Abstract
Background
The burden and prevalence of food allergy (FA), a condition known to negatively impact the quality of life (QoL) of patients and their families, vary across countries due to cultural habits and clinical practices and have been assessed differently among previous studies. To date, no validated scale exists to measure FA severity risk.
Objective
The study objectives were to develop a standardized methodology to estimate FA point prevalence in children and adults in 9 countries, to assess FA impact on QoL, and to construct a framework to define FA severity risk based on QoL and FA clinical severity.
Methods
This was a cross-sectional, international study that collected self-reported data through an online quantitative survey. Participants were recruited from general population panels: parents/caregivers of children aged 6 months-17 years, and adults aged 18–65 years. The survey instrument was developed by a panel of clinical FA experts and epidemiologists.
Results
Weighted prevalence rates were calculated for the 4 generated case definitions of FA (self-reported, physician-diagnosed, symptom-convincing, and confirmed FA). Several validated scales, including the Food Allergy Quality of Life Questionnaire (FAQLQ) and the Food Allergy Independent Measure (FAIM), were used to assess FA impact on QoL in patients with symptom-convincing FA. Four FA severity risk profiles were generated: non-severe, at-risk, highly impacted QoL, and severe patients.
Conclusion
The ASSESS FA study developed a methodology to estimate the prevalence of FA and its impact on QoL consistently across countries, age groups, and food allergens. The severity risk framework was based on both the clinical severity and impact on QoL aspects of FA.
Keywords: Food hypersensitivity, Epidemiology, Immunoglobulin E, Allergy and immunology, Cross-sectional studies, Methods
Introduction
Immunoglobulin E (IgE)-mediated food allergies (FA) are defined as adverse reactions after systemic exposure to a given allergen. From a pathophysiological point of view, they are an immune system response where food-specific immunoglobulin E antibodies are produced and released. Symptoms of FA can range from moderate (such as nausea or vomiting) to more severe (such as enterocolitis or anaphylaxis).1,2 The severity of symptoms is dependent on age, life exposure, and comorbidity with other allergy diagnoses such as atopic dermatitis, allergic rhinitis, or asthma.1,2
The overall prevalence of FA in the general population worldwide stands at approximately 2%, although rates vary from one region to another.3 In the United States of America (USA) and Canada, the prevalence rate of reported FA ranges between 6.5% and 8.7% in the pediatric population, and around 5.9%–10.8% in the adult population.4, 5, 6, 7 In Europe, a great variation can be observed across nations, ranging from 2% to up to 20% in children and 1%–4.7% in adults.8, 9, 10, 11, 12, 13, 14, 15 The only published data available in Japan relate to children (not adults), and it report a rate of around 5%.16 In China, pediatric FA rates are between 4% and 12%, while in adults between 7% and 14%.17,18 In general, the body of evidence for age-specific estimates suggests that the prevalence of FA is highest among younger children.10,16,19,20 Some children eventually develop natural tolerance through their lifetime, but others may retain their FA into adulthood.21 Alternatively, adults can still develop new FA by basis of cross-reactivity between inhalants and food proteins.22
Geographical and genetic variations may contribute to differences in FA prevalence. These variations may result from cultural differences and how different societies cope with food allergies or other conditions. Also, country-specific healthcare systems, access to healthcare, and nutritional assessment are highly dependent on socioeconomic variables and may play a role.23,24 In addition, different study designs, methodologies, FA prevalence definitions, and diagnostic tools used by researchers may contribute to the discrepancies in prevalence measurements, as there is no single, standardized way to diagnose and report food allergies. FA prevalence estimates for pediatric and adult populations have been based on self- or parent-reported FA or a diagnosis of FA through a skin-prick test, oral food challenge, double-blind placebo-controlled food challenge, or measured IgE.4,6,9,12,14
For both pediatric and adult patients, standard of care includes food avoidance and epinephrine auto-injection use in case of accidental exposure. Dietary restriction and constant fear of allergen exposure can cause anxiety and restrict daily activities, which consequently points to FA as a burden on quality of life (QoL) for both the patients and their families.25,26 Additionally, FA can result in significant medical and out-of-pocket costs that may vary according to disease severity, as patients with milder forms may not require all the therapeutic resources necessary for those who are at higher risk of life-threatening, food-induced anaphylaxis.27,28 Given the significant economic and QoL implications associated with FA management, it is crucial to evaluate the prevalence and impact of FA as reliably as possible to meet patients’ unmet needs.
To gain a comprehensive understanding of FA's burden, the International Cross-sectional Prevalence and Severity Study of Pediatric and Adult IgE-mediated Food Allergies (ASSESS FA) aimed to estimate the point prevalence of FA, FA's impact on health-related quality of life (HRQoL), and disease severity risk in the pediatric and adult populations in the United States, Canada, the United Kingdom (UK), France, Germany, Italy, Spain, Japan, and China. The study employed a standardized reporting protocol, utilizing definitions of FA and questionnaires that were refined with clinical experts and epidemiologists, and FA-specific HRQoL assessment tools. Since there is currently no gold standard in practice guidelines that defines or classifies patients' profiles based on the severity of their FA, the aim of the study was also to develop such a framework. Applying the same standardized methodology across the different countries included in this study would allow for meaningful comparisons across different geographical regions, socioeconomic, and clinical characteristics.
The primary objective of the study was to estimate the overall point prevalence of FA among children, adolescents, and adults in each of the included countries. The secondary objectives were to assess the impact of FA on participants' HRQoL using different patient reported outcomes’ validated instruments, and to develop a risk stratification framework to define severity risk profiles based on QoL impact and clinical severity risk of FA.
Methods
Study design
This was a cross-sectional, international, population-based study using an online survey, that collected self-reported data on FAs, between October 2022 and February 2023, from caregivers (including parents, legal guardians, or other caregivers) of children or adolescents, as well as from adults. Participants were recruited from general population panels in the United States, Canada, the United Kingdom, France, Germany, Italy, Spain, Japan, and China.
Study population
The study was planned to be conducted among caregivers with children between 6 months and <18 years of age living in the household, as well as adults (18 to ≤65 years) who were solicited from online general population panels.
The inclusion criteria for caregivers of children were: 1) having children aged between 6 months and <18 years who lived in the household, 2) having provided an online informed consent to participate; adolescents between 12 and less than 18 years (<20 years for Japan) who were able to read and understand the study specificities, 3) being able to read the native country language, and 4) being a member of an online panel.
The inclusion criteria for the adult population were: 1) being aged between 18 and 65 years old, 2) having provided an online informed consent to participate, 3) being able to read and write in the native country's language and 4) being a member of an online panel.
Participants who reported FA but also reported food protein-induced enterocolitis syndrome and/or inflammatory bowel disease and/or diabetes type I or II, and those who reported FA to only gluten were excluded from the survey to avoid the possibility of mistaking these conditions with FA-specific inflammatory responses. Participants who did not qualify as having FA confirmed through either a self-reported physician diagnosis or a through an allergic reaction symptom to at least 1 food allergen were also excluded from further participation in the survey.
Sample size calculation
The country-specific sample sizes were estimated based on the expected FA prevalence in each country with a high precision between 0.5% and 1%, a 95% confidence interval, and considering the need for a sufficient number of participants with FA that would allow for the analysis of HRQoL and the severity risk framework. The estimated sample sizes were as follows: United States (4212 children, 4525 adults), Canada (3971 children, 4016 adults), France (4249 children, 4258 adults), Germany (4154 children, 4305 adults, Italy (4143 children, 4656 adults), Spain (4267 children, 4467 adults), the United Kingdom (4573 children, 4797 adults), Japan (4263 children, 4263 adults), and China (4238 children, 4298 adults).
To optimize the representativeness of the sample of participants in each country, quotas were set up for gender and age of children or adults, region of residence, race/ethnicity/origin (United States, Canada, and United Kingdom only) and current residence (urban/rural split). A soft quota was established for caregivers’ employment status to recruit a balanced representation of this characteristic.
Ethical considerations
Participation in the study was voluntary, and prior to questionnaire initiation participants agreed to an online informed consent form (online disclaimer). Oral assent was requested from adolescents before their participation in the questionnaire after approval from their caregiver. After initiation, participants could stop answering the online survey at any time. The protocol, screener and questionnaire were submitted for review to a US-based institutional review board (IRB) (Sterling IRB), the Cerner Enviza independent IRB, and to local ethics committees (EC) in Spain and Japan (Comité Ético de Investigación Clínica del Hospital Clínic de Barcelona and Hillside Clinical EC, respectively). Participant responses remained confidential, and data were pseudonymized and held on a secure server.
Study instrument
The study instrument, consisting of a screener and a main questionnaire, was collaboratively developed by an expert panel of clinicians specializing in FA, health services researchers, and epidemiologists. The selection of items was based on the experts' knowledge and experience as well as the published literature.
The first section of the screener collected data on sociodemographic characteristics, such as country and region of residence, sex, age, ethnicity (United States, Canada, and United Kingdom only), employment, education level, area of residence (urban/rural), number and age of children for caregivers, and self-reported presence of medical conditions, including asthma, allergic rhinitis, and atopic dermatitis. The purpose of this section was to ensure that participants met the eligibility criteria and pre-defined quotas. The second section collected self-reported FA variables related to the definition of FA and calculation of FA prevalence, including type of food allergen the participant was allergic to, physician diagnosis of FA and type of tests used for diagnosis, allergic reactions experienced in the past, symptoms of the most severe allergic reaction, and the time for appearance of symptoms after ingesting the food.
Participants who met the inclusion criteria completed a 30-min online questionnaire which captured data on FA-related health resources use, medical history, health-related quality of life (measured with validated scales), and the management of FA.
Prior to survey launch, qualitative cognitive interviews were conducted with twelve eligible respondents (including caregivers of children with FA and adults with FA) from the United States and the United Kingdom to pre-test the questionnaire. The purpose of the cognitive interviews was to ensure that the questions were unambiguous and to establish face validity; the questionnaire was reviewed and adapted based on respondents’ feedback before launching the main survey.
Data collection
Recruitment was done through direct web-based invitations to participate in the study. The invitations did not contain any mention of the survey objective to avoid biasing individuals' responses. After accepting the invitation, participants were screened to assess their eligibility according to the inclusion and exclusion criteria. Participants who met the inclusion criteria were asked to sign an online informed consent form before initiation of the questionnaire. Responses from adults were collected directly, while responses relating to the pediatric group were collected by proxy from their caregiver. Adolescents were able to answer sections of the questionnaire directly, including perceived FA severity, FA diagnosis and severe reaction, FA-related hospitalization, emergency room and outpatient visits, epinephrine auto-injector use, and QoL/HRQoL for adolescents’ burden.
Study outcomes
Definition of the 4 types of evidence for FA
To estimate the prevalence of FA, the occurrence of FA was collected at an allergen-specific level. Participants were asked to report their history of allergies to specific foods, either 1 or multiple food allergens. Participants could select more than 1 food allergen from a predefined list (Table 1) which consisted of staple and non-staple foods as defined by the panel of experts and according to the literature. Staple foods were defined as those that were consumed most routinely and in such quantities that they constituted a predominant part of a standard diet for an individual or a population group in the countries included in the study. Staple and non-staple foods are listed in Table 1. The prevalence of FA was reported per food allergen for 6 prioritized allergens (peanuts, milk and/or dairy products, shrimp, shellfish, tree nuts, and eggs [raw or cooked]) identified by the panel of FA specialists.
Table 1.
List of food allergens including staple and non-staple foods.
| Food allergen main category | Food allergen subcategories |
|---|---|
| Fruits and vegetablesa | Celery |
| Kiwi | |
| Strawberry | |
| Apple | |
| Carrot | |
| Peach | |
| Other | |
| Eggs (raw or cooked) | Raw |
| Cooked | |
| Milk (fresh or cooked) and/or dairy products | Fresh |
| Cooked | |
| Dairy products | |
| Peanuts | |
| Soy/soya/soybeans | |
| Other legumes (lupin, peas, etc.)a | Lupin |
| Peas | |
| Other | |
| Tree nuts | Cashews |
| Pecans | |
| Walnuts | |
| Pistachios | |
| Hazelnuts | |
| Almonds | |
| Other | |
| Mustarda | |
| Seeds including sesamea | |
| Fin fish (cod, tuna, salmon, etc.) | |
| Shrimp | |
| Shellfish other than shrimp (lobster, mussels, etc.) | Mollusks (slugs, octopus, clams) |
| Crustaceans (crabs, lobsters, crayfish, krill, prawns, barnacles) | |
| Wheat and wheat varieties, rye, barley, and oats | |
Foods that are considered non-staples. Staple and non-staple foods were defined by the panel of experts based on publications showing the most consumed foods in the study countries. Staple foods were defined as those foods that are consumed most routinely and in such quantities that they constitute a predominant part of a standard diet for an individual or a population group in the countries included in the study.
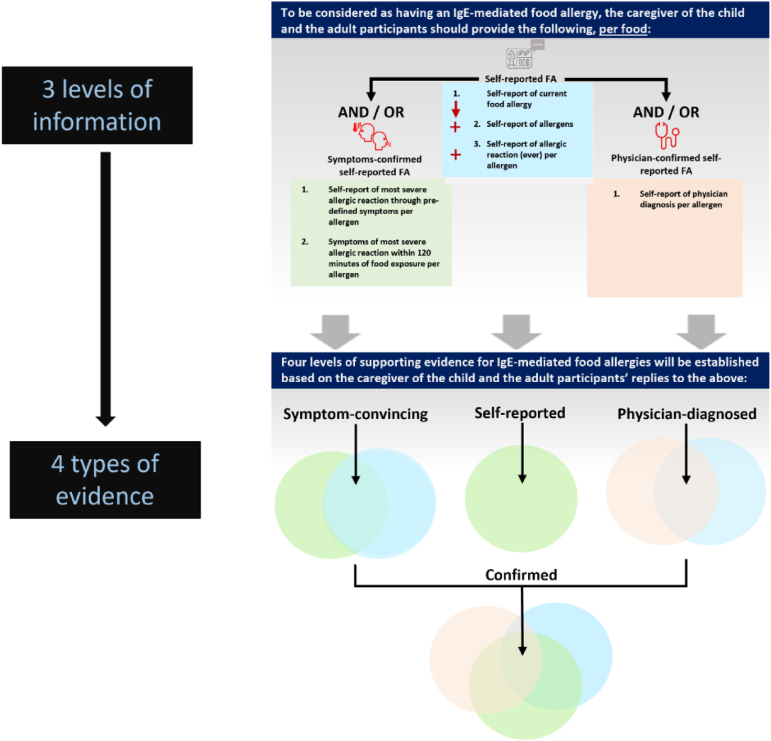
Based on the information collected, 4 case definitions of FA were created with increasing robustness (Fig. 1). To be considered as having "self-reported" IgE-mediated FA, participants had to self-report a current FA to 1 or more allergens. If a physician diagnosis of FA was self-reported along with the "self-reported" FA, they were considered as having "physician-diagnosed" FA. Participants were categorized as having "symptom-convincing" FA if they had a "self-reported" FA with symptoms indicative of FA defined as self-reported stringent symptoms of their most severe allergic reaction (based on a list of stringent symptoms adapted from Gupta et al, shown in Table 2)7 that occurred within 120 min of food exposure, with or without "physician-diagnosed" FA. A "symptom-convincing" FA, accompanied by a "physician-diagnosed" FA, was considered as "confirmed FA".
Fig. 1.
Flow chart of the criteria for the 4 types of evidence of FA (self-reported, physician-diagnosed, symptom-convincing, and confirmed). Note: Self-reported FA: Self-report of current FA to 1 or more allergens. Physician-diagnosed FA: Self-report of a current FA to 1 or more allergens with a physician diagnosis (regardless of the presence of symptoms indicative of FA). Symptom-convincing FA: Self-report of a current FA to 1 or more allergens with symptoms indicative of FA (regardless of the presence of a physician diagnosis). Confirmed FA: Self-report of current FA to 1 or more allergens with symptoms indicative of FA and with a physician diagnosis. Oral allergy syndrome (AOS) is excluded from the definition of the symptom-convincing FA and the confirmed FA, but it is included in the definition of self-reported and physician-diagnosed FA due to the absence of symptom-based FA confirmation.
Table 2.
List of food allergy related symptoms per organ system, including stringent symptoms
| Systems/organs involving symptoms | Presented symptoms within each system/organ |
|---|---|
| Skin and/or mouth symptoms | Hives (itchy, raised, red or skin-colored bumps or welts on the body, mouth, or throat) |
| Itchy skin - except lips, mouth, and/or tongue | |
| Itchy lips or mouth | |
| Rash (blotches on skin that may be itchy or red) | |
| Lip and/or tongue swelling | |
| Swelling - except lips, mouth, and/or tongue | |
| Flushing (hot feeling in skin with or without redness) | |
| Difficulty swallowing | |
| Sore or scratchy throat | |
| Throat tightening | |
| Mouth or throat tingling | |
| Numbness in mouth | |
| Respiratory symptoms | Chest tightening |
| Sudden appearance of hoarse voice | |
| Sneezing | |
| Shortness of breath | |
| Nasal congestion | |
| Sudden appearance of repetitive cough | |
| Trouble breathing | |
| Wheezing | |
| Gastrointestinal symptoms | Belly pain |
| Cramps | |
| Diarrhea | |
| Nausea | |
| Vomiting | |
| Dry heaving | |
| Cardiovascular and/or heart symptoms | Chest pain |
| Rapid heart rate | |
| Fainting, dizziness, or feeling light-headed | |
| Low blood pressure (hypotension) | |
| Other symptoms | Anxiety |
| Feeling of impending doom | |
| Headache | |
| Other | |
| Symptoms in bold represent stringent symptoms | |
Validated HRQoL scales used to evaluate FA impact on quality of life
FA burden on health-related quality of life was assessed among participants with symptom-convincing FA using several validated scales. These included the Food Allergy Quality of Life Questionnaire (FAQLQ),29, 30, 31 the Food Allergy Independent Measure (FAIM)32 and the Short Form Six-Dimensions health utility survey version 2 (SF-6Dv2).33,34 Caregivers of children aged between 6 months and 11 years completed the FAQLQ Parental Form (PF) and the FAIM PF.35 Adolescents aged between 12 and 17 years completed the FAQLQ Teenage Form (TF) and the FAIM TF themselves. Caregivers of children of all ages completed the Food Allergy QoL – Parent Form (FAQLQ-PF) and the SF-6Dv2. Adults completed the FAQLQ Adult Form (AF), the FAIM AF, and the SF-6Dv2.
Definition of the patient risk stratification framework
Currently, there is no gold standard for defining the severity of FA. To address this gap, this study aimed to classify patients into severity risk profiles based on 2 factors: 1) HRQoL impact of FA as a proxy for the degree of FA causing anxiety or substantially restricting patients’ QoL, and 2) the presence of risk factors indicative of having a lower or higher clinical severity of FA. The QoL impact of FA was dichotomized into 2 groups based on the total score of FAQLQ and FAIM, independently: low QoL impact, defined as a QoL score of less than 4 (scores range from 1 to 7, with a higher score indicating higher burden), and high QoL impact, defined as a score of 4 or more. Clinical severity was examined using a latent class analysis (LCA) designed to identify clusters of patients based on a variety of clinical characteristics. The LCA is described in detail in the statistical analysis section. As a final step of the framework, a matrix was developed that crossed the QoL impact scores independently from the FAQLQ and FAIM, and the clinical severity clusters to generate 4 severity risk profiles as follows:
-
-
Group 1: non-severe patients (low QoL impact + lower risk of clinical severity)
-
-
Group 2: at-risk patients (low QoL impact + higher risk of clinical severity)
-
-
Group 3: highly impacted QoL patients (high QoL impact + lower risk of clinical severity)
-
-
Group 4: severe patients (high QoL impact + higher risk of clinical severity)
Statistical analysis
Descriptive statistics
Analysis was mainly descriptive. Quantitative variables were summarized using means, standard deviations, medians, interquartile ranges (IQR), minimums, and maximums. Categorical variables were summarized using frequencies and percentages.
Prevalence of FA
Point prevalences (and 95% confidence intervals) were calculated for each type of evidence of FA: self-reported, physician-diagnosed, symptom-convincing, and confirmed FA. Estimates were based on the total number of participants who entered the study and who were part of the predefined quotas in each country. Since not all quotas were met during recruitment, minor weighting adjustments were applied at country level to have the structure of the participants best represent that of the general population in terms of sex, age, region of residence, race and ethnicity, and urban versus rural split in each country.
To avoid overestimating and underestimating the true prevalence of FA, prevalence was assessed in participants with symptom-convincing FA. Participants with self-reported FA only without an additional level of evidence would lead to an overestimation of prevalence. Physician-level evidence can be scarce among individuals who practice food avoidance rather than consulting a physician for a medical diagnosis.
Latent class analysis for clinical severity in the patient risk stratification framework
Clinical severity was explored using a latent class analysis (LCA), which is a statistical method used to identify subgroups within a larger population with similar characteristics or behaviors that are categorical rather than continuous. LCA has previously been used to identify phenotypes of FA based on risk indicators of developing severe allergic reactions.36 An LCA assumes that predictor variables are independent of one another since correlation can result in biased estimates of the class membership and misclassification errors. The strength of association between predictors was assessed using Phi and Cramer's coefficients. The limit for the coefficients was set at 0.2, which indicated a weak correlation and deemed the variables fitting to be included in the LCA model. Upon discussion with the panel of FA specialists and based on existing literature,36,37 5 factors were included in the LCA model: single/multiple FA, absence/presence of asthma, atopic dermatitis and allergic rhinitis, current epinephrine auto-injector use/prescription and number of severe FA reactions in the past year.
Following the rules involved in an LCA, and testing different models that included the potential predictors, the final best fitting LCA model clustered participants with self-reported FA into 2 clusters. As shown in Table 3, indicators of cluster 1, representing low clinical severity, were those having no severe FA reaction within the past year, being mono-allergic, not having any of the comorbidities, and not having current epinephrine auto-injector use or a prescription. In cluster 2, representing high clinical severity, membership included having at least 1 severe FA reaction within the past year, being allergic to 2 or more staple foods, or 3 or more non-staple foods, having 1 of the aforementioned comorbidities, and current epinephrine auto-injector use or prescription.
Table 3.
Indicators of class membership resulting from latent class analysis.
| Indicators of high risk for severe reactions | Indicators of lower risk (cluster 1) | Indicators of higher risk (cluster 2) |
|---|---|---|
| Number of severe food allergy reactions within past year | 0 | 1 or more |
| Multi-allergic | Mono-allergic | Allergic to 2 or more staplea foods |
| Allergic to 3 or more non-stapleb foods | ||
| Comorbid asthma | No | Yes |
| Comorbid allergic rhinitis | No | Yes |
| Comorbid atopic dermatitis | No | Yes |
| Current epinephrine auto-injector use or prescription | No | Yes |
Staple and non-staple foods were defined by the panel of experts based on publications showing the most consumed foods in the study countries.
Staple foods were defined as those foods that are consumed most routinely and in such quantities that they constitute a predominant part of a standard diet for an individual or a population group in the countries included in the study. See Table 1 for details.
Non-staple foods were foods other than staple foods. See Table 1 for details.
Discussion
The methodology of this study was developed in collaboration with a panel of FA specialists to standardize the reporting of FA prevalence globally, and its burden on HRQoL. The methodology used for recognizing FA relied on a survey structured with simple, straightforward, probes to assess FA reactions (list of stringent symptoms and timing of reaction), which may be applied universally regardless of the level of awareness to FA or access to specialized care. Unlike previously published studies reporting FA through a multitude of methods and diagnostic tools, this is a study where FA was reported in a systematic manner across the participating countries. From self-reported data, 4 different case definitions were created to categorize FA, increasing in robustness from self-reported FA to confirmed FA, with each prevalence level ensuring a lower impact of prevalence overestimation due to the self-reporting nature of the methodology. The 4 case definitions balance the risk of overestimating FA prevalence, such as with "self-reported FA", against the risk of underestimation if FA were recognized only through a physician diagnosis or a clinical test. Whilst biological confirmation (eg, oral food challenge, specific IgE levels) would strengthen the robustness of evidence, in the real-world setting where there are disparities in healthcare access, this is not always achievable. To address this limitation, the ASSESS FA study employed objective criteria to recognize FA based on a list of stringent symptoms indicative of FA as defined by FA experts.
Another objective of the study was to develop a risk stratification framework to identify different patient profiles, and eventually help researchers and clinicians in stratifying their patients based on their risk of experiencing severe FA reactions and the degree to which having food allergies impacts their quality of life.38 The FAQLQ and FAIM were used separately as building blocks of the framework rather than combined because they represent different facets of FA burden. FAQLQ is a disease-specific questionnaire that assesses the social and dietary limitations of living with FA caused by food safety concerns and negative emotions elicited by such restrictions.39 FAIM assesses the uncertainty and anxiety related to what could happen if patients were to have a FA reaction, which is a strong predictor of physical and mental health status.39 The LCA has been an innovative method, previously used to identify clinical severity groups among patients.36 The Akarsu et al study revealed 3 classes, or clinical phenotypes of FA: multi-allergic patients with asthma and eczema comorbidity, multi-allergic patients with persistent eczema, and mono-allergic patients with only an asthma comorbidity.36 Those results were similar to the results of the LCA in the ASSESS FA study. Recently, the Definition of Food Allergy Severity (DEFASE) project, which was conducted by the World Allergy Organization, developed a severity classification system based on 5 factors: symptoms of food allergy (FA) reactions, treatment needed, FA-related QoL, and economic burden.37 Taking a similar approach, the ASSESS FA risk stratification framework aimed to take a holistic perspective on patient risk assessment by including the psychological impact of FAs as evaluated by the FAQLQ/FAIM and the clinical presentation of FAs as evaluated by the LCA. In our framework, “severe” patients had to demonstrate a higher impact on their QoL and a higher risk of clinical severity. The latter includes experiencing at least 1 severe reaction within the past 12 months, being allergic to more than 1 staple food, having at least 1 atopy symptom, and currently having a prescription for or using an epinephrine auto-injector prescription. Due to the direct-to-patient, self-reported nature of this study, there is always a residual risk of misinformation bias leading to misclassification. Nonetheless, the framework is designed to help clinicians clearly classify their patients based on multiple factors. Nonetheless, the framework still needs to be tested and validated before it can be considered an approved clinical tool for assessing and managing FA patients. Once validated, this tool could help tailor management strategies for patients based on their risk profiles. The focus could be either on psychological interventions, such as cognitive behavioral therapy, or on pharmacological treatments that reduce the severity of allergic reactions, or on a combination of both.
This study has several limitations that pose a risk of multiple biases. The recruitment of participants from online consumer panels could have resulted in selection bias and limited generalizability to the general population. However, this bias was mitigated by setting recruitment quotas and applying weight adjustments at the country level during the analysis. Additionally, internet accessibility, specifically in the participating countries, was generally broad with rates ranging around 90%.40, 41, 42, 43 Selection bias may also have arisen from the scope countries that are high-income countries with relatively high levels of education, which could impact FA reporting. Nevertheless, the methodology used a simple questionnaire to objectively assess stringent symptoms of FA without requiring a physician diagnosis or test, which makes it universally applicable regardless of socio-economic settings or levels of FA awareness. This is why this study recommends the use of the ‘symptom-convincing FA’ case definition to report the prevalence of FA. The reliance on self-reported data directly from participants could have introduced potential recall bias or misclassification bias. However, relying upon self-reported data allows for a broader population to be reached which may better represent the general population where not all individuals have access to formal testing. Furthermore, some individuals may not seek a physician diagnosis but rather simply avoid allergens to which they previously experienced a reaction. However, some of the bias was limited by the use of validated tools such as FAQLQ, FAIM, and SF-6Dv2. Also, the use of clear, straightforward language in the questionnaire (which was reviewed for comprehensibility through cognitive interviews before the survey launch) facilitated correct interpretation of questions and enhanced accurate recall.
Conclusion
The ASSESS FA study methodology, developed with the input of food allergy specialists, established a strategy for estimating prevalence of FA in a consistent manner across countries. By utilizing 4 definitions of food allergy, it minimized an over-estimation of prevalence (when relying solely on self-reported FA) and an under-estimation of prevalence (when requiring a physician diagnosis of FA). Furthermore, a risk stratification framework categorized patients into severity risk profiles based on both clinical factors and the impact FA has on patients’ quality of life.
A strength of this study is that, to our knowledge, this is the first study to comprehensively assess the impact of FA on the HRQoL of pediatric and adult patients and their caregivers. A domain very specific to this condition due to its duration and impact on everyday life.
The applicability of the prevalence definitions and the risk stratification framework is multidimensional and flexible. The methodology of the ASSESS FA study may benefit patients and caregivers by enhancing the clinical assessment of patients, may increase awareness among patients and physicians of the deleterious effects of over- and under-estimating FA prevalence, and may have important implications for future research on the burden of FAs.
Abbreviations
DEFASE: Definition of Food Allergy Severity, FA: Food allergy; FAIM: Food Allergy Independent Measure; FAQLQ: Food Allergy Quality of Life Questionnaire, HRQoL: Health-related quality of life; IgE: Immunoglobulin-E; IQR: Interquartile range; LCA: Latent class analysis; QoL: Quality of life; SF-6Dv2: Short Form Six-Dimensions health utility survey version 2.
Ethics statement
The study was reviewed and approved by the Sterling Institutional Review Board, the Cerner Enviza independent Institutional Review Board, and the following local ethics committees: Comité Ético de Investigación Clínica del Hospital Clínic de Barcelona in Spain, and Hillside Clinical EC in Japan.
Authors’ contributions
Concept and design: Gupta, Fiocchi, Tassinari, Marvel, Mnif, Dritsa, Reed, LeCalvé
Analysis and data interpretation: Gupta, Fiocchi, Mnif, Reed, Hleyhel, Dritsa, LeCalvé, Moussallem, Tassinari, Marvel
Manuscript drafting: Hleyhel, Moussallem
Manuscript reviewing: Gupta, Mnif, Reed, Hleyhel, Dritsa, LeCalvé, Moussallem, Tassinari, Marvel, Fiocchi
Supervision: Marvel, Reed, Fiocchi
Disclosure statement on AI use
Nothing to disclose.
Funding
The study was sponsored by Novartis Pharma AG, Basel, Switzerland.
Declaration of competing interest
Ruchi Gupta receives research support from the National Institutes of Health (NIH) (R21 ID # AI135705, R01 ID # AI130348, U01 ID # AI138907), Food Allergy Research & Education (FARE), Melchiorre Family Foundation, Sunshine Charitable Foundation, The Walder Foundation, UnitedHealth Group, Thermo Fisher Scientific, Novartis, and Genentech. She serves as a medical consultant/advisor for Genentech, Novartis, Aimmune LLC, Allergenis LLC, and Food Allergy Research & Education (FARE). Dr. Gupta has ownership interest in Yobee Care, Inc. She is currently employed by Ann & Robert H. Lurie Children's Hospital of Chicago and is a Professor of Pediatrics & Medicine at Northwestern University Feinberg School of Medicine. Alessandro Fiocchi declares the receipt of honoraria or consulting fees by the following companies – Danone, Abbott, Aimmune, Ferrero, Novartis, Sanofi, Stallergenes. Tarek Mnif, Stefani Dritsa, Suzanne Reed, Mira Hleyhel, Perrine Le Calvé, Alissar Moussallem: Oracle Life Sciences, France (a company that received research funds from Novartis Pharma AG. during the conduct of the study) – employees. Paolo Tassinari, Jessica Marvel: Novartis Pharma AG – employees, may hold stock and/or stock options in the company.
Acknowledgment
We thank Manik Kalra from Novartis Pharma AG for coordinating the study manuscript, and Bastien Vincent from Oracle Life Sciences France for conducting all the statistical analyses.
Footnotes
Full list of author information is available at the end of the article.
References
- 1.Sicherer S.H., Sampson H.A. Food allergy. J Allergy Clin Immunol. 2010 Feb 1;125(2):S116–S125. doi: 10.1016/j.jaci.2009.08.028. [DOI] [PubMed] [Google Scholar]
- 2.Burks A.W., Tang M., Sicherer S., et al. ICON: food allergy. J Allergy Clin Immunol. 2012 Apr;129(4):906–920. doi: 10.1016/j.jaci.2012.02.001. [DOI] [PubMed] [Google Scholar]
- 3.Chafen J.J.S., Newberry S.J., Riedl M.A., Bravata D.M., Maglione M., Suttorp M.J., et al. Diagnosing and managing common food allergies: a systematic review. JAMA. 2010;303(18):1848–1856. doi: 10.1001/jama.2010.582. [DOI] [PubMed] [Google Scholar]
- 4.Gupta R.S., Warren C.M., Smith B.M., et al. The public health impact of parent-reported childhood food allergies in the United States. Pediatrics. 2018 Dec;142(6) doi: 10.1542/peds.2018-1235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Clarke A.E., Elliott S.J., St Pierre Y., Soller L., La Vieille S., Ben-Shoshan M. Temporal trends in prevalence of food allergy in Canada. J Allergy Clin Immunol Pract. 2020 Apr;8(4):1428–1430.e5. doi: 10.1016/j.jaip.2019.10.021. [DOI] [PubMed] [Google Scholar]
- 6.McGowan E.C., Keet C.A. Prevalence of self-reported food allergy in the national health and nutrition examination survey (NHANES) 2007-2010. J Allergy Clin Immunol. 2013 Nov;132(5):1216–1219.e5. doi: 10.1016/j.jaci.2013.07.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Gupta R.S., Warren C.M., Smith B.M., et al. Prevalence and severity of food allergies among US adults. JAMA Netw Open. 2019 Jan 4;2(1) doi: 10.1001/jamanetworkopen.2018.5630. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Grabenhenrich L., Trendelenburg V., Bellach J., et al. Frequency of food allergy in school-aged children in eight European countries-the EuroPrevall-iFAAM birth cohort. Allergy. 2020 Sep;75(9):2294–2308. doi: 10.1111/all.14290. [DOI] [PubMed] [Google Scholar]
- 9.Pénard-Morand C., Raherison C., Kopferschmitt C., et al. Prevalence of food allergy and its relationship to asthma and allergic rhinitis in schoolchildren. Allergy. 2005 Sep;60(9):1165–1171. doi: 10.1111/j.1398-9995.2005.00860.x. [DOI] [PubMed] [Google Scholar]
- 10.Caffarelli C., Coscia A., Ridolo E., et al. Parents' estimate of food allergy prevalence and management in Italian school-aged children. Pediatr Int. 2011 Aug;53(4):505–510. doi: 10.1111/j.1442-200X.2010.03294.x. [DOI] [PubMed] [Google Scholar]
- 11.Lyons S.A., Burney P.G.J., Ballmer-Weber B.K., et al. Food allergy in adults: substantial variation in prevalence and causative foods across Europe. J Allergy Clin Immunol Pract. 2019;7(6):1920–1928.e11. doi: 10.1016/j.jaip.2019.02.044. [DOI] [PubMed] [Google Scholar]
- 12.Pereira B., Venter C., Grundy J., Clayton C.B., Arshad S.H., Dean T. Prevalence of sensitization to food allergens, reported adverse reaction to foods, food avoidance, and food hypersensitivity among teenagers. J Allergy Clin Immunol. 2005 Oct;116(4):884–892. doi: 10.1016/j.jaci.2005.05.047. [DOI] [PubMed] [Google Scholar]
- 13.Kanny G., Moneret-Vautrin D.A., Flabbee J., Beaudouin E., Morisset M., Thevenin F. Population study of food allergy in France. J Allergy Clin Immunol. 2001 Jul 1;108(1):133–140. doi: 10.1067/mai.2001.116427. [DOI] [PubMed] [Google Scholar]
- 14.Steinke M., Fiocchi A., Kirchlechner V., et al. Perceived food allergy in children in 10 European nations. A randomised telephone survey. Int Arch Allergy Immunol. 2007;143(4):290–295. doi: 10.1159/000100575. [DOI] [PubMed] [Google Scholar]
- 15.Bergmann K.C., Heinrich J., Niemann H. Current status of allergy prevalence in Germany: position paper of the environmental medicine commission of the robert koch institute. Allergo J Int. 2016;25:6–10. doi: 10.1007/s40629-016-0092-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kaneko M., Miyoshi T., Miyashita Y., et al. Food allergy in nursery children of Kawasaki city, Japan. Asian Pac J Allergy Immunol. 2021 Jun;39(2):117–123. doi: 10.12932/AP-151118-0439. [DOI] [PubMed] [Google Scholar]
- 17.Wang J., Liu W., Zhou C., et al. Multi-perspective observation on the prevalence of food allergy in the general Chinese population: a meta-analysis. Nutrients. 2022 Dec 6;14(23):5181. doi: 10.3390/nu14235181. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Wang X.Y., Zhuang Y., Ma T.T., Zhang B., Wang X.Y. Prevalence of self-reported food allergy in six regions of Inner Mongolia, northern China: a population-based survey. Med Sci Monit. 2018 Apr 1;24:1902–1911. doi: 10.12659/MSM.908365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Rancé F., Grandmottet X., Grandjean H. Prevalence and main characteristics of schoolchildren diagnosed with food allergies in France. Clin Exp Allergy. 2005 Feb;35(2):167–172. doi: 10.1111/j.1365-2222.2005.02162.x. [DOI] [PubMed] [Google Scholar]
- 20.Roehr C.C., Edenharter G., Reimann S., et al. Food allergy and non-allergic food hypersensitivity in children and adolescents. Clin Exp Allergy. 2004 Oct;34(10):1534–1541. doi: 10.1111/j.1365-2222.2004.02080.x. [DOI] [PubMed] [Google Scholar]
- 21.Sicherer S.H., Warren C.M., Dant C., Gupta R.S., Nadeau K.C. Food allergy from infancy through adulthood. J Allergy Clin Immunol Pract. 2020 Jun;8(6):1854–1864. doi: 10.1016/j.jaip.2020.02.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ballmer-Weber B.K. Food allergy in adolescence and adulthood. Chem Immunol Allergy. 2015;101:51–58. doi: 10.1159/000371669. [DOI] [PubMed] [Google Scholar]
- 23.Warren C., Turner P.J., Chinthrajah R.S., Gupta R. Advancing food allergy through epidemiology: understanding and addressing disparities in food allergy management and outcomes. J Allergy Clin Immunol Pract. 2021 Jan;9(1):110–118. doi: 10.1016/j.jaip.2020.09.064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Merrill K.A., Abrams E.M., Good S.V., et al. Associations between food allergy, country of residence, and healthcare access. Allergy Asthma Clin Immunol. 2022 Dec 6;18(1):103. doi: 10.1186/s13223-022-00745-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Golding M.A., Gunnarsson N.V., Middelveld R., Ahlstedt S., Protudjer J.L.P. A scoping review of the caregiver burden of pediatric food allergy. Ann Allergy Asthma Immunol. 2021 Nov;127(5):536–547.e3. doi: 10.1016/j.anai.2021.04.034. [DOI] [PubMed] [Google Scholar]
- 26.Golding M.A., Batac A.L.R., Gunnarsson N.V., Ahlstedt S., Middelveld R., Protudjer J.L.P. The burden of food allergy on children and teens: a systematic review. Pediatr Allergy Immunol. 2022 Mar;33(3) doi: 10.1111/pai.13743. [DOI] [PubMed] [Google Scholar]
- 27.Fong A.T., Ahlstedt S., Golding M.A., Protudjer J.L.P. The economic burden of food allergy: what we know and what we need to learn. Curr Treat Options Allergy. 2022 Sep 1;9(3):169–186. doi: 10.1007/s40521-022-00306-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Bilaver L.A., Chadha A.S., Doshi P., O'Dwyer L., Gupta R.S. Economic burden of food allergy: a systematic review. Ann Allergy Asthma Immunol. 2019 Apr 1;122(4):373–380.e1. doi: 10.1016/j.anai.2019.01.014. [DOI] [PubMed] [Google Scholar]
- 29.Flokstra-de Blok B.M.J., van der Meulen G.N., DunnGalvin A., et al. Development and validation of the food allergy quality of life questionnaire - adult form. Allergy. 2009 Aug;64(8):1209–1217. doi: 10.1111/j.1398-9995.2009.01968.x. [DOI] [PubMed] [Google Scholar]
- 30.van der Velde J.L., Flokstra-de Blok B.M.J., Vlieg-Boerstra B.J., et al. Test-retest reliability of the food allergy quality of life questionnaires (FAQLQ) for children, adolescents and adults. Qual Life Res. 2009 Mar;18(2):245–251. doi: 10.1007/s11136-008-9434-2. [DOI] [PubMed] [Google Scholar]
- 31.van der Velde J.L., Flokstra-de Blok B.M.J., de Groot H., et al. Food allergy-related quality of life after double-blind, placebo-controlled food challenges in adults, adolescents, and children. J Allergy Clin Immunol. 2012 Nov;130(5):1136–1143.e2. doi: 10.1016/j.jaci.2012.05.037. [DOI] [PubMed] [Google Scholar]
- 32.van der Velde J.L., Flokstra-de Blok B.M.J., Vlieg-Boerstra B.J., et al. Development, validity and reliability of the food allergy independent measure (FAIM) Allergy. 2010 May;65(5):630–635. doi: 10.1111/j.1398-9995.2009.02216.x. [DOI] [PubMed] [Google Scholar]
- 33.Brazier J.E., Mulhern B.J., Bjorner J.B., et al. Developing a new version of the SF-6D health state classification system from the SF-36v2: SF-6Dv2. Medical Care. 2020 Jun;58(6):557–565. doi: 10.1097/MLR.0000000000001325. [DOI] [PubMed] [Google Scholar]
- 34.Dufresne É., Poder T.G., Samaan K., et al. SF-6Dv2 preference value set for health utility in food allergy. Allergy. 2021 Jan 1;76(1):326–338. doi: 10.1111/all.14444. [DOI] [PubMed] [Google Scholar]
- 35.Cohen B.L., Noone S., Muñoz-Furlong A., Sicherer S.H. Development of a questionnaire to measure quality of life in families with a child with food allergy. J Allergy Clin Immunol. 2004 Nov;114(5):1159–1163. doi: 10.1016/j.jaci.2004.08.007. [DOI] [PubMed] [Google Scholar]
- 36.Akarsu A., Oksel Karakus C., Ocak M., et al. Exploring the heterogeneity of IgE-Mediated food allergy through latent class analysis. Int Arch Allergy Immunol. 2023;184(3):228–235. doi: 10.1159/000527534. [DOI] [PubMed] [Google Scholar]
- 37.Arasi S., Nurmatov U., Dunn-Galvin A., et al. Consensus on DEfinition of food allergy SEverity (DEFASE) an integrated mixed methods systematic review. World Allergy Organ J. 2021 Mar;14(3) doi: 10.1016/j.waojou.2020.100503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Muraro A., Dubois A.E.J., DunnGalvin A., et al. EAACI food allergy and anaphylaxis guidelines. Food allergy health-related quality of life measures. Allergy. 2014 Jul;69(7):845–853. doi: 10.1111/all.12405. [DOI] [PubMed] [Google Scholar]
- 39.DunnGalvin A., De BlokFlokstra B.M.J., Burks A.W., Dubois A.E.J., Hourihane J.O. Food allergy QoL questionnaire for children aged 0–12 years: content, construct, and cross-cultural validity. Clin Exp Allergy. 2008;38(6):977–986. doi: 10.1111/j.1365-2222.2008.02978.x. [DOI] [PubMed] [Google Scholar]
- 40.L’usage des technologies de l’information et de la communication par les ménages entre 2009 et 2024 − L’usage des technologies de l’information et de la communication par les ménages entre 2009 et 2024 | Insee [Internet]. [cited 2025 Jul 3]. Available from: https://www.insee.fr/fr/statistiques/8278698?sommaire=8278710.
- 41.Internet users, UK - Office for national statistics [Internet]. [cited 2025 Jul 3]. Available from: https://www.ons.gov.uk/businessindustryandtrade/itandinternetindustry/bulletins/internetusers/2020.
- 42.Internet . Pew Research Center; 2024. Broadband Fact Sheet [Internet]https://www.pewresearch.org/internet/fact-sheet/internet-broadband/ [cited 2025 Jul 3]. Available from: [Google Scholar]
- 43.Government of Canada SC . 2023. Digital society - canada at a glance.https://www150.statcan.gc.ca/n1/pub/12-581-x/2023001/sec11-eng.htm [Internet]. 2022 [cited 2025 Jul 3]. Available from: [Google Scholar]