Abstract
Immune checkpoints including PD-1 and CTLA-4 help to regulate the intensity and timeframe of the immune response. Since they become upregulated in cancer and prevent sufficient antitumor immunity, monoclonal antibodies against these checkpoints have shown clinical promise for a range of cancers. Multimodal treatment plans combining immune checkpoint inhibitors with other therapies, including photodynamic therapy (PDT), may help to expand treatment efficacy and minimize side effects. PDT’s cytotoxic effects are spatially limited by the light activation process, constraining PDT direct effects to the treatment field. The production of damage-associated molecular patterns and tumor-associated antigens from PDT can encourage accumulation and maturation of antigen-presenting cells and reprogram the tumor microenvironment to be more susceptible to therapies targeting immune checkpoints.
INTRODUCTION
The immune system’s potential to respond to a tumor is curbed by inhibitory ligands and receptors on cancer and immune cells. These immune checkpoints are crucial for regulating self-tolerance and minimizing damage to tissues during the immune response to pathogens but can be upregulated in the tumor microenvironment and lymph nodes in the presence of local and/or systemic malignant disease.
PD-1 (CD279) is a transmembrane receptor expressed on activated T cells and B cells, as well as natural killer cells and myeloid cells in certain scenarios, including on macrophages during viral infection (1,2). The major positive regulator of PD-1 on T cells is T-cell receptor (TCR) engagement, although γ-chain cytokine stimulation (e.g. IL-2) and transcription factors including forkhead box protein O1 (FOXO1) also promote sustained PD-1 expression (2–4). Its ligands PD-L1 (B7-H1, CD274) and PD-L2 (B7-DC, CD273) are expressed on a wider variety of cell types−leukocytes, nonhematopoietic cells and nonlymphoid tissues including many tumors (5). PD-L2 is mostly expressed on dendritic cells and monocytes, but can be induced on other cell types (6). Type 1 and type 2 interferon signaling (particularly IFNγ), as well as other inflammatory cytokines including GM-CSF, IL-17 and TNF-α, act to induce PD-L1 and PD-L2 (7–9). Moreover, myeloid-derived suppressor cells (MDSCs) in the tumor microenvironment can induce PD-L1 expression on tumor cells through an EGFR/MAPK-dependent mechanism (10). Upon binding to one of its ligands, PD-1 inhibits kinases involved in T-cell activation, proliferation and effector functions through SHP2.
CTLA-4 (cytotoxic T-lymphocyte-associated protein 4) is an immune checkpoint receptor constitutively expressed in Tregs and upregulated in other activated T cells (11). It binds to CD80 (B7–1) or CD86 (B7–2) ligands on antigen-presenting cells (APCs) like dendritic cells. It is a member of the immunoglobulin superfamily and is homologous to CD28, a T-cell costimulatory protein, and competes with CD28 for the CD80 and CD86 ligands.
Naïve T-cell activation requires both an antigen-specific signal to the TCR by peptides presented on major histocompatibility (MHC) molecules expressed on APCs, as well as a costimulatory signal from B7 binding to CD28. The spatial expression patterns of the PD-1 and CTLA-4 pathways show that CTLA-4 primarily regulates T-cell priming in lymph node tissues, while PD-1 controls effector T-cell function in peripheral tissues (12) (Fig. 1). PD-1 and PD-L1 are expressed at higher levels within the tumor microenvironment compared to lymph nodes, so PD-1 pathway blockade functions mainly by reinvigorating intratumoral T cells expressing both PD-1 and CD28 (13,14). Together, these stimulatory and inhibitory immune checkpoint molecules regulate immune homeostasis and allow for rapid adjustments as needed.
Figure 1.
Spatial arrangement of immune checkpoints. APCs like dendritic cells capture tumor-associated antigen signals, present them on MHC complexes and carry them to T cells in lymph nodes. CTLA-4 on T cells competes with CD28 for binding with ligands CD80/CD86 on APCs. αCTLA-4 can prevent CTLA-4 binding to CD80/CD86, encouraging T-cell activation. Activated T cells migrate to the tumor, engaging with MHC-antigen complexes on cancer cells. PD-1 can interact with PD-L1/PD-L2 expressed on tumors or myeloid cells, inhibiting T-cell targeting. Monocytic and granulocytic myeloid-derived suppressor cells (M-MDSCs/G-MDSCs) within the tumor microenvironment also express immune checkpoints that inhibit T-cell activity. αPD-1 can prevent PD-1/PD-L1 interactions, encouraging continued cytotoxic T-cell response.
Several immune checkpoint antibodies have increased survival for cancer patients, adjusting the balance of the immune system toward promoting tumor recognition. Initial approval of immune checkpoint blockade by the United States Food and Drug Administration (FDA) was in 2011 for the CTLA-4 inhibitor ipilimumab in the treatment of metastatic melanoma. Since then, additional FDA approvals have included nivolumab and pembrolizumab for PD-1 blockade and atezolizumab and durvalumab for PD-L1 blockade. As the use of immune checkpoint therapy has grown, biomarkers predictive of an individual’s response are still needed. Only a minority of cancer patients responds to checkpoint inhibition due to presumed need for high expression of PD-L1 on tumors and pre-existing tumor-infiltrating activated CD8+ T cells expressing PD-1, together with the necessity of neoantigen expression (15).
Immune-related adverse events in response to these therapies are still being defined. Potential clinical toxicities include pneumonitis, diarrhea and other gastrointestinal inflammation, and rheumatologic toxicities (16). There is a 5% incidence rate of pneumonitis in conjunction with PD-1/PD-L1 blockade (17), and this approximately doubles when given in combination with CTLA-4 inhibition (18). Toxicity profiles of immune checkpoint inhibitors also vary as a function of combination with chemotherapies (19) and ionizing radiation (20,21). For example, a phase I study to test the tolerability of pembrolizumab and radiation therapy in patients with metastatic or locally advanced urothelial cancer of the bladder was paused due to grade 3 bladder toxicities although the radiation dosing schedule is normally well-tolerated alone. The investigators plan to mitigate these in field toxicities by de-escalating the radiation dose (20).
IMMUNOMODULATING EFFECTS OF PHOTODYNAMIC THERAPY
Strategies to enhance T-cell infiltration may sensitize tumors to checkpoint blockade and improve their response rates. A number of approaches are underway to reprogram the tumor microenvironment to improve the potential of immune checkpoint blockade, including combinations of these drugs with chemotherapy or radiation therapy. In photodynamic therapy (PDT), where a light-activated photosensitive drug generates reactive oxygen species within areas of tumor tissue exposed to light, direct cytotoxic effects are constrained to the treatment field. In addition to PDT’s direct cytotoxicity, it can also stimulate the innate and adaptive immune responses by releasing inflammatory mediators into the tumor microenvironment (22) (Fig. 2). This facilitates antigen uptake by antigen-presenting cells (APCs) and eventual presentation to T cells and their priming and activation within lymph nodes (23).
Figure 2.
PDT immunomodulation. PDT of a tumor causes direct cell death through various mechanisms, leading to release of antigen and damage-associated molecular patterns (DAMPs), vasculature damage/platelet aggregation and release of inflammatory cytokines such as IL8 and IL6. This promotes the recruitment of immune cells like neutrophils which then modulate the subsequent immune system response (maturation of dendritic cells and T-cell activation) that can promote antitumor immunity.
Treatment of a tumor by PDT can enhance tumor immunogenicity and visibility to immune cells. Depending on the mode of cell death after PDT, a variety of damage-associated molecular patterns (DAMPs) is exposed on the cell surface or released by dying cells. These include heat shock proteins and cell surface phosphatidylserine that help to regulate the subsequent immune response (22,24). Thus, as a function of the treatment regimen, PDT leads to immunogenic cell death characterized by calreticulin translocation to the cell membrane, HMGB1 release and secretion of ATP (25–27). Extracellular ATP acts as a DAMP because it can chemoattract immune cells. Surface calreticulin facilitates the uptake of antigens by APCs, while HMGB1 promotes the synthesis of inflammatory cytokines including type 1 interferons through TLR4 signaling (26). These DAMPs and inflammatory mediators stimulated by PDT serve to activate innate immune cells through processes that evolved as part of the body’s immune response to pathogens.
Inflammatory cytokines (IL-6, IL-8, etc.) produced by PDT-induced tissue damage recruit myeloid cells like neutrophils and functionally immunosuppressive neutrophil/monocyte populations (myeloid-derived suppressor cells; MDSCs). PDT has been shown to increase the population of intratumoral neutrophils to over 30% of the total viable cell population at 24 h after treatment (28,29), although this does depend on the PDT dose and other treatment-dependent variables (30). Neutrophils are then involved in the coordination of the subsequent immune response by secreting chemotactic signals that recruit immature dendritic cells (31) and can influence dendritic cell activation following degranulation through release of various alarmins (32) or by release of TNFα (33). As a result of all these inflammatory processes, PDT can induce recruitment of CD8+ T cells into tumors (34), and PDT’s prevention of both local and distant tumor regrowth notably depends on the presence of CD8+ T cells as part of an intact adaptive immune system (35).
Just as PDT changes the makeup of immune cells in the tumor microenvironment, there are also changes in the balance of immune checkpoint expression in this environment. We have studied this in the murine mesothelioma line AE17 that has been transfected with soluble ovalbumin and human mesothelin (AE17ova, meso). Photofrin-PDT at 135 J cm−2 promotes a dramatic accumulation of Ly6G+ cells into tumors at 24 h, which is consistent with previous studies (28,29) (Fig. 3B). Moreover, in the post-PDT environment, there is significantly less PD-L1 expressed on the surface of Ly6G+ cells than on this cell type in untreated tumors (Fig. 3C,D). Consequent of this, at 24 h after PDT, PD-L1 expression levels on tumor-resident CD11b+ LygG+ cells are lower than in controls; however, there are roughly 10× more of these cells present in the tumor (Fig. 3).
Figure 3.
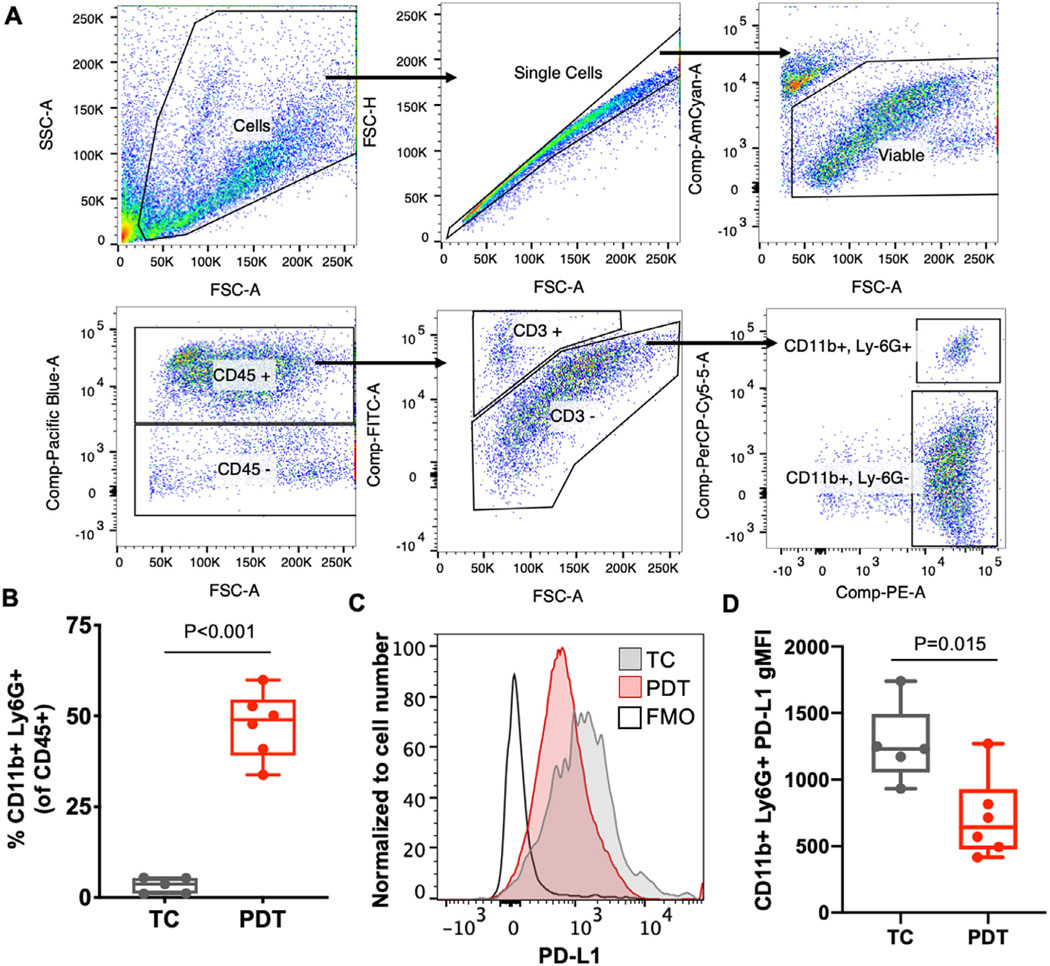
PDT increases the proportion Ly6G+ cells with low PD-L1 expression. AE17ova,meso (ovalbumin and mesothelin transfected) tumors were grown to 80–100 mm3 in C57BL/6 mice prior to Photofrin-PDT. PDT was delivered with Photofrin (i.v. at 5 mg kg−1) with a 24-h drug-light interval and a 632 nm light dose of 135 J cm−2 (75 mW cm−2). The next day, tumors were processed via enzymatic and mechanical digestion into single-cell suspensions for antibody staining and flow cytometry. Tumors were combined into groups of up to 2 for sufficient cell numbers, for a total of n = 6 for PDT and 5 for tumor control (TC). (A) Cells of interest were gated as single cells based on forward scattering, viable based on negative Aqua Live-Dead staining, CD45+, CD3−, CD11b+ and Ly6G+. (B) PDT significantly increases the percentage of CD11b+/Ly6G+ immune cells (as a percentage of all CD45+ immune cells) at 24 h after treatment compared to tumor control (TC) (P < 0.0001). (C) PD-L1 expression histograms show representative data for 1 of 2 independent experiments, including a PD-L1 fluorescence minus one (FMO) control. (D) TC tumors have an average PD-L1 geometric mean (gMFI) of 1265 (s.d. 294), and PDT-treated tumors have a PD-L1 gMFI of 713 (s.d. 309). Thus, Ly6G+ cells in PDT-treated tumors express significantly less PD-L1, albeit the presence of these cells is greatly increased (P = 0.015).
One possible explanation for a change in the context of immune checkpoint expression on tumor-resident Ly6G+ cells after PDT could be related to the large influx of myeloid cells that the treatment induces. If newly infiltrating CD11b+ Ly6G+ cells are neutrophils and therefore express less PD-L1 than pre-existing tumor-resistant MDSC cells, this would be consistent with a shift in balance to low PD-L1 expression on the tumor-resident population of CD11b+ Ly6G+ cells at the timepoint that was evaluated. Further support for this hypothesis could be provided by functional assays to differentiate between neutrophil and granulocytic MDSC classifications (36). Additional work to examine the roles and timecourse of neutrophil/MDSC involvement in PDT response as a function of their PD-L1 expression will be beneficial in informing the best strategies for combinations of PDT with immune checkpoint blockade.
POTENTIAL SYNERGISM OF PDT AND IMMUNE CHECKPOINT BLOCKADE
Ongoing research is exploring the potential synergism of PDT with inhibition of CTLA-4 and other immune checkpoints. For example, in double tumor models of colon carcinoma (either MC38 or CT26), addition of CTLA-4 blockade prior to Bremachlorin-PDT led to a significant reduction in tumor burden compared to either treatment alone (35). CTLA-4 blockade was more effective for the smaller secondary tumor than the primary tumor. Additionally, based on the results of CD8 depletion, this synergistic effect was also dependent on the presence of CD8 T cells.
Multiple factors could serve to promote synergy between PDT and immune checkpoint blockade. As noted earlier, PDT can lead to the release of DAMPs and tumor antigens that are essential to triggering an innate immune response, thereby serving as a good adjuvant to immunotherapy. Moreover, PDT can potentially alter immune checkpoint activity in the tumor microenvironment, sensitizing this environment to immune checkpoint blockade. For example, as found with other anticancer treatment modalities like fractionated radiotherapy (37), PDT could introduce changes in the tumor microenvironment that support upregulation of PD-L1 on tumors (38). Intratumoral PD-L1 expression has in some cases been correlated with higher response rates to PD-1/PD-L1 blockade, as reflected in PD-L1 threshold levels for FDA approval of immune checkpoint blockade antibodies for specific malignancies (39–41). Adjuvant IL6 produced by PDT-induced tissue damage specifically can augment PD-1 expression through STAT3 (42) and PD-L1 expression and stability through JAK1 (43).
Furthermore, tumor oxygenation often fluctuates during and shortly after PDT, and parameters such as fluence rate act to determine these fluctuations and the likelihood that oxygen levels will rebound after illumination (44–46). This transient hypoxia can potentially alter tumor and immune cell phenotype through PD-L1 upregulation dependent on HIF-1α signaling (47–49). Collectively, these potential changes in immune checkpoint homeostasis after PDT may act in combination with PDT’s promotion of immune cell infiltration to promote PD-1/PD-L1 blockade as a favorable complementary strategy. In the application of a multimodal treatment paradigm that combines PDT and immunotherapy, the primary tumor can be targeted with PDT and then immune checkpoint blockade may be used to reprogram the immune response to target remaining cancerous cells.
MULTIMODAL TREATMENT UTILIZING NANOMATERIALS
Third-generation PDT photosensitizers that employ antibodies or nanoparticle platforms have also been studied in combination with immune checkpoint blockade. Gao et al. developed an IRDye700 probe with streptavidin–biotin-HK peptide for use with diagnostic imaging and therapy of tumors by specific targeting of integrin α5β6 (named DSAB-HK) (50). Conjunction to a phthalocyanine photosensitizer provided for PDT targeting of α5β6-expressing cells in combination with near-infrared illumination. DSAB-HK itself allowed for imaging of primary subcutaneous 4T1 breast cancer tumors and lung metastases, while DSAB-HK PDT promoted accumulation and maturation of dendritic cells as shown by increases in CD11c+ CD83+ cells in tumor-draining lymph nodes and further supported by overall increases in serum levels of IL-1β and IL-12P70. When combined with anti-PD-1 immune checkpoint inhibition, DSAB-HK PDT significantly increased CD8 T-cell recruitment to the tumor and inhibited lung metastases (50). Additionally, Bao et al. have combined IRDye700 with the Fab fragment of an antibody targeted to CD276 (B7-H3), an immune checkpoint member of the B7/CD28 families that plays a regulatory role in T-cell priming. Targeting to tumor vasculature during PDT served to inhibit 4T1 primary and spontaneous lung metastatic tumor growth albeit it upregulated tumor PD-L1 levels (51). Anti-PD-1 or anti-PD-L1 further enhanced the efficacy of this treatment.
Nanoparticle delivery of photosensitizers can increase solubility of hydrophobic PDT photosensitizers or other therapies, prolong circulation in blood and allow for more selective accumulation in tumors (52,53). The nanoparticle surface can be modified with functional groups or targeting agents to improve biodistribution and tumor-specific uptake. A number of research groups have recently begun testing various nanoparticle–photosensitizer formulations for PDT in combination with immune checkpoint blockade against CTLA-4 (54,55) and PD-1/PD-L1 (52,56–58).
Immunogenic “nanoscale coordination polymer” (NCP) particles have a flexible design that permits multiple elements—for imaging, chemotherapy, molecular targets, etc., and are biodegradable in tissues. In a colon cancer model, delivery of oxaliplatin chemotherapy and photosensitizer (NCP@pyrolipid) PDT was an effective local therapy that also evoked systemic antitumor immunity and potentiated PD-L1 checkpoint blockade (58). Duan et al. also used a pyrolipid photosensitizer in combination with Zn-pyrophosphate (ZnP@pyro) nanoparticles, which significantly delayed orthotopic 4T1 tumor growth after 670 nm light activation. When combined with PD-L1 blockade, ZnP@pyrolipid PDT also prevented spontaneous lung metastases (52).
Upconversion nanoparticles (UCNPs) containing lanthanide ions can provide for PDT activation by converting long-wavelength NIR light through multiphoton absorption and single photon emission, which improves tissue penetration. Polymer-coated UCNPs coupled with the photosensitizer chlorin e6 and imiquimod (a TLR-7 agonist) stimulated the immune response against CT26 murine colorectal tumors after 980 nm light activation. In double tumor experiments, UCNP-Ce6-R837-based PDT temporarily controlled regrowth of the primary tumor, although effects on the distant tumor were more limited. Anti-CTLA-4 alone decreased tumor growth rates, but it did not lead to regression of either primary or contralateral tumors. Adding CTLA-4 blockade to PDT led to the control of both primary and contralateral flank tumors, with no regrowth of primary tumors and minimal regrowth of contralateral tumors (54).
Other studies have tested combinations of nanoparticle PDT with PD-1/PD-L1 intervention via RNA interference or small molecules (59) with the goal of improving response rates. Wang et al. developed a micelleplex with pheophorbide A as photosensitizer, an acid-activatable cationic micelle, and siRNA specific to PD-L1 (60). In B16-F10 melanoma tumors, PDT using a 671 nm laser or PD-L1 knockdown alone significantly inhibited over 60% of tumor growth, while the combination completely prevented tumor growth. Improvements in tumor control were revealingly accompanied by the recruitment of T cells to the tumor and an increase in their proliferation.
CONCLUSIONS
The cytotoxic effects of PDT are spatially limited by the light activation process, which constrains PDT’s direct effects to the local treatment field. PDT also serves to disrupt the pre-existing homeostatic balance in the tumor microenvironment, with the potential to support antitumor immunity. PDT-induced production of tumor-associated antigens and release of DAMPs can encourage maturation of antigen-presenting cells and reprogram the tumor microenvironment to be more susceptible to therapies targeting immune checkpoints. While the current monoclonal antibody strategy of immune checkpoint inhibition leads to a tumor response in only a small subset of patients, a multimodal treatment strategy combining PDT and immune checkpoint blockade may help to not only eradicate primary tumors but also control metastatic disease. These treatments can additionally be combined with nanoparticles to customize drug distribution and tumor-specific targeting. Revealingly, a clinical case report of a patient with head and neck squamous cell carcinoma receiving Redaporfin-PDT followed by nivolumab (anti-PD1) illustrates the potential success of combining PDT with immune checkpoint blockade and demonstrates the clinical relevance of the discussed combinations (61).
Acknowledgements−
This work was supported by NIH/NCI grants P01-CA087971, R21-CA223366 and R01-CA236362 and the Penn Translational Centers for Excellence in Mesothelioma Research and Radiation Oncology.
Biographies
AUTHOR BIOGRAPHIES

Gwendolyn M. Cramer is a postdoctoral researcher in the laboratories of Theresa Busch and Keith Cengel in the Department of Radiation Oncology at the University of Pennsylvania. She completed her Ph.D. at the University of Massachusetts Boston in 2017, where she studied the pancreatic cancer extracellular matrix as a determinant of cancer invasiveness and photodynamic therapy response. Her current research focuses on how surgically-induced inflammation in the mesothelioma tumor microenvironment can alter tumor response to photodynamic therapy.

Edmund K. Moon is Assistant Professor of Medicine at the Perelman School of Medicine at the University of Pennsylvania. He is an interventional pulmonologist with research focuses in the field of T cell-based immunotherapy and solid tumor immune microenvironment particularly in lung cancer and malignant pleural mesothelioma.

Keith A. Cengel is Associate Professor in the Department of Radiation Oncology at the University of Pennsylvania, the Director for the Photodynamic Therapy Program and the Executive Director of the Penn Mesothelioma and Pleural Diseases Program. His clinical practice centers on use of ionizing and non-ionizing (photodynamic therapy) radiation for thoracic and neuroendocrine cancers. His research centers on combining dosimetry, organ system physiology and immune/growth factor signaling to understand predict and modulate the therapeutic index of radiotherapy.

Theresa M. Busch, PhD is a professor and Associate Director of the Division of Research, Department of Radiation Oncology at the University of Pennsylvania. Her research interests encompass the study of tumor microenvironment as it relates to radiation therapy, including ionizing radiation and nonionizing radiation in the form of photodynamic therapy (PDT). Her laboratory has characterized how treatment parameters such as fluence rate, together with physiologic characteristics of the tumor microenvironment such as its oxygenation or blood flow, can individually or cooperatively act on radiotherapy response. Through elucidation of how PDT interacts with the tumor microenvironment, the Busch laboratory identifies photophysical, molecular targeted and immunological interventions with translational potential to optimize therapy outcome.
Footnotes
This article is part of a Special Issue dedicated to Dr. Thomas Dougherty.
REFERENCES
- 1.Ma CJ, Ni L, Zhang Y, Zhang CL, Wu XY, Atia AN, Thayer P, Moorman JP and Yao ZQ. (2011) PD-1 negatively regulates interleukin-12 expression by limiting STAT-1 phosphorylation in monocytes/macrophages during chronic hepatitis C virus infection. Immunology 132, 421–431. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Sharpe AH and Pauken KE. (2018) The diverse functions of the PD1 inhibitory pathway. Nat.. Rev. Immunol. 18, 153–167. [DOI] [PubMed] [Google Scholar]
- 3.Kinter AL, Godbout EJ, McNally JP, Sereti I, Roby GA, O’Shea MA and Fauci AS. (2008) The common gamma-chain cytokines IL-2, IL-7, IL-15, and IL-21 induce the expression of programmed death-1 and its ligands. J. Immunol. 181, 6738–6746. [DOI] [PubMed] [Google Scholar]
- 4.Staron MM, Gray SM, Marshall HD, Parish IA, Chen JH, Perry CJ, Cui G, Li MO and Kaech SM. (2014) The transcription factor FoxO1 sustains expression of the inhibitory receptor PD-1 and survival of antiviral CD8(+) T cells during chronic infection. Immunity 41, 802–814. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Buchbinder EI and Desai A. (2016) CTLA-4 and PD-1 pathways: Similarities, differences, and implications of their inhibition. Am. J. Clin. Oncol. 39, 98–106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Rozali EN, Hato SV, Robinson BW, Lake RA and Lesterhuis WJ. (2012) Programmed death ligand 2 in cancer-induced immune suppression. Clin. Dev. Immunol. 2012, 656340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Garcia-Diaz A, Shin DS, Moreno BH, Saco J, Escuin-Ordinas H, Rodriguez GA, Zaretsky JM, Sun L, Hugo W, Wang X, Parisi G, Saus CP, Torrejon DY, Graeber TG, Comin-Anduix B, Hu-Lieskovan S, Damoiseaux R, Lo RS and Ribas A. (2017) Interferon receptor signaling pathways regulating PD-L1 and PD-L2 expression. Cell Rep. 19, 1189–1201. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang TT, Zhao YL, Peng LS, Chen N, Chen W, Lv YP, Mao FY, Zhang JY, Cheng P, Teng YS, Fu XL, Yu PW, Guo G, Luo P, Zhuang Y and Zou QM. (2017) Tumour-activated neutrophils in gastric cancer foster immune suppression and disease progression through GM-CSF-PD-L1 pathway. Gut 66, 1900–1911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Wang X, Yang L, Huang F, Zhang Q, Liu S, Ma L and You Z. (2017) Inflammatory cytokines IL-17 and TNF-a up-regulate PD-L1 expression in human prostate and colon cancer cells. Immunol. Lett. 184, 7–14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhang Y, Velez-Delgado A, Mathew E, Li D, Mendez FM, Flannagan K, Rhim AD, Simeone DM, Beatty GL and Pasca di Magliano M. (2017) Myeloid cells are required for PD-1/PD-L1 checkpoint activation and the establishment of an immunosuppressive environment in pancreatic cancer. Gut 66, 124–136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wing K, Onishi Y, Prieto-Martin P, Yamaguchi T, Miyara M, Fehervari Z, Nomura T and Sakaguchi S. (2008) CTLA-4 control over Foxp3+ regulatory T cell function. Science 322, 271–275. [DOI] [PubMed] [Google Scholar]
- 12.Fife BT and Bluestone JA. (2008) Control of peripheral T-cell tolerance and autoimmunity via the CTLA-4 and PD-1 pathways. Immunol. Rev. 224, 166–182. [DOI] [PubMed] [Google Scholar]
- 13.Kamphorst AO, Wieland A, Nasti T, Yang S, Zhang R, Barber DL, Konieczny BT, Daugherty CZ, Koenig L, Yu K, Sica GL, Sharpe AH, Freeman GJ, Blazar BR, Turka LA, Owonikoko TK, Pillai RN, Ramalingam SS, Araki K and Ahmed R. (2017) Rescue of exhausted CD8 T cells by PD-1-targeted therapies is CD28-dependent. Science 355, 1423–1427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Zappasodi R, Merghoub T and Wolchok JD. (2018) Emerging concepts for immune checkpoint blockade-based combination therapies. Cancer Cell 34, 690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Fares CM, Van Allen EM, Drake CG, Allison JP and Hu-Lieskovan S. (2019) Mechanisms of resistance to immune checkpoint blockade: why does checkpoint inhibitor immunotherapy not work for all patients? Am. Soc. Clin. Oncol. Educ. Book 39, 147–164. [DOI] [PubMed] [Google Scholar]
- 16.Winer A, Bodor JN and Borghaei H. (2018) Identifying and managing the adverse effects of immune checkpoint blockade. J. Thorac. Dis. 10, S480–S489. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Puzanov I, Diab A, Abdallah K, Bingham CO, Brogdon C, Dadu R, Hamad L, Kim S, Lacouture ME, LeBoeuf NR, Lenihan D, Onofrei C, Shannon V, Sharma R, Silk AW, Skondra D, Suarez-Almazor ME, Wang Y, Wiley K, Kaufman HL and Ernstoff MS. (2017) Managing toxicities associated with immune checkpoint inhibitors: consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J. Immunother. Cancer 5, 95. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Naidoo J, Wang X, Woo KM, Iyriboz T, Halpenny D, Cunningham J, Chaft JE, Segal NH, Callahan MK, Lesokhin AM, Rosenberg J, Voss MH, Rudin CM, Rizvi H, Hou X, Rodriguez K, Albano M, Gordon RA, Leduc C, Rekhtman N, Harris B, Menzies AM, Guminski AD, Carlino MS, Kong BY, Wolchok JD, Postow MA, Long GV and Hellmann MD (2017) Pneumonitis in patients treated with anti-programmed death-1/programmed death ligand 1 therapy. J. Clin. Oncol. 35, 709–717. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Weber J, Hamid O, Amin A, O’Day S, Masson E, Goldberg SM, Williams D, Parker SM, Chasalow SD, Alaparthy S and Wolchok JD. (2013) Randomized phase I pharmacokinetic study of ipilimumab with or without one of two different chemotherapy regimens in patients with untreated advanced melanoma. Cancer Immun. 13, 7. [PMC free article] [PubMed] [Google Scholar]
- 20.Tree AC, Jones K, Hafeez S, Sharabiani MTA, Harrington KJ, Lalondrelle S, Ahmed M and Huddart RA. (2018) Dose-limiting urinary toxicity with pembrolizumab combined with weekly hypofractionated radiation therapy in bladder cancer. Int. J. Radiat. Oncol. Biol. Phys. 101, 1168–1171. [DOI] [PubMed] [Google Scholar]
- 21.Bang A, Wilhite TJ, Pike LRG, Cagney DN, Aizer AA, Taylor A, Spektor A, Krishnan M, Ott PA, Balboni TA, Hodi FS and Schoenfeld JD. (2017) Multicenter evaluation of the tolerability of combined treatment with PD-1 and CTLA-4 immune checkpoint inhibitors and palliative radiation therapy. Int. J. Radiat. Oncol. Biol. Phys. 98, 344–351. [DOI] [PubMed] [Google Scholar]
- 22.Reginato E, Wolf P and Hamblin MR. (2014) Immune response after photodynamic therapy increases anti-cancer and anti-bacterial effects. World J. Immunol. 4, 1–11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Nath S, Obaid G and Hasan T. (2019) The course of immune stimulation by photodynamic therapy: bridging fundamentals of photochemically induced immunogenic cell death to the enrichment of T-cell repertoire. Photochem. Photobiol. 95, 1288–1305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Zitvogel L, Kepp O and Kroemer G. (2010) Decoding cell death signals in inflammation and immunity. Cell 140, 798–804. [DOI] [PubMed] [Google Scholar]
- 25.Tanaka M, Kataoka H, Yano S, Sawada T, Akashi H, Inoue M, Suzuki S, Inagaki Y, Hayashi N, Nishie H, Shimura T, Mizoshita T, Mori Y, Kubota E, Tanida S, Takahashi S and Joh T. (2016) Immunogenic cell death due to a new photodynamic therapy (PDT) with glycoconjugated chlorin (G-chlorin). Oncotarget 7, 47242–47251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Galluzzi L, Buqué A, Kepp O, Zitvogel L and Kroemer G. (2017) Immunogenic cell death in cancer and infectious disease. Nat. Rev. Immunol. 17, 97–111. [DOI] [PubMed] [Google Scholar]
- 27.Turubanova VD, Balalaeva IV, Mishchenko TA, Catanzaro E, Alzeibak R, Peskova NN, Efimova I, Bachert C, Mitroshina EV, Krysko O, Vedunova MV and Krysko DV. (2019) Immunogenic cell death induced by a new photodynamic therapy based on photosens and photodithazine. J. Immunother. Cancer 7, 350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Davis RW, Snyder E, Miller J, Carter S, Houser C, Klampatsa A, Albelda SM, Cengel KA and Busch TM. (2019) Luminol chemiluminescence reports photodynamic therapy-generated neutrophil activity in vivo and serves as a biomarker of therapeutic efficacy. Photochem. Photobiol. 95, 430–438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Gollnick SO, Liu X, Owczarczak B, Musser DA and Henderson BW. (1997) Altered expression of interleukin 6 and interleukin 10 as a result of photodynamic therapy in vivo. Cancer Res. 57, 3904–3909. [PubMed] [Google Scholar]
- 30.Gollnick SO. (2012) Photodynamic therapy and antitumor immunity. J.. Natl. Compr. Canc. Netw. 10(Suppl 2), S40–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Kousis PC, Henderson BW, Maier PG and Gollnick SO. (2007) Photodynamic therapy enhancement of antitumor immunity is regulated by neutrophils. Cancer Res. 67, 10501–10510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Yang D, de la Rosa G, Tewary P and Oppenheim JJ. (2009) Alarmins link neutrophils and dendritic cells. Trends Immunol. 30, 531–537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Bennouna S and Denkers EY. (2005) Microbial antigen triggers rapid mobilization of TNF-alpha to the surface of mouse neutrophils transforming them into inducers of high-level dendritic cell TNF-alpha production. J. Immunol. 174, 4845–4851. [DOI] [PubMed] [Google Scholar]
- 34.Kabingu E, Vaughan L, Owczarczak B, Ramsey KD and Gollnick SO. (2007) CD8+ T cell-mediated control of distant tumours following local photodynamic therapy is independent of CD4+ T cells and dependent on natural killer cells. Br. J. Cancer 96, 1839–1848. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Kleinovink JW, Fransen MF, Löwik CW and Ossendorp F. (2017) Photodynamic-immune checkpoint therapy eradicates local and distant tumors by CD8. Cancer Immunol. Res. 5, 832–838. [DOI] [PubMed] [Google Scholar]
- 36.Bronte V, Brandau S, Chen SH, Colombo MP, Frey AB, Greten TF, Mandruzzato S, Murray PJ, Ochoa A, Ostrand-Rosenberg S, Rodriguez PC, Sica A, Umansky V, Vonderheide RH and Gabrilovich DI. (2016) Recommendations for myeloid-derived suppressor cell nomenclature and characterization standards. Nat. Commun. 7, 12150. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dovedi SJ, Adlard AL, Lipowska-Bhalla G, McKenna C, Jones S, Cheadle EJ, Stratford IJ, Poon E, Morrow M, Stewart R, Jones H, Wilkinson RW, Honeychurch J and Illidge TM. (2014) Acquired resistance to fractionated radiotherapy can be overcome by concurrent PD-L1 blockade. Cancer Res. 74, 5458–5468. [DOI] [PubMed] [Google Scholar]
- 38.O’Shaughnessy MJ, Murray KS, La Rosa SP, Budhu S, Merghoub T, Somma A, Monette S, Kim K, Corradi RB, Scherz A and Coleman JA. (2018) Systemic antitumor immunity by PD-1/PD-L1 inhibition is potentiated by vascular-targeted photodynamic therapy of primary tumors. Clin. Cancer Res. 24, 592–599. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Herbst RS, Soria JC, Kowanetz M, Fine GD, Hamid O, Gordon MS, Sosman JA, McDermott DF, Powderly JD, Gettinger SN, Kohrt HE, Horn L, Lawrence DP, Rost S, Leabman M, Xiao Y, Mokatrin A, Koeppen H, Hegde PS, Mellman I, Chen DS and Hodi FS. (2014) Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 515, 563–567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Topalian SL, Drake CG and Pardoll DM. (2012) Targeting the PD-1/B7-H1(PD-L1) pathway to activate anti-tumor immunity. Curr. Opin. Immunol. 24, 207–212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Davis AA and Patel VG. (2019) The role of PD-L1 expression as a predictive biomarker: an analysis of all US Food and Drug Administration (FDA) approvals of immune checkpoint inhibitors. J. Immunother. Cancer 7, 278. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Austin JW, Lu P, Majumder P, Ahmed R and Boss JM. (2014) STAT3, STAT4, NFATc1, and CTCF regulate PD-1 through multiple novel regulatory regions in murine T cells. J. Immunol. 192, 4876–4886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Chan LC, Li CW, Xia W, Hsu JM, Lee HH, Cha JH, Wang HL, Yang WH, Yen EY, Chang WC, Zha Z, Lim SO, Lai YJ, Liu C, Liu J, Dong Q, Yang Y, Sun L, Wei Y, Nie L, Hsu JL, Li H, Ye Q, Hassan MM, Amin HM, Kaseb AO, Lin X, Wang SC and Hung MC. (2019) IL-6/JAK1 pathway drives PD-L1 Y112 phosphorylation to promote cancer immune evasion. J. Clin. Invest. 129, 3324–3338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Busch TM, Hahn SM, Evans SM and Koch CJ. (2000) Depletion of tumor oxygenation during photodynamic therapy: detection by the hypoxia marker EF3 [2-(2-nitroimidazol-1[H]-yl)-N-(3,3,3-trifluoropropyl)acetamide ]. Cancer Res. 60, 2636–2642. [PubMed] [Google Scholar]
- 45.Busch TM, Wileyto EP, Emanuele MJ, Del Piero F, Marconato L, Glatstein E and Koch CJ. (2002) Photodynamic therapy creates fluence rate-dependent gradients in the intratumoral spatial distribution of oxygen. Cancer Res. 62, 7273–7279. [PubMed] [Google Scholar]
- 46.Henderson BW, Gollnick SO, Snyder JW, Busch TM, Kousis PC, Cheney RT and Morgan J. (2004) Choice of oxygen-conserving treatment regimen determines the inflammatory response and outcome of photodynamic therapy of tumors. Cancer Res. 64, 2120–2126. [DOI] [PubMed] [Google Scholar]
- 47.Corzo CA, Condamine T, Lu L, Cotter MJ, Youn JI, Cheng P, Cho HI, Celis E, Quiceno DG, Padhya T, McCaffrey TV, McCaffrey JC and Gabrilovich DI. (2010) HIF-1alpha regulates function and differentiation of myeloid-derived suppressor cells in the tumor microenvironment. J. Exp. Med. 207, 2439–2453. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Barsoum IB, Smallwood CA, Siemens DR and Graham CH. (2014) A mechanism of hypoxia-mediated escape from adaptive immunity in cancer cells. Cancer Res. 74, 665–674. [DOI] [PubMed] [Google Scholar]
- 49.Noman MZ, Desantis G, Janji B, Hasmim M, Karray S, Dessen P, Bronte V and Chouaib S. (2014) PD-L1 is a novel direct target of HIF-1 alpha., and its blockade under hypoxia enhanced MDSC-mediated T cell activation. J. Exp. Med. 211, 781–790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Gao L, Zhang C, Gao D, Liu H, Yu X, Lai J, Wang F, Lin J and Liu Z. (2016) Enhanced anti-tumor efficacy through a combination of integrin alphavbeta6-targeted photodynamic therapy and immune checkpoint inhibition. Theranostics 6, 627–637. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Bao R, Wang Y, Lai J, Zhu H, Zhao Y, Li S, Li N, Huang J, Yang Z, Wang F and Liu Z. (2019) Enhancing anti-PD-1/PD-L1 immune checkpoint inhibitory cancer therapy by CD276-targeted photodynamic ablation of tumor cells and tumor vasculature. Mol. Pharm. 16, 339–348. [DOI] [PubMed] [Google Scholar]
- 52.Duan X, Chan C, Guo N, Han W, Weichselbaum RR and Lin W. (2016) Photodynamic therapy mediated by nontoxic core-shell nanoparticles synergizes with immune checkpoint blockade to elicit antitumor immunity and antimetastatic effect on breast cancer. J. Am. Chem. Soc. 138, 16686–16695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Allison RR, Mota HC, Bagnato VS and Sibata CH. (2008) Bio-nanotechnology and photodynamic therapy–state of the art review. Photodiagnosis Photodyn. Ther. 5, 19–28. [DOI] [PubMed] [Google Scholar]
- 54.Xu J, Xu L, Wang C, Yang R, Zhuang Q, Han X, Dong Z, Zhu W, Peng R and Liu Z. (2017) Near-infrared-triggered photodynamic therapy with multitasking upconversion nanoparticles in combination with checkpoint blockade for immunotherapy of colorectal cancer. ACS Nano 11, 4463–4474. [DOI] [PubMed] [Google Scholar]
- 55.Wang Z, Zhang F, Shao D, Chang Z, Wang L, Hu H, Zheng X, Li X, Chen F, Tu Z, Li M, Sun W, Chen L and Dong WF. (2019) Janus nanobullets combine photodynamic therapy and magnetic hyperthermia to potentiate synergetic anti-metastatic immunotherapy. Adv Sci (Weinh) 6, 1901690. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Hu L, Cao Z, Ma L, Liu Z, Liao G, Wang J, Shen S, Li D and Yang X. (2019) The potentiated checkpoint blockade immunotherapy by ROS-responsive nanocarrier-mediated cascade chemo-photodynamic therapy. Biomaterials 223, 119469. [DOI] [PubMed] [Google Scholar]
- 57.Lan G, Ni K, Xu Z, Veroneau SS, Song Y and Lin W. (2018) Nanoscale metal-organic framework overcomes hypoxia for photodynamic therapy primed cancer immunotherapy. J. Am. Chem. Soc. 140, 5670–5673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.He C, Duan X, Guo N, Chan C, Poon C, Weichselbaum RR and Lin W. (2016) Core-shell nanoscale coordination polymers combine chemotherapy and photodynamic therapy to potentiate checkpoint blockade cancer immunotherapy. Nat. Commun. 7, 12499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Zhang R, Zhu Z, Lv H, Li F, Sun S, Li J and Lee CS. (2019) Immune checkpoint blockade mediated by a small-molecule nanoinhibitor targeting the PD-1/PD-L1 pathway synergizes with photodynamic therapy to elicit antitumor immunity and antimetastatic effects on breast cancer. Small 15, e1903881. [DOI] [PubMed] [Google Scholar]
- 60.Wang D, Wang T, Liu J, Yu H, Jiao S, Feng B, Zhou F, Fu Y, Yin Q, Zhang P, Zhang Z, Zhou Z and Li Y. (2016) Acid-activatable versatile micelleplexes for PD-L1 blockade-enhanced cancer photodynamic immunotherapy. Nano Lett. 16, 5503–5513. [DOI] [PubMed] [Google Scholar]
- 61.Santos LL, Oliveira J, Monteiro E, Santos J and Sarmento C. (2018) Treatment of head and neck cancer with photodynamic therapy with Redaporfin: A clinical case report. Case Reports in Oncology 11(3), 769–776. 10.1159/000493423 [DOI] [PMC free article] [PubMed] [Google Scholar]




