Abstract
Background
Ventilator-associated bacterial pneumonia (VABP) is a common infection in critically ill patients in intensive care units (ICU), with attributable mortality of up to 13%, and its etiological diagnosis remains challenging.
Materials and methods
We conducted a multicenter, prospective, observational study within the MULTI-SITA platform to assess the impact on relevant clinical and antimicrobial stewardship outcomes of the use of a molecular syndromic panel (BIOFIRE® FILMARRAY® Pneumonia plus), in addition to a standard approach based on culture. The primary outcome measure was 30-day mortality from VABP onset.
Results
Overall, 237 patients with VABP were included in the study. In multivariable analysis, SOFA score (hazard ratio [HR] 1.13, 95% confidence interval [CI] 1.04–1.22, p = 0.003), previous isolation of carbapenem-resistant Pseudomonas aeruginosa (HR 3.02, 95% CI 1.25–7.32, p = 0.015), and solid neoplasm (HR 2.15, 95% CI 1.12–4.14, p = 0.022) were associated with increased mortality, while no association was registered for the molecular syndromic panel performed (HR 1.07, 95% CI 0.59–1.93, p = 0.825). In secondary analyses, use of the molecular syndromic panel resulted in more events of either de-escalation or initiation of appropriate antibiotic therapy at day 1 from VABP onset in comparison with a standard approach based on culture only (41.3% vs. 27.8%, p = 0.041).
Conclusion
The use of a molecular syndromic panel in patients with VABP was able to impact antibiotic decisions, without an unfavorable effect on mortality. Further study is necessary to assess the long-term effects in terms of antimicrobial stewardship of molecular syndromic panels-based antibiotic treatment decisions.
Supplementary Information
The online version contains supplementary material available at 10.1186/s13054-025-05632-z.
Keywords: Rapid molecular tests, Antimicrobial resistance, Antimicrobial stewardship, Rapid diagnosis, Ventilator-associated pneumonia
Introduction
Ventilator-associated bacterial pneumonia (VABP) is a common infection in critically ill patients in intensive care units (ICU), with an incidence of more than 18 events per 1000 ventilator days and an attributable mortality of up to 13% [1–4].
The etiological diagnosis of VABP remains challenging and is classically achieved through culture of deep respiratory specimens, which may take up to more than 48–72 h between collection and complete culture results, including susceptibility testing [5]. Before culture results, and whenever cultures are negative despite a clinical and radiological picture compatible with VABP, clinicians usually rely on empiric broad-spectrum antibiotic therapy [6]. In this context, rapid molecular testing of deep respiratory specimens could help reduce the duration of either broad-spectrum or inappropriate antibiotic therapy, by inducing earlier de-escalation to more targeted and narrow therapy, thereby mitigating the complications of broad-spectrum antibiotic overuse in line with antimicrobial stewardship principles, or by identifying causative agents or resistance determinants requiring treatment escalation [6, 7].
The BIOFIRE® FILMARRAY® Pneumonia plus (PNplus) panel allows to identify 18 and 7 different bacteria and resistance genes from respiratory specimens (bronchoalveolar lavage [BAL]-like, sputum-like), respectively, with an approximate turnaround time of less than 2 h [5, 8], thus providing clinicians with an additional and earlier time point to consider revision (e.g., de-escalation, escalation) or direct initiation of targeted therapy when stable conditions allow waiting a few hours for PNplus panel results [9–12].
The present observational, prospective, multicenter study aimed to assess the real-world impact of the PNplus panel on relevant clinical outcomes and antibiotic use in critically ill patients with VABP.
Material and methods
Study setting and objectives
The MULTI-SITA project is a platform developed by the Italian Society of Anti-Infective Therapy (SITA) and dedicated to conduct observational studies on invasive bacterial and fungal diseases. RAPID-SITA PHENOTYPES is an observational, prospective, multicenter study conducted in Italian hospitals within the MULTI-SITA project, aimed to compare the impact of rapid molecular panels on the outcomes of consecutive critically ill adult patients in ICU with VABP and/or BSI. Etiological diagnosis could be achieved by either BIOFIRE® FILMARRAY® panels (PNplus panel on BAL samples and BCID2 panel on blood cultures for VABP and BSI, respectively), standard microbiological cultures (BAL cultures and blood cultures for VABP and BSI, respectively), or both, performed on samples collected within 24 h before and 24 h after the onset of the infection. All diagnostic tests were prescribed and performed according to local clinical practice, in line with the observational nature of the study. The prospective study period of the RAPID-SITA PHENOTYPES study was from 1 January 2023 to 31 October 2024.
Here, we report the results related to the comparison of patients with VABP (either with or without concomitant BSI) included in the RAPID-SITA PHENOTYPES study from 12 Italian centers. The primary outcome measure of this first analysis was 30-day mortality from VABP onset, considered as a time-to-event endpoint. Secondary endpoints were: (i) earlier antibiotic therapy discontinuation as time-to-event endpoint with death as competing risk (see statistical analysis below); (ii) time to appropriate antibiotic therapy from VABP onset in days; (iii) a composite outcome of antibiotic de-escalation in patients receiving appropriate therapy from VABP onset (day 0) or start of an appropriate therapy on day 1 after VABP onset; (iv) incidence rate of Clostridioides difficile infection (CDI) and candidemia. The composite outcome (iii) was defined by considering those clinical decisions that can be theoretically impacted by the availability of the PNplus panel, i.e., frequency of de-escalation in patients who started an appropriate empirical therapy since infection onset (potentially prompted by more rapid availability of PNplus panel results compared to culture) and frequency of initiation of an appropriate therapy from day 1 in patients who did not start an appropriate therapy on day 0 (including both rapid initiation and rapid escalation of therapy, again potentially prompted by more rapid availability of PNplus panel results compared to culture). Exclusion criteria were (i) age less than 18 years; (ii) already included in the study for a previous VABP episode.
The MULTI-SITA project was approved by the ethics committee of the coordinating center (Liguria Region Ethics Committee, registry number 390/2020), with a subsequent amendment authorizing the conduct of the RAPID-SITA PHENOTYPES study within the MULTI-SITA project. The other participating centers followed the local ethical committees requirements and started to enroll patients prospectively once activated. Conscious patients at time of enrollment signed an informed consent to participate in the study. A waiver of informed consent for data collection from unconscious patients at the time of enrollment due to severe clinical conditions was obtained within the ethics committee approval, in line with the observational nature of the analyses and in order not to bias research results towards low mortality prejudicing scientific validity.
Microbiological procedures
Identification of bacterial isolates from BAL specimens was performed by means of matrix-assisted laser desorption ionization time of flight mass spectrometry (MALDI Biotyper, Bruker Daltonics, Billerica, MA, USA; or Vitek MS MALDI-TOF mass spectrometry, bioMérieux, Craponne, France;) or automated systems, depending on standard local procedures. Antimicrobial susceptibility testing (AST) was performed by means of automated systems (MicroScan, Beckman Coulter, Brea, CA, USA; Vitek 2, bioMérieux, Craponne, France; or Phoenix, Becton Dickinson Diagnostics, Sparks, MD, USA) according to local standard procedures.
PNplus panel testing
The PNplus panel testing was performed following the manufacturer’s instructions. Briefly, approximately 200 µL of the BAL-like (i.e. BAL and mini-BAL) specimen was transferred to the sample injection vial using a sterile sample swab provided in the test kit. The sample was then mixed with the provided buffer and loaded into the FilmArray pouch (a closed system disposable that stores all the necessary reagents), that was subsequently inserted into the FilmArray instrument providing with: i) automated nucleic acid extraction; ii) multiplex PCR; and iii) post-amplification analysis. Each positive and negative assay result was interpreted by the BIOFIRE Software to provide results for the identification of specific bacteria (i.e., Acinetobacter calcoaceticus-baumannii complex, Enterobacter cloacae complex, Escherichia coli, Haemophilus influenzae, Klebsiella aerogenes, Klebsiella oxytoca, Klebsiella pneumoniae group, Moraxella catarrhalis, Proteus spp., Pseudomonas aeruginosa, Serratia marcescens, Staphylococcus aureus, Streptococcus agalactiae, Streptococcus pneumoniae, Streptococcus pyogenes), atypical bacteria (i.e., Chlamydia pneumoniae, Legionella pneumophila, Mycoplasma pneumoniae), viruses (i.e., adenovirus, coronavirus, human metapneumovirus, human rhinovirus/enterovirus, influenza A virus, influenza B virus, middle east respiratory syndrome coronavirus, parainfluenza virus, respiratory syncytial virus), and antimicrobial resistance markers (i.e. CTX-M, KPC, IMP, VIM, NDM, OXA-48-like, mecA/C and MREJ).
Definitions and data collected for the study
VABP was defined as ventilator-associated pneumonia with bacterial etiological diagnosis achieved on BAL by either PNplus panel or standard microbiological cultures. Ventilator-associated pneumonia was defined as new or changing chest X-ray or chest computerized tomography infiltrate/s occurring more than 48 h after initiation of invasive mechanical ventilation, plus both of the following: (i) leukocytosis (total white cell count ≥ 10,000 cells/μL)/leukopenia (total white cell count ≤ 4500 cells/μL)/> 15% immature neutrophils and/or new onset of fever (body temperature ≥ 38 °C)/hypothermia (body temperature ≤ 35 °C); (ii) new onset of suctioned respiratory secretions and/or need for acute ventilator support system changes to enhance oxygenation [13, 14]. Besides information regarding primary and secondary outcome measures, the following demographic and clinical variables were collected as they were at the time of VABP onset: age in years; sex; diabetes mellitus; chronic obstructive pulmonary disease (COPD); New York Heart Academy (NYHA) score; chronic liver disease (defined histologically as liver cirrhosis or in presence of a clinical diagnosis supported by laboratory, endoscopy, and radiologic findings [15]); chronic kidney disease (defined as estimated glomerular filtration rate < 60 mL/min/1.73 m2); chronic intermittent hemodialysis; solid neoplasm; metastatic solid neoplasm; hematological malignancy; previous hematopoietic stem cell transplantation (HSCT); previous solid organ transplantation (SOT); human immunodeficiency virus (HIV) infection; autoimmune disease; age-adjusted Charlson Comorbidity Index [16]; previous antibiotic therapy, overall and stratified according to different agents/classes (within 6 months); previous antifungal therapy, overall and stratified according to different agents/classes (within 6 months); previous chemotherapy (within 6 months); previous steroid therapy (within 6 months); previous therapy with other immunosuppressants (within 6 months); previous major surgery (within 3 months); previous isolation of carbapenemase-producing Enterobacterales (CPE); previous isolation of carbapenem-resistant Acinetobacter baumannii (CRAB); previous isolation of carbapenem-resistant Pseudomonas aeruginosa (CRPA); previous isolation of methicillin-resistant Staphylococcus aureus (MRSA); days from ICU admission to VABP onset; sequential organ failure assessment (SOFA) score [17]; presence of central venous catheter (CVC); presence of septic shock [18]; presence of at least mild acute respiratory distress syndrome (ARDS) [19]; neutropenia (defined as absolute neutrophil count < 500 cell/mm3); continuous renal replacement therapy (CRRT); extracorporeal membrane oxygenation (ECMO); presence of at least stage 1 of acute kidney injury (AKI) according to the Kidney Disease: Improving Global Outcome (KDIGO) criteria [20]; concomitant BSI (either by the same etiological agent/s of VABP or by other microorganisms); other concomitant infections requiring antibiotic therapy (besides BSI); etiological agents of VABP according to culture results and/or PNplus panel results; days from VABP onset to appropriate antibiotic therapy (defined as therapy with at least one agent displaying in vitro activity against the given BAL isolate/s, whenever BAL culture result were available; for patients with only positive PNplus panel, appropriate therapy was defined as therapy with at least one agent considered as presumably active based on the type of identified microorganism/s and presence/lack of identified resistance genes; appropriateness of antibiotic therapy based solely on positive PNplus panel was assessed by two independent investigators, with cases of disagreement being resolved by a third investigator); use of cytokine blood filter/s; administration of intravenous immunoglobulins. De-escalation was defined as a reduction in the spectrum of administered antibiotics through the discontinuation of antibiotics or switching to an agent with a narrower spectrum [21].
Statistical analysis
Demographic and clinical characteristics of the study population were compared between critically ill patients with VABP in whom the PNplus panel was performed on BAL (independent of concomitant performance of BAL culture) and critically ill patients with VABP in whom only BAL culture was performed (and the PNplus panel was not performed) for descriptive purposes, using the Fisher exact test or the chi-squared test for categorical variables, as appropriate, and the Wilcoxon test for continuous variables. The crude 30-day mortality from the onset of VABP was summarized graphically by means of the Kaplan–Meier method, and compared between critically ill patients with VABP in whom the PNplus panel was performed and critically ill patients with VABP in whom only BAL culture was performed using the log-rank test.
The primary study analysis was to assess the independent impact of the performance of the PNplus panel on 30-day mortality. In this regard, we first performed Rubin’s multiple imputation [22]. The association of performance of the PNplus panel and other demographics and clinical variables with 30-day mortality was first tested in univariable Cox regression models, with ICU discharge within 30 days from VABP onset as a right-censoring event. Then, all the variables potentially associated with mortality in univariable comparisons (P < 0.10) were included in an initial multivariable Cox regression model, with subsequent selection for inclusion in the final multivariable Cox regression model (multivariable model A) by means of a backward stepwise procedure. In line with the purpose of the study, the variable “PNplus panel performed” was included in multivariable model A independent of its selection by the stepwise procedure. Variables included in multivariable model A were also included in an additional multivariable Cox regression model (multivariable model B) also including center as shared frailty [23]. In all Cox regression models for the assessment of factors associated with 30-day mortality, the variable “days from VABP onset to appropriate antibiotic therapy” was considered as a time-dependent variable (i.e. increase of one unit each passing day without antibiotic therapy or with no appropriate antibiotic therapy).
A secondary study analysis was to assess the independent impact of the performance of the PNplus panel on anticipating discontinuation of antibiotic therapy (i.e., reducing length of VABP treatment). The association of performance of the PNplus panel and other demographics and clinical variables with earlier discontinuation of antibiotic therapy was first tested in univariable Fine-Gray models, with ICU discharge within 30 days from antibiotic therapy initiation for VABP as a right-censoring event, and death as a competing risk [24]. Then, all the variables potentially associated with mortality in univariable comparisons (P < 0.10) were included in an initial multivariable Fine-Gray model, with subsequent selection for inclusion in the final multivariable Fine-Gray model (multivariable model C) by means of a backward stepwise procedure. In line with the purpose of the study, the variable “PNplus panel performed” was included in multivariable model C independent of its selection by the stepwise procedure. Variables included in the multivariable model C were also included in an additional Fine–Gray competing risks model (multivariable model D), accounting for center-level clustering through robust variance estimation. Of note, in Fine-Gray models for antibiotic therapy discontinuation, a subdistribution hazard ratio (sHR) > 1.00 indicated a direction of effect towards reduction of the length of antibiotic therapy. For both the primary analysis and secondary analysis aimed at assessing predictors of mortality and earlier discontinuation, respectively, sensitivity analyses of models A, B, C, and D were conducted including only patients without concomitant BSI.
Other secondary analyses were aimed to compare the following descriptive outcomes between patients in whom the PNplus panel was performed and patients in whom only BAL culture was performed: (i) time to appropriate antibiotic therapy from VABP onset, in days (compared using the Wilcoxon test); (ii) composite outcome of antibiotic de-escalation in patients receiving appropriate therapy from infection onset or starting of an appropriate therapy from day 1 (compared using the Fisher exact test); (iii) incidence rate of CDI and candidemia per 1000 patient days in ICU (compared using Poisson regression).
The analyses were conducted using SAS software (version 9.4, SAS Institute Inc., Cary, NC, USA) and R Statistical Software (version 4.2.2, R Foundation for Statistical Computing, Vienna, Austria).
Results
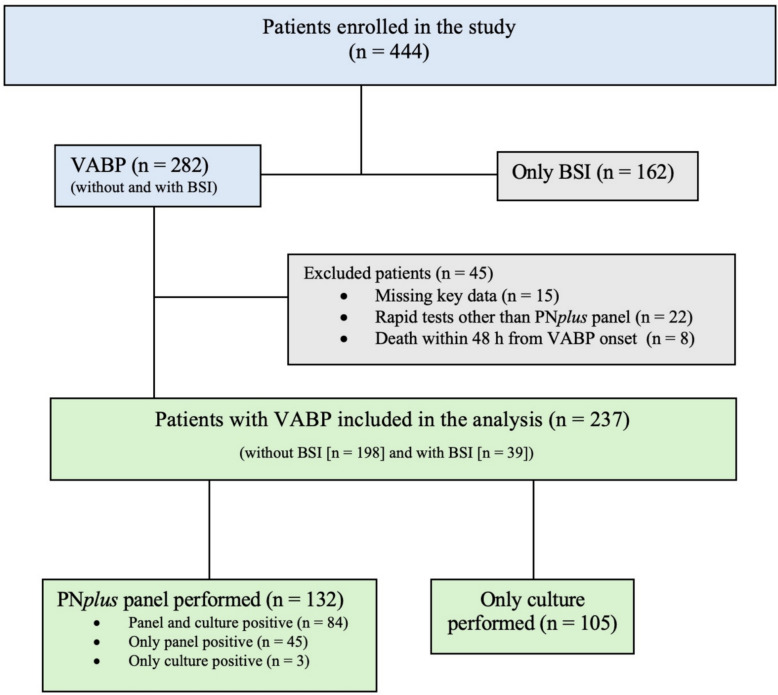
During the study period, 237 critically ill patients with VABP were included in the analyses, of whom 132 (55.7%) underwent PNplus panel testing on BAL and 105 (44.3%) underwent BAL culture only (Fig. 1).
Fig. 1.
Flow-chart of the patient inclusion process. BSI, bloodstream infection; VABP, ventilator-associated bacterial pneumonia
The demographic and clinical characteristics of the study population, overall and stratified according to performance of PNplus testing, are reported in Table 1. As shown in the table, the most frequent, non-mutually exclusive, etiological agents of VABP were members of the order Enterobacterales (131/237, 55.3%), followed by Pseudomonas aeruginosa (58/237, 24.5%) and Staphylococcus aureus (47/237, 19.8%). In patients who underwent PNplus panel testing, positivity of both PNplus panel and culture was registered in 63.6% of cases (84/132), while positivity of PNplus panel only and of culture only was registered in 34.1% (45/132) and 2.3% (3/132) of cases, respectively. In patients with positivity of both PNplus panel and BAL culture, the same bacteria identified in BAL culture were also detected by PNplus panel in 79/84 cases (94.0%). However, in 20 out of these 79 cases (25.3%), the PNplus panel also identified additional bacteria besides those grown in culture. Resistance genes were detected by the PNplus panel in 26.5% of cases (35/132) with the following non-mutually exclusive distribution: CTX-M (18/35, 51.4%); mecA/C – MREJ (9/35, 25.7%); KPC (5/35, 14.3%); VIM (5/35, 14.3%); NDM (1/35, 2.9%). BAL culture was positive in 24/35 cases in which resistance genes were detected by the PNplus panel, showing consistence between phenotypical resistance and the detected resistance gene/s in 14/24 cases (58.3%). In 30.0% (3/10) of the remaining cases with discordant phenotypical resistance and presumed resistance based on detected resistance gene/s, the PNplus panel identified at least an additional organism (compared with culture) that could express the detected resistance gene/s. Regarding the 3 cases in which only BAL culture was positive and the PNplus panel was negative, in two of them cultures yielded pathogens not included in the PNplus panel (Corynebacterium spp. and Morganella spp.), while in third case BAL culture was positive for Staphylococcus aureus (bacterial count in colony forming units not available).
Table 1.
Demographic and clinical characteristics of critically ill adult patients with VABP
| Variables* | Total (n = 237) | PNplus panel performed (n = 132) | Only BAL culture performed (n = 105) | P |
|---|---|---|---|---|
| No. of patients (%) | No. of patients (%) | No. of patients (%) | ||
| Demographics | ||||
| Age in years, median (IQR) | 63 (54–71) | 59 (52–70) | 65 (58–75) | 0.001 |
| Male sex | 162 (68.4) | 96 (72.7) | 66 (62.9) | 0.105 |
| Comorbidities and medical history | ||||
| Diabetes mellitus | 43/232 (18.5) | 22 (16.8) | 21 (20.8) | 0.437 |
| COPD | 41/227 (18.1) | 20 (15.3) | 21 (21.9) | 0.201 |
| NYHA score, median (IQR) | 2 (1–3) | 2 (1–3) | 2 (1–2) | 0.067 |
| Chronic liver disease | 9/229 (3.9) | 8 (6.2) | 1 (1.0) | 0.082 |
| Chronic kidney disease | 19/231 (8.2) | 7 (5.3) | 12 (12.0) | 0.068 |
| Chronic intermittent hemodialysis | 19/234 (8.1) | 10 (7.6) | 9 (8.7) | 0.759 |
| Solid neoplasm | 38/232 (16.4) | 18 (13.7) | 20 (19.8) | 0.216 |
| Metastatic solid neoplasm | 9/232 (3.9) | 4 (3.1) | 5 (5.0) | 0.508 |
| Hematological malignancy | 11/233 (4.7) | 5 (3.8) | 6 (5.9) | 0.541 |
| Previous HSCT | 5/233 (2.2) | 1 (0.8) | 4 (3.9) | 0.171 |
| Previous SOT | 10/233 (4.3) | 7 (5.3) | 3 (2.9) | 0.519 |
| HIV infection | 10/228 (4.4) | 9 (6.9) | 1 (1.0) | 0.047 |
| Autoimmune disease | 23/231 (10.0) | 12 (9.2) | 11 (11.0) | 0.644 |
| Age-adjusted Charlson Comorbidity Index, median (IQR) | 4 (2–5) | 3 (2–5) | 4 (2–6) | 0.056 |
| Previous antibiotic therapy | 71/183 (38.8) | 35 (32.7) | 36 (47.4) | 0.045 |
| Previous aminoglycosides | 5/183 (2.7) | 1 (0.9) | 4 (5.3) | 0.162 |
| Previous fluoroquinolones | 8/183 (4.4) | 4 (3.7) | 4 (5.3) | 0.720 |
| Previous macrolides | 3/183 (1.6) | 2 (1.9) | 1 (1.3) | 1.000 |
| Previous trimethoprim/sulfamethoxazole | 1/183 (0.6) | 1 (0.9) | 0 (0.0) | 1.000 |
| Previous metronidazole/clindamycin | 6/183 (3.3) | 3 (2.8) | 3 (4.0) | 0.694 |
| Previous antistaphylococcal penicillins | 2/183 (1.1) | 1 (0.9) | 1 (1.3) | 1.000 |
| Previous semisynthetic aminopenicillins | 12/183 (6.6) | 7 (6.5) | 5 (6.6) | 1.000 |
| Previous piperacillin/tazobactam | 33/183 (18.0) | 16 (15.0) | 17 (22.4) | 0.199 |
| Previous cefazolin | 5/183 (2.7) | 2 (1.9) | 3 (4.0) | 0.651 |
| Previous ceftriaxone/cefotaxime | 13/183 (7.1) | 8 (7.5) | 5 (6.6) | 0.816 |
| Previous ceftazidime/cefepime | 2/183 (1.1) | 2 (1.9) | 0 (0.0) | 0.512 |
| Previous ceftaroline/ceftobiprole | 9/183 (4.9) | 3 (2.8) | 6 (7.9) | 0.166 |
| Previous ceftolozane/tazobactam | 4/183 (2.2) | 0 (0.0) | 4 (5.3) | 0.028 |
| Previous ceftazidime/avibactam | 11/183 (6.0) | 8 (7.5) | 3 (4.0) | 0.366 |
| Previous cefiderocol | 4/183 (2.2) | 3 (2.8) | 1 (1.3) | 0.643 |
| Previous carbapenems | 24/183 (13.1) | 10 (9.4) | 14 (18.4) | 0.080 |
| Previous imipenem/relebactam | 0/183 (0.0) | 0 (0.0) | 0 (0.0) | - |
| Previous meropenem/vaborbactam | 0/183 (0.0) | 0 (0.0) | 0 (0.0) | - |
| Previous fosfomycin | 4/183 (2.2) | 2 (1.9) | 2 (2.6) | 1.000 |
| Previous polymyxins | 1/183 (0.6) | 1 (0.9) | 0 (0.0) | 1.000 |
| Previous tigecycline | 6/183 (3.3) | 4 (3.7) | 2 (2.6) | 1.000 |
| Previous glycopeptides | 10/183 (5.5) | 4 (3.7) | 6 (7.9) | 0.323 |
| Previous daptomycin | 20/183 (10.9) | 9 (8.4) | 11 (14.5) | 0.195 |
| Previous linezolid | 20/183 (10.9) | 13 (12.2) | 7 (9.2) | 0.530 |
| Previous antifungal therapy | 23/180 (12.8) | 13 (12.2) | 10 (13.7) | 0.760 |
| Previous azoles | 6/180 (3.3) | 2 (1.9) | 4 (5.5) | 0.225 |
| Previous echinocandins | 16/180 (8.9) | 9 (8.4) | 7 (9.6) | 0.785 |
| Previous polyenes | 3/180 (1.7) | 2 (1.9) | 1 (1.4) | 1.000 |
| Previous chemotherapy | 17/223 (7.6) | 9 (7.0) | 8 (8.4) | 0.700 |
| Previous steroid therapy | 37/181 (20.4) | 21 (19.8) | 16 (21.3) | 0.803 |
| Previous therapy with immunosuppressants | 13/178 (7.3) | 7 (6.7) | 6 (8.2) | 0.695 |
| Previous major surgery | 80/211 (37.9) | 33 (28.5) | 47 (49.5) | 0.002 |
| Previous isolation of CPE | 5/178 (2.8) | 4 (3.8) | 1 (1.4) | 0.650 |
| Previous KPC-producing CPE | 2/178 (1.1) | 1 (1.0) | 1 (1.4) | 1.000 |
| Previous MBL-producing CPE | 2/178 (1.1) | 1 (1.0) | 1 (1.4) | 1.000 |
| Previous OXA-48-producing CPE | 1/178 (0.6) | 1 (1.0) | 0 (0.0) | 1.000 |
| Previous isolation of CRAB | 1/178 (0.6) | 0 (0.0) | 1 (1.4) | 0.410 |
| Previous isolation of CRPA | 3/178 (1.7) | 0 (0.0) | 3 (4.1) | 0.067 |
| Previous isolation of MRSA | 2/178 (1.1) | 2 (1.9) | 0 (0.0) | 0.513 |
| Variables at VABP onset | ||||
| Days from ICU admission to VABP onset, median (IQR) | 5 (2–12) | 5 (2–13) | 5 (2–12) | 0.734 |
| SOFA score, median (IQR) | 7 (4–9) | 7 (4–9) | 6 (4–8) | 0.344 |
| Presence of CVC | 234 (98.7) | 129 (97.7) | 105 (100.0) | 0.257 |
| Presence of septic shock | 68/230 (29.6) | 35 (27.6) | 33 (32.0) | 0.459 |
| Presence of ARDS | 106/234 (45.3) | 59 (44.7) | 47 (46.1) | 0.833 |
| Neutropenia | 16/236 (6.8) | 5 (3.8) | 11 (10.6) | 0.039 |
| CRRT | 54/234 (23.1) | 33 (25.2) | 21 (20.4) | 0.387 |
| ECMO | 8/233 (3.4) | 5 (3.9) | 3 (2.9) | 1.000 |
| KDIGO stage of AKI, median (IQR) | 0 (0–2) | 0 (0–1) | 0 (0–2) | 0.464 |
| Concomitant BSI | 39 (16.5) | 13 (9.8) | 26 (24.8) | 0.002 |
| Other concomitant infections requiring antibiotic therapy | 41/230 (17.8) | 22 (16.8) | 19 (19.2) | 0.638 |
| Causative agents and VABP therapy | ||||
| Causative agents of VABP** | ||||
| Pseudomonas aeruginosa | 58 (24.5) | 29 (22.0) | 29 (27.6) | 0.315 |
| Acinetobacter spp. | 19 (8.0) | 8 (6.1) | 11 (10.5) | 0.214 |
| Members of Enterobacterales | 131 (55.3) | 79 (59.9) | 52 (49.5) | 0.112 |
| Staphylococcus aureus | 47 (19.8) | 34 (25.8) | 13 (12.4) | 0.010 |
| Streptococcus pneumoniae | 9 (3.8) | 8 (6.1) | 1 (1.0) | 0.046 |
| Haemophilus influenzae | 30 (12.7) | 24 (18.2) | 6 (5.7) | 0.004 |
| Stenotrophomonas maltophilia | 12 (5.1) | 2 (1.5) | 10 (9.5) | 0.005 |
| Days from VABP onset to appropriate antibiotic therapy | 1 (0–3) | 1 (0–3) | 1 (0–3) | 0.457 |
| Cytokine blood filtration | 4/224 (1.8) | 1 (0.8) | 3 (2.9) | 0.333 |
| Intravenous immunoglobulins | 0.628 | |||
| Polyclonal intravenous immunoglobulins | 4/223 (1.8) | 3 (2.5) | 1 (1.0) | |
| IgM-enriched intravenous immunoglobulins | 1/223 (0.5) | 1 (0.8) | 0 (0.0) |
AKI, acute kidney injury; ARDS, acute respiratory distress syndrome; BAL, bronchoalveolar lavage; BSI, bloodstream infection; COPD, chronic obstructive pulmonary disease; CPE, carbapenemase-producing Enterobacterales; CRAB, carbapenem-resistant Acinetobacter baumannii; CRPA, carbapenem-resistant Pseudomonas aeruginosa; CRRT, continuous renal replacement therapy; CVC, central venous catheter; ECMO, extracorporeal membrane oxygenation; HIV, human immunodeficiency virus; HSCT, hematopoietic stem cell transplantation; ICU, intensive care unit; IgM, immunoglobulin M; IQR, interquartile range; KDIGO, Kidney Disease: Improving Global Outcomes; KPC, Klebsiella pneumoniae carbapenemase; MBL, metallo β-lactamases; NYHA, New York Heart Association; OXA, oxacillinase; PNplus, Pneumonia Plus; SOFA, sequential organ failure assessment; SOT, solid organ transplantation; VABP, ventilator-associated bacterial pneumonia
* Results are presented as No. of patients/Total of patients unless otherwise indicated. Number of missing values per variable were as follows (reflected in frequencies calculations): Diabetes mellitus (n = 5/237); COPD (n = 10/237); NYHA score (n = 9/237); Chronic liver disease (n = 8/237); Chronic kidney disease (n = 6/237); Chronic intermittent hemodialysis (n = 3/237); Solid neoplasm (n = 5/237); Metastatic solid neoplasm (n = 5/237); Hematological malignancy (n = 4/237); Previous HSCT (n = 4/237); Previous SOT (n = 4/237); HIV infection (n = 9/237); Autoimmune disease (n = 6/237); Previous antibiotic therapy (n = 54/237); Previous antifungal therapy (n = 57/237); Previous Chemotherapy (n = 14/237); Previous steroid therapy (n = 56/237); Previous therapy with immunosuppressants (n = 59/237); Previous major surgery (n = 26/237); Previous isolation of CPE (n = 59/237); Previous isolation of CRAB (n = 59/237); Previous isolation of CRPA (n = 59/237); Previous isolation of MRSA (n = 59/237); Days from ICU admission to VABP onset (n = 27/237); Presence of septic shock (n = 7/237); Presence of ARDS (n = 3/237); Neutropenia (n = 1/237); CRRT (n = 3/237); ECMO (n = 4/237); Other concomitant infections requiring antibiotic therapy (n = 7/237); Days from VABP onset to appropriate antibiotic therapy (n = 21/237); Cytokine blood filtration (n = 13/237); Polyclonal intravenous immunoglobulins (n = 14/237); IgM-enriched intravenous immunoglobulins (n = 14/237). No missing values were registered for all other remaining variables
** Non-mutually exclusive
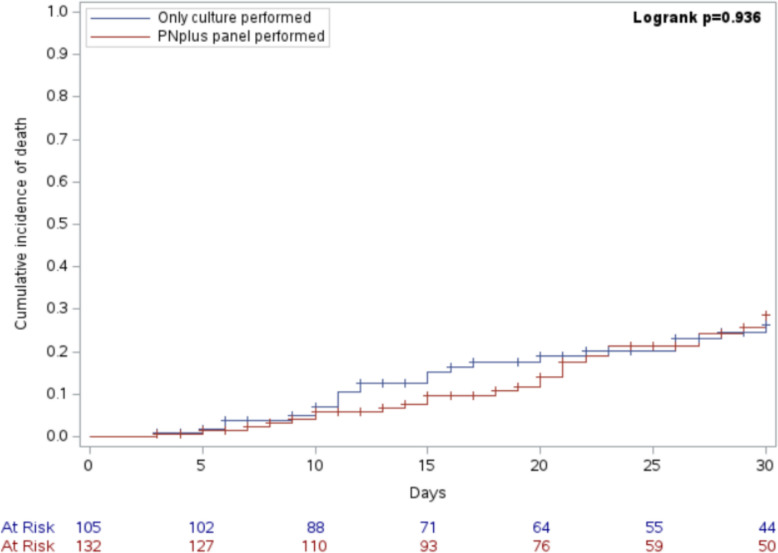
As shown in Fig. 2, unadjusted 30-day cumulative mortality was 28.7% (confidence interval [CI] 19.1%−38.3%) in patients with VABP in whom the PNplus panel was performed and 26.4% (CI 16.7%−36.1%) in patients with VABP in whom only BAL culture was performed (log-rank test, p = 0.936). Results of univariable and multivariable analyses of factors associated with 30-day mortality are reported in Supplementary Table S1 and Table 2, respectively. In the final multivariable model (model A), SOFA score (HR 1.13, 95% CI 1.04–1.22, p = 0.003), previous isolation of CRPA (HR 3.02, 95% CI 1.25–7.32, p = 0.015), and solid neoplasm (HR 2.15, 95% CI 1.12–4.14, p = 0.022) were associated with increased mortality, while no association was registered for PNplus panel performed (HR 1.07, 95% CI 0.59–1.93, p = 0.825). The direction of effects in model A was confirmed in model B, also including center as shared frailty (Supplementary Table S2).
Fig. 2.
Unadjusted cumulative mortality up to Day 30 in patients with VABP. Unadjusted cumulative mortality up to Day 30 in patients with VABP in whom the PNplus panel was performed (red line) and patients with VABP in whom only BAL culture was performed (blue line). The time of origin was set as the day of VABP onset. Death was the event of interest and right-censoring was applied at the end of follow-up (ICU discharge or Day 30, whichever came first). BAL, bronchoalveolar lavage; ICU, intensive care unit; VABP, ventilator-associated bacterial pneumonia
Table 2.
Multivariable analysis of factors associated with 30-day mortality
| Variable | HR (95% CI) | p-value |
|---|---|---|
| PNplus panel performed | 1.07 (0.59–1.93) | 0.825 |
| Solid neoplasm | 2.15 (1.12–4.14) | 0.022 |
| Previous isolation of CRPA | 3.02 (1.25–7.32) | 0.015 |
| SOFA score | 1.13 (1.04–1.22) | 0.003 |
Only variables retained in the final multivariable model after backward stepwise selection are presented. Variables included in the initial multivariable model were the following: PNplus panel performed; age; solid neoplasm; previous isolation of CRPA; SOFA score; ECMO; age-adjusted Charlson Comorbidity Index; previous therapy with immunosuppressants; presence of septic shock. The variable PNplus panel performed was retained in the final multivariable model independent of stepwise backward selection, in line with the purpose of the study. CI, confidence interval; CRPA, carbapenem-resistant Pseudomonas aeruginosa; ECMO, extracorporeal membrane oxygenation; HR, hazard ratio; PNplus, Pneumonia Plus; SOFA, sequential organ failure assessment
With regard to the secondary analyses of factors associated with timing of antibiotic discontinuation, results of univariable and multivariable models are reported in Supplementary Table S3 and Table 3, respectively. In the final multivariable model (model C), days from VABP onset to appropriate antibiotic therapy (sHR for one day increase 0.88, 95% CI 0.81–0.95, p = 0.002), concomitant BSI (sHR 0.50, 95% CI 0.28–0.88, p = 0.016), other concomitant infections requiring antibiotic therapy (sHR 0.53, 95% 0.34–0.84, p = 0.006), chronic intermittent hemodialysis (sHR 0.37, 95% CI 0.18–0.75, p = 0.006), solid neoplasm (sHR 0.54, 95% CI 0.31–0.93, p = 0.025), and Acinetobacter spp. as etiological agent (sHR 0.27; 95% CI 0.11–0.65, p = 0.004) were associated with delayed discontinuation, while no association was registered for PNplus panel performed (sHR 1.03, 95% CI 0.75–1.40, p = 0.871). The direction of effects in model C was confirmed in model D, also accounting for center-level clustering through robust variance estimation (Supplementary Table S4).
Table 3.
Multivariable analysis of factors associated with earlier antibiotic discontinuation
| Variable | sHR (95% CI) | p-value |
|---|---|---|
| PNplus panel performed | 1.03 (0.75–1.40) | 0.871 |
| Chronic intermittent hemodialysis | 0.37 (0.18–0.75) | 0.006 |
| Solid Neoplasm | 0.54 (0.31–0.93) | 0.025 |
| Concomitant BSI | 0.50 (0.28–0.88) | 0.016 |
| Other concomitant infections requiring antibiotic therapy | 0.53 (0.34–0.84) | 0.006 |
| Acinetobacter spp. as causative agent of VABP | 0.27 (0.11–0.65) | 0.004 |
| Days from VABP onset to appropriate antibiotic therapy* | 0.88 (0.81–0.95) | 0.002 |
Fine-Gray model with discontinuation of antibiotic therapy as the event of interest and death as competing risk. Only variables retained in the final multivariable model after backward stepwise selection are presented. Variables included in the initial multivariable model were the following: PNplus panel performed; chronic liver disease; chronic kidney diseases; chronic intermittent hemodialysis; solid neoplasm; previous isolation of CPE; previous KPC-producing CPE; previous isolation of CRPA; presence of septic shock; presence of ARDS; concomitant BSI; other concomitant infections requiring antibiotic therapy; Acinetobacter spp. as VABP causative agent; members of the Enterobacterales as VABP causative agents; Days from VABP onset to appropriate antibiotic therapy. The variable PNplus panel performed was retained in the final multivariable model independent of stepwise backward selection, in line with the purpose of the study. ARDS, acute respiratory distress syndrome; BSI, bloodstream infection; CI, confidence interval); CPE, carbapenemase-producing Enterobacterales; CRPA, carbapenem-resistant Pseudomonas aeruginosa; KPC, Klebsiella pneumoniae carbapenemase; PNplus, Pneumonia Plus; sHR, subdistribution hazard ratio; VABP, ventilator-associated bacterial pneumonia
* Modeled as fixed, despite the time of origin of the Fine-Gray analysis corresponding to the start of any therapy (either initially appropriate or inappropriate). This choice was supported by the non-significance of a time interaction term (p = 0.055)
The results of sensitivity analyses for predictors of mortality and earlier antibiotic discontinuation in patients without concomitant BSI were consistent in terms of direction of effect with those of the main analyses conducted in the entire study population (see Supplementary Tables S5 and S6).
Time to appropriate antibiotic therapy was 1 day (interquartile range [IQR] 0–3) both in patients who underwent PNplus panel testing on BAL and in patients who underwent BAL culture only (p = 0.457). However, the composite outcome (antibiotic de-escalation in patients who received appropriate therapy from day 0 or initiation of an appropriate therapy from day 1 in those who did not initiate an appropriate therapy on day 0) was registered more frequently in patients who underwent PNplus panel testing on BAL (41.3% [52/126] vs. 27.8% [25/90], p = 0.041). Results stratified for the two different components of the composite outcome are displayed graphically in Fig. 3. The incidence rate of candidemia was 14 per 1000 patient-days in ICU both in patients who underwent and in patients who did not underwent PNplus panel testing (incidence rate ratio 0.99; 95% CI 0.30–3.24, p = 0.986). No CDI episodes were reported.
Fig. 3.
Composite outcome of antibiotic de-escalation in patients who received appropriate therapy from VABP onset (day 0) or initiation of an appropriate therapy from day 1 in those who did not initiate an appropriate therapy on day 0. p values from chi square test. VABP, ventilator-associated bacterial pneumonia. In patients with PNplus panel performed, de-escalation was obtained through discontinuation of anti-Gram positive agents (15/24, 62.5%), discontinuation of anti-Gram negative agents (5/24, 20.8%), change to an agent with narrower spectrum (3/24, 12.5), or discontinuation of ani-anaerobes agents (1/24,4.2%). In patients with only culture performed, de-escalation was obtained through discontinuation of anti-Gram positive agents (4/8, 50.0%), discontinuation of anti-Gram negative agents (2/8, 25.0%), or change to an agent with narrower spectrum (2/8, 25.0%). In patients with PNplus panel performed, initiation of appropriate therapy from day 1 occurred because of initiation of any therapy on day 1 (19/28, 67.9%), or change of therapy on day 1 (9/28, 32.1%). In patients with only culture performed, initiation of appropriate therapy from day 1 occurred because of initiation of any therapy on day 1 (15/17, 88.2%), or change of therapy on day 1 (2/17, 11.8%)
Discussion
PNplus panel testing on BAL in patients with VABP resulted in higher frequencies of antibiotic therapy de-escalation and initiation of appropriate therapy on day 1 from VABP onset, compared with treatment decisions based on availability of BAL culture results only. The use of rapid molecular testing did not impact 30-day mortality and time to antibiotic therapy discontinuation.
The lack of an effect on mortality testifies to the safety of treatment decisions based on PNplus panel results and in line with antimicrobial stewardship principles (e.g., increased rates of de-escalation). Overall, our results are in line with those of the INHALE WP3 multicenter, randomized, controlled trial (RCT), in which, in a study population of 545 patients with suspected hospital-acquired/ventilator-associated pneumonia (eventually, 64% were ventilator-associated among confirmed pneumonia cases), 28-day mortality was similar in the intervention (diagnostic algorithm including PNplus panel) and standard of care arms (31.3% vs. 28.2%, HR 1.18; 95% CI 0.87–1.61) [25]. Moreover, in the INHALE WP3 RCT appropriate antibiotic therapy within 24 h from randomization was more frequent in the intervention arm than in the standard of care arm (76.5% vs. 55.9%; estimated difference 21%; 95% CI 13–28%). The same was not registered in our observational study, in which the median time to appropriate antibiotic therapy was 1 day from VABP onset (IQR 0-3) with both the PNplus-including approach and the only culture approach, possibly reflecting the high rate of appropriate empirical therapy since VABP onset (> 25%) in the study population. What differed between the two approaches in our study was the length of empirical appropriate therapy, that, owing to the higher frequency of de-escalation, was shorter in the PNplus-guided approach.
Time to appropriate antibiotic therapy was shorter and the rate of early de-escalation was higher by employing the BioFire FilmArray pneumonia panel in another single-center RCT conducted among patients with suspected pneumonia, although the eventual number of VABP in the study was low (< 20%) [26]. Of note, also in this latter study mortality was not different between the panel-guided and the conventional approaches (22.6% vs. 20.4%, p = 0.42) [26]. Overall, results from observational studies in terms of the impact of molecular syndromic panels on the timing of appropriate antibiotic therapy in patients with VABP are heterogenous and likely depending on local rates of resistance and empirical treatment protocols. Nonetheless these studies consistently demonstrate that rapid molecular tests are able to influence therapeutic decisions in patients with VABP, usually with a potential advantage in terms of antimicrobial stewardship-related endpoints (e.g., reduced time to diagnosis, reduced time to first antibiotic change) [27–32].
The apparent lack of impact of the PNplus panel on anticipating antibiotic therapy discontinuation in our study could again rely on the high rate of appropriate empirical therapy from VABP onset. Indeed, even if de-escalated less often in the culture only approach, appropriate empirical, broad-spectrum antibiotic therapies are likely not to impact the total duration of VABP treatment (differently from delayed appropriate therapy, as shown in our analysis). This introduces a first limitation of our study also shared by other experiences in the literature, that is, no data is available on the long term impact of PNplus guided treatment decisions. In our opinion, this would require dedicated study designs, able to capture any possible long-term favorable impact of the use of the PNplus panel, and represents the next step in its real-life assessment, now that current studies have frequently demonstrated a substantial lack of unfavorable effects in terms of relevant clinical outcomes, e.g., short-term mortality.
The current study has some limitations. The first one is the possible heterogeneity of clinicians’ decisions on the basis of PNplus panel results, that was not standardized across centers. To limit the relevance of between-center heterogeneity in our study, we used shared frailty models and center-level clustering. A second important limitation is the possible selection bias related to the real-life nature of the study. Indeed, PNplus panel testing might have been deemed unnecessary according to clinical judgment in some patients who underwent only conventional culture. Third, part of the clinical impact of PNplus panel testing could depend on the etiology of VABP and the rate/type of multidrug-resistant organisms. For example, we cannot exclude, also in line with some existent literature discussed above, that different settings with higher endemicity for carbapenemase-producing organisms and/or for some high-burden pathogens (e.g. Acinetobacter spp.) could benefit from the use of rapid molecular testing also in terms of time to appropriate therapy. Fourth, while we selected our primary and secondary endpoints to specifically investigate the possible impact of PNplus panel on survival and immediate stewardship-related outcomes, other outcomes such as possible impact on ICU length of stay and hospital length of stay were not investigated by design and may deserve dedicated post-hoc investigation. Fifth, it cannot be ruled out that, although PNplus panel results are communicated rapidly to clinicians via phone calls or notifications as part of institutional antimicrobial stewardship and diagnostic protocols at all participating centers, delays in communication may have occurred in some cases, such as the absence or limited number of dedicated staff at night, which represent a potential unmeasured confounder. Finally, the composite secondary outcome of antibiotic de-escalation in patients receiving appropriate therapy from VABP onset (day 0) and start of an appropriate therapy on day 1 after VABP onset, while having been rationally defined to capture those choices occurring before BAL culture results (i.e., those in which the impact of PNplus results on clinical decisions could be maximized), may still require external evaluation and validation. Regarding strengths of our study, the large sample of patients with VABP and not only of suspected pneumonia, conferring homogeneity to the entire study population, should be highlighted.
In conclusion, our study confirmed that PNplus panel testing in patients with VABP is able to impact antibiotic decisions, without affecting mortality. Further study is necessary to assess the long-term effects in terms of antimicrobial stewardship of PNplus panel-based antibiotic treatment decisions.
Supplementary Information
Acknowledgements
None.
RAPID-SITA PHENOTYPES investigators (collaborators): Ylenia Murgia; Gabriele Di Meco; Alice Cappello; Sabrina Guastavino; Cristina Campi; Michele Piana; Sara Mora; Nicola Rosso; Antonio Di Biagio; Giulia Viglietti; Annaflavia Lamarina; Luca De Paola; Stefano Antola; Selene Gallone; Federica Cosentino; Chiara Gullotta; Maria Vittoria Cossu; Marco Piscaglia; Federico D’Amico; Serena Sassi; Elena Simoncini; Maria Carolina Benvenuto; Giacomo Gonnelli; Anna Gidari; Lisa Malincarne; Elisabetta Blasi Vacca; Nicoletta Bobbio; Andrea Parisini; Annalisa Garlaschelli; Sara Grignolo; Fabiola Cammarota; Paola Del Giacomo; Angela Raffaella Losito; Brunella Posteraro; Andrea Lombardi; Antonio Muscatello; Carlo Peri; Mara Tomasello; Nicoletta Boffa; Elena Potenza; Flavia Badalucco Ciotta; Antonella Castagna; Roberta Monardo; Chiara Tassan Din; Giulia Catalisano; Michela Di Maio; Antonino Giarratano; Mariachiara Ippolito; Pierluigi Viale.
Author contributions
Daniele Roberto Giacobbe, Vincenzo Di Pilato, and Matteo Bassetti conceived the research question and all authors participated in design of the study. All authors participated in data acquisition in the different participating centers. Daniele Roberto Giacobbe, Marco Muccio, Greta Cattardico, and Claudia Bartalucci contributed to data analysis. Daniele Roberto Giacobbe, Greta Cattardico, Claudia Bartalucci, and Vincenzo Di Pilato drafted the manuscript, which all authors contributed to revising. The corresponding author attests that all listed authors meet authorship criteria and that no others meeting the criteria have been omitted. All authors read and approved the final manuscript.
Funding
The RAPID SITA PHENOTYPES project was funded by an investigator-initiated research grant (BFD-MU-23–002) from bioMérieux. The funder had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Data availability
The data presented in this study will be available from the corresponding author on reasonable request and provided all regulatory and privacy requirements are fulfilled.
Declarations
Ethical approval
The MULTI-SITA project was approved by the ethics committee of the coordinating center (Liguria Region Ethics Committee, registry number 390/2020), with a subsequent amendment authorizing the conduct of the RAPID-SITA PHENOTYPES study within the MULTI-SITA project. The other participating centers followed the local ethical committees requirements and started to enroll patients prospectively once activated. Conscious patients at time of enrollment signed an informed consent to participate in the study. A waiver of informed consent for data collection from unconscious patients at the time of enrollment due to severe clinical conditions was obtained within the ethics committee approval, in line with the observational nature of the analyses and in order not to bias research results towards low mortality prejudicing scientific validity.
Competing interests
Outside the submitted work, Matteo Bassetti has received funding for scientific advisory boards, travel, and speaker honoraria from Cidara, Gilead, Menarini, MSD, Mundipharma, Pfizer, and Shionogi. Outside the submitted work, Daniele Roberto Giacobbe reports investigator-initiated grants from Pfizer, Shionogi, Menarini, Tillotts Pharma, and Gilead Italia, travel support from Pfizer, and speaker/advisor fees from Pfizer, bioMérieux, Advanz Pharma, Menarini, and Tillotts Pharma. Outside the submitted work, Vincenzo Di Pilato reports travel and speaker honoraria from Arrow Diagnostics. Outside the submitted work, Andrea Cortegiani has received fees for lectures/advisory board membership from Gilead, MSD, Mundipharma, and Pfizer. Outside the submitted work, Gian Maria Rossolini has received research grants for the laboratory, funding for scientific advisory boards and/or speaker engagements from ADA, Advanz Pharma, Alifax, Arrow Diagnostics, bioMérieux, Cepheid, Diesse, Hain Life Sciences, Menarini, Meridian, MSD, Pfizer, Qiagen, Q-linea, Quantamatrix, Quidel, Qvella, SD Biosensor, Seegene, Shionogi, Syncells, Viatris, and Zambon. The other authors report no conflicts of interest.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Daniele Roberto Giacobbe, Email: danieleroberto.giacobbe@unige.it.
RAPID-SITA PHENOTYPES investigators:
Ylenia Murgia, Gabriele Di Meco, Alice Cappello, Sabrina Guastavino, Cristina Campi, Michele Piana, Sara Mora, Nicola Rosso, Antonio Di Biagio, Giulia Viglietti, Annaflavia Lamarina, Luca De Paola, Stefano Antola, Selene Gallone, Federica Cosentino, Chiara Gullotta, Marco Piscaglia, Federico D’Amico, Serena Sassi, Elena Simoncini, Giacomo Gonnelli, Anna Gidari, Lisa Malincarne, Nicoletta Bobbio, Andrea Parisini, Annalisa Garlaschelli, Sara Grignolo, Fabiola Cammarota, Paola Del Giacomo, Brunella Posteraro, Andrea Lombardi, Antonio Muscatello, Carlo Peri, Mara Tomasello, Nicoletta Boffa, Elena Potenza, Antonella Castagna, Roberta Monardo, Giulia Catalisano, Michela Di Maio, Antonino Giarratano, Mariachiara Ippolito, and Pierluigi Viale
References
- 1.Berteau F, Kouatchet A, Le Gall Y, et al. Epidemiology and prediction of non-targeted bacteria by the filmarray pneumonia plus panel in culture-positive ventilator-associated pneumonia: a retrospective multicentre analysis. Ann Intensive Care. 2025;15(1):57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Koulenti D, Tsigou E, Rello J. Nosocomial pneumonia in 27 ICUs in Europe: perspectives from the EU-VAP/CAP study. Eur J Clin Microbiol Infect Dis. 2017;36(11):1999–2006. [DOI] [PubMed] [Google Scholar]
- 3.Melsen WG, Rovers MM, Groenwold RH, et al. Attributable mortality of ventilator-associated pneumonia: a meta-analysis of individual patient data from randomised prevention studies. Lancet Infect Dis. 2013;13(8):665–71. [DOI] [PubMed] [Google Scholar]
- 4.Colaneri M, Montrucchio G, Scaglione G, et al. Incidence, microbiology and mortality of ventilation-associated pneumonia in a large Italian cohort of critically ill patients. Clin Microbiol Infect: Results PROSA proj; 2025. [DOI] [PubMed] [Google Scholar]
- 5.Giacobbe DR, Di Pilato V, Vena A, et al. Interpreting the results of rapid molecular diagnostic tests for carbapenem-resistant Enterobacterales infection: current clinical perspective while waiting for further evidence. Expert Rev Mol Diagn. 2024;24(7):583–90. [DOI] [PubMed] [Google Scholar]
- 6.Stewart SJ, Pandolfo AM, Jani Y, et al. Guidelines vs mindlines: a qualitative investigation of how clinicians’ beliefs influence the application of rapid molecular diagnostics in intensive care. Antimicrob Agents Chemother. 2025;69(3):e0115624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bassetti M, Giacobbe DR, Vena A, et al. Challenges and research priorities to progress the impact of antimicrobial stewardship. Drugs Context. 2019;8:212600. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Dumkow LE, Worden LJ, Rao SN. Syndromic diagnostic testing: a new way to approach patient care in the treatment of infectious diseases. J Antimicrob Chemother. 2021;76(Suppl 3):iii4-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Dessajan J, Timsit JF. Impact of multiplex PCR in the therapeutic management of severe bacterial pneumonia. Antibiotics (Basel). 2024. 10.3390/antibiotics13010095. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lombardi A, Renisi G, Liparoti A, et al. Application of Syndromic Panels for respiratory Tract Infections in Lung Transplantation: A Critical Review on Current Evidence and Future Perspectives. Transpl Infect 2025.Dis.:e14448. [DOI] [PubMed]
- 11.Mangioni D, Panigada M, Palomba E, et al. Incidence, microbiological and immunological characteristics of ventilator-associated pneumonia assessed by bronchoalveolar lavage and endotracheal aspirate in a prospective cohort of COVID-19 patients: CoV-AP study. Crit Care. 2023;27(1):369. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Posteraro B, Cortazzo V, Liotti FM, et al. Diagnosis and treatment of bacterial pneumonia in critically ill patients with COVID-19 using a multiplex PCR assay: a large Italian hospital’s five-month experience. Microbiol Spectr. 2021;9(3):e0069521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Food and Drug. Administration Hospital-Acquired Bacterial Pneumonia and Ventilator-Associated Bacterial Pneumonia: Developing Drugs for Treatment Guidance for Industry (2025). 2020 Available at: https://www.fda.gov/media/79516/download.
- 14.Giacobbe DR, Battaglini D, Enrile EM, et al. Incidence and prognosis of ventilator-associated pneumonia in critically ill patients with COVID-19: a multicenter study. J Clin Med. 2021. 10.3390/jcm10040555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Bartoletti M, Giannella M, Lewis R, et al. A prospective multicentre study of the epidemiology and outcomes of bloodstream infection in cirrhotic patients. Clin Microbiol Infect. 2018;24(5):546 e1-546 e8. [DOI] [PubMed] [Google Scholar]
- 16.Charlson M, Szatrowski TP, Peterson J, et al. Validation of a combined comorbidity index. J Clin Epidemiol. 1994;47(11):1245–51. [DOI] [PubMed] [Google Scholar]
- 17.Vincent JL, Moreno R, Takala J, et al. 1996. The SOFA (Sepsis-related Organ Failure Assessment) score to describe organ dysfunction/failure. On behalf of the Working Group on Sepsis-Related Problems of the European Society of Intensive Care Medicine. Intensive Care Med. 1996 22(7) 707–10. [DOI] [PubMed]
- 18.Singer M, Deutschman CS, Seymour CW, et al. The third international consensus definitions for sepsis and septic shock (Sepsis-3). JAMA. 2016;315(8):801–10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Force ADT, Ranieri VM, Rubenfeld GD, et al. Acute respiratory distress syndrome: the berlin definition. JAMA. 2012;307(23):2526–33. [DOI] [PubMed] [Google Scholar]
- 20.Kellum JA, Lameire N, Group KAGW. Diagnosis, evaluation, and management of acute kidney injury: a KDIGO summary (Part 1). Crit Care. 2013;17(1):204. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Garnacho-Montero J, Escoresca-Ortega A, Fernandez-Delgado E. Antibiotic de-escalation in the ICU: how is it best done? Curr Opin Infect Dis. 2015;28(2):193–8. [DOI] [PubMed] [Google Scholar]
- 22.Yuan YC. Multiple Imputation for Missing Data: Concepts and New Development (Version 9.0). SAS Institute Inc.; 2010 [2023]. Available at: https://support.sas.com/rnd/app/stat/papers/multipleimputation.pdf.
- 23.Balan TA, Putter H. A tutorial on frailty models. Stat Methods Med Res. 2020;29(11):3424–54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fine JP, Gray RJ. A proportional hazards model for the subdistribution of a competing risk. J Am Stat Assoc. 1999;94(446):496–509. [Google Scholar]
- 25.Enne VI, Stirling S, Barber JA, et al. Inhale WP3, a multicentre, open-label, pragmatic randomised controlled trial assessing the impact of rapid, ICU-based, syndromic PCR, versus standard-of-care on antibiotic stewardship and clinical outcomes in hospital-acquired and ventilator-associated pneumonia. Intensive Care Med. 2025;51(2):272–86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Virk A, Strasburg AP, Kies KD, et al. Rapid multiplex PCR panel for pneumonia in hospitalised patients with suspected pneumonia in the USA: a single-centre, open-label, pragmatic, randomised controlled trial. Lancet Microbe. 2024;5(12):100928. [DOI] [PubMed] [Google Scholar]
- 27.Chen CL, Tseng HY, Chen WC, et al. Application of a multiplex molecular pneumonia panel and real-world impact on antimicrobial stewardship among patients with hospital-acquired and ventilator-associated pneumonia in intensive care units. J Microbiol Immunol Infect. 2024;57(3):480–9. [DOI] [PubMed] [Google Scholar]
- 28.Esplund JN, Taylor AD, Stone TJ, et al. Clinical impact of a multiplex rapid diagnostic pneumonia panel in critically ill patients. Antimicrob Steward Healthc Epidemiol. 2023;3(1):e5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Stafylaki D, Maraki S, Vaporidi K, et al. Impact of molecular syndromic diagnosis of severe pneumonia in the management of critically ill patients. Microbiol Spectr. 2022;10(5):e0161622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Verroken A, Favresse J, Anantharajah A, et al. Optimized antibiotic management of critically ill patients with severe pneumonia following multiplex polymerase chain reaction testing: a prospective clinical exploratory trial. Antibiotics. 2024. 10.3390/antibiotics13010067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Bay P, Fihman V, Woerther PL, et al. Performance and impact of rapid multiplex PCR on diagnosis and treatment of ventilated hospital-acquired pneumonia in patients with extended-spectrum beta-lactamase-producing Enterobacterales rectal carriage. Ann Intensive Care. 2024;14(1):118. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Monard C, Pehlivan J, Auger G, et al. Multicenter evaluation of a syndromic rapid multiplex PCR test for early adaptation of antimicrobial therapy in adult patients with pneumonia. Crit Care. 2020;24(1):434. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The data presented in this study will be available from the corresponding author on reasonable request and provided all regulatory and privacy requirements are fulfilled.