model 2 did not account for width, which prevented a significant interaction effect between species and shape as indicated by Model 5. These results indicate that both shape and width have varying effects on temperature variation based on species (see below for further details). Models 14, 15, and 16 showed high multicollinearity between volume, shape, and width that resulted in unreliable inference caused by volume being included in the models.
In the stratified models, Model 5 (shape + width) best explained temperature variation for brown-headed cowbird eggs (AICc = 71.41; Table 2) and the predictors shape and width were both significant (β = −0.35, P = 0.019; β = 0.28, P = 0.016, respectively). In contrast, Model 7 (null) was the best model to explain temperature variation in the house sparrow eggs (AICc = 68.61; Table 2). The predictors within Model 5 indicated that a larger shape decreases temperature variation, while larger width increases temperature variation in brown-headed cowbird eggs. For example, if there are 2 brown-headed cowbird eggs whose width/length is 14.25/19 for egg 1 and 15.75/21 for egg 2, their shape would be 0.75. However, egg 2 would have more temperature variation because of its larger width. On the other hand, if egg 1 is 15/20 and egg 2 is 15/17.6, then egg 1 will have a shape of 0.75 and egg 2 a shape of 0.85. They both will have equal width, but egg 2 will have less temperature variation because it is more spherical (Table 3).
Table 3.
An illustration of the effects of shape and width in Model 5 using 2 sets of brown-headed cowbird eggs. Maintaining a constant egg shape (eggs A.1 and A.2) or width (eggs B.1 and B.2) yields changes in temperature variation (by a factor of 0.42 and −0.035, respectively) in cowbird eggs. Eggs with differing widths can have the same shape due to differences in length (eggs A.1 and A.2). Therefore, width only has an effect on temperature variation. Additionally, eggs with different shapes can have the same width (eggs B.1 and B.2), and as a result, shape only affects temperature variation
| Egg | Length | Width | Shape | Effects of egg shape on temperature variation between eggs | Effects of egg width on temperature variation between eggs |
|---|---|---|---|---|---|
| A.1 | 19 | 14.25 | 0.75 | −0.35 × 0 = 0 | 0.28 × 1.5 = 0.42 |
| A.2 | 21 | 15.75 | 0.75 | ||
| B.1 | 20 | 15 | 0.75 | −0.35 × 0.1 = −0.035 | 0.28 × 0 = 0 |
| B.2 | 17.6 | 15 | 0.85 |
In the second model set comparing mean egg temperature, Model 17 (null) was the best (AICc = 204.201, Table 1). The significant interaction term between species and shape found in the first model set for egg temperature variation was not observed for mean temperature (Model 31; β = −1.85, P = 0.136). Models 26, 30, and 32 showed multicollinearity issues between shape, volume, and width caused by volume being included in the models. In the stratified model tests for mean temperature (Table 2), the null models (Models 13 and 19) were the best models in both species. All the remaining models and predictors within the models were not significant.
Discussion
Brown-headed cowbird eggs experienced less temperature variation compared to house sparrow eggs. Our stratified models (Table 2), which accounted for differences in ambient temperature range, revealed that cowbird egg temperature variation was explained by differences in shape and width. The predictors within Model 5 also indicated that larger cowbird egg shape explained lower temperature variation and increased width explained larger temperature variation. The small surface-area-to-volume ratio of spherical eggs possessed by brown-headed cowbirds and other brood parasites has been proposed to be the optimal shape for promoting heat retention (Lack 1968; Wyllie 1981; Johnsgard 1997; see also Bán et al. 2011), and our study is the first to provide experimental evidence in support of this hypothesis. The best strategy for brood parasites may not be to increase mean incubation temperature because there could be a risk of overheating, but rather to reduce the temperature variation the embryo endures. Minimizing variation in incubation temperature would allow the brown-headed cowbird embryo to remain closer to the optimum temperature, thus yielding a shorter incubation period. In brown-headed cowbirds and other brood parasites, hatching earlier than hosts is critical especially in the nests of large hosts because it allows them to start being fed and to grow before the host nestlings emerge (Briskie and Sealy 1990; Peer and Bollinger 1997, 2000). There was, however, no relationship between brown-headed cowbird egg width and mean incubation temperature, suggesting that the greater width of cowbird eggs does not increase their contact with the brood patch of female red-winged blackbirds to enhance heat acquisition although future studies should measure the amount of contact eggs have with a host’s brood patch.
Stoddard et al. (2017) analyzed egg shape in 1,400 species of birds although they only included one brood parasite, the pallid cuckoo (Cuculus pallidus). They found that greater flight efficiency has selected for more elliptical eggs, while some flightless species such as the Ratites have more spherical eggs. Stoddard et al. (2017) also found that life history traits can influence egg shape at smaller taxonomic levels. In cowbirds, the brood parasitic lifestyle may favor less elliptical eggs that enhance heat retention, withstand the impact of host eggs when female cowbirds lay from an elevated perch (Lopez et al. 2018; Ellison et al. 2019), and protect against damage when multiple cowbirds routinely lay in a single nest (Lopez et al. 2021).
Theoretically, the heat-retention capabilities of brown-headed cowbird eggs we document here could also promote ambient incubation and embryonic development in the absence of host incubation (Griffith et al. 2016). Ambient incubation could allow brown-headed cowbirds to hatch even sooner depending on when they are laid relative to host eggs. Brown-headed cowbird eggs are typically laid early in the host’s laying period and often before the host has laid any eggs (Sealy 1995). If the brown-headed cowbird egg shape promotes ambient incubation, this would hasten their development compared to the hosts.
Our findings that brown-headed cowbird eggs demonstrate less temperature variation are significant because they are extreme host generalists. Brown-headed cowbirds have parasitized at least 248 species (Lowther 2024) and their eggs are exposed to a wide range of host nest environments compared to birds that care for their young and even specialist brood parasites that use a limited number of host species. Given that fluctuations in temperature can be expected across and within species due to various factors, safeguards against temperature variation may be vital for generalist brood parasites. Eggs with superior heat-retention properties would allow the embryo to develop in the wide variety of nests in which cowbirds lay. This is reflected in the results of Peer and Bollinger (2000) who added brown-headed cowbird and house sparrow eggs into the nests of a large host, the common grackle (Quiscalus quiscula). The incubation period of brown-headed cowbird eggs was within the range of what has been reported in other host species (12.1 days; Peer and Bollinger 2000). However, the incubation period of house sparrow eggs (13.5 days) was similar to that of common grackle eggs (13.2 days) and was significantly longer than the typical house sparrow incubation period (11 days; Lowther and Cink 2020). The findings of Peer and Bollinger (2000) and our study indicate that brown-headed cowbird eggs are adapted for incubation in the nests of other species, whereas house sparrow eggs apparently do not possess such adaptations.
Finally, the thicker eggshells typical of brown-headed cowbirds may further reduce temperature variation by acting as an insulator (see also Wyllie 1981) and cowbird eggshells (0.125 mm; Picman 1989) are thicker than house sparrow eggshells (0.102 mm; Ar et al. 1979). Similar to our results that brown-headed cowbird eggs had less temperature variation, Yang et al. (2018) found that parasitic common cuckoo (Cuculus canorus) eggs had lower temperature variation than host eggs as well as higher temperatures and attributed it to the thicker eggshells of the cuckoo. They did not, however, examine the effects of egg shape on temperature and indicated that this aspect required further study. The findings of Yang et al. (2018) suggest that there might be differences in the thermal properties of brown-headed cowbird and common cuckoo eggs although the studies are not directly comparable because different methods were used. Yang et al. (2018) measured temperatures in an incubator in the lab with a thermal imager and recorded 8 temperature readings over 4 separate days, whereas our study was conducted in the field in nests tended by birds, and our temperatures were recorded every 2.5 min for 3 days.
In summary, our results that decreased temperature variation in brown-headed cowbird eggs is related to their shape when accounting for width may provide cowbird embryos with the ability to develop rapidly in a variety of nest microclimates in which this extreme host generalist lays its eggs. Future studies are needed to elucidate the relative contributions of egg shape, width, and shell thickness in minimizing temperature variation, in addition to whether cowbird eggs experience ambient incubation. Importantly, experiments on the incubation properties of the eggs of closely related icterids should be conducted to determine whether decreased temperature variation is a trait evolved by brown-headed cowbirds for parasitism or whether it is simply a trait common to blackbirds in general (Mermoz and Ornelas 2004).
Acknowledgements
Kent McClure allowed us access to his property and David Westmoreland graciously allowed us to borrow his temperature loggers. We thank the editor, Dr. Zu-Shi Huang, along with Kevin Ellison, Nicholas Horrocks, Brandon Whitton, and the anonymous reviewers for constructive comments on the manuscript. Rick Anderson, Bob McCleery, Mike Romano, Eivin Røskaft, and Spencer Sealy provided comments to improve an earlier draft of the manuscript.
Contributor Information
Francisco R Magdaleno, Department of Psychology, California State University, Channel Islands, Camarillo, One University Dr., Camarillo, CA 93012, USA.
Lyndon R Hawkins, Department of Biological Sciences, Western Illinois University, 1 University Circle, Macomb, IL 61455, USA.
Isaac Quintanilla Salinas, Department of Mathematics, California State University, Channel Islands, One University Dr., Camarillo, CA 93012, USA.
Brian D Peer, Department of Biological Sciences, Western Illinois University, 1 University Circle, Macomb, IL 61455, USA.
Funding
This study was funded by a grant from Western Illinois University.
Conflict of Interest
The authors declare no conflicts of interest.
Authors’ Contributions
L.R.H and B.D.P. designed and conducted the study. B.D.P., L.R.H., F.R.M., and I.Q.S. analyzed the data, and B.D.P., L.R.H., F.R.M., and I.Q.S. wrote the manuscript.
Ethical Statement
All applicable guidelines for the care and use of animals were followed. The study was approved by the WIU IACUC (#09-42) and the U.S. Fish and Wildlife Service (#MB122718).
References
- Antonov A, Stokke BG, Fossøy F, Liang W, Moksnes A. et al. , 2013. Why do brood parasitic birds lay strong-shelled eggs? Chinese Birds 3:245–258. [Google Scholar]
- Ar A, Rahn H, Paganelli CV, 1979. The avian egg: Mass and strength. Condor 81:331–337. [Google Scholar]
- Bán M, Barta Z, Muñoz AR, Takasu F, Nakamura H. et al. , 2011. The analysis of common cuckoo’s egg shape in relation to its hosts’ in two geographically distant areas. J Zool 284:77–83. [Google Scholar]
- Barta Z, Székely T, 1997. The optimal shape of avian eggs. Funct Ecol 11:656–662. [Google Scholar]
- Biggins JD, Thompson JE, Birkhead TR, 2018. Accurately quantifying the shape of birds’ eggs. Ecol Evol 8:9728–9738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boersma PD, 1982. Why some birds take so long to hatch. Am Nat 120:733–750. [Google Scholar]
- Briskie JV, Sealy SG, 1990. Evolution of short incubation periods in the parasitic cowbirds, Molothrus spp. Auk 107:789–794. [Google Scholar]
- Cao P, Sun BJ, Wang LW, Liang W, Du WG, 2018. Proximate mechanisms of earlier hatching in parasitic cuckoos: Yolk energy and embryonic metabolism. Biol J Linn Soc 123:63–71. [Google Scholar]
- Davies NB, 2000. Cuckoos, Cowbirds and Other Cheats. London (UK): T & AD Poyser. [Google Scholar]
- Deeming DC, 2002. Avian Incubation Behaviour, Environment, and Evolution. New York (NY): Oxford University Press. [Google Scholar]
- Drent RH, 1975. Incubation. Avian Biology, vol. 5. New York (NY): Academic Press. [Google Scholar]
- Ellison KS, Fiorini VD, Gloag R, Sealy SG, 2019. Video recordings of brown-headed Molothrus ater) and shiny (M. bonariensis) cowbirds reveal oviposition from an elevated position: implications for host-parasite coevolution. Wilson J Ornithol 131:789–795. [Google Scholar]
- Griffith SC, Mainwaring MC, Sorato E, Beckmann C, 2016. High atmospheric temperatures and ’ambient incubation’ drive embryonic development and lead to earlier hatching in a passerine bird. R Soc Open Sci 3:150371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hargitai R, Moskát C, Bán M, Gil D, López-Rull I. et al. , 2010. Eggshell characteristics and yolk composition in the common cuckoo Cuculus canorus: Are they adapted to brood parasitism? J Avian Biol 41:177–185. [Google Scholar]
- Igic B, Braganza K, Hyland MM, Silyn-Roberts H, Cassey P. et al. , 2011. Alternative mechanisms of increased eggshell hardness of avian brood parasites relative to host species. J R Soc Interface 8:1654–1664. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaeckle WB, Kiefer M, Childs B, Harper RG, Rivers JW. et al. , 2012. Comparison of eggshell porosity and estimated gas flux between the brown-headed cowbird and two common hosts. J Avian Biol 43:486–490. [Google Scholar]
- Johnsgard PA, 1997. The Avian Brood Parasites. Deception at the Nest. New York (NY): Oxford University Press. [Google Scholar]
- Kattan GH, 1995. Mechanisms of short incubation period in the brood-parasitic cowbirds. Auk 112:335–342. [Google Scholar]
- Lack D, 1968. Ecological Adaptations for Breeding in Birds. London, England: Methuen & Co. [Google Scholar]
- Lopez AV, Bolmaro RE, Avalos M, Gerschenson LN, Reboreda JC. et al. , 2021. How to build a puncture- and breakage-resistant eggshell? Mechanical and structural analyses of avian brood parasites and their hosts. J Exp Biol 224:1–20. [DOI] [PubMed] [Google Scholar]
- Lopez AV, Fiorini VD, Ellison K, Peer BD, 2018. Thick eggshells of brood parasitic cowbirds protect their eggs and damage host eggs during laying. Behav Ecol 29:965–973. [Google Scholar]
- Lowther PE, 2020. Brown-headed cowbird (Molothrus ater), version 1.0. In: Poole AF, Gill FB, editors. Birds of the World. Ithaca (NY): Cornell Lab of Ornithology. [Google Scholar]
- Lowther PE, 2024. Lists of victims and hosts of the parasitic cowbirds (Molothrus). Available from: https://www.datocms-assets.com/44232/1715101068-host-list-molothrus-ver10apr2024.pdf (Accessed 20 July 2024). [Google Scholar]
- Lowther PE, Cink CL, 2020. House sparrow (Passer domesticus), version 1.0. In: Billerman SM, editor. Birds of the World. Ithaca (NY): Cornell Lab of Ornithology. [Google Scholar]
- McMaster DG, Sealy SG, 1998. Short incubation periods of brown-headed cowbirds: How do cowbird eggs hatch before yellow warbler eggs? Condor 100:102–111. [Google Scholar]
- Mermoz ME, Ornelas JF, 2004. Phylogenetic analysis of life-history adaptations in parasitic cowbirds. Behav Ecol 15:109–119. [Google Scholar]
- Nagy J, Hauber ME, Hartley IR, Mainwaring MC, 2019. Correlated evolution of nest and egg characteristics in birds. Anim Behav 158:211–225. [Google Scholar]
- Peer BD, Bollinger EK, 1997. Explanations for the infrequent cowbird parasitism on common grackles. Condor 99:151–161. [Google Scholar]
- Peer BD, Bollinger EK, 2000. Why do female brown-headed cowbirds remove host eggs? A test of the incubation efficiency hypothesis. In: Smith JNM, Cook TL, Rothstein SI, Robinson SK, Sealy SG, editors. Ecology and Management of Cowbirds and Their Hosts. Austin (TX): University of Texas Press, 187–192. [Google Scholar]
- Peer BD, McCleery RA, Jensen WE, 2018. Resistance is futile: prohibitive costs of egg ejection in an obligate avian brood parasite host. Anim Behav 144:45–51. [Google Scholar]
- Picman J, 1989. Mechanism of increased puncture resistance of eggs of brown-headed cowbird. Auk 106:577–583. [Google Scholar]
- Picman J, Pribil S, 1997. Is greater eggshell density an alternative mechanism by which parasitic cuckoos increase the strength of their eggs? J Ornithol 138:531–541. [Google Scholar]
- Portugal SJ, Hauber ME, Maurer G, Stokke BG, Grim T. et al. , 2014. Rapid development of brood‐parasitic cuckoo embryos cannot be explained by increased gas exchange through the eggshell. J Zool 293:219–226. [Google Scholar]
- R Core Team. 2023. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. [Google Scholar]
- Sealy SG, 1995. Burial of cowbird eggs by parasitized yellow warblers: An empirical and experimental study. Anim Behav 49:877–889. [Google Scholar]
- Sealy SG, McMaster DG, Peer BD, 2002. Tactics of obligate brood parasites to secure suitable incubators. In: Deeming DC, editor. Avian Incubation: Behaviour, Environment, and Evolution. New York (NY): Oxford University Press, 254–269. [Google Scholar]
- Simonoff JS, 2003. Analyzing Categorical Data. New York (NY): Springer. [Google Scholar]
- Stoddard MC, Yong EH, Akkaynak D, Sheard C, Tobias JA. et al. , 2017. Avian egg shape: form, function, and evolution. Science 356:1249–1254. [DOI] [PubMed] [Google Scholar]
- Wisocki PA, Kennelly P, Rivera IR, Cassey P, Burkey ML. et al. , 2020. The global distribution of avian eggshell colours suggest a thermoregulatory benefit of darker pigmentation. Nat Ecol Evol 4:148–155. [DOI] [PubMed] [Google Scholar]
- Wyllie I, 1981. The Cuckoo. New York (NY): Universe Books. [Google Scholar]
- Yang C, Huang Q, Wang L, Du WG, Liang W. et al. , 2018. Keeping eggs warm: thermal and developmental advantages for parasitic cuckoos of laying unusually thick-shelled eggs. Sci Nat 105(1-2):10. [DOI] [PubMed] [Google Scholar]
- Yasukawa K, Searcy WA, 2020. Red-winged blackbird (Agelaius phoeniceus), version 1.0. In: Rodewald PG, editor. Birds of the World. Ithaca (NY): Cornell Lab of Ornithology. [Google Scholar]