Abstract
Background
It is unknown whether there is an association between initial serum potassium level and short-term survival in out-of-hospital cardiac arrest (OHCA) survivors. The aim of this study was to describe potential associations between first recorded potassium level and 30-day survival in patients surviving OHCA.
Methods
We identified 4,894 patients who had return of spontaneous circulation (ROSC) at hospital arrival, and a registered post-OHCA serum-potassium value, using Danish nationwide registry data from 2001–2019. Potassium values were divided into seven predefined levels: < 2.5, 2.5–2.9, 3.0–3.4, 3.5–4.6, 4.7–5.5, 5.5–6.0, > 6.0 mmol/L. Thirty-day survival was estimated using a multivariable Cox regression (reference normokalemia 3.5–4.6 mmol/L). The multivariable model included age, sex, Charlson comorbidity index (including chronic kidney disease), witnessed status, performance of bystander cardiopulmonary resuscitation (CPR) and first registered heart rhythm.
Results
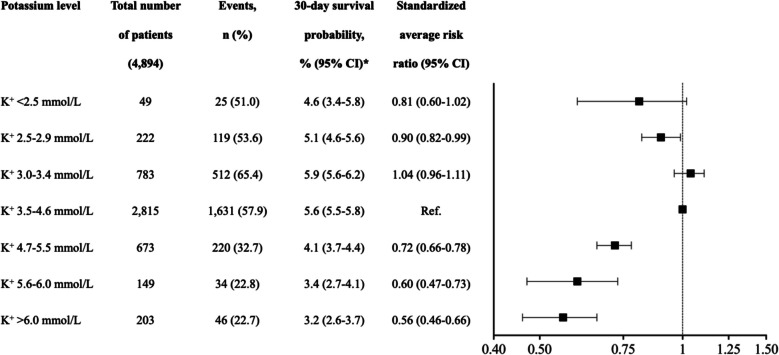
Over the 30-day follow-up period, survival rates in the seven strata were as follows: 25 (51.0%), 119 (53.6%), 512 (65.4%), 1,631 (57.9%), 220 (32.7%), 34 (22.8%), and 46 (22.7%), respectively. Thirty-day survival was significantly lower for all groups with hyperkalemia compared with normokalemia: 4.7–5.5 mmol/L: (average risk ratio (RR): 0.72, 95% confidence interval (95% CI): 0.66–0.78); 5.5–6.0 mmol/L: (average RR: 0.60, 95% CI: 0.47–0.73); > 6.0 mmol/L: (average RR: 0.56, 95% CI: 0.46–0.66). Survival did not differ significantly in patients with hypokalemia compared with normokalemia.
Conclusions
In OHCA survivors, hyperkalemia was associated with reduced 30-day survival compared with normokalemia, independent of age, sex, comorbidity burden and pre-hospital OHCA-characteristics. Conversely, hypokalemia was not associated with reduced 30-day survival.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13049-025-01454-8.
Keywords: Out-of-hospital cardiac arrest survivors, Potassium, Survival
Background
Globally, the yearly occurrence of out-of-hospital cardiac arrest (OHCA) treated by emergency medical services (EMS) is approximated to range from 30 to 97 individuals per 100,000 residents [1]. Following the return of spontaneous circulation (ROSC), over half of the patients remain in a comatose state upon arrival at the hospital, with a mortality rate exceeding 50% within this patient cohort [2, 3]. Prognostic biomarkers for these patients in the early stages have not been adequately established [4, 5]. Deviation of serum potassium levels below or above the normal range is associated with short-term mortality in individuals with acute cardiovascular conditions such as acute heart failure, acute myocardial infarction and other cardiac diseases [6–9]. Survivors of OHCA frequently present with initial serum potassium concentrations outside the normal range. Previous smaller studies on OHCA have identified a correlation between hyperkalemia and unfavorable survival outcomes, including neurological prognosis at discharge and 30-day post-event [10–12]. However, a large nationwide study with allowance to stratify into more potassium categories has not previously been performed. Therefore, the aim of this study was to explore potential associations between serum potassium levels and the probability of 30-day survival among OHCA patients with ROSC, in a large nationwide Danish population study.
Methods
Study setting
The study was conducted in Denmark between June 1, 2001, and December 31, 2019. Denmark holds a population of 5.8 million and covers ∼43,000 km.2. Mobile advanced life support emergency care units staffed with anesthesiologists, paramedics, or nurses and basic life support ambulances staffed with ambulance technicians are included in the emergency medical service. The mobile emergency care units are dispatched as rendezvous with basic life support ambulances. Patients were treated following the latest resuscitation guidelines during the study period [13–15].
Data sources
In Denmark, every resident possesses a distinct Civil Personal Registration Number, facilitating the integration of various nationwide administrative registries. The Danish Cardiac Arrest Registry encompasses all instances of OHCA where resuscitation efforts were initiated by either a bystander or EMS personnel [16]. Patients exhibiting late signs of death, wherein resuscitative measures are not commenced, are excluded from the study. Data are collected in accordance with Utstein-style guidelines [17, 18]. The categorization of presumed cause of arrest involved utilizing discharge diagnoses from hospitals and diagnosis codes from death certificates. Cardiac causes of arrest encompassed diagnosis codes indicative of cardiac disease, unexpected collapse, or unspecified illnesses. Non-cardiac causes encompassed events with diagnosis codes related to other medical disorders (i.e., excluding the aforementioned diagnoses), trauma, various accidents, violent attacks, attempted suicides, drowning incidents, and drug overdoses, irrespective of any accompanying diagnoses [19, 20].
Information regarding age and sex was obtained from the Danish Civil Personal Registry [21]. Data concerning potassium levels, renal function, pH, and lactate were extracted from the clinical laboratory information system (LABKA) [22, 23]. The first available values for potassium, pH, and lactate within 12 h following OHCA were recorded. Data concerning comorbidities, including the characterization of end-stage renal disease, were sourced from the Danish National Patient Registry. Diagnoses were documented following the International Classification of Diseases (ICD), with records ranging from the 8th revision (ICD-8) until 1994, and transitioning to the 10th revision (ICD-10) from 1994 onwards [24]. Charlson score17 was used. Scores range from 0 to 33, with higher scores indicating more coexisting comorbidities.
The Danish Registry of Causes of Death documents the causes of death, encompassing immediate, contributory, and underlying factors [25].
Study population and design
The study, conducted nationwide, was structured as a follow-up study utilizing registry data. We conducted retrospective identification of all individuals aged 18 years or older who experienced OHCA between 2001 and 2019 in Denmark. Specifically, we targeted patients who achieved ROSC upon arrival at the hospital and had recorded post-OHCA serum-potassium levels. Exclusion criteria comprised individuals declared dead prior to hospital arrival, those undergoing cardiopulmonary resuscitation (CPR) upon hospital arrival, or those lacking documented status upon hospital arrival.
Outcomes
The main study outcome was 30-day survival following OHCA. Patients were followed from occurrence of OHCA until death or censoring in case of emigration, end of follow-up, or end of study, whichever occurred first.
Statistical analysis
Categorical variables are depicted as counts and percentages, while continuous data are represented as medians with 25–75% percentiles. Ordinal data were treated as continuous. First recorded serum potassium levels were ordered into seven predefined potassium levels: < 2.5, 2.5–2.9, 3.0–3.4, 3.5–4.6, 4.7–5.5, 5.5–6.0, > 6.0 mmol/L. Thirty-day survival was analyzed according to the seven potassium levels. Survival probability for the different potassium groups was illustrated using Kaplan–Meier curves and compared using log-rank test. We used multivariable Cox regression models adjusted for age, sex, Charlson comorbidity index (which included chronic kidney disease), witnessed status, performance of bystander CPR, and the first registered heart rhythm. Furthermore, in patients with available lactate values, we performed a sensitivity analysis adding lactate to the multivariable Cox regression model. The association between potassium levels and 30-day survival was shown with standardized absolute risks and corresponding average risk ratios (RR) with 95% confidence intervals (CI) (normokalemia (3.5–4.6 mmol/L) serving as reference) [26]. The association of potassium with survival was also assessed using restricted cubic splines with knots at the 25th, 50th and 75th percentiles of potassium. Missing data was handled using multiple imputations. Analyses were performed based on 200 imputed data sets using the smcfcs package in R [27].
Statistical significance was established at a two-sided P-value < 0.05. Data management and analyses were conducted using SAS version 9.4 (Cary, NC, USA) and R version 4.4.1 [28].
Ethics
The study was approved by the Danish Data Protection Agency. Registry-based studies does not require ethical approval by The Danish National Committee on Health Research Ethics. For conducting the study, the use of the Danish Cardiac Arrest Registry and other national registries was approved by the Data Responsible Unit in the Capital Region of Denmark.
Results
Patients and characteristics
The study population selection flowchart is shown in Fig. 1. Of 68,500 OHCA patients registered between 2001 and 2019, 13,964 achieved ROSC and our analysis encompassed a cohort of 4,894 of those patients who had accessible post-OHCA serum potassium levels. None of the included patients had missing 30-day survival data. Over the 30-day follow-up period, survival rates within the seven strata were as follows: < 2.5: 253 (55.9%), 2.5–2.9: 107 (53.0%), 3.0–3.4: 462 (65.1%), 3.5–4.6: 1495 (57.7%), 4.7–5.5: 199 (32.3%), 5.5–6.0: 32 (22.5%), and > 6.0: 39 (21.6%) respectively. Baseline characteristics of patients with available post-OHCA potassium are shown in Table 1 and stratified according to potassium levels. Median time interval from OCHA recognition to potassium measurement was 75 min (IQR 56–105). A notable trend is the sharp decline in shockable rhythm proportion in patients with potassium > 4.7 mmol/L. Missing data are shown under Table 1. Characteristics of patients excluded from the study due to missing potassium blood samples, in comparison to those included, are detailed in Supplemental Table 1. Thirty-day and 1-year survival were both higher among patients with an available serum potassium concentration, i.e., 52.9% versus 45.5% and 47.7% versus 41.1%, respectively.
Fig. 1.
Selection of the study population. OHCA out-of-hospital cardiac arrest. ROSC return of spontaneous circulation
Table 1.
Patient characteristics stratification according to potassium levels
| K+ < 2.5 mmol/L n = 49 (1.0) |
K+ 2.5–2.9 mmol/L n = 222 (4.5) |
K+ 3.0–3.4 mmol/L n = 783 (16.0) |
K+ 3.5–4.6 mmol/L n = 2,815 (57.5) |
K+ 4.7–5.5 mmol/L n = 673 (13.8) |
K+ 5.6–6.0 mmol/L n = 149 (3.0) |
K+ > 6.0 mmol/L n = 203 (4.2) |
Total n = 4,894 (100.0) |
|
|---|---|---|---|---|---|---|---|---|
| Year of OHCA – n (%) | ||||||||
| ≤ 2010 | 0 (0.0) | 14 (6.3) | 33 (4.2) | 140 (5.0) | 22 (3.3) | 8 (5.4) | 11 (5.4) | 228 (4.6) |
| 2011–2015 | 18 (36.7) | 76 (34.2) | 251 (32.1) | 862 (30.6) | 184 (27.3) | 39 (26.2) | 70 (34.5) | 1500 (30.6) |
| 2016–2019 | 31 (63.2) | 132 (59.5) | 499 (63.7) | 1,813 (64.4) | 467 (69.4) | 102 (68.5) | 122 (60.1) | 3,166 (64.8) |
| Age, years – median (IQR) | 67 (57, 73) | 66 (55, 74) | 64 (54, 73) | 68 (57, 77) | 71 (61, 79) | 70 (62, 80) | 70 (60, 78) | 68 (57, 77) |
| Sex, Men – n (%) | 25 (51.0) | 135 (60.8) | 520 (66.4) | 2,001 (71.1) | 444 (66.0) | 93 (62.4) | 118 (58.1) | 3,336 (68.2) |
| Presumed cause of OHCA – n (%) | ||||||||
| Cardiac | 42 (85.7) | 174 (78.4) | 640 (81.7) | 2,195 (78.0) | 460 (68.4) | 99 (62.4) | 142 (70.0) | 3,752 (76.7) |
| Noncardiac | 7 (14.3) | 39 (17.6) | 112 (14.3) | 455 (16.2) | 141 (21.0) | 32 (21.5) | 45 (22.2) | 831 (17.0) |
| Missing | 0 (0.0) | 9 (4.1) | 31 (4.0) | 165 (5.9) | 72 (10.7) | 18 (12.1) | 16 (7.9) | 311 (6.4) |
| Charlson score – n (%) | ||||||||
| 0 | 28 (57.1) | 121 (54.5) | 415 (53.0) | 1,559 (55.4) | 409 (60.8) | 88 (59.1) | 113 (55.7) | 2,733 (55.8) |
| 1 | 8 (16.3) | 57 (25.7) | 200 (25.5) | 706 (25.1) | 156 (23.2) | 43 (28.9) | 58 (28.6) | 1,228 (25.1) |
| ≥ 2 | 13 (26.5) | 44 (19.8) | 168 (21.5) | 550 (19.5) | 108 (16.0) | 18 (12.1) | 32 (15.8) | 933 (19.1) |
| Status at hospital arrival – n (%) | ||||||||
| ROSC and comatose | 34 (69.4) | 157 (70.7) | 486 (62.1) | 1,857 (66.0) | 519 (77.1) | 122 (81.9) | 167 (82.3) | 3,342 (68.3) |
| ROSC and awake | 14 (28.6) | 53 (23.9) | 262 (33.5) | 854 (30.3) | 126 (18.7) | 22 (14.8) | 29 (14.3) | 1,360 (27.8) |
| ROSC and unknown status | 1 (2.0) | 12 (5.4) | 35 (4.5) | 104 (3.7) | 28 (4.2) | 5 (3.4) | 7 (3.4) | 192 (3.9) |
| Location of arrest – n (%) | ||||||||
| Arrest in private home | 30 (65.2) | 149 (68.7) | 427 (55.5) | 1,601 (57.6) | 448 (68.4) | 108 (75.0) | 155 (77.1) | 2,918 (60.6) |
| First registered rhythm – n (%) | ||||||||
| Shockable | 32 (68.1) | 110 (51.4) | 455 (59.5) | 1,454 (52.9) | 199 (30.0) | 32 (21.8) | 33 (16.6) | 2,315 (48.4) |
| Witnessed status of OHCA – n (%) | ||||||||
| Bystander-witnessed | 30 (61.2) | 142 (64.0) | 475 (61.1) | 1,805 (64.5) | 390 (58.3) | 82 (55.0) | 101 (49.8) | 3,025 (62.1) |
| EMS-witnessed | 10 (20.4) | 38 (17.1) | 156 (20.0) | 473 (16.9) | 102 (15.2) | 30 (20.1) | 48 (23.6) | 857 (17.6) |
| Unwitnessed | 9 (18.4) | 42 (18.9) | 147 (18.9) | 521 (18.9) | 177 (26.5) | 37 (24.9) | 54 (26.6) | 987 (20.3) |
| Bystander CPR – n (%) | ||||||||
| Bystander CPR | 27 (56.2) | 142 (64.0) | 503 (64.7) | 1,874 (66.8) | 433 (64.5) | 83 (55.7) | 105 (51.7) | 3,167 (65.0) |
| EMS-witnessed OHCA | 10 (20.8) | 38 (17.1) | 156 (20.1) | 473 (16.9) | 102 (15.2) | 30 (20.1) | 48 (23.7) | 857 (17.6) |
| Neither of the above | 11 (23.0) | 42 (18.9) | 118 (15.2) | 457 (16.3) | 136 (20.3) | 36 (24.2) | 50 (24.6) | 850 (17.4) |
| Pre-hospital defibrillation – n (%) | 34 (73.9) | 137 (64.3) | 531 (72.1) | 1,655 (62.3) | 262 (40.4) | 41 (29.3) | 51 (26.3) | 2,711 (58.5) |
| Physician present before hospital arrival – n (%) | 43 (91.5) | 178 (89.5) | 639 (89.5) | 2,300 (90.6) | 540 (92.2) | 132 (95.0) | 170 (89.3) | 4,002 (90.7) |
| Time interval from recognition of OHCA to EMS arrival, minutes – median (IQR)* | 10 (6–13) | 10 (7–13) | 10 (7–15) | 10 (7–14) | 10 (5–15) | 11 (8–16) | 10 (7–20) | 10 (6–14) |
| Time interval from recognition of OHCA to ROSC, minutes – median (IQR) | 12 (4.5–18.5) | 14 (5–23) | 14 (6–21) | 14 (6–21) | 15 (8–23) | 16 (9–27) | 19 (10–27) | 14 (7–27) |
| Time interval from recognition of OHCA to K measurement, minutes – median (IQR) | 71 (59–110) | 76 (54–104) | 70 (52–96) | 76 (56–110) | 77 (56–106) | 79 (62–114) | 77 (61–104) | 75 (56–105) |
| Inpatient contact post-OHCA | 49 (100.0) | 214 (96.4) | 757 (96.7) | 2,703 (96.7) | 635 (94.4) | 136 (91.3) | 190 (93.6) | 4,684 (95.7) |
| ICU-stay post-OHCA (within one week) | 36 (73.5) | 151 (68.0) | 486 (62.1) | 1,888 (67.1) | 498 (74.0) | 114 (76.5) | 150 (73.9) | 3,323 (67.9) |
| 30-day survival | 25 (51.0) | 119 (53.6) | 512 (65.4) | 1,631 (57.9) | 220 (32.7) | 34 (22.8) | 46 (22.7) | 2,587 (52.9) |
| 1-year survival | 22 (44.9) | 106 (47.7) | 476 (60.8) | 1,489 (52.9) | 178 (26.4) | 28 (18.8) | 36 (17.7) | 2,335 (47.7) |
| Potassium – median (IQR) | 2.2 (2.1, 2.4) | 2.8 (2.7, 2.9) | 3.2 (3.1, 3.3) | 3.9 (3.7, 4.2) | 5.0 (4.8, 5.2) | 5.8 (5.7, 5.9) | 6.7 (6.4, 7.2) | 3.9 (3.5, 4.5) |
| Lactate, mmol/L – median (IQR) | 4.4 (2.6, 10.2) | 5.6 (3.2, 9.4) | 5.0 (3.1, 8.3) | 5.1 (2.7, 8.7) | 7.4 (3.9, 10.9) | 7.8 (4.8, 11.3) | 8.5 (5.2, 13.7) | 5.7 (3.0, 9.4) |
| pH – median (IQR)** | 7.3 (7.2, 7.4) | 7.3 (7.2, 7.4) | 7.3 (7.2, 7.3) | 7.2 (7.1, 7.3) | 7.1 (7.0, 7.2) | 7.0 (6.9, 7.2) | 7.0 (6.9, 7.1) | 7.2 (7.1, 7.3) |
| eGFR post-OHCA, mL/min/1.73m2 – median (IQR)** | 66 (52, 81) | 64 (52, 85) | 68 (55, 85) | 63 (49, 79) | 51 (37, 66) | 46 (27, 63) | 33 (20, 52) | 61 (47, 77) |
| eGFR pre-OHCA, mL/min/1.73m2 – median (IQR)*** | 65 (44–82) | 74 (42–90) | 80 (61–90) | 78 (52–90) | 66 (30–90) | 56 (30–88) | 43 (20–76) | 75 (46–90) |
*EMS-witnessed excluded
**within 24 h after OHCA
*** within 30 days before OHCA
In the calculation of percentages, observations with missing value for the covariate involved in calculation were excluded. CPR cardiopulmonary resuscitation. eGFR estimated glomerular filtration rate. EMS emergency medical services. ICU intensive care unit. IQR interquartile range. OHCA out-of-hospital cardiac arrest. ROSC return of spontaneous circulation. Total number of missing: location of arrest n = 81 (1.7%); first registered rythm n = 112 (2.3%); witnessed status n = 25 (0.5%); bystander CPR n = 20 (0.4%); pre-hospital defibrillation n = 258 (5.3%); physician present before hospital arrival n = 481 (9.8%); time interval from recognition of OHCA to EMS arrival n = 3,037 (75.2%); time interval from recognition of OHCA to ROSC n = 1,122 (22.9%); lactate n = 1,840 (37.6%); pH n = 1,684 (34.4%); eGFR—post-OHCA n = 988 (20.2%); eGFR pre- OHCA n = 1,593 (32.5%)
Outcomes
Figure 2 illustrates a Kaplan–Meier curve depicting the survival probability across different potassium intervals, revealing significantly reduced 30-day survival rates in all patient groups with hyperkalemia (P < 0.001 for all groups). Figure 3 presents standardized survival probabilities and average RR of 30-day survival. Notably, standardized average RR was significantly lower for all hyperkalemic groups compared to normokalemia: 4.7–5.5 mmol/L (average RR: 0.72, 95% CI: 0.66–0.78); 5.5–6.0 mmol/L (average RR: 0.60, 95% CI: 0.47–0.73); > 6.0 mmol/L (average RR: 0.56, 95% CI: 0.46–0.66), indicating lower survival. Conversely, no significant survival trend was observed in patients with hypokalemia compared to normokalemia: < 2.5 mmol/L (average RR: 0.81, 95% CI: 0.60–1.02); 2.5–2.9 mmol/L (average RR: 0.90, 95% CI: 0.82–0.99); 3.0–3.4 mmol/L (average RR: 1.04, 95% CI: 0.96–1.11). Figure 4 presents a restricted cubic spline analysis illustrating the adjusted hazard ratios for 30-day survival in relation to potassium concentration. The data reveals an inverse J-shaped relationship between potassium levels and the outcome. The main results were confirmed in sub analyses including a) patients who had a post-OHCA inpatient hospital contact (4,684 patients, i.e. excluding 210 patients who died in the emergency room, Supplemental Table 2), and b) patients with an estimated glomerular filtration rate (eGFR) ≥ 60 within one month prior to OHCA (2,227 patients, Supplemental Table 3). Additionally, in a sensitivity analysis involving 3,054 patients (62.4% of the total study population), the inclusion of lactate data in the multivariable Cox regression analysis did not alter our significant findings regarding reduced survival in patients with hyperkalemia: 4.7–5.5 mmol/L (average RR: 0.83, 95% CI: 0.76–0.91); 5.5–6.0 mmol/L (average RR: 0.69, 95% CI: 0.53–0.85); > 6.0 mmol/L (average RR: 0.75, 95% CI: 0.62–0.89), (Supplemental Table 4). Furthermore, a sub analysis including only patients with presumed cardiac cause of arrest (3,755 patients, 76.7% of the total study population) yielded results consistent with those of the main analysis with reduced survival in hyperkalemic patients: 4.7–5.5 mmol/L (average RR: 0.76, 95% CI: 0.70–0.83); 5.5–6.0 mmol/L (average RR: 0.75, 95% CI: 0.63–0.74); > 6.0 mmol/L (average RR: 0.63, 95% CI: 0.53–0.74), (Supplemental Table 5). Due to the small number of patients with a non-cardiac cause and the heterogeneity of this group, a separate analysis could not be conducted. Time to ROSC was available in 3,772 (77.1%) patients. In a sensitivity the inclusion of ‘time to ROSC’ data in the multivariable Cox regression analysis our findings regarding reduced survival in patients with hyperkalemia remained significant: 4.7–5.5 mmol/L (average RR: 0.76, 95% CI: 0.70–0.83); 5.5–6.0 mmol/L (average RR: 0.65, 95% CI: 0.52–0.79); > 6.0 mmol/L (average RR: 0.55, 95% CI: 0.45–0.64), (Supplemental Table 6).
Fig. 2.

Log-rank test of the survival probability among the different potassium intervals (n = 4,894)
Fig. 3.
Standardized 30-day survival probability and average risk ratio in OHCA-patients stratified by potassium intervals. Reference interval represented by the interval K+: 3.5–4.6 mmol/l. * The models included age, sex, Charlson score, witnessed status, performance of bystander cardiopulmonary resuscitation, and first registered heart rhythm
Fig. 4.
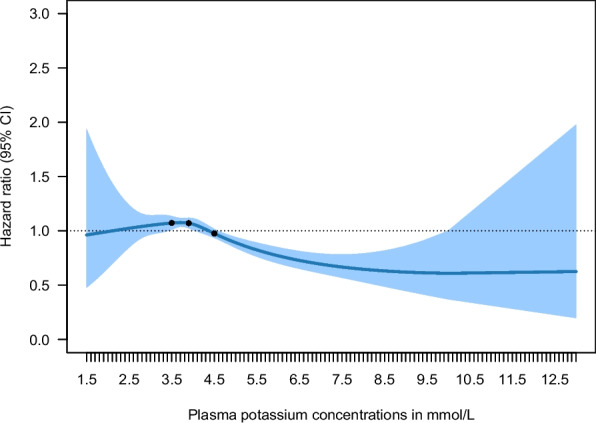
Restricted cubic splines showing the adjusted hazard ratios for 30-day survival as a function of potassium concentration. Knots at the 25th, 50th and 75th percentiles of potassium. Model adjusted for age, sex, Charlson score, witnessed status, performance of bystander cardiopulmonary resuscitation, and first registered heart rhythm
Discussion
Within this extensive nationwide cohort of OHCA survivors, we assessed the association between the probability of 30-day survival and various serum potassium levels, determined as the first value within the initial 12 h following OHCA. The findings indicated a significantly reduced 30-day survival rate in patients with any degree of hyperkalemia compared to those with normokalemia (3.5–4.6 mmol/L). Conversely, no significant difference in survival was observed among groups with hypokalemia. Preliminary data has previously been presented as a congress abstract [29]. Only two prior studies have investigated the initial serum potassium levels and survival in OHCA patients. Results were in alignment with ours [10, 11]. We extend the results of these studies by including a significantly larger population for analysis, allowing us to stratify into more potassium categories.
This analysis has several potential clinical implications. Serum potassium serves as a readily measurable biomarker in clinical settings, enabling medical personnel to swiftly assess its levels. However, this is one of the few studies incorporating laboratory values in an OHCA registry. Potassium stands as one of the most plentiful cations within the human body and holds significance in maintaining acid–base equilibrium [30, 31]. Therefore, it is important to note that hyperkalemia often accompanies acidosis, particularly in cases where patients were resuscitated but experienced severe acidosis due to inadequate oxygenation and prolonged no-flow time. Meanwhile, lactate has been shown to be the strongest determinant of acidosis in these patients [32]. It is well established that when blood flow is halted during cardiac arrest, anaerobic metabolism takes over, leading to lactic acid production, the development of metabolic acidosis, and a subsequent rise in serum potassium levels as potassium shifts out of the cells [33, 34]. While an elevated potassium level may serve as an indicator of a poor outcome, it may not necessarily be the direct cause of the unfavorable outcome. That said, our results showed a significant association between hyperkalemia and lower survival, and results remained significant when adjusting for lactate level. Another critical factor influencing survival and neurological outcomes following OHCA is time to ROSC. Previous studies have shown an inverse relationship between resuscitation duration and outcomes [35–38]. However, when adjusting for time to ROSC in our multivariable model, the association between hyperkalemia and reduced 30-day survival remained significant. Interestingly, we observed a sharp decline in shockable rhythm proportion in patients with hyperkalemia, supporting the association between hyperkalemia and poor outcomes. Further investigation is needed to determine whether targeting potassium levels could impact outcomes in patients initially surviving OHCA and those with cardiovascular disease in general [39, 40].
Another widely acknowledged association is the one between hyperkalemia and renal failure. The conditions are associated both in general and specifically in relation to OHCA [41, 42]. Importantly, all our results remained consistent both when adjusting for Charlson comorbidity index in the multivariable analysis, and when only examining patients with an eGFR ≥ 60 within one month before OHCA.
Since our data were observational, we can only propose hypotheses and are unable to assess causality. Furthermore, our data were collected retrospectively, and it is likely that by the time serum potassium levels become available, a considerable proportion of patients may have already deceased. Consequently, there exists an immortal bias in our findings, as patients are ensured to survive until the time of the sample collection. Furthermore, our data were collected over an extended period, which may have led to variations in intra- and post-arrest care. Nevertheless, the majority of the data were obtained within the past decade and within each time period, data were evenly distributed across potassium levels. Serum potassium was available in around one third of patients with ROSC from 2001–2019, and we did find worse outcomes in patients without available serum potassium compared with those included in our study. However, the large study population reduces this risk of bias. Furthermore, data on electrocardiographic findings and medications, including potential treatment of hyperkalemia prior to hospital arrival, were unavailable and could not be included in the adjustments. Our study did not include long term survival or neurological outcomes. Hence, the association between serum potassium and these outcomes remain to be thoroughly investigated in such a study population. A recent post hoc analysis of 1,133 patients from four trials reported that among comatose patients admitted to intensive care following cardiac arrest, hyperkalemia was associated with poor cerebral performance outcomes at 180 days, whereas hypokalemia was not, supporting our results [43]. Similar results on neurological outcomes were found in a previous prospective, multicenter observational study [11].
While the causality between hyperkalemia and decreased survival after OHCA is still uncertain, it could still prove valuable as an indicator for early prognostication regarding the outcome following OHCA with ROSC.
Conclusion
In patients surviving OHCA, hyperkalemia was significantly associated with reduced 30-day survival compared with potassium within the normal range. The association was independent of age, sex, Charlson comorbidity index, witnessed status, performance of bystander CPR, and first registered heart rhythm. Conversely, hypokalemia was not associated with reduced or increased 30-day survival compared with normokalemia in OHCA survivors.
Supplementary Information
Acknowledgements
Not applicable.
Abbreviations
- CI
Confidence interval
- CPR
Cardiopulmonary resuscitation
- eGFR
Estimated glomerular filtration rate
- EMS
Emergency medical services
- OHCA
Out of hospital cardiac arrest
- RR
Risk ratio
- ROSC
Return of spontaneous circulation
Authors' contributions
CB participated in the conception and design of the work, interpreted data and drafted the work. MLK participated in the design of the work analyzed data and substantively revised the work. KK participated in the conception and design of the work, interpreted data and substantively revised the work. MP participated in the conception and design of the work, interpreted data and substantively revised the work. GHM participated in the design of the work and substantively revised the work. KBR substantively revised the work. MW participated in the design of the work and substantively revised the work. SR substantively revised the work. MW substantively revised the work. FF substantively revised the work. GG participated in the acquisition and substantively revised the work. LK participated in the acquisition and substantively revised the work. CH participated in the design of the work and substantively revised the work. JK participated in the design of the work and substantively revised the work. CBF substantively revised the work. CTP participated in the acquisition and substantively revised the work. GYHL substantively revised the work. CAB participated in the conception and design of the work, analyzed and interpreted data and substantively revised the work. All authors read and approved the final manuscript.
Funding
Open access funding provided by Copenhagen University The Danish Heart Foundation and The Danish Foundation TrygFonden supported the study financially. The two institutions have no commercial interests in the cardiac arrest area and had no influence on study design or conduct; collection, management, analysis, or interpretation of the data; or preparation, review, or approval of the manuscript for submission.
Data availability
No datasets were generated or analysed during the current study.
Declarations
Ethics approval and consent to participate
The study was approved by the Danish Data Protection Agency. Registry-based studies does not require ethical approval by The Danish National Committee on Health Research Ethics.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Kiguchi T, Okubo M, Nishiyama C, Maconochie I, Ong MEH, Kern KB, et al. Out-of-hospital cardiac arrest across the World: first report from the International Liaison Committee on Resuscitation (ILCOR). Resuscitation. 2020;152:39–49. 10.1016/j.resuscitation.2020.02.044. [DOI] [PubMed] [Google Scholar]
- 2.Nielsen N, Wetterslev J, Cronberg T, Erlinge D, Gasche Y, Hassager C, et al. Targeted temperature management at 33°C versus 36°C after cardiac arrest. N Engl J Med. 2013;369:2197–206. 10.1056/NEJMoa1310519. [DOI] [PubMed] [Google Scholar]
- 3.Sondergaard KB, Riddersholm S, Wissenberg M, Moller Hansen S, Barcella CA, Karlsson L, et al. Out-of-hospital cardiac arrest: 30-day survival and 1-year risk of anoxic brain damage or nursing home admission according to consciousness status at hospital arrival. Resuscitation. 2020;148:251–8. 10.1016/j.resuscitation.2019.12.006. [DOI] [PubMed] [Google Scholar]
- 4.Nolan JP, Sandroni C, Böttiger BW, Cariou A, Cronberg T, Friberg H, et al. European Resuscitation Council and European Society of Intensive Care Medicine guidelines 2021: post-resuscitation care. Intensive Care Med. 2021;47:369–421. 10.1007/s00134-021-06368-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Berg KM, Cheng A, Panchal AR, Topjian AA, Aziz K, Bhanji F, et al. Part 7: systems of care: 2020 American Heart Association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation. 2020;142:S580-604. 10.1161/CIR.0000000000000899. [DOI] [PubMed] [Google Scholar]
- 6.Krogager ML, Torp-Pedersen C, Mortensen RN, Køber L, Gislason G, Søgaard P, et al. Short-term mortality risk of serum potassium levels in hypertension: a retrospective analysis of nationwide registry data. Eur Heart J. 2017;38:104–12. 10.1093/eurheartj/ehw129. [DOI] [PubMed] [Google Scholar]
- 7.Krogager ML, Eggers-Kaas L, Aasbjerg K, Mortensen RN, Køber L, Gislason G, et al. Short-term mortality risk of serum potassium levels in acute heart failure following myocardial infarction. Eur Heart J Cardiovasc Pharmacother. 2015;1:245–51. 10.1093/ehjcvp/pvv026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Hagengaard L, Søgaard P, Espersen M, Sessa M, Lund PE, Krogager ML, et al. Association between serum potassium levels and short-term mortality in patients with atrial fibrillation or flutter co-treated with diuretics and rate- or rhythm-controlling drugs. Eur Heart J Cardiovasc Pharmacother. 2020;6:137–44. 10.1093/ehjcvp/pvz024. [DOI] [PubMed] [Google Scholar]
- 9.Goyal A, Spertus JA, Gosch K, Venkitachalam L, Jones PG, Van Den Berghe G, et al. Serum potassium levels and mortality in acute myocardial infarction. JAMA. 2012;307:157. 10.1001/jama.2011.1967. [DOI] [PubMed] [Google Scholar]
- 10.Choi DS, Shin SD, Ro YS, Lee KW. Relationship between serum potassium level and survival outcome in out-of-hospital cardiac arrest using CAPTURES database of Korea: does hypokalemia have good neurological outcomes in out-of-hospital cardiac arrest? Adv Clin Exp Med. 2020;29:727–34. 10.17219/acem/122178. [DOI] [PubMed]
- 11.Shida H, Matsuyama T, Iwami T, Okabayashi S, Yamada T, Hayakawa K, et al. Serum potassium level on hospital arrival and survival after out-of-hospital cardiac arrest: the CRITICAL study in Osaka, Japan. Eur Heart J Acute Cardiovasc Care. 2020;9:S175–83. 10.1177/2048872619848883. [DOI] [PubMed] [Google Scholar]
- 12.Shin J, Lim YS, Kim K, Lee HJ, Lee SJ, Jung E, et al. Initial blood pH during cardiopulmonary resuscitation in out-of-hospital cardiac arrest patients: a multicenter observational registry-based study. Crit Care. 2017;21:322. 10.1186/s13054-017-1893-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Nolan JP, Neumar RW, Adrie C, Aibiki M, Berg RA, Böttiger BW, et al. Post-cardiac arrest syndrome: epidemiology, pathophysiology, treatment, and prognostication. A Scientific Statement from the International Liaison Committee on Resuscitation; the American Heart Association Emergency Cardiovascular Care Committee; the Council on Cardiovascular Surgery and Anesthesia; the Council on Cardiopulmonary, Perioperative, and Critical Care; the Council on Clinical Cardiology; the Council on Stroke. Resuscitation. 2008;79:350–79. 10.1016/j.resuscitation.2008.09.017. [DOI] [PubMed]
- 14.Deakin CD, Nolan JP, Soar J, Sunde K, Koster RW, Smith GB, et al. European resuscitation council guidelines for resuscitation 2010 section 4. Adult advanced life support. Resuscitation. 2010;81:1305–52. 10.1016/j.resuscitation.2010.08.017. [DOI] [PubMed] [Google Scholar]
- 15.Soar J, Nolan JP, Böttiger BW, Perkins GD, Lott C, Carli P, et al. European resuscitation council guidelines for resuscitation 2015: section 3. Adult advanced life support. Resuscitation. 2015;95:100–47. 10.1016/j.resuscitation.2015.07.016. [DOI] [PubMed] [Google Scholar]
- 16.Christensen DM, Rajan S, Kragholm K, Søndergaard KB, Hansen OM, Gerds TA, et al. Bystander cardiopulmonary resuscitation and survival in patients with out-of-hospital cardiac arrest of non-cardiac origin. Resuscitation. 2019;140:98–105. 10.1016/j.resuscitation.2019.05.014. [DOI] [PubMed] [Google Scholar]
- 17.Perkins GD, Jacobs IG, Nadkarni VM, Berg RA, Bhanji F, Biarent D, et al. Cardiac arrest and cardiopulmonary resuscitation outcome reports: update of the Utstein resuscitation registry templates for out-of-hospital cardiac arrest: a statement for healthcare professionals from a task force of the International Liaison Committee on Resuscitation (American Heart Association, European Resuscitation Council, Australian and New Zealand Council on Resuscitation, Heart and Stroke Foundation of Canada, InterAmerican Heart Foundation, Resuscitation Council of Southern Africa, Resuscitation Council of Asia); and the American Heart Association Emergency Cardiovascular Care Committee and the Council on Cardiopulmonary, critical care. Periop Resuscitation Circ. 2015;132:1286–300. 10.1161/CIR.0000000000000144. [DOI] [PubMed] [Google Scholar]
- 18.Wissenberg M, Lippert FK, Folke F, Weeke P, Hansen CM, Christensen EF, et al. Association of national initiatives to improve cardiac arrest management with rates of bystander intervention and patient survival after out-of-hospital cardiac arrest. JAMA. 2013;310:1377–84. 10.1001/jama.2013.278483. [DOI] [PubMed] [Google Scholar]
- 19.Jacobs I, Nadkarni V, Bahr J, Berg RA, Billi JE, Bossaert L, et al. Cardiac arrest and cardiopulmonary resuscitation outcome reports: update and simplification of the Utstein templates for resuscitation registries: a statement for healthcare professionals from a task force of the International Liaison Committee on Resuscitation (American Heart Association, European Resuscitation Council, Australian Resuscitation Council, New Zealand Resuscitation Council, Heart and Stroke Foundation of Canada, InterAmerican Heart Foundation, Resuscitation Councils of Southern Africa). Circulation. 2004;110:3385–97. 10.1161/01.CIR.0000147236.85306.15. [DOI] [PubMed] [Google Scholar]
- 20.Wissenberg M, Folke F, Hansen CM, Lippert FK, Kragholm K, Risgaard B, et al. Survival after out-of-hospital cardiac arrest in relation to age and early identification of patients with minimal chance of long-term survival. Circulation. 2015;131:1536–45. 10.1161/CIRCULATIONAHA.114.013122. [DOI] [PubMed] [Google Scholar]
- 21.Pedersen CB. The Danish civil registration system. Scand J Public Health. 2011;39:22–5. 10.1177/1403494810387965. [DOI] [PubMed] [Google Scholar]
- 22.Grann, Erichsen R, Nielsen, Frøslev, Thomsen R. Existing data sources for clinical epidemiology: the clinical laboratory information system (LABKA) research database at Aarhus University, Denmark. CLEP. 2011:133. 10.2147/CLEP.S17901. [DOI] [PMC free article] [PubMed]
- 23.Byrne C, Barcella CA, Krogager ML, Pareek M, Ringgren KB, Andersen MP, et al. External validation of the simple NULL-PLEASE clinical score in predicting outcomes of out-of-hospital cardiac arrest in the Danish population - a nationwide registry-based study. Resuscitation. 2022:S0300–9572(22)00644-X. 10.1016/j.resuscitation.2022.08.009. [DOI] [PubMed]
- 24.Lynge E, Sandegaard JL, Rebolj M. The Danish national patient register. Scand J Public Health. 2011;39:30–3. 10.1177/1403494811401482. [DOI] [PubMed] [Google Scholar]
- 25.Helweg-Larsen K. The Danish register of causes of death. Scand J Public Health. 2011;39:26–9. 10.1177/1403494811399958. [DOI] [PubMed] [Google Scholar]
- 26.Rustad P, Felding P, Franzson L, Kairisto V, Lahti A, Mårtensson A, et al. The Nordic Reference Interval Project 2000: recommended reference intervals for 25 common biochemical properties. Scand J Clin Lab Invest. 2004;64:271–84. 10.1080/00365510410006324. [DOI] [PubMed] [Google Scholar]
- 27.Bartlett JW, Seaman SR, White IR, Carpenter JR, for the Alzheimer’s Disease Neuroimaging Initiative*. Multiple imputation of covariates by fully conditional specification: accommodating the substantive model. Stat Methods Med Res. 2015;24:462–87. 10.1177/0962280214521348. [DOI] [PMC free article] [PubMed]
- 28.R Development Core Team. R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria; 2008. ISBN 3–900051–07–0, ISBN 3–900051–07–0. Available from: http://www.R-project.org.n.d.
- 29.Byrne C, Krogager M, Kragholm K, Pareek M, Mohr G, Ringgren K, et al. Association between serum potassium levels and short-term mortality in out-of-hospital cardiac arrest survivors. Eur Heart J Acute Cardiovasc Care. 2022;11:zuac041.082. 10.1093/ehjacc/zuac041.082.
- 30.Gumz ML, Rabinowitz L, Wingo CS. An integrated view of potassium homeostasis. N Engl J Med. 2015;373:60–72. 10.1056/NEJMra1313341. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.McDonough AA, Youn JH. Potassium homeostasis: the knowns, the unknowns, and the health benefits. Physiology (Bethesda). 2017;32:100–11. 10.1152/physiol.00022.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Makino J, Uchino S, Morimatsu H, Bellomo R. A quantitative analysis of the acidosis of cardiac arrest: a prospective observational study. Crit Care. 2005;9:R357-362. 10.1186/cc3714. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Brown RS. Potassium homeostasis and clinical implications. Am J Med. 1984;77:3–10. 10.1016/s0002-9343(84)80002-9. [DOI] [PubMed] [Google Scholar]
- 34.Geddes LA, Roeder RA, Rundell AE, Otlewski MP, Kemeny AE, Lottes AE. The natural biochemical changes during ventricular fibrillation with cardiopulmonary resuscitation and the onset of postdefibrillation pulseless electrical activity. Am J Emerg Med. 2006;24:577–81. 10.1016/j.ajem.2006.01.030. [DOI] [PubMed] [Google Scholar]
- 35.Nagao K, Nonogi H, Yonemoto N, Gaieski DF, Ito N, Takayama M, et al. Duration of prehospital resuscitation efforts after out-of-hospital cardiac arrest. Circulation. 2016;133:1386–96. 10.1161/CIRCULATIONAHA.115.018788. [DOI] [PubMed] [Google Scholar]
- 36.Reynolds JC, Grunau BE, Rittenberger JC, Sawyer KN, Kurz MC, Callaway CW. Association between duration of resuscitation and favorable outcome after out-of-hospital cardiac arrest: implications for prolonging or terminating resuscitation. Circulation. 2016;134:2084–94. 10.1161/CIRCULATIONAHA.116.023309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Matsuyama T, Kitamura T, Kiyohara K, Nishiyama C, Nishiuchi T, Hayashi Y, et al. Impact of cardiopulmonary resuscitation duration on neurologically favourable outcome after out-of-hospital cardiac arrest: a population-based study in Japan. Resuscitation. 2017;113:1–7. 10.1016/j.resuscitation.2017.01.005. [DOI] [PubMed] [Google Scholar]
- 38.Kashiura M, Hamabe Y, Akashi A, Sakurai A, Tahara Y, Yonemoto N, et al. Association between cardiopulmonary resuscitation duration and one-month neurological outcomes for out-of-hospital cardiac arrest: a prospective cohort study. BMC Anesthesiol. 2017;17:59. 10.1186/s12871-017-0351-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Winsløw U, Sakthivel T, Zheng C, Bosselmann H, Haugan K, Bruun N, et al. Targeted potassium levels to decrease arrhythmia burden in high risk patients with cardiovascular diseases (POTCAST): study protocol for a randomized controlled trial. Am Heart J. 2022;253:59–66. 10.1016/j.ahj.2022.07.003. [DOI] [PubMed] [Google Scholar]
- 40.Soeholm H, Kirkegaard H. Serum potassium changes during therapeutic hypothermia after out-of-hospital cardiac arrest-should it be treated? Ther Hypothermia Temp Manag. 2012;2:30–6. 10.1089/ther.2012.0004. [DOI] [PubMed] [Google Scholar]
- 41.Lin CH, Tu YF, Chiang WC, Wu SY, Chang YH, Chi CH. Electrolyte abnormalities and laboratory findings in patients with out-of-hospital cardiac arrest who have kidney disease. Am J Emerg Med. 2013;31:487–93. 10.1016/j.ajem.2012.09.021. [DOI] [PubMed] [Google Scholar]
- 42.Jain N, Kotla S, Little BB, Weideman RA, Brilakis ES, Reilly RF, et al. Predictors of hyperkalemia and death in patients with cardiac and renal disease. Am J Cardiol. 2012;109:1510–3. 10.1016/j.amjcard.2012.01.367. [DOI] [PubMed] [Google Scholar]
- 43.Holm A, Lascarrou JB, Cariou A, Reinikainen M, Laitio T, Kirkegaard H, et al. Potassium disorders at intensive care unit admission and functional outcomes after cardiac arrest. Resuscitation. 2024;205: 110439. 10.1016/j.resuscitation.2024.110439. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
No datasets were generated or analysed during the current study.