Abstract
The major part of the dUTPase-encoding region of the visna virus genome was deleted. Intracerebral injection of the mutant virus resulted in a somewhat reduced viral load compared to that resulting from injection of the wild type, especially in the lungs, but the neuropathogenic effects were comparable. The dUTPase gene is dispensable for induction of lesions in the brain.
The enzyme dUTPase (EC 3.6.1.23) has been demonstrated in various procaryotic and eucaryotic organisms (5, 6, 31). It hydrolyzes dUTP to dUMP and PPi and thus provides a substrate for thymidylate synthase in the major biosynthetic pathway to TTP. Its activity lowers the dUTP/TTP ratio and leads to decreased misincorporation of uracil into DNA. Cellular dUTPase activity is reported to be cell cycle regulated, high in dividing cells but low in terminally differentiated nondividing cells (9, 10, 16, 21, 23). It may correlate with the pool of intracellular deoxynucleoside triphosphates, which is reported to be very low in macrophages (24).
dUTPase activity has been demonstrated in several viruses, including herpesviruses (4, 32), poxviruses (3), type B and D retroviruses (11), and in a subset of lentiviruses, namely, equine infectious anemia virus (EIAV), feline immunodeficiency virus (FIV), caprine arthritis-encephalitis virus (CAEV), and visna virus of sheep (7, 14). Human immunodeficiency virus and other primate lentiviruses do not contain a dUTPase gene. Two main theories have been proposed to explain the role of the viral dUTPases. One is that this viral enzyme permits or facilitates viral DNA synthesis in cells with low levels of deoxynucleoside triphosphates and thus enables the virus to replicate in nondividing cells such as neurons or macrophages. Evidence supporting this theory has been published for the dUTPase-containing lentiviruses EIAV and FIV (13, 22, 25, 30). The second theory maintains that the viral dUTPase exerts an antimutator function by reducing the misincorporation of uracil into viral DNA. Such misincorporation could lead to perturbation of viral DNA replication (20) and decreased viral fitness, and uracil residues in viral DNA might affect the function of the DNA such as binding of transcription factors (19, 29).
We have previously reported that dUTPase-deficient mutants of CAEV and visna virus show delayed replication in nondividing goat macrophages (27). In the present study, we showed that dUTPase-deficient visna virus is still fully pathogenic after intracerebral infection of sheep.
Construction and propagation of dUTPase-deficient virus.
We constructed an in-frame deletion mutant of the dUTPase gene of the pathogenic proviral molecular clone of the neurovirulent visna virus strain KV 1772 (1, 2) as previously described (27), which resulted in the deletion of three of the five conserved domains of the dUTPase, including the central domain with a tyrosine residue which is thought to be at the active site of the enzyme (14, 15). The deleted provirus and the wild-type provirus were transfected into primary goat synovial cells (27). The activity of the virion-associated dUTPase was assayed in pelleted virus from clarified supernatants of infected cells by measuring the relative incorporation of tritium-labeled dUTP and dTTP into a poly(rA)-oligo(dT) template under the conditions of the classical reverse transcriptase (RT) assay as described previously (27). The dUTP/TTP incorporation ratio of the dUTPase-deficient (DU-2) virus was found to be 0.26, as compared to a ratio of 0.04 for the wild-type (WT) virus.
In vitro replication of DU-2 and WT visna viruses.
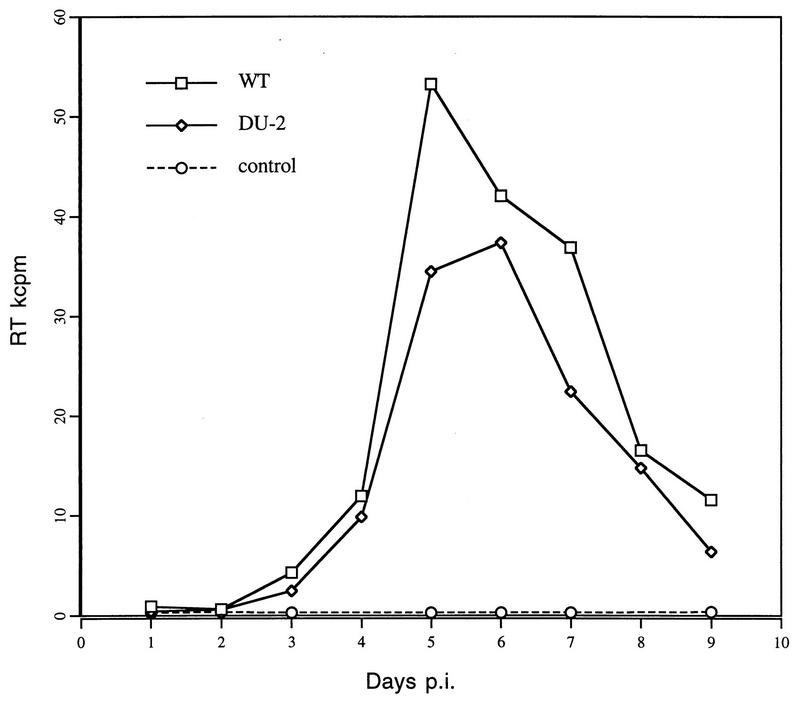
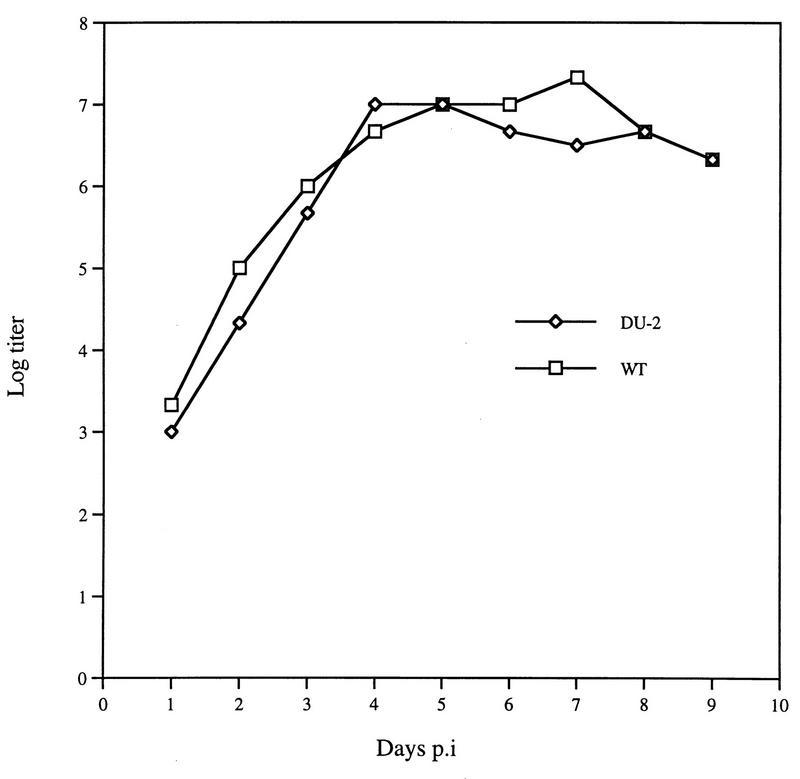
Replication of DU-2 and WT visna viruses was tested in sheep monocyte-derived macrophages in culture. Sheep monocyte-derived macrophages were prepared from heparinized blood by centrifugation on Histopaque-1077 as described previously (26), washed repeatedly, and seeded in plastic tissue culture vessels. After 24 h, adherent cells were washed repeatedly to remove unattached cells and incubated for 7 days in Dulbecco modified Eagle medium supplemented with 5 × 10−5 M mercaptoethanol and 10% lamb serum before they were used for infection. Viral replication was measured by RT assay as described previously (27). The replication of the DU-2 mutant virus in sheep macrophages was slightly delayed compared to that of the wild type when measured by this assay (Fig. 1). This difference, although slight, was consistently reproducible in repeated tests, whereas no significant difference in growth curves could be demonstrated when the infectivity of the macrophage-derived viruses was titrated in sheep choroid plexus cell cultures (Fig. 2).
FIG. 1.
Growth curves of DU-2 and WT visna viruses in sheep monocyte-derived macrophages as measured by RT activity (kcpm/0.5 ml). p.i., postinfection.
FIG. 2.
Growth curves of DU-2 and WT visna viruses in sheep monocyte-derived macrophages as measured by infectivity titration in sheep choroid plexus cells. Titration end points were calculated by the Reed-Muench method. p.i., postinfection.
Animal experiments.
To test the pathogenic potential of the DU-2 mutant, we inoculated five Icelandic 10-month-old sheep intracerebrally in the left hemisphere with 0.4 ml of DU-2 virus (titer, 107.5 tissue culture infective doses/ml) which had been grown in sheep choroid plexus cells; five sheep were also infected with the same dose of WT virus grown in the same way. The sheep were bled at regular intervals to test for serum antibodies to the virus by an enzyme-linked immunosorbent assay (ELISA) (28) and virus neutralization by standard methods, and circulating virus was tested by cocultivation of buffy coat cells with sheep choroid plexus cells. Samples of spinal fluid for cell counts and virus isolation were obtained at 4, 12, and 25 weeks after infection. All sheep were sacrificed at 25 weeks after infection, and virus isolations from the following tissues were attempted: plexus choroideus, cerebellum, medulla oblongata, spinal cord (cervical, thoracic, and lumbar parts), cervical, mediastinal, and mesenteric lymph nodes, spleen, bone marrow, and lungs. The histopathological lesions of the central nervous system were graded on a scale of 0 to 6 as described previously (17).
The frequency of virus isolation from sheep infected with DU-2 virus was somewhat lower than that of sheep inoculated with the WT, as shown in Table 1. The difference between the groups was statistically significant for determinations for the lungs and for all tissues taken together, but the frequencies of virus isolations from the central nervous systems of both groups were equal. The deletion of mutant virus reisolated from infected sheep was confirmed by PCR and sequencing (data not shown).
TABLE 1.
Frequency of virus isolations from sheep infected with DU-2 and WT viruses
| Tissue | No. of isolates/no. of samples tested
|
|
|---|---|---|
| DU-2 | WT | |
| Blood | 11/38 | 16/39 |
| Cerebrospinal fluid | 7/14 | 6/13 |
| Brain and spinal cord | 7/30 | 8/29 |
| Lymphoid tissue and bone marrow | 11/25 | 18/25 |
| Lungs | 4/20a | 11/20 |
| All tissues | 40/127b | 59/126 |
Significantly different from value for WT group (P = 0.0484, according to Fisher’s exact test).
Significantly different from value for WT group (P = 0.0145, according to Fisher’s exact test).
Virus-specific serum antibodies against whole virus antigen measured by an ELISA (26) were found to increase in titer with time after infection in both groups. The geometric means of the antibody titers were consistently lower in the sheep infected with the DU-2 virus than in the WT virus-infected sheep, although the range was rather wide (Table 2). As shown in Table 3, the virus-neutralizing titers were similar for both groups.
TABLE 2.
ELISA antibodies against whole visna virus antigen in sheep infected with DU-2 and WT viruses
| Time after infection (wk) | Geometric mean ELISA titer (range)
|
|
|---|---|---|
| DU-2 | WT | |
| 4 | 2 (0–6) | 2 (0–6) |
| 6 | 18 (6–54) | 35 (18–54) |
| 8 | 35 (18–162) | 43 (18–162) |
| 12 | 67 (18–162) | 111 (18–486) |
| 16 | 202 (54–486) | 313 (54–486) |
| 20 | 251 (54–486) | 390 (162–486) |
| 25 | 313 (162–486) | 605 (162–1,458) |
TABLE 3.
Neutralizing antibodies against visna virus
| Time postinfection (wk) | Neutralizing titera against:
|
|
|---|---|---|
| DU-2 | WT | |
| 6 | 2.8 (2–4) | 4 (4–4) |
| 8 | 4 (2–6) | 5.5 (4.5–7) |
| 12 | 7.5 (2.5–10) | 6.8 (5–8.5) |
| 25 | 8.5 (4.5–12) | 8.7 (6–12) |
Values are expressed as geometric means (ranges) of log base 2 titers (n = 5). Fifty percent end points were calculated by the Reed-Muench method.
Spinal fluid samples were obtained from the infected sheep at 4, 12, and 25 weeks postinfection. There was a brisk increase of mononuclear cells 1 month after infection (Table 4), as frequently observed in intracerebrally infected sheep (17). Although the group infected with the mutant virus showed more pleocytosis at this time point than the WT virus-infected group, the difference was statistically not quite significant (P = 0.11 with the Welch test); after 12 weeks and at the time of sacrifice, the sheep infected with the WT virus had a somewhat higher number of cells, but the difference was not significant.
TABLE 4.
Mononuclear cells in spinal fluid of sheep infected with DU-2 or WT virus
| Time postinfection (wk) | Avg no. of cells/μl (range)
|
|
|---|---|---|
| DU-2 | WT | |
| 4 | 572 (176–912) | 264 (62–434) |
| 12 | 84 (15–140) | 143 (49–270) |
| 25 | 33 (23–46) | 85 (10–318) |
At the time of sacrifice, histopathological changes were observed only in the brain (Fig. 3). They were, as shown in Fig. 3, in all respects like the classical lesions of visna virus described previously (8, 17), consisting of perivascular and periventricular infiltrates of mononuclear inflammatory cells (macrophages, lymphocytes, and some plasma cells). As shown in Table 5, there was no significant difference in the severity of lesions between sheep inoculated with the DU-2 mutant virus and those inoculated with the WT.
FIG. 3.
Photomicrographs of brain lesions in sheep infected with WT (A, B, and C) and DU-2 (D, E, and F) visna viruses. (A and D) Periventricular inflammation and discrete perivascular infiltrates in adjacent white matter are visible. (B and E) Almost confluent inflammation of the white matter is evident. (C and F) Inflammation of the choroid plexus with formation of lymph follicles is visible. Stain, hematoxylin-eosin. Magnification, ×90.
TABLE 5.
Histopathological grading of lesion severitya
| Virus | Lesion severity
|
|
|---|---|---|
| Grades | Avg grade | |
| DU-2 | 3.5, 3.5, 4.0, 4.5, 3.0 | 3.7 |
| WT | 5.0, 5.0, 4.0, 4.0, 2.0 | 4.0 |
Grading was done on a scale of 0 to 6.
These results show that visna virus lacking a functional dUTPase gene can replicate rather well in sheep macrophages in culture, unlike EIAV and FIV (13, 22, 25, 30), where deletion of the dUTPase gene practically abolished virus replication in equine and feline macrophages, respectively. CAEV seems to be intermediate in this respect, since dUTPase-negative mutants will replicate in goat macrophage cultures but at a lower rate than that of wild-type viruses (27). Ponies infected with dUTPase-negative EIAV and cats injected with FIV with the dUTPase-encoding gene deleted showed reduced viral loads (12, 13). We have found similar evidence of a reduced viral load in the lungs and possibly in the blood, lymphoid tissue, and bone marrow of sheep injected with the DU-2 visna virus. Recently, it has been found that intra-articular inoculation of dUTPase-negative mutants of CAEV resulted in systemic infection and dissemination of virus in a manner similar to that of the WT virus (28). The WT virus was pathogenic both locally in the injected joint and in the contralateral joint, whereas the lesions produced by the mutant were restricted to the inoculated joint and were somewhat less severe. In our experiments, lesions were found only in the central nervous system, which is in accordance with our previous experience with sheep inoculated intracerebrally, where lung lesions are rarely observed. The pathogenic potential of visna virus is unaffected by the loss of the viral dUTPase function, at least at the site of injection. The gene coding for dUTPase must, however, have some evolutionary advantage for the virus since it has been conserved, and CAEV with a point mutation in the gene coding for dUTPase has been shown to revert to the WT in an infected goat (28). This advantage seems to be too subtle to be detected by our experiment, possibly because of the rather high virus dose and the route of infection. The low frequency of virus isolation from the lungs of the sheep infected with the DU-2 virus may indicate that such mutants would be less readily transmissible in the course of natural infection, which is believed to be either by the respiratory route or lactogenic (18).
Acknowledgments
This study was supported by the Agence Nationale de Recherches sur le SIDA, Paris, France, the Icelandic Research Council Science Fund, and the University of Iceland Research Fund.
REFERENCES
- 1.Andrésson Ó, Elser J E, Tobin G J, Greenwood J D, Gonda M A, Georgsson G, Andrésdóttir V, Benediktsdóttir E, Carlsdóttir H M, Mäntylä E O, Rafnar B, Pálsson P A, Casey J W, Pétursson G. Nucleotide sequence and biological properties of a pathogenic proviral molecular clone of neurovirulent visna virus. Virology. 1993;193:89–105. doi: 10.1006/viro.1993.1106. [DOI] [PubMed] [Google Scholar]
- 2.Andrésson Ó, Elser J E, Georgsson G, Tobin G J, Greenwood J D, Gonda M A, Andrésdóttir V, Pálsson P A, Pétursson G. Pathogenic proviral molecular clone of neurovirulent visna virus. Ann N Y Acad Sci. 1994;724:133–139. doi: 10.1111/j.1749-6632.1994.tb38901.x. [DOI] [PubMed] [Google Scholar]
- 3.Broyles S S. Vaccinia virus encodes a functional dUTPase. Virology. 1993;195:863–865. doi: 10.1006/viro.1993.1446. [DOI] [PubMed] [Google Scholar]
- 4.Caradonna S J, Cheng Y-C. Induction of uracil-DNA glycosylase and dUTP nucleotidohydrolase in herpes simplex virus-infected human cells. J Biol Chem. 1981;256:9834–9837. [PubMed] [Google Scholar]
- 5.Cedergren-Zeppezauer E S, Larsson G, Nyman P O, Dauter Z, Wilson K S. Crystal structure of a dUTPase. Nature (London) 1992;355:440–443. doi: 10.1038/355740a0. [DOI] [PubMed] [Google Scholar]
- 6.Doignon F, Biteau N, Aigle M, Crouzet M. The complete sequence of a 6794 bp segment located on the right arm of chromosome II of Saccharomyces cerevisiae. Finding a putative dUTPase in a yeast. Yeast. 1993;9:1131–1137. doi: 10.1002/yea.320091014. [DOI] [PubMed] [Google Scholar]
- 7.Elder J H, Lerner D L, Hasselkus-Light C S, Fontenot D J, Hunter E, Luciw P A, Montelaro R C, Phillips T R. Distinct subsets of retroviruses encode dUTPase. J Virol. 1992;66:1791–1794. doi: 10.1128/jvi.66.3.1791-1794.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Georgsson G, Houwers D J, Pálsson P A, Pétursson G. Expression of viral antigens in the central nervous system of visna-infected sheep: an immunohistochemical study on experimental visna induced by virus strains of increased neurovirulence. Acta Neuropathol. 1989;77:299–306. doi: 10.1007/BF00687582. [DOI] [PubMed] [Google Scholar]
- 9.Hokari S, Hasegawa M, Sakagishi Y, Kikushi G. Deoxyuridine triphosphate nucleotidohydrolase activity and its correlation with multiplication of erythroid cells in rat spleen. Biochem Int. 1987;14:851–857. [PubMed] [Google Scholar]
- 10.Hokari S, Sakagishi Y, Tsukada K. Enhanced activity of deoxyuridine 5′-triphosphatase in regenerating rat liver. Biochem Biophys Res Commun. 1982;108:95–101. doi: 10.1016/0006-291x(82)91836-8. [DOI] [PubMed] [Google Scholar]
- 11.Köppe B, Menéndes-Arias L, Oroszlan S. Expression and purification of the mouse mammary tumor virus gag-pro transframe protein p30 and characterization of its dUTPase activity. J Virol. 1994;68:2313–2319. doi: 10.1128/jvi.68.4.2313-2319.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Lerner D L, Wagaman P C, Phillips T R, Prospero-Garcia O, Henriksen S J, Fox H S, Bloom F E, Elder J H. Increased mutation frequency of feline immunodeficiency virus lacking functional deoxyuridine-triphosphatase. Proc Natl Acad Sci USA. 1995;92:7480–7484. doi: 10.1073/pnas.92.16.7480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lichtenstein D L, Rushlow K E, Cook R F, Raabe M L, Swardson C J, Kociba G J, Issel C J, Montelaro R C. Replication in vitro and in vivo of an equine infectious anemia virus mutant deficient in dUTPase activity. J Virol. 1995;69:2881–2888. doi: 10.1128/jvi.69.5.2881-2888.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.McGeoch D J. Protein sequence comparisons show that the “pseudoproteases” encoded by poxviruses and certain retroviruses belong to the deoxyuridine triphosphatase family. Nucleic Acids Res. 1990;18:4105–4110. doi: 10.1093/nar/18.14.4105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.McIntosh E M, Ager D D, Gadsden M H, Haynes R H. Human dUTP pyrophoshatase: cDNA sequence and potential biological importance of the enzyme. Proc Natl Acad Sci USA. 1992;89:8020–8024. doi: 10.1073/pnas.89.17.8020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pardo E G, Gutierrez C. Cell cycle- and differentiation stage-dependent variation of dUTPase activity in higher plant cells. Exp Cell Res. 1990;186:90–98. doi: 10.1016/0014-4827(90)90214-u. [DOI] [PubMed] [Google Scholar]
- 17.Pétursson G, Nathanson N, Georgsson G, Panitch H, Pálsson P A. Pathogenesis of visna. I. Sequential virologic, serologic, and pathologic studies. Lab Invest. 1976;35:402–412. [PubMed] [Google Scholar]
- 18.Pétursson G, Andrésdóttir V, Andrésson Ó, Georgsson G, Pálsson P A, Rafnar B, Torsteinsdóttir S. Lentivirus diseases of sheep and goats: maedi-visna and caprine arthritis-encephalitis. In: Speedy A W, editor. Progress in sheep and goat research. Wallingford, United Kingdom: CAB International; 1990. pp. 107–129. [Google Scholar]
- 19.Pu W, Struhl K. Uracil interference, a rapid and general method for defining protein-DNA interaction involving the 5-methyl group of thymines: the GCN4-DNA complex. Nucleic Acids Res. 1992;20:771–775. doi: 10.1093/nar/20.4.771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Richards R G, Sowers L C, Lazslo J, Sedwick W D. The occurrence and consequences of deoxyuridine in DNA. Adv Enzyme Regul. 1986;22:157–185. doi: 10.1016/0065-2571(84)90013-x. [DOI] [PubMed] [Google Scholar]
- 21.Spector R, Boose B. Development and regional distribution of deoxyuridine 5′-triphosphatase in rabbit brain. J Neurochem. 1983;41:1192–1195. doi: 10.1111/j.1471-4159.1983.tb09073.x. [DOI] [PubMed] [Google Scholar]
- 22.Steagall W K, Robek M D, Perry S T, Fuller F J, Payne S L. Incorporation of uracil into viral DNA correlates with reduced replication of EIAV in macrophages. Virology. 1995;210:302–313. doi: 10.1006/viro.1995.1347. [DOI] [PubMed] [Google Scholar]
- 23.Strahler J R, Zhu X X, Nora N, Wang Y K, Andrewes P C, Roseman N A, Neel J V, Turka L, Payne S L. Maturation stage and proliferation-dependent expression of dUTPase in human T cells. Proc Natl Acad Sci USA. 1993;90:4991–4995. doi: 10.1073/pnas.90.11.4991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Terai C, Carson D A. Pyrimidine nucleotide and nucleic acid synthesis in human monocytes and macrophages. Exp Cell Res. 1991;193:375–381. doi: 10.1016/0014-4827(91)90110-g. [DOI] [PubMed] [Google Scholar]
- 25.Threadgill D S, Steagall W K, Flaherty M T, Flaherty F J, Fuller F J, Perry S T, Rushlow K E, Le Grice S F, Payne S L. Characterization of equine infectious anemia virus dUTPase: growth properties of a dUTPase-deficient mutant. J Virol. 1993;67:2592–2600. doi: 10.1128/jvi.67.5.2592-2600.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Torsteinsdóttir S, Agnarsdóttir G, Matthíasdóttir S, Rafnar B, Andrésdóttir V, Andrésson Ó, Staskus K, Pétursson G, Pálsson P A, Georgsson G. In vivo and in vitro infection with two different molecular clones of visna virus. Virology. 1997;229:370–380. doi: 10.1006/viro.1996.8428. [DOI] [PubMed] [Google Scholar]
- 27.Turelli P, Pétursson G, Guiguen F, Mornex J F, Vigne R, Quérat G. Replication properties of dUTPase-deficient mutants of caprine and ovine lentiviruses. J Virol. 1996;70:1213–1217. doi: 10.1128/jvi.70.2.1213-1217.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Turelli P, Guiguen F, Mornex J-F, Vigne R, Quérat G. dUTPase-minus caprine arthritis-encephalitis virus is attenuated for pathogenesis and accumulates G-to-A substitutions. J Virol. 1997;71:4522–4530. doi: 10.1128/jvi.71.6.4522-4530.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Verri A, Mazzarello P, Biamonti G, Spadari S, Focher F. The specific binding of nuclear protein(s) to the cAMP responsive element (CRE) sequence (TGACGTCA) is reduced by the misincorporation of U and increased by the deamination of C. Nucleic Acids Res. 1990;18:5775–5780. doi: 10.1093/nar/18.19.5775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Wagaman P C, Hasselkus-Light C S, Henson M, Lerner D L, Phillips T R, Elder J H. Molecular cloning and characterization of deoxyuridine triphosphatase from feline immunodeficiency virus (FIV) Virology. 1993;196:451–457. doi: 10.1006/viro.1993.1501. [DOI] [PubMed] [Google Scholar]
- 31.Williams M V, Cheng Y-C. Human deoxyuridine triphosphate nucleotidohydrolase. J Biol Chem. 1979;254:2897–2901. [PubMed] [Google Scholar]
- 32.Williams M V, Parris D S. Characterization of a herpes simplex virus type 2 deoxyuridine triphosphate nucleotidohydrolase and mapping of a gene conferring type specificity for the enzyme. Virology. 1987;156:282–292. doi: 10.1016/0042-6822(87)90408-9. [DOI] [PubMed] [Google Scholar]