Abstract
While the Southern Ocean represents a unique habitat, currently undergoing rapid environmental change, its biodiversity remains largely unknown, particularly at greater depths. Increased sampling efforts in the Amundsen Sea, a previously unexplored region of the Southern Ocean, combined with the use of an epibenthic sledge resulted in a large collection of mobile, scale-bearing worms from the family Polynoidae Kinberg, 1856. The greatest taxonomic novelty in the material collected from the Pine Island Bay, Amundsen Sea, was found within the exclusively deep-sea subfamily Macellicephalinae Hartmann-Schröder, 1971. Examination of this material has already led to formalization of six new species of Macellicephala (Neal et al. 2018). This study represents the continuation of such effort with formalization of Macellicephaloides veronikae sp. n. based on morphology and 16S and 18S molecular markers. In the phylogenetic analyses, the new species is sister taxon to Macellicephaloides moustachu from the abyssal equatorial Pacific Ocean, albeit based on very limited taxon sampling currently available. Macellicephaloides veronikae sp. n. shows the shallowest distribution (500–1000 m) of this genus recorded to date and may represent a case of polar emergence.
Keywords: Taxonomic novelty, Molecular phylogeny, 16S, 18S, Antarctic shelf, Deep sea
Introduction
The Southern Ocean, a unique and rapidly changing habitat, still has much of its biodiversity unexplored, especially at greater depths. The region’s deep-sea ecosystems remain poorly understood, highlighting the need for further research to uncover their biodiversity (e.g. Griffiths 2010). The Amundsen Sea (Fig. 1a) is a significant body of water situated in western Antarctica, bordered by the Bellingshausen Sea and the Ross Sea, undergoing rapid environmental change due to melting of the Pine Island Glacier, which flows into Pine Island Bay, the Amundsen Sea’s principal embayment (Reed et al. 2024). The past glacial actions have also created a complex bathymetry of the shelf by carving deep troughs up to 1500 m deep (Fig. 1b).
Fig. 1.
Map of sampling sites and background environmental information. a Map of sampling sites visited during the BIOPEARL II—JR179 cruise to the Amundsen Sea (black circles). b Depth cross-section of the shelf and slope in the Amundsen Sea, showing deep-trough sites on the inner shelf (Pine Island Bay) and outer shelf/slope. Green circle: samples with Macellicephaloides veronikae presented in this study. Black circle: overall sampling effort
The first benthic samples collected from the Amundsen Sea in the Southern Ocean, as part of the BIOPEARL programme (Biodiversity, Phylogeny, Evolution and Adaptive Radiation of Life) (Linse et al. 2013), revealed significant taxonomic novelty in several macrofaunal groups, including annelid worms (e.g. Kaiser et al. 2009; Moreau et al. 2013; Pabis et al. 2015; Neal et al. 2018). These discoveries were particularly notable in deep-shelf and slope areas (500–1500 m), which had been underexplored in previous studies that focused on shallower depths (Griffiths 2010). Only the ANDEEP expeditions (ANtarctic benthic DEEP-sea biodiversity) had previously targeted deep-sea areas of the Southern Ocean, also finding high levels of novel biodiversity (e.g. Brandt et al. 2007; Schüller et al. 2009). The BIOPEARL programme employed a relatively new sampling device, the Brenke Epibenthic sledge (Brenke 2005), which helped capture highly mobile epibenthic fauna, including scale-bearing worms of family Polynoidae Kinberg, 1856, which had often been under-represented in earlier studies using coring devices (Neal et al. 2017).
Polynoidae, the largest family of scale-bearing annelids (Aphroditiformia), includes around 1000 species across approximately 160 genera and 8 subfamilies, making it one of the most diverse families among polychaetes (Read and Fauchald 2025). These errant worms were once thought to have a broad geographic and bathymetric distribution, although recent molecular studies have challenged this view (e. g. Bogantes et al. 2020; Serpetti et al. 2017). In the Southern Ocean, there are approximately 60 valid species of Polynoidae, which represents about 7.5% of the known polychaete fauna in the region estimated at around 800 species (Schüller and Ebbe 2014). Most polynoid diversity in the Southern Ocean has been observed in shallow waters (< 500 m), primarily in the subfamily Polynoinae Kinberg, 1856. However, some subfamilies are restricted to deep-sea habitats, including bathyal to hadal depths and chemosynthetic environments or shallow deep-sea analogues such as submarine caves, with the subfamily Macellicephalinae Hartmann-Schröder, 1971, being particularly prominent in such habitats.
Neal et al. (2018) already described in part the novelty found within the Antarctic Macellicephalinae by formalizing six new species of Macellicephala. Here, we continue the efforts of describing the polynoid fauna of the Southern Ocean with the formalization of new species in the genus Macellicephaloides. Genus Macellicephaloides Ushakov, 1955, was erected to accommodate three species discovered from the Kuril-Kamchatka Trench—M. grandicirra Uschakov, 1955, M. verrucosa Uschakov, 1955, and M. vitiazi Uschakov, 1955. Subsequent discovery of further four species from the trenches supported the hypothesis that the genus Macellicephaloides is adapted to hadal depths – M. uschakovi Levenstein, 1971, from the Kuril-Kamchatka Trench; M. improvisa Levenstein, 1982; and M. villosa Levenstein, 1982, from the Trench of Japan and M. sandvichensis Levenstein, 1975, from the South Sandwich Trench in the Atlantic sector of the Southern Ocean. Pettibone (1989a) described M. alvini, a bathyal representative, from the bacterial mats at hydrothermal vents in the Guaymas Basin, Gulf of California, at a depth of 2004 m. It took nearly three decades to describe the next species in this genus—M. moustachu Bonifácio and Menot, 2018, from the polymetallic nodule fields of the Clarion-Clipperton Zone in the abyssal equatorial Pacific. The discovery of Macellicephaloides veronikae sp. n., from the depths of ~ 500–1000 m in the Amundsen Sea, Southern Ocean, formalized here, is thus the shallowest record of this genus to date and may be an example of polar emergence (e.g. Brandt 1992; Strugnell et al. 2011), recently suggested for some, but not all Antarctic polynoids (Cowart et al. 2022).
Material and methods
Field sampling and laboratory analysis
Samples were collected during the BIOPEARL II expedition to the Amundsen Sea in austral summer of 2007/2008 onboard RRS James Clark Ross and organized by the British Antarctic Survey (see Linse et al. 2013 for details). Targeted depths were 500 m, 1000 m, and 1500 m horizons. The detailed description of epibenthic sledge (EBS) is given in Brenke (2005) and Glover et al. (2016). Briefly, the EBS consists of a 500-µm epi- (lower) and a supra- (upper) net, each with an opening of 100 cm width and 33 cm height. Both nets end up in cod ends with a mesh size of 300 µm. The EBS was hauled over the seabed at 1 knot for 10 min. The study areas, field methods, and treatment of polychaete samples on board and in the laboratory were described in detail in Neal et al. (2017), using protocols detailed in Glover et al. (2016).
SEM
Paratype NHMUK. 2018.246 was examined using SEM. The specimen was dehydrated in 100% alcohol, critically point dried, coated with gold-vanadium, and examined using Phillips XL30 SEM at the Imaging and Analysis Centre, Natural History Museum, London.
Molecular analysis
In total, DNA was extracted from 15 specimens morphologically assigned to genus Macellicephaloides in Neal et al. (2017). Holotype NHMUK. 2018.915 was targeted in this study with GenBank accession numbers PV911683 for 16S and PV911684 for 18S. Other specimens were targeted by Brasier et al. (2016), with GenBank accession numbers KX867331- KX867344.
DNA was extracted from parapodia using DNeasy Blood and Tissue Kit (Qiagen) following the protocol supplied by the manufacturer, with final elution in 100 μl buffer. Two genes were amplified: the mt non-coding 16S and the nuclear (n) non-coding 18S gene. About 450 bp of 16S and 1800 bp of 18S were amplified using the following primers: Ann16SF and 16SbrH for 16S (Palumbi 1996; Sjölin et al. 2005), and 18SA, 18SB, 620 F, and 1324R for 18S (Cohen et al. 1998; Medlin et al. 1988; Nygren and Sundberg 2003).
PCR mixtures contained 1 μl of each primer (10 μM), 2 μl template DNA, and 21 μl Red Taq DNA Polymerase 1.1X MasterMix (VWR) in a mixture totalling 25 μl. The temperature profile was as follows: 96 °C for 240 s, followed by (94 °C for 30 s, 48 °C for 30 s, then 72 °C for 60 s)*35 cycles, followed by 72 °C for 480 s. PCR purification was performed using a Millipore Multiscreen 96-well PCR Purification System, and sequencing was performed on an ABI 3730XL DNA Analyser (Applied Biosystems) at the Natural History Museum Sequencing Facility, using the primers mentioned above.
Overlapping sequence fragments were merged into consensus sequences using Geneious Prime 2025.0.3 (https://www.geneious.com). Gene 18S was used for the phylogenetic analyses. The 18S sequences were aligned together with other Polynoidae sequences and with Neoleanira tetragona (Örsted 1845) from Sigalionidae as root, using MAFFT (Katoh et al. 2002) with default settings, provided as plug-in in Geneious. Maximum likelihood (ML) analysis was performed using IQTree 1.6.0 (Nguyen et al. 2015) where ModelFinder (Kalyaanamoorthy et al. 2017) selected the optimal model TIMe + I + G4 for 18S. ML was run with 1000 bootstrap replicates. Bayesian phylogenetic analyses (BAs) using the model GTR + I + G were conducted with MrBayes 3.2.6 (Ronquist et al. 2012). Analyses were run independently three times for 10,000,000 generations. Of these, 2,500 000 generations were discarded as burn-in. The tree files were interpreted with FigTree v1.4.2 (http://tree.bio.ed.ac.uk/software/figtree/) and edited using Sketch (https://www.sketch.com).
All sequences obtained in this study have been deposited in GenBank (http://www.ncbi.nlm.nih.gov/genbank), with sequences deposited by Brasier et al. (2016) already available. GenBank sequence accession numbers are summarized in Table 1. Type material was deposited in the Natural History Museum, London, UK (NHMUK).
Table 1.
List of taxa included in the phylogenetic analysis, their GenBank accession numbers and their current placement within subfamilies of Polynoidae
| Taxon name | Genbank acc. no | Family/subfamily |
|---|---|---|
| Acholoe squamosa (Delle Chiaje, 1827) | AY839567.1 | Polynoinae |
| Antarctinoe ferox (Baird, 1865) | MG905039.1 | Polynoinae |
| Bathyedithia retierei Bonifácio & Menot, 2018 | MH233215.1 | Macellicephalinae |
| Bathyeliasona mariaae Bonifácio & Menot, 2018 | MH233204.1 | Macellicephalinae |
| Bathyfauvelia glacigena Bonifácio & Menot, 2018 | MH233236.1 | Macellicephalinae |
| Bathyfauvelia ignigena Bonifácio & Menot, 2018 | MH233246.1 | Macellicephalinae |
| Bathykurila guaymasensis Pettibone, 1989a | DQ074765.1 | Macellicephalinae |
| Bathymoorea lucasi Bonifácio & Menot, 2018 | MH233224.1 | Eulagiscinae |
| Bathypolaria magnicirrata (Neal et. al., 2012) | JX863895.1 | Macellicephalinae |
| Branchipolynoe pettiboneae Miura & Hashimoto, 1991 | KU507074.1 | Macellicephalinae |
| Bruunilla nealae Bonifácio & Menot, 2018 | MH233216.1 | Macellicephalinae |
| Bylgides elegans (Théel, 1879) | JN852822.1 | Polynoinae |
| Cladopolynoe sandersi Hiley et al., 2024 | JN852821.1 | Macellicephalinae |
| Eulagisca gigantea Monro, 1939 | MG905040.1 | Eulagiscinae |
| Gastrolepidia clavigera Schmarda, 1861 | JN852825.1 | Arctonoinae |
| Gorgoniapolynoe caeciliae (Fauvel, 1913) | KU738172.1 | Polynoinae |
| Gorgoniapolynoe corralophila (Day, 1960) | KU738175.1 | Polynoinae |
| Halosydna brevisetosa (Kinberg, 1856) | JN852827.1 | Lepidonotinae |
| Halosydnella australis (Kinberg, 1856) | KY823449.1 | Lepidonotinae |
| Harmothoe imbricata (Linnaeus, 1767) | AY340434.1 | Polynoinae |
| Hermenia verruculosa Grube, 1856 | JN852830.1 | Lepidonotinae |
| Hodor anduril Bonifácio & Menot, 2018 | MH233239.1 | Macellicephalinae |
| Hodor hodor Bonifácio & Menot, 2018 | MH233238.1 | Macellicephalinae |
| Hyperhalosydna striata (Kinberg, 1856) | JN852831.1 | Lepidastheniinae |
| Intoshella dictyaulus Sui et al., 2018 | MG519807.1 | Polynoinae |
| Lepidonotus clava (Montagu, 1808) | JN852833.1 | Lepidonotinae |
| Macellicephala brenesorum Neal et al., 2018 | MG905041.1 | Macellicephalinae |
| Macellicephala clarionensis Bonifácio & Menot, 2018 | MH233235.1 | Macellicephalinae |
| Macellicephala gloveri Neal et al., 2018 | MG905042.1 | Macellicephalinae |
| Macellicephala linseae Neal et al., 2018 | MG905043.1 | Macellicephalinae |
| Macellicephala monroi Neal et al., 2018 | MG905044.1 | Macellicephalinae |
| Macellicephala parvafauces Bonifácio & Menot, 2018 | MH233225.1 | Macellicephalinae |
| Macellicephala patersoni Neal et al., 2018 | MG905045.1 | Macellicephalinae |
| Macellicephala violacea (Levinsen, 1886) | OP476757.1 | Macellicephalinae |
| Macellicephaloides alvini Pettibone, 1989a | OP651045.1 | Macellicephalinae |
| Macellicephaloides moustachu Bonifácio & Menot, 2018 | MH233212.1 | Macellicephalinae |
| Macellicephaloides veronikae sp. n | PV911683-84 | Macellicephalinae |
| Malmgrenia mcintoshi (Tebble & Chambers, 1982) | JN852834.1 | Polynoinae |
| Neoleanira tetragona (Örsted, 1845) | AY839570.1 | Sigalionidae |
| Neopolynoe acanellae (Verrill, 1882) | MN653050.1 | Polynoinae |
| Nu aakhu Bonifácio & Menot, 2018 | MH233209.1 | Macellicephalinae |
| Pelagomacellicephala iliffei Pettibone, 1985 | KY454411.1 | Macellicephalinae |
| Photinopolynoe elytropapillata (Zhang et al., 2018) | MG799378.1 | Macellicephalinae |
| Polaruschakov lamellae Bonifácio & Menot, 2018 | MH233226.1 | Macellicephalinae |
| Polaruschakov omnesae Bonifácio & Menot, 2018 | MH233213.1 | Macellicephalinae |
| Polynoe scolopendrina Savigny, 1822 | JN852839.1 | Polynoinae |
| Robertianella synophthalma McIntosh, 1885 | MN653053.1 | Polynoinae |
| Themis undomarginata (Zhang et al., 2018) | MG799379.1 | Macellicephalinae |
| Thormora jukesii Baird, 1865 | JN852840.1 | Lepidonotinae |
Bold values indicates outlined the new species
In total, 49 terminal taxa were used in the molecular phylogenetic analysis (Table 1). Polynoidae were represented with 28 taxa from the subfamily Macellicephalinae, 11 from Polynoinae, five from Lepidonotinae Willey, 1902, two from Eulagiscinae Pettibone, 1997, and one each from Arctonoinae Hanley, 1989, and Lepidastheniinae Pettibone, 1989a, b (in Pettibone 1989b).
Results
Phylogeny
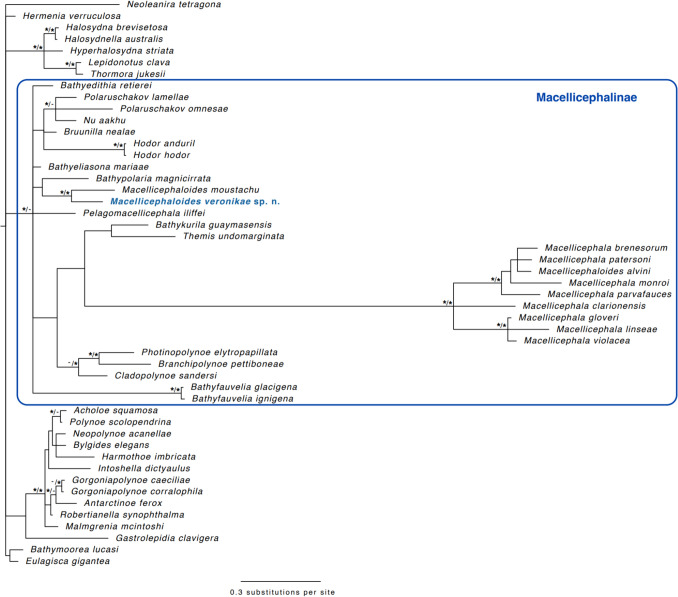
The phylogenetic analyses (Fig. 2) recovered Macellicephaloides veronikae sp. n. nested within the subfamily Macellicephalinae in agreement with Bonifácio and Menot (2019), as sister taxon to Macellicephaloides moustachu based on the currently available data. On GenBank, there are also sequences from a taxon labelled Macellicephaloides alvini Pettibone 1989a, b, which we included in our study. However, in our analyses, this taxon falls within the genus Macellicephala with strong support, which might be due to sequence contamination, or the sequenced specimen being misidentified.
Fig. 2.
Majority rule consensus tree from the Bayesian Analyses (BA) using 18S, with 48 taxa from Polynoidae and Neoleanira tetragona (Sigalionidae) used as root. The bootstrap support values from the maximum likelihood (ML) analyses are added in to the Bayesian tree as BA/ML on the nodes. Support values at or above 0.95 for the BA and at or above 80 from the ML are shown in the tree as asterisks. Support values from both analyses are shown as */* while if support is low from one of the analyses it is shown as */- or -/*. No symbols on nodes show there was low support in both analyses
Systematics
Family: Polynoidae Kinberg, 1856
Subfamily: Macellicephalinae Hartmann-Schröder, 1971.
Macellicephaloides Uschakov, 1955
Macellicephaloides veronikae sp. n.
Macellicephaloides sp. B in Brasier et al. (2016), Neal et al. (2017)
Fig. 3.
Macellicephaloides veronikae n. sp. Holotype (NHMUK. 2018.915). a Complete preserved specimen in dorsal view. b Complete preserved specimen in ventral view. c Anterior end in dorsal view. d Posterior end in dorsal view. All scale bars = 1000 µm
Fig. 7.
Comparison of shapes of parapodia of all known species of Macellicephaloides (a-i) and M. veronikae sp. n. (j), all parapodia cirrigerous with the exception of (e), line drawings not drawn to scale. a M. alvini after Pettibone (1989a, b), style of dorsal cirrus missing. b M. grandicirra after Uschakov (1955). c M. verrucosa after Uschakov (1955). d Anterior parapodium of M. vitiazi with reduced notopodium (top), posterior parpodium of M. vitiazi with elongated notopodium (bottom), both after Uschakov (1955). e elytragerous parapodium M. improvisa after Levenstein (1982). f M. sandwichensis after Levenstein (1975). g M. villosa after Levenstein (1982) showing papillae. h M. moustachu after Bonifácio and Menot (2019). i M. uschakovi after Levenstein (1971). j M. veronikae sp. n., cirrigerous parapodium showing papillae bordering cirrophore and neuropodium
Fig. 6.
Macellicephaloides veronikae n. sp. paratype (NHMUK. 2018.246, SEM specimen) light microscopy in a–c, SEM micrograph d–e. a Cirrigerous parapodium. b Detail of elytragerous parapodium, notopodial acicula (ntpa) marked by arrow; insert showing papillae on neuropodium as seen using stereomicroscopy. c Fascicle of neurochaetae. d Arrangement of alternating spines on neurochaetal shaft. e Detail of forked neurochaetal spine with unequal teeth. Scale bars: a, b = 100 µm, d = 5 µm, e = 1 µm
Material examined
Type material. Holotype NHMUK. 2018.915: Amundsen Sea (Pine Island Bay), RRS James Clarke Ross 179, station BIO4-EBS-3F, collected on 07 March 2008, epibenthic sledge, supra-net, latitude −74.38964, longitude −104.7645, depth 523 m. Paratype NHMUK. 2018.916: Amundsen Sea (Pine Island Bay), RRS James Clarke Ross 179, station BIO4-EBS-3F, collected on 07 March 2008, epibenthic sledge, supra-net, latitude −74.38964, longitude −104.7645, depth 523 m. Paratype (SEM specimen) NHMUK. 2018.246: Amundsen Sea (Pine Island Bay), RRS James Clarke Ross 179, station BIO6-EBS-2B, collected on 12 March 2008, epibenthic sledge supra-net, latitude −71.17799, longitude −109.88426, depth 1005 m. Other material: NHMUK.2018.154, Amundsen Sea (Pine Island Bay), RRS James Clarke Ross 179, station BIO6-EBS-3C, collected on 12 March 2008, epibenthic sledge, epi-net, latitude −71.34783, longitude −110.01908, depth 482 m, 1 specimen; NHMUK. 2018.915-916, Amundsen Sea (Pine Island Bay), RRS James Clarke Ross 179, station BIO4-EBS-3F, collected on 07 March 2008, epibenthic sledge, supra-net, latitude −74.38964, longitude −104.7645, depth 523 m, 2 specimens; NHMUK.2018.434-435, Amundsen Sea (Pine Island Bay), RRS James Clarke Ross 179, station BIO5-EBS-3F, collected on 10 March 2008, epibenthic sledge, epi-net, latitude −73.98821, longitude −107.39726, depth 543 m, 2 specimens; NHMUK.2018.898-902, Amundsen Sea (Pine Island Bay), RRS James Clarke Ross 179, station BIO4-EBS-3F, collected on 07 March 2008, epibenthic sledge, epi-net, latitude −74.38964, longitude −104.7645, depth 523 m, 5 specimens; NHMUK. 2018.1069-1078, Amundsen Sea (Pine Island Bay), RRS James Clarke Ross 179, station BIO4-EBS-3C, collected on 07 March 2008, epibenthic sledge, epi-net, latitude −74.39848, longitude −104.63748, depth 505 m, 10 specimens.
Description (based on holotype and paratypes)
Robust species up to 4.5 mm long with 16–19 segments (most specimens with 17 segments) (Figs. 3a, 4a) and 8 pairs of elytrophores (elytra missing); body integument smooth dorsally, but with papillae across ventrum arranged in five rows across each segment (Fig. 4c), on parapodia (Figs. 3b, 4a, 5a, d, 6b—insert, 7j) and around ceratophores of median antenna and tentacular cirri (Fig. 5b, c). Holotype NHMUK. 2018.915 complete (Fig. 3a–d), 4.1 mm long, 1.8 mm wide (including parapodia), and 0.9 mm wide (excluding parapodia) for 17 segments (including tentacular segment). Body robust, compact, dorsoventrally flattened; colour in alcohol pale yellow (Figs. 3a, b). Paratype NHMUK. 2018.916 complete (Fig. 4a–f), 4.2 mm long, 2.5 mm wide (including parapodia), and 1.1 mm wide (excluding parapodia) for 18 segments (including tentacular segment). Paratype (SEM specimen) NHMUK. 2018.246 (Fig. 5a–d), 3.5 mm long, 1.9 mm wide (including parapodia), and 0.8 mm wide (excluding parapodia) for 17 segments (including tentacular segment).
Fig. 4.
Macellicephaloides veronikae n. sp. Paratype (NHMUK. 2018.916). a Complete preserved specimen in dorsal view. b Complete preserved specimen in ventral view. c Complete preserved specimen in ventral view, specimen stained with Shirlastain A. d Anterior end in dorsal view, specimen stained with Shirlastain A. e Anterior end in ventral view, with ventral flap on segment 3 marked by arrow, specimen stained with Shirlastain A. f Posterior part of specimen in dorsal view. Scale bars = 1000 µm
Fig. 5.
Macellicephaloides veronikae n. sp. paratype (NHMUK. 2018.246, SEM specimen), SEM micrograph. a Complete specimen in dorsal view (ma – median antenna, tc – tentacular cirri, elt – elytrophore). b Anterior end with prostomium in dorsal view. c Ventral view of the anterior end, showing partially everted proboscis with long pharyngeal papillae (lpap), palps (p), and papillae around the base of median tentaculophores (arrows). d Detail of dorsal cirrus with papillated cirrophores (pap) and smooth style (dc). Scale bars a = 1000 µm, b = 500 µm, c = 400 µm, d = 100 µm
Prostomium bilobed, about as wide as long, with shallow median notch (Fig. 5b). Eyes absent. Median antenna (Fig. 5a–c) with large ceratophore, tufts of papillae laterally at the base of the ceratophore (Fig. 5c), style of antenna moderately long tapering filament, reaching to 2nd segment (longest of all prostomial appendages); lateral antennae absent; frontal filaments absent. Palps smooth, short and robust, wide and thick basally, tapering distally (Fig. 5a–c). Pharynx partially everted in both paratypes; one pair of very long lateral papillae was observed (Fig. 5c). Jaws not observed.
Tentacular cirri similar in size and form to palps, consisting of bulbous tentaculophores and short, smooth distally tapering styles; the bases of tentaculophores with tufts of papillae. Second segment with elytrophores, subbiramous parapodia, with chaetae and ventral cirri. On ventrum of segment 3, a small broadly rounded fleshy pad present (Fig. 4e).
Body always with 8 pairs of elytrophores on segments 2, 4, 5, 7, 9, 11, 13, and 15. Elytrophores inflated, knob-shaped (Fig. 5a). Elytra missing.
Parapodia subbiramous. Notopodia extremely reduced, with acicula (Fig. 6a, b). Neuropodia elongated, distally narrowly pointed, with acicular lobe (Figs. 6b, 7j); neuropodia dorsally and ventrally bordered by papillae (Figs. 3b, 4b, 6b-insert, 7j). Cirrigerous segments with massive, somewhat inflated cirrophores bordered by papillae (Figs. 4a, d, 5a, d, 7j); styles of dorsal cirri similar to the tentacular cirri and palps, somewhat flask-shaped (Figs. 4d, 5d, 6a, 7j) in the anterior part of the body, becoming slightly longer posteriorly. Dorsal tubercules not developed. Ventral cirri present from segment 2 to last segment; inserted basally on segment 2, where very short; in subsequent segments attached subdistally on neuropodium, short, cirriform, tapering, becoming longer posteriorly (Fig. 7j).
Notochaetae absent. Neurochaetae numerous, very long, slender capillaries (Fig. 6a) with alternating rose-bush like spines, each spine forked distally with 4–5 sharp unequal teeth (Fig. 6d–e). Nephridial papillae enlarged on ventrum of chaetigers 6–15, observed as raised bulges. Pygidium tubular, enclosed by the posteriormost segments, with two long thin, anal cirri (Figs. 3d, 4f).
DNA
16S and 18S genes were successfully sequenced for this species, GenBank accession numbers KX867331-KX867344 (Brasier et al. 2016) and PV911683-PV911684 for holotype NHMUK. 2018.915 (this study).
Distribution
Southern Ocean, Amundsen Sea, including deep-shelf troughs of Pine Island Bay, in depths of ~ 500– ~ 1000 m.
Etymology
This species is dedicated to Dr. Veronika Lukášová (https://www.veronikalukasova.com), artist, author, natural history enthusiast, and collector and to her incredible life.
Remarks
This is the tenth species to be described in the genus Macellicephaloides. The species has been assigned to this genus based on the combination of the following characters (see Bonifácio and Menot 2019): body with < 21 segments, 8 pairs of elytra, bilobed prostomium with median antenna, lateral antennae absent, parapodia with reduced notopodia, and well-developed neuropodia, with neurochaetae only.
Macellicephaloides veronikae sp. n. can be easily distinguished from all other known species by the presence of papillae, attached to the bases of ceratophores of median antenna and tentacular cirri, but most prominently observed on neuropodia and cirrophores of dorsal cirri, and ventrum, where arranged in five rows across each segment (Fig. 4c). Of the known species, only M. villosa has the body surface papillated, but the papillae are restricted to the dorsum and cirrophores of dorsal cirri (Fig. 7g), while all other species have a smooth integument (see comparative Fig. 7a–f, h, i).
With the exception of M. villosa, the forked nature of neurochaetal spines was never specifically mentioned in the previous descriptions, although it was depicted in SEM image of M. moustachu, (Fig. 16 J in Bonifácio and Menot 2019).While this structure of neurochaetae is best observed using SEM (Fig. 6d–e), this character can be approximated even under high level (oil) magnification using light microscopy [Neal pers. observation; Levenstein (1971, Fig. 2b)] and it is possible that the forking of spines is present in other Macellicephaloides species, but has previously been overlooked.
The availability of molecular data for M. veronikae sp. n. shows that number of segments is not fixed in this species, similar to the finding of Bonifácio and Menot (2019) for M. moustachu. Specimens belonging to M. veronikae sp. n. as determined by 16S marker (this study, Brasier et al. 2016) possess 16–19 segments, with most having 17 segments. Brasier et al. (2016) reported K2P distances between Macellicephaloides sp. (formalized here as M. veronikae n. sp.) sequences ranged from 0.00 to 3.56%, failing to find the barcoding gap in the absence of data from COI marker. Except for M. moustachu, all previously known species were described based on morphology only, often from a single specimen, and thus intraspecific variation in number of segments is unknown. A tabulated key to all known species of Macellicephaloides is provided below (Table 2).
Table 2.
Comparison of selected morphological characters among all known species of Macellicephaloides
| Type locality | Type depth (m) | No. of specimens examined | No. of segments | Integument | Development of notopodium (see Fig. 7) | Cirrophore of dorsal cirrus (see Fig. 7) | Neurochaetae | |
|---|---|---|---|---|---|---|---|---|
| Macellicephaloides alvini Pettibone, 1989a, b | Gulf of California, Eastern Pacific | 2004 | 1 | 17 | smooth | ~ 1/2 the length of neuropodium | quite robust, cylindrical, slightly shorter then neuropodium | slender, with two rows of delicate spines |
| Macellicephaloides grandicirra Uschakov, 1955 | Kuril-Kamchatka Trench, NW Pacific | 8100–9950 | 43 | 17 | smooth | the same length as notopodium | extremely thin and long, 3–5 × the length of neuropodium | slender, with two rows of spines |
| Macellicephaloides improvisa Levenstein, 1982 | Kuril-Kamchatka Trench, NW Pacific | ? | 1 | 18 | smooth | very reduced, much shorter than neuropodium | very short | unknown |
| Macellicephaloides moustachu Bonifácio & Menot, 2018 | CCZ, eastern Pacific | 4398 | 10 | 14–16 (11 in juvenile) | smooth | very reduced, much shorter than neuropodium | large, cylindrical, elongate, with pointed projection | slender, distally with spines along both margins, with pointed tips [forked spines imaged but not described in Bonifacio and Menot (2019)] |
| Macellicephaloides sandvichensis Levenstein, 1975 | South Sandwich Trench, South Atlantic | 7200–7934 | ? | 17 | smooth | small, much shorter than neuropodium | very slender, about the length of neuropodium | slender, with two rows of spines, distally with slender tooth (hook) |
| Macellicephaloides uschakovi Levenstein, 1971 | Kuril-Kamchatka Trench, NW Pacific | 8120 | 16 | 20–21 | smooth | the same length as notopodium | somewhat thickened, ~ 1/2 length of neuropodia | with two rows of distinct spines, distally gently curved |
| Macellicephaloides verrucosa Uschakov, 1955 | Kuril-Kamchatka Trench, NW Pacific | 7210–7230 | 3 | 16 | smooth | slightly longer then neuropodium | thick, slightly shorter then neuropodium | slender, with two rows of delicate spines |
| Macellicephaloides villosa Levenstein, 1982 | Japan Trench, NW Pacific | 7370–7380 | 1 | 21 | papillated—dorsum, parapodia | very long, longer then neuropodium | slender, elongated, slightly short then neuropodium, papillated | slender, distally with spines along both margins; spines forked with multiple teeth |
| Macellicephaloides vitiazi Uschakov, 1955 | Kuril-Kamchatka Trench, NW Pacific | 7210–8430 | 2 | 16 | smooth | variable along the length of the body, extremely short in the anterior in mid segments, very long in the posterior segments | very slender, about the length of neuropodium | slender, with two rows of delicate spines |
| Macellicephaloides veronikae n. sp. | Amundsen Sea, Southern Ocean | ~ 500–1000 | 27 (15*) | 16–19 | papillated—ventrum, parapodia | very reduced, much shorter than neuropodium | massive, somewhat inflated cirrophores bearing papillae on the edges | slender, distally with spines along both margins; spines forked into ~ 4 unequal teeth |
*Specimens used in an unpublished stable isotope analysis study, now represented by DNA extractions only with the associated sequences published by Brasier et al. (2016)
Macellicephaloides veronikae sp. n. is the third species of its genus to be described from a non-trench environment and represents the shallowest known record of this genus to date. While an evolutionary link between fauna of the deep-sea and polar shelves has been previously proposed (e. g. Strugnell et al. 2011), the complex bathymetry of the Pine Island Bay in the Amundsen Sea, with its deep (up to 1500 m) troughs carved by glaciers, is likely to harbour further taxa with deep-sea relatives (Riehl and Kaiser 2012; Neal et al. 2017, 2018). The Antarctic shelf can be considered a deep-water analogue, owing to the cold temperatures, darkness (even if seasonal) and greater depth of the shelf due to ice-loading. More species are likely to be discovered with further sampling effort from Antarctic deep-sea realms that can provide further insights into the biodiversity and evolutionary history of the region.
Acknowledgements
We would like to thank the British Antarctic Survey for organizing and leading the BIOPEARL expeditions, especially to Dr. Peter Enderlein—the lead scientist of BIOPEARL II expedition, and the crew of RRS James Clarke Ross. Very special thanks go to Dr. Helena Lukášová for her assistance with the line drawing in the Fig. 7j. We are also grateful to Callum Slade for proofreading this manuscript and to Dr. Katrin Linse who commented on this manuscript.
Author contributions
AGG conceived and designed research and collected the samples. LN contributed the morphological description and wrote the manuscript. HW provided the molecular analysis of the samples. All authors read, edited, and approved of the manuscript.
Funding
Open access funding provided by University of Gothenburg. BIOPEARL is part of the British Antarctic Survey Polar Science for Planet Earth Programme funded by The Natural Environment Research Council. Taxonomic research received financial support from the Systematics and Taxonomy research scheme (SynTax), 2011–2012.
Data availability
Sequence data that support the findings of this study have been deposited in GenBank. Specimens were deposited at the Natural History Museum London.
Declarations
Conflict of interest
The authors declare no competing interests.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Contributor Information
Lenka Neal, Email: l.nealova@nhm.ac.uk.
Helena Wiklund, Email: helena.wiklund@marine.gu.se.
References
- Baird W (1865) Contributions towards a monograph of the species of Annelides belonging to the Aphroditacea, containing a list of the known species, and a description of some new species contained in the National Collection of the British Museum. Zool J Linn Soc 8(31–32):172–202 [Google Scholar]
- Bogantes VE, Whelan NV, Webster K, Mahon AR, Halanych KM (2020) Unrecognized diversity of a scale worm, Polyeunoa laevis (Annelida: Polynoidae), that feeds on soft coral. Zool Scr 49(2):236–249. 10.1111/zsc.12400 [Google Scholar]
- Bonifácio P, Menot L (2019) New genera and species from the Equatorial Pacific provide phylogenetic insights into deep-sea Polynoidae (Annelida). Zool J Linn Soc 185(3):555–635. 10.1093/zoolinnean/zly063 [Google Scholar]
- Brandt A, Gooday AJ, Brandao SN, Brix S, Brökeland W, Cedhagen T, Choudhury M, Cornelius N, Danis B, De Mesel I, Diaz RJ (2007) First insights into the biodiversity and biogeography of the Southern Ocean deep sea. Nature 447(7142):307–311. 10.1038/nature05827 [DOI] [PubMed] [Google Scholar]
- Brandt A (1992) Origin of Antarctic Isopoda (Crustacea, Malacostraca). Mar Biol 113:415–423. 10.1007/BF00349167 [Google Scholar]
- Brasier MJ, Wiklund H, Neal L, Jeffreys R, Linse K, Ruhl H, Glover AG (2016) DNA barcoding uncovers cryptic diversity in 50% of deep-sea Antarctic polychaetes. R Soc Open Sci 3(11):160432. 10.1098/rsos.160432 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brenke N (2005) An epibenthic sledge for operations on marine soft bottom and bedrock. Mar Technol Soc J 39(2):10–21. 10.4031/002533205787444015 [Google Scholar]
- Cowart DA, Schiaparelli S, Alvaro MC, Cecchetto M, Le Port AS, Jollivet D, Hourdez S (2022) Origin, diversity, and biogeography of Antarctic scale worms (Polychaeta: Polynoidae): a wide-scale barcoding approach. Ecol Evol 12(7):e9093. 10.1002/ece3.9093 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cohen BL, Gawthrop A, Cavalier-Smith T (1998) Molecular phylogeny of brachiopods and phoronids based on nuclear–encoded small subunit ribosomal RNA gene sequences. Philos Trans R Soc Lond B Biol Sci 353(1378):2039–2061. 10.1098/rstb.1998.0351 [Google Scholar]
- Day JH (1960) The polychaete [sic] fauna of South Africa. Part 5. Errant species dredged off Cape coasts. Ann S Afr Mus 45(3):261–373 [Google Scholar]
- Delle Chiaje S (1825) [-1827] [POLYCHAETA context. Vol.2] Memorie sulla storia e notomia degli animali senza vertebre del Regno di Napoli: Volume 2. 185–444. Stamperia delle Societa Tipografica, Napoli, available online at http://www.biodiversitylibrary.org/item/40280#7
- Fauvel P (1913) Quatrième note préliminaire sur les polychètes provenant des campagnes de l’Hirondelle et de la Princesse-Alice, ou déposées dans le Musée Océanographique de Monaco. Bull Inst Océanogr (Monaco) 270:1–80 [Google Scholar]
- Glover AG, Dahlgren TG, Wiklund H, Mohrbeck I, Smith CR (2016) An end-to-end DNA taxonomy methodology for benthic biodiversity survey in the Clarion-Clipperton Zone, central Pacific abyss. J Mar Sci Eng 4(1):2. 10.3390/jmse4010002 [Google Scholar]
- Griffiths HJ (2010) Antarctic marine biodiversity–what do we know about the distribution of life in the Southern Ocean? PLoS ONE 5(8):e11683. 10.1371/journal.pone.0011683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grube AE (1856) (pub. 1857) Annulata Örstediana. Enumeratio Annulatorum, quae in itinere per Indiam occidentalem et Americam centralem annis 1845–1848 suscepto legit cl. A.S. Örsted, adjectis speciebus nonnullis a cl. H. Kröyero in itinere ad Americam meridionalem collectis. [Part 1] Videnskabelige Meddelelser fra Dansk naturhistorisk Forening i Köbenhavn 1856: 44–62, available online at http://www.biodiversitylibrary.org/page/35632990
- Hanley JR (1989) Revision of the scaleworm genera Arctonoe Chamberlin and Gastolepidia Schmarda (Polychaeta: Polynoidae) with the erection of a new subfamily Arctonoinae. The Beagle, Records of the Northern Territory Museum of Arts and Sciences 6(1):1–34 [Google Scholar]
- Hartmann-Schroder G (1971) Annelida, Borstenwurmer, Polychaeta. Die Tierwelt Deutschlands und der Angrenzenden Meeresteile Nach Ihren Merkmalen und Nach Ihrer Lebensweise 58:1–594 [Google Scholar]
- Hiley AS, Mongiardino Koch N, Rouse GW (2024) Phylogenetics of Lepidonotopodini (Macellicephalinae, Polynoidae, Annelida) and comparative mitogenomics of shallow-water vs. deep-sea scaleworms (Aphroditiformia). Biol 13(12):979. 10.3390/biology13120979 [Google Scholar]
- Kaiser S, Barnes DK, Sands CJ, Brandt A (2009) Biodiversity of an unknown Antarctic Sea: assessing isopod richness and abundance in the first benthic survey of the Amundsen continental shelf. Mar Biodivers 39:27–43. 10.1007/s12526-009-0004-9 [Google Scholar]
- Katoh K (2002) MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transformation. Nucleic Acids Res 30(14):3059–3066. 10.1093/nar/gkf436 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalyaanamoorthy S, Minh B, Wong T, von Haeseler A, Jermiin LS (2017) ModelFinder: fast model selection for accurate phylogenetic estimates. Nat Methods 14:587–589. 10.1038/nmeth.4285 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kinberg JGH (1856) Nya slägten och arter af Annelider. Overs K Vetenskaps-Akad Forh 12(9–10):381–388 [Google Scholar]
- Levenstein RY (1971) Polychaeta of the genus Macellicephala and Macellicephaloides (Aphroditidae) from the Pacific Ocean. Tr Inst Okeanol Im p p Shirshova, Akad Nauk USSR 92:18–35 [Google Scholar]
- Levenstein RY (1975) The polychaetous annelids of the deep-sea trenches of the Atlantic sector of the Antarctic Ocean. Tr Inst Okeanol Im P P Shirshova Akad Nauk SSSR 103:119–142 [Google Scholar]
- Levenstein RY (1982) On the polychaete fauna (fam. Polynoidae) from the Trench of Japan. Tr Inst Okeanol Im p p Shirshova, Akad Nauk USSR 17:59–62 [Google Scholar]
- Levinsen GMR (1886) Kara-Havets Ledorme (Annulata). [Book section] 288–303. In: Lütken CF (Ed) Djimphna-Togtets Zoologiske-botaniske Udbytte. J. Hagerup. Köbenhaven
- Linnaeus C (1767) Systema naturae per regna tria naturae: secundum classes, ordines, genera, species, cum characteribus, differentiis, synonymis, locis. Ed 12. 1., Regnum Animale. 1 & 2. Holmiae [Stockholm], Laurentii Salvii pp 1–532 [1766] pp 533–1327 [1767], available online at http://www.biodiversitylibrary.org/item/83650#5
- Linse K, Griffiths HJ, Barnes DK, Brandt A, Davey N, David B, De Grave S, Eléaume M, Glover AG, Hemery LG, Mah C (2013) The macro-and megabenthic fauna on the continental shelf of the eastern Amundsen Sea, Antarctica. Cont Shelf Res 68:80–90. 10.1016/j.csr.2013.08.012 [Google Scholar]
- McIntosh WC [M’Intosh] (1885) Report on the Annelida Polychaeta collected by H.M.S. Challenger during the years 1873–1876. Report on the Scientific Results of the Voyage of H.M.S. Challenger during the years 1873–76. Zool 12 (part 34):i–xxxvi, 1–554, pl. 1–55, 1A–39A, & Annelida stations map., available online at https://biodiversitylibrary.org/page/50688426
- Medlin L, Elwood H, Stickel S, Sogin M (1988) The characterization of enzymatically am-plified eukaryotic 16S-like rRNA-coding regions. Gene 71(2):491–499. 10.1016/0378-1119(88)90066-2 [DOI] [PubMed] [Google Scholar]
- Miura T, Hashimoto J (1991) Two new branchiate scale-worms (Polynoidae: Polychaeta) from the hydrothermal vent of the Okinawa Trough and the volcanic seamount off Chichijima Island. Proc Biol Soc Wash 104(1):166–174 [Google Scholar]
- Monro CCA (1939). Polychaeta. B.A.N.Z.Antarctic Research Expedition Reports, Ser B Zoology and Botany 4(4):87–156
- Montagu G (1808) Description of several marine animals found on the south coast of Devonshire. Trans Linn Soc Lond 9:81–114 [Google Scholar]
- Moreau C, Linse K, Griffiths H, Barnes D, Kaiser S, Glover A, Sands C, Strugnell J, Enderlein P, Geissler P (2013) Amundsen Sea Mollusca from the BIOPEARL II expedition. ZooKeys 294:1. 10.3897/zookeys.294.4796 [Google Scholar]
- Neal L, Barnich R, Wiklund H, Glover AG (2012) A new genus and species of Polynoidae (Annelida, Polychaeta) from Pine Island Bay, Amundsen Sea, Southern Ocean - a region of high taxonomic novelty. Zootaxa 3542:80–88. 10.11646/zootaxa.3542.1.4 [Google Scholar]
- Neal L, Linse K, Brasier MJ, Sherlock E, Glover AG (2017) Comparative marine biodiversity and depth zonation in the Southern Ocean: evidence from a new large polychaete dataset from Scotia and Amundsen seas. Mar Biodivers 48:581–601. 10.1007/s12526-017-0735-y [Google Scholar]
- Neal L, Brasier MJ, Wiklund H (2018) Six new species of Macellicephala (Annelida: Polynoidae) from the Southern Ocean and south Atlantic with re-description of type species. Zootaxa 4455(1):1–34. 10.11646/zootaxa.4455.1.1 [DOI] [PubMed] [Google Scholar]
- Nguyen LT, Schmidt HA, Von Haeseler A, Minh BQ (2015) IQ-TREE: a fast and effective stochastic algorithm for estimating maximum-likelihood phylogenies. Mol Biol Evol 32(1):268–274. 10.1093/molbev/msu300 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nygren A, Sundberg P (2003) Phylogeny and evolution of reproductive modes in Autolytinae (Syllidae, Annelida). Mol Phyl Evol 29(2):235–249. 10.1016/S1055-7903(03)00095-2 [Google Scholar]
- Örsted AS (1845) Fortegnelse over Dyr, samlede i Christianiafjord ved Drøbak fra 21–24 Juli, 1844. Naturhistorisk Tidsskrift, Kjøbenhavn, Ser 2.1:400–427, plate V., available online at https://biodiversitylibrary.org/page/2359713
- Pabis K, Błażewicz-Paszkowycz M, Jóźwiak P, Barnes DK (2015) Tanaidacea of the Amundsen and Scotia seas: an unexplored diversity. Antarct Sci 27(1):19–30. 10.1007/s12526-017-0735-y [Google Scholar]
- Palumbi SR (1996) Nucleic acids II: the polymerase chain reaction. Mol Syst 205–247
- Pettibone MH (1985) Polychaete worms from a cave in the Bahamas and from experimental wood panels in deep water of the North Atlantic (Polynoidae, Macellicephalinae, Harmothoinae). Proc Biol Soc Wash 98(1):127–149 [Google Scholar]
- Pettibone MH (1989a) Polynoidae and Sigalionidae (Polychaeta) from the Guaymas Basin, with descriptions of two new species, and additional records from hydrothermal vents of the Galapagos Rift, 21ºN, and seep-sites in the Gulf of Mexico (Florida and Louisiana). Proc Biol Soc Wash 102(1):154–168 [Google Scholar]
- Pettibone MH (1989b) A new species of Benhamipolynoe (Polychaeta: Polynoidae: Lepidastheniinae) from Australia, associated with the unattached stylasterid coral Conopora adeta. Proc Biol Soc Wash 102(2):300–304 [Google Scholar]
- Pettibone MH (1997) Revision of the scaleworm genus Eulagisca McIntosh (Polychaeta: Polynoidae) with the erection of the subfamily Eulagiscinae and the new genus Pareulagisca. Proc Biol Soc Wash 110(4):537–551 [Google Scholar]
- Read G, Fauchald K (Ed) (2025) World Polychaeta Database. Polynoidae Kinberg, 1856. Accessed through: World Register of Marine Species at: https://www.marinespecies.org/aphia.php?p=taxdetailsandid=939 on 2025–01–10
- Reed B, Green JAM, Jenkins A et al (2024) Recent irreversible retreat phase of Pine Island Glacier. Nat Clim Chang 14:75–81. 10.1038/s41558-023-01887-y [Google Scholar]
- Riehl T, Kaiser S (2012) Conquered from the deep sea? A new deep-sea isopod species from the Antarctic shelf shows pattern of recent colonization. PLoS One 7(11):e49354. 10.1371/journal.pone.0049354 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ronquist F, Teslenko M, Van Der Mark P, Ayres DL, Darling A, Höhna S, Larget Liu L, Suchard MA, Huelsenbeck JP (2012) MrBayes 3.2: efficient Bayesian phylogenetic inference and model choice across a large model space. Syst Biol 61(3):539–542. 10.1093/sysbio/sys029 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Savigny J-C (1822) Système des annélides, principalement de celles des côtes de l’Égypte et de la Syrie, offrant les caractères tant distinctifs que naturels des Ordres, Familles et Genres, avec la Description des Espèces. Description de l’Égypte ou Recueil des Observations et des Recherches qui ont été faites en Égypte pendant l’Expédition de l’Armée Française, publié par les Ordres de sa Majesté l’Empereur Napoléon le Grand, Histoire Naturelle, Paris 1(3):1–128, available online at http://biodiversitylibrary.org/page/4132989
- Schmarda LK (1861) Neue Wirbellose Thiere: Beobachted und Gesammelt auf einer Reise um die Erdr 1853 bis 1857. In Turbellarien, Rotatorien und Anneliden. Leipzig, Verlag von Wilhelm Engelmann. Erster Band, Zweite Hälfte, available online at http://www.biodiversitylibrary.org/ia/neuewirbelloseth21861schm
- Schüller M, Ebbe B, Wägele JW (2009) Community structure and diversity of polychaetes (Annelida) in the deep Weddell Sea (Southern Ocean) and adjacent basins. Mar Biodivers 39:95–108. 10.1007/s12526-009-0009-4 [Google Scholar]
- Schüller M, Ebbe B (2014) Polychaetes. In: De Broyer C, Koubbi P (eds) Biogeographic atlas of the Southern Ocean. Scientific Committee on Antarctic Research, Cambridge, pp 134–137 [Google Scholar]
- Serpetti N, Taylor ML, Brennan D, Green DH, Rogers AD, Paterson GL, Narayanaswamy BE (2017) Ecological adaptations and commensal evolution of the Polynoidae (Polychaeta) in the Southwest Indian Ocean Ridge: a phylogenetic approach. Deep Sea Res 2 Top Stud Oceanogr 137:273–281. 10.1016/j.dsr2.2016.06.004 [Google Scholar]
- Sjölin E, Erséus C, Källersjö M (2005) Phylogeny of Tubificidae (Annelida, Clitellata) based on mitochondrial and nuclear sequence data. Mol Phylogenet Evol 35(2):431–441. 10.1016/j.ympev.2004.12.018 [DOI] [PubMed] [Google Scholar]
- Sui J, Li X, Kou Q (2018) A new species of the genus Intoshella Darboux, 1899 (Polychaeta: Polynoidae) commensal with a deep-sea sponge from a seamount near the Mariana Trench. Mar Biodivers 49(3):1479–1488. 10.1007/s12526-018-0922-5 [Google Scholar]
- Strugnell JM, Cherel Y, Cooke IR, Gleadall IG, Hochberg FG, Ibáñez CM, Jorgensen E, Laptikhovsky VV, Linse K, Norman M, Vecchione M (2011) The Southern Ocean: source and sink? Deep Sea Res 2 Top Stud Oceanogr 58(1–2):196–204. 10.1016/j.dsr2.2010.05.015 [Google Scholar]
- Tebble N, Chambers S (1982) Polychaetes from Scottish Waters, Part 1. Family Polynoidae. RSM 73
- Théel HJ (1879) Les Annélides Polychètes des mers de la Nouvelle-Zemble. Kungliga Svenska Vetenskapsakademiens Handlingar 16(3):1–75, plates I–IV
- Uschakov PV (1955) Mnogoshchetinkovye chervi sem. Aphroditidae Kurilo-Kamshatskovpradinyi. Tr Inst Okeanol im P. P. Shirshova, Akad Nauk USSR 12:311–321 [Google Scholar]
- Verrill AE (1882) Notice of recent additions to the marine Invertebrata of the northeastern coast of America, with descriptions of new genera and species and critical remarks on others. Part III. catalogue of Mollusca recently added to the fauna of southern New England. Smithsonian Misc Collect 22:405–409 [Google Scholar]
- Willey A (1902) XII. Polychaeta In: Sharpe, B and Bell J (eds) Report on the collections of natural history made in the Antarctic regions during the voyage of the "Southern Cross",. pp. 262–283, plates 241–246. British Museum, London., available online at https://www.biodiversitylibrary.org/page/12554197
- Zhang Y, Chen C, Qiu J-W (2018) Sexually dimorphic scale worms (Annelida: Polynoidae) from hydrothermal vents in the Okinawa Trough: two new species and two new sex morphs. Front Mar Sci 5(112):1–20. 10.3389/fmars.2018.00112 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Sequence data that support the findings of this study have been deposited in GenBank. Specimens were deposited at the Natural History Museum London.